Abstract
Administration of retinoic acid, the active metabolite of vitamin A, is linked to the stimulation of nephrogenesis. The aim of this study was to determine whether early postnatal administration of retinoic acid (RA) could enhance ongoing nephrogenesis in a baboon model of premature birth. Unbiased stereological methods were employed to estimate kidney volume, renal corpuscle volume and nephron number. The percentage of abnormal glomeruli and the number of glomerular generations were also determined in the kidneys of preterm control (n=6) and preterm +RA (n=6) animals that received 500 μg/kg/day of all-trans retinoic acid following premature delivery. There was no significant difference between the preterm control and the preterm +RA groups in kidney size, nephron number (preterm control: 329,924 ± 41,752; preterm +RA: 354,041 ± 52,095; p = 0.59), renal corpuscle volume, number of glomerular generations, or the percentage of abnormal glomeruli. The proportion of abnormal glomeruli did not appear to be linked to any elements of postnatal care examined. The results of this study indicate that early postnatal administration of retinoic acid is unable to stimulate nephrogenesis in the kidney of the preterm baboon. Encouragingly, it does not appear to have any adverse effects on kidney development.
Keywords: Vitamin A, Preterm Birth, Papio hamadryas, Kidney, Nephron
Advances in neonatal care have markedly improved the survival of preterm infants such that neonates as young as 26 weeks gestation (term = 37 weeks) now have a 60–80% chance of survival (1, 2). There is limited knowledge to date, however, as to long-term adverse health consequences of preterm birth, and the possibility that these may be prevented through neonatal intervention.
A major organ that rapidly develops during late gestation is the kidney, with the formation of nephrons (nephrogenesis) continuing until approximately 36 weeks gestation (3). Furthermore, 60% of nephrons are developed during the third trimester of pregnancy (4). Infants born preterm, therefore, are delivered before nephrogenesis is completed; it is conceivable that this will impact upon their future nephron endowment and kidney function. Indeed, the results of a recent study by Gubhaju et al. (Gubhaju L et al. The effects of pre-term birth on nephrogenesis, 32nd Annual FNPS Meeting, September 25–28, 2005, Glenelg, Australia, Abstract O8), in a non-human primate model of preterm birth, has indicated that an attenuation of the nephrogenic potential of the individual may occur due to preterm delivery, suggesting that nephrogenesis in the extrauterine environment is impaired. In a human post-mortem analysis, Rodriguez et al. (5) determined that, although nephrogenesis is ongoing after preterm birth, infants had a significantly reduced number of glomerular generations which is potentially indicative of a nephron deficit. Furthermore, a number of the preterm infants exhibited glomerular abnormalities, such as cystic dilation of the Bowman’s space. This deficit in functional nephrons may in turn lead to long-term complications, as a reduced nephron endowment has been linked to an increased susceptibility to hypertension and the development of renal disease later in life (6–8). Therefore, it is that nephron number be maximized before the completion of nephrogenesis.
Retinoic acid (RA), the active metabolite of vitamin A, acts as a transcription factor and modulates the expression of the Ret tyrosine kinase receptor (9). Increased Ret signaling leads to an increase in ureteric bud branching in the developing kidney, and through this mechanism retinoic acid has been linked to the modulation of nephron number both in vitro (10) and in vivo. Importantly, a clear linear correlation has been shown between vitamin A status and the nephron endowment of newborns in the rodent model (11), and retinoic acid administration during mid-gestation has been shown to prevent the 30% nephron deficit expected due to intrauterine growth restriction in rat offspring (12).
Given the importance of retinoic acid signaling during kidney development, it is conceivable that the administration of vitamin A derivatives following preterm birth may act to augment ongoing nephrogenesis. In this regard, recommendations for vitamin A supplementation in neonates are currently in place (13), as it has previously been demonstrated that low birth weight and preterm neonates have low levels of both hepatic and serum retinol (14, 15). A Cochrane Review of clinical trials has since established that retinol supplementation in low birth weight infants results in improved vitamin A status leading to a reduction in neonatal mortality and oxygen requirement at one month of age (16). This may be due to a beneficial effect of vitamin A supplementation on lung development, as has been evidenced in a number of experimental studies (17–20).
The effects of postnatal vitamin A supplementation on kidney development, however, are currently unknown. The major aim of this study, therefore, was to determine whether early postnatal administration of retinoic acid could stimulate nephrogenesis in a non-human primate model of preterm birth. Fetal baboons were delivered at 125d gestation (term = 185d), equivalent to approximately 26 weeks gestation in humans, and placed in a neonatal intensive care unit after birth (21). Importantly, the ontogeny of the baboon kidney follows very closely to that of human renal development (22), with ongoing nephrogenesis following preterm birth (Gubhaju L et al. The effects of pre-term birth on nephrogenesis, 32nd Annual FNPS Meeting, September 25–28, 2005, Glenelg, Australia, Abstract O8).
Methods
Induction of Preterm Delivery
All animal experiments were performed at the Southwest Foundation for Biomedical Research (San Antonio, TX, USA), with animal handling and husbandry procedures reviewed and approved to conform to the American Association for Accreditation of Laboratory Animal Care guidelines.
At 48 and 24 hours before delivery, pregnant baboons received 6mg of betamethasone (Celestone Soluspan; Schering-Plough Pharmaceuticals, Kenilworth, NJ, USA) via intramuscular injection. Baboon neonates were delivered prematurely by caesarean section at 125 days gestation (term = 185d).
Postnatal Care
A detailed description of the postnatal intensive care protocols, including nutritional and ventilatory management, has been previously published (21). As described, respiration, cardiac function, fluid levels and urine output were all maintained within the normal parameters of the extremely low birth weight infant.
Medication
All preterm neonates were administered ampicillin and gentamicin for the first 7–10 days of life, at the recommended dosage for infants less than 29 weeks gestation (23). Prophylactic fluconazole (6 mg/kg) was administered at 12, 96 and 168 hours of age, then twice a week until postnatal day 21. Further doses of antibiotics were used in cases of clinically suspected infection. All neonates were treated with intravenous caffeine citrate (20 mg/kg) at 1 and 12 hours of age, followed by 10 mg/kg daily.
In the case of significant hypotension, the stepwise administration of additional volume, dopamine, dobutamine, and hydrocortisone was undertaken. Patent ductus arteriosus was treated with indomethacin or surgical ligation.
Retinoic Acid Treatment
Baboon neonates were randomly assigned to treatment groups before delivery. The animals in the preterm + RA treatment group (n=6) received 500 μg/kg/day of all-trans retinoic acid (17–19) in almond oil via gavage commencing within 24 hours of delivery. 2000 IU/kg of retinol (Aquasol A) was also administered via intramuscular injection every 3 days in order to maintain adequate serum retinol levels (> 20 μg/ml). The administration of exogenous retinoic acid is known to have a sparing effect on hepatic retinol stores (24), leading to decreased serum retinol levels. Retinol deficiency in these animals was thereby prevented by retinol supplementation. The animals in the preterm control group were matched for both gestational and postnatal age, as well as maternal betamethasone treatment. The majority received daily vehicle injections (n=4), and the remainder (n=2) were untreated. Serum retinol concentrations were measured at birth, then at 7, 14 and 21 days postnatally.
Animal Euthanasia and Tissue Processing
At postnatal day 21, baboons were euthanized. Body weights at birth and at the time of necropsy were recorded. Kidneys were cleaned of fat and connective tissue, cut into quarters and immersion-fixed in 10% buffered formalin. Researchers were blinded to the treatment group of each kidney during analysis.
Fixed kidney quarters were initially weighed then sliced into 2 mm slices using a razor blade device. Every second slice was then embedded in glycolmethacrylate (25) to be used in for the estimation of kidney volume and nephron number.
Estimation of Kidney Volume, Renal Corpuscle Volume and Nephron Number
Glycolmethacrylate blocks were serially sectioned at 20 μm with every 10th and 11th section collected and stained with haemotoxylin and eosin. Kidney volume was then estimated using the Cavalieri principle (26).
In each kidney, the number of glomeruli (thereby the number of nephrons) and renal corpuscle volume were estimated using an unbiased physical disector/fractionator technique (27, 28).
Determination of Glomerular Generation Number
Medullary ray glomerular generations are the number of glomeruli formed in succession from the corticomedullary junction (first to be formed) to the outer renal cortex (last to be formed). One complete section from every kidney block was used to count the number of glomeruli per ray (Figure 1), in five clearly distinguishable medullary rays per section (approximately 40 rays per kidney). An average number of glomerular generations for each kidney were then determined. This method is considered to be highly reproducible (29), and has previously been utilized in the study of nephrogenesis in preterm human neonates (5, 30).
Figure 1.
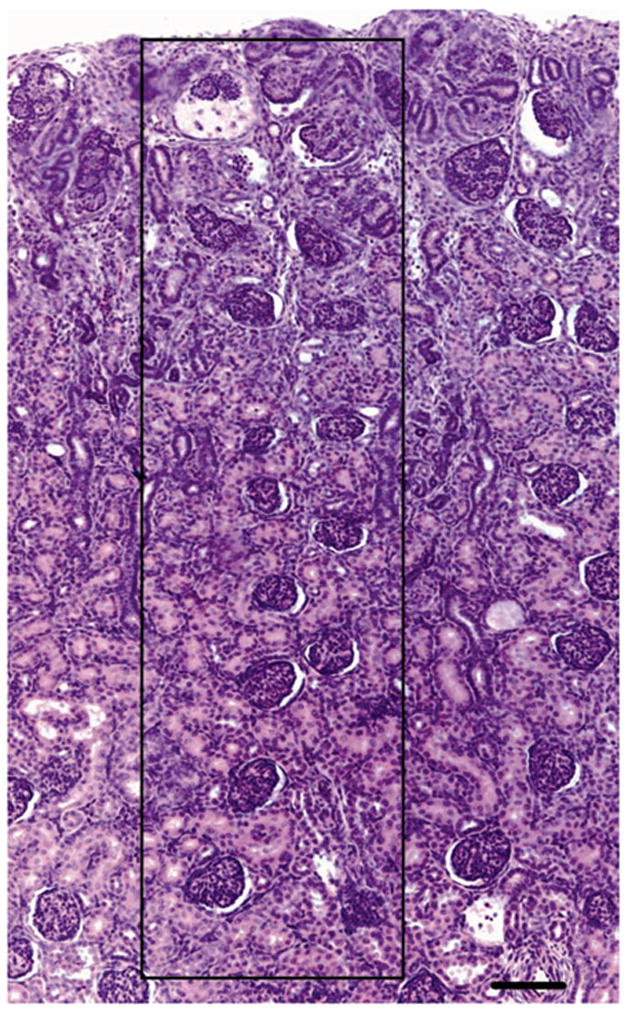
Photomicrograph of a neonatal baboon kidney indicating the glomerular generation counting method. Mature glomeruli were counted in a straight line along one side of a clearly distinguishable medullary ray (indicated by the black box) from the corticomedullary junction to the outer renal cortex. Scale bar = 100 μm.
Determination of the Percentage of Abnormal Glomeruli
A number of glomeruli situated in the outer renal cortex of several kidney sections were of abnormal appearance, with a dilated Bowman’s space and shrunken glomerular tuft. In every 10th section, previously used in the estimation of nephron number, the number of normal and abnormal glomeruli was recorded in each of the sampled fields of view. The percentage of abnormal glomeruli was then calculated.
Immunohistochemical Localization of the Endothelial Cell Marker, CD31 and the Podocyte Marker, Wilms Tumor Suppressor Gene 1 (WT-1)
4 μm paraffin sections, from both the preterm control and preterm +RA animals, were de-parrafinized and rehydrated, then underwent heat-induced antigen retrieval in Sodium Citrate buffer (10mM sodium citrate, pH6.0) for CD31 localization, and Tris-EDTA buffer (10mM Tris Base, 1mM EDTA, 0.05% Tween 20, pH 9.0) for the localization of WT-1. Endogenous peroxidase activity was blocked for 15 minutes with endogenous enzyme block solution (Dako, CA, USA) before incubation with 1% goat serum for 30 minutes. Sections were incubated overnight at room temperature with a monoclonal mouse anti-human WT-1 at 1:100 dilution, or CD31 antibody (Dako, CA, USA). The sections were subsequently incubated for 1 hour with the ‘Envision’ molecule (Dako, CA, USA), and 3′3′-diaminobenzidine tetrachloride (DAB) was used to detect antibody binding. All sections were counterstained with haematoxylin.
In-situ Hybridization of Vascular Endothelial Growth Factor (VEGF)
Synthesis of Riboprobes
A cDNA fragment of human VEGF121 (gift of Steven Stacker; Ludwig Institute, Melbourne, Australia) was cloned into BSKS plasmid (Stratagene, CA, USA) and linearized with HindIII. An anti-sense riboprobe was generated from the template incorporating digoxigenin (DIG)-UTP (Roche Applied Science, Mannheim, Germany) into run off transcripts using T7 RNA polymerase. A sense riboprobe was also generated.
In-situ Hybridization
In-situ hybridization of VEGF was performed on 4μm paraffin sections from both preterm control and preterm +RA animals. Paraffin sections were de-parafinized and rehydrated before being microwaved in 0.01M citrate buffer, pH 6.0, for 12 minutes and post-fixed in 1% formaldehyde in PBS. The sections were incubated in proteinase K (Roche Applied Science, Mannheim, Germany) for 20 minutes at 37°C, then prehybridized for one hour in hybridisation solution (50% deionised formamide, 5X SSC, 0.02% SDS, 0.1% N-laurosarcosine, DEPC H2O, 10% blocking reagent (Roche Applied Science, Mannheim, Germany). Sections were then hybridised overnight at 50°C with DIG-labelled VEGF antisense and sense riboprobes at 1/10 dilution in hybridisation buffer. Slides were then washed in 2X SSC, and treated with RNAse (150μg/mL in 2X SSC) for 30 minutes at 37°C. Following RNAse treatment, slides were washed in 1X SSC, 0.1X SSC, then maleic acid buffer (0.1M maleic acid, 0.15M NaCl; pH 7.5) for 5 minutes at room temperature. Sections were incubated in 10% blocking reagent (Boehringer, Mannheim, Germany) for 30 minutes, followed by 60 minutes incubation with 1/500 anti-DIG-AP, Fab fragments (Roche Applied Science, Mannheim, Germany) in blocking reagent. Sections were incubated overnight at room temperature with BCIP/NBT chromagen substrate (DakoCytomation, CA, USA), then coverslipped using Clearmount mounting medium (Zymed, CA, USA).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism Version 4.03 for Windows (GraphPad Software, CA, USA). An unpaired student’s t-test was used to compare data from the preterm control and preterm +RA groups. Linear regression analyses were performed to determine whether there were any significant correlations between birth/necropsy weight and kidney weight, nephron number and kidney weight/volume and renal corpuscle volume. An analysis of covariance (ANCOVA) was also performed to determine whether there were any significant differences in the linear regression of nephron number versus kidney volume and kidney weight between the preterm control and preterm +RA groups. Statistical significance was accepted as p < 0.05.
Results
Birth Weight, Necropsy Weight, and Kidney Weight
There was no significant difference between the preterm control and preterm +RA groups in body weights at birth or at the time of necropsy. There was also no significant difference in the weights of the fixed kidneys (Table 1).
Table 1.
Mean birth and necropsy weights, kidney weights and volumes, renal corpuscle volumes and nephron density data of the preterm control and preterm +RA groups.*
| Preterm Control (n=6) | Preterm +RA (n=6) | |
|---|---|---|
| Birth Weight (g) | 391.8 ± 20.6 | 382.7 ± 16.7 |
| Necropsy Weight (g) | 372.8 ± 24.8 | 383.3 ± 27.1 |
| Kidney Weight (g) | 1.85 ± 0.26 | 2.07 ± 0.30 |
| Kidney Volume (mm3) | 1156 ± 164 | 1229 ± 178 |
| Renal Corpuscle Volume (×10−4 mm3) | 2.60 ± 0.23 | 2.42 ± 0.17 |
| Nephron Density (glomeruli/mm3) | 298.8 ± 28.2 | 322.6 ± 23.6 |
Data is expressed as the mean ± SEM. There was no significant difference between groups in any parameter.
Linear regression analyses revealed a significant correlation between birth weight and kidney weight in the preterm control group (R2 = 0.771, p = 0.02). This association, however, was not evident in the preterm +RA group (R2 = 0.342, p = 0.22). Similarly, there was no significant correlation between necropsy weight and kidney weight in the preterm +RA group (R2 = 0.080, p = 0.59). The association was nearing significance, however, in the preterm controls (R2 = 0.627, p = 0.06).
Clinical Measurements
There were no statistically significant differences between the preterm control and preterm +RA animals in arterial blood gas pressure (PO2, PCO2), blood pressure, fluid intake or urine output at postnatal day 21 (Table 2).
Table 2.
Measurements of blood pressure, arterial blood gas pressures, fluid intake and urine output in preterm control and preterm +RA animals at postnatal day 21.*
| Preterm Control (n=6) | Preterm +RA (n=6) | |
|---|---|---|
| Systolic Blood Pressure (mmHg) | 53 ± 4 | 56 ± 13 |
| Diastolic Blood Pressure (mmHg) | 21 ± 4 | 25 ± 6 |
| Mean Blood Pressure (mmHg) | 33 ± 2 | 35 ± 8 |
| PO2 (mmHg) | 67.0 ± 5.1 | 77.2 ± 11.7 |
| PCO2 (mmHg) | 51.2 ± 1.0 | 44.2 ± 3.7 |
| Fluid Intake (ml/kg/day) | 167.8 ± 5.2 | 181.4 ± 5.6 |
| Urine Output (ml/kg/hour) | 3.1 ± 0.5 | 2.5 ± 0.8 |
Data is expressed as the mean ± SEM. There was no significant difference between groups in any parameter.
The serum retinol concentration of the preterm control (n=3) and preterm +RA (n=6) neonatal baboons did not differ significantly at birth, 7 days, or 21 days. There was, however, a significantly higher mean serum retinol concentration in animals in the preterm control group at postnatal day 14 (preterm control: 45.733 ± 5.385 mg/100ml; preterm +RA: 23.700 ± 3.071 mg/100ml; p = 0.01).
Kidney Volume and Renal Corpuscle Volume
Kidney volumes and renal corpuscle volumes are shown in Table 1. There was no significant difference between the two treatment groups in either parameter.
Nephron Number and Density
Average nephron number for the preterm control and preterm +RA groups is shown in Figure 2. There was no significant difference between two treatment groups. Similarly, there were no significant differences in nephron density (number of glomeruli per unit volume of kidney), as shown in Table 1.
Figure 2.
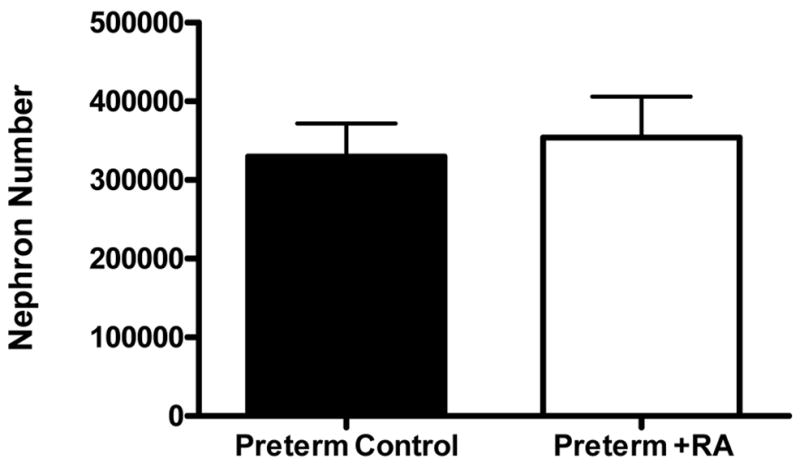
Bar graph depicting the average nephron number (± SEM) of preterm control (n=6) and preterm +RA (n=6) animals.
There was a significant correlation between kidney volume and nephron number within both the preterm control and preterm +RA groups, as depicted in Figure 3. Furthermore, a comparison between the two regression lines (ANCOVA) revealed no significant difference between the slope values, and predicted an overall increase of 236 nephrons per 1 mm3 increase in kidney volume across the pooled data.
Figure 3.
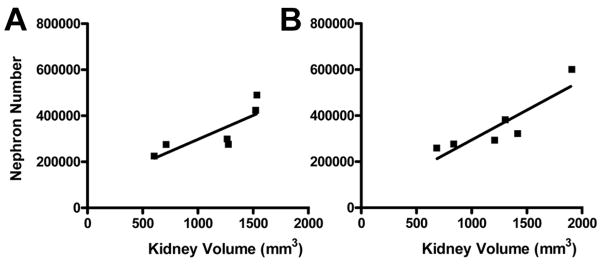
Linear regression analyses of nephron number versus kidney volume in A preterm control (R2 = 0.665, p = 0.047), and B preterm +RA (R2 = 0.785, p = 0.019) animals.
Correlations between kidney weight and nephron number within the preterm control (R2 = 0.613, p = 0.07) and the preterm +RA (R2 = 0.611, p = 0.07) treatment groups did not quite reach statistical significance. Since an ANCOVA analysis revealed no significant difference between the slope values of the two linear regressions, data from the two treatment groups were pooled and subsequently revealed a highly significant correlation between nephron number and kidney weight (R2 = 0.672, p = 0.001) across all preterm animals.
Further linear regression analyses revealed no significant correlations between body weight and nephron number or nephron number and renal corpuscle volume.
Glomerular Generations
There was no significant difference in the number of glomerular generations formed within the kidneys of the preterm control or preterm +RA animals (preterm control: 10.04 ± 0.26; preterm +RA: 9.99 ± 0.48; p = 0.91). The average number of glomerular generations per medullary ray was similar in all kidneys studied.
Abnormal Glomeruli
Abnormal glomeruli were classified as those with cystic dilation of the Bowman’s space. These glomeruli also commonly exhibited a shrunken glomerular tuft, as depicted in Figures 4, 5 and 6(C and D). Importantly, the abnormal glomeruli appeared only in the outer renal cortex of the kidney, whereas glomeruli located in the inner cortex of the same kidneys appeared to be normal.
Figure 4.
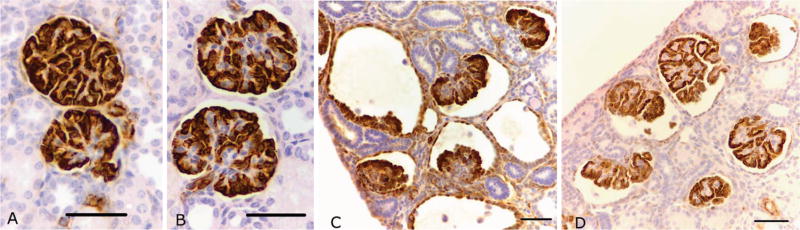
Representative photomicrographs of WT-1 immunostaining of podocytes in normal and abnormal glomeruli from both preterm control (A normal; C abnormal) and preterm +RA animals (B normal; D abnormal). Scale bar = 50 μm.
Figure 5.
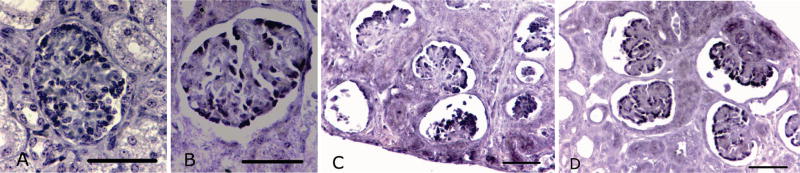
Representative photomicrographs of in-situ hybridization of VEGF expression (dark purple stained nuclei) in podocytes of normal and abnormal glomeruli from preterm control (A normal; C abnormal) and preterm +RA animals (B normal; D abnormal). Scale bar = 50 μm.
Figure 6.
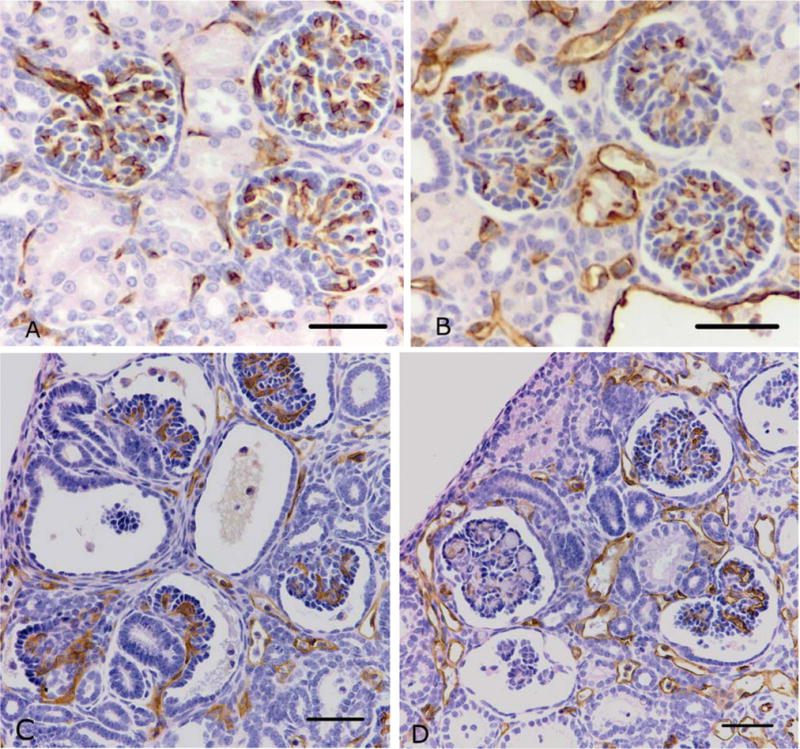
Representative photomicrographs of CD31 immunostaining of endothelial cells in normal and abnormal glomeruli from preterm control (A normal; C abnormal) and preterm +RA animals (B normal; D abnormal). Scale bar = 50 μm.
There was a wide range in the proportion of abnormal glomeruli in the kidneys of the preterm control (0.7–13.9%) and the preterm +RA (0.0–16.0%) animals. There was no significant difference in the proportion of abnormal glomeruli between the two treatment groups (preterm control: 3.54 ± 2.10%; preterm +RA: 5.83 ± 2.62%; p = 0.51). As shown in Table 3, there was no clear link between the administration of any of the postnatal medications and the percentage of abnormal glomeruli.
Table 3.
Medications administered postnatally to individual preterm control and preterm +RA animals, as compared to the percentage of abnormal glomeruli.
| Group | % Abnormal | Ampicillin | Gentamicin | Vancomycin | Fortaz | Indomethacin | Caffeine | Dopamine | Dobutamine | Hydrocortisone | Fluconazole | Cancidas | Nystatin |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Preterm Control | 13.93 | ✓ | ✓ | ✓ | ✓ | ✓ | |||||||
| 1.83 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| 1.04 | ✓ | ✓ | ✓ | ✓ | |||||||||
| 1.14 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||
| 0.73 | ✓ | ✓ | ✓ | ✓ | |||||||||
| 2.56 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| Preterm +RA | 4.12 | ✓ | ✓ | ✓ | ✓ | ||||||||
| 11.29 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| 16.03 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| 0.55 | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||||
| 0.00 | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ✓ | ||||||
| 2.99 | ✓ | ✓ | ✓ | ✓ | ✓ |
Immunohistochemical Localization and In-situ Hybridization
Podocyte marker WT-1
WT-1 immunostaining was localized to visceral and parietal podocytes in both normal and abnormal glomeruli from the preterm control and preterm +RA animals (Figure 4). In the abnormal glomeruli, podocytes were localized to a crescent of cells lining the glomerular tuft, indicating that the glomeruli are at an immature stage of development (31). Glomeruli that were grossly abnormal exhibited a glomerular tuft that was composed primarily of podocytes.
Vascular endothelial growth factor (VEGF)
In-situ hybridization of VEGF mRNA indicated that VEGF was expressed by the podocytes in all normal and abnormal glomeruli from the preterm control and preterm +RA animals (Figure 5).
Endothelial cell marker CD31
CD31 immunostaining was localized to the capillaries within the glomerular tuft of normal and abnormal glomeruli, in both the preterm control and preterm +RA animals (Figure 6). Glomeruli that were grossly abnormal lacked CD31 immunostaining indicating that they were not capillarized.
Discussion
The results of this study indicate that early postnatal administration of retinoic acid does not stimulate nephrogenesis in the kidney of the preterm baboon. Furthermore, retinoic acid treatment was shown to have no influence on postnatal growth or kidney size. It was observed, however, that a number of glomeruli located in the outer renal cortex of the preterm baboon kidney were of abnormal appearance, suggesting that nephrons formed in the extrauterine environment are at risk of impairment.
Contrary to our initial hypothesis, the results demonstrate that daily retinoic acid administration (500 μg/kg/day) has no significant effect on nephron number, the number of glomerular generations, kidney volume and weight, renal corpuscle volume, and the proportion of abnormal glomeruli formed. The lack of effect of retinoic acid administration on nephrogenesis in the present study may relate to the timing of administration. Retinoic acid administered during mid-gestation has been found to have a strong influence on branching morphogenesis in kidney development (11, 12), as it was administered at a time when the ureteric bud was undergoing its initial levels of branching. Conversely, in the present study retinoic acid was administered to the prematurely delivered baboons at a time point consistent with 26 weeks of gestation onwards, where up to 10 generations of glomeruli have already formed. In the human kidney, branching occurs only very rarely after the 22nd week of gestation (32) as the ampullae of the ureteric buds seldom divide and new nephrons are formed in an arcade pattern, attached to single ampullae (3, 32). Hence, nephrogenesis in the postnatal preterm kidney may not have been receptive to the influence of retinoic acid at this time. Taken together with findings from previous studies, the results suggest than an adequate maternal supply of vitamin A during gestation is of more importance in achieving optimal nephron endowment after premature birth than is the influence of postnatal supplementation.
In the current study there was a significant linear correlation between nephron number and both kidney weight and kidney volume across all preterm baboons, as has been shown previously (22). Therefore, the total number of nephrons formed in these animals is strongly linked to individual kidney size. In this regard, kidney size was not influenced by retinoic acid treatment and nor was nephron number. Nephron number in the preterm baboon kidney ranged between 273,697 and 599,320 which is consistent with previously published findings in the baboon (22), albeit in the higher range.
Furthermore, the number of glomerular generations was found to be very similar across all animals, with approximately 10 generations of glomeruli formed in the prematurely delivered baboon kidney by postnatal day 21 of development.
Although a nephron deficit as such has not been exhibited following preterm birth, a reduction in the functionality of the kidney is likely. In the current study, it was noted that a number of glomeruli exhibiting a dilated Bowman’s space and shrunken glomerular tuft were present in the outer renal cortex of kidneys from both the preterm groups. The overall proportion of abnormal glomeruli ranged from 0% to 16%, and these were seen to be situated exclusively in the outer renal cortex of the kidney, suggesting that only those glomeruli formed in the extrauterine environment are at risk of impairment. In support of this concept, our laboratory has recently shown that the number of abnormal glomeruli in the kidneys of term and gestational control animals is negligible (Gubhaju L et al. The effects of pre-term birth on nephrogenesis, 32nd Annual FNPS Meeting, September 25–28, 2005, Glenelg, Australia, Abstract O8), indicating that the glomerular abnormalities have occurred as a direct result of premature birth and/or its associated complications. Through immunohistochemical and in-situ hybridization analysis, we have determined that VEGF, the major angiogenic growth factor for glomerular capillaries, is expressed by the podocytes of the abnormal glomeruli and there is some evidence of capillarization of the tuft. In the grossly abnormal glomeruli, however, endothelial cell staining was negative suggesting that they are non-functional.
However, the cause of the abnormal glomerular morphology is, at the present time, unknown. In this regard, there was no link between the proportion of abnormal glomeruli in the preterm kidney and any irregularities in clinical parameters, such as PO2 and PCO2 levels. Furthermore, there was no apparent link between the percentage of abnormal glomeruli and medications administered postnatally (Table 3). Although medications such as aminoglycoside antibiotics and non-steroidal anti-inflammatory drugs have been reported to lead to renal injury in experimental studies (33–35) and impaired renal function in human neonates (36), there is no evidence to suggest that this was the cause of the abnormal glomeruli in the current study. All animals were routinely administered gentamicin for the first 7–10 days of life, and three animals received indomethacin; however, only one of these three neonates had a high proportion of abnormal glomeruli.
In conclusion, the results of this study indicate that early postnatal administration of retinoic acid is unable to enhance nephron endowment in the kidney of the preterm baboon, and this lack of effect is likely to be due to the timing of administration. As vitamin A supplementation is already being trialed in humans, however, it was encouraging to note that postnatal administration of retinoic acid and retinol does not appear to have an adverse effect on kidney development.
Acknowledgments
This work was supported by NIH HL52636 BPD Resource Center grant, P51RR13986 for facility support, the Vanderbilt Newborn Research Fund, and the National Health and Medical Research Council of Australia.
We wish to thank the Southwest Foundation for Biomedical Research (San Antonio, TX, USA) and in particular Professor Jacqueline Coalson and Ms. Vicki Winter from the University of Texas Health Science Center for their assistance in obtaining the baboon kidneys. We would also like to thank Associate Professor Darren Kelly and Ms. Alison Cox, from the Department of Medicine, St. Vincent’s Hospital, Melbourne, for undertaking the in-situ hybridization.
Abbreviations
- RA
Retinoic Acid
References
- 1.Noble L. Developments in neonatal technology continue to improve infant outcomes. Pediatr Ann. 2003;32:595–603. doi: 10.3928/0090-4481-20030901-08. [DOI] [PubMed] [Google Scholar]
- 2.Slattery MM, Morrison JJ. Preterm Delivery. Lancet. 2002;360:1489–1497. doi: 10.1016/S0140-6736(02)11476-0. [DOI] [PubMed] [Google Scholar]
- 3.Saxen L. Organogenesis of the Kidney. Cambridge University Press; Cambridge: 1987. [Google Scholar]
- 4.Hinchliffe SA, Sargent PH, Howard CV, Chan YF, Van Velzen D. Human intrauterine renal growth expressed in absolute number of glomeruli assessed by the disector method and cavalieri principle. Lab Invest. 1991;64:777–784. [PubMed] [Google Scholar]
- 5.Rodriguez MM, Gomez AH, Abitbol CL, Chandar JJ, Duara S, Zilleruelo GE. Histomorphometric analysis of postnatal glomerulogenesis in extremely preterm infants. Pediatr Dev Pathol. 2004;7:17–25. doi: 10.1007/s10024-003-3029-2. [DOI] [PubMed] [Google Scholar]
- 6.Hoy WE, Hughson MD, Bertram JF, Douglas-Denton R, Amann K. Nephron number, hypertension, renal disease, and renal failure. J Am Soc Nephrol. 2005;16:2557–2564. doi: 10.1681/ASN.2005020172. [DOI] [PubMed] [Google Scholar]
- 7.Brenner BM, Mackenzie HS. Nephron mass as a risk factor for progression of renal disease. Kidney Int Suppl. 1997;63:S124–S127. [PubMed] [Google Scholar]
- 8.Nenov VD, Taal MW, Sakharova OV, Brenner BM. Multi-hit nature of chronic renal disease. Curr Opin Nephrol Hypertens. 2000;9:85–97. doi: 10.1097/00041552-200003000-00001. [DOI] [PubMed] [Google Scholar]
- 9.Moreau E, Vilar J, Lelievre-Pegorier M, Merlet-Benichou C, Gilbert T. Regulation of c-ret expression by retinoic acid in rat metanephros: implication in nephron mass control. Am J Physiol. 1998;275:F938–F945. doi: 10.1152/ajprenal.1998.275.6.F938. [DOI] [PubMed] [Google Scholar]
- 10.Vilar J, Gilbert T, Moreau E, Merlet-Benichou C. Metanephros organogenesis is highly stimulated by vitamin A derivatives in organ culture. Kidney Int. 1996;49:1478–1487. doi: 10.1038/ki.1996.208. [DOI] [PubMed] [Google Scholar]
- 11.Lelievre-Pegorier M, Vilar J, Ferrier M-L, Moreau E, Freund N, Gilbert T, Merlet-Benichou C. Mild vitamin A deficiency leads to inborn nephron deficit in the rat. Kidney Int. 1998;54:1455–1462. doi: 10.1046/j.1523-1755.1998.00151.x. [DOI] [PubMed] [Google Scholar]
- 12.Makrakis J, Zimanyi MA, Black MJ. Retinoic acid enhances nephron endowment in rats exposed to maternal protein restriction. Pediatr Nephrol. 2007;22:1861–1867. doi: 10.1007/s00467-007-0572-5. [DOI] [PubMed] [Google Scholar]
- 13.Kennedy KA, Stoll BJ, Ehrenkranz RA, Oh W, Wright LL, Stevenson DK, Lemons JA, Sowell A, Mele L, Tyson JE, Verter J. Vitamin A to prevent bronchopulmonary dysplasia in very-low-birth-weight infants: has the dose been too low? The NICHD Neonatal Research Network. Early Hum Dev. 1997;49:19–31. doi: 10.1016/s0378-3782(97)01869-0. [DOI] [PubMed] [Google Scholar]
- 14.Shenai JP, Chytil F, Jhaveri A, Stahlman MT. Plasma vitamin A and retinol-binding protein in premature and term neonates. J Pediatr. 1981;99:302–305. doi: 10.1016/s0022-3476(81)80484-2. [DOI] [PubMed] [Google Scholar]
- 15.Shenai JP, Chytil F, Stahlman MT. Liver vitamin A reserves of very low birth weight neonates. Pediatr Res. 1985;19:892–893. doi: 10.1203/00006450-198509000-00003. [DOI] [PubMed] [Google Scholar]
- 16.Darlow BA, Graham PJ. Vitamin A supplementation to prevent mortality and short and long-term morbidity in very low birthweight infants. Cochrane Database Syst Rev. 2007;4:CD000501. doi: 10.1002/14651858.CD000501.pub2. [DOI] [PubMed] [Google Scholar]
- 17.Massaro GD, Massaro D. Postnatal treatment with retinoic acid increases the number of pulmonary alveoli in rats. Am J Physiol. 1996;270:L305–L310. doi: 10.1152/ajplung.1996.270.2.L305. [DOI] [PubMed] [Google Scholar]
- 18.Massaro GD, Massaro D. Retinoic acid treatment abrogates elastase-induced pulmonary emphysema in rats. Nat Med. 1997;3:675–677. doi: 10.1038/nm0697-675. [DOI] [PubMed] [Google Scholar]
- 19.Massaro GD, Massaro D. Retinoic acid treatment partially rescues failed septation in rats and in mice. Am J Physiol Lung Cell Mol Physiol. 2000;278:L955–L960. doi: 10.1152/ajplung.2000.278.5.L955. [DOI] [PubMed] [Google Scholar]
- 20.Veness-Meehan KA, Pierce RA, Moats-Staats BM, Stiles AD. Retinoic acid attenuates O2-induced inhibition of lung septation. Am J Physiol Lung Cell Mol Physiol. 2002;283:L971–L980. doi: 10.1152/ajplung.00266.2001. [DOI] [PubMed] [Google Scholar]
- 21.Thomson MA, Yoder BA, Winter VT, Martin H, Catland D, Siler-Khodr TM, Coalson JJ. Treatment of immature baboons for 28 days with early nasal continuous positive airway pressure. Am J Respir Crit Care Med. 2004;169:1054–1062. doi: 10.1164/rccm.200309-1276OC. [DOI] [PubMed] [Google Scholar]
- 22.Gubhaju L, Black MJ. The baboon as a good model for studies of human kidney development. Pediatr Res. 2005;58:505–509. doi: 10.1203/01.PDR.0000179397.20862.73. [DOI] [PubMed] [Google Scholar]
- 23.Young TE, Mangum OB. A manual of drugs used in neonatal care. Acorn Publishing; Raleigh, N.C: 2002. Neofax. [Google Scholar]
- 24.Gerlach TH, Zile MH. Effect of retinoic acid and apo-RBP on serum retinol concentration in acute renal failure. FASEB J. 1991;5:86–92. doi: 10.1096/fasebj.5.1.1991596. [DOI] [PubMed] [Google Scholar]
- 25.Siewert-Delle A, Ljungman S. The impact of birth weight and gestational age on blood pressure in adult life: a population-based study of 49-year-old men. Am J Hypertens. 1998;11:946–953. doi: 10.1016/s0895-7061(98)00090-9. [DOI] [PubMed] [Google Scholar]
- 26.Gundersen HJ, Jensen EB. The efficiency of systematic sampling in stereology and its prediction. J Microsc. 1987;147:229–263. doi: 10.1111/j.1365-2818.1987.tb02837.x. [DOI] [PubMed] [Google Scholar]
- 27.Bertram JF. Counting in the kidney. Kidney Int. 2001;59:792–796. doi: 10.1046/j.1523-1755.2001.059002792.x. [DOI] [PubMed] [Google Scholar]
- 28.Black MJ, Briscoe TA, Constantinou M, Kett MM, Bertram JF. Is there an association between level of adult blood pressure and nephron number or renal filtration surface area? Kidney Int. 2004;65:582–588. doi: 10.1111/j.1523-1755.2004.00406.x. [DOI] [PubMed] [Google Scholar]
- 29.Hinchliffe SA, Sargent PH, Chan YF, van Velzen D, Howard CV, Hutton JL, Rushton DI. Medullary ray glomerular counting” as a method of assessment of human nephrogenesis. Pathol Res Pract. 1992;188:775–782. doi: 10.1016/S0344-0338(11)80177-9. [DOI] [PubMed] [Google Scholar]
- 30.Rodriguez MM, Gomez A, Abitbol C, Chandar J, Montane B, Zilleruelo G. Comparative renal histomorphometry: a case study of oligonephropathy of prematurity. Pediatr Nephrol. 2005;20:945–949. doi: 10.1007/s00467-004-1800-x. [DOI] [PubMed] [Google Scholar]
- 31.Vernier RL, Birch-Andersen A. Studies of the human fetal kidney. I. Development of the glomerulus. J Pediatr. 1962;60:754–768. doi: 10.1016/s0022-3476(62)80103-6. [DOI] [PubMed] [Google Scholar]
- 32.Osathanondh V, Potter E. Development of human kidney as shown by microdissection. III. Formation and interrelationship of collecting tubules and nephrons. Arch Pathol. 1963;76:290–302. [PubMed] [Google Scholar]
- 33.Kent AL, Maxwell LE, Koina ME, Falk MC, Willenborg D, Dahlstrom JE. Renal glomeruli and tubular injury following indomethacin, ibuprofen, and gentamicin exposure in a neonatal rat model. Pediatr Res. 2007;62:307–312. doi: 10.1203/PDR.0b013e318123f6e3. [DOI] [PubMed] [Google Scholar]
- 34.Gilbert T, Gaonach S, Moreau E, Merlet-Benichou C. Defect of nephrogenesis induced by gentamicin in rat metanephric organ culture. Lab Invest. 1994;70:656–666. [PubMed] [Google Scholar]
- 35.Cullen LA, Young RJ, Bertram JF. Studies on the effects of gentamicin on rat metanephric development in vitro. Nephrology. 2001;5:115–123. [Google Scholar]
- 36.Andreoli SP. Acute renal failure in the newborn. Semin Perinatol. 2004;28:112–123. doi: 10.1053/j.semperi.2003.11.003. [DOI] [PubMed] [Google Scholar]


