Abstract
Obesity and its related metabolic diseases have reached a pandemic level worldwide. There are sex differences in the prevalence of obesity and its related metabolic diseases, with men being more vulnerable than women; however, the prevalence of these disorders increases dramatically in women after menopause, suggesting that sex steroid hormone estrogens play key protective roles against development of obesity and metabolic diseases. Estrogens are important regulators of several aspects of metabolism, including body weight and body fat, caloric intake and energy expenditure, and glucose and lipid metabolism in both males and females. Estrogens act in complex ways on their nuclear estrogen receptors (ERs) ERα and ERβ and transmembrane ERs such as G protein-coupled estrogen receptor. Genetic tools, such as different lines of knockout mouse models, and pharmacological agents, such as selective agonists and antagonists, are available to study function and signaling mechanisms of ERs. We provide an overview of the evidence for the physiological and cellular actions of ERs in estrogen-dependent processes in the context of energy homeostasis and body fat regulation and discuss its pathology that leads to obesity and related metabolic states.
1. INTRODUCTION
1.1. Obesity is a risk factor for metabolic diseases
Despite attempts to increase awareness of the deleterious health consequences associated with obesity, there continues to be a significant increase in the obese population worldwide.1 Obesity has been recognized as a major and an independent risk factor for metabolic syndrome, a group of interrelated metabolic abnormalities characterized by increased visceral fat accumulation, disturbed glucose homeostasis with hyperglycemia and insulin resistance, increased triglycerides and decreased high-density lipoprotein cholesterol, and hypertension.2 The economic costs of caring for patients with obesity and its metabolic complications are enormously high.3 Although the pathophysiologic basis of obesity and metabolic syndrome is largely unknown, evidence suggests that modern diets with high fat content and sedentary lifestyle progressively lead to the accumulation of saturated fatty acids in nonadipose tissue. The subsequent metabolism of this ectopic fatty acid deposition leads to decreased insulin-stimulated glucose uptake in skeletal muscle, unsuppressed hepatic glucose production, and altered glucose-stimulated insulin release from pancreatic β-cells.4 Once diabetes is declared, hyperglycemia and elevated free fatty acids cooperate to generate major oxidative stress in tissues, further aggravating insulin resistance and deficiency.
Development of adverse metabolic complications has been attributed to increased body fat, not just body weight, and the fat distributed particularly in the abdominal visceral compartment, a source of bioactive mediators that directly contribute to metabolic disorders.5 Intra-abdominal visceral adipose tissue carries a greater risk for the development of metabolic disorders than subcutaneous adipose tissue does. Therefore, body composition and body fat distribution are closely associated with metabolic state.
1.2. Sex differences in the development of obesity and metabolic diseases
There are important sex differences in the prevalence of obesity and metabolic diseases. The prevalence of obesity and its related metabolic diseases is particularly high among men and middle-aged women. Prior to menopause, women have much less obesity-related metabolic disorders than obese men. For example, hypercholesterolemia is more common in males, whose lipid profile is characterized by lower HDL, higher LDL and triglyceride levels, and larger VLDL particles. The incidence of liver steatosis is also higher in men.6 After menopause, however, women are at an increased risk of developing visceral obesity and metabolic syndrome7 due to the loss of - endogenous ovarian hormone production.
Risk for metabolic syndrome incidence generally relates more to the central distribution of fat with abdominal, omental, and visceral adiposity than to the total amount of fat, with individuals with large amounts of abdominal visceral fat having particularly high risk.8,9 Adipose tissue accumulation is sexually dimorphic.10 The amount and regional distribution of body fat is different between males and females, as males and females differ in terms of how and where they store body fat. Premenopausal women usually have a higher amount of body fat and more subcutaneous gluteal/femoral pattern of fat distribution, whereas men have a leaner body composition and have more fat is stored in the central intra-abdominal visceral depot.11,12 Because visceral fat is more likely to cause metabolic syndromes, incidence of obesity-related metabolic disorders is much lower in premenopausal women than men and women experiencing menopause.
1.3. Sex steroid estrogens play key roles in energy homeostasis and glucose regulation
1.3.1 Estrogens regulate energy homeostasis and body fat
The epidemiological and clinical evidence strongly suggests a major role for estrogens in the regulation of body weight and body fat. It is now evident that estrogens account for sexual dimorphism in body fat distribution and accretion. The differences in the fat distribution between premenopausal women and age-matched men are abolished between postmenopausal women and age-matched men.13 Estrogens exert important antiobesity effects in females. Postmenopausal women with decreased levels of estrogens are associated with an increased risk for developing obesity.14 Additionally, estrogens promote the accumulation of subcutaneous fat15 while visceral fat varies inversely with levels of estrogens.16 Consequently, the loss of endogenous estrogen production with menopause is associated with an increased risk to develop visceral obesity and symptoms of the metabolic syndrome.9 The increased visceral obesity in menopause women can be altered by exogenous hormone replacement therapy.17,18
Studies using experimental animal models have provided important insights into the pathogenesis of human diseases and new therapeutic approaches. While metabolic diseases and obesity have been shown in a number of animal species, in the laboratory they are most commonly investigated in two species of rodents, rats (Rattus norvegicus) and mice (Mus musculus/Mus domesticus).
Reductions in circulating estrogens and estrogen signaling, which occurs following removal of ovaries (i.e., ovariectomy), transiently increase caloric intake, and result in increased adiposity,19 specifically visceral fat with no change of subcutaneous fat.20 Interestingly, the transient hyperphagia induced by ovariectomy does not account for the development of obesity,21 as ovariectomized rats gain weight to a similar extent even when they are pair-fed to 17β–estradiol (E2)-treated rats,22 suggesting that endogenous estrogens regulate body weight homeostasis primarily by modulating energy expenditure.
E2 administration decreases energy intake and increases energy expenditure, 23 indicating that exogenous estrogens may promote a negative energy balance by influencing both energy intake and energy expenditure. The ovariectomy-induced increase in adiposity can be ameliorated by exogenous E2 administration.20 Furthermore, peripheral or central administration of E2 to ovariectomized rats changes their body fat distribution to mirror that of intact females. Additionally, altering the sex hormone milieu in males with E2 administration increases subcutaneous fat deposition.20 An important implication from these findings is that estrogens are critical determinates of body fat distribution and energy balance.
1.3.2 Estrogens regulate lipid metabolism
Epidemiological and prospective studies associate estrogens to several aspects of the metabolic syndrome (Fig. 6.1). There is increasing evidence both in humans and laboratory rodents suggesting that estrogens are not only involved in regulating adiposity and body fat distribution, but also directly involved in regulation of glucose and lipid metabolism. Thus, estrogen action seems to be implicated in adipose tissue regulation and prevention of obesity.
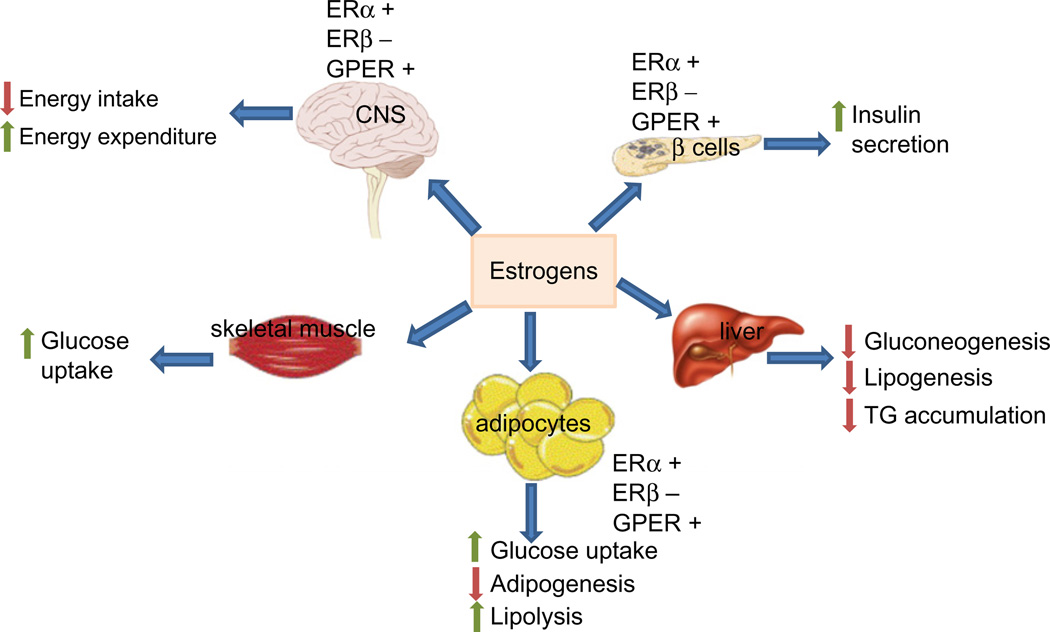
Figure 6.1.
Overview of estrogenic control of energy homeostasis, glucose, and lipid metabolism by regulatory actions in the CNS, adipose tissue, pancreatic β cells, skeletal muscles, and liver. Deficiency in estrogenic action leads to opposite events and subsequent disturbances in metabolic tissues. ERα, ERβ, and GPER play roles to regulate energy balance and glucose homeostasis. ERα and ERβ generally mediate opposing effects, while GPER generally has similar roles as ERα in the brain, adipose tissues, and pancreas.
Estrogens suppress lipogenesis in adipose tissue, skeletal muscle, and liver.24,25 In order to understand the role of estrogens in liver, studies evaluated the consequences of hormone replacement therapy to postmenopausal women and the effects of estrogen-suppression therapy in breast cancer patients. Estrogens increase HDL and triglycerides, decrease LDL and total cholesterol, and decrease fasting insulin and glucose levels, suggesting beneficial effects of estrogen on lipid and glucose metabolism.26 Recent studies demonstrate that the use of antiestrogen therapy, such as tamoxifen and ovariectomy, leads to abnormal lipid profile and liver steatosis.27 Furthermore, studies reported that islet activation of estrogen receptors (ERs), estrogen receptor alpha (ERα), estrogen receptor beta (ERβ), and G protein-coupled ER (GPER) by estrogens suppresses fatty acid synthase expression and enzymatic activity.28 This effect reduces islet lipid accumulation in rodent and human β-cells and prevents β-cell failure in rodent models of type 2 diabetes.
Another essential argument for estrogens’ antilipogenic roles on lipid metabolism comes from the study of men with estrogen deficiency. In both sexes, estrogens are biosynthesized by the cytochrome P450 enzyme complex called aromatase.29 In men, estrogen secretion comes principally from the conversion of androgens in adipose tissue by aromatase. The aromatase-deficient patients, who cannot synthesize estrogens thus lack endogenous estrogen secretion secondary to missense mutations in the aromatase gene, show impaired liver functions, hepatic steatosis, and altered lipid profile,30 as well as develop hypertriglyceridemia and insulin resistance.31,32 These metabolic abnormalities are improved or corrected by estrogen replacement therapy.30,32,33 In the E2-deficient aromatase knockout (ArKO) mice, insulin resistance develops due to hepatic steatosis.34
1.4. Focus of this chapter
In both humans and rodent models, estrogens are important sex hormones central to health in both genders that have protective effects in many physiological processes in mammals, including but not limited to reproduction, cardiovascular health, metabolic systems, bone integrity, cognition, and behavior.35 Given these widespread roles for estrogens, it is not surprising that estrogens are implicated in the development or progression of numerous diseases, which include but are not limited to obesity, insulin resistance, osteoporosis, cardiovascular disease, various types of cancer of reproductive tissues, and neurodegenerative diseases. Pertinent to this chapter is estrogens’ regulation in energy homeostasis, glucose and lipid metabolism, and insulin sensitivity (Fig. 6.1). Estrogens are protective against the development of visceral obesity and glucose intolerance, and thus contribute to the maintenance of insulin sensitivity. Deficiency in estrogen leads to development of visceral obesity and insulin resistance.
We discuss in this chapter the mechanisms of action by estrogens in metabolic processes, focusing on the central nervous system (CNS) and adipose tissues. We first introduce the estrogenic system, including the estrogens and their receptors. We then discuss the current knowledge and questions about estrogenic action in the brain and adipose tissue, and how these processes are involved in the regulation of energy homeostasis and lipid metabolism. We review studies that have investigated the roles of estrogens and their receptors in the regulation of such metabolism, focusing on GPER-dependent signaling and function. Controversies that have complicated our understanding of GPER, including interactions with nuclear and transmembrane ERs, will also be discussed.
2. ESTROGEN SIGNALING: ESTROGENS AND ERs
2.1. Estrogenic action through genomic and nongenomic signaling mechanisms
Estrogens are the best-characterized member of the family of steroid hormones that includes progesterone, testosterone, glucocorticoids, and mineralocorticoids. Human estrogens comprise a group of structurally related steroid molecules, namely 17β-estradiol (E2), estrone, and estriol, which are the most important regulators of the female and male reproductive systems. Estrogens are synthesized from cholesterol by aromatase via a series of chemical reactions that are part of the steroidogenic pathways predominantly in the ovaries. Estrogens are important hormones in mammalian physiology exerting important functions on the regulation of female secondary sexual characteristics. In males, relatively small amount of estrogens are produced by Leydig and germ cells of the testis. Additionally, estrogens are synthesized at multiple discrete sites throughout the body from the liver, adrenal glands, and adipocytes,36 where they have highly localized effects on various unrelated processes in many tissues.37 Estrogens also interact with and exert regulatory effects in a variety of tissues in the body that do not secrete estrogen, including the CNS, cardiovascular system, immune system, breast, uterus, and bone.38–42
Estrogenic actions are mediated by a number of different ERs (Fig. 6.2). The highly hydrophobic nature of estrogens allows them to pass through cellular membranes by passive diffusion43 and also allows its concentration in cellular membranes.44 Cellular responses to estrogens are mediated by receptors that initiate a complex array of cellular events upon ligand binding. These responses are divided into two broad categories. One is genomic responses that are characterized by changes in gene transcription and occur on the time frame of hours to days after estrogens bind to nuclear ERS, ERα and ERβ. The other one is nongenomic rapid signaling events that occur within seconds to minutes of cell stimulation after estrogens bind to novel GPER45 which is localized to the endoplasmic reticulum, membrane subpopulation ERs (mERα/β), or G protein-coupled receptor membrane ERs (Gq-mER), and mediates nongenomic estrogen signaling via cytosolic pathways involving second messengers.45,46 Thus, an organism’s responses to estrogens are generally the result of a complex interaction between genomic and nongenomic signaling (Fig. 6.2). The genomic mechanisms are relatively well characterized, while the nongenomic estrogenic signaling is less well understood and is beginning to be explored.
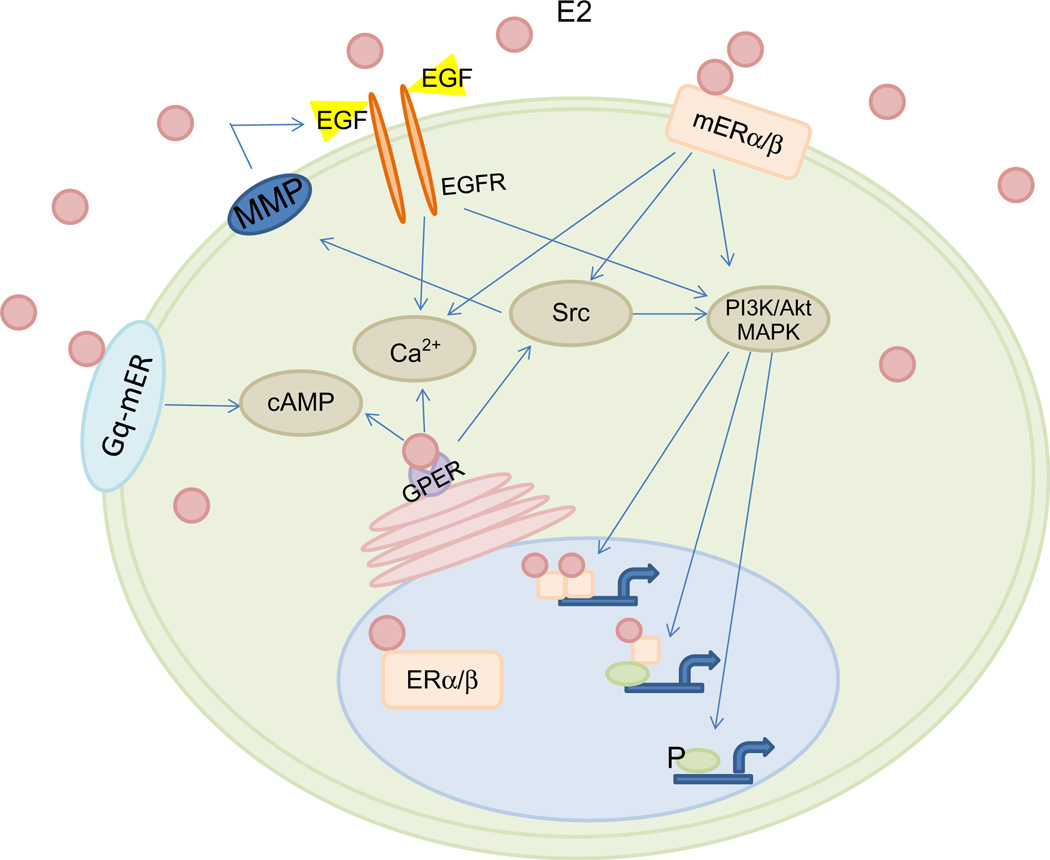
Figure 6.2.
Schematic overview of estradiol-mediated genomic signaling pathway via ERα/β and rapid nongenomic signaling pathways via GPER, Gq-mER, and membrane subpopulation mERα/β.
2.2. Estrogenic genomic actions via nuclear ERs
Estrogens activate their nuclear ERs in target cells, acting as transcription factors to regulate the expression of target genes, ultimately controlling cell growth, differentiation, and homeostasis.47 Two nuclear ERs located on distinct chromosomes have been identified.48–51 The first described classical ER, today known as ERα, was discovered and characterized in 1973, on the basis of specific binding activity in rat uterus/vagina extracts.52 The classical nuclear ER was cloned in 1985,51 its DNA sequence was determined in 1986,50 and the first crystal structure of an ER ligand-binding domain was described in 1997.53 ER was renamed ERα when a second nuclear ER, ERβ, was identified 10 years later.49
The major physiological estrogen is E2 that has a similar affinity for both ERs. Pharmacological reagents have been developed that permit the investigation of the specific role of ERs. ERs are activated by a range of ligands, including selective ER modulators (SERM) such as raloxifene and tamoxifen; the ERα-selective agonist 4,4′,4″-(4-propyl-[1H]-pyrazole-1,3,5-triyl) trisphenol (PPT),which has 410 times greater affinity for ERα than ERβ54; and two commonly used ERβ agonists, 2,3-bis(4-Hydroxyphenyl)-propionitrile (DPN) and 7-Bromo-2-(4-hydroxyphenyl)-1,3-benzoxazol-5-ol (WAY200070), which show similar receptor specificity and neither is as selective for ERβ as PPT is for ERα. DPN is 70-fold more selective for ERβ than ERα,55 while the affinity of WAY200070 (compound 92) for ERβ in mice is 68 times that for ERα.56
Estrogens’ genomic action on cell functions is via the activation of ERα and ERβ, both of which belong to the nuclear hormone receptor family and are transcription factors. They have classically been described as ligand-activated transcription factors mediating long-term genomic effects in central and peripheral estrogen-regulated tissues. ERα and ERβ exist primarily in the nucleus, where they are complexed with chaperones. Briefly, binding of estrogens to ERα and ERβ leads to conformational changes, chaperone dissociation from their inhibitory protein complexes, and dimerization of the receptor, followed by receptor translocation to the nucleus to recognize and binding by receptor dimers to cis-acting estrogen response elements (EREs) on the promoters of hormonally regulated genes, where the recruitment of coactivators and/or corepressors leads to transcription of important estrogen responsive genes and alterations in the rate of gene expression.57–59 Estrogen also modulates gene expression by a non-ERE-mediated mechanism in which ERα and ERβ dimers bind to other non-ERE promoter sites and interact with other transcription factors through protein–protein interactions, such as activating protein-1 and stimulating protein-1, Fos-Jun, and a number of coactivators to regulate estrogenic genomic actions, a process referred to as transcription factor cross talk. Furthermore, the ERs have some ligand-independent transcriptional responses and can also interact with other transcription factors in regulating gene expression.60
Although ERα and ERβ are highly homologous in their DNA- and ligand-binding domains, ERα and ERβ lack homology in their transcriptional activation domains and their splice variants differ in their affinities for E2.60,61 Additionally, ERα and ERβ differ in the tissue distribution.62 ERα is mainly expressed in reproductive tissues, kidney, bone, white adipose tissue, and liver, whereas ERβ is expressed in the ovary, prostate, lung, gastrointestinal tract, bladder, hematopoietic cells, and the CNS.63 These differences suggest that functional differences exist between the two subtypes ERα and ERβ, which is supported by characterization of ERα and ERβKO mice.64
Male and female ERα KO mice are diabetogenic and obese with severe hepatic insulin resistance induced by targeted deletion in the ERα gene.65 Thus, ERα is believed to mediate most estrogenic effects on energy homeostasis and glucose regulation. The cause of obesity in ERα KO mice is unclear; however, overeating does not appear to be essential,65–68 and the increased adiposity in ERα KO mice is attributed primarily to decreased energy expenditure.65,67 While E2 replacement prevents ovariectomized wild type mice from developing hyperphagia and obesity, such protective effects are blocked in ovariectomized ERα KO mice.66 Therefore, although no hyperphagia is observed in ERα KO mice,65–67 ERα is clearly required to mediate normal satiation process because E2-induced hypophagia and CCK-induced satiation in wild type mice are blocked in ERα KO mice.66
The role of ERβ in the central regulation of feeding is less known. In ovariectomized rats, there is increased food intake, body weight, and abdominal fat accumulation, which can be reversed by E2. However, intracerebroventricular administration of E2 together with ERβ antisense oligodeoxynucleotides abrogates the anorexic effects of E2. Ovariectomy of ERβ KO mice leads to normalized homeostasis of circulating glucose and insulin levels and reverses the obese phenotype, suggesting that ERβ activity may result in a diabetogenic and adipogenic phenotype, which is opposite to the effects of ERα. Additionally, ERβ KO mice display improved insulin sensitivity and glucose tolerance without increased body fat content, suggesting that ERβ also plays an important role in maintaining metabolic control.69 These studies suggest opposite effects of E2 via ERα and ERβ.70
2.3. Estrogenic nongenomic actions via GPER
Estrogens are best known for their long-term transcriptional effects involving the genomic mechanism of action that typically takes several hours for the effect to be manifested due to the time needed for transcription and translation of estrogen-regulated genes.71 However, a growing body of evidence has emerged to describe rapid estrogen signal transduction events in various cellular systems,72,73 which occur within minutes and thus cannot be attributed to a genomic mechanism of action. These rapid effects have thus been designated as nongenomic effects of estrogen.38,74–78 The earliest reports for nongenomic effects of estrogens date back to 1967.79 In 1977, Pietras and Szego discovered that estrogens bind to a cell surface receptor,80 introducing the possibility that estrogenic signaling may involve nongenomic nontranscriptional mechanisms or indirect actions to influence gene expression.
Membrane-bound ERs have been described recently. GPER, previously known as G protein-coupled receptor 30 (GPR30), has been identified as an estrogen-binding intracellular membrane receptor.45,81–84 GPER expression is required for rapid cellular responses to E2.85,86 In addition, another estradiol-responsive, G protein-coupled membrane ER has been described (Gq-mER).87 Even though its distribution is currently unknown, Gq-mER appears to be involved in maintaining homeostatic functions by reducing food intake and fat accumulation, as well as other functions such as increasing bone density.88–90 Finally, in 2002, the existence of an additional mER, named ER-X, was hypothesized.91 However, its gene sequence and amino acid structure are currently unknown.
It is noteworthy that membrane ERs have been the focus of a surge of studies over the past decade.92 A search in PubMed in May 2012 with the keywords “estrogen and nongenomic” yields 576 published papers since 1984, with 480 (83%) of these papers being published after 2000. This area has seen an explosion of interest in the past decade, and represents one of the fastest emerging areas in the field of estrogen research.
2.3.1 GPER distribution and expression in tissues
GPER is a seven transmembrane-domain G protein-coupled receptor. GPER was first identified and cloned by Carmechi et al. in 1997.81 Using a differential cDNA library screening technique, the investigators identified GPER as a differentially expressed gene in a screen of genes from an ER-positive breast cancer cell line (MCF-7) versus an ER-negative breast cancer cell line (MDA-MB-361).
Antibodies have been raised against GPER, facilitating a detailed analysis of its tissue distribution. Expression of GPER protein is not restricted to traditionally estrogen responsive tissues. Indeed, characterization of GPER using immunohistochemical analysis revealed ubiquitous expression of the receptor in normal and malignant human tissues. GPER expression is tissue and cell type specific, with high levels of expression found in numerous human organs, including male and female reproductive systems, heart, intestine, ovary, CNS, pancreatic islets, adipose tissue, neurons, and inflammatory cells. GPER is moderately positive at skeletal/cardiac muscle, but is negative at most smooth muscle.83,93–105 GPER is expressed in many regions of the brain with high expression noted in the hypothalamic–pituitary axis, hippocampus, brainstem autonomic nuclei, cortex, and striatum.106 Several primary breast cancers81 and lymphomas107 also expressed GPER transcripts, although many others were negative. There are discrepancies in the reported expression levels in some tissues, such as the rodent embryonic and fetal tissues, placenta, lung, and liver.81,107,108 Therefore a thorough analysis of the regulation of GPER expression remains to be completed.
GPER expression is developmentally regulated during the development of murine female reproductive tissues. For example, in the mammary gland, GPER expression in elongating ducts is low during puberty but rises once sexual maturity is reached. GPER expression in the mammary ductal epithelia exhibits estrous-cycle-dependent changes and differentiation-dependent changes during pregnancy, lactation, and involution. Similarly, GPER expression in both luminal and glandular uterine epithelia is regulated during the estrous cycle. Finally, Roy and colleagues recently demonstrated that the distribution and level of expression of GPER in the hamster ovary also varies with the estrous cycle; both granulosa and theca cell layers are rich in GPER, and expression is reduced at estrous.109
Interestingly, sexual dimorphisms for GPER expression and/or function have been described in the brain110 and in the pancreatic islets, where it is expressed at a much higher level in women than in men.95 Accordingly, a role for GPER in the regulation of obesity-associated metabolic functions has been proposed.
These studies have provided a framework in which to interpret many of the reported functions of GPER, and suggest that GPER has been implicated to be involved in estrogen-dependent physiology of immune function and cardiovascular systems.47,93 For example, GPER is associated with diseases such as obesity, insulin resistance, and hormone-sensitive cancers,93,98,99 and GPER plays a role in estrogen-sensitive tumor growth in the absence of ERα/β.81,111
2.3.2 GPER location in cells
GPER is expected to localize in the plasma membrane and its plasma membrane localization is reported by Thomas et al.84 However, other groups reported GPER locations in the membrane of intracellular compartments. Revankar et al. used novel fluorescent estrogen derivatives (E2-Alexas) to directly visualize the cellular and subcellular binding properties of GPER. Confocal microscopy revealed that, whereas E2-Alexas detected ERα and ERβ in the nucleus of cells, E2-Alexa binding to GPER occurred predominantly in the endoplasmic reticulum, with no detectable binding at the plasma membrane.45 In addition, binding of the fluorescent E2-Alexas was directly proportional to expression of endogenous GPER on a cell-by-cell basis, strongly supporting the idea that expression of GPER protein directly generates estrogen-binding sites. In all cell lines where GPER expression has been observed, the E2-Alexa or antibody staining is colocalized in the endoplasmic reticulum, with staining also present in the Golgi apparatus and nuclear membrane. These results, although they are in contrast to the expected localization of GPER in the plasma membrane, clearly demonstrate that GPER is localized in the membrane of intracellular compartments, such as the endoplasmic reticulum and the Golgi complex.45,81,83–85,93
2.3.3 GPER intracellular signaling
GPER binds E2 with high affinity to mediate subsequent nongenomic and rapid estrogen signaling via multiple pathways (Fig. 6.2).45,83–85 As a G protein-coupled receptor, GPER couples to G proteins and modulates second messenger pathways. Signaling mechanisms of this action include rapid increases in Ca2+,112 cAMP79,113 and phosphoinositide-3 kinase (PI3K)/Akt and mitogen-activated protein kinase (MAPK) activation47 (Fig. 6.2). cAMP and Ca2+ have emerged as the second messengers most frequently associated with GPER signaling. GPER is involved in the activation of downstream signaling cascades such as protein kinase A (PKA), protein kinase C, and MAPK via membrane-localized ERs82 and intracellular Ca2+ regulation.106 The GPER couples to both a Gαs protein yielding stimulation of cyclic AMP (cAMP) production and a pertussis toxin (PTX)- sensitive Gαi/o protein in part yielding attenuation of cAMP production. Ding et al.114 have reported that E2-promoted vascular smooth muscle cell apoptosis is dependent on GPER-mediated PKA inhibition and PI3K activation, both of which are PTX sensitive. Furthermore, Maggiolini et al.86 demonstrate GPER-mediated and PTX-sensitive c-fos upregulation in SkBr3 breast cancer cells, a cell line that has been shown to have GPER-dependent stimulation of cAMP production.82
The rapid effects from stimulation of GPER also include release of intracellular Ca2+. This signal has been described as phospholipase C-independent, 45 PTX-sensitive,45,105 and dependent on epidermal growth factor receptor (EGFR) activation.45 Increased cytosolic Ca2+ subsequently activates Ca2+–calmodulin-dependent kinases or activation of MAPK and PI3K pathways.
Filardo et al. demonstrated that in MCF-7 cells, which express ERα, ERβ, and GPER, estrogen mediated a rapid increase in MAPK phosphorylation but that in MDA-MB-231 breast cancer cells, which express ERβ but not ERα or GPER, this response was absent, suggesting a possible role for ERα or GPER. When MDA-MB-231 cells were transfected to express GPER, E2 stimulated both cAMP production and PTX-sensitive MAPK activation in the cells.85 Additionally, they identified that GPER is involved in the estrogen-mediated activation of MAPK82,85,115 and PI3K45 in SKBr3 breast cancer-derived cells that express only GPER but lack classical nuclear ERα and ERβ. Together, these results strongly implicated GPER is essential in estrogen-mediated cellular responses.
Stimulation of GPER also leads to PI3K and PTX-sensitive Src activation, which in turn yields phosphorylation of Akt and SHC, respectively. While Akt phosphorylation has been linked to antiapoptotic and cardioprotective effects,116 SHC phosphorylation triggers cleavage of membrane-tethered heparin-bound EGF, presumably through activation of a metalloproteinase, resulting in transactivation of the EGF receptor.85 However, there is a debate on whether the PI3K/Akt pathway lies downstream of Gαi/o or not. While several groups have shown that the GPER-coupled Src/SHC/EGF/MAPK pathway is PTX sensitive,85,114 and thus dependent on Gαi/o signaling, Revankar et al.45 reported that PI3K/Akt activation is downstream of the Src/SHC/EGF/MAPK pathway but insensitive to PTX.
2.3.4 Pharmacologic tools—GPER agonists and antagonists
Elucidation of the biological roles of GPER has been facilitated by the identification of specific pharmacological reagents. GPER agonists, including G-1, (±)-1-[(3aR*, 4S*,9bS*)-4-(6-Bromo-1,3-benzodioxol-5-yl)-3a,4,5,9b-tetrahydro-3H-cyclopenta[c]quinolin-8-yl]-ethanone,117 and 5408–0877, which behaved similarly to G-1,118 and an antagonist G-15, (3aS*)-4R*,9bR*-(6-Bromo-1,3-benzodioxol-5-yl)-3a,4,5,9b-3H-cyclopenta[c] quinoline,119 exhibit high affinity and high selectivity for GPER over classical ERα and ERβ, with none of them showing affinity for ERα or ERβ.117,119 These tools have revealed that GPER activation may have several beneficial effects in the metabolic system including stimulation of insulin release and protection against pancreatic β-cell apoptosis. Thus, GPER is emerging as a candidate therapeutic target in metabolic disease.
To demonstrate the selectivity of G-1 toward GPER as compared with classical ERs, Ca2+ mobilization in COS7 cells expressing either ERα or ERβ revealed no response to G-1 compared with estrogen, which yielded a rapid response with both ERα and ERβ. G-1 also activates transfected and endogenous GPER, resulting in PI3K activation. With the identification of the first GPER-selective agent, it is now possible to investigate the specific functions of GPER compared with those of ERs in complex systems expressing both receptor types.
The pharmacological agents are currently being widely employed to investigate GPER function and physiology in various systems.117,119 GPER agonists including G-1 and 5408-0877 and antagonist G-15 remain to be fully evaluated for unequivocal specificity for this receptor.
GPER can also be activated by tamoxifen and raloxifene, SERMs45,55,120,121 traditionally thought only to modulate the function of ERα and ERβ.45,55,85 These studies provide experimental evidence that support the concept that GPER activation has inhibitory effects on food intake, body weight, and fat mass.122,123
2.3.5 Genetic tools—GPER knockout mice
The availability of genetic tools (i.e., different mouse models lacking the GPER gene) and pharmacological agents (i.e., GPER-selective agonists and antagonists) has advanced our understanding on GPER. Four mouse models lacking the GPER gene (GPER knockout, GPER KO) have been used to study the pathophysiological role of this receptor.98,124–126
GPER KO mice are found to associate with increased visceral adiposity 100,127 whereas Mårtensson et al.98 using a different GPER KO strategy found changes in body weight that were limited to GPER KO females. The same investigators found no effect of GPER deficiency on the antiobesity effects of E2.128 By contrast, Isensee et al. found no effect of GPER deficiency on body weight in animals on either a normal or high-fat diet in another model, LacZ-GPER reporter mice, which represent a partial GPER deletion.125,129,130 Data obtained from different GPER KO models generated by different transgenic techniques do not show a homogenous phenotype under physiological conditions.126 One group was unable to demonstrate an E2-dependent phenotype in GPER KO mice indicating that GPER might not be relevant for estrogen signaling,130,131 whereas other groups did.98,101,124 Such discrepancy would not exclude the possibility that E2-mediated effects might involve overlapping signaling of various ERs, and might be due to cross talk effects between GPER and other ERs132 as described below.
2.4. Cross talk between GPER and other ERs
2.4.1 Cross talk between membrane subpopulation ERs (mERα/β) and GPER
Rapid estrogen effects are mediated by ERs localized to the plasma membrane, comprising subpopulations of the classical ERs, namely membrane subpopulations of ERα and ERβ (mERα/β).47,57,133 Activation of mERα/β induces a variety of intracellular signaling cascades and mediates rapid estrogenic actions that involve cell-signaling mechanisms, mostly the phosphorylation of various kinases134,135 and the regulation of intracellular Ca2+ levels.136,137
An mERα/β selective ligand, STX, [2-(4-hydroxyphenyl)-3-phenylpent-2-enoic acid [4-(2-dimethyl amino ethoxy)phenyl] amide, E-enantiomer], has been developed and used in studies on homeostasis.138 Most behavioral investigations with this drug have utilized systemic administrations.139,140 Less commonly, direct brain infusions have attempted to identify its specific site of action.141
Membrane-associated full-length ERα and two truncated isoforms, ERα 46 kDa and ERα 36 kDa, exist that are at least in part responsible for nongenomic estrogenic signaling.142–144 Wang and colleagues have identified ERα 36 as a novel splice variant of the human ER.145 In breast cancer cell lines lacking ERα, expression of ERα 36 is associated with improved survival.146 It has been recently proposed that ERα 36 is the main effector and mediator of GPER-dependent effects mediated by estradiol and that its induction is dependent on GPER.147 However, others found that regulation of ERα 36 or ERα 36 is not detectable despite effects of G-1 being observed.148 A recent report demonstrating protective effects of G-1 in hypertensive rats in the kidney did not find any expression of ERα 36 while ERα 46 and ERα 66 were abundantly expressed.148 At present, data—particularly in vivo data—are still scarce. A recent report found effects of GPER agonists tamoxifen and ICI 182,780 are dependent on ERα 36 in cancer cells.149 Levin has proposed that GPER might act as “collaborator” of ERα—which it might be under certain conditions, as cross talk between both receptors has been described.150,151 Recently, it has been suggested that GPER signaling requires and might even solely occur through ERα 36.143
Different experimental settings from in vitro observations, immortalized cells vs. experimental animals, cancer cell vs. normal kidney, may be applicable to certain but not all conditions. In addition, results from in vitro findings cannot be extrapolated to the in vivo functions of receptors and thus are only of limited relevance. Another aspect to consider is that, unlike GPER, rapid ERα 36-dependent signaling is also activated by estrone, estradiol, estriol, and estetrol, and that E2-induced MAPK activation is further potentiated by GPER agonists such as ICI or tamoxifen.143 By contrast, the ERα 36 agonist estriol is an antagonist for GPER.152 Importantly, in vivo evidence for a role of ERα 36 in mediating the E2-mediated effects of GPER under physiological or disease conditions is still lacking.
2.4.2 Cross talk between EGFRs and GPER
Cellular signaling by GPER activates EGFRs via a G protein-dependent pathway, which involves Src and metalloproteinase (MMP) cleavage of proheparin-binding epidermal growth factor-like growth factor. Filardo and colleagues described GPER-mediated elevation of cAMP by estrogen as a mechanism to restore EGFR-activated MAPK to basal levels through PKA-dependent inhibition of Raf-1 activity.82 Two questions arose from this observation: (a) Do different ligands specifically activate the two receptors? (b) Do the two receptors utilize identical signaling pathways? To answer the first question, the same cells are treated with 4-hydroxytamoxifen, a well-described ER partial antagonist previously shown to activate MAPK via GPER. Stimulation with 4-hydroxytamoxifen resulted in no activation of PI3K in ERα-expressing cells but did result in PI3K activation in GPER-expressing cells, demonstrating for the first time in a side-by-side comparison the differential effects of tamoxifen on ERα and GPER. To answer the second question, the EGFR inhibitor AG1478 is employed. Two groups have shown independently that both ERα and GPER mediated EGFR transactivation.85,153 When EGFR phosphorylation is blocked, GPER-, but not ERα-mediated PI3K activation is inhibited. These studies demonstrated the close link in cellular signaling between GPER and EGFR.
2.4.3 Cross talk between classical ER and GPER
There appears to be complex interplay between ERα and GPER although such interplay has not yet been fully defined.93,150,154 Classical ERs may couple to G protein signaling via an unknown mechanism to mediate rapid estrogen signaling. The coexpression of ERα and ERβ with GPER in breast cancer cells has suggested a possible role for interactions between these two receptors.132,155,156
Indeed, functional cross talk has been demonstrated in cancer cells and uterine epithelial cells,150 where GPER functions as inhibitor of ERα-dependent, estradiol-mediated cell growth.151 Functional cross talk between ERs is also evident from isolated vascular preparations, where the E2-induced acute vasodilator response is only seen with ERα selective agonists but completely abrogated when ERα, ERβ, and GPER are activated simultaneously by E2.157 Navarro et al. demonstrated by coimmunoprecipitation studies that ERα interacts with Gαi3 in immortalized GnRH neurons, and that estrogen-induced inhibition of cAMP can be blocked by pertussis toxin, a G protein signaling inhibitor.158 Additionally, Kelly and coworkers87,159 have provided evidence of a Gαq-coupled ER that regulates the PKC and PKA signaling pathway in hypothalamic neurons and is involved in hypothalamic control of energy homeostasis.
Mitochondrial protection in response to E2 unrelated to GPER160 has been described for ERβ.161 E2 effect on insulin is absent in GPER KO mice,28,98,101 and protective effects on pancreatic islets are comparable independent of whether selective activation of ERα, ERβ, or GPER occurs.28 Thus, possible cross talk between GPER and ERs (also possibly other steroid receptors) as well as redundancy of ER function must be considered when assessing possible pathogenic and therapeutic questions related to GPER activation or inhibition.132 This should also be considered when determining the specific activity of compounds or drugs targeting either one117,119 or several membrane ERs.162
3. ESTROGENIC ACTION IN THE BRAIN: CENTRAL REGULATION OF ENERGY HOMEOSTASIS
3.1. Estrogenic action in the CNS
3.1.1 Estrogens directly act on hypothalamic areas involved in energy balance
A series of complex systems regulate energy homeostasis in order to keep energy storage levels and body weight stable. Central brain circuits receive peripheral signals indicating satiety, energy levels, and energy stores. The brain is made aware of the metabolic state of the body through signals released from adipose tissue, skeletal muscle, liver, pancreas, and gut. There is a wide variety of these signals, including hormones (insulin, leptin, gut hormones), cytokines (IL-6, TNF-α), and nutrients (glucose, free fatty acids, lipids). The hypothalamus is a key regulator of food intake and maintenance of energy homeostasis.163 The hypothalamus processes afferent signals from the gut and brain stem and efferent signals that modulate food intake and energy expenditure. Anorexigenic and orexigenic signals from the CNS then induce behavioral and metabolic changes targeting energy homeostasis and survival.
The main hypothalamic areas involved in food intake and satiety are the arcuate nucleus (ARC), the lateral hypothalamus (LH), the paraventricular nucleus (PVN), and the ventromedial hypothalamus (VMH).134 Several melanocortin receptors composing the melanocortin system are expressed throughout these nuclei and are one of the main regulators of hunger and satiety.164
Estrogens are major effectors for the regulation of energy balance and body fat. Energy homeostasis and feeding behavior in the hypothalamus follows the menstrual cycle, and food intake in women varies across the cycle with lowest daily food intake during the periovulatory period when estrogen levels are maximum.165 In several mammals and in women, higher levels of E2 during the estrus of menstrual cycles and pregnancy lead to decreased food intake and fat accumulation. In addition to its central actions, E2 also regulates peripheral feedback hormones that influence feeding and meal size through their actions in the CNS.
In contrast, ovariectomy, antiestrogen treatment, and menopause lead to an increase in food consumption and meal size, a decrease in running wheel activities, and increased fat mass, which can be reversed by E2 replacement. 166 The importance of the brain for the observed phenotypes has been demonstrated by studies showing that direct intracranial injections of E2 into the PVN leads to anorexia, reduced food intake, and body weight and increased running activities.167,168 These observations indicate that E2 has direct anorexigenic action in the hypothalamus.134
3.1.2 Estrogens act on central melanocortin system
Estrogens act on central melanocortin system to regulate energy balance and body fat distribution. The melanocortin system is responsible for the opposing actions of these neurons; however, so far a clear relation between E2, the melanocortin system, and energy balance has not been established.169,170 In the ARC, several types of neurons and receptors of the melanocortin system (MC3R and MC4R) are involved in the regulation of appetite and respond to variations in glucose, lipids, leptin, and insulin. The proopiomelanocortin (POMC) neurons have an anorexigenic action and, when activated, reduce food intake through the release of two peptides, α-melanocyte stimulating hormone (α-MSH) and cocaine-and-amphetamine-regulated transcripts. The neuropeptide Y (NPY) neurons, on the other hand, release NPY hormone and agouti gene-related protein (AgRP), which prevent the binding of α-MSH to MC3R and MC4R, increasing food intake.164 Whole animal studies in ovariectomized rats have reported that a significant reduction in food intake by E2 treatment compared to oil treatment was not detected after administration of the melanocortin receptor antagonist SHU919.
A recent study demonstrated that NPY/AgRP neurons are required to mediate the anorexigenic effects of estrogens. In this study, Xu and colleagues showed that hypothalamic expression of NPY and AgRP in wild type mice is tightly regulated across the estrus cycle, with the lowest levels during the estrus, which coincides with the plasma estrogen peak and feeding nadir.171 They further showed that central E2 administration suppresses fasting-induced c-Fos activation in NPY/AgRP neurons and blunts the refeeding response.171 Importantly, the cyclic changes in food intake and E2-induced anorexia are blunted in mice with degenerated NPY/AgRP neurons. This study indicates that NPY/AgRP neurons are functionally required for the cyclic changes in feeding across estrous cycles. Surprisingly, these authors also found that ERα is not expressed in NPY/AgRP neurons,171 suggesting that estrogen may regulate these neurons indirectly via presynaptic neurons that express ERα (e.g., POMC neurons). Indeed, POMC neurons coexpress ERα.172–174 In addition, estrogens regulate excitability of POMC neurons. Using electron microcopy, Horvath and colleagues have reported that the number of excitatory synaptic inputs to ARC POMC neurons rises as mice enter proestrus when estrogen levels are high.23
Further, central E2 administration rapidly increases the excitatory synapses on POMC neurons, an effect that is also reflected by increased miniature excitatory postsynaptic current recorded from POMC neurons.23 These synaptological rearrangements in POMC neurons are tightly paralleled with the effects of E2 on food intake and body weight.23 Similarly, Ronnekleiv and colleagues reported that E2 administration in hypothalamic slices activates POMC neurons by rapidly uncoupling GABAB receptors from the G protein—gated inwardly rectifying K+ channels.175 Importantly, female mice lacking ERα in POMC neurons only develop hyperphagia.172 These observations, together with the findings from the Horvath and Ronnekleiv groups, indicate that estrogen signals in POMC neurons are physiologically relevant in the regulation of food intake.
3.1.3 Estrogens act on nuclear and membrane ER in the CNS
Classical ERα and ERβ and GPER are all highly expressed in the brain, their distribution being both overlapping and unique.61 The critical intracellular signals that mediate the anorexigenic effects of estrogens have been explored. Stimulation of nuclear and membrane ERs engages genomic and nongenomic events that may mediate distinct functions of estrogens, as detailed below.
3.2. Estrogen acts on nuclear ERs in the CNS
Genomic activation of ERs results from estrogens binding to EREs in promoter regions of target genes.176,177 ERα is ubiquitously expressed in rodent brains. Areas of predominant ERα expression are the VMH and ARC.178–181 ERβ is found in the same hypothalamic nuclei as ERα, but ERβ expression is significantly reduced relative to ERα. ERβ predominates in the suprachiasmatic, supraoptic, and PVN of the hypothalamus, the cerebellum, dorsal raphe, hippocampus, and cerebral cortex.68,179,182–184 ERα and ERβ expression overlaps in the ARC, LH, medial amygdala, preoptic area, bed nucleus of the stria terminalis, nucleus of the solitary tract and periaqueductal gray.
Pertinent to the estrogenic action on melanocortin system is that both ERα and ERβ have been identified in the ARC. Specifically, ERα is predominantly expressed in the POMC neurons, while both ERα and ERβ are present in the NPY neurons.134,173 The PVN and VMH are also mediators of energy ingestion and expenditure and are interconnected with the ARC. The PVN is highly responsive to glucose, lipids, NPY, and AgRP, and its activation leads to increased food intake.134,185 The PVN is the hypothalamic region with the highest expression of ERβ and is weakly ERα positive.182 Despite this apparent low level of ERα, in rats and mice, the ERα-selective agonist, PPT, rapidly results in a decrease in food intake and increase in expression of c-fos in the corticotrophin-releasing hormone-expressing cells in the PVN.186 The VMH is ERα regulated and has an important function in the inhibition of food intake through anorexigenic signals from the ARC. Silencing of ERα in the VMH by RNA interference leads to hyperphagia, obesity, decreased glucose tolerance, and reduced energy expenditure.187
The role of estrogens and ERα in the VMH in the regulation of energy balance has been reexamined using the ERα silencing approach.187 In this study, ERα in the VMH is knocked down with an AAV-shRNA.187 Animals with impaired ERα signaling in the VMH are less sensitive to E2-induced weight loss and develop obesity characteristic of increased visceral fat. The obesity syndrome is likely caused by decreased physical activity and impaired diet-induced thermogenesis, whereas food intake of these animals is not directly affected.
Xu et al. generated mice with ERα deleted in VMH SF1 neurons.172 They found that deletion of ERα in VMH SF1 neurons in female mice, while not affecting food intake, significantly reduces basal metabolic rate and diet-induced thermogenesis, which consequently results in increased body weight and adiposity. Interestingly, a significant increase in visceral fat deposition but not subcutaneous fat deposition is observed in these mutant females.172 Finally, they showed that the decreased energy expenditure and increased visceral fat distribution in mice lacking ERα in SF1 neurons presumably results from decreased sympathetic tone, as demonstrated by decreased plasma norepinephrine levels.172 These findings are largely consistent with those obtained from the VMH-specific ERα knockdown model. Collectively, these results support the hypothesis that ERα signaling in VMH neurons plays an important role in regulating energy expenditure and fat distribution.
To summarize, ERα and ERβ are expressed in brain regions implicated in the regulation of appetite, satiety, and energy expenditure,49,68,178–180,183 which can explain the involvement of these receptors in changes of feeding behavior and energy expenditure and thus regulation of energy homeostasis. The differential distribution further suggests that, like for other behaviors, the respective involvement of ERα and ERβ in regulation of energy homeostasis may be different.
3.3. Estrogen acts on membrane ERs in the CNS
3.3.1 Identification of three membrane ERs in the CNS
Recent evidence suggests that estrogen has at least three different transmembrane receptors in the brain.115,188
The first membrane ER identified and localized in the brain is the estrogen-binding G protein-coupled receptor GPER. Brain GPER expression in adult male and female rats was thoroughly characterized by Brailoiu et al. using immunohistochemical analysis.106 GPER in the hypothalamus is localized primarily to the PVN and the supraoptic nucleus but is also found in ARC neurons.106 GPER is also expressed in nonhypothalamic regions, including the anterior and posterior pituitary, striatum, hippocampus, and the autonomic nuclei of the brainstem including substantia nigra, area postrema, nucleus of the solitary tract (NTS), and dorsal motor nucleus of the vagus.106 These hypothalamic and brainstem areas are typically associated with energy homeostasis function. GPER immunostaining exhibits a similar pattern in male and female mice,189 suggesting that GPER expression in these autonomic areas is not regulated by estrogen status. In the NTS, GPER immunostaining is predominantly localized to neurons that coexpress ERα, whereas in the PVN, GPER displays moderate colocalization with nitric oxide synthase.190 Consistent with this study, Sakamoto and coworkers have also shown expression of GPER mRNA and protein in the oxytocin neurons of the rat PVN and supraoptic nuclei.191
The second membrane ER is the putative G protein-coupled receptor membrane estrogen receptor (Gq-mER) functionally characterized by Kelly and coworkers.87,88 Although the Gq-mER has not been cloned, it has been functionally identified in at least three types of ARC neurons, including POMC, dopamine, and γ-aminobutyric acid (GABA) neurons.
The third membrane ER is membrane subpopulation of ERα and ERβ has been identified. Ultrastructural electron microscopic studies have provided direct evidence of extranuclear localization of ERs in the brain.192 Milner et al.192 performed ultrastructural studies of ERα localization in the rat hippocampal formation and demonstrated that ERα, in addition to nuclear localization, is found in cytoplasm and plasma membrane of neurons, and in unmyelinated axons and axon terminals. Furthermore, approximately 25% of ERα is localized in dendritic spines, often associated with spine apparati and/or polyribosomes, suggesting it may act to regulate Ca2+ availability, local protein translation, and synaptic growth. Using Western blot and estrogen-horseradish protein-FITC or estrogen-BSA-FITC conjugates, Marin et al. showed plasma membrane localization of ERα in SN56 septal-derived cholinergic neurons, and that the estrogen conjugates protected the cells against A-beta toxicity as effectively as estrogen.193,194 Xu et al.195 transfected GFP-tagged human ERα in rat cortical neurons in vitro and determined its subcellular localization. The studies revealed that there was localization of the GFP-tagged human ERα in neurites of the cortical neurons and that the ERα appears to be directed to the neurites directly from its site of translation and not from nuclear stores. Furthermore, biotinylated membrane protein studies also support localization of ERα at the plasma membrane of rat cortical neurons. Finally, ultrastructural studies have also demonstrated that ERβ is also localized at extranuclear sites such as cytoplasm, plasma membrane, dendritic spines, axons, and axon terminals in the rat hippocampus.196 Thus, ERβ could also have a role in mediating nongenomic actions of estrogen in the brain. We should also mention that a novel plasma membrane-associated ER called ER-X has been proposed as well in neurons of the brain, but attempts to clone it have so far been unsuccessful.91
3.3.2 Estrogenic nongenomic signaling in the CNS
Estrogens can initiate rapid signaling cascade events within minutes to affect cell physiology and activate gene transcription through membrane-initiated steroid signaling via nongenomic mechanisms at non-ERE transcription factor promoter sites. Instead, estrogens activate transcription via complexes with other transcription factors through protein–protein interactions, including CREB, STATs, Elk-1-SRF, ATF-2-Jun, and NFκB-inducing transcription via their respective promoter sites.197
Reports of rapid E2 effects in the brain first began to appear in the literature over 40 years ago, when it was observed that estrogen rapidly modulated hypothalamic electrical activity.198–200 The latency of the estrogen effects was within minutes, and typically two types of effects were observed on hypothalamic neurons—some units had a decreased firing rate that lasted up to 150 min, whereas other units responded with a transitory increase in firing rates. The remaining units were unresponsive to E2.200 Kelly et al.201–204 showed that neurons from several hypothalamic regions displayed rapid electrophysiological responses to estrogen, including preoptic, ARC, and VMH neurons. These early observations set the stage for subsequent studies on the mechanisms underlying the rapid effects of estrogen in the brain. ERs in the plasma membrane are able to activate the MAPK cascade and PI3K pathway, causing a rise in intracellular Ca2+.205,206 ERs also activate PKB or Akt in neurons.207–209 The PI3K/Akt cascade mediates a variety of estrogens’ central actions, including neuronal excitability, neuroprotection, reductions in inflammation, neurite outgrowth, and potentially body weight regulation.210
The rapid effects of estrogen on Ca2+ and ion channel signaling and kinase activation suggest a potential membrane receptor-mediated effect of estrogen. One piece of evidence supporting a membrane receptor-mediated mechanism is the use of membrane impermeable conjugates of estrogen, such as estrogen-BSA and estrogen dendrimers. Similar to estrogen, estrogen-BSA was shown to rapidly enhance phosphorylation of MAPK in the human neuroblastoma SK-N-SHR cells,211 in rat hypothalamic neurons in vitro,212 and in the rat hippocampus in vivo.213 Estrogen-BSA conjugates have also been shown to rapidly enhance PKA activation in hippocampal neurons,214 increase intracellular Ca2+ levels within minutes in both astrocytes and neurons,215,216 and to rapidly modulate potassium channel activation in hypothalamic neurons.87 Nevertheless, it should be pointed out that there has been some criticism of estrogen-BSA conjugates, in that some estrogen (3–5%) can be found in the free unconjugated form in commercially available estrogen-BSA conjugates.217 For this reason, it is advisable that the estrogen-BSA be subjected to filtration in order to remove free estrogen, and that studies that fail to perform the filtration should be interpreted cautiously. Stevis et al.217 reported that estrogen-BSA did not bind ERα or ERβ in binding studies; however, a subsequent study demonstrated that estrogen-BSA indeed binds to purified ERs in vitro and to membrane ERs in intact cells, albeit at a slower rate than estrogen.218 Taken as a whole, the estrogen conjugate data suggest that rapid signaling effects of estrogen involve mediation via a membrane ER.
3.3.2.1 Rapid estrogen signaling in ER KO mice
One approach to determine the role of ERs in mediating rapid signaling effects of estrogen in the brain is to utilize mutant mice with one or both classical ERs knocked out. Using this approach, Chaban and Micevych demonstrated that rapid estrogen inhibition of ATP-induced Ca2+ signaling in dorsal root ganglion neurons is blocked by an ER antagonist, and that the effect of estrogen on Ca2+ signaling is lost in ERα KO mice.219 Likewise, Herbison and colleagues demonstrated that rapid estrogen-induced phospho-CREB responses in hypothalamic neurons, where ERβ is densely localized, is preserved in ERα KO mice, but lost in ERβ KO mice.220
With regard to MAPK regulation, there are contradicting reports on the estrogen regulation of phospho-MAPK in ER KO mice. Singh et al.221 reported that estrogen-induced phosphorylation of MAPK was not lost in cortical neurons from ERα knockout mice, suggesting that ERα may not mediate the rapid phospho-MAPK-enhancing effects of estrogen. In contrast, Abraham et al.220 reported that rapid estrogen induction of phosphorylation of MAPK in the medial preoptic nucleus was lost in both ERα and ERβ knockout mice, suggesting that both ERα and ERβ may mediate the MAPK regulatory effect of estrogen. Finally, estrogen can also rapidly induce c-Fos protein in the brain, an effect which has been shown to be lost in double ERα/β KO mice in several brain areas including the VMH.222
As a whole, results with the ER KO mice shed some light on involvement of ERα or ERβ in rapid signaling effects of estrogen in the brain. Additionally, they suggest that the role of ERα or ERβ in rapid estrogen signaling effects may differ from one brain region to another, and results in one region cannot be immediately extrapolated to another region or another kinase.
3.3.2.2 Rapid estrogen signaling in the brain via G protein-coupled receptors
G protein-coupled receptors are vital membrane mediators for a host of central and peripheral signals that control energy homeostasis in the hypothalamus.223 Because G protein-coupled receptors target potassium channels to control neuronal excitability, they may account for one of the rapid mechanisms estrogen utilizes to control energy homeostasis. A candidate for an estrogenic mechanism recently identified is the control of neuronal excitability through a G protein-coupled receptor membrane ER (Gq-mER).87,88 The Gq-mER affects neuronal activity, alters metabolic processes such as body weight, and regulates energy balance.
The Gq-mER was initially characterized in female guinea pigs, but has also been functionally examined in both male and female ERα KO mice and ERβ KO mice, and in male ERα/β double KO mice.88 Estrogen through Gq-mER activates multiple signaling pathways including PI3K, phospholipase C (PLC), MAPK and PKA and PKC pathways.210,224,225 Briefly, the Gq-mER activates a Gq-PLC-PKA pathway that inhibits the activation of G protein-coupled inwardly rectifying potassium channels. The inhibition of the potassium channel activity will depolarize the POMC neuron and increase neuronal activity and potentially add another mechanism for estrogen to control energy homeostasis.
The Gq-mER involved in estrogenic membrane-initiated steroid signaling is thought to be ERα,225 although recent data suggest that, in a transfected neuronal cell line, ERβ is also associated with the membrane and is translocated to the membrane by estrogen to activate MAPK signaling.226 In the hypothalamus, estrogen membrane-initiated steroid signaling has been identified in the VMH to potentiate the effects of estrogenic nuclear-initiated steroid signaling during particular reproductive behaviors such as lordosis227 and may also affect estrogen’s control of homeostasis via VMH neurons.
There is in vivo evidence suggesting a role for the activation of membrane-initiated estrogenic signaling events that affect energy homeostasis. E2 can attenuate food intake within 6 h of administration into the third ventricle via cannula after an overnight fast compared to saline in mice228 and between 4 and 14 h in fed rats.23 Estrogen can activate signaling cascades initiated through Gq-mER that would rapidly alter neuronal activity and initiate changes in feeding behavior.87,88 Indeed, acute administration of estrogen to hypothalamic slices will alter neuronal excitability of many different types of relevant neurons through the modulation of various ion channels.229,230
To elucidate this estrogen pathway, STX, a selective agonist for the GqmER, has been developed that has no binding capacity to classical ER.138 STX is more potent than estrogen in attenuating the activation of K+ channels.87,88 STX is an excellent tool for the elucidation of the role of the Gq-mER in energy homeostasis and gene regulation.89 The ability of STX to alter POMC neuronal excitability leads to the hypothesis that the putative Gq-mER has a role in energy homeostasis. Whole animal studies in which STX was systemically injected in ovariectomized female guinea pigs over a period of 4 weeks support this hypothesis. This study demonstrated a dose-dependent effect on the reduction in body weight gain by systemic STX treatment. Furthermore, the administration of systemic STX-generated new transcription in the ARC of these STX-treated female guinea pigs.89 The genes regulated include NPY gene involved in the control of energy homeostasis and Cav3.1 gene involved in neuronal activity. Therefore, Gq-mER may function in the estrogenic control of energy homeostasis, presumably through activation of POMC neurons in the ARC, although other hypothalamic nuclei may be involved.88
The molecular characterization of this Gq-mER, including cloning, localization, and regulation, is crucial for a full understanding of the role of the rapid, membrane-mediated effects of estrogen on energy homeostasis. Additionally, STX can be utilized to target the Gq-mER to determine whether Gq-mER actually contributes to the regulation of energy homeostasis in ERKO mice.
4. ESTROGEN ACTION IN ADIPOSE TISSUE: REGULATION OF LIPOGENESIS/LIPOLYSIS AND ADIPOSITY
Organisms store energy for later use during times of nutrient scarcity. Excess energy is stored as triacylglycerol (TG) in lipid droplets produced by lipogenesis. When energy is required, TG is catabolized into free fatty acids via lipolytic pathways. Obesity results from excess white adipose tissue, which is considered to be an endocrine organ and it is able to store and metabolize steroid hormones. ERs are expressed in adipose tissue, where they may influence adiposity and inflammation. Estrogens regulate both the metabolism and the location of white adipose tissue and play a role in adipose deposition, lipogenesis, lipolysis, and adipocyte proliferation.231
4.1. Estrogenic action in adipose tissue prevents obesity
Obesity, high body fat percentage, and increased hip circumference are all strongly correlated to increased E2 levels.232 Sex steroids are known to regulate lipid metabolism and are responsible for accumulation, distribution, and development and function of adipose tissues. The differences in body fat distribution suggest that the sex steroid hormones could have an effect on adipose tissues. Direct effects seem plausible, as sex steroid hormone receptors have now been found in adipose tissues. The amount of the receptor varies with the fat depot, with subcutaneous adipose tissue having a higher concentration of ERs than intra-abdominal adipose.
Estrogen activates lipogenesis under normal physiological conditions in lean animals but suppresses lipogenesis under pathologic conditions. In normal lean animals, increased intracellular E2 activates ERα and stimulates lipogenesis.233 Consequently, female mice with higher E2 level have greater lipogenic capacities in adipocytes compared with male mice.234 Specifically, E2 increase the deposition of subcutaneous fat on the hips, upper thighs, and lower abdomen, ectopic fat in the liver and muscle, and visceral fat in and about abdominal organs.235 As the enlarging fat cells produce more E2, they increase their leptin output,236 to cyclically upregulate 11β-hydroxysteroid dehydrogenase,237 increase intracellular E2 production.238 This stimulation of lipogenesis by E2 is ERα-dependent, as revealed by global gene expression analysis from ERα KO and wild type mice that demonstrated ERα dependent increased expression of lipogenic genes and decreased expression of genes regulating lipid transport.239 Interestingly, despite the increased lipogenesis, adipocytes from females are smaller than adipocytes from males.
It is noteworthy that androgen receptors (AR) are also expressed in adipose tissues.240 Subcutaneous adipose tissue has higher concentrations of ERs whereas visceral adipose tissue has higher concentrations of AR.241 Additionally, subcutaneous adipose tissue has few AR, and estrogens downregulate AR expression in subcutaneous fat.12 Adipose tissue-specific AR knockout mice have increased intra-adipose estrogens, which leads to increased subcutaneous obesity and hyperleptinemia.242 The sexual dimorphism in adipose tissue distribution may partially explain the greater risk for the metabolic syndrome in men compared with premenopausal women.
Estrogens suppress lipogenesis under certain conditions leading to obesity. E2 suppresses the expression of lipogenic genes, i.e. fatty acid synthase, stearoyl-coenzyme A desaturase 1, and glycerol-3-phosphate acyltransferase, and thus suppresses lipogenesis and TG accumulation in adipose tissue and liver in high-fat diet fed, leptin-deficient ob/ob mice.243 When circulating levels of estrogen are raised above the physiological range due to enlarged adipocytes, adipose tissue metabolism is altered resulting in reduced lipogenic rates and fat depot size to prevent further expansion of adipose tissue. E2-treated ovariectomized mice display reduced lipogenesis in adipocytes. 24 This is further supported by epidemiological observations that serum triglyceride levels increase in postmenopausal women with increased adiposity and that estrogen hormone replacement therapy reduces the activity of lipoprotein lipase (LPL),244 the enzyme that catalyzes the conversion of triglycerides into free fatty acids and glycerol.245 Therefore, normal amount of estrogen favors homeostasis in adipose tissues and estrogenic action in adipose tissue prevents the development of obesity by decreasing the lipogenic activity in adipose tissue. However, decreases in the amount of estrogen, as occur with menopause in women and ageing or gonadectomy in rodents, usually result in central obesity with increased lipid accumulation and decreased lipid utilization at visceral fat.246
Genetic mouse models of either E2 deficiency by knockout of the aromatase gene (ArKO) or E2 resistance in ERKO have provided important information on the role of E2 signaling in adipose tissue physiology and support roles of E2 and ER in adipose tissue.
ArKO, ERα KO, and mice with double knockout of both ERα and ERβ exhibit an increased white adipose tissue mass but not brown adipose tissue mass,65,67,247 whereas knockout of the ERβ gene alone does not develop obesity,248 suggesting that ERα plays an important role in adipogenesis. In addition, ArKO mice and ERα KO mice show increase in adipocyte size (hypertrophy) and number (hyperplasia), display dyslipidemia including an increase in serum cholesterol and a change in the lipoprotein profile, and insulin resistance, implicating a role for ERα in adipocyte growth and proliferation. By studying estrogen replacement in ovariectomized ERα and/or ERβ knockout mice, Lindberg et al.249 demonstrated a reduction of fat in these animals with estrogen replacement mediated mainly by ERα. These studies demonstrate the importance of E2 signaling through the ERα in adipocyte differentiation and metabolism.65,247,250 This is further supported by studies on 3T3-L1 preadipocytes with stably transfected ERα, which show decreased triglyceride accumulation and reduced expression of LPL. The observation from ArKO mice is consistent with men with aromatase deficiency who have central obesity, elevated blood lipids, and severe insulin resistance.251
ERβ KO female mice have a higher body weight under HFD feeding than wild type littermates.252 This could be a result from increased adipogenesis with subsequent increased mass of adipose tissue and improved insulin sensitivity. Furthermore, the key adipogenic and lipogenic factor PPARγ is negatively regulated by ERβ, suggesting that PPARγ could be a mediator of the metabolic effects observed in ERβ KO mice.252 Female ERβ KO mice on HFD displayed impaired food efficiency and increased respiratory quotient, which is an indication of disturbed fatty acid oxidation. PPARγ DNA-binding properties and target gene activation are markedly induced in the gonadal fat of the ERβ KO mice and inhibition of adipose PPARγ signaling reversed this metabolic phenotype.252 In summary, both ER isoforms participate in the antilipogenic actions of estrogens.
Estrogens carry out their action directly on adipose tissues by a combination of genomic (receptor DNA interactions) and nongenomic (membrane receptors) mechanisms in estrogen regulation of adipose accretion and metabolism.253,254 In the genomic mechanism, estrogens bind to their receptors and the steroid–receptor complex regulates the transcription of given genes. LPL is a key adipose protein regulated by estrogens in a classical genomic mechanism. Second messenger pathways via the cAMP and phosphoinositide cascades are modulated by membrane receptors for estrogens in a nongenomic mechanism. See below for details.
4.2. Estrogenic action on adipogenesis via genomic mechanism
4.2.1 Classical nuclear receptors ERα and ERβ in adipose tissue
The mechanism for the regulation of the amount and distribution of adipose tissues by estrogens is not completely clear. One possible mechanism would be the regulation of key proteins in adipose tissues at the genomic level by transcriptional means. This would require ERs to be present in adipose tissues.
Many groups have demonstrated that ERα and ERβ mRNA and protein are expressed in human adipose tissue.240,255–260 In initial experiments, it was not possible to detect ER in subcutaneous or omental fat from normal women.261,262 Later, low levels of ER have been found in human adipose tissues with increased sensitivity of molecular techniques.
Price & O’Brien258 demonstrated by PCR the presence of ER mRNA in human subcutaneous abdominal tissue. Mizutani et al.260 identified ER protein and ER mRNA in subcutaneous abdominal tissue. The ER protein had a molecular weight of 65 kDa, which is similar to the ER from other tissues including reproductive tissues. Human subcutaneous and visceral adipose tissues express both ERα and ERβ. Expression of ERα is similar in subcutaneous gluteal and subcutaneous abdominal adipose tissues, and is similar between men and women.256 The ER is higher in concentration in subcutaneous fat than in visceral fat in both males and females. These differences in concentrations of ER in visceral adipose tissue and subcutaneous adipose tissue may offer a possible explanation why men have a more central accumulation of fat, whereas women have a more gluteal/femoral accumulation.
Pedersen et al. demonstrated by ligand binding and RT-PCR the presence of ER in human adipose tissue and mature adipocytes.263 ERα and ERβ are present in cytosols of adipose tissues from both men and women. Cytosols from visceral adipose tissue and subcutaneous abdominal tissue of women showed about the same amount of ER binding, whereas cytosol of subcutaneous abdominal tissue from men showed a higher amount of binding than those from visceral adipose tissue. By immunohistochemistry and RT-PCR analyses, ERα and ERβ are detected in both stromal cells and adipocytes from subcutaneous and omental adipose tissues.257 A higher amount of ER mRNA was found in adipocytes than in adipose stromal cells. In mature human adipocytes, ERα is the more highly expressed isoform. ERβ is expressed in breast and subcutaneous abdominal tissues,240 and it appears to be more highly expressed in women than in men.259 Interestingly, only ERα, not ERβ, is expressed in human preadipocytes by RT-PCR, although the expression of ERα in preadipocytes is very low compared with that in mature adipocytes.264
E2 treatment regulates ERα and ERβ in stromal cells from subcutaneous and omental adipose tissues. In adipocytes from subcutaneous adipose tissue, E2 treatment decreases the expression of ERα, while increases ERβ expression. In omental adipocytes, the decrease in the expression of ERα with E2 treatment is not observed.
Laboratory rodents have been used to study the relationship of estrogens to obesity. Obesity in laboratory rodents is easily induced by overeating and reduced energy expenditure, and the animals develop many of the same deleterious effects, such as insulin resistance and hyperinsulinemia, as seen in human obesity. The major advantage of working with mice is the development of techniques that allow for the manipulation of their genome to generate transgenic mice that lack functional ER.265 Both ERs are expressed in rodent white adipose tissue256,266–269 with a predominance of ERα270 and colocalization of ERα and ERβ in the same nuclei of some cells,271 whereas only ERα but not ERβ mRNA has been identified in brown adipose tissue. 272 Additionally, brown adipocytes from male rats have higher ERα expression compared to those of female rats, suggesting that this receptor could have a role in thermogenesis.272
One of the first indication that ERs exists in adipose tissues was reported by Wade & Gray.266 They showed estrogen-specific, high affinity [3H]-E2 binding in cytosolic extracts of ovariectomized rat adipose tissues with similar properties to ER in reproductive tissue, but at a much lower concentration. Binding was highest in parametrial fat and lowest in inguinal fat. In a subsequent paper, Gray & Wade273 showed estrogen-binding sites in male rat adipose tissues. Gray et al.274 also demonstrated binding of injected [3H]-E2 in the nuclei in adipose tissues. Using ovariectomized and adrenalectomized rats for conditions of low endogenous sex steroid production, Rebuffe-Scrive275 demonstrated cytoplasmic E2 binding in adipose tissues from these animals that decreased dramatically after estrogen administration. Pedersen et al.276 showed nuclear binding of [3H]-E2 in isolated rat adipocytes. The binding is specific for E2 and showed regional variation. Using immunogold electron microscopic localization, Echeverria et al.277 demonstrated significant labeling of ER in adipose cells from various locations of both male and female rats. A higher density of label is found in the nucleus, but some label was observed in the cytoplasm of the adipose cells. All these results support the notion that estrogens may have a direct effect on adipose tissues via its binding to their receptors.
ER knockout mice have abnormalities in their adipose tissues.278 Indeed, ERα is now considered a candidate gene for obesity.279 In male mice, deletion of ERα causes progressive increase in epididymal, perirenal, and inguinal adipose tissue with advancing age. Epididymal and perirenal adipocyte size is increased and adipocyte number is greater in the fat pads. Female ERα KO mice have increased fat, adipocyte size, number, and leptin and cholesterol levels and smaller LDL particles. Both males and females are insulin resistant with impaired glucose tolerance.65 E2 administered to ovariectomized mice fed a high-fat diet preserved glucose tolerance and insulin sensitivity and improved glucose tolerance in wild type but not in ERα KO mice, suggesting that targeting of the ERα could represent a strategy to reduce the impact of high-fat diet-induced insulin resistance.280
ERs play roles on GLUT4 function in adipose tissue. The administration of tamoxifen to ERα KO or ERβ KO mice resulted in different responses on GLUT4 expression. In ERα KO mice, tamoxifen did not interfere with GLUT4 expression, whereas treatment of ERβ KO mice with tamoxifen substantially decreased it, suggesting that ERα is the main regulator of GLUT4 expression in adipose tissue.271 Ovariectomy of ERβ KO mice prevented accumulation of triglycerides and reduced body fat and adipocyte size, and preserved regular insulin signaling in liver and skeletal muscle, and improved whole body insulin sensitivity and glucose tolerance, suggesting that ERα and ERβ have opposite functions on lipid and glucose metabolism, with ERβ involved in adipogenesis.252,281 An imbalance between ERα and ERβ in adipose tissue may have important implications for the development of metabolic diseases.
4.2.2 Estrogenic genomic action on LPL in adipose tissue
In the classical mechanism of action of sex steroid hormones, the steroid enters the cell and binds with high affinity to a specific receptor. The steroid–receptor complex then undergoes a conformation change and binds to a specific response element on the DNA. Once bound, it regulates the transcription of a given gene. Transcription may be up- or downregulated depending upon the gene, the steroid, and the coregulatory proteins. Many genes in adipose tissues are transcriptionally regulated by sex steroid hormones. An ERE is present in the LPL genome and thus LPL gene is a direct transcriptional target of estrogen. LPL is the key protein involved in lipid deposition.
LPL is the key enzyme for the hydrolysis of circulating triglycerides into free fatty acids and glycerol. Its highest activity is located in adipose tissues, heart, and skeletal muscle, and it plays a major role in adipocyte lipid storage and muscle fuel supply and hence the regulation of lean muscle mass and obesity. It is synthesized and secreted by the adipocyte and then transported to the capillary endothelium, where it hydrolyses the circulating triglycerides into free fatty acids, which in turn are stored in the adipose tissues. LPL in adipocytes is activated by insulin. During feasting, increased insulin activates LPL to promote adipogenesis.
Although the mechanisms responsible for estrogen’s regulation of adipogenesis remain unclear,255 they may involve estrogens’ suppression of LPL, a lipogenic enzyme that regulates the metabolism of plasma triglycerides to free fatty acids and increases lipid storage by adipocytes. Specifically, the effect of estrogens on LPL activity may be concentration dependent, as Palin et al. found that a high concentration (10−7 mol L−1) of estrogen reduced LPL expression in isolated adipocytes from subcutaneous abdominal fat of women, while lower estrogen concentrations increased LPL expression.282 Consistent with this notion, ovariectomy of female rats results in increased adipose tissue LPL while estrogen replacement decreases the LPL activity.283–288 E2 administration to male rats also significantly reduces adipocyte LPL activity289 to suppress adipogenesis.
In obese women, plasma LPL activity has an inverse correlation with plasma E2 levels.244 The administration of estrogens alone or estrogen–progestin in women decreases LPL activity and increases serum triglyceride concentrations.290–292 A transdermal E2 patch placed in the gluteal region of women decrease LPL in adipose tissue from beneath the patch as compared to tissue beneath the placebo patch.293 However, there is no difference in LPL mRNA levels between these two groups, suggesting that a possible posttranscriptional modification of LPL led to the difference. In women, LPL levels increase after menopause.294 In addition to inhibiting fatty acid uptake (lipogenesis), estrogen also increases fatty acid and glycerol release (lipolysis) by increasing expression of hormone-sensitive lipase (HSL), which catalyzes the breakdown of stored TG and the release of fatty acids.282,295 However, estrogen has also been shown to inhibit lipolysis. Estrogen treatment of postmenopausal women increases expression of the α2A-adrenergic receptor in subcutaneous adipose tissue. Activation of this receptor inhibits lipolysis; therefore, increased expression due to estrogen would inhibit lipolysis.
This increase was recapitulated in vitro using cultured human adipose tissue fragments incubated with E2 and was correlated with inhibition of lipolysis,296 suggesting a direct role for ER in regulating lipolysis. The complex relationship between the ERs and adipogenesis remains to be elucidated and may provide critical information for the prevention and treatment of obesity.
4.3. Estrogenic nongenomic action via second messengers in adipose tissue
Another possible mechanism would be the regulation of second messengers at the cell membrane by nongenomic effects. Although transcriptional regulation by steroid hormones has been the most studied mechanism of steroid action, recent studies have shown that a nongenomic mechanism also exists for steroid hormone action.297 This would require a sex steroid receptor to be present in the membrane. There is also evidence for rapid nongenomic estrogen actions via a membrane-bound ER in adipocytes.112,298 Nongenomic actions of steroids are generally rapid responses occurring in less than 10 min, while transcriptional regulation requires several hours to days.197,299,300
In the nongenomic mechanism, estrogens bind to their receptors in the plasma membrane, and second messengers are formed to carry out their action. Estrogens appear to regulate various components of the membrane-signaling systems, including both the cAMP cascade and the phosphoinositide cascade.301,302
Activation of the cAMP cascade by sex steroid hormones would activate HSL leading to lipolysis in adipose tissues. In the phosphoinositide cascade, diacylglycerol and inositol 1,4,5-trisphosphate are formed as second messengers ultimately causing the activation of protein kinase C. Their activation appears to be involved in the control of preadipocyte proliferation and differentiation.
Sex steroid membrane receptors have been characterized in many different tissues,112 but little is known about the receptors in adipose tissues. Anwar et al.257 showed immunostaining for both ERα and ERβ in the cellular membranes of subcutaneous abdominal and omental human adipose tissues. Dos Santos et al.298 found immunoreactive ERα in the membrane fraction from cultured rat adipocytes, but not in the membrane fraction from preadipocytes.
These plasma membrane ERs are associated with caveolae endocytic vesicles. Caveolin-1 coimmunoprecipitate with ERα and bind directly to ERα.303 Caveolin-1 has also been reported to bind to endoplasmic reticulum membranes.304
4.3.1 cAMP cascade
Early studies showed an increase in cAMP and lipolysis within 5 min when isolated rat adipocytes were treated with E2305 and adenylate cyclase activity in fat cell mass increased by E2 treatment of ovariectomized rats.306 This response to estrogen appears to be depot specific as ovariectomy caused a decrease in cAMP production in rat parametrial adipocytes, but not in femoral subcutaneous adipocytes.307 These studies with rats would suggest that estrogen increased cAMP in adipose tissues, resulting in stimulation of HSL and increased lipolysis.
In humans, estrogens also appear to decrease lipolysis in adipose tissues. Lindberg et al. found that women who were given oral ethinyl E2 had a decrease in noradrenaline-stimulated lipolysis in subcutaneous abdominal adipocytes.308 To study the effect of estrogens on lipolysis in women, Pedersen et al. used adipocytes that were isolated from adipose tissue fragments, which were incubated in vitro with or without estradiol.296 A decrease in adrenaline-stimulated lipolysis was observed in adipocytes from subcutaneous E2-treated adipose tissue fragments. E2 also increased the number of antilipolytic α2A-adrenergic receptors in subcutaneous adipocytes. These results suggested that estrogen lowered lipolysis by increasing the number of antilipolytic α2A-adrenergic receptors. In contrast, no effect of estrogen was observed in adipocytes from intra-abdominal fat. This difference in the E2 effect on α2A-adrenergic receptors between subcutaneous fat and intra-abdominal fat may be one factor to explain how estrogen maintains the gynoid fat pattern in women. This increase in α2A-adrenergic receptors with estrogen in subcutaneous fat from women may also explain the difference in response to estrogen between humans and rats as adipose tissue from rats do not have α2A-adrenergic receptors.308
4.3.2 Phosphoinositide cascade
The phosphoinositide cascade also appears to be regulated by sex steroid hormones. Protein kinase C content of rat subcutaneous and parametrial fat cells are reduced by ovariectomy and restored to normal by E2 and progesterone treatment.309 Lacasa et al.310 demonstrated that ovariectomy increased proliferation, MAPK activity, and c-fos protein in preadipocytes from rat perirenal fat depots. Treatment of the ovariectomized animals with E2 and progesterone reversed the increased proliferation and c-fos protein induction, but not the MAPK activation. The ovarian status had no effect on these parameters in subcutaneous preadipocytes, indicating that this regulation is depot specific. Recently, Dos Santos et al.298 found that estrogen activated p42/p44 MAPK by phosphorylation in rat white adipocytes through a rapid nongenomic pathway. This pathway was postulated to involve membrane ERα, G protein, protein kinase C, raf, and MAPK kinase (MEK). The phosphoinositide cascade appears to be involved in adipose proliferation and differentiation.
5. ESTROGENS INTERACT WITH LEPTIN
5.1. Estrogen interact with leptin in the CNS
Leptin, the product of the obese gene, is a key adipocytokine hormone that is normally released by adipose tissue in direct proportion to fat content and is related to the amount and distribution of body fat.311 It is a potent anorexigenic and catabolic hormone in central regulation of metabolism by crossing the blood–brain barrier to interact with leptin receptors (lepr) in the hypothalamus. Although there are several isoforms of the lepr, the b form (leprb) is the critical variant for regulating energy balance.312 Leprb has been identified in the ARC, PVN, and VMH, where they colocalize with ERα.313 Leptin transfers a catabolic signal to the brain to inhibit food intake and increase energy expenditure.314–316 Thus, leptin plays a key role in the regulation of body fat and energy homeostasis. Estrogen not only modulates leptin receptor leprb mRNA in the ARC and VMH via an ERE in the lepr gene, but also increases hypothalamic sensitivity to leptin, altering peripheral fat distribution.317 In addition, raised levels of estrogens have been associated with increased leptin sensitivity in the brain,318 although ovariectomy reduces the sensitivity to leptin when compared with intact females, an effect that can be restored by E2 replacement. Furthermore, administration of E2 in male rats increases the sensitivity to leptin.20 Thus, interactions between E2 and leptin exist for the regulation of energy homeostasis.313,319
5.2. Estrogen interact with leptin in adipose tissue
Leptin levels are higher in females compared with males.320 After puberty, estrogenmodulates leptin synthesis and secretion via ER-dependent transcriptional mechanisms in adipose tissue.321 The presence of rising intracellular bioactive E2 stimulates leptin production by fat cells.236 In support of this, Hardie et al.322 reported a correlation between serum leptin and estrogen levels. As estrogen levels decrease with menopause, a decrease in leptin levels might be expected with menopause.320,323,324 Additionally, increase in leptin with estrogen replacement therapy has been reported.324,325 Thomas et al.326 found that bioavailable estrogen levels in postmenopausal women who were not on hormone replacement therapy are significantly and directly related to serum leptin levels, but in premenopausal women or in women on hormone replacement therapy, variations in estrogen level are not associated with the higher leptin levels, suggesting a possible threshold effect. The most convincing evidence that the sex steroid hormones affect blood leptin levels comes from studies on cross-sex administration of sex steroid hormones to transsexuals. Elbers et al.327 were able to show that estrogen and antiandrogen administration to male-to-female transsexuals greatly increased the serum leptin levels and testosterone administration to female-to-male transsexuals decreased the serum leptin levels. They concluded that the sex steroid milieu plays an important role in the regulation of blood leptin levels.
Studying human omental adipose tissue in vitro, Casabiell et al.328 demonstrated that leptin secretion was higher in tissue from women than in tissue from men. Dexamethasone and E2 stimulated leptin secretion in fat from women, but not in fat from men. The increased E2237,329 and the increased leptin330 both upregulate aromatase to further increase extra-glandular E2 production,237 to increase subcutaneous fat deposition331 and to potentiate the action of E2 on ERα332 and transmembrane GPER.333
As in humans, female rats have higher circulating levels of leptin than do males.334–336 Ovariectomy of female rats has been shown by some investigators to lower circulating leptin levels, and estrogen replacement in these animals has been found to return the levels back to normal.320,337 Other investigators, however, found no effect on circulating leptin levels after ovariectomy and estrogen replacement.321,334–336,338,339 These discrepancies could be caused by the time of the blood samples, as Chu et al.340 showed that leptin levels declined during the first 7 weeks after ovariectomy. Subsequently, its concentration increased, and by the end of 13 weeks the value was higher than the level before ovariectomy. The fluctuation of serum leptin levels with ovariectomy was eliminated by estrogen replacement. These discrepancies could also be caused by weight gain in the ovariectomized animals, as leptin levels are related to the amount of fat, which was not always controlled in these experiments.
Ovariectomy has also been reported by some authors to decrease leptin gene expression in white adipose tissues of rats, and this decrease can be reversed by estrogen replacement.320,321,337,339 In contrast, other authors have reported no effect of estrogen replacement on leptin gene expression in white adipose tissues.336,338 These discrepancies could be caused by differences in leptin secretion and regulation in various adipose sites, as Shimizu et al.320 showed that although leptin gene expression decreased in subcutaneous and retroperitoneal white adipose tissues of ovariectomized rats as compared to controls, leptin gene expression actually increased in mesenteric white adipose tissues of the ovariectomized rats.
Machinal et al.321 also observed that ovariectomy resulted in a 25% decrease in leptin gene expression in isolated perirenal adipocytes and a small but insignificant decrease in subcutaneous adipocytes. Therefore, the regional distribution of adipose tissues may greatly influence leptin levels and its regulation by sex steroid hormones. Basal leptin production is equal in epididymal fat fragments from males and parametrial fat fragments from females. E2 increased leptin secretion in both male and female adipose fragments, but the effect was greater in female adipose tissue. Parametrial fat from ovariectomized rats had lower leptin secretion compared to shamoperated controls, and E2 treatment of ovariectomized rats maintained a normal leptin secretion in fat fragments.
Sex steroid hormone regulation of leptin production has also been studied in normal and mutant mice. Nedvidkova et al. showed a decrease of serum leptin levels in estrogen-treated male mice.341 However, in female mice, Pelleymounter et al. reported that ovariectomy or E2 administration did not alter leptin levels.342 Kronfeld-Schor et al. demonstrated that subcutaneous implants of E2 or corticosterone into lactating mice stimulated leptin secretion rates of omental adipose tissue in vitro, and corticosterone, but not estradiol, stimulated leptin secretion rates when added to isolated omental adipose tissue.343 From this study, the authors suggest that E2 could be acting indirectly on the omental adipose tissue possibly through an increase in plasma corticosterone levels.
In ERα KO male mice, Ohlsson et al. demonstrated an enhanced serum leptin level as compared to controls that was not seen in ERβ KO mice.67 Jones et al. showed an elevation of leptin levels in ArKO male and female mice.247 However, both the ERα KO mice and ArKO mice had an increase in fat mass, which could account for the increased leptin levels.
In summary, estrogens appear to increase leptin production. However, many discrepancies exist in the data. If sex steroid hormones are going to directly regulate the production of leptin, an ERE should be present in the leptin gene. After reviewing the cDNA sequence of the leptin gene, Shimizu et al.320 concluded that this gene had a consensus sequence of the ERE in its promoter region. These results suggest that the leptin gene has an ERE; that leptin promoter activation may depend upon coactivators present in leptin-producing cells; and that different effects of estrogen may be due to the type of ER expressed in target tissue.
6. CONCLUSIONS
Obesity and its associated health-related problems have become so prevalent in developed countries that many programs and strategies have been developed to prevent and/or treat this affliction, but without much success.344,345 Although sex steroid hormones have a major role in lipid metabolism and deposition in adipose tissues, they or their derivatives have not been extensively used to prevent or treat obesity.
There is abundant evidence that estrogen can utilize both genomic and nongenomic signaling mechanisms to regulate energy homeostasis, body fat, and glucose regulation. Estrogens exert rapid nongenomic effects in the brain and adipose tissues to regulate second messengers and kinase signaling pathways, and play physiologically important roles in estrogen-induced regulation of homeostasis.
One area where sex steroid hormones appear to have a positive effect on obesity is in hormone replacement therapy in older women. Menopause in women causes a large decrease in endogenous estrogens, which leads to central obesity and an increased risk in metabolic disorders. A relationship of central obesity and its complications to the decrease in sex steroid hormones is unclear. If it is related, hormone replacement therapy in postmenopausal women should reduce the occurrence of these problems.346 Several studies were able to show that hormone replacement therapy did decrease central obesity in postmenopausal women and hence prevent at least some of the problems of menopause.18,347–354 In contrast, other studies have not been able to confirm the decrease in central obesity with hormone treatment. 17,355,356 In contrast, a recent report showed that hormone replacement therapy in postmenopausal women results in an increased risk for coronary heart disease, stroke, thromboembolic events, and breast cancer,357 suggesting caution when using sex steroid treatment. For the amelioration of obesity by sex steroid hormones, it would be helpful to develop compounds that would be specific for adipose tissues and that would not have the adverse side effects of estrogen. Considerable time was devoted to the need for SERMs that are adipose tissue specific and have pathways related to lipid metabolism for safe intervention in a number of endocrine-regulated conditions.358 SERMs, such as estren359 and CP-336,156,360 that were shown to prevent bone loss in animal models without serious side effects, and CP-336,156 that was shown to prevent the increase in body fat in ovariectomized rats, may be an option for obesity regulation in the future.
Regulation of some key proteins in adipose tissues by estrogens, such as downregulation of LPL in visceral adipose tissue, may also be a mechanism for the treatment and/or prevention of obesity.361 Estrogens are known to decrease LPL activity at least in gluteal adipose tissue, which may be important in body fat distribution.292
Leptin is another adipose protein that could be a target for regulation by estrogens to control central obesity. The extension of leptin therapy in experimental animals to humans has been only partially successful.362,363 In patients with a genetic defect of leptin production, leptin therapy greatly reduces the obesity in these individuals.364 However, these individuals only represent an extremely small percentage of the obese population, and in the common diet-induced obesity, leptin therapy remains questionable because leptin levels are very high in obese individuals. The high leptin levels in obese individuals might suggest that exogenous leptin therapy would be ineffective in decreasing fat stores. It is possible that regulation of endogenous leptin by sex steroid hormones would be more effective than exogenous leptin.
By gaining a more thorough understanding of estrogen’s effects on the CNS and peripheral adipose tissues, and a better grasp of the signaling cascade involved, we will gain new insights that lead to better diagnostic tools and improved therapeutics in humans.
ACKNOWLEDGMENTS
This work was supported by the National Institutes of Health National Institute of Diabetes and Digestive and Kidney Diseases (DK090823) and the American Heart Association (SDG4520028) to H. S., and doctoral fellowship from the Cellular, Molecular and Structural Biology program to S. P. D. S. K. and X. L.
ABBREVIATIONS
- α-MSH
α-melanocyte stimulating hormone
- AgRP
agouti-related protein
- ARC
arcuate nucleus
- cAMP
cyclic AMP
- CNS
central nervous system
- E2
17β-estradiol
- EGFRs
epidermal growth factor receptors
- ERs
estrogen receptors
- Erα
estrogen receptor alpha
- ERβ
estrogen receptor beta
- EREs
estrogen response elements
- GLUT4
glucose transporter 4
- GPER
G protein-coupled estrogen receptor
- HSL
hormone-sensitive lipase
- LH
lateral hypothalamus
- LPL
lipoprotein lipase
- mER
membrane subpopulation estrogen receptors
- NPY
neuropeptide Y
- PI3K
phosphoinositide-3 kinases
- PKA
protein kinase A
- PKB
protein kinase B (Akt)
- POMC
pro-opiomelanocortin
- PTX
pertussis toxin
- PVN
paraventricular nucleus
- SERM
selective estrogen receptor modulators
- STZ
streptozotocin
- VMH
ventromedial hypothalamus
REFERENCES
- 1.James WPT. The epidemiology of obesity: the size of the problem. J Intern Med. 2008;263(4):336–352. doi: 10.1111/j.1365-2796.2008.01922.x. [DOI] [PubMed] [Google Scholar]
- 2.Cummings DE, Schwartz MW. Genetics and Pathophysiology of Human Obesity. Annu Rev Med. 2003;54(1):453–471. doi: 10.1146/annurev.med.54.101601.152403. [DOI] [PubMed] [Google Scholar]
- 3.Alberti K, Zimmet P, Shaw J. IDF epidemiology task force consensus group: the metabolic syndrome—a worldwide definition. Lancet. 2005;366:1059–1062. doi: 10.1016/S0140-6736(05)67402-8. [DOI] [PubMed] [Google Scholar]
- 4.McGarry JD. Banting lecture 2001: dysregulation of fatty acid metabolism in the etiology of type 2 diabetes. Diabetes. 2002;51(1):7–18. doi: 10.2337/diabetes.51.1.7. [DOI] [PubMed] [Google Scholar]
- 5.Xu H, et al. Chronic inflammation in fat plays a crucial role in the development of obesity-related insulin resistance. J Clin Invest. 2003;112(12):1821–1830. doi: 10.1172/JCI19451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kolovou G, et al. Apolipoprotein E gene polymorphism and gender. Ann Clin Lab Sci. 2009;39(2):120–133. [PubMed] [Google Scholar]
- 7.Ford ES. Prevalence of the metabolic syndrome defined by the International Diabetes Federation among adults in the U.S. Diabetes Care. 2005;28(11):2745–2749. doi: 10.2337/diacare.28.11.2745. [DOI] [PubMed] [Google Scholar]
- 8.Kannel WB, et al. Regional obesity and risk of cardiovascular disease; the Framingham study. J Clin Epidemiol. 1991;44(2):183–190. doi: 10.1016/0895-4356(91)90265-b. [DOI] [PubMed] [Google Scholar]
- 9.Lee CG, et al. Adipokines, inflammation, and visceral adiposity across the menopausal transition: a prospective study. J Clin Endocrinol Metab. 2009;94(4):1104–1110. doi: 10.1210/jc.2008-0701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Regitz-Zagrosek V, Lehmkuhl E, Weickert M. Gender differences in the metabolic syndrome and their role for cardiovascular disease. Clin Res Cardiol. 2006;95(3):136–147. doi: 10.1007/s00392-006-0351-5. [DOI] [PubMed] [Google Scholar]
- 11.Bjoörntorp P. Body fat distribution, insulin resistance, and metabolic diseases. Nutrition. 1997;13(9):795–803. doi: 10.1016/s0899-9007(97)00191-3. [DOI] [PubMed] [Google Scholar]
- 12.Björntorp P. Hormonal control of regional fat distribution. Hum Reprod. 1997;12(Suppl. 1):21–25. doi: 10.1093/humrep/12.suppl_1.21. [DOI] [PubMed] [Google Scholar]
- 13.Kotani K, et al. Sexual dimorphism of age-related changes in whole-body fat distribution in the obese. Int J Obes Relat Metab Disord. 1994;18(4):207–212. [PubMed] [Google Scholar]
- 14.Carr MC. The emergence of the metabolic syndrome with menopause. J Clin Endocrinol Metab. 2003;88(6):2404–2411. doi: 10.1210/jc.2003-030242. [DOI] [PubMed] [Google Scholar]
- 15.Krotkiewski M, et al. Impact of obesity on metabolism in men and women. Importance of regional adipose tissue distribution. J Clin Invest. 1983;72(3):1150–1162. doi: 10.1172/JCI111040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Bouchard C, Despres J-P, Mauriege P. Genetic and nongenetic determinants of regional fat distribution. Endocr Rev. 1993;14(1):72–93. doi: 10.1210/edrv-14-1-72. [DOI] [PubMed] [Google Scholar]
- 17.Ryan AS, Nicklas BJ, Berman DM. Hormone replacement therapy, insulin sensitivity, and abdominal obesity in postmenopausal women. Diabetes Care. 2002;25(1):127–133. doi: 10.2337/diacare.25.1.127. [DOI] [PubMed] [Google Scholar]
- 18.Samaras K, et al. Effects of postmenopausal hormone replacement therapy on central abdominal fat, glycemic control, lipid metabolism, and vascular factors in type 2 diabetes: a prospective study. Diabetes Care. 1999;22(9):1401–1407. doi: 10.2337/diacare.22.9.1401. [DOI] [PubMed] [Google Scholar]
- 19.Blaustein JD, Wade GN. Ovarian influences on the meal patterns of female rats. Physiol Behav. 1976;17(2):201–208. doi: 10.1016/0031-9384(76)90064-0. [DOI] [PubMed] [Google Scholar]
- 20.Clegg DJ, et al. Gonadal hormones determine sensitivity to central leptin and insulin. Diabetes. 2006;55(4):978–987. doi: 10.2337/diabetes.55.04.06.db05-1339. [DOI] [PubMed] [Google Scholar]
- 21.Wallen WJ, Belanger MP, Wittnich C. Sex hormones and the selective estrogen receptor modulator tamoxifen modulate weekly body weights and food intakes in adolescent and adult rats. J Nutr. 2001;131(9):2351–2357. doi: 10.1093/jn/131.9.2351. [DOI] [PubMed] [Google Scholar]
- 22.Roy EJ, Wade GN. Role of food intake in estradiol-induced body weight changes in female rats. Horm Behav. 1977;8(3):265–274. doi: 10.1016/0018-506x(77)90001-0. [DOI] [PubMed] [Google Scholar]
- 23.Gao Q, et al. Anorectic estrogen mimics leptin’s effect on the rewiring of melanocortin cells and Stat3 signaling in obese animals. Nat Med. 2007;13(1):89–94. doi: 10.1038/nm1525. [DOI] [PubMed] [Google Scholar]
- 24.D’Eon TM, et al. Estrogen regulation of adiposity and fuel partitioning Evidence of genomic and non-genomic regulation of lipogenic and oxidative pathways. J Biol Chem. 2005;280(43):35983–35991. doi: 10.1074/jbc.M507339200. [DOI] [PubMed] [Google Scholar]
- 25.Gao H, et al. Long-term administration of estradiol decreases expression of hepatic lipogenic genes and improves insulin sensitivity in ob/ob Mice: a possible mechanism is through direct regulation of signal transducer and activator of transcription 3. Mol Endocrinol. 2006;20(6):1287–1299. doi: 10.1210/me.2006-0012. [DOI] [PubMed] [Google Scholar]
- 26.Espeland MA, et al. Effect of postmenopausal hormone therapy on lipoprotein(a) concentration. PEPI Investigators. Postmenopausal estrogen/progestin interventions. Circulation. 1998;97(10):979–986. doi: 10.1161/01.cir.97.10.979. [DOI] [PubMed] [Google Scholar]
- 27.Redig AJ, Munshi HG. Care of the cancer survivor: metabolic syndrome after hormone-modifying therapy. Am J Med. 2010;123(1):87.e1–87.e6. doi: 10.1016/j.amjmed.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Tiano JP, et al. Estrogen receptor activation reduces lipid synthesis in pancreatic islets and prevents b cell failure in rodent models of type 2 diabetes. J Clin Invest. 2011;121(8):3331–3342. doi: 10.1172/JCI44564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Simpson ER, et al. Estrogen—the good, the bad, the unexpected. Endocr Rev. 2005;26(3):322–330. doi: 10.1210/er.2004-0020. [DOI] [PubMed] [Google Scholar]
- 30.Maffei L, et al. Dysmetabolic syndrome in a man with a novel mutation of the aromatase gene: effects of testosterone, alendronate, and estradiol treatment. J Clin Endocrinol Metab. 2004;89(1):61–70. doi: 10.1210/jc.2003-030313. [DOI] [PubMed] [Google Scholar]
- 31.Morishima A, et al. Aromatase deficiency in male and female siblings caused by a novel mutation and the physiological role of estrogens. J Clin Endocrinol Metab. 1995;80(12):3689–3698. doi: 10.1210/jcem.80.12.8530621. [DOI] [PubMed] [Google Scholar]
- 32.Bilezikian JP, et al. Increased bone mass as a result of estrogen therapy in a man with aromatase deficiency. N Engl J Med. 1998;339(9):599–603. doi: 10.1056/NEJM199808273390905. [DOI] [PubMed] [Google Scholar]
- 33.Rochira V, et al. Oestradiol replacement treatment and glucose homeostasis in two men with congenital aromatase deficiency: evidence for a role of oestradiol and sex steroids imbalance on insulin sensitivity in men. Diabet Med. 2007;24(12):1491–1495. doi: 10.1111/j.1464-5491.2007.02304.x. [DOI] [PubMed] [Google Scholar]
- 34.Takeda K, et al. Progressive development of insulin resistance phenotype in male mice with complete aromatase (CYP19) deficiency. J Endocrinol. 2003;176(2):237–246. doi: 10.1677/joe.0.1760237. [DOI] [PubMed] [Google Scholar]
- 35.Deroo BJ, Korach KS. Estrogen receptors and human disease. J Clin Invest. 2006;116(3):561–570. doi: 10.1172/JCI27987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hess R. Estrogen in the adult male reproductive tract: a review. Reprod Biol Endocrinol. 2003;1(1):52. doi: 10.1186/1477-7827-1-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Baquedano MS, et al. Identification and developmental changes of aromatase and estrogen receptor expression in prepubertal and pubertal human adrenal tissues. J Clin Endocrinol Metab. 2007;92(6):2215–2222. doi: 10.1210/jc.2006-2329. [DOI] [PubMed] [Google Scholar]
- 38.Kelly MJ, Qiu J, Rønnekleiv OK. Gerald L. Vitamins & hormones. Academic Press; 2005. Estrogen signaling in the hypothalamus; pp. 123–145. [DOI] [PubMed] [Google Scholar]
- 39.McEwen BS, Alves SE. Estrogen actions in the central nervous system. Endocr Rev. 1999;20(3):279–307. doi: 10.1210/edrv.20.3.0365. [DOI] [PubMed] [Google Scholar]
- 40.Teede HJ. Sex hormones and the cardiovascular system: effects on arterial function in women. Clin Exp Pharmacol Physiol. 2007;34(7):672–676. doi: 10.1111/j.1440-1681.2007.04658.x. [DOI] [PubMed] [Google Scholar]
- 41.Turner RT, Riggs BL, Spelsberg TC. Skeletal effects of estrogen. Endocr Rev. 1994;15(3):275–300. doi: 10.1210/edrv-15-3-275. [DOI] [PubMed] [Google Scholar]
- 42.Gruber CJ, et al. Production and actions of estrogens. N Engl J Med. 2002;346(5):340–352. doi: 10.1056/NEJMra000471. [DOI] [PubMed] [Google Scholar]
- 43.Muller RE, et al. Studies on the mechanism of estradiol uptake by rat uterine cells and on estradiol binding to uterine plasma membranes. Adv Exp Med Biol. 1979;117:401–421. doi: 10.1007/978-1-4757-6589-2_22. [DOI] [PubMed] [Google Scholar]
- 44.Jackson V, Chalkley R. The binding of estradiol-17 beta to the bovine endometrial nuclear membrane. J Biol Chem. 1974;249(5):1615–1626. [PubMed] [Google Scholar]
- 45.Revankar CM, et al. A transmembrane intracellular estrogen receptor mediates rapid cell signaling. Science. 2005;307(5715):1625–1630. doi: 10.1126/science.1106943. [DOI] [PubMed] [Google Scholar]
- 46.Nadal A, et al. Nongenomic actions of estrogens and xenoestrogens by binding at a plasma membrane receptor unrelated to estrogen receptor α and estrogen receptor β. Proc Natl Acad Sci U S A. 2000;97(21):11603–11608. doi: 10.1073/pnas.97.21.11603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Meyer MR, et al. Non-genomic regulation of vascular cell function and growth by estrogen. Mol Cell Endocrinol. 2009;308(1–2):9–16. doi: 10.1016/j.mce.2009.03.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Walter P, et al. Cloning of the human estrogen receptor cDNA. Proc Natl Acad Sci U S A. 1985;82(23):7889–7893. doi: 10.1073/pnas.82.23.7889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kuiper GG, et al. Cloning of a novel receptor expressed in rat prostate and ovary. Proc Natl Acad Sci U S A. 1996;93(12):5925–5930. doi: 10.1073/pnas.93.12.5925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Greene G, et al. Sequence and expression of human estrogen receptor complementary DNA. Science. 1986;231(4742):1150–1154. doi: 10.1126/science.3753802. [DOI] [PubMed] [Google Scholar]
- 51.Green S, et al. Human oestrogen receptor cDNA: sequence, expression and homology to v-erb-A. Nature. 1986;320(6058):134–139. doi: 10.1038/320134a0. [DOI] [PubMed] [Google Scholar]
- 52.Jensen EV, DeSombre ER. Estrogen–receptor interaction. Science. 1973;182(4108):126–134. doi: 10.1126/science.182.4108.126. [DOI] [PubMed] [Google Scholar]
- 53.Brzozowski AM, et al. Molecular basis of agonism and antagonism in the oestrogen receptor. Nature. 1997;389(6652):753–758. doi: 10.1038/39645. [DOI] [PubMed] [Google Scholar]
- 54.Stauffer SR, et al. Pyrazole ligands: structure–affinity/activity relationships and estrogen receptor-alpha-selective agonists. J Med Chem. 2000;43(26):4934–4947. doi: 10.1021/jm000170m. [DOI] [PubMed] [Google Scholar]
- 55.Meyer MR, et al. Dilation of epicardial coronary arteries by the G protein-coupled estrogen receptor agonists G-1 and ICI 182,780. Pharmacology. 2010;86(1):58–64. doi: 10.1159/000315497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Malamas MS, et al. Design and synthesis of aryl diphenolic azoles as potent and selective estrogen receptor-beta ligands. J Med Chem. 2004;47(21):5021–5040. doi: 10.1021/jm049719y. [DOI] [PubMed] [Google Scholar]
- 57.Hammes SR, Levin ER. Extranuclear steroid receptors: nature and actions. Endocr Rev. 2007;28(7):726–741. doi: 10.1210/er.2007-0022. [DOI] [PubMed] [Google Scholar]
- 58.Hall JM, Couse JF, Korach KS. The multifaceted mechanisms of estradiol and estrogen receptor signaling. J Biol Chem. 2001;276(40):36869–36872. doi: 10.1074/jbc.R100029200. [DOI] [PubMed] [Google Scholar]
- 59.Nilsson S, et al. Mechanisms of estrogen action. Physiol Rev. 2001;81(4):1535–1565. doi: 10.1152/physrev.2001.81.4.1535. [DOI] [PubMed] [Google Scholar]
- 60.Thomas C, Gustafsson J-Å, et al. The different roles of ER subtypes in cancer biology and therapy. Nat Rev Cancer. 2011;11(8):597–608. doi: 10.1038/nrc3093. [DOI] [PubMed] [Google Scholar]
- 61.Handa RJ, et al. Roles for oestrogen receptor b in adult brain function. J Neuroendocrinol. 2012;24(1):160–173. doi: 10.1111/j.1365-2826.2011.02206.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kuiper GGJM, et al. Comparison of the ligand binding specificity and transcript tissue distribution of estrogen receptors alpha and beta. Endocrinology. 1997;138(3):863–870. doi: 10.1210/endo.138.3.4979. [DOI] [PubMed] [Google Scholar]
- 63.Matthews J, Gustafsson J-Å, et al. Estrogen signaling: a subtle balance between ER alpha and ER beta. Mol Interv. 2003;3(5):281–292. doi: 10.1124/mi.3.5.281. [DOI] [PubMed] [Google Scholar]
- 64.Hewitt S, Korach K. Oestrogen receptor knockout mice: roles for oestrogen receptors alpha and beta in reproductive tissues. Reproduction. 2003;125(2):143–149. doi: 10.1530/rep.0.1250143. [DOI] [PubMed] [Google Scholar]
- 65.Heine PA, et al. Increased adipose tissue in male and female estrogen receptor-a knockout mice. P Natl Acad Sci USA. 2000;97(23):12729–12734. doi: 10.1073/pnas.97.23.12729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Geary N, et al. Deficits in E2-dependent control of feeding, weight gain, and cholecystokinin satiation in ER-α null mice. Endocrinology. 2001;142(11):4751–4757. doi: 10.1210/endo.142.11.8504. [DOI] [PubMed] [Google Scholar]
- 67.Ohlsson C, et al. Obesity and disturbed lipoprotein profile in estrogen receptora- deficient male mice. Biochem Biophys Res Commun. 2000;278(3):640–645. doi: 10.1006/bbrc.2000.3827. [DOI] [PubMed] [Google Scholar]
- 68.Shughrue PJ, Lane MV, Merchenthaler I. Comparative distribution of estrogen receptor-alpha and -beta mRNA in the rat central nervous system. J Comp Neurol. 1997;388(4):507–525. doi: 10.1002/(sici)1096-9861(19971201)388:4<507::aid-cne1>3.0.co;2-6. [DOI] [PubMed] [Google Scholar]
- 69.Nilsson S, Gustafsson JA. Estrogen receptors: therapies targeted to receptor subtypes. Clin Pharmacol Ther. 2011;89(1):44–55. doi: 10.1038/clpt.2010.226. [DOI] [PubMed] [Google Scholar]
- 70.Liang YQ, et al. Estrogen receptor beta is involved in the anorectic action of estrogen. Int J Obes Relat Metab Disord. 2002;26(8):1103–1109. doi: 10.1038/sj.ijo.0802054. [DOI] [PubMed] [Google Scholar]
- 71.O’Lone R, et al. Genomic targets of nuclear estrogen receptors. Mol Endocrinol. 2004;18(8):1859–1875. doi: 10.1210/me.2003-0044. [DOI] [PubMed] [Google Scholar]
- 72.Edwards DP. Regulation of signal transduction pathways by estrogen and progesterone. Annu Rev Physiol. 2005;67(1):335–376. doi: 10.1146/annurev.physiol.67.040403.120151. [DOI] [PubMed] [Google Scholar]
- 73.Losel RM, et al. Nongenomic steroid action: controversies, questions, and answers. Physiol Rev. 2003;83(3):965–1016. doi: 10.1152/physrev.00003.2003. [DOI] [PubMed] [Google Scholar]
- 74.Brann DW, Hendry LB, Mahesh VB. Emerging diversities in the mechanism of action of steroid hormones. J Steroid Biochem Mol Biol. 1995;52(2):113–133. doi: 10.1016/0960-0760(94)00160-n. [DOI] [PubMed] [Google Scholar]
- 75.Manavathi B, Kumar R. Steering estrogen signals from the plasma membrane to the nucleus: two sides of the coin. J Cell Physiol. 2006;207(3):594–604. doi: 10.1002/jcp.20551. [DOI] [PubMed] [Google Scholar]
- 76.Moriarty K, Kim KH, Bender JR. Minireview: estrogen receptor-mediated rapid signaling. Endocrinology. 2006;147(12):5557–5563. doi: 10.1210/en.2006-0729. [DOI] [PubMed] [Google Scholar]
- 77.Mhyre AJ, Dorsa DM. Estrogen activates rapid signaling in the brain: role of estrogen receptor α and estrogen receptor β in neurons and glia. Neuroscience. 2006;138(3):851–858. doi: 10.1016/j.neuroscience.2005.10.019. [DOI] [PubMed] [Google Scholar]
- 78.Boonyaratanakornkit V, Edwards DP. Receptor mechanisms mediating non-genomic actions of sex steroids. Semin Reprod Med. 2007;25(03) doi: 10.1055/s-2007-973427. 139,153. [DOI] [PubMed] [Google Scholar]
- 79.Szego CM, Davis JS. Adenosine 3’,5’-monophosphate in rat uterus: acute elevation by estrogen. Proc Natl Acad Sci U S A. 1967;58(4):1711–1718. doi: 10.1073/pnas.58.4.1711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pietras RJ, Szego CM. Specific binding sites for oestrogen at the outer surfaces of isolated endometrial cells. Nature. 1977;265(5589):69–72. doi: 10.1038/265069a0. [DOI] [PubMed] [Google Scholar]
- 81.Carmeci C, et al. Identification of a gene (GPR30) with homology to the G-proteincoupled receptor superfamily associated with estrogen receptor expression in breast cancer. Genomics. 1997;45(3):607–617. doi: 10.1006/geno.1997.4972. [DOI] [PubMed] [Google Scholar]
- 82.Filardo EJ, et al. Estrogen action via the G protein-coupled receptor, GPR30: stimulation of adenylyl cyclase and cAMP-mediated attenuation of the epidermal growth factor receptor-to-MAPK signaling axis. Mol Endocrinol. 2002;16(1):70–84. doi: 10.1210/mend.16.1.0758. [DOI] [PubMed] [Google Scholar]
- 83.Prossnitz ER, et al. Estrogen signaling through the transmembrane G protein-coupled receptor GPR30. Annu Rev Physiol. 2008;70(1):165–190. doi: 10.1146/annurev.physiol.70.113006.100518. [DOI] [PubMed] [Google Scholar]
- 84.Thomas P, et al. Identity of an estrogen membrane receptor coupled to a G protein in human breast cancer cells. Endocrinology. 2005;146(2):624–632. doi: 10.1210/en.2004-1064. [DOI] [PubMed] [Google Scholar]
- 85.Filardo EJ, et al. Estrogen-induced activation of Erk-1 and Erk-2 requires the G protein-coupled receptor homolog, GPR30, and occurs via trans-activation of the epidermal growth factor receptor through release of HB-EGF. Mol Endocrinol. 2000;14(10):1649–1660. doi: 10.1210/mend.14.10.0532. [DOI] [PubMed] [Google Scholar]
- 86.Maggiolini M, et al. The G protein-coupled receptor GPR30 mediates c-fos upregulation by 17beta-estradiol and phytoestrogens in breast cancer cells. J Biol Chem. 2004;279(26):27008–27016. doi: 10.1074/jbc.M403588200. [DOI] [PubMed] [Google Scholar]
- 87.Qiu J, et al. Rapid signaling of estrogen in hypothalamic neurons involves a novel G-protein-coupled estrogen receptor that activates protein kinase C. J Neurosci. 2003;23(29):9529–9540. doi: 10.1523/JNEUROSCI.23-29-09529.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Qiu J, et al. A G-protein-coupled estrogen receptor is involved in hypothalamic control of energy homeostasis. J Neurosci. 2006;26(21):5649–5655. doi: 10.1523/JNEUROSCI.0327-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Roepke TA, et al. Genes associated with membrane-initiated signaling of estrogen and energy homeostasis. Endocrinology. 2008;149(12):6113–6124. doi: 10.1210/en.2008-0769. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Roepke TA, et al. Contribution of a membrane estrogen receptor to the estrogenic regulation of body temperature and energy homeostasis. Endocrinology. 2010;151(10):4926–4937. doi: 10.1210/en.2010-0573. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Toran-Allerand CD, et al. ER-X: a novel, plasma membrane-associated, putative estrogen receptor that is regulated during development and after ischemic brain injury. J Neurosci. 2002;22(19):8391–8401. doi: 10.1523/JNEUROSCI.22-19-08391.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang Z, et al. Identification, cloning, and expression of human estrogen receptor-α36, a novel variant of human estrogen receptor-α66. Biochem Biophys Res Commun. 2005;336(4):1023–1027. doi: 10.1016/j.bbrc.2005.08.226. [DOI] [PubMed] [Google Scholar]
- 93.Prossnitz ER, Barton M. Signaling, physiological functions and clinical relevance of the G protein-coupled estrogen receptor GPER. Prostaglandins Other Lipid Mediat. 2009;89(3–4):89–97. doi: 10.1016/j.prostaglandins.2009.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.O’Dowd BF, et al. Discovery of three novel G-protein-coupled receptor genes. Genomics. 1998;47(2):310–313. doi: 10.1006/geno.1998.5095. [DOI] [PubMed] [Google Scholar]
- 95.Balhuizen A, et al. Activation of G protein-coupled receptor 30 modulates hormone secretion and counteracts cytokine-induced apoptosis in pancreatic islets of female mice. Mol Cell Endocrinol. 2010;320(1–2):16–24. doi: 10.1016/j.mce.2010.01.030. [DOI] [PubMed] [Google Scholar]
- 96.Hugo ER, et al. Bisphenol A at environmentally relevant doses inhibits adiponectin release from human adipose tissue explants and adipocytes. Environ Health Perspect. 2008;116(12):1642–1647. doi: 10.1289/ehp.11537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kanda N, Watanabe S. 17[beta]-estradiol inhibits oxidative stress-induced apoptosis in keratinocytes by promoting Bcl-2 expression. J Invest Dermatol. 2003;121(6):1500–1509. doi: 10.1111/j.1523-1747.2003.12617.x. [DOI] [PubMed] [Google Scholar]
- 98.Mårtensson UEA, et al. Deletion of the G protein-coupled receptor 30 impairs glucose tolerance, reduces bone growth, increases blood pressure, and eliminates estradiolstimulated insulin release in female mice. Endocrinology. 2009;150(2):687–698. doi: 10.1210/en.2008-0623. [DOI] [PubMed] [Google Scholar]
- 99.Nadal A, et al. The role of oestrogens in the adaptation of islets to insulin resistance. J Physiol. 2009;587(21):5031–5037. doi: 10.1113/jphysiol.2009.177188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Haas E, et al. Regulatory role of G protein-coupled estrogen receptor for vascular function and obesity. Circ Res. 2009;104(3):288–291. doi: 10.1161/CIRCRESAHA.108.190892. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Liu S, et al. Importance of extranuclear estrogen receptor-alpha and membrane G protein-coupled estrogen receptor in pancreatic islet survival. Diabetes. 2009;58(10):2292–2302. doi: 10.2337/db09-0257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Blasko E, et al. Beneficial role of the GPR30 agonist G-1 in an animal model of multiple sclerosis. J Neuroimmunol. 2009;214(1–2):67–77. doi: 10.1016/j.jneuroim.2009.06.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Noel SD, et al. Involvement of G protein-coupled receptor 30 (GPR30) in rapid action of estrogen in primate LHRH neurons. Mol Endocrinol. 2009;23(3):349–359. doi: 10.1210/me.2008-0299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Rettew JA, McCall SH, Marriott I. GPR30/GPER-1 mediates rapid decreases in TLR4 expression on murine macrophages. Mol Cell Endocrinol. 2010;328(1–2):87–92. doi: 10.1016/j.mce.2010.07.017. [DOI] [PubMed] [Google Scholar]
- 105.Terasawa E, Noel SD, Keen KL. Rapid action of oestrogen in luteinising hormonereleasing hormone neurones: the role of GPR30. J Neuroendocrinol. 2009;21(4):316–321. doi: 10.1111/j.1365-2826.2009.01839.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Brailoiu E, et al. Distribution and characterization of estrogen receptor G proteincoupled receptor 30 in the rat central nervous system. J Endocrinol. 2007;193(2):311–321. doi: 10.1677/JOE-07-0017. [DOI] [PubMed] [Google Scholar]
- 107.Owman C, et al. Cloning of human cDNA encoding a novel heptahelix receptor expressed in Burkitt’s lymphoma and widely distributed in brain and peripheral tissues. Biochem Biophys Res Commun. 1996;228(2):285–292. doi: 10.1006/bbrc.1996.1654. [DOI] [PubMed] [Google Scholar]
- 108.Takada Y, et al. Cloning of cDNAs encoding G protein-coupled receptor expressed in human endothelial cells exposed to fluid shear stress. Biochem Biophys Res Commun. 1997;240(3):737–741. doi: 10.1006/bbrc.1997.7734. [DOI] [PubMed] [Google Scholar]
- 109.Wang C, Prossnitz ER, Roy SK. Expression of G protein-coupled receptor 30 in the hamster ovary: differential regulation by gonadotropins and steroid hormones. Endocrinology. 2007;148(10):4853–4864. doi: 10.1210/en.2007-0727. [DOI] [PubMed] [Google Scholar]
- 110.Canonaco M, et al. A sexually dimorphic distribution pattern of the novel estrogen receptor G-protein-coupled receptor 30 in some brain areas of the hamster. J Endocrinol. 2008;196(1):131–138. doi: 10.1677/JOE-07-0392. [DOI] [PubMed] [Google Scholar]
- 111.Wang D, et al. G protein-coupled receptor 30 in tumor development. Endocrine. 2010;38(1):29–37. doi: 10.1007/s12020-010-9363-z. [DOI] [PubMed] [Google Scholar]
- 112.Pietras R, Nemere I, Szego C. Steroid hormone receptors in target cell membranes. Endocrine. 2001;14(3):417–427. doi: 10.1385/ENDO:14:3:417. [DOI] [PubMed] [Google Scholar]
- 113.Aronica SM, Kraus WL, Katzenellenbogen BS. Estrogen action via the cAMP signaling pathway: stimulation of adenylate cyclase and cAMP-regulated gene transcription. Proc Natl Acad Sci U S A. 1994;91(18):8517–8521. doi: 10.1073/pnas.91.18.8517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Ding Q, et al. Estradiol-mediated ERK phosphorylation and apoptosis in vascular smooth muscle cells requires GPR 30. Am J Physiol Cell Physiol. 2009;297(5):C1178–C1187. doi: 10.1152/ajpcell.00185.2009. [DOI] [PubMed] [Google Scholar]
- 115.Filardo E, et al. Activation of the novel estrogen receptor G protein-coupled receptor30 (GPR30) at the plasma membrane. Endocrinology. 2007;148(7):3236–3245. doi: 10.1210/en.2006-1605. [DOI] [PubMed] [Google Scholar]
- 116.Weil BR, et al. Signaling via GPR30 protects the myocardium from ischemia/reperfusion injury. Surgery. 2010;148(2):436–443. doi: 10.1016/j.surg.2010.03.011. [DOI] [PubMed] [Google Scholar]
- 117.Bologa CG, et al. Virtual and biomolecular screening converge on a selective agonist for GPR30. Nat Chem Biol. 2006;2(4):207–212. doi: 10.1038/nchembio775. [DOI] [PubMed] [Google Scholar]
- 118.Broughton BRS, Miller AA, Sobey CG. Endothelium-dependent relaxation by G protein-coupled receptor 30 agonists in rat carotid arteries. Am J Physiol Heart Circ Physiol. 2010;298(3):H1055–H1061. doi: 10.1152/ajpheart.00878.2009. [DOI] [PubMed] [Google Scholar]
- 119.Dennis MK, et al. In vivo effects of a GPR30 antagonist. Nat Chem Biol. 2009;5(6):421–427. doi: 10.1038/nchembio.168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Chow RWY, Handelsman DJ, Ng MKC. Minireview: rapid actions of sex steroids in the endothelium. Endocrinology. 2010;151(6):2411–2422. doi: 10.1210/en.2009-1456. [DOI] [PubMed] [Google Scholar]
- 121.Lin BC, et al. Stimulating the GPR30 estrogen receptor with a novel tamoxifen analogue activates SF-1 and promotes endometrial cell proliferation. Cancer Res. 2009;69(13):5415–5423. doi: 10.1158/0008-5472.CAN-08-1622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Meli R, et al. Estrogen and raloxifene modulate leptin and its receptor in hypothalamus and adipose tissue from ovariectomized rats. Endocrinology. 2004;145(7):3115–3121. doi: 10.1210/en.2004-0129. [DOI] [PubMed] [Google Scholar]
- 123.Baptista T, et al. Tamoxifen prevents sulpiride-induced weight gain in female rats. Pharmacol Biochem Behav. 1997;57(1–2):215–222. doi: 10.1016/s0091-3057(96)00315-2. [DOI] [PubMed] [Google Scholar]
- 124.Wang C, et al. GPR30 contributes to estrogen-induced thymic atrophy. Mol Endocrinol. 2008;22(3):636–648. doi: 10.1210/me.2007-0359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Isensee J, et al. Expression pattern of G protein-coupled receptor 30 in LacZ reporter mice. Endocrinology. 2009;150(4):1722–1730. doi: 10.1210/en.2008-1488. [DOI] [PubMed] [Google Scholar]
- 126.Otto C, et al. GPR30 does not mediate estrogenic responses in reproductive organs in mice. Biol Reprod. 2009;80(1):34–41. doi: 10.1095/biolreprod.108.071175. [DOI] [PubMed] [Google Scholar]
- 127.Ford J, et al. GPR30 deficiency causes increased bone mass, mineralization, and growth plate proliferative activity in male mice. J Bone Miner Res. 2011;26(2):298–307. doi: 10.1002/jbmr.209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Windahl SH, et al. The role of the G protein-coupled receptor GPR30 in the effects of estrogen in ovariectomized mice. Am J Physiol EndocrinolMetab. 2009;296(3):E490–E496. doi: 10.1152/ajpendo.90691.2008. [DOI] [PubMed] [Google Scholar]
- 129.Olde B, Leeb-Lundberg LMF. GPR30/GPER1: searching for a role in estrogen physiology. Trends Endocrinol Metab. 2009;20(8):409–416. doi: 10.1016/j.tem.2009.04.006. [DOI] [PubMed] [Google Scholar]
- 130.Langer G, et al. A critical review of fundamental controversies in the field of GPR30 research. Steroids. 2010;75(8–9):603–610. doi: 10.1016/j.steroids.2009.12.006. [DOI] [PubMed] [Google Scholar]
- 131.Ohlsson C, Vandenput L. The role of estrogens for male bone health. Eur J Endocrinol. 2009;160(6):883–889. doi: 10.1530/EJE-09-0118. [DOI] [PubMed] [Google Scholar]
- 132.Prossnitz ER, Barton M. The G-protein-coupled estrogen receptor GPER in health and disease. Nat Rev Endocrinol. 2011;7(12):715–726. doi: 10.1038/nrendo.2011.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Pedram A, et al. Functional estrogen receptors in the mitochondria of breast cancer cells. Mol Biol Cell. 2006;17(5):2125–2137. doi: 10.1091/mbc.E05-11-1013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Roepke TA. Oestrogen modulates hypothalamic control of energy homeostasis through multiple mechanisms. J Neuroendocrinol. 2009;21(2):141–150. doi: 10.1111/j.1365-2826.2008.01814.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zhang C, Kelly MJ, Rønnekleiv OK. 17βb-Estradiol rapidly increases K(ATP) activity in GnRH via a protein kinase signaling pathway. Endocrinology. 2010;151(9):4477–4484. doi: 10.1210/en.2010-0177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Kenealy BP, et al. STX, a novel nonsteroidal estrogenic compound, induces rapid action in primate GnRH neuronal calcium dynamics and peptide release. Endocrinology. 2011;152(8):3182–3191. doi: 10.1210/en.2011-0097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Kuo J, et al. Membrane estrogen receptors stimulate intracellular calcium release and progesterone synthesis in hypothalamic astrocytes. J Neurosci. 2010;30(39):12950–12957. doi: 10.1523/JNEUROSCI.1158-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Tobias SC, et al. Synthesis and biological evaluation of SERMs with potent nongenomic estrogenic activity. ChemMedChem. 2006;1(5):565–571. doi: 10.1002/cmdc.200500098. [DOI] [PubMed] [Google Scholar]
- 139.Clipperton Allen AE, et al. Agonistic behavior in males and females: effects of an estrogen receptor beta agonist in gonadectomized and gonadally intact mice. Psychoneuroendocrinology. 2010;35(7):1008–1022. doi: 10.1016/j.psyneuen.2010.01.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Clipperton-Allen AE, et al. Effects of an estrogen receptor alpha agonist on agonistic behaviour in intact and gonadectomized male and female mice. Psychoneuroendocrinology. 2011;36(7):981–995. doi: 10.1016/j.psyneuen.2010.12.010. [DOI] [PubMed] [Google Scholar]
- 141.Chang Y-J, et al. Estrogen modulates sexually dimorphic contextual fear extinction in rats through estrogen receptor β. Hippocampus. 2009;19(11):1142–1150. doi: 10.1002/hipo.20581. [DOI] [PubMed] [Google Scholar]
- 142.Chen Z, et al. Estrogen receptor α mediates the nongenomic activation of endothelial nitric oxide synthase by estrogen. J Clin Invest. 1999;103(3):401–406. doi: 10.1172/JCI5347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Kang L, et al. Involvement of estrogen receptor variant ER-alpha36, not GPR30, in nongenomic estrogen signaling. Mol Endocrinol. 2010;24(4):709–721. doi: 10.1210/me.2009-0317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Chambliss KL, et al. Non-nuclear estrogen receptor α signaling promotes cardiovascular protection but not uterine or breast cancer growth in mice. J Clin Invest. 2010;120(7):2319–2330. doi: 10.1172/JCI38291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Wang Z, et al. A variant of estrogen receptor-α, hER-α36: transduction of estrogenand antiestrogen-dependent membrane-initiated mitogenic signaling. Proc Natl Acad Sci U S A. 2006;103(24):9063–9068. doi: 10.1073/pnas.0603339103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Pelekanou V, et al. ERα36, a new variant of the ERα is expressed in triple negative breast carcinomas and has a specific transcriptomic signature in breast cancer cell lines. Steroids. 2012;77:928–934. doi: 10.1016/j.steroids.2011.12.016. [DOI] [PubMed] [Google Scholar]
- 147.Wallacides A, et al. Estrogens promote proliferation of the seminoma-like TCam-2 cell line through a GPER-dependent ERα36 induction. Mol Cell Endocrinol. 2012;350(1):61–71. doi: 10.1016/j.mce.2011.11.021. [DOI] [PubMed] [Google Scholar]
- 148.Lindsey SH, et al. Estrogen receptor GPR30 reduces oxidative stress and proteinuria in the salt-sensitive female mRen2. Lewis rat. Hypertension. 2011;58(4):665–671. doi: 10.1161/HYPERTENSIONAHA.111.175174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhang X, et al. Estrogen receptor-alpha 36 mediates mitogenic antiestrogen signaling in ER-negative breast cancer cells. PLoS One. 2012;7(1):e30174. doi: 10.1371/journal.pone.0030174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Albanito L, et al. G protein-coupled receptor 30 (GPR30) mediates gene expression changes and growth response to 17beta-estradiol and selective GPR30 ligand G-1 in ovarian cancer cells. Cancer Res. 2007;67(4):1859–1866. doi: 10.1158/0008-5472.CAN-06-2909. [DOI] [PubMed] [Google Scholar]
- 151.Gao F, et al. GPR30 activation opposes estrogen-dependent uterine growth via inhibition of stromal ERK1/2 and estrogen receptor alpha (ERα) phosphorylation signals. Endocrinology. 2011;152(4):1434–1447. doi: 10.1210/en.2010-1368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Lappano R, et al. Estriol acts as a GPR30 antagonist in estrogen receptor-negative breast cancer cells. Mol Cell Endocrinol. 2010;320(1–2):162–170. doi: 10.1016/j.mce.2010.02.006. [DOI] [PubMed] [Google Scholar]
- 153.Razandi M, et al. Proximal events in signaling by plasma membrane estrogen receptors. J Biol Chem. 2003;278(4):2701–2712. doi: 10.1074/jbc.M205692200. [DOI] [PubMed] [Google Scholar]
- 154.Vivacqua A, et al. G protein-coupled receptor 30 expression is up-regulated by EGF and TGF alpha in estrogen receptor alpha-positive cancer cells. Mol Endocrinol. 2009;23(11):1815–1826. doi: 10.1210/me.2009-0120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Huang GS, et al. Co-expression of GPR30 and ERβ and their association with disease progression in uterine carcinosarcoma. Am J Obstet Gynecol. 2010;203(3):242.e1–242.e5. doi: 10.1016/j.ajog.2010.04.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Filardo EJ, et al. Distribution of GPR30, a seven membrane-spanning estrogen receptor, in primary breast cancer and its association with clinicopathologic determinants of tumor progression. Clin Cancer Res. 2006;12(21):6359–6366. doi: 10.1158/1078-0432.CCR-06-0860. [DOI] [PubMed] [Google Scholar]
- 157.Tica AA, et al. G protein-coupled estrogen receptor 1-mediated effects in the rat myometrium. Am J Physiol Cell Physiol. 2011;301(5):C1262–C1269. doi: 10.1152/ajpcell.00501.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Navarro CE, et al. Regulation of cyclic adenosine 3’,5’-monophosphate signaling and pulsatile neurosecretion by Gi-coupled plasma membrane estrogen receptors in immortalized gonadotropin-releasing hormone neurons. Mol Endocrinol. 2003;17(9):1792–1804. doi: 10.1210/me.2003-0040. [DOI] [PubMed] [Google Scholar]
- 159.Rønnekleiv OK, Kelly MJ. Diversity of ovarian steroid signaling in the hypothalamus. Front Neuroendocrinol. 2005;26(2):65–84. doi: 10.1016/j.yfrne.2005.05.001. [DOI] [PubMed] [Google Scholar]
- 160.Bopassa JC, et al. A novel estrogen receptor GPER inhibits mitochondria permeability transition pore opening and protects the heart against ischemia-reperfusion injury. Am J Physiol Heart Circ Physiol. 2010;298(1):H16–H23. doi: 10.1152/ajpheart.00588.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Jayachandran M, et al. Loss of estrogen receptor b decreases mitochondrial energetic potential and increases thrombogenicity of platelets in aged female mice. Age (Dordr) 2010;32(1):109–121. doi: 10.1007/s11357-009-9119-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Lappano R, et al. MIBE acts as antagonist ligand of both estrogen receptor alpha and GPER in breast cancer cells. Breast Cancer Res. 2012;14(1):R12. doi: 10.1186/bcr3096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Morton GJ, et al. Central nervous system control of food intake and body weight. Nature. 2006;443(7109):289–295. doi: 10.1038/nature05026. [DOI] [PubMed] [Google Scholar]
- 164.Pillot B, et al. Role of hypothalamic melanocortin system in adaptation of food intake to food protein increase in mice. PLoS One. 2011;6(4):e19107. doi: 10.1371/journal.pone.0019107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Asarian L, Geary N. Modulation of appetite by gonadal steroid hormones. Philos T Roy Soc B. 2006;361(1471):1251–1263. doi: 10.1098/rstb.2006.1860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Laudenslager ML, et al. Energy balance in ovariectomized rats with and without estrogen replacement. Am J Physiol. 1980;238(5):R400–R405. doi: 10.1152/ajpregu.1980.238.5.R400. [DOI] [PubMed] [Google Scholar]
- 167.Ahdieh HB, Wade GN. Effects of hysterectomy on sexual receptivity, food intake, running wheel activity, and hypothalamic estrogen and progestin receptors in rats. J Comp Physiol Psychol. 1982;96(6):886–892. [PubMed] [Google Scholar]
- 168.Colvin GB, Sawyer CH. Induction of running activity by intracerebral implants of estrogen in overiectomized rats. Neuroendocrinology. 1969;4(4–5):309–320. doi: 10.1159/000121762. [DOI] [PubMed] [Google Scholar]
- 169.Polidori C, Geary N. Estradiol treatment fails to affect the feeding responses to melanocortin-3/4 receptor agonism or antagonism in ovariectomized rats. Peptides. 2002;23(9):1697–1700. doi: 10.1016/s0196-9781(02)00112-2. [DOI] [PubMed] [Google Scholar]
- 170.Rocha M,et al. Physiologic estradiol levels enhance hypothalamic expression of the long form of the leptin receptor in intact rats. J Nutr Biochem. 2004;15(6):328–334. doi: 10.1016/j.jnutbio.2004.01.003. [DOI] [PubMed] [Google Scholar]
- 171.Olofsson LE, Pierce AA, Xu AW. Functional requirement of AgRP and NPY neurons in ovarian cycle-dependent regulation of food intake. Proc Natl Acad Sci U S A. 2009;106(37):15932–15937. doi: 10.1073/pnas.0904747106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Xu Y, et al. Distinct hypothalamic neurons mediate estrogenic effects on energy homeostasis and reproduction. Cell Metab. 2011;14(4):453–465. doi: 10.1016/j.cmet.2011.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.de Souza FSJ, et al. The estrogen receptor a colocalizes with proopiomelanocortin in hypothalamic neurons and binds to a conserved motif present in the neuron-specific enhancer nPE2. Eur J Pharmacol. 2011;660(1):181–187. doi: 10.1016/j.ejphar.2010.10.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174.Miller MM, et al. Effects of age and long-term ovariectomy on the estrogen–receptor containing subpopulations of beta-endorphin-immunoreactive neurons in the arcuate nucleus of female C57BL/6J mice. Neuroendocrinology. 1995;61(5):542–551. doi: 10.1159/000126878. [DOI] [PubMed] [Google Scholar]
- 175.Malyala A, et al. PI3K signaling effects in hypothalamic neurons mediated by estrogen. J Comp Neurol. 2008;506(6):895–911. doi: 10.1002/cne.21584. [DOI] [PubMed] [Google Scholar]
- 176.Pappas T, Gametchu B, Watson C. Membrane estrogen receptors identified by multiple antibody labeling and impeded-ligand binding. FASEB J. 1995;9(5):404–410. doi: 10.1096/fasebj.9.5.7896011. [DOI] [PubMed] [Google Scholar]
- 177.Razandi M, et al. Cell membrane and nuclear estrogen receptors (ERs) originate from a single transcript: studies of ERα and ERα expressed in Chinese hamster ovary cells. Mol Endocrinol. 1999;13(2):307–319. doi: 10.1210/mend.13.2.0239. [DOI] [PubMed] [Google Scholar]
- 178.Merchenthaler I, et al. Distribution of estrogen receptor α and β in the mouse central nervous system: in vivo autoradiographic and immunocytochemical analyses. J Comp Neurol. 2004;473(2):270–291. doi: 10.1002/cne.20128. [DOI] [PubMed] [Google Scholar]
- 179.Österlund M, et al. Differential distribution and regulation of estrogen receptor-α and -β mRNA within the female rat brain. Mol Brain Res. 1998;54(1):175–180. doi: 10.1016/s0169-328x(97)00351-3. [DOI] [PubMed] [Google Scholar]
- 180.Simerly RB, et al. Distribution of androgen and estrogen receptor mRNA-containing cells in the rat brain: an in situ hybridization study. J Comp Neurol. 1990;294(1):76–95. doi: 10.1002/cne.902940107. [DOI] [PubMed] [Google Scholar]
- 181.Shima N, Yamaguchi Y, Yuri K. Distribution of estrogen receptor β mRNA-containing cells in ovariectomized and estrogen-treated female rat brain. Anat Sci Int. 2003;78(2):85–97. doi: 10.1046/j.0022-7722.2003.00042.x. [DOI] [PubMed] [Google Scholar]
- 182.Laflamme N, et al. Expression and neuropeptidergic characterization of estrogen receptors (ERα and ERβ) throughout the rat brain: anatomical evidence of distinct roles of each subtype. J Neurobiol. 1998;36(3):357–378. doi: 10.1002/(sici)1097-4695(19980905)36:3<357::aid-neu5>3.0.co;2-v. [DOI] [PubMed] [Google Scholar]
- 183.Mitra SW, et al. Immunolocalization of estrogen receptor β in the mouse brain: comparison with estrogen receptor α. Endocrinology. 2003;144(5):2055–2067. doi: 10.1210/en.2002-221069. [DOI] [PubMed] [Google Scholar]
- 184.Shughrue PJ, et al. Comparative distribution of estrogen receptor-α (ER-α) and β (ER-β) mRNA in the rat pituitary, gonad, and reproductive tract. Steroids. 1998;63(10):498–504. doi: 10.1016/s0039-128x(98)00054-3. [DOI] [PubMed] [Google Scholar]
- 185.Horvath TL. Synaptic plasticity in energy balance regulation. Obesity (Silver Spring) 2006;14(S8):228S–233S. doi: 10.1038/oby.2006.314. [DOI] [PubMed] [Google Scholar]
- 186.Thammacharoen S, et al. Divergent effects of estradiol and the estrogen receptor- [alpha] agonist PPT on eating and activation of PVN CRH neurons in ovariectomized rats and mice. Brain Res. 2009;1268:88–96. doi: 10.1016/j.brainres.2009.02.067. [DOI] [PubMed] [Google Scholar]
- 187.Musatov S, et al. Silencing of estrogen receptor α in the ventromedial nucleus of hypothalamus leads to metabolic syndrome. Proc Natl Acad Sci U S A. 2007;104(7):2501–2506. doi: 10.1073/pnas.0610787104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Kelly MJ, et al. Estrogen modulation of G-protein-coupled receptor activation of potassium channels in the central nervous system. Ann N Y Acad Sci. 2003;1007(1):6–9. doi: 10.1196/annals.1286.001. [DOI] [PubMed] [Google Scholar]
- 189.Hazell GGJ, et al. Localisation of GPR30, a novel G protein-coupled oestrogen receptor, suggests multiple functions in rodent brain and peripheral tissues. J Endocrinol. 2009;202(2):223–236. doi: 10.1677/JOE-09-0066. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Spary EJ, Maqbool A, Batten TFC. Oestrogen receptors in the central nervous system and evidence for their role in the control of cardiovascular function. J Chem Neuroanat. 2009;38(3):185–196. doi: 10.1016/j.jchemneu.2009.05.008. [DOI] [PubMed] [Google Scholar]
- 191.Sakamoto H, et al. Expression of G protein-coupled receptor-30, a G protein-coupled membrane estrogen receptor, in oxytocin neurons of the rat paraventricular and supraoptic nuclei. Endocrinology. 2007;148(12):5842–5050. doi: 10.1210/en.2007-0436. [DOI] [PubMed] [Google Scholar]
- 192.Milner TA, et al. Ultrastructural evidence that hippocampal alpha estrogen receptors are located at extranuclear sites. J Comp Neurol. 2001;429(3):355–371. [PubMed] [Google Scholar]
- 193.Marin R, et al. An oestrogen membrane receptor participates in estradiol actions for the prevention of amyloid-α peptide 1—40-induced toxicity in septal-derived cholinergic SN56 cells. J Neurochem. 2003;85(5):1180–1189. doi: 10.1046/j.1471-4159.2003.01767.x. [DOI] [PubMed] [Google Scholar]
- 194.Marin R, et al. An ICI 182,780-sensitive, membrane-related estrogen receptor contributes to estrogenic neuroprotective actions against amyloid-beta toxicity. Ann N Y Acad Sci. 2003;1007(1):108–116. doi: 10.1196/annals.1286.011. [DOI] [PubMed] [Google Scholar]
- 195.Xu Y, et al. Neurite-localized estrogen receptor-α mediates rapid signaling by estrogen. J Neurosci Res. 2003;74(1):1–11. doi: 10.1002/jnr.10725. [DOI] [PubMed] [Google Scholar]
- 196.Milner TA, et al. Ultrastructural localization of estrogen receptor β immunoreactivity in the rat hippocampal formation. J Comp Neurol. 2005;491(2):81–95. doi: 10.1002/cne.20724. [DOI] [PubMed] [Google Scholar]
- 197.Kelly MJ, Levin ER. Rapid actions of plasma membrane estrogen receptors. Trends Endocrinol Metab. 2001;12(4):152–156. doi: 10.1016/s1043-2760(01)00377-0. [DOI] [PubMed] [Google Scholar]
- 198.Hukshorn CJ, et al. The effect of pegylated recombinant human leptin (PEG-OB) on weight loss and inflammatory status in obese subjects. Int J Obes Relat Metab Disord. 2002;26(4):504–509. doi: 10.1038/sj.ijo.0801952. [DOI] [PubMed] [Google Scholar]
- 199.Bueno J, Pfaff DW. Single unit recording in hypothalamus and preoptic area of estrogentreated and untreated ovariectomized female rats. Brain Res. 1976;101(1):67–78. doi: 10.1016/0006-8993(76)90988-4. [DOI] [PubMed] [Google Scholar]
- 200.Yagi K. Changes in firing rates of single preoptic and hypothalamic units following an intravenous administration of estrogen in the castrated female rat. Brain Res. 1973;53(2):343–352. doi: 10.1016/0006-8993(73)90219-9. [DOI] [PubMed] [Google Scholar]
- 201.Kelly MJ, Moss RL, Dudley CA. The effect of ovariectomy on the responsiveness of preoptic-septal neurons to microelectrophoresed estrogen. Neuroendocrinology. 1978;25(4):204–211. doi: 10.1159/000122742. [DOI] [PubMed] [Google Scholar]
- 202.Kelly MJ, Moss RL, Dudley CA. Differential sensitivity of preoptic-septal neurons to microelectrophoresed estrogen during the estrous cycle. Brain Res. 1976;114(1):152–157. doi: 10.1016/0006-8993(76)91017-9. [DOI] [PubMed] [Google Scholar]
- 203.Kelly MJ, Moss RL, Dudley CA. The effects of microelectrophoretically applied estrogen, cortisol and acetylcholine on medial preoptic-septal unit activity throughout the estrous cycle of the female rat. Exp Brain Res. 1977;30(1):53–64. doi: 10.1007/BF00237858. [DOI] [PubMed] [Google Scholar]
- 204.Kelly MJ, et al. The specificity of the response of preoptic-septal area neurons to estrogen:17α-estradiol versus 17β-estradiol and the response of extrahypothalamic neurons. Exp Brain Res. 1977;30(1):43–52. doi: 10.1007/BF00237857. [DOI] [PubMed] [Google Scholar]
- 205.Balthazart J, Baillien M, Ball GF. Phosphorylation processes mediate rapid changes of brain aromatase activity. J Steroid Biochem Mol Biol. 2001;79(1–5):261–277. doi: 10.1016/s0960-0760(01)00143-1. [DOI] [PubMed] [Google Scholar]
- 206.Sutter-Dub M-T. Rapid non-genomic and genomic responses to progestogens, estrogens, and glucocorticoids in the endocrine pancreatic B cell, the adipocyte and other cell types. Steroids. 2002;67(2):77–93. doi: 10.1016/s0039-128x(01)00142-8. [DOI] [PubMed] [Google Scholar]
- 207.Ivanova T, et al. Rapid stimulation of the PI3-kinase/Akt signalling pathway in developing midbrain neurones by oestrogen. J Neuroendocrinol. 2002;14(1):73–79. doi: 10.1046/j.0007-1331.2001.00742.x. [DOI] [PubMed] [Google Scholar]
- 208.Singh M. Ovarian hormones elicit phosphorylation of akt and extracellular-signal regulated kinase in explants of the cerebral cortex. Endocrine. 2001;14(3):407–415. doi: 10.1385/ENDO:14:3:407. [DOI] [PubMed] [Google Scholar]
- 209.Wilson ME, Liu Y, Wise PM. Estradiol enhances Akt activation in cortical explant cultures following neuronal injury. Mol Brain Res. 2002;102(1–2):48–54. doi: 10.1016/s0169-328x(02)00181-x. [DOI] [PubMed] [Google Scholar]
- 210.Vasudevan N, Pfaff DW. Non-genomic actions of estrogens and their interaction with genomic actions in the brain. Front Neuroendocrinol. 2008;29(2):238–257. doi: 10.1016/j.yfrne.2007.08.003. [DOI] [PubMed] [Google Scholar]
- 211.Watters JJ, et al. Rapid membrane effects of steroids in neuroblastoma cells: effects of estrogen on mitogen activated protein kinase signalling cascade and c-fos immediate early gene transcription. Endocrinology. 1997;138(9):4030–4033. doi: 10.1210/endo.138.9.5489. [DOI] [PubMed] [Google Scholar]
- 212.Carrer HF, Cambiasso MJ, Gorosito S. Effects of estrogen on neuronal growth and differentiation. J Steroid Biochem Mol Biol. 2005;93(2–5):319–323. doi: 10.1016/j.jsbmb.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 213.Kuroki Y, et al. Putative membrane-bound estrogen receptors possibly stimulate mitogen-activated protein kinase in the rat hippocampus. Eur J Pharmacol. 2000;400(2–3):205–209. doi: 10.1016/s0014-2999(00)00425-8. [DOI] [PubMed] [Google Scholar]
- 214.Shingo AS, Kito S. Estradiol induces PKA activation through the putative membrane receptor in the living hippocampal neuron. J Neural Transm. 2005;112(11):1469–1473. doi: 10.1007/s00702-005-0371-8. [DOI] [PubMed] [Google Scholar]
- 215.Beyer C, Raab H. Nongenomic effects of oestrogen: embryonic mouse midbrain neurones respond with a rapid release of calcium from intracellular stores. Eur J Neurosci. 1998;10(1):255–262. doi: 10.1046/j.1460-9568.1998.00045.x. [DOI] [PubMed] [Google Scholar]
- 216.Micevych PE, et al. Estradiol stimulates progesterone synthesis in hypothalamic astrocyte cultures. Endocrinology. 2007;148(2):782–789. doi: 10.1210/en.2006-0774. [DOI] [PubMed] [Google Scholar]
- 217.Stevis PE, et al. Differential effects of estradiol and estradiol-BSA conjugates. Endocrinology. 1999;140(11):5455–5458. doi: 10.1210/endo.140.11.7247. [DOI] [PubMed] [Google Scholar]
- 218.Taguchi Y, Koslowski M, Bodenner D. Binding of estrogen receptor with estrogen conjugated to bovine serum albumin (BSA) Nuclear Receptor. 2004;2(1):5. doi: 10.1186/1478-1336-2-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Chaban VV, Micevych PE. Estrogen receptor-α mediates estradiol attenuation of ATP-induced Ca2þ signaling in mouse dorsal root ganglion neurons. J Neurosci Res. 2005;81(1):31–37. doi: 10.1002/jnr.20524. [DOI] [PubMed] [Google Scholar]
- 220.Ábrahám IM, et al. Critical in vivo roles for classical estrogen receptors in rapid estrogen actions on intracellular signaling in mouse brain. Endocrinology. 2004;145(7):3055–3061. doi: 10.1210/en.2003-1676. [DOI] [PubMed] [Google Scholar]
- 221.Singh M, et al. Estrogen-induced activation of mitogen-activated protein kinase in cerebral cortical explants: convergence of estrogen and neurotrophin signaling pathways. J Neurosci. 1999;19(4):1179–1188. doi: 10.1523/JNEUROSCI.19-04-01179.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Dominguez-Salazar E, Shetty S, Rissman EF. Rapid neural Fos responses to oestradiol in oestrogen receptor alphabeta double knockout mice. J Neuroendocrinol. 2006;18(3):195–202. doi: 10.1111/j.1365-2826.2005.01408.x. [DOI] [PubMed] [Google Scholar]
- 223.Schioth HB. G protein-coupled receptors in regulation of body weight. CNS Neurol Disord Drug Targets. 2006;5(3):241–249. doi: 10.2174/187152706777452263. [DOI] [PubMed] [Google Scholar]
- 224.Björnström L, Sjöberg M. Mechanisms of estrogen receptor signaling: convergence of genomic and nongenomic actions on target genes. Mol Endocrinol. 2005;19(4):833–842. doi: 10.1210/me.2004-0486. [DOI] [PubMed] [Google Scholar]
- 225.Levin ER. Integration of the extranuclear and nuclear actions of estrogen. Mol Endocrinol. 2005;19(8):1951–1959. doi: 10.1210/me.2004-0390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Sheldahl LC, et al. Estrogen induces rapid translocation of estrogen receptor β, but not estrogen receptor α to the neuronal plasma membrane. Neuroscience. 2008;153(3):751–761. doi: 10.1016/j.neuroscience.2008.02.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Vasudevan N, Kow LM, Pfaff D. Integration of steroid hormone initiated membrane action to genomic function in the brain. Steroids. 2005;70(5–7):388–396. doi: 10.1016/j.steroids.2005.02.007. [DOI] [PubMed] [Google Scholar]
- 228.Qiu J, et al. Serotonin 5-hydroxytryptamine2C receptor signaling in hypothalamic proopiomelanocortin neurons: role in energy homeostasis in females. Mol Pharmacol. 2007;72(4):885–896. doi: 10.1124/mol.107.038083. [DOI] [PubMed] [Google Scholar]
- 229.Kelly MJ, et al. Rapid effects of estrogen on G protein-coupled receptor activation of potassium channels in the central nervous system (CNS) J Steroid Biochem Mol Biol. 2002;83(1–5):187–193. doi: 10.1016/s0960-0760(02)00249-2. [DOI] [PubMed] [Google Scholar]
- 230.Kow LM, et al. Acute estradiol application increases inward and decreases outward whole-cell currents of neurons in rat hypothalamic ventromedial nucleus. Brain Res. 2006;1116(1):1–11. doi: 10.1016/j.brainres.2006.07.104. [DOI] [PubMed] [Google Scholar]
- 231.Cooke PS, Naaz A. Role of estrogens in adipocyte development and function. Exp Biol Med. 2004;229(11):1127–1135. doi: 10.1177/153537020422901107. [DOI] [PubMed] [Google Scholar]
- 232.Group EHBCC. Body Mass Index, Serum Sex Hormones, and Breast Cancer Risk in Postmenopausal Women. J Natl Cancer Inst. 2003;95(16):1218–1226. doi: 10.1093/jnci/djg022. [DOI] [PubMed] [Google Scholar]
- 233.Iguchi T, et al. Developmental effects: oestrogen-induced vaginal changes and organotin-induced adipogenesis. Int J Androl. 2008;31(2):263–268. doi: 10.1111/j.1365-2605.2008.00863.x. [DOI] [PubMed] [Google Scholar]
- 234.Macotela Y, et al. Sex and depot differences in adipocyte insulin sensitivity and glucose metabolism. Diabetes. 2009;58(4):803–812. doi: 10.2337/db08-1054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Dyck DJ, Heigenhauser GJF, Bruce CR. The role of adipokines as regulators of skeletal muscle fatty acid metabolism and insulin sensitivity. Acta Physiol (Oxf) 2006;186(1):5–16. doi: 10.1111/j.1748-1716.2005.01502.x. [DOI] [PubMed] [Google Scholar]
- 236.Tanaka M, et al. Effects of estrogen on serum leptin levels and leptin mRNA expression in adipose tissue in rats. Horm Res. 2001;56(3–4):98–104. doi: 10.1159/000048099. [DOI] [PubMed] [Google Scholar]
- 237.Dieudonné M-N, et al. Sex steroids and leptin regulate 11β-hydroxysteroid dehydrogenase I and P450 aromatase expressions in human preadipocytes: sex specificities. J Steroid Biochem Mol Biol. 2006;99(4–5):189–196. doi: 10.1016/j.jsbmb.2006.01.007. [DOI] [PubMed] [Google Scholar]
- 238.Thornton MJ, et al. The modulation of aromatase and estrogen receptor alpha in cultured human dermal papilla cells by dexamethasone: a novel mechanism for selective action of estrogen via estrogen receptor beta? J Invest Dermatol. 2006;126(9):2010–2018. doi: 10.1038/sj.jid.5700344. [DOI] [PubMed] [Google Scholar]
- 239.Bryzgalova G, et al. Evidence that oestrogen receptor-α plays an important role in the regulation of glucose homeostasis in mice: insulin sensitivity in the liver. Diabetologia. 2006;49(3):588–597. doi: 10.1007/s00125-005-0105-3. [DOI] [PubMed] [Google Scholar]
- 240.Crandall DL, et al. Identification of estrogen receptor β RNA in human breast and abdominal subcutaneous adipose tissue. Biochem Biophys Res Commun. 1998;248(3):523–526. doi: 10.1006/bbrc.1998.8997. [DOI] [PubMed] [Google Scholar]
- 241.Lu SF, et al. Androgen receptor in mouse brain sex: differences and similarities in autoregulation. Endocrinology. 1998;139(4):1594–1601. doi: 10.1210/endo.139.4.5863. [DOI] [PubMed] [Google Scholar]
- 242.Yu I-C. Hyperleptinemia without obesity in male mice lacking androgen receptor in adipose tissue. Endocrinology. 2008;149(5):2361–2368. doi: 10.1210/en.2007-0516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243.Bryzgalova G, et al. Mechanisms of antidiabetogenic and body weight-lowering effects of estrogen in high-fat diet-fedmice. AmJ Physiol Endocrinol Metab. 2008;295(4):E904–E912. doi: 10.1152/ajpendo.90248.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 244.Iverius PH, Brunzell JD. Relationship between lipoprotein lipase activity and plasma sex steroid level in obese women. J Clin Invest. 1988;82(3):1106–1112. doi: 10.1172/JCI113667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Homma H, et al. Estrogen suppresses transcription of lipoprotein lipase gene. Existence of a unique estrogen response element on the lipoprotein lipase promoter. J Biol Chem. 2000;275(15):11404–11411. doi: 10.1074/jbc.275.15.11404. [DOI] [PubMed] [Google Scholar]
- 246.Wohlers LM, Spangenburg EE. 17β-estradiol supplementation attenuates ovariectomy-induced increases in ATGL signaling and reduced perilipin expression in visceral adipose tissue. J Cell Biochem. 2010;110(2):420–427. doi: 10.1002/jcb.22553. [DOI] [PubMed] [Google Scholar]
- 247.Jones MEE, et al. Aromatase-deficient (ArKO) mice have a phenotype of increased adiposity. Proc Natl Acad Sci U S A. 2000;97(23):12735–12740. doi: 10.1073/pnas.97.23.12735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Couse JF, Korach KS. Estrogen receptor null mice: what have we learned and where will they lead us? Endocr Rev. 1999;20(3):358–417. doi: 10.1210/edrv.20.3.0370. [DOI] [PubMed] [Google Scholar]
- 249.Lindberg M, et al. Estrogen receptor specificity for the effects of estrogen in ovariectomized mice. J Endocrinol. 2002;174(2):167–178. doi: 10.1677/joe.0.1740167. [DOI] [PubMed] [Google Scholar]
- 250.Misso ML, et al. Cellular and molecular characterization of the adipose phenotype of the aromatase-deficient mouse. Endocrinology. 2003;144(4):1474–1480. doi: 10.1210/en.2002-221123. [DOI] [PubMed] [Google Scholar]
- 251.Jones MEE, et al. Of mice and men: the evolving phenotype of aromatase deficiency. Trends Endocrinol Metab. 2006;17(2):55–64. doi: 10.1016/j.tem.2006.01.004. [DOI] [PubMed] [Google Scholar]
- 252.Foryst-Ludwig A, et al. Metabolic actions of estrogen receptor beta (ERβ) are mediated by a negative cross-talk with PPARγ. PLoS Genet. 2008;4(6):e1000108. doi: 10.1371/journal.pgen.1000108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Luconi M, Forti G, Baldi E. Genomic and nongenomic effects of estrogens: molecular mechanisms of action and clinical implications for male reproduction. J Steroid Biochem Mol Biol. 2002;80(4–5):369–381. doi: 10.1016/s0960-0760(02)00041-9. [DOI] [PubMed] [Google Scholar]
- 254.Pedram A, et al. Integration of the non-genomic and genomic actions of estrogen Membrane-initiated signaling by steroid to transcription and cell biology. J Biol Chem. 2002;277(52):50768–50775. doi: 10.1074/jbc.M210106200. [DOI] [PubMed] [Google Scholar]
- 255.Mayes JS, Watson GH. Direct effects of sex steroid hormones on adipose tissues and obesity. Obes Rev. 2004;5(4):197–216. doi: 10.1111/j.1467-789X.2004.00152.x. [DOI] [PubMed] [Google Scholar]
- 256.Pedersen SB, et al. Demonstration of estrogen receptor subtypes α and β in human adipose tissue: influences of adipose cell differentiation and fat depot localization. Mol Cell Endocrinol. 2001;182(1):27–37. doi: 10.1016/s0303-7207(01)00557-3. [DOI] [PubMed] [Google Scholar]
- 257.Anwar A, et al. Site-specific regulation of oestrogen receptor-α and -β by oestradiol in human adipose tissue. Diabetes Obes Metab. 2001;3(5):338–349. doi: 10.1046/j.1463-1326.2001.00145.x. [DOI] [PubMed] [Google Scholar]
- 258.Price TM, O’Brien SN. Determination of estrogen receptor messenger ribonucleic acid (mRNA) and cytochrome P450 aromatasemRNAlevels in adipocytes and adipose stromal cells by competitive polymerase chain reaction amplification. J Clin Endocrinol Metab. 1993;77(4):1041–1045. doi: 10.1210/jcem.77.4.8408452. [DOI] [PubMed] [Google Scholar]
- 259.Dieudonné MN, et al. Evidence for functional estrogen receptors α and β in human adipose cells: regional specificities and regulation by estrogens. Am J Physiol Cell Physiol. 2004;286(3):C655–C661. doi: 10.1152/ajpcell.00321.2003. [DOI] [PubMed] [Google Scholar]
- 260.Mizutani T, et al. Identification of estrogen receptor in human adipose tissue and adipocytes. J Clin Endocrinol Metab. 1994;78(4):950–954. doi: 10.1210/jcem.78.4.8157726. [DOI] [PubMed] [Google Scholar]
- 261.Rebuffé-Scrive M, et al. Steroid hormone receptors in human adipose tissues. J Clin Endocrinol Metab. 1990;71(5):1215–1219. doi: 10.1210/jcem-71-5-1215. [DOI] [PubMed] [Google Scholar]
- 262.Brönnegård M, et al. Lack of evidence for estrogen and progesterone receptors in human adipose tissue. J Steroid Biochem Mol Biol. 1994;51(5–6):275–281. doi: 10.1016/0960-0760(94)90040-x. [DOI] [PubMed] [Google Scholar]
- 263.Pedersen SB, et al. Identification of oestrogen receptors and oestrogen receptor mRNA in human adipose tissue. Eur J Clin Invest. 1996;26(4):262–269. doi: 10.1046/j.1365-2362.1996.145278.x. [DOI] [PubMed] [Google Scholar]
- 264.Joyner J, Hutley L, Cameron D. Estrogen receptors in human preadipocytes. Endocrine. 2001;15(2):225–230. doi: 10.1385/ENDO:15:2:225. [DOI] [PubMed] [Google Scholar]
- 265.Lubahn DB, et al. Alteration of reproductive function but not prenatal sexual development after insertional disruption of the mouse estrogen receptor gene. Proc Natl Acad Sci U S A. 1993;90(23):11162–11166. doi: 10.1073/pnas.90.23.11162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 266.Wade GN, Gray JM. Cytoplasmic 17beta-[3H] estradiol binding in rat adipose tissues. Endocrinology. 1978;103(5):1695–1701. doi: 10.1210/endo-103-5-1695. [DOI] [PubMed] [Google Scholar]
- 267.Watson GH, et al. Biochemical and immunological characterization of oestrogen receptor in the cytosolic fraction of gluteal, omental and perirenal adipose tissues from sheep. J Endocrinol. 1993;139(1):107–115. doi: 10.1677/joe.0.1390107. [DOI] [PubMed] [Google Scholar]
- 268.Mayes JS, et al. Regional differences and up-regulation of progesterone receptors in adipose tissues from oestrogen-treated sheep. J Endocrinol. 1996;148(1):19–25. doi: 10.1677/joe.0.1480019. [DOI] [PubMed] [Google Scholar]
- 269.McCann J, et al. Subcellular distribution and glycosylation pattern of androgen receptor from sheep omental adipose tissue. J Endocrinol. 2001;169(3):587–593. doi: 10.1677/joe.0.1690587. [DOI] [PubMed] [Google Scholar]
- 270.Rodriguez-Cuenca S, et al. Depot differences in steroid receptor expression in adipose tissue: possible role of the local steroid milieu. Am J Physiol Endocrinol Metab. 2005;288(1):E200–E207. doi: 10.1152/ajpendo.00270.2004. [DOI] [PubMed] [Google Scholar]
- 271.Barros RPA, et al. Participation of ERα and ERα in glucose homeostasis in skeletal muscle and white adipose tissue. Am J Physiol Endocrinol Metab. 2009;297(1):E124–E133. doi: 10.1152/ajpendo.00189.2009. [DOI] [PubMed] [Google Scholar]
- 272.Rodriguez-Cuenca S, et al. Sex steroid receptor expression profile in brown adipose tissue Effects of hormonal status. Cell Physiol Biochem. 2007;20(6):877–886. doi: 10.1159/000110448. [DOI] [PubMed] [Google Scholar]
- 273.Gray JM, Wade GN. Cytoplasmic estrogen, but not progestin, binding sites in male rat adipose tissues. Am J Physiol. 1980;239(4):E237. doi: 10.1152/ajpendo.1980.239.4.E237. [DOI] [PubMed] [Google Scholar]
- 274.Gray JM, Dudley SD, Wade GN. In vivo cell nuclear binding of 17 beta-[3H]estradiol in rat adipose tissues. Am J Physiol. 1981;240(1):E43–E46. doi: 10.1152/ajpendo.1981.240.1.E43. [DOI] [PubMed] [Google Scholar]
- 275.Rebuffe-Scrive M. Sex steroid hormones and adipose tissue metabolism in ovariectomized and adrenalectomized rats. Acta Physiol Scand. 1987;129(4):471–477. doi: 10.1111/j.1365-201x.1987.tb10620.x. [DOI] [PubMed] [Google Scholar]
- 276.Pedersen SB, et al. Nuclear estradiol binding in rat adipocytes Regional variations and regulatory influences of hormones. Biochim Biophys Acta. 1991;1093(1):80–86. doi: 10.1016/0167-4889(91)90141-j. [DOI] [PubMed] [Google Scholar]
- 277.Echeverría OM, et al. Immuno-electron microscopic localization of estradiol receptor in cells of male and female reproductive and non-reproductive organs. Biol Cell. 1994;81(3):257–265. doi: 10.1016/0248-4900(94)90008-6. [DOI] [PubMed] [Google Scholar]
- 278.Cooke PS, et al. The role of estrogen and estrogen receptor-α in male adipose tissue. Mol Cell Endocrinol. 2001;178(1–2):147–154. doi: 10.1016/s0303-7207(01)00414-2. [DOI] [PubMed] [Google Scholar]
- 279.Casazza K, Page GP, Fernandez JR. The association between the rs2234693 and rs9340799 estrogen receptor alpha gene polymorphisms and risk factors for cardiovascular disease: a review. Biol Res Nurs. 2010;12(1):84–97. doi: 10.1177/1099800410371118. [DOI] [PubMed] [Google Scholar]
- 280.Riant E, et al. Estrogens protect against high-fat diet-induced insulin resistance and glucose intolerance in mice. Endocrinology. 2009;150(5):2109–2117. doi: 10.1210/en.2008-0971. [DOI] [PubMed] [Google Scholar]
- 281.Naaz A, et al. Effect of ovariectomy on adipose tissue of mice in the absence of estrogen receptor alpha (ERα): a potential role for estrogen receptor beta (ERα) Horm Metab Res. 2002;34(11/12) doi: 10.1055/s-2002-38259. 758,763. [DOI] [PubMed] [Google Scholar]
- 282.Palin SL, et al. 17Beta-estradiol and anti-estrogen ICI:Compound 182,780 regulate expression of lipoprotein lipase and hormone-sensitive lipase in isolated subcutaneous abdominal adipocytes. Metabolism. 2003;52(4):383–388. doi: 10.1053/meta.2003.50088. [DOI] [PubMed] [Google Scholar]
- 283.Pedersen SB, et al. Effects of in vivo estrogen treatment on adipose tissue metabolism and nuclear estrogen receptor binding in isolated rat adipocytes. Mol Cell Endocrinol. 1992;85(1–2):13–19. doi: 10.1016/0303-7207(92)90120-u. [DOI] [PubMed] [Google Scholar]
- 284.Hamosh M, Hamosh P. The effect of estrogen on the lipoprotein lipase activity of rat adipose tissue. J Clin Invest. 1975;55(5):1132–1135. doi: 10.1172/JCI108015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Kim HJ, Kalkhoff RK. Sex steroid influence on triglyceride metabolism. J Clin Invest. 1975;56(4):888–896. doi: 10.1172/JCI108168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Wilson DE, et al. Estrogen treatment and gonadal function in the regulation of lipoprotein lipase. Atherosclerosis. 1976;24(3):491–499. doi: 10.1016/0021-9150(76)90141-6. [DOI] [PubMed] [Google Scholar]
- 287.Steingrimsdottir L, Brasel J, Greenwood MR. Hormonal modulation of adipose tissue lipoprotein lipase may alter food intake in rats. Am J Physiol. 1980;239(2):E162–E167. doi: 10.1152/ajpendo.1980.239.2.E162. [DOI] [PubMed] [Google Scholar]
- 288.Ramirez I. Estradiol-induced changes in lipoprotein lipase, eating, and body weight in rats. Am J Physiol. 1981;240(2):E533–E538. doi: 10.1152/ajpendo.1981.240.5.E533. [DOI] [PubMed] [Google Scholar]
- 289.Ellis GS, et al. Effects of estradiol on lipoprotein lipase activity and lipid availability in exercised male rats. J Appl Physiol. 1994;77(1):209–215. doi: 10.1152/jappl.1994.77.1.209. [DOI] [PubMed] [Google Scholar]
- 290.Hazzard WR, et al. Studies on the mechanism of increased plasma triglyceride levels induced by oral contraceptives. N Engl J Med. 1969;280(9):471–474. doi: 10.1056/NEJM196902272800904. [DOI] [PubMed] [Google Scholar]
- 291.Fabian E, et al. Lipoprotein lipase and postheparin esterase activity and postheparin increase in free fatty acids after the short-term administration of a sequential contraceptive. Am J Obstet Gynecol. 1971;109(8):1212–1213. doi: 10.1016/0002-9378(71)90670-3. [DOI] [PubMed] [Google Scholar]
- 292.Ence TJ, et al. Heparin metabolism and heparin-released lipase activity during longterm estrogen-progestin treatment. Metabolism. 1976;25(2):139–145. doi: 10.1016/0026-0495(76)90044-5. [DOI] [PubMed] [Google Scholar]
- 293.Price TM, et al. Estrogen regulation of adipose tissue lipoprotein lipase— possible mechanism of body fat distribution. Am J Obstet Gynecol. 1998;178(1, part 1):101–107. doi: 10.1016/s0002-9378(98)70634-9. [DOI] [PubMed] [Google Scholar]
- 294.Urabe M, et al. Effect of Estrogen Replacement Therapy on Hepatic Triglyceride Lipase, Lipoprotein Liase and lipids Including Apolipoprotein E in Climacteric and Elderly Women. Endocr J. 1996;43(6):737–742. doi: 10.1507/endocrj.43.737. [DOI] [PubMed] [Google Scholar]
- 295.Benz V, et al. Sexual dimorphic regulation of body weight dynamics and adipose tissue lipolysis. PLoS ONE. 2012;7(5):e37794. doi: 10.1371/journal.pone.0037794. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 296.Pedersen SB, et al. Estrogen controls lipolysis by up-regulating α2A-adrenergic receptors directly in human adipose tissue through the estrogen receptor α Implications for the female fat distribution. J Clin Endocrinol Metab. 2004;89(4):1869–1878. doi: 10.1210/jc.2003-031327. [DOI] [PubMed] [Google Scholar]
- 297.Simoncini T, Genazzani A. Non-genomic actions of sex steroid hormones. Eur J Endocrinol. 2003;148(3):281–292. doi: 10.1530/eje.0.1480281. [DOI] [PubMed] [Google Scholar]
- 298.Dos Santos EG, et al. Rapid nongenomic E2 effects on p42/p44 MAPK, activator protein-1, and cAMP response element binding protein in rat white adipocytes. Endocrinology. 2002;143(3):930–940. doi: 10.1210/endo.143.3.8678. [DOI] [PubMed] [Google Scholar]
- 299.Falkenstein E, et al. Multiple actions of steroid hormones—a focus on rapid, nongenomic effects. Pharmacol Rev. 2000;52(4):513–556. [PubMed] [Google Scholar]
- 300.Schmidt BMW, et al. Rapid, nongenomic steroid actions: a new age? Front Neuroendocrinol. 2000;21(1):57–94. doi: 10.1006/frne.1999.0189. [DOI] [PubMed] [Google Scholar]
- 301.Segars JH, Driggers PH. Estrogen action and cytoplasmic signaling cascades Part I: membrane-associated signaling complexes. Trends Endocrinol Metab. 2002;13(8):349–354. doi: 10.1016/s1043-2760(02)00633-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 302.Driggers PH, Segars JH. Estrogen action and cytoplasmic signaling pathways Part II: the role of growth factors and phosphorylation in estrogen signaling. Trends Endocrinol Metab. 2002;13(10):422–427. doi: 10.1016/s1043-2760(02)00634-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 303.Schlegel A, et al. Caveolin-1 Potentiates Estrogen Receptor α (ERα) Signaling. J Biol Chem. 1999;274(47):33551–33556. doi: 10.1074/jbc.274.47.33551. [DOI] [PubMed] [Google Scholar]
- 304.Schlegel A, Arvan P, Lisanti MP. Caveolin-1 binding to endoplasmic reticulum membranes and entry into the regulated secretory pathway are regulated by serine phosphorylation. Protein sorting at the level of the endoplasmic reticulum. J Biol Chem. 2001;276(6):4398–4408. doi: 10.1074/jbc.M005448200. [DOI] [PubMed] [Google Scholar]
- 305.Mitznegg P, Estler CJ, Schubert E. Effect of estradiol on lipolysis and adenosine 3’,5’- monophosphate accumulation in isolated rat adipocytes. Biochem Pharmacol. 1974;23(16):2337–2339. doi: 10.1016/0006-2952(74)90566-8. [DOI] [PubMed] [Google Scholar]
- 306.Pasquier Y-N, Pecquery R, Giudicelli Y. Increased adenylate cyclase catalytic activity explains how estrogens “in vivo” promote lipolytic activity in rat white fat cells. Biochem Biophys Res Commun. 1988;154(3):1151–1159. doi: 10.1016/0006-291x(88)90261-6. [DOI] [PubMed] [Google Scholar]
- 307.Lacasa D, et al. Influence of ovariectomy and regional fat distribution on the membranous transducing system controlling lipolysis in rat fat cells. Endocrinology. 1991;128(2):747–753. doi: 10.1210/endo-128-2-747. [DOI] [PubMed] [Google Scholar]
- 308.Lindberg U-B, et al. Regional adipose tissue metabolism in postmenopausal women after treatment with exogenous sex steroids. Horm Metab Res. 1990;22(06) doi: 10.1055/s-2007-1004917. 345,351. [DOI] [PubMed] [Google Scholar]
- 309.Lacasa D, et al. Influence of ovarian status and regional fat distribution on protein kinase C in rat fat cells. J Endocrinol. 1994;140(2):275–282. doi: 10.1677/joe.0.1400275. [DOI] [PubMed] [Google Scholar]
- 310.Lacasa D, et al. Control of rat preadipocyte adipose conversion by ovarian status: regional specificity and possible involvement of the mitogen-activated protein kinase-dependent and c-fos signaling pathways. Endocrinology. 1997;138(7):2729–2734. doi: 10.1210/endo.138.7.5246. [DOI] [PubMed] [Google Scholar]
- 311.Friedman JM, Halaas JL. Leptin and the regulation of body weight in mammals. Nature. 1998;395(6704):763–770. doi: 10.1038/27376. [DOI] [PubMed] [Google Scholar]
- 312.Chen S-J, et al. Estrogen reduces myointimal proliferation after balloon injury of rat carotid artery. Circulation. 1996;93(3):577–584. doi: 10.1161/01.cir.93.3.577. [DOI] [PubMed] [Google Scholar]
- 313.Diano S, et al. Leptin receptors in estrogen receptor-containing neurons of the female rat hypothalamus. Brain Res. 1998;812(1–2):256–259. doi: 10.1016/s0006-8993(98)00936-6. [DOI] [PubMed] [Google Scholar]
- 314.Ahima RS, et al. Regulation of neuronal and glial proteins by leptin: implications for brain development. Endocrinology. 1999;140(6):2755–2762. doi: 10.1210/endo.140.6.6774. [DOI] [PubMed] [Google Scholar]
- 315.Elias CF, et al. Leptin differentially regulates NPY and POMC neurons projecting to the lateral hypothalamic area. Neuron. 1999;23(4):775–786. doi: 10.1016/s0896-6273(01)80035-0. [DOI] [PubMed] [Google Scholar]
- 316.Elmquist JK, Elias CF, Saper CB. From lesions to leptin: hypothalamic control of food intake and body weight. Neuron. 1999;22(2):221–232. doi: 10.1016/s0896-6273(00)81084-3. [DOI] [PubMed] [Google Scholar]
- 317.Chakraborty S, et al. Stereological analysis of estrogen receptor expression in the hypothalamic arcuate nucleus of ob/ob and agouti mice. Brain Res. 2008;1217:86–95. doi: 10.1016/j.brainres.2008.04.031. [DOI] [PubMed] [Google Scholar]
- 318.Ainslie DA, et al. Estrogen deficiency causes central leptin insensitivity and increased hypothalamic neuropeptide Y. Int J Obes Relat Metab Disord. 2001;25(11):1680–1688. doi: 10.1038/sj.ijo.0801806. [DOI] [PubMed] [Google Scholar]
- 319.Lindell K, et al. Leptin receptor 5′ untranslated regions in the rat: relative abundance, genomic organization and relation to putative response elements. Mol Cell Endocrinol. 2001;172(1–2):37–45. doi: 10.1016/s0303-7207(00)00382-8. [DOI] [PubMed] [Google Scholar]
- 320.Shimizu H, et al. Estrogen increases in vivo leptin production in rats and human subjects. J Endocrinol. 1997;154(2):285–292. doi: 10.1677/joe.0.1540285. [DOI] [PubMed] [Google Scholar]
- 321.Machinal F, et al. In vivo and in vitro ob gene expression and leptin secretion in rat adipocytes: evidence for a regional specific regulation by sex steroid hormones. Endocrinology. 1999;140(4):1567–1574. doi: 10.1210/endo.140.4.6617. [DOI] [PubMed] [Google Scholar]
- 322.Hardie L, et al. Circulating leptin in women: a longitudinal study in the menstrual cycle and during pregnancy. Clin Endocrinol. 1998;53(2):87–89. doi: 10.1046/j.1365-2265.1997.2441017.x. [DOI] [PubMed] [Google Scholar]
- 323.Rosenbaum M, et al. Effects of gender, body composition, and menopause on plasma concentrations of leptin. J Clin Endocrinol Metab. 1996;81(9):3424–3427. doi: 10.1210/jcem.81.9.8784109. [DOI] [PubMed] [Google Scholar]
- 324.Konukoglu D, Serin Ö, Ercan M. Plasma leptin levels in obese and non-obese postmenopausal women before and after hormone replacement therapy. Maturitas. 2000;36(3):203–207. doi: 10.1016/s0378-5122(00)00153-5. [DOI] [PubMed] [Google Scholar]
- 325.Elbers JMH, et al. Effects of administration of 17β-oestradiol on serum leptin levels in healthy postmenopausal women. Clin Endocrinol. 1999;51(4):449–454. doi: 10.1046/j.1365-2265.1999.00813.x. [DOI] [PubMed] [Google Scholar]
- 326.Thomas T, et al. Relationship of serum leptin levels with body composition and sex steroid and insulin levels in men and women. Metabolism. 2000;49(10):1278–1284. doi: 10.1053/meta.2000.9519. [DOI] [PubMed] [Google Scholar]
- 327.Elbers JMH, et al. Effects of sex steroid hormones on regional fat depots as assessed by magnetic resonance imaging in transsexuals. Am J Physiol Endocrinol Metab. 1999;276(2):E317–E325. doi: 10.1152/ajpendo.1999.276.2.E317. [DOI] [PubMed] [Google Scholar]
- 328.Casabiell X, et al. Gender differences in both spontaneous and stimulated leptin secretion by human omental adipose tissue in vitro: dexamethasone and estradiol stimulate leptin release in women, but not in men. J Clin Endocrinol Metab. 1998;83(6):2149–2155. doi: 10.1210/jcem.83.6.4849. [DOI] [PubMed] [Google Scholar]
- 329.Santner SJ, et al. Aromatase activity and expression in breast cancer and benign breast tissue stromal cells. J Clin Endocrinol Metab. 1997;82(1):200–208. doi: 10.1210/jcem.82.1.3672. [DOI] [PubMed] [Google Scholar]
- 330.Geisler J, et al. Total body aromatization in postmenopausal breast cancer patients is strongly correlated to plasma leptin levels. J Steroid Biochem Mol Biol. 2007;104(1–2):27–34. doi: 10.1016/j.jsbmb.2006.09.040. [DOI] [PubMed] [Google Scholar]
- 331.Shin J-H, et al. The ratio of estrogen receptor α to estrogen receptor β in adipose tissue is associated with leptin production and obesity. Steroids. 2007;72(6–7):592–599. doi: 10.1016/j.steroids.2007.03.013. [DOI] [PubMed] [Google Scholar]
- 332.Catalano S, et al. Leptin induces, via ERK1/ERK2 signal, functional activation of estrogen receptor α in MCF-7 cells. J Biol Chem. 2004;279(19):19908–19915. doi: 10.1074/jbc.M313191200. [DOI] [PubMed] [Google Scholar]
- 333.Vecchione C, et al. Leptin effect on endothelial nitric oxide is mediated through Akt–endothelial nitric oxide synthase phosphorylation pathway. Diabetes. 2002;51(1):168–173. doi: 10.2337/diabetes.51.1.168. [DOI] [PubMed] [Google Scholar]
- 334.Pinilla L, et al. Regulation of serum leptin levels by gonadal function in rats. Eur J Endocrinol. 1999;140(5):468–473. doi: 10.1530/eje.0.1400468. [DOI] [PubMed] [Google Scholar]
- 335.Watanobe H, Suda T. A detailed study on the role of sex steroid milieu in determining plasma leptin concentrations in adult male and female rats. Biochem Biophys Res Commun. 1999;259(1):56–59. doi: 10.1006/bbrc.1999.0718. [DOI] [PubMed] [Google Scholar]
- 336.Wu-Peng S, et al. Effects of exogenous gonadal steroids on leptin homeostasis in rats. Obes Res. 1999;7(6):586–592. doi: 10.1002/j.1550-8528.1999.tb00718.x. [DOI] [PubMed] [Google Scholar]
- 337.Brann DW, et al. Regulation of leptin gene expression and secretion by steroid hormones. Steroids. 1999;64(9):659–663. doi: 10.1016/s0039-128x(99)00049-5. [DOI] [PubMed] [Google Scholar]
- 338.Luukkaa V, et al. Effects of estrous cycle and steroid replacement on the expression of leptin and uncoupling proteins in adipose tissue in the rat. Gynecol Endocrinol. 2001;15(2):103–112. [PubMed] [Google Scholar]
- 339.Yoneda N, et al. The influence of ovariectomy on ob gene expression in rats. Horm Metab Res. 1998;30(05):263–265. doi: 10.1055/s-2007-978880. [DOI] [PubMed] [Google Scholar]
- 340.Chu S-C, et al. Fluctuation of serum leptin level in rats after ovariectomy and the influence of estrogen supplement. Life Sci. 1999;64(24):2299–2306. doi: 10.1016/s0024-3205(99)00181-2. [DOI] [PubMed] [Google Scholar]
- 341.Nedvídková J, Haluzík M, Schreiber V. The decrease of serum leptin levels in oestrogen-treated male mice. Physiol Res. 1997;46(4):291–294. [PubMed] [Google Scholar]
- 342.Pelleymounter MA, Baker MB, McCaleb M. Does estradiol mediate leptin’s effects on adiposity and body weight? Am J Physiol Endocrinol Metab. 1999;276(5):E955–E963. doi: 10.1152/ajpendo.1999.276.5.E955. [DOI] [PubMed] [Google Scholar]
- 343.Kronfeld-Schor N, et al. Steroid-dependent up-regulation of adipose leptin secretion in vitro during pregnancy in mice. Biol Reprod. 2000;63(1):274–280. doi: 10.1095/biolreprod63.1.274. [DOI] [PubMed] [Google Scholar]
- 344.Halpern A, Mancini MC. Treatment of obesity: an update on anti-obesity medications. Obes Rev. 2003;4(1):25–42. doi: 10.1046/j.1467-789x.2003.00083.x. [DOI] [PubMed] [Google Scholar]
- 345.Swinburn B, Egger G. Preventive strategies against weight gain and obesity. Obes Rev. 2002;3(4):289–301. doi: 10.1046/j.1467-789x.2002.00082.x. [DOI] [PubMed] [Google Scholar]
- 346.Tchernof A, et al. Menopause, central body fatness, and insulin resistance: effects of hormone-replacement therapy. Coron Artery Dis. 1998;9(8):503–512. doi: 10.1097/00019501-199809080-00006. [DOI] [PubMed] [Google Scholar]
- 347.Gambacciani M, et al. Body weight, body fat distribution, and hormonal replacement therapy in early postmenopausal women. J Clin Endocrinol Metab. 1997;82(2):414–417. doi: 10.1210/jcem.82.2.3735. [DOI] [PubMed] [Google Scholar]
- 348.Haarbo J, et al. Postmenopausal hormone replacement therapy prevents central distribution of body fat after menopause. Metabolism. 1991;40(12):1323–1326. doi: 10.1016/0026-0495(91)90037-w. [DOI] [PubMed] [Google Scholar]
- 349.Espeland MA, et al. Effect of postmenopausal hormone therapy on body weight and waist and hip girths. Postmenopausal Estrogen-Progestin Interventions Study Investigators. J Clin Endocrinol Metab. 1997;82(5):1549–1556. doi: 10.1210/jcem.82.5.3925. [DOI] [PubMed] [Google Scholar]
- 350.Samaras K, et al. Tobacco smoking and oestrogen replacement are associated with lower total and central fat in monozygotic twins. Int J Obes. 1998;22(2):149–156. doi: 10.1038/sj.ijo.0800558. [DOI] [PubMed] [Google Scholar]
- 351.Hänggi W, et al. Differential impact of conventional oral or transdermal hormone replacement therapy or tibolone on body composition in postmenopausal women. Clin Endocrinol. 1998;48(6):691–699. doi: 10.1046/j.1365-2265.1998.00481.x. [DOI] [PubMed] [Google Scholar]
- 352.Gambacciani M, et al. Prospective evaluation of body weight and body fat distribution in early postmenopausal women with and without hormonal replacement therapy. Maturitas. 2001;39(2):125–132. doi: 10.1016/s0378-5122(01)00194-3. [DOI] [PubMed] [Google Scholar]
- 353.Sites CK, et al. Relationship between hormone replacement therapy use with body fat distribution and insulin sensitivity in obese postmenopausal women. Metabolism. 2001;50(7):835–840. doi: 10.1053/meta.2001.24878. [DOI] [PubMed] [Google Scholar]
- 354.Chmouliovsky L, et al. Beneficial effect of hormone replacement therapy on weight loss in obese menopausal women. Maturitas. 1999;32(3):147–153. doi: 10.1016/s0378-5122(99)00037-7. [DOI] [PubMed] [Google Scholar]
- 355.Kritz-Silverstein D, Barrett-Connor E. Long-term postmenopausal hormone use, obesity, and fat distribution in older women. JAMA. 1996;275(1):46–49. [PubMed] [Google Scholar]
- 356.Kanaley JA, et al. Abdominal fat distribution in pre- and postmenopausal women: the impact of physical activity, age, menopausal status. Metabolism. 2001;50(8):976–982. doi: 10.1053/meta.2001.24931. [DOI] [PubMed] [Google Scholar]
- 357.Nelson HD, et al. Postmenopausal hormone replacement therapy: scientific review. JAMA. 2002;288(7):872–881. doi: 10.1001/jama.288.7.872. [DOI] [PubMed] [Google Scholar]
- 358.Smith CL, O’Malley BW. Coregulator function: a key to understanding tissue specificity of selective receptor modulators. Endocr Rev. 2004;25(1):45–71. doi: 10.1210/er.2003-0023. [DOI] [PubMed] [Google Scholar]
- 359.Kousteni S, et al. Reversal of bone loss in mice by nongenotropic signaling of sex steroids. Science. 2002;298(5594):843–846. doi: 10.1126/science.1074935. [DOI] [PubMed] [Google Scholar]
- 360.Ke HZ, et al. Effects of CP-336,156, a new, nonsteroidal estrogen agonist/antagonist, on bone, serum cholesterol, uterus and body composition in rat models. Endocrinology. 1998;139(4):2068–2076. doi: 10.1210/endo.139.4.5902. [DOI] [PubMed] [Google Scholar]
- 361.McCarty MF. Modulation of adipocyte lipoprotein lipase expression as a strategy for preventing or treating visceral obesity. Medical Hypotheses. 2001;57(2):192–200. doi: 10.1054/mehy.2001.1317. [DOI] [PubMed] [Google Scholar]
- 362.Mantzoros CS, Flier JS. Leptin as a therapeutic agent—trials and tribulations. J Clin Endocrinol Metab. 2000;85(11):4000–4002. doi: 10.1210/jcem.85.11.7062. [DOI] [PubMed] [Google Scholar]
- 363.Lee DW, et al. Leptin and the treatment of obesity: its current status. Eur J Pharmacol. 2002;440(2–3):129–139. doi: 10.1016/s0014-2999(02)01424-3. [DOI] [PubMed] [Google Scholar]
- 364.Farooqi IS, et al. Effects of recombinant leptin therapy in a child with congenital leptin deficiency. N Engl J Med. 1999;341(12):879–884. doi: 10.1056/NEJM199909163411204. [DOI] [PubMed] [Google Scholar]