Abstract
Diminished constriction of arteries and veins following exposure to microgravity or bed rest is associated with a reduced ability to augment peripheral vascular resistance (PVR) and stroke volume during orthostasis. We tested the hypothesis that small mesenteric arteries and veins, which are not exposed to large pressure shifts during simulated microgravity via head-down tail suspension (HDT), will exhibit decrements in adrenergic constriction after HDT in rats. Small mesenteric arteries and veins from control (Con; n= 41) and HDT (n= 35) male Sprague-Dawley rats were studied in vitro. Vasoactive responsiveness to norepinephrine (NE) in arteries (10−9 to 10−4 M) and veins (pressure-diameter responses from 2–12 cm H2O after incubation in 10−6 or 10−4 M NE) were evaluated. Plasma concentrations of atrial (ANP) and brain (NT-proBNP) natriuretic peptides were also measured. In mesenteric arteries, sensitivity and maximal responsiveness to NE were reduced with HDT. In mesenteric veins there was a diminished venoconstriction to NE at any given pressure in HDT. Plasma concentrations of both ANP and NT-proBNP were increased with HDT, and maximal arterial and venous constrictor responses to NE after incubation with 10−7 M ANP or NT-proBNP were diminished. These data demonstrate that, in a vascular bed not subjected to large hydrodynamic differences with HDT, both small arteries and veins have a reduced responsiveness to adrenergic stimulation. Elevated levels of circulating ANP or NT-proBNP could adversely affect the ability of these vascular beds to constrict in vivo, and conceivably could alter the intrinsic constrictor properties of these vessels with long-term exposure.
Keywords: arteries, veins, hindlimb unloading
INTRODUCTION
Human cardiovascular deconditioning resulting from bed rest or exposure to microgravity induces several adaptations affecting both central (e.g., attenuated baroreflex sensitivity (8) and stroke volume (33)) and peripheral (e.g., altered skeletal muscle vascular reactivity (53)) homeostasis. These perturbations manifest in a diminished capacity to perform and sustain exercise (50), as well as orthostatic intolerance in some individuals (4, 38, 40). Key mechanisms contributing to an altered regulation of MAP during orthostasis include an inability to augment peripheral vascular resistance (PVR) and stroke volume (4, 40, 57), resulting in reduced cerebral perfusion (2) and, if not corrected, syncope.
Animal models have been used to simulate the effects of microgravity on the cardiovascular system of humans. Head-down tilt (HDT) in rats limits postural and locomotor activity, and HDT elicits many of the hemodynamic alterations that characterize the cardiovascular deconditioning that occurs in humans after prolonged bed rest or exposure to microgravity, including hypovolemia (35), resting and exercising tachycardia (16, 37) altered tissue perfusion (37, 60), and arterial hypotension during head-up tilt (36, 58). The diminished capacity to elevate PVR with simulated microgravity is due, in part, to reductions in vasoconstrictor responsiveness that have been shown to occur in a number of arterial segments, including abdominal and thoracic aorta (11, 13, 46, 47), carotid and femoral arteries (47, 63), and skeletal muscle arterioles (10) from HDT rats. In addition, vasoconstrictor responses of mesenteric arteries to elevations in transmural pressure (34) and norepinephrine (43) have been reported to be lower in HDT rats. The diminished cardiac output with HDT-induced cardiovascular deconditioning is principally due to a profound reduction in stroke volume (33, 60). Stoke volume is primarily determined by the contractility of the heart and venous filling pressure (48), the latter of which is dependent upon central mobilization of venous volume via constriction of large capacitance peripheral veins. Therefore, diminished adrenergic venoconstriction would result in an increased functional capacitance of veins, thereby attenuating venous filling pressure and reducing stroke volume via the Starling mechanism.
Using the HDT rat as a model of the cardiovascular deconditioning induced by microgravity, previous investigators have suggested that the hypo-responsiveness of the arterial vasculature is primarily the result of changes in the pressure or flow within blood vessels (62). Therefore, this study was undertaken to investigate the effects of simulated microgravity upon vaso- and venoconstriction within a vascular bed not exposed to pressure or flow alterations with HDT, i.e., the mesenteric circulation (37). Specifically, we tested the hypothesis that vasoactive responses to adrenergic stimulation will be diminished in vessels from the arterial and venous mesenteric circulation. Furthermore, we hypothesized that circulating levels of atrial (ANP) and brain (NT-proBNP) natriuretic peptides, which are sensitive to postural fluid shifts and have the potential to alter the intrinsic vasomotor properties of peripheral arteries and veins, will be elevated in HDT rats. Based upon findings from the latter set of experiments, we hypothesized that exposure to either ANP or NT-proBNP reduces the contractile responses evoked by adrenergic stimulation in arteries and veins in the mesenteric circulation.
METHODS
Materials and Methods
All procedures performed in this study were approved by the Texas A&M University and West Virginia University Animal Care Committees and conformed to the National Institutes of Health (NIH) Guide for the Care and Use of Laboratory Animals.
Animals
Six month old male Sprague-Dawley rats were housed in a temperature-controlled (23 ± 2 °C) room with a 12:12-h light-dark cycle. Water and rat chow were provided ad libitum. The animals were randomly assigned to either cage control (Con, n = 41) or 14-days head-down tail suspension (HDT; n = 35) groups. The hindlimbs of the HDT group were elevated to an approximate spinal angle of 40–45° via orthopedic tape adhered to the tail, which is a modification of techniques previously described (12, 59, 61). Briefly, the animals were injected with sodium pentobarbital (30 mg/kg i.p.) to induce anesthesia. While anesthetized, the animal’s tail was washed and dried, and a length of breathable nonelastic adhesive tape (Curity Porous tape, Kendall) with a hook attached to the end was placed on the proximal two-thirds of the tail, which allowed the end of the tail to remain unattached. The ends of the adhesive tape were further bonded to the tail with an additional adhesive (Goop) and allowed to dry for 20 min before suspension. Daily inspection of each animal’s tail was performed, checking for discoloration of tissue damage from the suspension apparatus. The hook attached to the adhesive tape was connected by a small chain to a swivel apparatus fixed at the top of the cage. The height of the hindlimb elevation was adjusted to prevent the hindlimbs from touching supportive surfaces while the forelimbs maintained contact with the cage floor. This allowed free range of movement around the cage while achieving the desired experimental results. Control animals were individually housed and maintained in a normal cage environment while HDT rats were kept in the head-down position for 14 days. This time period has been shown to be sufficient to induce cephalic fluid shifts (25, 36) and produce cardiovascular alterations in HDT animals (10, 14, 35, 37, 43, 44, 58, 60). After the experimental period, HDT and control animals were weighed and euthanized by injecting 40 mg/kg i.p. Euthanasia-5 Solution (Henry-Schein, Inc. Port Washington, NY) without allowing the hindlimbs of the HDT rats to become weight bearing. The gastrointestinal tract was then carefully dissected free and placed in a 4 °C filtered physiological saline solution (PSS) (pH 7.4).
Microvessel Preparation
Under a dissecting microscope, resistance arteries (~200 µm) and veins (~300 µm) from the mesentery were identified, dissected free from surrounding tissue, and transferred to a Lucite vessel chamber containing PSS-albumin solution (pH 7.4) equilibrated with room air and ambient temperature. One end of the microvessel was cannulated with a glass micropipette (~50 µm in tip diameter) and the outside of the vessel was securely tied to the pipette with 11-O opthalmic suture. Any residual blood within the vessel was then flushed out with PSS solution under low positive pressure (~ 5 cm H2O). The other end of each vessel was then cannulated with a second pipette and secured with suture. After cannulation, the isolated vessel in the tissue chamber was transferred to the stage of an inverted microscope (Olympus IX71) coupled to a video camera (Panasonic BP310) and videomicrometer (307A, Colorado Video, Boulder, CO). Mesenteric arteries were then pressurized at 108 cm H2O, which corresponds to in vivo arterial pressures in these vessels (18). Mesenteric veins were pressurized at 4 cm H2O, which is similar to that used by Dunbar et al. (15). The vessels were allowed to equilibrate for 1 hour at 37 °C (PSS solution changed every 15 min) before vasomotor properties were characterized. Internal diameters were measured continuously throughout the experiment using the video caliper.
Experimental Design
Protocol I: Vasoconstrictor properties of mesenteric resistance arteries were characterized by determining maximal responsiveness and sensitivity to NE. Following cannulation and equilibration in PSS, concentration-response relations were determined by the cumulative addition of NE (10−9 – 10−4 M).
Protocol II: Venoconstrictor properties of mesenteric veins were characterized by determining pressure-diameter relations before and after exposure to NE. Specifically, following cannulation and equilibration in PSS, intraluminal pressure was lowered to 2 cm H2O and subsequently increased at 2 cm H2O increments to 12 cm H2O. A starting pressure of 2 cm H2O was used because at 0 cm H2O the veins tended to collapse. Vessels were allowed 5 min to equilibrate at a given pressure prior to measuring luminal diameter. After the active pressure diameter response was quantified, intraluminal pressure was set at 4 cm H2O, and the vessel was incubated in 10−6 or 10−4 M NE (random order) for 20 min and the pressure-diameter response was repeated as described above. Upon completion of all active responses, the bathing solution was replaced with Ca2+-free PSS for 60 minutes upon which the passive pressure-diameter response (same protocol as the active response) was quantified.
Plasma Atrial and Brain Natriuretic Peptide Concentration
Both ANP and NT-proBNP were measured using radioimmunoassay from plasma samples (ALPCO Diagnostics, Salem, NH). Blood was obtained from control (n=28) and 14-day HDT (n=26) rats in the morning between 7 and 9 AM via puncture of the abdominal aorta and collected in EDTA tubes containing aprotinine (200 KIU/ml). The mean intra-assay variance for ANP was 5.2%, with an interassay variance of 6.6%. The sensitivity of the assay was 3.5 pg/ml. The mean intra-assay variance for NT-proBNP was 4.0%, with an interassay variance of 3.8%. The sensitivity of the assay was 4 fmol/ml.
Effects of Atrial (ANP) and Brain (NT-proBNP) Natriuretic Peptide on Vascular Responsiveness
In a separate group of six month old male Sprague-Dawley rats (n=10), mesenteric arteries and veins were dissected, cannulated, and pressurized as described above. Vasoconstrictor properties of mesenteric resistance arteries and veins were characterized by determining maximal responsiveness and sensitivity to NE before and after incubation in either ANP or NT-proBNP. Following cannulation and equilibration in PSS, concentration-response relations were determined by the cumulative addition of NE (10−9 – 10−4 M). After completion of the control response, the PSS bathing solution was replaced every 15 min for 1 hour to allow the vessel to re-establish spontaneous tone. Vessels were then incubated either ANP (10−7M) or NT-proBNP (10−7M) (random order) for 20 min and the maximal responsiveness and sensitivity to NE was repeated as described above. Preliminary studies were conducted to assure that vascular responses to NE were not altered by multiple dose response tests.
Solutions and Drugs
The PSS buffer contained (in mM) 145 NaCl, 4.7 KCl, 1.2 NaH2PO4, 1.17 MgSO4, 2.0 CaCl2, 5.0 glucose, 2.0 pyruvate, 0.02 EDTA, and 3.0 MOPS at a pH of 7.4. Ca2+-free PSS buffer was similar to the PSS buffer except it contained 2 mM EDTA and the CaCl2 was replaced with 2.0 mM NaCl. Concentrated stock solutions of NE were prepared in PSS buffer.
Statistical Analysis
Intraluminal diameters were measured throughout the duration of each experimental protocol. At the end of each experiment the vessel was placed in Ca2+-free bathing solution and rinsed several times over the course of an hour to obtain a maximal diameter and wall thickness. The development of spontaneous tone is expressed as the percent constriction relative to maximal diameter and calculated as (Dmax − DB)/ Dmax × 100, where Dmax is the maximal diameter and DB is the starting baseline diameter. Within veins, pressure-diameter response curves are expressed as intraluminal diameter (µm) as well as the percent change versus the diameter at a given pressure during the active or passive protocols. Pressure-diameter and concentration-response curves were evaluated using repeated-measures analysis of variance with one within (intraluminal pressure or concentration) and one between (experimental groups) factor. Planned contrasts were conducted at each intraluminal pressure or concentration level to determine whether differences exist between experimental groups (control vs. HDT). To detect differences in sensitivity to NE, EC50 values were designated as the concentration of NE which produces 50% of its maximal response. Student’s unpaired t-test was used to determine whether differences in body mass, soleus muscle mass, soleus muscle-to-body weight ratio, developed spontaneous tone, maximal diameter, compliance and EC50 values were significantly different between groups. All values are presented as the mean ± standard error. A value of P < 0.05 was required for significance.
RESULTS
Soleus Muscle-to-Body Weight Ratio
The HDT rats weighed significantly less than their control counterparts (HDT, 381 ± 17 g vs. Con 423 ± 11 g; P<0.05). There was a significant atrophy of the soleus muscle in the HDT animals compared to controls (HDT 149 ± 12 mg, Con 245 ± 14 mg; P<0.05), which resulted in a ~35 % reduction in the soleus-to-body weight ratio (HDT 0.39 ± 0.01 mg/g, Con 0.58 ± 0.03 mg/g; P<0.05). The latter is a hallmark of reduced skeletal muscle weight-bearing activity and confirms the efficacy of the HDT procedure (56).
Vessel Characteristics
The maximal intraluminal diameter (determined in Ca2+-free solution) of the mesenteric arteries (at 108 cm H2O) and veins (at 4 cm H2O) were not different between Con and HDT (Table 1). In arteries, both groups displayed a similar amount of spontaneous tone (~20%; Table 1). However, at 4 cm H2O the mesenteric veins from Con exhibited a greater level of spontaneous tone (Con 16 ± 6 % vs. HDT 2 ± 1 %, P<0.05). HDT did not affect the wall thickness or wall-to-lumen ratio in either mesenteric arteries or veins (Table 1).
Table 1.
Mesenteric artery and vein morphology and vasoactive properties in Control animals and rats exposed to Head-Down Tilt (HDT).
| Artery | Vein | |||
|---|---|---|---|---|
| Con n=11 |
HDT n=9 |
Con n=7 |
HDT n=6 |
|
| Max. Diameter (µm) | 243 ± 14 | 251 ± 19 | 244 ± 32 | 236 ± 11 |
| Wall Thickness (µm) | 44 ± 5 | 39 ± 6 | 21 ± 2 | 20 ± 5 |
| Wall-to-Lumen | 0.18 ± 0.1 | 0.16 ± 0.2 | 0.09 ± 0.01 | 0.09 ± 0.01 |
| Initial Tone (%) | 24 ± 4 | 21 ± 7 | 16 ± 6 | 2 ± 1* |
Max. Diameter, intraluminal diameter measured in Ca2+-free solution at an intraluminal pressure of 108 cm H2O for arteries or 4 cm H2O for veins; Wall-to-Lumen, the wall intimal thickness divided by the maximal luminal diameter; Initial tone, the percent difference between the luminal diameter (in solution containing Ca2+) and the baseline diameter. Values are means + SE.
P<0.05 vs. Con for the same vessel type.
Mesenteric Artery Vasoconstrictor Responses
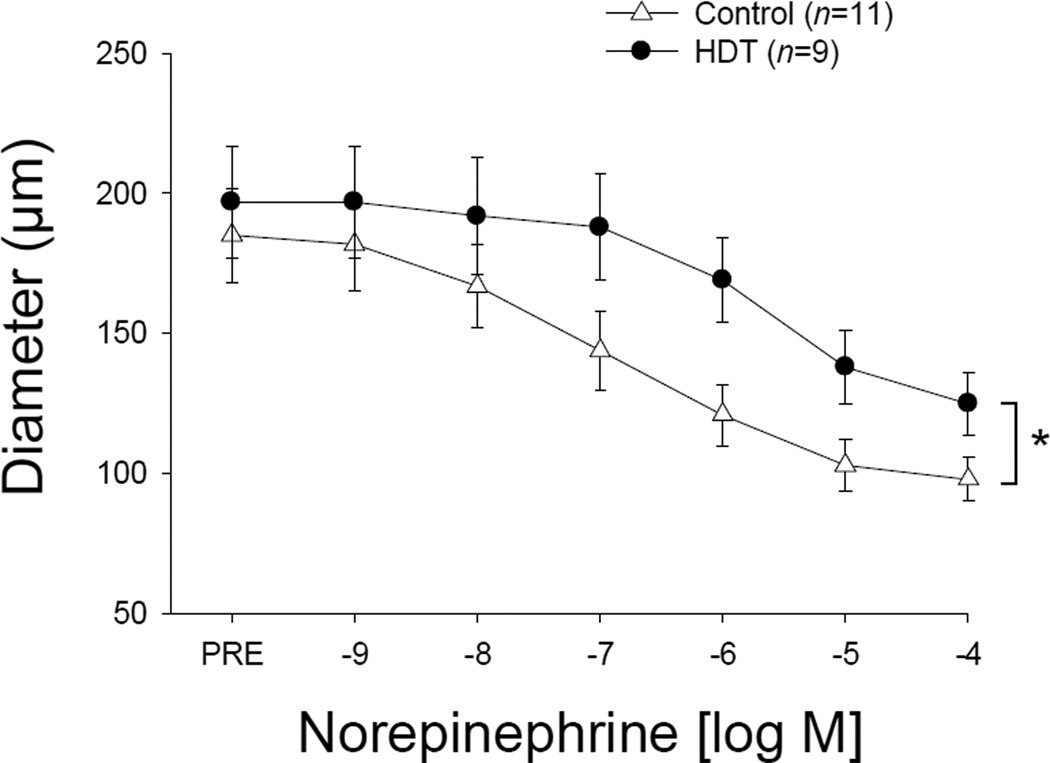
Norepinephrine (NE) produced dose-dependent decreases in luminal diameter in mesenteric arteries from both Con and HDT (Figure 1). However, the contractile responses evoked by 10−9 to 10−4 M NE were lower in HDT. The maximal constriction induced by NE was significantly attenuated in arteries from HDT versus Con (Table 1, Figure 1). In addition, the sensitivity of mesenteric arteries from HDT to NE was lower than that from Con animals, as indicated by a higher EC50.
Figure 1.
Concentration-response relation of norepinephrine in mesenteric resistance arteries from Control and Head-down tilt (HDT) animals. Values are means ± SE. *HDT significantly different from that of Control (P<0.05).
Mesenteric Vein Constrictor Responses
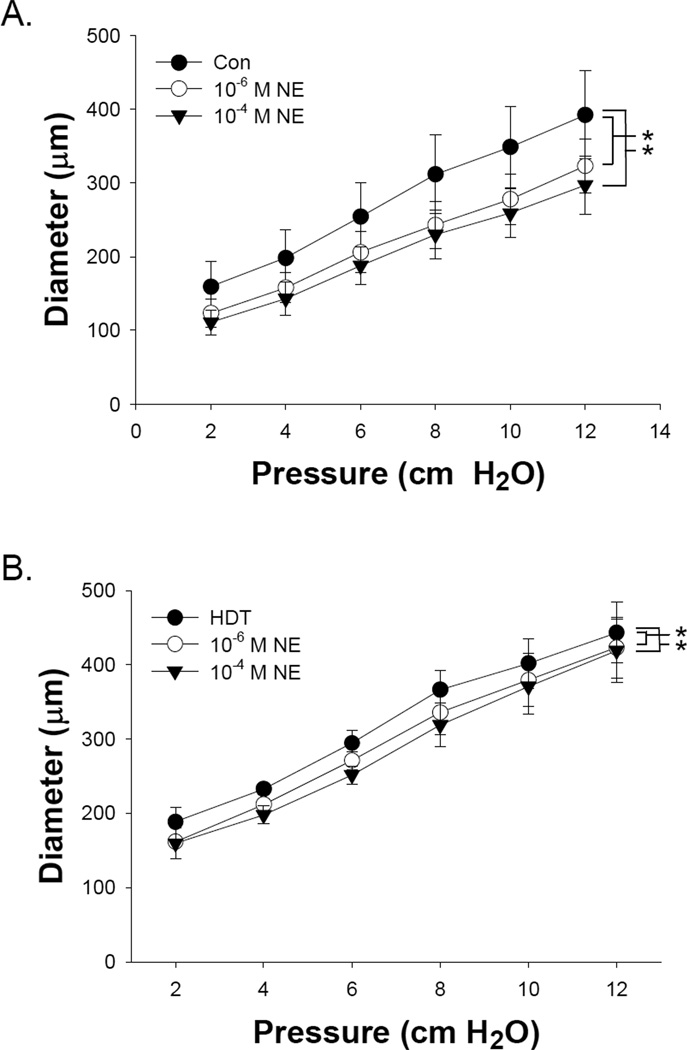
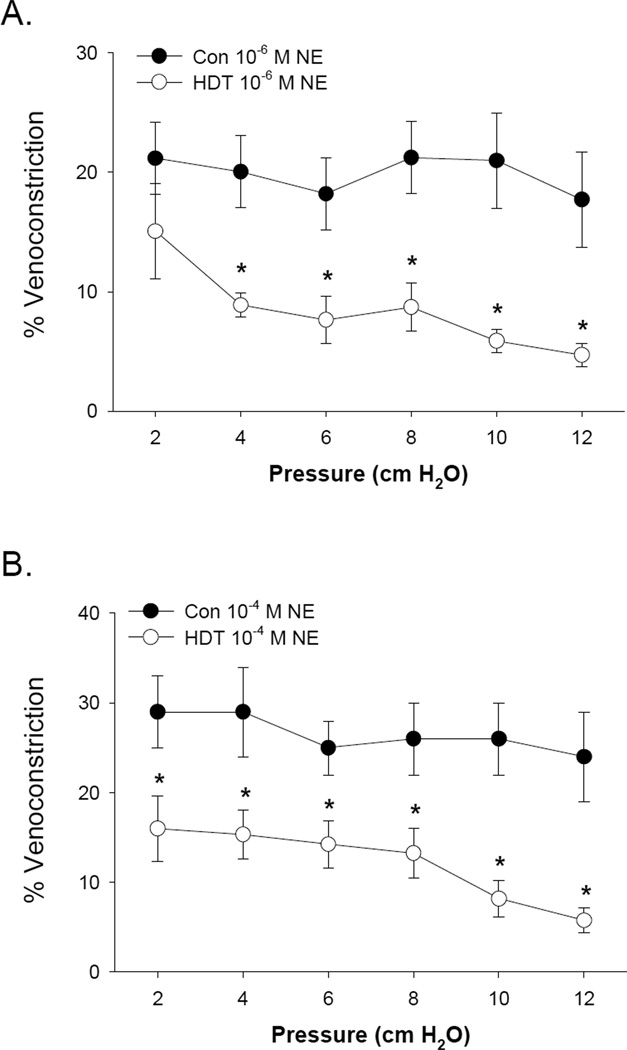
Lumen diameter increased near-linearly (R2, Con 0.986 ± 0.006, HDT 0.969 ± 0.007) as a function of intraluminal pressure, and there was a downward shift in the pressure-diameter response curve with exposure to NE (Figures 2A and 2B). However, at any given pressure the percent venoconstriction with both 10−6 and 10−4 M NE was significantly reduced in HDT versus Con vessels (Figure 3A and 3B).
Figure 2.
Pressure-diameter responses after exposure to norepinephrine in small mesenteric veins from Control and HDT animals. A) Diameter was smaller in mesenteric veins from Control animals at all pressures with norepinephrine, and exposure to 10−4 M norepinephrine resulted in smaller diameters versus those at 10−6 M norepinephrine (P<0.05). B) Mesenteric veins from HDT animals showed smaller diameters at all pressures with exposure to norepinephrine (P<0.05). Only at 4, 6, and 8 cm H20 was a difference observed between the diameters measured at 10−6 versus 10−4 M norepinephrine (P<0.05). Values are means ± SE.
Figure 3.
The percent constriction of mesenteric veins from Control and HDT rats to A) 10−6 M norepinephrine and B) 10−4 M norepinephrine. Values are means ± SE. *HDT significantly different from that of Control (P<0.05).
Plasma ANP and NT-proBNP Concentrations
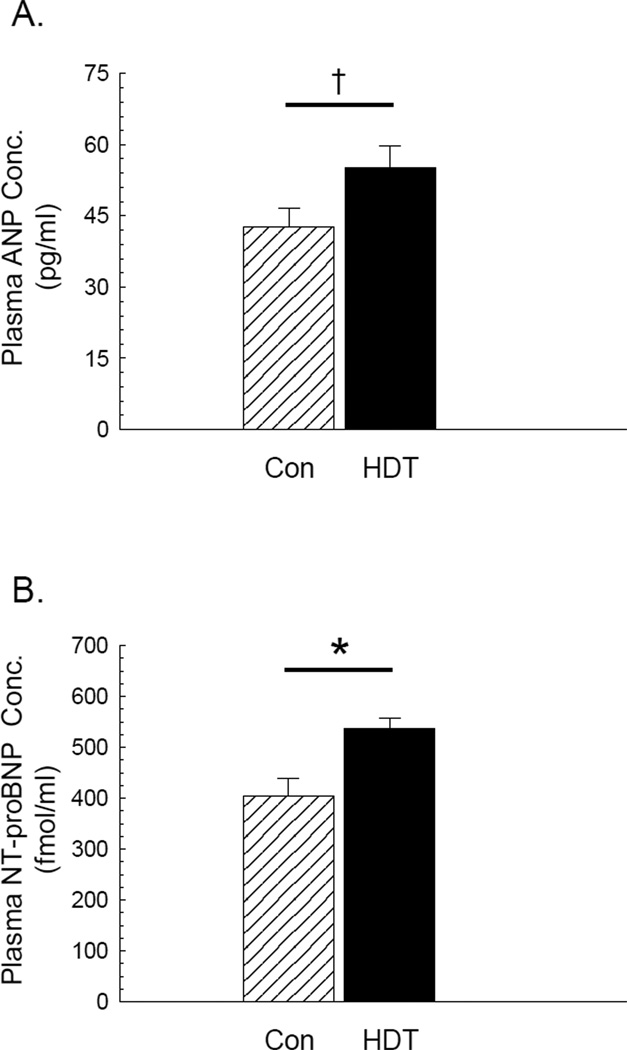
Plasma ANP concentration was greater in HDT rats relative to control animals (Figure 4A). Likewise, plasma NT-proBNP concentration was elevated in HDT rats (Figure 4B).
Figure 4.
A) Plasma atrial natriuretic peptide (ANP) and B) brain-type natriuretic peptide (NT-proBNP) concentrations in Control (n=28) and HDT (n=26) rats. Values are means ± SE. †P=0.052 between groups. *P<0.05 between groups.
ANP and NT-proBNP Mesenteric Vaso- and Venoconstrictor Responses
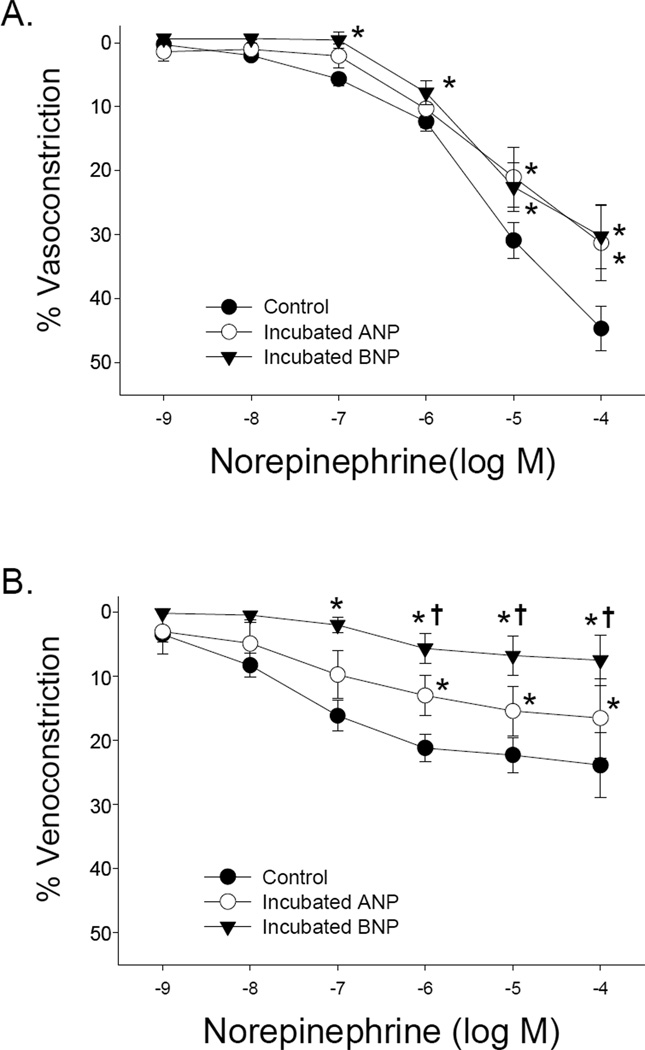
Incubation in either ANP or NT-proBNP reduced the maximal NE-induced constriction in mesenteric arteries (Figure 5A) and veins (Figure 5B). In addition, the attenuation of NE-mediated venoconstriction was greatest in the presence of NT-proBNP (Figure 5B). Contrary to the diminished maximal responsiveness, there were no differences observed in sensitivity to NE (EC50) with ANP or NT-proBNP in either mesenteric arteries or veins. Incubation in ANP or NT-proBNP had no effect on spontaneous tone developed in either arteries or veins as well (data not shown).
Figure 5.
A) Dose-response relations of mesenteric arteries to norepinephrine pre- and post-incubation in either ANP (10−7 M) or BNP (10−7 M). B) Dose-response relations of mesenteric veins to norepinephrine pre- and post-incubation in either ANP (10−7 M) or BNP (10−7 M). Values are means ± SE. *P<0.05 vs. control constrictor response at a given concentration of norepinephrine. †P<0.05 vs. constrictor response after incubation in ANP at a given concentration of norepinephrine.
DISCUSSION
This is the first study to demonstrate that cardiovascular deconditioning induced by simulated microgravity causes intrinsic deficits in vasoconstrictor function that are manifest in both arteries and veins of a tissue where arterial pressure and blood flow are not altered by the HDT intervention (37, 59). Pathognomonic of the cardiovascular deconditioning induced by simulated (19, 35) and actual (4, 38, 40) microgravity is an attenuated ability to elevate vascular resistance, including that in the splanchnic circulation (37, 44, 60). As mean arterial pressure (MAP) is the product of peripheral vascular resistance (PVR) and cardiac output, alterations in either variable without a compensatory change in the other would adversely affect the maintenance of MAP. Thus, the diminished vasoconstriction observed in mesenteric arteries could reduce the ability of the cardiovascular system to increase PVR during orthostatic stress and redirect blood flow from the viscera to active skeletal muscles during exercise. The diminished venoconstriction would compromise peripheral mechanisms to centrally mobilize blood volume to positively affect venous return, atrial filling pressure and consequently, stroke volume. Given the large capacitance of the splanchnic vascular bed (49), the results presented herein provide a likely mechanism for the deconditioning induced inability to mobilize blood volume from this region to support cardiac output and elevate splanchnic vascular resistance in the maintenance of MAP, both of which are critical cardiovascular adjustments to orthostatic and exercise stress.
Results from the present study, as well as those previously published demonstrating compromised vasoconstrictor function of mesenteric lymphatic vessels to HDT (21), gives important insight into the mechanism of smooth muscle contractile dysfunction associated with microgravity in large segments of the arterial, venous and lymphatic circulations, and potentially microgravity-induced smooth muscle contractile dysfunction associated with the uterus (5). For example, it was first proposed by Hargens et al. (24, 26) that headward fluid shifts associated with the removal of gravitational stress or head-down bed rest could alter vascular morphology and consequently vascular function. Indeed, increases in arterial pressure to the head of HDT rats stimulates increases in cerebral artery wall thickness (58, 59) and enhancement of cerebral artery vasoconstrictor function (22, 58, 63). Conversely, decreases in arterial pressure to the hindlimbs of HDT rats induces a thinning of arteriolar wall thickness (12) and a reduction in arteriolar vasoconstrictor function (10). Thus, microgravity or simulated microgravity-induced alterations in the mechanical environment of vessels (i.e., vascular transmural pressure and shear stress) can serve as a potent stimulus to alter vascular structure and function. However, arterial, venous and lymphatic smooth muscle contractile hypo-responsiveness (present study, 21) is unlikely to be the result of altered hydrostatic pressure or intravascular shear stress, since the mesenteric circulation is located at or near the hydrostatic indifference point (46, 59) and exhibits no alterations in blood flow during simulated microgravity (37); the absence of changes in arterial transmural pressure and shear stress are further demonstrated by the lack of change in mesenteric arterial structure with chronic HDT (present study, 34, 59). These results demonstrate that another factor, likely a systemic factor(s), is working to diminish smooth muscle contractile function in the arterial (Figure 1; (10, 11, 47)), venous (Figure 3; (15)) and lymphatic (21) circulations, and possibly smooth muscle contractile function throughout the body (e.g., uterine tissues (5)), with one notable exception. The cerebral circulation, where smooth muscle cells are shielded from circulating agents via the blood-brain barrier is the only vascular bed to consistently show that smooth muscle vasoconstrictor function is not diminished in HDT rats (22, 58, 63). Thus, we propose that in addition to microgravity-induced alterations in vascular transmural pressure and shear stress, some circulating substance whose release is also likely associated with headward fluid shifts may stimulate alterations in intrinsic vascular function independent of the abovementioned mechanical influences.
Smooth Muscle Hypo-Responsiveness: Role of Natriuretic Peptides
Although alterations in a number of circulating factors accompany headward fluid shifts (7), ANP and NT-proBNP are two endogenous hormones released in response to fluid volume shifts which regulate fluid volume via Na+ excretion (3, 9) and have vasoactive properties (45). Within smooth muscle, ANP and NT-proBNP selectively bind to the natriuretic peptide receptor Npra (20) and activate guanylyl cyclase to increase intracellular levels of the second messenger cGMP (45). cGMP and its associated cGMP-dependent protein kinase are important modulators of vascular tone (28). Specifically, cGMP-dependent protein kinase decreases cytosolic Ca2+ by activating Ca2+ATPase, thereby lowering cytosolic Ca2+ in the cell. cGMP also acts as a negative modulator of protein kinase C (PKC) activity, such that long-term exposure of smooth muscle cells to ANP inhibits PKC activity in a dose-dependent manner (31).
ANP and NT-proBNP are also both potent inhibitors of ANG-II stimulated ET-1 production (23), the latter of which potentiates the vasoconstrictive action of NE (55). The adrenoreceptor agonist NE binds to a G-protein receptor and activates phospholipase C resulting in the hydrolysis of phosphoinositide and the formation of inositol 1,4,5-trisphosphate (IP3) and diacylglycerol (DAG). Subsequently, DAG activates PKC (42) and IP3 induces the release of Ca2+ from intracellular stores (1), resulting in smooth muscle contraction. Therefore, we propose that the elevated circulating levels of ANP and NT-proBNP associated with simulated microgravity in rats could serve to attenuate smooth muscle cell vasoconstrictor responsiveness to NE by inhibiting both PKC activity (51) and the formation of IP3 (29). Furthermore, smooth muscle cells throughout the body, with the exception of those protected by the blood-brain barrier, may be susceptible to contractile dysfunction via this systemic mechanism.
To our knowledge, the present observation that NT-proBNP is elevated with HDT is the first reported in the literature. However, the effects of HDT on ANP have previously been examined. Our observation that increases in circulating plasma levels of ANP occur during HDT is consistent with the ANP stimulated elevation in cGMP concentration in the rat choroids plexus with HDT and spaceflight (6). However, others have also reported that plasma ANP concentrations are unchanged (41) or decreased (17) with HDT. The reasons for this discrepancy is unclear, but may involve the length of HDT (14 days in the current study versus 9 days (17)) or the hydration state of the animals.
Based upon the findings of elevations in ANP and NT-proBNP with HDT (Figure 4), we tested the hypothesis that acute exposure to either of these peptides would reduce the contractile responses evoked by adrenergic stimulation. Indeed, exposure to either ANP or NT-proBNP reduced the maximal responsiveness of mesenteric arteries (Figure 5A) and veins (Figure 5B) to NE. Therefore, it is likely that the elevated concentrations of circulating ANP or NT-proBNP found in the present study and after spaceflight (6) contribute to the compromised ability to lower vascular conductance in the mesenteric circulation with postural changes. It should also be noted that these acute effects of ANP and NT-proBNP on vessel function will likely differ from those of chronic exposure in vivo, since mesenteric arteries and veins from HDT rats demonstrated an intrinsic hypo-responsiveness to NE with no ANP or NT-proBNP in the bathing solution (Figures 1 and 3). We propose that chronic exposure of smooth muscle cells to ANP, NT-proBNP or some other systemic circulating factor associated with HDT impairs their intrinsic contractile function (31).
Mechanisms of Altered Mesenteric Vascular Function
Although not all studies have shown that HDT adversely affects arterial adrenergic vasoconstrictor responsiveness (34), the preponderance of evidence demonstrates that simulated microgravity impairs arterial vasoconstrictor function (10, 11, 13, 27, 43, 47). Likewise, there is not uniform agreement in the literature regarding responses of HDT on the venous circulation. For example, Purdy and colleagues (47) found that simulated microgravity had no effect on femoral veins, whereas Dunbar and colleagues (15) found in large mesenteric veins that simulated microgravity abolished the NE induced downward shift in the pressure-diameter response. Venular pressure-diameter responses in vivo are regulated by sympathetic activity (54) primarily through α1-adrenoreceptors (30, 32). HDT-induced decreases in α1-adrenoreceptors (52) may be partly responsible for the decreased sensitivity to NE observed in veins from several vascular beds (e.g., mesenteric veins: present study - Figure 3, (15); portal vein (39); vena cava (52)). Therefore, much of the current literature supports the notion that HDT reduces the contractile potential of veins to adrenergic stimuli regardless of an increased passive compliance which may (52) or may not (15) occur.
Ramifications of Diminished Mesenteric Vaso- and Venoconstriction
The effects of diminished arterial and venular contraction could have several functional consequences. Within arteries, the attenuated vasoconstrictor responsiveness of splanchnic tissue could reduce the ability of the cardiovascular system to elevate PVR by as much as 25% during orthostasis (48), which in accordance with Ohm’s Law, could diminish the fidelity in which arterial pressure and regional blood flow are regulated. In support of this, rats exposed to simulated microgravity exhibit hypotension during an orthostatic challenge (35, 58). Furthermore, following HDT there is a diminished ability to redistribute cardiac output during exercise such that the splanchnic bed receives a greater proportion of cardiac output than that of control rats (37, 44); this functional over-perfusion of the splanchnic bed with exercise could limit the ability to match oxygen-consumption to O2 delivery in active skeletal muscles, resulting in premature fatigue. Constriction of peripheral veins is also requisite to augment atrial filling pressure to increase stroke volume (48). Therefore, an attenuated venoconstriction (Figures 3 and 5B) could diminish venous return and lower venous filling pressure. Indeed, a lower stroke volume has been estimated in HDT rats (33, 60), which would contribute to the reduced VO2max observed in these animals (44).
Conclusion
Results from the present study demonstrate that, within the same vascular bed, there is a reduced adrenergic constriction of mesenteric arteries and veins. This adrenergic hypo-responsiveness occurs in a vascular bed where blood flow and arterial pressure alterations do not occur as a result of the HDT intervention (37, 59). Therefore, this suggests that the HDT-induced vascular effects on smooth muscle cells in this region are mediated by a systemic circulating factor(s) rather than altered hydrodynamic mechanical influences. Consistent with this hypothesis is an elevation of plasma ANP and NT-proBNP concentrations in HDT rats (Figure 4), which can inhibit constrictor responses of mesenteric arteries (Figure 5A) and veins (Figure 5B) to adrenergic stimulation. These acute effects or the possible chronic effects of ANP and NT-proBNP to alter intrinsic vasomotor properties could occur through several mechanisms, including inhibition of PKC activity and the formation of IP3 in smooth muscle cells. The diminished vaso- and venoconstriction observed (Figures 1 and 3) would likely affect the ability to augment peripheral vascular resistance and cardiac output during orthostatic and exercise stress. Thus, the functional consequences of these vascular alterations may include orthostatic and exercise intolerance commonly associated with actual and simulated microgravity.
ACKNOWLEGEMENTS
This study was supported, in part, by National Aeronautics and Space Administration Grants NAG2-1340, NCC2-1166 and NAG 9-1034, and National Institutes of Health Grants F32 AG25622, DC-006459, HL-58503 and P01HL077670.
Reference
- 1.Berridge MJ, Irvine RF. Inositol trisphosphate, a novel second messenger in cellular signal transduction. Nature. 1984;312:315–321. doi: 10.1038/312315a0. [DOI] [PubMed] [Google Scholar]
- 2.Bondar RL, Kassam MS, Stein F, Dunphy PT. Cerebrovascular response to standing post spaceflight. Aviat Space Environ Med. 1993;64:430. [Google Scholar]
- 3.Brenner BM, Ballermann BJ, Gunning ME, Zeidel ML. Diverse biological actions of atrial natriuretic peptide. Physiol Rev. 1990;70:665–699. doi: 10.1152/physrev.1990.70.3.665. [DOI] [PubMed] [Google Scholar]
- 4.Buckey JC, Jr, Lane LD, Levine BD, Watenpaugh DE, Wright SJ, Moore WE, Gaffney FA, Blomqvist CG. Orthostatic intolerance after spaceflight. J Appl Physiol. 1996;81:7–18. doi: 10.1152/jappl.1996.81.1.7. [DOI] [PubMed] [Google Scholar]
- 5.Burden HW, Poole MC, Zary J, Jeansonne B, Alberts JR. The effects of space flight during gestation on rat uterine smooth muscle. J Gravit Physiol. 1998;5:23–29. [PubMed] [Google Scholar]
- 6.Carcenac C, Herbute S, Masseguin C, Mani-Ponset L, Maurel D, Briggs R, Guell A, Gabrion JB. Hindlimb-suspension and spaceflight both alter cGMP levels in rat choroid plexus. J Gravit Physiol. 1999;6:17–24. [PubMed] [Google Scholar]
- 7.Convertino VA. Conditions of reduced gravity. In: Low PA, editor. Clinical Autonomic Disorders. Philadelphia, PA: Lippincott-Raven; 1997. pp. 429–440. [Google Scholar]
- 8.Convertino VA, Doerr DF, Eckberg DL, Fritsch JM, Vernikos-Danellis J. Head-down bed rest impairs vagal baroreflex responses and provokes orthostatic hypotension. J Appl Physiol. 1990;68:1458–1464. doi: 10.1152/jappl.1990.68.4.1458. [DOI] [PubMed] [Google Scholar]
- 9.de Bold AJ. Atrial natriuretic factor: a hormone produced by the heart. Science. 1985;230:767–770. doi: 10.1126/science.2932797. [DOI] [PubMed] [Google Scholar]
- 10.Delp MD. Myogenic and vasoconstrictor responsiveness of skeletal muscle arterioles is diminished by hindlimb unloading. J Appl Physiol. 1999;86:1178–1184. doi: 10.1152/jappl.1999.86.4.1178. [DOI] [PubMed] [Google Scholar]
- 11.Delp MD, Brown M, Laughlin MH, Hasser EM. Rat aortic vasoreactivity is altered by old age and hindlimb unloading. J Appl Physiol. 1995;78:2079–2086. doi: 10.1152/jappl.1995.78.6.2079. [DOI] [PubMed] [Google Scholar]
- 12.Delp MD, Colleran PN, Wilkerson MK, McCurdy MR, Muller-Delp J. Structural and functional remodeling of skeletal muscle microvasculature is induced by simulated microgravity. Am J Physiol Heart Circ Physiol. 2000;278:H1866–H1873. doi: 10.1152/ajpheart.2000.278.6.H1866. [DOI] [PubMed] [Google Scholar]
- 13.Delp MD, Holder-Binkley T, Laughlin MH, Hasser EM. Vasoconstrictor properties of rat aorta are diminished by hindlimb unweighting. J Appl Physiol. 1993;75:2620–2628. doi: 10.1152/jappl.1993.75.6.2620. [DOI] [PubMed] [Google Scholar]
- 14.Desplanches D, Mayet MH, Sempore B, Frutoso J, Flandrois R. Effect of spontaneous recovery or retraining after hindlimb suspension on aerobic capacity. J Appl Physiol. 1987;63:1739–1743. doi: 10.1152/jappl.1987.63.5.1739. [DOI] [PubMed] [Google Scholar]
- 15.Dunbar SL, Berkowitz DE, Brooks-Asplund EM, Shoukas AA. The effects of hindlimb unweighting on the capacitance of rat small mesenteric veins. J Appl Physiol. 2000;89:2073–2077. doi: 10.1152/jappl.2000.89.5.2073. [DOI] [PubMed] [Google Scholar]
- 16.Dunn CD, Johnson PC, Lange RD. Regulation of hematopoiesis in rats exposed to antiorthostatic hypokinetic/hypodynamia: II. Mechanisms of the "anemia". Aviat Space Environ Med. 1986;57:36–44. [PubMed] [Google Scholar]
- 17.Fareh J, Bayard B, Gabrion J, Thibault G, Oliver J, Bouille C, Gauquelin G, Gharib C. Cardiac and plasma atrial natriuretic peptide after 9-day hindlimb suspension in rats. J Appl Physiol. 1994;76:641–649. doi: 10.1152/jappl.1994.76.2.641. [DOI] [PubMed] [Google Scholar]
- 18.Fenger-Gron J, Mulvany MJ, Christensen KL. Mesenteric blood pressure profile of conscious, freely moving rats. J Physiol. 1995;488(Pt 3):753–760. doi: 10.1113/jphysiol.1995.sp021006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Gaffney FA, Nixon JV, Karlsson ES, Campbell W, Dowdey AB, Blomqvist CG. Cardiovascular deconditioning produced by 20 hours of bedrest with head-down tilt (−5 degrees) in middle-aged healthy men. Am J Cardiol. 1985;56:634–638. doi: 10.1016/0002-9149(85)91025-2. [DOI] [PubMed] [Google Scholar]
- 20.Garbers DL. Guanylyl cyclase receptors and their endocrine, paracrine, and autocrine ligands. Cell. 1992;71:1–4. doi: 10.1016/0092-8674(92)90258-e. [DOI] [PubMed] [Google Scholar]
- 21.Gashev AA, Delp MD, Zawieja DC. Inhibition of the active lymph pump by simulated microgravity in rats. Am J Physiol Heart Circ Physiol. 2006;290:H2295–H2308. doi: 10.1152/ajpheart.00260.2005. [DOI] [PubMed] [Google Scholar]
- 22.Geary GG, Krause DN, Purdy RE, Duckles SP. Simulated microgravity increases myogenic tone in rat cerebral arteries. J Appl Physiol. 1998;85:1615–1621. doi: 10.1152/jappl.1998.85.5.1615. [DOI] [PubMed] [Google Scholar]
- 23.Hanehira T, Kohno M, Yoshikawa J. Endothelin production in cultured vascular smooth muscle cells--modulation by the atrial, brain, and C-type natriuretic peptide system. Metabolism. 1997;46:487–493. doi: 10.1016/s0026-0495(97)90182-7. [DOI] [PubMed] [Google Scholar]
- 24.Hargens AR. Introduction and historical perspectives. In: Hargens AR, editor. Tissue Fluid Pressures and Composition. Baltimore: Williams and Wilkins; 1981. pp. 1–9. [Google Scholar]
- 25.Hargens AR, Steakai J, Johannson C, Tipton CM. Tissue fluid shift, forelimb loading, and tail tension in tail-suspended rats. Physiologist. 1984;27:S37–S38. [Google Scholar]
- 26.Hargens AR, Watenpaugh DE, Breit GA. Control of circulatory function in altered gravitational fields. Physiologist. 1992;35:S80–S83. [PubMed] [Google Scholar]
- 27.Hatton DC, Yue Q, Chapman J, Xue H, Dierickx J, Roullet C, Coste S, Roullet JB, McCarron DA. Blood pressure and mesenteric resistance arterial function after spaceflight. J Appl Physiol. 2002;92:13–17. doi: 10.1152/jappl.2002.92.1.13. [DOI] [PubMed] [Google Scholar]
- 28.Ignarro LJ, Kadowitz PJ. The pharmacological and physiological role of cyclic GMP in vascular smooth muscle relaxation. Annu Rev Pharmacol Toxicol. 1985;25:171–191. doi: 10.1146/annurev.pa.25.040185.001131. [DOI] [PubMed] [Google Scholar]
- 29.Khurana ML, Pandey KN. Atrial natriuretic peptide inhibits the phosphoinositide hydrolysis in murine Leydig tumor cells. Mol Cell Biochem. 1996;158:97–105. doi: 10.1007/BF00225834. [DOI] [PubMed] [Google Scholar]
- 30.Kong JQ, Taylor DA, Fleming WW. Functional distribution and role of alpha-1 adrenoceptor subtypes in the mesenteric vasculature of the rat. J Pharmacol Exp Ther. 1994;268:1153–1159. [PubMed] [Google Scholar]
- 31.Kumar R, Cartledge WA, Lincoln TM, Pandey KN. Expression of guanylyl cyclase-A/atrial natriuretic peptide receptor blocks the activation of protein kinase C in vascular smooth muscle cells. Role of cGMP and cGMP-dependent protein kinase. Hypertension. 1997;29:414–421. doi: 10.1161/01.hyp.29.1.414. [DOI] [PubMed] [Google Scholar]
- 32.Leech CJ, Faber JE. Different alpha-adrenoceptor subtypes mediate constriction of arterioles and venules. Am J Physiol. 1996;270:H710–H722. doi: 10.1152/ajpheart.1996.270.2.H710. [DOI] [PubMed] [Google Scholar]
- 33.Levine BD, Zuckerman JH, Pawelczyk JA. Cardiac atrophy after bed-rest deconditioning: a nonneural mechanism for orthostatic intolerance. Circulation. 1997;96:517–525. doi: 10.1161/01.cir.96.2.517. [DOI] [PubMed] [Google Scholar]
- 34.Looft-Wilson RC, Gisolfi CV. Rat small mesenteric artery function after hindlimb suspension. J Appl Physiol. 2000;88:1199–1206. doi: 10.1152/jappl.2000.88.4.1199. [DOI] [PubMed] [Google Scholar]
- 35.Martel E, Champeroux P, Lacolley P, Richard S, Safar M, Cuche JL. Central hypervolemia in the conscious rat: a model of cardiovascular deconditioning. J Appl Physiol. 1996;80:1390–1396. doi: 10.1152/jappl.1996.80.4.1390. [DOI] [PubMed] [Google Scholar]
- 36.Maurel D, Ixart G, Barbanel G, Mekaouche M, Assenmacher I. Effects of acute tilt from orthostatic to head-down antiorthostatic restraint and of sustained restraint on the intra-cerebroventricular pressure in rats. Brain Res. 1996;736:165–173. doi: 10.1016/0006-8993(96)00676-2. [DOI] [PubMed] [Google Scholar]
- 37.McDonald KS, Delp MD, Fitts RH. Effect of hindlimb unweighting on tissue blood flow in the rat. J Appl Physiol. 1992;72:2210–2218. doi: 10.1152/jappl.1992.72.6.2210. [DOI] [PubMed] [Google Scholar]
- 38.Michel EL, Johnston RS, Dietlein LF. Biomedical results of the Skylab Program. Life Sci Space Res. 1976;14:3–18. [PubMed] [Google Scholar]
- 39.Morel JL, Boittin FX, Halet G, Arnaudeau S, Mironneau C, Mironneau J. Effect of a 14-day hindlimb suspension on cytosolic Ca2+ concentration in rat portal vein myocytes. Am J Physiol. 1997;273:H2867–H2875. doi: 10.1152/ajpheart.1997.273.6.H2867. [DOI] [PubMed] [Google Scholar]
- 40.Mulvagh SL, Charles JB, Riddle JM, Rehbein TL, Bungo MW. Echocardiographic evaluation of the cardiovascular effects of short-duration spaceflight. J Clin Pharmacol. 1991;31:1024–1026. doi: 10.1002/j.1552-4604.1991.tb03666.x. [DOI] [PubMed] [Google Scholar]
- 41.Neri G, Bova S, Malendowicz LK, Mazzocchi G, Nussdorfer GG. Simulated microgravity impairs aldosterone secretion in rats: possible involvement of adrenomedullin. Am J Physiol Regul Integr Comp Physiol. 2002;283:R832–R836. doi: 10.1152/ajpregu.00099.2002. [DOI] [PubMed] [Google Scholar]
- 42.Nishizuka Y. Studies and perspectives of protein kinase C. Science. 1986;233:305–312. doi: 10.1126/science.3014651. [DOI] [PubMed] [Google Scholar]
- 43.Overton JM, Tipton CM. Effect of hindlimb suspension on cardiovascular responses to sympathomimetics and lower body negative pressure. J Appl Physiol. 1990;68:355–362. doi: 10.1152/jappl.1990.68.1.355. [DOI] [PubMed] [Google Scholar]
- 44.Overton JM, Woodman CR, Tipton CM. Effect of hindlimb suspension on VO2 max and regional blood flow responses to exercise. J Appl Physiol. 1989;66:653–659. doi: 10.1152/jappl.1989.66.2.653. [DOI] [PubMed] [Google Scholar]
- 45.Pandey KN. Vascular action and interaction of natriuretic peptide receptors. In: Sowers JR, editor. Contemporary Endocrinology: Endocrinology of Vasculature. Totawa, NJ: Human Press, INC.; 1996. pp. 255–267. [Google Scholar]
- 46.Papadopoulos A, Delp MD. Effects of hindlimb unweighting on the mechanical and structure properties of the rat abdominal aorta. J Appl Physiol. 2003;94:439–445. doi: 10.1152/japplphysiol.00734.2002. [DOI] [PubMed] [Google Scholar]
- 47.Purdy RE, Duckles SP, Krause DN, Rubera KM, Sara D. Effect of simulated microgravity on vascular contractility. J Appl Physiol. 1998;85:1307–1315. doi: 10.1152/jappl.1998.85.4.1307. [DOI] [PubMed] [Google Scholar]
- 48.Rowell LB. Human Cardiovascular Control. New York: Oxford University Press; 1993. [Google Scholar]
- 49.Rowell LB, Detry JM, Blackmon JR, Wyss C. Importance of the splanchnic vascular bed in human blood pressure regulation. J Appl Physiol. 1972;32:213–220. doi: 10.1152/jappl.1972.32.2.213. [DOI] [PubMed] [Google Scholar]
- 50.Saltin B, Blomqvist G, Mitchell JH, Johnson RL, Jr, Wildenthal K, Chapman CB. Response to exercise after bed rest and after training. Circulation. 1968;38:VII1–VII78. [PubMed] [Google Scholar]
- 51.Sauro MD, Fitzpatrick DF. Atrial peptides inhibit protein kinase C-mediated contraction in rat aorta. Eur J Pharmacol. 1990;180:373–376. doi: 10.1016/0014-2999(90)90325-z. [DOI] [PubMed] [Google Scholar]
- 52.Sayet I, Neuilly G, Mironneau J, Mironneau C. Influence of spaceflight, hindlimb suspension, and venous occlusion on alpha 1-adrenoceptors in rat vena cava. J Appl Physiol. 1995;78:1882–1888. doi: 10.1152/jappl.1995.78.5.1882. [DOI] [PubMed] [Google Scholar]
- 53.Shoemaker JK, Hogeman CS, Silber DH, Gray K, Herr M, Sinoway LI. Head-down-tilt bed rest alters forearm vasodilator and vasoconstrictor responses. J Appl Physiol. 1998;84:1756–1762. doi: 10.1152/jappl.1998.84.5.1756. [DOI] [PubMed] [Google Scholar]
- 54.Shoukas AA, Bohlen HG. Rat venular pressure-diameter relationships are regulated by sympathetic activity. Am J Physiol. 1990;259:H674–H680. doi: 10.1152/ajpheart.1990.259.3.H674. [DOI] [PubMed] [Google Scholar]
- 55.Tabuchi Y, Nakamaru M, Rakugi H, Nagano M, Ogihara T. Endothelin enhances adrenergic vasoconstriction in perfused rat mesenteric arteries. Biochem Biophys Res Commun. 1989;159:1304–1308. doi: 10.1016/0006-291x(89)92252-3. [DOI] [PubMed] [Google Scholar]
- 56.Thomason DB, Booth FW. Atrophy of the soleus muscle by hindlimb unweighting. J Appl Physiol. 1990;68:1–12. doi: 10.1152/jappl.1990.68.1.1. [DOI] [PubMed] [Google Scholar]
- 57.Watenpaugh DE, Hargens AR. Handbook of Physiology. Bethesda, MD: American Physiological Society; 1996. The cardiovascular system in microgravity; pp. 631–674. [Google Scholar]
- 58.Wilkerson MK, Lesniewski LA, Golding EM, Bryan RM, Jr, Amin A, Wilson E, Delp MD. Simulated microgravity enhances cerebral artery vasoconstriction and vascular resistance through endothelial nitric oxide mechanism. Am J Physiol Heart Circ Physiol. 2005;288:H1652–H1661. doi: 10.1152/ajpheart.00925.2004. [DOI] [PubMed] [Google Scholar]
- 59.Wilkerson MK, Muller-Delp J, Colleran PN, Delp MD. Effects of hindlimb unloading on rat cerebral, splenic, and mesenteric resistance artery morphology. J Appl Physiol. 1999;87:2115–2121. doi: 10.1152/jappl.1999.87.6.2115. [DOI] [PubMed] [Google Scholar]
- 60.Woodman CR, Sebastian LA, Tipton CM. Influence of simulated microgravity on cardiac output and blood flow distribution during exercise. J Appl Physiol. 1995;79:1762–1768. doi: 10.1152/jappl.1995.79.5.1762. [DOI] [PubMed] [Google Scholar]
- 61.Wronski TJ, Morey-Holton ER. Skeletal response to simulated weightlessness: a comparison of suspension techniques. Aviat Space Environ Med. 1987;58:63–68. [PubMed] [Google Scholar]
- 62.Zhang L-F, Papadopoulos A, Delp MD. Vascular adaptation to microgravity. J Appl Physiol. 2004;97:1584–1587. doi: 10.1152/japplphysiol.00534.2003. [DOI] [PubMed] [Google Scholar]
- 62.Zhang LN, Zhang L-F, Ma J. Simulated microgravity enhances vasoconstrictor responsiveness of rat basilar artery. J Appl Physiol. 2001;90:2296–2305. doi: 10.1152/jappl.2001.90.6.2296. [DOI] [PubMed] [Google Scholar]