Abstract
Cellular therapy utilizing adult mesenchymal stromal/stem cells (MSCs) may very well revolutionize the treatment of a variety of head and neck diseases through the restoration of normal structure and function. Transplanting allogeneic or autologous MSCs into damaged tissues can serve multiple regenerative functions through their self-renewal, differentiation capacity, immune modulation and secretion of bioactive molecules. Further, trophic factors expressed by MSCs have been shown to influence their microenvironment through the promotion of extracellular matrix remodeling, angiogenesis and wound healing needed to regenerate or replace injured tissues. Although clinical applications of MSC based therapies in Otolaryngology-Head and Neck Surgery are still in their infancy, efforts are being made to understand and exploit MSCs for tissue repair as well as engineering strategies. In this review, we highlight pre clinical and clinical investigations employing MSC based therapies for the reconstruction of bone, cartilage, soft tissue and vocal fold defects.
Keywords: Mesenchymal stem cells, tissue engineering, otolaryngology-head and neck surgery
Introduction
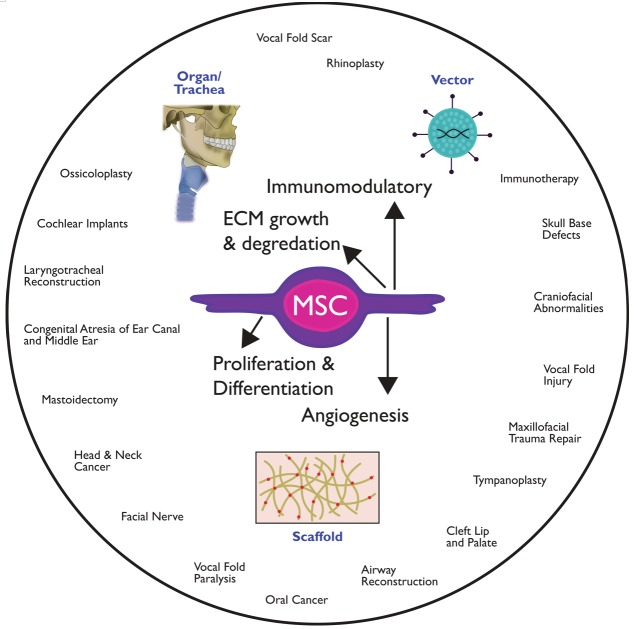
Mesenchymal stromal/stem cells (MSCs) provide numerous possibilities for “off the shelf” clinical application, because of their ease of production via ex vivo expansion, ability to self-renew with high proliferative capacity, and ability for multilineage differentiation [1]. MSCs are also an attractive cell choice as they produce numerous bioactive molecules that can suppress inflammatory responses and influence regeneration in injured tissues [2-5]. Moreover, one of MSCs most beneficial therapeutic effects is their immunomodulatory capacity that appears to facilitate their use for allogeneic transplantation [6]. MSCs can be transplanted into the body directly from freshly isolated tissues or after culture expansion, through degradable scaffolds or as complete three-dimensional tissues for organ replacement (Figure 1). MSCs can secrete various tropic factors, including molecules that regulate cell growth, proliferation, fibrosis, angiogenesis and immune suppression [7]. However, their long-term engraftment and differentiation capacity into damaged tissues are still not well understood. MSCs can be directly delivered to the site to treat inflammatory injuries or to provide potent bioactive molecules that can suppress the immune response during periods of inflammation. Lastly, combination of MSCs with biomaterial scaffolds or decellularized allografts could extend their long-term engraftment and differentiation in tissue, which can be clinically beneficial for the treatment of non-healing wounds, scarring or functional replacement of tissue [8-10]. There are diverse therapeutic applications utilizing MSC in Otolaryngology-Head and Neck Surgery including plastic reconstruction, tissue regeneration and revascularization of ischemic tissues (Table 1).
Figure 1.
Schematic of the potential therapeutic applications utilizing mesenchymal stem cells in Otolaryngology–Head and Neck Surgery.
Table 1.
Mesenchymal Stem Cell Based Therapies in Otolaryngology-Head and Neck Surgery
| Disorder | MSC Source | Differentiation | TE Approach | Biomaterials | Study | Outcome | Ref # |
|---|---|---|---|---|---|---|---|
| Vocal fold scar | Allogenic, Mouse BM | undifferentiated | cells + scaffold | Carbylan-GSX (HA hydrogel) | In vivo | Promote ECM production and degradation, suppressed myofibroblast differentiation; no cytotoxicity | [8] |
| Vocal fold scar | Allogenic, human AT/BM | undifferentiated | cells + scaffold | Carbylan-GSX | In vitro | MSCs suppressed inflammatory response against hydrogels | [47] |
| Vocal fold scar | Human AT | undifferentiated | cells + scaffold | Hydrogel (HA, collagen, fibrin, or co-gels) | In vitro | Fibrin co-gels + HA or Col had enhanced proliferation, differentiation and elastin expression | [44] |
| Vocal fold scar | Mice BM | undifferentiated | cells + scaffold | 1% HCL atelocollagen gel from calf dermis | In vitro | MSCs maintained undifferentiated state; however, there was low cell survival on matrix (20%) | [51] |
| Vocal fold scar | Human AT | differentiation induced in gel | cells + scaffold | Fibrin hydrogels | In vitro | Created bi-layer construct to replace vocal fold cover | [48,49] |
| Vocal fold scar | Rabbit AT | undifferentiated | cells + scaffold | Collagen or HA gel | In vitro | Safe stable engraftment, by 12 months lamina propria appeared to be normally distributed | [52] |
| Vocal fold scar | Autologous, Dog BM | undifferentiated | cells + scaffold | Atelocollagen sponge | in vivo | Histologic evaluations showed increased HA distribution and decreased dense collagen deposition | [53] |
| Injured vocal folds | Autologous, Dog BM | undifferentiated | cells + carrier | 1% HCL atelocollagen gel from porcine skin | In vivo | Histology showed MSC engraftment at 2 months post-surgery. | [54] |
| Vocal fold scar | Human BM | undifferentiated | cells | Saline | In vivo | Histology at 3 months showed reduced lamina propria thickness and collagen content. Improved viscoelastic properties. | [42] |
| Injured vocal folds | Allogenic, murine BM | undifferentiated | cells | N/A | In vivo | MSCs appeared to differentiate into multiple lineages in vivo | [55] |
| Vocal fold scar | Human BM | undifferentiated | cells | Saline | In vivo | Improved viscoelasticity, but engraftment was poor (0.18%) at 4 weeks post-surgery | [41] |
| Tracheal stenosis | Autologous Rabbit AT | undifferentiated | cells + graft | Porcine small intestinal submucosa | In vivo | Reduced inflammatory cell infiltration and granulation; promoted pseudostratified columnar epithelium; detected MSCs12 weeks post | [56] |
| Tracheal stenosis | Rat AT | undifferentiated | MSC + gingival fibroblasts + scaffold | Collagen scaffold | In vivo | Both cells together had greatest effect on tracheal epithelial regeneration | [57] |
| Tracheal stenosis | Autologous, Human BM | chondrocytes | cells + graft | Decellularized donor trachea | In situ | MSCs differentiated into chondrocytes and seeded onto the matrix with bioreactor | [10] |
| Tracheal stenosis | Rat AT | chondrocytes | cells + scaffold | Collagen gel and sponge | In vitro/ in vivo | Pseudostratified columnar epithelium with well-differentiated ciliated and goblet cells and neovascularization | [30] |
| Tracheal stenosis | Allogenic, Rat BM | osteogeneic | cells + scaffold | Collagen I hydrogel tubes | In vivo | Ring shaped tissue with MSCs reinforced tubular construct and enhanced stiffness of construct preventing tissue collapse | [31] |
| Tracheal stenosis | Allogenic, Rat BM | undifferentiated | cells | Systemic | In vivo | Labeled MSCs found at implant site with pseudostriated ciliated columnar epithelium covering lumen and increased VEGF levels at 8 weeks post-surgery. | [32] |
| Congenital high airway obstruction syndrome | Autologous, Lamb amniotic | chondrocytes | cells + graft | Xenologous decellularized tracheas | in vivo | MSC grafts enhanced remodeling by promoting epithelialization and increases in elastin levels | [58] |
| Long-segment airway stenosis | Autologous, Pig BM | chondrocytes | MSC + mucosal epithelial cells + graft | Decellularized 12 cm trachea | in vivo | Decellularized matrix with both cell types showed no signs of airway collapse or ischemia 60 days post | [29] |
| Long-segment congenital tracheal stenosis | Autologous, Human BM | seeded undifferentiated, induced chondrocyte diff. intraoperatively | MSC + patches of autologous epithelium + graft | Acellular allogeneic trachea | in situ | Reepithelialization and proximal rigidity took ~2yrs, regular bronchoscopy to remove secretions and balloon dilations performed. No development of anti-donor antibodies or rejection. | [59] |
| Cleft lip/palate | Dog, BM from iliac bone | undifferentiated | cell + carrier | Carbonated hydroxyapatite particles | in vivo | MSCs contributed to new bone formation and increase in capillary vessels and particle absorption | [60] |
| Craniofacial abnormalities | human BM | osteogeneic | cell + scaffold | Calcium phosphate cements-chitosan scaffold | in vitro | MSC had elevated alkaline phosphatase activity and mineralization. Improving the strength of the scaffold | [61] |
| Mandibular osteoradionecrosis | Autologous, mini pig BM | undifferentiated | cells + carrier | Hydroxyapatite/ tricalcium phosphate particles | in vivo | Bone and vessel regeneration, substantial reconstruction 6 months post | [52] |
| Bone distraction surgery | Autologous, dog BM | transduced with adenovirus BMP-2 | cells + vector + matrix | Demineralized human bone matrix | n vitro/ in vivo | Improved mandible remodeling and bone maturation | [62] |
| Craniofacial bony defects | Allogeniec, rat BM | osteogeneic | cells + growth factor + scaffold | Chitosan from crab shells and BMP-2 | in vivo | MSC and growth factors together enhanced bone growth in defects | [63] |
| Hemimaxillectomy-large keratocyst | Autologous, human AT | osteogeneic | cells + carrier | Titanium cage filled with beta-tricalcium phosphate | In situ | situ Created custom-made ectopic bone implant inside rectus abdominis muscle free flap | [64] |
| Maxillary Bone Defects | miniature swine BM | recombinant adenovirus BMP-2 | cell + scaffold | Collagen type I gel | in vivo | White solid bone formation with mineralized mature woven bone and similar biomechanics as normal bone | [65] |
| Maxillary Bone Defects | human BM | undifferentiated | cells + OP-1 DNA plasmid + scaffold | Chitosan-alginate gel & polyethylenimine | in vitro/ in vivo | MSC + scaffold maintained volumetric shape of gel in vivo; microscopic bone formation was found after 8 wks | [66] |
MSCs are most commonly isolated from bone marrow (BM), but can be derived from other tissue sources, including adipose tissue (AT) [11], umbilical cord [12], vocal fold tissue [13] , dental [14] or periodontal ligaments [15]. MSCs are defined by standards proposed by the International Society for Cellular Therapy (ISCT) [16]. Due to the number of MSC sources and variation in cell culture methods, there is a lack of consensus on their phenotypic and functional properties. Minimum criteria set by the ISCT for characterization of MSCs requires adherence to tissue culture plastic, reproducible expression of stem cell markers and multipotent differentiation [16]. MSCs are distinguished from hematopoietic cells through presence of a combination of cell surface markers, positive for CD29, CD44, CD73, CD90, and CD105 and lack of CD11b, CD14, CD34, HLA-DR and CD45. MSCs must also be able to differentiate into osteogenic, adipogenic, and chondrogenic lineages under standard in vitro differentiating conditions. Ex vivo culture expansion of MSCs results in a heterogeneous population of MSCs exhibiting a broad range of functional properties. Many factors can influence the yield and function of MSCs, such as surgical procedure, harvested anatomical region, isolation methods, culturing conditions (i.e. media, cell density, polystyrene dish) and cell cryopreservation [12,17]. Recent research has indicated MSCs from different tissue sources exhibit distinct phenotypes associated with their tissue microenvironment, which could potentially limit their range of application [18-21]. As a result, pre-clinical studies are needed to determine the optimal MSCs tissue source and processing technique necessary for individual therapeutic uses prior to clinical application.
MSCs based therapies for tissue replacement
Bone replacement
Bone replacement for clinical diseases treated by Otolaryngology-Head and Neck Surgeons are usually highly complex, requiring extensive skeletal reconstruction due to trauma, tumor resection or congenital abnormalities. Autologous bone graft or vascularized flap reconstruction are commonly used procedures to address large bone deficits as the procedures can provide all the biological prerequisites for healing, such as growth factors needed for osteoinduction, osteoprogenitor cells and an osteoconductive support scaffold [22]. However, these are highly invasive surgical procedures with the addition of a donor site and potential complications or morbidity at both the donor and recipient reconstructions. Recently cell based tissue engineering strategies have been explored in select clinical scenarios to avoid complications associated with harvesting of the bone. Biomaterial scaffolds can be osteoconductive, such as hydroxyapatite or other ceramics, which can further be enhanced by the addition of osteogenic cell lineages. Alone, bone marrow aspirates have been added to fracture sites for decades to promote healing or treat fracture non-union repair [23,24]. Engineered bone constructs are gaining support in maxillofacial applications, as an alternative to bone grafting for treatment of small defects or cysts, fracture support, or congenital anomalies. The combination of genetically engineered MSCs or those differentiated into osteocytes with synthetic bone substitutes could enhance the mechanical stability of the bone graft during formation by stimulating the secretion of bone morphogenetic proteins (BMP) and regenerating the ECM [25].
Perhaps most promising is the cellular augmentation of deficient bone stock for dental implants. The use of dental implants is limited by the volume of viable bone in the posterior maxillary floor and often an autologous bone graft or synthetic bone mineral is used to supplement the maxillary ridge for future implantation. The addition of MSCs to any implants may improve clinical outcomes. To this end, one study compared 12 consecutive patients undergoing sinus floor augmentation with each patient serving as their own control [26]. Both sides of the patients’ mouth were augmented with a hydroxyapatite based scaffold with one side receiving “standard therapy” of the scaffold combined with autologous bone graft, while the other received the scaffold combined with autologous BM MSCs (at passage 0). It was demonstrated that there was significantly more new bone formation in the area treated with MSCs compared to standard bone graft and no differences in overall healing, complication rate, or ability to place dental implants between either groups. In another investigation, Shayesteh and colleagues demonstrated similar success in six consecutive patients treated with hydroxyapatite - β tricalcium phosphate ceramic loaded with culture expanded autologous BM MSCs for maxillary sinus elevation (at passage 2-3) [27]. In both examples MSCs were differentiated into osteogenic lineages prior to seeding in the graft.
Demineralized bone matrix with calcium sulphate loaded with culture expanded MSCs has also been used clinically for reconstruction of alveolar defects in two patients with unilateral cleft palates [28]. There were no acute complications in the two patients reported. MSCs were added to osteoinductive scaffolds and implanted into defects in their undifferentiated state, demonstrating new bone formation as the grafts were incorporated.
An alternative strategy from those described above is to culture MSCs on scaffolds in osteogenic media to induce bone development de novo and then implant the constructs. Meijer and colleagues harvested BM MSCs via bone marrow aspirate from the iliac crest, expanded these to P3, seeded the MSCs onto hydroxyapatite particles, and cultured the composite for an additional seven days in osteogenic culture media [9]. The bone substitutes were placed directly into the defect site and covered with a local periosteal flap. Four months post-implantation, patients had dental implants placed and biopsies taken. There was bone formation observed in three of six patients with the remaining three patients failing to show new bone formation. Interestingly, constructs from each of the patients were implanted in a subcutaneous pocket on the backs of athymic mice at the time of their original surgery as well. At six weeks, all of the constructs in the mice exhibited new bone formation on histological analysis, although this was not observed in each of the clinical correlates. This investigation is noteworthy as it illustrates the inconsistent nature of translational research when developing complex tissue engineered constructs, particularly when using rodents as preclinical models.
Cartilage replacement
MSCs can be utilized in various capacities for cartilage replacement involving local engraftment into decellularized allografts or artificial scaffolds, as well as drug delivery methods to prevent allograft rejection. Most promising approaches in Otolaryngology-Head and Neck Surgery are in the generation of functional trachea replacements after extensive resection or reconstruction due to damage from intubation or malignancies obstructing the airway. Macciarini and colleagues were the first group to perform a human transplantation of a tissue engineered trachea using a decellularized donor trachea seeded with BM MSCs differentiated into chondrocytes on the external surface and epithelial cells in the lumen [10]. They designed a novel bioreactor with separate outer and inner surface compartments and rotated the three-dimensional tubular matrix between liquid and gas phases exposing cells to hydrodynamic shear stress. Animal studies confirming the applicability of this approach have demonstrated that both cell lines work synergistically to prevent graft failure by controlling the rate of bacterial/fungal contamination, preventing stenosis, and retaining functional strength needed to maintain a competent airway [29] (Figure 2). Although this approach was clinically successful providing a functional airway to the recipient, the shortages in donor tracheas limit its wide spread application. Development of artificial tracheas using cell and tissue engineered products would allow scaffolds to be fabricated to the patient, easily reproduced in the clinic and have high survival rate.
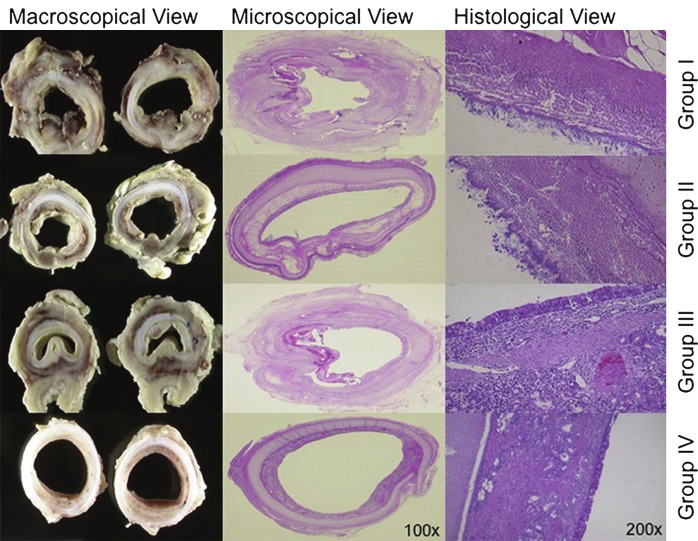
Figure 2.
First column demonstrates formalin-embedded macroscopic views (transversal sections, group specific). The second (1003) and third (2003) columns display a microscopic view of hematoxylin and eosin histologic transversal sections (group specific). For group I (decellularized matrix only), both stenosis and the inflammatory process are visible. For group II (decellularized matrix with external, autologous mesenchymal stem cell-derived chondrocytes), less stenosis but a high grade of bacterial/fungal contamination is shown. For group III (decellularized matrix with internal, autologous epithelial cells), less inflammatory signs and no bacterial/fungal contamination are shown (high stenosis caused by weakness). For group IV (decellularized matrix with both cell types), no stenosis or contamination is shown. (Reprinted from Go T., Jungebluth P., Baiguero S., et al (2010). Both epithelial cells and mesenchymal stem cell-derived chondrocytes contribute to the survival of tissue-engineered airway transplants in pigs. J Thorac Cardiovasc Surg 139: p441 with permission).
MSCs can greatly improve the long-term survival and efficiency of synthetic scaffolds designed for trachea replacement [30]. For example, Suzuki et al created a fabricated trachea graft from a collagen sponge with polypropylene mesh stratified with AT MSCs embedded in a collagen gel [30]. After transplantation into tracheal defects of rats they found that addition of AT MSCs helped to promote proliferation, migration, differentiation, and vascularization of subepithelial cells. Fourteen days after implantation the luminal surface of the trachea constructs embedded with AT MSCs had pseudostratified columnar, ciliated epithelium and goblet cells growing similar to normal tracheas. It was concluded that AT MSCs can accelerate tissue regeneration and improve survival of bioengineered scaffolds for trachea replacement.
Mechanical factors may influence the long-term survival of functional airway replacements, especially after extensive tracheal resections. Naito el al developed a bioartificial trachea that is strong enough to handle the pressure differences that occur during respiration without collapsing [31]. The novel tube-shaped construct is enclosed by a ring shaped cartilage derived from BM MSCs embedded in a collagen matrix to provide reinforcement to the multi-layer tube composed of fibroblasts embedded in a collagen hydrogel. To enhance the strength of the ring shaped cartilages researchers differentiated BM MSCs into osteogenic lineages prior to seeding into the scaffold. Results from in vitro biomechanical testing demonstrated that the rings around the tube improved the stiffness of the tissue and in vivo implantation into the rat showed that the artificial trachea was able to tolerate negative pressures during inspiration for at least twenty-four hours. However, the implanted bioartificial trachea failed to adequately transport secretions, resulting in mucous adhering to the anastomotic site. These finding demonstrate the need for multiple tissue layers to provide scaffold strength and epithelial cell growth on the luminal surface of the tube to improve survival and efficiency of an artificial trachea.
More recently, researchers have investigated the use of MSCs as drug delivery vehicles in an effort to reduce the antigenicity of tracheal allografts. Han et al investigated the use of intravenous injection of BM MSCs with cryopreserved allograft trachea transplantation in rat animal models [32]. Histologic observations 4 and 8 weeks post-surgery showed the greatest fluorescence at the anastomosis region, suggesting that the labeled BM MSCs migrated from the tail vein to the transplanted trachea and localized at the implant site. Additionally, BM MSC compared to saline conditions had greater pseudostriated ciliated columnar epithelium growth covering the tracheal lumen and higher VEGF expression levels, a marker for angiogenesis. These results suggest that MSCs can help reduce allograft rejection and enhance tracheal transplant survival by promoting epithelium regeneration and revascularization.
Adipose and dermal matrix replacement
Free tissue transfers and regional flaps are commonly employed for reconstruction of oral cavity and oropharynx defects, often requiring skin grafts to cover the large soft tissue defects at the donor site. A number of factors including age, radiation effects, donor site, cardiac disease, diabetes can affect the success of these procedures and may cause delayed wound healing, flap perfusion, hypercoagulation and/or infection. MSC therapies may be a viable clinical approach for enhancing flap or graft survival and recovery of damaged tissues. Local delivery of MSCs into cutaneous wounds followed by staged reconstruction could help enhance tissue reperfusion through their secretion of trophic factors that can inhibit T-cell function and promote neovascularization [33]. Alternatively, MSCs can be embedded in biomaterial constructs and used as an adjunct therapy or reconstruction. Off the shelf soft tissue constructs, though not clinically available at this time, offer the desired shape and volume of a given defect while reducing the need for additional tissue transfer.
Much of the interest in MSCs for Otolaryngology-Head & Neck surgery focuses on wound healing and the paracrine effects of these cells, including stimulation of extracellular matrix (ECM) deposition, angiogenesis, and native cell recruitment. To date, MSC are most frequently used for soft tissue augmentation or filler as part of a fat graft due to the relative ease of the procedure and possible anti-inflammatory effects of adipose tissue transfer [34]. However, there is a paucity of clinical studies indicating problems associated with long-term safety and efficacy of autologous fat grafts. Nevertheless, clinical research has been focused on enhancing their results through the addition of adipose-derived stem cells to improve graft survival. Matsumoto and colleagues developed a novel method of concurrent transfer of lipoaspirated fat with adipose derived progenitor cells termed cell-assisted lipotransfer (CAL) [35]. In this technique, a portion of the lipoaspirated fat is processed to isolate the heterogeneous mixture of cells (i.e. vascular endothelial cells, pericytes, blood cells and MSCs) from the stromal vascular fraction (SVF); the remaining lipoaspirate is processed for fat grafting, serving as a biological scaffold for the cells. The foundation of this technology is that the enrichment of SVF containing AT MSC to lipoaspirate will improve graft survival and reduce postoperative atrophy or resorption through enhanced angiogenesis and cell self-renewal.
Cell-assisted lipotransfer has been used for augmentation or facial contouring [36]. In a small study, groups of patients with facial lipoatrophy from lupus erythematosus profundus or Parry-Romberg syndrome (idiopathic hemifacial lipoatrophy) were treated with fat injections, with or without additional SVF containing AT MSC or CAL (n=3 per group). The average volume of lipoinjection was 100 ml with cell processing taking 90 minutes. The CAL-treatment group had a better clinical improvement scores, however this was not statistically significant given the small study size. One patient in the non-CAL group was treated for fat necrosis. Here, the authors established safety of the technique in soft tissue augmentation, though larger, structured clinical trials are necessary to make further conclusions.
Purified lipoaspirate has also been used to treat wounds in 20 patients resulting from radiation therapy to the supraclavicular region [37]. In this study, lipoaspirate was centrifuged, the oil/liquid layer discarded, and remaining cell-augmented adipose tissue was injected into the wounded tissue; patients received from one to six injections, based on the severity of their wound. Outcomes measured included clinical healing, symptom improvement, and recurrence. In only one case was there no sign of improvements.
While the majority of wounds treated clinically with cell based therapies have been chronic in nature, there are reports’ of severe radiation burn injuries successfully treated with a combination of serial debridements, split thickness skin graft and MSCs injection [38]. Cells were cultured from autologous BM aspirate, and injected directly into the wound following a two-step expansion process. Cells administered were positive for surface markers characteristic of MSCs, and pluripotency confirmed with differentiation assays. Complete healing was observed within six months with no functional impairments noted. Although encouraging, the single case report nature of this study and combined use of other modalities to treat this case should be considered.
Taken together, this literature shows that the addition of MSCs to soft tissue or wounds is associated with dermal rebuilding (in addition to remodeling), an increase in vascularity, and reduced fibrosis or scarring. While these reports demonstrate the heterogeneity of the type of wounds treated with MSCs, they also illustrate the variations in culture and application techniques that limit the current body of evidence in support of MSC therapy.
Vocal fold replacement
The application of cell-scaffold tissue engineering methods may revolutionize the management of a variety of vocal fold disorders that preclude normal structure and function. Not only can the physical and chemical components of the biomaterial be augmented to have favorable viscous and elastic properties, but the addition of MSC can provide growth factors and additional ECM proteins that are needed to enhance the long-term tissue regeneration and improve the pliability of the vocal fold lamina propria.
Although BM MSCs administered alone have been shown to provide antifibrotic benefits for treatment of vocal fold scarring, their long-term engraftment and differentiation capacity in vivo is unclear. AT MSCs can directly modulate scar vocal fold fibroblasts phenotype by increasing their expression of hyaluronic acid (HA) and hepatocyte growth factor (HGF) and decreasing their α- smooth muscle actin (SMA) expression, collagen secretion and proliferation [39,40]. Consistent with in vitro studies, BM MSCs have been shown to decrease the collagen type I expression in injured rabbit vocal folds, lowering their viscoelastic properties one month after treatment [41]. By three months the viscoelastic properties and thickness of the lamina propria were similar to unscarred rabbit vocal folds [42]. However, results showed that MSCs failed to engraft into the tissue as no MSCs were found after three months. These studies demonstrate that MSCs alone can provide functional short-term benefits to the injured vocal fold, which were most likely due to the variety of bioactive molecules MSCs secrete. However, this method does not provide long-term engraftment or differentiation needed for therapeutic benefit throughout the wound healing process, which typically takes 5 to 6 months in humans.
Extensive research has been conducted in the last decade to establish an appropriate composite which resembles the biomechanical properties of the vocal fold and can regenerate a damaged tissue. Several cell-scaffold composites have been suggested using various sources of MSCs encapsulated into hydrogels composed of collagen, fibrin, hyaluronic acid (HA), or a combination [43,44]. Tissue engineered HA hydrogels, such as Carbylan-GSX have been previously shown in animal models to produce optimal biomechanical properties for the superficial layer of the lamina propria and are biocompatible and non-toxic [45]. Unlike other HA based biomaterials, the semisynthetic HA derivative in Carbylan-GSX is cross-linked to gelatin using a poly (ethylene glycol) diacrylate (PEGDA), which can increase the bioactivity of the HA material alone and create a surface that promotes cellular attachment, migration, and proliferation [46]. Previous animal studies have shown that HA based hydrogels in combination with BM MSCs can enhance tissue regeneration compared to the biomaterial alone [8]. These studies found that BM MSCs embedded in Carbylan-GSX expressed high levels of CD44 a hyaluronan receptor and increased ECM gene expression, such as HA, hyaluronidase, fibronectin, collagen, and TGF-β without increasing myofibroblast differentiation. Results suggest that the addition of BM MSCs to the hydrogel construct may facilitate ECM production and remodeling which could improve the regeneration of injured vocal folds. Further studies have also supported these findings, demonstrating that higher concentrations of HA in hydrogel scaffolds can control the differentiation of MSCs resulting in high expression of CD44 and CD105 [44]. Hanson et al, also investigated MSCs pleiotropic functions while embedded in Carbylan-GSX to compare the effects of MSCs derived from multiple tissue sources on macrophages immunophenotype [47]. They reported that macrophages in the presence of MSCs derived from BM, AT or VF tissue embedded in Carbylan-GSX displayed a more anti-inflammatory phenotype including decreased expression of CD16 and human leukocyte antigen (HLA) - DR and increase expression of CD 206 [47]. Therefore, MSCs from multiple tissue sources maintain their immunosuppressive responses while embedded in the HA hydrogel construct that could potentially expand the long-term regeneration of tissue by reducing the inflammatory response associated with the biomaterial and directly promoting tissue repair. These studies demonstrate that the inclusion of MSCs into a HA hydrogel construct may provide an optimal treatment approach for vocal fold scarring by accelerating the wound healing process through the production of bioactive molecules and restoring volume to the superficial lamina propria.
In cases where surgical damage or fibrosis affects the vocal fold cover i.e. epithelium, basement membrane and superficial lamina propria, there has been one novel approach tested in vitro as a means of replacing the entire vocal fold cover. Long et al, created a gelliquid interface bilayer by encapsulating AT MSCs in a fibrin gel and submerged it in media supplemented with epidermal growth factor (EGF) [48]. To replicate the epithelial layer AT MSCs were seeded directly on top of the gel. Preliminary findings found that AT MSCs seeded on the surface of the gel differentiated into simple epithelial phenotype expressing E-cadherin and keratin 8. However, AT MSCs inside the gel did not appear to differentiate by expressing vimentin, signifying a mesenchymal phenotype [48]. Functional preliminary studies by this group showed that the AT MSCs-fibrin construct produced similar elastic properties as the vocal fold cover from a cadaveric larynx [49]. However, it should be noted that the stiffness of the construct would not adequately represent the superficial lamina propria and could potential effect the biomechanical properties of the tissue. This work is highly innovative, but findings are incomplete and the current approach may not be ideal for clinical application as it lacks many of the unique attributes specific to the vocal fold cover. Further work is necessary in this area.
Taken together these studies collectively suggest that MSC based therapies hold great promise for prophylactic treatment of vocal fold scar or atrophy. Preclinical in vitro and in vivo studies have shown no adverse risks with MSCs transplantation into the vocal fold [8,43,45]. Interestingly, a newly developed bioreactor capable of mimicking vocal fold vibration showed that BM MSCs do not increase their production of ECM genes after eight hours of 200Hz vibration [50] (Figure 3). Therefore, MSCs transplantation into the vocal fold should be able to withstand short-periods of vibration without modulating their expression of fibrous proteins. However, further risk assessments are needed to determine the differentiation and proliferation capacity of MSCs under long-term vibratory conditions.
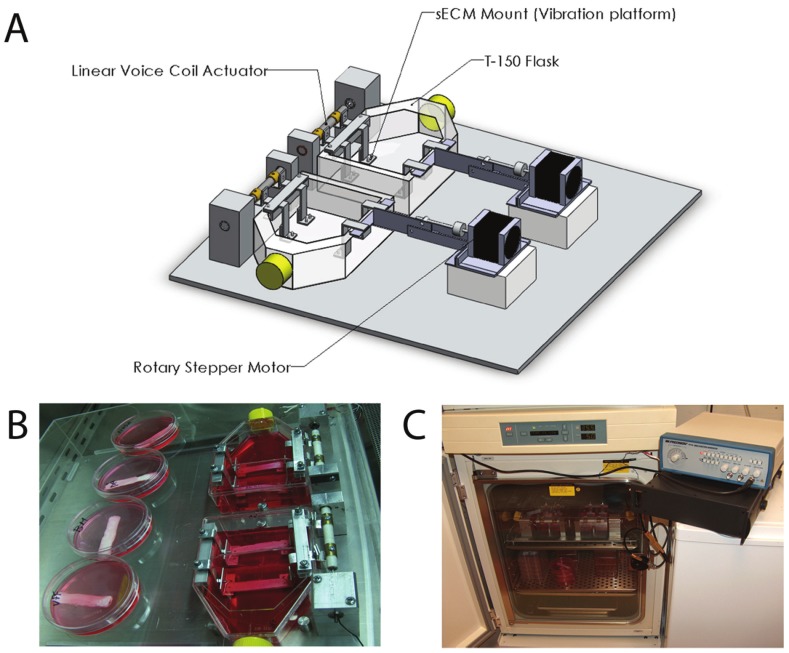
Figure 3.
Schematic of developed bioreactor. A: Bioreactor including T-flask, substrate, voice-coil actuator, linear stepper motors, rotary stepper motors, and scissor bars. B: Experimental setup, with static attachment in place of stepper motors. Non-vibrated controls can also be seen. C: Bioreactor within incubator. Wave-form generator is next to the incubator, sitting on top of the power amplifier. (Reprinted from Gaston J., Rios B.Q., Bartlett R., et al (2011). The response of vocal fold fibroblasts and mesenchymal stromal cells to vibration. PLoS ONE 7(2): p2).
Conclusions
Studies presented in this review clearly support further investigation of MSC based therapies in combination with biomaterial scaffolds or decellularized allografts to repair or replace injured tissue of the head and neck. Significant obstacles have been shown with locally injected MSCs alone for treatment of long-term regeneration, such as vocal fold scarring as cells can die or migrate away from the site prior to tissue healing. MSC only approaches may be more advantageous for suppressing the immune response caused by inflammatory diseases, allograft rejection, or autoimmune disorders.
Restoration of normal structure and function is the primary goal after head and neck surgery. Despite the numerous pre-clinical studies that have been conducted with MSCs, many of the published evidence in Otolaryngology-Head and Neck Surgery is based on animal research. As a result, the true impact of MSCs for clinical treatment of head and neck disorders will not be known until human clinical studies provide evidence of functional outcomes.
Acknowledgements
This project was supported by the NIDCD - NIH grants R01 DC4336, R01 DC9600 (S.L. Thibeault) and T32 DC009401 (S.N. King).
Conflict of interest statement
None.
References
- 1.Caplan AI. Adult mesenchymal stem cells for tissue engineering versus regenerative medicine. J Cell Physiol. 2007;213:341–347. doi: 10.1002/jcp.21200. [DOI] [PubMed] [Google Scholar]
- 2.Kim J, Hematti P. Mesenchymal stem cell-educated macrophages: A novel type of alternatively activated macrophages. Exp Hematol. 2009;37:1445–1453. doi: 10.1016/j.exphem.2009.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Phinney DG, Prockop DJ. Concise review: Mesenchymal stem/multipotent stromal cells: The state of transdifferentiation and modes of tissue repair--current views. Stem Cells. 2007;25:2896–2902. doi: 10.1634/stemcells.2007-0637. [DOI] [PubMed] [Google Scholar]
- 4.Prockop DJ. Repair of tissues by adult stem/progenitor cells (MSCs): Controversies, myths, and changing paradigms. Mol Ther. 2009;17:939–946. doi: 10.1038/mt.2009.62. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Prockop DJ, Oh JY. Mesenchymal stem/stromal cells (MSCs): Role as guardians of inflammation. Mol Ther. 2012;20:14–20. doi: 10.1038/mt.2011.211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Aggarwal S, Pittenger MF. Human mesenchymal stem cells modulate allogeneic immune cell responses. Blood. 2005;105:1815–1822. doi: 10.1182/blood-2004-04-1559. [DOI] [PubMed] [Google Scholar]
- 7.Chamberlain G, Fox J, Ashton B, Middleton J. Concise review: Mesenchymal stem cells: Their phenotype, differentiation capacity, immunological features, and potential for homing. Stem Cells. 2007;25:2739–2749. doi: 10.1634/stemcells.2007-0197. [DOI] [PubMed] [Google Scholar]
- 8.Johnson BQ, Fox R, Chen X, Thibeault S. Tissue regeneration of the vocal fold using bone marrow mesenchymal stem cells and synthetic extracellular matrix injections in rats. Laryngoscope. 2010;120:537–545. doi: 10.1002/lary.20782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Meijer GJ, de Bruijn JD, Koole R, van Blitterswijk CA. Cell based bone tissue engineering in jaw defects. Biomaterials. 2008;29:3053–3061. doi: 10.1016/j.biomaterials.2008.03.012. [DOI] [PubMed] [Google Scholar]
- 10.Macchiarini P, Jungebluth P, Go T, Asnaghi MA, Rees LE, Cogan TA, Dodson A, Martorell J, Bellini S, Parnigotto PP, Dickinson SC, Hollander AP, Mantero S, Conconi MT, Birchall MA. Clinical transplantation of a tissue-engineered airway. Lancet. 2008;372:2023–2030. doi: 10.1016/S0140-6736(08)61598-6. [DOI] [PubMed] [Google Scholar]
- 11.Zuk PA, Zhu M, Ashjian P, De Ugarte DA, Huang JI, Mizuno H, Alfonso ZC, Fraser JK, Benhaim P, Hedrick MH. Human adipose tissue is a source of multipotent stem cells. Mol Biol Cell. 2002;13:4279–4295. doi: 10.1091/mbc.E02-02-0105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wagner W, Wein F, Seckinger A, Frankhauser M, Wirkner U, Krause U, Blake J, Schwager C, Eckstein V, Ansorge W, Ho AD. Comparative characteristics of mesenchymal stem cells from human bone marrow, adipose tissue, and umbilical cord blood. Exp Hematol. 2005;33:1402–1416. doi: 10.1016/j.exphem.2005.07.003. [DOI] [PubMed] [Google Scholar]
- 13.Hanson SE, Kim J, Johnson BH, Bradley B, Breunig MJ, Hematti P, Thibeault SL. Characterization of mesenchymal stem cells from human vocal fold fibroblasts. Laryngoscope. 2010;120:546–551. doi: 10.1002/lary.20797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pierdomenico L, Bonsi L, Calvitti M, Rondelli D, Arpinati M, Chirumbolo G, Becchetti E, Marchionni C, Alviano F, Fossati V, Staffolani N, Franchina M, Grossi A, Bagnara GP. Multipotent mesenchymal stem cells with immunosuppressive activity can be easily isolated from dental pulp. Transplantation. 2005;80:836–842. doi: 10.1097/01.tp.0000173794.72151.88. [DOI] [PubMed] [Google Scholar]
- 15.Wada N, Menicanin D, Shi S, Bartold PM, Gronthos S. Immunomodulatory properties of human periodontal ligament stem cells. J Cell Physiol. 2009;219:667–676. doi: 10.1002/jcp.21710. [DOI] [PubMed] [Google Scholar]
- 16.Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. the international society for cellular therapy position statement. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905. [DOI] [PubMed] [Google Scholar]
- 17.Carrancio S, Lopez-Holgado N, Sanchez-Guijo FM, Villaron E, Barbado V, Tabera S, Diez-Campelo M, Blanco J, San Miguel JF, Del Canizo MC. Optimization of mesenchymal stem cell expansion procedures by cell separation and culture conditions modification. Exp Hematol. 2008;36:1014–1021. doi: 10.1016/j.exphem.2008.03.012. [DOI] [PubMed] [Google Scholar]
- 18.Rebelatto CK, Aguiar AM, Moretao MP, Senegaglia AC, Hansen P, Barchiki F, Oliveira J, Martins J, Kuligovski C, Mansur F, Christofis A, Amaral VF, Brofman PS, Goldenberg S, Nakao LS, Correa A. Dissimilar differentiation of mesenchymal stem cells from bone marrow, umbilical cord blood, and adipose tissue. Exp Biol Med (Maywood) 2008;233:901–913. doi: 10.3181/0712-RM-356. [DOI] [PubMed] [Google Scholar]
- 19.Kern S, Eichler H, Stoeve J, Kluter H, Bieback K. Comparative analysis of mesenchymal stem cells from bone marrow, umbilical cord blood, or adipose tissue. Stem Cells. 2006;24:1294–1301. doi: 10.1634/stemcells.2005-0342. [DOI] [PubMed] [Google Scholar]
- 20.Li G, Zhang XA, Wang H, Wang X, Meng CL, Chan CY, Yew DT, Tsang KS, Li K, Tsai SN, Ngai SM, Han ZC, Lin MC, He ML, Kung HF. Comparative proteomic analysis of mesenchymal stem cells derived from human bone marrow, umbilical cord, and placenta: Implication in the migration. Proteomics. 2009;9:20–30. doi: 10.1002/pmic.200701195. [DOI] [PubMed] [Google Scholar]
- 21.Shen JL, Huang YZ, Xu SX, Zheng PH, Yin WJ, Cen J, Gong LZ. Effectiveness of human mesenchymal stem cells derived from bone marrow cryopreserved for 23-25years. Cryobiology. 2012;64:167–75. doi: 10.1016/j.cryobiol.2012.01.004. [DOI] [PubMed] [Google Scholar]
- 22.Giannoudis PV, Einhorn TA, Marsh D. Fracture healing: The diamond concept. Injury. 2007;38(Suppl 4):S3–6. doi: 10.1016/s0020-1383(08)70003-2. [DOI] [PubMed] [Google Scholar]
- 23.Connolly JF, Guse R, Tiedeman J, Dehne R. Autologous marrow injection for delayed unions of the tibia: A preliminary report. J Orthop Trauma. 1989;3:276–282. doi: 10.1097/00005131-198912000-00002. [DOI] [PubMed] [Google Scholar]
- 24.Connolly JF, Guse R, Tiedeman J, Dehne R. Autologous marrow injection as a substitute for operative grafting of tibial nonunions. Clin Orthop Relat Res. 1991:259–270. [PubMed] [Google Scholar]
- 25.Dimitriou R, Jones E, McGonagle D, Giannoudis PV. Bone regeneration: Current concepts and future directions. BMC Med. 2011;9:66. doi: 10.1186/1741-7015-9-66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Rickert D, Sauerbier S, Nagursky H, Menne D, Vissink A, Raghoebar GM. Maxillary sinus floor elevation with bovine bone mineral combined with either autogenous bone or autogenous stem cells: A prospective randomized clinical trial. Clin Oral Implants Res. 2011;22:251–258. doi: 10.1111/j.1600-0501.2010.01981.x. [DOI] [PubMed] [Google Scholar]
- 27.Shayesteh YS, Khojasteh A, Soleimani M, Alikhasi M, Khoshzaban A, Ahmadbeigi N. Sinus augmentation using human mesenchymal stem cells loaded into a beta-tricalcium phosphate/hydroxyapatite scaffold. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2008;106:203–209. doi: 10.1016/j.tripleo.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 28.Behnia H, Khojasteh A, Soleimani M, Tehranchi A, Khoshzaban A, Keshel SH, Atashi R. Secondary repair of alveolar clefts using human mesenchymal stem cells. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2009;108:e1–6. doi: 10.1016/j.tripleo.2009.03.040. [DOI] [PubMed] [Google Scholar]
- 29.Go T, Jungebluth P, Baiguero S, Asnaghi A, Martorell J, Ostertag H, Mantero S, Birchall M, Bader A, Macchiarini P. Both epithelial cells and mesenchymal stem cell-derived chondrocytes contribute to the survival of tissue-engineered airway transplants in pigs. J Thorac Cardiovasc Surg. 2010;139:437–443. doi: 10.1016/j.jtcvs.2009.10.002. [DOI] [PubMed] [Google Scholar]
- 30.Suzuki T, Kobayashi K, Tada Y, Suzuki Y, Wada I, Nakamura T, Omori K. Regeneration of the trachea using a bioengineered scaffold with adipose-derived stem cells. Ann Otol Rhinol Laryngol. 2008;117:453–463. doi: 10.1177/000348940811700609. [DOI] [PubMed] [Google Scholar]
- 31.Naito H, Tojo T, Kimura M, Dohi Y, Zimmermann WH, Eschenhagen T, Taniguchi S. Engineering bioartificial tracheal tissue using hybrid fibroblast-mesenchymal stem cell cultures in collagen hydrogels. Interact Cardiovasc Thorac Surg. 2011;12:156–161. doi: 10.1510/icvts.2010.253559. [DOI] [PubMed] [Google Scholar]
- 32.Han Y, Lan N, Pang C, Tong X. Bone marrow-derived mesenchymal stem cells enhance cryopreserved trachea allograft epithelium regeneration and vascular endothelial growth factor expression . Transplantation. 2011;92:620–626. doi: 10.1097/TP.0b013e31822a4082. [DOI] [PubMed] [Google Scholar]
- 33.Hanson SE, Bentz ML, Hematti P. Mesenchymal stem cell therapy for nonhealing cutaneous wounds. Plast Reconstr Surg. 2010;125:510–516. doi: 10.1097/PRS.0b013e3181c722bb. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Gutowski KA, ASPS Fat Graft Task Force. Current applications and safety of autologous fat grafts: A report of the ASPS fat graft task force. Plast Reconstr Surg. 2009;124:272–280. doi: 10.1097/PRS.0b013e3181a09506. [DOI] [PubMed] [Google Scholar]
- 35.Matsumoto D, Sato K, Gonda K, Takaki Y, Shigeura T, Sato T, Aiba-Kojima E, Iizuka F, Inoue K, Suga H, Yoshimura K. Cell-assisted lipotransfer: Supportive use of human adiposederived cells for soft tissue augmentation with lipoinjection. Tissue Eng. 2006;12:3375–3382. doi: 10.1089/ten.2006.12.3375. [DOI] [PubMed] [Google Scholar]
- 36.Yoshimura K, Sato K, Aoi N, Kurita M, Hirohi T, Harii K. Cell-assisted lipotransfer for cosmetic breast augmentation: Supportive use of adipose-derived stem/stromal cells. Aesthetic Plast Surg. 2008;32:48–55. doi: 10.1007/s00266-007-9019-4. discussion 56-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rigotti G, Marchi A, Galie M, Baroni G, Benati D, Krampera M, Pasini A, Sbarbati A. Clinical treatment of radiotherapy tissue damage by lipoaspirate transplant: A healing process mediated by adipose-derived adult stem cells. Plast Reconstr Surg. 2007;119:1409–22. doi: 10.1097/01.prs.0000256047.47909.71. discussion 1423-4. [DOI] [PubMed] [Google Scholar]
- 38.Lataillade JJ, Doucet C, Bey E, Carsin H, Huet C, Clairand I, Bottollier-Depois JF, Chapel A, Ernou I, Gourven M, Boutin L, Hayden A, Carcamo C, Buglova E, Joussemet M, de Revel T, Gourmelon P. New approach to radiation burn treatment by dosimetry-guided surgery combined with autologous mesenchymal stem cell therapy. Regen Med. 2007;2:785–794. doi: 10.2217/17460751.2.5.785. [DOI] [PubMed] [Google Scholar]
- 39.Kumai Y, Kobler JB, Park H, Galindo M, Herrera VL, Zeitels SM. Modulation of vocal fold scar fibroblasts by adipose-derived stem/stromal cells. Laryngoscope. 2010;120:330–337. doi: 10.1002/lary.20753. [DOI] [PubMed] [Google Scholar]
- 40.Kumai Y, Kobler JB, Park H, Lopez-Guerra G, Karajanagi S, Herrera VL, Zeitels SM. Crosstalk between adipose-derived stem/stromal cells and vocal fold fibroblasts in vitro. Laryngoscope. 2009;119:799–805. doi: 10.1002/lary.20149. [DOI] [PubMed] [Google Scholar]
- 41.Hertegard S, Cedervall J, Svensson B, Forsberg K, Maurer FH, Vidovska D, Olivius P, Ahrlund-Richter L, Le Blanc K. Viscoelastic and histologic properties in scarred rabbit vocal folds after mesenchymal stem cell injection. Laryngoscope. 2006;116:1248–1254. doi: 10.1097/01.mlg.0000224548.68499.35. [DOI] [PubMed] [Google Scholar]
- 42.Svensson B, Nagubothu RS, Cedervall J, Le Blanc K, Ahrlund-Richter L, Tolf A, Hertegard S. Injection of human mesenchymal stem cells improves healing of scarred vocal folds: Analysis using a xenograft model. Laryngoscope. 2010;120:1370–1375. doi: 10.1002/lary.20926. [DOI] [PubMed] [Google Scholar]
- 43.Duflo S, Thibeault SL, Li W, Shu XZ, Prestwich G. Effect of a synthetic extracellular matrix on vocal fold lamina propria gene expression in early wound healing. Tissue Eng. 2006;12:3201–3207. doi: 10.1089/ten.2006.12.3201. [DOI] [PubMed] [Google Scholar]
- 44.Park H, Karajanagi S, Wolak K, Aanestad J, Daheron L, Kobler JB, Lopez-Guerra G, Heaton JT, Langer RS, Zeitels SM. Three-dimensional hydrogel model using adipose-derived stem cells for vocal fold augmentation. Tissue Eng Part A. 2010;16:535–543. doi: 10.1089/ten.TEA.2009.0029. [DOI] [PubMed] [Google Scholar]
- 45.Chen X, Thibeault SL. Biocompatibility of a synthetic extracellular matrix on immortalized vocal fold fibroblasts in 3-D culture. Acta Biomater. 2010;6:2940–2948. doi: 10.1016/j.actbio.2010.01.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Duflo S, Thibeault SL, Li W, Shu XZ, Prestwich GD. Vocal fold tissue repair in vivo using a synthetic extracellular matrix. Tissue Eng. 2006;12:2171–2180. doi: 10.1089/ten.2006.12.2171. [DOI] [PubMed] [Google Scholar]
- 47.Hanson SE, King SN, Kim J, Chen X, Thibeault SL, Hematti P. The effect of mesenchymal stromal cell-hyaluronic acid hydrogel constructs on immunophenotype of macrophages. Tissue Eng Part A. 2011;17:2463–2471. doi: 10.1089/ten.tea.2010.0716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Long JL, Zuk P, Berke GS, Chhetri DK. Epithelial differentiation of adipose-derived stem cells for laryngeal tissue engineering. Laryngoscope. 2010;120:125–131. doi: 10.1002/lary.20719. [DOI] [PubMed] [Google Scholar]
- 49.Long JL, Neubauer J, Zhang Z, Zuk P, Berke GS, Chhetri DK. Functional testing of a tissue-engineered vocal fold cover replacement. Otolaryngol Head Neck Surg. 2010;142:438–440. doi: 10.1016/j.otohns.2009.11.020. [DOI] [PubMed] [Google Scholar]
- 50.Gaston J, Quinchia Rios B, Bartlett R, Berchtold C, Thibeault SL. The response of vocal fold fibroblasts and mesenchymal stromal cells to vibration. PLoS One. 2012;7:e30965. doi: 10.1371/journal.pone.0030965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ohno S, Hirano S, Tateya I, Kanemaru S, Umeda H, Suehiro A, Kitani Y, Kishimoto Y, Kojima T, Nakamura T, Ito J. Atelocollagen sponge as a stem cell implantation scaffold for the treatment of scarred vocal folds. Ann Otol Rhinol Laryngol. 2009;118:805–810. [PubMed] [Google Scholar]
- 52.Xu W, Hu R, Fan E, Han D. Adipose-derived mesenchymal stem cells in collagen-hyaluronic acid gel composite scaffolds for vocal fold regeneration. Ann Otol Rhinol Laryngol. 2011;120:123–130. doi: 10.1177/000348941112000209. [DOI] [PubMed] [Google Scholar]
- 53.Ohno S, Hirano S, Kanemaru S, Kitani Y, Kojima T, Tateya I, Nakamura T, Ito J. Implantation of an atelocollagen sponge with autologous bone marrow-derived mesenchymal stromal cells for treatment of vocal fold scarring in a canine model. Ann Otol Rhinol Laryngol. 2011;120:401–408. doi: 10.1177/000348941112000610. [DOI] [PubMed] [Google Scholar]
- 54.Kanemaru S, Nakamura T, Omori K, Kojima H, Magrufov A, Hiratsuka Y, Hirano S, Ito J, Shimizu Y. Regeneration of the vocal fold using autologous mesenchymal stem cells. Ann Otol Rhinol Laryngol. 2003;112:915–920. doi: 10.1177/000348940311201101. [DOI] [PubMed] [Google Scholar]
- 55.Kanemaru S, Nakamura T, Yamashita M, Magrufov A, Kita T, Tamaki H, Tamura Y, Iguchi F, Kim TS, Kishimoto M, Omori K, Ito J. Destiny of autologous bone marrow-derived stromal cells implanted in the vocal fold. Ann Otol Rhinol Laryngol. 2005;114:907–912. doi: 10.1177/000348940511401203. [DOI] [PubMed] [Google Scholar]
- 56.Du XF, Kwon SK, Song JJ, Cho CG, Park SW. Tracheal reconstruction by mesenchymal stem cells with small intestine submucosa in rabbits. Int J Pediatr Otorhinolaryngol. 2012;76:345–351. doi: 10.1016/j.ijporl.2011.12.005. [DOI] [PubMed] [Google Scholar]
- 57.Kobayashi K, Suzuki T, Nomoto Y, Tada Y, Miyake M, Hazama A, Wada I, Nakamura T, Omori K. A tissue-engineered trachea derived from a framed collagen scaffold, gingival fibroblasts and adipose-derived stem cells. Biomaterials. 2010;31:4855–4863. doi: 10.1016/j.biomaterials.2010.02.027. [DOI] [PubMed] [Google Scholar]
- 58.Gray FL, Turner CG, Ahmed A, Calvert CE, Zurakowski D, Fauza DO. Prenatal tracheal reconstruction with a hybrid amniotic mesenchymal stem cells-engineered construct derived from decellularized airway. J Pediatr Surg. 2012;47:1072–1079. doi: 10.1016/j.jpedsurg.2012.03.006. [DOI] [PubMed] [Google Scholar]
- 59.Elliott MJ, De Coppi P, Speggiorin S, Roebuck D, Butler CR, Samuel E, Crowley C, McLaren C, Fierens A, Vondrys D, Cochrane L, Jephson C, Janes S, Beaumont NJ, Cogan T, Bader A, Seifalian AM, Hsuan JJ, Lowdell MW, Birchall MA. Stem-cell-based, tissue engineered tracheal replacement in a child: A 2-year followup study. Lancet. 2012;380:994–1000. doi: 10.1016/S0140-6736(12)60737-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yoshioka M, Tanimoto K, Tanne Y, Sumi K, Awada T, Oki N, Sugiyama M, Kato Y, Tanne K. Bone regeneration in artificial jaw cleft by use of carbonated hydroxyapatite particles and mesenchymal stem cells derived from iliac bone. Int J Dent. 2012;2012:352510. doi: 10.1155/2012/352510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Weir MD, Xu HH. Culture human mesenchymal stem cells with calcium phosphate cement scaffolds for bone repair. J Biomed Mater Res B Appl Biomater. 2010;93:93–105. doi: 10.1002/jbm.b.31563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Castro-Govea Y, Cervantes-Kardasch VH, Borrego-Soto G, Martinez-Rodriguez HG, Espinoza-Juarez M, Romero-Diaz V, Marino-Martinez IA, Robles-Zamora A, Alvarez-Lozano E, Padilla-Rivas GR, Ortiz-Lopez R, Lara-Arias J, Vazquez-Juarez J, Rojas-Martinez A. Human bone morphogenetic protein 2-transduced mesenchymal stem cells improve bone regeneration in a model of mandible distraction surgery. J Craniofac Surg. 2012;23:392–396. doi: 10.1097/SCS.0b013e318240fe9b. [DOI] [PubMed] [Google Scholar]
- 63.Stephan SJ, Tholpady SS, Gross B, Petrie-Aronin CE, Botchway EA, Nair LS, Ogle RC, Park SS. Injectable tissue-engineered bone repair of a rat calvarial defect. Laryngoscope. 2010;120:895–901. doi: 10.1002/lary.20624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mesimaki K, Lindroos B, Tornwall J, Mauno J, Lindqvist C, Kontio R, Miettinen S, Suuronen R. Novel maxillary reconstruction with ectopic bone formation by GMP adipose stem cells. Int J Oral Maxillofac Surg. 2009;38:201–209. doi: 10.1016/j.ijom.2009.01.001. [DOI] [PubMed] [Google Scholar]
- 65.Chang SC, Chuang HL, Chen YR, Chen JK, Chung HY, Lu YL, Lin HY, Tai CL, Lou J. Ex vivo gene therapy in autologous bone marrow stromal stem cells for tissue-engineered maxillofacial bone regeneration. Gene Ther. 2003;10:2013–2019. doi: 10.1038/sj.gt.3302106. [DOI] [PubMed] [Google Scholar]
- 66.Park DJ, Choi JH, Leong KW, Kwon JW, Eun HS. Tissue-engineered bone formation with gene transfer and mesenchymal stem cells in a minimally invasive technique. Laryngoscope. 2007;117:1267–1271. doi: 10.1097/MLG.0b013e31805f680e. [DOI] [PubMed] [Google Scholar]