Abstract
OBJECTIVE
Although many studies have shown that carotid intima-media thickness (IMT) is associated with coronary artery disease (CAD), it remains inconclusive whether assessment of carotid IMT is useful as a screening test for asymptomatic but severe CAD in diabetic patients.
RESEARCH DESIGN AND METHODS
A total of 333 asymptomatic type 2 diabetic patients without history of CAD underwent exercise electrocardiogram or myocardial perfusion scintigraphy for detection of silent myocardial ischemia, and those whose test results were positive were subjected to coronary computed tomography angiography or coronary angiography. The ability of carotid IMT to identify severe CAD corresponding to treatment with revascularization was examined by receiver-operating characteristic (ROC) curve analyses.
RESULTS
Among the 333 subjects, 17 were treated with revascularization. A multiple logistic regression analysis showed that maximum IMT was an independent predictor of severe CAD even after adjustment for conventional risk factors. ROC curve analyses revealed that the addition of maximum IMT to conventional risk factors significantly improved the prediction ability for severe CAD (from area under the curve, 0.67 to 0.79; P = 0.039). The greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.45 mm (pretest probability, 5%; posttest probability, 11%; sensitivity, 71%). When we applied age-specific cut-off values, the sensitivity of screening further increased in both the nonelderly (pretest probability, 6%; posttest probability, 10%; sensitivity, 100%) and the elderly subjects (pretest probability, 5%; posttest probability, 15%; sensitivity, 100%).
CONCLUSIONS
Our study suggests that carotid maximum IMT is useful for screening asymptomatic type 2 diabetic patients with severe CAD equivalent to revascularization.
Coronary artery disease (CAD) remains the main cause of death in diabetic patients and once it has developed, diabetic patients have worse outcome as compared with nondiabetic patients. One reason for this is the difficulty of early diagnosis of CAD. Many cases of coronary atherosclerosis develop without symptoms, especially in diabetic patients. It is reported that in patients with development of ischemic heart disease, frequency of silent myocardial ischemia (SMI) is approximately three-times to six-times higher in diabetic patients than in nondiabetic patients (1). According to Detection of Ischemia in Asymptomatic Diabetes study, as much as 22% of asymptomatic type 2 diabetic patients aged 50–70 years were revealed to have SMI when they were screened for myocardial ischemia with myocardial perfusion scintigraphy (2). Therefore, early detection of asymptomatic severe CAD and rapid intervention are important to reduce mortality in the management of diabetes.
Sophisticated cardiac study such as exercise electrocardiogram (ECG), myocardial perfusion scintigraphy, coronary computed tomography (CT) angiography, and coronary angiography can determine disease severity with a high degree of sensitivity and specificity. However, it is unrealistic to screen for SMI with these tools in all diabetic patients, because these tests are limited by the potential of significant adverse effects, technical difficulty, availability, and cost. Therefore, a noninvasive and inexpensive risk prediction tool of SMI is required for identifying individuals at high-risk for CAD.
Cardiovascular risk assessment based on conventional risk factors is recommended for predicting cardiovascular risk, although validation studies showed moderate performance (3–5). However, carotid intima media thickness (IMT) has been shown to be a reliable marker of risk for CAD and cardiovascular events in previous studies (6). Moreover, measurement of carotid IMT with high-resolution ultrasound can be assessed quickly, noninvasively, and inexpensively. Some studies have shown that carotid IMT could improve the prediction ability for cardiovascular disease over and above conventional coronary risk factors (7,8). These findings suggest that measurement of carotid IMT could be useful for identifying individuals at high-risk for CAD who should undergo SMI screening tests.
In this study, we investigated the usefulness of carotid IMT for identifying severe CAD equivalent to coronary revascularization based on the cross-sectional analysis in 333 asymptomatic type 2 diabetic patients who underwent screening tests for SMI.
RESEARCH DESIGN AND METHODS
Subjects
Middle-aged and older Japanese type 2 diabetic subjects without apparent cardiovascular disease participated in this study. We considered subjects eligible when they fulfilled the following criteria: age 40 years or older at the time of enrollment and diagnosis of type 2 diabetes based on Japan Diabetes Society’s criteria. Exclusion criteria were: angina pectoris or anginal equivalent symptoms; history of myocardial infarction, heart failure, or coronary revascularization; history of sophisticated cardiac study (exercise ECG, echocardiography, myocardial perfusion scintigraphy, coronary CT angiography, or coronary angiography); elevated liver enzymes (glutamate oxaloacetate transaminase or glutamic pyruvic transaminase ≥2.5-times the normal range); and renal insufficiency (serum creatinine ≥2.0 mg/dL). Screening of the study subjects was performed consecutively during the registration period (from April 2007 to December 2009) at the outpatient clinics of diabetes in Osaka Police Hospital. All the patients who met eligibility criteria were asked if they could participate in the current study, and all the patients who agreed to participate were registered. A total of 333 subjects were consecutively enrolled in this study.
We also prepared another cohort of asymptomatic type 2 diabetic subjects for the validation study. A total of 99 subjects were enrolled at Osaka University Medical Hospital. All the participants underwent carotid ultrasonography, rest or exercise ECG, and coronary CT angiography. In this validation cohort, 20 out of 99 study subjects underwent revascularization after the screening test for SMI (9). Their clinical and biochemical characteristics are shown in Supplementary Table 1.
The study protocol was approved by the committees on the ethics of human research of Osaka Police Hospital. A written informed consent was obtained from all the participants after a full explanation of the study.
Clinical and biochemical analysis
Blood samples were collected after an overnight fast for analysis of serum concentrations of glucose, total cholesterol, triglycerides, HDL cholesterol, LDL cholesterol, and HbA1c. Diagnosis of type 2 diabetes was made by the principal investigator in accordance with the standards of the Japan Diabetes Society (10). Dyslipidemia was defined as LDL cholesterol ≥120 mg/dL, HDL cholesterol <40 mg/dL, triglycerides ≥150 mg/dL, or current use of lipid-lowering agents. Hypertension was defined as systolic blood pressure ≥130 mmHg or diastolic blood pressure ≥80 mmHg, or current use of antihypertensive agents. Smoking status was categorically defined on the basis of self-reports.
Measurement of carotid IMT
B-mode ultrasonography of the carotid artery was performed using an ultrasound machine (Toshiba SSA-790CE; Toshiba Medical Systems, Tokyo, Japan) with a 7.5-MHz linear transducer. Scanning of the extracranial common carotid artery (CCA), the carotid bulb, and the internal carotid artery (ICA) in the neck was performed bilaterally in three different longitudinal projections as well as transverse projections. The IMT was measured as the distance between two parallel echogenic lines corresponding to the blood–intima and media–adventitia interface on the posterior wall of the artery using manual reading system equipped with the ultrasonograph. The greatest thickness of IMT (including plaque lesions) in the CCA, the carotid bulb, and the ICA were measured separately, and the greatest value of them was defined as maximum IMT and used as the representative value for each individual (11). In addition, three determinations of IMT were conducted at the site of the thickest point and two adjacent points (located 1 cm upstream and 1 cm downstream from the thickest point) in the CCA. These three determinations were averaged (mean IMT) and the greatest value among the six mean IMTs (three from the left and three from the right) was used as the representative value for each individual. Carotid plaque was defined as a focal structure encroaching into the arterial lumen or demonstrating a thickness >1.5 mm as measured from the media–adventitia interface to the intralumen interface.
To confirm the utility of maximum IMT in predicting severe CAD in asymptomatic type 2 diabetic subjects, we performed similar analyses using another cohort of asymptomatic type 2 diabetic subjects who were recruited at Osaka University Hospital (9). In this hospital, all scans were conducted by experienced laboratory physicians using the same ultrasound system and the same measuring method of IMT that were used in Osaka Police Hospital. Reproducibility analysis of 20 replicate measurements yielded absolute mean differences of 0.03 ± 0.05 and 0.05 ± 0.05 mm for maximum IMT and mean IMT, respectively. The interobserver coefficient of variation for measurement of maximum IMT and mean IMT were 0.7% and 2.3%, respectively.
Assessment of CAD
In this study, all the participants underwent exercise ECG or myocardial perfusion scintigraphy for detection of SMI, and those with positive results for these tests were subject to coronary CT angiography or coronary angiography; exercise ECG was most frequently performed (n = 324), followed by coronary CT angiography (n = 170), myocardial perfusion scintigraphy (n = 72), and coronary angiography (n = 31).
Symptom-limited bicycle exercise test was performed with a Bruce protocol. The 12-lead ECG was monitored continuously during the test. Exercise was continued for 3 min at each stage. Tests were terminated because of chest pain, fatigue, dyspnea, arrhythmias, or marked ST-segment changes on ECG. The following ST-segment changes were considered positive for ischemia: horizontal or down-sloping ST-segment depression ≥0.1 mV persisting ≥0.08 s and ST-segment elevation ≥0.1 mV compared with control tracing in any lead except augmented vector right (aVR). The test was considered negative only when ≥85% of the maximum heart rate predicted was achieved without positive ST-segment changes. The predicted maximum heart rate was determined by patient age − 220.
Other results were considered borderline, and the patients with borderline results were evaluated with stress ECG-gated 201Tl myocardial perfusion imaging. Exercise stress was performed with the same protocol and vasodilator stress was performed by intravenous infusion of adenosine (120 μg/kg/min). Rest and stress images were obtained on the same day. Data were acquired with dual-head single-photon emission CT camera (Infinia; GE Healthcare, Little Chalfont, England), followed by reconstruction into long-axis and short-axis projection perpendicular to the heart axis. Perfusion defect (segmental tracer activity <50% of maximum) was identified on the stress image first and was divided into ischemia (reversible defects: >10% increase in tracer uptake on the rest image compared with stress image) or scar (irreversible defects).
Patients were judged as having ischemia by stress test when his or her exercise ST-segment response was positive or SPECT images presented ischemic change.
Coronary CT angiography was performed with the use of a 64-slice multidetector CT scanner (LightSpeed VCT; GE Medical Systems) with 64- × 0.625-mm section collimation, 350-ms rotation time, 100- to 120-kV tube voltage, and 280- to 750-mA tube using ECG modulation. Patients with at least one stenotic lesion with ≥50% obstruction of the coronary artery were considered to have significant coronary artery stenosis.
All results of CAD screening were evaluated by the cardiologists participating in this study. Indications for coronary revascularization for patients with CAD were decided based on the American College of Cardiology/American Heart Association guidelines for percutaneous coronary intervention (12).
Statistical analyses
All statistical analyses were performed with Dr. SPSS II for Windows (SPSS). All values are reported as means ± SD, median and interquartile range, or real numbers of subjects with the percentage in parentheses. Between-group differences of the average were compared using the unpaired t test for parametric data, and the Mann-Whitney U test was used for nonparametric data. Between-group differences of numbers and percentages were compared using a χ2 test. Multivariate logistic regression analysis was used to detect independent predictors of severe CAD. The prediction ability of maximum IMT to identify severe CAD was examined by receiver-operating characteristic (ROC) curve analyses. Furthermore, to examine whether the addition of maximum IMT to conventional coronary risk factors could improve the prediction ability for severe CAD, ROC curves were plotted for conventional coronary risk factors, with and without maximum IMT. The incremental effect of adding maximum IMT to the conventional risk factors for identifying severe CAD was evaluated with the use of the Z test. For all tests, P < 0.05 was considered statistically significant.
RESULTS
Results of SMI screening
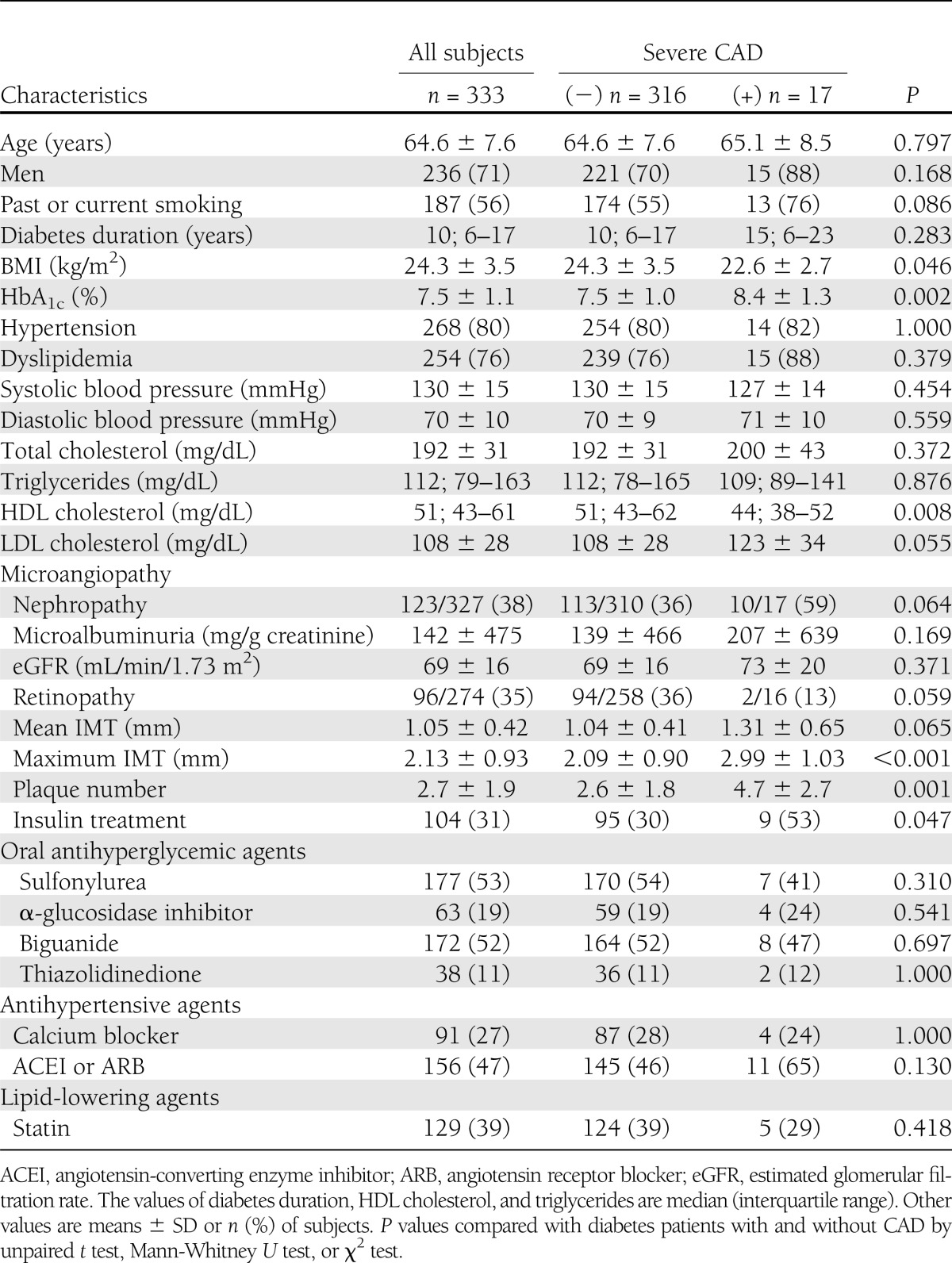
Among a total of 333 subjects (males, 71%; age, 64.6 ± 7.6 years [mean ± SD]; diabetes duration, 12.6 ± 8.7 years; HbA1c, 7.5 ± 1.1%), 268 (80%) patients had hypertension and 254 (76%) had dyslipidemia (Table 1).
Table 1.
Baseline characteristics of the study population
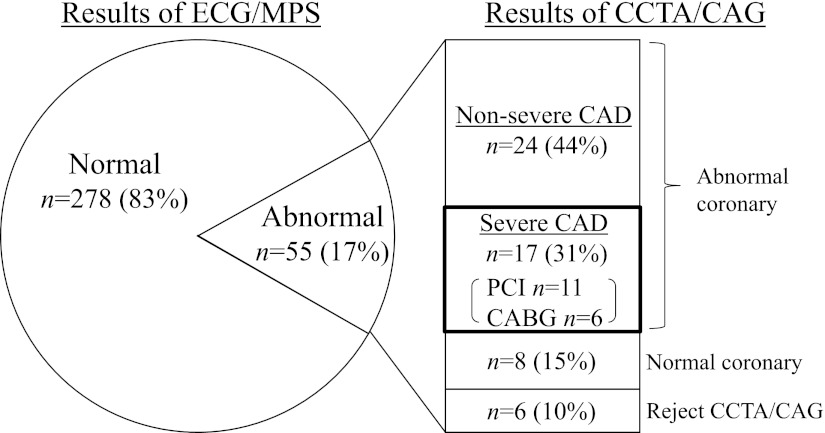
The results of SMI screening are shown in Fig. 1. Among the 333 subjects, 55 (17%) had SMI on exercise ECG or myocardial perfusion scintigraphy; 24 patients had positive ECG alone, 16 patients had positive myocardial perfusion scintigraphy alone, and 15 patients had concordant positive findings on both tests. Among the 55 patients with SMI, 41 (75%) were revealed to have significant coronary artery stenosis (≥50%) diagnosed by subsequent coronary CT angiography or coronary angiography. Then, 17 (31%) patients had severe CAD diagnosed, corresponding to the treatment with coronary revascularization, and all of them underwent percutaneous coronary intervention (n = 11) or coronary bypass graft surgery (n = 6).
Figure 1.
Results of SMI screening. Among 55 patients having SMI, 41 patients had significant coronary artery stenosis (≥50%), and 17 were treated with coronary revascularizations. MPS, myocardial perfusion scintigraphy; CCTA, coronary CT angiography; CAG, coronary angiography; PCI, percutaneous coronary intervention; CABG, coronary artery bypass grafting.
Relationship between carotid IMT and severe CAD
Patients with severe CAD had greater mean IMT than patients without it, but it was not significant. However, maximum IMT and plaque number were significantly greater in patients with severe CAD equivalent to coronary revascularization (Table 1). Furthermore, all the patients who were treated with revascularization had thickening of maximum IMT >1.6 mm.
To demonstrate that maximum IMT is a determinant of severe CAD independent of the other risk factors, we performed a multivariate logistic regression analysis including gender, age, hypertension, dyslipidemia, smoking status, HbA1c, and maximum IMT as independent variables. We found two independent predictors of severe CAD in the final discrimination function: maximum IMT (OR, 2.36; 95% CI, 1.37–4.08; P = 0.002) and HbA1c (OR, 1.64; 95% CI, 1.09–2.45; P = 0.017).
The ability of carotid IMT to identify severe CAD
We tried to assess the impact of maximum IMT in the CCA, the carotid bulb, and the ICA, separately. However, IMT measurements in the ICA area were obtainable in only 50% of all participants mainly because of the difficulty of observation. Therefore, we set the following three indices: CCA, carotid bulb ICA, and combined maximum IMT (CCA or carotid bulb ICA, whichever is greater). The prediction abilities of CCA, carotid bulb ICA, and combined maximum IMT estimated by the area under the ROC curve (AUC) were 0.62 (95% CI, 0.46–0.78), 0.72 (95% CI, 0.60–0.83), and 0.74 (95% CI, 0.63–0.86), respectively. In addition, the prediction ability of plaque numbers was 0.74 (95% CI, 0.62–0.86; P = 0.001). We also estimated the prediction ability of mean IMT. The AUC for mean IMT was 0.64 (95% CI, 0.49–0.79; P = 0.051). Even when measurement of mean IMT was performed in a region free of plaque, the AUC for mean IMT was 0.66 (95% CI, 0.51–0.80; P = 0.031).
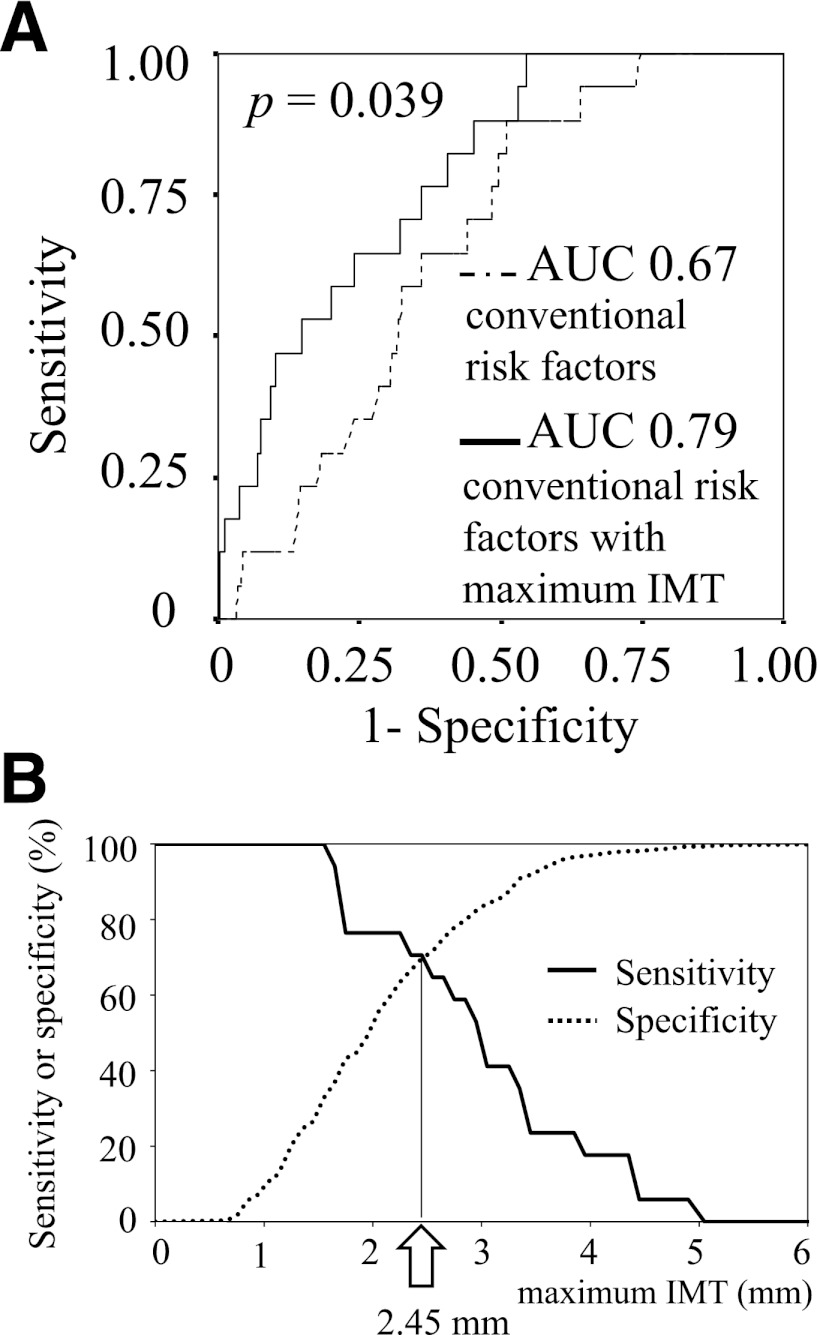
Next, we evaluated the prediction ability of severe CAD by ROC curves plotted for conventional coronary risk factors (age, gender, smoking status, hypertension, and dyslipidemia) with maximum IMT and for those without maximum IMT. This analysis revealed that the AUC significantly increased after the addition of maximum IMT to conventional coronary risk factors (0.67, 95% CI, 0.57–0.77 with conventional coronary risk factors versus. 0.79, 95% CI, 0.70–0.88 with conventional coronary risk factors and maximum IMT; P = 0.039), suggesting that the addition of maximum IMT to conventional coronary risk factors could improve the risk estimation for severe CAD (Fig. 2A).
Figure 2.
ROC curve analyses to identify severe CAD. A: The AUC significantly increased after the addition of maximum IMT to conventional coronary risk factors including age, gender, smoking status, hypertension, and dyslipidemia (from AUC 0.67 to 0.79; P = 0.039). B: The greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.45 mm.
The cut-off value of maximum IMT to identify severe CAD
To determine cut-off value of maximum IMT for screening of severe CAD, ROC curve was plotted for maximum IMT alone. This analysis revealed that the greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.45 mm (sensitivity, 0.71; specificity, 0.70; positive predictive value [PPV], 0.11; negative predictive value [NPV], 0.98) (Fig. 2B).
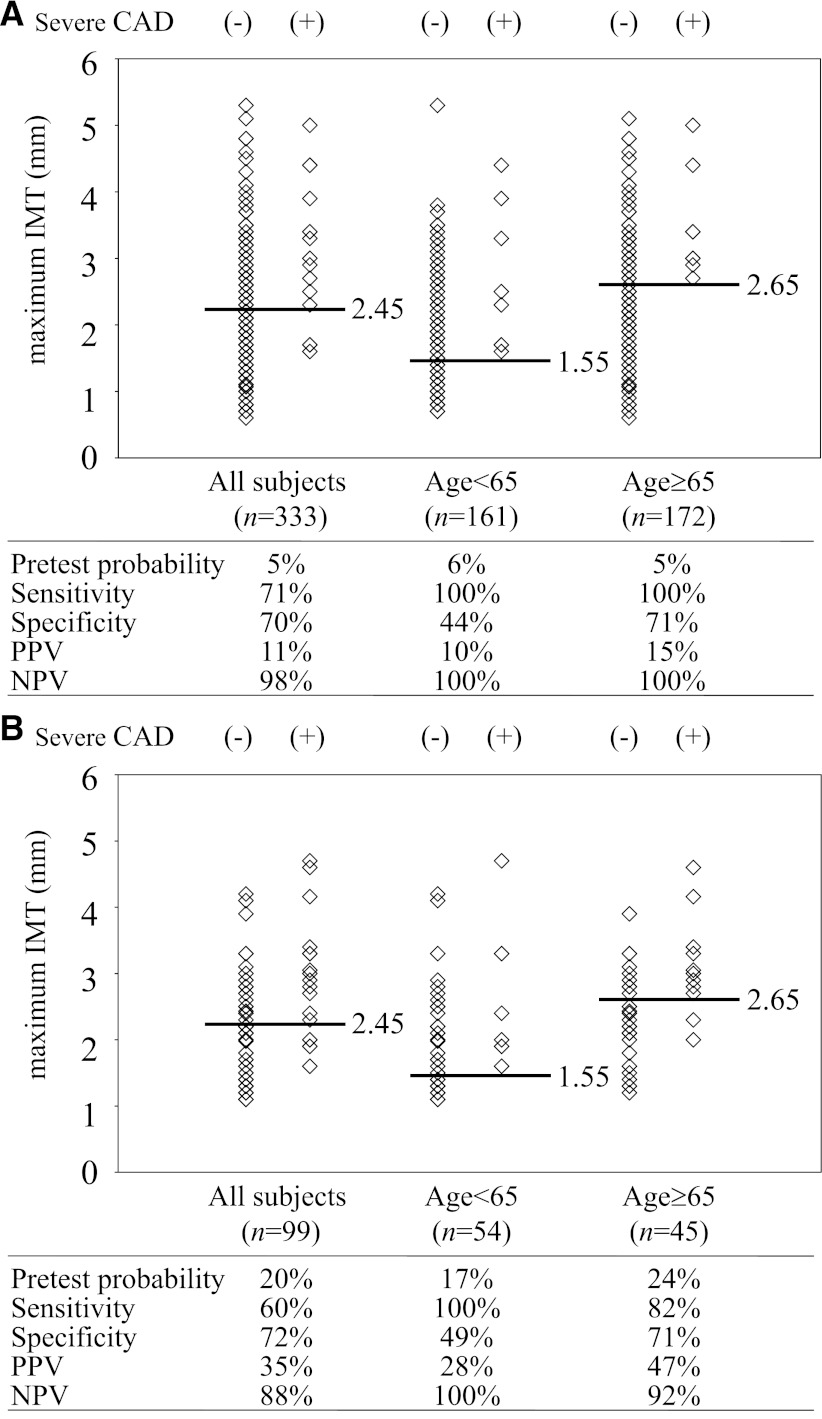
Next, we divided the study subjects into the elderly (65 years or older) group (n = 162) and the nonelderly (younger than 65 years) group (n = 171), because the value of maximum IMT was significantly correlated with age (r = 0.274; P < 0.001) (Supplementary Fig. 1). ROC curve analyses revealed that the greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 1.55 mm in the nonelderly (sensitivity, 1.00; specificity, 0.44; PPV, 0.10; NPV, 1.00) and 2.65 mm in the elderly group (sensitivity, 1.00; specificity, 0.71; PPV, 0.15; NPV, 1.00) (Fig. 3A).
Figure 3.
Sensitivity, specificity, PPV, and NPV of maximum IMT to identify severe CAD. A: The greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.45 mm in all subjects, 1.55 mm in the nonelderly (younger than 65 years old), and 2.65 mm in the elderly (65 years or older) group. B: The performance of the cut-off values of maximum IMT was validated in another cohort.
Confirmation study in another cohort
To confirm the utility of maximum IMT in identifying severe CAD in asymptomatic type 2 diabetic subjects, we performed similar analyses using another cohort of asymptomatic type 2 diabetic subjects who were recruited at Osaka University Hospital (12). In this cohort, 20 out of 99 study subjects underwent revascularization after the screening test for SMI. ROC curve analyses showed that the AUC for maximum IMT (CCA or carotid bulb ICA, whichever is greater) to identify severe CAD was 0.75 (95% CI, 0.63–0.87; P < 0.001) and that the greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.65 mm (sensitivity, 0.60; specificity, 0.80; PPV, 0.43; NPV, 0.89).
Next, to evaluate the performance of the cut-off value of maximum IMT derived from the derivation cohort (n = 333; cut-off level of maximum IMT ≥2.45 mm), we applied this value to identify severe CAD in this validation cohort (n = 99) and examined its predictive ability. In this validation cohort, as a whole, 12 out of 34 (35%) patients whose maximum IMT was above cut-off value (≥2.45 mm) underwent revascularization (pretest probability, 0.20; sensitivity, 0.60; specificity, 0.72; PPV, 0.35; NPV, 0.88). Furthermore, we divided the subjects in this validation cohort into the nonelderly (younger than 65 years old, n = 54) and the elderly (65 years or older, n = 45) groups and evaluated the performance of the cut-off levels of maximum IMT derived from the derivation cohort (1.55 mm and 2.65 mm, respectively). In the nonelderly group, 9 out of 32 (28%) whose maximum IMT was above cut-off value (≥1.55 mm) were treated with revascularization (pretest probability, 0.17; sensitivity, 1.00; specificity, 0.49; PPV, 0.28; NPV, 1.00). In the elderly group, 9 out of 19 (47%) whose maximum IMT was above the cut-off value (≥2.65 mm) were treated with revascularization (pretest probability, 0.24; sensitivity, 0.82; specificity, 0.71; PPV, 0.47; NPV, 0.92) (Fig. 3B). These results indicate the validity and the reproducibility of maximum IMT as a tool for screening the high-risk subjects with severe CAD.
CONCLUSIONS
It has been thought that carotid IMT is associated with future risk for cardiovascular disease, but it remains controversial whether measurement of carotid IMT is clinically useful as a screening test for CAD. Our study showed that the addition of maximum IMT to conventional coronary risk factors could substantially improve the prediction ability for severe CAD equivalent to coronary revascularization in asymptomatic type 2 diabetic patients. This finding suggests that measurement of carotid IMT in these patients is clinically useful for screening individuals at high-risk for severe CAD.
It has been reported that in healthy populations, 2.5–10% of middle-aged men have asymptomatic but active CAD (13–15). In diabetic patients, the prevalence of SMI determined by noninvasive imaging is reported to be 12–31% (2,16–19). In this study, we found that the prevalence of abnormal SMI screening findings was 17% in our asymptomatic type 2 diabetic patients, which was consistent with the previous findings. It should be noted that many of them had coronary arteriosclerosis; 75% of them were revealed to have significant coronary artery stenosis (≥50%) by subsequent coronary CT angiography or coronary angiography, and 31% of them actually were treated with revascularization (Fig. 1). Therefore, it would be clinically worthwhile to screen patients with asymptomatic CAD (especially those who should be treated with revascularization) in the management of diabetes, although the utility in identifying severe but asymptomatic CAD is still debated.
First, we confirmed that maximum IMT was significantly associated with severe CAD equivalent to revascularization. Multiple logistic regression analyses showed that maximum IMT was an independent predictor of severe CAD in asymptomatic type 2 diabetic patients without history of CAD. Next, we evaluated the incremental effect of adding maximum IMT to the conventional risk factors for identifying severe CAD by ROC curves. The predictive abilities of CCA, carotid bulb ICA, and combined maximum IMT (CCA or carotid bulb ICA, whichever is greater) estimated by the AUC were 0.62, 0.72, and 0.74, respectively. These results suggest that carotid bulb ICA maximum IMT is more closely associated with severe CAD rather than CCA maximum IMT, and that combined maximum IMT most directly reflects it. Taken together, our findings are consistent with previous reports showing that the ICA IMT is more useful for predicting cardiovascular events than mean IMT (20). The AUC significantly increased after the addition of maximum IMT to conventional risk factors, suggesting that maximum IMT substantially improves the prediction ability of severe CAD in these patients (Fig. 2A).
Next, we investigated the cut-off level of maximum IMT for identifying the presence of severe CAD. The greatest sensitivity and specificity were obtained when the cut-off value of maximum IMT was set at 2.45 mm (Fig. 2B). When we limited CAD screening with exercise ECG or myocardial perfusion scintigraphy to the patients whose maximum IMT was above this cut-off level, the prevalence of severe CAD in the subjects who underwent CAD screening tests increased from 5% (pretest probability) to 11% (posttest probability), indicating that maximum IMT provided greater efficiency to detect severe CAD (Fig. 3A).
Furthermore, when we applied age-specific cut-off values of maximum IMT, the sensitivity of screening for the severe CAD further increased in both the nonelderly (pretest probability, 6%; posttest probability, 10%; and sensitivity, 100%) and the elderly subjects (pretest probability, 5%; posttest probability, 15%; sensitivity, 100%). The difference in cut-off levels of maximum IMT between the elderly and the nonelderly groups became >1.0 mm (2.65 and 1.55 mm, respectively), because maximum IMT of patients with severe CAD showed relatively high values with broad range (1.6–4.4 mm) in the nonelderly group but much higher values (>2.7 mm) in the elderly group in the current study. Interestingly, mean difference of maximum IMT between the elderly and the nonelderly group was calculated to be 0.91 mm in the patients with severe CAD but 0.37 mm in the patients without it, suggesting that the progression of maximum IMT had been more accelerated in those who had development of severe CAD. It also has been reported that patients with CAD have more rapid progression of IMT than patients without CAD and that conventional risk factors are related to progression in them (21).
The utility of maximum IMT in identifying severe CAD in asymptomatic type 2 diabetic subjects also was validated in another cohort of asymptomatic type 2 diabetic subjects. In this validation cohort, all the participants underwent coronary CT angiography. Although exercise ECG and myocardial perfusion scintigraphy are most commonly performed for detection of SMI, they have limited sensitivity and specificity in diagnosing of CAD (22,23). However, coronary CT angiography is highly accurate for the detection of CAD at an early stage, whereas it has the potential of significant adverse effects (24). Interestingly, the cut-off levels of maximum IMT and their performance in this cohort were comparable with those in the derivation cohort. These results support the utility of the measurement of maximum IMT and the validity of cut-off levels of maximum IMT proposed in the current study.
To the best of our knowledge, there are few reports that show a cut-off level of carotid IMT, and this is the first report showing the cut-off level of maximum IMT for identifying severe CAD.
Generally, excess body weight is regarded as one of the most important risk factors for CAD and can be a predictor for future CAD. In this study, however, the subjects with severe CAD showed significantly lower BMI than that in the subjects without severe CAD (Table 1). This phenomenon might be attributable to poor diabetic control in the subjects with severe CAD, because poor diabetic control causes body weight loss. Actually, these patients showed significantly higher HbA1c, irrespective of higher insulin treatment ratio, as compared with those without severe CAD.
Several limitation of our study should be discussed. First, this is a study with a relatively small number of subjects and there is a gender imbalance of the participants (out of 333 subjects, 71% were male). Furthermore, the actual number of the subjects included in the current study was approximately one-fourth the estimated number of the patients who met the eligibility criteria, which holds a certain risk of yielding some kind of selection biases. Therefore, the clinical utility of maximum IMT as a screening tool should be verified in studies with larger sample size. Especially, the cut-off level of maximum IMT should be confirmed by further studies.
Second, the utility of screening patients with type 2 diabetes for asymptomatic CAD is still controversial. Previous studies showed long-term safety and efficacy of intensive medication therapy and coronary revascularization in type 2 diabetic patients (25,26). However, many cardiologists are now skeptical of revascularization of asymptomatic type 2 diabetic patients, because the BARI2D study did not show any substantial difference between intensive medical therapy and revascularization, such as coronary artery bypass grafting or percutaneous coronary intervention in type 2 diabetic patients with stable CAD (26). Thus, it is still controversial regarding which patients benefit from coronary artery revascularization and how it is clinically important to identify severe but asymptomatic CAD. Therefore, it is unclear if a long-term clinical benefit was achieved in 17 subjects who had coronary revascularization in the current study. To evaluate whether carotid ultrasonography really leads to the improvement of difficult clinical outcomes (e.g., myocardial infarction), another trial should be performed.
Third, among 55 subjects with SMI, 6 patients rejected coronary CT angiography and coronary angiography. In this study, they were classified into the nonsevere CAD group, because our purpose was to examine whether carotid IMT is useful as a screening test for asymptomatic type 2 diabetic patients with severe CAD equivalent to revascularization therapy. However, the results of statistical analyses did not change even though these six patients were excluded.
Fourth, previous studies have shown that the natural history, patterns of risk factors, and the prediction of cardiac and cerebral events are different for “carotid plaque,” which is a focal structure encroaching into the arterial lumen, and the “traditional IMT,” which is measured in a region free of plaque, whereas both share some common atherosclerosis risk factors. In European countries, it is recommended that measurement of IMT should be performed in a region free of plaque (27) and the distinction between IMT and plaque should be clearly made. However, the Japan Society of Ultrasonics in Medicine recommends that measurement of IMT should be performed at the site of greatest thickness, including plaque lesions (28). A few studies performed in Japan adopted this method and reported that this IMT was useful as a marker for cardiovascular events and CAD (29). The current study was performed in Japan and the IMT was measured at the site of greatest thickness including plaque lesions according to Japanese guideline. As a result, in 88% cases of all measurements, maximum IMT represented plaque lesion. Thus, the definition of maximum IMT in our study and those of previous studies are different, and it would not be appropriate to make a simple comparison between our findings and previous ones. Interestingly, the results of our study indicates that maximum IMT including plaque lesion and the number of carotid plaques were both useful for identifying severe CAD (AUC, 0.74 and 0.74, respectively). On the contrary, even when measurement of mean IMT was performed, whether in a region free of plaque, the prediction ability of mean IMT was relatively poor (AUC, 0.66 and 0.64, respectively). These findings seem to be consistent with previous reports suggesting that presence of plaque was more strongly associated with cardiovascular events than “traditional IMT” excluding plaque lesion (30).
Fifth, although carotid ultrasonography can be performed noninvasively and relatively inexpensively, its use for universal screening in type 2 diabetic patients is not feasible. Therefore, the kind of diabetic population that should be given preference in receiving this clinical examination also must be clarified in further studies.
Finally, it is noted that the subjects of this study were Japanese type 2 diabetic patients. Thus, it would be premature to generalize our findings to other race or ethnic groups and nondiabetic subjects.
Notwithstanding these limitations, our study indicates that measurement of carotid maximum IMT can provide useful information for identifying subjects at high risk for CAD.
In conclusion, our study suggests that carotid maximum IMT is useful for screening asymptomatic type 2 diabetic patients with severe CAD equivalent to revascularization.
Acknowledgments
No potential conflicts of interest relevant to this article were reported.
Y.I., N.K., H.K., M.N., R.K., K.S., Y.Um., and S.S. researched data. Y.I., N.K., and H.K. wrote the manuscript. Y.Ue., K.K., and I.S. contributed to the interpretation of the results and the discussion. N.K. had full access to the data and takes full responsibility for this work as a whole, including the study design and the decision to submit and publish the manuscript.
Footnotes
This article contains Supplementary Data online at http://care.diabetesjournals.org/lookup/suppl/doi:10.2337/dc12-1327/-/DC1.
References
- 1.Nesto RW, Phillips RT. Asymptomatic myocardial ischemia in diabetic patients. Am J Med 1986;80(4C):40–47 [DOI] [PubMed] [Google Scholar]
- 2.Wackers FJ, Young LH, Inzucchi SE, et al. Detection of Ischemia in Asymptomatic Diabetics Investigators. Detection of silent myocardial ischemia in asymptomatic diabetic subjects: the DIAD study. Diabetes Care 2004;27:1954–1961 [DOI] [PubMed] [Google Scholar]
- 3.van Dieren S, Peelen LM, Nöthlings U, et al. External validation of the UK Prospective Diabetes Study (UKPDS) risk engine in patients with type 2 diabetes. Diabetologia 2011;54:264–270 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Simmons RK, Coleman RL, Price HC, et al. Performance of the UK Prospective Diabetes Study Risk Engine and the Framingham Risk Equations in Estimating Cardiovascular Disease in the EPIC- Norfolk Cohort. Diabetes Care 2009;32:708–713 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Stephens JW, Ambler G, Vallance P, Betteridge DJ, Humphries SE, Hurel SJ. Cardiovascular risk and diabetes. Are the methods of risk prediction satisfactory? Eur J Cardiovasc Prev Rehabil 2004;11:521–528 [DOI] [PubMed] [Google Scholar]
- 6.O’Leary DH, Polak JF, Kronmal RA, Manolio TA, Burke GL, Wolfson SK, Jr, Cardiovascular Health Study Collaborative Research Group Carotid-artery intima and media thickness as a risk factor for myocardial infarction and stroke in older adults. N Engl J Med 1999;340:14–22 [DOI] [PubMed] [Google Scholar]
- 7.Irie Y, Katakami N, Kaneto H, et al. Maximum carotid intima-media thickness improves the prediction ability of coronary artery stenosis in type 2 diabetic patients without history of coronary artery disease. Atherosclerosis 2012;221:438–444 [DOI] [PubMed] [Google Scholar]
- 8.Nambi V, Chambless L, Folsom AR, et al. Carotid intima-media thickness and presence or absence of plaque improves prediction of coronary heart disease risk: the ARIC (Atherosclerosis Risk In Communities) study. J Am Coll Cardiol 2010;55:1600–1607 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kasami R, Kaneto H, Katakami N, et al. Relationship between carotid intima-media thickness and the presence and extent of coronary stenosis in type 2 diabetic patients with carotid atherosclerosis but without history of coronary artery disease. Diabetes Care 2011;34:468–470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Japan Diabetes Society. Diabetes mellitus treatment guideline based on scientific ground (revised edition 2). Nankodo: Japan Diabetes Society; 2007:257–272 [Google Scholar]
- 11.Katakami N, Kaneto H, Osonoi T, et al. Usefulness of lipoprotein ratios in assessing carotid atherosclerosis in Japanese type 2 diabetic patients. Atherosclerosis 2011;214:442–447 [DOI] [PubMed] [Google Scholar]
- 12.Smith SC, Jr, Dove JT, Jacobs AK, et al. American College of Cardiology. American Heart Association Task Force on Practice Guidelines. Committee to Revise the 1993 Guidelines for Percutaneous Transluminal Coronary Angioplasty ACC/AHA guidelines of percutaneous coronary interventions (revision of the 1993 PTCA guidelines)—executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (committee to revise the 1993 guidelines for percutaneous transluminal coronary angioplasty). J Am Coll Cardiol 2001;37:2215–2239 [DOI] [PubMed] [Google Scholar]
- 13.Thaulow E, Erikssen J, Sandvik L, Erikssen G, Jorgensen L, Cohn PF. Initial clinical presentation of cardiac disease in asymptomatic men with silent myocardial ischemia and angiographically documented coronary artery disease (the Oslo Ischemia Study). Am J Cardiol 1993;72:629–633 [DOI] [PubMed] [Google Scholar]
- 14.Froelicher VF, Callaham PR, Angelo J, Lehmann KG. Treadmill exercise testing and silent myocardial ischemia. Isr J Med Sci 1989;25:495–502 [PubMed] [Google Scholar]
- 15.Cohn PF. Silent myocardial ischemia. Ann Intern Med 1988;109:312–317 [DOI] [PubMed] [Google Scholar]
- 16.Bacci S, Villella M, Villella A, et al. Screening for silent myocardial ischaemia in type 2 diabetic patients with additional atherogenic risk factors: applicability and accuracy of the exercise stress test. Eur J Endocrinol 2002;147:649–654 [DOI] [PubMed] [Google Scholar]
- 17.Naka M, Hiramatsu K, Aizawa T, et al. Silent myocardial ischemia in patients with non-insulin-dependent diabetes mellitus as judged by treadmill exercise testing and coronary angiography. Am Heart J 1992;123:46–53 [DOI] [PubMed] [Google Scholar]
- 18.Milan Study on Atherosclerosis and Diabetes (MiSAD) Group Prevalence of unrecognized silent myocardial ischemia and its association with atherosclerotic risk factors in noninsulin-dependent diabetes mellitus. Milan Study on Atherosclerosis and Diabetes (MiSAD) Group. Am J Cardiol 1997;79:134–139 [DOI] [PubMed] [Google Scholar]
- 19.Janand-Delenne B, Savin B, Habib G, Bory M, Vague P, Lassmann-Vague V. Silent myocardial ischemia in patients with diabetes: who to screen. Diabetes Care 1999;22:1396–1400 [DOI] [PubMed] [Google Scholar]
- 20.O’Leary DH, Polak JF, Kronmal RA, et al. The CHS Collaborative Research Group Distribution and correlates of sonographically detected carotid artery disease in the Cardiovascular Health Study. Stroke 1992;23:1752–1760 [DOI] [PubMed] [Google Scholar]
- 21.Crouse JR, 3rd, Tang R, Espeland MA, Terry JG, Morgan T, Mercuri M. Associations of extracranial carotid atherosclerosis progression with coronary status and risk factors in patients with and without coronary artery disease. Circulation 2002;106:2061–2066 [DOI] [PubMed] [Google Scholar]
- 22.Iwase M, Fukui M, Tamagaki H, et al. Advantages and disadvantages of dobutamine stress echocardiography compared with treadmill exercise electrocardiography in detecting ischemia. Jpn Circ J 1996;60:954–960 [DOI] [PubMed] [Google Scholar]
- 23.Gibbons RJ, Balady GJ, Bricker JT, et al. American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Committee to Update the 1997 Exercise Testing Guidelines ACC/AHA 2002 guideline update for exercise testing: summary article. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Update the 1997 Exercise Testing Guidelines). J Am Coll Cardiol 2002;40:1531–1540 [DOI] [PubMed] [Google Scholar]
- 24.Schroeder S, Achenbach S, Bengel F, et al. Working Group Nuclear Cardiology and Cardiac CT. European Society of Cardiology. European Council of Nuclear Cardiology Cardiac computed tomography: indications, applications, limitations, and training requirements: report of a Writing Group deployed by the Working Group Nuclear Cardiology and Cardiac CT of the European Society of Cardiology and the European Council of Nuclear Cardiology. Eur Heart J 2008;29:531–556 [DOI] [PubMed] [Google Scholar]
- 25.Kapur A, Hall RJ, Malik IS, et al. Randomized comparison of percutaneous coronary intervention with coronary artery bypass grafting in diabetic patients. 1-year results of the CARDia (Coronary Artery Revascularization in Diabetes) trial. J Am Coll Cardiol 2010;55:432–440 [DOI] [PubMed] [Google Scholar]
- 26.Frye RL, August P, Brooks MM, et al. BARI 2D Study Group A randomized trial of therapies for type 2 diabetes and coronary artery disease. N Engl J Med 2009;360:2503–2515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Touboul PJ, Hennerici MG, Meairs S, et al. Mannheim carotid intima-media thickness consensus (2004-2006). An update on behalf of the Advisory Board of the 3rd and 4th Watching the Risk Symposium, 13th and 15th European Stroke Conferences, Mannheim, Germany, 2004, and Brussels, Belgium, 2006. Cerebrovasc Dis 2007;23:75–80 [DOI] [PubMed] [Google Scholar]
- 28.Terminology and Diagnostic Criteria Committee, Japan Society of Ultrasonics in Medicine Subcommittee for Preparing Guidelines for Ultrasound Diagnosis of Carotid Artery. Standard method for ultrasound evaluation of carotid artery lesions. Jpn J Med Ultrasonics 2009;36:501–518 [Google Scholar]
- 29.Mitsuhashi N, Onuma T, Kubo S, Takayanagi N, Honda M, Kawamori R. Coronary artery disease and carotid artery intima-media thickness in Japanese type 2 diabetic patients. Diabetes Care 2002;25:1308–1312 [DOI] [PubMed] [Google Scholar]
- 30.Salonen JT, Salonen R. Ultrasonographically assessed carotid morphology and the risk of coronary heart disease. Arterioscler Thromb 1991;11:1245–1249 [DOI] [PubMed] [Google Scholar]