Abstract
Research in the vestibular field has revealed the existence of a central process, called ‘velocity storage’, that is activated by both visual and vestibular rotation cues and is modified by gravity, but whose functional relevance during natural motion has often been questioned. In this review, we explore spatial orientation in the context of a Bayesian model of vestibular information processing. In this framework, deficiencies/ambiguities in the peripheral vestibular sensors are compensated for by central processing to more accurately estimate rotation velocity, orientation relative to gravity, and inertial motion. First, an inverse model of semicircular canal dynamics is used to reconstruct rotation velocity by integrating canal signals over time. However, its low-frequency bandwidth is limited to avoid accumulation of noise in the integrator. A second internal model uses this reconstructed rotation velocity to compute an internal estimate of tilt and inertial acceleration. The bandwidth of this second internal model is also restricted at low frequencies to avoid noise accumulation and drift of the tilt/translation estimator over time. As a result, low-frequency translation can be erroneously misinterpreted as tilt. The time constants of these two integrators (internal models) can be conceptualized as two Bayesian priors of zero rotation velocity and zero linear acceleration, respectively. The model replicates empirical observations like ‘velocity storage’ and ‘frequency segregation’ and explains spatial orientation (e.g., ‘somatogravic’) illusions. Importantly, the functional significance of this network, including velocity storage, is found during short-lasting, natural head movements, rather than at low frequencies with which it has been traditionally studied.
Keywords: Spatial orientation, Velocity storage, Vestibular, Internal model, Bayes rule, Eye movement, Somatogravic illusion, Vestibulo-ocular reflex, Prior, Bayesian model
Introduction
In addition to the classical five senses, all vertebrates possess the ability to detect motion of the head and body in space through a set of specialized sensory organs located in the inner ear. This sense is crucial for a variety of functions (Angelaki and Cullen 2008). It is fundamental for navigation and spatial orientation. It contributes to equilibrium, in particular while walking on uncertain or slippery terrains. It also drives a gaze stabilization reflex called the vestibulo-ocular reflex (VOR), which maintains a sharp vision when moving the head. In general, it allows tracking head and body position in space when performing complex sequences of movements. Unlike with other senses, one is usually not aware of the existence of ‘vestibular perception’. The significance of this sense becomes apparent after lesions of the vestibular periphery or when self-motion illusions are induced. One well-known illusion happens when one rotates for a while and then stops: this leads to a feeling of spinning in a direction opposite to the initial rotation. Erroneous judgments of body orientation in space are also typically experienced aboard a turning aircraft. In this situation, the influence of the vestibular organs, which misperceive earth-vertical, is so strong that one watching the ground will perceive it as tilted. Vision may also contribute to self-motion perception, as illustrated by the feeling of self-motion experienced when one, sitting in a train, watches an adjacent moving train.
These illusions are attributable to the physical properties of the vestibular sensors and to the organization of central vestibular processing. The various components of self-motion—head rotations and translations—are transduced by two set of vestibular sensors, the semicircular canals and the otolith organs, respectively, as well as the visual system. These sensors have intrinsic limitations: for instance, the canals respond reliably only to brief rotations and the visual system responds with a certain delay. However, the various sensory channels complement themselves efficiently. For example, the canals and the visual system together can reliably detect all rotations, and the canals can contribute to graviception by measuring rotations of the head relative to gravity; both of these ideas are further developed in the next sections. These multisensory interactions are implemented through central processing elements taking into account the specificity of each sensor and the physical relationships between motion variables—e.g., between head velocity and position. One of these key elements processes angular velocity information: Various sources of rotation information converge towards a multi-sensory process which has been called ‘velocity storage’ (see reviews by Hess and Angelaki 1997; MacNeilage et al. 2008; Raphan and Cohen 1985, 2002), and this concept will be central to this review.
But what is the function of velocity storage? Its most apparent feature in laboratory settings is its ability to memorize, or store, rotation information. But why this is functionally important has remained obscure. This lack of intuition is exasperated by the observation that gravity information influences velocity storage properties in a complex and rather unintuitive manner. In this review, we attempt to rationalize why velocity storage and other central vestibular properties exist from a theoretical point of view, while emphasizing intuition rather than presenting mathematical formulas. We start by describing the main sensory organs, their limitations, and how these limitations can theoretically be overcome by a set of simple processing elements. This will lead towards a simple but comprehensive schematic model incorporating the influences of the otolith organs and the semicircular canals, as well as visual rotation information (Part I). We will show how this model relates to previous works and, in particular, to a recent modeling approach based on Bayesian inference (Part II). Once such a model is constructed out of basic principles, as necessary to overcome the peripheral limitations of the vestibular system, it can also explain multiple perceptual illusions and the bizarre gravity influences on velocity storage (Part III). Because our goal is to introduce the central processing elements intuitively, as important and necessary computational solutions to the deficiencies in the peripheral sensors, references to previous work in Part I are limited—but are fully acknowledged in Parts II and III.
Part I: building a model of vestibular processing for spatial orientation and gaze stabilization
The ability of the vestibular organs to detect motion of the head is limited by their physical properties. These limitations can be improved by dynamic processing and multi-sensory integration. In this section, we show how a set of central processing stages can use vestibular information to compute an accurate estimate of head motion. We will illustrate this process by a series of simple simulations, focusing on motion stimuli that are representative of natural movements. Note that the concepts we present here represent a synthesis from a variety of theoretical studies performed in the last decades, e.g., (Bos and Bles 2002; Glasauer and Merfeld 1997; Hess and Angelaki 1997; Laurens and Droulez 2007; MacNeilage et al. 2008; Merfeld et al. 1993a; Merfeld 1995; Merfeld et al. 1999; Mergner and Glasauer 1999; Raphan et al. 1977; Zupan et al. 2002). Its relationship to previous modeling studies will be detailed in Part II.
Rotation information from the vestibular system
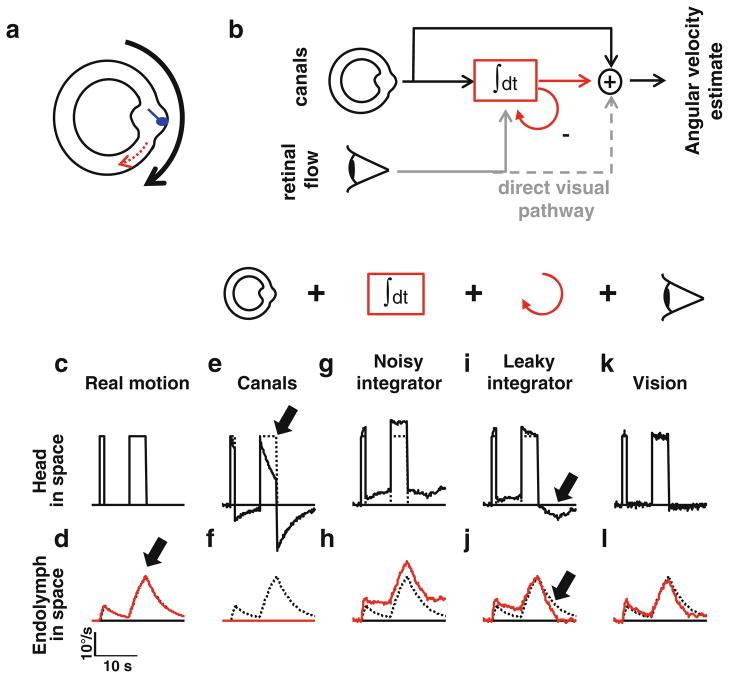
Rotations of the head are primarily sensed by the semicircular canals. Schematically, these organs are tubes filled with a liquid called endolymph. Whenever the head rotates, the canals themselves rotate together with the head, while the endolymph remains stationary because of its own inertia—as an analogy, one may imagine the motion of tea inside of a rotating teacup. This creates a relative motion between the canals and the endolymph, which is detected by a specialized structure, the cupula: the cupula is deflected by the action of the lagging endolymph, which causes a neural signal indicating the rotation. The canals can accurately report brief rotations of the head in all directions. However, they suffer from one limitation: whenever a rotation lasts for more than a fraction of a second, the endolymph gets progressively dragged by the friction with the canal wall; thus, it has a non-zero velocity in space (Fig. 1a). As the endolymph accelerates in space, its motion relative to the canals decreases and consequently the rotation signal decays (Fernández and Goldberg 1971; Goldberg and Fernández 1971; Mayne 1974).
Fig. 1.
Perception of angular velocity. a: principle of the semicircular canals: the cupula (blue) detects the difference between head velocity (black arrow) and endolymph velocity in space (red). b: model of the central visual and vestibular processing of rotation. c, d: head and endolymph velocity in space during a sequence of rotations. e–l: information about head velocity and endolymph velocity provided by the canals alone (e, f), the canals and the velocity storage (g–j), and the full visuo-vestibular model (k, l). Dashed black lines in (e, g, i, k) superimpose trace from (c). Dashed black lines in (f, h, j, l) superimpose trace from (d). See Fig. 4 for simulation parameters
This is illustrated in Fig. 1c, d, in which a sequence of one brief rotation (0.5 s) and a longer one (3 s) are simulated. Note that the endolymph reaches a substantial velocity, particularly for the latter (Fig. 1d, arrow), and consequently, the rotation signal generated by the canals decreases (Fig. 1e, arrow). Furthermore, endolymph rotation persists for a while when the rotation stops (Fig. 1d, red line), which causes a post-rotatory activation of the canals (rebound activity in Fig. 1e). These properties of canal afferents have been described by a high-pass filter, with a time constant of 4 s (Goldberg and Fernández 1971). It is reasonable to assume that these deficiencies in the dynamics of the semicircular canals can be ‘known’ by the brain, such that the real angular velocity can be centrally reconstructed by inverting the transfer function of the canals. This concept can be thought of as the brain having an ‘inverse internal model’ of canal dynamics, such that the real angular velocity of the head can be ideally reconstructed. We will show that what is known as velocity storage can be interpreted as this internal model. Unfortunately, this ideal solution cannot be implemented completely, as will be shown next, because of biological noise. It is for this reason that although the velocity storage improves canal dynamics, the real angular velocity is never reconstructed ideally.
But to proceed further with our discussion, we will first show how the inverse internal model idea can be implemented using the most widely known Raphan-Cohen model of velocity storage (Raphan et al. 1977) (Fig. 1b). It is possible to design a system that overcomes the deficient dynamics of canal afferents, by feeding their signal into an integrator. This velocity storage integrator can easily be tuned in such a way that its output will closely match the velocity of the endolymph in space.1 Thus, by summing the output of the velocity storage with the canal signal, one can compute head velocity in space (Fig. 1b). In other words, the combination of these two pathways implements an internal model of the canals. This solution is, however, impractical for one reason: any biological system is subject to a certain amount of noise. This noise can affect the canal signal or the integrator itself and would invariably accumulate in the integrator (Fig. 1h), causing a drift in its output and thus also in the rotation estimate (Fig. 1g).
This problem can be surmounted by adding a leak to the integrator, i.e., a process that slowly and continuously discharges it, which is represented by a negative feedback loop on Fig. 1b. This leak ‘flushes’ the noise that continuously accumulates in the integrator’s output and thus counteracts the drift. This comes at the expense of the efficiency of the system during sustained rotations: the leak causes the integrator’s signal to undershoot endolymph motion (Fig. 1j, arrow). Therefore, the final estimate decays to a certain extent during continuous rotation and suffers from a slight aftereffect (Fig. 1i, arrow). Despite this limitation, the leaky integrator improves considerably the rotation signal extracted from the canals (compare Fig. 1e, g, i). Importantly, the contribution of velocity storage to the angular velocity reconstruction is not only limited to very low frequencies and long-duration, constant velocity rotations, as often erroneously presumed (because these are the conditions typically tested in behavioral experiments). As illustrated in Fig. 1, its effects are already seen for 1–3 s constant velocity rotations, which are certainly in the range of motions experienced during natural behavior.
Rotation information from the visual system
Whenever the centrally reconstructed signal does not match endolymph velocity—either because of sensory noise or because of the integrator leak, the visual system is activated. The resulting retinal flow signal (generated because of a less than ideal VOR) can be added with an appropriate gain to the velocity storage such that its output now more closely reflects endolymph velocity (Fig. 1l, red versus gray dotted lines). This in turn improves the rotational velocity estimate (Fig. 1k).
Retinal flow can also be used to correct the output of the rotation estimator itself (Fig. 1b, direct visual pathway, dashed gray line), e.g., this pathway is recruited for gaze stabilization by efficiently suppressing vestibulo-ocular commands during fixation on a head-stationary target. Importantly, however, while this direct pathway improves the final estimate, the correction through the velocity storage is actually the one that minimizes the mismatch between the estimated and the real velocity of the endolymph in space. By doing so, it allows the VOR to provide a more accurate gaze stabilization command and reduces the contribution of visual fixation and tracking. This is ecologically beneficial since ocular tracking is typically weary—one can appreciate this by attempting to watch a movie filmed by an unsteady camera or read a book while slowly moving it in front of one’s head. In other words, the visual projection through the velocity storage allows the rotation estimator to operate accurately indefinitely, thereby minimizing the need to suppress the retinal flow by recruiting the direct pathway.
Note that once the velocity storage is inserted inside the visual feedback loop, its tendency to leak becomes rather unimportant, since the stored signal is continuously updated by the visual system. Whenever a change in the velocity storage output is necessary to match the visual signal, the stored velocity will increase or decrease exponentially towards the intended value. It is for this reason that the time constant of velocity storage is reduced during rotation in the light when compared with darkness (Cohen et al. 1977; Gizzi and Harper 2003; Waespe and Schwarz 1986). In the light, the time constant depends mostly on the gain of the visual feedback loop and only marginally on the leak of the velocity storage. In other words, the time constant measured in darkness becomes functionally unimportant, because the visual loop automatically updates the stored signal and therefore dominates its dynamics. As we will see next, there is a similar feedback loop from a processed otolith-driven signal to the velocity storage.
Inertial information from the otolith organs
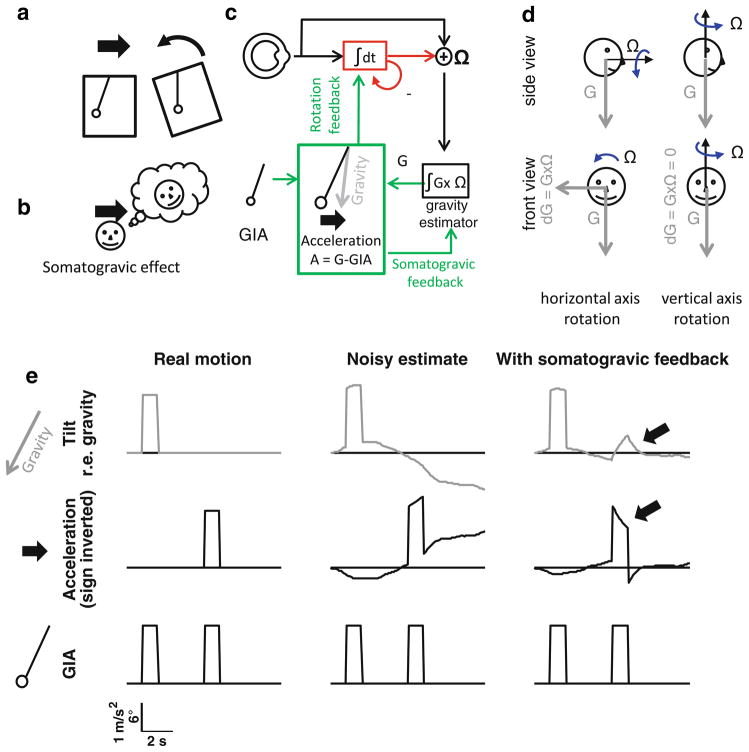
Another component of the vestibular system arises from the otolith organs, which are specialized to detect gravito-inertial acceleration (GIA), i.e., translation in space and tilt relative to gravity. The information provided by the otolith organs can be compared schematically with that of a pendulum (Fig. 2a), and we will use this analogy here. Translational acceleration of the head in a given direction (i.e., towards the right) causes the pendulum to be deviated in the opposite direction (i.e., the left), generating a sensory signal. However, a tilt of the head towards the left also deviates the pendulum in exactly the same manner (Fig. 2a). This is because the physical variable sensed by the otolith organs is the GIA, which is equal to the gravity vector minus the linear (inertial) acceleration. Thus, extracting the linear acceleration from the GIA requires knowledge of head orientation relative to gravity. This problem is called ‘the gravito-inertial ambiguity’, which can physically not be resolved (Einstein 1907) without the use of additional sensory signals.
Fig. 2.
Processing of inertial information. a: gravito-inertial ambiguity (the otoliths are represented as a pendulum inside of a box). b:somatogravic effect: a prolonged linear acceleration is interpreted as a tilt. c: model of canal and otolith information processing. d: Integration of angular velocity by the tilt estimator. The vector cross-product G × Ω allows computation of the rate of change of gravity (left). Note that this product is equal to zero during earth-vertical axis rotation (right). e: Motion estimates (top: tilt relative to gravity; middle: linear (inertial) acceleration; bottom: gravito-inertial acceleration (GIA)) during a tilt of the head followed by a step of inertial acceleration. The sign of the acceleration trace is inverted for clarity. Arrows illustrate soma-togravic effect. See Fig. 4 for simulation parameters
Under most circumstances, such signals are, however, readily available to the brain: a tilt of the head is a rotation which activates the semicircular canals. Therefore, tilt can be distinguished from translation by merging the signal of the canals and the otolith organs. A convenient way to do so consists of integrating rotation velocity over time, in order to keep track of head orientation relative to gravity (or equivalently of head tilt). This allows the estimation of the deviation of the GIA relative to the head attributable to gravity (Fig. 2c). Subtraction from the actual net deviation of the GIA allows computation of the linear acceleration (translation). We will refer to the element that integrates rotation velocity as the ‘tilt estimator’ (block “∫G × Ω” in Fig. 2c). Mathematically, the change of the estimated gravity vector relative to the head can be computed by the vectorial cross-product G × Ω (Fig. 2d), where Ω is the angular velocity vector and G the estimated gravity vector. Notice that this product is equal to zero if the head rotates around an earth-vertical axis, since this rotation does not affect head orientation relative to gravity (it is for this reason that the tilt estimator uses ∫G × Ω, rather than ∫Ω; the latter operation would erroneously estimate a non-zero tilt during earth-vertical axis rotations, e.g., yaw in upright). By integrating this product, G × Ω, over time, the tilt estimator delivers an estimate of the gravity vector that can be subtracted from the GIA in order to compute the translation of the head. Note that this second integrator can be thought of as another internal model, this time not of a sensor but of the physical equations relating signals Ω and G from the two vestibular sensors. This is a direct (forward) model, since it uses an estimate of the variable that causes the tilt (the rotation velocity) in order to compute the tilt.
A simple simulation of head tilt and translation is presented in Fig. 2e, in which the head is tilted for 1 s and then undergoes a linear acceleration for 1 s. Both motions induce the same variation of the GIA (Fig. 2e, bottom row). If the tilt estimator operates noiselessly, then tilt and translation can be discriminated perfectly. In practice, however, this approach is compromised by the same problem as the internal model of canal dynamics, i.e., error accumulation due to the integration of a noisy signal. The G × Ω signal is subject to noise and simply integrating it will cause the estimate of tilt to drift away from the real value, leading to an increasing error in the estimate of translation (Fig. 2e, ‘noisy estimate’).
This problem can be solved in a way which is similar to the leak introduced in the velocity storage, i.e., by adding a feedback loop that slowly and continuously drags the tilt estimate towards the direction indicated by the pendulum (i.e., the GIA). For reasons that will appear obvious soon, this loop is named the ‘somatogravic feedback’ (Fig. 2c) and its function is to bring the gravity estimate towards the GIA, a process that efficiently cancels the drift. Note that the signal fed back to the tilt estimator is simply the difference between GIA and gravity, times a coefficient, and it implements a simple low-pass filter.
But this important functional advantage also comes at a cost. During translation, the tilt estimate increases over time as the estimated gravity moves towards the GIA. This phenomenon, which has been known as the ‘somatogravic illusion’ (Graybiel 1952; Graybiel and Clark 1965; Graybiel et al. 1979; Paige and Seidman 1999), causes a decrease in the translation estimate, as well as an aftereffect at the end of the translational acceleration. The somatogravic illusion could occur rapidly and could be appreciable even during a simulation of a 1 s long translation (Fig. 2e, arrows). Indeed, tilt perception during translation can be sensed at frequencies as high as 0.5 Hz (Merfeld et al. 2005). For reflexive eye movements, torsional modulation during lateral translation in darkness can be seen at frequencies of ~0.5 Hz (Angelaki 1998; Paige and Tomko 1991a, b).
Thus, the caveat of this loop is that it can cause a misinterpretation of translational accelerations as tilt relative to gravity. However, the benefit is important. The somatogravic feedback ensures that the gravity estimate does not drift over time because of noise accumulation, i.e., the system ensures most accurate perception of brief translations and tilts at the expense of less commonly experienced prolonged constant accelerations. Note that this somatogravic feedback essentially generates Gary Paige’s ‘frequency segregation’ filtering seen in behavioral output (Paige and Tomko 1991a, b; Seidman and Paige 1996; Seidman et al. 1998). Importantly, though, the functional significance of this filtering is not to segregate tilt from translation (this is performed by integrating canal, or other rotation-coding, cues). This filtering rather improves the fidelity of mid/high-frequency motion perception at the expense of low-frequency tilt (somatogravic) illusions experienced during low-frequency translations.
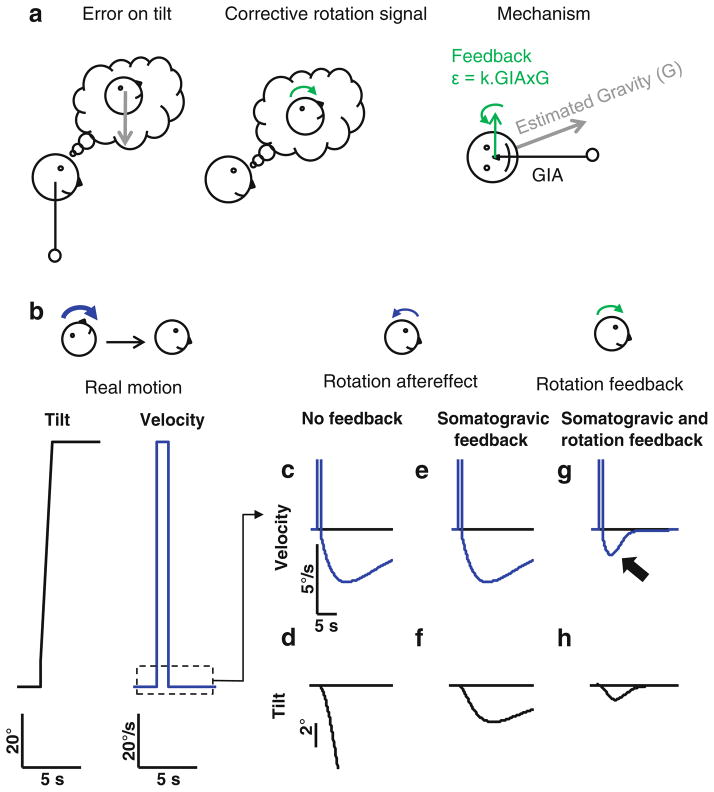
Although the somatogravic feedback improves the noise-driven drift of the tilt estimator, there is another problem that needs to be dealt with. If the angular velocity estimate (input to the tilt estimator) is itself incorrect, then it will cause the tilt estimator to generate new errors continuously. Such inaccurate rotation signals, for example, would commonly exist because of the canal aftereffect (Fig. 1e). In this case, it is more efficient to also directly correct the rotation estimate itself, rather than correcting only the gravity estimate through the somatogravic feedback. This is performed by a second feedback loop that acts on the velocity storage (Fig. 2c). This ‘rotation feedback’ is an angular velocity signal that represents a virtual rotation that would bring the estimated gravity vector back in alignment with the otolith signal (GIA). Such signal adequately opposes incorrect rotation estimates that drive the estimated gravity vector away from the GIA.
The mechanism underlying this rotation feedback is illustrated with an example in Fig. 3a: suppose that the head is upright but that, for some reason, the tilt estimator indicates that it is tilted backwards. This error can be corrected by a forward rotation. This virtual rotation can be computed as the cross-vector product of the estimated gravity (G) and the GIA. Thus, estimated gravity and GIA are continuously monitored and their cross-product, GIA × G, yields a rotation vector in the appropriate direction that would bring the estimated gravity back into alignment with the otolith signal. Note that this rotation feedback signal to the velocity storage allows the system to more accurately compute a rotation estimate using otolith information. Indeed, as will be further discussed in part III, this feedback loop accounts for the ability of the otolith system to generate a steady-state velocity and to improve the low-frequency dynamics of the VOR during off-vertical axis rotations (Angelaki and Hess 1996a, b; Angelaki et al. 2000; Benson and Bodin 1966a; Correia and Guedry 1966; Kushiro et al. 2002; Laurens et al. 2010).
Fig. 3.
Need for somatogravic and rotation feedback loops for inertial processing. a: principle of the rotation feedback signal to the velocity storage. b: example of motion inducing a canal aftereffect. c–h: rotation velocity (top) and tilt estimate (bottom) caused by the canal aftereffect in the absence of any feedback (c, d), in the presence of somatogravic feedback only (e, f; note that, due to its nature, the somatogravic feedback does not affect the velocity estimate (e) but only the tilt estimate (f)), and in the presence of both somatogravic and rotation feedback loops (g, h). See Fig. 4 for simulation parameters (the noise is omitted for clarity)
This velocity feedback acts in synergy with the somatogravic feedback. Its specificity is that it can correct errors in the angular velocity signal coming from the semicircular canals. But, importantly, the functional significance of this velocity feedback loop from the tilt estimator to the velocity storage is not found in low-frequency or constant velocity rotations, where it has been typically tested. Rather, it lies on the need to appropriately estimate tilt and translation under natural, everyday conditions. Consider, for instance, a rotation from supine to upright (Fig. 3b), performed in 1 s (such a movement happens daily as we get up from bed). Such a rotation is strong enough to induce a significant post-rotatory rotation signal (Fig. 3c, note that the noise is omitted from this simulation for clarity, as we are focusing on the consequences of the post-rotatory effect). This erroneous signal from the canals, which indicates a backward rotation, dramatically perturbs the gravity estimate (Fig. 3d). Without correction, the erroneous tilt and translation estimates would cause a considerable disorientation of the subject and compromise equilibrium. Although the somatogravic feedback somewhat counteracts the problem (compare Fig. 3d, f), an erroneous estimate of tilt would remain as long as the rotation signal persists (Fig. 3e). This problem is exasperated because, as explained above, the velocity storage would normally increase the duration of the erroneous rotation estimate. However, because of the conflict between the tilt estimate and the otolith GIA signal, the velocity feedback loop will carry a forward rotation signal to the velocity storage, which will strongly reduce the rotation estimate by ‘dumping’ the post-rotatory response (Fig. 3g, black arrow) and thus also reduce the incorrect tilt estimate (Fig. 3h).
Note that the influence of this velocity feedback on the velocity storage is similar to the influence of visual cues. Thus, the earth-horizontal component of the stored rotation velocity signal is continuously updated by the inertial system, and the exact value of the leak (i.e., time constant of velocity storage in darkness) becomes unimportant. Indeed, this inertial contribution reduces the time constant of the velocity storage during earth-horizontal axis rotations, as shown by e.g., Benson and Bodin (1966b), Dai et al. (1991), Laurens et al. (2010), Merfeld et al. (1993b), and Raphan and Cohen (1985). This is because the functional need to lengthen the canal time constant no longer exists.
Conclusion of part I: importance of spatial orientation processing for normal, every-day activities
An accurate perception of rotation, translation, and spatial orientation relative to gravity is primordial for everyday life. These signals must be correctly computed not only for stabilizing gaze, but also for avoiding illusions, loosing balance, and causing disorientation. The main take-home message from part I of this review is the following: Because of the physical limitations of the peripheral sensors, two important central integrators ensure proper evaluation of vestibular information:
-
The velocity storage is a multisensory element whose function is to compute an accurate estimate of rotation velocity using multiple sensory cues, e.g., canal signals, otolith signals, and vision. An accurate estimate of rotation is critically important not only for itself but also for accurate estimation of tilt and translation. During earth-vertical axis rotations in darkness, where contributions from the other cues are minimized, the velocity storage can be thought of improving the temporal response of the canals by compensating for their dynamics, although the extent of this compensation is limited because of error accumulation.
Traditionally, the function of velocity storage has been regarded as improving rotation estimates at low frequencies (i.e., below ~0.1 Hz) or during long-duration constant velocity rotations (i.e., >5 s). Ironically, though, it is the other way around. An internal model of canal dynamics would provide a frequency-independent, perfect estimate of rotation. This is not possible, however, because of noise accumulation in the integrator, and this is why the integrator is leaky. This leak reduces the accumulation of noise and improves the overall accuracy of the rotation estimator. Thus, a fine compromise is reached.
If low frequencies were important, the time constant of the velocity storage would be increased (reduced leak). The low-frequency response would be improved but the overall amount of integrated noise would be higher. Thus, the time constant of the velocity storage integrator is not adapted to the need to assist low-frequency rotation estimation, but rather to improve accuracy during mid-frequency rotation estimation (0.2–1 Hz, or constant velocity rotations lasting 1–3 s) and consequently also tilt and translation estimation, while compromising low frequencies. This is a fundamental difference in how one thinks of the system.
Importantly, the ecological relevance of the time constant of this integrator is trivialized by the fact that the velocity storage is a multisensory integrator, which allows visual and otolith information to contribute to the estimation of rotation. Accordingly, the velocity storage is continuously updated by feedback from the visual and otolith (inertial) systems and its storage ability allows an accurate estimate of head rotation even during brief interruptions of this feedback. Thus, under normal circumstances, the dynamics of the velocity storage is dominated by the strength of these feedback loops, so that the actual value of the time constant measured during earth-vertical axis rotations in darkness has little impact on its function.
-
A second integrator uses canal and otolith information to compute a robust central estimate of tilt. Like the velocity storage integrator, however, the tilt estimator is also limited dynamically in order to improve the signal-to-noise ratio. To avoid drift because of error accumulation, a somatogravic feedback loop makes the tilt and translation estimates more robust for short-duration movements, at the expense of compromising spatial orientation during prolonged linear accelerations (somatogravic illusion).
Importantly, the functional range of both components, and the central vestibular processing in general, is mid/high frequencies (e.g., 0.2–0.5 Hz or 1–3 s constant velocity head movements). Proper estimation of tilt and translation is of the greatest importance for spatial orientation. To ensure this during everyday activities given the deficiencies of the peripheral vestibular sensors, feedback from the tilt estimator to the velocity storage ensures proper operation and avoids illusions and disorientations, which otherwise would be frequent in everyday life. In fact, without these two interconnected integrators, spatial orientation would be compromised for head movements lasting as little as, or less than, 1 s (Fig. 3), something which would be detrimental for everyday activities. Next, we relate these ideas to more traditional models that have been proposed over the years, with a special emphasis on Bayesian processing. Unlike parametric models where gains and time constants are set arbitrarily or through curve fitting, the Bayesian framework provides a unique intuition, where coefficients approximating pathway gains are set based on signal-to-noise properties and integrator time constants are viewed as priors influencing sensory signals.
Part II: comparison with published models
The full model outlined above, which is presented in Fig. 4a, includes elements from several previous theoretical studies, which we will briefly review and comment in this section. The model merges three modeling approaches: Specifically, the model of Raphan and Cohen (Cohen et al. 1977; Raphan et al. 1977) has defined the velocity storage and shown how it interacts with visual signals. Another line of models, initiated by Oman and Merfeld (Merfeld et al. 1993a; Merfeld 1995; Oman 1982), has shown that the storage activity could be created by the use of an internal model of the canals and has focused on explaining the influence of inertial signals on rotation estimation. Here, we have merged the inertial pathway of Merfeld’s model with the velocity storage element defined by Raphan and Cohen, which is simpler and more intuitive than the internal model of the canals used in Merfeld’s model. The third approach is the Bayesian model (Laurens 2006; Laurens and Droulez 2007, 2008) that directly addresses the issue of sensory noise and takes into account the statistical distribution of head movements in everyday life. We will show in this section how the Bayesian approach relates to the model presented here.
Fig. 4.
Modeling visuo-vestibular processing. a: full representation of the model developed in the previous sections and used for all simulations; blue lines: vestibular pathways; grey lines: visual pathways; green lines: inertial pathways. The parameters used in all simulations are: kV = 0.2, ko = 0.6, τVS = 15 s, go = 2, kF = 0.38, l = 0.65. The time constant of the canals is 4 s. For the simulations of Figs. 1, 2. Gaussian noise with standard deviation of 0.1° (canals), 1° (vision) and 0.4° (velocity storage) is added every 0.1 s. b: schematic model of the principles of Bayesian inference applied to vestibular processing; black lines: deterministic model; lightning bolts: sources of noise; question marks: points of error accumulation; blue lines: influence of the zero velocity prior; green lines: influence of the zero translation prior; grey lines: incorporation of visual information
The inputs of the model are the canal signal, the otolith signal (GIA), and rotational retinal flow, whereas its outputs are: rotation velocity (Ω), the direction of gravity (G), and translation (A = G − GIA). Its architecture can be summarized into three main components:
The vestibular rotation pathway (blue lines in Fig. 4a) is dedicated to detecting fast rotations of the head. Its main feature is the velocity storage (inverse dynamic model), which improves the signal of the canals. Note that there are a number of ways to describe this phenomenon. Here, we have opted for the formalism introduced by Raphan and Cohen (Cohen et al. 1977; Raphan et al. 1977). Other modeling studies have reproduced this improvement of canal signals by various other means, without explicitly using a velocity storage element (e.g., Robinson 1977). The notion of velocity storage as an internal model of canal dynamics can also be found in the studies by Droulez and Perez (1993), MacNeilage et al. (2008), Merfeld et al. (1993a), Merfeld (1995), and Zupan et al. (2002).
The visual pathways (grey lines in Fig. 4a) allow the rotational retinal flow to influence the velocity storage and participate directly to the estimate of rotation. These pathways appear in an identical form as in the model of Raphan and Cohen (Cohen et al. 1977; Raphan et al. 1977), see also (Gizzi and Harper 2003), although other representations are identical (Robinson 1977; Zupan et al. 2002).
The inertial pathways (green lines in Fig. 4a), where rotation information is integrated into an estimate of head tilt, disambiguate the otolith signal. This idea is identical to previous studies putting forward the notion that the brain processes vestibular information using an internal model of the physical equations relating gravity, linear acceleration, and GIA (Angelaki et al. 1999, 2004; Merfeld et al. 1999, see Green and Angelaki 2010; MacNeilage et al. 2008; Poon and Merfeld 2005 for reviews). The implementation of tilt/translation discrimination has been included in several models of vestibular information processing (Borah et al. 1988; Bos and Bles 2002; Droulez and Perez 1993; Glasauer and Merfeld 1997; Green and Angelaki 2004; Laurens 2006; Laurens and Droulez 2007, 2008; Mayne 1974; Merfeld et al. 1993a; Merfeld 1995; Mergner and Glasauer 1999; Oman 1982; Reymond et al. 2002; Zupan et al. 2002). These models also include feedback loops to the tilt estimator and to the estimate of rotation velocity (Droulez and Perez 1993; Glasauer and Merfeld 1997; Merfeld et al. 1993a; Merfeld 1995; Reymond et al. 2002; Zupan et al. 2002).
These models all rely on similar equations, i.e., the tilt estimate is computed as ∫G × Ω and the feedback to the velocity storage as ε = kFGIA × G, where G is the tilt estimate, GIA represents the otolith signal, kF is a coefficient, and x is the vectorial cross-product (note that variants of this formula have been used in certain models but are all approximately equivalent during most motion simulations). Of these, the least well-appreciated signal is the feedback from the inertial system to the velocity storage integrator; this feedback is a virtual rotation vector pointing in such a way that applying this rotation brings G towards GIA (Fig. 3). One should point out that ε is always orthogonal to the GIA. Therefore, if the GIA is aligned with earth-vertical—i.e., when the head is not translating, then ε always indicates a rotation around an earth-horizontal axis. We will show how this feedback affects the rotation estimate during various motion paradigms in part III of this review.
Bayesian model of vestibular processing
In addition to being in line with previous deterministic models using classical control systems and observer theory, the ideas governing central vestibular processing are more intuitively appreciated using the Bayesian framework (Laurens 2006; Laurens and Droulez 2007, 2008; Laurens et al. 2010). One of the many features of the Bayesian approach is that, rather than making hypotheses on how the brain processes sensory information, it makes hypotheses on which information is available to the brain. Once this has been defined, the rules of Bayesian inference determine how the estimate of motion should be computed, thus directly guiding our understanding of central vestibular processing. Another important feature is the handling of biological noise, which is intuitively incorporated into the definition of Bayesian variables.
Importantly, the model outlined in Fig. 4a becomes a direct result from the rules of Bayesian inference (rather than arbitrarily assigning loops and pathways). For example, one can show (Laurens 2006) that, when restricted to rotations around an earth-vertical axis, the Bayesian model is mathematically equivalent to the Raphan and Cohen model of visuo-vestibular interactions (Raphan et al. 1977), i.e., the model of Fig. 1b (blue, red, and gray lines in Fig. 4a) computes optimal estimates of rotation, as predicted by Bayesian inference. As explained by Laurens (Laurens 2006; Laurens et al. 2010), the inertial pathway of Fig. 4a (green) can also approximate optimal processing of otolith information based on Bayesian inference. Next, we first present the fundamental hypotheses of the Bayesian model developed by Laurens (2006) (Fig. 4b) and then describe in more detail the correspondence with the model of Fig. 4a.
Building blocks of a Bayesian model
Deterministic components
The first assumption of the model is that the brain processes information according to the physical and geometrical relationships between the motion and sensory variables. This includes, for instance, the dynamics of the endolymph or the fact that rotation velocity integrated over time yields the orientation of the head in space. These relations are represented in Fig. 4b by black lines. In the absence of noise, a model restricted to these black lines could compute a perfect estimate of head motion indefinitely.
Sensory and processing noise
As we have pointed out earlier, the factor that limits the ability of the brain to compute spatial orientation indefinitely is the noise present at the level of sensory organs or of central processing (note that, for simplicity, the noise in otolith organs has not been considered here since its effect on the dynamics of the model is marginal). This noise accumulates in the integrators and would cause an increasing uncertainty on the estimate of endolymph velocity and head tilt in the absence of additional information.
Priors on head motion
Even in the absence of sensory information, head motion can be expected to follow certain statistical rules, in particular the fact that high velocity or high acceleration movements are infrequent in everyday life. This represents a source of information which is used by the Bayesian model when it is uncertain about head motion. There are two such priors in the Bayesian model of Fig. 4b:
A zero angular velocity prior: High head velocities are uncommonly experienced and, when averaged across time, angular velocity is generally close to zero. Thus, in the absence of robust sensory information, it is more likely that we are stationary than rotating at constant velocity. Similarly, when based on sensory information two angular velocities are equally probable, the prior would pull the estimate towards the lower value. For example, the fact that this prior is responsible for the leak of the velocity storage can be shown by the following example: suppose that the model integrates the signal of the canals for a certain period in order to compute endolymph motion in space. At the end of this period, the model estimates that this velocity is 10°/s, with an error margin of 2°/s due to the noise. Since the error margin is symmetric, the values of 9 and 11°/s are so far equiprobable. However, an endolymph velocity of 11°/s would yield a higher estimate of head velocity, and the prior indicates that high rotation velocities are less probable than low ones. The Bayesian model takes this prior into account and concludes that a low endolymph velocity is more probable. This lowers the average velocity estimate. As this operation is repeated over time, the estimate of endolymph velocity, i.e., the output of the velocity storage, acquires a tendency to decrease continuously. By this process, the prior on zero velocity together with the presence of noise are responsible for the leak of the velocity storage.
A zero linear acceleration prior: High linear accelerations are also unlikely; thus, the second Bayesian prior is that translational acceleration is generally close to zero. This prior is responsible for the somatogravic effect: the noise on the rotation velocity results in uncertainty on head orientation relative to gravity—or equivalently on the direction of the gravity vector relative to the head. Since the linear acceleration estimate is minimized when the estimated gravity vector is close to the GIA, this prior influences the estimate of head tilt relative to gravity.
The fact that this prior affects the velocity storage can be explained as follows. In the example of Fig. 3, the post-rotatory rotation signal indicated that the head was tilting away from gravity, although this was not the case. This led to an estimate of linear acceleration which was unlikely according to this prior. Higher values of the rotation velocity estimate drive the head tilt estimate further away from the earth-vertical and therefore correspond to higher linear acceleration estimates. Thus, when the Bayesian model computes the probability distribution of the rotation velocity during the motion simulated in Fig. 3, higher rotation velocities are disfavored by the prior on linear acceleration. As a result, the average estimate of rotation velocity is shifted towards zero, i.e., the post-rotatory rotation estimate is suppressed. This effect can be modeled by the rotation feedback described earlier. For the same reasons, the Bayesian model also predicts that if the head actually rotates relative to gravity at a certain velocity, then the estimate of rotation will be shifted towards that velocity. The rotation feedback is also performing this function, as will be shown in part III.
Visual information
Whenever the eye velocity generated by the vestibulo-ocular reflex does not match head velocity relative to the environment, the difference generates a retinal flow that can charge or discharge the velocity storage (Fig. 4b, gray solid lines) and also affect directly the final estimate (Fig. 4b, gray dashed line). Laurens (2006) formulated the Bayesian model by using a steady-state Kalman filter, which is a simple optimal estimator applicable to linear problems such as visuo-vestibular interaction. Mathematical analysis demonstrated that this Kalman filter is equivalent to the Raphan and Cohen’s model (Cohen et al. 1977; Raphan et al. 1977); therefore, this model can compute a Bayesian optimal motion estimate.
Conclusion of part II: modeling
Bayesian inference focuses on the information available to the brain and uncovers the principles by which this information should be processed optimally. These principles have been included in a variety of previous deterministic control system models. Nevertheless, whether using classical control system approaches, internal model, and observer theory or Bayesian inference, the main principles that have emerged are similar (MacNeilage et al. 2008), with the velocity storage having a central role.
Importantly, the spatial orientation system of Fig. 4a, b has evolved to extract spatial orientation parameters from ambiguous sensory information during short- and medium-duration movements (<3 s). Indeed, all simulations shown already were based on such short-lasting head movements (Figs. 1, 2, 3). The compromise solutions in Fig. 4a, b occur often at the expense of low frequencies (e.g., <0.1 Hz, or when a rotation is maintained for more than 5 s), where spatial orientation can be critically compromised and where spatial orientation illusions are often experienced. As shown in the studies by MacNeilage et al. (2008) and Laurens (2006), relaxing the Bayesian priors on zero rotational velocity and zero linear acceleration increases the time constant of the velocity storage and the somatogravic effect and therefore improves the performance of the model at low frequencies. However, it also increases the amount of noise on the motion estimates. Thus, although the fact that velocity storage can be best observed during prolonged rotations lasting more than 10 s may suggest that it has emerged to operate at low frequencies, such a concept could not be more incorrect and misleading.
Having made this point, however, we now show next that this same model can nevertheless predict the low-frequency properties of velocity storage that have been described previously by many laboratories, e.g., the steady-state VOR velocity during off-vertical axis rotation (Angelaki and Hess 1996a, b; Angelaki et al. 2000; Kushiro et al. 2002; Laurens et al. 2010) and tilt dumping during post-rotatory nystagmus (Angelaki and Hess 1994; Benson and Bodin 1966b; Laurens et al. 2010; Raphan et al. 1981).
Part III: experimental paradigms of gravity influences on velocity storage
Several experimental studies have demonstrated that the velocity storage is influenced by the orientation or the motion of the head relative to gravity (Angelaki and Hess 1994, 1995a, b, Angelaki and Hess 1996a, b; Dai et al. 1991; Laurens et al. 2010; Raphan and Cohen 1985). The results of these paradigms have inspired a variety of models (Droulez and Perez 1993; Laurens and Droulez 2007; Merfeld et al. 1993a; Merfeld 1995; Raphan and Sturm 1991; Zupan et al. 2002). Several of these rely on the notion that the signal of the otoliths tends to be interpreted as indicating gravity and that the estimated rotation of the head relative to gravity should match this signal. This principle conforms to the Bayesian prior in favor of zero linear acceleration and can be implemented by the rotation feedback loop which we have described. We will review a couple of experimental paradigms in light of this theory and use simulations performed with the model of Fig. 4a to illustrate them.
Off-vertical axis rotation
As previously described, the signal emitted by the canals and the output of the velocity storage fade away during a constant velocity rotation (Fig. 5a). As a consequence, the feeling of rotation totally vanishes within 30 s to 1 min during a constant velocity rotation around an earth-vertical axis in darkness. However, if one rotates relative to gravity in darkness, then the perception of rotation and the VOR persist indefinitely (Angelaki and Hess 1995b, 1996a, b; Angelaki et al. 2000; Benson and Bodin 1966b; Cohen et al. 1983; Correia and Guedry 1966; Darlot et al. 1988; Denise et al. 1988; Guedry 1965; Harris 1987; Haslwanter et al. 2000; Kushiro et al. 2002; Laurens et al. 2010). This paradigm is called off-vertical axis rotation (OVAR) or ‘barbecue rotation’ in the particular case in which a subject is tilted 90° and rotated around the body’s vertical axis (Fig. 5b).
Fig. 5.

Simulations of off-vertical axis rotation (OVAR). a, b: angular velocity estimate during earth-vertical axis rotation (a) and OVAR (b). c: motion of the GIA (black) and the gravity estimate relative to the head during OVAR (gray), and resulting rotation feedback (green). See Fig. 4 for simulation parameters
The persistence of the rotation estimate is easily explained by the model of Fig. 4. During OVAR, the orientation of the head relative to gravity is continuously changing (Fig. 5b). In an egocentric frame of reference, the GIA is continuously rotating around the head (Fig. 5c: blue arrow). Immediately at the beginning of the rotation (Fig. 5b, arrow), the canals indicate the rotation velocity accurately. By integrating the velocity signal, the tilt estimator can track the motion of the head relative to gravity; therefore, the gravity estimate rotates together with the GIA.
After a few seconds, however, the rotation estimate (originating from the canals but through the velocity storage) begins to decay. Since the velocity of the rotation relative to gravity is underestimated, the estimated gravity vector begins to rotate slower and therefore to lag behind the GIA (Fig. 5c, gray). The difference between the two (G estimate versus GIA) generates a feedback signal computed as the cross-vector product GIA × G (Fig. 5c, green) that is sent to the velocity storage. This feedback compensates for the leak and allows the velocity storage to keep a constant velocity estimate, such as to ensure that the estimate of tilt, and thus of translation, is not compromised. This estimate is typically lower than the real rotation velocity and depends on how much the rotation axis is tilted relative to gravity, in a way which can be predicted by the Bayesian model (Laurens 2006; Laurens et al. 2010). The steady-state estimate of rotation represents an equilibrium where the angle between the estimated gravity and the GIA remains constant.
Post-rotatory tilt
The post-rotatory tilt is one of the clearest demonstrations of the influence of gravity on the velocity storage (Angelaki and Hess 1994, 1995a; Benson and Bodin 1966b; Dai et al. 1991; Fetter et al. 1996; Furman and Koizuka 1994; Hess and Angelaki 1995; 1997; Hess et al. 2005; Jaggi-Schwarz et al. 2000; Laurens et al. 2010; Merfeld et al. 1993b; Raphan et al. 1981; Waespe et al.1985). It consists of rotating a subject until the signal of the canals (and velocity storage) has subsided to zero. The subject is then abruptly stopped: this generates a post-rotatory stimulation of the canals, indicating a rotation in the direction opposite to the initial rotation (Fig. 6a). Immediately after the stop, the subject is tilted to the side.
Fig. 6.
Post-rotatory tilt paradigm. a: illustration of the paradigm and of the rotation indicated by the semicircular canals. b: motion of the estimated gravity caused by the rotation signal (grey arrows) and resulting rotation feedback (green). c, e: rotation estimates during the paradigm, in space-fixed (c) or head-fixed (e) coordinates. d, f: effects of the feedback signal (green arrow) in space-fixed (d) or head-fixed (f) coordinates. See Fig. 4 for simulation parameters
The tilt generates a canal/otolith conflict, where the signal of the canals indicates a yaw rotation around an axis which is now off-vertical. This yaw rotation signal is processed by the tilt estimator, which predicts that the head should rotate from an ear-down towards a nose-down orientation, although the head is in fact not moving (Fig. 5a). Consequently, the tilt estimate (G) rotates away from the GIA, which remains stationary (Fig. 6b). This discrepancy generates a corrective rotation feedback signal corresponding to a virtual rotation about an axis perpendicular to GIA, as is illustrated in Fig. 6b (green).
The effect of this feedback is shown in Fig. 6d. The rotation initially indicated by the canals is represented by the black arrow which is tilted relative to earth-vertical. The feedback brings the rotation estimate towards alignment with the earth-vertical. The time course of the resulting rotation estimate is shown in Fig. 6c in an earth-fixed reference frame. Immediately after the tilt, the earth-vertical and earth-horizontal components of the rotation signal are equal, since the tilt angle is 45°. Then the feedback loop, which provides an earth-horizontal correction signal to the velocity storage, shortens the horizontal component (Fig. 6c, grey arrow), while the earth-vertical component is essentially unaffected.
It is also noteworthy to consider the response to post-rotatory tilt in a head-fixed reference frame (Fig. 6e, f), as is the case when examining the VOR (where eye velocity is measured relative to the head). The rotation signal is initially aligned with the head-vertical (z) axis. By shifting it towards earth-vertical, the feedback loop induces a horizontal (y) component in the rotation estimate. Examination of the time course (Fig. 6e) shows that the post-rotatory tilt results in a rapid dumping of the head-vertical component, together with the appearance of an orthogonal (horizontal) component. Once the resulting vector has aligned with the earth-vertical, the rotation estimate decays slowly, with a time constant, which is characteristic of the velocity storage (Laurens et al. 2010).
In summary, the response to post-rotatory tilt can be expressed in two frames of reference. In the earth-fixed reference frame, the brain resolves the conflict induced by the post-rotatory canal signal by dumping the earth-horizontal component, which is responsible for the conflict. In a head-fixed reference frame, the same response can be expressed as a dumping and a realignment, both of which happen simultaneously. The realignment induces a rotation estimate around an axis (the y axis in this example), which is orthogonal to the axis of the initial canal activation (z). By realigning the estimated rotation vector, this induced component participates in resolving the canal-otolith conflict.
Conclusion of part III
The two paradigms presented here have been the most widely used to study the influence of the otolith (gravity) signals on the velocity storage. We have shown that they generate responses that can be explained by the velocity feedback loop, which is designed to correct mismatches between the internal estimate of rotation and the motion of the head relative of gravity. Importantly, the responses in Figs. 5, 6 are of little, if any, functional significance per se; they simply manifest the existence of the velocity feedback loop between the tilt estimator and velocity storage. Recall that this feedback signal is needed in order to improve the rotation estimate used for the calculation of tilt and translation. As illustrated in Fig. 3, without this feedback, an erroneous tilt, and thus an erroneous translation, signal would be generated every day we get up from bed.
In summary, with these examples, we hope to have illustrated the following paradox: On one hand, research in the field has used for decades the low-frequency regime during OVAR and post-rotatory tilt to study gravity influences on velocity storage (Figs. 5, 6). These responses, when considered in isolation, have little ecological relevance. On the other hand, the elements of central vestibular processing (Fig. 4) have emerged based on functional demands during natural head movements (Figs. 1, 2, 3). Yet these natural conditions have not been studied in the laboratory, as findings would be boring: when all elements of central vestibular processing are intact (Fig. 4), spatial orientation works perfectly, perception is robust, and the VOR is as it should be. Thus, a group of researchers focused on the low-frequency regime where findings become ‘interesting’, as the influence of these loops manifests themselves clearly on behavior (e.g., gravity-dependent VOR responses). However, the relation between these paradigms and ecologically relevant conditions has remained obscure, and most researchers might not have appreciated that these seemingly complex, low-frequency properties reflect the existence of a beautiful spatial orientation system that represents a superb example of neural computation.
We have shown how erroneous and problematic spatial orientation and motion estimation would be during natural head movements if any one (or all) of the elements of central vestibular processing (i.e., velocity storage, tilt estimator, somatogravic, and rotation feedbacks) were not in place (e.g., see Figs. 1, 2, 3). We do hope that this review achieves its goal to bring the two groups of researchers together, by helping those who have been skeptics understand that there is a treasure of extreme functional significance hidden behind the gravity-dependent low-frequency properties of the velocity storage.
Function of velocity storage: multisensory rotation estimator
In this review, we have also advocated the Bayesian framework and emphasized how Bayesian inference allows linking the central vestibular processing with the physical laws which govern motion in space and sensor properties. The fact that this link has been recognized by a few is not new, as shown by the following lines written in 1977 by David A Robinson regarding velocity storage: “[…] this is just a restatement of Newton’s second law of motion. Whenever a body (you) is set into motion, it is presumed that you will continue in that motion until you are acted upon by another force, at which time your canals will tell you. […] Thus, even without vision, the brain is using the properties of positive feedback to recognize and simulate simple laws of physics to improve on the design of the canals” (Robinson 1977).
In the following decades, several theoretical studies have been based on the notion that internal models of the laws of physics and of sensory organ dynamics are key to how the brain interprets vestibular information. The Bayesian model is the heir to this line of work, which was refined by removing from the model’s design everything other than assumptions about the physical world. By introducing sensory noise and priors about body motion into a rigorous mathematical formalism, the Bayesian model predicts how vestibular processing is shaped by these constraints. Finally, by focusing on the processing of rotation information during stimulations of the canals, the otolith organs, and the visual system, the model has presented the velocity storage as an optimal multisensory integrator placed at the center of spatial orientation processing.
Under most natural activities, the velocity storage receives continuous inputs from the visual and inertial systems (Fig. 4). These inputs determine its activity to such a large extent that we should think of the velocity storage primarily as a multisensory integrator rather than as a process dedicated to lengthening the time constant of rotation perception. Importantly, this lengthening of the time constant is not ecologically relevant because it only exists during earth-vertical axis rotations in darkness (i.e., in the absence of otolith and visual information). In contrast, there is no time constant lengthening during either rotations relative to gravity (post-rotatory tilt and OVAR experiments have demonstrated that the VOR time constant drops to ~3–7 s, see Benson and Bodin 1966b; Dai et al. 1991; Laurens et al. 2010) or in the presence of vision (Cohen et al. 1977; Gizzi and Harper 2003; Waespe and Schwarz 1986).
Thus, an important take-home message from this review is that lengthening of the semicircular canal time constant, which has been used to define the concept of velocity storage (Raphan et al. 1977), does not exist during 99% (if not more) of our daily lives when the velocity storage is under the influence of vision and otolith signals. Instead, velocity storage should be viewed as a central, multisensory rotation estimator that represents a critical element of spatial orientation as it provides input signals for tilt and translation estimation. But its storage ability during earth-vertical axis rotation in darkness should be considered as an anecdotic aspect of its function which, in our view, is optimal multi-sensory processing.
General comments
The motivation for this review came after a promise one of us made to Jay after a flight from Reykjavik to Boston in August 2010. For those who have not had the opportunity to spend 5 h sitting and talking to Jay undisturbed, it is an experience not to be missed. There are a few topics that we and Jay disagree and the velocity storage is (or was?) one of them. So, this review has been written for Jay and for others who, like Jay, have not appreciated the significance of velocity storage. On that flight, it became apparent that it has been our fault (i.e., those of us who have studied extensively and remain intrigued by the mysteries of velocity storage) not articulating in a comprehensive way what has been so obvious to us for so long. We hope that this review has at least partly achieved this goal.
In closing, it is worth pointing out that those of us who have always been intrigued and allured by the mysteries of velocity storage do so because we view the system as a prime example of neural computation that rivals (and probably wins) over other sensory systems. It is also worth highlighting the fact that it is often beneficial to study a system in the laboratory using experimental protocols that are artificial and far from everyday, natural movements. This is because such artificial stimuli are often the best to reveal the underlying mechanisms. But such a statement should be of little surprise to most, as this is how all major discoveries in other systems have been made (e.g., Hubel and Wiesel (Hubel and Wiesel 1962) did not discover orientation columns using natural visual stimuli!). In this respect, we can only speculate that experimental protocols like OVAR and post-rotatory tilt (as well as ‘pitch while rotating’, a stimulus that we have not considered here; but see Hess and Angelaki 1993; Raphan et al. 1983) will prove extremely important in revealing how these computations are made by neural populations. Of course, if one uses unnatural experimental protocols, it is also important to put findings into proper perspective and to be able to see the whole picture, and this is where modeling and modelers have an advantage. But the modelers must communicate better the basic intuitions to the rest of the field. What is written here is not of any surprise to the modelers of velocity storage and spatial orientation, but we do hope it is of use to those who are not.
Acknowledgments
This work was supported by NIH grant EY12814
Footnotes
It is so because the acceleration of the endolymph in space is due to the friction between the endolymph and the canals. This friction is proportional to the difference in velocity between the canal and the endolymph, which is what is reported by the canal. Therefore, a perfect integration of the sensory signal of the canals yields a signal which is proportional to endolymph velocity in space.
References
- Angelaki DE. Three-dimensional organization of otolith-ocular reflexes in rhesus monkeys. III. Responses to translation. J Neurophysiol. 1998;80:680–695. doi: 10.1152/jn.1998.80.2.680. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Cullen KE. Vestibular system: the many facets of a multimodal sense. Annu Rev Neurosci. 2008;31:125–150. doi: 10.1146/annurev.neuro.31.060407.125555. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Hess BJ. Inertial representation of angular motion in the vestibular system of rhesus monkeys. I. Vestibuloocular reflex. J Neurophysiol. 1994;71:1222–1249. doi: 10.1152/jn.1994.71.3.1222. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Hess BJ. Inertial representation of angular motion in the vestibular system of rhesus monkeys. II. Otolith-controlled transformation that depends on an intact cerebellar nodulus. J Neurophysiol. 1995a;73:1729–1751. doi: 10.1152/jn.1995.73.5.1729. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Hess BJ. Lesion of the nodulus and ventral uvula abolish steady-state off-vertical axis otolith response. J Neurophysiol. 1995b;73:1716–1720. doi: 10.1152/jn.1995.73.4.1716. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Hess BJ. Three-dimensional organization of otolith-ocular reflexes in rhesus monkeys. I. Linear acceleration responses during off-vertical axis rotation. J Neurophysiol. 1996a;75:2405–2424. doi: 10.1152/jn.1996.75.6.2405. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Hess BJ. Three-dimensional organization of otolith-ocular reflexes in rhesus monkeys. II. Inertial detection of angular velocity. J Neurophysiol. 1996b;75:2425–2440. doi: 10.1152/jn.1996.75.6.2425. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, McHenry MQ, Dickman JD, Newlands SD, Hess BJ. Computation of inertial motion: neural strategies to resolve ambiguous otolith information. J Neurosci. 1999;19:316–327. doi: 10.1523/JNEUROSCI.19-01-00316.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angelaki DE, Merfeld DM, Hess BJ. Low-frequency otolith and semicircular canal interactions after canal inactivation. Exp Brain Res. 2000;132:539–549. doi: 10.1007/s002210000364. [DOI] [PubMed] [Google Scholar]
- Angelaki DE, Shaikh AG, Green AM, Dickman JD. Neurons compute internal models of the physical laws of motion. Nature. 2004;430:560–564. doi: 10.1038/nature02754. [DOI] [PubMed] [Google Scholar]
- Benson A, Bodin M. Interaction of linear and angular accelerations on vestibular receptors in man. Aerospace Med. 1966a;37:144–154. [PubMed] [Google Scholar]
- Benson A, Bodin M. Effect of orientation to the gravitational vertical on nystagmus following rotation about a horizontal axis. Acta Otolaryngol. 1966b;7:1136–1140. doi: 10.3109/00016486609127090. [DOI] [PubMed] [Google Scholar]
- Borah J, Young LR, Curry RE. Optimal estimator model for human spatial orientation. Ann N Y Acad Sci. 1988;545:51–73. doi: 10.1111/j.1749-6632.1988.tb19555.x. [DOI] [PubMed] [Google Scholar]
- Bos JE, Bles W. Theoretical considerations on canal-otolith interaction and an observer model. Biol Cybern. 2002;86:191–207. doi: 10.1007/s00422-001-0289-7. [DOI] [PubMed] [Google Scholar]
- Cohen B, Matsuo V, Raphan T. Quantitative analysis of the velocity characteristics of optokinetic nystagmus and optokinetic after-nystagmus. J Physiol. 1977;270:321–344. doi: 10.1113/jphysiol.1977.sp011955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen B, Suzuki JI, Raphan T. Role of the otolith organs in generation of horizontal nystagmus: effects of selective labyrinthine lesions. Brain Res. 1983;276:159–164. doi: 10.1016/0006-8993(83)90558-9. [DOI] [PubMed] [Google Scholar]
- Correia M, Guedry F. Modification of vestibular responses as a function of rate of rotation about an earth-horizontal axis. Acta Otolaryngol. 1966;62:297–308. doi: 10.3109/00016486609119575. [DOI] [PubMed] [Google Scholar]
- Dai M, Raphan T, Cohen B. Spatial orientation of the vestibular system: dependence of optokinetic after-nystagmus on gravity. J Neurophysiol. 1991;66:1422–1439. doi: 10.1152/jn.1991.66.4.1422. [DOI] [PubMed] [Google Scholar]
- Darlot C, Denise P, Droulez J, Cohen B, Berthoz A. Eye movements induced by off-vertical axis rotation (OVAR) at small angles of tilt. Exp Brain Res. 1988;73:91–105. doi: 10.1007/BF00279664. [DOI] [PubMed] [Google Scholar]
- Denise P, Darlot C, Droulez J, Cohen B, Berthoz A. Motion perceptions induced by off-vertical axis rotation (OVAR) at small angles of tilt. Exp Brain Res. 1988;73:106–114. doi: 10.1007/BF00279665. [DOI] [PubMed] [Google Scholar]
- Droulez J, Perez VC. Application of the coherence scheme to the multisensory fusion problem. In: Berthoz A, editor. Multisensory control of movement. Oxford University Press; Oxford: 1993. pp. 485–501. [Google Scholar]
- Einstein A. Uber das Relativitatsprinzip und die aus demselben gezogenen Folgerungen. Jahrbuch der Radioaktivität und Elek-tronik. 1907;4:411–462. [Google Scholar]
- Fernández C, Goldberg JM. Physiology of peripheral neurons innervating semicircular canals of the squirrel monkey. II. Response to sinusoidal stimulation and dynamics of peripheral vestibular system. J Neurophysiol. 1971;34:661–675. doi: 10.1152/jn.1971.34.4.661. [DOI] [PubMed] [Google Scholar]
- Fetter M, Heimberger J, Black R, Hermann W, Sievering F, Dichgans J. Otolith-semicircular canal interaction during postrotatory nystagmus in humans. Exp Brain Res. 1996;108:463–472. doi: 10.1007/BF00227269. [DOI] [PubMed] [Google Scholar]
- Furman J, Koizuka I. Reorientation of poststimulus nystagmus in tilted humans. J Vestib Res. 1994;4:421–428. [PubMed] [Google Scholar]
- Gizzi MS, Harper HW. Suppression of the human vestibulo-ocular reflex by visual fixation or forced convergence in the dark, with a model interpretation. Curr Eye Res. 2003;26:281–290. doi: 10.1076/ceyr.26.4.281.15426. [DOI] [PubMed] [Google Scholar]
- Glasauer S, Merfeld DM. Modelling three-dimensional vestibular responses during complex motion stimulation. In: Fetter M, Haslwanter T, Misslisch H, editors. Three-dimensional kinematics of eye head and limb movements. Harwood academic; Amsterdam: 1997. pp. 387–398. [Google Scholar]
- Goldberg JM, Fernández C. Physiology of peripheral neurons innervating semicircular canals of the squirrel monkey. I. Resting discharge and response to constant angular accelerations. J Neurophysiol. 1971;34:635–660. doi: 10.1152/jn.1971.34.4.635. [DOI] [PubMed] [Google Scholar]
- Graybiel A. Oculogravic illusion. Arch Ophthal. 1952;48:605–615. doi: 10.1001/archopht.1952.00920010616007. [DOI] [PubMed] [Google Scholar]
- Graybiel A, Clark B. Validity of the oculogravic illusion as a specific indicator of otolith function. Aerospace Med. 1965;36:1173–1181. [Google Scholar]
- Graybiel A, Johnson W, Money K, Malcolm R, Jennings G. Oculogravic illusion in response to straight-ahead acceleration of CF-104 aircraft. Aviat Space Environ Med. 1979;50:382–386. [PubMed] [Google Scholar]
- Green AM, Angelaki DE. An integrative neural network for detecting inertial motion and head orientation. J Neurophysiol. 2004;92:905–925. doi: 10.1152/jn.01234.2003. [DOI] [PubMed] [Google Scholar]
- Green AM, Angelaki DE. Internal models and neural computation in the vestibular system. Exp Brain Res. 2010;200:197–222. doi: 10.1007/s00221-009-2054-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guedry F. Orientation of the rotation-axis relative to gravity: its influence on nystagmus and the sensation of rotation. Acta Otolaryngol. 1965;60:30–48. doi: 10.3109/00016486509126986. [DOI] [PubMed] [Google Scholar]
- Harris L. Vestibular and optokinetic eye movements evoked in the cat by rotation about a tilted axis. Exp Brain Res. 1987;66:522–532. doi: 10.1007/BF00270685. [DOI] [PubMed] [Google Scholar]
- Haslwanter T, Jaeger R, Mayr S, Fetter M. Three-dimensional eye-movement responses to off-vertical axis rotations in humans. Exp Brain Res. 2000;134:96–106. doi: 10.1007/s002210000418. [DOI] [PubMed] [Google Scholar]
- Hess BJ, Angelaki DE. Angular velocity detection by head movements orthogonal to the plane of rotation. Exp Brain Res. 1993;95:77–83. doi: 10.1007/BF00229656. [DOI] [PubMed] [Google Scholar]
- Hess BJ, Angelaki DE. Inertial representation of visual and vestibular self-motion signals. In: Mergner T, Hlavacka F, editors. Multi-sensory Control of Posture. Plenum Press; New York: 1995. pp. 183–190. [Google Scholar]
- Hess BJ, Angelaki DE. Inertial vestibular coding of motion: concepts and evidence. Curr Opin Neurobiol. 1997;7:860–866. doi: 10.1016/s0959-4388(97)80147-x. [DOI] [PubMed] [Google Scholar]
- Hess BJ, Jaggi-Schwarz K, Misslisch H. Canal-otolith interactions after off-vertical axis rotations. II. Spatiotemporal properties of roll and pitch postrotatory vestibuloocular reflexes. J Neurophysiol. 2005;93:1633–1646. doi: 10.1152/jn.00383.2004. [DOI] [PubMed] [Google Scholar]
- Hubel DH, Wiesel TN. Receptive fields, binocular interaction and functional architecture in the cat’s visual cortex. J Physiol. 1962;160:106–154. doi: 10.1113/jphysiol.1962.sp006837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaggi-Schwarz K, Misslisch H, Hess BJ. Canal-otolith interactions after off-vertical axis rotations I. Spatial reorientation of horizontal vestibuloocular reflex. J Neurophysiol. 2000;83:1522–1535. doi: 10.1152/jn.2000.83.3.1522. [DOI] [PubMed] [Google Scholar]
- Kushiro K, Dai M, Kunin M, Yakushin SB, Cohen B, Raphan T. Compensatory and orienting eye movements induced by off-vertical axis rotation (OVAR) in monkeys. J Neurophysiol. 2002;88:2445–2462. doi: 10.1152/jn.00197.222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laurens J. Dissertation. Université Pierre et Marie Curie (Paris 6); 2006. modelisation bayesienne de interactions visuo-vestibulaires. [Google Scholar]
- Laurens J, Droulez J. Bayesian processing of vestibular information. Biol Cybern. 2007;96:389–404. doi: 10.1007/s00422-006-0133-1. [DOI] [PubMed] [Google Scholar]
- Laurens J, Droulez J. Bayesian modelling of visuo-vestibular interactions. In: Bessière P, Laugier C, Siegwart R, editors. Probabilistic reasoning and decision making in sensory-motor systems. Springer; New York: 2008. pp. 279–300. [Google Scholar]
- Laurens J, Straumann D, Hess BJ. Processing of angular motion and gravity information through an internal model. J Neurophysiol. 2010;104:1370–1381. doi: 10.1152/jn.00143.2010. [DOI] [PubMed] [Google Scholar]
- MacNeilage PR, Ganesan N, Angelaki DE. Computational approaches to spatial orientation: from transfer functions to dynamic Bayesian inference. J Neurophysiol. 2008;100:2981–2996. doi: 10.1152/jn.90677.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mayne R. A systems concept of the vestibular organs. In: Kornhuber HH, editor. Handbook of Sensory Physiology. Springer; Berlin: 1974. pp. 493–580. [Google Scholar]
- Merfeld DM. Modeling the vestibulo-ocular reflex of the squirrel monkey during eccentric rotation and roll tilt. Exp Brain Res. 1995;106:123–134. doi: 10.1007/BF00241362. [DOI] [PubMed] [Google Scholar]
- Merfeld DM, Young LR, Oman CM, Shelhamer MJ. A multidimensional model of the effect of gravity on the spatial orientation of the monkey. J Vestib Res. 1993a;3:141–161. [PubMed] [Google Scholar]
- Merfeld DM, Young LR, Paige G, Tomko DL. Three dimensional eye movements of squirrel monkeys following postrotatory tilt. J Vestib Res. 1993b;3:123–139. [PubMed] [Google Scholar]
- Merfeld DM, Zupan LH, Peterka RJ. Humans use internal models to estimate gravity and linear acceleration. Nature. 1999;398:615–618. doi: 10.1038/19303. [DOI] [PubMed] [Google Scholar]
- Merfeld DM, Park S, Gianna-Poulin C, Black FO, Wood SJ. Vestibular perception and action employ qualitatively different mechanisms. I. Frequency response of VOR and perceptual responses during Translation and Tilt. J Neurophysiol. 2005;94:186–198. doi: 10.1152/jn.00904.2004. [DOI] [PubMed] [Google Scholar]
- Mergner T, Glasauer S. A simple model of vestibular canal-otolith signal fusion. Ann N Y Acad Sci. 1999;871:430–434. doi: 10.1111/j.1749-6632.1999.tb09211.x. [DOI] [PubMed] [Google Scholar]
- Oman CM. A heuristic mathematical model for the dynamics of sensory conflict and motion sickness. Acta Otolaryngol Supp. 1982;392:1–44. [PubMed] [Google Scholar]
- Paige G, Seidman S. Characteristics of the VOR in response to linear acceleration. Ann N Y Acad Sci. 1999;871:123–135. doi: 10.1111/j.1749-6632.1999.tb09179.x. [DOI] [PubMed] [Google Scholar]
- Paige G, Tomko DL. Eye movement responses to linear head motion in the squirrel monkey. I. Basic characteristics. J Neurophysiol. 1991a;65:1170–1182. doi: 10.1152/jn.1991.65.5.1170. [DOI] [PubMed] [Google Scholar]
- Paige G, Tomko DL. Eye movement responses to linear head motion in the squirrel monkey. II. Visual-vestibular interactions and kinematic considerations. J Neurophysiol. 1991b;65:1183–1196. doi: 10.1152/jn.1991.65.5.1183. [DOI] [PubMed] [Google Scholar]
- Poon C, Merfeld DM. Internal models: the state of the art. (Editorial for special issue: Sensory integration, state estimation, and motor control in the brain: role of internal models) Journal of Neural Eng. 2005:2. [Google Scholar]
- Raphan T, Cohen B. Velocity storage and the ocular response to multidimensional vestibular stimuli. Rev Oculomotor Res. 1985;1:123–143. [PubMed] [Google Scholar]
- Raphan T, Cohen B. The vestibulo-ocular reflex in three dimensions. Exp Brain Res. 2002;145:1–27. doi: 10.1007/s00221-002-1067-z. [DOI] [PubMed] [Google Scholar]
- Raphan T, Sturm D. Modelling the spatiotemporal organization of velocity storage in the vestibuloocular reflex by optokinetic studies. J Neurophysiol. 1991;66:1410–1421. doi: 10.1152/jn.1991.66.4.1410. [DOI] [PubMed] [Google Scholar]
- Raphan T, Cohen B, Matsuo V. A velocity storage mechanism responsible for optokinetic nystagmus (OKN), optokinetic after-nystagmus (OKAN) and vestibular nystagmus. In: Baker R, Berthoz A, editors. Control of gaze by brain stem neurons. Elsevier; Amsterdam: 1977. pp. 37–47. [Google Scholar]
- Raphan T, Cohen B, Henn V. Effects of gravity on rotatory nystagmus in monkeys. Ann N Y Acad Sci. 1981;374:44–55. doi: 10.1111/j.1749-6632.1981.tb30859.x. [DOI] [PubMed] [Google Scholar]
- Raphan T, Cohen B, Suzuki JI, Henn V. Nystagmus generated by sinusoidal pitch while rotating. Brain Res. 1983;276:165–172. doi: 10.1016/0006-8993(83)90559-0. [DOI] [PubMed] [Google Scholar]
- Reymond G, Droulez J, Kemeny A. Visuovestibular perception of self-motion modeled as a dynamic optimization process. Biol Cybern. 2002;87:301–314. doi: 10.1007/s00422-002-0357-7. [DOI] [PubMed] [Google Scholar]
- Robinson D. Vestibular and optokinetic symbiosis: an example of explaining by modeling. In: Baker R, Berthoz A, editors. Control of gaze by brain stem neurons. Elsevier; Amsterdam: 1977. pp. 49–58. [Google Scholar]
- Seidman S, Paige G. Perception and eye movement during low-frequency centripetal acceleration. Ann N Y Acad Sci. 1996;781:693–695. doi: 10.1111/j.1749-6632.1996.tb15762.x. [DOI] [PubMed] [Google Scholar]
- Seidman S, Telford L, Paige G. Tilt perception during dynamic linear acceleration. Exp Brain Res. 1998;119:307–314. doi: 10.1007/s002210050346. [DOI] [PubMed] [Google Scholar]
- Waespe W, Schwarz U. Characteristics of eye velocity storage during periods of suppression and reversal of eye velocity in monkeys. Exp Brain Res. 1986;65:49–58. doi: 10.1007/BF00243829. [DOI] [PubMed] [Google Scholar]
- Waespe W, Cohen B, Raphan T. Dynamic modification of the vestibulo-ocular reflex by the nodulus and uvula. Science. 1985;217:151–160. doi: 10.1126/science.3871968. [DOI] [PubMed] [Google Scholar]
- Zupan LH, Merfeld DM, Darlot C. Using sensory weighting to model the influence of canal, otolith and visual cues on spatial orientation and eye movements. Biol Cybern. 2002;86:209–230. doi: 10.1007/s00422-001-0290-1. [DOI] [PubMed] [Google Scholar]