Key Points
The KKO-inhibition ELISA and DT40-luciferase tests are novel laboratory assays for HIT.
They showed better discrimination than 2 commercial immunoassays and may improve the specificity and feasibility of HIT laboratory testing.
Abstract
Laboratory testing for heparin-induced thrombocytopenia (HIT) has important shortcomings. Immunoassays fail to discriminate platelet-activating from nonpathogenic antibodies. Specific functional assays are impracticable due to the need for platelets and radioisotope. We describe 2 assays that may overcome these limitations. The KKO-inhibition test (KKO-I) measures the effect of plasma on binding of the HIT-like monoclonal antibody KKO to platelet factor 4 (PF4)/heparin. DT40-luciferase (DT40-luc) is a functional test comprised of a B-cell line expressing FcγRIIa coupled to a luciferase reporter. We compared these assays to polyspecific and immunoglobulin (Ig)G-specific PF4/heparin enzyme-linked immunosorbent assays (ELISAs) in samples from 58 patients with suspected HIT and circulating anti-PF4/heparin antibodies. HIT was defined as a 4Ts score ≥ 4 and positive 14C-serotonin release assay. HIT-positive plasma demonstrated greater mean inhibition of KKO binding than HIT-negative plasma (78.9% vs 26.0%; P < .0001) and induced greater luciferase activity (3.14-fold basal vs 0.96-fold basal; P < .0001). The area under the receiver-operating characteristic curve was greater for KKO-I (0.93) than for the polyspecific (0.82; P = .020) and IgG-specific ELISA (0.76; P = .0044) and for DT40-luc (0.89) than for the IgG-specific ELISA (P = .046). KKO-I and DT40-luc showed better discrimination than 2 commercially available immunoassays, are simple to perform, and hold promise for improving the specificity and feasibility of HIT laboratory testing.
Introduction
Heparin-induced thrombocytopenia (HIT) is a prothrombotic disorder mediated by platelet-, monocyte-, and endothelial cell-activating antibodies that preferentially recognize ultra-large complexes of platelet factor 4 (PF4) and heparin.1,2 Laboratory testing plays a key role in the diagnosis of HIT but is associated with important shortcomings.3 Immunoassays such as the PF4/heparin enzyme-linked immunosorbent assay (ELISA) frequently yield false-positive results due to their inability to discriminate cell-activating and potentially pathogenic antibodies from their nonpathogenic counterparts. Functional tests such as the 14C-serotonin release assay (SRA) are more specific but are unfeasible for most clinical laboratories due to the requirement for radioisotope and fresh platelets from reactive donors.3
KKO is a murine monoclonal anti-PF4/heparin immunoglobulin (Ig)G that induces a HIT-like thrombotic thrombocytopenic disorder in a mouse model. RTO, an isotype-matched anti-PF4 antibody not dependent on heparin for comparable binding in an ELISA, does not activate platelets in vitro or cause thrombocytopenia in vivo.4 Binding of KKO (but not RTO) to immobilized PF4/heparin is inhibited by human HIT plasma but not by plasma from patients with nonplatelet-activating (SRA-negative) anti-PF4/heparin antibodies.5 We leveraged this property of KKO to develop a KKO-inhibition (KKO-I) ELISA for selective detection of platelet-activating antibodies. We also recently described a system to measure cellular activation by HIT antibodies using DT40 (chicken B lymphocyte) cells transfected with human FcγRIIa coupled to a luciferase reporter.6 We hypothesized that this system (DT40-luciferase [DT40-luc]) could be used to identify cell-activating anti-PF4/heparin antibodies without need for donor platelets or radioactivity. Herein, we compare the performance of the KKO-I and DT40-luc assays to 2 commercially available immunoassays in samples from 58 patients with suspected HIT and circulating anti-PF4/heparin antibodies.
Methods
Patient samples consecutively referred to the University of Pennsylvania for laboratory assessment of HIT that tested positive [i.e. optical density (OD) ≥ 0.40] in a polyspecific PF4/heparin ELISA (Hologic Gen-Probe, San Diego, CA) were included. Citrated plasma samples from all patients were also tested using an IgG-specific PF4/heparin ELISA (Hologic Gen-Probe), an in-house SRA, and the investigational KKO-I and DT40-luc assays. The polyspecific and IgG-specific ELISAs were performed in accordance with the manufacturer’s instructions. The SRA was performed with platelet-rich plasma (PRP) as previously described (hereafter referred to as PRP-SRA)7 and was considered positive if there was <5% 14C-serotonin release after patient plasma was added to platelets in the absence of heparin and >20% release after the addition of 0.1 or 0.5 U/mL of heparin.
The KKO-I assay was performed as previously described.5 Briefly, Immulon 4 HBx 96-well plates (Thermo Fisher Scientific, Waltham, MA) coated with PF4 and heparin (Sagent Pharmaceuticals, Schaumburg, IL) were incubated with human plasma (1:50 dilution) for 30 min at 37°C followed by incubation with KKO for an additional 10 min at 37°C. Recombinant PF4 was expressed in Drosophila Schneider 2 cells and purified as previously described.6 KKO binding was measured as absorbance at 405 nm (A405) after incubation with horseradish peroxidase-conjugated goat anti-mouse IgG-Fc (Jackson ImmunoResearch Laboratories, West Grove, PA) and the horseradish peroxidase substrate ABTS (Roche Applied Science, Penzberg, Germany). Absorbance was measured with a SpectraCount plate reader (Packard BioScience, Waltham, MA). Data are presented as percent inhibition of KKO binding, calculated as ([A405max −A405patient]/A405max) × 100, with A405max set in the absence of plasma. “0%” represents no inhibition of KKO binding and 100% represents complete inhibition.
The DT40-luc assay was performed as previously described.6 In brief, DT40 chicken B cells were transiently transfected to express human FcγRIIA (pEF6-FcγRIIA) as well as a reporter molecule (NFAT-Luc), which consists of the luciferase gene under control of the IL-2 promoter. On the day following transfection, the resultant DT40-luc cells were placed in 96-well culture plates. PF4/heparin complexes were formed first by incubating recombinant PF4 with heparin for 15 min at 37°C, followed by the addition of patient plasma (1:800 final dilution). The PF4/heparin/plasma mixtures were then added to the cells for 6 h at 37°C in an atmosphere containing 5% CO2. Plates were frozen at −80°C to terminate the activation reaction. To measure activation, cells were thawed and lysed with 5× Passive Lysis Buffer (Promega, Madison, WI) for 15 min. Luciferase activity was measured on a Berthold (Pforzheim, Germany) MultiLumat LB 9506 Luminometer (10-s readings) using Luciferase Assay Reagent (Promega) following the manufacturer’s instructions. Data are reported as the ratio of luciferase signal induced by patient plasma relative to the absence of plasma (fold-basal). Normalization of the luciferase signal in the absence of plasma was performed with each transfection to account for potential differences in transfection efficiency.
We determined the 4Ts score for each subject by retrospective chart review to estimate the clinical likelihood of HIT at the time of laboratory testing.8 The investigator performing 4Ts scoring (A.C.) was blinded to the results of all HIT laboratory assays. Investigators performing the KKO-I and DT40-luc assays (A.H.R., J.L.H.) were blinded to the 4Ts score and the results of the PRP-SRA and the polyspecific and IgG-specific PF4/heparin ELISAs. The protocol was approved by the University of Pennsylvania institutional review board. The study was conducted in accordance with the Declaration of Helsinki.
HIT was defined as the combination of an intermediate or high-probability 4Ts score (≥4) and a positive PRP-SRA. The performances of the polyspecific ELISA, IgG-specific ELISA, KKO-I, and DT40-luc assay were evaluated with respect to this reference standard by receiver-operating characteristic (ROC) analysis. Areas under the ROC curves (AUCs) were calculated and compared by the DeLong method for correlated samples.9 Analyses were carried out using GraphPad Prism 5 (GraphPad Software, La Jolla, CA) and Analyze-it (Analyze-it Software, Leeds, UK). A P value < 0.05 was considered statistically significant.
Results
Plasma samples from 58 subjects were studied. Each of the 21 subjects testing positive by PRP-SRA had a 4Ts score ≥4 and thus met the prespecified criteria for HIT. Plasma samples from 37 subjects tested negative by PRP-SRA and were considered negative for HIT. Demographic and clinical characteristics of HIT-positive and -negative subjects are summarized in Table 1. The median 4Ts score was significantly greater in HIT-positive subjects (6 vs 4; P < .0001), although 21 (57%) HIT-negative subjects had an intermediate or high probability (≥4) score, highlighting the limited specificity of clinical diagnosis.3 More patients in the HIT-positive group were on the cardiovascular surgical service (66.7% vs 37.8%) and received treatment for HIT (90.5% vs 67.6%), although these differences did not meet statistical significance (P = .06 for both comparisons). Other demographic and clinical features were similar between the 2 groups (Table 1).
Table 1.
Demographic and clinical characteristics of HIT-positive and HIT-negative subjects
| Characteristic | HIT positive (n = 21) | HIT negative (n = 37) | P value |
|---|---|---|---|
| Age, mean (range) | 66 (39-89) | 63 (39-80) | .47 |
| Female gender, n (%) | 9 (42.9) | 19 (51.4) | .59 |
| Race, n (%) | .39 | ||
| White | 16 (76.2) | 23 (62.2) | |
| Black | 1 (4.8) | 6 (16.2) | |
| Other/unknown | 4 (19.0) | 8 (21.6) | |
| Patient population, n (%) | .06 | ||
| Cardiovascular surgery | 14 (66.7) | 14 (37.8) | |
| Surgery (noncardiovascular) | 5 (23.8) | 9 (24.3) | |
| Medicine | 2 (9.5) | 13 (35.1) | |
| Other | 0 | 1 (2.7) | |
| Setting, n (%) | .22 | ||
| Tertiary care hospital | 18 (85.7) | 26 (70.3) | |
| Community hospital | 3 (14.2) | 11 (29.7) | |
| Platelet count × 109/L, median (range)* | 50 (19-139) | 69 (3-883) | .29 |
| Recognized thrombosis, n (%)* | 14 (66.7) | 20 (54.1) | .41 |
| 4Ts score, median (interquartile range) | 6 (5-7) | 4 (3-5) | <.0001 |
| Received treatment for HIT, n (%) | 19 (90.5) | 25 (67.6) | .06 |
At time of HIT laboratory testing.
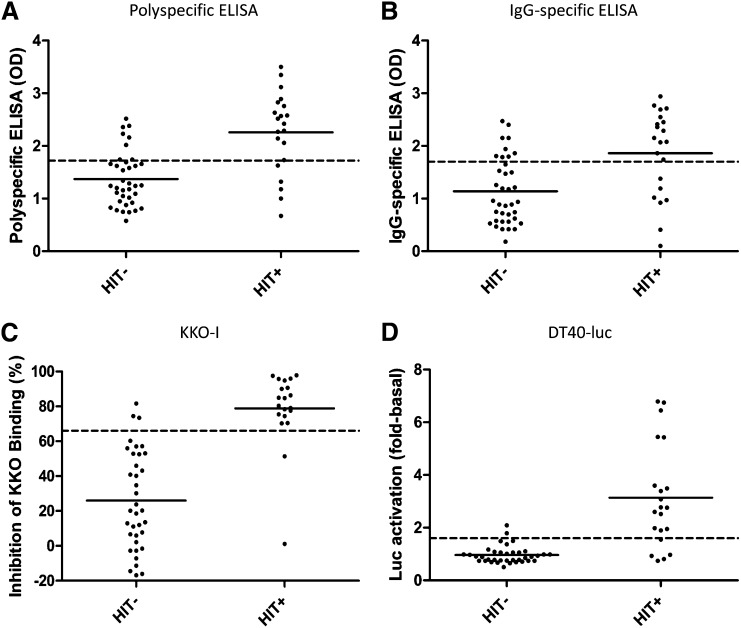
All 58 patient samples were tested using the polyspecific ELISA, IgG-specific ELISA, and DT40-luc. One HIT-positive and one HIT-negative sample were not tested by KKO-I due to insufficient sample volume. The ability of each assay to discriminate HIT-positive from HIT-negative subjects is shown in Figure 1 (A-D, respectively). Consistent with previous studies,10 the mean OD was higher among HIT-positive than HIT-negative subject samples by both polyspecific (2.26 vs 1.37; P < .0001) and IgG-specific ELISA (1.86 vs 1.14; P = .0004). However, a significant overlap in OD values among HIT-positive and HIT-negative subjects was observed with both assays (Figure 1A-B), underscoring their limited capacity to discriminate cell-activating (and presumably pathogenic) from nonactivating PF4/heparin antibodies at the level of the individual patient.
Figure 1.
Assay results in HIT-negative and HIT-positive subjects. Results from HIT-negative and -positive subjects are shown for the polyspecific ELISA (A), IgG-specific ELISA (B), KKO-I (C), and DT40-luc (D). Solid horizontal lines represent mean values. Dashed horizontal lines represent the cutoff associated with the most northwest point on the receiver operating characteristic curve (ie, the cutoff at which sensitivity and specificity are optimized) for each assay.
KKO-I and DT40-luc showed better diagnostic discrimination than the commercially available ELISAs (Figure 1C-D). HIT-positive plasma samples exhibited significantly greater mean inhibition of KKO binding (78.9% vs 26.0%; P < .0001) (Figure 1C) and induced significantly greater luciferase activity (3.14-fold basal vs 0.96-fold basal; P < .0001) than HIT-negative samples (Figure 1D).
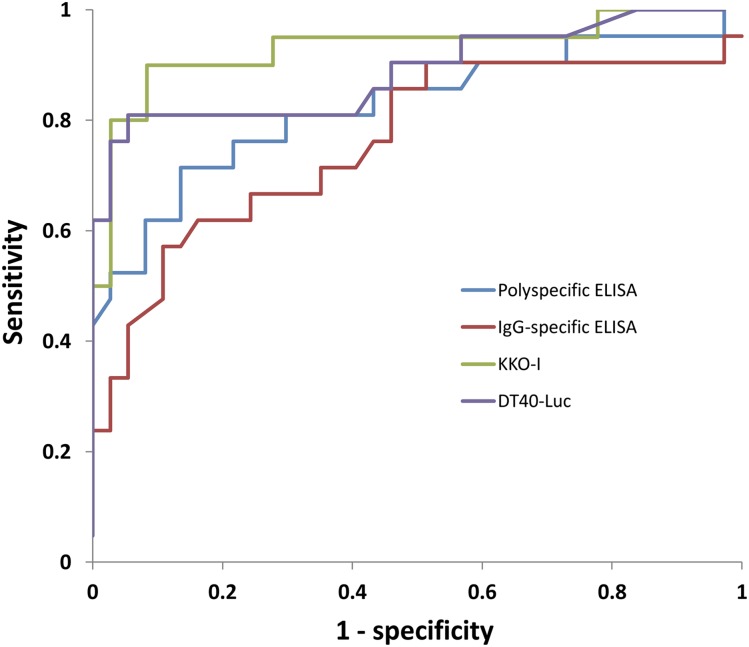
ROC curves for each assay are shown in Figure 2. The AUC for KKO-I (0.93, 95% CI: 0.85-1.00) was significantly greater than the AUC for the polyspecific (0.82, 0.70-0.95; P = .020) and IgG-specific (0.76, 0.62-0.90; P = .0044) ELISAs, but not the DT40-luc (0.89, 0.79-0.99; P = .40). The AUC for DT40-luc was significantly greater than the AUC for the IgG-specific (P = .046) but not the polyspecific ELISA (P = .28).
Figure 2.
ROC curves. ROC curves are shown for the polyspecific ELISA, IgG-specific ELISA, KKO-I, and DT40-luc. The AUCs for these assays were 0.82, 0.76, 0.93, and 0.89, respectively. The AUC for KKO-I was significantly greater than the AUC for the polyspecific (P = .020) and IgG-specific ELISAs (P = .0044). The AUC for DT40-luc was significantly greater than the AUC for the IgG-specific (P = .046) but not the polyspecific ELISA (P = .28).
Table 2 shows the sensitivity/specificity pairs at the most northwest point on the ROC curve (the point at which sensitivity and specificity are optimized) for each assay. The cutoffs associated with these points are denoted by dashed horizontal lines in Figure 1. At a cutoff of 66%, the sensitivity and specificity of KKO-I was 0.90 and 0.92, respectively, and correctly classified 91% (51/56) of samples with respect to the reference standard. A lower cutoff of 50% improved sensitivity (0.95) at the cost of reduced specificity (0.72). The sensitivity and specificity of DT40-luc at a cutoff of 1.6-fold basal were 0.81 and 0.95, respectively, and were associated with correct classification of 88% (51/58) of samples. The operating characteristics of KKO-I and DT40-luc compared favorably with those of the polyspecific and IgG-specific ELISA. At cutoffs of 1.72 OD and 1.70 OD, the sensitivity of the latter assays was 0.76 and 0.67 and the specificity 0.78 and 0.76, respectively (Table 2). At these optimized cutoffs, the polyspecific ELISA correctly classified 78% (45/58) and the IgG-specific ELISA 72% (42/58) of samples.
Table 2.
Operating characteristics of the assays at the most northwest point on the ROC curve
| Assay | Cutoff | Sensitivity (95% CI) | Specificity (95% CI) |
|---|---|---|---|
| Polyspecific ELISA | 1.72 OD | 0.76 (0.53-0.92) | 0.78 (0.62-0.90) |
| IgG-specific ELISA | 1.70 OD | 0.67 (0.43-0.85) | 0.76 (0.59-0.88) |
| KKO-I | 66% | 0.90 (0.68-0.99) | 0.92 (0.78-0.98) |
| DT40-luc | 1.6-fold basal | 0.81 (0.58-0.95) | 0.95 (0.82-0.99) |
| Combinatorial | KKO-I ≥75% or DT40-luc ≥ 2-fold basal | 0.95 (0.74-1.00) | 0.94 (0.80-0.99) |
Combinatorial analysis showed that the performance of KKO-I and DT40-luc could be further improved when both assays are integrated within a single diagnostic algorithm. A combinatorial strategy in which a sample was considered positive if it demonstrated either KKO-I ≥75% or DT40-luc ≥2.0-fold basal would have resulted in the correct classification of 55 of the 58 patients and a sensitivity and specificity of 0.95 and 0.94, respectively (Table 2). Conjunctive combinatorial strategies (ie, Boolean operations using “AND” rather than “OR”) did not result in improved performance.
Because the polyspecific PF4/heparin ELISA has very high negative predictive value for HIT,3,10 we chose to confine our study cohort to the more diagnostically challenging patients who test positive by polyspecific ELISA (OD ≥ 0.4). Nevertheless, samples from 9 additional subjects with suspected HIT who tested negative in the polyspecific ELISA were analyzed as negative controls. As expected, all 9 samples showed negligible inhibition of KKO binding (range: −16.1% to 11.9%) and luciferase activity (0.75-fold basal to 1.37-fold basal) (data not shown).
Discussion
HIT laboratory testing is associated with limitations in specificity and feasibility. In this report, we describe 2 novel assays for HIT that may overcome these limitations: KKO-I, a modification of the PF4/heparin ELISA, and DT40-luc, a cell-based functional assay.
We previously reported that binding of KKO to immobilized PF4/heparin is inhibited to a greater extent by PRP-SRA–positive plasma than by plasma from patients with non–cell-activating anti-PF4/heparin antibodies.5 The KKO-I assay was designed to leverage this property of KKO, which possesses in vitro and in vivo platelet-activating activity, for the purpose of discriminating cell-activating and potentially pathogenic HIT antibodies from their nonpathogenic counterparts. In the present study, plasma from HIT-positive subjects demonstrated significantly greater mean inhibition of KKO binding than HIT-negative plasma (78.9% vs 26.0%; P < .0001) by KKO-I (Figure 1C). These findings suggest that human cell-activating antibodies in HIT plasma bind to epitopes on PF4 that overlap to a greater extent with KKO’s binding site(s) than epitopes recognized by nonplatelet-activating antibodies, such as the isotype-matched monoclonal anti-PF4 antibody RTO,5 and point to epitope specificity as a potential major determinant of anti-PF4/heparin antibody pathogenicity.
KKO-I is not the only diagnostic assay for HIT to make use of KKO. HemosIL HIT-Ab(PF4-H) (Instrumentation Laboratory, Bedford, MA) is a latex particle-enhanced immunoturbidimetric assay in which agglutination of KKO-coated latex beads to PF4/polyvinylsulfonate complexes in solution is inhibited in the presence of human anti-PF4/heparin antibodies. Like the polyspecific PF4/heparin ELISA, HemosIL HIT-Ab(PF4-H) has high sensitivity but appears to be limited in its capacity to discriminate cell-activating from nonpathogenic antibodies. The assay was studied in a cohort of 102 subjects with suspected HIT, in which HIT was defined as an intermediate or high clinical suspicion coupled with a positive aggregometry-based functional assay. All 17 samples meeting this definition tested positive by HemosIL HIT-Ab(PF4-H) (sensitivity 100%), but 16 additional HIT-negative samples also tested positive (specificity 81%).11 Although KKO-I and HemosIL HIT-Ab(PF4-H) have not been compared head to head, our data suggest that the former may be more specific for platelet-activating antibodies. The reasons for this apparent discrepancy in specificity are unknown but may relate to differences in the geometry and kinetics of KKO. The spatial orientation and intermolecular organization of KKO on the latex bead may permit steric inhibition by a higher proportion of anti-PF4/heparin antibodies of diverse epitope specificities compared with more stringent requirements to compete with the same antibody in solution. In addition, the epitope(s) recognized by KKO on complexes of PF4/polyvinylsulfonate in solution may sufficiently differ from those on immobilized complexes of PF4/heparin that nonpathogenic antibodies can compete effectively. A better understanding of these differences may lead to more detailed characterization of the pathogenic epitopes in HIT, which in turn could be used to further improve assay specificity.
Previous work has shown that FcγRIIa is required for the platelet activation central to the pathogenesis of HIT.12 To obviate the need for fresh, reactive, donor platelets, we developed a DT40 (chicken B lymphocyte) cell line transfected with human FcγRIIa coupled to a luciferase reporter to measure cellular activation by HIT antibodies.6 In this platelet-free system (DT40-luc), plasma from HIT-positive subjects induced significantly greater luciferase activity than plasma from HIT-negative subjects (3.14-fold basal vs 0.96-fold basal; P < .0001) (Figure 1D). Thus, this assay recapitulates the salient requirements to induce HIT, namely PF4-heparin-IgG complexes capable of activating cells via FcγRIIa,12-15 without the need for platelet-specific antigens. This is an important feature in light of emerging evidence that activation of other cell types (eg, endothelial cells, monocytes) may contribute to the prothrombotic phenotype of HIT through this and other Fcγ receptors.16-18
In the current study, KKO-I and DT40-luc exhibited better discrimination than a commercially available polyspecific and IgG-specific ELISA (Figure 1). At the most northwest point on their respective ROC curves (Figure 2), the sensitivity and specificity of the novel assays were superior to those of the commercial tests (Table 2). The sensitivity of the polyspecific and IgG-specific ELISA is lower than that reported in other studies (0.95-1.00),19 primarily because we used the most northwest point on the ROC curve in our analysis rather than the manufacturer-recommended cutoff of 0.40. The specificity of all 4 assays would likely have been enhanced had samples from patients testing negative by polyspecific PF4/heparin ELISA, a group that typically comprises 70% to 90% of a reference laboratory’s test population,19 been included in the study cohort.
Our findings have potentially important clinical implications. The diagnosis of HIT is a high stakes enterprise. Delays in diagnosis and institution of appropriate therapy are associated with an initial 6.1% daily risk of thromboembolism, amputation, and death.20 Misdiagnosis, conversely, may result in unnecessary exposure of thrombocytopenic patients without HIT to direct thrombin inhibitors and their attendant 1% daily risk of major hemorrhage.21,22 The latter is a particularly prevalent problem in current practice,23 as highlighted by the observation that two-thirds of the 37 HIT-negative subjects in our study nonetheless received treatment for HIT (Table 1) with potential bleeding complications and unlikely benefit.
The limited specificity of currently available immunoassays and the limited availability of more specific functional assays contribute to the problem of overdiagnosis and unnecessary treatment. Immunoassays such as the polyspecific PF4/heparin ELISA are simple to perform and widely used but are unable to discriminate cell-activating and potentially pathogenic from nonpathogenic antibodies (Figure 1A). Depending on the patient population, only 28% to 59% of samples testing positive by polyspecific ELISA also test positive by a more specific functional assay.8,24-26 The OD of a positive ELISA is a helpful, but relatively crude predictor of pathogenicity (Figures 1A-B).10 Modifications of the PF4/heparin ELISA that detect only antibodies of the IgG class (eg, IgG-specific ELISA) or antibodies inhibited by excess heparin (eg, high-dose heparin confirmatory test) enhance specificity,24,26,27 but false-positive results remain common with these approaches and reductions in sensitivity have also been reported.19,28
KKO-I and DT40-luc demonstrated better diagnostic discrimination than a commercially available polyspecific and IgG-specific ELISA in the present study (Figure 2; Table 2) and hold promise for improving the specificity of laboratory diagnosis and curtailing the current trend of overdiagnosis. Of the 37 HIT-negative subjects in our study, all would have been misdiagnosed with (and potentially treated for) HIT based on the manufacturer-recommended polyspecific ELISA OD cutoff of 0.40. Even the use of an optimized cutoff corresponding to the most northwest point on the ROC curve would have resulted in misclassification of 13 of 58 (22%) subjects by polyspecific ELISA and 16 of 58 (28%) by IgG-specific ELISA. In contrast, KKO-I and DT40-luc correctly classified 33 of 36 (92%) and 35 of 37 (95%) HIT-negative patients, respectively, suggesting the potential for these novel assays to curb the current problem of overdiagnosis and unnecessary treatment.
Functional assays for HIT such as the SRA and the heparin-induced platelet activation test are sensitive and more specific than commercial immunoassays, but they are impracticable for most clinical laboratories due to the need for fresh reactive donor platelets, radioisotope (for the SRA), and meticulous technique and platelet aggregometry (for the heparin-induced platelet activation test).7,29 KKO-I and DT40-luc may represent more feasible options for laboratories seeking to improve the specificity of the testing they offer. As a modification of the PF4/heparin ELISA, KKO-I could be relatively easily adopted by laboratories that currently perform the former assay. DT40-luc is a test of cellular activation in which donor platelets are replaced by a cell line that can be stored at −80°C and retrieved as needed for testing. The endpoint for cellular activation is luciferase activity, which can be measured by a standard spectrophotometer without the need for radioactivity or platelet aggregometry.
KKO and the transiently transfected DT40 cell line are not widely available at present. We are currently developing a stably transfected cell line. Should the value of KKO-I and DT40-luc be validated in a larger prospective study, recombinant monoclonal antibody and a stably transfected cell line would provide the opportunity for laboratories to employ shared antigenic and functional assay systems.
Several limitations of our study deserve mention. First, the study population was relatively small and was recruited from 2 hospitals within a single health system. Validation in a larger multicenter study is required. Second, the PRP-SRA employed in the current study7 differs from the more widely used washed platelet SRA.30 PRP-based aggregometry assays may be less specific than washed platelet functional assays,31 though SRAs utilizing PRP and washed platelets have not been directly compared. Third, there is no universally accepted gold standard for HIT. In a recent meta-analysis, a 4Ts score ≥4 was associated with a positive predictive value of only 22%.32 Positive results by SRA in the absence of clinical HIT may also occur, particularly after cardiac surgery.33 To overcome these limitations in the positive predictive value of the 4Ts and SRA, we used a reference standard (4Ts score ≥ 4 and positive PRP-SRA) incorporating both clinical and laboratory criteria, as recommended by an international expert consensus panel.34 Nevertheless, misclassification of some subjects in our study cannot be excluded.
With this caveat in mind, 2 subjects that met the prespecified definition of HIT tested negative by KKO-I at a cutoff of 66% (Figure 1C). One was a 43-year-old woman who developed thrombocytopenia, lower extremity deep vein thrombosis, and pulmonary embolism following resection of locally advanced ovarian cancer. Her 4Ts score was 7 (high probability) and her platelet count normalized rapidly on argatroban. Her plasma showed only 1% inhibition by KKO-I and 0.81-fold basal activation by DT40-luc. Interestingly, weakly positive results were obtained by polyspecific ELISA, IgG-specific ELISA, and PRP-SRA (0.67, 0.41, and 43%, respectively). Values in this range have been reported to occur in the absence of clinical HIT,10,35 suggesting that the patient may have been misclassified by the reference standard rather than by KKO-I. The second patient was a 72-year-old man who developed thrombocytopenia, lower extremity deep vein thrombosis, and necrotic skin lesions at heparin injection sites following percutaneous repair of a celiac artery aneurysm (4Ts score = 7). Thrombocytopenia resolved with cessation of heparin and initiation of argatroban. The results of the polyspecific ELISA, IgG-specific ELISA, and PRP-SRA were 1.32, 0.97, and 93%, respectively. Although the patient tested negative by KKO-I (51% inhibition), his plasma induced strong luciferase activity (6.45-fold basal) by the DT40-luc assay. This laboratory profile suggests the possibility of an uncommon variant of HIT in which cell-activating antibodies recognize complexes of heparin and cationic proteins other than PF4 such as interleukin-8 or neutrophil-activating peptide-2.36 Such antibodies might be detectable using functional assays (eg, DT40-luc) but not by PF4/heparin immunoassays (eg, KKO-I).
As suggested by this second case in which discrepant results were obtained by KKO-I and DT40-luc, a combinatorial approach may yield better performance than use of either assay alone. Indeed, an integrated algorithm in which a sample was considered positive if it demonstrated either KKO-I ≥75% or DT40-luc ≥2.0-fold basal would have resulted in correct classification of 55 of the 58 patients in our study and a sensitivity and specificity of 0.95 (0.74-1.00) and 0.94 (0.80-0.99), respectively (Table 2). The operating characteristics of such an approach require validation in an independent test population.
In summary, we describe a novel immunoassay (KKO-I) and a novel functional assay (DT40-luc) for the diagnosis of HIT. KKO-I and DT40-luc discriminated cell-activating and potentially pathogenic antibodies from nonactivating antibodies more effectively than 2 commercially available ELISAs in a small cohort of patients with suspected HIT. These assays are simple to perform; do not require donor platelets, radioactivity, or platelet aggregometry; and hold promise for improving the specificity and feasibility of HIT laboratory testing. Validation of KKO-I and DT40-luc in a larger prospective study is underway.
Acknowledgments
This work was supported by HL112903 to A.C., HL099973 to B.A.K. and D.B.C., HL110860 to G.M.A., D.B.C., and B.S.S., HL109825 and AI101992 to G.M.A., and HL078726 and HL078726-S1 to B.S.S. S.P.W. holds a British Heart Foundation Chair.
Footnotes
The publication costs of this article were defrayed in part by page charge payment. Therefore, and solely to indicate this fact, this article is hereby marked “advertisement” in accordance with 18 USC section 1734.
Authorship
Contribution: A.C., D.B.C., and B.S.S. conceived and designed the study, analyzed and interpreted the data, and wrote the manuscript; A.H.R. performed assays, analyzed the data, and edited the manuscript; J.L.H., M.D.C., S.V.Y., and I.A.M.B. performed assays and edited the manuscript; W.Y. analyzed the data and edited the manuscript; and B.A.K., G.M.A., and S.P.W. provided critical reagents and edited the manuscript.
Conflict-of-interest disclosure: A.C. has provided consulting services to Baxter, Bayer, Biogen-Idec, Canyon Pharmaceuticals, CSL Behring, Daiichi-Sankyo, and Genzyme; has received research funding from Baxter, Bayer, and Novo Nordisk; and has provided expert witness testimony relating to HIT. G.M.A. has provided consulting services to Teva Pharmaceuticals. D.B.C. has provided consulting services to Bayer. KKO-I and DT40-luc are protected by provisional US patents 61/640 960 and 61/614 729, respectively. The remaining authors declare no competing financial interests.
Correspondence: Adam Cuker, Hospital of the University of Pennsylvania, 3 Dulles, 3400 Spruce St, Philadelphia, PA 19104; e-mail: adam.cuker@uphs.upenn.edu.
References
- 1.Amiral J, Bridey F, Dreyfus M, Vissoc AM, Fressinaud E, Wolf M, Meyer D. Platelet factor 4 complexed to heparin is the target for antibodies generated in heparin-induced thrombocytopenia. Thromb Haemost. 1992;68(1):95–96. [PubMed] [Google Scholar]
- 2.Rauova L, Poncz M, McKenzie SE, et al. Ultralarge complexes of PF4 and heparin are central to the pathogenesis of heparin-induced thrombocytopenia. Blood. 2005;105(1):131–138. doi: 10.1182/blood-2004-04-1544. [DOI] [PubMed] [Google Scholar]
- 3.Cuker A, Cines DB. How I treat heparin-induced thrombocytopenia. Blood. 2012;119(10):2209–2218. doi: 10.1182/blood-2011-11-376293. [DOI] [PubMed] [Google Scholar]
- 4.Arepally GM, Kamei S, Park KS, et al. Characterization of a murine monoclonal antibody that mimics heparin-induced thrombocytopenia antibodies. Blood. 2000;95(5):1533–1540. [PubMed] [Google Scholar]
- 5.Sachais BS, Litvinov RI, Yarovoi SV, et al. Dynamic antibody-binding properties in the pathogenesis of HIT. Blood. 2012;120(5):1137–1142. doi: 10.1182/blood-2012-01-407262. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sachais BS, Rux AH, Cines DB, et al. Rational design and characterization of platelet factor 4 antagonists for the study of heparin-induced thrombocytopenia. Blood. 2012;119(25):5955–5962. doi: 10.1182/blood-2012-01-406801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cines DB, Kaywin P, Bina M, Tomaski A, Schreiber AD. Heparin-associated thrombocytopenia. N Engl J Med. 1980;303(14):788–795. doi: 10.1056/NEJM198010023031404. [DOI] [PubMed] [Google Scholar]
- 8.Lo GK, Juhl D, Warkentin TE, Sigouin CS, Eichler P, Greinacher A. Evaluation of pretest clinical score (4 T’s) for the diagnosis of heparin-induced thrombocytopenia in two clinical settings. J Thromb Haemost. 2006;4(4):759–765. doi: 10.1111/j.1538-7836.2006.01787.x. [DOI] [PubMed] [Google Scholar]
- 9.DeLong ER, DeLong DM, Clarke-Pearson DL. Comparing the areas under two or more correlated receiver operating characteristic curves: a nonparametric approach. Biometrics. 1988;44(3):837–845. [PubMed] [Google Scholar]
- 10.Warkentin TE, Sheppard JI, Moore JC, Sigouin CS, Kelton JG. Quantitative interpretation of optical density measurements using PF4-dependent enzyme-immunoassays. J Thromb Haemost. 2008;6(8):1304–1312. doi: 10.1111/j.1538-7836.2008.03025.x. [DOI] [PubMed] [Google Scholar]
- 11.Legnani C, Cini M, Pili C, Boggian O, Frascaro M, Palareti G. Evaluation of a new automated panel of assays for the detection of anti-PF4/heparin antibodies in patients suspected of having heparin-induced thrombocytopenia. Thromb Haemost. 2010;104(2):402–409. doi: 10.1160/TH10-01-0002. [DOI] [PubMed] [Google Scholar]
- 12.Reilly MP, Taylor SM, Hartman NK, et al. Heparin-induced thrombocytopenia/thrombosis in a transgenic mouse model requires human platelet factor 4 and platelet activation through FcgammaRIIA. Blood. 2001;98(8):2442–2447. doi: 10.1182/blood.v98.8.2442. [DOI] [PubMed] [Google Scholar]
- 13.Visentin GP, Ford SE, Scott JP, Aster RH. Antibodies from patients with heparin-induced thrombocytopenia/thrombosis are specific for platelet factor 4 complexed with heparin or bound to endothelial cells. J Clin Invest. 1994;93(1):81–88. doi: 10.1172/JCI116987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Greinacher A, Pötzsch B, Amiral J, Dummel V, Eichner A, Mueller-Eckhardt C. Heparin-associated thrombocytopenia: isolation of the antibody and characterization of a multimolecular PF4-heparin complex as the major antigen. Thromb Haemost. 1994;71(2):247–251. [PubMed] [Google Scholar]
- 15.Kelton JG, Smith JW, Warkentin TE, Hayward CP, Denomme GA, Horsewood P. Immunoglobulin G from patients with heparin-induced thrombocytopenia binds to a complex of heparin and platelet factor 4. Blood. 1994;83(11):3232–3239. [PubMed] [Google Scholar]
- 16.Blank M, Shoenfeld Y, Tavor S, et al. Anti-platelet factor 4/heparin antibodies from patients with heparin-induced thrombocytopenia provoke direct activation of microvascular endothelial cells. Int Immunol. 2002;14(2):121–129. doi: 10.1093/intimm/14.2.121. [DOI] [PubMed] [Google Scholar]
- 17.Rauova L, Hirsch JD, Greene TK, et al. Monocyte-bound PF4 in the pathogenesis of heparin-induced thrombocytopenia. Blood. 2010;116(23):5021–5031. doi: 10.1182/blood-2010-03-276964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kasthuri RS, Glover SL, Jonas W, et al. PF4/heparin-antibody complex induces monocyte tissue factor expression and release of tissue factor positive microparticles by activation of FcγRI. Blood. 2012;119(22):5285–5293. doi: 10.1182/blood-2011-06-359430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cuker A, Ortel TL. ASH evidence-based guidelines: is the IgG-specific anti-PF4/heparin ELISA superior to the polyspecific ELISA in the laboratory diagnosis of HIT? Hematology Am Soc Hematol Educ Program. 2009:250–252. doi: 10.1182/asheducation-2009.1.250. [DOI] [PubMed] [Google Scholar]
- 20.Greinacher A, Eichler P, Lubenow N, Kwasny H, Luz M. Heparin-induced thrombocytopenia with thromboembolic complications: meta-analysis of 2 prospective trials to assess the value of parenteral treatment with lepirudin and its therapeutic aPTT range. Blood. 2000;96(3):846–851. [PubMed] [Google Scholar]
- 21.Lubenow N, Eichler P, Lietz T, Greinacher A Hit Investigators Group. Lepirudin in patients with heparin-induced thrombocytopenia: results of the third prospective study (HAT-3) and a combined analysis of HAT-1, HAT-2, and HAT-3. J Thromb Haemost. 2005;3(11):2428–2436. doi: 10.1111/j.1538-7836.2005.01623.x. [DOI] [PubMed] [Google Scholar]
- 22.Lewis BE, Wallis DE, Hursting MJ, Levine RL, Leya F. Effects of argatroban therapy, demographic variables, and platelet count on thrombotic risks in heparin-induced thrombocytopenia. Chest. 2006;129(6):1407–1416. doi: 10.1378/chest.129.6.1407. [DOI] [PubMed] [Google Scholar]
- 23.Cuker A. Heparin-induced thrombocytopenia (HIT) in 2011: an epidemic of overdiagnosis. Thromb Haemost. 2011;106(6):993–994. doi: 10.1160/TH11-09-0677. [DOI] [PubMed] [Google Scholar]
- 24.Juhl D, Eichler P, Lubenow N, Strobel U, Wessel A, Greinacher A. Incidence and clinical significance of anti-PF4/heparin antibodies of the IgG, IgM, and IgA class in 755 consecutive patient samples referred for diagnostic testing for heparin-induced thrombocytopenia. Eur J Haematol. 2006;76(5):420–426. doi: 10.1111/j.1600-0609.2005.00621.x. [DOI] [PubMed] [Google Scholar]
- 25.Pouplard C, Gueret P, Fouassier M, Ternisien C, Trossaert M, Régina S, Gruel Y. Prospective evaluation of the ‘4Ts’ score and particle gel immunoassay specific to heparin/PF4 for the diagnosis of heparin-induced thrombocytopenia. J Thromb Haemost. 2007;5(7):1373–1379. doi: 10.1111/j.1538-7836.2007.02524.x. [DOI] [PubMed] [Google Scholar]
- 26.Bakchoul T, Giptner A, Najaoui A, Bein G, Santoso S, Sachs UJ. Prospective evaluation of PF4/heparin immunoassays for the diagnosis of heparin-induced thrombocytopenia. J Thromb Haemost. 2009;7(8):1260–1265. doi: 10.1111/j.1538-7836.2009.03465.x. [DOI] [PubMed] [Google Scholar]
- 27.Whitlatch NL, Kong DF, Metjian AD, Arepally GM, Ortel TL. Validation of the high-dose heparin confirmatory step for the diagnosis of heparin-induced thrombocytopenia. Blood. 2010;116(10):1761–1766. doi: 10.1182/blood-2010-01-262659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Althaus K, Strobel U, Warkentin TE, Greinacher A. Combined use of the high heparin step and optical density to optimize diagnostic sensitivity and specificity of an anti-PF4/heparin enzyme-immunoassay. Thromb Res. 2011;128(3):256–260. doi: 10.1016/j.thromres.2011.05.003. [DOI] [PubMed] [Google Scholar]
- 29.Greinacher A, Michels I, Kiefel V, Mueller-Eckhardt C. A rapid and sensitive test for diagnosing heparin-associated thrombocytopenia. Thromb Haemost. 1991;66(6):734–736. [PubMed] [Google Scholar]
- 30.Sheridan D, Carter C, Kelton JG. A diagnostic test for heparin-induced thrombocytopenia. Blood. 1986;67(1):27–30. [PubMed] [Google Scholar]
- 31.Warkentin TE. Heparin-induced thrombocytopenia in the ICU: a transatlantic perspective. Chest. 2012;142(4):815–816. doi: 10.1378/chest.12-0979. [DOI] [PubMed] [Google Scholar]
- 32.Cuker A, Gimotty PA, Crowther MA, Warkentin TE. Predictive value of the 4Ts scoring system for heparin-induced thrombocytopenia: a systematic review and meta-analysis. Blood. 2012;120(20):4160–4167. doi: 10.1182/blood-2012-07-443051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bauer TL, Arepally G, Konkle BA, et al. Prevalence of heparin-associated antibodies without thrombosis in patients undergoing cardiopulmonary bypass surgery. Circulation. 1997;95(5):1242–1246. doi: 10.1161/01.cir.95.5.1242. [DOI] [PubMed] [Google Scholar]
- 34.Warkentin TE, Greinacher A, Gruel Y, Aster RH, Chong BH Scientific and Standardization Committee of the International Society on Thrombosis and Haemostasis. Laboratory testing for heparin-induced thrombocytopenia: a conceptual framework and implications for diagnosis. J Thromb Haemost. 2011;9(12):2498–2500. doi: 10.1111/j.1538-7836.2011.04536.x. [DOI] [PubMed] [Google Scholar]
- 35.Warkentin TE, Levine MN, Hirsh J, Horsewood P, Roberts RS, Gent M, Kelton JG. Heparin-induced thrombocytopenia in patients treated with low-molecular-weight heparin or unfractionated heparin. N Engl J Med. 1995;332(20):1330–1335. doi: 10.1056/NEJM199505183322003. [DOI] [PubMed] [Google Scholar]
- 36.Amiral J, Marfaing-Koka A, Wolf M, et al. Presence of autoantibodies to interleukin-8 or neutrophil-activating peptide-2 in patients with heparin-associated thrombocytopenia. Blood. 1996;88(2):410–416. [PubMed] [Google Scholar]