Abstract
Compounds that delay aging in model organisms may be of significant interest to anti-aging medicine, since these substances potentially provide pharmaceutical approaches to promote healthy lifespan in humans. We here aimed to test whether pharmaceutical concentrations of three fibrates, pharmacologically established serum lipid-lowering drugs and ligands of the nuclear receptor PPARalpha in mammals, are capable of extending lifespan in a nematodal model organism for aging processes, the roundworm Caenorhabditis elegans.
Adult C. elegans (wild-type N2 as well as two nhr-49-deficient strains, RB1716 and VC870) were maintained on agar plates and were fed E. coli strain OP50 bacteria. Bezafibrate, clofibrate, and fenofibrate were applied to the agar, respectively, to test whether they may promote longevity by quantifying survival in the presence and absence of the respective compounds.
All three fibrates extended C. elegans N2 lifespan when applied at a concentration of 10 micromolar. Bezafibrate additionally extended C. elegans N2 lifespan at concentrations of 1 micromolar and 0.1 micromolar. In strains deficient for nhr-49, a functional orthologue of the mammalian peroxisome proliferator-activated receptor alpha (PPARalpha), all three compounds were incapable of extending lifespan.
Taken together, fibrates promote C. elegans longevity in an NHR-49-dependent manner possibly by promoting mitohormesis and suggesting that these compounds may promote lifespan also in mammals.
Keywords: ageing, lifespan, healthspan, C. elegans, nematodes, pharmacology, nuclear receptors, peroxisomes, peroxisome proliferator activated receptor alpha, PPARalpha, beta oxidation
INTRODUCTION
Promotion of longevity and in particular extension of healthy lifespan (also named ‘healthspan’) is of eminent interest to most humans. Specific mutations have been shown to extend the lifespan of model organisms dramatically [1-8], while more readily available interventions, including calorie restriction, also extend life expectancy of model organisms [9, 10].
Accordingly, considerable effort has been invested to identify naturally occurring and/or pharmaceutical compounds that promote longevity in model organisms. A number of such compounds have been identified in recent years, including rapamycin [11-15], resveratrol [13, 16-19], 2-deoxy-D-glucose [20], lithium [21, 22], glaucarubinone [23], lonidamine [24], rotenone [25], and others reviewed elsewhere.
Fibrates are amphipatic carboxylic acids that are used to treat metabolic disorders, primarily hypercholesterolemia and/or hypertriglyceridemia [26]. As lipid-modifying substances they are capable of increasing HDL cholesterol levels and decreasing triglycerides and LDL in plasma [26]. Like certain fatty acids, they are agonists of the peroxisome proliferator activated receptors (PPARs). Activation of PPAR by fibrates leads to increased hydrolysis of triglycerides, stimulation of cellular fatty acid uptake and conversion to acyl-CoA derivatives, decreased synthesis of triglycerides and fatty acids as well as VLDL, and finally increased peroxisomal and mitochondrial beta oxidation [26].
PPARs are nuclear receptors that act as transcription factors. They typically heterodimerize with the retinoic X receptor (RXR) and regulate expression of genes involved in development, metabolism, and cellular differentiation after binding to their respective response elements (PPREs) [27]. PPARs regulate expression of genes involved in intra- and extracellular lipid metabolism, especially genes implicated in beta oxidation [28]. PPARalpha is a master regulator of lipid metabolism. Activation induces the expression of the liver-X-receptor and ACBA1, a transporter that mediates cholesterol efflux from macrophages [29]. Furthermore, it controls adaptive response processes to calorie restriction due to its ability to activate ketogenesis [30].
The nematodal nuclear hormone receptor 49 (NHR-49) has sequence homology to the human hepatocyte nuclear factor 4 (HNF4), but is assumed to act as the functional orthologue of mammalian PPARalpha since it shares most of the biological activities of the latter [31].
In this study we have tested whether various fibrates, namely bezafibrate, clofibrate, and fenofibrate, at pharmaceutical doses may be capable of extending the life span of the nematodal model organism C. elegans.
RESULTS
Fibrates extend C. elegans life span
By continuously exposing nematodes starting at young adult age for their entire lifespan to defined concentrations of three different fibrates, which in mammals serve as ligands for the nuclear receptor PPARalpha, we tested whether and to which extent these compounds affect C. elegans lifespan.
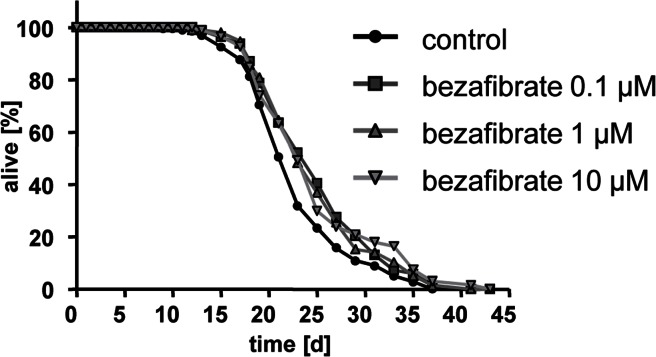
Bezafibrate extended nematodal life span at three different concentrations (0.1, 1, and 10 micromolar) (Fig. 1). The maximum observable effect on mean life span was 2.8 days which occurred at a concentration of 10 micromolar (pls. see Table 1 for details, also applies to all following life span assays).
Figure 1. Bezafibrate extends lifespan of adult C. elegans.
Life span analyses with several hundred N2 wild type nematodes each at three different concentrations of bezafibrate (0.1, 1, and 10 micromolar) versus control (0.1% DMSO).
Table 1. Results and statistical analyses of life span assays.
| Strain | Treatment | Max Life Span in Days ± SD | Mean Life Span in Days ± SD | n | p-Value versus Control | |
|---|---|---|---|---|---|---|
| N2 | 0.1 % DMSO control | 28.1 ± 1.5 | 21.6 ± 1.2 | 11 | ||
| clofibrate 0.1 μM | 25.9 | 22.0 | 1 | n. s. | 0.8248 | |
| clofibrate 1 μM | 27.2 | 22.6 | 1 | n. s. | 0.1877 | |
| clofibrate 10 μM | 33.8 | 25.1 | 1 | *** | < 0.0001 | |
| clofibrate 10 μM | 34.2 ± 0.9 | 23.0 ± 0.5 | 3 | *** | < 0.0001 | |
| bezafibrate 0.1 μM | 32.0 | 23.3 | 1 | * | 0.0138 | |
| bezafibrate 1 μM | 33.1 | 22.8 | 1 | * | 0.0137 | |
| bezafibrate 10 μM | 34.4 | 22.9 | 1 | ** | 0.0078 | |
| bezafibrate 10 μM | 32.5 ± 1.8 | 24.4 ± 0.7 | 3 | *** | 0.0002 | |
| fenofibrate 0.1 μM | 34.6 | 22.5 | 1 | * | 0.0143 | |
| fenofibrate 1 μM | 28.8 | 22.3 | 1 | n. s. | 0.1824 | |
| fenofibrate 10 μM | 32.8 | 25.6 | 1 | *** | < 0.0001 | |
| fenofibrate 10 μM | 30.2 ± 0.3 | 23.7 ± 1.1 | 3 | * | 0.0218 | |
| nhr-49 | ||||||
| ok2165 | 0.1 % DMSO control | 17.2 ± 1.2 | 12.2 ± 0.4 | 3 | ||
| bezafibrate 10 μM | 17.7 ± 0.8 | 11.5 ± 0.2 | 3 | n. s. | 0.1606 | |
| clofibrate 10 μM | 15.7 ± 0.4 | 11.0 ± 0.2 | 3 | neg. s. | < 0.0001 | |
| fenofibrate 10 μM | 13.5 ± 0.2 | 9.5 ± 0.0 | 3 | neg. s. | < 0.0001 | |
| gk405 | 0.1 % DMSO control | 16.7 ± 3.6 | 11.0 ± 0.4 | 3 | ||
| bezafibrate 10 μM | 17.7 ± 3.7 | 11.4 ± 0.8 | 3 | n. s. | 0.0679 | |
| clofibrate 10 μM | 17.5 ± 3.4 | 11.0 ± 0.3 | 3 | n. s. | 0.8962 | |
| fenofibrate 10 μM | 14.5 ± 1.6 | 10.8 ± 0.2 | 3 | n. s. | 0.3035 | |
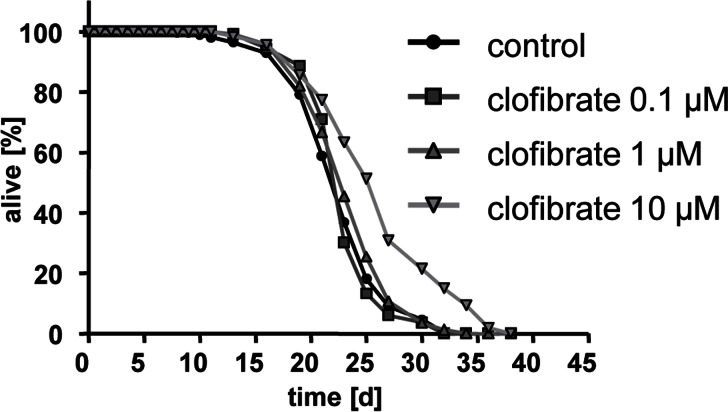
Clofibrate cause extension of C. elegans lifespan at a concentration of 10 micromolar (Fig. 2) reflected by a mean life span of 23.0 days equaling an increase of 1.4 days.
Figure 2. Clofibrate extends lifespan of adult C. elegans.
Life span analyses with several hundred N2 wild type nematodes each at three different concentrations of clofibrate (0.1, 1, and 10 micromolar) versus control (0.1% DMSO).
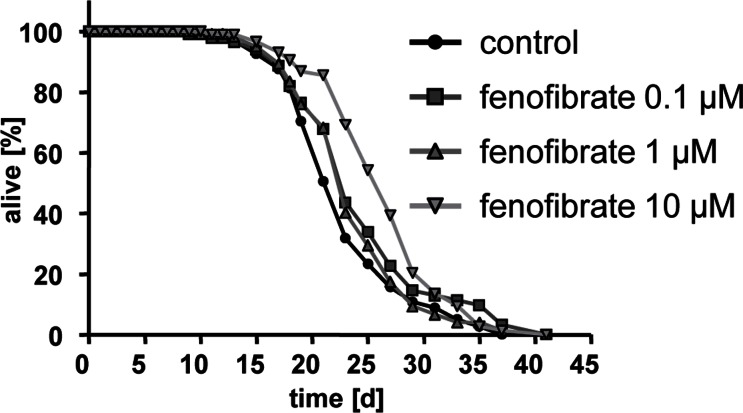
Fenofibrate was capable of promoting life expectancy at concentrations of 0.1 and 10 micomolar (Fig. 3) with the most pronounced increase in mean life span at a concentration of 10 micromolar as reflected by an increase of 2.1 days in comparison to wild-type worms.
Figure 3. Fenofibrate extends lifespan of adult C. elegans.
Life span analyses with several hundred N2 wild type nematodes each at three different concentrations of fenofibrate (0.1, 1, and 10 micromolar) versus control (0.1% DMSO).
The effects on maximum life span (80th percentile) were an extension by 4.4 days for bezafibrate (10 micomolar), an extension by 6.1 days for clofibrate (10 micromolar), and an extension by 6.5 days for fenofibrate (0.1 micromolar).
Taken together, these findings indicate that three different fibrates are capable of extending both mean and maximum lifespan of wild type C. elegans.
Life span extension through fibrates is PPA-Ralpha/NHR-49 dependent
As stated in the introductory section fibrates serve as well-accepted ligands for the mammalian PPARalpha, a nuclear receptor known to heterodimerize with the retinoid-X-receptor (RXR) to promote a number of catabolic processes. The nematodal nuclear hormone receptor 49 (NHR-49) is commonly accepted to serve as a functional orthologue of mammalian PPARalpha since it shares most of the biological activities of the latter [31].
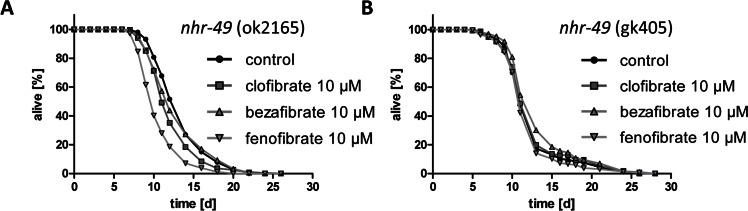
To test the hypothesis whether fibrates act as agonists of PPARalpha and therefore extent life span in C. elegans, the life span assays were repeated using two strains that lack functional nhr-49, variation ok2165 and variation gk405 (strains RB1716 and VC870, respectively). For this, the most effective life span extending fibrates concentration in wild type, 10 micromolar, was used. Consistent with the hypothesis, clofibrate, bezafibrate, and fenofibrate failed to extend nematodal life span in absence of NHR-49 (Fig. 4A and 4B).
Figure 4. Life span extension through fibrates is NHR-49/PPARalpha-dependent.
A Life span analyses with several hundred nhr-49 knockout nematodes (variation ok2165, strain RB1716) at 10 micromolar bezafibrate, clofibrate, and fenofibrate versus control (0.1% DMSO). B Life span analyses with several hundred nhr-49 knockout nematodes (variation gk405, strain VC870) at 10 micromolar bezafribrate, clofibrate, and fenofibrate versus control (0.1% DMSO).
DISCUSSION
To potentially support the ongoing search for compounds that may promote human health especially at higher age, we here show that the fibrates clofibrate, bezafibrate, and fenofibrate induce longevity in a nematodal model organism, the roundworm C. elegans. Interestingly, these effects are dependent on the nematodal orthologue of PPARalpha, NHR-49, mediating the promotion of life span.
The C. elegans PPARalpha orthologue NHR-49 induces the expression of genes involved in energy metabolism, more precisely in fatty acid beta oxidation (acs-2, ech-1), desaturation (fat-5, fat-7), transport, and synthesis of mono-methyl branched-chain fatty acids [31, 32]. It remains to be elevated, which of these mechanism are responsible for the life span extending effect of fibrates. Due to stimulation of fatty acid beta oxidation an increase in ROS formation may occur [33], which could promote formation of reactive oxygen species (ROS) in nematodes [20, 23]. This increase in ROS may act as a signal to increase stress response and antioxidant defense resulting in longevity resembling an adaptive response signaling process that was named mitochondrial hormesis or mitohormesis [20, 34, 35]. Based on the current findings, it appears feasible that fibrates act by employing a similar mechanism, especially since it is shown that PPARalpha agonists increase the expression of superoxide dismutase, a major enzyme in antioxidative defense [36].
It is unclear whether our results can be extrapolated to mammals or even humans since the current study has been performed in the model organism C. elegans. However, other compounds that have been identified by using a similar, metazoan-based approach have been shown to be effective also in rodents [11-19].
Summarizing these findings, it is likely that fibrates acting as agonists of PPARalpha to promote health and life span through modulating beta oxidation and ROS formation in a mitohormetic manner, suggesting that these substances may be potential to prevent aging and age-associated diseases also in higher organisms.
METHODS
Compounds
Bezafibrate, clofibrate and fenofibrate were obtained from Sigma-Aldrich (Munich, Germany).
C. elegans maintenance
The C. elegans strains used were Bristol N2, as well as the mutant strains nhr-49(ok2165) and nhr-49(gk405). These were obtained from CGC. Maintenance was performed as previously described [24]. The E. coli OP50 strain was used as food source.
Life span assays
Compounds were admitted to the agar as previously described [24]. E.coli OP50 bacteria were heat-inactivated for 45 minutes as previously described to avoid interference by the xenobiotic-metabolizing activity of E. coli, and used as the only food source [37].
Acknowledgments
The authors thank Beate Laube, Annett Müller and Waltraud Scheiding for excellent technical assistance. Sven Brandstädt did his parts of the experiments to fulfill parts of the requirements for his M.D. thesis work. This work is part of the research programme of the Jena Centre for Systems Biology of Ageing – JenAge funded by the German Ministry for Education and Research (Bundesministerium für Bildung und Forschung – BMBF; support code: 0315581[A-D]). Funding for this project was denied by the German Research Association (Deutsche Forschungsgemeinschaft, DFG), grant application number RI 1976/3-1.
Footnotes
The authors of this manuscript have no conflict of interests to declare.
REFERENCES
- Friedman DB, Johnson TE. A mutation in the age-1 gene in Caenorhabditis elegans lengthens life and reduces hermaphrodite fertility. Genetics. 1988;118:75–86. doi: 10.1093/genetics/118.1.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kenyon C, Chang J, Gensch E, Rudner A, Tabtiang R. A C. elegans mutant that lives twice as long as wild type. Nature. 1993;366:461–464. doi: 10.1038/366461a0. [DOI] [PubMed] [Google Scholar]
- Kimura KD, Tissenbaum HA, Liu Y, Ruvkun G. daf-2, an insulin receptor-like gene that regulates longevity and diapause in Caenorhabditis elegans. Science. 1997;277:942–946. doi: 10.1126/science.277.5328.942. [DOI] [PubMed] [Google Scholar]
- Clancy DJ, Gems D, Harshman LG, Oldham S, Stocker H, Hafen E, Leevers SJ, Partridge L. Extension of life-span by loss of CHICO, a Drosophila insulin receptor substrate protein. Science. 2001;292:104–106. doi: 10.1126/science.1057991. [DOI] [PubMed] [Google Scholar]
- Tatar M, Kopelman A, Epstein D, Tu MP, Yin CM, Garofalo RS. A mutant Drosophila insulin receptor homolog that extends life-span and impairs neuroendocrine function. Science. 2001;292:107–110. doi: 10.1126/science.1057987. [DOI] [PubMed] [Google Scholar]
- Brown-Borg HM, Borg KE, Meliska CJ, Bartke A. Dwarf mice and the ageing process. Nature. 1996;384:33. doi: 10.1038/384033a0. [DOI] [PubMed] [Google Scholar]
- Holzenberger M, Dupont J, Ducos B, Leneuve P, Geloen A, Even PC, Cervera P, Le Bouc Y. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature. 2003;421:182–187. doi: 10.1038/nature01298. [DOI] [PubMed] [Google Scholar]
- Blüher M, Kahn BB, Kahn CR. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science. 2003:299572–574. doi: 10.1126/science.1078223. [DOI] [PubMed] [Google Scholar]
- Weindruch R, Walford RL. The retardation of aging and disease by dietary restriction. Springfield, Illinois: Charles C Thomas Pub Ltd. 1988 [Google Scholar]
- Colman RJ, Anderson RM, Johnson SC, Kastman EK, Kosmatka KJ, Beasley TM, Allison DB, Cruzen C, Simmons HA, Kemnitz JW, et al. Caloric restriction delays disease onset and mortality in rhesus monkeys. Science. 2009;325:201–204. doi: 10.1126/science.1173635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vellai T, Takacs-Vellai K, Zhang Y, Kovacs AL, Orosz L, Muller F. Genetics: influence of TOR kinase on lifespan in C. elegans. Nature. 2003;426:620. doi: 10.1038/426620a. [DOI] [PubMed] [Google Scholar]
- Harrison DE, Strong R, Sharp ZD, Nelson JF, Astle CM, Flurkey K, Nadon NL, Wilkinson JE, Frenkel K, Carter CS, et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature. 2009;460:392–395. doi: 10.1038/nature08221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaeberlein M. Resveratrol and rapamycin: are they anti-aging drugs? Bioessays. 2010;32:96–99. doi: 10.1002/bies.200900171. [DOI] [PubMed] [Google Scholar]
- Robida-Stubbs S, Glover-Cutter K, Lamming DW, Mizunuma M, Narasimhan SD, Neumann-Haefelin E, Sabatini DM, Blackwell TK. TOR Signaling and Rapamycin Influence Longevity by Regulating SKN-1/Nrf and DAF-16/FoxO. Cell Metab. 2012;15:713–724. doi: 10.1016/j.cmet.2012.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller RA, Harrison DE, Astle CM, Baur JA, Boyd AR, de Cabo R, Fernandez E, Flurkey K, Javors MA, Nelson JF, et al. Rapamycin, but not resveratrol or simvastatin, extends life span of genetically heterogeneous mice. J Gerontol A Biol Sci Med Sci. 2011;66:191–201. doi: 10.1093/gerona/glq178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Howitz KT, Bitterman KJ, Cohen HY, Lamming DW, Lavu S, Wood JG, Zipkin RE, Chung P, Kisielewski A, Zhang LL, et al. Small molecule activators of sirtuins extend Saccharomyces cerevisiae lifespan. Nature. 2003;425:191–196. doi: 10.1038/nature01960. [DOI] [PubMed] [Google Scholar]
- Wood JG, Rogina B, Lavu S, Howitz K, Helfand SL, Tatar M, Sinclair D. Sirtuin activators mimic caloric restriction and delay ageing in metazoans. Nature. 2004;430:686–689. doi: 10.1038/nature02789. [DOI] [PubMed] [Google Scholar]
- Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, Prabhu VV, Allard JS, Lopez-Lluch G, Lewis K, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444:337–342. doi: 10.1038/nature05354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pearson KJ, Baur JA, Lewis KN, Peshkin L, Price NL, Labinskyy N, Swindell WR, Kamara D, Minor RK, Perez E, et al. Resveratrol Delays Age-Related Deterioration and Mimics Transcriptional Aspects of Dietary Restriction without Extending Life Span. Cell Metab. 2008;8:157–168. doi: 10.1016/j.cmet.2008.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schulz TJ, Zarse K, Voigt A, Urban N, Birringer M, Ristow M. Glucose restriction extends Caenorhabditis elegans life span by inducing mitochondrial respiration and increasing oxidative stress. Cell Metab. 2007;6:280–293. doi: 10.1016/j.cmet.2007.08.011. [DOI] [PubMed] [Google Scholar]
- Zarse K, Terao T, Tian J, Iwata N, Ishii N, Ristow M. Low-dose lithium uptake promotes longevity in humans and metazoans. Eur J Nutr. 2011;50:387–389. doi: 10.1007/s00394-011-0171-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McColl G, Killilea DW, Hubbard AE, Vantipalli MC, Melov S, Lithgow GJ. Pharmacogenetic analysis of lithium-induced delayed aging in Caenorhabditis elegans. J Biol Chem. 2008;283:350–357. doi: 10.1074/jbc.M705028200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zarse K, Bossecker A, Muller-Kuhrt L, Siems K, Hernandez MA, Berendsohn WG, Birringer M, Ristow M. The phytochemical glaucarubinone promotes mitochondrial metabolism, reduces body fat, and extends lifespan of Caenorhabditis elegans. Horm Metab Res. 2011;43:241–243. doi: 10.1055/s-0030-1270524. [DOI] [PubMed] [Google Scholar]
- Schmeisser S, Zarse K, Ristow M. Lonidamine extends lifespan of adult Caenorhabditis elegans by increasing the formation of mitochondrial reactive oxygen species. Horm Metab Res. 2011;43:687–692. doi: 10.1055/s-0031-1286308. [DOI] [PubMed] [Google Scholar]
- Schmeisser S, Priebe S, Groth M, Monajembashi S, Hemmerich P, Guthke R, Platzer M, Ristow M. Neuronal ROS Signaling Rather Than AMPK/Sirtuin-Mediated Energy Sensing Links Dietary Restriction to Lifespan Extension. Molecular Metabolism. 2013 doi: 10.1016/j.molmet.2013.02.002. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Staels B, Dallongeville J, Auwerx J, Schoonjans K, Leitersdorf E, Fruchart JC. Mechanism of action of fibrates on lipid and lipoprotein metabolism. Circulation. 1998;98:2088–2093. doi: 10.1161/01.cir.98.19.2088. [DOI] [PubMed] [Google Scholar]
- Berger J, Moller DE. The mechanisms of action of PPARs. Annu Rev Med. 2002;53:409–435. doi: 10.1146/annurev.med.53.082901.104018. [DOI] [PubMed] [Google Scholar]
- Schoonjans K, Staels B, Auwerx J. The peroxisome proliferator activated receptors (PPARS) and their effects on lipid metabolism and adipocyte differentiation. Biochim Biophys Acta. 1996;1302:93–109. doi: 10.1016/0005-2760(96)00066-5. [DOI] [PubMed] [Google Scholar]
- Chinetti G, Lestavel S, Bocher V, Remaley AT, Neve B, Torra IP, Teissier E, Minnich A, Jaye M, Duverger N, et al. PPAR-alpha and PPAR-gamma activators induce cholesterol removal from human macrophage foam cells through stimulation of the ABCA1 pathway. Nat Med. 2001;7:53–58. doi: 10.1038/83348. [DOI] [PubMed] [Google Scholar]
- Kersten S, Seydoux J, Peters JM, Gonzalez FJ, Desvergne B, Wahli W. Peroxisome proliferator-activated receptor alpha mediates the adaptive response to fasting. J Clin Invest. 1999;103:1489–1498. doi: 10.1172/JCI6223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Gilst MR, Hadjivassiliou H, Jolly A, Yamamoto KR. Nuclear hormone receptor NHR-49 controls fat consumption and fatty acid composition in C. elegans. PLoS Biol. 2005;3:e53. doi: 10.1371/journal.pbio.0030053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kniazeva M, Crawford QT, Seiber M, Wang CY, Han M. Monomethyl branched-chain fatty acids play an essential role in Caenorhabditis elegans development. PLoS Biol. 2004;2:E257. doi: 10.1371/journal.pbio.0020257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murphy MP. How mitochondria produce reactive oxygen species. Biochem. J. 2009;417:1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tapia PC. Sublethal mitochondrial stress with an attendant stoichiometric augmentation of reactive oxygen species may precipitate many of the beneficial alterations in cellular physiology produced by caloric restriction, intermittent fasting, exercise and dietary phytonutrients: ‘Mitohormesis’ for health and vitality. Med Hypotheses. 2006;66:832–843. doi: 10.1016/j.mehy.2005.09.009. [DOI] [PubMed] [Google Scholar]
- Ristow M, Schmeisser S. Extending life span by increasing oxidative stress. Free Radic Biol Med. 2011;51:327–336. doi: 10.1016/j.freeradbiomed.2011.05.010. [DOI] [PubMed] [Google Scholar]
- Inoue I, Goto S, Matsunaga T, Nakajima T, Awata T, Hokari S, Komoda T, Katayama S. The ligands/activators for peroxisome proliferator-activated receptor alpha (PPARalpha) and PPARgamma increase Cu2+,Zn2+-superoxide dismutase and decrease p22phox message expressions in primary endothelial cells. Metabolism. 2001;50:3–11. doi: 10.1053/meta.2001.19415. [DOI] [PubMed] [Google Scholar]
- Gruber J, Ng LF, Poovathingal SK, Halliwell B. Deceptively simple but simply deceptive -Caenorhabditis elegans lifespan studies: Considerations for aging and antioxidant effects. FEBS Lett. 2009;583:3377–3387. doi: 10.1016/j.febslet.2009.09.051. [DOI] [PubMed] [Google Scholar]