Abstract
Police officers are required to work irregular hours, which induces stress, fatigue, and sleep disruption, and they have higher rates of chronic disease and mortality. Cortisol is a well-known “stress hormone” produced via activation of the hypothalamic-pituitary-adrenal axis. An abnormal secretion pattern has been associated with immune system dysregulation and may serve as an early indicator of disease risk. This study examined the effects of long- and short-term shiftwork on the cortisol awakening response among officers (n = 68) in the Buffalo Cardio-Metabolic Occupational Police Stress (BCOPS) pilot study (2001–2003). The time each officer spent on day (start time: 04:00–11:59 h), afternoon (12:00–19:59 h), or night (20:00–03:59 h) shifts was summarized from 1994 to examination date to characterize long-term (mean: 14 ± 9 yrs) and short-term (3, 5, 7, or 14 days prior to participation) shiftwork exposures. The cortisol awakening response was characterized by summarizing the area under the curve (AUC) for samples collected on first awakening, and at 15-, 30-, and 45-min intervals after waking. Data were collected on a scheduled training or off day. The cortisol AUC with respect to ground (AUCG) summarized total cortisol output after waking, and the cortisol AUC with respect to increase (AUCI) characterized the waking cortisol response. Officers also completed the Center for Epidemiologic Studies Depression scale. Waking cortisol AUC values were lower among officers working short-term night or afternoon shifts than day shifts, with maximal differences occurring after 5 days of shiftwork. The duration of long-term shiftwork was not associated with the cortisol awakening response, although values were attenuated among officers with more career shift changes.
Keywords: Cortisol awakening response, Police, Shiftwork, Stress
Introduction
Police officers suffer disproportionately from a variety of adverse health outcomes, including sleep disruption, psychological stress, depression, suicide, metabolic disorders, heart disease, diabetes, and higher mortality (Franke et al., 1997; Vena et al., 1986; Vila, 2006; Violanti et al., 1998, 2006). Increased risks for developing heart disease among police likely arise, at least in part, from a higher prevalence of hypertension and obesity in this occupational group compared to the general population (Franke et al., 1997; Ramey et al., 2008). In addition, higher rates of cancer, including those of the colon, bladder, digestive tract, brain, esophagus, hematopoietic tissues, as well as lymphomas and melanomas have been associated with police work (Forastiere et al., 1994; Vena et al., 1986). The lengthy, stressful, and sometimes life-threatening work that officers face often requires working irregular hours, including nights. There is considerable overlap between diseases observed among police officers and those observed in other occupations that include shiftwork. For example, shiftwork has been associated with higher risks of prostate, breast, and colorectal cancer, non-Hodgkin's lymphoma, elevated blood pressure, and circulating cholesterol, cardiovascular disease (CVD), and increased rates of occupational accidents and injuries (Boggild & Knutsson, 1999; Chen et al., 2010; Conlon et al., 2007; Dembe et al., 2006; Ha & Park, 2005; Horwitz & McCall, 2004; Karlsson et al., 2003; Kubo et al., 2006; Lavie & Lavie, 2007; Schernhammer et al., 2001, 2003). However, the extent to which excesses in morbidity and mortality among police officers are due to shiftwork is not completely understood, and research on this subject has been limited.
Shiftwork can result in circadian dysregulation, sleep insufficiency, and disruption of familial and social life (Åkerstedt, 1990, 1998; Burch et al., 2005,2009; Camerino et al., 2010; Gordon et al., 1986; Harada et al., 2005; Ohayon et al., 2010; Shields, 2002), which can lead to increased allostatic load (defined as strain on the body and changes in metabolism produced by repeated cycling of the stress response) (McEwen & Stellar, 1993), altered homeostasis, plus immune, endocrine, and autonomic dysfunction (Navara & Nelson, 2007). These changes may contribute to the pathophysiological mechanisms underlying the elevated disease risks that have been observed among shiftworkers. Psychological impacts, in some instances manifested as increases in depressive symptoms, have also been observed among shiftworkers through use of the Center for Epidemiologic Studies Depression (CES-D) scale or other instruments that ascertain mood (Cavallo et al., 2002; Coffey et al., 1988; Gordon et al., 1986; Harada et al., 2005; Takahashi et al., 2006). The specific biological processes that are linked with these adverse outcomes have not been fully characterized, but are likely to be critical to the development of effective disease prevention and intervention strategies among police officers, as are the temporal aspects of shiftwork-related physiological or psychological perturbations.
Cortisol is an adrenal steroid classically referred to as a “stress hormone” (Henry, 1992) that can be conveniently measured in saliva where its secretion pattern mimics concentrations in circulation (Lippi et al., 2009). Acute exposure to a stressor can result in elevated circulating cortisol levels for periods of 45 to 60 min after initiation of the stress response (Clow et al., 1997; Khalfa et al., 2003). There are a number of regulatory processes that facilitate maintenance of basal cortisol levels after initiation of the stress response (McEwen, 2006; McEwen & Stellar, 1993). However, during prolonged periods of stress, the ability of the hypothalamic-pituitary-adrenal (HPA) axis to self-regulate cortisol secretion may become compromised, which can result in an “exhaustion stage” or inability of the body to regulate the stress response. This breakdown in control of the stress response, sometimes referred to as allostatic overload (McEwen, 2008), can be manifested as a change in the pattern of the awakening response or a failure of the body to control the release of cortisol after stimulation (Motzer & Hertig, 2004). The cortisol awakening response (CAR) is characterized by a 40–75% increase of salivary cortisol levels, with maximum values occurring about 30 min after awakening (de Weerth et al., 2003; Fries et al., 2009; Kudielka et al., 2006). The CAR can be characterized via collection of saliva samples within the first hour of awakening and computation of the area under the curve (AUC) for the salivary cortisol profile (Fekedulegn et al., 2007; Pruessner et al., 1997; Wust et al., 2000). The CAR is a discrete and dynamic component of daily cortisol secretion that is heritable and remains stable within individuals over at least several days under normal circumstances (Clow et al., 2004; Pruessner et al., 1997; Wust et al., 2000). A recent meta-analysis of 62 studies concluded that job stress and overall life stress were positively associated with waking cortisol AUC measures (Chida & Steptoe, 2009). In contrast, decreases in waking cortisol measures have been observed in populations where chronic stress might be expected, including depressed individuals and among police officers with symptoms of posttraumatic stress disorder (PTSD) (Huber et al., 2006; Neylan et al., 2005). Changes in cortisol secretion in response to various stressors have been linked with perturbations in physiological processes that facilitate inflammation, and with other risk factors for CVD, type II diabetes, stroke, and cancer (Gidron & Ronson, 2008; Goh et al., 2000; Scheer et al., 2008; Zuzewicz et al., 2000). This indicates that salivary cortisol biomonitoring may be useful for characterizing the pathophysiological effects of stress and shiftwork, as suggested by a recent study (Oginska et al., 2010).
The Buffalo Cardio-Metabolic Occupational Police Stress (BCOPS) cohort is a prospective study that is examining biological processes by which the stressors associated with police work may mediate adverse health outcomes. The BCOPS protocol has combined the characterization of stress biomarkers, including the CAR, with subclinical CVD measures and psychosocial factors to examine their potential associations with irregular work hours, psychological disturbances, and chronic diseases afflicting police officers (Charles et al., 2007; Violanti et al., 2006, 2007, 2008, 2009a, 2009b). The objective of the current analysis was to evaluate the effect of long- and short-term shiftwork on the CAR and depressive symptoms among officers participating in the BCOPS pilot study, and to estimate the extent of shiftwork required to exert impacts on these endpoints. Specifically, this study tested the hypotheses that officers working predominantly night or afternoon shifts had altered salivary waking cortisol AUC measures and increased depressive symptoms compared to those working days. It is still uncertain whether night work per se, more frequent shift changes, or extended work hours may be more detrimental to long-term worker health (Crowley et al., 2004; Quera-Salva et al., 1997; Reinberg & Ashkenazi, 2008; Reinberg et al., 2007). For this reason, we used several different summary measures, including the cumulative number of work hours, or cumulative shift changes experienced by each officer, as well as the cumulative amount of time on afternoon and/or night shifts, summarized over two different time frames, to examine these possibilities. The short-term shiftwork period summarized the work schedule between 3 and 14 days prior to participation and sample collection, and the long-term shiftwork period summarized each subject's cumulative irregular work hours from 1994 until their participation in the study in 2001.
Methods
Study Population
The study population was comprised of male and female officers from the Buffalo New York Police Department enrolled in the BCOPS pilot study. A computer-generated random sample stratified on sex was obtained from all police officers in the department to recruit an initial sample of 100 participants (42 females, 58 males) (Charles et al., 2007; Violanti et al., 2006). The participation rate was 100%. The study received Institutional Review Board approval, and all subjects provided informed consent (Portaluppi et al., 2010). Data collection included long- and short-term shiftwork histories, serial saliva sample collection upon awakening on a single day, and completion of several validated instruments, including the CES-D (Ratloff, 1977), the Impact of Events Scale (IES), which measures stress related to traumatic events and symptoms of PTSD (Horowitz et al., 1979), the Life Events Scale (Paykel et al., 1971), and basic demographic characteristics (age, sex, education, race, marital status, rank, years of service, and baseline alcohol and tobacco consumption).
Shiftwork History
Daily work histories were obtained for each participant from 1994 or initiation of employment to the date of study participation in 2001 using payroll records. Records were accessible through an electronic archive beginning in 1994; administrative changes resulting in a switch from rotating to fixed work schedules occurred at that time. The typical work schedule after 1994 consisted of 4 work days, 3 days off, 3 work days, 3 days off, and then the cycle was repeated. Each shift was examined to summarize work activities and quantify the amount of time officers spent on each shift. If an individual had to appear in court or work overtime and had a work shift on the same day, then the court or overtime hours were added to the shift hours. If a subject had to appear in court or worked overtime hours and did not have a work shift on the same day, the court or overtime hours were counted as a separate workshift according to the time of day they were logged. Long-term shiftwork variables included the cumulative number of shift changes,and the cumulative hours on day, afternoon, or night shifts. In addition, a categorical shift status variable (day, afternoon, or night) was assigned to each participant based on long-term work history, which was defined as the shift on which each participant spent >50% of her or his time working during the study period. Shifts were classified as day, afternoon, or night based on start times between 04:00 and 11:59 h, 12:00 and 19:59 h, and 20:00 and 03:59 h, respectively. A “combined” category was created that included both afternoon and night shiftworkers. A total of 55 (85%) subjects spent ≥70% of their total work time on one specific shift, 9 (14%) spent 50–69% of their total work time on a single shift, and 1 (2%) spent 42% of their total work time on the night shift (classified as a night worker). Using this approach, good consistency with a previous study summarizing shift status over 30, 60, and 90 days, and 5 yrs was achieved. (Violanti et al., 2008). Although officers worked fixed shifts during the study period, they occasionally worked for an absent colleague and may have been assigned to a different shift schedule. The frequency of shift changes was defined as the number of times a participant switched between any two of the shift types (day, afternoon, or night) during the study period. Total work hours also were summed for each participant (regardless of shift) as an indicator of long-term workload. For short-term shiftwork, the cumulative number of hours on day, afternoon, night, or combined shifts was reconstructed over periods of 3, 5, 7, and 14 days prior to the date of saliva sample collection, and participants were assigned to a shift status category for each short-term period using the 50% cut-point as described above. Over each of these periods, participating officers spent ≥55% of their time working in the shift category to which they were assigned (i.e., day, afternoon, or night). The total number of hours worked over these periods was used to estimate total workload. Fourteen subjects who did not work during the 3-day period prior to saliva collection were not included in the 3-day shiftwork analyses. The same is true for the 5-, 7-, and 14-day analyses where 8, 7, and 6 subjects did not work, respectively. Six subjects who did not work over the 14-day period prior to saliva collection also did not work over the 3-, 5-, or 7-day periods prior to saliva collection.
Saliva Collection and Questionnaires
Although cortisol can also be measured in urine and blood, saliva has come to be preferred because collection is simple (thus facilitating participant compliance), and because salivary cortisol is unbound (thus providing a biologically active hormone concentration), and it can be readily quantified via immunoassay (Violanti et al., 2009a). Participants collected four serial saliva samples for characterization of the CAR, the first upon awakening, and then at 15-, 30-, and 45-min intervals after waking. Cortisol levels based on the time of awakening have been found to have a higher test-retest stability (Neylanet al., 2005; Pruessner et al., 1997) compared to cortisol levels at specific clock times (Coste et al., 1994). Participants were studied during a clinic examination on a scheduled training or off day. A majority of the participating officers (88%) were either working a day shift or had a day off prior to their clinic visit. On the morning after the clinic visit, sample collection was achieved by placing Salivettes (a dental roll provided in a centrifuge tube; Sarstedt Aktiengesellschaft & Company, Numbrecht, Germany) into the mouth for 3 min to allow for saturation. Subjects were instructed to refrigerate the saliva samples until they could be delivered by the participant to the research laboratory for processing. The average time of the first saliva sample collection was 07:29 h ± 104 min, 07:55 h ± 104 min, and 08:13 h ± 123 min for day, afternoon, and night shiftworkers, respectively. Samples were shipped to the National Institute for Occupational Safety and Health (Toxicology and Molecular Biology Branch, Health Effects Laboratory, Morgantown, WV) where they were centrifuged and archived at −20°C (Fekedulegn et al., 2007; Violanti et al., 2007). Cortisol determinations were performed at the Technical University of Dresden, Dresden, Germany, using a sensitive and specific chemiluminescence immunoassay (CLIA; IBL-Hamburg, Germany). Each batch of processed samples included quality control (QC) samples at low and high concentrations on each assay plate, which allowed for assessment of assay drift. If QC samples were outside the range of expected concentrations, the assay was repeated. The intra- and interassay coefficients of variation were ≤8.0% for either the high (25 nmol/L) or low (3 nmol/ L) cortisol control concentrations. Blind replicate samples of participants' saliva (10%) were also included for QC purposes, and these replicate samples had a coefficient of variation of 15.3%.
The waking cortisol rhythm was summarized using two measures, the cortisol AUC with respect to increased secretion above baseline (cortisol AUCI), and the cortisol AUC above ground (i.e., above zero, cortisol AUCG). These measures independently characterize different aspects of the CAR (Fekedulegn et al., 2007; Jolliffe, 2010). The cortisol AUCI measures the amount of cortisol secreted above the waking baseline; it represents changes of cortisol secretion over time and is related to its reactivity (Fekedulegn et al., 2007). The cortisol AUCG measures the total amount of cortisol secreted during the sampling period and is representative of total hormonal output (Fekedulegn et al., 2007). These measures were applied in this study because the AUC captures the cortisol secretion pattern at the individual level (rather than grouping participant data at each time point as with repeated-measures analysis of variance [ANOVA]). This was considered more physiologically meaningful in this context since it provides for a more parsimonious statistical approach while still integrating the temporal aspects of salivary cortisol secretion over the 45-min waking time frame. This approach has been previously applied in this cohort and in other populations (Chida & Steptoe, 2009; Violanti et al., 2008).
Questionnaires to ascertain depressive symptoms, life events, and their impact, and demographic characteristics were completed by each officer at the same time of day during their clinic visit. The CES-D is a self-administered instrument originally designed to measure depressive symptoms in the general population. It is comprised of 20 questions that ascertain depressive mood, feelings of guilt and worthlessness, psychomotor retardation, loss of appetite, and sleep disturbance (Ratloff, 1977). CES-D scores are highly correlated with other depression scales (Fava, 1983; Roberts & Vernon, 1983). For example, the CES-D was well correlated with the Hamilton Rating Scale for Depression among depressed and nondepressed individuals (correlation coefficients [r]=0.61 and 0.65, respectively). The CES-D has also exhibited good internal validity (Cronbach's α = 0.91) and test-retest reliability (intraclass correlation coefficient = 0.87) (Miller et al., 2008). Although not considered diagnostic, a total score of ≥16 is the suggested cut-point to identify depressed individuals (Radloff, 1977). The IES measures current subjective distress related to a specific event. It contains a series of 15 questions that ascertain intrusive and avoidance symptoms, both of which have excellent reliability (Cronbach αs of 0.79 and 0.82, respectively) (Horowitz et al., 1979).
Statistical Analysis
Analyses were performed using the SAS computer software package (version 9.2, Cary, NC). Relationships between each dependent variable (AUCI, AUCG, and CES-D scores) and potential confounding factors were evaluated univariately using the generalized linear models (GLM) procedure in SAS. Variables were selected for further evaluation if their statistical significance was p ≤ .15. Variables with >10% missing data were excluded from the analysis. CES-D scores were log-transformed for statistical analysis and means were back-transformed for presentation in tables. Analyses of cortisol AUCI and AUCG were performed without transformation, since these data were normally distributed. Spearman correlation coefficients (r) were calculated among dependent variables, and among long- and short-term cumulative shiftwork variables in order to evaluate the extent to which night and/or afternoon workers maintained their work schedule over an extended period, which could have implications for future analyses of chronic disease in this cohort. To evaluate possible linear trends in the relationship between cortisol and shiftwork or total workload measures, regression analyses were performed using the continuous form of each shiftwork variable (Proc GLM in SAS). The GLM procedure was used to compute the least squares (LS) means of cortisol AUCI and AUCG and CES-D among quartiles of each long-term shiftwork variable, after adjustment for selected potential confounding factors (AUCI, AUCG, and CES-D were adjusted for sex, rank, and IES score, respectively).
The difference between the LS means in the upper and lower quartiles was then evaluated using the least significant differences statistic. Similar analyses were performed using the categorical shiftwork variables with day workers as referents. Relationships between cortisol (AUCI or AUCG) and depressive symptoms (CES-D) were evaluated using similar methods. To examine whether a heavy workload or many shift changes combined with the effects of shiftwork (i.e., cumulative hours on afternoon, night, or combined shifts) to elicit changes in the CAR, interaction terms between shiftwork and total work hours or total shift changes were included in separate statistical models. Ancillary analyses were performed using logistic regression (Proc Logistic in SAS) to determine whether those excluded from the analysis differed from those with complete data. Participants excluded due to missing cortisol or shiftwork data did not differ in age, sex, race, education, marital status, rank, years of police work, IES scores, alcohol or tobacco consumption at baseline, or shift status compared to those included in the study, and were thus considered missing at random. Adjustment for time of awakening (first saliva collection) did not alter the interpretation of the results presented below.
Results
Complete data were available for 65 participants with long-term shiftwork data (n = 32 missing one or more of four waking cortisol samples, n = 3 missing long-term shiftwork data) and 58 subjects with short-term shiftwork data (n = 32 missing cortisol, n=10 missing short-term shiftwork data). Characteristics of officers grouped according to their long-term shift status (days, afternoons, or nights) are presented in Table 1. The average duration of police work (± standard deviation [SD]) in this population was 14 ± 9 yrs with a range of 1 to 33 yrs. The mean age was 43 ± 8 yrs with a range of 29 to 63 yrs. Males comprised 65% of the study participants. European Americans (EAs) comprised 71% of the study population, and 29% were African American (AA). Those working mainly afternoon or night shifts tended to be younger, male, EA, and had lower IES scores and fewer years of service compared to those working day shifts. A greater proportion of participants working night shifts held the rank of police officer (100%) compared to those on day shifts (47%). On average, participants classified as afternoon or night workers spent >85% of their time working irregular (night or afternoon) shifts (Table 1). Long-term cumulative night work was modestly correlated with night work summarized over the preceding 5 or 14 days (r = 0.39, p ≤ .01 for both) (Table 2) and long-term afternoon work was correlated with afternoon work over the prior 5 or 14 days (r = 0.63 to 0.52, both p ≤ .01) (Table 2). Shift changes were correlated with long-term work on afternoon shifts (r = 0.64, p ≤ .01) but not with long-term night work, total work hours, or recent work on afternoon or night shifts (Table 2).
Table 1. Population characteristics by long-term shift status.
| Characteristic | Day Shifts (n = 34) | Afternoon Shifts (n = 23) | Night Shifts (n = 8) | Afternoon + Night Shifts (n = 31) |
|---|---|---|---|---|
| Age group (yrs) | ||||
| <40 (n = 25) | 6 (18%) | 16 (70%) | 3 (38%) | 19 (61%) |
| 40–49 (n = 25) | 16 (47%) | 5 (22%) | 4 (50%) | 9 (29%) |
| >50 (n = 15) | 12 (35%) | 2 (9%) | 1 (13%) | 3 (10%) |
| Sex | ||||
| Male (n = 42) | 18 (53%) | 17 (74%) | 7 (88%) | 24 (77%) |
| Female (n = 23) | 16 (47%) | 6 (26%) | 1 (13%) | 7 (23%) |
| Race | ||||
| European American (n = 46) | 19 (56%) | 21 (91%) | 6 (75%) | 27 (87%) |
| African American (n = 19) | 15 (44%) | 2 (9%) | 2 (25%) | 4 (13%) |
| Education | ||||
| ≤High School (n = 10) | 5 (15%) | 3 (13%) | 2 (25%) | 5 (16%) |
| College (n = 19) | 9 (26%) | 6 (26%) | 4 (50%) | 10 (32%) |
| >College (n = 36) | 20 (59%) | 14 (61%) | 2 (25%) | 16 (52%) |
| Marital status | ||||
| Single (n = 12) | 6 (18%) | 3 (13%) | 3 (38%) | 6 (19%) |
| Married (n = 43) | 21 (62%) | 17 (74%) | 5 (63%) | 22 (71%) |
| Divorced (n = 10) | 7 (21%) | 3 (13%) | 0 (0%) | 3 (10%) |
| Rank | ||||
| Police officer (n = 40) | 16 (47%) | 16 (70%) | 8 (100%) | 24 (77%) |
| Sergeant/Lieutenant (n = 10) | 8 (24%) | 2 (9%) | 0 (0%) | 2 (6%) |
| Captain/Detective (n = 15) | 10 (29%) | 5 (22%) | 0 (0%) | 5 (16%) |
| Years worked | ||||
| 1–5 (n = 12) | 2 (6%) | 6 (26%) | 4 (50%) | 10 (32%) |
| 6–10 (n = 7) | 2 (6%) | 4 (17%) | 1 (13%) | 5 (16%) |
| 11–15 (n = 18) | 9 (26%) | 7 (30%) | 2 (25%) | 9 (29%) |
| >15 (n = 28) | 21 (62%) | 6 (26%) | 1 (13%) | 7 (23%) |
| Shift changes/year (mean ± SD) | 5.0 ± 8.4 | 7.4 ± 6.7 | 2.9 ± 3.0 | 6.3 ± 6.2 |
| Mean % time on shift (mean ± SD) | 90.1 ± 13.5 | 80.0 ± 17.5 | 77.7 ± 17.6 | 85.1 ± 13.1 |
| Impact of Events Scale (mean ± SD) | 29 ± 15 | 18 ± 15 | 13 ± 12 | 17 ± 15 |
| Life Events Scale (mean ± SD) | 3 ± 3 | 2 ± 2 | 3 ± 2 | 2 ± 2 |
Shift assigned represents >50% of work hours during study period. SD = standard deviation. Column percentages not totaling 100% are due to rounding.
Table 2. Correlations between long-term and short-term shiftwork.
| Long-term | 5-Day | 14-Day | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|||||||||
| Daytime | Afternoon shifts | Night shifts | Total hours | Shift changes | Daytime | Afternoon shifts | Night shifts | Daytime | Afternoon shifts | ||
| Long-term | Afternoon | −0.60 | — | — | — | — | — | — | — | — | — |
| shifts | (<0.01) | ||||||||||
| Night shifts | −0.47 | 0.10 | — | — | — | — | — | — | — | — | |
| (<0.01) | (0.44) | ||||||||||
| Total hours | 0.13 | 0.17 | −0.01 | — | — | — | — | — | — | — | |
| (0.32) | (0.18) | (0.96) | |||||||||
| Shift changes | −0.17 | 0.64 | 0.05 | 0.10 | — | — | — | — | — | — | |
| (0.17) | (<.01) | (0.70) | (.042) | ||||||||
| 5-Day | Daytime | 0.34 | −0.26 | <−0.01 | 0.09 | −0.17 | — | — | — | — | — |
| (<.01) | (0.03) | (0.99) | (0.50) | (0.17) | |||||||
| Afternoon | −0.49 | 0.63 | 0.12 | 0.11 | 0.26 | −0.25 | — | — | — | — | |
| shifts | (<0.01) | (<0.01) | (0.34) | (0.38) | (0.03) | (0.04) | |||||
| Night shifts | −0.13 | −0.22 | 0.39 | <0.01 | −0.07 | −0.23 | −0.04 | — | — | — | |
| (0.31) | (0.08) | (<0.01) | (0.98) | (0.55) | (0.06) | (0.73) | |||||
| 14-Day | Daytime | 0.29 | −0.17 | 0.03 | 0.11 | −0.18 | 0.90 | −0.12 | −0.26 | — | — |
| (0.02) | (0.17) | (0.84) | (0.37) | (0.16) | (<0.01) | (0.34) | (0.03) | ||||
| Afternoon | −0.55 | 0.52 | 0.19 | 0.10 | 0.17 | −0.22 | 0.88 | 0.05 | −0.14 | — | |
| shifts | (<0.01) | (<0.01) | (0.13) | (0.44) | (0.18) | (0.08) | (<0.01) | (0.71) | (0.27) | ||
| Night shifts | −0.12 | −0.24 | 0.39 | −0.07 | −0.05 | −0.20 | −0.04 | 0.85 | −0.27 | 0.07 | |
| (0.36) | (0.06) | (<0.01) | (0.57) | (0.73) | (0.10) | (0.73) | (<0.01) | (0.03) | (0.57) | ||
Spearman correlation coefficients (p values) for relationships among cumulative long-term and short-term (5- and 14-day) shiftwork measures.
The salivary cortisol values for participating officers were generally representative of those found in other investigations, with higher levels in the morning and declining values over the day (Griefahn & Robens, 2008; Kudielka et al., 2007; Stone et al., 2001). Table 3 provides a summary of crude mean cortisol AUCI and AUCG, and CES-D values by population characteristic. Mean cortisol AUCI values were elevated among females (229 vs. −6 among males, p ≤ .01), and there was a tendency for AUCI values to be higher among those 40 to 49 yrs of age, although differences among age groups were not statistically significant. Cortisol AUCG values tended to be elevated among those with a higher rank (captain/ detective: 955 vs. police officer: 679, p = .06). CES-D scores were not correlated with the cortisol AUCI or AUCG (both, r < 0.10, p > .45). Higher CES-D scores were observed among those with more education (>college: 5.2 vs. ≤high school: 2.9, p = .12), and elevated IES scores (>19) (8.1 vs. 3.2 among those with IES values ≤19, p<.01) (Table 3).
Table 3. Summary statistics for measures of waking cortisol and depressive symptoms.
| Characteristics | AUCI | p value | AUCG | p value | CES-D | p value |
|---|---|---|---|---|---|---|
| Age group (yrs) | ||||||
| <40 | 43 (280) | 0.791 | 807 (621) | 0.721 | 5.5 (2.4) | 0.821 |
| 40–49 | 160 (269) | 0.132 | 635 (356) | 0.202 | 4.3 (2.9) | 0.372 |
| >49 | 20 (277) | 0.113 | 751 (420) | 0.453 | 5.2 (3.1) | 0.583 |
| Sex | ||||||
| Male | −6 (266) | <0.01 | 749 (687) | 0.61 | 4.7 (2.5) | 0.80 |
| Female | 229 (237) | 687 (359) | 5.1 (3.3) | |||
| Race | ||||||
| European American | 68 (279) | 0.45 | 689 (427) | 0.32 | 5.0 (2.8) | 0.76 |
| African American | 125 (280) | 820 (606) | 4.6 (2.6) | |||
| Education4 | ||||||
| ≤High School | 127 (178) | 0.33 | 645 (471) | 0.98 | 2.9 (4.3) | 0.12 |
| College | 160 (264) | 899 (582) | 5.7 (2.6) | |||
| >College | 30 (301) | 649 (405) | 5.2 (2.4) | |||
| Marital status5 | ||||||
| Single | 80 (304) | 0.92 | 691 (487) | 0.82 | 5.7 (3.4) | 0.95 |
| Married | 84 (266) | 727 (497) | 4.8 (2.5) | |||
| Divorced | 93 (329) | 766 (453) | 4.7 (3.7) | |||
| Rank6 | ||||||
| Police officer | 95 (268) | 0.83 | 679 (491) | 0.06 | 5.6 (2.8) | 0.22 |
| Sergeant/Lieutenant | 48 (173) | 583 (160) | 4.2 (2.3) | |||
| Captain/Detective | 77 (366) | 955 (545) | 3.8 (2.9) | |||
| Years worked7 | ||||||
| 1–5 | 44 (192) | 0.81 | 525 (312) | 0.24 | 6.4 (2.8) | 0.18 |
| 6–10 | 69 (219) | 807 (420) | 4.5 (2.4) | |||
| 11–15 | 222 (342) | 875 (605) | 5.6 (2.7) | |||
| >15 | 22 (264) | 710 (465) | 4.1 (2.9) | |||
| Impact of Events Scale | ||||||
| ≤19 | 56 (283) | 0.38 | 723 (456) | 0.96 | 3.2 (2.6) | <0.01 |
| >19 | 116 (274) | 729 (519) | 8.1 (2.3) | |||
| Life Events Scale | ||||||
| ≤2 | 82 (297) | 0.94 | 729 (482) | 0.95 | 4.3 (2.8) | 0.22 |
| >2 | 87 (261) | 722 (491) | 5.8 (2.7) |
Crude means (SD in parentheses, GSD for CES-D) are presented for AUCI = area under the curve with respect to increase; AUCG = area under the curve with respect to ground; units for AUCI and AUCG are nmol/L·min; CES-D = Center for Epidemiologic Studies Depression scale.
p value is for difference between <40 and >49 yrs;
p value for difference between <40 and 40–49 yrs;
p value for difference between 40–49 and >49 yrs;
p value is for the difference between ≤high school and >college;
p value is for the difference between married and divorced;
p value is for the difference between police officer and captain/detective;
p value is for the difference between 1–5 yr and >15 yr groups.
No statistically significant differences in adjusted-mean cortisol AUCI or AUCG were observed between officers classified as long-term afternoon, night, or combined shiftworkers compared to those working long-term day shifts (data not shown). Similarly, there were no statistically significant differences in adjusted-mean cortisol AUCI or AUCG between the lowest and highest quartiles of total work hours, or among quartiles of cumulative time on afternoon, night, or combined shifts (data not shown). There were no associations between continuous long-term shiftwork variables and the waking cortisol measures, nor were interaction terms between these variables and shift changes or with total work hours statistically significant (data not shown). Adjusted-mean cortisol AUCI and AUCG values were reduced among participants in the highest quartile of cumulative shift changes compared to those in the lowest quartile (AUCI: −10 vs. 156, p = .07, and AUCG: 568 vs. 826, p = .13, for upper vs. lower quartile, respectively). A statistically significant difference in the mean cortisol AUCI was observed when these analyses were adjusted for time of awakening (AUCI: −22 vs. 155, p = .05, and AUCG: 786 vs. 517, p = .12, for upper vs. lower quartile, respectively).
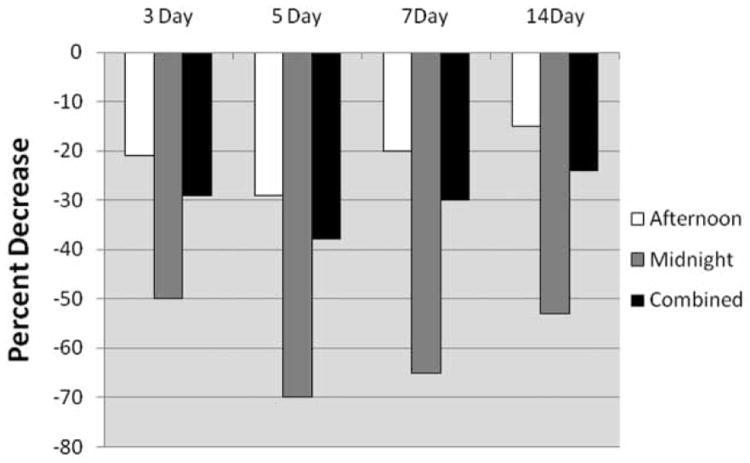
When short-term shiftwork was evaluated, there was a statistically significant reduction in adjusted-mean AUCG cortisol levels among officers working nights compared to those working day shifts over the preceding 3-, 5-, 7-, or 14-day periods (Table 4). Cortisol AUCI was reduced in a manner similar as AUCG, predominantly among those working nights or combined shifts, although none of those comparisons were statistically significant (Table 4). Figure 1 shows the percent difference in mean cortisol AUCG between officers on irregular shifts compared to those working days. In all instances, mean cortisol AUCG levels were greatest among day workers and lower among afternoon or night-shift workers, with a peak effect occurring when data were summarized over the 5 days immediately preceding study participation. In addition, we used repeated-measures analysis of variance with a spatial power covariance structure (to account for minor differences in data collection time points among subjects) to model cortisol concentrations as a function of collection time, shift, and the interaction between time and shift (5-day). The results from that analysis were consistent with the results described above; cortisol levels among night or afternoon workers were lower compared to those working days, and the time × shift interaction term was not statistically significant. There were no statistically significant differences in cortisol AUCG or AUCI between the upper and lower quartiles of total hours worked over 3, 5, 7, or 14 days, and the continuous forms of those variables were also not statistically significant. Furthermore, the interaction terms between irregular shift hours and total work hours summarized over the respective short-term work periods (3, 5, 7, or 14 days) did not suggest a combined effect among these variables (data not shown).
Table 4. Relationship between short-term police shiftwork and mean waking cortisol or CES-D summary measures.
| Dependant | Day shifts | Afternoon shifts | Night shifts | Afternoon + night shifts |
|---|---|---|---|---|
| 3 Days | ||||
| (n = 21) | (n = 16) | (n = 7) | (n = 23) | |
| AUCG | 841 — | 665 (0.28) | 424 (0.05) | 592 (0.09) |
| AUCI | 111 — | 107 (0.96) | 67 (0.69) | 96 (0.83) |
| CES-D | 4.9 — | 4.9 (0.97) | 6.4 (0.50) | 5.3 (0.75) |
| 5 Days | ||||
| (n = 22) | (n = 21) | (n = 7) | (n = 28) | |
| AUCG | 843 — | 598 (0.09) | 256 (<0.01) | 521 (0.02) |
| AUCI | 124 — | 105 (0.80) | 50 (0.50) | 92 (0.65) |
| CES-D | 4.4 — | 4.8 (0.78) | 7.0 (0.26) | 5.3 (0.51) |
| 7 Days | ||||
| (n = 24) | (n = 20) | (n = 7) | (n = 27) | |
| AUCG | 785 — | 625 (0.28) | 271 (0.02) | 550 (0.08) |
| AUCI | 110 — | 103 (0.92) | 48 (0.57) | 90 (0.77) |
| CES-D | 4.6 — | 4.8 (0.87) | 7.0 (0.29) | 5.3 (0.58) |
| 14 Days | ||||
| (n = 25) | (n = 19) | (n = 8) | (n = 27) | |
| AUCG | 774 — | 661 (0.46) | 366 (0.04) | 585 (0.16) |
| AUCI | 96 — | 99 (0.99) | 66 (0.75) | 87 (0.89) |
| CES-D | 4.2 — | 5.1 (0.53) | 7.4 (0.14) | 5.7 (0.26) |
Note. Data for AUCG (area under the curve with respect to ground); AUCI (area under the curve with respect to increase); and CES-D (Center for Epidemiologic Studies Depression) are presented as least squares means adjusted for rank, sex, and Impact of Events Scale, respectively. (p value vs. day shift in parentheses). Shift assigned represents >50% of work hours during that period. Units for AUCI and AUCG are nmol/L·min.
Figure 1.
Percent decrease in cortisol awakening response (AUCG) by shift status over 3, 5, 7, and 14 days. Percent decrease refers to the difference in least squares mean AUCG values (adjusted for rank) among day workers compared to those working afternoon (white), night (gray), or combined (afternoon + night) shifts (black) for work periods summarized over 3, 5, 7, or 14 days.
There were no statistically significant differences in adjusted-mean CES-D scores by shift status, between the upper and lower quartiles, or for the continuous forms of long-term afternoon, night, or combined shiftwork hours, total work hours, or shift changes (data not shown). There also were no statistically significant differences in adjusted-mean CES-D scores between those working primarily night shifts compared to those working days over the 3-, 5-, 7-, or 14-day periods, although there was a tendency for CES-D scores among night shiftworkers to be greater than day shiftworkers over 14 days (7.4 vs. 4.2 respectively, p = .14) (Table 4).
Discussion
Diseases observed among police officers are similar to those seen in other occupations that include shiftworkers. However, the extent to which shiftwork or other types of occupational stressors contribute to the development of disease in this understudied, high-risk population remains uncertain. This study quantified shiftwork, total work hours, and shift changes over a period encompassing 972 person-years of police work in order to examine potential effects on the CAR and depressive symptoms. We observed shift-related reductions in the CAR compared to day workers. The largest differences occurred with cortisol AUCG, but the patterns of response were similar for cortisol AUCI. Cortisol secretion typically increases with the acute onset of different types of stressors (Kudielka et al., 2009) and with repeated challenges cortisol can remain elevated for extended periods with consequent flattening of the diurnal slope (McEwen, 2006). Thus, one might have expected CAR measures in this study to be elevated among shiftworking police officers. For example, in a study that compared different types of shift rotation, workers on a schedule that included three different rotating shifts over 7 days, with 5 subsequent days off, were compared with those on a schedule that included 3 working days on a given shift with 2 subsequent days off. Those on the 7-day rotating shifts had elevated diurnal cortisol AUCs (measured across the entire day) and a flattened diurnal cortisol slope compared to those with more frequent shift rotations (Lac & Chamoux, 2004). Other studies have observed reductions in waking as well as diurnal cortisol levels in response to shiftwork. For example, average waking cortisol concentrations were reduced by 44% after 2 wks of simulated night work (Griefahn & Robens, 2008). A reduction in mean waking salivary cortisol concentrations was reported among night workers after transitioning from daytime work in an electronics manufacturing facility (Kudielka et al., 2007). Among permanent nightworkers, waking salivary cortisol profiles were reduced both on night shifts and on days off (Kudielka et al., 2007).
In the present study, a maximal reduction in the CAR was observed when irregular work hours were averaged over 5 days, indicating that nearly a full week of afternoon or night shifts was required before maximal dysregulation of the CAR occurred. Typically, the officers repeatedly worked 4 days on, 3 days off, then 3 days on and 3 days off on a fixed shift. Thus, it is unlikely that officers routinely worked 5 continuous days during the week, but did so when they needed to cover a shift for a fellow employee. Other studies have observed changes in cortisol secretion patterns within a similar shiftwork time frame. For example, a reversal in salivary cortisol levels at 06:00 and 21:00 h was observed among nurses after 5 to 7 consecutive night shifts compared to those working days. After 7 days of night work, mean cortisol levels at 06:00 h decreased from about 16 nmol/L down to 4 nmol/L, whereas the 21:00 h cortisol values only increased from 3 to 8 nmol/L (Hennig et al., 1998). In another study, mean serum cortisol concentrations among night workers on a 3–4-day rotating schedule were elevated at the expected trough of circadian cortisol secretion (00:00 and 02:00 h) compared to controls (Touitou et al., 1990). There was a progressive increase in cortisol levels among controls from 02:00 to 08:00 h, but this pattern was not observed among the night workers, resulting in reduced cortisol amplitudes (Touitou et al., 1990). Changes in the rhythm or concentration of cortisol secretion have been associated with shiftwork in other studies (Goh et al., 2000; Leese et al., 1996; Munakata et al., 2001; Scheer et al., 2008; Zuzewicz et al., 2000), although not all results have been consistent (Axelsson et al., 2003; Boquet et al., 2004; Roden et al., 1993). For example, reductions in the waking cortisol AUC were recently reported among police officers with PTSD symptoms but not among night workers, although the type or duration of night work was unspecified (Neylan et al., 2005).
A strength of the current study is that shiftwork was objectively characterized by accessing payroll records and quantifying each participant's work history. The effects of irregular shifts varied by duration of shiftwork, and were independent of other potential confounding or modifying factors, including extended work hours and PTSD (IES) symptoms. Discrepancies between this and other studies may be due to certain uncharacterized factors, including differences in ambient light exposures among participants. Light exposure, particularly during the late dark phase or predawn hours, can activate cortisol production or alter the normal cortisol nadir (Clow et al., 2004; Sephton & Spiegel, 2003). Direct comparisons between our results and those reported by others also are difficult due to potential differences in the type of work performed, length, direction, or type of shift schedule that was used, time-of-day of sample collection or type of cortisol measure that was employed, or because of other differences, such as potential confounding factors that may not have been consistently characterized and evaluated among the published studies (Clow et al., 2004). One limitation of the present analysis is that some covariate information was not available (e.g., current tobacco, coffee, or alcohol consumption, and light exposure following awakening). In addition, saliva collection in the present study occurred on a training or off day, so officers working nights or afternoons had time to revert back to a typical diurnal schedule, which may have resulted in an attenuation of the effects we observed. In any case, our results were generally consistent with other studies indicating that waking cortisol levels or other aspects of cortisol secretion are disrupted in response to shiftwork (Goh et al., 2000; Griefahn & Robens, 2008; Kudielka et al., 2007; Leese et al., 1996; Munakata et al., 2001; Scheer et al., 2008; Touitou et al., 1990; Zuzewicz et al., 2000).
The shift change metric was the only long-term measure associated with the CAR, and the cortisol AUCI was essentially ablated among officers in that group. Individuals in the highest quartile of shift changes had an average with SD of 105 ± 73 shift changes and an average work duration of 19 ± 9 yrs compared with an average of 5 ± 3 shift changes and 14 ± 9 yrs work duration among those in the lowest quartile. It is interesting to note that recent shiftwork over several days and frequent shift changes over periods of years both contributed to a reduction in the CAR. This diminution of cortisol secretion is more suggestive of the exhaustion stage of the stress response, when maladaptation and breakdown of the HPA's compensatory mechanisms are thought to occur due to frequent or excessive exposure to stressors (Motzer & Hertig, 2004). Reductions in the CAR have been linked with a variety of circumstances that may relate to adverse health, including advanced age (Kudielka & Kirschbaum, 2003), poor sleep quality among insomnia patients (Backhaus et al., 2004), chronic fatigue syndrome and symptoms of burnout (Pruessner et al., 1999; Roberts et al., 2004), PTSD (Rohleder et al., 2004), early loss experiences due to death or separation (Meinlschmidt & Heim, 2005), and depression (Stetler & Miller, 2005), as well as measures of adverse cardiovascular health (Hurwitz Eller et al., 2001) or reduced bone density in women (Brooke-Wavell et al., 2002; Clow et al., 2004). Continued research is needed to develop a better understanding of temporal relationships between stressor exposures, chronic dysregulation of the HPA axis and cortisol secretion, and associations with chronic disease risks among police officers or other shiftworking occupations, including evaluations of the intermediate time periods not included in this analysis.
Our study did not find that depressive symptoms were associated with cortisol AUCI or AUCG, which is not consistent with results from some previous studies. A recent meta-analysis found that depressed individuals had lower baseline cortisol levels than those without depression (Burke et al., 2005). Using the CES-D scale, elevated scores for depressive symptoms have been associated with shiftwork and increased sleepiness among rotating shiftworkers compared to those on permanent day shifts (Takahashi et al., 2006). A greater prevalence of depressive symptoms also was observed among pediatric residents working night float shifts (43%) when compared to working daytime rotations (14%) (Cavallo et al., 2002). In the present study, only about 6% of the population met the suggested definition of depression (CES-D scores ≥16), which is similar to what is expected in the general population (∼5%) (Weissman et al., 1996). It is possible that depressive symptoms were not frequent or severe enough in this population to detect shiftwork-related changes in cortisol AUCG or AUCI, or that coping measures or other psychological adaptation processes were effectively in place among the participants.
In summary, the BCOPS study is the first-ever population-based prospective cohort to characterize biomarkers of stress, subclinical CVD, body composition, and psychological symptoms in this high-risk occupation (Violanti et al., 2006). Measurement of the salivary CAR provides a valid and efficient procedure for biomonitoring of the HPA axis. The CAR exhibits a relatively a high degree of heritability, good intraindividual stability over several days (Bartels et al., 2003; Pruessner et al., 1997; Wust et al., 2000), and altered values have been consistently associated with psychometric measures of stress (Chida & Steptoe, 2009; Maina et al., 2009). Results from this investigation build upon our previous analyses showing a dysregulation of salivary cortisol secretion among officers with PTSD symptoms (Violanti et al., 2007). Disrupted sleep (Charles et al., 2007), increased suicide ideation (Violanti et al., 2008), and an elevated prevalence of metabolic syndrome (Violanti et al., 2009b) have also been observed among officers in the BCOPS cohort working irregular shifts. Our current results indicate that short-term night work and possibly long-term shift changes may lead to dysregulation of the CAR among police officers, which may help inform future analyses within the entire BCOPS cohort and in other populations that seek to determine whether changes in the CAR may be associated with the development of chronic disease.
Acknowledgments
This project was supported by Contract No. 212-2008-M-24052 from the Biostatistics and Epidemiology Branch of the National Institute for Occupational Safety and Health, Centers for Disease Control and Prevention, Morgan-town, West Virginia. Dr. Hébert is supported by an Established Investigator Award in Cancer Prevention and Control from the Cancer Training Branch of the National Cancer Institute (K05 CA136975). Michael Wirth's participation in this research was supported by the University of South Carolina's Behavioral-Biomedical Interface Program, which is funded in part by training grant T32-5R18CE001240 from the National Institute of General Medical Sciences. The findings and conclusions in this report are those of the authors and do not necessarily represent the views of the National Institute for Occupational Safety and Health.
Footnotes
This work has been presented at the New York Academy of Sciences, “Shift Work Duration and Patterns of Salivary Cortisol Among Police Officers” [poster], June 2009, New York, New York; and at the South Carolina Public Health Association, “Shift Work Duration and Patterns of Salivary Cortisol among Police Officers” [oral presentation), May 2010, Myrtle Beach, South Carolina.
Declaration of Interest: The authors report no conflicts of interest. The authors alone are responsible for the content and writing of the paper.
References
- Åkerstedt T. Psychological and psychophysiological effects of shift work. Scand J Work Environ Health. 1990;16(Suppl 1):67–73. doi: 10.5271/sjweh.1819. [DOI] [PubMed] [Google Scholar]
- Åkerstedt T. Shift work and disturbed sleep/wakefulness. Sleep Med Rev. 1998;2:117–128. doi: 10.1016/s1087-0792(98)90004-1. [DOI] [PubMed] [Google Scholar]
- Axelsson J, Akerstedt T, Kecklund G, Lindqvist A, Attefors R. Hormonal changes in satisfied and dissatisfied shift workers across a shift cycle. J Appl Physiol. 2003;95:2099–2105. doi: 10.1152/japplphysiol.00231.2003. [DOI] [PubMed] [Google Scholar]
- Backhaus J, Junghanns K, Hohagen F. Sleep disturbances are correlated with decreased morning awakening salivary cortisol. Psychoneuroendocrinology. 2004;29:1184–1191. doi: 10.1016/j.psyneuen.2004.01.010. [DOI] [PubMed] [Google Scholar]
- Bartels M, Van den Berg M, Sluyter F, Boomsma DI, de Geus EJ. Heritability of cortisol levels: review and simultaneous analysis of twin studies. Psychoneuroendocrinology. 2003;28:121–137. doi: 10.1016/s0306-4530(02)00003-3. [DOI] [PubMed] [Google Scholar]
- Boggild H, Knutsson A. Shift work, risk factors and cardiovascular disease. Scand J Work Environ Health. 1999;25:85–99. doi: 10.5271/sjweh.410. [DOI] [PubMed] [Google Scholar]
- Boquet A, Cruz C, Nesthus T, Detwiler C, Knecht W, Holcomb K. Clockwise and counterclockwise rotating shifts: effects on temperature and neuroendocrine measures. Aviat Space Environ Med. 2004;75:898–904. [PubMed] [Google Scholar]
- Brooke-Wavell K, Clow A, Ghazi-Noori S, Evans P, Hucklebridge F. Ultrasound measures of bone and the diurnal free cortisol cycle: a positive association with the awakening cortisol response in healthy premenopausal women. Calcif Tissue Int. 2002;70:463–468. doi: 10.1007/s00223-001-2085-8. [DOI] [PubMed] [Google Scholar]
- Burch JB, Yost MG, Johnson W, Allen E. Melatonin, sleep, and shift work adaptation. J Occup Environ Med. 2005;47:893–901. doi: 10.1097/01.jom.0000177336.21147.9f. [DOI] [PubMed] [Google Scholar]
- Burch JB, Tom J, Zhai Y, Criswell L, Leo E, Ogoussan K. Shift-work impacts and adaptation among health care workers. Occup Med (Lond) 2009;59:159–166. doi: 10.1093/occmed/kqp015. [DOI] [PubMed] [Google Scholar]
- Burke HM, Davis MC, Otte C, Mohr DC. Depression and cortisol responses to psychological stress: a meta-analysis. Psychoneuroendocrinology. 2005;30:846–856. doi: 10.1016/j.psyneuen.2005.02.010. [DOI] [PubMed] [Google Scholar]
- Camerino D, Sandri M, Sartori S, Conway PM, Campanini P, Costa G. Shiftwork, work-family conflict among Italian nurses, and prevention efficacy. Chronobiol Int. 2010:1105–1123. doi: 10.3109/07420528.2010.490072. [DOI] [PubMed] [Google Scholar]
- Cavallo A, Jaskiewicz J, Ris MD. Impact of night-float rotation on sleep, mood, and alertness: the resident's perception. Chronobiol Int. 2002;19:893–902. doi: 10.1081/cbi-120014106. [DOI] [PubMed] [Google Scholar]
- Charles LE, Burchfiel CM, Fekedulegn D, Vila B, Hartley TA, Slaven J, Mnatsakanova A, Violanti JM. Shift-work and sleep: the Buffalo police health study. Policing Int J Police Strat Mgmt. 2007;30:215–227. [Google Scholar]
- Chen JD, Lin YC, Hsiao ST. Obesity and high blood pressure of 12-hour night shift female clean-room workers. Chronobiol Int. 2010;27:334–344. doi: 10.3109/07420520903502242. [DOI] [PubMed] [Google Scholar]
- Chida Y, Steptoe A. Cortisol awakening response and psychosocial factors: a systematic review and meta-analysis. Biol Psychol. 2009;80:265–278. doi: 10.1016/j.biopsycho.2008.10.004. [DOI] [PubMed] [Google Scholar]
- Clow A, Patel S, Najafi M, Evans PD, Hucklebridge F. The cortisol response to psychological challenge is preceded by a transient rise in endogenous inhibitor of monoamine oxidase. Life Sci. 1997;61:567–575. doi: 10.1016/s0024-3205(97)00416-5. [DOI] [PubMed] [Google Scholar]
- Clow A, Thorn L, Evans P, Hucklebridge F. The awakening cortisol response: methodological issues and significance. Stress. 2004;7:29–37. doi: 10.1080/10253890410001667205. [DOI] [PubMed] [Google Scholar]
- Coffey LC, Skipper JK, Jr, Jung FD. Nurses and shift work: effects on job performance and job-related stress. J Adv Nurs. 1988;13:245–254. doi: 10.1111/j.1365-2648.1988.tb01414.x. [DOI] [PubMed] [Google Scholar]
- Conlon M, Lightfoot N, Kreiger N. Rotating shift work and risk of prostate cancer. Epidemiology. 2007;18:182–183. doi: 10.1097/01.ede.0000249519.33978.31. [DOI] [PubMed] [Google Scholar]
- Coste J, Strauch G, Letrait M, Bertagna X. Reliability of hormonal levels for assessing the hypothalamic-pituitary-adrenocortical system in clinical pharmacology. Br J Clin Pharmacol. 1994;38:474–479. doi: 10.1111/j.1365-2125.1994.tb04386.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crowley SJ, Lee C, Tseng CY, Fogg LF, Eastman CI. Complete or partial circadian re-entrainment improves performance, alertness, and mood during night-shift work. Sleep. 2004;27:1077–1087. doi: 10.1093/sleep/27.6.1077. [DOI] [PubMed] [Google Scholar]
- de Weerth C, Zijl RH, Buitelaar JK. Development of cortisol circadian rhythm in infancy. Early Hum Dev. 2003;73:39–52. doi: 10.1016/s0378-3782(03)00074-4. [DOI] [PubMed] [Google Scholar]
- Dembe AE, Erickson JB, Delbos RG, Banks SM. Nonstandard shift schedules and the risk of job-related injuries. Scand J Work Environ Health. 2006;32:232–240. doi: 10.5271/sjweh.1004. [DOI] [PubMed] [Google Scholar]
- Fava GA. Assessing depressive symptoms across cultures: Italian validation of the CES-D self-rating scale. J Clin Psychol. 1983;39:249–251. doi: 10.1002/1097-4679(198303)39:2<249::aid-jclp2270390218>3.0.co;2-y. [DOI] [PubMed] [Google Scholar]
- Fekedulegn DB, Andrew ME, Burchfiel CM, Violanti JM, Hartley TA, Charles LE, Miller DB. Area under the curve and other summary indicators of repeated waking cortisol measurements. Psychosom Med. 2007;69:651–659. doi: 10.1097/PSY.0b013e31814c405c. [DOI] [PubMed] [Google Scholar]
- Forastiere F, Perucci CA, Di Pietro A, Miceli M, Rapiti E, Bargagli A, Borgia P. Mortality among urban policemen in Rome. Am J Ind Med. 1994;26:785–798. doi: 10.1002/ajim.4700260607. [DOI] [PubMed] [Google Scholar]
- Franke WD, Cox DF, Schultz DP, Anderson DF. Coronary heart disease risk factors in employees of Iowa's Department of Public Safety compared to a cohort of the general population. Am J Ind Med. 1997;31:733–737. doi: 10.1002/(sici)1097-0274(199706)31:6<733::aid-ajim10>3.0.co;2-z. [DOI] [PubMed] [Google Scholar]
- Fries E, Dettenborn L, Kirschbaum C. The cortisol awakening response (CAR): facts and future directions. Int J Psychophysiol. 2009;72:67–73. doi: 10.1016/j.ijpsycho.2008.03.014. [DOI] [PubMed] [Google Scholar]
- Gidron Y, Ronson A. Psychosocial factors, biological mediators, and cancer prognosis: a new look at an old story. Curr Opin Oncol. 2008;20:386–392. doi: 10.1097/CCO.0b013e3282fbcd0d. [DOI] [PubMed] [Google Scholar]
- Goh VH, Tong TY, Lim CL, Low EC, Lee LK. Circadian disturbances after night-shift work onboard a naval ship. Mil Med. 2000;165:101–105. [PubMed] [Google Scholar]
- Gordon NP, Cleary PD, Parker CE, Czeisler CA. The prevalence and health impact of shiftwork. Am J Public Health. 1986;76:1225–1258. doi: 10.2105/ajph.76.10.1225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griefahn B, Robens S. The cortisol awakening response: a pilot study on the effects of shift work, morningness and sleep duration. Psychoneuroendocrinology. 2008;33:981–988. doi: 10.1016/j.psyneuen.2008.04.004. [DOI] [PubMed] [Google Scholar]
- Ha M, Park J. Shiftwork and metabolic risk factors of cardiovascular disease. J Occup Health. 2005;47:89–95. doi: 10.1539/joh.47.89. [DOI] [PubMed] [Google Scholar]
- Harada H, Suwazono Y, Sakata K, Okubo Y, Oishi M, Uetani M, Kobayashi E, Nogawa K. Three-shift system increases job-related stress in Japanese workers. J Occup Health. 2005;47:397–404. doi: 10.1539/joh.47.397. [DOI] [PubMed] [Google Scholar]
- Hennig J, Kieferdorf P, Moritz C, Huwe S, Netter P. Changes in cortisol secretion during shiftwork: implications for tolerance to shiftwork? Ergonomics. 1998;41:610–21. doi: 10.1080/001401398186784. [DOI] [PubMed] [Google Scholar]
- Henry JP. Biological basis of the stress response. Integr Physiol Behav Sci. 1992;27:66–83. doi: 10.1007/BF02691093. [DOI] [PubMed] [Google Scholar]
- Horowitz M, Wilner M, Alvarez W. Impact of event scale: a measure of subjective stress. Psychosom Med. 1979;41:209–218. doi: 10.1097/00006842-197905000-00004. [DOI] [PubMed] [Google Scholar]
- Horwitz IB, McCall BP. The impact of shift work on the risk and severity of injuries for hospital employees: an analysis using Oregon workers' compensation data. Occup Med (Lond) 2004;54:556–563. doi: 10.1093/occmed/kqh093. [DOI] [PubMed] [Google Scholar]
- Huber TJ, Issa K, Schik G, Wolf OT. The cortisol awakening response is blunted in psychotherapy inpatients suffering from depression. Psychoneuroendocrinology. 2006;31:900–904. doi: 10.1016/j.psyneuen.2006.03.005. [DOI] [PubMed] [Google Scholar]
- Hurwitz Eller N, Netterstrom B, Hansen AM. Cortisol in urine and saliva: relations to the intima media thickness, IMT. Atherosclerosis. 2001;159:175–185. doi: 10.1016/s0021-9150(01)00487-7. [DOI] [PubMed] [Google Scholar]
- Jolliffe IT. Principal components analysis. New York: Springer-Verlag New York, Inc; 2010. [Google Scholar]
- Karlsson BH, Knutsson AK, Lindahl BO, Alfredsson LS. Metabolic disturbances in male workers with rotating three-shift work. Results of the WOLF study. Int Arch Occup Environ Health. 2003;76:424–430. doi: 10.1007/s00420-003-0440-y. [DOI] [PubMed] [Google Scholar]
- Khalfa S, Bella SD, Roy M, Peretz I, Lupien SJ. Effects of relaxing music on salivary cortisol level after psychological stress. Ann N Y Acad Sci. 2003;999:374–376. doi: 10.1196/annals.1284.045. [DOI] [PubMed] [Google Scholar]
- Kubo T, Ozasa K, Mikami K, Wakai K. Prospective cohort of the risk of prostate cancer among rotating-shift workers: findings from the Japan collaborative cohort study. Am J Epidemiol. 2006;164:549–555. doi: 10.1093/aje/kwj232. [DOI] [PubMed] [Google Scholar]
- Kudielka BM, Kirschbaum C. Awakening cortisol responses are influenced by health status and awakening time but not by menstrual cycle phase. Psychoneuroendocrinology. 2003;28:35–47. doi: 10.1016/s0306-4530(02)00008-2. [DOI] [PubMed] [Google Scholar]
- Kudielka BM, Federenko IS, Hellhammer DH, Wüst S. Morningness and eveningness: the free cortisol rise after awakening in “early birds” and “night owls”. Biol Psychol. 2006;72:141–146. doi: 10.1016/j.biopsycho.2005.08.003. [DOI] [PubMed] [Google Scholar]
- Kudielka BM, Buchtal J, Uhde A, Wust S. Circadian cortisol profiles and psychological self-reports in shift workers with and without recent change in the shift rotation system. Biol Psychol. 2007;74:92–103. doi: 10.1016/j.biopsycho.2006.08.008. [DOI] [PubMed] [Google Scholar]
- Kudielka BM, Hellhammer DH, Wust S. Why do we respond so differently? Reviewing determinants of human salivary cortisol responses to challenge. Psychoneuroendocrinology. 2009;34:2–18. doi: 10.1016/j.psyneuen.2008.10.004. [DOI] [PubMed] [Google Scholar]
- Lac G, Chamoux A. Biological and psychological responses to two rapid shiftwork schedules. Ergonomics. 2004;47:1339–1349. doi: 10.1080/00140130410001724237. [DOI] [PubMed] [Google Scholar]
- Lavie L, Lavie P. Elevated plasma homocysteine in older shiftworkers: a potential risk factor for cardiovascular morbidity. Chronobiol Int. 2007;24:115–128. doi: 10.1080/07420520601139797. [DOI] [PubMed] [Google Scholar]
- Leese G, Chattington P, Fraser W, Vora J, Edwards R, Williams G. Short-term night-shift working mimics the pituitary-adrenocortical dysfunction in chronic fatigue syndrome. J Clin Endocrinol Metab. 1996;81:1867–1870. doi: 10.1210/jcem.81.5.8626849. [DOI] [PubMed] [Google Scholar]
- Lippi G, De Vita F, Salvagno GL, Gelati M, Montagnana M, Guidi GC. Measurement of morning saliva cortisol in athletes. Clin Biochem. 2009;42:904–906. doi: 10.1016/j.clinbiochem.2009.02.012. [DOI] [PubMed] [Google Scholar]
- Maina G, Palmas A, Bovenzi M, Filon FL. Salivary cortisol and psychosocial hazards at work. Am J Ind Med. 2009;52:251–260. doi: 10.1002/ajim.20659. [DOI] [PubMed] [Google Scholar]
- McEwen BS. Protective and damaging effects of stress mediators: central role of the brain. Dialogues Clin Neurosci. 2006;8:367–381. doi: 10.31887/DCNS.2006.8.4/bmcewen. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen BS. Central effects of stress hormones in health and disease: understanding the protective and damaging effects of stress and stress mediators. Eur J Pharmacol. 2008;583:174–185. doi: 10.1016/j.ejphar.2007.11.071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McEwen BS, Stellar E. Stress and the individual—mechanisms leading to disease. Arch Int Med. 1993;153:2093–2101. [PubMed] [Google Scholar]
- Meinlschmidt G, Heim C. Decreased cortisol awakening response after early loss experience. Psychoneuroendocrinology. 2005;30:568–576. doi: 10.1016/j.psyneuen.2005.01.006. [DOI] [PubMed] [Google Scholar]
- Miller WC, Anton HA, Townson AF. Measurement properties of the CESD scale among individuals with spinal cord injury. Spinal Cord. 2008;46:287–292. doi: 10.1038/sj.sc.3102127. [DOI] [PubMed] [Google Scholar]
- Motzer SA, Hertig V. Stress, stress response, and health. Nurs Clin North Am. 2004;39:1–17. doi: 10.1016/j.cnur.2003.11.001. [DOI] [PubMed] [Google Scholar]
- Munakata M, Ichi S, Nunokawa T, Saito Y, Ito N, Fukudo S, Yoshinaga K. Influence of night shift work on psychologic state and cardiovascular and neuroendocrine responses in healthy nurses. Hypertens Res. 2001;24:25–31. doi: 10.1291/hypres.24.25. [DOI] [PubMed] [Google Scholar]
- Navara KJ, Nelson RJ. The dark side of light at night: physiological, epidemiological, and ecological consequences. J Pineal Res. 2007;43:215–224. doi: 10.1111/j.1600-079X.2007.00473.x. [DOI] [PubMed] [Google Scholar]
- Neylan TC, Brunet A, Pole N, Best SR, Metzler TJ, Yehuda R, Marmar CR. PTSD symptoms predict waking salivary cortisol levels in police officers. Psychoneuroendocrinology. 2005;30:373–381. doi: 10.1016/j.psyneuen.2004.10.005. [DOI] [PubMed] [Google Scholar]
- Oginska H, Fafrowicz, Golonka K, Marek T, Mojsa-Kaja J, Tuscholska K. Chronotype, sleep loss, and diurnal pattern of salivary cortisol in a simulated daylong driving. Chronobiol Int. 2010;27:959–974. doi: 10.3109/07420528.2010.489412. [DOI] [PubMed] [Google Scholar]
- Ohayon MM, Smolensky MH, Roth T. Consequences of shiftworking on sleep duration, sleepiness and sleep attacks. Chronobiol Int. 2010;27:573–589. doi: 10.3109/07420521003749956. [DOI] [PubMed] [Google Scholar]
- Paykel ES, Prusoff BA, Uhlenhuth EH. Scaling of life events. Arch Gen Psychiatry. 1971;25:340–347. doi: 10.1001/archpsyc.1971.01750160052010. [DOI] [PubMed] [Google Scholar]
- Portaluppi F, Smolensky MH, Touitou Y. Ethics and methodsfor biological rhythm research on animals and human beings. Chronobiol Int. 2010;27:1911–1929. doi: 10.3109/07420528.2010.516381. [DOI] [PubMed] [Google Scholar]
- Pruessner JC, Hellhammer DH, Kirschbaum C. Burnout, perceived stress, and cortisol responses to awakening. Psychosom Med. 1999;61:197–204. doi: 10.1097/00006842-199903000-00012. [DOI] [PubMed] [Google Scholar]
- Pruessner JC, Wolf OT, Hellhammer DH, Buske-Kirschbaum A, von Auer K, Jobst S, Kaspers F, Kirschbaum C. Free cortisol levels after awakening: a reliable biological marker for the assessment of adrenocortical activity. Life Sci. 1997;61:2539–2549. doi: 10.1016/s0024-3205(97)01008-4. [DOI] [PubMed] [Google Scholar]
- Quera-Salva MA, Guilleminault C, Claustrat B, Defrance R, Gajdos P, McCann CC, De Lattre J. Rapid shift in peak melatonin secretion associated with improved performance in short shift work schedule. Sleep. 1997;20:1145–1150. [PubMed] [Google Scholar]
- Radloff LS. The CES-D Scale: a self-report depression scale for research in the general populations. Appl Psych Meas. 1977;1:385–401. [Google Scholar]
- Ramey SL, Downing NR, Knoblauch A. Developing strategic interventions to reduce cardiovascular disease risk among law enforcement officers: the art and science of data triangulation. AAOHN J. 2008;56:54–62. doi: 10.1177/216507990805600202. [DOI] [PubMed] [Google Scholar]
- Ratloff LS. The CES-D scale: a self-report depression scale for research in the general population. Appl Psychol Meas. 1977;1:385–401. [Google Scholar]
- Reinberg A, Ashkenazi I. Internal desynchronization of circadian rhythms and tolerance to shift work. Chronobiol Int. 2008;25:625–643. doi: 10.1080/07420520802256101. [DOI] [PubMed] [Google Scholar]
- Reinberg AE, Ashkenazi I, Smolensky MH. Euchronism, allochronism, and dyschronism: is internal desynchronization of human circadian rhythms a sign of illness? Chronobiol Int. 2007;24:553–588. doi: 10.1080/07420520701534624. [DOI] [PubMed] [Google Scholar]
- Roberts AD, Wessely S, Chalder T, Papadopoulos A, Cleare AJ. Salivary cortisol response to awakening in chronic fatigue syndrome. Br J Psychiatry. 2004;184:136–141. doi: 10.1192/bjp.184.2.136. [DOI] [PubMed] [Google Scholar]
- Roberts RE, Vernon SW. The Center for Epidemiologic Studies Depression Scale: its use in a community sample. Am J Psychiatry. 1983;140:41–46. doi: 10.1176/ajp.140.1.41. [DOI] [PubMed] [Google Scholar]
- Roden M, Koller M, Pirich K, Vierhapper H, Waldhauser F. The circadian melatonin and cortisol secretion pattern in permanent night shift workers. Am J Physiol. 1993;265:R261–R267. doi: 10.1152/ajpregu.1993.265.1.R261. [DOI] [PubMed] [Google Scholar]
- Rohleder N, Joksimovic L, Wolf JM, Kirschbaum C. Hypocortisolism and increased glucocorticoid sensitivity of pro-inflammatory cytokine production in Bosnian war refugees with posttraumatic stress disorder. Biol Psychiatry. 2004;55:745–751. doi: 10.1016/j.biopsych.2003.11.018. [DOI] [PubMed] [Google Scholar]
- Scheer FA, Shea TJ, Hilton MF, Shea SA. An endogenous circadian rhythm in sleep inertia results in greatest cognitive impairment upon awakening during the biological night. J Biol Rhythms. 2008;23:353–361. doi: 10.1177/0748730408318081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Colditz GA. Rotating night shifts and risk of breast cancer in women participating in the nurses' health study. J Natl Cancer Inst. 2001;93:1563–1568. doi: 10.1093/jnci/93.20.1563. [DOI] [PubMed] [Google Scholar]
- Schernhammer ES, Laden F, Speizer FE, Willett WC, Hunter DJ, Kawachi I, Fuchs CS, Colditz GA. Night-shift work and risk of colorectal cancer in the nurses' health study. J Natl Cancer Inst. 2003;95:825–828. doi: 10.1093/jnci/95.11.825. [DOI] [PubMed] [Google Scholar]
- Sephton S, Spiegel D. Circadian disruption in cancer: a neuroendocrine-immune pathway from stress to disease? Brain Behav Immun. 2003;17:321–328. doi: 10.1016/s0889-1591(03)00078-3. [DOI] [PubMed] [Google Scholar]
- Shields M. Shift work and health. Health Rep. 2002;13:11–33. [PubMed] [Google Scholar]
- Stetler C, Miller GE. Blunted cortisol response to awakening in mild to moderate depression: regulatory influences of sleep patterns and social contacts. J Abnorm Psychol. 2005;114:697–705. doi: 10.1037/0021-843X.114.4.697. [DOI] [PubMed] [Google Scholar]
- Stone AA, Schwartz JE, Smyth J, Kirschbaum C, Cohen S, Hellhammer D, Grossman S. Individual differences in the diurnal cycle of salivary free cortisol: a replication of flattened cycles for some individuals. Psychoneuroendocrinology. 2001;26:295–306. doi: 10.1016/s0306-4530(00)00057-3. [DOI] [PubMed] [Google Scholar]
- Takahashi M, Nakata A, Haratani T, Otsuka Y, Kaida K, Fukasawa K. Psychosocial work characteristics predicting daytime sleepiness in day and shift workers. Chronobiol Int. 2006;23:1409–1422. doi: 10.1080/07420520601100963. [DOI] [PubMed] [Google Scholar]
- Touitou Y, Motohashi Y, Reinberg A, Touitou C, Bourdeleau P, Bogdan A, Auzeby A. Effect of shift work on the night-time secretory patterns of melatonin, prolactin, cortisol and testosterone. Eur J Appl Physiol Occup Physiol. 1990;60:288–292. doi: 10.1007/BF00379398. [DOI] [PubMed] [Google Scholar]
- Vena JE, Violanti JM, Marshall J, Fiedler RC. Mortality of a municipal worker cohort: III. Police officers. Am J Ind Med. 1986;10:383–397. doi: 10.1002/ajim.4700100406. [DOI] [PubMed] [Google Scholar]
- Vila B. Impact of long work hours on police officers and the communities they serve. Am J Ind Med. 2006;49:972–980. doi: 10.1002/ajim.20333. [DOI] [PubMed] [Google Scholar]
- Violanti JM, Vena JE, Petralia S. Mortality of a police cohort: 1950–1990. Am J Ind Med. 1998;33:366–373. doi: 10.1002/(sici)1097-0274(199804)33:4<366::aid-ajim6>3.0.co;2-s. [DOI] [PubMed] [Google Scholar]
- Violanti JM, Burchfiel CM, Miller DB, Andrew ME, Dorn J, Wactawki-Wende J, Beighley CM, Pierino K, Joseph PN, Vena JE, Sharp DS, Trevisan M. The Buffalo Cardio-Metabolic Occupational Police Stress (BCOPS) pilot study: methods and participant characteristics. Ann Epidemiol. 2006;16:148–156. doi: 10.1016/j.annepidem.2005.07.054. [DOI] [PubMed] [Google Scholar]
- Violanti JM, Andrew M, Burchfiel CM, Hartley TA, Charles LE, Miller DB. Post-traumatic stress symptoms and cortisol patterns among police officers. Policing Int J Police Strat Mgmt. 2007;30:189–202. [Google Scholar]
- Violanti JM, Charles LE, Hartley TA, Mnatsakanova A, Andrew ME, Fekedulegn D, Vila B, Burchfiel CM. Shift-work and suicide ideation among police officers. Am J Ind Med. 2008;51:758–768. doi: 10.1002/ajim.20629. [DOI] [PubMed] [Google Scholar]
- Violanti JM, Burchfiel CM, Fekedulegn D, Andrew ME, Dorn J, Hartley TA, Charles LE, Miller DB. Cortisol patterns and brachial artery reactivity in a high stress environment. Psychiatry Res. 2009a;169:75–81. doi: 10.1016/j.psychres.2008.06.012. [DOI] [PubMed] [Google Scholar]
- Violanti JM, Burchfiel CM, Hartley TA, Mnatsakanova A, Fekedulegn D, Andrew ME, Charles LE, Vila BJ. Atypical work hours and metabolic syndrome among police officers. Arch Environ Occup Health. 2009b;64:194–201. doi: 10.1080/19338240903241259. [DOI] [PubMed] [Google Scholar]
- Weissman MM, Bland RC, Canino GJ, Faravelli C, Greenwald S, Hwu HG, Joyce PR, Karam EG, Lee CK, Lellouch J, Lépine JP, Newman SC, Rubio-Stipec M, Wells JE, Wickramaratne PJ, Wttchen H, Yeh EK. Cross-national epidemiology of major depression and bipolar disorder. JAMA. 1996;276:293–299. [PubMed] [Google Scholar]
- Wust S, Wolf J, Hellhammer DH, Federenko I, Schommer N, Kirschbaum C. The cortisol awakening response—normal values and confounds. Noise Health. 2000;2:79–88. [PubMed] [Google Scholar]
- Zuzewicz K, Kwarecki K, Waterhouse J. Circadian rhythm of heart rate, urinary cortisol excretion, and sleep in civil air traffic controllers. Int J Occup Saf Ergon. 2000;6:383–392. doi: 10.1080/10803548.2000.11076462. [DOI] [PubMed] [Google Scholar]