Abstract
The asymmetric unit of the title compound, C30H48O2, contains two independent molecules, the main difference between them being that the isopropenyl group is rotated by approximately 180°. In each molecule, the fused six-membered rings have chair–chair–chair–chair conformations and the cyclopentane ring adopts an envelope conformation with the C atom bearing the hydroxymethyl group as the flap. All ring junctions are trans-fused. With the exception of one of the methyl groups adjacent to the C=O group, all the methyl groups are in axial positions. The isopropenyl group is equatorial and the hydroxymethyl group is in an axial orientation. In the crystal, weak C—H⋯O interactions link the molecules into chains along [010]. Weak intramolecular C—H⋯O hydrogen bonds are also observed but the hydroxy groups are not involved in hydrogen bonds.
Related literature
For the synthesis of betulone, see: Hase et al. (1981 ▶). For the isolation of betulone from plants, see: Cole et al. (1991 ▶); Reyes et al. (2006 ▶); Diouf et al. (2009 ▶); Liu et al. (2010 ▶); Kim et al. (2002 ▶); Garcez et al. (2003 ▶); Fuchino et al. (1996 ▶). For the biological activity of betulone, see: Alakurtti et al. (2010 ▶); Hata et al. (2002 ▶); Reyes et al. (2006 ▶). For related structures, see: Mohamed et al. (2006 ▶); Ding et al. (2009 ▶); Drebushchak et al. (2010 ▶); Boryczka et al. (2011 ▶, 2012a
▶,b
▶).
Experimental
Crystal data
C30H48O2
M r = 440.71
Orthorhombic,

a = 9.4447 (3) Å
b = 19.1818 (6) Å
c = 28.1141 (7) Å
V = 5093.3 (3) Å3
Z = 8
Mo Kα radiation
μ = 0.07 mm−1
T = 100 K
0.60 × 0.56 × 0.20 mm
Data collection
Oxford Diffraction diffractometer with a Sapphire3 detector
Absorption correction: multi-scan (CrysAlis RED; Oxford Diffraction, 2008 ▶) T min = 0.960, T max = 0.986
61473 measured reflections
5036 independent reflections
4401 reflections with I > 2σ(I)
R int = 0.055
Refinement
R[F 2 > 2σ(F 2)] = 0.051
wR(F 2) = 0.141
S = 1.03
5036 reflections
609 parameters
H atoms treated by a mixture of independent and constrained refinement
Δρmax = 0.55 e Å−3
Δρmin = −0.50 e Å−3
Data collection: CrysAlis CCD (Oxford Diffraction, 2008 ▶); cell refinement: CrysAlis RED (Oxford Diffraction, 2008 ▶); data reduction: CrysAlis RED; program(s) used to solve structure: SHELXS97 (Sheldrick, 2008 ▶); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008 ▶); molecular graphics: Mercury (Macrae et al., 2006 ▶); software used to prepare material for publication: publCIF (Westrip, 2010 ▶).
Supplementary Material
Crystal structure: contains datablock(s) I, global. DOI: 10.1107/S1600536813011008/lh5603sup1.cif
Structure factors: contains datablock(s) I. DOI: 10.1107/S1600536813011008/lh5603Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Table 1. Hydrogen-bond geometry (Å, °).
| D—H⋯A | D—H | H⋯A | D⋯A | D—H⋯A |
|---|---|---|---|---|
| C13A—H13A⋯O2A | 1.04 (4) | 2.52 (3) | 3.186 (4) | 122 (2) |
| C13B—H13B⋯O2B | 1.02 (3) | 2.47 (3) | 3.165 (4) | 125 (2) |
| C19A—H19A⋯O2A | 0.95 (4) | 2.45 (4) | 3.006 (5) | 118 (3) |
| C22B—H22C⋯O1B i | 1.05 (4) | 2.56 (4) | 3.567 (4) | 160 (3) |
Symmetry code: (i)  .
.
Acknowledgments
This work was supported by the Medical University of Silesia in Katowice, Poland (grant No. KNW-1–006/P/2/0).
supplementary crystallographic information
Comment
Betulone (lup-20 (29)-en-28-ol-3-one) also known as betulonic alcohol, is a pentacyclic triterpene of the lupane type which was first isolated as a natural product from Betula lenta in 1991 (Cole et al., 1991). Betulone can be also isolated from various plants for example Maytenus cuzcoina and Maytenus chiapensis (Reyes et al., 2006), Betula alleghaniens (Diouf et al., 2009), Excoecaria agallocha (Liu et al., 2010), Ilex macropoda (Kim et al., 2002) and Terminalia glabrescens (Garcez et al., 2003). The continually growing interest in betulone and its derivatives results from their wide spectrum of biological activities such as anti-inflammatory (Reyes et al., 2006), anti-leishmanial (Alakurtti et al., 2010) and anticancer (Hata et al., 2002).
The structure of betulone is based on the 30-carbon skeleton comprising of four 6-membered rings and one cyclopentane ring. It has three available sites for simple chemical modification, namely: keto group at C3, primary hydroxy group at C28 and isopropenyl side chain at C19. These groups and their positions, mutual distances and orientation with respect to the rings can influence hydrogen bonding and the interactions of betulone with other active sites of surrounding species.
Betulone is also known as a derivative of betulin, which is one of the most plentiful triterpenes comprising up to 30% dry weight of the outer bark of the white birch. In comparison to betulin, the content of betulone in the outer bark of different tree species is very low, e.g. about of 0.03% in Betula platyphylla (Fuchino et al., 1996) and for this reason the isolation from raw plant material is poorly profitable. A more effective method to obtain betulone with high yield is to carry out synthesis from betulin (Hase et al., 1981). The crystal structure of betulone has not been reported until now. However, the crystal structures of betulonic acid-DMSO and betulonic acid-DMF solvates (Boryczka et al., 2012b) were earlier described. In addition, the structure of betulinic acid-DMSO solvate (Boryczka et al., 2012a) has also been reported. In the present work, we describe the crystal structure of betulone in order to gain a better understanding of the structure-activity relationships of this important molecule. Betulonic alcohol was obtained by oxidation of naturally occurring betulin in a one-step reaction utilizing Jones-oxidation (CrO3/H2SO4 in acetone-water solution) as the side product.
The asymmetric unit contains two independent molecules (IA and IB). A schematic drawing of the ring and atom labeling is shown in Fig. 1 and the asymmetric unit is shown in Fig. 2. All bond lengths and the angles show normal values. The cyclopentane ring adopts an envelope conformation and the cyclohexane rings are all in chair conformations. All the ring junctions in the lupane nucleus are trans-fused. A similar ring conformation is also observed in 20 (29)-lupene-3β,28β-diacetate (Mohamed et al., 2006), 3,28-diacetoxy-29-bromo-betulin (Ding et al., 2009) and also in betulin-ethanol (Drebushchak et al., 2010), betulin-DMSO (Boryczka et al., 2011), betulinic acid-DMSO (Boryczka et al., 2012a) solvates. However, the conformation of ring A in betulone differs significantly from that observed earlier for betulonic acid-DMSO and DMF solvate, where ring A adopts a boat conformation. The molecules are packed along the a axis, in a zigzag fashion, parallel to the bc plane (Fig. 3). The cyclopentane ring is in an envelope conformation with the C17 atom being displaced from C18/C19/C21/C22 plane by 0.656 (4) Å (IA) and 0.674 (4) Å (IB). The C17—C18—C19—C21 and C19—C21—C22—C17 torsion angles are 27.0 (4)°, -25.4 (4)° (for IA) and 27.5 (3)°, -26.1 (3)° (for IB) respectively. The methyl groups C24, C25, C26, C27 occupy axial positions, but the methyl group C23 and isopropenyl group at C19 are equatorial. Fig. 4 shows the different orientations of the isopropenyl groups in the two independent molecules, (IA and IB). The value of the C21—C19—C20—C29 torsion angle describes the orientation of the isopropenyl group is equal to 92.3 (4)° (IA) and -98.8 (4)° (IB). The value of the C21—C19—C20—C29 torsion angle for betulin-DMSO and betulin-ethanol solvates are -96.8 (5)° and 88.6 (5)°, respectively. The hydroxymethyl group is attached to atom C17 of ring D in an axial orientation. No classical hydrogen bonding involving the hydroxy groups is observed. In the crystal, weak intermolecular C—H···O interactions link molecules into chains along [010]. Weak intramolecular C—H···O hydrogen bonds are also observed.
Experimental
Betulonic alcohol was obtained by oxidation of naturally occurring betulin in one-step reaction utilizing Jones-oxidation (CrO3/H2SO4 in acetone-water solution) as the side product. The crude material was subjected to column chromatography on silica gel and eluted with CH2Cl2/C2H5OH (40:1, v/v). Betulonic alcohol [m.p. 367-369K, lit. m.p. 367-369K, Hata et al., 2002), Rf=0.68 (silica gel, CH2Cl2/C2H5OH, 40:1, v/v)] was crystallized from methanol, yielding colorless single crystals suitable for the X-ray analysis. 1H NMR (500 MHz, CDCl3) and EI MS data were identical with reported data (Hata et al., 2002).
Refinement
The aromatic hydrogen atoms were treated as riding on their parent carbon atoms with d(C—H) = 0.95 Å and assigned isotropic atomic displacement parameters equal to 1.2 times the value of the equivalent atomic displacement parameters of the parent carbon atom [Uiso(H)= 1.2Ueq(C)]. The methylene H atoms were constrained to an ideal geometry with d(C—H) = 0.99 A° or d(C—H) = 0.95 Å (for terminal methylene group) and Uiso(H) = 1.2Ueq(C). Methyl H atoms were constrained as riding atoms, fixed to the parent atoms with distance of 0.98 A° and Uiso(H) = 1.5Ueq(C). hydroxy H atoms were constrained as riding atoms with d(O—H) = 0.84 Å and Uiso(H) = 1.5Ueq(O). Hydrogen atoms involved in weak hydrogen bonds were refined freely with Uiso(H) equal to 1.2Ueq of the parent atom. In the absence of significant anomalous dispersion effects the Friedel pairs were merged.
Figures
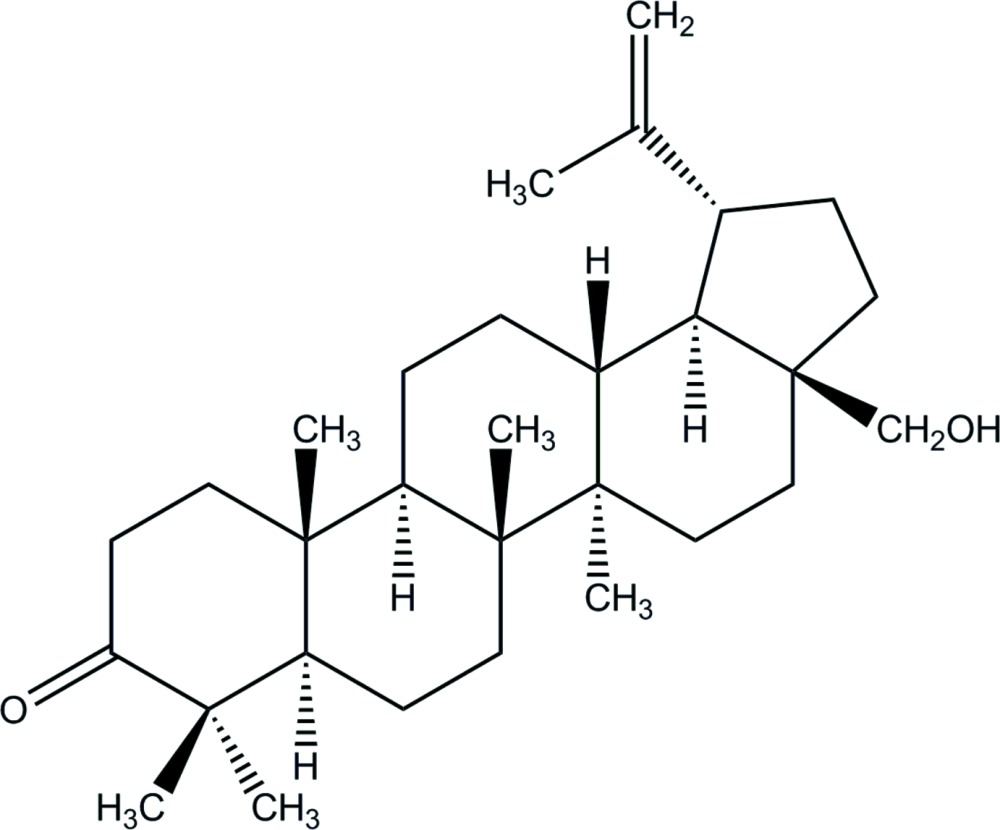
Fig. 1.
Atom and ring numbering scheme for the title compound.
Fig. 2.
The asymmetric unit of the title compound. Displacement ellipsoids are drawn at the 50% level.
Fig. 3.
View of the unit cell along the crystallographic a axis.
Fig. 4.

Different orientation of isopropenyl group virewd towards the C19/C20/C29/C30 plane in IA and IB.
Crystal data
| C30H48O2 | F(000) = 1952 |
| Mr = 440.71 | Dx = 1.149 Mg m−3 |
| Orthorhombic, P212121 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2ac 2ab | Cell parameters from 24992 reflections |
| a = 9.4447 (3) Å | θ = 2.5–34.6° |
| b = 19.1818 (6) Å | µ = 0.07 mm−1 |
| c = 28.1141 (7) Å | T = 100 K |
| V = 5093.3 (3) Å3 | Plate, colourless |
| Z = 8 | 0.60 × 0.56 × 0.20 mm |
Data collection
| Oxford Diffraction diffractometer with a Sapphire3 detector | 5036 independent reflections |
| Radiation source: fine-focus sealed tube | 4401 reflections with I > 2σ(I) |
| Graphite monochromator | Rint = 0.055 |
| Detector resolution: 16.0328 pixels mm-1 | θmax = 25.1°, θmin = 2.5° |
| ω scan | h = −8→11 |
| Absorption correction: multi-scan (CrysAlis RED; Oxford Diffraction, 2008) | k = −22→22 |
| Tmin = 0.960, Tmax = 0.986 | l = −33→33 |
| 61473 measured reflections |
Refinement
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.051 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.141 | H atoms treated by a mixture of independent and constrained refinement |
| S = 1.03 | w = 1/[σ2(Fo2) + (0.1099P)2] where P = (Fo2 + 2Fc2)/3 |
| 5036 reflections | (Δ/σ)max < 0.001 |
| 609 parameters | Δρmax = 0.55 e Å−3 |
| 0 restraints | Δρmin = −0.50 e Å−3 |
Special details
| Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
| Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R- factors based on ALL data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| O1A | 1.1431 (3) | 0.77637 (14) | −0.03486 (10) | 0.0509 (8) | |
| O2A | 0.6372 (4) | 0.28035 (18) | 0.08472 (12) | 0.0727 (11) | |
| H2A | 0.7190 | 0.2945 | 0.0914 | 0.109* | |
| C1A | 1.0227 (4) | 0.60249 (18) | −0.04338 (11) | 0.0299 (8) | |
| H1AA | 0.9311 | 0.6232 | −0.0531 | 0.036* | |
| H1AB | 1.0404 | 0.5616 | −0.0640 | 0.036* | |
| C2A | 1.1415 (4) | 0.6566 (2) | −0.05138 (13) | 0.0385 (9) | |
| H2AA | 1.2343 | 0.6339 | −0.0461 | 0.046* | |
| H2AB | 1.1383 | 0.6727 | −0.0848 | 0.046* | |
| C3A | 1.1299 (4) | 0.71811 (19) | −0.01944 (13) | 0.0320 (8) | |
| C4A | 1.1037 (4) | 0.70497 (18) | 0.03356 (12) | 0.0309 (8) | |
| C5A | 0.9920 (3) | 0.64490 (17) | 0.03958 (11) | 0.0255 (7) | |
| H5A | 0.9010 | 0.6656 | 0.0282 | 0.031* | |
| C6A | 0.9629 (4) | 0.62503 (19) | 0.09145 (11) | 0.0329 (8) | |
| H6AA | 1.0405 | 0.5951 | 0.1035 | 0.039* | |
| H6AB | 0.9594 | 0.6677 | 0.1113 | 0.039* | |
| C7A | 0.8220 (4) | 0.58598 (19) | 0.09519 (11) | 0.0332 (8) | |
| H7AA | 0.7447 | 0.6175 | 0.0850 | 0.040* | |
| H7AB | 0.8050 | 0.5737 | 0.1289 | 0.040* | |
| C8A | 0.8157 (4) | 0.51877 (18) | 0.06497 (10) | 0.0267 (7) | |
| C9A | 0.8698 (3) | 0.53544 (17) | 0.01340 (10) | 0.0218 (7) | |
| H9A | 0.7955 | 0.5666 | −0.0005 | 0.026* | |
| C10A | 1.0118 (3) | 0.57800 (17) | 0.00863 (11) | 0.0251 (7) | |
| C11A | 0.8664 (4) | 0.46982 (17) | −0.01759 (11) | 0.0264 (7) | |
| H11A | 0.9318 | 0.4347 | −0.0041 | 0.032* | |
| H11B | 0.8998 | 0.4816 | −0.0500 | 0.032* | |
| C12A | 0.7174 (3) | 0.43854 (17) | −0.02076 (10) | 0.0249 (7) | |
| H12A | 0.7214 | 0.3943 | −0.0388 | 0.030* | |
| H12B | 0.6548 | 0.4710 | −0.0382 | 0.030* | |
| C13A | 0.6552 (3) | 0.42468 (17) | 0.02894 (10) | 0.0240 (7) | |
| H13A | 0.723 (4) | 0.3924 (18) | 0.0478 (12) | 0.029* | |
| C14A | 0.6565 (3) | 0.49143 (17) | 0.06015 (10) | 0.0244 (7) | |
| C15A | 0.5923 (4) | 0.4738 (2) | 0.10962 (11) | 0.0359 (9) | |
| H15A | 0.6601 | 0.4439 | 0.1270 | 0.043* | |
| H15B | 0.5823 | 0.5177 | 0.1278 | 0.043* | |
| C16A | 0.4469 (4) | 0.4367 (2) | 0.10862 (12) | 0.0374 (9) | |
| H16A | 0.4186 | 0.4238 | 0.1414 | 0.045* | |
| H16B | 0.3742 | 0.4686 | 0.0955 | 0.045* | |
| C17A | 0.4564 (4) | 0.37156 (19) | 0.07795 (12) | 0.0341 (8) | |
| C18A | 0.5063 (4) | 0.39119 (18) | 0.02774 (11) | 0.0268 (7) | |
| H18A | 0.4399 | 0.4280 | 0.0160 | 0.032* | |
| C19A | 0.4791 (4) | 0.32596 (18) | −0.00271 (13) | 0.0341 (8) | |
| H19A | 0.558 (4) | 0.296 (2) | 0.0026 (13) | 0.041* | |
| C20A | 0.4436 (4) | 0.33884 (19) | −0.05530 (13) | 0.0352 (8) | |
| C21A | 0.3519 (5) | 0.2879 (2) | 0.02319 (15) | 0.0510 (11) | |
| H21A | 0.2689 | 0.2846 | 0.0018 | 0.061* | |
| H21B | 0.3799 | 0.2403 | 0.0331 | 0.061* | |
| C22A | 0.3172 (5) | 0.3332 (2) | 0.06692 (15) | 0.0489 (11) | |
| H22A | 0.2406 | 0.3667 | 0.0596 | 0.059* | |
| H22B | 0.2878 | 0.3039 | 0.0941 | 0.059* | |
| C23A | 1.0466 (4) | 0.77220 (19) | 0.05630 (14) | 0.0409 (9) | |
| H23A | 1.1071 | 0.8115 | 0.0473 | 0.061* | |
| H23B | 1.0465 | 0.7673 | 0.0910 | 0.061* | |
| H23C | 0.9498 | 0.7807 | 0.0451 | 0.061* | |
| C24A | 1.2508 (4) | 0.6891 (2) | 0.05663 (15) | 0.0453 (10) | |
| H24A | 1.2978 | 0.6518 | 0.0388 | 0.068* | |
| H24B | 1.2372 | 0.6745 | 0.0897 | 0.068* | |
| H24C | 1.3095 | 0.7312 | 0.0558 | 0.068* | |
| C25A | 1.1442 (4) | 0.5352 (2) | 0.02094 (14) | 0.0364 (8) | |
| H25A | 1.1341 | 0.4879 | 0.0083 | 0.055* | |
| H25B | 1.1554 | 0.5331 | 0.0556 | 0.055* | |
| H25C | 1.2278 | 0.5574 | 0.0068 | 0.055* | |
| C26A | 0.9099 (4) | 0.46436 (19) | 0.09009 (12) | 0.0342 (8) | |
| H26A | 1.0051 | 0.4836 | 0.0946 | 0.051* | |
| H26B | 0.9155 | 0.4221 | 0.0705 | 0.051* | |
| H26C | 0.8690 | 0.4528 | 0.1211 | 0.051* | |
| C27A | 0.5578 (4) | 0.54781 (17) | 0.03828 (12) | 0.0291 (8) | |
| H27A | 0.4591 | 0.5325 | 0.0408 | 0.044* | |
| H27B | 0.5823 | 0.5547 | 0.0047 | 0.044* | |
| H27C | 0.5699 | 0.5918 | 0.0556 | 0.044* | |
| C28A | 0.5492 (5) | 0.3188 (2) | 0.10332 (14) | 0.0447 (10) | |
| H28A | 0.6013 | 0.3454 | 0.1279 | 0.054* | |
| H28B | 0.4835 | 0.2877 | 0.1207 | 0.054* | |
| C29A | 0.3848 (4) | 0.3965 (2) | −0.07222 (14) | 0.0407 (9) | |
| H29A | 0.3618 | 0.3998 | −0.1050 | 0.049* | |
| H29B | 0.3656 | 0.4345 | −0.0515 | 0.049* | |
| C30A | 0.4735 (6) | 0.2800 (3) | −0.08691 (16) | 0.0631 (13) | |
| H30A | 0.4304 | 0.2886 | −0.1181 | 0.095* | |
| H30B | 0.4341 | 0.2373 | −0.0732 | 0.095* | |
| H30C | 0.5762 | 0.2749 | −0.0906 | 0.095* | |
| O1B | −0.3496 (3) | 0.80561 (15) | 0.28884 (10) | 0.0480 (7) | |
| O2B | 0.6246 (3) | 1.01860 (12) | 0.13685 (8) | 0.0344 (6) | |
| H2B | 0.6040 | 1.0023 | 0.1637 | 0.052* | |
| C1B | 0.0161 (4) | 0.84111 (17) | 0.28726 (11) | 0.0261 (7) | |
| H1BA | 0.1042 | 0.8236 | 0.3022 | 0.031* | |
| H1BB | −0.0123 | 0.8840 | 0.3044 | 0.031* | |
| C2B | −0.1003 (4) | 0.78616 (18) | 0.29336 (12) | 0.0323 (8) | |
| H2BA | −0.0657 | 0.7410 | 0.2809 | 0.039* | |
| H2BB | −0.1203 | 0.7802 | 0.3277 | 0.039* | |
| C3B | −0.2362 (4) | 0.80529 (17) | 0.26795 (12) | 0.0286 (7) | |
| C4B | −0.2235 (3) | 0.82674 (16) | 0.21577 (11) | 0.0222 (7) | |
| C5B | −0.0983 (3) | 0.88076 (15) | 0.21148 (11) | 0.0185 (6) | |
| H5B | −0.1308 | 0.9221 | 0.2302 | 0.022* | |
| C6B | −0.0769 (3) | 0.90864 (16) | 0.16122 (10) | 0.0205 (6) | |
| H6BA | −0.1700 | 0.9196 | 0.1469 | 0.025* | |
| H6BB | −0.0307 | 0.8725 | 0.1414 | 0.025* | |
| C7B | 0.0147 (3) | 0.97415 (15) | 0.16204 (10) | 0.0197 (6) | |
| H7BA | −0.0361 | 1.0111 | 0.1798 | 0.024* | |
| H7BB | 0.0278 | 0.9908 | 0.1290 | 0.024* | |
| C8B | 0.1626 (3) | 0.96323 (15) | 0.18508 (10) | 0.0164 (6) | |
| C9B | 0.1422 (3) | 0.92689 (14) | 0.23454 (10) | 0.0175 (6) | |
| H9BA | 0.0894 | 0.9616 | 0.2543 | 0.021* | |
| C10B | 0.0471 (3) | 0.85976 (15) | 0.23497 (10) | 0.0191 (6) | |
| C11B | 0.2853 (3) | 0.91725 (16) | 0.26026 (10) | 0.0211 (6) | |
| H11C | 0.2675 | 0.8986 | 0.2925 | 0.025* | |
| H11D | 0.3423 | 0.8824 | 0.2427 | 0.025* | |
| C12B | 0.3709 (3) | 0.98473 (15) | 0.26455 (10) | 0.0214 (6) | |
| H12C | 0.3226 | 1.0167 | 0.2869 | 0.026* | |
| H12D | 0.4656 | 0.9739 | 0.2777 | 0.026* | |
| C13B | 0.3881 (3) | 1.02087 (15) | 0.21660 (10) | 0.0173 (6) | |
| H13B | 0.435 (3) | 0.9873 (17) | 0.1934 (11) | 0.021* | |
| C14B | 0.2394 (3) | 1.03694 (15) | 0.19426 (10) | 0.0177 (6) | |
| C15B | 0.2574 (3) | 1.07779 (16) | 0.14691 (10) | 0.0212 (6) | |
| H15C | 0.2956 | 1.0456 | 0.1226 | 0.025* | |
| H15D | 0.1627 | 1.0932 | 0.1360 | 0.025* | |
| C16B | 0.3544 (3) | 1.14170 (16) | 0.14973 (11) | 0.0242 (7) | |
| H16C | 0.3116 | 1.1772 | 0.1708 | 0.029* | |
| H16D | 0.3656 | 1.1624 | 0.1177 | 0.029* | |
| C17B | 0.4999 (3) | 1.11996 (15) | 0.16926 (10) | 0.0212 (7) | |
| C18B | 0.4813 (3) | 1.08681 (15) | 0.21843 (10) | 0.0200 (6) | |
| H18B | 0.4304 | 1.1216 | 0.2387 | 0.024* | |
| C19B | 0.6342 (3) | 1.08099 (16) | 0.23816 (11) | 0.0218 (6) | |
| H19B | 0.677 (4) | 1.0405 (19) | 0.2266 (12) | 0.026* | |
| C20B | 0.6521 (4) | 1.08491 (17) | 0.29161 (12) | 0.0287 (7) | |
| C21B | 0.7161 (4) | 1.14185 (17) | 0.21277 (12) | 0.0276 (7) | |
| H21C | 0.7537 | 1.1751 | 0.2365 | 0.033* | |
| H21D | 0.7962 | 1.1232 | 0.1940 | 0.033* | |
| C22B | 0.6084 (4) | 1.17815 (17) | 0.18003 (12) | 0.0262 (7) | |
| H22C | 0.551 (4) | 1.2189 (18) | 0.1957 (12) | 0.031* | |
| H22D | 0.662 (4) | 1.1975 (18) | 0.1499 (13) | 0.031* | |
| C23B | −0.3632 (3) | 0.86105 (17) | 0.20088 (13) | 0.0296 (7) | |
| H23D | −0.4416 | 0.8286 | 0.2066 | 0.044* | |
| H23E | −0.3594 | 0.8729 | 0.1670 | 0.044* | |
| H23F | −0.3779 | 0.9036 | 0.2195 | 0.044* | |
| C24B | −0.2054 (4) | 0.76017 (17) | 0.18541 (13) | 0.0297 (8) | |
| H24D | −0.1275 | 0.7322 | 0.1982 | 0.045* | |
| H24E | −0.1842 | 0.7732 | 0.1525 | 0.045* | |
| H24F | −0.2931 | 0.7329 | 0.1863 | 0.045* | |
| C25B | 0.1199 (3) | 0.79668 (15) | 0.21113 (12) | 0.0237 (7) | |
| H25D | 0.2216 | 0.7979 | 0.2178 | 0.035* | |
| H25E | 0.1047 | 0.7987 | 0.1767 | 0.035* | |
| H25F | 0.0794 | 0.7534 | 0.2237 | 0.035* | |
| C26B | 0.2521 (3) | 0.91803 (16) | 0.15102 (10) | 0.0218 (6) | |
| H26D | 0.1926 | 0.8813 | 0.1374 | 0.033* | |
| H26E | 0.3306 | 0.8968 | 0.1687 | 0.033* | |
| H26F | 0.2899 | 0.9473 | 0.1254 | 0.033* | |
| C27B | 0.1513 (3) | 1.08464 (15) | 0.22700 (11) | 0.0237 (7) | |
| H27D | 0.2041 | 1.1276 | 0.2334 | 0.036* | |
| H27E | 0.1322 | 1.0605 | 0.2570 | 0.036* | |
| H27F | 0.0615 | 1.0961 | 0.2114 | 0.036* | |
| C28B | 0.5697 (3) | 1.07459 (16) | 0.13205 (11) | 0.0231 (7) | |
| H28C | 0.4967 | 1.0677 | 0.1072 | 0.028* | |
| H28D | 0.6433 | 1.1044 | 0.1174 | 0.028* | |
| C29B | 0.7480 (5) | 1.0447 (2) | 0.31341 (14) | 0.0483 (11) | |
| H29C | 0.7636 | 1.0495 | 0.3466 | 0.058* | |
| H29D | 0.8004 | 1.0114 | 0.2957 | 0.058* | |
| C30B | 0.5679 (5) | 1.1375 (2) | 0.31777 (13) | 0.0491 (11) | |
| H30D | 0.6030 | 1.1412 | 0.3505 | 0.074* | |
| H30E | 0.4682 | 1.1233 | 0.3182 | 0.074* | |
| H30F | 0.5768 | 1.1828 | 0.3020 | 0.074* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| O1A | 0.0601 (19) | 0.0390 (16) | 0.0537 (18) | −0.0081 (14) | 0.0087 (15) | −0.0007 (13) |
| O2A | 0.082 (2) | 0.081 (2) | 0.055 (2) | 0.034 (2) | 0.027 (2) | 0.0391 (18) |
| C1A | 0.0320 (18) | 0.0340 (18) | 0.0237 (16) | −0.0067 (16) | 0.0070 (14) | −0.0063 (14) |
| C2A | 0.042 (2) | 0.044 (2) | 0.0295 (18) | −0.0090 (19) | 0.0083 (17) | −0.0069 (16) |
| C3A | 0.0208 (17) | 0.034 (2) | 0.041 (2) | −0.0040 (15) | −0.0020 (15) | −0.0043 (16) |
| C4A | 0.0280 (17) | 0.0333 (19) | 0.0314 (18) | 0.0025 (15) | −0.0057 (15) | −0.0109 (15) |
| C5A | 0.0208 (16) | 0.0335 (17) | 0.0223 (16) | 0.0038 (14) | −0.0046 (13) | −0.0046 (13) |
| C6A | 0.0369 (19) | 0.042 (2) | 0.0191 (16) | 0.0074 (16) | −0.0075 (14) | −0.0092 (15) |
| C7A | 0.0346 (18) | 0.049 (2) | 0.0157 (15) | 0.0052 (17) | 0.0010 (14) | −0.0076 (15) |
| C8A | 0.0298 (17) | 0.0379 (19) | 0.0125 (14) | 0.0085 (15) | 0.0004 (13) | 0.0003 (13) |
| C9A | 0.0215 (15) | 0.0292 (17) | 0.0145 (14) | 0.0062 (14) | −0.0005 (12) | −0.0001 (12) |
| C10A | 0.0228 (16) | 0.0330 (18) | 0.0195 (15) | 0.0030 (14) | −0.0002 (13) | −0.0038 (13) |
| C11A | 0.0289 (17) | 0.0330 (18) | 0.0173 (14) | 0.0018 (15) | 0.0046 (13) | −0.0011 (13) |
| C12A | 0.0299 (17) | 0.0297 (17) | 0.0152 (15) | 0.0001 (14) | 0.0053 (13) | 0.0011 (13) |
| C13A | 0.0254 (16) | 0.0304 (17) | 0.0163 (14) | 0.0050 (15) | 0.0035 (13) | 0.0064 (13) |
| C14A | 0.0249 (16) | 0.0348 (18) | 0.0135 (14) | 0.0058 (15) | 0.0039 (12) | 0.0038 (13) |
| C15A | 0.040 (2) | 0.053 (2) | 0.0140 (15) | 0.0062 (18) | 0.0062 (14) | 0.0019 (15) |
| C16A | 0.0355 (19) | 0.053 (2) | 0.0235 (17) | 0.0058 (18) | 0.0145 (15) | 0.0084 (16) |
| C17A | 0.0359 (19) | 0.041 (2) | 0.0254 (17) | 0.0050 (17) | 0.0120 (15) | 0.0129 (15) |
| C18A | 0.0303 (18) | 0.0296 (17) | 0.0205 (16) | 0.0043 (14) | 0.0067 (14) | 0.0097 (13) |
| C19A | 0.037 (2) | 0.0275 (18) | 0.038 (2) | −0.0004 (16) | 0.0083 (16) | 0.0074 (15) |
| C20A | 0.0259 (17) | 0.041 (2) | 0.038 (2) | −0.0069 (16) | 0.0049 (15) | −0.0043 (16) |
| C21A | 0.059 (3) | 0.048 (2) | 0.046 (2) | −0.016 (2) | 0.015 (2) | 0.0079 (19) |
| C22A | 0.046 (2) | 0.052 (2) | 0.049 (2) | −0.007 (2) | 0.022 (2) | 0.016 (2) |
| C23A | 0.040 (2) | 0.037 (2) | 0.046 (2) | 0.0024 (18) | −0.0033 (18) | −0.0132 (17) |
| C24A | 0.028 (2) | 0.051 (2) | 0.057 (3) | 0.0003 (18) | −0.0116 (18) | −0.011 (2) |
| C25A | 0.0232 (17) | 0.038 (2) | 0.048 (2) | 0.0055 (16) | 0.0005 (16) | −0.0091 (17) |
| C26A | 0.0326 (19) | 0.045 (2) | 0.0247 (17) | 0.0053 (17) | −0.0061 (14) | 0.0085 (15) |
| C27A | 0.0260 (17) | 0.0286 (17) | 0.0327 (18) | 0.0050 (14) | −0.0003 (15) | 0.0025 (14) |
| C28A | 0.051 (2) | 0.048 (2) | 0.035 (2) | 0.005 (2) | 0.0139 (19) | 0.0185 (18) |
| C29A | 0.041 (2) | 0.044 (2) | 0.037 (2) | −0.0020 (18) | −0.0088 (17) | 0.0047 (17) |
| C30A | 0.074 (3) | 0.071 (3) | 0.044 (3) | 0.019 (3) | 0.008 (2) | −0.008 (2) |
| O1B | 0.0398 (15) | 0.0614 (18) | 0.0428 (16) | −0.0095 (14) | 0.0111 (13) | 0.0146 (13) |
| O2B | 0.0411 (14) | 0.0317 (13) | 0.0302 (12) | −0.0012 (12) | 0.0049 (11) | 0.0028 (10) |
| C1B | 0.0302 (17) | 0.0276 (16) | 0.0206 (16) | −0.0045 (14) | 0.0005 (13) | 0.0040 (13) |
| C2B | 0.040 (2) | 0.0317 (18) | 0.0254 (17) | −0.0080 (16) | −0.0008 (15) | 0.0100 (14) |
| C3B | 0.0295 (18) | 0.0253 (17) | 0.0310 (18) | −0.0106 (15) | 0.0048 (15) | 0.0057 (14) |
| C4B | 0.0228 (16) | 0.0197 (15) | 0.0242 (16) | −0.0009 (13) | 0.0003 (13) | 0.0021 (13) |
| C5B | 0.0184 (15) | 0.0148 (14) | 0.0222 (15) | −0.0008 (12) | 0.0001 (12) | 0.0008 (12) |
| C6B | 0.0181 (15) | 0.0229 (15) | 0.0206 (15) | 0.0020 (13) | −0.0040 (12) | 0.0019 (12) |
| C7B | 0.0232 (16) | 0.0181 (14) | 0.0177 (14) | 0.0000 (13) | −0.0025 (12) | 0.0055 (12) |
| C8B | 0.0175 (14) | 0.0178 (15) | 0.0138 (13) | −0.0010 (12) | −0.0003 (11) | 0.0023 (11) |
| C9B | 0.0228 (15) | 0.0154 (14) | 0.0143 (13) | −0.0011 (13) | 0.0006 (12) | 0.0022 (11) |
| C10B | 0.0226 (15) | 0.0164 (14) | 0.0185 (15) | −0.0007 (13) | −0.0016 (12) | 0.0051 (11) |
| C11B | 0.0272 (16) | 0.0199 (15) | 0.0162 (14) | −0.0015 (13) | −0.0017 (12) | 0.0053 (12) |
| C12B | 0.0257 (16) | 0.0220 (15) | 0.0165 (14) | −0.0007 (14) | −0.0022 (13) | 0.0019 (12) |
| C13B | 0.0201 (15) | 0.0178 (14) | 0.0139 (13) | −0.0010 (13) | −0.0006 (12) | −0.0019 (12) |
| C14B | 0.0193 (15) | 0.0156 (14) | 0.0180 (14) | −0.0005 (13) | 0.0001 (12) | 0.0028 (12) |
| C15B | 0.0224 (15) | 0.0207 (15) | 0.0206 (15) | −0.0008 (13) | −0.0015 (12) | 0.0083 (13) |
| C16B | 0.0285 (17) | 0.0216 (15) | 0.0225 (15) | −0.0010 (14) | −0.0019 (14) | 0.0072 (12) |
| C17B | 0.0248 (17) | 0.0176 (15) | 0.0211 (15) | −0.0007 (13) | −0.0009 (13) | 0.0029 (12) |
| C18B | 0.0257 (16) | 0.0164 (14) | 0.0180 (14) | −0.0009 (13) | −0.0019 (12) | 0.0009 (12) |
| C19B | 0.0228 (16) | 0.0175 (15) | 0.0250 (15) | −0.0039 (13) | −0.0040 (13) | −0.0005 (13) |
| C20B | 0.0319 (18) | 0.0265 (16) | 0.0277 (17) | −0.0101 (15) | −0.0076 (14) | 0.0008 (14) |
| C21B | 0.0304 (17) | 0.0230 (16) | 0.0295 (17) | −0.0081 (14) | −0.0042 (14) | 0.0010 (14) |
| C22B | 0.0292 (17) | 0.0193 (16) | 0.0300 (17) | −0.0044 (14) | 0.0006 (14) | 0.0037 (13) |
| C23B | 0.0208 (16) | 0.0268 (17) | 0.041 (2) | −0.0004 (14) | 0.0019 (15) | 0.0018 (14) |
| C24B | 0.0271 (17) | 0.0227 (17) | 0.039 (2) | −0.0002 (14) | 0.0004 (15) | −0.0040 (14) |
| C25B | 0.0241 (16) | 0.0159 (14) | 0.0309 (17) | 0.0001 (13) | −0.0053 (14) | 0.0021 (13) |
| C26B | 0.0270 (16) | 0.0217 (15) | 0.0166 (14) | −0.0026 (14) | 0.0027 (13) | −0.0002 (12) |
| C27B | 0.0268 (16) | 0.0161 (14) | 0.0282 (16) | 0.0013 (13) | −0.0004 (13) | 0.0017 (13) |
| C28B | 0.0204 (15) | 0.0269 (17) | 0.0219 (16) | −0.0097 (15) | −0.0022 (13) | 0.0015 (13) |
| C29B | 0.065 (3) | 0.047 (2) | 0.033 (2) | −0.011 (2) | −0.022 (2) | 0.0082 (17) |
| C30B | 0.053 (3) | 0.068 (3) | 0.0266 (19) | −0.005 (2) | −0.0048 (18) | −0.0162 (19) |
Geometric parameters (Å, º)
| O1A—C3A | 1.205 (4) | O1B—C3B | 1.221 (4) |
| O2A—C28A | 1.228 (5) | O2B—C28B | 1.200 (4) |
| O2A—H2A | 0.8400 | O2B—H2B | 0.8400 |
| C1A—C10A | 1.539 (4) | C1B—C2B | 1.533 (5) |
| C1A—C2A | 1.546 (5) | C1B—C10B | 1.541 (4) |
| C1A—H1AA | 0.9900 | C1B—H1BA | 0.9900 |
| C1A—H1AB | 0.9900 | C1B—H1BB | 0.9900 |
| C2A—C3A | 1.486 (5) | C2B—C3B | 1.514 (5) |
| C2A—H2AA | 0.9900 | C2B—H2BA | 0.9900 |
| C2A—H2AB | 0.9900 | C2B—H2BB | 0.9900 |
| C3A—C4A | 1.531 (5) | C3B—C4B | 1.528 (4) |
| C4A—C23A | 1.537 (5) | C4B—C23B | 1.533 (5) |
| C4A—C24A | 1.562 (5) | C4B—C24B | 1.546 (4) |
| C4A—C5A | 1.572 (5) | C4B—C5B | 1.577 (4) |
| C5A—C6A | 1.532 (5) | C5B—C6B | 1.524 (4) |
| C5A—C10A | 1.562 (4) | C5B—C10B | 1.576 (4) |
| C5A—H5A | 1.0000 | C5B—H5B | 1.0000 |
| C6A—C7A | 1.531 (5) | C6B—C7B | 1.526 (4) |
| C6A—H6AA | 0.9900 | C6B—H6BA | 0.9900 |
| C6A—H6AB | 0.9900 | C6B—H6BB | 0.9900 |
| C7A—C8A | 1.545 (5) | C7B—C8B | 1.554 (4) |
| C7A—H7AA | 0.9900 | C7B—H7BA | 0.9900 |
| C7A—H7AB | 0.9900 | C7B—H7BB | 0.9900 |
| C8A—C26A | 1.543 (5) | C8B—C26B | 1.543 (4) |
| C8A—C9A | 1.570 (4) | C8B—C9B | 1.567 (4) |
| C8A—C14A | 1.598 (5) | C8B—C14B | 1.610 (4) |
| C9A—C11A | 1.531 (4) | C9B—C11B | 1.544 (4) |
| C9A—C10A | 1.576 (4) | C9B—C10B | 1.570 (4) |
| C9A—H9A | 1.0000 | C9B—H9BA | 1.0000 |
| C10A—C25A | 1.536 (5) | C10B—C25B | 1.545 (4) |
| C11A—C12A | 1.532 (5) | C11B—C12B | 1.531 (4) |
| C11A—H11A | 0.9900 | C11B—H11C | 0.9900 |
| C11A—H11B | 0.9900 | C11B—H11D | 0.9900 |
| C12A—C13A | 1.539 (4) | C12B—C13B | 1.525 (4) |
| C12A—H12A | 0.9900 | C12B—H12C | 0.9900 |
| C12A—H12B | 0.9900 | C12B—H12D | 0.9900 |
| C13A—C18A | 1.546 (5) | C13B—C18B | 1.542 (4) |
| C13A—C14A | 1.552 (4) | C13B—C14B | 1.569 (4) |
| C13A—H13A | 1.04 (4) | C13B—H13B | 1.02 (3) |
| C14A—C27A | 1.554 (4) | C14B—C27B | 1.542 (4) |
| C14A—C15A | 1.554 (4) | C14B—C15B | 1.554 (4) |
| C15A—C16A | 1.547 (5) | C15B—C16B | 1.532 (4) |
| C15A—H15A | 0.9900 | C15B—H15C | 0.9900 |
| C15A—H15B | 0.9900 | C15B—H15D | 0.9900 |
| C16A—C17A | 1.521 (5) | C16B—C17B | 1.538 (4) |
| C16A—H16A | 0.9900 | C16B—H16C | 0.9900 |
| C16A—H16B | 0.9900 | C16B—H16D | 0.9900 |
| C17A—C28A | 1.517 (5) | C17B—C28B | 1.512 (4) |
| C17A—C18A | 1.535 (4) | C17B—C18B | 1.532 (4) |
| C17A—C22A | 1.538 (6) | C17B—C22B | 1.546 (4) |
| C18A—C19A | 1.538 (5) | C18B—C19B | 1.551 (4) |
| C18A—H18A | 1.0000 | C18B—H18B | 1.0000 |
| C19A—C20A | 1.536 (5) | C19B—C20B | 1.514 (4) |
| C19A—C21A | 1.582 (5) | C19B—C21B | 1.572 (4) |
| C19A—H19A | 0.95 (4) | C19B—H19B | 0.93 (4) |
| C20A—C29A | 1.326 (5) | C20B—C29B | 1.338 (5) |
| C20A—C30A | 1.463 (6) | C20B—C30B | 1.480 (5) |
| C21A—C22A | 1.541 (6) | C21B—C22B | 1.538 (5) |
| C21A—H21A | 0.9900 | C21B—H21C | 0.9900 |
| C21A—H21B | 0.9900 | C21B—H21D | 0.9900 |
| C22A—H22A | 0.9900 | C22B—H22C | 1.05 (4) |
| C22A—H22B | 0.9900 | C22B—H22D | 1.05 (4) |
| C23A—H23A | 0.9800 | C23B—H23D | 0.9800 |
| C23A—H23B | 0.9800 | C23B—H23E | 0.9800 |
| C23A—H23C | 0.9800 | C23B—H23F | 0.9800 |
| C24A—H24A | 0.9800 | C24B—H24D | 0.9800 |
| C24A—H24B | 0.9800 | C24B—H24E | 0.9800 |
| C24A—H24C | 0.9800 | C24B—H24F | 0.9800 |
| C25A—H25A | 0.9800 | C25B—H25D | 0.9800 |
| C25A—H25B | 0.9800 | C25B—H25E | 0.9800 |
| C25A—H25C | 0.9800 | C25B—H25F | 0.9800 |
| C26A—H26A | 0.9800 | C26B—H26D | 0.9800 |
| C26A—H26B | 0.9800 | C26B—H26E | 0.9800 |
| C26A—H26C | 0.9800 | C26B—H26F | 0.9800 |
| C27A—H27A | 0.9800 | C27B—H27D | 0.9800 |
| C27A—H27B | 0.9800 | C27B—H27E | 0.9800 |
| C27A—H27C | 0.9800 | C27B—H27F | 0.9800 |
| C28A—H28A | 0.9900 | C28B—H28C | 0.9900 |
| C28A—H28B | 0.9900 | C28B—H28D | 0.9900 |
| C29A—H29A | 0.9500 | C29B—H29C | 0.9500 |
| C29A—H29B | 0.9500 | C29B—H29D | 0.9500 |
| C30A—H30A | 0.9800 | C30B—H30D | 0.9800 |
| C30A—H30B | 0.9800 | C30B—H30E | 0.9800 |
| C30A—H30C | 0.9800 | C30B—H30F | 0.9800 |
| C28A—O2A—H2A | 109.5 | C28B—O2B—H2B | 109.5 |
| C10A—C1A—C2A | 113.1 (3) | C2B—C1B—C10B | 113.7 (3) |
| C10A—C1A—H1AA | 109.0 | C2B—C1B—H1BA | 108.8 |
| C2A—C1A—H1AA | 109.0 | C10B—C1B—H1BA | 108.8 |
| C10A—C1A—H1AB | 109.0 | C2B—C1B—H1BB | 108.8 |
| C2A—C1A—H1AB | 109.0 | C10B—C1B—H1BB | 108.8 |
| H1AA—C1A—H1AB | 107.8 | H1BA—C1B—H1BB | 107.7 |
| C3A—C2A—C1A | 113.1 (3) | C3B—C2B—C1B | 112.9 (3) |
| C3A—C2A—H2AA | 109.0 | C3B—C2B—H2BA | 109.0 |
| C1A—C2A—H2AA | 109.0 | C1B—C2B—H2BA | 109.0 |
| C3A—C2A—H2AB | 109.0 | C3B—C2B—H2BB | 109.0 |
| C1A—C2A—H2AB | 109.0 | C1B—C2B—H2BB | 109.0 |
| H2AA—C2A—H2AB | 107.8 | H2BA—C2B—H2BB | 107.8 |
| O1A—C3A—C2A | 120.7 (3) | O1B—C3B—C2B | 121.2 (3) |
| O1A—C3A—C4A | 121.3 (3) | O1B—C3B—C4B | 121.9 (3) |
| C2A—C3A—C4A | 118.0 (3) | C2B—C3B—C4B | 116.8 (3) |
| C3A—C4A—C23A | 108.9 (3) | C3B—C4B—C23B | 108.1 (3) |
| C3A—C4A—C24A | 107.0 (3) | C3B—C4B—C24B | 108.4 (3) |
| C23A—C4A—C24A | 107.6 (3) | C23B—C4B—C24B | 107.4 (3) |
| C3A—C4A—C5A | 109.5 (3) | C3B—C4B—C5B | 108.0 (2) |
| C23A—C4A—C5A | 109.5 (3) | C23B—C4B—C5B | 110.0 (2) |
| C24A—C4A—C5A | 114.2 (3) | C24B—C4B—C5B | 114.7 (3) |
| C6A—C5A—C10A | 110.3 (3) | C6B—C5B—C10B | 111.3 (2) |
| C6A—C5A—C4A | 113.9 (3) | C6B—C5B—C4B | 113.6 (2) |
| C10A—C5A—C4A | 117.5 (3) | C10B—C5B—C4B | 117.0 (2) |
| C6A—C5A—H5A | 104.5 | C6B—C5B—H5B | 104.5 |
| C10A—C5A—H5A | 104.5 | C10B—C5B—H5B | 104.5 |
| C4A—C5A—H5A | 104.5 | C4B—C5B—H5B | 104.5 |
| C7A—C6A—C5A | 110.0 (3) | C5B—C6B—C7B | 110.5 (2) |
| C7A—C6A—H6AA | 109.7 | C5B—C6B—H6BA | 109.6 |
| C5A—C6A—H6AA | 109.7 | C7B—C6B—H6BA | 109.6 |
| C7A—C6A—H6AB | 109.7 | C5B—C6B—H6BB | 109.6 |
| C5A—C6A—H6AB | 109.7 | C7B—C6B—H6BB | 109.6 |
| H6AA—C6A—H6AB | 108.2 | H6BA—C6B—H6BB | 108.1 |
| C6A—C7A—C8A | 113.8 (3) | C6B—C7B—C8B | 113.9 (2) |
| C6A—C7A—H7AA | 108.8 | C6B—C7B—H7BA | 108.8 |
| C8A—C7A—H7AA | 108.8 | C8B—C7B—H7BA | 108.8 |
| C6A—C7A—H7AB | 108.8 | C6B—C7B—H7BB | 108.8 |
| C8A—C7A—H7AB | 108.8 | C8B—C7B—H7BB | 108.8 |
| H7AA—C7A—H7AB | 107.7 | H7BA—C7B—H7BB | 107.7 |
| C26A—C8A—C7A | 106.9 (3) | C26B—C8B—C7B | 108.0 (2) |
| C26A—C8A—C9A | 111.9 (3) | C26B—C8B—C9B | 111.6 (2) |
| C7A—C8A—C9A | 109.0 (3) | C7B—C8B—C9B | 108.6 (2) |
| C26A—C8A—C14A | 111.1 (3) | C26B—C8B—C14B | 110.3 (2) |
| C7A—C8A—C14A | 110.9 (3) | C7B—C8B—C14B | 110.7 (2) |
| C9A—C8A—C14A | 107.1 (2) | C9B—C8B—C14B | 107.7 (2) |
| C11A—C9A—C8A | 110.6 (3) | C11B—C9B—C8B | 111.2 (2) |
| C11A—C9A—C10A | 113.3 (2) | C11B—C9B—C10B | 113.5 (2) |
| C8A—C9A—C10A | 117.4 (2) | C8B—C9B—C10B | 116.2 (2) |
| C11A—C9A—H9A | 104.7 | C11B—C9B—H9BA | 104.9 |
| C8A—C9A—H9A | 104.7 | C8B—C9B—H9BA | 104.9 |
| C10A—C9A—H9A | 104.7 | C10B—C9B—H9BA | 104.9 |
| C25A—C10A—C1A | 108.8 (3) | C1B—C10B—C25B | 108.5 (2) |
| C25A—C10A—C5A | 114.3 (3) | C1B—C10B—C9B | 107.8 (2) |
| C1A—C10A—C5A | 106.6 (3) | C25B—C10B—C9B | 112.6 (2) |
| C25A—C10A—C9A | 113.4 (3) | C1B—C10B—C5B | 107.1 (2) |
| C1A—C10A—C9A | 107.2 (2) | C25B—C10B—C5B | 113.9 (2) |
| C5A—C10A—C9A | 106.0 (2) | C9B—C10B—C5B | 106.6 (2) |
| C9A—C11A—C12A | 112.0 (3) | C12B—C11B—C9B | 113.5 (2) |
| C9A—C11A—H11A | 109.2 | C12B—C11B—H11C | 108.9 |
| C12A—C11A—H11A | 109.2 | C9B—C11B—H11C | 108.9 |
| C9A—C11A—H11B | 109.2 | C12B—C11B—H11D | 108.9 |
| C12A—C11A—H11B | 109.2 | C9B—C11B—H11D | 108.9 |
| H11A—C11A—H11B | 107.9 | H11C—C11B—H11D | 107.7 |
| C11A—C12A—C13A | 111.4 (3) | C13B—C12B—C11B | 111.8 (2) |
| C11A—C12A—H12A | 109.3 | C13B—C12B—H12C | 109.3 |
| C13A—C12A—H12A | 109.3 | C11B—C12B—H12C | 109.3 |
| C11A—C12A—H12B | 109.3 | C13B—C12B—H12D | 109.3 |
| C13A—C12A—H12B | 109.3 | C11B—C12B—H12D | 109.3 |
| H12A—C12A—H12B | 108.0 | H12C—C12B—H12D | 107.9 |
| C12A—C13A—C18A | 113.5 (3) | C12B—C13B—C18B | 113.9 (2) |
| C12A—C13A—C14A | 111.6 (3) | C12B—C13B—C14B | 110.3 (2) |
| C18A—C13A—C14A | 111.2 (3) | C18B—C13B—C14B | 111.3 (2) |
| C12A—C13A—H13A | 109.4 (19) | C12B—C13B—H13B | 109.1 (17) |
| C18A—C13A—H13A | 108.9 (19) | C18B—C13B—H13B | 106.8 (18) |
| C14A—C13A—H13A | 101.5 (19) | C14B—C13B—H13B | 105.0 (18) |
| C13A—C14A—C27A | 110.2 (3) | C27B—C14B—C15B | 105.7 (2) |
| C13A—C14A—C15A | 108.9 (3) | C27B—C14B—C13B | 111.2 (2) |
| C27A—C14A—C15A | 105.7 (3) | C15B—C14B—C13B | 110.1 (2) |
| C13A—C14A—C8A | 109.0 (2) | C27B—C14B—C8B | 112.0 (2) |
| C27A—C14A—C8A | 111.7 (3) | C15B—C14B—C8B | 110.8 (2) |
| C15A—C14A—C8A | 111.2 (3) | C13B—C14B—C8B | 107.1 (2) |
| C16A—C15A—C14A | 115.5 (3) | C16B—C15B—C14B | 115.1 (2) |
| C16A—C15A—H15A | 108.4 | C16B—C15B—H15C | 108.5 |
| C14A—C15A—H15A | 108.4 | C14B—C15B—H15C | 108.5 |
| C16A—C15A—H15B | 108.4 | C16B—C15B—H15D | 108.5 |
| C14A—C15A—H15B | 108.4 | C14B—C15B—H15D | 108.5 |
| H15A—C15A—H15B | 107.5 | H15C—C15B—H15D | 107.5 |
| C17A—C16A—C15A | 109.6 (3) | C15B—C16B—C17B | 109.6 (2) |
| C17A—C16A—H16A | 109.7 | C15B—C16B—H16C | 109.7 |
| C15A—C16A—H16A | 109.7 | C17B—C16B—H16C | 109.7 |
| C17A—C16A—H16B | 109.7 | C15B—C16B—H16D | 109.7 |
| C15A—C16A—H16B | 109.7 | C17B—C16B—H16D | 109.7 |
| H16A—C16A—H16B | 108.2 | H16C—C16B—H16D | 108.2 |
| C28A—C17A—C16A | 108.4 (3) | C28B—C17B—C18B | 115.8 (2) |
| C28A—C17A—C18A | 114.7 (3) | C28B—C17B—C16B | 107.4 (3) |
| C16A—C17A—C18A | 109.7 (3) | C18B—C17B—C16B | 109.4 (2) |
| C28A—C17A—C22A | 105.6 (3) | C28B—C17B—C22B | 105.2 (3) |
| C16A—C17A—C22A | 117.2 (3) | C18B—C17B—C22B | 101.5 (2) |
| C18A—C17A—C22A | 101.2 (3) | C16B—C17B—C22B | 117.8 (2) |
| C17A—C18A—C19A | 105.1 (3) | C17B—C18B—C13B | 112.1 (2) |
| C17A—C18A—C13A | 111.2 (3) | C17B—C18B—C19B | 104.2 (2) |
| C19A—C18A—C13A | 120.2 (3) | C13B—C18B—C19B | 119.0 (2) |
| C17A—C18A—H18A | 106.5 | C17B—C18B—H18B | 107.0 |
| C19A—C18A—H18A | 106.5 | C13B—C18B—H18B | 107.0 |
| C13A—C18A—H18A | 106.5 | C19B—C18B—H18B | 107.0 |
| C20A—C19A—C18A | 116.2 (3) | C20B—C19B—C18B | 117.1 (3) |
| C20A—C19A—C21A | 110.6 (3) | C20B—C19B—C21B | 111.0 (3) |
| C18A—C19A—C21A | 104.2 (3) | C18B—C19B—C21B | 104.0 (2) |
| C20A—C19A—H19A | 115 (2) | C20B—C19B—H19B | 110 (2) |
| C18A—C19A—H19A | 105 (2) | C18B—C19B—H19B | 110 (2) |
| C21A—C19A—H19A | 104 (2) | C21B—C19B—H19B | 104 (2) |
| C29A—C20A—C30A | 120.4 (4) | C29B—C20B—C30B | 121.9 (3) |
| C29A—C20A—C19A | 124.8 (3) | C29B—C20B—C19B | 120.1 (3) |
| C30A—C20A—C19A | 114.7 (3) | C30B—C20B—C19B | 117.8 (3) |
| C22A—C21A—C19A | 105.6 (3) | C22B—C21B—C19B | 106.4 (3) |
| C22A—C21A—H21A | 110.6 | C22B—C21B—H21C | 110.4 |
| C19A—C21A—H21A | 110.6 | C19B—C21B—H21C | 110.4 |
| C22A—C21A—H21B | 110.6 | C22B—C21B—H21D | 110.4 |
| C19A—C21A—H21B | 110.6 | C19B—C21B—H21D | 110.4 |
| H21A—C21A—H21B | 108.8 | H21C—C21B—H21D | 108.6 |
| C17A—C22A—C21A | 104.4 (3) | C21B—C22B—C17B | 103.2 (2) |
| C17A—C22A—H22A | 110.9 | C21B—C22B—H22C | 115.4 (19) |
| C21A—C22A—H22A | 110.9 | C17B—C22B—H22C | 106 (2) |
| C17A—C22A—H22B | 110.9 | C21B—C22B—H22D | 109 (2) |
| C21A—C22A—H22B | 110.9 | C17B—C22B—H22D | 114.5 (19) |
| H22A—C22A—H22B | 108.9 | H22C—C22B—H22D | 109 (3) |
| C4A—C23A—H23A | 109.5 | C4B—C23B—H23D | 109.5 |
| C4A—C23A—H23B | 109.5 | C4B—C23B—H23E | 109.5 |
| H23A—C23A—H23B | 109.5 | H23D—C23B—H23E | 109.5 |
| C4A—C23A—H23C | 109.5 | C4B—C23B—H23F | 109.5 |
| H23A—C23A—H23C | 109.5 | H23D—C23B—H23F | 109.5 |
| H23B—C23A—H23C | 109.5 | H23E—C23B—H23F | 109.5 |
| C4A—C24A—H24A | 109.5 | C4B—C24B—H24D | 109.5 |
| C4A—C24A—H24B | 109.5 | C4B—C24B—H24E | 109.5 |
| H24A—C24A—H24B | 109.5 | H24D—C24B—H24E | 109.5 |
| C4A—C24A—H24C | 109.5 | C4B—C24B—H24F | 109.5 |
| H24A—C24A—H24C | 109.5 | H24D—C24B—H24F | 109.5 |
| H24B—C24A—H24C | 109.5 | H24E—C24B—H24F | 109.5 |
| C10A—C25A—H25A | 109.5 | C10B—C25B—H25D | 109.5 |
| C10A—C25A—H25B | 109.5 | C10B—C25B—H25E | 109.5 |
| H25A—C25A—H25B | 109.5 | H25D—C25B—H25E | 109.5 |
| C10A—C25A—H25C | 109.5 | C10B—C25B—H25F | 109.5 |
| H25A—C25A—H25C | 109.5 | H25D—C25B—H25F | 109.5 |
| H25B—C25A—H25C | 109.5 | H25E—C25B—H25F | 109.5 |
| C8A—C26A—H26A | 109.5 | C8B—C26B—H26D | 109.5 |
| C8A—C26A—H26B | 109.5 | C8B—C26B—H26E | 109.5 |
| H26A—C26A—H26B | 109.5 | H26D—C26B—H26E | 109.5 |
| C8A—C26A—H26C | 109.5 | C8B—C26B—H26F | 109.5 |
| H26A—C26A—H26C | 109.5 | H26D—C26B—H26F | 109.5 |
| H26B—C26A—H26C | 109.5 | H26E—C26B—H26F | 109.5 |
| C14A—C27A—H27A | 109.5 | C14B—C27B—H27D | 109.5 |
| C14A—C27A—H27B | 109.5 | C14B—C27B—H27E | 109.5 |
| H27A—C27A—H27B | 109.5 | H27D—C27B—H27E | 109.5 |
| C14A—C27A—H27C | 109.5 | C14B—C27B—H27F | 109.5 |
| H27A—C27A—H27C | 109.5 | H27D—C27B—H27F | 109.5 |
| H27B—C27A—H27C | 109.5 | H27E—C27B—H27F | 109.5 |
| O2A—C28A—C17A | 126.3 (4) | O2B—C28B—C17B | 128.7 (3) |
| O2A—C28A—H28A | 105.8 | O2B—C28B—H28C | 105.1 |
| C17A—C28A—H28A | 105.8 | C17B—C28B—H28C | 105.1 |
| O2A—C28A—H28B | 105.8 | O2B—C28B—H28D | 105.1 |
| C17A—C28A—H28B | 105.8 | C17B—C28B—H28D | 105.1 |
| H28A—C28A—H28B | 106.2 | H28C—C28B—H28D | 105.9 |
| C20A—C29A—H29A | 120.0 | C20B—C29B—H29C | 120.0 |
| C20A—C29A—H29B | 120.0 | C20B—C29B—H29D | 120.0 |
| H29A—C29A—H29B | 120.0 | H29C—C29B—H29D | 120.0 |
| C20A—C30A—H30A | 109.5 | C20B—C30B—H30D | 109.5 |
| C20A—C30A—H30B | 109.5 | C20B—C30B—H30E | 109.5 |
| H30A—C30A—H30B | 109.5 | H30D—C30B—H30E | 109.5 |
| C20A—C30A—H30C | 109.5 | C20B—C30B—H30F | 109.5 |
| H30A—C30A—H30C | 109.5 | H30D—C30B—H30F | 109.5 |
| H30B—C30A—H30C | 109.5 | H30E—C30B—H30F | 109.5 |
| C10A—C1A—C2A—C3A | −54.6 (4) | C10B—C1B—C2B—C3B | −53.5 (4) |
| C1A—C2A—C3A—O1A | −134.1 (4) | C1B—C2B—C3B—O1B | −127.3 (4) |
| C1A—C2A—C3A—C4A | 47.1 (4) | C1B—C2B—C3B—C4B | 50.4 (4) |
| O1A—C3A—C4A—C23A | 20.0 (5) | O1B—C3B—C4B—C23B | 11.8 (4) |
| C2A—C3A—C4A—C23A | −161.2 (3) | C2B—C3B—C4B—C23B | −165.9 (3) |
| O1A—C3A—C4A—C24A | −96.0 (4) | O1B—C3B—C4B—C24B | −104.3 (4) |
| C2A—C3A—C4A—C24A | 82.8 (4) | C2B—C3B—C4B—C24B | 78.0 (3) |
| O1A—C3A—C4A—C5A | 139.8 (3) | O1B—C3B—C4B—C5B | 130.8 (3) |
| C2A—C3A—C4A—C5A | −41.5 (4) | C2B—C3B—C4B—C5B | −46.9 (4) |
| C3A—C4A—C5A—C6A | 177.3 (3) | C3B—C4B—C5B—C6B | −178.0 (3) |
| C23A—C4A—C5A—C6A | −63.4 (4) | C23B—C4B—C5B—C6B | −60.2 (3) |
| C24A—C4A—C5A—C6A | 57.3 (4) | C24B—C4B—C5B—C6B | 61.0 (3) |
| C3A—C4A—C5A—C10A | 46.0 (4) | C3B—C4B—C5B—C10B | 50.2 (3) |
| C23A—C4A—C5A—C10A | 165.3 (3) | C23B—C4B—C5B—C10B | 167.9 (3) |
| C24A—C4A—C5A—C10A | −73.9 (4) | C24B—C4B—C5B—C10B | −70.9 (3) |
| C10A—C5A—C6A—C7A | −64.5 (3) | C10B—C5B—C6B—C7B | −61.5 (3) |
| C4A—C5A—C6A—C7A | 160.8 (3) | C4B—C5B—C6B—C7B | 164.0 (2) |
| C5A—C6A—C7A—C8A | 58.6 (4) | C5B—C6B—C7B—C8B | 57.8 (3) |
| C6A—C7A—C8A—C26A | 72.9 (4) | C6B—C7B—C8B—C26B | 70.7 (3) |
| C6A—C7A—C8A—C9A | −48.2 (4) | C6B—C7B—C8B—C9B | −50.5 (3) |
| C6A—C7A—C8A—C14A | −165.8 (3) | C6B—C7B—C8B—C14B | −168.5 (2) |
| C26A—C8A—C9A—C11A | 61.5 (4) | C26B—C8B—C9B—C11B | 63.4 (3) |
| C7A—C8A—C9A—C11A | 179.5 (3) | C7B—C8B—C9B—C11B | −177.7 (2) |
| C14A—C8A—C9A—C11A | −60.5 (3) | C14B—C8B—C9B—C11B | −57.8 (3) |
| C26A—C8A—C9A—C10A | −70.6 (4) | C26B—C8B—C9B—C10B | −68.5 (3) |
| C7A—C8A—C9A—C10A | 47.4 (4) | C7B—C8B—C9B—C10B | 50.4 (3) |
| C14A—C8A—C9A—C10A | 167.4 (2) | C14B—C8B—C9B—C10B | 170.3 (2) |
| C2A—C1A—C10A—C25A | −68.0 (4) | C2B—C1B—C10B—C25B | −69.9 (3) |
| C2A—C1A—C10A—C5A | 55.7 (4) | C2B—C1B—C10B—C9B | 167.9 (3) |
| C2A—C1A—C10A—C9A | 169.0 (3) | C2B—C1B—C10B—C5B | 53.5 (3) |
| C6A—C5A—C10A—C25A | −66.3 (4) | C11B—C9B—C10B—C1B | 60.4 (3) |
| C4A—C5A—C10A—C25A | 66.6 (4) | C8B—C9B—C10B—C1B | −168.8 (2) |
| C6A—C5A—C10A—C1A | 173.5 (3) | C11B—C9B—C10B—C25B | −59.2 (3) |
| C4A—C5A—C10A—C1A | −53.7 (3) | C8B—C9B—C10B—C25B | 71.6 (3) |
| C6A—C5A—C10A—C9A | 59.4 (3) | C11B—C9B—C10B—C5B | 175.1 (2) |
| C4A—C5A—C10A—C9A | −167.7 (3) | C8B—C9B—C10B—C5B | −54.1 (3) |
| C11A—C9A—C10A—C25A | −57.7 (3) | C6B—C5B—C10B—C1B | 173.1 (2) |
| C8A—C9A—C10A—C25A | 73.2 (3) | C4B—C5B—C10B—C1B | −54.0 (3) |
| C11A—C9A—C10A—C1A | 62.5 (3) | C6B—C5B—C10B—C25B | −66.9 (3) |
| C8A—C9A—C10A—C1A | −166.6 (3) | C4B—C5B—C10B—C25B | 66.0 (3) |
| C11A—C9A—C10A—C5A | 176.1 (2) | C6B—C5B—C10B—C9B | 57.9 (3) |
| C8A—C9A—C10A—C5A | −53.0 (3) | C4B—C5B—C10B—C9B | −169.2 (2) |
| C8A—C9A—C11A—C12A | 58.3 (3) | C8B—C9B—C11B—C12B | 53.0 (3) |
| C10A—C9A—C11A—C12A | −167.5 (3) | C10B—C9B—C11B—C12B | −173.7 (2) |
| C9A—C11A—C12A—C13A | −54.3 (4) | C9B—C11B—C12B—C13B | −52.1 (3) |
| C11A—C12A—C13A—C18A | −178.4 (3) | C11B—C12B—C13B—C18B | −176.5 (2) |
| C11A—C12A—C13A—C14A | 54.9 (4) | C11B—C12B—C13B—C14B | 57.5 (3) |
| C12A—C13A—C14A—C27A | 64.4 (3) | C12B—C13B—C14B—C27B | 59.7 (3) |
| C18A—C13A—C14A—C27A | −63.5 (3) | C18B—C13B—C14B—C27B | −67.7 (3) |
| C12A—C13A—C14A—C15A | 179.9 (3) | C12B—C13B—C14B—C15B | 176.5 (2) |
| C18A—C13A—C14A—C15A | 52.0 (3) | C18B—C13B—C14B—C15B | 49.1 (3) |
| C12A—C13A—C14A—C8A | −58.5 (3) | C12B—C13B—C14B—C8B | −62.9 (3) |
| C18A—C13A—C14A—C8A | 173.6 (2) | C18B—C13B—C14B—C8B | 169.7 (2) |
| C26A—C8A—C14A—C13A | −62.2 (3) | C26B—C8B—C14B—C27B | 178.3 (2) |
| C7A—C8A—C14A—C13A | 179.1 (2) | C7B—C8B—C14B—C27B | 58.9 (3) |
| C9A—C8A—C14A—C13A | 60.3 (3) | C9B—C8B—C14B—C27B | −59.7 (3) |
| C26A—C8A—C14A—C27A | 175.8 (3) | C26B—C8B—C14B—C15B | 60.6 (3) |
| C7A—C8A—C14A—C27A | 57.1 (3) | C7B—C8B—C14B—C15B | −58.9 (3) |
| C9A—C8A—C14A—C27A | −61.7 (3) | C9B—C8B—C14B—C15B | −177.5 (2) |
| C26A—C8A—C14A—C15A | 57.9 (3) | C26B—C8B—C14B—C13B | −59.6 (3) |
| C7A—C8A—C14A—C15A | −60.8 (3) | C7B—C8B—C14B—C13B | −179.0 (2) |
| C9A—C8A—C14A—C15A | −179.6 (3) | C9B—C8B—C14B—C13B | 62.4 (3) |
| C13A—C14A—C15A—C16A | −51.1 (4) | C27B—C14B—C15B—C16B | 70.0 (3) |
| C27A—C14A—C15A—C16A | 67.3 (4) | C13B—C14B—C15B—C16B | −50.2 (3) |
| C8A—C14A—C15A—C16A | −171.3 (3) | C8B—C14B—C15B—C16B | −168.5 (2) |
| C14A—C15A—C16A—C17A | 54.3 (4) | C14B—C15B—C16B—C17B | 55.1 (3) |
| C15A—C16A—C17A—C28A | 69.0 (3) | C15B—C16B—C17B—C28B | 68.1 (3) |
| C15A—C16A—C17A—C18A | −57.0 (4) | C15B—C16B—C17B—C18B | −58.4 (3) |
| C15A—C16A—C17A—C22A | −171.7 (3) | C15B—C16B—C17B—C22B | −173.5 (3) |
| C28A—C17A—C18A—C19A | 70.4 (4) | C28B—C17B—C18B—C13B | −60.6 (3) |
| C16A—C17A—C18A—C19A | −167.2 (3) | C16B—C17B—C18B—C13B | 60.9 (3) |
| C22A—C17A—C18A—C19A | −42.8 (3) | C22B—C17B—C18B—C13B | −173.8 (2) |
| C28A—C17A—C18A—C13A | −61.1 (4) | C28B—C17B—C18B—C19B | 69.4 (3) |
| C16A—C17A—C18A—C13A | 61.3 (4) | C16B—C17B—C18B—C19B | −169.1 (2) |
| C22A—C17A—C18A—C13A | −174.3 (3) | C22B—C17B—C18B—C19B | −43.9 (3) |
| C12A—C13A—C18A—C17A | 174.0 (3) | C12B—C13B—C18B—C17B | 178.0 (3) |
| C14A—C13A—C18A—C17A | −59.1 (3) | C14B—C13B—C18B—C17B | −56.5 (3) |
| C12A—C13A—C18A—C19A | 50.7 (4) | C12B—C13B—C18B—C19B | 56.2 (4) |
| C14A—C13A—C18A—C19A | 177.6 (3) | C14B—C13B—C18B—C19B | −178.4 (2) |
| C17A—C18A—C19A—C20A | 149.0 (3) | C17B—C18B—C19B—C20B | 150.4 (3) |
| C13A—C18A—C19A—C20A | −84.9 (4) | C13B—C18B—C19B—C20B | −83.9 (3) |
| C17A—C18A—C19A—C21A | 27.0 (4) | C17B—C18B—C19B—C21B | 27.5 (3) |
| C13A—C18A—C19A—C21A | 153.2 (3) | C13B—C18B—C19B—C21B | 153.2 (3) |
| C18A—C19A—C20A—C29A | −26.2 (5) | C18B—C19B—C20B—C29B | 142.0 (3) |
| C21A—C19A—C20A—C29A | 92.3 (4) | C21B—C19B—C20B—C29B | −98.8 (4) |
| C18A—C19A—C20A—C30A | 156.6 (4) | C18B—C19B—C20B—C30B | −42.1 (4) |
| C21A—C19A—C20A—C30A | −84.8 (4) | C21B—C19B—C20B—C30B | 77.1 (4) |
| C20A—C19A—C21A—C22A | −126.5 (4) | C20B—C19B—C21B—C22B | −127.4 (3) |
| C18A—C19A—C21A—C22A | −0.9 (4) | C18B—C19B—C21B—C22B | −0.6 (3) |
| C28A—C17A—C22A—C21A | −78.2 (4) | C19B—C21B—C22B—C17B | −26.1 (3) |
| C16A—C17A—C22A—C21A | 161.0 (3) | C28B—C17B—C22B—C21B | −78.0 (3) |
| C18A—C17A—C22A—C21A | 41.7 (4) | C18B—C17B—C22B—C21B | 43.0 (3) |
| C19A—C21A—C22A—C17A | −25.4 (4) | C16B—C17B—C22B—C21B | 162.4 (3) |
| C16A—C17A—C28A—O2A | −140.3 (5) | C16B—C17B—C28B—O2B | −130.4 (3) |
| C18A—C17A—C28A—O2A | −17.2 (6) | C18B—C17B—C28B—O2B | −7.8 (5) |
| C22A—C17A—C28A—O2A | 93.3 (5) | C22B—C17B—C28B—O2B | 103.3 (4) |
Hydrogen-bond geometry (Å, º)
| D—H···A | D—H | H···A | D···A | D—H···A |
| C13A—H13A···O2A | 1.04 (4) | 2.52 (3) | 3.186 (4) | 122 (2) |
| C13B—H13B···O2B | 1.02 (3) | 2.47 (3) | 3.165 (4) | 125 (2) |
| C19A—H19A···O2A | 0.95 (4) | 2.45 (4) | 3.006 (5) | 118 (3) |
| C22B—H22C···O1Bi | 1.05 (4) | 2.56 (4) | 3.567 (4) | 160 (3) |
Symmetry code: (i) −x, y+1/2, −z+1/2.
Footnotes
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: LH5603).
References
- Alakurtti, S., Bergstrom, P., Sacerdoti-Sierra, N., Jaffe, C. L. & Yli-Kauhaluoma, J. (2010). J. Antibiot. 63, 123–126. [DOI] [PubMed]
- Boryczka, S., Bębenek, E., Jastrzębska, M., Kusz, J. & Zubko, M. (2012a). Z. Kristallogr. 227, 379–384.
- Boryczka, S., Jastrzębska, M., Bębenek, E., Kusz, J., Zubko, M., Kadela, M. & Michalik, E. (2012b). J. Pharm. Sci. 101, 4458–4471. [DOI] [PubMed]
- Boryczka, S., Michalik, E., Jastrzębska, M., Kusz, J., Zubko, M. & Bębenek, E. (2011). J. Chem. Crystallogr. 42, 345–351.
- Cole, B. J. W., Bentley, M. D. & Hua, Y. (1991). Hölzforschung, 45, 265–268.
- Ding, W.-M., Jing, L.-J., Yu, T., Wang, Y. & Yan, X.-F. (2009). Acta Cryst. E65, o1982. [DOI] [PMC free article] [PubMed]
- Diouf, P. N., Stevanovic, T. & Boutin, Y. (2009). Ind. Crops Prod. 30, 297–303.
- Drebushchak, T. N., Mikhailenko, M. A., Brezgunova, M. E., Shakhtshneider, T. P. & Kuznetsova, S. A. (2010). J. Struct. Chem. 51, 798–801.
- Fuchino, H., Konishi, S., Satoh, T., Yagi, A., Saitsu, K., Tatsumi, T. & Tanaka, N. (1996). Chem. Pharm. Bull. 44, 1033–1038.
- Garcez, F. R., Garcez, W. S., Miguel, D. L. S., Serea, A. A. T. & Prado, F. C. (2003). J. Braz. Chem. Soc. 14, 461–465.
- Hase, T. A., Suokas, E. & Weckman, A. (1981). Synth. Commun. 11, 489–492.
- Hata, K., Hori, K. & Takahashi, S. (2002). J. Nat. Prod. 65, 645–648. [DOI] [PubMed]
- Kim, D. K., Nam, I. Y., Kim, J. W., Shin, T. Y. & Lim, J. P. (2002). Arch. Pharm. Res. 25, 617–620. [PubMed]
- Liu, Z., Jiang, W., Deng, Z.-W. & Lin, W.-H. (2010). J. Chin. Pharm. Sci. 19, 387–392.
- Macrae, C. F., Edgington, P. R., McCabe, P., Pidcock, E., Shields, G. P., Taylor, R., Towler, M. & van de Streek, J. (2006). J. Appl. Cryst. 39, 453–457.
- Mohamed, I. E., Choudhary, M. I., Ali, S., Anjum, S. & Atta-ur-Rahman, (2006). Acta Cryst. E62, o1352–o1354.
- Oxford Diffraction (2008). CrysAlis CCD and CrysAlis RED Oxford Diffraction Ltd, Abington, England.
- Reyes, C. P., Núnez, M. J., Jiménez, I. A., Busserolles, J., Alcaraz, M. J. & Bazzocchi, I. L. (2006). Bioorg. Med. Chem. 14, 1573–1579. [DOI] [PubMed]
- Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. [DOI] [PubMed]
- Westrip, S. P. (2010). J. Appl. Cryst. 43, 920–925.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablock(s) I, global. DOI: 10.1107/S1600536813011008/lh5603sup1.cif
Structure factors: contains datablock(s) I. DOI: 10.1107/S1600536813011008/lh5603Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report





