Abstract
Background and purpose:
QT and Tpeak-Tend (Te) intervals are associated with sudden cardiac death in patients with chronic heart failure (CHF). We studied age-dependent influence on short-term temporal dispersion of these two variables in patients with postischemic CHF.
Method:
We grouped 75 CHF and 53 healthy control subjects into three age subsets: ≤50 years, >50 years and ≤65 years, and >65 years. We then calculated the following indices: QT and Te variability index (QTVI and TeVI), the ratio between the short-term variability (STV) of QT or Te, and the STV of resting rate (RR) (QT/RR STV and Te/RR STV).
Results:
In all different age subgroups, patients with CHF showed a higher level of QTVI than age-matched control subjects (≤50 years: P < 0.0001; >50 years and ≤65 years: P < 0.05; >65 years: P < 0.05). Patients with CHF < 50 years old also had all repolarization variability indices higher than normal age-matched controls (TeVI, P < 0.05; QT/RR STV, P < 0.05; Te/RR STV, P < 0.05), whereas we did not find any difference between the two older classes of subjects. Both QTVI (r2: 0.178, P < 0.05) and TeVI (r2: 0.433, P < 0.001) were positively related to age in normal subjects, even if the first correlation was weaker than the second one.
Conclusion:
Our data showed that QTVI could be used in all ages to evaluate repolarization temporal liability, whereas the other indices are deeply influenced by age. Probably, the age-dependent increase in QTVI was more influenced by a reduction of RR variability reported in older normal subjects.
Keywords: aging, QT variability, heart rate variability, chronic heart failure, sudden death
Introduction
Malignant ventricular arrhythmias are common complications of chronic heart failure (CHF) induced by myocardial infarction. Senescence1,2 and CHF3,4 strongly influence the cardiovascular autonomic control and myocardial repolarization phase.5–9 Indeed, both these conditions are able to increase α-adrenoreceptor-mediated peripheral resistance1 and to reduce β-adrenergic adrenoreceptor10 and baroreflex function,10,11 with a consistent decrease of heart rate variability.12–16 The increase of temporal dispersion of the repolarization ventricular phase17–25 and the reduction of heart rate variability25–27 are risk factors for ventricular malignant arrhythmias and sudden cardiac death (SCD) and non-SCD. Aging28–30 and CHF31 are clear pathogenetic factors able to induce ventricular arrhythmogenesis. Neurohumoral activation,4,6,32 electroanatomical remodeling,33,34 chronic inflammatory condition,35 and endothelial dysfunction36–39 are well-recognized causes of ventricular arrhythmia in patients with CHF. Elderly patients with CHF show higher proneness for the malignant ventricular arrhythmias because these subjects have lower levels of functional reserve capable of resisting arrhythmogenesis. Although the influence of CHF and aging on ventricular arrhythmias is well known, the underlying pathogenic mechanisms are not yet completely understood. Particularly, the impact of aging on markers of repolarization’s temporal dispersion during CHF has not been clarified. In a recent large community-based study, an increased risk of SCD has been found related to a prolonged Tpeak-Tend interval (Te).40 In addition, in patients with CHF and SCD, a higher temporal dispersion was reported.5
All in all, we analyzed QT interval (from Q to end T wave electrocardiogram [ECG]) variability index (QTVI) and short-term variability (STV) of QT in CHF subjects with different ages, in order to individuate a possible age-dependent influence on these markers. Finally, we calculated the same indices of variability on the last part of the T wave (considering the peak and end of the T wave) to observe a peculiar age-related effect on this important part of repolarization.
Methods
Study subjects
For this study we selected 75 outpatients (63 men and 12 woman) who had stable CHF secondary to ischemic dilated cardiomyopathy, and 53 healthy control subjects (41 men and 12 women). We defined clinically stable patients as those who had not been hospitalized or had their therapy adjusted or had experienced any other acute coronary artery or noncoronary event during the past 3 months. All participants had undergone revascularization either cutaneously or by aortocoronary artery bypass at least 3 months before the study. None of the patients had malignancy, primary valve disease, atrial fibrillation, premature complexes (one premature complex per minute was permitted), or other arrhythmias likely to interfere with heart rate and QT analysis. None of the patients was New York Heart Association class IV. Before the study, none of the subjects had a documented history of cardiac arrest, ventricular tachycardia, or fibrillation. We suggested that all patients with ejection fraction ≤35% underwent an implant of implantable cardioverter/defibrillator device.
To detect possible statistical differences related to age we divided each of the two study groups into three age subsets: ≤50 years, >50 years and ≤65 years, and >65 years.
Study protocol and offline data analysis
After a 10-minute rest lying down, each subject underwent a 5-minute, single ECG lead recording during controlled breathing (15 breaths per minute, 0.25 Hz). All digitized signal recordings were analyzed by a single physician (GP) blinded to the subjects’ circumstances.
We measured the following intervals from the respective time series of ECG recordings: resting rate (RR), QT (from the Q wave to the T wave end), and Te (from the T peak to T wave end). We therefore calculated mean and variance values of each of these intervals and then used the original formula proposed by Berger et al41 to calculate three different QT variability indices:
Software for data acquisition and storage and for spectral analysis were designed and produced by our research group and are described in detail elsewhere.7–9,42–45
Finally, we used all ECG recordings to measure the STV of the aforementioned intervals. This variable was calculated following the standard method, namely by using the first 60 consecutive beats (STV60) 6,46,47 and also by using the total number of beats in the whole 5-minute recording (STVT). The formula used was:
where D was the duration of RR or QTe, or Te interval. Consequently, we were able to obtain the following six STV indices: RR STV60, QT STV60, Te STV60, RR STVT, QTe STVT, and Te STVT.
Finally, we calculated the following ratio between the different STVs:
Moreover, from the same 5-minute ECG segment, the corrected QT and Te intervals were obtained according to the formulas proposed by Bazett (QT/RR0.5; QT; Te/RR0.5), Friedericia (QT/RR0.33; Te/RR0.33), Lilly (QT/RR0.4; Te/RR0.4), and Framingham (QT+ [0.154*{1000-RR}]; Te+ [0.154*{1000–RR}]).
Statistical analysis
Unless otherwise indicated, all data are expressed as means ± standard deviation. Data with skewed distribution are given as median and interquartile range (75th percentile–25th percentile). Categorical variables were analyzed with the χ2 test. One-way analysis of variance and the Bonferroni test were used to compare data for the normally distributed variables. Kruskal–Wallis and Mann–Whitney tests were used to compare non-normally distributed variables (as evaluated by the Kolmogorov–Smirnov test). To detect possible statistical differences related to age, we divided each of the two study groups into three age subsets: ≤50 years, >50 years and ≤65 years, and ≥65 years.
Stepwise multiple regression analysis was used to determine possible relationships between the studied variables. P-values ≤ 0.05 were considered statistically significant. All data were evaluated with the database SPSS-PC+ (SPSS-PC+ Inc, Chicago, IL, USA).
Results
We examined 128 subjects, 75 with CHF and 53 healthy controls. The clinical characteristics of subjects enrolled in the study are shown in Table 1.
Table 1.
General characteristics of study sample
| Characteristics |
Chronic heart failure
|
Healthy controls
|
P values |
|---|---|---|---|
| N = 75 | N = 53 | ||
| Age, years | 64 ± 13 | 60 ± 15 | 0.452 |
| Male/female | 63/12 | 41/12 | 0.275 |
| BMI, kg/m2 | 26 ± 4 | 26 ± 3 | 0.155 |
| HR, beats/min | 66 ± 11 | 68 ± 10 | 0.329 |
| Systolic blood pressure, mmHg | 123 ± 26 | 122 ± 17 | 0.700 |
| Diastolic blood pressure, mmHg | 66 ± 15 | 75 ± 11 | 0.002 |
| Left ventricular ejection fraction, % | 40 ± 10 | 57 ± 4 | 0.0001 |
| QT, ms | 375 ± 58 | 353 ± 26 | 0.007 |
| QTBazett, ms | 389 ± 51 | 372 ± 20 | 0.017 |
| QTFridericia, ms | 384 ± 51 | 366 ± 19 | 0.008 |
| QTLilly, ms | 386 ± 51 | 361 ± 19 | 0.011 |
| QTFramingham, ms | 385 ± 51 | 368 ± 18 | 0.012 |
| NYHA class, I/II/III | 10/46/19 | 0 | 0.0001 |
| Serum K+ mmEq/L | 4.2 | 4.1 | 0.842 |
| β-blockers | 35 | 0 | 0.0001 |
| Furosemide | 33 | 0 | 0.0001 |
| ACEi/Sartans | 56 | 5 | 0.0001 |
| Spironolactone | 22 | 0 | 0.0001 |
| Digoxin | 13 | 0 | 0.0001 |
| Amiodarone | 11 | 0 | 0.017 |
Note: Data are expressed as means ± standard deviation.
Abbreviations: BMI, body mass index; HR, heart rate; NYHA, New York Heart Association; ACEi, angiotensin converting enzyme; ms, milliseconds; mmEq/L milliequivalents per liter.
Age, body mass index, and sex distribution did not differ significantly between the two groups, whereas heart rate, diastolic arterial pressure, left ventricular ejection fraction, and QT interval differed significantly (Table 1). Both subgroups had a similar mean intergroup age (Table 2).
Table 2.
Mean age of subjects in the three age-subgroups
| Age subgroups |
Chronic heart failure
|
Healthy controls
|
P values |
|---|---|---|---|
| N = 75 | N = 53 | ||
| ≤50 years | 46 ± 4 (n = 19) | 44 ± 3 (n = 17) | 0.371 |
| >50 ≤ 65 years | 58 ± 3 (n = 21) | 57 ± 4 (n = 18) | 0.342 |
| ≥65 years | 75 ± 6 (n = 35) | 77 ± 4 (n = 18) | 0.317 |
Note: Data are expressed as means ± standard deviation.
The younger CHF group showed a reduction of RR variance in comparison with the age-matched healthy controls, whereas only the older CHF group reported a longer RRmean than controls (Table 3). Only elderly subjects had a significantly larger QTmean (P < 0.05), QT variance (P < 0.05), and Temean (P < 0.05) than age-matched healthy controls. Furthermore, the middle-aged group showed a QTvariance (P < 0.05) significantly higher than age-matched controls (Table 3). In all subsets, independently from age, the CHF group showed a significantly higher QTVI (P < 0.001) than age-matched controls, but only the younger CHF group had all other QT variability values significantly higher (TeVI: P < 0.05, QT/RR STV60: P < 0.05, Te/RR STV60: P < 0.05, QT/RR STVT: P < 0.05, and Te/RR STVT: P < 0.05) in comparison with age-matched controls (Table 4). In the middle-aged and elderly CHF groups we did not find any differences for the other repolarization variables (Table 4).
Table 3.
RR interval and QT dynamics data according to age and presence of chronic heart failure
| Variables |
Group 1 (≤50 years)
|
P |
Group 2 (>50 ≤
65 years)
|
P |
Group 3 (≥65 years)
|
P | |||
|---|---|---|---|---|---|---|---|---|---|
| CHF (N = 19) | Controls (N = 17) | CHF (N = 21) | Controls (N = 18) | CHF (N = 35) | Controls (N = 18) | ||||
| RR mean, ms | 899 ± 154 | 945 ± 142 | 0.354 | 918 ± 140 | 906 ± 127 | 0.785 | 966 (164) | 873 (126) | 0.040 |
| RR variance, ms2 | 574 (1124) | 1627 (2244)*,# | 0.0001 | 805 ± 1309 | 885 ± 990§ | 0.349 | 611 (820) | 580 (1340) | 0.992 |
| QT mean, ms | 363 ± 42 | 359 ± 30 | 0.801 | 371 ± 57 | 355 ± 31 | 0.294 | 385 ± 66 | 347 ± 18 | 0.008 |
| QT variance, ms2 | 36 (84) | 18 (14) | 0.066 | 43 (76) | 19 (10) | 0.011 | 38 (67) | 14 (52) | 0.004 |
| Te mean, ms | 85 ± 29 | 86 ± 13# | 0.847 | 81 ± 18 | 87 ± 22† | 0.402 | 91 ± 43 | 70 ± 13 | 0.019 |
| Te variance, ms2 | 23 (80) | 20 (16)# | 0.397 | 38 (85) | 17 (33)† | 0.349 | 33 (80) | 39 (35) | 0.696 |
Notes: Data are expressed as median and interquartile range (75th–25th).
P < 0.05 Group 1 vs Group 2;
P < 0.05 Group 1 vs Group 3;
P < 0.05 Group 2 vs Group 3;
P < 0.001 Group 2 vs Group 3.
Abbreviations: CHF, chronic heart failure; TeVI, Tpeak-Tend variability index; RR mean or variance, waves interval; ms, milliseconds; QT interval mean or variance.
Table 4.
QT variability indices according to age and presence of chronic heart failure
| Variables |
Group 1 (≤50 years)
|
P |
Group 2 (>50 ≤
65 years)
|
P |
Group 3 (≥65 years)
|
P | |||
|---|---|---|---|---|---|---|---|---|---|
| CHF (N = 19) | Controls (N = 17) | CHF (N = 21) | Controls (N = 18) | CHF (N = 35) | Controls (N = 18) | ||||
| QTVI | −0.40 (1.00) | −1.22 (0.35)*,# | 0.0001 | −0.54 (0.88) | −1.09 (0.28)† | 0.001 | −0.38 (0.90) | −0.77 (0.62) | 0.033 |
| TeVI | 0.96 (1.00) | 0.20 (0.66)¥ | 0.003 | 0.72 (1.02) | 0.47 (0.69)§ | 0.133 | 0.77 (1.38) | 1.12 (0.72) | 0.551 |
| QT/RR STV60 | 0.49 (0.58) | 0.17 (0.63)# | 0.002 | 0. 38 (0.75) | 0.27 (0.46) | 0.438 | 0.53 (0.70) | 0.37 (0.51) | 0.393 |
| Te/RR STV60 | 0.54 (1.28) | 0.26 (0.28)# | 0.030 | 0.31 (0.65) | 0.33 (0.52) | 0.982 | 0.48 (0.81) | 0.58 (0.79) | 0.794 |
| QT/RR STVT | 0.59 (0.38) | 0.18 (0.20)¥ | 0.001 | 0.40 (0.33) | 0.31 (0.21)† | 0.256 | 0.44 (0.62) | 0.47 (0.71) | 0.460 |
| Te/RR STVT | 0.53 (1.09) | 0.25 (0.26)¥ | 0.022 | 0.37 (0.36) | 0.33 (0.34)† | 0.707 | 0.46 (0.76) | 0.81 (1.11) | 0.223 |
Notes: Data are expressed as median and interquartile range (75th–25th).
P < 0.05 Group 1 vs Group 2;
P < 0.05 Group 1 vs Group 3;
P < 0.001 Group 1 vs Group 3;
P < 0.05 Group 2 vs Group 3;
P < 0.001 Group 2 vs Group 3.
Abbreviations: QTVI, QT variability index; TeVI, Tpeak-Tend variability index; STV, short term variability.
We did not find any difference between the three CHF groups in respect of the RR and QT variables (Tables 3 and 4). RRvariance (P < 0.001) was significantly higher in younger and middle-aged control subjects than older subjects (Table 3), and this variable was also significantly higher in younger controls compared with healthy middle-aged subjects. Te (P < 0.05) was higher in younger and middle-aged control subjects than older subjects (Table 3). On the other hand, Tevariance (P < 0.05) was higher in the two younger groups than the older normal subjects.
In the control group, all temporal dispersion variables (QTVI, TeVI, QT/RR STV60, Te/RR STV60, QT/RR STVT, Te/RR STVT) were significantly lower in the younger group in comparison with the older subjects (P < 0.05) (Table 4). QTVI, TeVI, QT/RR STVT, and Te/RR STVT were lower in the middle-aged subjects than in the older ones. Only QTVI results were significantly lower in the younger control group compared with the middle-aged group (Table 4).
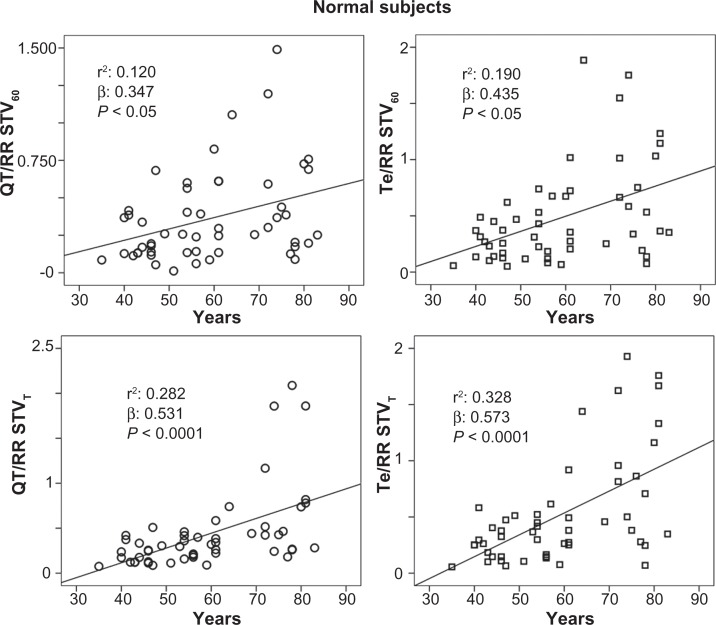
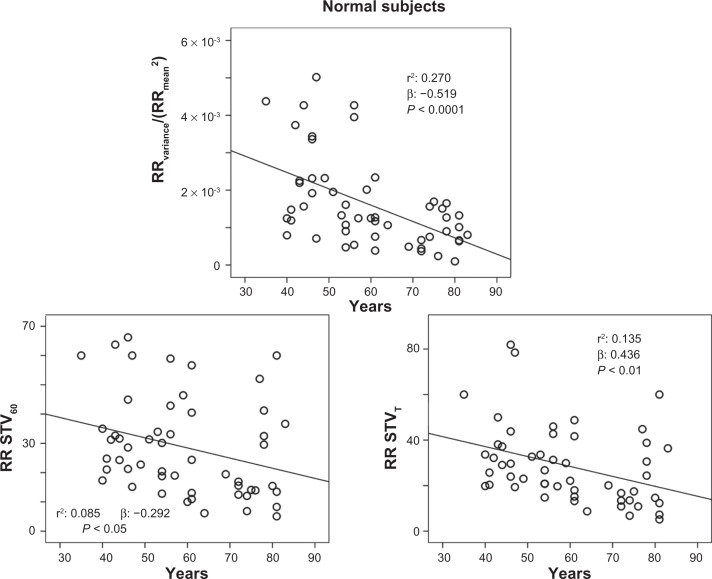
The stepwise multiple regression analysis found a significant relation with QTVI and ejection fraction (R: 0.364; R2: 0.132, β: −0.364, P: 0.001) in CHF subjects. Instead, QTVI (P < 0.05), TeVI (P < 0.001), QT/RR STV60 (P < 0.05), Te/RR STV60 (P < 0.05), QT/RR STVT (P < 0.001), and Te/RR STVT (P < 0.001) showed a significant positive relation with aging in control groups (Figures 1 and 2). We also found a negative significant correlation between all measures of RR variability and age (Figure 3) in the same subjects. Except for Tevariance/(Tmean2) (Figure 4), we did not find any significant correlation between age and repolarization variables without RR normalization in the control group.
Figure 1.
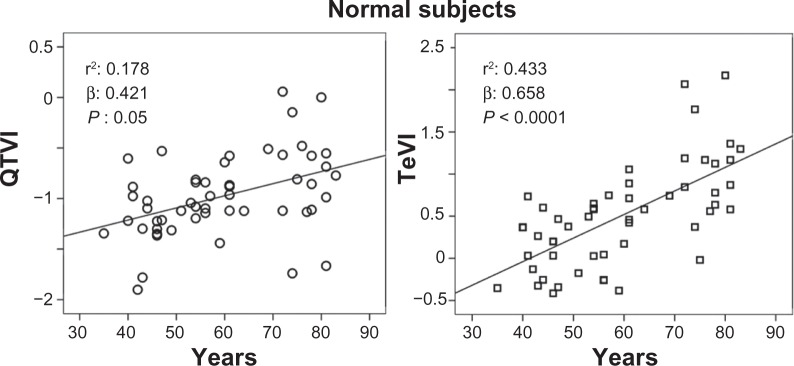
Relationship between QT variability index (QTVI) or Tpeak-Tend variability index (TeVI) and age in healthy control subjects.
Abbreviation: QT, from the Q wave to the T wave end.
Figure 2.
Relationship between age and ratio of short-term QT or Tpeak-Tend (Te) variability (STV) and resting rate (RR) variability indices, calculated on 60 consecutive beats (QT/RR STV60, Te/RRv STV60) or on 5-minute electrocardiogram recordings (QT/RR STVT, Te/RRv STVT) in healthy control subjects.
Abbreviations: QT, from the Q wave to the T wave end; v, variance.
Figure 3.
Relationship between age and the resting rate (RR) variability indices (ratio denominator of QTVI, TeVi, and short-term variability [STV], see Methods section), calculated on 5-minute electrocardiogram recordings (RRvariance/RRmean2 and RR STVT) or on 60 consecutive QRSs (RR STV60) in healthy control subjects.
Abbreviations: TeVI, Tend Variability index; Q-T, waves interval.
Figure 4.
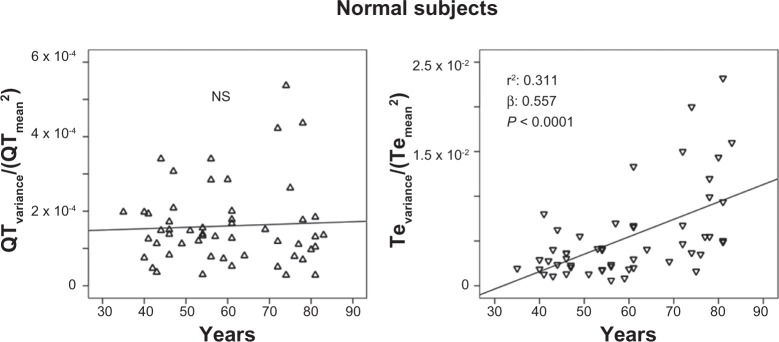
Relationship between age and Te variance/Te mean2 (ratio numerator of Tpeak-Tend variability index, see Methods section).
Notes: No significant relation was reported between age and QTvariance/QTmean2 (ratio numerator of TeVI, see Methods section) in healthy control subjects. Abbreviations: TeVI, Tend Variability index; Q-T, waves interval.
Discussion
Our main and original finding was that only QTVI values remained significantly higher in patients with CHF regardless of their age, whereas all other indices of temporal myocardial repolarization dispersion were significantly higher only in those patients with CHF belonging to the youngest category, namely those characterized with an age ≤50 years. Our second, somewhat confirmatory, finding was the significant positive correlation between QTVI and left ventricular ejection fraction in patients with CHF,10 whereas, in healthy control subjects, all repolarization variability parameters showed a significant relation only with age. Thus, although QTVI suffers a certain age influence, this index seems to be the only useful variable to measure temporal dispersion of myocardial repolarization independently from age of the patients with CHF.
An increase in temporal myocardial repolarization dispersion, as assessed by several QT interval-derived indices, has been proven to be significantly associated with an increased SCD risk in patients with structural heart disease.17–21,41–44 However, it is well known that, particularly in patients with CHF, aging represents an additional feature able to magnify this risk, most likely through a further increase in myocardial repolarization lability. Indeed, aging typically leads to a prolongation of action potential duration due to different mechanisms, such as an increased number and overactivity of cardiac L-type Ca2+ channels with a consequent slow inactivation of calcium influx and a reduction in outward potassium current.48,49 As a result, with aging, the QT length and variability tends to increase. Nonetheless, it should be emphasized that CHF leads to a prolongation and dispersion of action potential duration that is quantitatively more pronounced in respect of pure aging. Specifically, functional downregulation of potassium current and an alteration in depolarizing sodium and calcium currents are responsible for electrical remodeling, with a consequent increase in QT temporal dispersion in CHF.50 Our data, besides confirming that QT variability-derived indices are significantly higher in patients with CHF compared with healthy controls, suggest that aging might represent a confounding factor for a number of them but not for QTVI. Initially, our data also suggested through which mechanisms QTVI remains a solid marker of myocardial repolarization lability despite aging. Indeed, in patients with CHF aged ≤50 years, QTVI suffers because of extremely low RR variance values, whereas, considering those patients with CHF aged >50 years, QTVI was much worse, as indicated by a marked increase in QT variance. Thus, it could be hypothesized that in relatively young patients with CHF, an increased QTVI might mirror a sinus node dysfunction due to a prevalent autonomic nervous system control derangement, whereas, with aging, QTVI mainly reflects an altered myocardial repolarization phase. A possible reason underlying a major role of sinus node dysfunction in worsening QTVI of young patients with CHF could be represented by the well-known age-related decline in heart rate variability.11–16
Evidence indicates that an increased Te interval40 is related to SCD, and also recent research from our group showed an increased temporal dispersion of the last part of repolarization (TeVI) in patients with CHF5 and SCD, as well as in an animal model with pacing-induced heart failure (TeVI, Te/STVT).6 However, the pathophysiologic meaning of this ECG interval still remains controversial. Some authors believe that Te is a reliable noninvasive marker of transmural repolarization gradient, whereas others affirm that this period reflects the total spatial ventricular repolarization dispersion.41,46,51 Notably, clinical and experimental evidence indicates that Te should be considered as a marker of Iks function, especially during sympathetic activation.52,53 Indeed, an enhancement in IKs was reported after a β-adrenergic stimulation, the latter leading to an elevation of intracellular cyclic adenosine monophosphate (cAMP) and an activation of protein kinase A.54 Supporting this datum, it was reported that subjects with concealed type 1 long QT reported a significant increase of Te interval during an infusion of epinephrine,55 again favoring a close link between adrenergic burst and IKs activity. Our current data seem to identify a possible role of Te-derived indices only when considering those patients with CHF aged <50 years. However, a real comparison is impossible given the different population enrolled and the lack of a follow-up. A possible explanation, albeit merely speculative, could be that aging per se, due to a downregulation of β-adrenoreceptor expression and function, could affect Te-derived indices, leading to an age-related loss of their diagnostic utility in patients with CHF. Accordingly, we can hypothesize that, between all indices of myocardial repolarization dispersion, QTVI might remain the only one that could be used to point out an increased SCD risk regardless of patients’ age, whereas Te indices should be useful just in middle-aged patients.
Conclusion
Our data demonstrate a deep influence of age on the short-term variability of repolarization phases, and tend to suggest that only QTVI is able to characterize temporal myocardial repolarization lability in patients with CHF regardless of aging.
Footnotes
Disclosure
The authors report no conflicts of interest in this work.
References
- 1.Hotta H, Uchida S. Aging of the autonomic nervous system and possible improvements in autonomic activity using somatic afferent stimulation. Geriatr Gerontol Int. 2010;10:S127–S136. doi: 10.1111/j.1447-0594.2010.00592.x. [DOI] [PubMed] [Google Scholar]
- 2.Kaye DM, Esler MD. Autonomic control of the aging heart. Neuro Molecular Medicine. 2008;10:179–186. doi: 10.1007/s12017-008-8034-1. [DOI] [PubMed] [Google Scholar]
- 3.Kishi T. Heart failure as an autonomic nervous system dysfunction. J Cardiol. 2012;59:117–122. doi: 10.1016/j.jjcc.2011.12.006. [DOI] [PubMed] [Google Scholar]
- 4.Piccirillo G, Ogawa M, Song J, Chong VJ, Joung B, Han S, et al. Power spectral analysis of heart rate variability and autonomic nervous system activity measured directly in healthy dogs and dogs with tachycardia-induced heart failure. Heart Rhythm. 2009;6:546–552. doi: 10.1016/j.hrthm.2009.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Piccirillo G, Rossi P, Mitra M, et al. Indexes of temporal myocardial repolarization dispersion and sudden cardiac death in heart failure: any difference? Ann Noninv Electrocardiol. 2012 doi: 10.1111/anec.12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Piccirillo G, Magrì D, Pappadà MA, Maruotti A, Ogawa M, Han S, et al. Autonomic nerve activity and the short term variability of Tpeak-Tend interval in dogs with pacing-induced heart failure. Heart Rhythm. 2012;9:2044–2050. doi: 10.1016/j.hrthm.2012.08.030. [DOI] [PubMed] [Google Scholar]
- 7.Piccirillo G, Magrì D, Ogawa M, Song J, Chong VJ, Han S, et al. Autonomic nervous system activity measured directly and QT interval variability in normal and pacing-induced tachycardia heart failure dogs. J Am Coll Cardiol. 2009;54:840–850. doi: 10.1016/j.jacc.2009.06.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Piccirillo G, Cacciafesta M, Lionetti M, Nocco M, Di Giuseppe V, Moisè A, et al. The influence of age, the autonomic nervous system and anxiety on QT interval variability. Clin Sci. 2001;101:429–438. [PubMed] [Google Scholar]
- 9.Piccirillo G, Magnanti M, Matera S, Di Carlo S, De Laurentis T, Torrini A, et al. Age and QT variability index during free breathing, controlled breathing and tilt in patients with chronic heart failure and healthy control subjects. Transl Res. 2006;148:72–78. doi: 10.1016/j.trsl.2006.02.001. [DOI] [PubMed] [Google Scholar]
- 10.Janczewski AJ, Lakatta EG. Modulation of sarcoplasmic reticulum Ca2+ cycling in systolic and diastolic heart failure associated with aging. Heart Fail Rev. 2010;15:431–445. doi: 10.1007/s10741-010-9167-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Piccirillo G, Cacciafesta M, Viola E, et al. Influence of aging on cardiac baroreflex sensitivity determined noninvasively by power spectral analysis. Clin Sci. 2001;100:267–274. [PubMed] [Google Scholar]
- 12.Piccirillo G, Di Giuseppe V, Nocco M, Lionetti M, Naso C, Tallarico D, et al. Influence of aging and other cardiovascular risk factors on baroreflex sensitivity. J Am Geriatr Soc. 2001;49:1059–1065. doi: 10.1046/j.1532-5415.2001.49209.x. [DOI] [PubMed] [Google Scholar]
- 13.Piccirillo G, Fimognari FL, Viola E, Marigliano V. Age-adjusted normal confidence intervals for heart rate variability in healthy subjects during head-up tilt. Int J Cardiol. 1995;50:117–124. doi: 10.1016/0167-5273(95)93680-q. [DOI] [PubMed] [Google Scholar]
- 14.Colosimo A, Giuliani A, Mancini AM, Piccirillo G, Marigliano V. Estimating a cardiac age by means of heart rate variability. Am J Physiol. 1997;273:H1841–H1847. doi: 10.1152/ajpheart.1997.273.4.H1841. [DOI] [PubMed] [Google Scholar]
- 15.Piccirillo G, Bucca C, Bauco C, Cinti AM, Michele D, Fimognari FL, et al. Power spectral analysis of heart rate in subjects over a hundred years old. Int J Cardiol. 1998;63:53–61. doi: 10.1016/s0167-5273(97)00282-9. [DOI] [PubMed] [Google Scholar]
- 16.Piccirillo G, Magrì D, Naso C, di Carlo S, Moisè A, De Laurentis T, et al. Factors influencing heart rate variability power spectral analysis during controlled breathing in patients with chronic heart failure or hypertension and in healthy normotensive subjects. Clin Sci (Lond) 2004;107:183–190. doi: 10.1042/CS20030401. [DOI] [PubMed] [Google Scholar]
- 17.Tereshchenko LG, Cygankiewicz I, McNitt S, Vazquez R, Bayes-Genis A, Han L, et al. Predictive value of beat-to-beat QT variability index across the continuum of left ventricular dysfunction: competing risks of noncardiac or cardiovascular death and sudden or nonsudden cardiac death. Circ Arrhythm Electrophysiol. 2012;5:719–727. doi: 10.1161/CIRCEP.112.970541. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dobson CP, La Rovere MT, Pinna GD, Goldstein R, Olsen C, Bernardinangeli M, et al. QT variability index on 24-hour Holter independently predicts mortality in patients with heart failure: analysis of GISSI-HF trial data. Heart Rhythm. 2011;8:1237–1242. doi: 10.1016/j.hrthm.2011.03.055. [DOI] [PubMed] [Google Scholar]
- 19.Dobson CP, La Rovere MT, Olsen C, Berardinangeli M, Veniani M, Midi P, et al. 24-hour QT variability in heart failure. J Electrocardiol. 2009;42:500–504. doi: 10.1016/j.jelectrocard.2009.06.021. [DOI] [PubMed] [Google Scholar]
- 20.Haigney MC, Zareba W, Nasir JM, McNitt S, McAdams D, Gentlesk PJ, et al. MADIT II investigators Gender differences and risk of ventricular tachycardia or ventricular fibrillation. Heart Rhythm. 2009;6:180–186. doi: 10.1016/j.hrthm.2008.10.045. [DOI] [PubMed] [Google Scholar]
- 21.Piccirillo G, Magrì D, Matera S, Magnanti M, Torrini A, Pasquazzi E, et al. QT variability strongly predicts sudden cardiac death in asymptomatic subjects with mild or moderate left ventricular systolic dysfunction: a prospective study. Eur Heart J. 2007;28:1344–1350. doi: 10.1093/eurheartj/ehl367. [DOI] [PubMed] [Google Scholar]
- 22.Haigney MC, Zareba W, Gentlesk PJ, Goldstein RE, Illovsky M, McNitt S, et al. Multicenter Automatic Defibrillator Implantation Trial II investigators QT interval variability and spontaneous ventricular tachycardia or fibrillation in the Multicenter Automatic Defibrillator Implantation Trial [MADIT] II. J Am Coll Cardiol. 2004;44:1481–1487. doi: 10.1016/j.jacc.2004.06.063. [DOI] [PubMed] [Google Scholar]
- 23.Atiga WL, Calkins H, Lawrence JH, Tomaselli GF, Smith JM, Berger RD. Beat-to-beat repolarization lability identifies patients at risk for sudden cardiac death. J Cardiovasc Electrophysiol. 1998;9:899–908. doi: 10.1111/j.1540-8167.1998.tb00130.x. [DOI] [PubMed] [Google Scholar]
- 24.Piccirillo G, Magrì D, Matera S, Magnanti M, Pasquazzi E, Schifano E, et al. Effects of pink grapefruit juice on QT variability in patients with dilated or hypertensive cardiomyopathy and in healthy subjects. Transl Res. 2008;151:267–272. doi: 10.1016/j.trsl.2008.03.002. [DOI] [PubMed] [Google Scholar]
- 25.Piccirillo G, Magrì D, di Carlo S, De Laurentis T, Torrini A, Matera S, et al. Influence of cardiac-resynchronization therapy on heart rate and blood pressure variability: 1-year follow-up. Eur J Heart Fail. 2006;8:716–722. doi: 10.1016/j.ejheart.2006.01.008. [DOI] [PubMed] [Google Scholar]
- 26.Huikuri HV, Stein PK. Clinical application of heart rate variability after acute myocardial infarction. Front Physiol. 2012;3:41. doi: 10.3389/fphys.2012.00041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Thayer JF, Yamamoto SS, Brosschot JF. The relationship of autonomic imbalance, heart rate variability and cardiovascular disease risk factors. Int J Cardiol. 2010;141:122–131. doi: 10.1016/j.ijcard.2009.09.543. [DOI] [PubMed] [Google Scholar]
- 28.Cooper LL, Odening KE, Hwang MS, Chaves L, Schofield L, Taylor CA, et al. Electromechanical and structural alterations in the aging rabbit heart and aorta. Am J Physiol Heart Circ Physiol. 2012;302:H1625–H1635. doi: 10.1152/ajpheart.00960.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Bapat A, Nguyen TP, Lee JH, Sovari AA, Fishbein MC, Weiss JN, Karagueuzian HS. Enhanced sensitivity of aged fibrotic hearts to angiotensin II- and hypokalemia-induced early after depolarization-mediated ventricular arrhythmias. Am J Physiol Heart Circ Physiol. 2012;302:H2331–H2340. doi: 10.1152/ajpheart.00094.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Morita N, Sovari AA, Xie Y, Fishbein MC, Mandel WJ, Garfinkel A, et al. Increased susceptibility of aged hearts to ventricular fibrillation during oxidative stress. Am J Physiol Heart Circ Physiol. 2009;297:H1594–H1605. doi: 10.1152/ajpheart.00579.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Aiba T, Tomaselli GF. Electrical remodelling of the failing heart. Curr Opin Cardiol. 2010;25:29–36. doi: 10.1097/HCO.0b013e328333d3d6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Zucker IH, Patel KP, Schultz HD. Neurohumoral stimulation. Heart Fail Clin. 2012;8:87–99. doi: 10.1016/j.hfc.2011.08.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Han S, Kobayashi K, Joung B, Piccirillo G, Maruyama M, Vinters HV, et al. Electroanatomic remodeling of the left stellate ganglion after myocardial infarction. J Am Coll Cardiol. 2012;59:954–961. doi: 10.1016/j.jacc.2011.11.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Ajijola OA, Wisco JJ, Lambert HW, Mahajan A, Stark E, Fishbein MC, Shivkumar K. Extracardiac neural remodeling in humans with cardiomyopathy. Circ Arrhythm Electrophysiol. 2012;5:1010–1116. doi: 10.1161/CIRCEP.112.972836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Rossi P, Ricci A, De Paulis R, Papi E, Pavaci H, Porcelli D, et al. Epicardial ganglionated plexus stimulation decreases post-operative inflammatory response in humans. Heart Rhythm. 2012;9:943–950. doi: 10.1016/j.hrthm.2012.01.025. [DOI] [PubMed] [Google Scholar]
- 36.Brack KE, Coote JH, Ng GA. Vagus nerve stimulation protects against ventricular fibrillation independent of muscarinic receptor activation. Cardiovasc Res. 2011;91:437–446. doi: 10.1093/cvr/cvr105. [DOI] [PubMed] [Google Scholar]
- 37.Hassanabad ZF, Furman BL, Parratt JR, Aughey E. Coronary endothelial dysfunction increases the severity of ischaemia-induced ventricular arrhythmias in rat isolated perfused hearts. Basic Res Cardiol. 1998;93:241–249. doi: 10.1007/s003950050091. [DOI] [PubMed] [Google Scholar]
- 38.Piccirillo G, Quaglione R, Fimognari F, Moisè A, Mario M, Lionetti M, et al. Influence of L-arginine and vitamin C on the autonomic nervous system in chronic heart failure secondary to ischemic cardiomyopathy. Am J Cardiol. 2004;93:650–654. doi: 10.1016/j.amjcard.2003.11.043. [DOI] [PubMed] [Google Scholar]
- 39.Piccirillo G, Nocco M, Moisè A, et al. Influence of vitamin C on baroreflex sensitivity in chronic heart failure. Hypertension. 2003;41:1240–1245. doi: 10.1161/01.HYP.0000073581.74107.22. [DOI] [PubMed] [Google Scholar]
- 40.Panikkath R, Reinier K, Uy-Evanado A, Teodorescu C, Hattenhauer J, Mariani R, et al. Prolonged Tpeak-to-tend interval on the resting ECG is associated with increased risk of sudden cardiac death. Circ Arrhythm Electrophysiol. 2011;4:441–447. doi: 10.1161/CIRCEP.110.960658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Berger RD, Kasper EK, Baughman KL, Marban E, Calkins H, Tomaselli GF. Beat-to-beat QT interval variability. Novel evidence for repolarization lability in ischemic and nonischemic dilated cardiomyopathy. Circulation. 1997;96:1557–1565. doi: 10.1161/01.cir.96.5.1557. [DOI] [PubMed] [Google Scholar]
- 42.Magrì D, Piccirillo G, Bucci E, Pignatelli G, Cauti FM, Morino S, et al. Increased temporal dispersion of myocardial repolarization in myotonic dystrophy type 1: beyond the cardiac conduction system. Int J Cardiol. 2012;156:259–264. doi: 10.1016/j.ijcard.2010.10.132. 3. [DOI] [PubMed] [Google Scholar]
- 43.Piccirillo G, Magrì D, Mitra M, Rufa A, Zicari E, Stromillo ML, et al. Increased QT variability in cerebral autosomal dominant arteriopathy with subcortical infarcts and leukoencephalopathy. Eur J Neurol. 2008;15:1216–1221. doi: 10.1111/j.1468-1331.2008.02300.x. [DOI] [PubMed] [Google Scholar]
- 44.Magrì D, Sciomer S, Fedele F, Gualdi G, Casciani E, Pugliese P, et al. Increased QT variability in young asymptomatic patients with beta-thalassemia major. Eur J Haematol. 2007;79:322–329. doi: 10.1111/j.1600-0609.2007.00921.x. [DOI] [PubMed] [Google Scholar]
- 45.Magrì D, Piccirillo G, Quaglione R, Dell’armi A, Mitra M, Velitti S, et al. Effect of acute mental stress on heart rate and QT variability in postmyocardial infarction patients. ISRN Cardiol. 2012:912672. doi: 10.5402/2012/912672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Thomsen MB, Oros A, Schoenmakers M, et al. Proarrhythmic electrical remodelling is associated with increased beat-to-beat variability of repolarization. Cardiovasc Res. 2007;73:521–530. doi: 10.1016/j.cardiores.2006.11.025. [DOI] [PubMed] [Google Scholar]
- 47.Oosterhoff P, Tereshchenko LG, van der Heyden MA, et al. Short-term variability of repolarization predicts ventricular tachycardia and sudden cardiac death in patients with structural heart disease: a comparison with QT variability index. Heart Rhythm. 2011;8:1584–1590. doi: 10.1016/j.hrthm.2011.04.033. [DOI] [PubMed] [Google Scholar]
- 48.Janczewski AM, Lakatta EG. Modulation of sarcoplasmic reticulum Ca2+ in systolic and diastolic heart failure. Heart Rev. 2010;15:431–445. doi: 10.1007/s10741-010-9167-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ocorr K, Reeves NL, Wessells RJ, Fink M, Chen HS, Akasaka T, et al. KCNQ potassium channel mutations cause cardiac arrhythmias in Drosophila that mimic the effects of aging. PNAS. 2007;104:3943–3948. doi: 10.1073/pnas.0609278104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Aiba T, Tomaselli GF. Electrical remodeling in the failing heart. Curr Opin Cardiol. 2010;25:26–36. doi: 10.1097/HCO.0b013e328333d3d6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Patel C, Burke JF, Patel H, et al. Is there a significant transmural gradient in repolarization time in the intact heart? Cellular basis of the T wave: a century of controversy. Circ Arrhythm Electrophysiol. 2009;2:80–88. doi: 10.1161/CIRCEP.108.791830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Xia Y, Liang Y, Kongstad O, et al. In vivo validation of the coincidence of the peak and end of the T wave with full repolarization of the epicardium and endocardium in swine. Heart Rhythm. 2005;2:162–169. doi: 10.1016/j.hrthm.2004.11.011. [DOI] [PubMed] [Google Scholar]
- 53.Izumi D, Chinushi M, Iijima K, et al. The peak-to-end of the T wave in the limb ECG leads reflects total spatial rather than transmural dispersion of ventricular repolarization in an anthopleurin-A model of prolonged QT interval. Heart Rhythm. 2012;9:796–803. doi: 10.1016/j.hrthm.2011.11.046. [DOI] [PubMed] [Google Scholar]
- 54.Cheng JH, Kodama I. Two components of delayed rectifier K+ current in heart: molecular basis, functional diversity, and contribution to repolarization. Acta Pharmacol Sin. 2004;25:137–145. [PubMed] [Google Scholar]
- 55.Shimizu W, Noda T, Takaki H, Kurita T, Nagaya N, Satomi K, et al. Epinephrine unmasks latent mutation carriers with LQT1 form of congenital long-QT syndrome. J Am Coll Cardiol. 2003;41:633–642. doi: 10.1016/s0735-1097(02)02850-4. [DOI] [PubMed] [Google Scholar]