Abstract
The neurotransmitter acetylcholine (ACh) can regulate neuronal excitability throughout the nervous system by acting on both the cys-loop ligand-gated nicotinic ACh receptor channels (nAChRs) and the G protein-coupled muscarinic ACh receptors (mAChRs). The hippocampus is an important area in the brain for learning and memory, where both nAChRs and mAChRs are expressed. The primary cholinergic input to the hippocampus arises from the medial septum and diagonal band of Broca, the activation of which can activate both nAChRs and mAChRs in the hippocampus and regulate synaptic communication and induce oscillations that are thought to be important for cognitive function. Dysfunction in the hippocampal cholinergic system has been linked with cognitive deficits and a variety of neurological disorders and diseases, including Alzheimer's disease and schizophrenia. My lab has focused on the role of the nAChRs in regulating hippocampal function, from understanding the expression and functional properties of the various subtypes of nAChRs, and what role these receptors may be playing in regulating synaptic plasticity. Here, I will briefly review this work, and where we are going in our attempts to further understand the role of these receptors in learning and memory, as well as in disease and neuroprotection.
The hippocampus is critical for learning and memory [13, 14, 30, 63] and is an important site for cognitive dysfunction in a variety of neurodegenerative diseases including Alzheimer's disease (AD) [62]. The hippocampal formation is divided into four main subregions: the dentate gyrus, the hippocampal proper (including CA1, CA2, and CA3 regions), the subicular complex, and the entorhinal cortex (EC, including layers I–VI). The main cholinergic input to the hippocampus is from the medial septum and diagonal band of Broca (MSDB) [40, 41], which innervates both principal glutamatergic cells and inhibitory GABAergic interneurons. In addition, the stimulation of the cholinergic inputs to the hippocampus activates muscarinic acetylcho-line receptors (mAChRs) on astrocytes in the CA1 stratum oriens layer [3]. Besides the cholinergic input from the MSDB, there is also a significant GABAergic input, and the activation of the cholinergic and GABAergic inputs from the MSDB is known to initiate and sustain network oscillations (e.g., hippocampal theta rhythm) in vivo and in vitro [5, 19, 24, 41, 75, 76, 102]. Additionally, inputs to the hippocampus from the EC are thought to regulate hippocampal theta rhythm [5, 19].
Cholinergic receptors in the hippocampus—focus on nicotinic acetylcholine receptor channels
The activation of acetylcholine (ACh) release will exert its effect on a variety of different cys-loop ligand-gated nicotinic ACh receptor channels (nAChRs) and G protein-coupled mAChRs that are expressed on both neurons and nonneuronal cells. The nAChRs are permeable to cations, the activation of which depolarizes the cell and may induce electrical firing. Some nAChRs are also permeable to calcium ions [21], and this increase in cytoplasmic calcium levels can affect neurotransmitter release, signal transduction cascades, plasticity, cell survival, apoptosis, and gene transcription [8, 23, 61, 107, 108, 111].
A variety of different subtypes of G protein-coupled mAChRs have been shown to be expressed and regulate a variety of ionic conductances (both depolarizing and hyper-polarizing responses) and signal transduction cascades in hippocampal pyramidal cells, interneurons, and astrocytes [3, 24, 75, 76, 94, 131]. Presently, it is unclear how the activation of both mAChRs and nAChRs, working in concert, can modulate the oscillatory properties of neurons within the hippocampus. Understanding how cholinergic receptor signaling regulates hippocampal network activity is critical since dysregulation of normal oscillations may induce seizures [11, 27, 123], and cognitive deficits linked with AD [39].
Structure–function aspects of nAChRs and related cys-loop receptor proteins
The focus of my lab has been on understanding the properties of nAChRs in the rodent hippocampus, the activation of which is thought to be involved in regulating excitability, plasticity, and cognitive function [59, 67, 78, 100]. Thus far, at least nine different nAChR subunits are known to be expressed in rodent brain, resulting in multiple functional subtypes of nAChRs. In the hippocampus, the most prevalent subtypes of functional nAChRs that are expressed are comprised of the α7 and α4β2 subtypes [1, 2, 68, 71, 103, 116, 127]. The neuronal nAChRs are known to be differentially permeable to calcium [9, 21, 43, 105]. For example, activation of the α7-containing (but not the non-α7) nAChRs will elicit local changes in cytoplasmic calcium levels in interneurons [35, 36, 70] and astrocytes [107, 110, 126], and it is this calcium influx that is thought to underlie the role of α7 nAChRs in synaptic plasticity and memory processes. However, this influx of calcium can also have deleterious consequences by inducing neurotoxicity [44, 85, 96].
The nAChRs are in the superfamily of cys-loop receptors (Fig. 1), which also includes the acetylcholine-binding proteins (AChBPs), a soluble protein from mollusks and the marine annelid Capitella teleta [17, 91] that is analogous to the extracellular ligand-binding domain of the cys-loop receptors. The binding of ACh to the extracellular interface between two nAChR subunits induces channel opening [51] (Fig. 1). While the precise steps between ligand binding and channel gating are presently unknown, the Auerbach Lab has provided some of the most complete data and models on the stepwise interactions that may be occurring from ligand binding to channel gating in the adult-type mouse muscle nAChR. As recently reported [64], they are proposing a mechanism similar to that of glutamate ligand-gated ion channels whereby after the diffusion of agonist to the binding site (docking), the binding states undergo two conformational changes (catch and hold). The first conformational changes were estimated, using Φ value analysis [4], to be residues near the binding loops (A–D), including the capping of loop C. In addition, there are conformational changes near the C terminus of the M2 helix; it is uncertain if the changes in these two regions are coupled or are independent. Finally, there is an anticlockwise twist of the extracellular domain β sandwich, a downward motion of loop 2, an upward movement of the M2–M3 linker, and a tilting of the pore-lining M2 helix, which may result in channel gating.
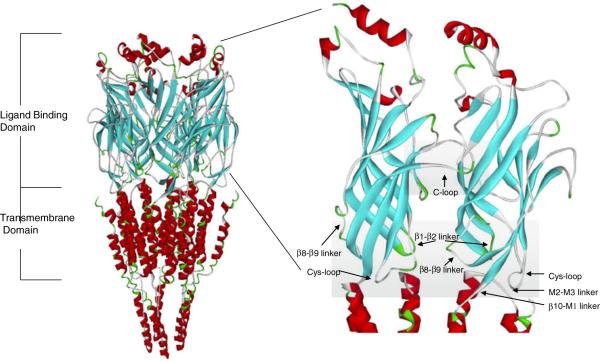
Fig. 1.
Molecular model of the rat α7 nAChR with ligand-binding domain. A side view of the pentameric α7 receptor model is shown on the left. The α-helices are shown in red and the β-strands in blue. The extracellular ligand-binding domain is shown up close on the right. The ligand-binding site is composed of a cluster of aromatic residues from both the principal and complementary subunits and is capped by the C-loop. The transition domain consists of several loops as shown
Some structural information has been obtained from the 4-Å resolution electron microscopic images of the Torpedo nAChR [125], higher resolution X-ray crystal structures of the AChBPs [12, 17, 22], and the extracellular domain of the α7 nAChR that has recently been crystallized [80]. Although the AChBPs do not contain an ion channel pore or intracellular domains, they bind nAChR ligands, and these crystal structures may help in elucidating the tertiary structure of the extracellular ligand-binding domain of nAChRs and the transitions that may occur in response to ligand binding. In addition, when AChBP was attached to the pore domain of the serotonin 5-HT3AR, ACh can activate the opening of this hybrid channel [15], further suggesting that AChBPs may be a good model system for studying nAChR gating.
The rat α7 nAChR can undergo rapid onset of desensitization, the process whereby the channel closes even in the continued presence of an agonist [53]. Although the mechanism of desensitization is not completely understood, it might be important in controlling cholinergic signaling and perhaps in certain nAChR-related diseases [34]. Furthermore, therapeutic drugs that potentiate α7 receptors through the removal of desensitization (e.g., through the use of type II α7-positive allosteric modulators such as PNU-120596) [10] are currently being developed to treat Alzheimer's disease and other neurological disorders [66, 96, 119, 133], highlighting the importance in understanding better the function of the α7 nAChRs.
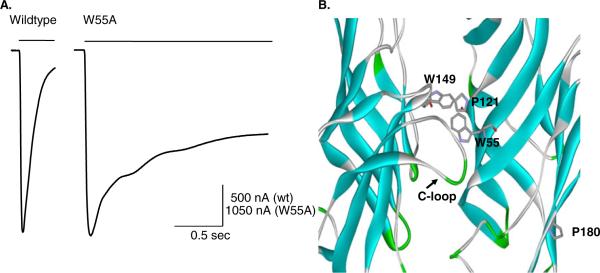
Previously, we found that the tryptophan residue at position 55 of the rat α7 nAChR (W55) was the site where synthetic peptides derived from apolipoprotein E noncompetitively inhibited α7 receptors through hydrophobic interactions [49]. Furthermore, when W55 was mutated to alanine, the receptor desensitized more slowly [50] (Fig. 2). We probed the accessibility of W55 by mutating it to cysteine and tested the ability of various sulfhydryl reagents to react with this cysteine. Modification with several positively charged sulfhydryl reagents, including 2-(trimethylammonium)ethyl methanethiosulfonate (MTSET+), produced α7 nAChRs that were unresponsive to ACh, whereas a neutral sulfhydryl reagent, methyl methanethiolsulfonate (MMTS), enhanced ACh-induced responses by nearly 60 %. These data suggested that W55 plays an important role in desensitization and suggested that W55 may be a potential target for modulatory agents operating via hydrophobic interactions.
Fig. 2.
The rate of α7 nAChR desensitization is altered by mutagenesis. Inward current responses (left) due to the rapid application of ACh for wild-type (left) and W55A mutant α7 receptor (right) expressed in Xenopus oocytes. Close up of the ligand-binding domain highlighting residues W55 and P180
However, these data left unresolved the question of how modification of W55 of the α7 nAChR with either MTSET+ or MMTS had two clearly distinct functional effects. To test this, we analyzed X-ray crystal structures of AChBP of the homologous Y53C mutant modified either with MTSET+ alone or MMTS in the presence of ACh [16]. Crystal structures of AChBP showed that MTSET+ modification stabilized loop C in an extended conformation that resembled the antagonist-bound state, which paralleled our observation that MTSET+ modification produced unresponsive W55C α7 nAChRs [50]. In contrast, the MMTS-modified mutant in complex with ACh was characterized by a contracted loop C, similar to other agonist-bound complexes. Surprisingly, we found that two ACh molecules were bound in the ligand-binding site, which might explain the potentiating effect of MMTS modification on W55C nAChRs. These results demonstrated that residue W55 of the α7 nAChR may be a critical regulatory site for channel gating and interactions with ligands of potential therapeutic benefit. In addition, our data show that there was a good correlation between structure and function of the nAChRs and AChBP and further suggest that crystal structures of the AChBP may yield valuable structural information into how ligand binding leads to channel opening and desensitization of the nAChRs.
The rat α7 nAChR has a proline residue near the middle of the β9 strand at position 180 (P180, Fig. 2b). The replacement of this proline residue by either threonine (α7-P180T) or serine (α7-P180S) also slowed the rate of onset of desensitization dramatically, similar to that of the W55A mutation [90]. We investigated the importance of the hydroxyl group on the position 180 side chains. While the hydroxyl group may contribute to the slow desensitization rates, our data suggested that increased backbone to backbone H bonding expected in the absence of proline at position 180 would likely exert a greater effect on desensitization. Molecular dynamics simulations were used to provide insight into likely H-bond interactions within the outer β-sheet that occur when the P180 residue is mutated. Our results indicate that rapid desensitization of the wild-type rat α7 nAChR is facilitated by the presence of the proline residue within the β9 strand.
Tobacco use remains the leading cause of preventable death in the USA, and the desire to use tobacco is thought to be due in part to the reinforcing actions of nicotine on the α4β2 nAChR subtype; therefore, ligands targeting this receptor (e.g., varenicline) may be useful for smoking cessation [12, 25]. To understand better how ligands bind into the pocket of the nAChRs, we crystallized AChBP in complex with varenicline [12] and found contact residues that are likely responsible for mediating its molecular actions. Electrophysiological studies confirmed these key molecular interactions, and this information should certainly help in the continual development of therapeutic drugs for smoking cessation with improved efficacy and reduced side effects.
Involvement of nAChRs in synaptic plasticity and oscillations
The nAChRs and mAChRs have both previously been associated with different forms of synaptic plasticity [24, 46, 65, 89, 92]. For the α7 nAChR, the activation of these receptors with exogenous ligands in the CA1 and dentate gyrus regions enhanced synaptic plasticity [45, 87, 129, 130]. Furthermore, activation of the α7 nAChRs on presynaptic terminals in the hippocampus can enhance the activation of long-term potentiation (LTP), which is thought to be a cellular form of learning and memory, and can block short-term potentiation (STP) and LTP in the pyramidal cells [65]. The activation of either pre- or postsynaptic mAChRs can either enhance or inhibit LTP in the hippocampus [18, 24, 77, 97, 104]. However, whether the activation of the endogenous cholinergic inputs to the hippocampus induces synaptic plasticity remains to be determined. The vast majority of prior knowledge has been derived from the use of exogenously applied receptor agonists or blockers, where critical information about the timing and context of neurotransmitter action is usually lacking [28, 54, 113]. For example, small shifts in the timing of the same glutamatergic input could result in either LTP or long-term depression (LTD) in the case of spike-timing-dependent plasticity [135]. In addition, previous studies have also shown that the timing of exogenously applied ACh is important in modulating high-frequency stimulation-induced hippocampal synaptic plasticity [52, 65], suggesting the potential capability of ACh to execute physiological functions with high temporal precision. Furthermore, the regulation of nAChRs in the hippocampus has also been linked to LTD [46, 52].
We investigated how the activation of the endogenous cholinergic inputs from the septum to the hippocampus [57], either electrically or through an optogenetic approach, could regulate hippocampal synaptic plasticity. We found that activation of the cholinergic input to the hippocampus with single electrical or light pulses can directly induce different forms of hippocampal synaptic plasticity with a timing precision in the millisecond range. When the cholinergic input to the CA1 hippocampal region was activated 100 ms prior to activation of the Schaffer collateral (SC) pathway, this induced an α7 nAChR-dependent LTP; this was likely due to the prolongation of the NMDAR-mediated calcium transients in the CA1 pyramidal cell spines, and synaptic insertion of the GluR2-containing AMPARs into these spines. When the cholinergic input was activated only 10 ms (rather than 100 ms) prior to the SC pathway, this induced an α7 nAChR-dependent short-term depression (STD) that was likely mediated in part through the presynaptic inhibition of glutamate release. Both of these forms of plasticity were absent if either the α7 receptors were blocked or, in mice, the α7 receptor has been knocked out (α7-KO mice). If however the cholinergic input was activated 10 ms after the SC pathway, this induced a mAChR-dependent LTP. Therefore, altering the timing of activation of the septal cholinergic input to the hippocampus induced three different forms of plasticity that depended solely on the timing of the input relative to the stimulation of the SC pathway.
Genetically encoded calcium indicators (GECIs) have recently been developed that have provided the ability to directly monitor neuronal activities (by measuring changes in cytoplasmic calcium levels) at either the synapse or network level. Furthermore, differently colored GECIs have provided excellent tools to monitor presynaptic and postsynaptic components at the same time facilitating our understanding of the coordinated activities that mediate synaptic plasticity [120, 121, 136]. Synaptic plasticity is known to be best induced when both pre- and postsynaptic activities are coordinated, as in spike-timing-dependent plasticity [7, 33, 37, 86, 88, 135]. However, little is known about the physiological events that could mediate such coordination, and yet it is fundamental to our understanding of neuronal communication and synaptic plasticity. Cholinergic receptors, as with other modulatory neurotransmitter receptors, are localized to both pre- and postsynaptic sites and thus provide a potential mechanism to coordinate preand postsynaptic activities to induce synaptic plasticity.
We used a septo-hippocampal coculture system [38, 47, 48, 101] with GECIs to monitor the pre- and postsynaptic activities of hippocampal SC to CA1 synapses during the α7 nAChR-dependent LTP and STD protocols [56]. During the LTP, we observed a prolonged enhancement of the SC-induced calcium responses both post- and presynaptically, while during the STD, we observed a short-term depression of the calcium responses both pre- and postsynaptically. Next, we found that the presence of the α7 nAChRs to both pre- and postsynaptic sites appeared to be required to induce LTP; the presence of α7 nAChRs at either pre- or postsynaptic sites alone induced only short-term potentiation of presynaptic or postsynaptic activities, respectively. Furthermore, for STD, α7 nAChRs were also required both pre- and postsynaptically. In α7-KO mice, both the α7 receptor-dependent LTP and STD were absent. Dual-color calcium imaging revealed a differential time course and pattern of post- versus presynaptic modulation during both LTP and STD, suggesting the existence of independent postsynaptic modulatory mechanisms. We propose that α7 nAChRs appear to be able to coordinate pre- and postsynaptic activities to induce glutamatergic synaptic plasticity and thus provide a novel mechanism underlying physiological neuronal communication that could lead to timing-dependent synaptic plasticity [56].
Although nicotine has been known to enhance cognitive function for decades, the mechanisms whereby cholinergic receptor activation can influence hippocampal neuronal networks are not understood. To gain more insights into which regions in the hippocampal complex are responsible for the initiation and spreading of information involving cholinergic receptors during smoking, we utilized voltage-sensitive dye imaging techniques in combination with electrophysiological recordings to investigate spatial-temporal aspects of cholinergic responses in the hippocampus [122]. The bath application of nicotine (to emulate systematic administration during smoking), at a concentration comparable to that achieved through smoking (i.e., as low as 100 nM), depolarized neurons in the deep EC cortical layers (layer VI) via activation of the α4β2 nAChR subtype. We found that subicular neurons also contained functional non-α7 nAChRs that were activated by the bath-applied nicotine. Interestingly, both of these nAChR-expressing ECVI and Sb groups of neurons were primarily glutamatergic. Furthermore, when we recorded from ECVI neurons directly and evoked glutamatergic EPSCs (eEPSCs) to the ECVI neurons by stimulating the Sb near the CA1 region, nicotine (100 nM) enhanced synaptic transmission by enhancing the amplitude of these eEPSCs. This low dose of bath-applied nicotine also enhanced synaptic plasticity in the ECVI neurons since it was able to convert STP (that was induced by tetanus stimulation of the Sb) to LTP, suggesting that this nicotine-induced plasticity could help in understanding the procognitive effects of nicotine. Therefore, neurons in deep layers of the EC not only contain diverse subtypes of functional nAChRs but these neurons may also be important regulators of hippocampal excitability and plasticity during smoking.
The cholinergic and GABAergic inputs from the MSDB are known to initiate and sustain network oscillations (e.g., hippocampal theta rhythm) in vivo and in vitro [5, 19, 24, 41, 75, 76, 102]. Additionally, inputs to the hippocampus from the EC are thought to regulate hippocampal theta rhythm [5, 19]. Presently, it is unclear precisely how the activation of both mAChRs and nAChRs, working in concert, can modulate the oscillatory properties of neurons within the hippocampus. Understanding how cholinergic receptor signaling regulates hippocampal network activity is critical since dysregulation of normal oscillations may induce seizures [11, 27, 123] and cognitive deficits linked with AD [39].
Role of nAChRs in development
The different subtypes of neuronal nAChRs are known to be developmentally expressed in the rat brain [134] and (particularly for the α7 subtype) are known to have important roles in the development of the hippocampal circuitry. For example, α7 receptor signaling is thought to promote the maturation and survival [20] of adult-born neurons, and the critical transformation of GABAergic currents from excitatory in early development to inhibitory later in development was shown to be due to the α7 receptor-dependent change in chloride transporter levels [82]. Recently, it was found that glutamatergic synapse formation in the hippocampus was promoted by the α7 nAChRs [83]. In mice where the α7 receptors were knocked out (α7-KO), or their expression level reduced by RNA interference, glutamatergic synapse numbers were adversely affected, whereas the activation of endogenous α7 receptors increases the number of functional glutamatergic synapses [83].
Role of nAChRs in disease and neuroprotection
Cholinergic dysfunction has long been associated with the cognitive deficit in AD [6, 118]. Furthermore, deficits in cholinergic signaling, in particular in the hippocampus, produce an array of disorders in learning and memory and have been linked with a variety of neurological disorders and diseases, including Alzheimer's disease, schizophrenia, and epilepsy [29, 118]. It should be noted that while rodent models have been very useful in helping to understand the cholinergic system and that there is a high degree of sequence homology between human and rodent nAChRs, even small numbers of amino acid differences have been shown to alter functional properties of these receptors, and therefore caution must be taken [29, 115]
A clear pathological feature of AD is the presence of amyloid plaques comprised of the β-amyloid peptide (Aβ1–42 or Aβ peptide), and recent studies have suggested that the soluble oligomeric rather than the fibrillar form of Aβ causes synaptic and cognitive dysfunction in AD [58, 60, 84, 93, 106]. While Aβ has been shown by several labs to affect nAChR function, conflicting reports of inhibition or upregulation of various nAChR subtypes have been reported [32, 42, 55, 74, 99, 132]. Interestingly, in cultured rat basal forebrain cholinergic neurons, the α7-containing receptors, which appeared to be heteromeric α7β2 receptors [71, 95], were blocked by low concentrations of oligomeric Aβ1–42 [81].
Since hippocampal α7 nAChRs have previously been shown to have a critical role in working and reference memory [78, 79], and the cognitive deficits associated with AD may be related to dysfunction of the α7 nAChRs [31, 69, 72, 98], we tested whether the α7 nAChR-dependent LTP and STD that we had recently reported [57] were sensitive to exposure to the soluble oligomeric form of Aβ peptide. We found that both the LTP and STD were both completely blocked in slices preexposed to a low dose of Aβ [57]. These results suggest a possible mechanism whereby Aβ could impair cholinergic-related synaptic plasticity and potentially cognitive functions.
Much evidence indicates that the α7 nAChR subtype is participating in various mechanisms of neuroprotection and inflammation [31, 98, 109]. For example, the nicotine-mediated neuroprotection against either glutamate excitotoxicity or the Aβ peptide is thought to be acting through the α7 nAChR [26, 69, 73, 114, 117]. Furthermore, in the brain (and in particular the hippocampus), nicotine or ACh activation of the α7 nAChR blocked the release of proinflammatory cytokines [112, 124], while in the periphery, ACh activation of the α7 nAChR inhibits cytokine synthesis [128]. Therefore, the α7 nAChR may be an essential regulator of inflammation and further highlights the therapeutic potential of α7 nAChR-selective ligands.
Conclusion
The cholinergic neurotransmitter system is involved in regulating synaptic excitability and plasticity and clearly has a role in a variety of physiological functions including learning and memory. Furthermore, deficits in cholinergic signaling are directly linked with a variety of different neurological disorders and diseases, including Alzheimer's disease and schizophrenia. It is critical that we learn more about the basic biology of the cholinergic system and the brain circuitry involved, and new and emerging tools such as optogenetics (along with more classical tools of electro-physiology and imaging) are providing us with an unprecedented ability to investigate these roles. These are very exciting times in the study of the brain and neuromodulator systems, and it is very likely that new breakthroughs that will help in the mitigation and treatment of disease are just around the corner.
Acknowledgments
This work was supported by the Intramural Research Program of the NIH, National Institute of Environmental Health Sciences.
References
- 1.Alkondon M, Albuquerque EX. Diversity of nicotinic acetylcholine receptors in rat hippocampal neurons. I. Pharmacological and functional evidence for distinct structural subtypes. J Pharmacol Exp Ther. 1993;265(3):1455–1473. [PubMed] [Google Scholar]
- 2.Alkondon M, Albuquerque EX. The nicotinic acetylcholine receptor subtypes and their function in the hippocampus and cerebral cortex. Prog Brain Res. 2004;145:109–120. doi: 10.1016/S0079-6123(03)45007-3. doi:10.1016/S0079-6123(03)45007-3. [DOI] [PubMed] [Google Scholar]
- 3.Araque A, Martin ED, Perea G, Arellano JI, Buno W. Synaptically released acetylcholine evokes Ca2+ elevations in astrocytes in hippocampal slices. J Neurosci. 2002;22(7):2443–2450. doi: 10.1523/JNEUROSCI.22-07-02443.2002. doi:20026212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Auerbach A. The gating isomerization of neuromuscular acetylcholine receptors. J Physiol. 2010;588(Pt 4):573–586. doi: 10.1113/jphysiol.2009.182774. doi:10.1113/jphysiol.2009.182774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Barry C, Heys JG, Hasselmo ME. Possible role of acetylcholine in regulating spatial novelty effects on theta rhythm and grid cells. Front Neural Circuits. 2012;6:5. doi: 10.3389/fncir.2012.00005. doi:10.3389/fncir.2012.00005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bartus RT, Dean RL, 3rd, Beer B, Lippa AS. The cholinergic hypothesis of geriatric memory dysfunction. Science. 1982;217(4558):408–414. doi: 10.1126/science.7046051. [DOI] [PubMed] [Google Scholar]
- 7.Bell CC, Han VZ, Sugawara Y, Grant K. Synaptic plasticity in a cerebellum-like structure depends on temporal order. Nature. 1997;387(6630):278–281. doi: 10.1038/387278a0. doi:10.1038/387278a0. [DOI] [PubMed] [Google Scholar]
- 8.Berger F, Gage FH, Vijayaraghavan S. Nicotinic receptor-induced apoptotic cell death of hippocampal progenitor cells. J Neurosci. 1998;18(17):6871–6881. doi: 10.1523/JNEUROSCI.18-17-06871.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bertrand D, Galzi JL, Devillers-Thiery A, Bertrand S, Changeux JP. Mutations at two distinct sites within the channel domain M2 alter calcium permeability of neuronal alpha 7 nicotinic receptor. Proc Natl Acad Sci U S A. 1993;90(15):6971–6975. doi: 10.1073/pnas.90.15.6971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bertrand D, Gopalakrishnan M. Allosteric modulation of nicotinic acetylcholine receptors. Biochem Pharmacol. 2007;74(8):1155–1163. doi: 10.1016/j.bcp.2007.07.011. doi:10.1016/j.bcp.2007.07.011. [DOI] [PubMed] [Google Scholar]
- 11.Bertrand S, Weiland S, Berkovic SF, Steinlein OK, Bertrand D. Properties of neuronal nicotinic acetylcholine receptor mutants from humans suffering from autosomal dominant nocturnal frontal lobe epilepsy. Br J Pharmacol. 1998;125(4):751–760. doi: 10.1038/sj.bjp.0702154. doi:10.1038/sj.bjp.0702154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Billen B, Spurny R, Brams M, van Elk R, Valera-Kummer S, Yakel JL, Voets T, Bertrand D, Smit AB, Ulens C. Molecular actions of smoking cessation drugs at alpha4beta2 nicotinic receptors defined in crystal structures of a homologous binding protein. Proc Natl Acad Sci U S A. 2012;109(23):9173–9178. doi: 10.1073/pnas.1116397109. doi:10.1073/pnas.1116397109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Blozovski D. Deficits in passive avoidance learning in young rats following mecamylamine injections in the hippocampoentorhinal area. Exp Brain Res. 1983;50(2–3):442–448. doi: 10.1007/BF00239211. [DOI] [PubMed] [Google Scholar]
- 14.Blozovski D. Mediation of passive avoidance learning by nicotinic hippocampo-entorhinal components in young rats. Dev Psychobiol. 1985;18(4):355–366. doi: 10.1002/dev.420180408. doi:10.1002/dev.420180408. [DOI] [PubMed] [Google Scholar]
- 15.Bouzat C, Gumilar F, Spitzmaul G, Wang HL, Rayes D, Hansen SB, Taylor P, Sine SM. Coupling of agonist binding to channel gating in an ACh-binding protein linked to an ion channel. Nature. 2004;430(7002):896–900. doi: 10.1038/nature02753. doi:10.1038/nature02753. [DOI] [PubMed] [Google Scholar]
- 16.Brams M, Gay EA, Saez JC, Guskov A, van Elk R, van der Schors RC, Peigneur S, Tytgat J, Strelkov SV, Smit AB, Yakel JL, Ulens C. Crystal structures of a cysteine-modified mutant in loop D of acetylcholine-binding protein. J Biol Chem. 2011;286(6):4420–4428. doi: 10.1074/jbc.M110.188730. doi:10.1074/jbc.M110.188730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Brejc K, van Dijk WJ, Klaassen RV, Schuurmans M, van Der Oost J, Smit AB, Sixma TK. Crystal structure of an ACh-binding protein reveals the ligand-binding domain of nicotinic receptors. Nature. 2001;411(6835):269–276. doi: 10.1038/35077011. doi:10.1038/35077011. [DOI] [PubMed] [Google Scholar]
- 18.Buchanan KA, Petrovic MM, Chamberlain SE, Marrion NV, Mellor JR. Facilitation of long-term potentiation by muscarinic M(1) receptors is mediated by inhibition of SK channels. Neuron. 2010;68(5):948–963. doi: 10.1016/j.neuron.2010.11.018. doi:10.1016/j.neuron.2010.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Buzsaki G. Theta oscillations in the hippocampus. Neuron. 2002;33(3):325–340. doi: 10.1016/s0896-6273(02)00586-x. [DOI] [PubMed] [Google Scholar]
- 20.Campbell NR, Fernandes CC, Halff AW, Berg DK. Endogenous signaling through alpha7-containing nicotinic receptors promotes maturation and integration of adult-born neurons in the hippocampus. J Neurosci. 2010;30(26):8734–8744. doi: 10.1523/JNEUROSCI.0931-10.2010. doi:10.1523/JNEUROSCI.0931-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Castro NG, Albuquerque EX. Alpha-bungarotoxin-sensitive hippocampal nicotinic receptor channel has a high calcium permeability. Biophys J. 1995;68(2):516–524. doi: 10.1016/S0006-3495(95)80213-4. doi:10.1016/S0006-3495(95)80213-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Celie PH, van Rossum-Fikkert SE, van Dijk WJ, Brejc K, Smit AB, Sixma TK. Nicotine and carbamylcholine binding to nicotinic acetylcholine receptors as studied in AChBP crystal structures. Neuron. 2004;41(6):907–914. doi: 10.1016/s0896-6273(04)00115-1. [DOI] [PubMed] [Google Scholar]
- 23.Chang KT, Berg DK. Voltage-gated channels block nicotinic regulation of CREB phosphorylation and gene expression in neurons. Neuron. 2001;32(5):855–865. doi: 10.1016/s0896-6273(01)00516-5. [DOI] [PubMed] [Google Scholar]
- 24.Cobb SR, Davies CH. Cholinergic modulation of hippocampal cells and circuits. J Physiol. 2005;562(Pt 1):81–88. doi: 10.1113/jphysiol.2004.076539. doi:10.1113/jphysiol.2004.076539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Coe JW, Brooks PR, Vetelino MG, Wirtz MC, Arnold EP, Huang J, Sands SB, Davis TI, Lebel LA, Fox CB, Shrikhande A, Heym JH, Schaeffer E, Rollema H, Lu Y, Mansbach RS, Chambers LK, Rovetti CC, Schulz DW, Tingley FD, 3rd, O'Neill BT. Varenicline: an alpha4beta2 nicotinic receptor partial agonist for smoking cessation. J Med Chem. 2005;48(10):3474–3477. doi: 10.1021/jm050069n. doi:10.1021/jm050069n. [DOI] [PubMed] [Google Scholar]
- 26.Dajas-Bailador FA, Lima PA, Wonnacott S. The alpha7 nicotinic acetylcholine receptor subtype mediates nicotine protection against NMDA excitotoxicity in primary hippocampal cultures through a Ca(2+) dependent mechanism. Neuropharmacology. 2000;39(13):2799–2807. doi: 10.1016/s0028-3908(00)00127-1. [DOI] [PubMed] [Google Scholar]
- 27.Damaj MI, Glassco W, Dukat M, Martin BR. Pharmacological characterization of nicotine-induced seizures in mice. J Pharmacol Exp Ther. 1999;291(3):1284–1291. [PubMed] [Google Scholar]
- 28.Dan Y, Poo MM. Spike timing-dependent plasticity of neural circuits. Neuron. 2004;44(1):23–30. doi: 10.1016/j.neuron.2004.09.007. doi:10.1016/j.neuron.2004.09.007. [DOI] [PubMed] [Google Scholar]
- 29.Dani JA, Bertrand D. Nicotinic acetylcholine receptors and nicotinic cholinergic mechanisms of the central nervous system. Annu Rev Pharmacol Toxicol. 2007;47:699–729. doi: 10.1146/annurev.pharmtox.47.120505.105214. doi:10.1146/annurev.pharmtox.47.120505.105214. [DOI] [PubMed] [Google Scholar]
- 30.Davis JA, Kenney JW, Gould TJ. Hippocampal alpha4beta2 nicotinic acetylcholine receptor involvement in the enhancing effect of acute nicotine on contextual fear conditioning. J Neurosci. 2007;27(40):10870–10877. doi: 10.1523/JNEUROSCI.3242-07.2007. doi:10.1523/JNEUROSCI.3242-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dineley KT. Beta-amyloid peptide—nicotinic acetylcho-line receptor interaction: the two faces of health and disease. Front Biosci. 2007;12:5030–5038. doi: 10.2741/2445. [DOI] [PubMed] [Google Scholar]
- 32.Dougherty JJ, Wu J, Nichols RA. Beta-amyloid regulation of presynaptic nicotinic receptors in rat hippocampus and neo-cortex. J Neurosci. 2003;23(17):6740–6747. doi: 10.1523/JNEUROSCI.23-17-06740.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Egger V, Feldmeyer D, Sakmann B. Coincidence detection and changes of synaptic efficacy in spiny stellate neurons in rat barrel cortex. Nat Neurosci. 1999;2(12):1098–1105. doi: 10.1038/16026. doi:10.1038/16026. [DOI] [PubMed] [Google Scholar]
- 34.Fayuk D, Yakel JL. Regulation of nicotinic acetylcholine receptor channel function by acetylcholinesterase inhibitors in rat hippocampal CA1 interneurons. Mol Pharmacol. 2004;66(3):658–666. doi: 10.1124/mol.104.000042. doi:10.1124/mol.104.000042. [DOI] [PubMed] [Google Scholar]
- 35.Fayuk D, Yakel JL. Ca2+ permeability of nicotinic acetylcholine receptors in rat hippocampal CA1 interneurones. J Physiol. 2005;566(Pt 3):759–768. doi: 10.1113/jphysiol.2005.089789. doi:10.1113/jphysiol.2005.089789. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Fayuk D, Yakel JL. Dendritic Ca2+ signalling due to activation of alpha 7-containing nicotinic acetylcholine receptors in rat hippocampal neurons. J Physiol. 2007;582(Pt 2):597–611. doi: 10.1113/jphysiol.2007.135319. doi:10.1113/jphysiol.2007.135319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Feldman DE. Timing-based LTP and LTD at vertical inputs to layer II/III pyramidal cells in rat barrel cortex. Neuron. 2000;27(1):45–56. doi: 10.1016/s0896-6273(00)00008-8. [DOI] [PubMed] [Google Scholar]
- 38.Fischer Y, Gahwiler BH, Thompson SM. Activation of intrinsic hippocampal theta oscillations by acetylcholine in rat septo-hippocampal cocultures. J Physiol. 1999;519(Pt 2):405–413. doi: 10.1111/j.1469-7793.1999.0405m.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Fodale V, Quattrone D, Trecroci C, Caminiti V, Santamaria LB. Alzheimer's disease and anaesthesia: implications for the central cholinergic system. Br J Anaesth. 2006;97(4):445–452. doi: 10.1093/bja/ael233. doi:10.1093/bja/ael233. [DOI] [PubMed] [Google Scholar]
- 40.Frotscher M, Drakew A, Heimrich B. Role of afferent innervation and neuronal activity in dendritic development and spine maturation of fascia dentata granule cells. Cereb Cortex. 2000;10(10):946–951. doi: 10.1093/cercor/10.10.946. [DOI] [PubMed] [Google Scholar]
- 41.Frotscher M, Leranth C. Cholinergic innervation of the rat hippocampus as revealed by choline acetyltransferase immunocytochemistry: a combined light and electron microscopic study. J Comp Neurol. 1985;239(2):237–246. doi: 10.1002/cne.902390210. doi:10.1002/cne.902390210. [DOI] [PubMed] [Google Scholar]
- 42.Fu W, Jhamandas JH. Beta-amyloid peptide activates non-alpha7 nicotinic acetylcholine receptors in rat basal forebrain neurons. J Neurophysiol. 2003;90(5):3130–3136. doi: 10.1152/jn.00616.2003. doi:10.1152/jn.00616.2003. [DOI] [PubMed] [Google Scholar]
- 43.Fucile S. Ca2+ permeability of nicotinic acetylcholine receptors. Cell Calcium. 2004;35(1):1–8. doi: 10.1016/j.ceca.2003.08.006. [DOI] [PubMed] [Google Scholar]
- 44.Fucile S, Sucapane A, Grassi F, Eusebi F, Engel AG. The human adult subtype ACh receptor channel has high Ca2+ permeability and predisposes to endplate Ca2+ overloading. J Physiol. 2006;573(Pt 1):35–43. doi: 10.1113/jphysiol.2006.108092. doi:10.1113/jphysiol.2006.108092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Fujii S, Ji Z, Morita N, Sumikawa K. Acute and chronic nicotine exposure differentially facilitate the induction of LTP. Brain Res. 1999;846(1):137–143. doi: 10.1016/s0006-8993(99)01982-4. [DOI] [PubMed] [Google Scholar]
- 46.Fujii S, Sumikawa K. Nicotine accelerates reversal of long-term potentiation and enhances long-term depression in the rat hippocampal CA1 region. Brain Res. 2001;894(2):340–346. doi: 10.1016/s0006-8993(01)02058-3. [DOI] [PubMed] [Google Scholar]
- 47.Gahwiler BH, Brown DA. Functional innervation of cultured hippocampal neurones by cholinergic afferents from co-cultured septal explants. Nature. 1985;313(6003):577–579. doi: 10.1038/313577a0. [DOI] [PubMed] [Google Scholar]
- 48.Gahwiler BH, Hefti F. Guidance of acetylcholinesterase-containing fibres by target tissue in co-cultured brain slices. Neuroscience. 1984;13(3):681–689. doi: 10.1016/0306-4522(84)90088-5. [DOI] [PubMed] [Google Scholar]
- 49.Gay EA, Bienstock RJ, Lamb PW, Yakel JL. Structural determinates for apolipoprotein E-derived peptide interaction with the alpha7 nicotinic acetylcholine receptor. Mol Pharmacol. 2007;72(4):838–849. doi: 10.1124/mol.107.035527. doi:10.1124/mol.107.035527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gay EA, Giniatullin R, Skorinkin A, Yakel JL. Aromatic residues at position 55 of rat alpha7 nicotinic acetylcholine receptors are critical for maintaining rapid desensitization. J Physiol. 2008;586(4):1105–1115. doi: 10.1113/jphysiol.2007.149492. doi:10.1113/jphysiol.2007.149492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Gay EA, Yakel JL. Gating of nicotinic ACh receptors; new insights into structural transitions triggered by agonist binding that induce channel opening. J Physiol. 2007;584(Pt 3):727–733. doi: 10.1113/jphysiol.2007.142554. doi:10.1113/jphysiol.2007.142554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Ge S, Dani JA. Nicotinic acetylcholine receptors at glutamate synapses facilitate long-term depression or potentiation. J Neurosci. 2005;25(26):6084–6091. doi: 10.1523/JNEUROSCI.0542-05.2005. doi:10.1523/JNEUROSCI.0542-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Giniatullin R, Nistri A, Yakel JL. Desensitization of nicotinic ACh receptors: shaping cholinergic signaling. Trends Neurosci. 2005;28(7):371–378. doi: 10.1016/j.tins.2005.04.009. doi:10.1016/j.tins.2005.04.009. [DOI] [PubMed] [Google Scholar]
- 54.Gradinaru V, Zhang F, Ramakrishnan C, Mattis J, Prakash R, Diester I, Goshen I, Thompson KR, Deisseroth K. Molecular and cellular approaches for diversifying and extending optogenetics. Cell. 2010;141(1):154–165. doi: 10.1016/j.cell.2010.02.037. doi:10.1016/j.cell.2010.02.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Grassi F, Palma E, Tonini R, Amici M, Ballivet M, Eusebi F. Amyloid beta(1–42) peptide alters the gating of human and mouse alpha-bungarotoxin-sensitive nicotinic receptors. J Physiol. 2003;547(Pt 1):147–157. doi: 10.1113/jphysiol.2002.035436. doi:10.1113/jphysiol.2002.035436. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Gu Z, Lamb PW, Yakel JL. Cholinergic coordination of presynaptic and postsynaptic activity induces timing-dependent hippocampal synaptic plasticity. J Neurosci. 2012;32(36):12337–12348. doi: 10.1523/JNEUROSCI.2129-12.2012. doi:10.1523/JNEUROSCI.2129-12.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Gu Z, Yakel JL. Timing-dependent septal cholinergic induction of dynamic hippocampal synaptic plasticity. Neuron. 2011;71(1):155–165. doi: 10.1016/j.neuron.2011.04.026. doi:10.1016/j.neuron.2011.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Haass C, Selkoe DJ. Soluble protein oligomers in neuro-degeneration: lessons from the Alzheimer's amyloid beta-peptide. Nat Rev Mol Cell Biol. 2007;8(2):101–112. doi: 10.1038/nrm2101. doi:10.1038/nrm2101. [DOI] [PubMed] [Google Scholar]
- 59.Hasselmo ME. Neuromodulation and the hippocampus: memory function and dysfunction in a network simulation. Prog Brain Res. 1999;121:3–18. doi: 10.1016/s0079-6123(08)63064-2. [DOI] [PubMed] [Google Scholar]
- 60.Hsieh H, Boehm J, Sato C, Iwatsubo T, Tomita T, Sisodia S, Malinow R. AMPAR removal underlies Abeta-induced synaptic depression and dendritic spine loss. Neuron. 2006;52(5):831–843. doi: 10.1016/j.neuron.2006.10.035. doi:10.1016/j.neuron.2006.10.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Hu M, Liu QS, Chang KT, Berg DK. Nicotinic regulation of CREB activation in hippocampal neurons by glutamatergic and nonglutamatergic pathways. Mol Cell Neurosci. 2002;21(4):616–625. doi: 10.1006/mcne.2002.1202. [DOI] [PubMed] [Google Scholar]
- 62.Hurst R, Rollema H, Bertrand D. Nicotinic acetylcholine receptors: from basic science to therapeutics. Pharmacol Ther. 2012 doi: 10.1016/j.pharmthera.2012.08.012. doi:10.1016/j.pharmthera.2012.08.012. [DOI] [PubMed] [Google Scholar]
- 63.Izquierdo I, Cammarota M, Da Silva WC, Bevilaqua LR, Rossato JI, Bonini JS, Mello P, Benetti F, Costa JC, Medina JH. The evidence for hippocampal long-term potentiation as a basis of memory for simple tasks. An Acad Bras Cienc. 2008;80(1):115–127. doi: 10.1590/s0001-37652008000100007. [DOI] [PubMed] [Google Scholar]
- 64.Jadey S, Auerbach A. An integrated catch-and-hold mechanism activates nicotinic acetylcholine receptors. J Gen Physiol. 2012;140(1):17–28. doi: 10.1085/jgp.201210801. doi:10.1085/jgp.201210801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Ji D, Lape R, Dani JA. Timing and location of nicotinic activity enhances or depresses hippocampal synaptic plasticity. Neuron. 2001;31(1):131–141. doi: 10.1016/s0896-6273(01)00332-4. [DOI] [PubMed] [Google Scholar]
- 66.Johnstone TB, Gu Z, Yoshimura RF, Villegier AS, Hogenkamp DJ, Whittemore ER, Huang JC, Tran MB, Belluzzi JD, Yakel JL, Gee KW. Allosteric modulation of related ligand-gated ion channels synergistically induces long-term potentiation in the hippocampus and enhances cognition. J Pharmacol Exp Ther. 2011;336(3):908–915. doi: 10.1124/jpet.110.176255. doi:10.1124/jpet.110.176255. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Jones S, Sudweeks S, Yakel JL. Nicotinic receptors in the brain: correlating physiology with function. Trends Neurosci. 1999;22(12):555–561. doi: 10.1016/s0166-2236(99)01471-x. [DOI] [PubMed] [Google Scholar]
- 68.Jones S, Yakel JL. Functional nicotinic ACh receptors on interneurones in the rat hippocampus. J Physiol. 1997;504(Pt 3):603–610. doi: 10.1111/j.1469-7793.1997.603bd.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Jonnala RR, Buccafusco JJ. Relationship between the increased cell surface alpha7 nicotinic receptor expression and neuroprotection induced by several nicotinic receptor agonists. J Neurosci Res. 2001;66(4):565–572. doi: 10.1002/jnr.10022. [DOI] [PubMed] [Google Scholar]
- 70.Khiroug L, Giniatullin R, Klein RC, Fayuk D, Yakel JL. Functional mapping and Ca2+ regulation of nicotinic acetylcholine receptor channels in rat hippocampal CA1 neurons. J Neurosci. 2003;23(27):9024–9031. doi: 10.1523/JNEUROSCI.23-27-09024.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Khiroug SS, Harkness PC, Lamb PW, Sudweeks SN, Khiroug L, Millar NS, Yakel JL. Rat nicotinic ACh receptor alpha7 and beta2 subunits co-assemble to form functional heteromeric nicotinic receptor channels. J Physiol. 2002;540(Pt 2):425–434. doi: 10.1113/jphysiol.2001.013847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Kihara T, Shimohama S, Sawada H, Honda K, Nakamizo T, Shibasaki H, Kume T, Akaike A. Alpha 7 nicotinic receptor transduces signals to phosphatidylinositol 3-kinase to block A beta-amyloid-induced neurotoxicity. J Biol Chem. 2001;276(17):13541–13546. doi: 10.1074/jbc.M008035200. doi:10.1074/jbc.M008035200. [DOI] [PubMed] [Google Scholar]
- 73.Kihara T, Shimohama S, Sawada H, Kimura J, Kume T, Kochiyama H, Maeda T, Akaike A. Nicotinic receptor stimulation protects neurons against beta-amyloid toxicity. Ann Neurol. 1997;42(2):159–163. doi: 10.1002/ana.410420205. doi:10.1002/ana.410420205. [DOI] [PubMed] [Google Scholar]
- 74.Lamb PW, Melton MA, Yakel JL. Inhibition of neuronal nicotinic acetylcholine receptor channels expressed in Xenopus oocytes by beta-amyloid1-42 peptide. J Mol Neurosci. 2005;27(1):13–21. doi: 10.1385/JMN:27:1:013. doi:10.1385/JMN:27:1:013. [DOI] [PubMed] [Google Scholar]
- 75.Lawrence JJ, Grinspan ZM, Statland JM, McBain CJ. Muscarinic receptor activation tunes mouse stratum oriens inter-neurones to amplify spike reliability. J Physiol. 2006;571(Pt 3):555–562. doi: 10.1113/jphysiol.2005.103218. doi:10.1113/jphysiol.2005.103218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Lawrence JJ, Statland JM, Grinspan ZM, McBain CJ. Cell type-specific dependence of muscarinic signalling in mouse hippocampal stratum oriens interneurones. J Physiol. 2006;570(Pt 3):595–610. doi: 10.1113/jphysiol.2005.100875. doi:10.1113/jphysiol.2005.100875. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Leung LS, Shen B, Rajakumar N, Ma J. Cholinergic activity enhances hippocampal long-term potentiation in CA1 during walking in rats. J Neurosci. 2003;23(28):9297–9304. doi: 10.1523/JNEUROSCI.23-28-09297.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Levin ED. Nicotinic receptor subtypes and cognitive function. J Neurobiol. 2002;53(4):633–640. doi: 10.1002/neu.10151. doi:10.1002/neu.10151. [DOI] [PubMed] [Google Scholar]
- 79.Levin ED, Petro A, Rezvani AH, Pollard N, Christopher NC, Strauss M, Avery J, Nicholson J, Rose JE. Nicotinic alpha7- or beta2-containing receptor knockout: effects on radial-arm maze learning and long-term nicotine consumption in mice. Behav Brain Res. 2009;196(2):207–213. doi: 10.1016/j.bbr.2008.08.048. doi:10.1016/j.bbr.2008.08.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Li SX, Huang S, Bren N, Noridomi K, Dellisanti CD, Sine SM, Chen L. Ligand-binding domain of an alpha7-nicotinic receptor chimera and its complex with agonist. Nat Neurosci. 2011;14(10):1253–1259. doi: 10.1038/nn.2908. doi:10.1038/nn.2908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Liu Q, Huang Y, Xue F, Simard A, DeChon J, Li G, Zhang J, Lucero L, Wang M, Sierks M, Hu G, Chang Y, Lukas RJ, Wu J. A novel nicotinic acetylcholine receptor subtype in basal forebrain cholinergic neurons with high sensitivity to amyloid peptides. J Neurosci. 2009;29(4):918–929. doi: 10.1523/JNEUROSCI.3952-08.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Liu Z, Neff RA, Berg DK. Sequential interplay of nicotinic and GABAergic signaling guides neuronal development. Science. 2006;314(5805):1610–1613. doi: 10.1126/science.1134246. doi:10.1126/science.1134246. [DOI] [PubMed] [Google Scholar]
- 83.Lozada AF, Wang X, Gounko NV, Massey KA, Duan J, Liu Z, Berg DK. Glutamatergic synapse formation is promoted by alpha7-containing nicotinic acetylcholine receptors. J Neurosci. 2012;32(22):7651–7661. doi: 10.1523/JNEUROSCI.6246-11.2012. doi:10.1523/JNEUROSCI.6246-11.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Lue LF, Kuo YM, Roher AE, Brachova L, Shen Y, Sue L, Beach T, Kurth JH, Rydel RE, Rogers J. Soluble amyloid beta peptide concentration as a predictor of synaptic change in Alzheimer's disease. Am J Pathol. 1999;155(3):853–862. doi: 10.1016/s0002-9440(10)65184-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lukas RJ, Lucero L, Buisson B, Galzi JL, Puchacz E, Fryer JD, Changeux JP, Bertrand D. Neurotoxicity of channel mutations in heterologously expressed alpha7-nicotinic acetylcholine receptors. Eur J Neurosci. 2001;13(10):1849–1860. doi: 10.1046/j.0953-816x.2001.01560.x. [DOI] [PubMed] [Google Scholar]
- 86.Magee JC, Johnston D. A synaptically controlled, associative signal for Hebbian plasticity in hippocampal neurons. Science. 1997;275(5297):209–213. doi: 10.1126/science.275.5297.209. [DOI] [PubMed] [Google Scholar]
- 87.Mann EO, Greenfield SA. Novel modulatory mechanisms revealed by the sustained application of nicotine in the guinea-pig hippocampus in vitro. J Physiol. 2003;551(Pt 2):539–550. doi: 10.1113/jphysiol.2003.045492. doi:10.1113/jphysiol.2003.045492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Markram H, Lubke J, Frotscher M, Sakmann B. Regulation of synaptic efficacy by coincidence of postsynaptic APs and EPSPs. Science. 1997;275(5297):213–215. doi: 10.1126/science.275.5297.213. [DOI] [PubMed] [Google Scholar]
- 89.Maylie J, Adelman JP. Cholinergic signaling through synaptic SK channels: it's a protein kinase but which one? Neuron. 2010;68(5):809–811. doi: 10.1016/j.neuron.2010.11.037. doi:10.1016/j.neuron.2010.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.McCormack TJ, Melis C, Colon J, Gay EA, Mike A, Karoly R, Lamb PW, Molteni C, Yakel JL. Rapid desensitization of the rat alpha7 nAChR is facilitated by the presence of a proline residue in the outer beta-sheet. J Physiol. 2010;588(Pt 22):4415–4429. doi: 10.1113/jphysiol.2010.195495. doi:10.1113/jphysiol.2010.195495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.McCormack T, Petrovich RM, Mercier KA, DeRose EF, Cuneo MJ, Williams J, Johnson KL, Lamb PW, London RE, Yakel JL. Identification and functional characterization of a novel acetylcholine-binding protein from the marine annelid Capitella teleta. Biochemistry. 2010;49(10):2279–2287. doi: 10.1021/bi902023y. doi:10.1021/bi902023y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.McGehee DS. Nicotinic receptors and hippocampal synaptic plasticity … it's all in the timing. Trends Neurosci. 2002;25(4):171–172. doi: 10.1016/s0166-2236(00)02127-5. [DOI] [PubMed] [Google Scholar]
- 93.McLean CA, Cherny RA, Fraser FW, Fuller SJ, Smith MJ, Beyreuther K, Bush AI, Masters CL. Soluble pool of Abeta amyloid as a determinant of severity of neurodegeneration in Alzheimer's disease. Ann Neurol. 1999;46(6):860–866. doi: 10.1002/1531-8249(199912)46:6<860::aid-ana8>3.0.co;2-m. [DOI] [PubMed] [Google Scholar]
- 94.McQuiston AR, Madison DV. Nicotinic receptor activation excites distinct subtypes of interneurons in the rat hippocampus. J Neurosci. 1999;19(8):2887–2896. doi: 10.1523/JNEUROSCI.19-08-02887.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Murray TA, Bertrand D, Papke RL, George AA, Pantoja R, Srinivasan R, Liu Q, Wu J, Whiteaker P, Lester HA, Lukas RJ. Alpha7beta2 nicotinic acetylcholine receptors assemble, function, and are activated primarily via their alpha7–alpha7 interfaces. Mol Pharmacol. 2012;81(2):175–188. doi: 10.1124/mol.111.074088. doi:10.1124/mol.111.074088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ng HJ, Whittemore ER, Tran MB, Hogenkamp DJ, Broide RS, Johnstone TB, Zheng L, Stevens KE, Gee KW. Nootropic alpha7 nicotinic receptor allosteric modulator derived from GABAA receptor modulators. Proc Natl Acad Sci U S A. 2007;104(19):8059–8064. doi: 10.1073/pnas.0701321104. doi:10.1073/pnas.0701321104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Ovsepian SV, Anwyl R, Rowan MJ. Endogenous acetylcholine lowers the threshold for long-term potentiation induction in the CA1 area through muscarinic receptor activation: in vivo study. Eur J Neurosci. 2004;20(5):1267–1275. doi: 10.1111/j.1460-9568.2004.03582.x. doi:10.1111/j.1460-9568.2004.03582.x. [DOI] [PubMed] [Google Scholar]
- 98.Parri HR, Hernandez CM, Dineley KT. Research update: alpha7 nicotinic acetylcholine receptor mechanisms in Alzheimer's disease. Biochem Pharmacol. 2011;82(8):931–942. doi: 10.1016/j.bcp.2011.06.039. doi:10.1016/j.bcp.2011.06.039. [DOI] [PubMed] [Google Scholar]
- 99.Pettit DL, Shao Z, Yakel JL. Beta-amyloid(1–42) peptide directly modulates nicotinic receptors in the rat hippocampal slice. J Neurosci. 2001;21(1):RC120. doi: 10.1523/JNEUROSCI.21-01-j0003.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Reis HJ, Guatimosim C, Paquet M, Santos M, Ribeiro FM, Kummer A, Schenatto G, Salgado JV, Vieira LB, Teixeira AL, Palotas A. Neuro-transmitters in the central nervous system & their implication in learning and memory processes. Curr Med Chem. 2009;16(7):796–840. doi: 10.2174/092986709787549271. [DOI] [PubMed] [Google Scholar]
- 101.Rimvall K, Keller F, Waser PG. Development of cholinergic projections in organotypic cultures of rat septum, hippocampus and cerebellum. Brain Res. 1985;351(2):267–278. doi: 10.1016/0165-3806(85)90198-1. [DOI] [PubMed] [Google Scholar]
- 102.Royer S, Zemelman BV, Losonczy A, Kim J, Chance F, Magee JC, Buzsaki G. Control of timing, rate and bursts of hippocampal place cells by dendritic and somatic inhibition. Nat Neurosci. 2012;15(5):769–775. doi: 10.1038/nn.3077. doi:10.1038/nn.3077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Sargent PB. The diversity of neuronal nicotinic acetylcholine receptors. Annu Rev Neurosci. 1993;16:403–443. doi: 10.1146/annurev.ne.16.030193.002155. doi:10.1146/annurev.ne.16.030193.002155. [DOI] [PubMed] [Google Scholar]
- 104.Seeger T, Fedorova I, Zheng F, Miyakawa T, Koustova E, Gomeza J, Basile AS, Alzheimer C, Wess J. M2 muscarinic acetylcholine receptor knock-out mice show deficits in behavioral flexibility, working memory, and hippocampal plasticity. J Neurosci. 2004;24(45):10117–10127. doi: 10.1523/JNEUROSCI.3581-04.2004. doi:10.1523/JNEUROSCI.3581-04.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Seguela P, Wadiche J, Dineley-Miller K, Dani JA, Patrick JW. Molecular cloning, functional properties, and distribution of rat brain alpha 7: a nicotinic cation channel highly permeable to calcium. J Neurosci. 1993;13(2):596–604. doi: 10.1523/JNEUROSCI.13-02-00596.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Selkoe DJ. Alzheimer's disease is a synaptic failure. Science. 2002;298(5594):789–791. doi: 10.1126/science.1074069. doi:10.1126/science.1074069. [DOI] [PubMed] [Google Scholar]
- 107.Sharma G, Vijayaraghavan S. Nicotinic cholinergic signaling in hippocampal astrocytes involves calcium-induced calcium release from intracellular stores. Proc Natl Acad Sci U S A. 2001;98(7):4148–4153. doi: 10.1073/pnas.071540198. doi:10.1073/pnas.071540198. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Sharma G, Vijayaraghavan S. Modulation of presynaptic store calcium induces release of glutamate and postsynaptic firing. Neuron. 2003;38(6):929–939. doi: 10.1016/s0896-6273(03)00322-2. [DOI] [PubMed] [Google Scholar]
- 109.Shen JX, Yakel JL. Nicotinic acetylcholine receptor-mediated calcium signaling in the nervous system. Acta Pharmacol Sin. 2009;30(6):673–680. doi: 10.1038/aps.2009.64. doi:10.1038/aps.2009.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Shen JX, Yakel JL. Functional alpha7 nicotinic ACh receptors on astrocytes in rat hippocampal CA1 slices. J Mol Neurosci. 2012;48(1):14–21. doi: 10.1007/s12031-012-9719-3. doi:10.1007/s12031-012-9719-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Shimohama S, Kihara T. Nicotinic receptor-mediated protection against beta-amyloid neurotoxicity. Biol Psychiatry. 2001;49(3):233–239. doi: 10.1016/s0006-3223(00)01100-8. [DOI] [PubMed] [Google Scholar]
- 112.Shytle RD, Mori T, Townsend K, Vendrame M, Sun N, Zeng J, Ehrhart J, Silver AA, Sanberg PR, Tan J. Cholinergic modulation of microglial activation by alpha 7 nicotinic receptors. J Neurochem. 2004;89(2):337–343. doi: 10.1046/j.1471-4159.2004.02347.x. doi:10.1046/j.1471-4159.2004.02347.x. [DOI] [PubMed] [Google Scholar]
- 113.Silberberg G, Wu C, Markram H. Synaptic dynamics control the timing of neuronal excitation in the activated neocortical microcircuit. J Physiol. 2004;556(Pt 1):19–27. doi: 10.1113/jphysiol.2004.060962. doi:10.1113/jphysiol.2004.060962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Stevens TR, Krueger SR, Fitzsimonds RM, Picciotto MR. Neuroprotection by nicotine in mouse primary cortical cultures involves activation of calcineurin and L-type calcium channel inactivation. J Neurosci. 2003;23(31):10093–10099. doi: 10.1523/JNEUROSCI.23-31-10093.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Stokes C, Papke JK, Horenstein NA, Kem WR, McCormack TJ, Papke RL. The structural basis for GTS-21 selectivity between human and rat nicotinic alpha7 receptors. Mol Pharmacol. 2004;66(1):14–24. doi: 10.1124/mol.66.1.14. doi:10.1124/mol.66.1.14. [DOI] [PubMed] [Google Scholar]
- 116.Sudweeks SN, Yakel JL. Functional and molecular characterization of neuronal nicotinic ACh receptors in rat CA1 hippocampal neurons. J Physiol. 2000;527(Pt 3):515–528. doi: 10.1111/j.1469-7793.2000.00515.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Svensson AL, Nordberg A. Beta-estradiol attenuate amyloid beta-peptide toxicity via nicotinic receptors. Neuroreport. 1999;10(17):3485–3489. doi: 10.1097/00001756-199911260-00004. [DOI] [PubMed] [Google Scholar]
- 118.Terry AV, Jr, Buccafusco JJ. The cholinergic hypothesis of age and Alzheimer's disease-related cognitive deficits: recent challenges and their implications for novel drug development. J Pharmacol Exp Ther. 2003;306(3):821–827. doi: 10.1124/jpet.102.041616. doi:10.1124/jpet.102.041616. [DOI] [PubMed] [Google Scholar]
- 119.Thomsen MS, Mikkelsen JD. Type I and II positive allosteric modulators differentially modulate agonist-induced up-regulation of alpha7 nicotinic acetylcholine receptors. J Neurochem. 2012;123(1):73–83. doi: 10.1111/j.1471-4159.2012.07876.x. doi:10.1111/j.1471-4159.2012.07876.x. [DOI] [PubMed] [Google Scholar]
- 120.Tian L, Akerboom J, Schreiter ER, Looger LL. Neural activity imaging with genetically encoded calcium indicators. Prog Brain Res. 2012;196:79–94. doi: 10.1016/B978-0-444-59426-6.00005-7. doi:10.1016/B978-0-444-59426-6.00005-7. [DOI] [PubMed] [Google Scholar]
- 121.Tian L, Hires SA, Mao T, Huber D, Chiappe ME, Chalasani SH, Petreanu L, Akerboom J, McKinney SA, Schreiter ER, Bargmann CI, Jayaraman V, Svoboda K, Looger LL. Imaging neural activity in worms, flies and mice with improved GCaMP calcium indicators. Nat Methods. 2009;6(12):875–881. doi: 10.1038/nmeth.1398. doi:10.1038/nmeth.1398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Tu B, Gu Z, Shen JX, Lamb PW, Yakel JL. Characterization of a nicotine-sensitive neuronal population in rat entorhinal cortex. J Neurosci. 2009;29(33):10436–10448. doi: 10.1523/JNEUROSCI.2580-09.2009. doi:10.1523/JNEUROSCI.2580-09.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Turski L, Ikonomidou C, Turski WA, Bortolotto ZA, Cavalheiro EA. Review: cholinergic mechanisms and epileptogenesis. The seizures induced by pilocarpine: a novel experimental model of intractable epilepsy. Synapse. 1989;3(2):154–171. doi: 10.1002/syn.890030207. doi:10.1002/syn.890030207. [DOI] [PubMed] [Google Scholar]
- 124.Tyagi E, Agrawal R, Nath C, Shukla R. Inhibitory role of cholinergic system mediated via alpha7 nicotinic acetylcholine receptor in LPS-induced neuro-inflammation. Innate Immun. 2010;16(1):3–13. doi: 10.1177/1753425909104680. doi:10.1177/1753425909104680. [DOI] [PubMed] [Google Scholar]
- 125.Unwin N. Refined structure of the nicotinic acetylcholine receptor at 4 Å resolution. J Mol Biol. 2005;346(4):967–989. doi: 10.1016/j.jmb.2004.12.031. doi:10.1016/j.jmb.2004.12.031. [DOI] [PubMed] [Google Scholar]
- 126.Velez-Fort M, Audinat E, Angulo MC. Functional alpha 7-containing nicotinic receptors of NG2-expressing cells in the hippocampus. Glia. 2009;57(10):1104–1114. doi: 10.1002/glia.20834. doi:10.1002/glia.20834. [DOI] [PubMed] [Google Scholar]
- 127.Wada E, Wada K, Boulter J, Deneris E, Heinemann S, Patrick J, Swanson LW. Distribution of alpha 2, alpha 3, alpha 4, and beta 2 neuronal nicotinic receptor subunit mRNAs in the central nervous system: a hybridization histochemical study in the rat. J Comp Neurol. 1989;284(2):314–335. doi: 10.1002/cne.902840212. doi:10.1002/cne.902840212. [DOI] [PubMed] [Google Scholar]
- 128.Wang H, Yu M, Ochani M, Amella CA, Tanovic M, Susarla S, Li JH, Yang H, Ulloa L, Al-Abed Y, Czura CJ, Tracey KJ. Nicotinic acetylcholine receptor alpha7 subunit is an essential regulator of inflammation. Nature. 2003;421(6921):384–388. doi: 10.1038/nature01339. doi: 10.1038/nature01339. [DOI] [PubMed] [Google Scholar]
- 129.Welsby P, Rowan M, Anwyl R. Nicotinic receptor-mediated enhancement of long-term potentiation involves activation of metabotropic glutamate receptors and ryanodine-sensitive calcium stores in the dentate gyrus. Eur J Neurosci. 2006;24(11):3109–3118. doi: 10.1111/j.1460-9568.2006.05187.x. doi:10.1111/j.1460-9568.2006.05187.x. [DOI] [PubMed] [Google Scholar]
- 130.Welsby PJ, Rowan MJ, Anwyl R. Beta-amyloid blocks high frequency stimulation induced LTP but not nicotine enhanced LTP. Neuropharmacology. 2007;53(1):188–195. doi: 10.1016/j.neuropharm.2007.05.013. doi:10.1016/j.neuropharm.2007.05.013. [DOI] [PubMed] [Google Scholar]
- 131.Widmer H, Ferrigan L, Davies CH, Cobb SR. Evoked slow muscarinic acetylcholinergic synaptic potentials in rat hippocampal interneurons. Hippocampus. 2006;16(7):617–628. doi: 10.1002/hipo.20191. doi:10.1002/hipo.20191. [DOI] [PubMed] [Google Scholar]
- 132.Wu J, Kuo YP, George AA, Xu L, Hu J, Lukas RJ. beta-Amyloid directly inhibits human alpha4beta2-nicotinic acetylcholine receptors heterologously expressed in human SHEP1 cells. J Biol Chem. 2004;279(36):37842–37851. doi: 10.1074/jbc.M400335200. doi:10.1074/jbc.M400335200. [DOI] [PubMed] [Google Scholar]
- 133.Young GT, Zwart R, Walker AS, Sher E, Millar NS. Potentiation of alpha7 nicotinic acetylcholine receptors via an allosteric transmembrane site. Proc Natl Acad Sci U S A. 2008;105(38):14686–14691. doi: 10.1073/pnas.0804372105. doi:10.1073/pnas.0804372105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Zhang X, Liu C, Miao H, Gong ZH, Nordberg A. Postnatal changes of nicotinic acetylcholine receptor alpha 2, alpha 3, alpha 4, alpha 7 and beta 2 subunits genes expression in rat brain. Int J Dev Neurosci. 1998;16(6):507–518. doi: 10.1016/s0736-5748(98)00044-6. [DOI] [PubMed] [Google Scholar]
- 135.Zhang LI, Tao HW, Holt CE, Harris WA, Poo M. A critical window for cooperation and competition among developing retinotectal synapses. Nature. 1998;395(6697):37–44. doi: 10.1038/25665. doi:10.1038/25665. [DOI] [PubMed] [Google Scholar]
- 136.Zhao Y, Araki S, Wu J, Teramoto T, Chang YF, Nakano M, Abdelfattah AS, Fujiwara M, Ishihara T, Nagai T, Campbell RE. An expanded palette of genetically encoded Ca (2)(+) indicators. Science. 2011;333(6051):1888–1891. doi: 10.1126/science.1208592. doi:10.1126/science.1208592. [DOI] [PMC free article] [PubMed] [Google Scholar]