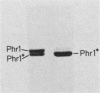Abstract
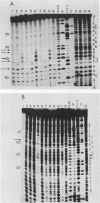
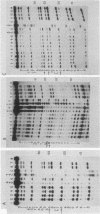
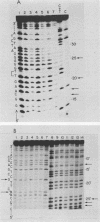
DNA photolyases catalyze the light-dependent repair of pyrimidine dimers in DNA. The results of nucleotide sequence analysis and spectroscopic studies demonstrated that photolyases from Saccharomyces cerevisiae and Escherichia coli share 37% amino acid sequence homology and contain identical chromophores. Do the similarities between these two enzymes extend to their interactions with DNA containing pyrimidine dimers, or does the organization of DNA into nucleosomes in S. cerevisiae necessitate alternative or additional recognition determinants? To answer this question, we used chemical and enzymatic techniques to identify the contacts made on DNA by S. cerevisiae photolyase when it is bound to a pyrimidine dimer and compared these contacts with those made by E. coli photolyase and by a truncated derivative of the yeast enzyme when bound to the same substrate. We found evidence for a common set of interactions between the photolyases and specific phosphates in the backbones of both strands as well as for interactions with bases in both the major and minor grooves of dimer-containing DNA. Superimposed on this common pattern were significant differences in the contributions of specific contacts to the overall binding energy, in the interactions of the enzymes with groups on the complementary strand, and in the extent to which other DNA-binding proteins were excluded from the region around the dimer. These results provide strong evidence both for a conserved dimer-binding motif and for the evolution of new interactions that permit photolyases to also act as accessory proteins in nucleotide excision repair. The locations of the specific contacts made by the yeast enzyme indicate that the mechanism of nucleotide excision repair in this organism involves incision(s) at a distance from the pyrimidine dimer.
Full text
PDF





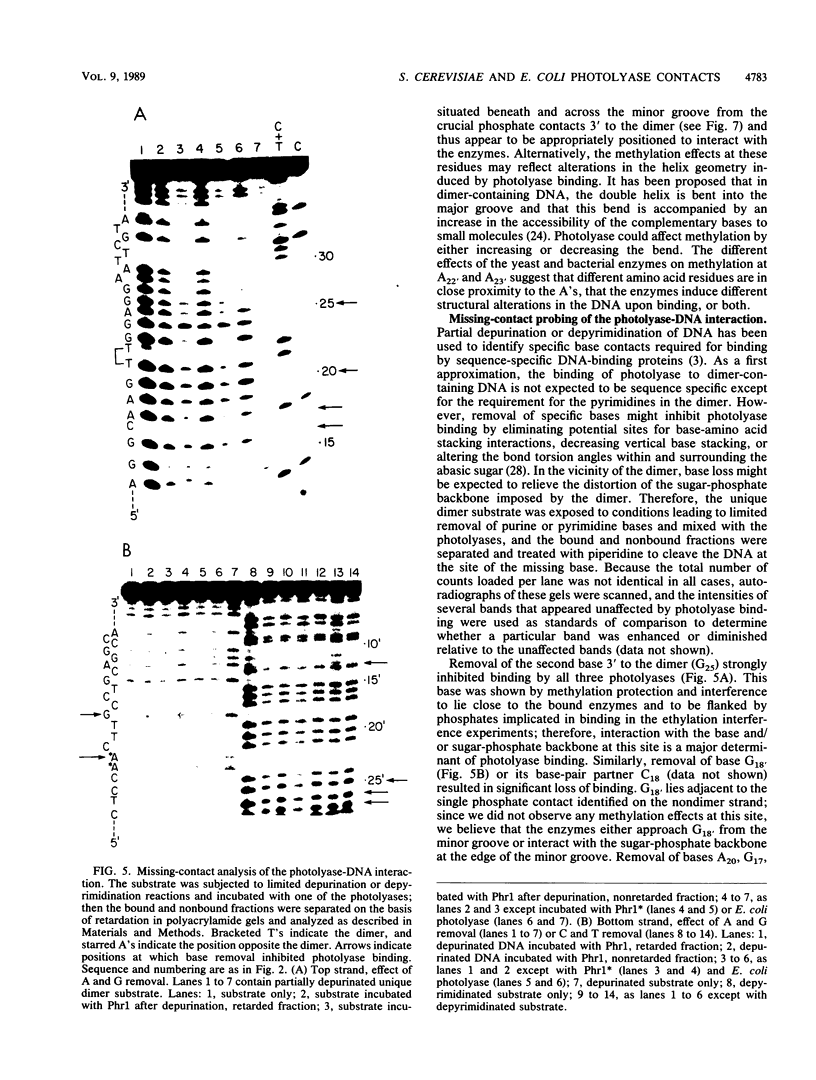

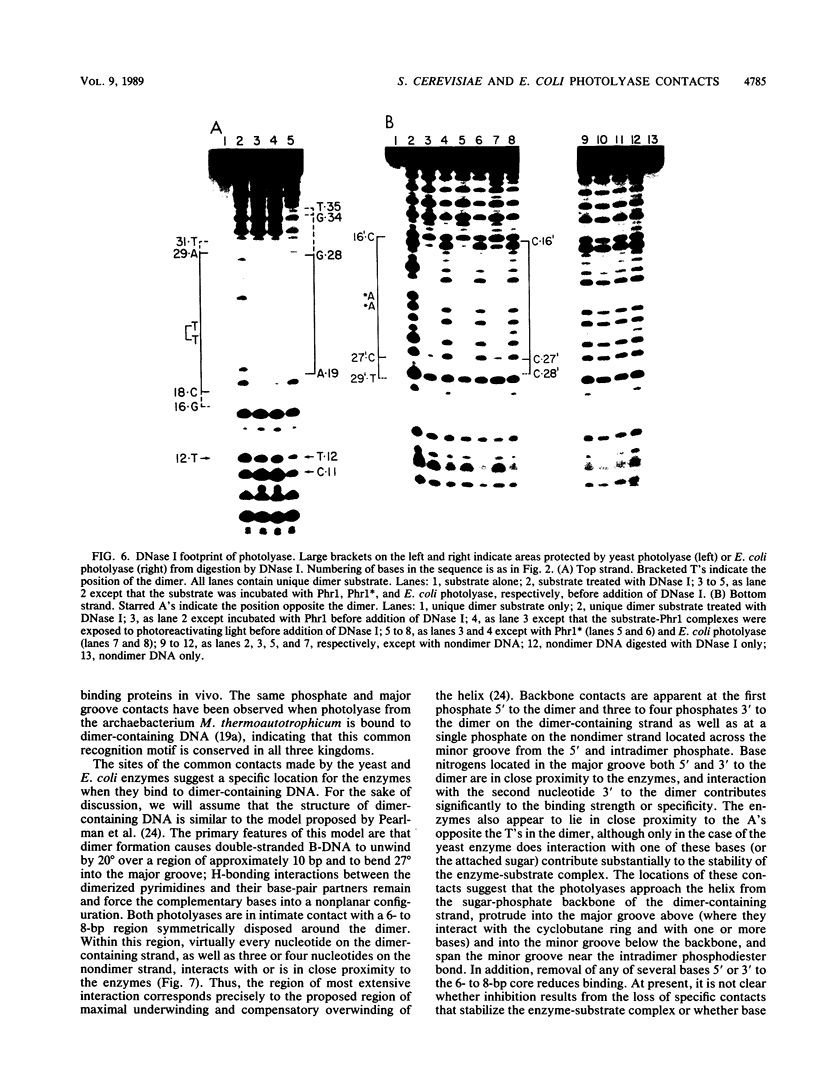



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Banerjee S. K., Christensen R. B., Lawrence C. W., LeClerc J. E. Frequency and spectrum of mutations produced by a single cis-syn thymine-thymine cyclobutane dimer in a single-stranded vector. Proc Natl Acad Sci U S A. 1988 Nov;85(21):8141–8145. doi: 10.1073/pnas.85.21.8141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brunelle A., Schleif R. F. Missing contact probing of DNA-protein interactions. Proc Natl Acad Sci U S A. 1987 Oct;84(19):6673–6676. doi: 10.1073/pnas.84.19.6673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Eker A. P., Dekker R. H., Berends W. Photoreactivating enzyme from Streptomyces griseus-IV. On the nature of the chromophoric cofactor in Streptomyces griseus photoreactivating enzyme. Photochem Photobiol. 1981 Jan;33(1):65–72. doi: 10.1111/j.1751-1097.1981.tb04298.x. [DOI] [PubMed] [Google Scholar]
- Eker A. P., Hessels J. K., Dekker R. H. Photoreactivating enzyme from Streptomyces griseus--VI. Action spectrum and kinetics of photoreactivation. Photochem Photobiol. 1986 Aug;44(2):197–205. doi: 10.1111/j.1751-1097.1986.tb03586.x. [DOI] [PubMed] [Google Scholar]
- Ferguson L. R., Cox B. S. Excision of bases accompanying the excision of dimers from DNA of UV-irradiated yeast. Mol Gen Genet. 1974;135(1):87–90. doi: 10.1007/BF00433904. [DOI] [PubMed] [Google Scholar]
- Fried M., Crothers D. M. Equilibria and kinetics of lac repressor-operator interactions by polyacrylamide gel electrophoresis. Nucleic Acids Res. 1981 Dec 11;9(23):6505–6525. doi: 10.1093/nar/9.23.6505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galas D. J., Schmitz A. DNAse footprinting: a simple method for the detection of protein-DNA binding specificity. Nucleic Acids Res. 1978 Sep;5(9):3157–3170. doi: 10.1093/nar/5.9.3157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garner M. M., Revzin A. A gel electrophoresis method for quantifying the binding of proteins to specific DNA regions: application to components of the Escherichia coli lactose operon regulatory system. Nucleic Acids Res. 1981 Jul 10;9(13):3047–3060. doi: 10.1093/nar/9.13.3047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grafstrom R. H., Park L., Grossman L. Enzymatic repair of pyrimidine dimer-containing DNA. A 5' dimer DNA glycosylase: 3'-apyrimidinic endonuclease mechanism from Micrococcus luteus. J Biol Chem. 1982 Nov 25;257(22):13465–13474. [PubMed] [Google Scholar]
- Hertzberg R. P., Dervan P. B. Cleavage of DNA with methidiumpropyl-EDTA-iron(II): reaction conditions and product analyses. Biochemistry. 1984 Aug 14;23(17):3934–3945. doi: 10.1021/bi00312a022. [DOI] [PubMed] [Google Scholar]
- Husain I., Sancar A. Binding of E. coli DNA photolyase to a defined substrate containing a single T mean value of T dimer. Nucleic Acids Res. 1987 Feb 11;15(3):1109–1120. doi: 10.1093/nar/15.3.1109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Husain I., Sancar G. B., Holbrook S. R., Sancar A. Mechanism of damage recognition by Escherichia coli DNA photolyase. J Biol Chem. 1987 Sep 25;262(27):13188–13197. [PubMed] [Google Scholar]
- Johnson J. L., Hamm-Alvarez S., Payne G., Sancar G. B., Rajagopalan K. V., Sancar A. Identification of the second chromophore of Escherichia coli and yeast DNA photolyases as 5,10-methenyltetrahydrofolate. Proc Natl Acad Sci U S A. 1988 Apr;85(7):2046–2050. doi: 10.1073/pnas.85.7.2046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorns M. S., Sancar G. B., Sancar A. Identification of oligothymidylates as new simple substrates for Escherichia coli DNA photolyase and their use in a rapid spectrophotometric enzyme assay. Biochemistry. 1985 Apr 9;24(8):1856–1861. doi: 10.1021/bi00329a008. [DOI] [PubMed] [Google Scholar]
- Kiener A., Husain I., Sancar A., Walsh C. Purification and properties of Methanobacterium thermoautotrophicum DNA photolyase. J Biol Chem. 1989 Aug 15;264(23):13880–13887. [PubMed] [Google Scholar]
- Liuzzi M., Weinfeld M., Paterson M. C. Selective inhibition by methoxyamine of the apurinic/apyrimidinic endonuclease activity associated with pyrimidine dimer-DNA glycosylases from Micrococcus luteus and bacteriophage T4. Biochemistry. 1987 Jun 16;26(12):3315–3321. doi: 10.1021/bi00386a011. [DOI] [PubMed] [Google Scholar]
- Maxam A. M., Gilbert W. Sequencing end-labeled DNA with base-specific chemical cleavages. Methods Enzymol. 1980;65(1):499–560. doi: 10.1016/s0076-6879(80)65059-9. [DOI] [PubMed] [Google Scholar]
- Mirzabekov A. D., Rich A. Asymmetric lateral distribution of unshielded phosphate groups in nucleosomal DNA and its role in DNA bending. Proc Natl Acad Sci U S A. 1979 Mar;76(3):1118–1121. doi: 10.1073/pnas.76.3.1118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Myles G. M., Van Houten B., Sancar A. Utilization of DNA photolyase, pyrimidine dimer endonucleases, and alkali hydrolysis in the analysis of aberrant ABC excinuclease incisions adjacent to UV-induced DNA photoproducts. Nucleic Acids Res. 1987 Feb 11;15(3):1227–1243. doi: 10.1093/nar/15.3.1227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearlman D. A., Holbrook S. R., Pirkle D. H., Kim S. H. Molecular models for DNA damaged by photoreaction. Science. 1985 Mar 15;227(4692):1304–1308. doi: 10.1126/science.3975615. [DOI] [PubMed] [Google Scholar]
- RUPERT C. S., GOODGAL S. H., HERRIOTT R. M. Photoreactivation in vitro of ultraviolet-inactivated Hemophilus influenzae transforming factor. J Gen Physiol. 1958 Jan 20;41(3):451–471. doi: 10.1085/jgp.41.3.451. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RUPERT C. S. Photoenzymatic repair of ultraviolet damage in DNA. I. Kinetics of the reaction. J Gen Physiol. 1962 Mar;45:703–724. doi: 10.1085/jgp.45.4.703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RUPERT C. S. Photoenzymatic repair of ultraviolet damage in DNA. II. Formation of an enzyme-substrate complex. J Gen Physiol. 1962 Mar;45:725–741. doi: 10.1085/jgp.45.4.725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saito N., Werbin H. Purification of a blue-green algal deoxyribonucleic acid photoreactiving enzyme. An enzyme requiring light as a physical cofactor to perform its catalytic function. Biochemistry. 1970 Jun 23;9(13):2610–2620. doi: 10.1021/bi00815a008. [DOI] [PubMed] [Google Scholar]
- Sancar A., Franklin K. A., Sancar G. B. Escherichia coli DNA photolyase stimulates uvrABC excision nuclease in vitro. Proc Natl Acad Sci U S A. 1984 Dec;81(23):7397–7401. doi: 10.1073/pnas.81.23.7397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancar A., Rupp W. D. A novel repair enzyme: UVRABC excision nuclease of Escherichia coli cuts a DNA strand on both sides of the damaged region. Cell. 1983 May;33(1):249–260. doi: 10.1016/0092-8674(83)90354-9. [DOI] [PubMed] [Google Scholar]
- Sancar A., Sancar G. B. DNA repair enzymes. Annu Rev Biochem. 1988;57:29–67. doi: 10.1146/annurev.bi.57.070188.000333. [DOI] [PubMed] [Google Scholar]
- Sancar A., Sancar G. B. Escherichia coli DNA photolyase is a flavoprotein. J Mol Biol. 1984 Jan 15;172(2):223–227. doi: 10.1016/s0022-2836(84)80040-6. [DOI] [PubMed] [Google Scholar]
- Sancar A., Smith F. W., Sancar G. B. Purification of Escherichia coli DNA photolyase. J Biol Chem. 1984 May 10;259(9):6028–6032. [PubMed] [Google Scholar]
- Sancar G. B. Sequence of the Saccharomyces cerevisiae PHR1 gene and homology of the PHR1 photolyase to E. coli photolyase. Nucleic Acids Res. 1985 Nov 25;13(22):8231–8246. doi: 10.1093/nar/13.22.8231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancar G. B., Smith F. W., Heelis P. F. Purification of the yeast PHR1 photolyase from an Escherichia coli overproducing strain and characterization of the intrinsic chromophores of the enzyme. J Biol Chem. 1987 Nov 15;262(32):15457–15465. [PubMed] [Google Scholar]
- Sancar G. B., Smith F. W. Interactions between yeast photolyase and nucleotide excision repair proteins in Saccharomyces cerevisiae and Escherichia coli. Mol Cell Biol. 1989 Nov;9(11):4767–4776. doi: 10.1128/mcb.9.11.4767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancar G. B., Smith F. W., Lorence M. C., Rupert C. S., Sancar A. Sequences of the Escherichia coli photolyase gene and protein. J Biol Chem. 1984 May 10;259(9):6033–6038. [PubMed] [Google Scholar]
- Sancar G. B., Smith F. W., Reid R., Payne G., Levy M., Sancar A. Action mechanism of Escherichia coli DNA photolyase. I. Formation of the enzyme-substrate complex. J Biol Chem. 1987 Jan 5;262(1):478–485. [PubMed] [Google Scholar]
- Sancar G. B., Smith F. W., Sancar A. Binding of Escherichia coli DNA photolyase to UV-irradiated DNA. Biochemistry. 1985 Apr 9;24(8):1849–1855. doi: 10.1021/bi00329a007. [DOI] [PubMed] [Google Scholar]
- Setlow J. K., Bollum F. J. The minimum size of the substrate for yeast photoreactivating enzyme. Biochim Biophys Acta. 1968 Apr 22;157(2):233–237. doi: 10.1016/0005-2787(68)90077-4. [DOI] [PubMed] [Google Scholar]
- Setlow R. B., Carrier W. L. Pyrimidine dimers in ultraviolet-irradiated DNA's. J Mol Biol. 1966 May;17(1):237–254. doi: 10.1016/s0022-2836(66)80105-5. [DOI] [PubMed] [Google Scholar]
- Siebenlist U., Gilbert W. Contacts between Escherichia coli RNA polymerase and an early promoter of phage T7. Proc Natl Acad Sci U S A. 1980 Jan;77(1):122–126. doi: 10.1073/pnas.77.1.122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Siebenlist U., Simpson R. B., Gilbert W. E. coli RNA polymerase interacts homologously with two different promoters. Cell. 1980 Jun;20(2):269–281. doi: 10.1016/0092-8674(80)90613-3. [DOI] [PubMed] [Google Scholar]
- Suck D., Oefner C. Structure of DNase I at 2.0 A resolution suggests a mechanism for binding to and cutting DNA. Nature. 1986 Jun 5;321(6070):620–625. doi: 10.1038/321620a0. [DOI] [PubMed] [Google Scholar]
- Unrau P., Wheatcroft R., Cox B. S. The excision of pyrimidine dimers from DNA of ultraviolet irradiated yeast. Mol Gen Genet. 1971;113(4):359–362. doi: 10.1007/BF00272336. [DOI] [PubMed] [Google Scholar]
- Wang J. C. Helical repeat of DNA in solution. Proc Natl Acad Sci U S A. 1979 Jan;76(1):200–203. doi: 10.1073/pnas.76.1.200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weinfeld M., Gentner N. E., Johnson L. D., Paterson M. C. Photoreversal-dependent release of thymidine and thymidine monophosphate from pyrimidine dimer-containing DNA excision fragments isolated from ultraviolet-damaged human fibroblasts. Biochemistry. 1986 May 6;25(9):2656–2664. doi: 10.1021/bi00357a055. [DOI] [PubMed] [Google Scholar]
- Weinfeld M., Paterson M. C. DNA cyclobutane pyrimidine dimers with a cleaved internal phosphodiester bond can be photoenzymatically reversed by Escherichia coli PhrB photolyase. Nucleic Acids Res. 1988 Jun 24;16(12):5693–5693. doi: 10.1093/nar/16.12.5693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto K., Satake M., Shinagawa H. A multicopy phr-plasmid increases the ultraviolet resistance of a recA strain of Escherichia coli. Mutat Res. 1984 Jan;131(1):11–18. doi: 10.1016/0167-8817(84)90042-7. [DOI] [PubMed] [Google Scholar]
- Yasui A., Langeveld S. A. Homology between the photoreactivation genes of Saccharomyces cerevisiae and Escherichia coli. Gene. 1985;36(3):349–355. doi: 10.1016/0378-1119(85)90190-8. [DOI] [PubMed] [Google Scholar]
- Yasui A., Takao M., Oikawa A., Kiener A., Walsh C. T., Eker A. P. Cloning and characterization of a photolyase gene from the cyanobacterium Anacystis nidulans. Nucleic Acids Res. 1988 May 25;16(10):4447–4463. doi: 10.1093/nar/16.10.4447. [DOI] [PMC free article] [PubMed] [Google Scholar]