Abstract
The eye and its associated tissues including the lacrimal system and lids have evolved several defence mechanisms to prevent microbial invasion. Included among this armory are several host-defence peptides. These multifunctional molecules are being studied not only for their endogenous antimicrobial properties but also for their potential therapeutic effects. Here the current knowledge of host-defence peptide expression in the eye will be summarised. The role of these peptides in eye disease will be discussed with the primary focus being on infectious keratitis, inflammatory conditions including dry eye and wound healing. Finally the potential of using host-defence peptides and their mimetics/derivatives for the treatment and prevention of eye diseases is addressed.
Keywords: Defensin, Cathelicidin, Keratitis, Dry eye, Contact lens
Expression and distribution of host-defence peptides in the eye and associated structures
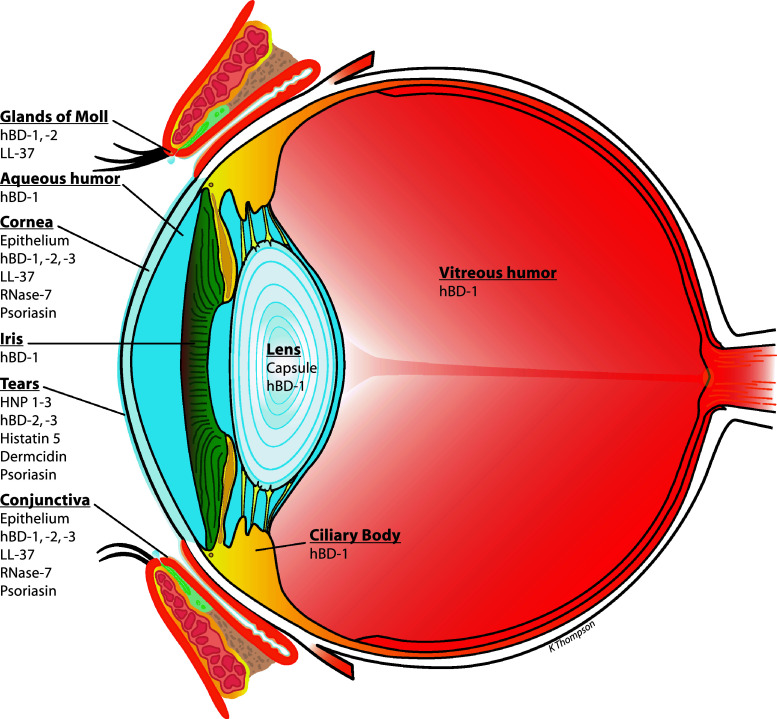
Several studies over the past 10–15 years have confirmed that, like many tissues in the body, the eye and its associated structures (lids and lacrimal system for example) express a variety of host-defence peptides (HDPs). Prior review articles have primarily focused on HDPs at the ocular surface; here current knowledge of their expression by related structures and internal components is also addressed. Figure 1 summarises the current understanding of HDP distribution in the human eye. As can be readily seen, HDPs of the β-defensin (hBD) family and the cathelicidin LL-37 are commonly found.
Fig. 1.
Expression profile of host-defence peptides in the eye. The diagram summarises the pattern of expression of the major HDPs in the human eye. These data are derived primarily from studies to detect peptide expression that have been performed on cultured cells in vitro and on tissue/tear fluid samples from human donors (these studies are summarised and references given in the section “Expression and distribution of host-defence peptides in the eye and associated structures”). Lacrimal gland and lacrimal drainage are not included but are discussed in the section “Associated ocular structures”
For details on the chemical characteristics, basic biology and general mechanisms of antimicrobial, immunomodulatory and other actions of these and other HDPs, the reader is referred to other articles in this volume and recent reviews [1–5].
Ocular surface
The ocular surface comprises the cornea, conjunctiva and tear film. The cornea is the transparent window covering the pupil and provides the major refracting surface of the eye. The conjunctiva is a thin mucous membrane harbouring immune cells, which covers the sclera, the white tough covering of the eye. The tear film coats the entire exposed surface of the eye providing lubrication and protection.
A squamous epithelium forms the outermost layer of both the cornea and conjunctiva, and these cells are known to express a number of HDPs in keeping with their role as a major barrier to invading organisms. Both corneal and conjunctival epithelial cells constitutively express hBD-1, whereas expression of hBD-2 is variable with it being found in normal tissue only occasionally [6–11]. hBD-2 is found more frequently in infected/inflamed ocular surface tissue, which is in keeping with in vitro studies with cultured corneal and conjunctival epithelial cells showing that bacterial products such as lipopolysaccharide (LPS) and pro-inflammatory cytokines such as interleukin (IL)-1β upregulate hBD-2 expression [8, 10–17]. These ocular surface epithelial cells also constitutively express hBD-3, which some have found to be upregulated in response to cytokines [10, 11, 17, 18]. Expression of hBD-4 has also been investigated in a number of studies, and while its expression is common in cultured ocular surface cells, it was found only infrequently in actual tissue samples [17–19]. hBD-9 was also recently found to be expressed by corneal and conjunctival epithelial cells [20, 21]. However, while expression of several other members of the β-defensin family has been sought, none appear to be present [18, 19].
Although members of the α-defensin family, human neutrophil peptides (HNP)-1, 2, 3, have been detected in the corneal stroma (the thick collagen layer that forms the bulk of the cornea), their source is the neutrophils infiltrating the cornea in response to inflammatory signals rather than the resident corneal cells themselves [22]. Other α-defensins, human defensin (HD)-5 and 6, were not present [7, 23].
LL-37, the sole member of the human cathelicidin family, is also expressed by corneal and conjunctival epithelial cells [18, 24]. This peptide is derived from a larger precursor, hCAP-18, and like hBD-2, its expression is upregulated in response to bacterial challenge [15, 16, 25]. LL-37 is also found in neutrophil granules [26] so recruitment of these cells to the ocular surface in response to inflammation/infection would be expected to contribute to an increase in local levels of LL-37 (and indeed possibly several other neutrophil-derived antimicrobial molecules).
While defensins and LL-37 are the most widely studied of the ocular surface HDPs, the expression of several other peptides and proteins with antimicrobial activity has been reported. These include liver-expressed antimicrobial peptide (LEAP)-1 (also known as hepcidin) and LEAP-2 [18], although another study failed to detect these HDPs [19]. The antimicrobial phosphoprotein statherin is also expressed as are MIP-3α and thymosin β-4 [19, 27] and some “antimicrobial cytokines” such as CCL28 and CXCL-1 [28–30]. Psoriasin (S100A7) is constitutively expressed in cornea and conjunctival epithelial cells, and its expression is upregulated in response to some bacterial products [31]. Most recently, Mohammed et al. 2011, reported the expression of RNase-7 in cornea and conjunctiva and observed its upregulation in response to IL-1β [32]. It should be cautioned that some of these other molecules have only been reported at the mRNA level, and so confirmation of their expression awaits experiments addressing protein levels. Further, for many of these, while they have been shown to have antimicrobial activity, this is sometimes relatively weak activity and it is likely that contributing to innate defence is not their primary role at the ocular surface. They may, however, be more directly involved in other activities recently ascribed to HDPs such as immunomodulation.
Studies in laboratory animals also confirm the existence of HDPs at the ocular surface with mice expressing a range of corneal and conjunctival defensins as well as cathelin-related antimicrobial peptide (CRAMP), the homologue of human LL-37 [17, 33–36]. However, care should be taken in extrapolating from mouse to human as mice have many more β-defensins than humans, and equivalency in terms of numerical designation cannot be assumed, for example, the mouse orthologue of hBD-3 is mBD-14 [37], not mBD-3.
The tear film is a complex structure comprising an outermost lipid layer to limit evaporation; it overlays a mix of possibly over 500 different proteins, electrolytes and mucins, with the latter increasing in concentration toward the ocular surface and interacting with the glycocalyx of the superficial corneal and conjunctival epithelial cells so adhering the tears to the cells [38]. It has long been established that tears contain important antimicrobial molecules such as lysozyme, phospholipase A2 and lactoferrin [39]. Recent studies also show that HDPs are present. These may arise from the main and/or accessory lacrimal glands, ocular surface epithelial cells or come from neutrophils that are naturally found in the tears in small numbers, particularly upon eye-opening [40]. α-Defensins HNP1–3, whose source is neutrophils, are present at low levels in normal human tears and are increased after ocular surface surgery [7, 41]. Recently the presence of hBD-2 and 3 has also been reported [17]. Again, here the levels were low and were less than would be needed for antimicrobial activity based on the results of in vitro studies, but these samples were normal tears, and it is possible that much higher levels may be seen in samples from patients with active infections (unfortunately such samples are very hard to come by). Preliminary reports have shown that tears contain psoriasin [31] and the antifungal peptide histatin 5 [42] and most recently dermcidin was detected [43].
Internal ocular components
HDP expression by internal ocular structures has not been intensively studied. A study by Lehmann et al. [9] revealed that most samples of human lens capsule (which surrounds the crystalline lens) and iris (the coloured part of the eye) expressed mRNA for hBD-1 but not hBD-2. hBD-1 message was also detected in ciliary body (a highly vascularised structure surrounding the ciliary muscle that is involved in aqueous humour production and anchors the lens via the zonular fibers), but again hBD-2 and also α-defensins HD-5 and 6 were absent [44]. Retinal pigment epithelium and ciliary body epithelial cell lines did not express hBD-1 or 2 at baseline, but the latter was upregulated after cytokine treatment. Further, immunoblotting showed that while samples of both vitreous and aqueous humour were positive for hBD-1 (although levels were estimated to be quite low), no hBD-2 was present. These studies suggest that hBD-1 is constitutively expressed inside the eye, while hBD-2 is not normally present but may be expressed in response to inflammation. Elucidation of the specific roles of these HDPs inside the eye awaits further investigation, but it was suggested that, owing to its broader spectrum of activity and higher potency, hBD-2 is a stronger candidate for antimicrobial defence inside the eye than hBD-1 [44].
Associated ocular structures
A number of structures external to the eye are important for normal ocular function, including the main and accessory lacrimal glands which supply the bulk of the aqueous component of the tear film, the lacrimal drainage system which conducts tears away from the ocular surface and the eyelids which serve a protective role and have glands that supply some tear film constituents. Haynes et al. [7] reported the presence of hBD-1 message in lacrimal gland, but hBD-2 and α-defensins HD-5 and 6 were not found. The lacrimal sac and nasolacrimal duct are major components of the lacrimal drainage system and harbour mucosa-associated lymphoid tissue. In keeping with this protective role, they also express HDPs [45]. hBD-1 was detected in several samples of nasolacrimal duct epithelium obtained by biopsy from both healthy patients and those with dacryocystitis. hBD-2 on the other hand was only detected in dacryocystitis patients. HNP1–3 were also detectable, with infiltrating neutrophils being the source in both healthy and diseased tissue. Messenger RNA for defence proteins bactericidal/permeability-increasing protein and cationic antimicrobial protein-37 was present, however that for HD-5 and 6 was not, and interestingly mRNA for hCAP-18, the precursor to LL-37, was absent too. Some of these data have recently been confirmed, and the studies extended to include hBD-3 and 4 [17]. hBD-4 was not found in the lacrimal gland or nasolacrimal ducts. hBD-3 protein, however, could be detected by ELISA and immunostaining, although RT-PCR failed to detect any mRNA. The glands of Moll (also referred to as ciliary glands) are apocrine glands located at the margin of the eyelids and secrete lipid onto the lid, which becomes incorporated into the tear film. Recently hBD-1 and 2 and LL-37 were detected in the secretory end-pieces of the glands and are presumed to protect the glands themselves from infection, with the material being released into the gland lumen being protective in the ducts, eyelash shaft and ocular surface [46]. β-Defensin expression has also been observed in the glands of Moll in non-human primates [47] and in various glands of the porcine eyelid [48].
This brief overview reveals a broad expression of HDPs in ocular and related structures. The value of some studies is a little limited due to experiments being restricted to investigation of message rather than actual protein, also a number were conducted before the study of HDPs came into vogue and before the discovery of the extent of the HDP repertoire. Thus, it will be of interest to revisit the area of HDP expression in intra-ocular and associated tissues and extend these investigations to additional defensins such as hBD-9 and other HDPs. The expression of HDPs suggests their participation in maintaining normal ocular health and function through their antimicrobial action as well as any/all of the myriad of activities they have been found to perform in other tissues such as modulating immune responses and wound healing [3, 49]. However, in the eye, only defensins and cathelicidin have been studied in any depth, and the focus of these investigations has been the ocular surface and in particular the cornea. Therefore, these particular HDPs and this location form the basis for the following discussions on the role of endogenously expressed HDPs in eye diseases and their clinical potential as ocular therapeutics.
Role for endogenous host-defence peptides in eye diseases
Defence against infection
The majority of studies investigating the role of HDPs in preventing ocular infection have focused on microbial keratitis. This affects the cornea, the primary refracting surface of the eye, and if not treated promptly and adequately may result in permanent vision loss. Microbial keratitis may be caused by bacteria, viruses, fungi and protozoa, with contact lens wear and corneal trauma being common risk factors.
Pseudomonas aeruginosa and Staphylococcus aureus are two of the most common causes of bacterial keratitis with P. aeruginosa being the most common cause in contact lens wearers [50, 51]. P. aeruginosa is a particularly dangerous and dreaded ocular pathogen and induces very severe disease, which, if not quickly treated or if unresponsive to treatment, rapidly progresses to inflammation, neovascularisation and liquefactive necrosis of the cornea. As discussed above (section “Ocular surface”), ocular surface epithelial cells express defensins and the cathelicidin LL-37, and infection and exposure to bacterial products in vitro increase expression of some of these, although in contrast hBD-9 is decreased in infection [6–12, 15–17, 20]. Ocular surface–expressed HDPs are also active, to varying degrees, against common ocular bacterial pathogens [19, 24]. hBD-1 is the least effective defensin having relatively moderate antimicrobial activity against P. aeruginosa but being poorly effective against Staphylococcal strains. hBD-2 has good antimicrobial activity against P. aeruginosa although its activity towards S. aureus is limited. hBD-3 and LL-37 on the other hand have good bactericidal activity against both P. aeruginosa and S. aureus. The level of HDPs detected in vivo and in cultured cells is generally much lower than is required for antimicrobial activity in vitro. For example, Zhou et al. [41] reported that normal tear film levels of HNPs are in the range 0.2–1 μg/ml and rise to a maximum of 20 μg/ml after ocular surgery. This latter level would be expected to result in mild to at best moderate antibacterial activity depending on the target strain. Garreis et al. [17] recently quantitated levels of hBD-2 and hBD-3 in human cornea, conjunctiva, lacrimal gland and tears. The peptides were present in picogram amounts, and highest levels were in the cornea. Unpublished data from our laboratory also show hBD-2 to be present in normal tear fluid at 329 ± 154 pg/ml. These levels are far below those required for antibacterial activity in vitro, but it should be noted they are reflective of the normal ocular surface, and levels would be expected to rise somewhat in response to infection. However, in vitro HDPs show effective antibacterial activity at concentrations of several micrograms per millilitre and above, and so it is questionable that such high amounts are actually present at the ocular surface in vivo even after induction by pathogens. Rather, as has been suggested for other tissues [52, 53], within the environment of the tear film and epithelial layer as yet undefined mechanisms are likely to exist to raise the local concentration of HDPs to effective levels.
It has long been recognised that because of their mode of antibacterial action, i.e. electrostatic interaction with negatively charged microbial membranes, cationic HDPs are subject to varying degrees of sensitivity to physiological salt concentrations [3]. This is of particular concern at the ocular surface, which is bathed in tear fluid. Huang et al. investigated the antibacterial activity of various HDPs expressed by ocular surface epithelial cells in the presence of human tears and observed that activity of hBD-1 and hBD-2 was markedly reduced, whereas that for hBD-3 and LL-37 was much less affected [19, 25]. Furthermore, while some of the loss of activity was attributable to salt effects, MUC5AC, the major gel-forming mucin of the tear film, was also found to inhibit the antipseudomonal activity of hBD-2 [54]. Together, these current in vitro findings suggest that, at the human ocular surface in vivo, hBD-3 and LL-37 are the HDPs most likely to have significant independent antibacterial activity. Others such as hBD-1, hBD-2 and HNP1-3 may have roles to play under circumstances when high concentrations can be achieved or when they can interact synergistically with other HDPs and antimicrobial molecules as both situations would be expected to help temper the effects of high salt and perhaps that of other interfering tear film components [55–57].
A number of animal studies provide compelling evidence to support a role for HDPs in defence against P. aeruginosa infection at the ocular surface. Huang et al. [33] showed that mice deficient in CRAMP, the murine homologue of LL-37, have increased susceptibility to bacterial keratitis as manifest by delayed P. aeruginosa clearance, increased numbers of neutrophils, an altered cytokine profile and worse clinical score. Subsequently, roles for some defensins have also been revealed, as mice in which expression of mBD-2 or mBD-3 (an orthologue of hBD-2), but not mBD-1 or mBD-4, was knocked down with siRNA also showed increased susceptibility to P. aeruginosa infection [35, 36]. Further, flagellin pretreatment has been found to reduce inflammation and improve clinical outcome in experimental murine P. aeruginosa keratitis, an effect that, at least in part, was attributable to the upregulation of antimicrobial molecules including CRAMP [58]. In these aforementioned models, the cornea is scratched to facilitate infection. A recent study by Augustin et al. [59] shows that the unscratched ocular surface of mice deficient in mBD-3 has reduced capacity to clear P. aeruginosa and increased susceptibility to colonisation, indicating for the first time that HDPs are important in prevention of bacterial keratitis and not just after the infection has been initiated. Thus, while more in depth investigation is required to fully understand the role of specific HDPs in bacterial keratitis in humans, animal models conclusively show that despite potential confounding factors of low concentrations and interference from tear film components, HDPs do function to defend the cornea against P. aeruginosa infection.
The role of HDPs in other forms of microbial keratitis has not yet been investigated in depth. Herpes simplex virus (HSV) and adenovirus are two common ocular viral pathogens. There are some 20,000 new cases of ocular HSV infection in the USA each year and, owing to viral latency, an additional 28,000 reactivations [60]. Infection can lead to a very robust inflammatory response with the formation of classic dendritic corneal ulcers making HSV keratitis the most frequent cause of corneal blindness in the USA. Adenoviruses are the most common cause of viral conjunctivitis and keratoconjunctivitis. While typically not as serious as HSV keratitis, these infections are highly contagious and cause significant morbidity and discomfort. Despite the serious nature and frequency of viral infections, a role for HDPs has not been investigated. It is known however that α-defensins and LL-37 have activity against both HSV-1 and adenoviridae in vitro suggesting they may be active against these pathogens in vivo [24, 61, 62].
Fungi are a significant cause of keratitis particularly in warm humid climates and developing countries where they may account for 40% or more of microbial keratitis cases [51, 63–66]. Common culprits are Fusarium solani, Aspergillus fumigatus and Candia albicans [64, 66–71]. These infections are very difficult to treat and are characterised by corneal oedema and ulceration leading to scarring and profound vision loss. That various HDPs have activity against a range of fungi is well established, and fungi have been shown to modulate the expression of HDPs in a variety of epithelial cells [72–86]. However mice deficient in CRAMP, the homologue of LL-37, did not show increased susceptibility to C. albicans in an intradermal infection model, leading the authors to conclude that cathelicidins may be most effective as a superficial barrier to infection [77]. Recent studies have started to address a role for HDPs in fungal keratitis. Yuan et al. [87] investigated the expression of various HDPs in a mouse model of Candida keratitis and found that CRAMP was markedly increased in response to the infection. Further, Gao et al. [88] have shown that mice deficient in CRAMP have more severe Candida keratitis than wild type mice. These positive results in comparison to the earlier study of Lopez-Garcia et al. [77] in a skin model of C. albicans infection may reflect the more “superficial”/mucosal nature of the corneal infection [88]. Overall, these studies indicate a potential role for LL-37 in particular, but possibly also other HDPs, in preventing/limiting fungal keratitis in humans.
Acanthamoeba keratitis is relatively uncommon, but is of interest as there was a recent outbreak of A. castellanii keratitis associated with a particular contact lens solution [89]. An early study investigated amphibian magainins and found them to have activity against the trophic and cystic stages of A. polyphaga [90]. More recently Otri et al. have observed that A. castellanii stimulated the expression of several HDPs including hBD-3 and LL-37 in cultured corneo-limbal epithelial cells but decreased expression of hBD-1 [91]. Together these studies suggest that HDPs may play a role in the innate response to Acanthamoeba keratitis, but this needs to be investigated further.
That external risk factors such as contact lens wear and trauma predispose an individual to corneal infection are well established. The issues relating contact lens wear to infection are complex, and currently patient compliance with cleaning routines is an area of intense study. Interesting results from a recent in vitro study also reveal additional novel avenues for investigation [92]. Here it was found that contact lenses impaired the ability of human corneal epithelial cells to secrete hBD-2 in response to exposure to P. aeruginosa antigens, implying that lens wear per se may interfere with the ocular surface HDP innate response. Also, little is known about individual genetic variation in the host response that increases susceptibility to corneal infection. Recent research suggests copy number variation and/or polymorphisms in HDP genes (defensins in particular) contribute to susceptibility to infection and may underlie the onset of chronic inflammatory diseases such as psoriasis and Crohn’s disease [93–95]. Interestingly, Carter et al. [96] reported a case of bilateral endophthalmitis associated with the hBD-1 genotype -44CC, an allele previously shown to be associated with a predisposition to infection. Endophthalmitis is a rare complication of cataract surgery. Most cases are due to infection and result in vision-threatening inflammation of the intraocular cavities. A subsequent study by the same group with a larger patient cohort showed the -44CC genotype to be more common in patients who have had endophthalmitis suggesting a potential genetic risk factor that reduces innate immune defences in this intra-ocular infection [97]. An association between such genetic variations and microbial keratitis has not yet been investigated but could provide novel insight into an individual’s predisposition to infection that could be incorporated into a genome-based approach to treatment in which care (e.g. the recommendation to wear contact lenses or not) is tailored to individuals based on their specific genetic make-up.
Inflammatory conditions
Dry eye syndrome is a very common ocular surface condition resulting from decreased tear secretion or increased tear evaporation and is one of the leading causes for visits to an eye doctor [98]. It is a multifactorial condition in which inflammation plays a major role. Tear fluid hyperosmolality and tear film instability stimulate an increase in pro-inflammatory cytokines and the resulting inflammation further exacerbates the response leading to damage to the ocular surface and symptoms of dryness, irritation and pain [99–104]. We observed that hBD-2 was specifically upregulated in the conjunctiva of patients with dry eye [11]. This was also confirmed in patients with Sjogren’s syndrome, which results in a very severe form of dry eye due to autoimmune destruction of the lacrimal gland [105]. Pro-inflammatory cytokines IL-1β and TNFα are known to be elevated in dry eye and were able to stimulate hBD-2 expression in human conjunctival epithelial cells in culture leading to the suggestion that they may be responsible for the elevated expression in dry eye patients [11]. However, when this was investigated further, a hyperosmolar culture environment was actually found to blunt IL-1β stimulation of hBD-2 production [106] suggesting that this cytokine may not have a major role to play in modulating ocular surface hBD-2 expression in dry eye. Interestingly, while hBD-1 and 3 were unchanged and hBD-2 upregulated in patients with dry eye, another defensin, hBD-9, was found to be decreased [20]. Murine models have been important in elucidating the mechanisms underlying dry eye, however when defensin expression was examined in the cornea and conjunctiva of mice with experimental dry eye, the changes compared to normal mice were not dramatic [34]. Thus, the mechanisms underlying modulated defensin expression in dry eye have yet to be established. Further, the specific role of these HDPs in dry eye is also still a subject for investigation. That enhanced expression of hBD-2 will give additional antimicrobial protection is a logical speculation and is in keeping with observations that dry eye patients (with the exception perhaps of those with Sjogren’s syndrome [107]) do not commonly present with infection despite having a disrupted ocular surface. On the other hand, hBD-9 is not expected to have a major antimicrobial effect, therefore its reduced expression in dry eye is not likely to compromise innate immunity [20]. HDPs are of course known to have a wide range of activities in addition to their antimicrobial actions, thus their altered expression in dry eye may influence several aspects of the disease process, for example through their chemotactic effects, they may help mediate T cell recruitment [108].
Hida et al. [109] found that α-defensins were elevated in the tears of patients with ocular allergies complicated by the presence of corneal lesions and speculated that this may help prevent secondary infection in these patients. However, as the elevated levels were only seen in patients with lesions, this observation appears to be linked to corneal injury (discussed in section “Wound healing”) rather than the allergic response per se. Ikeda et al. [110] noted that mRNA expression for mBD-1 was unchanged but that of mBD-2 was decreased compared to normal mice in a model of allergic conjunctivitis. The authors note that patients with ocular allergies are susceptible to infection and extrapolate from their findings to suggest that this may be associated with reduced hBD-2 expression. However as mBD-2 does not appear to be an orthologue of hBD-2, the ultimate significance of the findings in this mouse model are unknown.
An early-discovered property of LL-37 is that it binds and inactivates LPS, a component of the cell wall of Gram-negative bacteria that stimulates a strong inflammatory response through activation of TLR4 [111, 112]. Thus, endogenously expressed LL-37 may actually help reduce ocular inflammation associated with bacterial infection. Ohgami et al. [113] reported that hCAP-18 (the LL-37 precursor) suppressed the acute inflammatory response in a rat model of LPS-induced uveitis and suggested that this peptide may be a promising agent for the treatment of inflammation. Interestingly, Jager et al. [114, 115] recently detected the presence of cationic amino acid transporters at the ocular surface and lacrimal apparatus and suggested there is a functional relationship between them and β-defensins in a regulatory circuit via which the balance toward β-defensin production could be shifted to treat atopic and inflammatory diseases. An alternative role for endogenous HDPs, although one that has yet to be investigated, is that some may actually exacerbate an inflammatory response so contributing to disease processes. This may be mediated through HDP cytotoxic effects (cytotoxic levels may be reached for peptides whose expression is upregulated by pro-inflammatory cytokines), ability to induce mast cell degranulation [116, 117] and influence cytokine secretion [25, 118–121] and leukocyte migration [122–124].
Wound healing
The most commonly studied ocular tissue in terms of wound healing is the ocular surface, in particular the cornea. This is because rapid healing of the cornea is essential to restore a smooth surface for refraction of light and barrier function to prevent pathogen penetration, which may otherwise lead to fulminant infection and permanent vision loss. Studies carried out over more than two decades have shown a substantial redundancy in terms of endogenous factors capable of modulating wound healing at the ocular surface, a testament to the importance of having healing happen rapidly and effectively [125]. Recent studies provide evidence that HDPs may also contribute to these processes.
The expression of some HDPs, such as hBD-2 and LL-37, is increased, and neutrophils, a source of α-defensins and LL-37, are known to infiltrate the cornea after injury [25, 126]. Notably several studies have found that these HDPs can modulate activities such as migration, proliferation and cytokine production, all of which are essential for accurate and timely ocular surface healing [125]. For example, through activation of formyl peptide receptor-like 1 and transactivation of the epidermal growth factor receptor, LL-37 stimulated migration and cytokine secretion by corneal epithelial cells [25]. Notably, LL-37 also promoted wound closure in a corneal epithelial cell monolayer scratch model and promoted healing in a corneal organ culture model in which a high glucose environment was used to slow healing so mimicking the delayed corneal re-epithelialisation seen in diabetic patients [127]. Further, HNP-1, hBD-2 and hBD-3 were found to stimulate cytokine production by conjunctival epithelial cells, and HNP-1 and hBD-2 enhanced proliferation and collagen gene expression and modulated matrix metalloproteinase production by conjunctival fibroblasts [128, 129]. Overall these findings support a role for defensins and LL-37 in modulating cell migration and proliferation during corneal wound healing and tissue remodeling, effects that may be direct receptor-mediated actions or indirect via cytokine production. In most tissues, neovascularisation is an important component of wound healing. However, the development of new blood vessels in the cornea reduces corneal transparency resulting in visual dysfunction. It is of significance therefore that, while LL-37 is pro-angiogenic, HNPs are antiangiogenic [130–132]. Thus, the relative levels of LL-37 (from epithelial cells and infiltrating neutrophils) and HNPs (from infiltrating neutrophils) may contribute to maintaining corneal avascularity after injury. Although not tested at the ocular surface, HNPs inhibited neovascularisation in the retina, another ocular tissue in which pathological vessel growth in conditions such as diabetic retinopathy can lead to profound visual loss, even blindness [132].
That endogenously expressed peptides can influence and stimulate the various cell behaviours important for wound healing at the ocular surface also suggests that HDPs or their derivatives/mimetics may be useful as topical agents to promote corneal healing responses in diseases such as diabetes and following accidental injury or elective procedures such as refractive surgeries that involve damage to the ocular surface.
Host-defence peptides as therapeutics for eye diseases
To date the majority of studies of HDPs as potential therapeutics for eye diseases have focused on their anti-infective properties for microbial keratitis. As discussed earlier (section “Defence against infection”) this affects the cornea, the major refracting surface of the eye, and if not adequately and rapidly treated may result in permanent loss of vision. As is the case in many infectious diseases, the emergence of resistant strains is a growing issue in the treatment of microbial keratitis and is driving the search for novel antimicrobial agents. As discussed by others, through their mechanism of action, HDPs have reduced potential for development of resistance [133], thus several studies have investigated the potential of using HDPs or their derivatives/mimetics in a variety of therapeutic paradigms for treating or preventing corneal infection.
Topical application
Ease of access to the cornea makes it an ideal candidate for topical drug application, and there have been a small number of studies investigating HDP efficacy in animal models of microbial keratitis. Employing synthetic peptides containing partial sequences from cecropin A (an insect peptide) and melittin (from bee venom), Nos-Barbera et al. [134] noted reduced inflammation and less corneal damage compared to vehicle control in a rabbit model of P. aeruginosa keratitis. Also, retrocyclin 2, a synthetic θ-defensin, had antiviral activity in a murine model of HSV-1 keratitis when given prophylactically, although not when administered for an established infection [135]. However, in contrast to these at least somewhat successful studies, a synthetic peptide COL-1, which had potent antimicrobial activity in vitro, did not show a clinical benefit in a rabbit model of P. aeruginosa keratitis. Rather the peptide-treated animals showed more inflammation than controls [136]. This latter study highlights two major issues, namely lack of efficacy and toxicity. Although the reason for the lack of effect with COL-1 is not known, it is possible that it may have been the result of inactivation of the peptide by components in the tear film. Studies have shown that tears do interfere with the antimicrobial activity of some HDPs [19, 25, 137]. Also as discussed earlier (section “Defence against infection”), while this is in part attributable to the salt content of tears, MUC5AC also dramatically reduced the activity of hBD-2 against P. aeruginosa [54]. Notably tears, over and above the effects of salt, also inhibited the antibacterial activity of hBD-1, whereas hBD-3 and LL-37 were substantially less affected [19, 25]. These observations highlight the need for thorough investigation into the potential detrimental effects of tears on HDP activity before embarking on in vivo studies. We also observed that various artificial tear solutions also reduced HDP antimicrobial activity [138]. This is of some concern as these over-the-counter preparations are commonly used to relieve the symptoms of dry eye, and patients may inadvertently be suppressing the innate response so increasing the risk for ocular infection. Another issue with the use of HDPs as ocular therapeutics is toxicity. Although HDPs show selectivity for microbial membranes, for some peptides, this is lost at higher concentrations and they become toxic to mammalian cells [3]. This is an obvious issue as high peptide concentrations may be warranted to overcome the effects of interfering entities such as tear film salt and mucins. Recent studies have shown that lowering the hydrophobicity of linear hBD-3 analogues reduces cytotoxicity to ocular surface epithelial cells while maintaining antimicrobial efficacy, so revealing one possible approach to overcoming the toxicity issue [139, 140]. Covalent dimerisation and multivalent forms of HDPs are a further strategy to help address issues such as cytotoxicity [141]. We have observed that tethering of an HDP (in our case a derivative of the cathelicidin LL-37) to a “surface” greatly enhances its efficacy against ocular pathogens compared to peptide in solution [142–144]. Thus, we believe that attachment of an HDP to a surface (such as a contact lens as described in section “Antimicrobial contact lenses”) or a nanoparticle scaffold such as a liposome or dendrimer is much more likely to result in potent non-cytotoxic antimicrobial activity at the ocular surface than simply applying the peptide in solution and so represents a very promising mode of delivery.
Corneal preservation media and contact lens care solutions
Corneal transplants are the most common and most successful solid tissue transplant performed in the US, with more than 40,000 being performed per year. After removal from the donor, the tissue is placed into a preservation medium which helps prevent bacterial growth while the cornea is in storage at 4°C until it is used for transplantation. Again, the spread of organisms resistant to the traditional antibacterial agents contained within these preservation media has led to a search for alternative components. Schwab et al. [145] tested defensin antimicrobial activity against common ocular pathogenic bacteria in the corneal preservation medium Optisol™ and found it to be effective at 4°C so revealing the potential of defensins as preservatives for corneal storage. In a related study, the bovine peptide BNP-1 was found to help preserve endothelial cell (essential for normal corneal transparency) viability in stored corneas [146]. Thus HDPs, when added to corneal storage media, may have dual benefits. Notably, BNP-1 as part of a mix of trophic factors has also been found to improve kidney preservation [147].
As noted previously, contact lens wear is a major risk factor for the development of microbial keratitis, particularly that caused by P. aeruginosa. An important factor in minimising this risk is adequate disinfection of the lenses during cleaning and overnight storage. Cecropin D5C, a synthetic insect antimicrobial peptide, has been shown to augment the antimicrobial activity of contact lens solutions against P. aeruginosa so supporting a potential use of HDPs as effective contact lens disinfectants [148]. The mid-2000s saw outbreaks of both Fusarium and Acanthamoeba keratitis related to contact lens wear and use of specific contact lens solutions, highlighting the need for improved contact lens hygiene practices and contact lens disinfectant test regimens [89]. Notably, a number of HDPs have potent antifungal activity [72–80], and the amphibian peptide magainin has been found to have activity against Acanthamoeba [90]; thus, they may be useful as novel disinfectants for elimination of potentially vision-threatening pathogens such as Fusarium and Acanthamoeba.
One potential factor limiting the use of HDPs in these sorts of clinical applications is that relatively large volumes would be required and this is likely to be prohibitively expensive. It may however be possible to circumvent some of this cost by tethering an HDP to a nanoparticle so enhancing its efficacy and reducing the amount of peptide needed and/or using the shortest peptide sequence required for optimal antimicrobial activity.
Antimicrobial contact lenses
As contact lens wear is associated with increased risk for corneal infection there has been significant interest in the development of contact lenses with antimicrobial, and specifically antipseudomonal, properties. Contact lens–associated microbial keratitis is the result of colonisation of the lens surface which then leads to contamination of the cornea upon insertion of the lens into the eye. Thus, major efforts are being directed toward development of lenses which prevent bacterial adherence and biofilm formation. Many potential agents have been investigated but development of microbial resistance is an issue as is loss of activity after covalent attachment, inability to sterilise the lenses after attachment of the antimicrobial and toxicity to ocular surface cells. Nevertheless, some agents have been identified that hold particular promise, and those currently under investigation include selenium [149], quorum-sensing inhibitors such as fimbrolides [150] and antimicrobial peptides [151, 152]. In regards to the latter, Willcox et al. [151] developed a synthetic cationic peptide termed melimine which combined active but minimally cytotoxic sequences from mellitin (from bee venom) and protamine (from salmon sperm). This hybrid peptide, when adsorbed or covalently linked to contact lenses, reduced colonisation by P. aeruginosa and S. aureus by 70–80%. Notably, melimine did not induce resistance after repeated exposure of the organisms to sub-inhibitory concentrations of the peptide and was heat-stable. Subsequently, the effectiveness of melimine covalently incorporated into silicone hydrogel lenses was tested in animal models in vivo [152]. In a guinea pig model of contact lens acute red eye induced by P. aeruginosa, animals fitted with melimine-coated lenses had significantly less ocular inflammation, chemosis and discharge compared to animals fitted with control lenses. Corneal infiltrates were also markedly reduced. Similarly, melimine-coated lenses performed favourably in a rabbit model of contact lens–induced peripheral ulcer caused by S. aureus. Here rabbits fitted with the melimine-coated lenses had significantly lower scores for ocular inflammation and a threefold reduction in the presence of a corneal epithelial defect compared to those wearing control lenses. These studies highlight the potential of HDPs and derivatives thereof as coatings for antimicrobial contact lenses. Also it is encouraging that, although no antimicrobial contact lens has yet been approved, in a very closely related arena, antimicrobial contact lens cases, which exploit the antibacterial activity of the well-known biocide silver, are now on the market.
Summary
In summary, eye and associated tissues such as the lacrimal apparatus express a range of HDPs which help prevent infection and are likely involved in wound-healing responses and tissue remodeling. These endogenous HDPs may also be involved in ocular inflammatory and immune responses although much remains to be discovered about such roles. Development of an HDP or derivative/mimic thereof for therapeutic purposes is an attractive proposition, and because of ease of application (topical or local injection), which reduces some of the confounding factors associated with systemic delivery, is indeed a viable option.
Acknowledgments
The authors thank Ms. Kimberly Thompson of the University of Houston, College of Optometry Audio Visual Services for drawing Fig. 1. The authors’ research on HDPs in the eye is supported by grants from the National Eye Institute (EY13175, EY7551) and collaborative grants with Dr. C. Cai awarded by the National Science Foundation (DMR-0706627) and National Eye Institute (EY18303).
Abbreviations
- HDP
Host-defence peptide
- hBD
Human β-defensin
- mBD
Mouse β-defensin
- HNP
Human neutrophil peptide
- LL-37
Human cathelicidin
- CRAMP
Cathelin-related antimicrobial peptide
- LPS
Lipopolysaccharide
References
- 1.Hale JD, Hancock RE. Alternative mechanisms of action of cationic antimicrobial peptides on bacteria. Expert Rev Anti Infect Ther. 2007;5:951–959. doi: 10.1586/14787210.5.6.951. [DOI] [PubMed] [Google Scholar]
- 2.Radek K, Gallo R. Antimicrobial peptides: natural effectors of the innate immune system. Semin Immunopathol. 2007;29:27–43. doi: 10.1007/s00281-007-0064-5. [DOI] [PubMed] [Google Scholar]
- 3.McDermott AM. Antimicrobial peptides. In: Howl J, Jones S, editors. Bioactive peptides. Boca Raton: CRC Press; 2009. [Google Scholar]
- 4.Nijnik A, Hancock RE. The roles of cathelicidin LL-37 in immune defences and novel clinical applications. Curr Opin Hematol. 2009;16:41–47. doi: 10.1097/MOH.0b013e32831ac517. [DOI] [PubMed] [Google Scholar]
- 5.Steinstraesser L, Kraneburg U, Jacobsen F, Al-Benna S. Host defense peptides and their antimicrobial-immunomodulatory duality. Immunobiology. 2011;216(3):322–333. doi: 10.1016/j.imbio.2010.07.003. [DOI] [PubMed] [Google Scholar]
- 6.Hattenbach LO, Gumbel H, Kippenberger S. Identification of beta-defensins in human conjunctiva. Antimicrob Agents Chemother. 1998;42:3332. doi: 10.1128/aac.42.12.3332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Haynes RJ, Tighe PJ, Dua HS. Antimicrobial defensin peptides of the human ocular surface. Br J Ophthalmol. 1999;83:737–741. doi: 10.1136/bjo.83.6.737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.McNamara NA, Van R, Tuchin OS, Fleiszig SMJ. Ocular surface epithelia express mRNA for human beta defensin-2. Exp Eye Res. 1999;69:483–490. doi: 10.1006/exer.1999.0722. [DOI] [PubMed] [Google Scholar]
- 9.Lehmann OJ, Hussain IR, Watt PJ. Investigation of β-defensin gene expression in the ocular anterior segment by semiquantitative RT-PCR. Br J Ophthalmol. 2000;84:523–526. doi: 10.1136/bjo.84.5.523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McDermott AM, Redfern RL, Zhang B, Pei Y, Huang L, Proske RJ. Defensin expression by the cornea: multiple signaling pathways mediate IL-1 beta stimulation of hBD-2 expression by corneal epithelial cells. Invest Ophthalmol Vis Sci. 2003;44:1859–1865. doi: 10.1167/iovs.02-0787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Narayanan S, Miller WL, McDermott AM. Expression of human beta-defensins in conjunctival epithelium: relevance to dry eye disease. Invest Ophthalmol Vis Sci. 2003;44:3795–3801. doi: 10.1167/iovs.02-1301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kumar A, Zhang J, Yu FS. Innate immune response of corneal epithelial cells to Staphylococcus aureus infections: role of peptidoglycan in stimulating proinflammatory cytokine secretion. Invest Ophthalmol Vis Sci. 2004;45:3513–3522. doi: 10.1167/iovs.04-0467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shin JS, Kim CW, Kwon YS, Kim JC. Human beta-defensin 2 is induced by interleukin-1 beta in the corneal epithelial cells. Exp Mol Med. 2004;36:204–210. doi: 10.1038/emm.2004.28. [DOI] [PubMed] [Google Scholar]
- 14.Kumar A, Zhang J, Yu FS. Toll-like receptor 2-mediated expression of beta-defensin-2 in human corneal epithelial cells. Microbes Infect. 2006;8:380–389. doi: 10.1016/j.micinf.2005.07.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kumar A, Yin J, Zhang J, Yu F-SX. Modulation of corneal epithelial innate immune response to Pseudomonas infection by flagellin pretreatment. Invest Ophthalmol Vis Sci. 2007;48:4664–4670. doi: 10.1167/iovs.07-0473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li Q, Kumar A, Gui JF, Yu FS. Staphylococcus aureus lipoproteins trigger human corneal epithelial innate response through toll-like receptor-2. Microb Pathog. 2008;44:426–434. doi: 10.1016/j.micpath.2007.11.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Garreis F, Schlorf T, Worlitzsch D, Steven P, Bräuer L, Jäger K, Paulsen FP. Roles of human beta-defensins in innate immune defense at the ocular surface: arming and alarming corneal and conjunctival epithelial cells. Histochem Cell Biol. 2010;134:59–73. doi: 10.1007/s00418-010-0713-y. [DOI] [PubMed] [Google Scholar]
- 18.McIntosh RS, Cade JE, Al-Abed M, Shanmuganathan V, Gupta R, Bhan A, Tighe PJ, Dua HS. The spectrum of antimicrobial peptide expression at the ocular surface. Invest Ophthalmol Vis Sci. 2005;46:1379–1385. doi: 10.1167/iovs.04-0607. [DOI] [PubMed] [Google Scholar]
- 19.Huang LC, Jean D, Proske RJ, Reins RY, McDermott AM. Ocular surface expression and in vitro activity of antimicrobial peptides. Curr Eye Res. 2007;32:595–609. doi: 10.1080/02713680701446653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abedin A, Mohammed I, Hopkinson A, Dua HS. A novel antimicrobial peptide on the ocular surface shows decreased expression in inflammation and infection. Invest Ophthalmol Vis Sci. 2008;49:28–33. doi: 10.1167/iovs.07-0645. [DOI] [PubMed] [Google Scholar]
- 21.Mohammed I, Suleman H, Otri A, Kulkarni BB, Chen P, Hopkinson A, Dua HS. Localization and gene expression of human β-defensin 9 at the human ocular surface epithelium. Invest Ophthalmol Vis Sci. 2010;51:4677–4682. doi: 10.1167/iovs.10-5334. [DOI] [PubMed] [Google Scholar]
- 22.Gottsch JD, Li Q, Ashraf MF, O’Brien TP, Stark WJ, Liu SH. Defensin gene expression in the cornea. Curr Eye Res. 1998;17:1082–1086. doi: 10.1076/ceyr.17.11.1082.5235. [DOI] [PubMed] [Google Scholar]
- 23.Haynes RJ, Tighe PJ, Dua HS. Innate defence of the eye by antimicrobial defensin peptides. Lancet. 1998;352:451–452. doi: 10.1016/S0140-6736(05)79185-6. [DOI] [PubMed] [Google Scholar]
- 24.Gordon YJ, Huang LC, Romanowski EG, Yates KA, Proske RJ, McDermott AM. Human cathelicidin (LL-37), a multifunctional peptide, is expressed by ocular surface epithelia and has potent antibacterial and antiviral activity. Curr Eye Res. 2005;30:1–10. doi: 10.1080/02713680490522399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Huang LC, Petkova TD, Reins RY, Proske RJ, McDermott AM. Multifunctional roles of human cathelicidin (LL-37) at the ocular surface. Invest Ophthalmol Vis Sci. 2006;47:2369–2380. doi: 10.1167/iovs.05-1649. [DOI] [PubMed] [Google Scholar]
- 26.Sorensen O, Arnljots K, Cowland JB, Bainton DF, Borregaard N. The human antibacterial cathelicidin, hCAP18, is synthesized in myelocytes and metamyelocytes and localized to specific granules in neutrophils. Blood. 1997;90:2796–2803. [PubMed] [Google Scholar]
- 27.Steele PS, Jumblatt MM (2004) Defense proteins of the ocular surface. Invest Ophthalmol Vis Sci 45: e-abstract 3792
- 28.Cole AM, Ganz T, Liese AM, Burdick MD, Liu L, Strieter RM. Cutting edge: IFN-inducible ELR-CXC chemokines display defensin-like antimicrobial activity. J Immunol. 2001;167:623–627. doi: 10.4049/jimmunol.167.2.623. [DOI] [PubMed] [Google Scholar]
- 29.Yang D, Chen Q, Hoover DM, Staley P, Tucker KD, Lubkowski J, Oppenheim JJ. Many chemokines including CCL20/MIP-3alpha display antimicrobial activity. J Leukoc Biol. 2003;74:448–455. doi: 10.1189/jlb.0103024. [DOI] [PubMed] [Google Scholar]
- 30.Spandau UH, Toksoy A, Verhaart S, Gillitzer R, Kruse FE. High expression of chemokines Gro-alpha (CXCL-1), IL-8 (CXCL-8), and MCP-1 (CCL-2) in inflamed human corneas in vivo. Arch Ophthalmol. 2003;121:825–831. doi: 10.1001/archopht.121.6.825. [DOI] [PubMed] [Google Scholar]
- 31.Garreis F, Gottschalt M, Glaser R, Harder J, Worlitzsch D, Paulsen FP. Expression and regulation of antimicrobial peptide psoriasin (S100A7) in the lacrimal apparatus. Invest Ophthalmol Vis Sci. 2009;50:5515. doi: 10.1167/iovs.10-6598. [DOI] [PubMed] [Google Scholar]
- 32.Mohammed I, Yeung A, Abedin A, Hopkinson A, Dua HA (2010) Signalling pathways involved in ribonuclease-7 expression. Cell Mol Life Sci. doi:10.1007/s00018-010-0540-2 [DOI] [PMC free article] [PubMed]
- 33.Huang LC, Reins RY, Gallo RL, McDermott AM. Cathelicidin-deficient (Cnlp−/−) mice show increased susceptibility to Pseudomonas aeruginosa keratitis. Invest Ophthalmol Vis Sci. 2007;48:4498–4508. doi: 10.1167/iovs.06-0320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Narayanan S, Corrales RM, Farley W, McDermott AM, Pflugfelder SC. Interleukin-1 receptor-1-deficient mice show attenuated production of ocular surface inflammatory cytokines in experimental dry eye. Cornea. 2008;27:811–817. doi: 10.1097/ICO.0b013e31816bf46c. [DOI] [PubMed] [Google Scholar]
- 35.Wu M, McClellan SA, Barrett RP, Hazlett LD. Beta-defensin-2 promotes resistance against infection with P. aeruginosa . J Immunol. 2009;182:1609–1616. doi: 10.4049/jimmunol.182.3.1609. [DOI] [PubMed] [Google Scholar]
- 36.Wu M, McClellan SA, Barrett RP, Zhang Y, Hazlett LD. Beta-defensin-2 and 3 together promote resistance to Pseudomonas aeruginosa keratitis. J Immunol. 2009;183:8054–8060. doi: 10.4049/jimmunol.0902140. [DOI] [PubMed] [Google Scholar]
- 37.Hinrichsen K, Podschun R, Schubert S, Schroder JM, Harder J, Proksch E. Mouse beta-defensin-14, an antimicrobial ortholog of human beta-defensin-3. Antimicrob Agents Chemother. 2008;52:1876–1879. doi: 10.1128/AAC.01308-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Tsubota K. Tear film overview. In: Dartt DA, editor. Encyclopedia of the eye. Amsterdam: Elsevier; 2010. [Google Scholar]
- 39.McDermott AM. Defense mechanisms of tears and ocular surface. In: Dartt DA, editor. Encyclopedia of the eye. Amsterdam: Elsevier; 2010. [Google Scholar]
- 40.Tan KO, Sack RA, Holden BA, Swarbrick HA. Temporal sequence of changes in tear film composition during sleep. Curr Eye Res. 1993;12:1001–1007. doi: 10.3109/02713689309029226. [DOI] [PubMed] [Google Scholar]
- 41.Zhou L, Huang LQ, Beuerman RW, Grigg ME, Li SF, Chew FT, Ang L, Stern ME, Tan D. Proteomic analysis of human tears: defensin expression after ocular surface surgery. J Proteome Res. 2004;3:410–416. doi: 10.1021/pr034065n. [DOI] [PubMed] [Google Scholar]
- 42.Steele PS, Jumblatt MM, Smith NB, Pierce WM. Detection of Histatin 5 in normal human Schirmer strip samples by mass spectroscopy. Invest Ophthalmol Vis Sci. 2002;43:98. [Google Scholar]
- 43.You Y, Fitzgerald A, Cozzi PJ, Zhao Z, Graham P, Russell PJ, Walsh BJ, Willcox M, Zhong L, Wasinger V, Li Y. Post-translation modification of proteins in tears. Electrophoresis. 2010;31:1853–1861. doi: 10.1002/elps.200900755. [DOI] [PubMed] [Google Scholar]
- 44.Haynes RJ, McElveen JE, Dua HS, Tighe PJ, Liversidge J. Expression of human beta-defensins in intraocular tissues. Invest Ophthalmol Vis Sci. 2000;41:3026–3031. [PubMed] [Google Scholar]
- 45.Paulsen FP, Pufe T, Schaudig U, Held-Feindt J, Lehmann J, Schroder JM, Tillmann BN. Detection of natural peptide antibiotics in human nasolacrimal ducts. Invest Ophthalmol Vis Sci. 2001;42:2157–2163. [PubMed] [Google Scholar]
- 46.Stoeckelhuber M, Messmer EM, Schubert C, Stoeckelhuber BM, Koehler C, Welsch U, Bals R. Immunolocalization of defensins and cathelicidin in human glands of Moll. Ann Anat. 2008;190:230–237. doi: 10.1016/j.aanat.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 47.Stoeckelhuber M, Stoeckelhuber BM, Welsch U. Apocrine glands in the eyelids of primates contribute to the ocular host defense. Cells Tissues Organs. 2004;176:187–194. doi: 10.1159/000077035. [DOI] [PubMed] [Google Scholar]
- 48.Yasui T, Tsukise A, Nara T, Kuwahara Y, Meyer W. Morphological, histochemical and immunohistochemical characterization of secretory production of the ciliary glands in the porcine eyelid. Eur J Histochem. 2006;50:99–108. [PubMed] [Google Scholar]
- 49.McDermott AM. The role of antimicrobial peptides at the ocular surface. Ophthalmic Res. 2009;41:60–75. doi: 10.1159/000187622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Pachigolla G, Blomquist P, Cavanagh HD. Microbial keratitis pathogens and antibiotic susceptibilities: a 5-year review of cases at an urban county hospital in north Texas. Eye Contact Lens. 2007;33:45–49. doi: 10.1097/01.icl.0000234002.88643.d0. [DOI] [PubMed] [Google Scholar]
- 51.Green M, Apel A, Stapleton F. Risk factors and causative organisms in microbial keratitis. Cornea. 2008;27:22–27. doi: 10.1097/ICO.0b013e318156caf2. [DOI] [PubMed] [Google Scholar]
- 52.Oren A, Ganz T, Liu L, Meerloo T. In human epidermis, β-defensin 2 is packaged in lamellar bodies. Exp Mol Pathol. 2003;74:180–182. doi: 10.1016/S0014-4800(02)00023-0. [DOI] [PubMed] [Google Scholar]
- 53.Braff MH, Di Nardo A, Gallo RL. Keratinocytes store the antimicrobial peptide cathelicidin in lamellar bodies. J Invest Dermatol. 2005;124:394–400. doi: 10.1111/j.0022-202X.2004.23443.x. [DOI] [PubMed] [Google Scholar]
- 54.Huang LC, Redfern RL, Narayanan S, Reins RY, McDermott AM. In vitro activity of human beta-defensin 2 against Pseudomonas aeruginosa in the presence of tear fluid. Antimicrob Agents Chemother. 2007;51:3853–3860. doi: 10.1128/AAC.01317-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Singh PK, Tack BF, McCray PB, Jr, Welsh MJ. Synergistic and additive killing by antimicrobial factors found in human airway surface liquid. Am J Physiol Lung Cell Mol Physiol. 2000;279:L799–L805. doi: 10.1152/ajplung.2000.279.5.L799. [DOI] [PubMed] [Google Scholar]
- 56.Nagaoka I, Hirota S, Yomogida S, Ohwada A, Hirata M. Synergistic actions of antibacterial neutrophil defensins and cathelicidins. Inflamm Res. 2000;49:73–79. doi: 10.1007/s000110050561. [DOI] [PubMed] [Google Scholar]
- 57.Chen X, Niyonsaba F, Ushio H, Okuda D, Nagaoka I, Ikeda S, Okumura K, Ogawa H. Synergistic effect of antibacterial agents human beta-defensins, cathelicidin LL-37 and lysozyme against Staphylococcus aureus and Escherichia coli . J Dermatol Sci. 2005;40:123–132. doi: 10.1016/j.jdermsci.2005.03.014. [DOI] [PubMed] [Google Scholar]
- 58.Kumar A, Hazlett LD, Yu FS. Flagellin suppresses the inflammatory response and enhances bacterial clearance in a murine model of Pseudomonas aeruginosa keratitis. Infect Immun. 2008;76:89–96. doi: 10.1128/IAI.01232-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Augustin DK, Heimer SR, Tan C, Li WY, Le Due JM, Evans DJ, Fleiszig SM (2011) Role of defensins in corneal epithelial barrier function against P. aeruginosa traversal. Infect Immun 9(2):595–605 [DOI] [PMC free article] [PubMed]
- 60.Liesegang TJ, Melton LJ, Daly PJ, Ilstrup DM. Epidemiology of ocular herpes simplex. Incidence in Rochester, Minn, 1950 through 1982. Arch Ophthalmol. 1989;107:1155–1159. doi: 10.1001/archopht.1989.01070020221029. [DOI] [PubMed] [Google Scholar]
- 61.Daher KA, Selsted ME, Lehrer RI. Direct inactivation of viruses by human granulocyte defensins. J Virol. 1986;60:1068–1074. doi: 10.1128/jvi.60.3.1068-1074.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bastian A, Schafer H. Human alpha-defensin 1 (HNP-1) inhibits adenoviral infection in vitro. Regul Pept. 2001;101:157–161. doi: 10.1016/S0167-0115(01)00282-8. [DOI] [PubMed] [Google Scholar]
- 63.Liesegang TJ, Forster RK. Spectrum of microbial keratitis in south Florida. Am J Ophthalmol. 1980;90:38–47. doi: 10.1016/s0002-9394(14)75075-5. [DOI] [PubMed] [Google Scholar]
- 64.Ritterband DC, Seedor JA, Shah MK, Koplin RS, McCormick SA. Fungal keratitis at the New York Eye and Ear Infirmary. Cornea. 2006;25:264–267. doi: 10.1097/01.ico.0000177423.77648.8d. [DOI] [PubMed] [Google Scholar]
- 65.Sirikul T, Prabriputaloong T, Smathivat A, Chuck RS, Vongthongsri A. Predisposing factors and etiologic diagnosis of ulcerative keratitis. Cornea. 2008;27:283–287. doi: 10.1097/ICO.0b013e31815ca0bb. [DOI] [PubMed] [Google Scholar]
- 66.Gopinathan U, Sharma S, Garg P, Rao GN. Review of epidemiological features, microbiological diagnosis and treatment outcome of microbial keratitis: experience of over a decade. Indian J Ophthalmol. 2009;57:273–279. doi: 10.4103/0301-4738.53051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Tanure MA, Cohen EJ, Sudesh S, Rapuano CJ, Laibson PR. Spectrum of fungal keratitis at Wills Eye Hospital, Philadelphia, Pennsylvania. Cornea. 2000;19:307–312. doi: 10.1097/00003226-200005000-00010. [DOI] [PubMed] [Google Scholar]
- 68.Thew MR, Todd B. Fungal keratitis in far north Queensland, Australia. Clin Exp Ophthalmol. 2008;36:721–724. doi: 10.1111/j.1442-9071.2008.01879.x. [DOI] [PubMed] [Google Scholar]
- 69.Xie L, Zhai H, Zhao J, Sun S, Shi W, Dong X. Antifungal susceptibility for common pathogens of fungal keratitis in Shandong Provice, China. Am J Ophthalmol. 2008;146:260–265. doi: 10.1016/j.ajo.2008.04.019. [DOI] [PubMed] [Google Scholar]
- 70.Perez-Balbuena AL, Vanzzini-Rosano V, Valadez-Virgen J, Campos-Möller X. Fusarium keratitis in Mexico. Cornea. 2009;28:626–630. doi: 10.1097/ICO.0b013e31819bc2ea. [DOI] [PubMed] [Google Scholar]
- 71.Furlanetto RL, Andreo EG, Finotti IG, Arcieri ES, Ferreira MA, Rocha FJ. Epidemiology and etiologic diagnosis of infectious keratitis in Uberlandia, Brazil. Eur J Ophthalmol. 2010;20:498–503. doi: 10.1177/112067211002000312. [DOI] [PubMed] [Google Scholar]
- 72.Lehrer RI, Ganz T, Szklarek D, Selsted ME. Modulation of the in vitro candidacidal activity of human neutrophil defensins by target cell metabolism and divalent cations. J Clin Invest. 1998;81:1829–1835. doi: 10.1172/JCI113527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Wilde CG, Griffith JE, Marra MN, Snable JL, Scott RW. Purification and characterization of human neutrophil peptide 4, a novel member of the defensin family. J Biol Chem. 1989;264:11200–11203. [PubMed] [Google Scholar]
- 74.Raj PA, Antonyraj KJ, Karunakaran T. Large-scale synthesis and functional elements for the antimicrobial activity of defensins. Biochem J. 2000;347:633–641. doi: 10.1042/0264-6021:3470633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Harder J, Bartels J, Christophers E, Schroder JM. Isolation and characterization of human beta-defensin-3, a novel human inducible peptide antibiotic. J Biol Chem. 2001;276:5707–5713. doi: 10.1074/jbc.M008557200. [DOI] [PubMed] [Google Scholar]
- 76.Hoover DM, Wu Z, Tucker K, Lu W, Lubkowski J. Antimicrobial characterization of human beta-defensin 3 derivatives. Antimicrob Agents Chemother. 2003;47:2804–2809. doi: 10.1128/AAC.47.9.2804-2809.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.López-García B, Lee PH, Yamasaki K, Gallo RL. Anti-fungal activity of cathelicidins and their potential role in Candida albicans skin infection. J Invest Dermatol. 2005;125:108–115. doi: 10.1111/j.0022-202X.2005.23713.x. [DOI] [PubMed] [Google Scholar]
- 78.den Hertog AL, van Marle J, van Veen HA, Van’t Hof W, Bolscher JG, Veerman EC, Nieuw Amerongen AV. Candidacidal effects of two antimicrobial peptides: histatin 5 causes small membrane defects, but LL-37 causes massive disruption of the cell membrane. Biochem J. 2005;388:689–695. doi: 10.1042/BJ20042099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Feng Z, Jiang B, Chandra J, Ghannoum M, Nelson S, Weinberg A. Human beta-defensins: differential activity against candidal species and regulation by Candida albicans . J Dent Res. 2005;84:445–450. doi: 10.1177/154405910508400509. [DOI] [PubMed] [Google Scholar]
- 80.Kiehne K, Brunke G, Meyer D, Harder J, Herzig KH. Oesophageal defensin expression during Candida infection and reflux disease. Scand J Gastroenterol. 2005;40:501–507. doi: 10.1080/00365520510015386. [DOI] [PubMed] [Google Scholar]
- 81.Ooi EH, Wormald P-J, Carney AS, James CL, Tan LW. Fungal allergens induce cathelicidin LL-37 expression in chronic rhinosinusitis patients in a nasal explant model. Am J Rhinol. 2007;21:367–372. doi: 10.2500/ajr.2007.21.3025. [DOI] [PubMed] [Google Scholar]
- 82.Krishnakumari V, Rangaraj N, Nagaraj R. Antifungal activities of human beta-defensins HBD-1 to HBD-3 and their C-terminal analogs Phd1 to Phd3. Antimicrob Agents Chemother. 2009;53:256–260. doi: 10.1128/AAC.00470-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Alekseeva L, Huat D, Femenia F, Mouyna I, Abdelouahab M, Cagna A, Guerrier D, Tichanné-Seltzer V, Baeza-Squiban A, Chermette R, Latgé JP, Berkova N. Inducible expression of beta defensins by human respiratory epithelial cells exposed to Aspergillus fumigatus organisms. BMC Microbiol. 2009;9:33. doi: 10.1186/1471-2180-9-33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Steubesand N, Kiehne K, Brunke G, Pahl R, Reiss K, Herzig KH, Schubert S, Schreiber S, Fölsch UR, Rosenstiel P, Arlt A. The expression of the beta-defensins hBD-2 and hBD-3 is differentially regulated by NFkB and MAPK/AP-1 pathways in an in vitro model of Candida esophagitis . BMC Immunol. 2009;10:36. doi: 10.1186/1471-2172-10-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bahri R, Saidane-Mosbahi D, Rouabhia M. Candida famata modulates toll-like receptor, beta-defensin, and proinflammatory cytokine expression by normal human epithelial cells. J Cell Physiol. 2010;222:209–218. doi: 10.1002/jcp.21939. [DOI] [PubMed] [Google Scholar]
- 86.Bahri R, Curt S, Saidane-Mosbahi D, Rouabhia M (2010) Normal human gingival epithelial cells sense C. parapsilosis by toll-like receptors and modulate its pathogenesis through antimicrobial peptides and proinflammatory cytokines. Mediators Inflamm. doi:10.1155/2010/940383 [DOI] [PMC free article] [PubMed]
- 87.Yuan X, Hua X, Wilhelmus KR. The corneal expression of antimicrobial peptides during experimental fungal keratitis. Curr Eye Res. 2010;35:872–879. doi: 10.3109/02713683.2010.495812. [DOI] [PubMed] [Google Scholar]
- 88.Gao N, Kumar A, Guo H, Wu X, Wheater M, Yu F-S (2011) Topical flagellin mediated innate defence against Candida albicans keratitis. Invest Ophthalmol Vis Sci (in press) [DOI] [PMC free article] [PubMed]
- 89.Tu EY, Joslin CE. Recent outbreaks of atypical contact lens-related keratitis: what have we learned? Am J Ophthalmol. 2010;150:602–608. doi: 10.1016/j.ajo.2010.06.045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Schuster FL, Jacobs LS. Effects of magainins on ameba and cyst stages of Acanthamoeba polyphaga . Antimicrob Agents Chemother. 1992;36:1263–1271. doi: 10.1128/aac.36.6.1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Otri AM, Mohammed I, Abedin A, Cao Z, Hopkinson A, Panjwani N, Dua HS. Antimicrobial peptides expression by ocular surface cells in response to Acanthamoeba castellanii: an in vitro study. Br J Ophthalmol. 2010;94:1523–1527. doi: 10.1136/bjo.2009.178236. [DOI] [PubMed] [Google Scholar]
- 92.Maltseva IA, Fleiszig SM, Evans DJ, Kerr S, Sidhu SS, McNamara NA, Basbaum C. Exposure of human corneal epithelial cells to contact lenses in vitro suppresses the upregulation of human beta-defensin-2 in response to antigens of Pseudomonas aeruginosa . Exp Eye Res. 2007;85:142–153. doi: 10.1016/j.exer.2007.04.001. [DOI] [PubMed] [Google Scholar]
- 93.Linzmeier RM, Ganz T. Human defensin gene copy number polymorphisms: comprehensive analysis of independent variation in alpha- and beta-defensin regions at 8p22–p23. Genomics. 2005;86:423–430. doi: 10.1016/j.ygeno.2005.06.003. [DOI] [PubMed] [Google Scholar]
- 94.Hollox EJ. Copy number variation of beta-defensins and relevance to disease. Cytogenet Genome Res. 2008;123:148–155. doi: 10.1159/000184702. [DOI] [PubMed] [Google Scholar]
- 95.Milanese M, Segat L, Arraes LC, Garzino-Demo A, Crovella S. Copy number variation of defensin genes and HIV infection in Brazilian children. J Acquir Immune Defic Syndr. 2009;50:331–333. doi: 10.1097/QAI.0b013e3181945f39. [DOI] [PubMed] [Google Scholar]
- 96.Carter J, Churchill AJ, Gorman C, Haynes R. A case of bilateral endophthalmitis and carriage of beta-defensin 1 44CC genotype. Br J Ophthalmol. 2007;91:1249–1250. doi: 10.1136/bjo.2006.111708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Carter JG, West SK, Painter S, Haynes RJ, Churchill AJ. β-Defensin 1 haplotype associated with postoperative endophthalmitis. Acta Ophthalmol. 2010;88:786–790. doi: 10.1111/j.1755-3768.2009.01534.x. [DOI] [PubMed] [Google Scholar]
- 98.Brewitt H, Sistani F. Dry eye disease: the scale of the problem. Surv Ophthalmol. 2001;45(s2):S199. doi: 10.1016/S0039-6257(00)00202-2. [DOI] [PubMed] [Google Scholar]
- 99.Gilbard JP, Farris RL, Santamaria J., III Osmolarity of tear microvolumes in keratoconjunctivitis sicca. Arch Ophthalmol. 1978;96:677–681. doi: 10.1001/archopht.1978.03910050373015. [DOI] [PubMed] [Google Scholar]
- 100.Farris RL. Tear osmolarity—a new gold standard? Adv Exp Med Biol. 1994;350:495–503. doi: 10.1007/978-1-4615-2417-5_83. [DOI] [PubMed] [Google Scholar]
- 101.Afonso AA, Obrin L, Monroy DC, Selzer M, Lokeshwar B, Pflugfelder SC. Tear fluid gelatinase B activity correlates with IL-alpha concentrations and fluorescein clearance in ocular rosacea. Invest Ophthalmol Vis Sci. 1999;40:2506–2512. [PubMed] [Google Scholar]
- 102.Pflugfelder SC, Jones D, Ji Z, Afonso A, Monroy D. Altered cytokine balance in tear fluid and conjunctiva of patients with Sjogrens syndrome keratoconjunctivitis sicca. Curr Eye Res. 1999;19:201–211. doi: 10.1076/ceyr.19.3.201.5309. [DOI] [PubMed] [Google Scholar]
- 103.Solomon A, Dursun D, Liu Z, Xie Y, Macri A, Pflugfelder SC. Pro- and anti-inflammatory forms of interleukin-1 in the tear fluid and conjunctiva of patients with dry-eye disease. Invest Ophthalmol Vis Sci. 2001;42:2283–2292. [PubMed] [Google Scholar]
- 104.Bron AJ, Tiffany JM, Yokoi N, Gouveia SM. Using osmolarity to diagnose dry eye: a compartmental hypothesis and review of our assumptions. Adv Exp Med Biol. 2002;506:1087–1095. doi: 10.1007/978-1-4615-0717-8_153. [DOI] [PubMed] [Google Scholar]
- 105.Kawasaki S, Kawamoto S, Yokoi N, Connon C, Minesaki Y, Kinoshita S, Okubo K. Up-regulated gene expression in the conjunctival epithelium of patients with Sjogren’s syndrome. Exp Eye Res. 2003;77:17–26. doi: 10.1016/S0014-4835(03)00087-3. [DOI] [PubMed] [Google Scholar]
- 106.Narayanan S, Manning J, Proske R, McDermott AM. Effect of hyperosmolality on beta-defensin gene expression by human corneal epithelial cells. Cornea. 2006;25:1063–1068. doi: 10.1097/01.ico.0000228785.84581.35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Vivino FB, Minerva P, Huang CH, Orlin SE. Corneal melt as the initial presentation of primary Sjogren’s syndrome. J Rheumatol. 2001;28:379–382. [PubMed] [Google Scholar]
- 108.Stern ME, Gao J, Schwalb TA, Ngo M, Tieu D, Chan CC, Reis BL, Whitcup SM, Thompson D, Smith JA. Conjunctival T-cell subpopulations in Sjorgren’s and no-Sjogren’s patients with dry eye. Invest Ophthalmol Vis Sci. 2002;43:2609–2614. [PubMed] [Google Scholar]
- 109.Hida RY, Ohashi Y, Takano Y, Dogru M, Goto E, Fujishima H, Saito I, Saito K, Fukase Y, Tsubota K. Elevated levels of human α-defensin in tears of patients with allergic conjunctival disease complicated by corneal lesions: detection by SELDI protein chip systems and quantification. Curr Eye Res. 2005;30:723–730. doi: 10.1080/02713680591005986. [DOI] [PubMed] [Google Scholar]
- 110.Ikeda A, Nakanishi Y, Sakimoto T, Shoji J, Sawa M, Nemoto N. Expression of β defensins in ocular surface tissue of experimentally developed allergic conjunctivitis mouse model. Jpn J Ophthalmol. 2006;50:106. doi: 10.1007/s10384-005-0262-4. [DOI] [PubMed] [Google Scholar]
- 111.Larrick JW, Morgan JG, Palings I, Hirata M, Yen MH. Complementary DNA sequence of rabbit CAP18—a unique lipopolysaccharide binding protein. Biochem Biophys Res Commun. 1991;179:170–175. doi: 10.1016/0006-291X(91)91350-L. [DOI] [PubMed] [Google Scholar]
- 112.Larrick J, Hirata M, Balint R, Lee J, Zhong J, Wright SC. Human CAP 18: a novel antimicrobial lipopolysaccharide-binding protein. Infect Immun. 1995;63:1291–1297. doi: 10.1128/iai.63.4.1291-1297.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ohgami K, Ilieva IB, Shiratori K, Isogai E, Yoshida K, Kotake S, Nishida T, Mizuki N, Ohno S. Effect of human cationic antimicrobial peptide protein 18 peptide on endotoxin-induced uveitis in rats. Invest Ophthalmol Vis Sci. 2003;44:4412–4418. doi: 10.1167/iovs.03-0246. [DOI] [PubMed] [Google Scholar]
- 114.Jager K, Garreis F, Posa A, Dunse M, Paulsen FP. Functional relationship between cationic amino acid transporters and beta-defensins: implications for dry skin diseases and the dry eye. Ann Anat. 2010;192:65–69. doi: 10.1016/j.aanat.2010.01.006. [DOI] [PubMed] [Google Scholar]
- 115.Jager K, Bonisch U, Risch M, Worlitzsch D, Paulsen F. Detection and regulation of cationic amino acid transporters in healthy and diseased ocular surface. Invest Ophthalmol Vis Sci. 2009;50:1112–1121. doi: 10.1167/iovs.08-2368. [DOI] [PubMed] [Google Scholar]
- 116.Befus AD, Mowat C, Gilchrist M, Hu J, Solomon S, Bateman A. Neutrophil defensins induce histamine secretion from mast cells: mechanisms of action. J Immunol. 1999;163:947–953. [PubMed] [Google Scholar]
- 117.Niyonsaba F, Someya A, Hirata M, Ogawa H, Nagaoka I. Evaluation of the effects of peptide antibiotics human beta-defensins-1/-2 and LL-37 on histamine release and prostaglandin D(2) production from mast cells. Eur J Immunol. 2001;31:1066–1075. doi: 10.1002/1521-4141(200104)31:4<1066::AID-IMMU1066>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- 118.van Wetering S, Mannesse-Lazeroms SPG, van Sterkenburg MA, Daha MR, Dijkman JH, Hiemstra PS. Effect of defensins on interleukin-8 synthesis in airway epithelial cells. Am J Physiol. 1997;272:L888–L896. doi: 10.1152/ajplung.1997.272.5.L888. [DOI] [PubMed] [Google Scholar]
- 119.van Wetering S, Mannesse-Lazeroms SPG, van Sterkenburg MA, Hiemstra PS. Neutrophil defensins stimulate the release of cytokines by airway epithelial cells: modulation by dexamethasone. Inflamm Res. 2002;51:8–15. doi: 10.1007/PL00000282. [DOI] [PubMed] [Google Scholar]
- 120.Tjabringa GS, Aarbiou J, Ninaber DK, Drijfhout JW, Sorensen OE, Borregaard N, Rabe KF, Hiemstra PS. The antimicrobial peptide LL-37 activates innate immunity at the airway epithelial surface by transactivation of the epidermal growth factor receptor. J Immunol. 2003;171:6690–6696. doi: 10.4049/jimmunol.171.12.6690. [DOI] [PubMed] [Google Scholar]
- 121.Niyonsaba F, Ushio H, Nagaoka I, Okumura K, Ogawa H. The human beta-defensins (-1, -2, -3, -4) and cathelicidin LL-37 induce IL-18 secretion through p38 and ERK MAPK activation in primary human keratinocytes. J Immunol. 2005;175:1776–1784. doi: 10.4049/jimmunol.175.3.1776. [DOI] [PubMed] [Google Scholar]
- 122.Garcia JR, Jaumann F, Schulz S, Krause A, Rodriguez-Jimenez J, Forssmann U, Adermann K, Kluver E, Vogelmeier C, Becker D, Hedrich R, Forssmann WG, Bals R. Identification of a novel, multifunctional beta-defensin (human beta-defensin 3) with specific antimicrobial activity. Its interaction with plasma membranes of Xenopus oocytes and the induction of macrophage chemoattraction. Cell Tissue Res. 2001;306:257–264. doi: 10.1007/s004410100433. [DOI] [PubMed] [Google Scholar]
- 123.Yang D, Chertov O, Bykovskaia SN, Chen Q, Buffo MJ, Shogan J, Anderson M, Schroder JM, Wang JM, Howard OM, Oppenheim JJ. Beta-defensins: linking innate and adaptive immunity through dendritic and T cell CCR6. Science. 1999;286:525–528. doi: 10.1126/science.286.5439.525. [DOI] [PubMed] [Google Scholar]
- 124.Yang De, Chen Q, Schmidt AP, Andersin GM, Wang JM, Wooters J, Oppenheim JJ, Chertov O. LL-37, the neutrophil granule- and epithelial cell-derived cathelicidin, utilizes formyl peptide receptor-like 1 (FPRL-1) as a receptor to chemoattract human peripheral blood neutrophils, monocytes and T cells. J Exp Med. 2000;192:1069–1074. doi: 10.1084/jem.192.7.1069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Kurpakus-Wheater M, Kernacki KA, Hazlett LD. Maintaining corneal integrity. How the “window” stays clear. Prog Histochem Cytochem. 2001;36:185–259. [PubMed] [Google Scholar]
- 126.McDermott AM, Redfern RL, Zhang B. Human β-defensin 2 is up-regulated during re-epithelialization of the cornea. Curr Eye Res. 2001;22:64–67. doi: 10.1076/ceyr.22.1.64.6978. [DOI] [PubMed] [Google Scholar]
- 127.Yin J, Yu FS. LL-37 promotes high glucose-attenuated epithelial wound healing via EGFR transactivation in organ cultured corneas. Invest Ophthalmol Vis Sci. 2010;51:1891–1897. doi: 10.1167/iovs.09-3904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Li J, Raghunath M, Tan D, Lareu RR, Chen ZC, Beuerman RW. Defensins HNP1 and HBD2 stimulation of wound-associated responses in human conjunctival fibroblasts. Invest Ophthalmol Vis Sci. 2006;47:3811–3819. doi: 10.1167/iovs.05-1360. [DOI] [PubMed] [Google Scholar]
- 129.Li J, Zhu HY, Beuerman RW. Stimulation of specific cytokines in human conjunctival epithelial cells by defensins HNP1, HBD-2 and HBD-3. Invest Ophthalmol Vis Sci. 2009;50:644–653. doi: 10.1167/iovs.08-1838. [DOI] [PubMed] [Google Scholar]
- 130.Koczulla R, von Degenfeld G, Kupatt C, Krotz F, Zahler S, Gloe T, Issbrucker K, Unterberger P, Zaiou M, Lebherz C, Karl A, Raake P, Pfosser A, Boekstegers P, Welsch U, Heimstra PS, Vogelmeier C, Gallo RL, Clauss M, Bals R. An angiogenic role for the human peptide antibiotic LL-37/hCAP-18. J Clin Invest. 2003;111:1665–1672. doi: 10.1172/JCI17545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Chavakis T, Cines DB, Rhee JS, Liang OD, Schubert U, Hammes HP, Higazi AA, Nawroth PP, Preissner KT, Bdeir K. Regulation of neovascularisation by human neutrophil peptides (alpha-defensins): a link between inflammation and angiogenesis. FASEB J. 2004;18:1306–1308. doi: 10.1096/fj.03-1009fje. [DOI] [PubMed] [Google Scholar]
- 132.Economopoulou M, Bdeir K, Cines DB, Fogt F, Bdeir Y, Lubkowski J, Lu W, Preissner KT, Hammes HP, Chavakis T. Inhibition of pathologic retinal neovascularisation by alpha-defensins. Blood. 2005;106:3831–3838. doi: 10.1182/blood-2005-03-0889. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Hanckock RE, Sahl HG. Antimicrobial and host-defence peptides as new anti-infective therapeutic strategies. Nat Biotechnol. 2006;24:1551–1557. doi: 10.1038/nbt1267. [DOI] [PubMed] [Google Scholar]
- 134.Nos-Barbera S, Portoles M, Morilla A, Ubach J, Andreu D, Paterson CA. Effect of hybrid peptides of cecropin A and melittin in an experimental model of bacterial keratitis. Cornea. 1997;16:101–106. doi: 10.1097/00003226-199701000-00017. [DOI] [PubMed] [Google Scholar]
- 135.Brandt CR, Akkarawongsa R, Altmann S, Jose G, Kolb AW, Waring AJ, Lehrer RI. Evaluation of a theta-defensin in a murine model of herpes simplex virus type 1 keratitis. Invest Ophthalmol Vis Sci. 2007;48:5118–5124. doi: 10.1167/iovs.07-0302. [DOI] [PubMed] [Google Scholar]
- 136.Mannis M. The use of antimicrobial peptides in ophthalmology: an experimental study in corneal preservation and the management of bacterial keratitis. Trans Am Ophthalmol Soc. 2002;100:243–271. [PMC free article] [PubMed] [Google Scholar]
- 137.McDermott AM, Rich D, Cullor J, Mannis MJ, Smith W, Reid T, Murphy CJ. The in vitro activity of selected defensins against an isolate of Pseudomonas in the presence of human tears. Br J Ophthalmol. 2006;90:609–611. doi: 10.1136/bjo.2005.083428. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Huang LC, Jean D, McDermott AM. Effect of preservative free artificial tears on the antimicrobial activity of human beta-defensin-2 and cathelicidin LL-37 in vitro. Eye Contact Lens. 2005;31:34–38. doi: 10.1097/01.ICL.0000146320.64438.8C. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Liu S, Zhou L, Li J, Suresh A, Verma C, Foo YH, Yap EP, Tan DT, Beuerman RW. Linear analogues of human beta-defensin 3: concepts for design of antimicrobial peptides with reduced toxicity to mammalian cells. ChemBioChem. 2008;9:964–973. doi: 10.1002/cbic.200700560. [DOI] [PubMed] [Google Scholar]
- 140.Zhou L, Liu SP, Chen LY, Li J, Ong LB, Guo L, Wohland T, Tang CC, Lakshminarayanan R, Mavinahalli J, Verma C, Beuerman RW. The structural parameters for antimicrobial activity, human epithelial cell cytotoxicity and killing mechanism of synthetic monomer and dimer analogues derived from hBD-3 C-terminal region. Amino Acids. 2011;40:123–133. doi: 10.1007/s00726-010-0565-8. [DOI] [PubMed] [Google Scholar]
- 141.Liu SP, Zhou L, Lakshminarayanan R, Beuerman RW. Multivalent antimicrobial peptides as therapeutics: design principles and structural diversities. Int J Pept Res Ther. 2010;16:199–213. doi: 10.1007/s10989-010-9230-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Qin G, Cai C, McDermott AM. Influence of local concentration on the antimicrobial efficacy of the cathelicidin LL-37. Invest Ophthalmol Vis Sci. 2008;49:4790. [Google Scholar]
- 143.McDermott AM, Santos CM, Kolar SS, Kumar A, Cai C. Antimicrobial activity of a cathelicidin derivative covalently attached to fluorous surfaces. Invest Ophthalmol Vis Sci. 2010;51:3433. [Google Scholar]
- 144.Kumar A, Kolar SS, Zao M, McDermott AM, Cai C. Localization of antimicrobial peptides on polymerized liposomes leading to their enhanced efficacy against Pseudomonas aeruginosa. Mol BioSyst. 2011;7:711–713. doi: 10.1039/c0mb00207k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Schwab IR, Dries D, Cullor J, Smith W, Mannis M, Reid T, Murphy CJ. Corneal storage medium preservation with defensins. Cornea. 1992;11:370–375. doi: 10.1097/00003226-199209000-00002. [DOI] [PubMed] [Google Scholar]
- 146.Reid TW, Steward K, Johnson M, McCartney DL, Mannis MJ, Murphy CJ (1998) Studies on the effect of the bovine neutrophil antibiotic dodeca-peptide (BNP-1) on the viability of human corneal endothelial cells stored in optisol. Invest Ophthalmol Vis Sci 39: ARVO Abstract 358
- 147.McAnulty JF, Reid TW, Waller KR, Murphy CJ. Successful six-day kidney preservation using trophic factor supplemented media and simple cold storage. Am J Transplant. 2002;2:712–718. doi: 10.1034/j.1600-6143.2002.20805.x. [DOI] [PubMed] [Google Scholar]
- 148.Sousa LB, Mannis MJ, Schwab IR, Cullor J, Hosotani H, Smith W, Jaynes J. The use of synthetic cecropin (D5C) in disinfecting contact lens solutions. CLAO J. 1996;22:114–117. [PubMed] [Google Scholar]
- 149.Mathews SM, Spallholz JE, Grimson MJ, Dubielzig RR, Gray T, Reid TW. Prevention of bacterial colonization of contact lenses with covalently attached selenium and effects on the rabbit cornea. Cornea. 2006;25:806–814. doi: 10.1097/01.ico.0000224636.57062.90. [DOI] [PubMed] [Google Scholar]
- 150.Zhu H, Kumar A, Ozkan J, Bandara R, Ding A, Perera I, Steinberg P, Kumar N, Lao W, Griesser SS, Britcher L, Griesser HJ, Willcox MD. Fimbrolide-coated antimicrobial lenses: their in vitro and in vivo effects. Optom Vis Sci. 2008;85:292–300. doi: 10.1097/OPX.0b013e31816bea0f. [DOI] [PubMed] [Google Scholar]
- 151.Willcox MD, Hume EB, Aliwarga Y, Kumar N, Cole N. A novel cationic-peptide coating for the prevention of microbial colonization on contact lenses. J Appl Microbiol. 2008;105:1817–1825. doi: 10.1111/j.1365-2672.2008.03942.x. [DOI] [PubMed] [Google Scholar]
- 152.Cole N, Human EB, Vijay AK, Sankaridurg P, Kumar N, Willcox MD. In vivo performance of melamine as an antimicrobial coating for contact lenses in models of CLARE and CLPU. Invest Ophthalmol Vis Sci. 2010;51:390–395. doi: 10.1167/iovs.09-4068. [DOI] [PubMed] [Google Scholar]