Dear Editor,
Mevalonic aciduria (MA, OMIM #610377), a rare autosomal recessive disease caused by mutations affecting an enzyme of the cholesterol pathway, represents the most severe form of a periodic fever called mevalonate kinase deficiency.1 MA patients show recurrent fever episodes with associated inflammatory symptoms, severe neurologic impairments or death in early childhood.2
Literature data suggest that neurons are able to produce cholesterol3 even if the mechanisms regulating this pathway remain unclear. The evidence that cholesterol biosynthesis deregulation is involved in neurodegenerative diseases gave new strength to the studies focused on the cholesterol pathway in the brain.
Recently, we showed that the SH-SY5Y neuroblastoma cell line was susceptible to lovastatin (Lova). Lova blocks the cholesterol pathway by inhibiting 3-hydroxy-3-methylglutaryl-CoA reductase and thus causing apoptosis. We demonstrated that the mitochondrial pathway promotes apoptosis in a neuronal cell model, with caspase-9 as initiator and caspase-3 as effector.4, 5
We then verified the role of caspase-3 and caspase-9 in apoptosis using cell-permeable caspase-specific inhibitors. We evaluated the programmed cell death (PCD) and the mitochondrial dysfunction, characterized by the change of transmembrane potential, which is directly proportional to mean fluorescence intensity (MFI).6
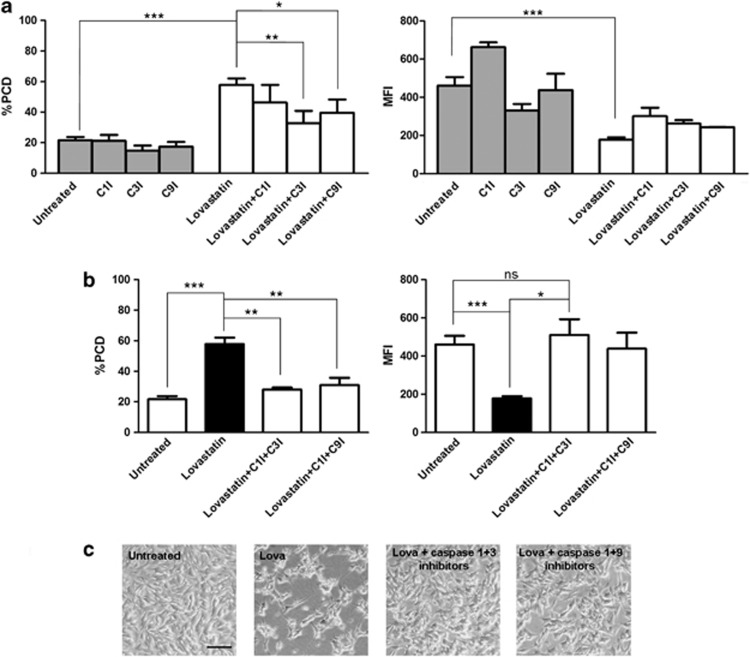
We observed that SH-SY5Y pre-treated with caspase-3 inhibitor (C3I) and subsequently treated with Lova showed a considerable decrease of PCD and an increase of MFI when compared to cells treated only with Lova. The same results, even though less remarkable, were obtained by pre-treating with caspase-9 inhibitor (C9I) (Figure 1a).
Figure 1.
Effects of caspase inhibitors and Lovastatin (Lova) treatments on SH-SY5Y undifferentiated cell line. (a) Forty-eight hours after seeding, cells were treated for 1 h at 37 °C with inhibitors of caspase-3 (Z-DEVD-FMK) (50 μM), caspase-9 (Z-LEHD-FMK) (50 μM), caspase-1 (Z-WEHD-FMK) (50 μM) (R&D Systems, Minnepolis, MN, USA) and then stimulated for 24 h with Lova (10 μM) (compounds were assayed alone or in combination). (b) Lova-treated cells with caspase 1+3 inhibitors or 1+9 inhibitors were compared to untreated or lovastatin conditions. (c) Images were acquired before the PCD assay. The horizontal bar indicates 0.1 mm (Leica DM IL inverted microscope, Leica Microsystem, Milano, Italy). PCD was analyzed with flow cytometry (Annexin V-FITC Apoptosis Detection Kit, Immunostep, Spain): bars represent the means of apoptosis percentages±standard error of the mean (S.E.M.) of three independent experiments. MFI was analyzed with flow cytometry (Rhodamine 123, Sigma-Aldrich, St Louis, MO, USA): bars represent the mean fluorescent intensity of Rhodamine 123±S.E.M. of 3 independent experiments. Fluorescence was acquired with CyAn ADP analyzer and Summit software (Beckman Coulter, Brea, CA, USA), then analyzed with FlowJo software (version 7.6, Treestar, Inc., St Ashland, OR, USA). Statistical significance was calculated using analysis of variance (ANOVA, one- or two-way where appropriate) and Bonferroni post-test correction in the case of multiple comparisons. Analysis was performed using GraphPad Prism software (version 5.0, GraphPad Software, Inc., La Jolla, CA, USA). *P<0.05; **P<0.01, ***P<0.001, ns: nonsignificant.
These findings confirm that apoptosis induced by Lova follows the intrinsic (mitochondrial) pathway in neuronal cell lines. Since C3I and C9I are not able to completely restore the physiological condition, we hypothesized the intervention of caspase-1, inasmuch as it is active at systemic level in MA monocytes.7 To understand its potential involvement, we pre-treated cells with the caspase-1 inhibitor (C1I): pre-treated determined significant changes neither in PCD nor in MFI values (Figure 1a).
Inhibition of the mevalonate pathway induces cell death, which is in part sustained by the activation of caspase-3 and in part by caspase-1. C1I and C3I were able together to nullify the Lova-induced effect on MFI, which indeed showed levels similar to those of untreated cells. Even though less evident, the inhibition of Lova-induced effect on MFI was observed also after the administration of C1I plus C9I (Figure 1b). These findings suggest that caspase-9 may act as an accelerator for caspase-1 in triggering apoptosis.
It is crucial to highlight that C3I and C9I used alone or in combination are not able to rescue cells from PCD, whereas C1I does, when given in combination with the other inhibitors (data not shown).
We therefore hypothesized that cell death induced by inhibition of the mevalonate pathway could be related to pyroptosis besides apoptosis, considering the role played by caspase-1 and caspase-3 (Figure 1b). The anti-apoptotic effect of caspase inhibitors in SH-SY5Y Lova-treated cells was also evident by microscope pictures (Figure 1c). The cell density, after Lova-caspase1/3 inhibitor and Lova-caspase1/9 inhibitor treatments, was comparable to that of untreated cells, while the difference in density and morphology was significant in Lova-treated cells.
These results allow to hypothesize that apoptosis in SH-SY5Y Lova-treated cells follows the mitochondrial pathway; that is, it is caspase-9 and caspase-3 dependent; however, we also demonstrated that caspase-1 plays a role in this still unclear mechanism.
As statins induce the translocation of cytosolic Bax to the mitochondria and allow to permeabilize the mitochondrial outer membrane during apoptosis,8 further studies will be carried out on the expression of proapoptotic genes, such as Bax or Bcl2.
These findings, even if obtained in an SH-SY5Y undifferentiated cell line, surely represent a first step towards the understanding of the neuronal damage caused by inflammation processes in MA.
Acknowledgments
This study was supported by a grant from the Institute for Maternal and Child Health—IRCCS ‘Burlo Garofolo'—Trieste, Italy (RC 42/2011).
The authors declare no conflict of interest.
References
- Haas D, Hoffmann GF. N Engl J Med. 2007. pp. 2671–2673. [DOI] [PubMed]
- Haas D, Hoffmann GF. Orphanet J Rare Dis. 2006. pp. 1–13. [DOI] [PMC free article] [PubMed]
- Pfrieger FW, Ungerer N. Prog Lipid Res. 2011. pp. 357–371. [DOI] [PubMed]
- Marcuzzi A, et al. Int J Dev Neurosci. 2012. pp. 451–456. [DOI] [PubMed]
- Arnold DE, et al. Mol Cell Biochem. 2010. pp. 1–11. [DOI] [PubMed]
- Morabito R, et al. Toxicol In Vitro. 2012. pp. 694–699. [DOI] [PubMed]
- Marcuzzi A, et al. Apoptosis. 2011. pp. 882–888. [DOI] [PubMed]
- Herrero-Martin G, Lòpez-Rivas A. FEBS Lett. 2008. pp. 2589–2594. [DOI] [PubMed]