Abstract
Hydrogels may be the ideal vitreous substitutes due to their wonderful physical features and biocompatibility. However, their drawbacks, short residence time, and biodegradation in vivo, have led to the fact that none of them have been approved for clinical use. In this study, we developed a novel approach of using a foldable capsular vitreous body (FCVB) injected with polyvinylalcohol (PVA) hydrogel as a vitreous substitute for long-term tamponade. The 3% PVA hydrogel that was cross-linked by gamma irradiation showed good rheological and physical properties and had no toxicity in vitro. After 180 days retention, the 3% PVA hydrogel inside FCVB remained transparent and showed good viscoelasticity without biodegradation and showed good biocompatibility and retina support. This new approach may develop into a valuable tool to improve the stability performance of PVA hydrogel as a vitreous substitute and to extend the application function of FCVB for long-term implantation in vitreous cavity.
Vitreous substitutes are essential for restoring the tamponade to the vitreous cavity after vitrectomy due to some form of vitreous retinopathy, such as diabetic retinopathy (DR), proliferative vitreoretinopathy (PVR), or retinal detachment (RD), and the research on developing a long-term ideal vitreous substitute has been carried on by scientists for about a century. However, there is no ideal material yet available to function as a permanent replacement for the vitreous. Clinically, several materials, including air, sulphur hexafluoride (SF6), perfluoropropane (C3F8), perfluorocarbon liquids, silicone oil, and heavy silicone oil are commonly used as temporary vitreous substitutes. Although these substitutes have some important characteristics, such as optical clarity and chemical inertness, they are also associated with many drawbacks and limitations, such as elevating intraocular pressure (IOP) and fast intraocular absorption1, post-operative cataract, oil emulsification, secondary glaucoma and keratopathy2,3,4,5,6, irreversible cell damage7, higher ametropia8,9, creating non-effective tamponade if the inferior retina breaks10,11, and so on.
Due to the drawbacks and limitations of the currently used substitutes, a variety of materials have been investigated in the search for an ideal artificial vitreous. Many polymers, including natural, synthetic, and semi-synthetic materials, such as collagen, hyaluronic acid, methylated collagen, gelatin hydrogels, poly (1-vinyl-2-pyrrolidinone) hydrogels, polyvinylalcohol (PVA) hydrogels, and polyacrylamide (PAA) hydrogels, have been proposed12,13,14,15,16,17,18,19,20,21. These polymers have numerous advantages in biomedical applications, good optical properties, and mechanical properties close to those of the natural vitreous. However, they have one main defect, which is their short residence time, or fast degradation in vivo12,16,17,21,22,23, as well as unknown long-term toxicity14,15,24. Consequently, none of these polymers have been approved for clinical use.
In our previous studies, we invented a new device to replace the natural vitreous with a novel foldable capsular vitreous body (FCVB)25,26,27, which consisted of a thin and vitreous-shaped capsule with a tube-valve system. After its installation into the vitreous cavity, a balanced salt solution (BSS) or silicone oil (SO) could be injected into the capsule to support the retina and even control the intraocular pressure (IOP) through the tube-valve system25,28. Also, the FCVB has good mechanical, optical, and biocompatible properties in vitro and in vivo26,27,28 and has been shown to be flexible, effective, and safe as a vitreous substitute in the treatment of severe human retinal detachment after 3-month and 12-month follow-ups29,30. Compared with the traditional method of tamponade (directly injecting the material into the vitreous cavity), this novel FCVB device has been proven to avoid many disadvantages, such as intraocular toxicity, emulsification, high IOP, and keratopathy28,31.
Therefore, in this study, we evaluated the long-term retina support effect and biocompatibility of cross-linked PVA hydrogel combined with FCVB via implanting it into the vitreous cavity of rabbit.
Results
In vitro
Rheological measurements
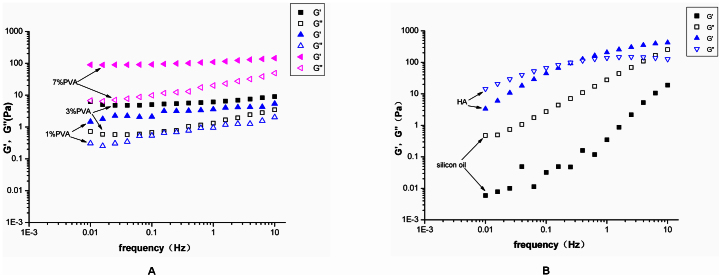
The oscillatory shear studies were performed on the PVA hydrogels, HA, and silicone oil. The storage modulus (G′) represents the elastic or solid-like component, while the loss modulus (G″) represents the viscous or liquid-like component. For all the hydrogels, the storage modulus (G′) was greater than the loss modulus (G″) at all frequencies (Fig. 1A), which showed “gel-like” behavior. Also, the plots of G′ and G″ were almost parallel, and there was no crossover in the range of frequencies used. The behaviour is like that of a type IV gel32. This class of gels would be most suitable for vitreous substitution.
Figure 1.
(A) The storage modulus G′ and the loss modulus G″ were plotted logarithmically against frequency (0.01–10 Hz at 37°C) for the 1% PVA, 3% PVA, and 7% PVA hydrogels. (B) The storage modulus G′ and the loss modulus G″ were plotted logarithmically against frequency (0.01–10 Hz at 37°C) for the silicon oil and HA.
However, for the silicon oil, the G″ is greater than the G′ for all the frequencies analysed, which means that the oil has the appearance of a viscous solution. Meanwhile, the HA behaviour is like a type III gel32. The G′ is generally higher than G″, but a G′-G″ crossover occurs at low frequencies, which suggest that the HA behaves as an entanglement network and that the sample is mainly viscous rather than elastic (Fig. 1B).
The G′ and G″ values for all the samples at 1 Hz are reported in Table 1. The values of the human vitreous are similar to those of the porcine vitreous. The average steady state moduli for the porcine vitreous are G′ = 2.8 ± 0.9 Pa and G″ = 0.7 ± 0.4 Pa33. Therefore, the 7% PVA hydrogel is not suitable as a substitute, due to the fact that its rheological parameters are much higher than porcine rheological parameters.
Table 1. Storage and loss moduli of fully hydrated 1% PVA, 3% PVA, 7% PVA hydrogels at 1 Hz compared with porcine vitreous, silicon oil and hyaluronic acid.
| Samples | G′(Pa) | G″(Pa) |
|---|---|---|
| Silicon oil | 2.4 ± 1.6 | 44.8 ± 28.3 |
| Hyaluronic acid | 155.2 ± 147.2 | 92.9 ± 49.1 |
| Porcine vitreous[*] | 2.8 ± 0.9 | 0.7 ± 0.4 |
| 1% PVA hydrogel | 3.2 ± 1.1 | 0.8 ± 0.5 |
| 3% PVA hydrogel | 6.1 ± 1.3 | 1.3 ± 0.9 |
| 7% PVA hydrogel | 106.5 ± 18.6 | 18.3 ± 12.8 |
*Nickerson CS, Park J, Kornfield JA, Karageozian H: Rheological properties of the vitreous and the role of hyaluronic acid. J Biomech 2008, 41(9):1840–1846.
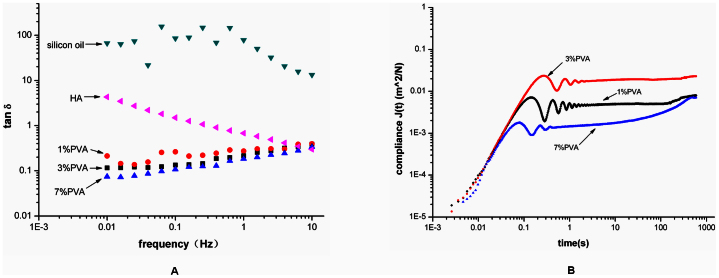
The loss tangent plots of all samples are recorded as a function of frequency (Fig. 2A). The lower the loss tangent, the higher the resilience is. Therefore, according to resilience, the hydrogels can be ranked as follows: 7% PVA > 3% PVA > 1% PVA. A material with a high resilience may perform better mechanically in a vitreous cavity32.
Figure 2.
(A) The loss tangent was plotted logarithmically against frequency (0.01–10 Hz at 37°C) for the 1% PVA, 3% PVA, and 7% PVA hydrogels and for a silicon oil, HA. (B) Creep compliance was plotted logarithmically against time for the 1% PVA, 3% PVA, and 7% PVA hydrogels.
All the hydrogels were subjected to creep analysis. The representative plots of the creep compliance are shown in Fig. 2B. The compliance of the three analysed hydrogels increases initially and tends to level off and approach a compliance plateau after intermediate periods of time. Therefore, the creep behaviours of these hydrogels are similar to those of very lightly cross-linked amorphous polymers32. This behaviour would indicate that there are prominent instantaneous elastic responses followed by substantial retarded elastic responses over time under constant shear stress. The values of the instantaneous elastic deformation of 3% PVA is higher than those of 1% PVA and 7% PVA, indicating that the 3% PVA is more elastic.
Therefore, among the three hydrogels, the 3% PVA hydrogel would be the best candidate as a vitreous substitute according to the rheological analysis of G′, G″, loss tangent, and creep behaviour.
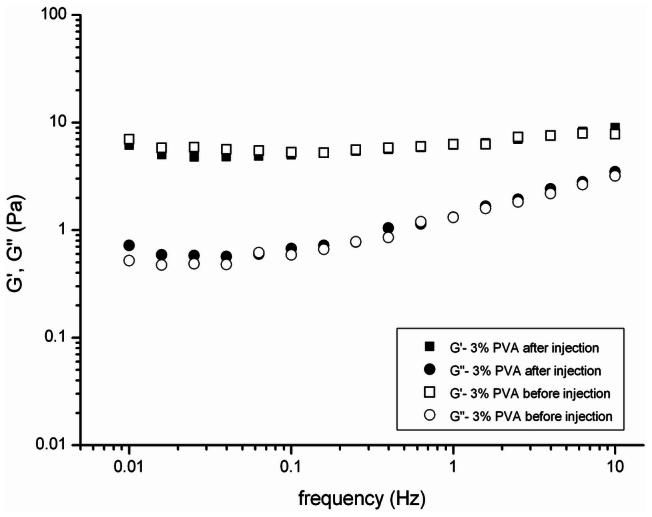
Additionally, stable mechanical properties are essential for the hydrogel after injection. In Fig. 3 the storage modulus G′ and the loss modulus G″ of the 3% PVA hydrogel before and after injection through a-19 gauge needle at 37°C are compared. The G′ value decreased from 6.3 ± 0.9 to 6.1 ± 1.3 Pa, and the G″ value increased from 1.2 ± 0.8 to 1.3 ± 0.9 Pa. The 3% PVA hydrogel shows approximately the same rheological behavior before and after the injection, indicating that the injection does not affect the cross-linked structure of the hydrogel.
Figure 3. The storage modulus G′ and the loss modulus G″ were plotted logarithmically against frequency (0.01–10 Hz at 37°C) for 3% PVA hydrogel before and after injection through a 19-gauge needle.
Physical properties of the hydrogels
The physical properties of the PVA hydrogels are summarized in Table 2, including water content, pH, density, refractive index, and light transmittance. After injection, minor fragmentation occurred in only the 7% PVA hydrogel, not in the 1% and 3% PVA hydrogels. All the hydrogels had a high water content and were transparent. The 1% and 3% PVA hydrogels had similar parameters for these physical features and were close to those of the natural human vitreous.
Table 2. Physical Properties of fully hydrated 1% PVA, 3% PVA, 7% PVA hydrogels compared with human vitreous.
| Samples | Water content | Refractive index | Light transmittance | pH | Density (g/cm3) |
|---|---|---|---|---|---|
| Human vitreous[**] | 98–99% | 1.3345–1.3348 | >90% | 7.0–7.4 | 1.0053–1.0089 |
| 1% PVA hydrogel | 98.9% | 1.3355 | 94% | 7.22 | 1.0039 |
| 3% PVA hydrogel | 98.1% | 1.3361 | 93% | 7.25 | 1.0144 |
| 7% PVA hydrogel | 93.5% | 1.3425 | 88% | 7.41 | 1.1147 |
**Baino F: Towards an ideal biomaterial for vitreous replacement: Historical overview and future trends. Acta Biomater 2011, 7(3):921–935.
Cytotoxicity tests
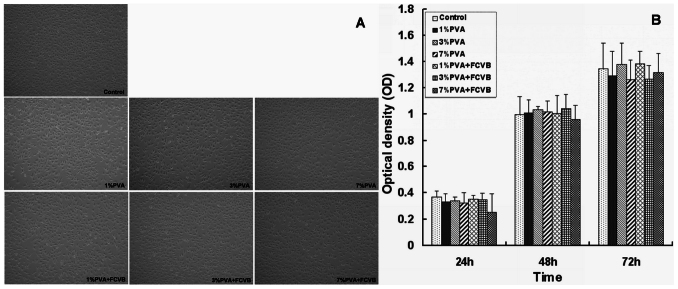
The in vitro cytotoxicity of the experimental groups (1% PVA, 3% PVA, 7% PVA, and 1% PVA + FCVB, 3% PVA + FCVB, 7% PVA + FCVB) was tested on the L929 mouse fibroblast cell. No changes in cell morphology, detachment, and membrane lysis were observed in the culture with the tested materials and the negative control (Fig. 4A). Cell cytotoxicity was evaluated via MTT assay. After 24, 48, and 72 hours of incubation, the OD values did not show a significant difference among the experimental groups (P > 0.05). Also, there was no significant difference between the experimental groups and the control group (P > 0.05) (Fig. 4B). The extractions of the experimental groups induced neither cell viability reduction nor the inhibition of cell growth, resulting in no cytotoxic effects.
Figure 4.
(A) Cytotoxicity using L929 fibroblasts cell culture in different samples extracts (1% PVA, 3% PVA, 7% PVA, 1% PVA + FCVB, 3% PVA + FCVB, and 7% PVA + FCVB). No changes in cell morphology were observed in any groups (200×). (B) MTT assay at 24, 48 and 72 hours. No significant difference among sample extracts as compared to the negative control (P > 0.05) was observed.
To sum up, the 3% PVA hydrogel had good rheological properties and good biocompatibility in vitro, so it was selected as a vitreous substitute for further analysis of the long-term biocompatibility and residence time in vivo.
In vivo
Slit lamp
Anterior chamber inflammation was visible in all groups postoperatively. However, the inflammation subsided on the third day after the operation in the BSS group and in the PVA group. Although eyes of PVA + FCVB group had relatively severe inflammation (fibrinous exudation) in the anterior chamber, they recovered within seven days with intensive anti-inflammatory treatments. With the exception of cataracts, no serious complications, such as corneal opacity, keratopathy, or posterior synechia, were observed over 180 days (Fig. 5).
Figure 5. Ninety days and one hundred eighty days follow-up examinations using a slit-lamp.
(A1–A1′) The anterior segment was shown to be normal after 90 days in the BSS group. (A2–A2′) The anterior segment was shown normal after 180 days in the BSS group; (B1–B1′) No inflammation reaction or other disease emerged in the anterior segments of the eyes after 90 days in the PVA group, except for a slight complicated cataract. (B2–B2′) The complicated caratact was aggravated after 180 days in the PVA group. (C1–C1′) The eye was aphakic without any abnormalities in cornea or anterior chamber after 90 days in the PVA + FCVB group. (C2–C2′) The cornea and anterior chamber of the eye remained very clear after 180 days in the PVA + FCVB group.
The groups showed varying degrees of lens opacity. In the BSS group, one of six eyes developed cataracts during the 180 days of observation. In the PVA group, one eye and two eyes developed cataracts at 90 and 180 days, respectively. In the PVA + FCVB group, lensectomy was performed during par plana vitrectomy (PPV) surgery. Our prior study28 revealed that severe cataracts occurred most frequently in FCVB implantated eyes and that this led to a failure to observe the fundus. In order to avoid lesions from a second surgery, the lens and vitreous were removed at the same time during vitrectomy.
Funduscopic examination
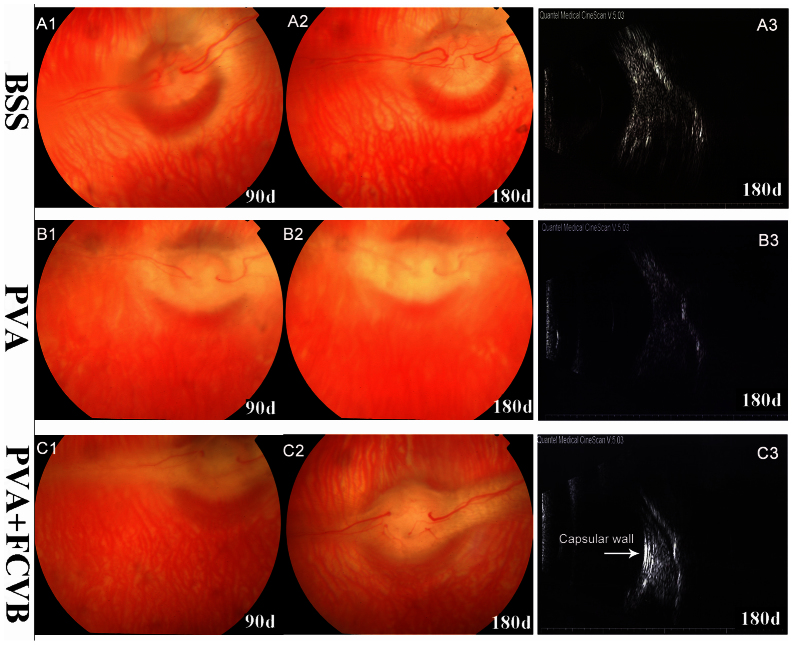
Funduscopic examination revealed that there was no evidence of vitritis, uveitis, retinitis, endophthalmitis, vitreous hemorrhage, or retina detachment in the rabbit eyes of all groups (Fig. 6). The retina and the optic nerve appeared to be normal. In the BSS group and the PVA + FCVB group, the vitreous cavity remained optically clear on postoperative day 90 and 180. However, in the PVA group, though the eyes showed clarity in their vitreous cavities on postoperative day 90, the vitreous cavities appeared to be relatively blurry on postoperative day 180 due to the complication of cataracts.
Figure 6. Postoperative posterior fundus photograph of the three groups' eyes.
All eyes showed clarity in the vitreous cavities, except that the eyes of PVA group appeared to be mildly blurry after 180 days due to complicated cataracts. B-scan ultrasonography showed all eyes' had good retina attachment and a smooth curve epiretinal echo enhancement in the FCVB eye (white arrow), indicating that the FCVB has a good retina-support function.
B-scan ultrasonography
The B-scan ultrasonography showed no retinal detachment in any groups during follow-up. In the PVA + FCVB group, slightly reflective signals of a capsule-like membrane were observed in the vitreous cavity, as shown in Fig. 6-C3. The FCVB injected with PVA hydrogel was apparently in good contact with the inner retina and can support it well.
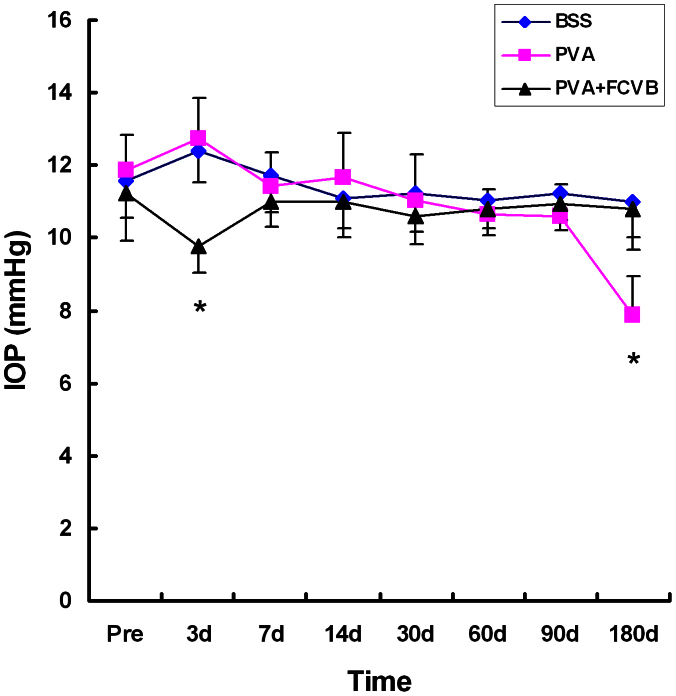
IOP
The changes in the IOP after operation are shown in Fig. 7. A downward trend was observed at postoperative day 3 in the PVA + FCVB group, which may be due to a leakage of the aqueous humor at the incision for the FCVB implantation before the incision healed. There were upward trends in the IOP after 3 days in the other two groups, but the IOP reached a plateau level at 7, 14, 30, 60, and 90 days. No statistically significant differences were found in the IOP among the three groups preoperatively or at 7, 14, 30, 60, and 90 days postoperatively, but a significant difference was found after 180 days. The IOP of the PVA group was significantly decreased at 180 days (P < 0.05). This may be due to the fact that some of the PVA hydrogel was dissolved in the vitreous cavities.
Figure 7. Intraocular pressure changes during the 180-day follow-up.
On postoperative day 3, an increasing tendency in the BSS group and in PVA group and a decreasing tendency in the PVA + FCVB group were observed, and the differences were significant among the three groups (*P < 0.05). No significant elevation of intraocular pressure was observed after 7, 14, 30, 60, and 90 days among the three group (P > 0.05).
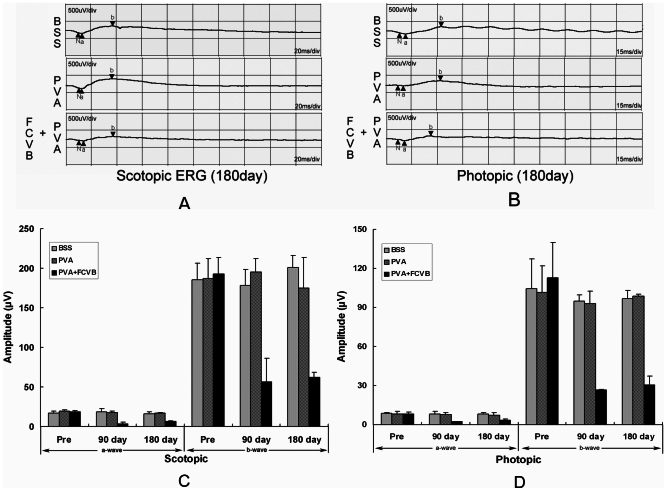
Electroretinography
Full-field ERG measurements were obtained pre- and post-operatively in all eyes (Fig. 8). In the BSS group and PVA groups, the ERG recordings appeared to be similar in terms of the a-wave and b-wave amplitudes before and after operation. There were no significant differences between the BSS group and the PVA group eyes in terms of a- or b-wave amplitudes at postoperative day 90 (P > 0.05) and day 180 (P > 0.05). By contrast, in the PVA + FCVB group, a decrease in a-wave and b-wave amplitudes was evident postoperatively, and the differences were significant when compared to the BSS group or PVA group at postoperative day 90 (P < 0.05) and day 180 (P < 0.05).
Figure 8.
Scotopic ERG waveforms (A) and the photopic ERG waveforms (B) of the three groups after 180 days, as well as the amplitude of the ERG after operation. (C) Change in the amplitude of the scotopic ERG after 90 days and 180 days. The decrease in a-wave and b-wave amplitudes was significant in the PVA + FCVB group after 90 days and 180 days as compared to the other two groups (P < 0.05). (D) Change in the amplitude of the photopic ERG after 90 days and 180 days. The decrease in the a-wave and b-wave amplitudes was significant in the PVA + FCVB group after 90 days and 180 days as compared to the other two groups (P < 0.05).
Histological findings
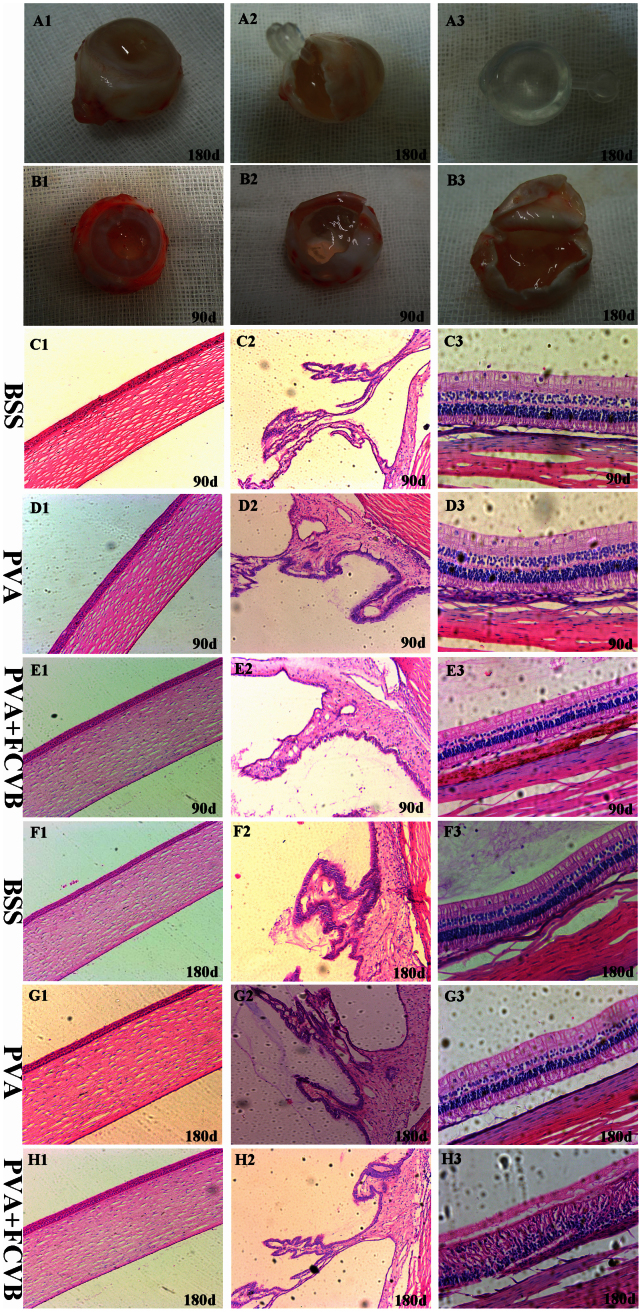
The eyes were enucleated at postoperative day 90 and day 180. In the PVA + FCVB group, a gross examination of eye specimens showed that the capsular wall of the FCVB could fit perfectly with the retina in the vitreous cavity and that the FCVBs injected with 3% PVA hydrogel remained transparent and well-rounded (Fig. 9 A1–A3). In the PVA group, the vitreous cavities were still filled with 3% PVA hydrogel at postoperative day 90. Also, the transparency of the 3% PVA hydrogel was still very clear, and the viscoelasticity did not seem to have obviously changed (Fig. 9 B1–B2). However, the vitreous cavities were filled with 3% PVA hydrogel and water-like solution at postoperative day 180, and some of the 3% PVA hydrogel was dissolved and degenerated (Fig. 9 B3). Although the 3% PVA hydrogel remained transparent and showed no apparent fragmentation, the viscoelasticity appeared to be poorer than it was before implantation.
Figure 9. Appearance of the eyeball specimens in the PVA + FCVB group and the PVA group.
(A1–A3) The FCVB-supported retina operates perfectly in the vitreous cavity after 180 days, and the FCVB injected with 3% PVA hydrogel was clear and transparent. (B1–B3) The vitreous cavity in the PVA group was still full of 3% PVA hydrogel after 90 days, and the 3% PVA hydrogel remained very clear and transparency. (C1–H2) Histology revealed the normal structure and cell morphology of the cornea, ciliary body, and retina in the three groups' eyes after 90 days and 180 days. H3: Retinal disorder was seen in the PVA + FCVB group eyes after 180 days. The retina displayed an aggregation of the inner nuclear layer and the outer nuclear layer and a thinning of the ganglion cells layer. (H&E staining: conera and ciliary body, ×100; retina, ×200).
The histological examination of H&E-stained retinal sections from the BSS group and PVA group eyes showed that the integrity of the retinal layers was good and that no loss of tissue was evident. There was no evidence of pathological changes, such as deformations, degeneration, or inflammation (Fig. 9 C3, D3, F3, G3). In the PVA + FCVB group eyes terminated on postoperative day 90, the H&E-stained sections displayed a normal retinal morphology (Fig. 9 E3). In eyes terminated at day 180, retinal disorder was seen. The retinas displayed an aggregation of the inner nuclear layer and the outer nuclear layer and a thinning of the ganglion cells layer (Fig. 9 H3). No signs of inflammation were seen. Additionally, in all three groups, there were no pathological changes in other parts of the eye, including the cornea and ciliary body.
Oscillatory shear measurements after in vivo experiments
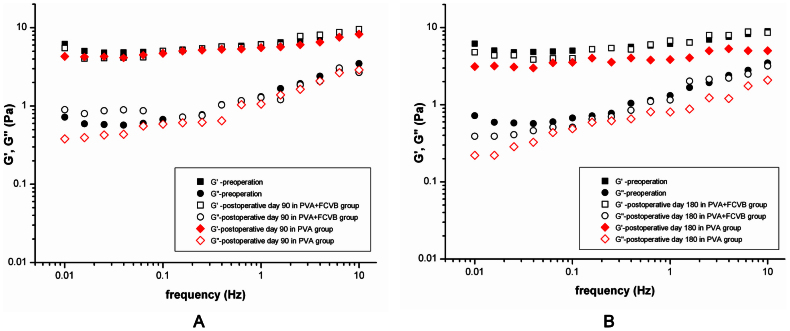
After sacrifice and enucleation, the 3% PVA hydrogel of the vitreous cavity in the PVA group and the PVA + FCVB group was collected and subjected to oscillatory shear measurements, as described before. The mechanical spectrum of the hydrogels is shown in Figure 10. In the PVA + FCVB group, on postoperative day 90 and day 180, the G′ and G″ values remained similar to those before surgery. In the PVA group, though a mild decrease in the G′ and G″ values was observed at postoperative day 90, a conspicuous decrease was observed at postoperative day 180 as compared to preoperative values. The G′ decreased from 6.1 ± 1.3 to 3.9 ± 0.8 Pa and G″ decreased from 1.3 ± 0.9 to 0.8 ± 0.5 Pa. This indicated that the 3% PVA hydrogel had undergone obvious biodegradation in the vitreous cavity after a 180-day retention.
Figure 10.
(A) Comparison of the dynamic viscoelastic moduli for the 3% PVA hydrogel before and after 90 days tamponade. (B) Comparison of the dynamic viscoelastic moduli for the 3% PVA hydrogel before and after 180 days tamponade.
Discussion
Our study demonstrated that the FCVB injected with PVA hydrogel as a vitreous substitute had good biocompatibility and retina support function in the vitreous cavity after a long-term (180 days) tamponade. This revealed that the new approach of using FCVB combined with hydrophilic hydrogels, like PVA, may yield an ideal artificial vitreous substitute for long-term vitreous replacement. This new approach greatly decreased the biodegradation of PVA hydrogel and improved its stability performance as a vitreous substitute.
An ideal vitreous substitute should mimic the native human vitreous in both form and function. It should have similar viscoelastic properties to the native vitreous in order to maintain a physiologic-range intraocular pressure and support the retina in a proper position15. The substitute should be also be clear and transparent. It should be stable, permanent, and biocompatible, without any toxic reactions during long-term use9. Meanwhile, the ideal substitute would be easily manipulable during surgery and injectable through a small syringe23.
Currently, only silicone oil is accepted as a long-term vitreous replacement. However, silicone oil has many disadvantages as a vitreous substitute, such as secondary glaucoma, cataract formation, hyperopic shift, oil emulsification, keratopathy, and in cases of noneffective tamponade, the inferior retina breaks2,3,4,5,6,8,9,10,11. Short-term persistence of a vitreous substitute in a vitreous cavity will not be sufficient efficacy in the treatment of complicated retinal detachments or retinal detachments with proliferative vitreoretinopathy. Therefore, researchers have been trying to find an ideal alternative material that may be left safely in the vitreous cavity as a long-term tamponade. The hydrogels (e.g., PVA, PAA) can closely mimic the light transmittance of the natural vitreous humour, as well as its physical and mechanical properties, and may therefore be the best candidates for vitreous substitution10. However, to our knowledge, they have the primary drawbacks of a short residence time and biodegradation in vivo.
The PVA hydrogel is deemed to be one of the most promising candidates for vitreous substitution due to its optical properties, rheological features, and long-term biocompatibility10. In previous studies14,15,24,34,35, the PVA hydrogel has been confirmed to have excellent optical properties and safety in vitro or in vivo, but further studies need to be performed in order to show their retention time, mechanical properties, and ability to reattach the retina within the eye.
In this study, we found that the 3% PVA hydrogel had proper rheological behavior and similar physical properties (including water content, pH, density, refractive index, and light transmittance) to the human vitreous and had good biocompatibility, without any toxic reactions. However, there appeared to be obvious biodegradation, along with a decrease of viscoelastic properties, after a long-term (180 days) tamponade in vivo. By contrast, when the PVA hydrogel was injected into the FCVB as a vitreous substitute, it not only showed biocompatibility, transparency, and retina support function but also had good rheological features without biodegradation at 180 days follow-up. At postoperative day 180, some of the hydrogels were obviously absorbed in the PVA group eyes, and their G′ and G″ values had significantly decreased as compared to preoperative level or those of the PVA + FCVB group. In the PVA + FCVB group, the G′ and G″ values of the PVA hydrogel remained similar to their preoperative values, without any obvious absorption after 180 days tamponade. The decrease of G′ and G″ (lost of viscoelasticity) may due to the breakage of crosslinking points, caused by biodegradation. Our previous studies demonstrated that the FCVB had cellular barrier function and could avoid silicone oil emulsification and migration28,31. Therefore, the FCVB could also serve as an isolator in order to avoid PVA hydrogel biodegradation or absorption and displayed better all-round retina support.
Our study revealed biocompatibility and relative stability of PVA hydrogel, and fewer complications were found in the PVA group during 90 days follow-up. The results were similar to those of previous research projects15,24.
For application during vitrectomy, the hydrogel should be injectable through a needle. During injection, the molecular chains of the polymer will be subjected to an external shear stress, which may result in the massive mechanical breakage of the crosslinks, inducing a significant loss of elasticity (G′ decreases), causing the hydrogel to become more fluid-like and viscous. Our rheologic experiments showed that after their injection through a 19-gauge needle, the storage modulus G′ and the loss modulus G″ of the 3% PVA hydrogel were approximately equivalent to those before injection. This indicated that the injection did not affect the network structure of the 3% PVA hydrogel and that it had similar rheological behavior before and after injection.
Previous study showed that the fragmentation of a PVA hydrogel in the vitreous cavity and the resulting muddiness of the vitreous cavity occurred after the implantation of the PVA hydrogel24. However, in our research, because of the moderate viscosity of the 3% PVA hydrogel and the sufficient removal of non-cross-linked PVA remnants, no fragmentation occurred, and the vitreous cavity remained transparent until enucleation.
In our previous study28, we found that most of FCVB-implanted eyes developed cataracts after a long-term tamponade. In order to better observe fundus, we had to perform lensectomy which lead to secondary lesion. Therefore, in this study, lensectomy was performed during vitrectomy in the PVA + FCVB -treated eyes.
No significant changes in IOP were observed after 7, 14, 30, 60, and 90 days, but there was an increasing IOP at 3 days after PVA hydrogel and BSS implantation, which may have resulted from anterior chamber inflammation after the operation. In contrast, the IOP in the PVA + FCVB group decreased at 3 days after operation. This may have been due to a leakage of the aqueous humor at the incision for the FCVB implantation. Meanwhile, a significant decrease in IOP was observed at 180 days after PVA hydrogel direct implantation, which may have occurred because of hydrogel degradation and absorption.
The ERG demonstrated that there were no significant differences between the amplitude of the PVA group and the BSS group during 180 days follow-up period. However, in the PVA + FCVB group, there were significant reductions of the a- and b-wave amplitudes in both dark- and light-adapted ERGs during the observation period. This was perhaps due to the insulator material of the FCVB, which could hinder current conduction.
In the PVA + FCVB-implanted eyes, a histopathologic examination showed that the structure of retina was intact without obvious pathological changes on postoperative 90 days. However, retinal disorder and proliferation were observed at 180 days after implantation, which may have been caused by a long-term FCVB capsule-induced mechanical pressure to the retina. Therefore, in future study, we further improve FCVB biocompatibility via hydrophilic modification, and select a more appropriate target intraocular pressure to avoid the retinal mechanical damage. Meanwhile, the retinal subcellular structures, haemodynamics and oxyhemoglobin saturation should be evaluated after a long-term FCVB implantation.
In conclusion, the FCVB tamponade with 3% PVA hydrogel seems to have good stability and transparency in the vitreous cavity for at least 180 days. The FCVB can safely prolong the PVA hydrogel retention time and effectively improve its stability performance in the vitreous cavity. Also this novel approach may indicate that the FCVB is suitable for other hydrogels (e.g. PVP, PAA) tamponade. Moreover, many nanometer-wide apertures exist in the FCVB, and drugs can be released from the FCVB36,37,38,39,40, so a drug may be added to the hydrogels, providing clinicians with a slow-release drug device. Therefore, this novel approach may develop into a new ideal vitreous substitute and open new options for the prevention and treatment of vitreous retinal diseases.
Methods
In vitro experiments
Preparation of PVA hydrogel
All of solvents used in this experiment were of analytical grade. Poly-vinyl alcohol (PVA) (Sigma Aldrich, St. Louis, MO) was purchased and used without further purification. Three concentrations (1%, 3%, and 7% w/v) of PVA solution were prepared in ultra-purified water and were stirred until fully dissolved at 80°C. Then, the solution was irradiated by γ-rays (7 kGy, Co60) to form a hydrogel. The hydrogel obtained was dialysed in distilled water for 48 hours to remove the remnants of the non-cross-linked PVA.
Rheological measurements
The rheological analyses were carried out at 37 ± 0.1°C on a strain-controlled ARES-RFS rheometer (TA Instruments Inc., New Castle, DE), using a cone and plate geometry of 50 mm in diameter and a cone angle of 0.04 rad. The cross-linked hydrogels were injected onto the plate through a 19-gauge needle.
Dynamic strain sweep tests were performed on the hydrogels at a 1.0 Hz oscillation frequency to discover which strain amplitude was in the linear viscoelastic region14. The mechanical properties of PVA hydrogels were studied by analysing the storage modulus (G′) and the loss modulus (G″) according to the frequency of the oscillatory shear stress. A dynamic frequency sweep test was conducted at a strain amplitude γ0 = 1.0% over an oscillating frequency range of 10 to 0.01 Hz14.
The relative contribution of the elastic and viscous properties can be quantified by the loss tangent (tanδ), which is the ratio of the loss to the storage modulus (tanδ = G″/G′). The higher the tanδ, the more liquid-like the sample, with a value of 1 considered to be the threshold between liquid and gel behavior. Also, because the sufficient resilience of the material, when subjected to shear stress, is also desirable in a vitreous substitute, this parameter was estimated by recording the loss tangent. The resilience (R) is an inverse measure of the damping property and is usually estimated as R = 1 − 2π tanδ32.
In a creep experiment, a constant shear stress (σ0) of 0.1 Pa was imposed on the hydrogel samples for a nominal creep time of 500 s. In viscoelastic materials, the strain (γ) response is linear. The ratio γ/σ0 is called the creep compliance J(t)32.
The same analyses were also performed on a commercial silicone oil (Oxane 5700; Bauch&Lomb, Rochester, NY) and a hyaluronic acid (HA) (Medical Hyaluronan Gel; Iviz, Shangdong, China).
Water content
The hydrogel samples (10 mg) were immersed in PBS in a thermostatic bath at 37°C until equilibrium was reached. At equilibrium swelling, the water content of the hydrogel was determined by the following formula: W.C. = (Ws − Wd/Ws) × 100 (Ws: weight of swollen hydrogel, Wd: weight of dried hydrogel)32.
Refractive index
The Abbe refractometer (WYA-2W, Lumsail Industrial Inc., Shanghai, China) was used to measure the refractive index of each hydrogel sample. The temperature of the testing was kept at 37°C.
Transmission
The transmission of light was measured using a UV/VIS spectrophotometer (DU800; BeckmanCoulter Inc., Brea, CA) at 37°C. Distilled water was used as a blank, and light transmission was measured over the 380–700 nm range.
pH value
The pH was measured by using a digital pH probe (Thermo Fisher Scientific Inc., Waltham, MA).
Cytotoxicity
The experiment groups included 1%, 3%, and 7% (w/v) PVA hydrogels and a combination of FCVBs that were injected with 1%, 3%, and 7% (w/v) PVA hydrogels. These groups were defined as 1% PVA, 3% PVA, 7% PVA, 1% PVA + FCVB, 3% PVA + FCVB, and 7% PVA + FCVB. The extraction medium was prepared by incubating the varying samples (1% PVA, 3% PVA, 7% PVA, 1% PVA + FCVB, 3% PVA + FCVB, and 7% PVA + FCVB) with standard culture medium (Dulbecco's modified Eagle's medium with 10% fetal calf serum) for 72 hours at 37°C. The extraction medium (200 μl) was tested on a monolayer of murine fibroblast L929 cells. The murine fibroblast L929 cells were seeded in 96-well culture plates at a cell density of 1.0 × 103 cells/well and fed with standard culture medium for 24 hours at 37°C in a 5% CO2 atmosphere. Then, cell cultures were incubated with the extraction medium and a negative control (standard culture medium) at the same conditions. After 24, 48, and 72 hours of incubation, the cytotoxicity was assessed via a 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyltetrazoliumbro-mide [MTT, Sigma Aldrich, St. Louis, MO] assay. The absorbance was measured at 570 nm, and the cytotoxicity was calculated via the following formulae:
In vivo experiments
The rabbit FCVB
The rabbit FCVB (Guangzhou Vesber Biotechnology LTD) was made of tailor-made modified liquid silicone rubber. The basic material was Dow Corning Class VI elastomers (Dow Corning company, Midland, MI, USA), and the shape was manipulated according to the vitreous parameters of the rabbit by using a computer. The FCVB consists of a vitreous-shaped capsule, tube, and valve, as described in a previous study27. The FCVBs were sterilized by heating in double-distilled water at 100°C for 30 min prior to surgery.
Animal preparation
All experiments were approved by the Hospital Animal Research Review Committee and performed in accordance with the Association for Research in Vision and Ophthalmology (ARVO) Resolution on the Use of Animals in Vision and Ophthalmic Research. Eighteen New Zealand albino rabbits weighing 2.5 to 3.5 kg were divided into three groups: the FCVBs were implanted into the vitreous cavity and then injected with 3% PVA hydrogel (PVA + FCVB group, n = 6), 3% PVA hydrogel only (PVA group, n = 6), or balanced salt solution (BSS) (BSS group, n = 6).
Surgical procedures
All surgeries were performed with sterile technique in the right eyes of the rabbits. After pupillary dilatation, the rabbits were anesthetized via an intramuscular injection of a mixture of ketamine hydrochloride (30 mg/kg) and chlorpromazine hydrochloride (15 mg/kg). Under the standard ophthalmic operating microscope (OPMI VISU 200 plus, Carl Zeiss, Jena, Germany), a standard three-port PPV, 20-gauge Alcon Accurus vitrectomy system was used. The sclerotomy was created approximately 3.5 mm posterior to the limbs. A core vitrectomy was performed and followed by a gentle vitrectomy of the peripheral vitreous base. After removing the vitreous as much as possible, PVA hydrogel (1.1 ml) or BSS (1.1 ml) was directly injected into the vitreous cavity through a 19-gauge needle. In the PVA + FCVB group, the lens was cut during vitrectomy for a better long-term observation of the fundus. At the end of the procedure, the FCVB was folded into three petals and implanted into the vitreous cavity through a 3 mm incision at 12 o'clock without fluid-air exchange. Then, 1.4 ml of PVA hydrogel was injected into the capsule through the tube-valve device fixed under the conjunctiva (Fig. 11). After surgery, the eyes were treated with tobramycin as antibiotic eyedrops and dexamethasone sodium phosphate as steroid eyedrops. This was done three times a day for 2 weeks.
Figure 11. An FCVB was folded and implanted into a vitreous cavity during PPV surgery.
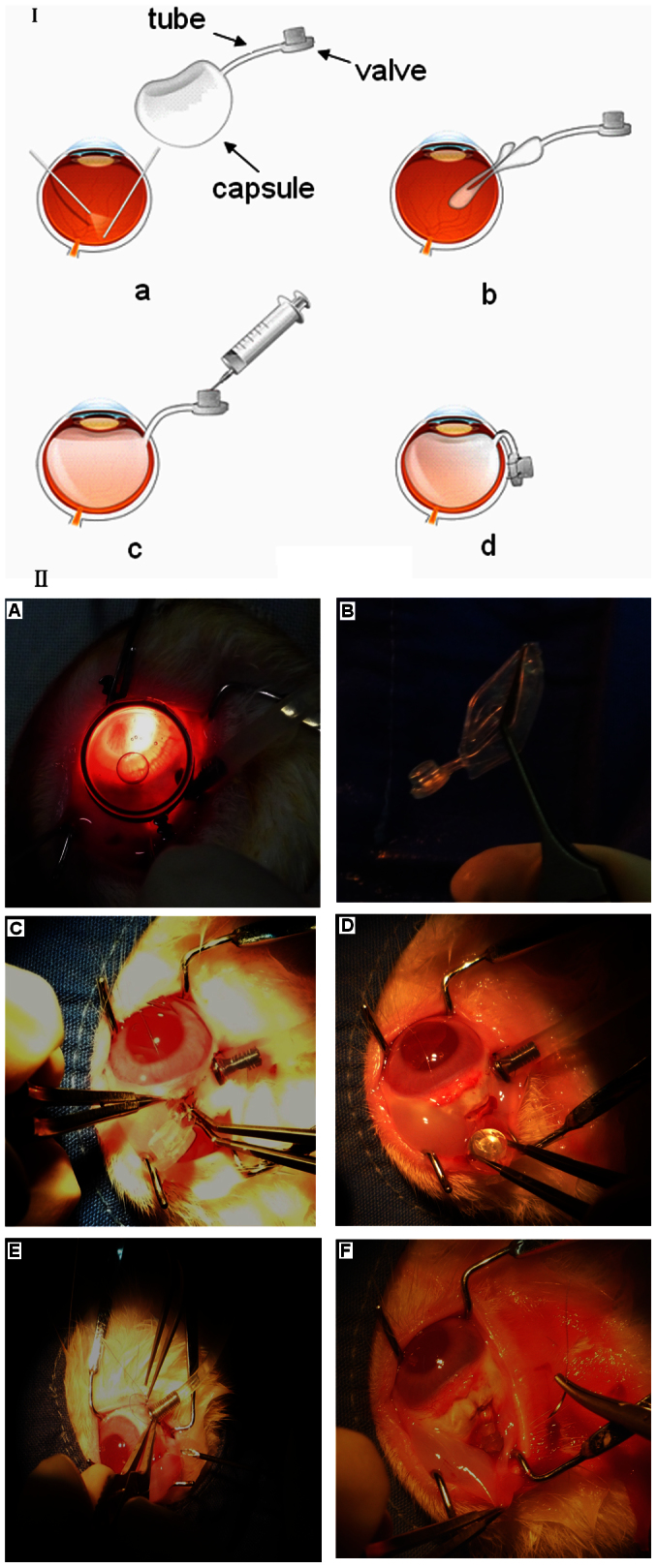
I: Illustration of FCVB implantation. II: Rabbit surgical procedures. (A) A standard three-port PPV was performed to remove the vitreous. (B–C) The FCVB was folded and implanted into the vitreous cavity. (D–E) The PVA hydrogel was injected into the capsule through the valve. (F) fixing the tube-valve device under the conjunctiva.
Postoperative examinations
A slit lamp (SL-D7; Topcon Co., Japan) and a fundus camera (TRL-50DX; Topcon Co., Japan) were used to examine and record the anterior segment, ocular media, and fundus at day 3, 7, 14, 30, 60, 90, and 180 postoperatively. The measurement of intraocular pressure (IOP) with a Tono-Pen (Tonopen avia; Reichert Co., U.S.A.) was performed at day 3, 7, 14, 30, 60, 90, and 180 postoperatively. Also, to evaluate the anatomical position of the retina, a B-scan ultrasound (CineScan A/B, Quantel Medical, Clermont-Ferrand, France) was performed at day 14, 30, 60, 90, and 180 postoperatively.
Electroretinogram (ERG)
A standardized full-field ERG was recorded preoperatively and 90 days and 180 days postoperatively on the right eye by using the Roland Ganzfeld system and PC-based signal acquisition and analysis software (Fa. Roland Consult, Brandeburg, Germany). The eyes were dilated with 0.5% tropicamide and dark-adapted for at least 30 min. Rabbits were anesthetized as described for surgery. The positive unipolar contact lens was placed on the cornea with methylcellulose (2%). The negative needle electrode was inserted into the subcutaneous in the forehead, and the ground electrode was clipped to the earlobe with some electric gel. The bright flash response was elicited by using the ISCEV standard flash of 2.4 cds/m2. The photopic ERG was recorded after 10 min of continuous light adaptation with a background illumination of 10 cds/m2. The a-wave and b-wave amplitudes were evaluated at different time points.
Pathologic examination
The rabbits were euthanatized via an intramuscular injection of an overdose of ketamine and chlorpromazine (1:1) at 90 days postoperative (n = 3, BSS group; n = 3, PVA group; n = 3, PVA + FCVB group) and 180 days postoperative (n = 3, BSS group; n = 3, PVA group; n = 3, PVA + FCVB group). Then, the experimental (right) eyes were harvested from these animals. After enucleation, the PVA hydrogels were removed from the vitreous cavities in order to analyse their elastic and viscous properties. Then, the eyeballs were immediately fixed with 10% paraformaldehyde, and they were embedded in paraffin and stained with hematoxylin and eosin (HE) for a light microscope study.
Statistical analysis
All data were analyzed using the SPSS statistical software version 13.0 (SPSS, Cary, NC, USA). Data are expressed as means ± SD. Statistical differences between groups were tested using a one-way analysis of variance (ANOVA). A P value of less than 0.05 was considered statistically significant.
Author Contributions
S.F.F., X.H.L. and Q.Y.G. conceived and designed the study, and wrote the paper; S.F.F., H.C., Y.Q.L. and Z.H. performed animal experiment. S.F.F., H.C. and X.Y.S. performed the experiments of characteristic and reological property of PVA hydrogel in vitro. L.Z. fabricated the FCVB.
Acknowledgments
This study was supported by Natural Science Foundation of China (81271008) and the Project of Scientific and Technique Plan funded by Sun Yat-sen University (90035-3283315). And we gratefully acknowledge the assistance of Polymer Physics Laboratory of the South China University of Technology, and Analysis and Testing Center of Jinan University.
References
- Silvanus M. T., Moldzio P., Bornfeld N. & Peters J. Visual loss following intraocular gas injection. Dtsch Arztebl Int. 105, 108–112 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Federman J. L. & Schubert H. D. Complications associated with the use of silicone oil in 150 eyes after retina-vitreous surgery. Ophthalmology. 95, 870–876 (1988). [DOI] [PubMed] [Google Scholar]
- Valone Jr J. & McCarthy M. Emulsified anterior chamber silicone oil and glaucoma. Ophthalmology. 101, 1908–1912 (1994). [DOI] [PubMed] [Google Scholar]
- Ichhpujani P., Jindal A. & Jay Katz L. Silicone oil induced glaucoma: a review. Graefes Arch Clin Exp Ophthalmol. 247, 1585–1593 (2009). [DOI] [PubMed] [Google Scholar]
- Foster W. J. Vitreous Substitutes. Expert Rev Ophthalmol 3, 211–218 (2008). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W. et al. Clinical complications of Densiron 68 intraocular tamponade for complicated retinal detachment. Eye (Lond). 24, 21–28 (2010). [DOI] [PubMed] [Google Scholar]
- Orzalesi N., Migliavacca L., Bottoni F. & Miglior S. Experimental short-term tolerance to perfluorodecalin in the rabbit eye: a histopathological study. Curr Eye Res. 17, 828–835 (1998). [DOI] [PubMed] [Google Scholar]
- Stefansson E., Anderson M. M. Jr, Landers M. B. III, Tiedeman J. S. & McCuen B. W. II Refractive changes from use of silicone oil in vitreous surgery. Retina. 8, 20–23 (1988). [DOI] [PubMed] [Google Scholar]
- Kleinberg T. T., Tzekov R. T., Stein L., Ravi N. & Kaushal S. Vitreous substitutes: a comprehensive review. Surv Ophthalmol. 56, 300–323 (2011). [DOI] [PubMed] [Google Scholar]
- Baino F. Towards an ideal biomaterial for vitreous replacement: Historical overview and future trends. Acta Biomater. 7, 921–935 (2011). [DOI] [PubMed] [Google Scholar]
- Pastor J. C. Proliferative vitreoretinopathy: an overview. Surv Ophthalmol. 43, 3–18 (1998). [DOI] [PubMed] [Google Scholar]
- Nakagawa M., Tanaka M. & Miyata T. Evaluation of collagen gel and hyaluronic acid as vitreous substitutes. Ophthalmic Res. 29, 409–420 (1997). [DOI] [PubMed] [Google Scholar]
- Pruett R. C., Schepens C. L. & Freeman H. M. Collagen vitreous substitute. II. Preliminary clinical trials. Arch Ophthalmol. 91, 29–32 (1974). [DOI] [PubMed] [Google Scholar]
- Leone G. et al. PVA/STMP based hydrogels as potential substitutes of human vitreous. J Mater Sci Mater Med. 21, 2491–2500 (2010). [DOI] [PubMed] [Google Scholar]
- Maruoka S. et al. Biocompatibility of polyvinylalcohol gel as a vitreous substitute. Curr Eye Res. 31, 599–606 (2006). [DOI] [PubMed] [Google Scholar]
- Hong Y. et al. Crosslinked poly(1-vinyl-2-pyrrolidinone) as a vitreous substitute. J Biomed Mater Res. 30, 441–448 (1996). [DOI] [PubMed] [Google Scholar]
- Hong Y. et al. Biodegradation in vitro and retention in the rabbit eye of crosslinked poly(1-vinyl-2-pyrrolidinone) hydrogel as a vitreous substitute. J Biomed Mater Res. 39, 650–659 (1998). [DOI] [PubMed] [Google Scholar]
- Liang C. et al. An evaluation of methylated collagen as a substitute for vitreous and aqueous humor. Int Ophthalmol. 22, 13–18 (1998). [DOI] [PubMed] [Google Scholar]
- Swindle K. E., Hamilton P. D. & Ravi N. In situ formation of hydrogels as vitreous substitutes: Viscoelastic comparison to porcine vitreous. J Biomed Mater Res A. 87, 656–665 (2008). [DOI] [PubMed] [Google Scholar]
- Mueller-Jensen K. Polyacrylamide as an alloplastic vitreous implant. Albrecht Von Graefes Arch Klin Exp Ophthalmol. 189, 147–158 (1973). [DOI] [PubMed] [Google Scholar]
- Lai J. Y. Biocompatibility of chemically cross-linked gelatin hydrogels for ophthalmic use. J Mater Sci Mater Med. 21, 1899–1911 (2010). [DOI] [PubMed] [Google Scholar]
- Bruining M. J. et al. New biodegradable networks of poly(N-vinylpyrrolidinone) designed for controlled nonburst degradation in the vitreous body. J Biomed Mater Res. 47, 189–197 (1999). [DOI] [PubMed] [Google Scholar]
- Swindle-Reilly K. E. et al. Rabbit study of an in situ forming hydrogel vitreous substitute. Invest Ophthalmol Vis Sci. 50, 4840–4846 (2009). [DOI] [PubMed] [Google Scholar]
- Hara Y. et al. Biocompatibility of polyvinylalcohol gel as a vitreous substitute. Nihon Ganka Gakkai Zasshi. 102, 247–255 (1998). [PubMed] [Google Scholar]
- Gao Q. et al. A new strategy to replace the natural vitreous by a novel capsular artificial vitreous body with pressure-control valve. Eye (Lond). 22, 461–468 (2008). [DOI] [PubMed] [Google Scholar]
- Gao Q. et al. Refractive shifts in four selected artificial vitreous substitutes based on Gullstrand-Emsley and Liou-Brennan schematic eyes. Invest Ophthalmol Vis Sci. 50, 3529–3534 (2009). [DOI] [PubMed] [Google Scholar]
- Liu Y. et al. Technical standards of a foldable capsular vitreous body in terms of mechanical, optical, and biocompatible properties. Artif Organs. 34, 836–845 (2010). [DOI] [PubMed] [Google Scholar]
- Wang P. et al. Biocompatibility and retinal support of a foldable capsular vitreous body injected with saline or silicone oil implanted in rabbit eyes. Clin Experiment Ophthalmol. 40, e67–75 (2012). [DOI] [PubMed] [Google Scholar]
- Lin X. et al. Evaluation of the flexibility, efficacy, and safety of a foldable capsular vitreous body in the treatment of severe retinal detachment. Invest Ophthalmol Vis Sci. 52, 374–381 (2011). [DOI] [PubMed] [Google Scholar]
- Lin X. et al. Preliminary efficacy and safety of a silicone oil-filled foldable capsular vitreous body in the treatment of severe retinal detachment. Retina. 32, 729–741 (2012). [DOI] [PubMed] [Google Scholar]
- Chen J. et al. Clinical device-related article evaluation of morphology and functions of a foldable capsular vitreous body in the rabbit eye. J Biomed Mater Res B Appl Biomater. 97, 396–404 (2011). [DOI] [PubMed] [Google Scholar]
- Chirila T. V. & Hong Y. Poly(1-vinyl-2-pyrrolidinone) hydrogels as vitreous substitutes: a rheological study. Polymer International. 46, 183–195 (1998). [Google Scholar]
- Nickerson C. S., Park J., Kornfield J. A. & Karageozian H. Rheological properties of the vitreous and the role of hyaluronic acid. J Biomech. 41, 1840–1846 (2008). [DOI] [PubMed] [Google Scholar]
- Yamauchi A. Synthetic vitreous body of PVA hydrogel. In: De Rossi, D., Kajiwara, K., Osada, Y., Yamauchi, A. (eds). Polymer Gels – Fundamentals and Biomedical Applications. New York: Plenum Press. pp 127–134 (1991). [Google Scholar]
- Benlian W., Zhang J. & Huibin W. The development of synthetic vitreous body and its experiment in rabbits. In: Feng, H., Han, Y., Huang, L. (eds). Polymers and Biomaterials. Amsterdam: Elsevier Scientific Publishers, pp 397–400 (1991). [Google Scholar]
- Jiang Z. et al. Evaluation of the levofloxacin release characters from a rabbit foldable capsular vitreous body. Int J Nanomedicine. 7, 1–10 (2012). [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang Z. et al. Evaluation of levofloxacin release characteristics from a human foldable capsular vitreous body in vitro. J Ocul Pharmacol Ther. 28, 33–40 (2012). [DOI] [PubMed] [Google Scholar]
- Liu Y. et al. Sustained mechanical release of dexamethasone sodium phosphate from a foldable capsular vitreous body. Invest Ophthalmol Vis Sci. 51, 1636–1642 (2010). [DOI] [PubMed] [Google Scholar]
- Zheng H. et al. Evaluation of 5-fluorouracil released from a foldable capsular vitreous body in vitro and in vivo. Graefes Arch Clin Exp Ophthalmol. 250, 751-759 (2012). [DOI] [PubMed] [Google Scholar]
- Chen X. et al. Protein kinase Calpha downregulation via siRNA-PKCalpha released from foldable capsular vitreous body in cultured human retinal pigment epithelium cells. Int J Nanomedicine. 6, 1303–1311 (2011). [DOI] [PMC free article] [PubMed] [Google Scholar]