Abstract
The development of a preventative HIV/AIDS vaccine is challenging due to the diversity of viral genome sequences, especially in the viral envelope (Env160). Since it is not possible to directly match the vaccine strain to the vast number of circulating HIV-1 strains, it is necessary to develop an HIV-1 vaccine that can protect against a heterologous viral challenge. Previous studies from our group demonstrated that a mixture of wild type clade B Envgp160s were able to protect against a heterologous clade B challenge more effectively than a consensus clade B Envgp160 vaccine. In order to broaden the immune response to other clades of HIV, in this study rhesus macaques were vaccinated with a polyvalent mixture of purified HIV-1 trimerized consensus Envgp140 proteins representing clades A, B, C, and E. The elicited immune responses were compared to a single consensus Envgp140 representing all isolates in group M (Con M). Both vaccines elicited anti- Envgp140 IgG antibodies that bound an equal number of HIV-1 Envgp160 proteins representing clades A, B and C. In addition, both vaccines elicited antibodies that neutralized the HIV-1SF162 isolate. However, the vaccinated monkeys were not protected against SHIVSF162p4 challenge. These results indicate that consensus Envgp160 vaccines, administered as purified Envgp140 trimers, elicit antibodies that bind to Envgp160s from strains representing multiple clades of HIV-1, but these vaccines did not protect against heterologous SHIV challenge.
Introduction
One of the greatest struggles for developing a preventative human immunodeficiency virus (HIV)/acquired immunodeficiency syndrome (AIDS) vaccine is overcoming the diversity of viral isolates [1]. The Envgp160 sequences can differ up to 35% between clades and ~15% within a specific clade [2]. Viruses classified as clade B are responsible for ≥40% of infections in the Americas and Europe, but in Asia and sub-Saharan Africa, where most new infections are recorded each year, other clades are dominant. Most new infections in these regions are classified as clades A, C, or A/E viruses [1,3]. Any HIV vaccine that will prevent infection must be able to overcome the diversity of HIV sequences.
To overcome the HIV sequence diversity, polyvalent mixture of antigens and consensus proteins were designed [4-7]. Polyvalent vaccines increase breadth by including multiple copies of a target (s) or epitopes into a single formulation. Polyvalent vaccine strategies have been employed to increase the breadth of the humoral and cellular immune responses [8,9]. Polyvalent mixtures of Envgp140 or HIV proteins (Gag-Pol, Tat and trimeric Envgp140) elicit a degree of protection against heterologous challenge [8,10]. Consensus-based vaccines rely on a centralized antigen designed to reduce sequence diversity by using the most common amino acid at each position of the protein. Consensus vaccines are designed to reduce the genetic differences between the vaccine and the primary isolate and increase the breadth of immune responses [11-14].
To overcome the diversity in Envgp160 sequences and to design a more effective AIDS vaccine, consensus Envgp140 sequences were designed for 4 clades of HIV-1 (A, B, C, and E), as well as a single consensus Envgp160 representing isolates from all of Group M. For the first time, in the same study, consensus A, B, C, and E Envgp140 sequences were used in a polyvalent vaccine mixture, and compared to a Con M Envgp160, to assess the ability to elicit a broadly reactive anti-Envgp160 immune response. The immunological responses of the polyvalent mixture in vaccinated rhesus macaques were compared to that of the single Con M Envgp140 vaccine. Both vaccines elicited anti-Env immune responses against multiple clades of HIV; however neither vaccine strategy efficiently protected monkeys against a SHIVSF162p4 challenge.
Results
Characterization of consensus envelopes
The goal of this study was to design a HIV Envgp160 vaccine that elicits broadly reactive immune responses in an effort to overcome the inherent diversity in the Envgp160. Therefore, an HIV-1 group M consensus Envgp140 vaccine was compared to a polyvalent mixture of clade consensus Envgp140s representing 4 individual clades of HIV-1 (A, B, C, and E). The env gene sequences were then truncated at the transmembrane domain, and the cleavage site mutated, to generate a Envgp140[15]. To stabilize the truncated Envgp140 trimers, the bacteriophage fibronectin domain (FT) was added to the 3’ end of the Envgp140 sequence, as previously described [15].
Purified trimerized Envs were detected at ~480kDa size indicating oligomerization as trimer proteins (Figure 1A). Some Env dimers were observed in consensus C, E and M Envgp140 protein fractions. To probe the antigenic structure, the broadly reactive monoclonal antibody b12 [16] was used to determine binding kinetics to each consensus envelope by surface plasmon resonance (SPR) on a Biacore 3000 (Figure 1B and Additional file 1: Figure S1). The rate of association between the consensus Envgp140 trimers and b12 was similar to the rate of association between b12 and primary Envgp140 trimers. The rate of dissociation of b12 from all the Envgp140 trimers was similar, except for consensus B, which had a slower rate of disassociation. Each Envgp140 bound to the primary HIV receptor, human CD4 (hCD4) (Figure 1C). The MAb b12 is a monoclonal isolated from an infected patient with a consensus B wild-type Envgp160 on its viral surfaces and therefore may recognize clade B Envgp160 with more efficiency than non-clade B Envgp160s. In addition, consensus B envelopes are bound with higher affinity antibodies to clade B Envgp160s, but less so with consensus C Envgp140. The reverse is true as well. A polyclonal serum, HIV-Ig, was obtained from the AIDS Reference and Reagent Program. This polyclonal serum, collected from a clade B infected person, may not recognize the consensus E Envgp160 with the same affinity and efficiency as the consensus B Envgp160.
Figure 1.
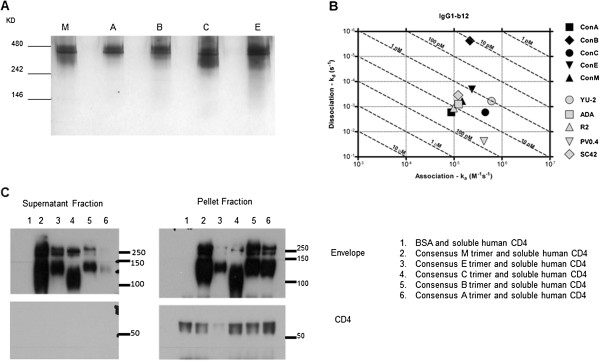
Characterization of consensus Envgp140s. A) 1 μg of each purified consensus Envgp140 was loaded onto a native PAGE and then sliver stained. The top of the gel is labeled with the consensus Envgp140 present in the lane and the protein ladder values are present on the y-axis. B) Using surface plasmon resonance, the interaction of the consensus Envgp160s and b12 was investigated in solution. The x-axis gives the rates of association between b12 and the Envgp140 and the y-axis gives the dissociation between the Envgp160s and b12. The consensus trimers are indicated by the darker symbols on the graph and the primary envelope trimers are indicated by the lighter symbols. C) The consensus Envgp140s and negative control BSA was pre-incubated with Histag soluble human CD4 at 37°C then mixed in with magnetic beads that were pre-incubated with the anti-his antibody. Using immunoprecipitation (IP), sCD4 and anything bound to it was pulled down in the pellet fraction. Supernatants and pellet fraction was analyzed by SDS-PAGE and western blot using anti-his and anti- Envgp160 antibodies. Left panel: supernatants fraction. Right panel: pellet fraction (sCD4 and proteins bound). The protein ladder values on y-axis. The upper blots were probed with rabbit polyclonal anti- Envgp160 antibody and the bottom blots were probed with mouse anti-human CD4 antibody (Clone RFT-4g mouse IgG, SouthernBiotech). Lane 1: BSA, Lane 2: Con M Envgp140, Lane 3: Consensus E Envgp140, Lane 4: Consensus C Envgp140, Lane 5: Consensus B Envgp140, Lane 6: Consensus A Envgp140.
Vaccination of non-human primates with consensus envelopes
To determine the ability of the vaccines to induce a protective response in non-human primates, rhesus macaques were vaccinated intramuscularly with 300 μg of total Envgp140 protein either Con M or the polyvalent Con Mixture (equal amounts of each Envgp140 protein). Animals were treated according to the guidelines of the IACUC of the University of Pittsburgh. All the protocols used were approved by the IACUC of the University of Pittsburgh (#1002617). Monkeys were administered 3 vaccinations of Envgp140 formulated with Imject® alum adjuvant. Mock vaccinated monkeys were administered PBS formulated with adjuvant (Table 1). Monkeys vaccinated with Con M or polyvalent consensus developed anti-Envgp140 antibody titers greater than 1:400 dilution against all consensus Envgp160s following the three vaccinations (Figure 2A). Collected sera were tested for the ability to bind to a set of primary Envgp160s representing clades A, B, C, and E (Table 2). The diversity of the Envgp160s chosen, as well as their similarity to the consensus Envgp160s, is displayed in the phylogenetic tree where Envgp160s cluster into their identified clades (Figure 2B). The Con M did not cluster with any one clade, but as expected, was located in a more central position on the tree.
Table 1.
Vaccine groups and regimen of non-human primate study
| Vaccine group | Animal numbers | Vaccine given | Other treatment |
|---|---|---|---|
| Group 1 |
N1,N2,N3,N4 |
Adjuvant only |
|
| Group 2 |
P1,P2,P3,P4 |
Mixture of Con A,B,C,E Env gp140 |
|
| Group 3 | M1,M2,M3,M4 | Con M Env gp140 | Depletion of CD8+ T cells |
Each animal was vaccinated 3 times week 0, 4 and 8. Each vaccine was given in combination alum imject© as an adjuvant. The table gives the vaccine groups, the identification number of animals in each group and the vaccine and treatment (in the case of group 3) received by the animals.
Figure 2.
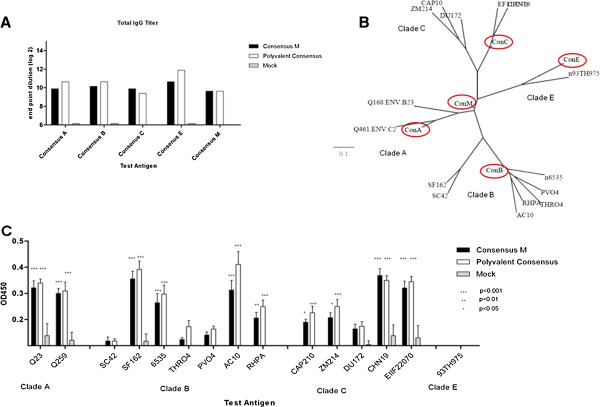
Binding breadth of vaccinated NHPs sera. NHP were vaccinated at weeks 0, 4 and 8 with blood collected 14 days after each vaccination. Vaccines were formulated with 300 μg of purified protein and Imject® alum and delivered intramuscularly. A) Sera collected on day 35 was pooled and used to determine total IgG via ELISA for each vaccine group. Bar values represent the geometric mean titer (+95% confidence interval) of log2 transformed titers. The endpoint titer is described on the y–axis and the identified Envgp140 trimer antigen is described on the x-axis. B) The unrooted phylogenetic tree was produced using Phylogeny.fr web service and the 14 HIV-1 Envgp160 sequences based upon the list of Envgp160s in Table 2 showing. The Envgp160s were from clades A, B, C, E from 1993-2005. The clades are indicated on the tree and the consensus Envgp160s are circled. C) At day 35 post-vaccination, anti-Envgp160 IgG was detected in the serum samples (1:100 dilution) against a panel of primary Envgp120s from clades A, B, C and E via ELISA. Bar values represent the geometric mean titer (95% confidence interval) at an OD450. The OD450 values are displayed on the y-axis and the Envgp120s used are listed on the x-axis. A two-way ANOVA with Bonferroni’s post-test was used to evaluate Statistical significance between the vaccines for each test antigen. A p-value of less than 0.05 was considered significant.
Table 2.
Information of envelopes used for assays
| Env ID | Clade | Location | Mode of transmission | Length of infection | Mo/yr isolated | Corecepter |
|---|---|---|---|---|---|---|
| Du172.17 |
C |
South Africa |
M-F |
12 weeks |
Nov-98 |
R5 |
| ZM214M.PL15 |
C |
Zambia |
F-M |
<13 weeks |
Jul-03 |
R5 |
| CAP210.2.00.E8 |
C |
South Africa |
M-F |
5 weeks |
May-05 |
R5 |
| CHN19 |
C |
China |
|
|
|
R5 |
| HIV16936-2 EF117270 |
C |
India |
F-M |
1 week |
Nov-00 |
R5 |
| Q168.ENV.B23 |
A |
Kenya |
M-F |
1 week |
|
R5 |
| Q461.ENV.C2 |
A |
Kenya |
M-F |
4 weeks |
|
R5 |
| HIV env 6235 clone 3 |
B |
USA |
M-M |
6 weeks |
Mar-95 |
R5 |
| PVO clone 4 |
B |
Italy |
M-M |
4 weeks |
Jan-96 |
R5 |
| pRHPA 4259 clone7 |
B |
USA |
M-F |
< weeks |
Dec-00 |
R5 |
| pTHRO4156 clone 18 |
B |
USA |
M-M |
1 week |
Aug-00 |
R5 |
| SC 422661.8 |
B |
Trinidad |
F-M |
4 weeks |
Jan-95 |
R5 |
| SF162 |
B |
USA |
|
|
|
R5 |
| 93TH975-15 | E | Haiti | F-M | <18 weeks | Jan-93 | R5 |
Primary envelopes from various clade was chosen based to have a diversity of locations and mode of mocusal transmission. Envelopes were used to test the presence of binding and neutralizing antibodies elicited by vaccination of consensus vaccines in non-human primates. SF162 was included because it is the challenge virus envelope.
Both Con M and polyvalent consensus Envgp140 vaccines elicited anti- Envgp140 antibodies that recognized Envgp160s from clade A, B and C. However, the Envgp160 SC42, THRO4, PVO4, (clade B), DU172 (clade C) and 93TH975 (clade E) were not significantly recognized by sera collected from vaccinated animals (Figure 2C). Overall, there was no binding preference of the elicited anti-Envgp160 antibodies to primary Envgp160s based on clade, location, or year of Envgp160 isolation.
SHIVSF162p4 challenge
To evaluate the protective efficacy of each vaccine, monkeys were challenged rectally four weeks after final vaccination with SHIVSF162p4 (dose: 640TCID50) to ensure all mock vaccinated animals become infected. All mock vaccinated and polyvalent consensus vaccinated monkeys were infected following challenge (Figure 3A and B). Viral loads peaked at day 14 post-challenge at ~1 × 10e+6 RNA copies/ml and then declined to undetectable levels by day 80 post-infection as seen in previous studies using the same stock of virus (unpublished and [8]. Two out of four monkeys (M1 and M4) vaccinated with Con M vaccine had no detectable virus at any time point post-challenge (Figure 3C). Monkeys M2 and M3 had a similar viral pattern as mock vaccinated monkeys with a peak at day 14, followed by a rapid decline.
Figure 3.
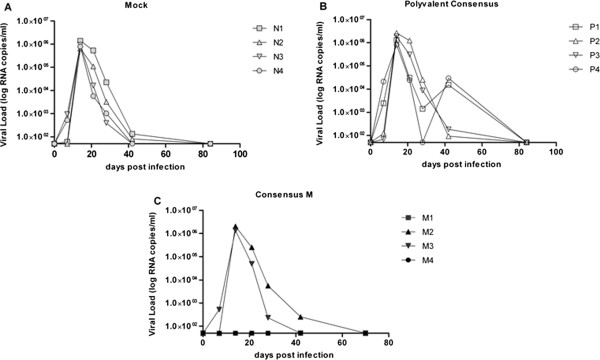
Heterologous virus challenge. Monkeys were challenged rectally with 640 TCID50 SHIV SF162p4 at week 12 post initial vaccination. Viral titers were determined from collected sera. Viral titers for each individual monkey are displayed as RNA copies/ml of blood on the y-axis by days post challenge on the x-axis. A) Mock vaccinated animals (Imject© adjuvant only). B) Polyvalent Consensus Envgp140 vaccine C) Con M Envgp140 vaccine.
To determine whether Con M Envgp140 vaccinated monkeys were protected from infection, at day 70 post-challenge all Con M Envgp140 vaccinated monkeys were depleted of CD8+ cells by administering monoclonal antibody M-T807R1 intravenously [17]. Seven days following antibody administration, no CD8+ cells were detected in the peripheral blood that was sustained for an additional 18 days (Figure 4A). Previously infected monkeys M2 and M3 had a re-emergence of virus during this CD8+-depletion period (Figure 4B). Monkey M4 who was initially aviremic after challenge had an emergence of virus after CD8+-depletion. In contrast, monkey M1 maintained undetectable viral loads following CD8+ depletion.
Figure 4.
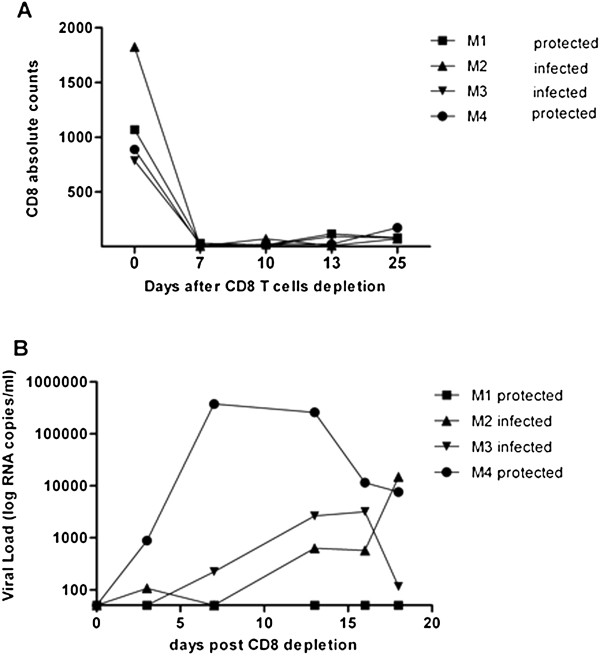
CD8+ T cell depletion. Animals vaccinated with Con M Envgp140 were all depleted of CD8+ cells by administering the M-T807R1 antibody subcutaneously (50 mg/Kg) at day 0 (day 70 post challenge). A) Number of CD8+ cells following antibody administration over the 25 day period of observation. B) Viral titers for each individual monkey are displayed as RNA copies/ml of blood on the y-axis by days post challenge on the x-axis.
Responses to the challenge envelope SF162
At 2 weeks after final vaccination (day -14 prior to challenge), anti-EnvSF162 IgG antibodies were detected in monkeys vaccinated with either vaccine (Figure 2C). In addition, these antibodies were able to neutralize the ability of HIVSF162 to infect cells in vitro (Figure 5A). The presence of neutralization titers to all Envgp160s used in the ELISA binding assay were investigated, however, only titers to HIVSF162 Envgp160 was observed. All vaccinated monkeys had a neutralizing titer of 1:40, except for monkey M4 that had a titer of 1:320. Two weeks following challenge, only monkey P4 had an increase in neutralizing titers. There were detectable neutralization titers in 3 of the 4 mock vaccinated monkeys two weeks post-challenge (Figure 5A). No monkey had antibodies that recognized a set of overlapping SF162 Envgp160 peptides. These pools of peptides represented the SF162 Envgp160 regions V1/V2, V1 only, or V2 only (data not shown). Interestingly, there was no detectable IFN-γ Envgp160 or gag-specific T-cell responses prior to challenge or 7 days post-challenge. At day 14 and 21 post-challenge, IFN-γ T cell responses were detected in all monkeys (Figure 5B), with no significant differences in the number of cells between groups.
Figure 5.
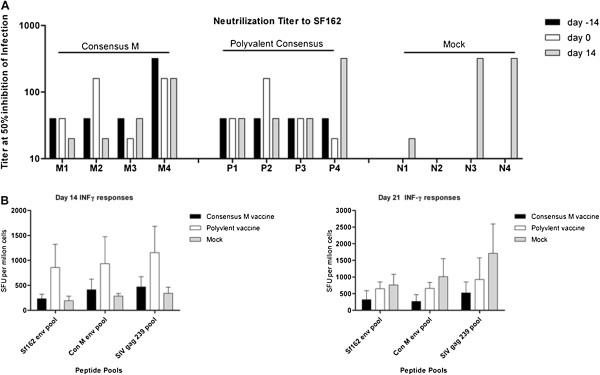
Neutralizing titers to the SF162 Envgp160 and post challenge cellular responses. A) Neutralization of HIVSF162 by serum collected at day 21 and 35 post-vaccination and 14 post-challenge. The pooled sera dilution at which 50% of virus infection is inhibited is displayed on the y-axis. Neutralizing titers for individuals monkeys are listed on the x-axis by each vaccine group. B) INF-γ ELISPOTs were performed using monkey PBMCs collected 14 (left panel) and 21 (right panel) days post-challenge. 1x105 PBMCs were stimulated with 30 ug of Gag peptide pools representing SIVmac239 or Envgp160 pools representing either Con M or SF162p3 (overlapping peptides, 15-mers with11 amino acid overlap NIH AIDS Research and Reference Reagent program Env 211 peptides and Gag 125 peptides). Spot forming units (SFU) per one million PBMCs are listed on the y-axis and the peptide pools are listed on the x-axis.
Discussion
HIV-1 Envgp160 based vaccines protect monkeys against a homologous SHIV challenges [18,19]. The ability to match the gene sequences used in the vaccine to the possible exposure virus in humans is not possible. Therefore, studies that use a matched Envgp160 in the vaccine to the challenge strain is appropriate for proof-of concept studies, but our group set a more challenging goal to protect against a challenge virus with a mismatched vaccine with a limited number of vaccinations. We report here that each consensus sequence representing clade A, B, C, E, in a polyvalent mixture or as a single consensus Envgp160 representing the entire Group M elicited anti-Envgp140 antibodies that bound to a broad panel of HIV-1 Envgp160s. The immunization schedule used was not optimal for antibody affinity maturation; nonetheless, the regimen did induce modest neutralizing antibody titers to the challenge Envgp160. However, the elicited immunity did not prevent infection by SHIVSF162p4.
In previous studies, consensus sequences designed for clades B and C Gag and Envgp140 elicited increased breadth of humoral and cellular immune responses [12,20-22]. Consensus Envgp160 sequences representing Group M, termed CON-S, elicited antibodies that neutralized multiple Envgp160s, as well as eliciting cross-clade cellular immune responses [13,14]. However, viral challenges of CON-S vaccinated monkeys were not reported and therefore the efficacy of the induced immune response elicited by these vaccines is unknown.
Compared to Envgp120 monomers, Envgp140 trimers may expose binding and neutralizing epitopes that are present only in Env’s quaternary state [23,24]. Our consensus Envgp140 trimers have similar antigenic properties as wild-type Envgp160s, as demonstrated by attaching to human CD4 and binding to the monoclonal antibody b12. The b12 antibody recognizes a conserved region on gp120 mapped to a discontinuous epitope overlapping the CD4 binding site [25].
Following three intramuscular vaccinations, all monkeys seroconverted by day 14 following the final vaccination. Nonetheless, there were differences in the vaccine efficacy following challenge between the two vaccine groups. Both non-neutralizing and neutralizing antibodies have been implicated in reducing rates of infection by HIV-1 [26,27]. A report based on the analysis of the sera samples of vaccinated volunteers in the RV144 clinical trial stated that the vaccine elicited antibodies against the V2 region of the HIV-1 Envgp160 were correlated with lower rates of HIV infection [28]. Antisera collected from these vaccinated individuals did not neutralize the infection in vitro. Additionally, vaccine induced protection against a neutralization resistant virus in macaques was correlated with antibodies to the V2 region of Envgp160[29]. Whether antibodies that bind to the V2 region are correlated with protection against SHIVSF162p4 infection in this study is unclear. There were no antibodies elicited in monkeys vaccinated with Con M or polyvalent consensus Envgp140 vaccines that recognized SF162 Envgp160 linear peptides, including those specific to V2 (data not shown). Further studies are necessary to determine if the two protected animals in the Con M Envgp140 group elicited antibodies recognizing conformational epitopes, such as the V1/V2 scaffold proteins. The V1/V2 scaffold was used to analyze human sera collected from vaccinated volunteers in the RV144 clinical trial [30,31]. Determining if antibodies specific to various conformational epitopes on Envgp160 may explain the differences observed in vaccinated animals following SHIV challenge.
Two monkeys that had no detectable viral levels following SHIV infection were M1 and M4. M2 and M3 had detectable viral levels and therefore were not protected against infection. Monkeys vaccinated with either polyvalent consensus or Con M Envgp140 trimers had neutralizing titers to HIV-1SF162. Neutralizing antibodies against Envgp160 can protect monkeys against viral challenge [32,33]. However, only one monkey (M4) in the present study had high neutralizing antibodies (1:320) against SHIVSF162p4 and had undetectable viral titer 14 days after challenge. However, following CD8+ T cell depletion, virus was detected (<1×105 RNA copies /ml) in the blood indicating that infection was not blocked, but may have been controlled by the vaccine elicited antibodies. T cell responses did not appear to play a role in protecting the monkeys from infection. There was no difference in the number or kinetics in the elicitation of Envgp160 or Gag specific IFNγ producing cells following challenge in any of the vaccinated monkeys compared to mock vaccinated animals.
Upon CD8+ depletion, it was not unexpected that M2 and M3 had a rebound in blood titer virus, but the detection M4 was unexpected. In contrast to monkey M4, no virus was detected in monkey M1 even after depletion of CD8+ T cells. Both IFNγ specific T cells and neutralizing antibodies were detected, but it is unclear which of these immune responses may have contributed to the protection. In addition, the MHC class I haplotype did not appear to correlate with protection. Even though no viremia was ever detected in monkey M1, it is possible that virus could be located in reservoirs, such as the bone marrow or gut mucosa [34]. The M-T807R1 monoclonal antibody used for CD8+ T cell depletion is specific for cells in the serum and lymph nodes [35], therefore, it may have not depleted cells in reservoirs of hidden virus. In an effort to identify possible reasons for M1 protection the animals’ halotypes were determined. Monkey M1 had a Mamu-B*008 MHC class I haplotype, which has been associated with control of SIVmac239 virus; the parent virus of the challenge SHIVSF162p4[36]. Therefore, a combination of the neutralizing antibodies, non-neutralizing antibodies and the Mamu-B*008 MHC class I haplotype may have resulted in “sterilizing” protection after viral challenge. However, the Mamu-B*008 MHC class I haplotype was also present in monkey M3, which had similar binding and neutralizing antibody titers as monkey M1, but was not protected from SHIV infection. Interestingly, two monkeys vaccinated with the polyvalent consensus vaccine, P2 and P4, had a rebound in viral titers at day 40 post-infection before returning to undetectable levels (Figure 3B). The rebound virus could have been a variant that escaped the vaccine elicited immune response, however, sequencing of the virus in the blood collected at day 40, did not show any significant variation of the viral sequence compared to the input virus on day 0.
While Envgp140 only vaccines have been successful against homologous challenge, both the human RV144 trial and previous monkey studies showed significant protection from heterologous challenge, included other HIV protein components [8,37,38]. Including Tat in the vaccine formulation induces strong and persistent CD4+ T cells [39] and broadens T cell responses directed against Gag and Envgp160[40,41]. Gag is known for inducing strong cellular responses that may lead to reduced viral loads [42,43]. Addition of Gag and/or Tat to our Con M vaccine may have prevented infection or controlled undetectable virus in vaccinated animals more effectively than Con M Envgp140 alone. Even though some of these studies use Envgp140 proteins, they are combined with other HIV proteins to elicit a broadly reactive response. In our vaccine presented here, the purified VLPs only have Gag and Envgp140 expressed in different modalities than VLPs and we achieve a broadly reactive anti-Envgp140 response using our consensus Envgp160s. For example, viral vectors are used to express Gag and Envgp160 independently in the RV144 human trial, which really does not allow for comparison with our VLP strategy.
Conclusion
Rhesus macaques were vaccinated with trimerized Envgp140 proteins representing consensus sequences for clade A, B, C, E, in a polyvalent mixture or as a single consensus Envgp160 representing the entire Group M. These consensus Envgp140 elicited antibodies with cross-clade anti-Envgp140 binding against a panel of HIV-1 Envgp160s. However, this breadth of antibody binding to HIV-1 Envgp160s only partially correlated with the prevention of infection by SHIVSF162p4.
Materials and methods
Vaccines design
The consensus sequences represent the most common amino acids found at each position of the aligned envelope sequences used. One hundred Envgp160s sequences per clade were used to design consensus sequences of clades A, B, C, and E. The design of the group M consensus Envgp160 was based on two hundred Envgp160 sequences representing clades A, B, C, D, E, F and H. Envgp160s chosen were isolated following mucosal transmission, within weeks after infection and included a diversity of viruses that were isolated in different locations around the world between 1995 and 2005. Each Envgp160 used the CCR5 co-receptor. Vaccine immunogens were designed as Envgp140 trimers as previously described [15].
Protein purification
Human embryonic kidney (HEK) 293T cells were transiently transfected with DNA (8 μg) expressing one of the consensus HIV-1 Envgp140 proteins or wild type Envgp120 6X-HIS tagged proteins. Following DNA transfection as previously described [44], secreted Envgp140 proteins were purified using lectin columns made from agarose galanthus nivalis (snowdrop) lectin (Vector laboratories, Burlingame, CA, USA) [45] and the Envgp120 6X-HIS tagged proteins were purified using nickel columns [44]. Other purified Envgp160s used for ELISAs were purchased from eEnzyme (Gaithersburg, MD, USA). Each purified consensus Envgp140 trimer protein (1 μg) was loaded on to NativePAGE native gel (Invitrogen, Carlsbad, CA, USA) and separated by electrophoresis in the manufacture’s recommended buffers. After separation, the proteins were detected using the ProteoSilver Sliver Stain kit (Sigma, St. Louis, MO, USA) following manufacturer’s protocol [44].
CD4 binding assay
The CD4 binding assay was performed to demonstrate Envgp160 binding to its primary receptor using a similar protocol as previously described [46]. Protein G Dynabeads (Invitrogen) were mixed with anti-his antibody. The tubes with the mixture were placed on the magnets to remove all unbound antibody. Then soluble human CD4-6XHIS tagged protein (eEnzyme, Gaithersburg, MD, USA) and consensus Envgp140 mixtures were then mixed with the beads. Following pellet fractionation, samples were separated on a 10% SDS PAGE gel, transferred unto a nitrocellulose membrane and probed for sCD4 or Envgp160 using specific antibodies. Following secondary IgG-HRP antibodies which were used to detect proteins by Western blot.
Surface plasmon resonance
MAb b12 binding kinetic analyses of the HIV rgp140(s) was performed by surface plasmon resonance (SPR) on a Biacore 3000 (GE/Biacore AB, Inc., Uppsala, Sweden) as previously described [47].
Animals and vaccination
Animals were treated according to the guidelines of the IACUC of the University of Pittsburgh. All the protocols used were approved by the IACUC of the University of Pittsburgh (#1002617). Rhesus macaques (Macaca mulatta) were used for all non-human primate experiments. All animals were cared for adhering to USDA guidelines for laboratory animals. Rhesus macaques were anesthetized using 10-20 mg/kg ketamine and vaccinated intramuscularly in the quadriceps and formulated with Imject® alum adjuvant (Imject® Alum, Pierce Biotechnology; Rockford, IL, USA). Vaccinations were completed at weeks 0, 4 and 8. Twelve animals were divided into three (3) groups, four animals per group (Table 1). For blood sample collection animals were anesthetized with a mixture of ketamine/xylazine. Sera was then harvested and stored at -80°C until needed.
Immune assays
Total anti-Envgp160 IgG was detected by enzyme-linked immunosorbent assay (ELISA) using Concanavalin A (50 μg/μl) per well as previously described [8]. End point titer for assay was determined as the reciprocal of the dilution at which the optical density reading was above the mean plus two standard deviations of naïve sera. For In vitro neutralization, antisera were tested for the ability to neutralize virus infection in vitro using TZM-Bl indicator cells [8]. The sera dilution necessary to neutralize virus was calculated by the following formula (relative light units (RLU) of virus only-RLU of cell only)/2 + RLU cell only. For assessment of T cell responses, NHP IFN-γ ELISPOT was used to enumerate anti-Envgp160 specific cellular responses. The number of anti-Envgp160 (SF162p3 and Con M) and Gag (SIVmac239) specific IFN-γ secreting cells were determined using the non-human primate enzyme-linked immunospot (ELISPOT) assay (R&D Systems, Minneapolis, MN, USA).
SHIV viral load determination
Real time PCR-based SIV viral detection assay was used to determine the viral titers post-challenge as described in [48]. cDNA (10 μl) generated by the RT-RCR reaction was then used for PCR using the ABI 7000 Gene detection system (Applied Bioscience, Carlsbad, California, USA).
Anti-CD8 cell depletion by antibody administration
All animals in the Con M group were depleted of CD8+ T cells. The antibody M-T807R1 (NIH NHP Reagent Source, Beth Israel Deacones Medical Center, Boston, MA, USA) was administered subcutaneously (50 mg/Kg) on day 0 (Day 70 post infection). CD8+ T cell depletion was verified using TruCOUNT tubes (BD Bioscience, San Jose, CA, USA).
Statistical analysis
Statistical tests were performed using Graph Pad Prism software. Statistical significance of antibody test was determined by two-way analysis of variance (ANOVA) followed by the Bonferroni’s post-hoc test. Post-test was used to analyze differences between the vaccine groups. Significance was determined to be a p<0.05.
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
HSE designed and performed most of the experiments analyze data and wrote the paper. BRP was instrumental in animal care and processing of animal samples. JKC performed and analyzed all Surface plasmon resonance data. She was also involve in discussion on results and implications and commented on the manuscript. Finally TMR was the supervisor on the project and was involved in discussions on the results and its implications. He was also instrumental in editing of the manuscript at all stages. All authors read and approved the final manuscript.
Supplementary Material
Representative sensograms of IgG b12 SPR data. SPR binding isotherms detailing the interaction of each recombinant trimeric Envgp140 with the monoclonal antibody b12 are displayed. Indicated concentrations of each Envgp140 were flowed over captured b12 at 37°C on CM5 chips as detailed in Materials and Methods. Association rates, dissociation rates, and affinity constants were calculated with BIA evaluation 4.1.1 software (GE/Biacore AB, Uppsala, Sweden). A, ConA; B, ConB; C, ConC; D, Con M; E, ConE; F, ADA; G, PV0.04; H, R2; I, SC42; and J, YU2. Black lines are determined 1:1 Langmuir kinetic fits. RU, resonance units.
Contributor Information
Hermancia S Eugene, Email: hee2@pitt.edu.
Brooke R Pierce-Paul, Email: brp42@pitt.edu.
Jodi K Cragio, Email: craigoj@pitt.edu.
Ted M Ross, Email: tmr15@pitt.edu.
Acknowledgements
This research was primarily supported by an award from the National Institute of Health/National Institute of Allergy and Infectious Diseases R01AI068507 to T.M.R. The HIV and SIV specific peptides and reagents were obtained through the AIDS Research and Reference Reagent Program, Division of AIDS, NIAID, NIH including TZM-bl cells (also called JC57BL-13) (#8129). The authors would also thank investigators that provided specific HIV-1 isolates (Rob Center, Dennis Ellenberger, Phalguni Gupta, Beatrice Hahn, David Montefiori, Gary Nabel, Yiming Shao, and Carolyn Williamson). We are also grateful to Amara Luckay and Jim Smith at the CDC for determination of viral loads. This work was supported, in part, under a grant with the Pennsylvania Department of Health. The department specifically disclaims responsibility for any analyses, interpretations or conclusions.
References
- Hemelaar J. The origin and diversity of the HIV-1 pandemic. Trends Mol Med. 2012;18(3):182–192. doi: 10.1016/j.molmed.2011.12.001. [DOI] [PubMed] [Google Scholar]
- Gaschen B, Taylor J, Yusim K, Foley B, Gao F, Lang D, Novitsky V, Haynes B, Hahn BH, Bhattacharya T. Diversity Considerations in HIV-1 Vaccine Selection. Science (New York, NY) 2002;296:2354–2360. doi: 10.1126/science.1070441. [DOI] [PubMed] [Google Scholar]
- Spira S, Wainberg MA, Loemba H, Turner D, Brenner BG. Impact of clade diversity on HIV-1 virulence, antiretroviral drug sensitivity and drug resistance. J Antimicrob Chemother. 2003;51(2):229–240. doi: 10.1093/jac/dkg079. [DOI] [PubMed] [Google Scholar]
- McBurney SP, Ross TM. Viral sequence diversity: challenges for AIDS vaccine designs. Expert Rev Vaccines. 2008;7(9):1405–1417. doi: 10.1586/14760584.7.9.1405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu S, Grimes Serrano JM, Wang S. Polyvalent AIDS Vaccines. Curr HIV Res. 2010;8(8):622–629. doi: 10.2174/157016210794088290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arenas M, Posada D. Computational Design of Centralized HIV-1 Genes. Curr HIV Res. 2010;8(8):613–621. doi: 10.2174/157016210794088263. [DOI] [PubMed] [Google Scholar]
- Gao F, Liao H-X, Hahn BH, Letvin NL, Korber BT, Haynes BF. Centralized HIV-1 Envelope Immunogens and Neutralizing Antibodies. Curr HIV Res. 2007;5(6):572–577. doi: 10.2174/157016207782418498. [DOI] [PubMed] [Google Scholar]
- McBurney SP LG, Forthal DN, Ross TM. Evaluation of heterologous vaginal SHIV SF162p4 infection following vaccination with a polyvalent clade B virus-like particle vaccine. AIDS Res Hum Retroviruses. 2012. in Press. [DOI] [PMC free article] [PubMed]
- Wang S, Kennedy JS, West K, Montefiori DC, Coley S, Lawrence J, Shen S, Green S, Rothman AL, Ennis FA. Cross-subtype antibody and cellular immune responses induced by a polyvalent DNA prime–protein boost HIV-1 vaccine in healthy human volunteers. Vaccine. 2008;26(8):1098–1110. doi: 10.1016/j.vaccine.2007.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakhashe SK, Wang W, Siddappa NB, Hemashettar G, Polacino P, Hu SL, Villinger F, Else JG, Novembre FJ, Yoon JK. Vaccination against Heterologous R5 Clade C SHIV: Prevention of Infection and Correlates of Protection. PLoS One. 2011;6(7):e22010. doi: 10.1371/journal.pone.0022010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yan J, Yoon H, Kumar S, Ramanathan MP, Corbitt N, Kutzler M, Dai A, Boyer JD, Weiner DB. Enhanced Cellular Immune Responses Elicited by an Engineered HIV-1 Subtype B Consensus-based Envelope DNA Vaccine. Mol Ther. 2007;15(2):411–421. doi: 10.1038/sj.mt.6300036. [DOI] [PubMed] [Google Scholar]
- McBurney SP, Ross TM. Human immunodeficiency virus-like particles with consensus envelopes elicited broader cell-mediated peripheral and mucosal immune responses than polyvalent and monovalent Env vaccines. Vaccine. 2009;27(32):4337–4349. doi: 10.1016/j.vaccine.2009.04.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santra S, Korber BT, Muldoon M, Barouch DH, Nabel GJ, Gao F, Hahn BH, Haynes BF, Letvin NL. A centralized gene-based HIV-1 vaccine elicits broad cross-clade cellular immune responses in rhesus monkeys. PNAS. 2008;105(30):10489–10494. doi: 10.1073/pnas.0803352105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liao H-X, Sutherland LL, Xia S-M, Brock ME, Scearce RM, Vanleeuwen S, Alam SM, McAdams M, Weaver EA, Camacho ZT. A group M consensus envelope glycoprotein induces antibodies that neutralize subsets of subtype B and C HIV-1 primary viruses. Virology. 2006;353(2):268–282. doi: 10.1016/j.virol.2006.04.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bower JF, Li Y, Wyatt R, Ross TM. HIV-1 Envgp140 trimers elicit neutralizing antibodies without efficient induction of conformational antibodies. Vaccine. 2006;24(26):5442–5451. doi: 10.1016/j.vaccine.2006.03.063. [DOI] [PubMed] [Google Scholar]
- Pantophlet R, Wilson IA, Burton DR. Improved design of an antigen with enhanced specificity for the broadly HIV-neutralizing antibody b12. PEDS. 2004;17(10):749–758. doi: 10.1093/protein/gzh085. [DOI] [PubMed] [Google Scholar]
- Watkins JD, Siddappa NB, Lakhashe SK, Humbert M, Sholukh A, Hemashettar G, Wong YL, Yoon JK, Wang W, Novembre FJ. An Anti-HIV-1 V3 Loop Antibody Fully Protects Cross-Clade and Elicits T-Cell Immunity in Macaques Mucosally Challenged with an R5 Clade C SHIV. PLoS One. 2011;6(3):e18207. doi: 10.1371/journal.pone.0018207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett SW, Srivastava IK, Kan E, Zhou F, Goodsell A, Cristillo AD, Ferrai MG, Weiss DE, Letvin NL, Montefiori D. Protection of macaques against vaginal SHIV challenge by systemic or mucosal and systemic vaccinations with HIV-envelope. AIDS. 2008;22(3):339–348. doi: 10.1097/QAD.0b013e3282f3ca57. [DOI] [PubMed] [Google Scholar]
- Barnett SW, Burke B, Sun Y, Kan E, Legg H, Lian Y, Bost K, Zhou F, Goodsell A, zur Megede J. Antibody-Mediated Protection against Mucosal Simian-Human Immunodeficiency Virus Challenge of Macaques Immunized with Alphavirus Replicon Particles and Boosted with Trimeric Envelope Glycoprotein in MF59 Adjuvant. J Virol. 2010;84(12):5975–5985. doi: 10.1128/JVI.02533-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kothe DL, Decker JM, Li Y, Weng Z, Bibollet-Ruche F, Zammit KP, Salazar MG, Chen Y, Salazar-Gonzalez JF, Moldoveanu Z. Antigenicity and immunogenicity of HIV-1 consensus subtype B envelope glycoproteins. Virology. 2007;360(1):218–234. doi: 10.1016/j.virol.2006.10.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kothe DL, Li Y, Decker JM, Bibollet-Ruche F, Zammit KP, Salazar MG, Chen Y, Weng Z, Weaver EA, Gao F. Ancestral and consensus envelope immunogens for HIV-1 subtype C. Virology. 2006;352(2):438–449. doi: 10.1016/j.virol.2006.05.011. [DOI] [PubMed] [Google Scholar]
- Barouch DH, Liu J, Li H, Maxfield LF, Abbink P, Lynch DM, Iampietro MJ, SanMiguel A, Seaman MS, Ferrari G. Vaccine protection against acquisition of neutralization-resistant SIV challenges in rhesus monkeys. Nature. 2012. advance online publication. [DOI] [PMC free article] [PubMed]
- Sharma VA, Kan E, Sun Y, Lian Y, Cisto J, Frasca V, Hilt S, Stamatatos L, Donnelly JJ, Ulmer JB. Structural characteristics correlate with immune responses induced by HIV envelope glycoprotein vaccines. Virology. 2006;352(1):131–144. doi: 10.1016/j.virol.2006.04.030. [DOI] [PubMed] [Google Scholar]
- Harris A, Borgnia MJ, Shi D, Bartesaghi A, He H, Pejchal R, Kang Y, Depetris R, Marozsan AJ, Sanders RW. Trimeric HIV-1 glycoprotein gp140 immunogens and native HIV-1 envelope glycoproteins display the same closed and open quaternary molecular architectures. Proc Natl Acad Sci U S A. 2011;108:11440–11445. doi: 10.1073/pnas.1101414108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saphire EO, Parren PWHI, Pantophlet R, Zwick MB, Morris GM, Rudd PM, Dwek RA, Stanfield RL, Burton DR, Wilson IA. Crystal Structure of a Neutralizing Human IgG Against HIV-1: A Template for Vaccine Design. Science. 2001;293(5532):1155–1159. doi: 10.1126/science.1061692. [DOI] [PubMed] [Google Scholar]
- Holl V, Peressin M, Moog C. Antibody-Mediated Fcψ Receptor-Based Mechanisms of HIV Inhibition: Recent Findings and New Vaccination Strategies. Viruses. 2009;1:1265–1294. doi: 10.3390/v1031265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burton DR. Antibodies, viruses and vaccines. Nat Rev Immunol. 2002;2(9):706–713. doi: 10.1038/nri891. [DOI] [PubMed] [Google Scholar]
- Haynes BF, Gilbert PB, McElrath MJ, Zolla-Pazner S, Tomaras GD, Alam SM, Evans DT, Montefiori DC, Karnasuta C, Sutthent R, Liao HX. Immune-correlates analysis of an HIV-1 vaccine efficacy trial. N Engl J Med. 2012;366(14)):1275–86. doi: 10.1056/NEJMoa1113425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barouch DH, Liu J, Li H, Maxfield LF, Abbink P, Lynch DM, Iampietro MJ, SanMiguel A, Seaman MS, Ferrari G. Vaccine protection against acquisition of neutralization-resistant SIV challenges in rhesus monkeys. Nature. advance online publication. [DOI] [PMC free article] [PubMed]
- Rerks-Ngarm S, Pitisuttithum P, Nitayaphan S, Kaewkungwal J, Chiu J, Paris R, Premsri N, Namwat C, de Souza M, Adams E. Vaccination with ALVAC and AIDSVAX to Prevent HIV-1 Infection in Thailand. N Eng J Med. 2009;361:2209–2220. doi: 10.1056/NEJMoa0908492. [DOI] [PubMed] [Google Scholar]
- Haynes BF, Gilbert PB, McElrath MJ, Zolla-Pazner S, Tomaras GD, Alam SM, Evans DT, Montefiori DC, Karnasuta C, Sutthent R. Immune-Correlates Analysis of an HIV-1 Vaccine Efficacy Trial. N Eng J Med. 2012;366(14):1275–1286. doi: 10.1056/NEJMoa1113425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hessell AJ, Rakasz EG, Tehrani DM, Huber M, Weisgrau KL, Landucci G, Forthal DN, Koff WC, Poignard P, Watkins DI. Broadly Neutralizing Monoclonal Antibodies 2F5 and 4E10 Directed against the Human Immunodeficiency Virus Type 1 gp41 Membrane-Proximal External Region Protect against Mucosal Challenge by Simian-Human Immunodeficiency Virus SHIVBa-L. J Virol. 2009;84:1302–1313. doi: 10.1128/JVI.01272-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barnett SW, Srivastava IK, Kan E, Zhou F, Goodsell A, Cristillo AD, Ferrai MG, Weiss DE, Letvin NL, Montefiori D. Protection of macaques against vaginal SHIV challenge by systemic or mucosal and systemic vaccinations with HIV-envelope. AIDS. 2008;22(3):339–348. doi: 10.1097/QAD.0b013e3282f3ca57. [DOI] [PubMed] [Google Scholar]
- Smith M, Wightman F, Lewin S. HIV Reservoirs and strategies for eradication. Curr HIV/AIDS Rep. 2012;9:5–15. doi: 10.1007/s11904-011-0108-2. [DOI] [PubMed] [Google Scholar]
- Veazey RS, Acierno PM, McEvers KJ, Baumeister SHC, Foster GJ, Rett MD, Newberg MH, Kuroda MJ, Williams K, Kim E-Y. Increased Loss of CCR5+ CD45RA− CD4+ T Cells in CD8+ Lymphocyte-Depleted Simian Immunodeficiency Virus-Infected Rhesus Monkeys. J Virol . 2008;82:5618–5630. doi: 10.1128/JVI.02748-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loffredo JT, Maxwell J, Qi Y, Glidden CE, Borchardt GJ, Soma T, Bean AT, Beal DR, Wilson NA, Rehrauer WM. Mamu-B*08-Positive Macaques Control Simian Immunodeficiency Virus Replication. J Virol. 2007;81:8827–8832. doi: 10.1128/JVI.00895-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lakhashe SK, Velu V, Siddappa NB, Dipasquale JM, Hemashettar G, Yoon JK, Rasmussen RA, Yang F, Lee SJ, Montefiori DC. Prime-boost vaccination with heterologous live vectors encoding SIV gag and multimeric HIV-1 gp160 protein: efficacy against repeated mucosal R5 clade C SHIV challenges. Vaccine. 2011;29(34):5611–24. doi: 10.1016/j.vaccine.2011.06.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sundling C, Forsell MN, O'Dell S, Feng Y, Chakrabarti B, Rao SS, Loré K, Mascola JR, Wyatt RT, Douagi I. Soluble HIV-1 Env trimers in adjuvant elicit potent and diverse functional B cell responses in primates. J Exp Med. 2010;207(9):2003–2017. doi: 10.1084/jem.20100025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leroux-Roels I, Koutsoukos M, Clement F, Steyaert S, Janssens M, Bourguignon P, Cohen K, Altfeld M, Vandepapelière P, Pedneault L. Strong and persistent CD4+ T-cell response in healthy adults immunized with a candidate HIV-1 vaccine containing gp120, Nef and Tat antigens formulated in three Adjuvant Systems. Vaccine. 2010;28(43):7016–7024. doi: 10.1016/j.vaccine.2010.08.035. [DOI] [PubMed] [Google Scholar]
- Gavioli R, Cellini S, Castaldello A, Voltan R, Gallerani E, Gagliardoni F, Fortini C, Cofano EB, Triulzi C, Cafaro A. The Tat protein broadens T cell responses directed to the HIV-1 antigens Gag and Env: Implications for the design of new vaccination strategies against AIDS. Vaccine. 2008;26(5):727–737. doi: 10.1016/j.vaccine.2007.11.040. [DOI] [PubMed] [Google Scholar]
- Ferrantelli F, Maggiorella MT, Schiavoni I, Sernicola L, Olivieri E, Farcomeni S, Pavone-Cossut MR, Moretti S, Belli R, Collacchi B. A combination HIV vaccine based on Tat and Env proteins was immunogenic and protected macaques from mucosal SHIV challenge in a pilot study. Vaccine. 2011;29(16):2918–2932. doi: 10.1016/j.vaccine.2011.02.006. [DOI] [PubMed] [Google Scholar]
- Ferre AL, Lemongello D, Hunt PW, Morris MM, Garcia JC, Pollard RB, Yee HF, Martin JN, Deeks SG, Shacklett BL. Immunodominant HIV-Specific CD8+ T-Cell Responses Are Common to Blood and Gastrointestinal Mucosa, and Gag-Specific Responses Dominate in Rectal Mucosa of HIV Controllers. J Virol. 2010;84(19):10354–10365. doi: 10.1128/JVI.00803-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ranasinghe S, Flanders M, Cutler S, Soghoian DZ, Ghebremichael M, Davis I, Lindqvist M, Pereyra F, Walker BD, Heckerman D. HIV-Specific CD4 T Cell Responses to Different Viral Proteins Have Discordant Associations with Viral Load and Clinical Outcome. J Virol. 2011;86(1):277–283. doi: 10.1128/JVI.05577-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toapanta FR, DeAlmeida DR, Dunn MD, Ross TM. C3d adjuvant activity is reduced by altering residues involved in the electronegative binding of C3d to CR2. Immunol Lett. 2010;129(1):32–38. doi: 10.1016/j.imlet.2009.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bower JF, Ross TM. In: Current Topics in Complement vol. 586. Lambris JD, editor. US: Springer; 2006. A Minimum CR2 Binding Domain of C3d Enhances Immunity Following Vaccination; pp. 249–264. [DOI] [PubMed] [Google Scholar]
- Young KR, Smith JM, Ross TM. Characterization of a DNA vaccine expressing a human immunodeficiency virus-like particle. Virology. 2004;327(2):262–272. doi: 10.1016/j.virol.2004.07.009. [DOI] [PubMed] [Google Scholar]
- Steckbeck JD, Orlov I, Chow A, Grieser H, Miller K, Bruno J, Robinson JE, Montelaro RC, Cole KS. Kinetic Rates of Antibody Binding Correlate with Neutralization Sensitivity of Variant Simian Immunodeficiency Virus Strains. J Virol. 2005;79(19):12311–12320. doi: 10.1128/JVI.79.19.12311-12320.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Subbarao S, Otten RA, Ramos A, Kim C, Jackson E, Monsour M, Adams DR, Bashirian S, Johnson J, Soriano V. Chemoprophylaxis with Tenofovir Disoproxil Fumarate provided partial protection against infection with simian human immunodeficiency virus in macaques given multiple virus challenges. J Infect Dis. 2006;194(7):904–911. doi: 10.1086/507306. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Representative sensograms of IgG b12 SPR data. SPR binding isotherms detailing the interaction of each recombinant trimeric Envgp140 with the monoclonal antibody b12 are displayed. Indicated concentrations of each Envgp140 were flowed over captured b12 at 37°C on CM5 chips as detailed in Materials and Methods. Association rates, dissociation rates, and affinity constants were calculated with BIA evaluation 4.1.1 software (GE/Biacore AB, Uppsala, Sweden). A, ConA; B, ConB; C, ConC; D, Con M; E, ConE; F, ADA; G, PV0.04; H, R2; I, SC42; and J, YU2. Black lines are determined 1:1 Langmuir kinetic fits. RU, resonance units.


