Abstract
The posterior lateral line primordium (PLLp) migrates caudally and periodically deposits neuromasts. Coupled, but mutually inhibitory, Wnt-FGF signaling systems regulate proto-neuromast formation in the PLLp: FGF ligands expressed in response to Wnt signaling activate FGF receptors and initiate proto-neuromast formation. FGF receptor signaling, in turn, inhibits Wnt signaling. However, mechanisms that determine periodic neuromast formation and deposition in the PLLp remain poorly understood. Previous studies showed that neuromasts are deposited closer together and the PLLp terminates prematurely in lef1-deficient zebrafish embryos. It was suggested that this results from reduced proliferation in the leading domain of the PLLp and/or premature incorporation of progenitors into proto-neuromasts. We found that rspo3 knockdown reduces proliferation in a manner similar to that seen in lef1 morphants. However, it does not cause closer neuromast deposition or premature termination of the PLLp, suggesting that such changes in lef1-deficient embryos are not linked to changes in proliferation. Instead, we suggest that they are related to the role of Lef1 in regulating the balance of Wnt and FGF functions in the PLLp. Lef1 determines expression of the FGF signaling inhibitor Dusp6 in leading cells and regulates incorporation of cells into neuromasts; reduction of Dusp6 in leading cells in lef1-deficient embryos allows new proto-neuromasts to form closer to the leading edge. This is associated with progressively slower PLLp migration, reduced spacing between deposited neuromasts and premature termination of the PLLp system.
Keywords: Lateral line, Wnt, FGF, Lef1, R-spondin, Dusp6, Zebrafish
INTRODUCTION
The lateral line in zebrafish has emerged as an attractive model system with which to understand molecular mechanisms underlying the coordination of cell proliferation, migration and differentiation during organ morphogenesis in vertebrate embryonic development (reviewed by Chitnis et al., 2012; Ma and Raible, 2009). In this study we describe a new role for Lef1 in inhibiting FGF signaling and show how the balance of Wnt and FGF signaling influences neuromast formation and deposition.
The lateral line is a mechanosensory system that has evolved to detect patterns of water movement (Coombs and van Netten, 2006). It contains neuromasts, which are small sensory organs that are distributed in a stereotyped pattern on the surface of the fish. Formation of the posterior lateral line system in zebrafish is pioneered by the posterior lateral line primordium (PLLp), a cluster of ~100 cells formed near the ear that migrates under the skin along the horizontal myoseptum and periodically deposits five to six neuromasts during its migration (reviewed by Ghysen and Dambly-Chaudière, 2007). The PLLp eventually dissociates to form two to three terminal neuromasts at the tip of the tail.
Formation of nascent neuromasts, or ‘proto-neuromasts’, is initiated by FGF signaling in the migrating PLLp (Lecaudey et al., 2008; Nechiporuk and Raible, 2008). Fgf3 and Fgf10 are expressed in response to Wnt signaling in a leading domain of the PLLp (Aman and Piotrowski, 2008). However, the FGFs are not very effective at activating FGF receptor (FGFR) signaling in the leading domain, at least in part because factors that determine FGF expression also determine the expression of Sef (Il17rd – Zebrafish Information Network), an inhibitor of FGFR signaling (Aman and Piotrowski, 2008; Tsang et al., 2002). As a result, FGFs produced in the leading domain in response to Wnt signaling activate FGFR signaling in an adjacent trailing domain, where there is less inhibition of FGFR signaling. FGFR signaling also inhibits Wnt signaling and restricts expression of the Wnt effector lef1 to a leading domain of the PLLp. This inhibition is mediated in part by a diffusible Wnt antagonist, Dkk1, the expression of which at the trailing end of the leading lef1 expression domain is dependent on FGFR signaling (Aman and Piotrowski, 2008). In this manner, coupled, but mutually inhibitory, Wnt-FGF signaling systems are established in the PLLp.
FGF signaling induces proto-neuromast formation by initiating epithelial rosette formation and by inducing the expression of atoh1a, which gives cells the potential to become sensory hair cell progenitors (Lecaudey et al., 2008; Nechiporuk and Raible, 2008). Lateral inhibition mediated by Notch signaling restricts atoh1a expression and sensory hair cell progenitor fate to a central cell in each forming proto-neuromast (Itoh and Chitnis, 2001; Sarrazin et al., 2006). While Wnt signaling initiates fgf10 expression at the leading end of the PLLp, Atoh1a begins to drive fgf10 expression in the trailing part of the PLLp (Matsuda and Chitnis, 2010). In this manner, the central cell maintains a restricted source of FGF ligand in each maturing proto-neuromast.
Proto-neuromasts are formed periodically toward the leading end of the PLLp and as they mature they are deposited from the trailing end. Chemokine signaling and the polarized expression of two chemokine receptors in the PLLp determine the directional migration of the PLLp (David et al., 2002; Haas and Gilmour, 2006; Dambly-Chaudière et al., 2007; Valentin et al., 2007). Whereas cells in a leading domain of the PLLp express cxcr4b and undergo collective migration, cells in the trailing part of the PLLp express cxcr7b during and after deposition (Dambly-Chaudière et al., 2007; Valentin et al., 2007). Although previous studies have examined mechanisms that initiate proto-neuromast formation and determine PLLp migration, mechanisms that regulate periodic neuromast formation and their deposition remain poorly defined.
It has been suggested that cell proliferation in the PLLp is a key determinant of periodic neuromast deposition (Aman et al., 2011). These authors suggest that a regulatory mechanism keeps the cxcr7b-free leading domain relatively constant in size. In this context, the rate at which the PLLp grows by proliferation determines how quickly cells enter the trailing domain, where they express cxcr7b and are eventually deposited. This hypothesis provides a potential explanation for why manipulations that either broadly increase cell death or broadly reduce proliferation in the PLLp decrease the rate of proto-neuromast deposition in the migrating PLLp (Aman et al., 2011).
Wnt and FGFR signaling both regulate proliferation in the PLLp (Aman et al., 2011). Loss of lef1, a transcriptional co-factor that mediates a subset of Wnt-dependent processes in the PLLp, reduces proliferation in the PLLp (Gamba et al., 2010; Valdivia et al., 2011; McGraw et al., 2011). However, the reduced proliferation is not accompanied by a delay in neuromast deposition. Instead, loss of lef1 function is associated with closer neuromast deposition and premature termination of the PLLp system. It has been suggested that this is because proliferation is specifically reduced in a leading domain of the PLLp when lef1 function is lost (Valdivia et al., 2011). This, the authors suggest, results in rapid shrinking of the leading domain, which is a source of hypothetical factors that determine both the length of the column of cells undergoing collective migration and how close to the leading end new proto-neuromasts form. Proliferation-related shrinking of the leading domain in lef1 mutants is suggested to accelerate the loss of these hypothetical factors, which reduces the length of the migrating column of PLLp cells, accelerates the deposition of neuromasts and eventually contributes to premature termination of the PLLp system (Valdivia et al., 2011).
Alternatively, it has been suggested that the premature termination of the PLLp system is related less to reduced proliferation and more to a failure in the maintenance of progenitor cells in the leading domain of the PLLp (McGraw et al., 2011). These authors show that loss of lef1 allows premature incorporation of leading progenitors into forming proto-neuromasts, and loss of progenitors results in the premature termination of the PLLp system in lef1 mutants. However, the molecular mechanisms by which Lef1 maintains the progenitor state were not defined.
We found that knockdown of rspo3, which encodes a potential facilitator of Wnt signaling (Binnerts et al., 2007; Hendrickx and Leyns, 2008), reduces proliferation in the PLLp in a manner similar to that seen following lef1 knockdown. However, it does not reduce the spacing of deposited neuromasts or result in premature termination of the PLLp system. This suggested that changes in the pattern of proliferation might not be at the heart of why neuromasts are deposited closer or why the PLLp terminates prematurely when lef1 function is lost. In this study, we have explored the possibility that these changes are more related to a distinct role of lef1 in regulating the balance of Wnt and FGF signaling and to FGF signaling-dependent formation of proto-neuromasts in the PLLp. We show that the balance of Wnt and FGF signaling can influence the neuromast deposition rate and that Lef1 determines the expression of an antagonist of FGF signaling, Dusp6, in the leading domain of the PLLp. Consistent with the role for lef1 suggested by McGraw et al. (McGraw et al., 2011), we show that Dusp6 operates downstream of lef1 to deter incorporation of cells into proto-neuromasts. Taken together, our study shows how Lef1 regulates the balance of Wnt and FGF signaling and the incorporation of progenitors into nascent proto-neuromasts.
MATERIALS AND METHODS
Fish maintenance and mutant strains
Zebrafish were maintained under standard conditions. The embryos were staged according to Kimmel et al. (Kimmel et al., 1995). The tg[cldnb:lynGFP] transgenic strain was described previously (Haas and Gilmour, 2006). Embryos were treated with 1-50 μM IWR-1 (Calbiochem), 10 μM XAV939 (Sigma), 1-2.5 μM BCI (Calbiochem) and 20 μM SU5402 (Calbiochem) from 26 hpf to the time of fixation.
Morpholino (MO) injections
MOs used were (5′-3′): fgf10-MO1, GCTTTACTCACTGTACGGATCGTCC (Nechiporuk and Raible, 2008); fgf10a-MO2, GAAAATGATGCTCACCGCCCCGTAG (Norton et al., 2005); rspo3-MO, TTGCGGCTCTTACCTTTTGTTCAAG; lef1-MO, CTCCACCTGACAACTGCGGCATTTC (Valdivia et al., 2011). Unless stated otherwise in figure legends, 2 ng fgf10, rspo3 or lef1 MO was injected. All MO injections were performed with co-injection of 1.5 ng p53-MO (Robu et al., 2007). Standard Control Oligo (Gene Tools) was used as control-MO and injected at the same concentration as the experimental MO(s).
Whole-mount in situ hybridization and immunohistochemistry
Whole-mount in situ hybridization was performed as described (Matsuda and Chitnis. 2010). RNA probes were synthesized using a DIG labeling kit (Roche). BCIP/NBT substrate (Vector Laboratories) was used as a chromogen. Immunohistochemistry was performed as described (Matsuda and Chitnis, 2009). Antibodies were mouse anti-ZO-1 (Invitrogen; 1:500) and rabbit anti-Cldnb produced by injection of peptide based on published sequence (López-Schier et al., 2004) (1:500). Images were captured with a Zeiss LSM510.
Quantification of expression domains
For quantification of gene expression visualized with in situ hybridization, the length of the expression domain or its integrated density was measured. To measure integrated density, images were converted to grayscale, inverted, and the sum of the grayscale values of pixels within a region of interest (ROI) measured using the integrated density measurement plug-in in ImageJ software (NIH). For statistical analysis, an unpaired Student's t-test was performed using QuickCalcs software (GraphPad).
BrdU incorporation and TUNEL staining
tg[cldnb:lynGFP] embryos were dechorionated and placed in 10 mM BrdU (Roche) in a solution of egg water with 15% DMSO for 1 hour with constant agitation at 28°C. Staining was performed as described (Dalle Nogare et al., 2009), and imaging was performed on a Zeiss LSM510. Images were analyzed in ImageJ using the cell counter plug-in (Kurt De Vos, University of Sheffield, UK). PLLp were divided into thirds by length, based on the Cldnb staining, and total cells and BrdU+ nuclei were counted for each segment to obtain the BrdU index. TUNEL staining was performed using the ApopTag Fluorescein Detection Kit (Millipore), according to the manufacturer's directions, except that embryos were incubated overnight in labeling solution at 37°C. TUNEL-stained tg[cldnb:lynGFP] embryos were counterstained with anti-GFP antibody (Abcam).
Time-lapse
For time-lapse acquisition, tg[cldnb:lynGFP] embryos were anesthetized at ~28 hpf in 600 μM Tricaine (Sigma) and embedded in 0.75% low melting point agarose (Nusieve GTG, Lonza) in glass-bottom chamber slides (Nunc). Movies were acquired for 14-18 hours at 5-minute intervals using an inverted Leica SP5 confocal microscope with a 20× objective lens. Quantification of divisions and migration speed was performed in ImageJ. To calculate migration speed, the distance traveled between successive neuromast depositions was divided by the total time between depositions as measured from the time-lapse movie.
Distribution of cells with ectopic expression of Dusp6
cDNA encoding mCherry or Dusp6-T2A-mCherry red fluorescent protein was inserted downstream of the hsp70 promoter. Plasmid DNA was injected into 1- to 2-cell stage tg[cldnb:lynGFP] embryos. Embryos were heat treated at 38°C for 45 minutes at 24, 30 and 36 hpf. The distribution of red fluorescent cells in deposited neuromasts and terminal neuromasts at the tail tip was counted at 56 hpf. A total of 142 cells in eight embryos expressing mCherry and 94 cells in six embryos expressing Dusp6-mCherry were analyzed. Imaging was performed on a Leica TCS SP5 with a 40× objective lens.
RESULTS
rspo3 is expressed in a Wnt signaling-dependent manner in the PLLp
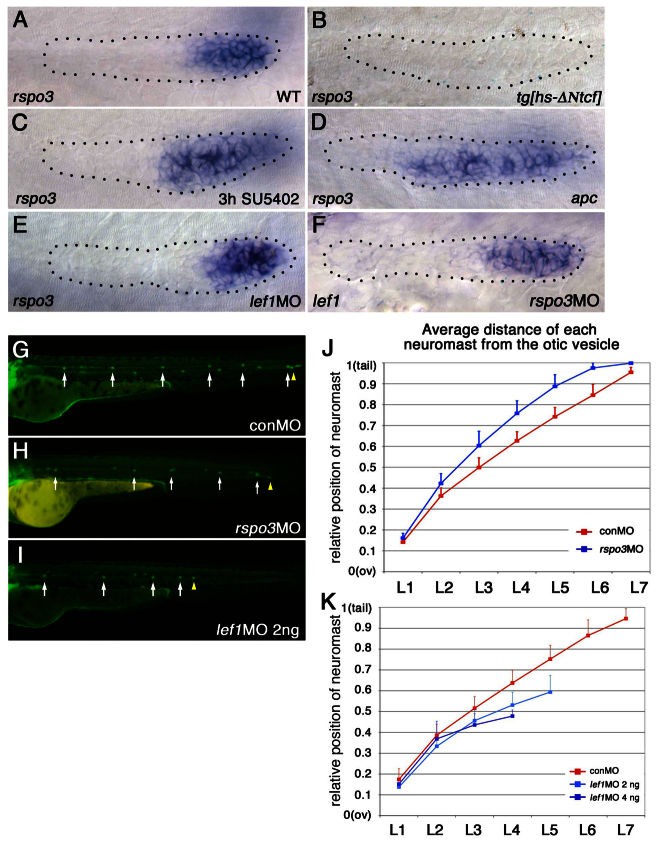
Like lef1, which is a key mediator of Wnt signaling, rspo3 is expressed in the leading part of the PLLp (Fig. 1A). As shown previously for lef1 (Aman and Piotrowski, 2008), the expression of a dominant-repressor form of TCF, ΔNTcf, eliminated expression of rspo3 (Fig. 1B), and inhibition of FGF signaling with SU5402 was associated with slightly expanded expression of rspo3 (Fig. 1C). However, the expansion was less than that of lef1 expression, which typically expands to fill almost the entire PLLp following SU5402 treatment (Aman and Piotrowski, 2008) (data not shown). Expanded rspo3 expression was also seen in apc mutants (Fig. 1D), associated with exaggerated Wnt signaling (Hurlstone et al., 2003) and expanded lef1 expression (Aman and Piotrowski, 2008). These observations suggest that expression of rspo3, like that of lef1, is promoted by Wnt signaling. However, knockdown of lef1, which significantly reduces GFP expression in the PLLp of Wnt reporter tg(7xTCF-Xla.Siam:GFP)ia4 transgenic fish (Valdivia et al., 2011; Moro et al., 2012) (supplementary material Fig. S2), does not reduce the expression level of rspo3 (Fig. 1E). This suggests that Wnt signaling determines rspo3 expression in a Lef1-independent manner in the PLLp. In addition, knockdown of rspo3 (see supplementary material Fig. S1 for validation of the MO) did not cause any obvious change in lef1 expression (Fig. 1F). However, it did slightly reduce fluorescence in the primordium of Wnt reporter tg(7xTCF-Xla.Siam:GFP)ia4 transgenic fish (supplementary material Fig. S2), supporting the role of Rspo3 as a facilitator of Wnt signaling.
Fig. 1.
Expression of rspo3 and lef1 is determined by similar mechanisms but knockdown of rspo3 and of lef1 have distinct effects on the pattern of neuromast deposition. (A-I) The leading end of the PLLp and caudal end of the zebrafish embryo are to the right. (A-F) The PLLp is outlined (dotted line). (A-D) Expression of rspo3 in the PLLp at 31 hpf. (A) rspo3 is expressed in the leading domain of the wild-type (WT) PLLp. (B) Heat shock-induced expression of a dominant-repressor form of TCF (ΔNTcf) inhibits rspo3 expression. (C,D) Exaggeration of Wnt activity, following inhibition of FGFR signaling with SU5402 (C) or in apc mutants (D), expands rspo3 expression toward the trailing domain. (E,F) rspo3 expression in the lef1 morphant PLLp (E) and lef1 expression in the rspo3 morphant PLLp (F) at 31 hpf. (G-K) The pattern of neuromast deposition in lef1 morphants and rspo3 morphants. (G-I) Neuromast deposition in tg[cldnb:lynGFP] embryos injected with lef1-MO, rspo3-MO or control-MO (conMO) at 48 hpf. White arrows indicate individual neuromasts and yellow arrowheads indicate PLLp termination. (J,K) Average position of each deposited neuromast in lef1 or rspo3 morphants relative to the distance from the otic vesicle (ov) to the tip of the tail. (H,J) Neuromasts are spaced further apart in rspo3 morphants. See supplementary material Fig. S4 for suppression of this phenotype by rspo3 mRNA co-injection. (I,K) Neuromasts are spaced closer together in lef1 morphants. Error bars indicate s.d.
Knockdown of lef1 and of rspo3 have distinct effects on neuromast deposition
As expression of lef1 and rspo3 appeared to be regulated by similar mechanisms, we compared changes in the rspo3 morphants with those in lef1 morphants to look for similarities in function. Although there was some cell death in both lef1 and rspo3 morphants, co-injection of p53-MO minimized p53 (Tp53)-dependent cell death in the PLLp (supplementary material Fig. S3). After confirming that p53-MO on its own does not affect PLLp development, p53-MO was co-injected with experimental or control MO to suppress p53-dependent cell death in all subsequent MO experiments.
In wild-type embryos, the PLLp migrates and deposits five to six neuromasts along the horizontal myoseptum and stops migration at the tip of the tail (Fig. 1G), where the PLLp dissociates to form two to three terminal neuromasts. In rspo3 morphants neuromast spacing was increased and the PLLp system rarely (1/43) terminated prematurely (Fig. 1H,J; supplementary material Fig. S4). By contrast, as shown previously (Valdivia et al., 2011; McGraw et al., 2011), neuromasts were deposited closer to each other and the PLLp system consistently terminated prematurely in lef1 morphants (Fig. 1I,K; supplementary material Fig. S5). Co-injection of rspo3 mRNA helped bring neuromast spacing and the number of deposited neuromasts closer to wild-type patterns in rspo3 morphants (supplementary material Fig. S4), supporting the specificity of rspo3-MO effects. Taken together, these studies show that knockdown of lef1 and of rspo3 have different effects on the spacing of neuromasts and on PLLp termination.
Knockdown of lef1 and of rspo3 result in a similar reduction in proliferation
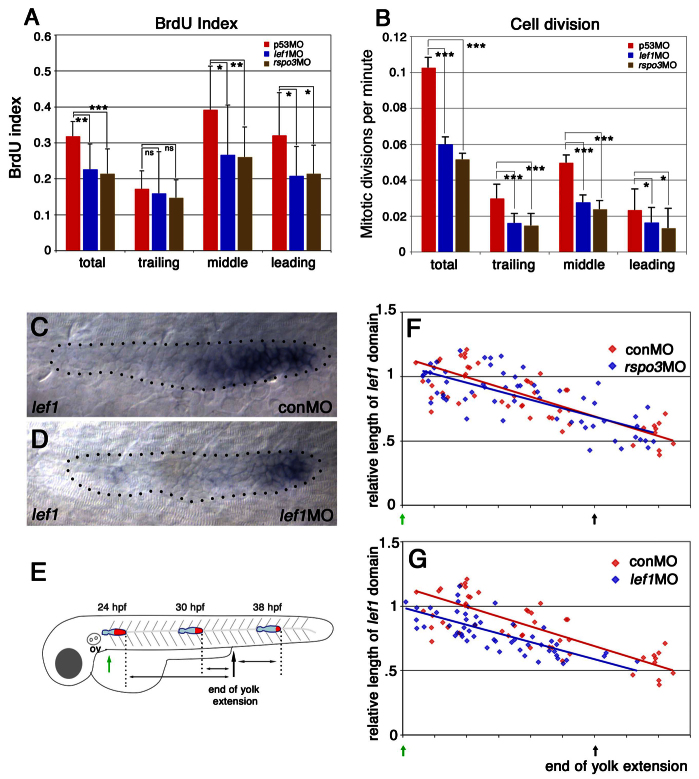
We compared proliferation patterns in lef1 and rspo3 morphants to determine whether distinct effects on proliferation might account for the differences in their patterns of neuromast deposition. lef1 and rspo3 morphants showed similar reductions in BrdU incorporation (Fig. 2A; supplementary material Table S1); the reduction was primarily in the leading and middle domains of the PLLp, which is consistent with previous analyses of lef1 mutants (McGraw et al., 2011; Valdivia et al., 2011).
Fig. 2.
Although rspo3 and lef1 morphants have similar changes in cell proliferation, the size of the lef1 expression domain is only reduced in lef1 morphants. (A,B) Knockdown of lef1 and of rspo3 reduce proliferation in a similar manner. (A) Total and regional BrdU incorporation index in the PLLp of control, lef1 morphant and rspo3 morphant embryos (see also supplementary material Table S1). (B) Total and regional cell division in the PLLp of control, lef1 morphant and rspo3 morphant embryos. ns, non-significant (P>0.05); *P<0.05, **P<0.01, ***P<0.001 (see also supplementary material Table S2). Error bars indicate s.d. (C,D) The lef1 expression domain is smaller in lef1 morphants at 31 hpf. For statistical analysis, see supplementary material Fig. S5D. (E) The progressive change in the size of the lef1 domain (red) over the course of PLLp migration. (F,G) Size of the lef1 domain (relative to its average length in control embryos at 24 hpf) plotted against the position of the PLLp along the trunk for a collection of embryos fixed between 24 and 38 hpf (see schematic in E). Red dots indicate control-MO-injected embryos and blue dots indicate either rspo3 (F) or lef1 (G) morphant embryos. Red or blue lines indicate the associated linear regression lines for each data set. A simple linear regression model and a paired t-test confirmed that the length of the lef1 domain is significantly reduced in lef1 morphants (P<0.001).
As changes in cell number following cell division, not just the potential for division indicated by BrdU incorporation, are expected to determine proliferation-dependent changes in neuromast deposition, time-lapse imaging of the PLLp was used to record every cell division during ~15 hours of PLLp migration, starting from just before L1 deposition (supplementary material Movies 1-3). Although there was no significant difference in the pattern of cell divisions between lef1 and rspo3 morphants, the analysis confirmed that the number of cell divisions in both lef1 and rspo3 morphants was significantly lower than in controls (Fig. 2B; supplementary material Table S2). It is important to note that, although BrdU incorporation was most prominent in the leading and middle thirds of the wild-type PLLp (Fig. 2A; supplementary material Table S1), cell divisions were relatively infrequent in the leading third and were most frequent in the middle third (Fig. 2B; supplementary material Table S2). Furthermore, the reduction in cell division was least in the leading third of the PLLp in both lef1 and rspo3 morphants (Fig. 2B). Changes in the pattern of proliferation were similar in lef1 and rspo3 morphants, but effects on neuromast deposition and PLLp termination were different. Therefore, it appeared unlikely that reduced proliferation was specifically responsible for the closer spacing of neuromasts and premature termination of the PLLp in lef1 morphants.
Knockdown of lef1, but not rspo3, reduces the size of the lef1 expression domain
As previous studies suggested that reduced proliferation following loss of lef1 function results in a reduction in the size of the leading domain of the PLLp (Valdivia et al., 2011), we compared the size of the leading domain in lef1 and rspo3 morphants, as defined by the domain of lef1 expression. As shown previously, lef1 expression was not lost in lef1 morphants (Valdivia et al., 2011; McGraw et al., 2011) (Fig. 2C,D). However, examination of the PLLp at various points along its migration (Fig. 2E) suggested that the lef1 domain is significantly smaller in lef1 morphants (Fig. 2G; supplementary material Fig. S5D). By contrast, there were no obvious changes in the size of the lef1 domain in rspo3 morphants (Fig. 2F). This suggested that a reduction in the size of the leading domain, as defined by lef1 expression, might indeed be a key change that contributes to the closer neuromast spacing and premature termination of the PLLp in lef1 morphants. It should be noted, however, that the lef1 domain did not shrink faster in lef1 morphants, it was just smaller from the beginning (Fig. 2G).
lef1 knockdown is associated with slower PLLp migration prior to premature termination
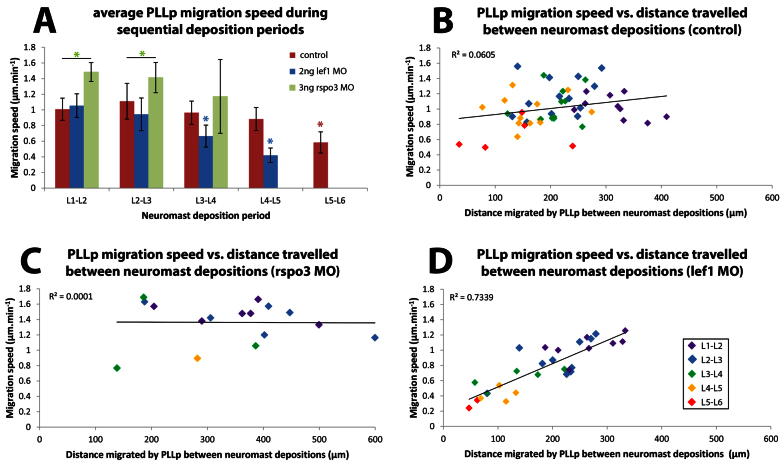
To determine whether differences in neuromast spacing are linked to differences in how far the PLLp migrates between deposition events, we used long-term time-lapse movies (supplementary material Movies 1-3), originally collected for cell division analysis, to compare migration speeds in lef1 and rspo3 morphants. As previously reported (Valdivia et al., 2011; McGraw et al., 2011), there was little change in the migration speed during early migration; however, the lef1 morphant PLLp became progressively slower during the later phase of migration (Fig. 3A; supplementary material Table S3). Furthermore, whereas there was no significant correlation between the migration speed and the distance migrated by the PLLp between neuromast depositions in wild-type embryos, there was a strong positive correlation in lef1 morphants (Fig. 3B,D; supplementary material Table S3). This suggests that, whereas other factors play a greater role in determining neuromast spacing in wild-type embryos, reduced speed is a significant determinant of reduced inter-neuromast spacing in lef1 morphants. However, although the PLLp migrated significantly faster in rspo3 morphants than in wild type and lef1 morphants during the L1-L2 and L2-L3 deposition cycles, there was no significant overall correlation between migration speed and the distance between neuromast depositions in rspo3 morphants (Fig. 3C; supplementary material Table S3). These results suggest that, as in wild-type embryos, factors other than migration speed play a more important role in determining spacing.
Fig. 3.
Correlation of PLLp migration speed with spacing of deposited neuromasts. (A) PLLp migration speed compared in control, lef1 and rspo3 morphant embryos in successive periods between neuromast deposition. Red asterisk indicates that the speed between L5-L6 depositions was significantly slower than between preceding L4-L5 depositions. Blue asterisks indicate that PLLp speed in lef1 morphants between L3-L4 and L4-L5 depositions was significantly slower than in the respective, preceding deposition cycles. In addition, PLLp speed in lef1 morphants between L3-L4 and L4-L5 depositions was significantly slower than in controls during the same deposition cycle. Green asterisks indicate statistical significance between the indicated measurements (black bar). *P<0.05. Error bars indicate s.d. (B-D) PLLp migration speed plotted against distance traveled between neuromast depositions in wild-type (B), rspo3 morphant (C) and lef1 morphant (D) embryos. Points are shown in different colors for successive pairs of neuromasts (color key in D). Linear regression lines are shown for each data set. The associated coefficient of determination (R2) indicates that the correlation between PLLp migration speed and distance traveled between neuromast depositions is strong only for lef1 morphants. See supplementary material Table S3 for additional information.
Reduced Wnt activity promotes proto-neuromast maturation and deposition
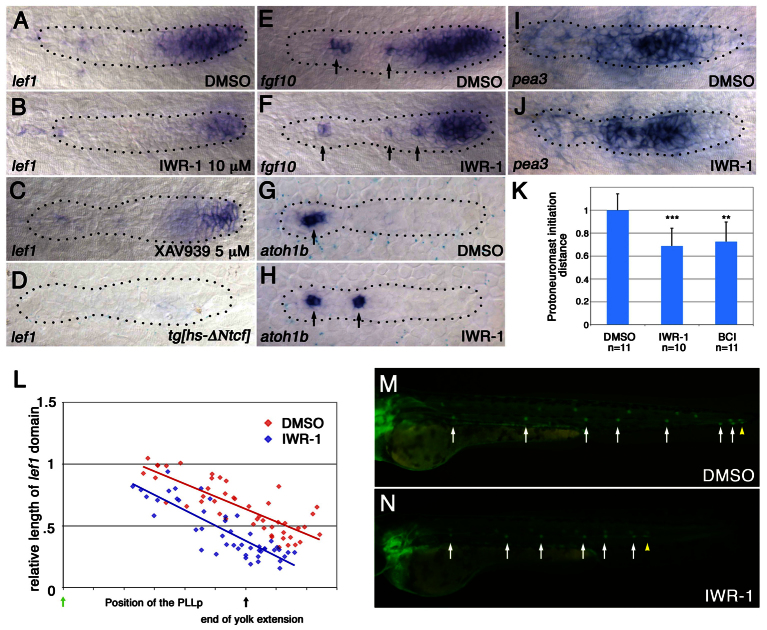
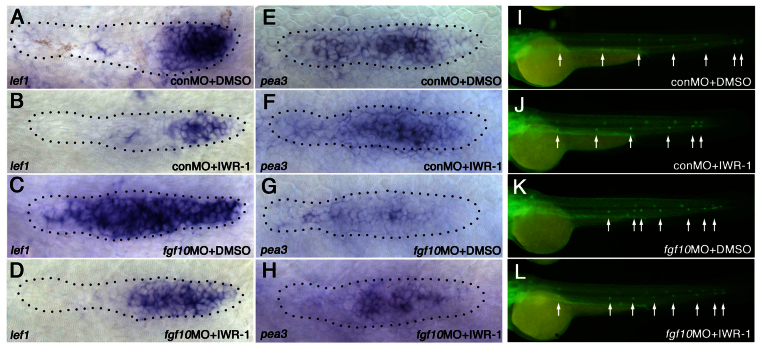
To determine whether changes in lef1 morphants reflect changes in the balance of Wnt and FGFR signaling, we examined how weakening the Wnt system with other manipulations would affect neuromast deposition and PLLp termination. The pharmacological Wnt signaling inhibitors IWR-1 and XAV939 stabilize Axin, which is an essential component of the β-catenin degradation complex, and reduce the amount of β-catenin available for Wnt signaling (Chen et al., 2009; Huang et al., 2009). Treatment with either IWR-1 or XAV939 reduced the size and intensity of the leading lef1 domain in the PLLp (Fig. 4A-C; supplementary material Fig. S6A), but did not completely repress lef1 expression, as follows expression of ΔNTcf (Aman and Piotrowski, 2008) (Fig. 4D). When examined at different points along its migration, the lef1 domain was consistently smaller in the PLLp of IWR-1-treated embryos (Fig. 4L).
Fig. 4.
Weakening Wnt signaling facilitates proto-neuromast maturation and deposition. (A-D) Wnt inhibitors reduce lef1 expression. lef1 expression is shown in the PLLp following exposure to DMSO control (A), IWR-1 (B) and XAV939 (C), and following heat shock-induced expression of ΔNTcf (D) at 31 hpf. (E,F) IWR-1 shrinks the broad leading fgf10 expression domain while exposing more spots of trailing fgf10 expression (arrows). (G,H) Premature appearance of atoh1b expression (arrows) in IWR-1-treated PLLp at ~31 hpf. (I,J) pea3 expression is not obviously altered in IWR-1-treated embryos. (K) Proto-neuromast formation is initiated closer to the leading edge in IWR-1-treated and BCI-treated embryos. **P<0.01, ***P<0.001. Error bars indicate s.d. (L) Comparison of the relative size of the lef1 expression domain at different stages of migration in embryos treated with DMSO (red) or 10 μM IWR-1 (blue), as in Fig. 2F,G. A simple linear regression model and a paired t-test confirmed that the length of the lef1 domain is significantly smaller in the IWR-1-treated embryos. P<0.0001. (M,N) Neuromast deposition in embryos treated with DMSO (M) or IWR-1 (N).
IWR-1 also reduced the size of the broad Wnt-dependent fgf10 domain at the leading end of the PLLp (Fig. 4E,F; supplementary material Fig. S6B), which was accompanied by the premature appearance of Atoh1a-dependent focal fgf10 expression (arrows in Fig. 4E,F; supplementary material Fig. S6D,E). Furthermore, shrinking of the Wnt active domain resulted in premature atoh1b expression, which was seen in more proto-neuromasts and closer to the leading edge of the PLLp (Fig. 4G,H; supplementary material Fig. S6G). Knockdown of lef1 had a similar effect to IWR-1 on fgf10 and atoh1b expression (supplementary material Fig. S6C,F,H). Although we did not see statistically significant differences in the size or intensity of the pea3 (etv4 – Zebrafish Information Network) expression domain, as a readout of FGF signaling, in the IWR-1-treated PLLp (Fig. 4I,J; supplementary material Fig. S6I), we did observe a central accumulation of ZO-1 (Tjp1 – Zebrafish Information Network), which marks proto-neuromast formation, closer to the leading end of the PLLp (Fig. 4K).
In IWR-1-treated embryos, the PLLp deposited neuromasts at shorter intervals and the terminated prematurely (Fig. 4M,N). Progressively higher concentrations of IWR-1 resulted in earlier termination of the system and the deposition of fewer neuromasts (supplementary material Fig. S7). Taken together, these results suggest that reduced Wnt signaling induced by IWR-1 allows FGF signaling-dependent proto-neuromast formation closer to the leading end, reduces neuromast spacing and prematurely terminates the PLLp system, as is seen when lef1 function is compromised.
fgf10 knockdown delays the establishment of FGFR signaling and the shrinking of the lef1 domain
We next asked whether weakening the FGF system, by knocking down fgf3 or fgf10, delays neuromast formation and deposition. Whereas knockdown of fgf3 had little effect on pea3 expression (Lecaudey et al., 2008; Nechiporuk and Raible, 2008) (data not shown), knockdown of fgf10 was associated with an expanded lef1 domain and a failure to establish robust pea3 expression at 31 hpf (Fig. 5A-D; supplementary material Fig. S8A,B). By 35 hpf, however, pea3 expression was well established in the trailing domain and lef1 had become restricted to a much smaller leading domain, as in control embryos (Fig. 5I-L).
Fig. 5.
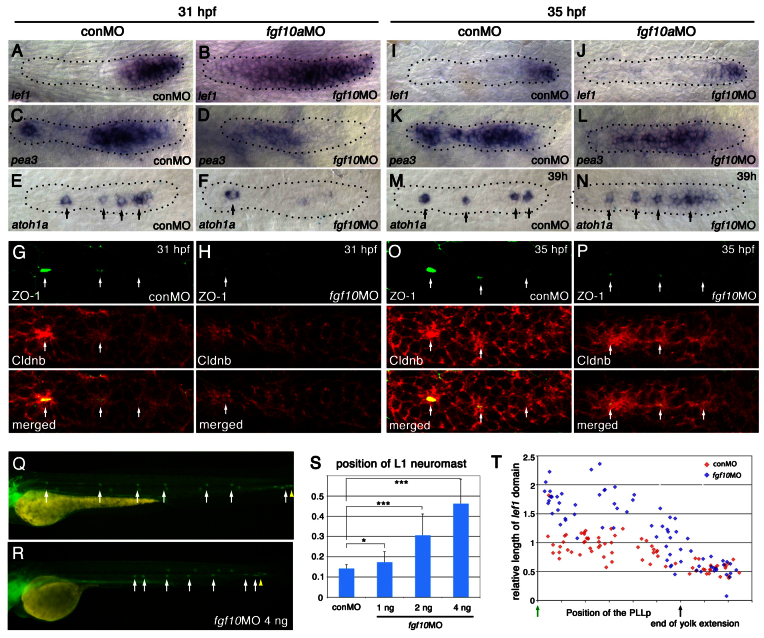
Knockdown of fgf10 delays initial neuromast formation and deposition. (A-H) Knockdown of fgf10 delays the initial restriction of the lef1 domain, the establishment of FGFR signaling and the formation of proto-neuromasts. lef1 (A,B), pea3 (C,D), and atoh1a (E,F) expression is shown in control (conMO) and fgf10 morphant embryos at 31 hpf. For statistical analysis, see supplementary material Fig. S8. (G,H) Epithelial rosette formation visualized by central accumulation (arrows) of ZO-1 (green) and Cldnb (red). (I-P) By 35 hpf, however, there is restriction of lef1 (I,J), recovery of pea3 (K,L) and establishment of multiple proto-neuromasts, as shown by focal atoh1a (M,N) and central accumulation of ZO-1 (green) and Cldnb (red) (O,P). (Q,R) Deposition of the first neuromast is delayed in fgf10 morphants and subsequent neuromasts are deposited at closer intervals. (S) Dose-dependent delay in L1 neuromast deposition. *P<0.05, ***P<0.001. Error bars indicate s.d. (T) Comparison of the relative size of the lef1 expression domain at different stages of migration in control (red) and fgf10 morphant (blue) embryos, as in Fig. 2E.
fgf10 knockdown delays initial proto-neuromast formation and deposition
The delay in establishing FGFR signaling in fgf10 morphants was accompanied by the absence of atoh1a expression in the leading domain of the PLLp at 31 hpf (Fig. 5E,F) (Nechiporuk and Raible, 2008). This is likely to reflect a delay in the initial formation of proto-neuromasts, which, in the wild-type PLLp, starts at the trailing end and then proceeds sequentially toward the leading end (Nechiporuk and Raible, 2008). Consistent with this interpretation, absence of atoh1a expression was accompanied by a severe delay in the accumulation of ZO-1 and Cldnb in the fgf10 morphant PLLp (Fig. 5G,H). Once initiated, periodic proto-neuromast formation in the fgf10 morphant PLLp progressed from the trailing end toward the leading end of the PLLp, as in wild type. This was marked by the appearance of restricted atoh1a expression and the accumulation of ZO-1 and Cldnb in more leading domains of the PLLp (Fig. 5M-P).
The neuromast formation and deposition rate correlates with shrinking of the lef1 domain
Delay in proto-neuromast formation in fgf10 morphants was accompanied by a dose-dependent delay in deposition of the first stable neuromast (Fig. 5Q-S; supplementary material Fig. S9). However, after L1 neuromast deposition, subsequent neuromasts were deposited closer to each other (Fig. 5Q,R; supplementary material Fig. S9). The initial delay and subsequent recovery of neuromast deposition correlated with an initial delay in the restriction of the lef1 domain and a subsequent recovery in shrinking of the lef1 domain in the fgf10 morphant PLLp (Fig. 5A,B,I,J,T). Although these changes were not seen in fgf10 mutants (Lecaudey et al., 2008), the effects of fgf10 knockdown on PLLp development were confirmed with two independent previously characterized MOs [fgf10-MO1 (Nechiporuk and Raible, 2008) and fgf10a-MO2 (Norton et al., 2005)] (supplementary material Fig. S10). Furthermore, by comparing the effects of knockdown of fgf10a, fgf10b, or both in combination, we confirmed that these effects are specific to fgf10a (data not shown).
Time-lapse imaging showed that PLLp migration was significantly slower in fgf10 morphants between the deposition of the L2 and L3 neuromasts; however, as in wild-type embryos, there was no overall significant correlation between migration speed and distance traveled between neuromast depositions (supplementary material Movie 4, Fig. S11). This suggests that reduced speed is not the primary factor that determines the closer spacing of deposited neuromasts in fgf10 morphants, and that other factors appear to play a more important role.
The balance of Wnt and FGF signaling determines the initiation of proto-neuromast formation
Progressive shrinking of the lef1 expression domain in the PLLp could be a secondary effect of sequential proto-neuromast formation from progenitors in the leading domain that lose lef1 expression as they are incorporated into proto-neuromasts; alternatively, it could reflect attenuation of the Wnt system, which serves as a pre-condition for effective proto-neuromast formation. Delay in formation of the first stable proto-neuromast in fgf10 morphants is associated with persistent broad lef1 expression. If the expanded lef1 expression simply reflects failure of proto-neuromast formation due to ineffective FGF signaling, then attenuation of Wnt signaling would not be expected to have any effect on when proto-neuromast formation is initiated in this context. If, however, shrinking of the Wnt active domain defines a pre-condition for effective FGF-dependent proto-neuromast formation, then attenuating Wnt signaling should facilitate earlier proto-neuromast formation in the fgf10 morphant PLLp.
Weakening the Wnt system with IWR-1 in fgf10 morphants facilitated earlier shrinking of the lef1 expression domain (Fig. 6A-D; supplementary material Fig. S12A,B). This was associated with earlier establishment of the trailing FGF system, as indicated by pea3 expression (Fig. 6E-H; supplementary material Fig. S12C), and earlier deposition of the first neuromast (Fig. 6I-L; supplementary material Fig. S13A,B). Similarly, knockdown of lef1 in fgf10 morphants facilitated earlier deposition of the first neuromast (supplementary material Fig. S13C,D). Together, these observations support our interpretation that the dynamic balance of Wnt and FGFR signaling, in some way regulated by lef1, influences the initiation of proto-neuromast formation in the PLLp.
Fig. 6.
Attenuation of Wnt signaling facilitates neuromast formation in fgf10 morphants. (A-L) Attenuation of Wnt signaling with IWR-1 facilitates earlier shrinking of the Wnt system, earlier establishment of effective FGFR signaling and earlier initiation of neuromast deposition in the PLLp of fgf10 morphants. lef1 expression (A-D), pea3 expression (E-H) and the neuromast deposition pattern (I-L, arrows) are shown in control morphants (A,E,I), control morphants with IWR-1 treatment (B,F,J), fgf10 morphants (C,G,K) and fgf10 morphants with IWR-1 treatment (D,H,L). For statistical analysis, see supplementary material Figs S12, S13.
Lef1 is responsible for expression of dusp6 in the leading Wnt active zone
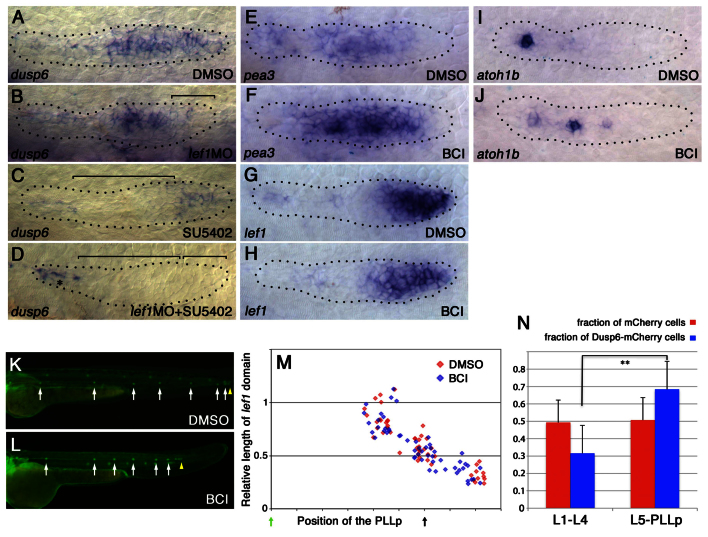
Next, we asked whether Lef1 influences the balance of Wnt and FGF signaling by regulating the expression of factors that either facilitate Wnt or inhibit FGF signaling. Previous studies have shown that Lef1 is not essential for driving its own expression nor is it required for expression of Sef, an inhibitor of FGFR signaling in the leading domain (McGraw et al., 2011; Valdivia et al., 2011). However, we found that Lef1 is required for the expression of dusp6 [previously known as map kinase phosphatase 3 (mkp3)], which encodes a member of the dual specificity protein phosphatase subfamily that functions as an intracellular inhibitor of ERK signaling (Tsang and Dawid, 2004). Unlike sef, the Wnt-dependent expression of which is more restricted in the leading compartment of the PLLp (Aman and Piotrowski, 2008), dusp6 is expressed in approximately the leading two-thirds of the PLLp (Fig. 7A).
Fig. 7.
Lef1 determines leading dusp6 expression and inhibition of Lef1 function influences the pattern of proto-neuromast formation and neuromast deposition. (A-D) dusp6 expression is regulated by Lef1 and FGFR signaling. (A) dusp6 is expressed in the leading two-thirds of the PLLp. (B) lef1 knockdown eliminates dusp6 expression in a leading domain. (C) SU5402 treatment eliminates trailing dusp6 expression. (D) SU5402 treatment in lef1 morphants eliminates dusp6 expression in the PLLp. Bracket shows where dusp6 expression is lost. Asterisk indicates persistent dusp6 expression in prospective inter-neuromast cells. (E,F) pea3 expression shows that FGFR signaling is enhanced in BCI-treated embryos. For statistical analysis, see supplementary material Fig. S15B. (G,H) lef1 expression suggests that Wnt signaling is not obviously altered in BCI-treated embryos. For statistical analysis, see supplementary material Fig. S15A. (I,J) atoh1b expression is seen in more proto-neuromasts closer to the lead edge of the PLLp. For statistical analysis, see supplementary material Fig. S6E. (K,L) Neuromasts are deposited closer together in BCI-treated embryos. For statistical analysis, see supplementary material Fig. S16. (M) There is no apparent difference between DMSO-treated controls and BCI-treated embryos in the size of the lef1 domain over the course of PLLp migration. (N) mCherry or Dusp6-mCherry expression was induced by heat shock treatment during PLLp migration. After the PLLp had completed migration, the numbers of red fluorescent cells were counted in each neuromast (see supplementary material Fig. S17). Cells expressing Dusp6-mCherry were less likely to be deposited in neuromasts L1-4 and more likely to be deposited in L5-6 or terminal neuromasts of the PLLp. **P<0.01. Error bars indicate s.d.
In lef1 morphants there is a specific loss of dusp6 expression in the leading end, while some expression in the trailing end is retained (Fig. 7B). When embryos were exposed to the FGF signaling inhibitor SU5402, trailing expression was lost, whereas expression in the leading zone was retained (Fig. 7C). When embryos were both injected with lef1 MO and exposed to SU5402, dusp6 expression was lost throughout the PLLp (Fig. 7D). These observations suggest that dusp6 expression is under the dual control of lef1-dependent Wnt signaling in the leading zone and FGF signaling in the trailing zone. Dual regulation of dusp6 by Wnt and FGF signaling has been described previously in zebrafish late gastrula embryos (Tsang and Dawid, 2004). It should be noted, however, that exposure to SU5402 resulted in a specific loss of dusp6 in the trailing domain, in spite of the fact that inhibition of FGF signaling causes an expansion of Wnt-dependent lef1 expression to the trailing end (Aman and Piotrowski, 2008). Restricted expression of dusp6 in the leading zone following exposure to SU5402 suggests that some additional factors regulate dusp6 expression in the PLLp. Knockdown of rspo3 did not alter the expression of dusp6 (supplementary material Fig. S14).
Inhibition of Dusp6 function facilitates neuromast formation and deposition
If loss of lef1-dependent dusp6 expression is responsible for closer neuromast deposition, inhibition of dusp6 itself should cause similar changes. To test this, we used a specific chemical inhibitor of Dusp6, BCI (Molina et al., 2009). Consistent with the function of Dusp6 as an inhibitor of FGF signaling, BCI treatment increased FGF signaling-dependent pea3 expression in the PLLp (Fig. 7E,F; supplementary material Fig. S15B). It also facilitated proto-neuromast formation and maturation: central ZO-1 accumulation was first seen closer to the leading edge of the PLLp (Fig. 4K) and atoh1b expression was prematurely induced in the trailing part of the PLLp (Fig. 7I,J; supplementary material Fig. S6G). In addition, exposure to BCI reduced neuromast spacing and led to premature termination of PLLp migration (Fig. 7K,L; supplementary material Fig. S16), as in lef1 morphants. However, unlike lef1 morphants, the lef1 domain was not consistently smaller in BCI-treated embryos (Fig. 7G,H,M). This is consistent with Dusp6 functioning downstream of lef1 and with changes in Dusp6 function not feeding back on lef1 expression.
As inhibition of Dusp6 with BCI facilitated proto-neuromast formation, we asked whether induced expression of Dusp6 would reduce the probability of cells being incorporated into proto-neuromasts and being deposited by the migrating PLLp. Plasmids encoding heat shock-inducible mCherry red fluorescent protein or Dusp6 in conjunction with mCherry were injected into 1- to 2-cell stage tg[cldnb:lynGFP] embryos. Embryos were heat-treated at 38°C for 45 minutes at 24, 30 and 36 hpf to maintain induced expression of mCherry or Dusp6-mCherry. The distribution of mCherry+ or Dusp6-mCherry+ cells in deposited neuromasts or terminal neuromasts of the PLLp was compared at 56 hpf (supplementary material Fig. S17). For effective statistical comparison of their relative distribution, only embryos with at least ten positive cells in the PLLp system were counted. Cells with induced expression of Dusp6 were significantly less likely to be deposited in neuromasts L1-4 and more likely to be deposited in L5-6 or terminal neuromasts of the PLLp (Fig. 7N). Together, these observations show that Lef1 determines the expression of Dusp6 in a leading domain of the PLLp and that Lef1-dependent Dusp6 expression regulates FGF signaling-dependent incorporation of progenitors into proto-neuromasts.
DISCUSSION
This study began with an examination of Rspo3 function in the PLLp. It has shown that, like Lef1, Rspo3 determines Wnt-dependent proliferation in the PLLp, and that Rspo3 loss results in a pattern of reduced proliferation similar to that seen following loss of Lef1 function. It was initially thought that reduced proliferation could account for both the premature termination of the PLLp system and the closer spacing of deposited neuromasts when Lef1 function is lost. However, our studies suggest that these changes are more likely to be related to an additional role of lef1 in determining dusp6 expression and in influencing where FGF signaling can effectively determine the morphogenesis of nascent proto-neuromasts. Our observations reveal how Wnt signaling, acting via distinct target genes, simultaneously promotes proliferation and inhibits proto-neuromast formation, ensuring that both processes are coordinated in the migrating PLLp.
Our study shows that the balance of Wnt and FGF signaling influences when and where proto-neuromasts form and that this can influence the pattern of neuromast deposition by the migrating PLLp. Previous studies have suggested that Sef, which broadly inhibits FGF signaling by interfering with the function of the FGF receptor, is expressed in response to Wnt signaling in a leading domain (Aman and Piotrowski, 2008). We have now shown that Dusp6, a phosphatase that inhibits ERK signaling, is expressed in response to Lef1-mediated Wnt/β-catenin signaling in the leading domain of the PLLp. This is the branch of the FGF signaling pathway that is specifically responsible for mediating morphogenetic changes involved in neuromast formation (Harding and Nechiporuk, 2012).
The sequential formation of proto-neuromasts is accompanied by the complementary progressive restriction of lef1 expression to a smaller leading domain of the PLLp. This can be interpreted in a number of ways. The leading lef1 domain can be thought to represent a progenitor population from which cells are progressively lost as they are incorporated into neuromasts. From this perspective, the shrinking of the lef1 domain is a secondary consequence of proto-neuromast formation/differentiation. Alternatively, reduction of Wnt signaling in response to FGF signaling might serve as a pre-condition for effective proto-neuromast formation in the trailing domain. In this context, shrinking of the lef1 expression domain may indirectly reflect the dynamic tug-of-war between the Wnt and FGF signaling pathways that determines when and how close to the leading edge the formation of proto-neuromasts is allowed. We have shown that knockdown of fgf10 delays the establishment of a robust FGF signaling system and consequently delays the formation and deposition of stable neuromasts. In this context, weakening the Wnt system with IWR-1 or by knockdown of the Wnt effector lef1 allows the earlier formation of proto-neuromasts, supporting our interpretation that the balance of Wnt and FGF signaling determines when proto-neuromasts are formed.
Weakening the Wnt system or loss of lef1 results in both a shorter leading lef1 expression domain and the formation of nascent proto-neuromasts closer to the leading end. But how is the shorter leading lef1 expression domain linked to the shorter distance between deposited neuromasts? Inhibition of Dusp6 function results in many changes similar to those seen when lef1 function is lost: the spacing between deposited neuromasts is closer and nascent proto-neuromasts are formed closer to the leading edge of the migrating PLLp. Nevertheless, there is no obvious reduction in the size of the leading domain as revealed by lef1 expression. This suggests that reduction in the length of the leading lef1 domain might not be directly related to changes in spacing between deposited neuromasts. What might be more relevant is the reduction in the size of the domain between the most recently formed proto-neuromast and the leading edge of the PLLp, as this is clearly smaller in lef1-deficient, IWR-1-treated and Dusp6-inhibited embryos.
We have also shown that the spacing of deposited neuromasts correlates with the migration speed between deposition events in lef1 morphants. This suggests that the size of the leading proto-neuromast-free domain might influence the PLLp migration speed, but how remains unclear. One possibility is that it may be related to changes in cell behavior and morphology that accompany nascent proto-neuromast formation; the relatively mesenchymal leading cells might become progressively less migratory as they form epithelial rosettes. In this manner, the size of the proto-neuromast-free leading domain may determine the efficacy of PLLp migration.
Preliminary studies suggest that there is also an expansion of cxcr7b expression and a reduction of the leading cxcr7b-free domain in the PLLp of lef1 morphants and BCI-treated or IWR-1 treated embryos (supplementary material Fig. S18). As initiation of cxcr7b expression has been suggested to correlate with cells preparing for deposition (Aman et al., 2011), this too might contribute to slower PLLp migration. However, the mechanism by which potential changes in cxcr7b expression and the formation of proto-neuromasts closer to the leading end influence migration speed remains unclear and will be addressed in future studies.
The initially broad lef1 domain progressively shrinks as the PLLp migrates from the ear to the tip of the tail, where its eventual demise terminates the PLLp system. Inhibiting lef1 function facilitates the premature loss of the lef1 domain and termination of the PLLp system when it has only completed about half its journey to the tip of the tail. Although there is significant variation in the precise pattern of neuromast deposition, the shrinking and eventual demise of the lef1 domain are tightly linked to its arrival near the tip of the tail. How the environment or internal dynamics of the PLLp regulates the size of the lef1 domain to reproducibly determine termination of the PLLp system as it reaches the end of its journey also remains an interesting puzzle for future studies.
Our study illustrates how lef1 and the balance of Wnt and FGF signaling regulates migration, growth and differentiation in a developing organ. The insights from this model system are likely to be of general interest, as interactions between Wnt and FGF signaling regulate development in a wide range of contexts (Aulehla et al., 2008; ten Berge et al., 2008; Olivera-Martinez and Storey, 2007).
Supplementary Material
Acknowledgements
We thank James Hudspeth (Rockefeller University, NY, USA) for providing anti-Claudin b antibody, and all members of the A.B.C. laboratory for suggestions.
Footnotes
Funding
This work was supported by the Intramural Research Program of the Eunice Kennedy Shriver National Institute of Child Health and Human Development, National Institutes of Health (NIH) [HD001012-15]. M.M. was also supported by an NIH grant [HD062561]. Deposited in PMC for release after 12 months.
Competing interests statement
The authors declare no competing financial interests.
Supplementary material
Supplementary material available online at http://dev.biologists.org/lookup/suppl/doi:10.1242/dev.091348/-/DC1
References
- Aman A., Piotrowski T. (2008). Wnt/beta-catenin and Fgf signaling control collective cell migration by restricting chemokine receptor expression. Dev. Cell 15, 749-761 [DOI] [PubMed] [Google Scholar]
- Aman A., Nguyen M., Piotrowski T. (2011). Wnt/β-catenin dependent cell proliferation underlies segmented lateral line morphogenesis. Dev. Biol. 349, 470-482 [DOI] [PubMed] [Google Scholar]
- Aulehla A., Wiegraebe W., Baubet V., Wahl M. B., Deng C., Taketo M., Lewandoski M., Pourquié O. (2008). A beta-catenin gradient links the clock and wavefront systems in mouse embryo segmentation. Nat. Cell Biol. 10, 186-193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Binnerts M. E., Kim K. A., Bright J. M., Patel S. M., Tran K., Zhou M., Leung J. M., Liu Y., Lomas W. E., 3rd, Dixon M., et al. (2007). R-Spondin1 regulates Wnt signaling by inhibiting internalization of LRP6. Proc. Natl. Acad. Sci. USA 104, 14700-14705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen B., Dodge M. E., Tang W., Lu J., Ma Z., Fan C. W., Wei S., Hao W., Kilgore J., Williams N. S., et al. (2009). Small molecule-mediated disruption of Wnt-dependent signaling in tissue regeneration and cancer. Nat. Chem. Biol. 5, 100-107 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chitnis A. B., Nogare D. D., Matsuda M. (2012). Building the posterior lateral line system in zebrafish. Dev. Neurobiol. 72, 234-255 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coombs S., van Netten S. M. (2006). The hydrodynamics and structural mechanics of the lateral line system. Fish Biomechanics 23, 103-139 [Google Scholar]
- Dalle Nogare D. E., Pauerstein P. T., Lane M. E. (2009). G2 acquisition by transcription-independent mechanism at the zebrafish midblastula transition. Dev. Biol. 326, 131-142 [DOI] [PubMed] [Google Scholar]
- Dambly-Chaudière C., Cubedo N., Ghysen A. (2007). Control of cell migration in the development of the posterior lateral line: antagonistic interactions between the chemokine receptors CXCR4 and CXCR7/RDC1. BMC Dev. Biol. 7, 23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- David N. B., Sapède D., Saint-Etienne L., Thisse C., Thisse B., Dambly-Chaudière C., Rosa F. M., Ghysen A. (2002). Molecular basis of cell migration in the fish lateral line: role of the chemokine receptor CXCR4 and of its ligand, SDF1. Proc. Natl. Acad. Sci. USA 99, 16297-16302 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gamba L., Cubedo N., Lutfalla G., Ghysen A., Dambly-Chaudiere C. (2010). Lef1 controls patterning and proliferation in the posterior lateral line system of zebrafish. Dev. Dyn. 239, 3163-3171 [DOI] [PubMed] [Google Scholar]
- Ghysen A., Dambly-Chaudière C. (2007). The lateral line microcosmos. Genes Dev. 21, 2118-2130 [DOI] [PubMed] [Google Scholar]
- Haas P., Gilmour D. (2006). Chemokine signaling mediates self-organizing tissue migration in the zebrafish lateral line. Dev. Cell 10, 673-680 [DOI] [PubMed] [Google Scholar]
- Harding M. J., Nechiporuk A. V. (2012). Fgfr-Ras-MAPK signaling is required for apical constriction via apical positioning of Rho-associated kinase during mechanosensory organ formation. Development 139, 3130-3135 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hendrickx M., Leyns L. (2008). Non-conventional Frizzled ligands and Wnt receptors. Dev. Growth Differ. 50, 229-243 [DOI] [PubMed] [Google Scholar]
- Huang S. M., Mishina Y. M., Liu S., Cheung A., Stegmeier F., Michaud G. A., Charlat O., Wiellette E., Zhang Y., Wiessner S., et al. (2009). Tankyrase inhibition stabilizes axin and antagonizes Wnt signalling. Nature 461, 614-620 [DOI] [PubMed] [Google Scholar]
- Hurlstone A. F. L., Haramis A. P., Wienholds E., Begthel H., Korving J., Van Eeden F., Cuppen E., Zivkovic D., Plasterk R. H. A., Clevers H. (2003). The Wnt/β-catenin pathway regulates cardiac valve formation. Nature 425, 633-637 [DOI] [PubMed] [Google Scholar]
- Itoh M., Chitnis A. B. (2001). Expression of proneural and neurogenic genes in the zebrafish lateral line primordium correlates with selection of hair cell fate in neuromasts. Mech. Dev. 102, 263-266 [DOI] [PubMed] [Google Scholar]
- Kimmel C. B., Ballard W. W., Kimmel S. R., Ullmann B., Schilling T. F. (1995). Stages of embryonic development of the zebrafish. Dev. Dyn. 203, 253-310 [DOI] [PubMed] [Google Scholar]
- Lecaudey V., Cakan-Akdogan G., Norton W. H., Gilmour D. (2008). Dynamic Fgf signaling couples morphogenesis and migration in the zebrafish lateral line primordium. Development 135, 2695-2705 [DOI] [PubMed] [Google Scholar]
- López-Schier H., Starr C. J., Kappler J. A., Kollmar R., Hudspeth A. J. (2004). Directional cell migration establishes the axes of planar polarity in the posterior lateral-line organ of the zebrafish. Dev. Cell 7, 401-412 [DOI] [PubMed] [Google Scholar]
- Ma E. Y., Raible D. W. (2009). Signaling pathways regulating zebrafish lateral line development. Curr. Biol. 19, R381-R386 [DOI] [PubMed] [Google Scholar]
- Matsuda M., Chitnis A. B. (2009). Interaction with Notch determines endocytosis of specific Delta ligands in zebrafish neural tissue. Development 136, 197-206 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda M., Chitnis A. B. (2010). Atoh1a expression must be restricted by Notch signaling for effective morphogenesis of the posterior lateral line primordium in zebrafish. Development 137, 3477-3487 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGraw H. F., Drerup C. M., Culbertson M. D., Linbo T., Raible D. W., Nechiporuk A. V. (2011). Lef1 is required for progenitor cell identity in the zebrafish lateral line primordium. Development 138, 3921-3930 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molina G., Vogt A., Bakan A., Dai W., Queiroz de Oliveira P., Znosko W., Smithgall T. E., Bahar I., Lazo J. S., Day B. W., et al. (2009). Zebrafish chemical screening reveals an inhibitor of Dusp6 that expands cardiac cell lineages. Nat. Chem. Biol. 5, 680-687 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moro E., Ozhan-Kizil G., Mongera A., Beis D., Wierzbicki C., Young R. M., Bournele D., Domenichini A., Valdivia L. E., Lum L., et al. (2012). In vivo Wnt signaling tracing through a transgenic biosensor fish reveals novel activity domains. Dev. Biol. 366, 327-340 [DOI] [PubMed] [Google Scholar]
- Nechiporuk A., Raible D. W. (2008). FGF-dependent mechanosensory organ patterning in zebrafish. Science 320, 1774-1777 [DOI] [PubMed] [Google Scholar]
- Norton W. H. J., Ledin J., Grandel H., Neumann C. J. (2005). HSPG synthesis by zebrafish Ext2 and Extl3 is required for Fgf10 signalling during limb development. Development 132, 4963-4973 [DOI] [PubMed] [Google Scholar]
- Olivera-Martinez I., Storey K. G. (2007). Wnt signals provide a timing mechanism for the FGF-retinoid differentiation switch during vertebrate body axis extension. Development 134, 2125-2135 [DOI] [PubMed] [Google Scholar]
- Robu M. E., Larson J. D., Nasevicius A., Beiraghi S., Brenner C., Farber S. A., Ekker S. C. (2007). p53 activation by knockdown technologies. PLoS Genet. 3, e78 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sarrazin A. F., Villablanca E. J., Nuñez V. A., Sandoval P. C., Ghysen A., Allende M. L. (2006). Proneural gene requirement for hair cell differentiation in the zebrafish lateral line. Dev. Biol. 295, 534-545 [DOI] [PubMed] [Google Scholar]
- ten Berge D., Brugmann S. A., Helms J. A., Nusse R. (2008). Wnt and FGF signals interact to coordinate growth with cell fate specification during limb development. Development 135, 3247-3257 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsang M., Friesel R., Kudoh T., Dawid I. B. (2002). Identification of Sef, a novel modulator of FGF signalling. Nat. Cell Biol. 4, 165-169 [DOI] [PubMed] [Google Scholar]
- Tsang M., Maegawa S., Kiang A., Habas R., Weinberg E., Dawid I. B. (2004). A role for MKP3 in axial patterning of the zebrafish embryo. Development 131, 2769-2779 [DOI] [PubMed] [Google Scholar]
- Valdivia L. E., Young R. M., Hawkins T. A., Stickney H. L., Cavodeassi F., Schwarz Q., Pullin L. M., Villegas R., Moro E., Argenton F., et al. (2011). Lef1-dependent Wnt/β-catenin signalling drives the proliferative engine that maintains tissue homeostasis during lateral line development. Development 138, 3931-3941 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Valentin G., Haas P., Gilmour D. (2007). The chemokine SDF1a coordinates tissue migration through the spatially restricted activation of Cxcr7 and Cxcr4b. Curr. Biol. 17, 1026-1031 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.