Opinion statement
Solid organ transplantation is frequently complicated by a spectrum of seizure types, including single partial-onset or generalized tonic-clonic seizures, acute repetitive seizures or status epilepticus, and sometimes the evolution of symptomatic epilepsy. There is currently no specific evidence involving the transplant patient population to guide the selection, administration, or duration of antiepileptic drug (AED) therapy, so familiarity with clinical AED pharmacology and application of sound judgment are necessary for successful patient outcomes. An initial detailed search for symptomatic seizure etiologies, including metabolic, infectious, cerebrovascular, and calcineurin inhibitor treatment-related neuro-toxic complications such as posterior reversible encephalopathy syndrome (PRES), is imperative, as underlying central nervous system disorders may impose additional serious risks to cerebral or general health if not promptly detected and appropriately treated. The mainstay for post-transplant seizure management is AED therapy directed toward the suspected seizure type. Unfavorable drug interactions could place the transplanted organ at risk, so choosing an AED with limited interaction potential is also crucial. When the transplanted organ is dysfunctional or vulnerable to rejection, AEDs without substantial hepatic metabolism are favored in post-liver transplant patients, whereas after renal transplantation, AEDs with predominantly renal elimination may require dosage adjustment to prevent adverse effects. Levetiracetam, gabapentin, pregabalin, and lacosamide are drugs of choice for treatment of partial-onset seizures in post-transplant patients given their efficacy spectrum, generally excellent tolerability, and lack of drug interaction potential. Levetiracetam is the drug of choice for primary generalized seizures in post-transplant patients. When intravenous drugs are necessary for acute seizure management, benzodiazepines and fosphenytoin are the traditional and best evidence-based options, although intravenous levetiracetam, valproate, and lacosamide are emerging options. Availability of several newer AEDs has greatly expanded the therapeutic armamentarium for safe and efficacious treatment of post-transplant seizures, but future prospective clinical trials and pharmacokinetic studies within this specific patient population are needed.
Keywords: Transplant, Seizure, Epilepsy, Status epilepticus, Acute repetitive seizures, Posterior reversible encephalopathy syndrome, PRES, Infection, Antiepileptic drugs, Cyclosporine toxicity, Tacrolimus toxicity, Treatment
Introduction
Solid organ transplantation (SOT) has become a common and crucial life-saving procedure. In 2010, nearly 17,000 Americans received kidney transplants, more than 6,000 received liver transplants, and more than 2,300 heart and 1,750 lung transplants were performed, while pancreas and intestine transplants occurred less frequently [1]. Additional transplants include bone marrow, peripheral blood stem cell, and umbilical cord blood, of which a combined total of more than 5,500 were facilitated in 2011 by the National Marrow Donor Program alone [2]. SOT is a major operation and carries many risks, including acute risks of severe perioperative blood loss [3–6], infection [7–12, 13••, 14], and acute organ rejection [15, 16].
Antirejection therapy is almost universally implemented following SOT, with the exception of isografts involving genetically identical donor organs. Antirejection regimens commonly combine a cornerstone calcineurin inhibitor (CNI) such as tacrolimus or cyclosporine with adjunctive agents, including corticosteroids, anti-proliferatives such as mycophenolate or azathioprine, and sometimes antibody therapy [13••, 17]. Although imperative for the prevention of organ rejection, immunosuppressive drugs, particularly the CNIs, can induce central nervous system (CNS) complications following SOT [18–22].
New-onset seizures occur in 2 % to 24 % of SOT patients, making seizures the second most common post-transplant CNS complication next to neurotoxicity [13••]. Seizures after SOT may be partial-onset or primary generalized, and are usually tonic-clonic [13••]. The largest study investigating seizure types, causes, and outcomes in a transplant population examined 630 liver transplant patients and found that generalized tonic-clonic seizures were the most common seizure type, occurring in 28 patients [21]. Of these, more than half were attributed to CNI neurotoxicity due to tacrolimus or cyclosporine toxicity following abrupt dosing increases or abnormally high blood levels. Other causes implicated were CNS infection and major CNS catastrophe.
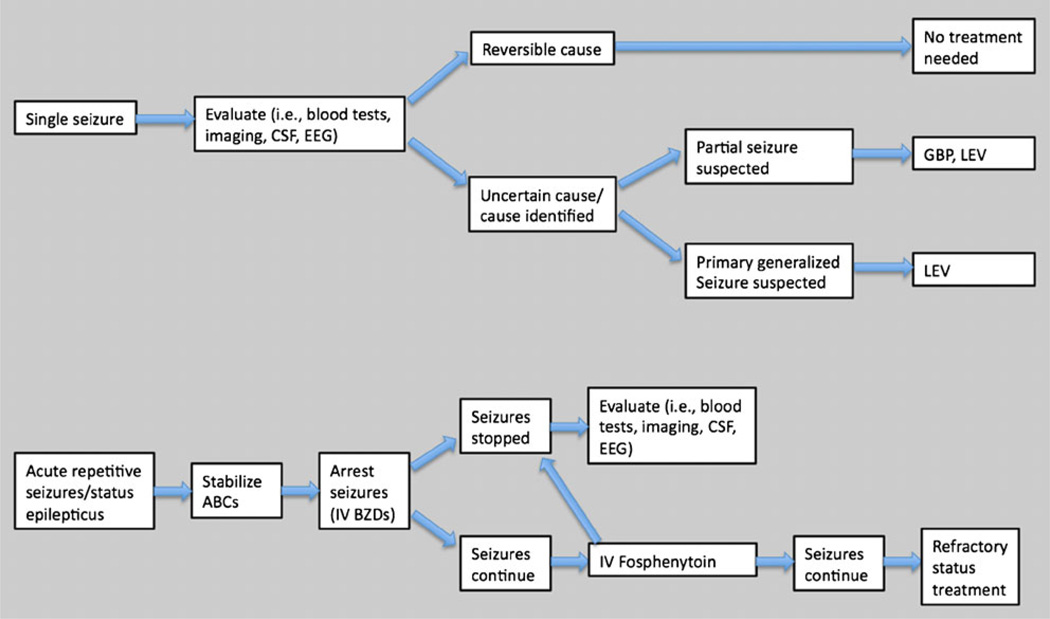
Post-transplant patients may experience a spectrum of different seizure types that require different diagnostic and treatment approaches (Fig. 1). Despite the frequency of seizures following SOT, there is currently no good evidence basis to guide AED selection, administration, or duration specifically within the transplant patient population. Therefore, antiepileptic drug (AED) therapeutics for the post-transplant patient require knowledge of clinical pharmacology and applied clinical wisdom to ensure successful treatment outcomes. This article considers the diagnosis and treatment of seizures in post-transplant patients.
Figure 1.
Algorithm for treatment of seizures in transplant patients. Treatment of seizures in transplant patients must take into account the transplanted organ and antirejection comedications so that antiepileptic drugs (AEDs) do not place the transplanted organ at further risk. For single partial-onset or secondary generalized tonic-clonic seizures, investigation for an underlying symptomatic etiology should be promptly undertaken, and treatment may be deferred if a correctable etiology is determined and seizures do not recur. If seizures recur, or when seizure recurrence risk is uncertain or an underlying epileptogenic cause is found, treatment with levetira-cetam or gabapentin should be initiated as appropriate for suspected seizure type. For status epilepticus or acute repetitive seizures, intravenous lorazepam, followed, if necessary, by fosphenytoin should be administered. ABCs airway, breathing, and circulation; BZD benzodiazepine; CSF cerebrospinal fluid; EEG electroencephalography; GBP gabapentin; IV intravenous; LEV levetiracetam.
Diagnostic considerations for symptomatic seizures in transplant patients
Because many acute symptomatic seizures are self-limited and do not recur following elimination or correction of the underlying cause, determination of the etiology for seizures should be expeditiously sought in every patient. Equally important, prompt identification and treatment of a serious underlying symptomatic neurological disorder may prevent evolving neurologic damage.
Measurement of vital signs is important, as malignant hypertension may provoke seizures in some renal transplant patients [23]. Prompt investigation should be considered in most transplant patients who experience new-onset seizures, including measurement of serum calcium, glucose, magnesium, phosphorus, sodium, complete blood count, antirejection medication blood levels, and blood and urine cultures in selected patients. Brain MRI should also be strongly considered to exclude newly evolved lesional pathologies, with contrast administration whenever possible from the standpoint of organ status and general health. Cerebrospinal fluid (CSF) examination is necessary to exclude primary CNS infection in immuno-compromised transplant patients when there are signs of infection or meningismus [24]. In selected cases, skin or bronchoscopic lung biopsy may provide ancillary clues for CNS infection.
While urgent electroencephalography (EEG) is necessary in selected encephalopathic patients to exclude non-convulsive status epilepticus, and EEG may aid in determining the underlying seizure mechanism, most patients with new-onset seizures following transplant are presumed to have a partial-onset seizure mechanism. Additionally, a lack of prompt EEG availability and the option of selecting a broad-spectrum AED for empiric treatment may render EEG relatively unnecessary in most patients who promptly recover consciousness following seizure episodes. Common causes of seizures following SOT, including metabolic dyscrasias, infection, cerebrovascular disorders, and posterior reversible encephalopathy syndrome (PRES), will now be further reviewed.
Metabolic dyscrasias
Prompt exclusion and correction of common seizure-provoking electrolyte imbalances such as hyponatremia, hypomagnesemia, hypocalcemia, and hypoglycemia should be considered in all patients with new-onset seizures following transplant [23, 25–27]. Selected transplant patients may be selectively vulnerable to specific dyselectrolytemias. Hypomagnesemia due to cyclosporine-induced renal wasting is important to consider in the transplant population [28], and hypoglycemia may occur in pediatric renal transplant recipients [29]. Hepatic, renal, or marrow insufficiencies should also be considered in patients with organ rejection.
Infection
Infections of the CNS occur in 5 % to 10 % of patients after SOT [8], with Aspergillus fumigatus, Listeria monocytogenes, and Cryptococcus neoformans accounting for the vast majority [9]. Early detection and initiation of treatment is crucial to increase the likelihood of survival. The timeline of symptom onset can aid diagnosis; the median time of onset of CNS aspergillosis is 21 to 24 days after SOT [10, 12], while bacterial infections usually occur in the first 2 months following transplantation, and viral and opportunistic infections occur most frequently over 6 months after transplantation [7, 13••].
Most post-transplant brain abscesses are fungal, with Aspergillus most commonly responsible [10–12]. In a prospective study of 60 brain lesions following liver transplant, 11 (18 %) were fungal brain abscesses, with Aspergillus implicated in more than half, and seizures the presenting clinical feature in 4 of these patients [10]. Among bacterial CNS infections following transplant, Nocardia are most common, affecting 1 % to 6 % of SOT recipients and usually occurring 1 to 6 months post-transplant, although Nocardia abscesses are rare [14].
Posterior reversible encephalopathy syndrome
Prompt recognition and diagnosis of posterior reversible encephalopathy syndrome (PRES) is crucial in transplant patients given its frequency and risk of permanent neurological complications. The reported incidence of PRES after SOT is 0.4 % to 0.5 % [30]. While antirejection therapy has greatly improved survival rates after SOT, CNI-induced PRES is a severe complication, presenting most often in the setting of CNI neurotoxicity with seizures, altered mental status, headache, focal neurological deficits including visual loss, or stupor, and is associated with posteriorly predominant, vasogenic, gyriform cerebral edema on brain MRI [22, 31•]. Significant associations in more than 50 % of PRES patients include hypertension, high cyclosporine levels, hypomagnesemia, and hypocholesterolemia [18]. Significant risk factors for CNI-induced neurotoxicity after liver transplant include pretransplant hepatic encephalopathy, post-transplant hyponatremia, and surgical time greater than 7 hours [32]. The etiology of PRES is still not well-understood but is thought to result from drug-induced hypertension and capillary leakage due to compromised endothelial tissue integrity [22, 33].
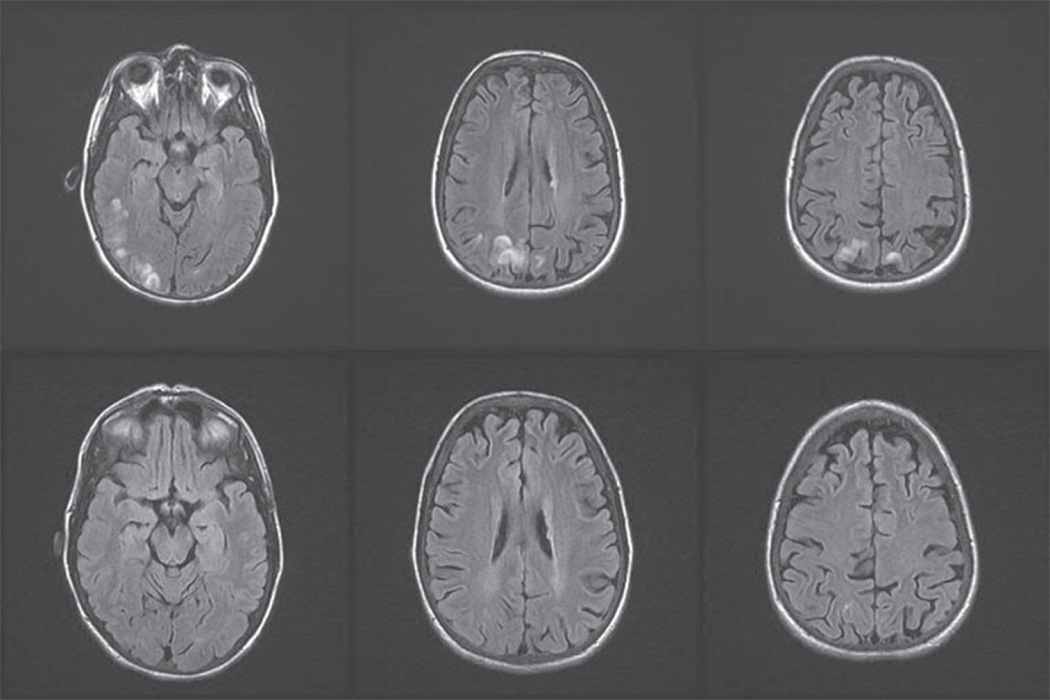
The neuroimaging manifestations of PRES are typically characterized by parieto-occipital cortical and subcortical edema but may involve the frontal lobe or cerebellum while less commonly affecting the basal ganglia and brainstem [31•]. Affected regions appear on CT as white-matter hypodensity and on T1- and T2-weighted MRI as hypo- and hyperintense areas, respectively. The MRI changes in PRES are typically reversible and improve following resolution of the clinical syndrome (Fig. 2).
Figure 2.
MRI Findings in posterior reversible encephalopathy syndrome (PRES). A 48-year-old liver transplant patient receiving cyclosporine at toxic levels developed a generalized tonic-clonic seizure. Blood pressure was elevated at 160/90. Cyclo-sporine was held for 2 days, then restarted at a lower dose, and judicious lowering of blood pressure with antihypertensive therapy was administered. Levetiracetam, 500 mg twice daily, was initiated. Shortly following his seizure, brain MRI axial FLAIR images showed hyperintense gyriform signal abnormality in the right more than left posterior temporo-parietal and occipital regions (top row) that nearly completely resolved on repeat imaging 2 months later (bottom row). No further seizures occurred, and levetiracetam was tapered and discontinued.
Acute symptomatic seizures occur in 1.5 % to 6 % of CNI-treated patients, often as part of the PRES presentation but sometimes without other apparent clinical or imaging characteristics of PRES [18, 19, 21, 34, 35]. Seizures often present within 4 weeks of CNI initiation [36]. Generalized tonic-clonic seizures or complex partial status epilepticus may occur [21, 35, 37], while periodic lateralized epileptiform discharges (PLEDs) on EEG during PRES may precede the development of chronic epilepsy [38]. CNI neurotoxicity may lead to future development of refractory epilepsy with hippocampal sclerosis [38–40]. While such cases are rare, they underscore the importance of aggressive management of PRES.
Neurotoxicity causing PRES is a major concern when CNI levels are high, but PRES can also occur at normal CNI levels. Transplant patients receiving cyclosporine or tacrolimus should have drug levels measured frequently to avoid evolution of neurotoxicity and seizures. Fortunately, PRES usually resolves within 2 weeks following dose reduction or withdrawal of the CNIs, and sometimes without a change in dosage [18, 22]. If hypertension is concurrent, antihypertensive medication should be administered to slow the progression of edema and accelerate reversal of PRES.
Cerebrovascular disorders
Recent studies have suggested that post-transplant patients whose CNI levels are carefully maintained rarely experience infections or PRES; rather, given the reduction in CNI-induced complications in these patients, cerebrovascular disorders, including acute cerebral infarctions, hemorrhages, and subdural hematomas, are the most common neurological complications within 30 days of transplantation [41]. Because acute symptomatic seizures may complicate cerebrovascular events, prompt brain MRI should be strongly considered in all new-onset seizure cases, especially those having focal neurological examination abnormalities.
Treatment
Pharmacologic treatment
Goals of treatment in transplant patients are similar to those in other patients with seizures and epilepsy. First, seizures must be and controlled and prevented while limiting adverse effects or drug–drug interactions due to AED treatment [42–47], with particular respect to avoidance of adverse impacts on the transplanted organ as well as drug interactions that could lead to altered immunosuppressive drug levels that place the organ at further risk. Second, underlying causes should be promptly recognized and treated to prevent seizure recurrence and further neurological complications.
The approach to treatment of acute repetitive seizures or status epilepticus differs from that of a self-limited, brief seizure. Because most seizures are shorter than 5 min in duration, seizures lasting 5 min or longer should be considered as evolving status epilepticus and terminated promptly using intravenous AEDs (Fig. 1). After controlling seizures, provoking factors such as toxic CNI levels, abnormal electrolyte or glucose levels, hypertension, or infection should be identified and corrected. If the underlying seizure cause can be controlled, the patient should be monitored carefully without commencing AED therapy if seizures are not sustained and do not recur. Brain MRI and EEG results may help direct further management. When investigations are normal and the patient is clinically stable, observation alone can be considered. For recurrent seizures or a single seizure with potentially epileptogenic abnormalities on brain imaging or EEG, AED therapy should be initiated. AEDs can usually be discontinued after 1 to 3 months without significant risk of seizure recurrence [21, 48]. However, patients with potentially epileptogenic brain lesions [49], uncontrollable metabolic imbalances, or those too ill to tolerate recurrent seizures may benefit from continued AED therapy [48].
The primary considerations when selecting an AED for treatment of a SOT patient are efficacy for seizure type and safety and tolerability profile, as in other patient groups. However, given the availability of many newer broad-spectrum AEDs (effective for a broad range of seizure types) with favorable overall tolerability, as well as the complexity of comorbid medical illnesses (including hepatic, renal, and bone marrow dysfunction) and complex pharmacologic regimens including antirejection drugs, pharmacokinetic properties of AEDs supersede most other considerations in transplant patients. Given a dearth of direct evidence basis for choosing between drugs with largely equivalent efficacy and tolerability, factors such as metabolism, excretion, and extent of protein binding that determine the prospect for drug interactions or organ injury become paramount [40].
Limiting potential drug–drug interactions is crucial in SOT patients, so choosing AEDs with minimal protein binding and minimal hepatic enzyme metabolism, induction, or inhibition is particularly desirable (Table 1) [42–47, 50]. Generally, most older and some newer AEDs that have extensive hepatic metabolism, enzyme induction, significant protein binding, and drug–drug interaction potential should be avoided in transplant patients, except for the use of intravenous lorazepam or fosphenytoin for reliable, prompt termination of acute repetitive seizures or status epilepticus. The older AEDs phenytoin, phenobarbital, and carbamazepine all have significant drug interactions with immunosuppressive agents, increasing metabolism of CNIs and corticosteroids via induction of the hepatic cytochrome P450 enzymes [48]. For this reason, valproate was previously recommended for treatment of seizures in renal transplant patients [51, 52] prior to the advent of newer, safer, and more tolerable AEDs. However, in current practice, valproate is highly discouraged in liver transplant patients, as it is primarily hepatically metabolized [53] and has been associated with hepatic failure [53– 56]. Cautious use of intravenous valproate can be considered as an alternative choice for treatment of status epilepticus in hemodynamically unstable patients.
Newer AEDs are typically preferred for their favorable adverse effect profiles and minimal drug interactions [23, 43–47, 50, 51, 57–61] in transplant patients. In addition to better patient tolerability, newer AEDs provide greater flexibility for treating partial and primary generalized seizure types. However, certain newer AEDs are still relatively undesirable for use in transplant patients. Clobazam, felbamate, rufinamide, and tiagabine are generally best avoided for transplant patients given hepatic metabolism or protein binding, which may produce drug interactions or hepatic dysfunction. Lamotrigine, topiramate, and zonisamide may be useful in carefully selected transplant patients with primary generalized and partial-onset seizures, but given hepatic metabolism with each of these drugs, these should be used with caution in liver transplant patients, and topiramate has enzyme-inducing properties at doses of 200 mg and above and could cause undesirable drug–drug interactions. Oxcarbazepine might be useful in some transplant patients with partial-onset seizures, although caution must be exercised to carefully monitor for evolving hyponatremia with this drug. Felbamate and vigabatrin are typically reserved for the most refractory patients who have continued seizures despite previous trials of all other older and newer AEDs, given their association with devastating adverse effects, including the potential for fatal aplastic anemia [62, 63] and hepatotoxicity [63] with felbamate, and a significant risk of irreversible visual field loss with vigabatrin [64]. Ezogabine has extremely limited safety and tolerability data thus far and is thus an undesirable choice for most transplant patients.
While a comprehensive review of AED pharmacology is beyond the scope of this review, details of the clinical pharmacology for four drugs of choice for transplant patients—levetiracetam, gabapentin, pregabalin, and lacosamide—are now provided.
Table 1.
Summary of practical clinical pharmacology of the AEDs of choice for transplant patients
| Drug | Use for | Available formulations |
Standard dosage |
Main drug interactions |
Typical adverse effects |
Cost |
|---|---|---|---|---|---|---|
| Levetiracetam | Partial onset or primary generalized |
Tablet, oral solution, IV |
Oral: 1,000–3,000 mg/d; IV: 1,000- to 3,000-mg loading dose over 15–60 min |
None | Dizziness, drowsiness, irritability |
Expensive (especially IV) |
| Gabapentin | Partial onset | Capsule, tablet, oral solution |
300–3,600 mg/d | None | Dizziness, drowsiness, nausea, ataxia, edema, increased appetite |
Moderate |
| Pregabalin | Partial onset | Capsule | 300–600 mg/d | None | Dizziness, drowsiness, nausea, ataxia, edema, weight increase, euphoria |
Expensive |
| Lacosamide | Partial onset, possibly primary generalized |
Tablet | 200–400 mg/d | None | Dizziness, drowsiness, nausea, ataxia |
Expensive |
| Lorazepam | Acute repetitive seizures or status epilepticus |
IV | IV: begin with 0.1 mg/kg (1- to 2-mg IV push at 2 mg/min), may increase to 8 mg maximum |
Multiple; both enzymatic induction and protein-binding displacement |
Dizziness, drowsiness, nausea, ataxia, rash, hepatic or marrow impairments |
Inexpensive |
| Fosphenytoin | Acute repetitive seizures or status epilepticus |
IV | IV: loading dose of 18 mg/kg at 100–150 mg/min, maintain at 10–20 µg/mL (total PHT levels of 1–2 µg/mL) |
Multiple; both enzymatic induction and protein-binding displacement |
Dizziness, drowsiness, nausea, ataxia, rash, hepatic or marrow impairments |
Moderate |
| Valproic acid | Acute repetitive seizures or status epilepticus |
IV | IV: loading dose of 20 mg/kg at 20–50 mg/min to total loading dose of 1,000–3,000 mg |
Multiple; both enzymatic inhibition and protein-binding displacement |
Dizziness, drowsiness, nausea, ataxia, hepatic or marrow impairments, pancreatitis |
Expensive |
While there are other available formulations for the older AEDs lorazepam and valproate, only intravenous formulations and dosing guidelines are provided to underscore that use of these drugs should be reserved for acute repetitive seizures or status epilepticus in post-transplant settings given undesirable pharmacokinetics for chronic longitudinal administration. Also, while a broader range of adverse effects occurs with each of these drugs, only the most common and distinctive side effects associated with each particular drug are emphasized
AED antiepileptic drug; IV intravenous; PHT phenytoin
Levetiracetam
| Levetiracetam is the drug of choice for post-transplant seizures given its broad-spectrum efficacy across a broad range of seizure types throughout the lifespan, including infants, children, and adults, as well as its rapid and linear oral absorption conferring rapid efficacy; favorable side effect profile; flexible availability as oral tablet, syrup, and intravenous formulations; and lack of significant hepatic metabolism or drug–drug interactions [57, 60, 65, 66]. The CNS target for levetiracetam is binding of intravesicular synaptic vesicle protein 2A, thereby modulating neuro-transmitter release in rapidly discharging neurons [67]. | |
| Standard dosage | Initiate levetiracetam at 1,000 mg/d(2×500-mg tablets once daily) and titrate in increments of 1,000 mg every 1 to 2 weeks up to 3,000 mg/d. Intravenously, 1,000 mg infusions can be given safely and rapidly over 15 to 60 min or faster. |
| Contraindications | Hypersensitivity to levetiracetam. |
| Main drug interactions | No interactions were found between levetiracetam and 11 different drug-metabolizing enzymes [68]. Of particular relevance to transplant patients, levetiracetam does not affect cyclosporine metabolism [58]. |
| Main side effects | Somnolence, asthenia, infection, and dizziness are the most frequent adverse effects. Behavioral abnormalities such as irritability, personality change, and psychosis, and mild leukopenia are possible. The US Food and Drug Administration has issued a class warning for antiepileptic drugs that an increase in suicidal thoughts may occur during treatment, applying generally to every AED [69]. |
| Special points | Patients with impaired renal function may require a dosage decrease, as levetiracetam is excreted largely unchanged and almost entirely renally, and the rate of elimination correlates with creatinine clearance [42–47, 50, 60, 70, 71]. An intravenous formulation has been demonstrated as bioequivalent to the oral formulation, providing another potential option for acute seizure management, preferably for those who are not in convulsive status epilepticus, as the evidence basis for acute seizure termination remains limited and uncontrolled [72, 73]. Ongoing randomized trials of intravenous levetiracetam in status epilepticus may yield more definitive data on efficacy in the near future [74]. |
| Cost | Expensive (especially the intravenous formulation). |
Gabapentin
| Indicated as adjunctive therapy to control partial-onset seizures in patients 3 years of age or older. The presumed mechanism of action for both gabapentin and pregabalin is binding of the alpha-2-delta subunit of presynaptic voltage-gated calcium channels [75]. | |
| Standard dosage | Start with 100 to 300 mg one to three times daily, titrating by 100 to 300 mg increments per dose every week to a goal target between 900 and 3,600 mg/d in three divided doses. One study of hospitalized inpatients with refractory epilepsy demonstrated that initial dosing at 3,600 mg/d was well-tolerated, suggesting that in selected acute seizure situations, rapid oral dosing can be safely achieved [76]. |
| Contraindications | Hypersensitivity to gabapentin. |
| Main drug interactions | None. |
| Main side effects | Dizziness, headache, nausea, fatigue, ataxia, abnormal vision, and somnolence, edema, and increased appetite. |
| Special points | Patients with impaired renal function usually require a lower initial dose and more gradual dosage titration with lower target doses [43–47, 50], as gabapentin is excreted largely unchanged and almost entirely renally, and the rate of elimination correlates with creatinine clearance [71]. |
| Cost | Moderate. |
Pregabalin
| Pregabalin is indicated as adjunctive therapy to control partial-onset seizures in patients 12 years of age or older. | |
| Standard dosage | Initiate 75 mg twice daily, titrating by 75 mg increments per dose every week to a goal target between 300 and 600 mg/d in two divided doses. Pregabalin is also available as an oral solution (20 mg/mL). |
| Contraindications | Hypersensitivity to gabapentin or pregabalin. |
| Main drug interactions | None [75]. |
| Main side effects | Dizziness, headache, nausea, fatigue, ataxia, abnormal vision, somnolence, euphoria, edema, weight gain, and rare angioedema. Two recent reports have suggested that very rare idiosyncratic hepatotoxicity may occur with pregabalin, suggesting caution for use of this drug in patients with hepatic insufficiency [77, 78]. |
| Special points | Patients with impaired renal function may require a dosage decrease, as pregabalin is excreted largely unchanged and almost entirely renally, and the rate of pregabalin elimination correlates with creatinine clearance [43–47, 50] |
| Cost | Expensive. |
Lacosamide
| Lacosamide is indicated as adjunctive therapy for refractory partial-onset seizures in patients 17 years of age or older. Lacosamide is a functionalized amino acid that selectively enhances slow inactivation of voltage-gated sodium channels and interacts with collapsin-response mediator protein-2 [79]. The median percentage reduction in seizure frequency was significantly greater for patients taking 400 or 600 mg lacosamide per day than for those taking the placebo (39 % and 40 % vs 10 % reductions, respectively) [80]. | |
| Standard dosage | Starting with 50 mg twice daily, titrate by increments of 100 mg/wk, up to the therapeutic dosing range of 200 to 400 mg/d, split between morning and evening doses, based on response and tolerability. |
| Contraindications | Hypersensitivity to lacosamide. Classified as a controlled substance (C–V) due to a low potential for euphoria. Use of oral lacosamide solution is heavily cautioned in phenylketonuric patients, as it contains aspartame, a source of phenylalanine. |
| Main drug interactions | No clinically relevant drug–drug interactions are known [81]. |
| Main side effects | Dizziness, headache, nausea, fatigue, ataxia, abnormal vision, vomiting, diplopia, somnolence, and nystagmus. |
| Special points | Available in oral tablets, oral solution, and as intravenous injection. |
| Cost | Expensive. |
Drugs limited to use in acute repetitive seizures or status epilepticus management
| Standard procedure | The treatment algorithm in Fig. 1 demonstrates the approach for treating acute repetitive seizures and status epilepticus. To terminate an acute prolonged seizure of 5 minutes or greater duration, initially administer intravenous lorazepam, 0.1 mg/kg (typically 1–2 mg in adults), with an additional 2 mg/min until seizure cessation or maximum of 8 mg is reached [82–84]. Lorazepam is the initial drug of choice for status given its greater lipid solubility and a longer duration of antiseizure effect than diazepam [83–85]. If seizure activity continues despite lorazepam, intravenous fosphenytoin (an esterified phenytoin prodrug safe for intravenous or intramuscular administration [86–88]) remains the drug of choice for status epilepticus. Infuse fosphenytoin at 100 to 150 mg phenytoin equivalents (PE)/min to a maximum of 18 mg/kg PE. Maintenance doses of 314± 61.2 mg/d support target-free phenytoin levels between 1 and 2 µg/mL (~10–20 µg/mL total phenytoin) [89]. Precautions during intravenous infusion include necessary blood pressure and ECG monitoring. Alternatively, intramuscular fosphenytoin may be administered if there is limited or no intravenous access. Measure levels 2 h following intravenous infusion and 4 h following intramuscular injection. As alternatives, intravenous valproate, 1,000 to 3,000 mg; levetiracetam, 1,000 to 3,000 mg; or lacosamide, 200 to 600 mg, may be considered for patients who are hemodynamically unstable. |
| Contraindications | Hypersensitivity to lorazepam, phenytoin, or other aromatic amine compounds. Relative contraindications include severe hepatic or bone marrow dysfunction, although the priority of prompt convulsive seizure termination by these proven effective drugs trumps other safety concerns in emergency situations. Be prepared to intubate patient to protect airway or mechanically ventilate in status epilepticus. |
| Main drug interactions | Blood levels and therapeutic effect of numerous medications metabolized by hepatic cytochrome P450 may be reduced by lorazepam and fosphenytoin, which are enzymatic inducers, and highly protein bound medications may be displaced by lorazepam or phenytoin, increasing their free unbound fraction. |
| Main side effects/complications | Genital pruritus is a common and transient adverse effect of fosphenytoin. Respiratory depression occurs with higher doses of lorazepam. Rash, including serious hypersensitivity allergic reactions (Stevens-Johnson syndrome, toxic epidermal necrolysis) are possible. Hepatic insufficiency or failure and hematologic dyscrasias, including aplastic anemia, rarely occur. |
| Special points | Increased clearance and earlier peak phenytoin concentrations are seen in patients with active hepatic or renal disease, likely due to decreased plasma protein concentrations and binding in these disease states, resulting in increased unbound fosphenytoin fractions [90]. Phenytoin half-life is decreased in uremia, so doses should be administered every 8 h at minimum [91]. Additionally, phenytoin reduces plasma cyclosporine concentration [85] and can also alter the metabolism of corticosteroids [65]. Management of refractory status epilepticus is beyond the scope of this review, but alternatives for management include infusions of additional 10 mg/kg PE fosphenytoin; midazolam or propofol; ketamine; phenobarbital, pentobarbital, or pentothal; or administration of inhalation anesthetics such as isoflurane [92]. |
| Cost | Inexpensive (lorazepam); moderate (fosphenytoin); expensive (valproate). |
Nonpharmacologic treatments (limited to use in refractory epilepsy management)
While approximately two thirds of individuals with epilepsy have well-controlled seizures, another one third (or ~3 million individuals in the United States) suffer from refractory epilepsy with recurring seizures that resist control by drugs. Refractory seizures can lead to significant impairments in quality of life and societal function, and constitute a risk for major morbidity and mortality [93–95]. Refractory epilepsy may be practically defined as the failure of two or three appropriately selected and administered AED trials [93, 94]. In refractory epilepsy patients, nonpharmacologic options should be considered, including epilepsy surgery, vagus nerve stimulation (VNS), ketogenic or modified Atkins diets, or treatment of comorbid sleep disorders [43–47]. Intensive evaluation at a comprehensive epilepsy center is recommended to determine which option might be best for each individual patient and should include prolonged ictal video-EEG monitoring, as well as volumetric brain MRI and functional neuroimaging procedures, if indicated.
Epilepsy surgery
| Special points | When possible and safe, epilepsy surgery is the most effective option for patients with refractory mesial temporal lobe epilepsy [16]. However, not all patients are suitable candidates, and extratemporal epilepsy surgeries, especially for nonlesional patients, are less robustly successful. |
| Complications | Focal neurological deficits such as visual field loss, language/memory impairment. |
| Cost | Expensive over a short term but highly cost-effective longitudinally. |
Vagal nerve stimulation
| Special points | VNS is effective in reducing seizure frequency in patients with chronic refractory epilepsy, with more than 40 % of implanted patients achieving a 50 % or greater reduction in seizure frequency [97], and often leads to improved quality of life, possibly due to reduction in AED load [43–47, 98, 99]. |
| Complications | Dysphonia, dysphagia, throat pain/dysesthesias. |
| Cost | Expensive over a short term but highly cost-effective longitudinally. |
Modified diet
| Special points | The ketogenic diet is a high-fat, low-carbohydrate diet [98, 99] that may mediate direct anticonvulsant effects via ketone bodies and/or glucose restriction [100, 101]. However, compliance is extremely difficult, especially for adults; thus, other low glycemic diets, such as a modified Atkins diet, have recently been explored as adjunctive epilepsy therapies in children and adults [102–106]. The ketogenic diet is contraindicated in patients with cardiovascular or mitochondrial disease. |
| Complications | Hyperlipidemia (ketogenic diet). |
| Cost | Inexpensive. |
Treatment of comorbid sleep disorders
Acknowledgment
This publication was made possible by grant no. 1 UL1 RR024150 from the National Center for Research Resources (NCRR), a component of the National Institutes of Health (NIH), and the NIH Roadmap for Medical Research. Its contents are solely the responsibility of the author and do not necessarily represent the official view of the NCRR or NIH. Information on NCRR is available at http://www.ncrr.nih.gov. Information on Reengineering the Clinical Research Enterprise can be obtained from http://nihroadmap.nih.gov.
Footnotes
Disclosure
Dr. St. Louis has served as a consultant for Inspire Inc. and received grant support from the Mayo Clinic. Mr. Shepard reported no potential conflicts of interest relevant to this article.
References and Recommended Reading
Papers of particular interest, published recently, have been highlighted as:
• Of importance
•• Of major importance
- 1.Organ Procurement and Transplant Network, US. Based on OPTN data as of February 2012.
- 2. [Accessed February 2012];National Marrow Donor Program® Key Messages, Facts & Figures. Available at http://marrow.org/News/Media/Facts_and_Figures_(PDF).aspx.
- 3.Mor E, Jennings L, Gonwa TA, et al. The impact of operative bleeding on outcome in transplantation of the liver. Surg Gynecol Obstet. 1993;176(3):219–227. [PubMed] [Google Scholar]
- 4.Boylan JF, Klinck JR, Sandler AN, et al. Tranexa-mic acid reduces blood loss, transfusion requirements, and coagulation factor use in primary orthotopic liver transplantation. Anesthesiol. 1996;85(5):1043–1048. doi: 10.1097/00000542-199611000-00012. [DOI] [PubMed] [Google Scholar]
- 5.Karkouti K, Wijeysundera DN, Yau TM, et al. The independent association of massive blood loss with mortality in cardiac surgery. Transfusion. 2004;44(10):1453–1462. doi: 10.1111/j.1537-2995.2004.04144.x. [DOI] [PubMed] [Google Scholar]
- 6.Mannucci PM, Levi M. Prevention and treatment of major blood loss. N Engl J Med. 2007;356(22):2301–2311. doi: 10.1056/NEJMra067742. [DOI] [PubMed] [Google Scholar]
- 7.Fishman JA, Rubin RH. Infection in organ-transplant recipients. N Engl J Med. 1998;338(24):1741–1751. doi: 10.1056/NEJM199806113382407. [DOI] [PubMed] [Google Scholar]
- 8.Conti DJ, Rubin RH. Infection of the central nervous system in organ transplant patients. Neurol Clin. 1988;6(2):241–260. [PubMed] [Google Scholar]
- 9.Hooper DC, Pruitt AA, Rubin RH. Central nervous system infections in the chronically immunosuppressed. Medicine (Baltimore) 1982;61(3):166–188. doi: 10.1097/00005792-198205000-00004. [DOI] [PubMed] [Google Scholar]
- 10.Bonham CA, Dominguez EA, Fukui MB, et al. Central nervous system lesions in liver transplant recipients: prospective assessment of indications for biopsy and implications for management. Transplantation. 1998;66(12):1596–1604. doi: 10.1097/00007890-199812270-00005. [DOI] [PubMed] [Google Scholar]
- 11.Martinez AJ, Puglia J. The neuropathology of liver, heart, and heart-lung transplantation. Transplant Proc. 1988;20(1 Suppl 1):806–809. [PubMed] [Google Scholar]
- 12.Selby R, Ramirez CB, Singh R, et al. Brain abscess in solid organ transplant recipients receiving cyclo-sporine-based immunosuppression. Arch Surg. 1997;132:304–310. doi: 10.1001/archsurg.1997.01430270090019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Marco S, Cecilia F, Patrizia B. Neurologic complications after solid organ transplantation. Transpl Int. 2008;22(3):269–278. doi: 10.1111/j.1432-2277.2008.00780.x. This article provides the most current and comprehensive review of incidence and causes for the range of neurological complications following heart, lung, kidney, liver, pancreas, and intestine transplantations.
- 14.Singh N, Husain S. Infections of the central nervous system in transplant recipients. Transpl Infect Dis. 2000;2:101–111. doi: 10.1034/j.1399-3062.2000.020302.x. [DOI] [PubMed] [Google Scholar]
- 15.Wiesner RH, Demetris AJ, Belle SH. Acute hepatic allograft rejection: incidence, risk factors, and impact on outcome. Hepatology. 1998;28(3):638–645. doi: 10.1002/hep.510280306. [DOI] [PubMed] [Google Scholar]
- 16.Sankaran D, Asderakis A, Ashraf S, et al. Cytokine gene polymorphisms predict acute rejection following renal transplantation. Kidney Int. 1999;56:281–288. doi: 10.1046/j.1523-1755.1999.00536.x. [DOI] [PubMed] [Google Scholar]
- 17.Pascual M, Theruvath T, Kawai T, et al. Strategies to improve long-term outcomes after renal transplantation. N Engl J Med. 2002;346(8):580–590. doi: 10.1056/NEJMra011295. [DOI] [PubMed] [Google Scholar]
- 18.Gijtenbeek JM, van den Bent MJ, Vecht CJ. Cyclo-sporine neurotoxicity: a review. J Neurol. 1999;246(5):339–346. doi: 10.1007/s004150050360. [DOI] [PubMed] [Google Scholar]
- 19.De Groen PC. Cyclosporine: a review and its specific use in liver transplantation. Mayo Clin Proc. 1989;64(6):680–689. doi: 10.1016/s0025-6196(12)65348-8. [DOI] [PubMed] [Google Scholar]
- 20.Teh LK, Dom SH, Zakaria ZA, et al. A systematic review of the adverse effects of tacrolimus in organ transplant patients. Afr J Pharm Pharmacol. 2011;4(6):764–771. [Google Scholar]
- 21.Wijdicks EF, Plevek DJ, Wiesner RH, et al. Causes and outcome of seizures in liver transplant recipients. Neurology. 1996;47:1523–1525. doi: 10.1212/wnl.47.6.1523. [DOI] [PubMed] [Google Scholar]
- 22.Hinchey J, Chaves C, Appignani B, et al. A reversible posterior leukoencephalopathy syndrome. N Engl J Med. 1996;334(8):494–500. doi: 10.1056/NEJM199602223340803. [DOI] [PubMed] [Google Scholar]
- 23.Lacerda G, Krummel T, Sabourdi C, et al. Optimizing therapy of seizures in patients with renal or hepatic dysfunction. Neurology. 2006;67(12 Suppl 4):S28–S33. doi: 10.1212/wnl.67.12_suppl_4.s28. [DOI] [PubMed] [Google Scholar]
- 24.Conti DJ, Rubin RH. Infection of the central nervous system in organ transplant patients. Neurol Clin. 1988;6(2):241–260. [PubMed] [Google Scholar]
- 25.Sarnaik AP, Meert K, Hackbarth R, et al. Management of hyponatremic seizures in children with hypertonic saline: a safe and effective strategy. Crit Care Med. 1991;19:758–762. doi: 10.1097/00003246-199106000-00005. [DOI] [PubMed] [Google Scholar]
- 26.Adrogue HJ, Madias NE. Hyponatremia. N Engl J Med. 2000;342(21):1581–1589. doi: 10.1056/NEJM200005253422107. [DOI] [PubMed] [Google Scholar]
- 27.Thompson CB, June CH, Sullivan KM, et al. Association between cyclosporin neurotoxicity and hypo-magnesaemia. Lancet. 1984;2:1116–1120. doi: 10.1016/s0140-6736(84)91556-3. [DOI] [PubMed] [Google Scholar]
- 28.Nozue T, Kobayashi A, Sako A. Evidence that cyclo-sporine causes both intracellular migration and in-appropriate urinary excretion of magnesium in rats. Transplantation. 1993;55(2):346–349. doi: 10.1097/00007890-199302000-00022. [DOI] [PubMed] [Google Scholar]
- 29.Wells TG, Ulstrom RA, Nevins TE. Hypoglycemia in pediatric renal allograft recipients. J Pediatr. 1988;113(6):1002–1007. doi: 10.1016/s0022-3476(88)80571-7. [DOI] [PubMed] [Google Scholar]
- 30.Bartynski WS, Tan HP, Boardman JF, et al. Posterior reversible encephalopathy syndrome after solid organ transplantation. AJNR Am J Neuroradiol. 2008;29(5):924–930. doi: 10.3174/ajnr.A0960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Bartynski WS. Posterior reversible encephalopathy syndrome, part 1: fundamental imaging and clinical features. Am J Neuroradiol. 2008;29(6):1036–1042. doi: 10.3174/ajnr.A0928. This review provides an excellent basis for the clinical identification of PRES, reviewing imaging patterns, clinical and laboratory markers, associated clinical conditions, histopathology, and cerebral blood flow in PRES.
- 32.Balderramo D, Prieto J, Cardenas A, et al. Hepatic encephalopathy and post-transplant hyponatremia predict early calcineurin inhibitor-induced neurotoxicity after liver transplantation. Transpl Int. 2011;24(8):812–819. doi: 10.1111/j.1432-2277.2011.01280.x. [DOI] [PubMed] [Google Scholar]
- 33.Hinchey J. Reversible posterior leukoencephalopathy syndrome: what have we learned in the last 10 years? Arch Neurol. 2008;65(2):175–176. doi: 10.1001/archneurol.2007.62. [DOI] [PubMed] [Google Scholar]
- 34.O’Sullivan DP. Convulsions associated with cyclosporin A. Br Med J (Clin Res Ed) 1985;290(6471):858. doi: 10.1136/bmj.290.6471.858-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Wijdicks EF. Neurotoxicity of immunosuppressive drugs. Liver Transpl. 2001;7(11):937–942. doi: 10.1053/jlts.2001.27475. [DOI] [PubMed] [Google Scholar]
- 36.Mueller AR, Platz KP, Bechstein WO, et al. Neurotoxicity after orthotopic liver transplantation. A comparison between cyclosporine and FK506. Transplantation. 1994;58(2):155–170. [PubMed] [Google Scholar]
- 37.Appleton RE, Farrell K, Teal P, et al. Complex partial status epilepticus associated with cyclosporin A therapy. J Neurol Neurosurg Psychiatry. 1989;52(9):1068–1071. doi: 10.1136/jnnp.52.9.1068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Skiba V, Etienne M, Miller JA. Development of chronic epilepsy after recurrent episodes of posterior reversible encephalopathy syndrome associated with periodic lateralized discharges. Seizure. 2011;20(1):93–95. doi: 10.1016/j.seizure.2010.10.005. [DOI] [PubMed] [Google Scholar]
- 39.Baldini M, Bartolini B, Gori S, et al. Epilepsy after neuroimaging in a woman with tacrolimus-related posterior reversible encephalopathy syndrome. Epilepsy Behav. 2010;17(4):558–560. doi: 10.1016/j.yebeh.2010.01.014. [DOI] [PubMed] [Google Scholar]
- 40.Gaggero R, Haupt R, Paola Fondelli M, et al. Intractable epilepsy secondary to cyclosporine toxicity in children undergoing allogeneic hematopoietic bone marrow transplantation. J Child Neurol. 2006;21(10):861–866. doi: 10.1177/08830738060210100501. [DOI] [PubMed] [Google Scholar]
- 41.Vizzini G, Asaro M, Miraglia R, et al. Changing picture of central nervous system complications in liver transplant recipients. Liver Transpl. 2011;17(11):1279–1285. doi: 10.1002/lt.22383. [DOI] [PubMed] [Google Scholar]
- 42.Garnett WR, St. Louis EK, Henry TR, et al. Transitional polytherapy: tricks of the trade for monotherapy to monotherapy antiepileptic drug conversions. Curr Neuropharmacol. 2009;7(2):83–95. doi: 10.2174/157015909788848884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.St. Louis EK. Minimizing AED adverse effects: improving quality of life in the interictal state in epilepsy care. Curr Neuropharmacol. 2009;7(2):106–114. doi: 10.2174/157015909788848857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.St. Louis EK. From mono- to polytherapy: antiepileptic drug conversions through the spectrum of epilepsy care. Curr Neuropharmacol. 2009;7(2):75–76. doi: 10.2174/157015909788848910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.St. Louis EK, Rosenfeld WE, Bramley T. Antiepileptic drug monotherapy: the initial approach in epilepsy management. Curr Neuropharmacol. 2009;7(2):77–82. doi: 10.2174/157015909788848866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.St. Louis EK. Truly “rational” polytherapy: maximizing efficacy and minimizing drug interactions, drug load, and adverse effects. Curr Neuropharmacol. 2009;7(2):96–105. doi: 10.2174/157015909788848929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.St Louis EK. The art of managing conversions between antiepileptic drugs: maximizing patient toler-ability and quality of life. Pharmaceuticals (Basel) 2010;3(9):2956–2969. doi: 10.3390/ph3092956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Chabolla DR, Wszolek ZK. Pharmacologic management of seizures in organ transplant. Neurology. 2006;67(12 Suppl 4):S34–S38. doi: 10.1212/wnl.67.12_suppl_4.s34. [DOI] [PubMed] [Google Scholar]
- 49.Grigg MM, Costanzo-Nordin MR, Celesia GG, et al. The etiology of seizures after cardiac transplantation. Transplant Proc. 1988;20(Suppl 3):937–944. [PubMed] [Google Scholar]
- 50.St. Louis EK, Gidal BE, Henry TR, et al. Conversions between monotherapies in epilepsy: expert consensus. Epilepsy Behav. 2007;11(2):222–234. doi: 10.1016/j.yebeh.2007.04.007. [DOI] [PubMed] [Google Scholar]
- 51.Mignat C. Clinically significant drug interactions with new immunosuppressive agents. Drug Saf. 1997;16(4):267–278. doi: 10.2165/00002018-199716040-00004. [DOI] [PubMed] [Google Scholar]
- 52.Hillebrand G, Castro LA, van Scheidt W, et al. Valproate for epilepsy in renal transplant recipients receiving cyclosporine. Transplantation. 1987;43(6):915–916. [PubMed] [Google Scholar]
- 53.Cotarlu D, Zaldman JL. Valproic acid and the liver. Clin Chem. 1988;34(5):890–897. [PubMed] [Google Scholar]
- 54.Scheffner D, König S, Rauterberg-Ruland I, et al. Fatal liver failure in 16 children with valproate therapy. Epilepsia. 1988;29(5):530–542. doi: 10.1111/j.1528-1157.1988.tb03757.x. [DOI] [PubMed] [Google Scholar]
- 55.Le Bihan G, Bourreille J, Sampson M, et al. Fatal hepatic failure and sodium valproate. Lancet. 1980;316(8207):1298–1299. doi: 10.1016/s0140-6736(80)92359-4. [DOI] [PubMed] [Google Scholar]
- 56.Chabrol B, Mansini J, Chretien D, et al. Valproate-induced hepatic failure in a case of cytochrome C oxidase deficiency. Eur J Pediatr. 1994;153(2):133–135. doi: 10.1007/BF01959226. [DOI] [PubMed] [Google Scholar]
- 57.Brodie MJ, Perucca E, Ryvlin P, et al. Comparison of levetiracetam and controlled-release carbamazepine in newly diagnosed epilepsy. Neurology. 2007;68(6):402–408. doi: 10.1212/01.wnl.0000252941.50833.4a. [DOI] [PubMed] [Google Scholar]
- 58.Franzoni E, Sarajlija J, Garone C, et al. No kinetic interaction between levetiracetam and cyclosporine. J Child Neurol. 2007;22(4):440–442. doi: 10.1177/0883073807301920. [DOI] [PubMed] [Google Scholar]
- 59.French JA, Kanner AM, Bautista J, et al. Efficacy and tolerability of the new antiepileptic drugs II: treatment of refractory epilepsy: report of the Therapeutics and Technology Assessment Subcommittee and Quality Standards Subcommittee of the American Academy of Neurology and the American Epilepsy Society. Neurology. 2004;62(8):1261–73. doi: 10.1212/01.wnl.0000123695.22623.32. [DOI] [PubMed] [Google Scholar]
- 60.Patsalos PN. Pharmacokinetic profile of levetirace-tam: toward ideal characteristics. Pharmacol Ther. 2000;85(2):77–85. doi: 10.1016/s0163-7258(99)00052-2. [DOI] [PubMed] [Google Scholar]
- 61.Smith BJ, St. Louis EK, Stern JM, et al. Concerns with AED conversion: comparison of patient and physician perspectives. Curr Neuropharmacol. 2009;7(2):120–124. doi: 10.2174/157015909788848947. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kaufman DW, Kelly JP, Anderson T, et al. An evaluation of case reports of aplastic anemia among individuals using felbamate. Epilepsia. 1997;38(12):1265–1269. doi: 10.1111/j.1528-1157.1997.tb00062.x. [DOI] [PubMed] [Google Scholar]
- 63.Pellock JM, Brodie MJ. Felbamate: 1997 update. Epilepsia. 1997;38(12):1261–1264. doi: 10.1111/j.1528-1157.1997.tb00061.x. [DOI] [PubMed] [Google Scholar]
- 64.Wild JM, Chiron C, Ahn H, et al. Visual field loss in patients with refractory partial epilepsy treated with vigabatrin: final results from an open-label, observational, multicentre study. CNS Drugs. 2009;23(11):965–982. doi: 10.2165/11317650-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 65.Chabolla DR, Harnois DM, Meschia JF. Levetirace-tam monotherapy for liver transplant patients with seizures. Transplant Proc. 2003;35(4):1480–1481. doi: 10.1016/s0041-1345(03)00361-0. [DOI] [PubMed] [Google Scholar]
- 66.Glass GA, Stankiewicz J, Mithoefer A, et al. Levetira-cetam for seizures after liver transplantation. Neurology. 2005;64(6):1084–1085. doi: 10.1212/01.WNL.0000154598.03596.40. [DOI] [PubMed] [Google Scholar]
- 67.Meehan AL, Yang X, McAdams BD, et al. A new mechanism for antiepileptic drug action: vesicular entry may mediate the effects of levetiracetam. J Neurophysiol. 2011;106(3):1227–1239. doi: 10.1152/jn.00279.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Nicolas JM, Collart P, Gerin B, et al. In vitro evaluation of potential drug interactions with levetirace-tam, a new antiepileptic agent. Drug Metab Dispos. 1999;27(2):250–254. [PubMed] [Google Scholar]
- 69. FDA Website.
- 70.Patsalos PN, Perucca E. Clinically important drug interactions in epilepsy: general features and interactions between antiepileptic drugs. Lancet Neurol. 2003;2(6):347–356. doi: 10.1016/s1474-4422(03)00409-5. [DOI] [PubMed] [Google Scholar]
- 71.LaRoche SM. A new look at the second-generation antiepileptic drugs. Neurologist. 2007;13(3):133–139. doi: 10.1097/01.nrl.0000256353.14257.7c. [DOI] [PubMed] [Google Scholar]
- 72.Misra UK, Kalita J, Maurya PK. Levetiracetam versus lorazepam in status epilepticus: a randomized, open labeled pilot study. J Neurol. 2011 doi: 10.1007/s00415-011-6227-2. [Epub ahead of print] [DOI] [PubMed] [Google Scholar]
- 73.Ramael S, De Smedt F, Toublanc N, et al. Single-dose bioavailability of levetiracetam intravenous infusion relative to oral tablets and multiple-dose pharmaco-kinetics and tolerability of levetiracetam intravenous infusion compared with placebo in healthy subjects. Clin Ther. 2006;28(5):734–744. doi: 10.1016/j.clinthera.2006.05.004. [DOI] [PubMed] [Google Scholar]
- 74.Navarro V, Dagron C, Demeret S, et al. A prehospital randomized trial in convulsive status epilepticus. Epilepsia. 2011;52(Suppl 8):48–49. doi: 10.1111/j.1528-1167.2011.03236.x. [DOI] [PubMed] [Google Scholar]
- 75.Ben-Menachem E. Pregabalin pharmacology and its relevance to clinical practice. Epilepsia. 2004;45(Suppl 6):13–18. doi: 10.1111/j.0013-9580.2004.455003.x. [DOI] [PubMed] [Google Scholar]
- 76.Bergey GK, Morris HH, Rosenfeld W, et al. Gaba-pentin monotherapy: I. An 8-day double-blinddose-controlled multicenter study in hospitalized patients with refractory complex partial or secondarily generalized seizures. The US Gabapentin Study Group 88/89. Neurology. 1997;49(3):739–745. doi: 10.1212/wnl.49.3.739. [DOI] [PubMed] [Google Scholar]
- 77.Sendra JM, Junyent TT, Pellicer MJ. Pregabalin-induced hepatotoxicity. Ann Pharmacother. 2011;45(6):e32. doi: 10.1345/aph.1Q032. [DOI] [PubMed] [Google Scholar]
- 78.Doğan S, Ozberk S, Yurci A. Pregabalin-induced hepatotoxicity. Eur J Gastroenterol Hepatol. 2011;23(7):628. doi: 10.1097/MEG.0b013e328346df7a. [DOI] [PubMed] [Google Scholar]
- 79.Harris JA, Murphy JA. Lacosamide: an adjunctive agent for partial-onset seizures and potential therapy for neuropathic pain. Ann Pharmacother. 2009;43(11):1809–1817. doi: 10.1345/aph.1M303. [DOI] [PubMed] [Google Scholar]
- 80.Ben-Menachem E, Biton V, Jatuzis D, et al. Efficacy and safety of oral lacosamide as adjunctive therapy in adults with partial-onset seizures. Epilepsia. 2007;48(7):1308–1317. doi: 10.1111/j.1528-1167.2007.01188.x. [DOI] [PubMed] [Google Scholar]
- 81.Thomas D, Scharfenecker U, Schiltmeyer B, et al. Low potential for drug-drug interaction of lacosamide. Epilepsia. 2006;47(Suppl 4):200. [Google Scholar]
- 82.Bleck TP. Intensive care unit management of patients with status epilepticus. Epilepsia. 2007;48(Suppl 8):59–60. doi: 10.1111/j.1528-1167.2007.01352.x. [DOI] [PubMed] [Google Scholar]
- 83.Leppik IE, Derivan AT, Homan RW, et al. Double-blind study of lorazepam and diazepam in status epilepticus. JAMA. 1983;249(11):1452–1454. [PubMed] [Google Scholar]
- 84.Treiman DM, Meyers PD, Walton NY, et al. A comparison of four treatments for generalized convulsive status epilepticus. N Engl J Med. 1998;339(12):792–798. doi: 10.1056/NEJM199809173391202. [DOI] [PubMed] [Google Scholar]
- 85.D’Souza MJ, Pollock SH, Solomon HM. Cyclospor-ine-phenytoin interaction. Drug Metab Dispos. 1988;16(2):256–258. [PubMed] [Google Scholar]
- 86.Boucher BA, Feler CA, Dean JC, et al. The safety, tolerability, and pharmacokinetics of fosphenytoin after intramuscular and intravenous administration in neurosurgery patients. Pharmacotherapy. 1996;16(4):638–645. [PubMed] [Google Scholar]
- 87.Ramsay RE, Wilder BJ, Uthman BM, et al. Intramuscular fosphenytoin (Cerebyx) in patients requiring a loading dose of phenytoin. Epilepsy Res. 1997;28(3):181–187. doi: 10.1016/s0920-1211(97)00054-5. [DOI] [PubMed] [Google Scholar]
- 88.Wilder BJ, Campbell K, Ramsay RE, et al. Safety and tolerance of multiple doses of intramuscular fos-phenytoin substituted for oral phenytoin in epilepsy or neurosurgery. Arch Neurol. 1996;53(8):764–768. doi: 10.1001/archneur.1996.00550080082016. [DOI] [PubMed] [Google Scholar]
- 89.Morita DA, Glauser TA. Phenytoin and fosphenytoin. In: Wyllie E, editor. The treatment of epilepsy: principles and practice. Philadelphia: Williams & Wilkins; 2005. pp. 785–803. [Google Scholar]
- 90.Aweeka FT, Gottwald MD, Gambertoglio JG, et al. Pharmacokinetics of fosphenytoin in patients with hepatic orrenal disease. Epilepsia. 1999;40(6):777–782. doi: 10.1111/j.1528-1157.1999.tb00778.x. [DOI] [PubMed] [Google Scholar]
- 91.Boggs JG. Seizures in medically complex patients. Epilepsia. 1997;38(Suppl 4):S55–S59. doi: 10.1111/j.1528-1157.1997.tb04540.x. [DOI] [PubMed] [Google Scholar]
- 92.Rossetti AO, Lowenstein DH. Management of refractory status epilepticus in adults: still more questions than answers. Lancet Neurol. 2011;10(10):922–930. doi: 10.1016/S1474-4422(11)70187-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Kwan P, Brodie MJ. Early identification of refractory epilepsy. N Engl J Med. 2000;342(5):314–319. doi: 10.1056/NEJM200002033420503. [DOI] [PubMed] [Google Scholar]
- 94.Kwan P, Sander JW. The natural history of epilepsy: an epidemiological view. J Neurol Neurosurg Psychiatry. 2004;75(10):1376–1381. doi: 10.1136/jnnp.2004.045690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Sillanpää M, Schmidt D. Natural history of treated childhood-onset epilepsy: prospective long term population-based study. Brain. 2006;129(3):617–624. doi: 10.1093/brain/awh726. [DOI] [PubMed] [Google Scholar]
- 96.Wiebe S, Blume WT, Girvin JP, et al. A randomized, controlled trial of surgery for temporal-lobe epilepsy. N Engl J Med. 2001;345(5):311–318. doi: 10.1056/NEJM200108023450501. [DOI] [PubMed] [Google Scholar]
- 97.Morris 3rd GL, Mueller WM. Long-term treatment with vagus nerve stimulation in patients with refractory epilepsy. Neurology. 1999;53(8):1731–1735. doi: 10.1212/wnl.53.8.1731. [DOI] [PubMed] [Google Scholar]
- 98.Cramer J. Exploration of changes in health-related quality of life after 3 months of vagus nerve stimulation. Epilepsy Behav. 2001;2(5):460–465. doi: 10.1006/ebeh.2001.0248. [DOI] [PubMed] [Google Scholar]
- 99.Labar DR. Antiepileptic drug use during the first 12 months of vagus nerve stimulation therapy: a registry study. Neurology. 2002;59(6 Suppl 4):S38–S43. doi: 10.1212/wnl.59.6_suppl_4.s38. [DOI] [PubMed] [Google Scholar]
- 100.Freeman JM, Kossoff EH, Hartman AL. The ketogenic diet: one decade later. Pediatrics. 2007;119(3):535–543. doi: 10.1542/peds.2006-2447. [DOI] [PubMed] [Google Scholar]
- 101.Wilder RM. The effect of ketonemia on the course of epilepsy. Mayo Clin Bull. 1921;2:307–308. [Google Scholar]
- 102.Freeman JM, Vining EP, Pillas DJ, et al. The efficacy of the ketogenic diet—1998: a prospective evaluation of intervention in 150 children. Pediatrics. 1998;102(6):1358–1363. doi: 10.1542/peds.102.6.1358. [DOI] [PubMed] [Google Scholar]
- 103.Hemingway C, Freeman JM, Pillas DJ, et al. The ketogenic diet: a 3- to 6-year follow-up of 150 children enrolled prospectively. Pediatrics. 2001;108(4):898–905. doi: 10.1542/peds.108.4.898. [DOI] [PubMed] [Google Scholar]
- 104.Kossoff EH, McGrogan JR, Bluml RM, et al. A modified atkins diet is effective for the treatment of intractable pediatric epilepsy. Epilepsia. 2006;47(2):421–424. doi: 10.1111/j.1528-1167.2006.00438.x. [DOI] [PubMed] [Google Scholar]
- 105.Kossoff EH, Rowley H, Sinha SR, et al. A prospective study of the modified Atkins diet for intractable epilepsy in adults. Epilepsia. 2008;49(2):316–319. doi: 10.1111/j.1528-1167.2007.01256.x. [DOI] [PubMed] [Google Scholar]
- 106.Sirven J, Whedon B, Caplan D, et al. The ketogenic diet for intractable epilepsy in adults: preliminary results. Epilepsia. 2005;40(12):1721–1726. doi: 10.1111/j.1528-1157.1999.tb01589.x. [DOI] [PubMed] [Google Scholar]
- 107.St Louis EK. Diagnosing and treating co-morbid sleep apnea in neurological disorders; part 1. Pract Neurol (Fort Wash Pa) 2010;9(4):26–30. [PMC free article] [PubMed] [Google Scholar]
- 108.St. Louis EK. Diagnosing and treating co-morbid sleep apnea in neurological disorders; part 2. Pract Neurol (Fort Wash Pa) 2010;9(5):26–31. [PMC free article] [PubMed] [Google Scholar]
- 109.St Louis EK. Sleep and epilepsy: strange bedfellows no more. Minerva Pneumol. 2011;50(3):159–176. [PMC free article] [PubMed] [Google Scholar]