Abstract
Background: Decreased 25-hydroxyvitamin D [25(OH)D] concentrations have been associated with an increased prevalence and severity of asthma and a lower response to inhaled corticosteroids.
Objective: The objective was to determine the association between serum 25(OH)D concentrations and asthma prevalence, severity, and response to asthma treatment.
Design: Secondary analyses were conducted in 2 samples of adolescents 12–20 y of age: 1) NHANES 2001–2006 (n = 6487), a cross-sectional nationally representative sample of the US population, and 2) a cohort of inner-city adolescents with asthma managed prospectively for 46 wk with guidelines-based therapy in the Asthma Control Evaluation (ACE; n = 226) trial.
Results: Mean (±SD) serum 25(OH)D concentrations in the NHANES and ACE samples were lower in African Americans than in non–African Americans (NHANES: 14.9 ± 6.5 compared with 23.0 ± 8.4 ng/mL, P < 0.0001; ACE: 11.2 ± 6.9 compared with 15.8 ± 7.1 ng/mL, P < 0.0001). In the NHANES sample, mean concentrations did not differ between participants without and with asthma (African Americans: 14.9 ± 6.4 compared with 15.0 ± 6.6 ng/mL, respectively, P = 0.87; non–African Americans: 23.0 ± 8.5 compared with 23.6 ± 8.2 ng/mL, respectively, P = 0.16). In the ACE models that used either a predefined cutoff (<20 ng/mL) or linear regression, 25(OH)D concentrations showed either no relation or minor contradictory correlations with indicators of asthma severity, treatment requirements, spirometry, or atopy/inflammation.
Conclusion: In 2 samples of adolescents, overall serum 25(OH)D concentrations were low and were not consistently associated with the presence of asthma, multiple asthma characteristics, asthma morbidity, or response to treatment. The ACE trial was registered at clinicaltrials.gov as NCT0011441.
INTRODUCTION
Interest in the role of vitamin D, serum 25-hydroxyvitamin D [25(OH)D]4, and health has expanded from a focus on bone health to a variety of chronic disorders and the innate and adaptive immune systems (1). Links have been made to increased asthma prevalence (2), morbidity (3–5), and lower response to inhaled corticosteroids for those with decreased 25(OH)D concentrations (6). Unfortunately, many methodologic issues limit the interpretation of these studies. For example, none of the studies used standardized therapy to ensure that the observed variation in morbidity was not due to treatment differences and none were able to follow the participants across seasons to observe seasonal variations in asthma morbidity (7).
The objective of this report was to examine the relation between serum 25(OH)D concentrations and asthma in 2 groups of adolescents. The first group was from NHANES 2001–2006, a cross-sectional nationally representative sample of the US population. The second group was from the Asthma Control Evaluation (ACE) trial, which examined the role of the fraction of exhaled nitric oxide (FENO) as a biomarker of inflammation in asthma. In this study, conducted by the National Institute of Allergy and Infectious Diseases, Inner City Asthma Consortium, participants were extensively characterized before randomization and for the following 46 wk; their treatment was guided by a closely monitored algorithm designed to maximize control (8).
MATERIALS AND METHODS
NHANES 2001–2006 population
The national prevalence of asthma and serum 25(OH)D concentrations were obtained from NHANES 2001–2006 data. The NHANES sample consisted of 7171 participants aged 12–20 y who were eligible to have blood drawn for serum 25(OH)D concentrations. The NHANES 2001–2006 protocols were approved by the National Center for Health Statistics Institutional Review Board (www.cdc.gov/nchs/nhanes.htm). Asthma was defined as positive responses to both of the following questions: 1) Has a doctor or other health professional ever told you that you have asthma? and 2) Do you still have asthma?
ACE study population
The primary objective of the ACE study was to evaluate whether the effectiveness of the clinical management of asthma would be increased by the incorporation of a biomarker (exhaled nitric oxide) into guidelines-based care among inner-city adolescents and young adults. A total of 546 participants 12–20 y of age with asthma, who were residents of urban census tracts in which ≥20% of households had incomes below the federal poverty level, were enrolled in the ACE trial at 10 large urban research centers. Participants had a physician diagnosis of asthma for >1 y or evidence of asthma symptoms for >1 y. The protocol was approved by the Institutional Review Boards at each research center. Written informed consent was obtained from each participant or their parent or legal guardian. Adolescents 12–17 y of age provided assent.
Assessment of asthma in ACE
The ACE trial had a randomized, double-blind, parallel-group design with a 3-wk run-in period to characterize participants and to establish treatment. At the initial visit, study physicians and staff assessed the participants’ current medication regimens and clinical status. Physicians then selected 1 of 6 protocol-defined treatment steps (8). Participants were randomly assigned to either a reference group or to a biomarker FENO group and were followed for 46 wk. Participants were evaluated at 7 additional visits, at 6–8-wk intervals. Treatment was adjusted according to guideline-based algorithms (9) with or without consideration of the FENO values. All medications were provided by the study, and a 24-h telephone number was made available for medical advice.
The primary outcome in ACE evaluated at each study visit during the 46-wk treatment period was the number of maximum days with asthma symptoms during the previous 2 wk. Other outcomes included exacerbations (systemic corticosteroids or hospitalization or emergency room visit or unscheduled visit), the dose of inhaled corticosteroids needed to maintain asthma control, spirometric measurements, the Asthma Control Test (ACT), and the Composite Asthma Severity Index, which can quantify the burden of asthma—even in the presence of good control—because it accounts for the level of required treatment (10).
Measurement of 25(OH)D
NHANES 2001–2006
Serum 25(OH)D concentrations were measurements by using a radioimmunoassay kit (DiaSorin) (11).
ACE trial
Serum 25(OH)D concentrations were determined by reversed-phase HPLC in the University of Wisconsin Osteoporosis Clinical Research Laboratory. This laboratory meets proficiency standards of the vitamin D External Quality Assessment Scheme. The limit of quantitation for this assay is 3 ng/mL for 25-hydroxyvitamin D2 and 25-hydroxyvitamin D3. The intraassay CV for this assay ranges from 1.9% at a total 25(OH)D concentration of 61.5 ng/mL to 6.3% at a total 25(OH)D concentration of 14.3 ng/mL. The interassay CV is 3.2% at a 25(OH)D concentration of 59.8 ng/mL (12).
Statistical analyses
25(OH)D concentrations were categorized as <20 ng/mL because the Institute of Medicine Panel states that “…practically all persons are sufficient at serum 25OHD levels of at least 50 nmol/L (20 ng/mL) …” (13). Data are reported as means ± SDs, medians with IQRs, or percentage. For baseline demographic characteristics, differences between racial-ethnic groups were analyzed by using chi-square tests for categorical variables and t tests for continuous data. The effect of 25(OH)D concentrations on postbaseline asthma-related outcomes in ACE was analyzed with a multivariate regression (linear for continuous and logistic for dichotomous outcomes) with adjustment for intervention status, age, sex, season of measurement, and BMI (in kg/m2) percentile at baseline. Log transformations of skewed data (FENO, total IgE) were used for multivariate analyses. Separate models were run for the African American and non–African American samples, not because we found an interaction between race and 25(OH)D concentrations, but rather because the non–African Americans had significantly higher 25(OH)D concentrations and because the non–African American sample was a very heterogeneous group in NHANES, including all those not self-identifying as African American. We felt that separate models would most clearly show the risk factors for 25(OH)D concentrations for the African American sample. The separate models also allow a comparison of the risk factors between the groups. A P value <0.05 was considered statistically significant. All statistical analyses were performed by using SAS statistical software version 9.2 (SAS Institute Inc) and the R system for statistical computing version 2.15.2.
Power analyses
We believed that our sample size was large enough to be confident in our findings, even in the analyses that separated African Americans and non–African Americans. Using the sample size obtained by dichotomizing 25(OH) concentrations at 20 ng/mL, we retrospectively calculated power for the ACE study primary endpoint (ie, maximum days with asthma symptoms) for the African American and non–African American samples. Using the observed SDs for maximum days with asthma symptoms in the ACE of 1.7 for African Americans and 1.3 for non–African Americans, we had 80% power to detect a difference of 1.1 symptom days for African Americans and of 0.9 symptom days for non–African Americans. These calculations were obtained by using the observed sample sizes of 114/20 and 70/22 (</>20 ng/mL) for African Americans and non–African Americans, respectively.
RESULTS
NHANES 2001–2006 sample
Serum 25(OH) concentrations were available for 6487 of 7171 participants (90.5%) 12–20 y of age. A total of 50% were male, 28% were non-Hispanic whites, 33% were non-Hispanic African Americans, and 35% were Mexican Americans.
25(OH)D concentrations and association with asthma in NHANES 2001–2006
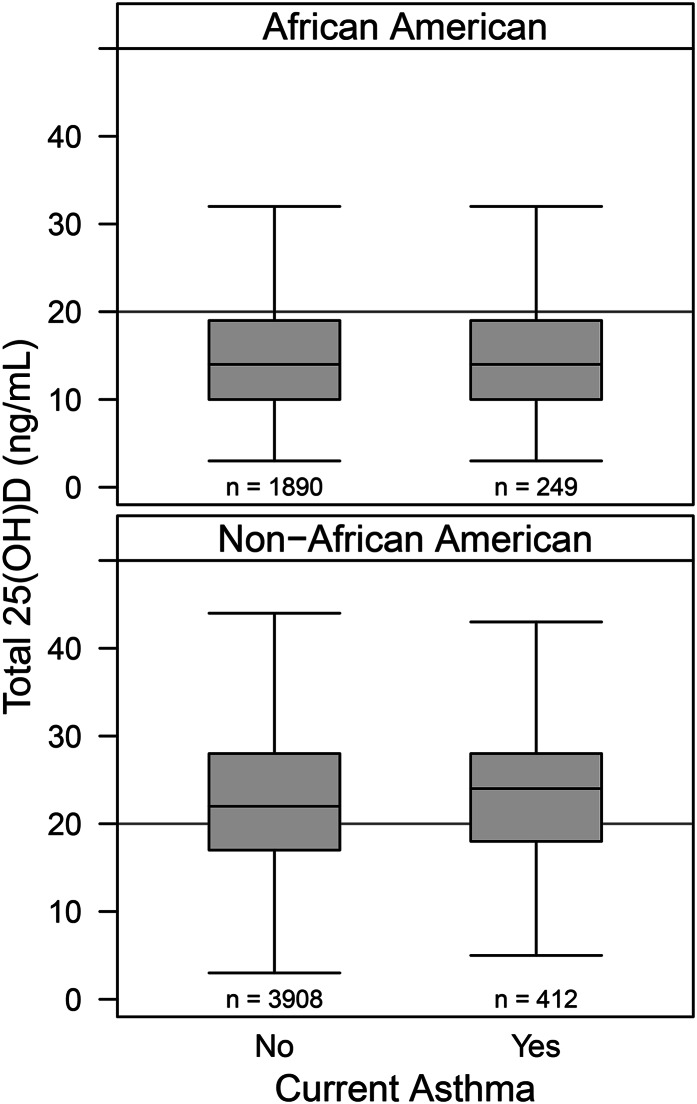
In participants aged 12–20 y, the mean (±SD) serum 25(OH)D concentration was 20.3 ± 8.7 ng/mL. Concentrations were significantly lower in African Americans than in non–African Americans (14.9 ± 6.5 compared with 23.0 ± 8.4 ng/mL, respectively; P < 0.0001). Asthma in African Americans and in non–African Americans was not found to be related to mean 25(OH)D concentrations. Nonasthmatic African Americans had 25(OH)D concentrations of 14.9 ± 6.4 ng/mL, whereas African Americans with asthma had mean 25(OH)D concentrations of 15.0 ± 6.6 ng/mL (P = 0.87). Although 25(OH)D concentrations, overall, were higher for non–African Americans than for African Americans, we again observed no difference between nonasthmatic (23.0 ± 8.5 ng/mL) and asthmatic (23.6 ± 8.2 ng/mL) non–African American participants (P = 0.16) (Figure 1).
FIGURE 1.
Serum 25(OH)D concentrations in African American and non–African American participants 12–20 y of age, in NHANES 2001–2006, with and without asthma. P > 0.05 for comparison between participants with and without asthma within each race. 25(OH)D, 25-hydroxyvitamin D.
ACE sample
Serum 25(OH)D concentrations were available for 226 of the 546 (41.4%) ACE participants. Because the 25(OH)D analyses were not a planned assay, but rather were performed on excess serum, a large number of sample had insufficient serum. However, participants with did not differ significantly from those without 25(OH)D measurements, except for being slightly younger (14.2 ± 2.0 compared with 14.6 ± 2.1 y, P = 0.024) and having a slightly higher predicted forced expiratory volume in 1 s (FEV1; 97% compared with 94%, P = 0.052) (Table 1).
TABLE 1.
Characteristics of the ACE trial population1
| 25(OH)D sample (n = 226) | Non-25(OH)D sample (n = 320) | P2 | |
| Demographics | |||
| FENO group [% (n)] | 49 (111) | 52 (165) | 0.57 |
| Age at recruitment (y) | 14.2 ± 2.03 | 14.6 ± 2.1 | 0.024 |
| Male sex [% (n)] | 52 (118) | 53 (170) | 0.83 |
| Race-ethnicity [% (n)] | 0.19 | ||
| Black | 59 (134) | 67 (213) | |
| Hispanic | 25 (56) | 22 (69) | |
| Other | 16 (36) | 12 (38) | |
| BMI percentile | 91 (68–97)4 | 87 (61–98) | 0.24 |
| Asthma morbidity5 | |||
| Maximum days with asthma symptoms | 2.0 ± 1.6 | 1.9 ± 1.7 | 0.42 |
| Composite Asthma Severity Index | 4.6 ± 2.0 | 4.8 ± 2.2 | 0.33 |
| Hospitalizations [% (n)] | 3.1 (7) | 4.1 (13) | 0.55 |
| Prednisone use [% (n)] | 37 (83) | 38 (123) | 0.68 |
| Exacerbations [% (n)] | 41 (92) | 42 (133) | 0.84 |
| Fall exacerbations [% (n)] | 15 (34) | 19 (59) | 0.22 |
| Treatment5 | |||
| Medication adherence (%) | 89 (76–99) | 89 (78–99) | 0.54 |
| Steroids prescribed (mg) | 500 (170–900) | 420 (160–1000) | 0.96 |
| Treatment level | 3.5 ± 1.9 | 3.5 ± 2.0 | 0.94 |
| Lung function5 | |||
| FEV1% | 97 ± 15 | 94 ± 15 | 0.052 |
| FEV1/FVC | 80 ± 8 | 80 ± 8 | 0.20 |
| FENO (ppb) | 29 (14–62) | 27 (15–51) | 0.52 |
ACE, Asthma Control Evaluation; FENO, fraction of exhaled nitric oxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; 25(OH)D, 25-hydroxyvitamin D.
Means, medians, and percentages were compared by using Wilcoxon's rank-sum test for continuous variables and the chi-square test for categorical variables.
Mean ± SD (all such values).
Median; first to third quartiles in parentheses (all such values).
Double-blind outcomes.
During the 46 treatment weeks, the group of participants with serum 25(OH)D concentrations experienced asthma symptoms on an average of 2.0 ± 1.6 d per each 2-wk assessment, and 41% had at least one exacerbation during the study period, including 15% who experienced an exacerbation in the fall (September to November). The median dose of controller medication (fluticasone by dry powder inhaler) was 500 μg (IQR: 170–900), with 89% adherence to treatment. The mean baseline predicted FEV1 was 97% ±15%, the mean baseline FEV1/forced vital capacity (FVC) was 80% ± 8%, and the median baseline FENO was 29 ppb (IQR: 14–62).
25(OH)D concentrations and association with asthma in ACE
The mean 25(OH)D concentration was 13.1 ± 7.3 ng/mL. The African American ACE participants had a lower mean 25(OH)D concentration than did the non–African American ACE participants (11.2 ± 6.9 compared with 15.8 ± 7.1 ng/mL, respectively; P < 0.0001]. Concentrations of 25(OH)D were higher in all samples collected during the summer in African Americans with lower BMIs and in non–African Americans in the younger age group (Table 2).
TABLE 2.
Mean serum 25(OH)D concentrations of participants in the ACE trial, stratified by race1
| African American |
Non–African American |
|||||
| Subject characteristics | n | Mean ± SD | P2 | n | Mean ± SD | P2 |
| ng/mL | ng/mL | |||||
| Overall | 134 | 11.2 ± 6.9 | 92 | 15.8 ± 7.13 | ||
| Study group | ||||||
| Non-FENO | 63 | 11.4 ± 7.0 | 0.73 | 52 | 15.4 ± 8.1 | 0.61 |
| FENO | 71 | 11.0 ± 6.8 | 40 | 16.2 ± 5.8 | ||
| Age | ||||||
| 12–15 y | 113 | 11.5 ± 7.0 | 0.21 | 66 | 16.7 ± 7.7 | 0.04 |
| 16–20 y | 21 | 9.5 ± 6.2 | 26 | 13.3 ± 4.8 | ||
| Sex | ||||||
| Male | 75 | 12.0 ± 6.7 | 0.13 | 43 | 16.1 ± 7.6 | 0.69 |
| Female | 59 | 10.2 ± 7.0 | 49 | 15.5 ± 6.7 | ||
| Summer collection time | ||||||
| No | 92 | 9.5 ± 6.2 | <0.0001 | 72 | 14.4 ± 5.8 | 0.0003 |
| Yes | 42 | 14.9 ± 7.0 | 20 | 20.8 ± 9.2 | ||
| Percentile BMI | ||||||
| <85 kg/m2 | 58 | 12.8 ± 7.0 | 0.01 | 36 | 17.2 ± 7.6 | 0.32 |
| 85–95 kg/m2 | 32 | 11.5 ± 7.4 | 18 | 14.9 ± 4.8 | ||
| >95 kg/m2 | 44 | 8.8 ± 5.6 | 38 | 14.8 ± 7.5 | ||
ACE, Asthma Control Evaluation; FENO, fraction of exhaled nitric oxide; 25(OH)D, 25-hydroxyvitamin D.
One-factor ANOVA was used to compare differences in mean serum 25(OH)D between groups.
Significantly different from African American, P < 0.0001.
We examined the relations between 25(OH)D concentrations and a range of asthma severity indicators. All models were controlled for intervention status, age, sex, season of measurement, and BMI percentile at baseline. Because the African American and non–African American samples had significantly different 25(OH)D concentrations, the models were run separately in each group. When 25(OH)D concentrations were dichotomized at a concentration of <20 ng/mL, higher concentrations of 25(OH)D were associated with more hospitalizations during the follow-up period (OR: 13.4; 95% CI: 1.6, 110.2) among African Americans and with higher ACT values (OR: 1.36; 95% CI: 0.39, 2.32) and lower total IgE (OR: −0.45; 95% CI: −0.86, −0.05) among non–African Americans. When 25(OH)D was used as a continuous variable, increasing 25(OH)D concentrations were associated with an increase in hospitalizations in African Americans, a decrease in FEV1/FVC, and an increase in the number of positive aeroallergen skin tests. No associations were found among non–African Americans. The hospitalization association was based on a relatively few occurrences (2 hospitalizations in the group with 25(OH)D concentrations <20 ng/mL compared with 3 hospitalizations in the group with concentrations ≥20 ng/mL) (Table 3). Additional analyses were performed if 25(OH)D had a nonlinear association with any of the outcomes. No significant associations were found.
TABLE 3.
Asthma and total 25(OH)D concentrations from a multivariate analysis of participants in the ACE trial1
| 25(OH)D (≥20 compared with <20 ng/mL) |
25(OH)D (continuous variable) |
|||||||
| African American (n = 134) |
Non–African American (n = 92) |
African American (n = 134) |
Non–African American (n = 92) |
|||||
| Effect (95% CI) | P | Effect (95% CI) | P | Effect (95% CI) | P | Effect (95% CI) | P | |
| Asthma morbidity | ||||||||
| Maximum days with asthma symptoms | 0.03 (−0.82, 0.88) | 0.94 | −0.42 (−1.14, 0.30) | 0.25 | 0.03 (−0.02, 0.07) | 0.24 | −0.02 (−0.07, 0.02) | 0.35 |
| Composite Asthma Severity Index | 0.36 (−0.72, 1.43) | 0.51 | −0.72 (−1.58, 0.14) | 0.10 | 0.05 (−0.01, 0.10) | 0.13 | −0.02 (−0.08, 0.03) | 0.39 |
| Asthma control test | 0.18 (−1.00, 1.35) | 0.77 | 1.36 (0.39, 2.32) | 0.006 | −0.03 (−0.09, 0.04) | 0.38 | 0.05 (−0.01, 0.11) | 0.086 |
| Asthma utilization2 | ||||||||
| Hospitalizations | 13.4 (1.6, 110.2) | 0.015 | NA3 | 1.16 (1.00, 1.34) | 0.047 | NA3 | ||
| Prednisone use | 1.22 (0.44, 3.37) | 0.71 | 1.36 (0.44, 4.16) | 0.59 | 1.01 (0.95, 1.06) | 0.84 | 0.99 (0.92, 1.06) | 0.81 |
| Exacerbations | 1.38 (0.50, 3.82) | 0.54 | 1.10 (0.36, 3.35) | 0.87 | 1.01 (0.96, 1.07) | 0.68 | 1.02 (0.95, 1.09) | 0.66 |
| Fall exacerbations | 0.59 (0.11, 3.10) | 0.54 | 0.46 (0.04, 5.50) | 0.54 | 0.96 (0.88, 1.04) | 0.34 | 0.97 (0.85, 1.11) | 0.67 |
| Treatment | ||||||||
| Inhaled corticosteroids prescribed (μg) | 100.5 (−79.9, 280.8) | 0.27 | −111.9 (−304.4, 80.6) | 0.25 | 8.17 (−1.68, 18.0) | 0.10 | 0.57 (−11.5, 12.6) | 0.93 |
| Treatment level | 0.49 (−0.47, 1.45) | 0.32 | −0.6 (−1.58, 0.39) | 0.23 | 0.05 (−0.01, 0.10) | 0.092 | 0.00 (−0.06, 0.06) | 0.94 |
| Lung function | ||||||||
| FEV1% predicted | 0.30 (−6.91, 7.52) | 0.93 | −0.45 (−7.77, 6.87) | 0.90 | −0.02 (−0.42, 0.38) | 0.93 | −0.04 (−0.49, 0.41) | 0.86 |
| FEV1/FVC | −2.53 (−6.65, 1.59) | 0.23 | 1.44 (−1.64, 4.51) | 0.36 | –0.23 (−0.45, −0.0) | 0.049 | 0.02 (−0.17, 0.21) | 0.85 |
| Atopy/inflammation | ||||||||
| Total log10 IgE (kU/L) | 0.02 (−0.27, 0.30) | 0.92 | –0.45 (−0.86, −0.05) | 0.03 | 0.01 (−0.01, 0.02) | 0.26 | −0.01 (−0.03, 0.02) | 0.64 |
| No. of positive allergen skin tests | 0.40 (−1.28, 2.08) | 0.64 | 0.90 (−2.70, 0.89) | 0.32 | 0.11 (0.02, 0.20) | 0.02 | −0.01 (−0.12, 0.10) | 0.86 |
| log10 FENO | 0.07 (−0.12, 0.25) | 0.47 | −0.07 (−0.27, 0.13) | 0.49 | 0.01 (0.0, 0.020) | 0.22 | 0.00 (−0.02, 0.01) | 0.63 |
Values were adjusted for control compared with intervention status, age, sex, season of measurement (summer compared with other), and BMI percentile at baseline. ACE, Asthma Control Evaluation; FENO, fraction of exhaled nitric oxide; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; NA, not applicable; 25(OH)D, 25-hydroxyvitamin D.
Values are ORs (95% CIs). There were a total of 5, 53, 59, and 22 (2, 30, 33, and 12) occurrences of hospitalizations, prednisone use, exacerbations, and fall exacerbations, respectively, in the African American (non–African American) sample.
Only 2 hospitalizations occurred in the non–African American group.
Comparison of 25(OH)D concentrations between ACE and NHANES
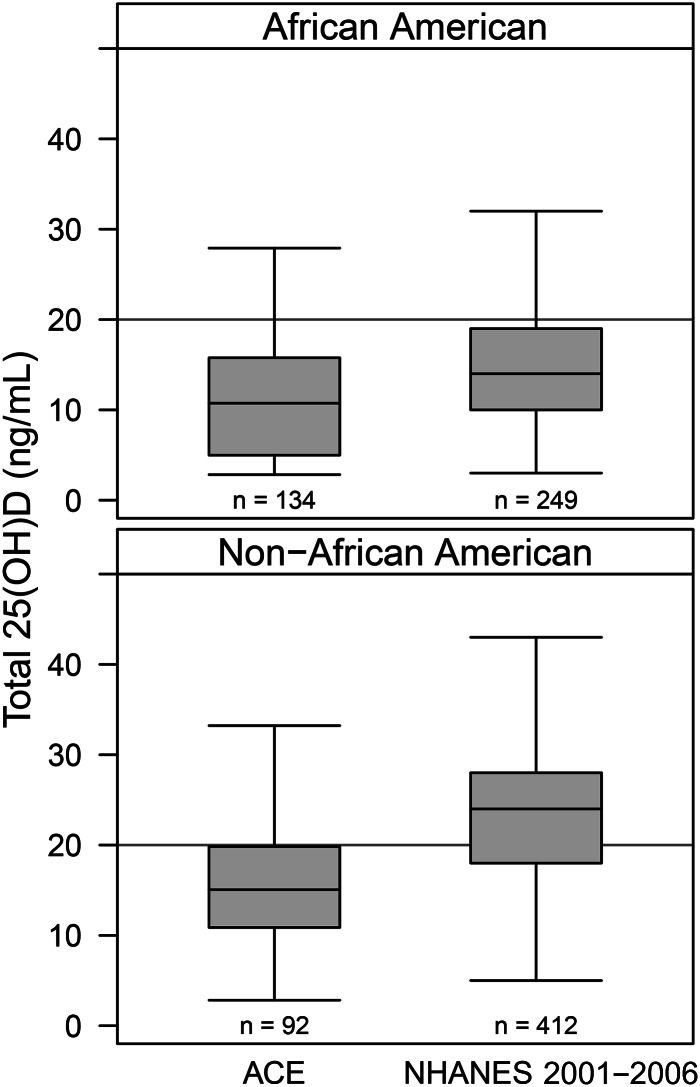
The 25(OH)D concentrations in African American adolescents with asthma in ACE were slightly lower than the values reported among the African American NHANES participants (11.2 ± 6.9 compared with 14.9 ± 6.5 ng/mL; P < 0.0001) (Figure 2). A larger difference in the 25(OH)D concentration was seen in the non–African American adolescents with asthma in ACE compared with the non–African American NHANES participants (15.8 ± 7.1 compared with 23.0 ± 8.4 ng/mL, respectively; P < 0.0001.
FIGURE 2.
Serum 25(OH)D concentrations in asthmatic African American and non–African American participants 12–20 y of age in ACE and NHANES 2001–2006. P < 0.0001 for comparison between African Americans and non–African Americans within each study. ACE, Asthma Control Evaluation; 25(OH)D, 25-hydroxyvitamin D.
DISCUSSION
The 2 studies available for these analyses offered unique opportunities to evaluate the role of serum 25(OH)D in asthma. The NHANES data provided the opportunity to evaluate asthma prevalence in an unbiased national sample, whereas the ACE data provided the opportunity evaluate asthma morbidity as a function of serum 25(OH)D in a closely followed and highly adherent cohort of asthmatic participants receiving standardized treatment over 46 wk. Analysis of the NHANES sample found no relation between serum 25(OH)D concentrations and reported asthma among African American or non–African American adolescents. The serum 25(OH)D concentrations in ACE were not consistently related to multiple measures of asthma severity, including the frequency of asthma exacerbations, the severity of symptoms, the amount of asthma therapy required to achieve control, predicted FEV1, or markers of inflammation/total IgE. The few statistically significant associations were either clinically insignificant [eg, failed to reach an ACT score difference of 3 points—the clinically important difference (14)] or, while having a large effect (OR for hospitalization: 13.4), were based on very few events [2 hospitalizations in the group with 25(OH)D concentrations <20 ng/mL compared with 3 hospitalizations in the group with concentrations ≥20 ng/mL]. In addition, no associations were observed between serum 25(OH)D concentrations and steroid use—the composite measure of asthma exacerbations—or unscheduled visits for asthma.
With respect to the relation between serum 25(OH)D concentrations and the presence of asthma, most studies have found little evidence of an association. A New Zealand birth cohort followed until 5 y of age found no relation between cord 25(OH)D concentrations and incident asthma (15). The results of cross-sectional studies have been conflicting: no association was found between 25(OH)D concentrations and the presence of asthma in NHANES III participants ≥12 y of age (1988–1994, n = 18,883) (16), whereas the 25(OH)D concentration was lower among the children with asthma than among those without asthma in African American children 6–20 y of age who participated in an asthma genetics study (2). A review of the association between 25(OH)D and asthma concluded that “..there is insufficient evidence of a causal association between vitamin D status and asthma per se…” (17).
Conversion of 25(OH)D to its active form 1,25 dihydroxyvitamin D in the respiratory tract has been associated with an increase in mediators of innate immune function (notably defensins and cathelicidin), which play a role in preventing respiratory infections (18). Because most asthma exacerbations are triggered by viral infections (19), it is reasonable to speculate that the greatest effect of 25(OH)D would be seen with exacerbations. The ACE findings showed little effect on the multiple indicators of exacerbations used in this study, except for hospitalizations, which were based on a small number of events.
Other studies that have examined 25(OH)D and the risk of exacerbations found lower concentrations associated with an increase in exacerbations. In Costa Rica, 616 children 6–14 y of age with asthma were identified as part of a family-based genetic study (3). Mean 25(OH)D concentrations in this sample were much higher than those in ACE, and concentrations were inversely associated with some markers of asthma exacerbations (asthma hospitalizations in the previous year) but not others (asthma unscheduled visits). There was no standardization of therapy in this study. Over 4 y of follow-up in 7–11-y-olds with mild to moderate persistent asthma who participated in the Childhood Asthma Management Program and who received standardized therapy (n = 1024), concentrations of 25(OH) ≤30 ng/mL were associated with increased exacerbations (hospitalizations and emergency department visits) in the group randomly assigned to receive budesonide but not in the placebo or nedocromil arms. However, the concentration of 25(OH)D was not associated with the risk of receiving a course of prednisone for an exacerbation (4). In a small study of predominantly white participants with severe (n = 36) or moderate (n = 26) asthma and nonasthmatic controls (n = 24), decreased 25(OH)D concentrations were associated with increased exacerbations (5).
Abnormalities in pulmonary flow rates and reactivity are frequently found in asthma and are predictors of asthma exacerbations (20). The ACE study found a minimal relation between an increase in 25(OH)D concentrations and a decrease in FEV1/FCV—a sensitive indicator of airway obstruction in asthma (21). In previous studies, FEV1/FVC was found to have no relation to 25(OH)D concentration in most (3, 4, 22), but not all (6), studies In a small study (n = 19) that evaluated airway remodeling in children (mean age: 11.7 y) with severe asthma (≥800 μg beclomethasone or equivalent per day), a decrease in 25(OH)D was associated with an increase in the volume fraction of airway smooth muscle but not in other measures of remodeling (reticular basement membrane thickness or smooth muscle proliferation) (5).
The amount of therapy necessary to control asthma over 46 wk was not related to 25(OH)D concentrations in the ACE trial. In the Costa Rican study, 25(OH)D concentrations were inversely related to the use of any antiinflammatory therapy (3). However, this was a generally mild group of children with asthma, because only 39% reported any use of antiinflammatory medication in the past year. Other studies found that decreasing 25(OH)D concentrations were associated with increased resistance to corticosteroids (6), especially in children (23).
The ACE analyses found conflicting associations between 25(OH)D concentrations and atopy, eg, total IgE decreased with increasing concentrations of 25(OH)D in non–African Americans, whereas the number of positive skin tests increased with increasing concentrations of 25(OH)D in African Americans. In the published literature, the reported relation between 25(OH)D concentrations and atopy/ inflammation is inconsistent. In the Childhood Asthma Management Program (4) and NHANES III (24), 25(OH)D concentrations were not associated with allergen skin test results. However, in the Costa Rican sample, the 25(OH)D concentration was found to have no relation to sensitivity to cockroach or Ascaris but to have decreased sensitivity to house dust mite (3). Total IgE has been reported to be unrelated (4), to be inversely related (6), or to have a U-shaped relation (25, 26). No significant associations between FENO and 25(OH)D have been reported (27).
Our study had many limitations. The very low serum concentrations of 25(OH)D found in our ACE trial appeared to be a threat to the generalizability of our findings with respect to asthma severity in the United States. However, when compared with concentrations found among African Americans in NHANES, the values were similar. Whereas larger differences were found between the non–African American samples, the difference in racial/ethnic composition of the groups made this comparison tenuous at best. Although the assays used in NHANES (radioimmunoassay) and ACE (HPLC) were different, they have been shown to produce similar results (12). In addition, 25(OH)D concentrations in the ACE cohort had relations to the selected variables that were similar to those reported previously, eg, lowest among African Americans (28), varied inversely with BMI (28), and higher in the spring and summer (29). In ACE, 25(OH)D concentrations were measured once; however, other studies have shown that a single measurement categorizes 25(OH)D status well. Over 12 mo of follow-up in 94 Norwegian adults, a comparison of values determined at the beginning and end of the study found that 71.3% of the sample had a change in 25(OH)D values of <4 ng/mL, and 94.7% had a change of <8 ng/mL (30). Saliba et al (31) reported a Pearson's correlation coefficient of 0.83 between 25(OH)D values measured as part of clinical care (n = 8881 individuals) 1 y apart, who had the tests performed in the same month of the year and had not used supplements in the 6 mo before the blood draw. Only 41.4% of the ACE sample had sufficient serum available for 25(OH)D concentrations. Whereas no important differences were found in the comparison of ACE participants with and without 25(OH)D measurements, it is possible that unmeasured characteristics that differed between the sample introduced bias. Finally, the 25(OH)D values in both the ACE and NHANES samples were predominately low. This limited our ability to determine whether there is a relation between higher concentrations of 25(OH)D and asthma. In summary, secondary analyses of 2 samples of African American adolescents found little to no association between serum 25(OH)D concentrations and reported asthma, asthma severity, or the response to asthma treatment.
Acknowledgments
The authors’ responsibilities were as follows—PJG, SJT, HEM, RFF, AC, EM, MK, GRB, AHL, CK, GO, JP, YR-S, WJM, CAS, and WB: designed the research; SJT, RFF, EM, MK, GRB, AHL, CK, GO, JP, YR-S, and WJM: conducted the research; NB: provided the essential reagents or the essential materials; PJG, SJT, HEM, RFF, and AC: analyzed the data or performed the statistical analyses; PJG, SJT, HEM, RFF, AC, NB, and WB: wrote the manuscript; and PJG, SJT, HEM, RFF, AC, EM, MK, GRB, AHL, CK, GO, JP, YR-S, WJM, CAS, NB, and WB: had primary responsibility for the final content. There were no real or apparent conflicts of interest with this article. The NIH played a role in the study design; the collection, analysis, and interpretation of the data; the writing of the report; and the decision to submit the manuscript. Neither GlaxoSmithKline nor Lincoln Diagnostic had any input in the study design; the collection, analysis, or interpretation of the data; the writing of the report; or the decision to submit the manuscript.
Footnotes
Abbreviations used: ACE, Asthma Control Evaluation; ACT, Asthma Control Test; FENO, fraction of exhaled nitric oxide in parts per billion; FEV1, forced expiratory volume in 1 s; FVC, forced vital capacity; 25(OH)D, 25-hydroxyvitamin D.
REFERENCES
- 1.van Etten E, Stoffels K, Gysemans C, Mathieu C, Overbergh L. Regulation of vitamin D homeostasis: implications for the immune system. Nutr Rev 2008;66:S125–34 [DOI] [PubMed] [Google Scholar]
- 2.Freishtat RJ, Iqbal SF, Pillai DK, Klein CJ, Ryan LM, Benton AS, Teach SJ. High prevalence of vitamin D deficiency among inner-city African American youth with asthma in Washington, DC. J Pediatr 2010;156:948–52 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Brehm JM, Celedon JC, Soto-Quiros ME, Avila L, Hunninghake GM, Forno E, Laskey D, Sylvia JS, Hollis BW, Weiss ST, et al. Serum vitamin D levels and markers of severity of childhood asthma in Costa Rica. Am J Respir Crit Care Med 2009;179:765–71 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Brehm JM, Schuemann B, Fuhlbrigge AL, Hollis BW, Strunk RC, Zeiger RS, Weiss ST, Litonjua AA. Serum vitamin D levels and severe asthma exacerbations in the Childhood Asthma Management Program study. J Allergy Clin Immunol 2010;126:52–8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Gupta A, Sjoukes A, Richards D, Banya W, Hawrylowicz C, Bush A, Saglani S. Relationship between serum vitamin D, disease severity and airway remodeling in children with asthma. Am J Respir Crit Care Med 2011;184:1342–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Searing DA, Zhang Y, Murphy JR, Hauk PJ, Goleva E, Leung DY. Decreased serum vitamin D levels in children with asthma are associated with increased corticosteroid use. J Allergy Clin Immunol 2010;125:995–1000 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gergen PJ, Mitchell H, Lynn H. Understanding the seasonal pattern of childhood asthma: results from the National Cooperative Inner-City Asthma Study (NCICAS). J Pediatr 2002;141:631–6 [DOI] [PubMed] [Google Scholar]
- 8.Szefler SJ, Mitchell H, Sorkness CA, Gergen PJ, O'Connor GT, Morgan WJ, Kattan M, Pongracic JA, Teach SJ, Bloomberg GR, et al. Management of asthma based on exhaled nitric oxide in addition to guideline-based treatment for inner-city adolescents and young adults: a randomised controlled trial. Lancet 2008;372:1065–72 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.National Heart, Lung, and Blood Institute. Expert Panel Report 3 (EPR-3): guidelines for the diagnosis and management of asthma - full report 2007. 28 August 2007. Available from: www.nhlbi.nih.gov/guidelines/asthma/asthgdln.htm (accessed 7 December 2009)
- 10.Wildfire JJ, Gergen PJ, Sorkness CA, Mitchell HE, Calatroni A, Kattan M, Szefler SJ, Teach SJ, Bloomberg GR, Wood RA, et al. Development and validation of the Composite Asthma Severity Index—an outcome measure for use in children and adolescents. J Allergy Clin Immunol 2012;129:694–701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Looker AC, Pfeiffer CM, Lacher DA, Schleicher RL, Picciano MF, Yetley EA. Serum 25-hydroxyvitamin D status of the US population: 1988-1994 compared with 2000-2004. Am J Clin Nutr 2008;88:1519–27 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lensmeyer GL, Wiebe DA, Binkley N, Drezner MK. HPLC method for 25-hydroxyvitamin D measurement: comparison with contemporary assays. Clin Chem 2006;52:1120–6 [DOI] [PubMed] [Google Scholar]
- 13.IOM Dietary Reference Intakes for calcium and vitamin D. Washington, DC: National Academy Press, 2011:13–4 [PubMed] [Google Scholar]
- 14.Schatz M, Kosinski M, Yarlas AS, Hanlon J, Watson ME, Jhingran P. The minimally important difference of the Asthma Control Test. J Allergy Clin Immunol 2009;124:719–23 e1 [DOI] [PubMed] [Google Scholar]
- 15.Camargo CA, Jr, Ingham T, Wickens K, Thadhani R, Silvers KM, Epton MJ, Town GI, Pattemore PK, Espinola JA, Crane J. Cord-blood 25-hydroxyvitamin D levels and risk of respiratory infection, wheezing, and asthma. Pediatrics 2011;127:e180–7 [DOI] [PubMed] [Google Scholar]
- 16.Ginde AA, Mansbach JM, Camargo CA., Jr Association between serum 25-hydroxyvitamin D level and upper respiratory tract infection in the third National Health and Nutrition Examination Survey. Arch Intern Med 2009;169:384–90 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paul G, Brehm JM, Alcorn JF, Holguin F, Aujla SJ, Celedon JC. Vitamin D and asthma. Am J Respir Crit Care Med 2012;185:124–32 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Hansdottir S, Monick MM, Hinde SL, Lovan N, Look DC, Hunninghake GW. Respiratory epithelial cells convert inactive vitamin D to its active form: potential effects on host defense. J Immunol 2008;181:7090–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Johnston SL, Pattemore PK, Sanderson G, Smith S, Lampe F, Josephs L, Symington P, O'Toole S, Myint SM, Tyrrell DAJ, et al. Community study of role of viral infections in exacerbations of asthma in 9-11 year old children. BMJ 1995;310:1225–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fuhlbrigge AL, Kitch BT, Paltiel AD, Kuntz KM, Neumann PJ, Dockery DW, Weiss ST. FEV1 is associated with risk of asthma attacks in a pediatric population. J Allergy Clin Immunol 2001;107:61–7 [DOI] [PubMed] [Google Scholar]
- 21.Bacharier LB, Strunk RC, Mauger D, White D, Lemanske RF, Jr, Sorkness CA. Classifying asthma severity in children: mismatch between symptoms, medication use, and lung function. Am J Respir Crit Care Med 2004;170:426–32 [DOI] [PubMed] [Google Scholar]
- 22.Black PN, Scragg R. Relationship between serum 25-hydroxyvitamin d and pulmonary function in the third national health and nutrition examination survey. Chest 2005;128:3792–8 [DOI] [PubMed] [Google Scholar]
- 23.Goleva E, Searing DA, Jackson LP, Richers BN, Leung DY. Steroid requirements and immune associations with vitamin D are stronger in children than adults with asthma. J Allergy Clin Immunol 2012;129:1243–51 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wjst M, Hypponen E. Vitamin D serum levels and allergic rhinitis. Allergy 2007;62:1085–6 [DOI] [PubMed] [Google Scholar]
- 25.Hyppönen E, Berry DJ, Wjst M, Power C. Serum 25-hydroxyvitamin D and IgE - a significant but nonlinear relationship. Allergy 2009;64:613–20 [DOI] [PubMed] [Google Scholar]
- 26.Rothers J, Wright AL, Stern DA, Halonen M, Camargo CA., Jr Cord blood 25-hydroxyvitamin D levels are associated with aeroallergen sensitization in children from Tucson, Arizona. J Allergy Clin Immunol 2011;128:1093–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Devereux G, Litonjua AA, Turner SW, Craig LC, McNeill G, Martindale S, Helms PJ, Seaton A, Weiss ST. Maternal vitamin D intake during pregnancy and early childhood wheezing. Am J Clin Nutr 2007;85:853–9 [DOI] [PubMed] [Google Scholar]
- 28.Dong Y, Pollock N, Stallmann-Jorgensen IS, Gutin B, Lan L, Chen TC, Keeton D, Petty K, Holick MF, Zhu H. Low 25-hydroxyvitamin D levels in adolescents: race, season, adiposity, physical activity, and fitness. Pediatrics 2010;125:1104–11 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Cole CR, Grant FK, Tangpricha V, Swaby-Ellis ED, Smith JL, Jacques A, Chen H, Schleicher RL, Ziegler TR. 25-hydroxyvitamin D status of healthy, low-income, minority children in Atlanta, Georgia. Pediatrics 2010;125:633–9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Jorde R, Sneve M, Hutchinson M, Emaus N, Figenschau Y, Grimnes G. Tracking of serum 25-hydroxyvitamin D levels during 14 years in a population-based study and during 12 months in an intervention study. Am J Epidemiol 2010;171:903–8 [DOI] [PubMed] [Google Scholar]
- 31.Saliba W, Barnett O, Stein N, Kershenbaum A, Rennert G. The longitudinal variability of serum 25(OH)D levels. Eur J Intern Med 2012;23:e106–11 [DOI] [PubMed] [Google Scholar]