Abstract
Methylene blue and a series of recently developed 1,4-naphthoquinones, including 3-[4-(substituted)benzyl]-menadiones, are potent antimalarial agents in vitro and in vivo. The activity of these structurally diverse compounds against the human malaria parasite Plasmodium falciparum might involve their peculiar redox properties. According to the current theory, redox-active methylene blue and 3-[4-(trifluoromethyl)benzyl]-menadione are “subversive substrates.” These agents are thought to shuttle electrons from reduced flavoproteins to acceptors such as hemoglobin-associated or free Fe(III)-protoporphyrin IX. The reduction of Fe(III)-protoporphyrin IX could subsequently prevent essential hemoglobin digestion and heme detoxification in the parasite. Alternatively, owing to their structures and redox properties, methylene blue and 1,4-naphthoquinones might also affect the mitochondrial electron transport chain. Here, we tested the latter hypothesis using an established system of transgenic P. falciparum cell lines and the antimalarial agents atovaquone and chloroquine as controls. In contrast to atovaquone, methylene blue and 3-[4-(trifluoromethyl)benzyl]-menadione do not inhibit the mitochondrial electron transport chain. A systematic comparison of the morphologies of drug-treated parasites furthermore suggests that the three drugs do not share a mechanism of action. Our findings support the idea that methylene blue and 3-[4-(trifluoromethyl)benzyl]-menadione exert their antimalarial activity as redox-active subversive substrates.
INTRODUCTION
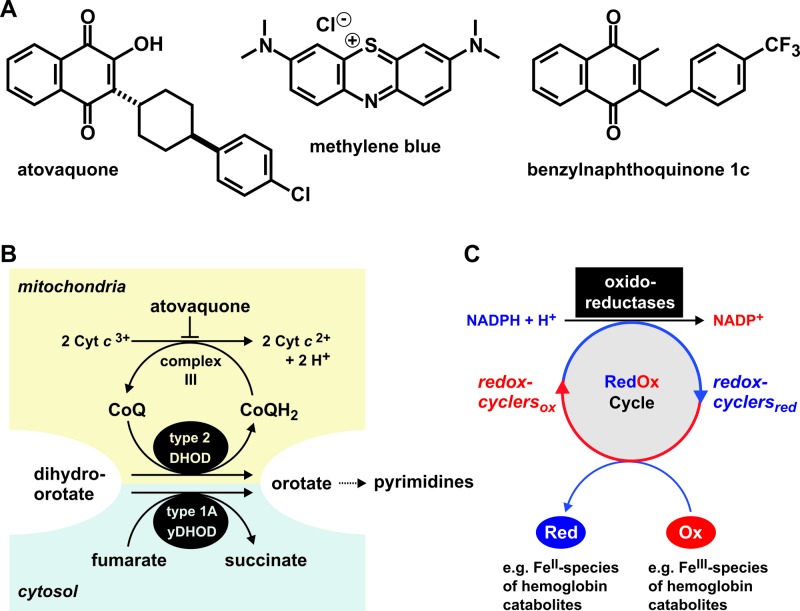
Two established drug targets of malaria blood-stage parasites are the mitochondrial electron transport chain (mETC) and the biocrystallization of hemoglobin-derived Fe(III)-protoporphyrin IX (heme) in the parasite's digestive vacuole. The commercial drug atovaquone, as well as other nonapproved agents and compounds under development block complex III of the mETC (1–5), whereas chloroquine and related 4-aminoquinoline derivatives inhibit the conversion of Fe(III)-protoporphyrin IX to an insoluble biocrystalline pigment called hemozoin (6–8). Atovaquone is a 3-hydroxy-1,4-naphthoquinone substituted at C-2 by a trans-4-(4-chlorophenyl)cyclohexane chain (Fig. 1A). It binds to the ubiquinol oxidation site of cytochrome b which is a central component of complex III (3, 9, 10). As a result, the ubiquinone pool becomes depleted, the mitochondrial membrane potential decreases, and parasite growth is arrested (1–4, 11). Even though oxidative phosphorylation seems to be significantly reduced or absent in parasite blood-stage cultures (4), the unusual ATP synthase of the most important human malaria parasite Plasmodium falciparum is likely to be essential for its survival (12). Furthermore, the mETC helps to maintain a proton gradient: an evolutionarily conserved prerequisite for protein and metabolite transport across the inner mitochondrial membrane (13). However, the major function of the mETC in asexual blood-stage cultures of P. falciparum is to regenerate ubiquinone as the electron acceptor of the mitochondrial dihydroorotate dehydrogenase (DHODH), which catalyzes a key step in pyrimidine biosynthesis (14) (Fig. 1B).
Fig 1.
Structures and (potential) mechanisms of the antimalarial agents analyzed in the present study. (A) Comparison of the structures of the 1,4-naphthoquinone tautomer of atovaquone, methylene blue, and benzylnaphthoquinone 1c from reference 22. (B) Scheme of the targeted mitochondrial electron transport in atovaquone-sensitive P. falciparum parasites. Expression of a gene encoding yDHOD provides a cytosolic bypass in transgenic P. falciparum strains (14, 18). Such strains become independent of the mitochondrial electron transport chain and are therefore not only resistant to atovaquone but also to other antimalarials that target the mETC. (C) Model of the NADPH-dependent bioactivation and mode of action of methylene blue and benzylnaphthoquinone 1c (22, 23). See the text for details.
P. falciparum DHODH has a predicted N-terminal matrix-targeting signal, followed by a single transmembrane helix in accordance with a stop-transfer pathway (15), and probably localizes to the outer side of the inner mitochondrial membrane (16). Since pyrimidine biosynthesis is essential, a functional DHODH and mETC are necessary for parasite survival (4, 14, 17). Accordingly, atovaquone was demonstrated to lose its antimalarial activity in the presence of an episomal (5, 14) or chromosomal (18) copy of a yeast gene encoding an alternative cytosolic DHODH (yDHODH) that does not require the mETC as an electron acceptor (Fig. 1B). Transgenic P. falciparum strains with or without yDHODH therefore provide an excellent tool for the study of potential inhibitors of the mETC. Such candidates include redox-active 1,4-naphthoquinones and methylene blue.
The dye methylene blue (Fig. 1A) and 3-[4-(trifluoromethyl)benzyl]-menadione were shown to be efficient antimalarial agents in cell culture and, with limitations, in vivo (19–23). The agent 3-[4-(trifluoromethyl)benzyl]-menadione—henceforth referred to as benzylnaphthoquinone 1c (Fig. 1A)—is the lead compound of a series of novel redox-active 1,4-naphthoquinones. Their peculiar redox properties support the theory that these agents and methylene blue act as catalysts, also defined as “turncoat inhibitors” or “subversive substrates” (22–24), for example, by shuttling electrons in a continuous NADPH-dependent redox cycle between the cytosol and the digestive vacuole of the malarial parasite (Fig. 1C). In vitro, methylene blue and certain 1,4-naphthoquinones accept electrons from the flavoprotein glutathione reductase (GR), which is itself reduced by NADPH (22, 25). After reduction, these compounds can transfer single electrons to key oxidants of P. falciparum, e.g., to the hemoglobin catabolites methemoglobin and Fe(III)-protoporphyrin IX (22, 24, 26). The generated Fe(II) species are toxic to the parasite since Fe(II)-containing oxyhemoglobin is a poor substrate of the hemoglobinase falcipain-2 (27), and Fe(II)-protoporphyrin IX was shown to inhibit the crystallization of β-hematin in vitro (28). As a result of drug action, NADPH is consumed, and damaging Fe(II) species are constantly generated. This could not only contribute to the production of harmful reactive oxygen species owing to Fenton chemistry but also prevent both the digestion of methemoglobin and the formation of hemozoin (22, 23). However, owing to their redox properties and structural similarities with mETC inhibitors, methylene blue and the lead 1,4-naphthoquinones might also alter or inhibit the mETC. For example, the structure of methylene blue shares similarities with rhodamine-123 and related cationic mitochondrial dyes (29), and the 1,4-naphthoquinone vitamin K2 was recently identified in a genetic screen as an alternative mitochondrial electron carrier in Drosophila (30). In addition, structurally related quinolones were shown to be efficient inhibitors of the mETC in P. falciparum (5, 31). We therefore investigated the involvement of the P. falciparum mETC in the antimalarial activities of methylene blue and of the recently characterized benzylnaphthoquinone 1c (22).
MATERIALS AND METHODS
Inhibitors.
Atovaquone was kindly provided by Vanessa Yardley. Chloroquine and methylene blue trihydrate were from Sigma. The benzylnaphthoquinone 1c was prepared as previously described (22). Stock solutions of methylene blue and chloroquine were prepared in pure water. Stock solutions of atovaquone and benzylnaphthoquinone 1c were prepared in dimethyl sulfoxide. All stock solutions (6 mM) were stored in aliquots at −20°C.
Growth inhibition assays.
P. falciparum wild-type strain 3D7 was cultured at 37°C according to standard protocols (32) in RPMI medium containing 9% human serum and type A erythrocytes at a hematocrit of 3.3% under a low-oxygen atmosphere (3% CO2, 5% O2, 92% N2, and 95% humidity). For strains 3D7attB and 3D7attB-yDHODH (18), human serum was replaced with 0.45% (wt/vol) Albumax II. Cultures were synchronized using the sorbitol method (33). Growth inhibition was determined in a SYBR green assay as previously described (34). Inhibitors were added to synchronized ring stage parasite cultures in microtiter plates (0.5% parasitemia, 1.25% hematocrit) and incubated for 72 h. Final inhibitor concentrations in each assay ranged from 22 pM to 5 μM.
Light microscopy.
Pictures of Giemsa-stained blood smears of treated and untreated parasite cultures of strain 3D7attB-yDHODH were taken at different time points with an AxioCam ICc 3 at an Axioplan microscope using the software ZEN 2011 blue edition (Zeiss). Individual images were imported into Fiji/ImageJ (35) (version 1.47c [http://fiji.sc/Downloads]), cropped, and stored as a TIF file. The TIF file was imported into CorelDraw, labels were added, and the figure was saved without any gamma, contrast, or color adjustments.
RESULTS AND DISCUSSION
In order to test the importance of the parasite mETC to the antimalarial activities of methylene blue and benzylnaphthoquinone 1c, we determined the 50% inhibitory concentrations (IC50s) for these drugs in the parasite strains 3D7attB-yDHODH, 3D7attB, and 3D7. The first two strains contain a chromosomal attB site for the site-specific integration of a segment of DNA, which disrupts the gene encoding P. falciparum glutaredoxin-like protein 3 (PfGlp3) (36, 37). The first strain, 3D7attB-yDHODH, contains the gene encoding yDHODH (18), which confers resistance to atovaquone, as well as other mETC inhibitors, whereas the second strain, 3D7attB, has no insert at the attB site. The wild-type strain 3D7 was included as a control to assess the possibility that the agents affect a PfGlp3-dependent redox pathway. Furthermore, we determined the IC50s of chloroquine and atovaquone as negative and positive controls, respectively (Fig. 2).
Fig 2.
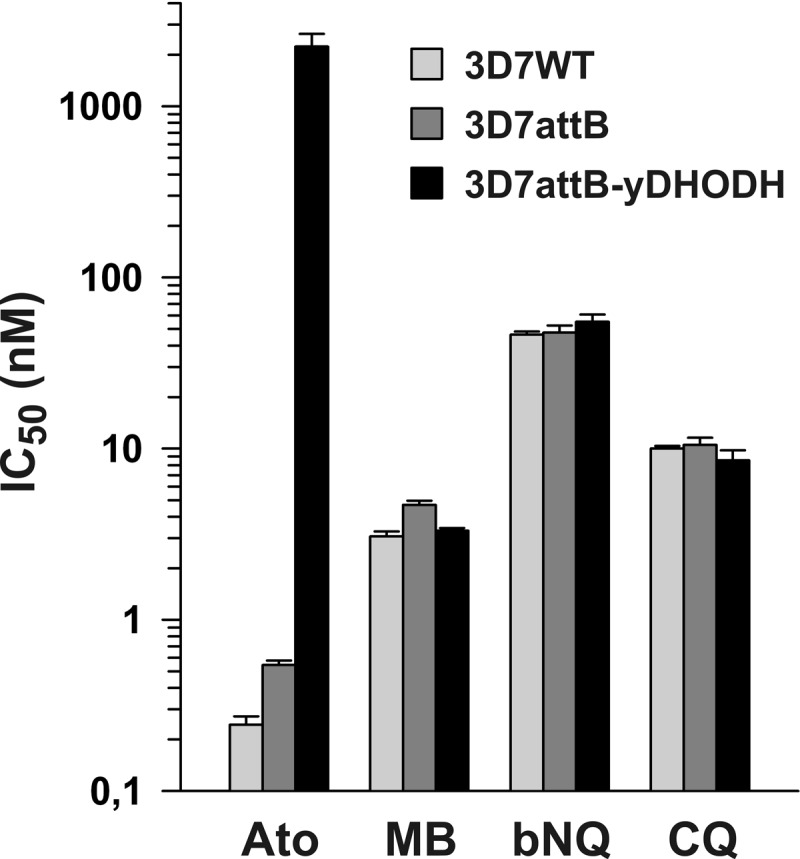
Average IC50s for antimalarial agents determined from growth inhibition assays with P. falciparum strains 3D7, 3D7attB, and 3D7attB-yDHODH. Ato, atovaquone; MB, methylene blue; bNQ, benzylnaphthoquinone 1c from reference 22; CQ, chloroquine. All values are means ± the standard errors of the mean from three independent growth inhibition assays. The IC50s are summarized in Table 1.
The IC50s for the negative control chloroquine were very similar for all three strains (Table 1). In contrast, the antimalarial activity of the positive control atovaquone was significantly reduced against the strain 3D7attB-yDHODH, and the IC50 increased by almost 4 orders of magnitude (please note the logarithmic scale in Fig. 2). Methylene blue and benzylnaphthoquinone 1c displayed potent antiplasmodial activities against all of the strains. The IC50s for atovaquone, methylene blue, and chloroquine presented in Table 1 correspond well with values reported elsewhere (5, 18, 38, 39). A similar low nanomolar IC50 was previously reported for benzylnaphthoquinone 1c, as well as a series of five analogues that were tested against the P. falciparum Dd2 strain (22). It is worth noting that the presence or absence of PfGlp3 did not affect the IC50s of the redox-active agents. Of particular importance, the presence of cytosolic yDHODH altered the activity of the mETC inhibitor atovaquone but had no effect on the antimalarial activities of chloroquine, methylene blue, or benzylnaphthoquinone 1c. Hence, the latter three agents do not exert their activity as inhibitors of the mETC in asexual blood-stage parasites.
Table 1.
Averaged IC50s determined from growth inhibition assays with strains 3D7, 3D7attB, and 3D7attB-yDHODH
| Agenta | Mean IC50 (nM) ± SEMb for strain: |
||
|---|---|---|---|
| 3D7 | 3D7attB | 3D7attB-yDHODH | |
| Ato | 0.24 ± 0.03 | 0.54 ± 0.03 | 2,230 ± 419 |
| MB | 3.07 ± 0.21 | 4.70 ± 0.27 | 3.31 ± 0.12 |
| bNQ | 46.3 ± 2.04 | 47.6 ± 4.79 | 55.1 ± 5.6 |
| CQ | 10.0 ± 0.34 | 10.5 ± 1.07 | 8.55 ± 1.24 |
Ato, atovaquone; MB, methylene blue; bNQ, benzylnaphthoquinone 1c from reference 22; CQ, chloroquine.
All values are means from three independent growth inhibition assays.
It is interesting that the enzymatic activities of the mETC were reported to increase in cultured gametocytes, and these stages also possess mitochondria with more cristae (40). Furthermore, recent experiments on a mutant strain of the rodent malaria parasite P. berghei that lacks a functional complex II revealed that succinate dehydrogenase and the mETC are crucial for ookinete development in vivo (41). Since methylene blue was reported to have a strong gametocytocidal effect (20), we cannot entirely exclude the possibility that it acts on the mETC in gametocytes and other stages. However, considering the complete absence of effects against blood stage parasites (Fig. 2), such a scenario seems unlikely, unless the mode of action is not due to a classical inhibition of the mETC but due to an activity as a subversive substrate (Fig. 1C). If methylene blue or benzylnaphthoquinone 1c target the mETC as a subversive substrate, the mETC could act as their electron donor/acceptor regardless of the presence or absence of cytosolic yDHODH. For instance, both inhibitors might be suited to accept electrons from the endogenous type 2 DHODH or from complex III. The reduced redox cycler could subsequently catalyze the formation of reactive oxygen species and/or of Fe(II) species as suggested earlier (22) - without depletion of the pyrimidine pool. Reduced menadione species with antitrypanosomal activities were, for example, shown to generate O2·− and to reduce cytochrome c [Fe(III)] in vitro (42, 43). Future work is needed to address whether methylene blue or benzylnaphthoquinone 1c are subversive substrates of the mETC of malaria parasites.
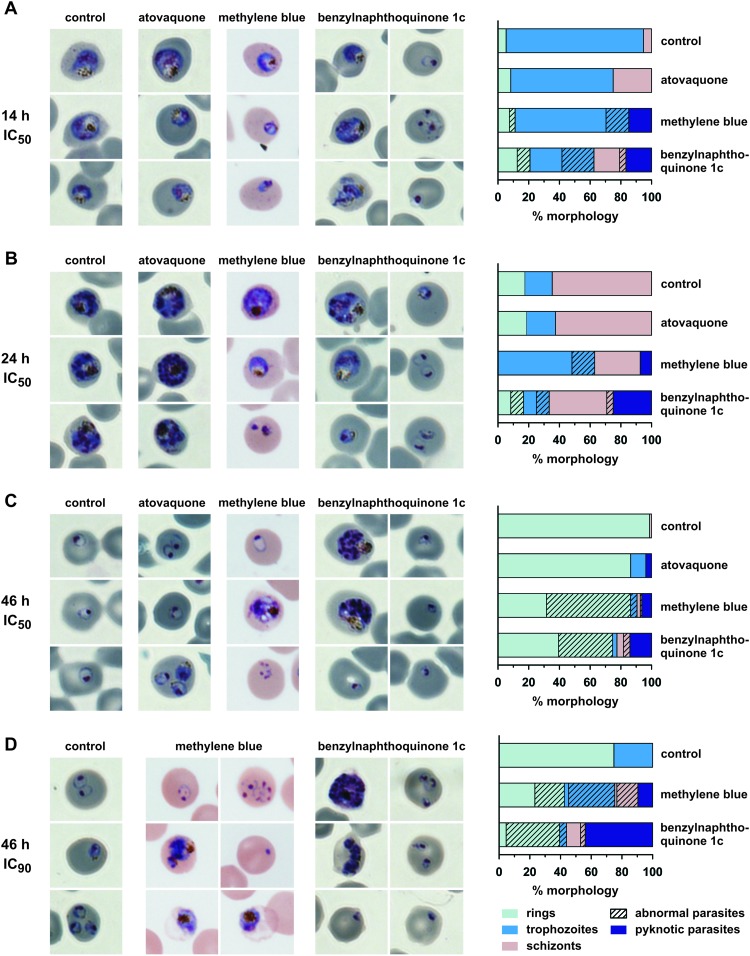
Certain antimalarial agents and oxidants, such as H2O2, can alter the morphology of P. falciparum (44), which can sometimes provide clues to the mode of action of the drug. We therefore analyzed the morphology of synchronized parasite cultures during 46 h of incubation with benzylnaphthoquinone 1c, methylene blue, or atovaquone (Fig. 3). The morphologies of atovaquone-treated parasites were very similar to the untreated controls, which is in agreement with previous reports (11). In contrast, the growth of parasites that were treated with benzylnaphthoquinone 1c was retarded, and a significant increase of abnormal parasite morphologies was observed (Fig. 3). After 14 h of drug treatment, fewer trophozoite-stage parasites and more ring-stage parasites were detected than in the control (Fig. 3A). Some of the abnormal ring-stage parasites had a distorted mesh-like morphology, which has been previously observed in stressed cultures and usually indicates that the parasite is incapable of further development (44). Other ring-stage parasites developed into residual pyknotic bodies as previously described (22). After 24 h of drug treatment, trophozoites and schizonts appeared to be younger than in the control, which contained predominantly older mid- to late-stage schizonts (Fig. 3B). After 46 h, the benzylnaphthoquinone-treated culture contained remaining schizonts from the first infection cycle and numerous very small ring-stage parasites from the second infection cycle, whereas the controls did not contain schizonts and instead consisted of late ring-stage parasites from the second infection cycle (Fig. 3C). The effect was more pronounced at higher drug concentrations, resulting in a significant increase of residual pyknotic bodies and disordered schizont-stage parasites (Fig. 3D). Thus, parasites treated with benzylnaphthoquinone 1c present a distinct morphology and appear to become predominantly pyknotic at the ring stage. An even more pronounced effect on ring-stage parasites was observed for methylene blue, as indicated by the clearance of these parasite stages after 24 h of drug treatment (Fig. 3B). In addition, for methylene blue, the ratio between mesh-like and pyknotic ring-stage parasites seemed to be shifted toward the mesh-like morphology. Another difference between the methylene blue-treated parasites and the other cultures was that ca. 40% of trophozoite- and schizont-stage parasites were surrounded by faintly stained erythrocytes after methylene blue treatment at IC90 concentrations (Fig. 3D). Since the surrounding uninfected erythrocytes in the blood smears remained intact, the results suggest a selective lysis of infected red blood cells after prolonged treatment with methylene blue. In summary, the observed morphological differences might be indicative of differences in drug action or drug metabolism during the course of the intraerythrocytic life cycle. A more detailed investigation is required to explore a putative methylene blue-mediated lysis of infected erythrocytes, as well as the observed growth retardation and the apparent death of young ring-stage parasites for benzylnaphthoquinone 1c. Taken together, the data of Fig. 2 and 3 support the conclusion that atovaquone, methylene blue, and benzylnaphthoquinone 1c do not share a mechanism of action.
Fig 3.
Parasite morphology after treatment with atovaquone, methylene blue, or benzylnaphthoquinone 1c. Synchronized parasite cultures were treated with one of the drugs at the ring stage (approximately 16 h postinfection). An untreated control culture was analyzed in parallel. (A to C) Treatment with the IC50 concentrations for 14 h (A), 24 h (B), and 46 h (C). (D) Treatment with the IC90 concentration for 46 h. Quantifications of the observed morphologies of the remaining parasites at each time point are shown on the right side. Color differences between the microscopic images are due to white balance settings and were not adjusted in order to avoid image manipulation.
What might be the crucial properties that underlie the differences in the modes of action of atovaquone, methylene blue, benzylnaphthoquinone 1c, and related redox agents? When we recently studied the ability of human and P. falciparum GR to reduce either atovaquone or 2-hydroxy-1,4-naphthoquinone at substrate concentrations of up to 25 or 100 μM, respectively, no NADPH consumption was observed (23, 45). In contrast, methylene blue and certain 1,4-naphthoquinones were shown to be effective substrates (24, 46) with catalytic efficiencies—i.e., the kcat/Km values—as high as 13.7 mM−1 s−1 for methylene blue and 12.5 mM−1 s−1 for the predicted 3-[4-(substituted)benzoyl]-menadione metabolites (22). These findings add further support to the idea that atovaquone, methylene blue, and 3-[4-(substituted)benzyl]-menadione derivatives do not share the same mechanism(s) of action (23, 45). Apart from obvious structural differences (Fig. 1A), an important property seems to be the redox potential. The low redox potential of atovaquone (−0.51 V) indicates that, under physiological conditions, atovaquone reduction is considerably less favored compared to menadione reduction (−0.14 V) (47). Even under highly reducing intracellular conditions with an estimated half-cell redox potential for NADPH around −0.34 V (48), the redox potential for atovaquone will be too low for efficient reduction by two-electron reduced GRs, which have redox potentials around −0.24 V at pH 7 (49). In contrast, the two-electron reduction potentials for methylene blue and menadione at pH 7 are +0.01 V (50) and −0.23 V (51), respectively, and cyclic voltammetry measurements show that one-electron and two-electron transfer reactions are kinetically reversible (46). These properties allow a continuous reduction and oxidation under physiological conditions, which is a prerequisite for a subversive substrate-driven redox cycle (Fig. 1C).
Is GR the electron donor for methylene blue and benzylnaphthoquinone 1c in vivo? A key study by Pastrana-Mena et al. revealed that the absence of P. berghei GR has no significant effect on the sensitivity of the rodent malaria parasite to methylene blue (52). Even though these findings point to an alternative electron donor for this drug (52), it is important to note that there seem to be substantial differences between the glutathione metabolisms of human and rodent malaria parasites. For example, a recent study by Patzewitz et al. suggests that glutathione biosynthesis is essential for the asexual blood stages of P. falciparum in contrast to P. berghei (53). To date, there is no P. falciparum GR knockout strain. Thus, we could not analyze the IC50s for methylene blue and benzylnaphthoquinone 1c in such a genetic background. In addition, P. falciparum-infected red blood cells have two GRs: the host and the parasite enzyme. Previous studies on methylene blue and benzylnaphthoquinone 1c clearly showed that these redox cyclers (or their metabolites) are subversive substrates of both GRs in NADPH-consuming in vitro assays (22). The redox cyclers could therefore act synergistically on the human and the P. falciparum enzyme, which might result in a disturbed redox homeostasis of the host-parasite unit. None of the previous studies on P. berghei GR knockout strains (50, 54) or human GR deficiency (55) targeted both enzymes of the host-parasite unit together (in contrast to the redox cyclers [22, 24]). Nevertheless, the genetic studies raise the question of alternative targets for these drugs, and the redox cyclers might indeed have pleiotropic effects, e.g., by altering the NADPH flux, which is crucial for several flavoenzymes that are involved in the parasite life cycle.
Glucose-6-phosphate dehydrogenase (G6PDH), a key enzyme of the pentose phosphate shunt, generates NADPH, which is subsequently consumed by GR and other enzymes. Conditions that affect the host G6PDH activity induce an increased sensitivity to oxidants, and homozygous or hemizygous G6PDH-deficient individuals often suffer from hemolytic episodes, particularly when they have to cope with additional exogenous oxidants such as drugs or ingested fava beans. On the other hand, the global distribution of G6PDH deficiency is very similar to that seen with malaria, which supports the so-called malaria protection hypothesis (56). A far less prevalent GR deficiency or drug-induced GR inhibition may also protect from malaria due to an enhanced phagocytosis of ring-stage parasites (55). Redox-active compounds such as dapsone and primaquine can cause clinical cases of methemoglobinemia and hemolytic anemia in G6PDH-deficient patients, and the evaluation of the hemolytic risk is an essential requirement at the lead optimization stage in any antimalarial drug discovery project (57). Among the arsenal of antimalarial drugs, methylene blue proved to be safe and effective in the treatment of uncomplicated falciparum malaria when combined with other antimalarials (19). Very recently, methylene blue treatment was reported to be associated with slightly reduced hemoglobin values in children with hemi- and homozygous G6PDH deficiency. Since no evidence for an excess of severe anemia was observed, the effect appears to be of limited clinical relevance (58). Our cell culture experiments suggest that methylene blue can lead to red blood cell lysis; however, this was only detected for infected erythrocytes, whereas uninfected erythrocytes remained intact (Fig. 3D). In the case of benzylnaphthoquinone 1c, no hemolysis was induced by the compound (22). Furthermore, methemoglobinemia is not expected to be a side effect of benzylnaphthoquinone 1c or methylene blue treatment because both compounds were shown to convert methemoglobin to oxyhemoglobin in the presence of GR (22, 24).
In conclusion, benzylnaphthoquinone 1c and methylene blue do not inhibit the mETC of the asexual blood stages of P. falciparum. Further work is required to analyze a potential stage specificity and to elucidate the modes of action of these agents in cell culture and in vivo. This includes the identification and validation of the relevant electron donors and acceptors.
ACKNOWLEDGMENTS
This study was supported by the Centre National de la Recherche Scientifique (CNRS, France, UMR 7509 [E.D.-C.]), the University of Strasbourg, the International Center for Frontier Research in Chemistry (www.icFRC.fr [E.D.-C.]) in Strasbourg, by the ANRémergence program (grant SCHISMAL [E.D.-C.]), by the Laboratoire d'Excellence (LabEx) ParaFrap (grant LabEx ParaFrap ANR-11-LABX-0024 [E.D.-C.]), and by a grant from National Institutes of Health (grant AI028398 [A.B.V.]). K.E. is grateful to the CNRS, France, and to Alain van Dorsselaer for her cofunded CNRS doctoral fellowship (BDI).
We thank Sophia Deil for help with Fiji/ImageJ.
Footnotes
Published ahead of print 25 February 2013
REFERENCES
- 1. Fry M, Pudney M. 1992. Site of action of the antimalarial hydroxynaphthoquinone, 2-[trans-4-(4′-chlorophenyl)cyclohexyl]-3-hydroxy-1,4-naphthoquinone (566C80). Biochem. Pharmacol. 43:1545–1553 [DOI] [PubMed] [Google Scholar]
- 2. Srivastava IK, Rottenberg H, Vaidya AB. 1997. Atovaquone, a broad spectrum antiparasitic drug, collapses mitochondrial membrane potential in a malarial parasite. J. Biol. Chem. 272:3961–3966 [DOI] [PubMed] [Google Scholar]
- 3. Srivastava IK, Morrisey JM, Darrouzet E, Daldal F, Vaidya AB. 1999. Resistance mutations reveal the atovaquone-binding domain of cytochrome b in malaria parasites. Mol. Microbiol. 33:704–711 [DOI] [PubMed] [Google Scholar]
- 4. Vaidya AB, Mather MW. 2009. Mitochondrial evolution and functions in malaria parasites. Annu. Rev. Microbiol. 63:249–267 [DOI] [PubMed] [Google Scholar]
- 5. Biagini GA, Fisher N, Shone AE, Mubaraki MA, Srivastava A, Hill A, Antoine T, Warman AJ, Davies J, Pidathala C, Amewu RK, Leung SC, Sharma R, Gibbons P, Hong DW, Pacorel B, Lawrenson AS, Charoensutthivarakul S, Taylor L, Berger O, Mbekeani A, Stocks PA, Nixon GL, Chadwick J, Hemingway J, Delves MJ, Sinden RE, Zeeman AM, Kocken CH, Berry NG, O'Neill PM, Ward SA. 2012. Generation of quinolone antimalarials targeting the Plasmodium falciparum mitochondrial respiratory chain for the treatment and prophylaxis of malaria. Proc. Natl. Acad. Sci. U. S. A. 109:8298–8303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Sullivan DJ, Jr, Gluzman IY, Russell DG, Goldberg DE. 1996. On the molecular mechanism of chloroquine's antimalarial action. Proc. Natl. Acad. Sci. U. S. A. 93:11865–11870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Pagola S, Stephens PW, Bohle DS, Kosar AD, Madsen SK. 2000. The structure of malaria pigment beta-haematin. Nature 404:307–310 [DOI] [PubMed] [Google Scholar]
- 8. Petersen I, Eastman R, Lanzer M. 2011. Drug-resistant malaria: molecular mechanisms and implications for public health. FEBS Lett. 585:1551–1562 [DOI] [PubMed] [Google Scholar]
- 9. Kessl JJ, Ha KH, Merritt AK, Lange BB, Hill P, Meunier B, Meshnick SR, Trumpower BL. 2005. Cytochrome b mutations that modify the ubiquinol-binding pocket of the cytochrome bc1 complex and confer anti-malarial drug resistance in Saccharomyces cerevisiae. J. Biol. Chem. 280:17142–17148 [DOI] [PubMed] [Google Scholar]
- 10. Kessl JJ, Moskalev NV, Gribble GW, Nasr M, Meshnick SR, Trumpower BL. 2007. Parameters determining the relative efficacy of hydroxy-naphthoquinone inhibitors of the cytochrome bc1 complex. Biochim. Biophys. Acta 1767:319–326 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Painter HJ, Morrisey JM, Vaidya AB. 2010. Mitochondrial electron transport inhibition and viability of intraerythrocytic Plasmodium falciparum. Antimicrob. Agents Chemother. 54:5281–5287 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Balabaskaran Nina P, Morrisey JM, Ganesan SM, Ke H, Pershing AM, Mather MW, Vaidya AB. 2011. ATP synthase complex of Plasmodium falciparum: dimeric assembly in mitochondrial membranes and resistance to genetic disruption. J. Biol. Chem. 286:41312–41322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Eckers E, Cyrklaff M, Simpson L, Deponte M. 2012. Mitochondrial protein import pathways are functionally conserved among eukaryotes despite compositional diversity of the import machineries. Biol. Chem. 393:513–524 [DOI] [PubMed] [Google Scholar]
- 14. Painter HJ, Morrisey JM, Mather MW, Vaidya AB. 2007. Specific role of mitochondrial electron transport in blood-stage Plasmodium falciparum. Nature 446:88–91 [DOI] [PubMed] [Google Scholar]
- 15. Deponte M, Hoppe HC, Lee M, Maier AG, Richard D, Rug M, Spielmann T, Przyborski JM. 2012. Wherever I may roam: protein and membrane trafficking in Plasmodium falciparum-infected red blood cells. Mol. Biochem. Parasitol. 186:95–116 [DOI] [PubMed] [Google Scholar]
- 16. Krungkrai J. 1995. Purification, characterization, and localization of mitochondrial dihydroorotate dehydrogenase in Plasmodium falciparum, human malaria parasite. Biochim. Biophys. Acta 1243:351–360 [DOI] [PubMed] [Google Scholar]
- 17. Booker ML, Bastos CM, Kramer ML, Barker RH, Jr, Skerlj R, Sidhu AB, Deng X, Celatka C, Cortese JF, Guerrero Bravo JE, Crespo Llado KN, Serrano AE, Angulo-Barturen I, Jimenez-Diaz MB, Viera S, Garuti H, Wittlin S, Papastogiannidis P, Lin JW, Janse CJ, Khan SM, Duraisingh M, Coleman B, Goldsmith EJ, Phillips MA, Munoz B, Wirth DF, Klinger JD, Wiegand R, Sybertz E. 2010. Novel inhibitors of Plasmodium falciparum dihydroorotate dehydrogenase with anti-malarial activity in the mouse model. J. Biol. Chem. 285:33054–33064 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Ke H, Morrisey JM, Ganesan SM, Painter HJ, Mather MW, Vaidya AB. 2011. Variation among Plasmodium falciparum strains in their reliance on mitochondrial electron transport chain function. Eukaryot. Cell 10:1053–1061 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Zoungrana A, Coulibaly B, Sie A, Walter-Sack I, Mockenhaupt FP, Kouyate B, Schirmer RH, Klose C, Mansmann U, Meissner P, Muller O. 2008. Safety and efficacy of methylene blue combined with artesunate or amodiaquine for uncomplicated falciparum malaria: a randomized controlled trial from Burkina Faso. PLoS One 3:e1630 doi:10.1371/journal.pone.0001630 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Coulibaly B, Zoungrana A, Mockenhaupt FP, Schirmer RH, Klose C, Mansmann U, Meissner PE, Muller O. 2009. Strong gametocytocidal effect of methylene blue-based combination therapy against falciparum malaria: a randomised controlled trial. PLoS One 4:e5318 doi:10.1371/journal.pone.0005318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Bountogo M, Zoungrana A, Coulibaly B, Klose C, Mansmann U, Mockenhaupt FP, Burhenne J, Mikus G, Walter-Sack I, Schirmer RH, Sie A, Meissner P, Muller O. 2010. Efficacy of methylene blue monotherapy in semi-immune adults with uncomplicated falciparum malaria: a controlled trial in Burkina Faso. Trop. Med. Int. Health 15:713–717 [DOI] [PubMed] [Google Scholar]
- 22. Müller T, Johann L, Jannack B, Bruckner M, Lanfranchi DA, Bauer H, Sanchez C, Yardley V, Deregnaucourt C, Schrevel J, Lanzer M, Schirmer RH, Davioud-Charvet E. 2011. Glutathione reductase-catalyzed cascade of redox reactions to bioactivate potent antimalarial 1,4-naphthoquinones: a new strategy to combat malarial parasites. J. Am. Chem. Soc. 133:11557–11571 [DOI] [PubMed] [Google Scholar]
- 23. Davioud-Charvet E, Lanfranchi DA. 2011. Subversive substrates of glutathione reductases from Plasmodium falciparum-infected red blood cells as antimalarial agents, p 375–396 In Selzer P. (ed), Drug discovery in infectious diseases, vol 2 Wiley-VCH, Weinheim, Germany [Google Scholar]
- 24. Blank O, Davioud-Charvet E, Elhabiri M. 2012. Interactions of the antimalarial drug methylene blue with methemoglobin and heme targets in Plasmodium falciparum: a physico-biochemical study. Antioxid. Redox Signal. 17:544–554 [DOI] [PubMed] [Google Scholar]
- 25. Buchholz K, Schirmer RH, Eubel JK, Akoachere MB, Dandekar T, Becker K, Gromer S. 2008. Interactions of methylene blue with human disulfide reductases and their orthologues from Plasmodium falciparum. Antimicrob. Agents Chemother. 52:183–191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Johann L, Lanfranchi DA, Davioud-Charvet E, Elhabiri M. 2012. A physico-biochemical study on potential redox-cyclers as antimalarial and anti-schistosomal drugs. Curr. Pharm. Des. 18:3539–3566 [PMC free article] [PubMed] [Google Scholar]
- 27. Hogg T, Nagarajan K, Herzberg S, Chen L, Shen X, Jiang H, Wecke M, Blohmke C, Hilgenfeld R, Schmidt CL. 2006. Structural and functional characterization of falcipain-2, a hemoglobinase from the malarial parasite Plasmodium falciparum. J. Biol. Chem. 281:25425–25437 [DOI] [PubMed] [Google Scholar]
- 28. Monti D, Vodopivec B, Basilico N, Olliaro P, Taramelli D. 1999. A novel endogenous antimalarial: Fe(II)-protoporphyrin IX alpha (heme) inhibits hematin polymerization to beta-hematin (malaria pigment) and kills malaria parasites. Biochemistry 38:8858–8863 [DOI] [PubMed] [Google Scholar]
- 29. Johnson LV, Walsh ML, Chen LB. 1980. Localization of mitochondria in living cells with rhodamine 123. Proc. Natl. Acad. Sci. U. S. A. 77:990–994 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Vos M, Esposito G, Edirisinghe JN, Vilain S, Haddad DM, Slabbaert JR, Van Meensel S, Schaap O, De Strooper B, Meganathan R, Morais VA, Verstreken P. 2012. Vitamin K2 is a mitochondrial electron carrier that rescues pink1 deficiency. Science 336:1306–1310 [DOI] [PubMed] [Google Scholar]
- 31. Winter RW, Kelly JX, Smilkstein MJ, Dodean R, Hinrichs D, Riscoe MK. 2008. Antimalarial quinolones: synthesis, potency, and mechanistic studies. Exp. Parasitol. 118:487–497 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Trager W, Jensen JB. 1976. Human malaria parasites in continuous culture. Science 193:673–675 [DOI] [PubMed] [Google Scholar]
- 33. Lambros C, Vanderberg JP. 1979. Synchronization of Plasmodium falciparum erythrocytic stages in culture. J. Parasitol. 65:418–420 [PubMed] [Google Scholar]
- 34. Beez D, Sanchez CP, Stein WD, Lanzer M. 2011. Genetic predisposition favors the acquisition of stable artemisinin resistance in malaria parasites. Antimicrob. Agents Chemother. 55:50–55 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A. 2012. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9:676–682 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Deponte M, Becker K, Rahlfs S. 2005. Plasmodium falciparum glutaredoxin-like proteins. Biol. Chem. 386:33–40 [DOI] [PubMed] [Google Scholar]
- 37. Nkrumah LJ, Muhle RA, Moura PA, Ghosh P, Hatfull GF, Jacobs WR, Jr, Fidock DA. 2006. Efficient site-specific integration in Plasmodium falciparum chromosomes mediated by mycobacteriophage Bxb1 integrase. Nat. Methods 3:615–621 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Vennerstrom JL, Makler MT, Angerhofer CK, Williams JA. 1995. Antimalarial dyes revisited: xanthenes, azines, oxazines, and thiazines. Antimicrob. Agents Chemother. 39:2671–2677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Baniecki ML, Wirth DF, Clardy J. 2007. High-throughput Plasmodium falciparum growth assay for malaria drug discovery. Antimicrob. Agents Chemother. 51:716–723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Krungkrai J, Prapunwattana P, Krungkrai SR. 2000. Ultrastructure and function of mitochondria in gametocytic stage of Plasmodium falciparum. Parasite 7:19–26 [DOI] [PubMed] [Google Scholar]
- 41. Hino A, Hirai M, Tanaka TQ, Watanabe YI, Matsuoka H, Kita K. 2012. Critical roles of the mitochondrial complex II in oocyst formation of rodent malaria parasite Plasmodium berghei. J. Biochem. 152:259–268 [DOI] [PubMed] [Google Scholar]
- 42. Blumenstiel K, Schöneck R, Yardley V, Croft SL, Krauth-Siegel RL. 1999. Nitrofuran drugs as common subversive substrates of Trypanosoma cruzi lipoamide dehydrogenase and trypanothione reductase. Biochem. Pharmacol. 58:1791–1799 [DOI] [PubMed] [Google Scholar]
- 43. Salmon-Chemin L, Buisine E, Yardley V, Kohler S, Debreu MA, Landry V, Sergheraert C, Croft SL, Krauth-Siegel LR, Davioud-Charvet E. 2001. 2- and 3-substituted-1,4-naphthoquinone derivatives as subversive substrates of trypanothione reductase and lipoamide dehydrogenase from Trypanosoma cruzi: synthesis and correlation between redox-cycling activities and in vitro cytotoxicity. J. Med. Chem. 44:548–565 [DOI] [PubMed] [Google Scholar]
- 44. Deponte M, Becker K. 2004. Plasmodium falciparum-do killers commit suicide? Trends Parasitol. 20:165–169 [DOI] [PubMed] [Google Scholar]
- 45. Lanfranchi DA, Belorgey D, Müller T, Vezin H, Lanzer M, Davioud-Charvet E. 2012. Exploring the trifluoromenadione core as a template to design antimalarial redox-active agents interacting with glutathione reductase. Org. Biomol. Chem. 10:4795–4806 [DOI] [PubMed] [Google Scholar]
- 46. Morin C, Besset T, Moutet JC, Fayolle M, Bruckner M, Limosin D, Becker K, Davioud-Charvet E. 2008. The aza-analogues of 1,4-naphthoquinones are potent substrates and inhibitors of plasmodial thioredoxin and glutathione reductases and of human erythrocyte glutathione reductase. Org. Biomol. Chem. 6:2731–2742 [DOI] [PubMed] [Google Scholar]
- 47. Lopez-Shirley K, Zhang F, Gosser D, Scott M, Meshnick SR. 1994. Antimalarial quinones: redox potential dependence of methemoglobin formation and heme release in erythrocytes. J. Lab. Clin. Med. 123:126–130 [PubMed] [Google Scholar]
- 48. Deponte M. 2012. Glutathione catalysis and the reaction mechanisms of glutathione-dependent enzymes. Biochim. Biophys. Acta. PMID 23036594. doi:10.1016/j.bbagen.2012.09.018 [DOI] [PubMed] [Google Scholar]
- 49. Veine DM, Arscott LD, Williams CH., Jr 1998. Redox potentials for yeast, Escherichia coli, and human glutathione reductase relative to the NAD+/NADH redox couple: enzyme forms active in catalysis. Biochemistry 37:15575–15582 [DOI] [PubMed] [Google Scholar]
- 50. Impert O, Katafias A, Kita P, Mills A, Pietkiewiez-Graczyk A, Wrzeszcz G. 2003. Kinetics and mechanism of a fast leuco-Methylene Blue oxidation by copper(II)-halide species in acidic aqueous media. Dalton Trans. 3:348–353 [Google Scholar]
- 51. Patriarche GJ, Lingane JJ. 1970. Electrochemical characteristics of 2-methyl-I,4-naphthoquinone (vitamin K3): a coulometric micromethod of determination. Anal. Chim. Acta 49:241–246 [DOI] [PubMed] [Google Scholar]
- 52. Pastrana-Mena R, Dinglasan RR, Franke-Fayard B, Vega-Rodríguez J, Fuentes-Caraballo M, Baerga-Ortiz A, Coppens I, Jacobs-Lorena M, Janse CJ, Serrano AE. 2010. Glutathione reductase-null malaria parasites have normal blood stage growth but arrest during development in the mosquito. J. Biol. Chem. 285:27045–27056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Patzewitz EM, Wong EH, Müller S. 2012. Dissecting the role of glutathione biosynthesis in Plasmodium falciparum. Mol. Microbiol. 83:304–318 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Buchholz K, Putrianti ED, Rahlfs S, Schirmer RH, Becker K, Matuschewski K. 2010. Molecular genetics evidence for the in vivo roles of the two major NADPH-dependent disulfide reductases in the malaria parasite. J. Biol. Chem. 285:37388–37395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Gallo V, Schwarzer E, Rahlfs S, Schirmer RH, van Zwieten R, Roos D, Arese P, Becker K. 2009. Inherited glutathione reductase deficiency and Plasmodium falciparum malaria: a case study. PLoS One 4:e7303 doi:10.1371/journal.pone.0007303 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Cappellini MD, Fiorelli G. 2008. Glucose-6-phosphate dehydrogenase deficiency. Lancet 371:64–74 [DOI] [PubMed] [Google Scholar]
- 57. Belorgey D, Lanfranchi DA, Davioud-Charvet E. 2012. 1,4-Naphthoquinones and others NADPH-dependent glutathione reductase-catalyzed redox cyclers as antimalarial agents. Curr. Pharm. Des. [Epub ahead of print.] PMID 23116403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Müller O, Mockenhaupt FP, Marks B, Meissner P, Coulibaly B, Kuhnert R, Buchner H, Schirmer RH, Walter-Sack I, Sié A, Mansmann U. 2013. Haemolysis risk in methylene blue treatment of G6PD-sufficient and G6PD-deficient West-African children with uncomplicated falciparum malaria: a synopsis of four RCTs. Pharmacoepidemiol. Drug Safety. PMID 23135803. doi:10.1002/pds.3370 [DOI] [PubMed] [Google Scholar]