Abstract
Background
In previous studies with male and female rhesus monkeys withdrawal of access to oral phencyclidine (PCP) self administration reduced responding for food under a high fixed-ratio (FR) schedule more in males than females and with a delay discounting (DD) task with saccharin (SACC) as the reinforcer. Impulsive choice for SACC increased during PCP withdrawal more than females.
Objectives
The goal of the present study was to examine the effect of PCP (0.25 or 0.5 mg/ml) withdrawal on impulsive choice for SACC in females during the follicular and luteal phases of the menstrual cycle.
Materials and methods
In Component 1 PCP and water were available from 2 drinking spouts for 1.5 h sessions under concurrent FR 16 schedules. In Component 2 a SACC solution was available for 45 min under a DD schedule. Monkeys had a choice of one immediate SACC delivery (0.6 ml) or 6 delayed SACC deliveries, and the delay was increased by 1 sec after a response on the delayed lever and decreased by 1 sec after a response on the immediate lever. There was then a 10-day water substitution phase, or PCP-withdrawal, that occurred during the mid-folllicular phase (Days 7–11) or the late-luteal (Days 24–28) phase of the menstrual cycle. Access to PCP and concurrent water was then restored, and the PCP withdrawal procedure was repeated over several follicular and luteal menstrual phases.
Results
PCP deliveries were higher during the luteal vs the follicular phase. Impulsive choice was greater during the luteal (vs follicular) phase during withdrawal of the higher PCP concentration.
Conclusions
PCP withdrawal was associated with elevated impulsive choice for SACC, especially in the luteal (vs follicular) phase of the menstrual cycle in female monkeys.
Keywords: Delay discounting, follicular phase, impulsive choice, luteal phase, menstrual cycle, phencyclidine (PCP), rhesus monkeys, saccharin (SACC), withdrawal
Introduction
In a previous study we found when daily oral access to PCP was discontinued, impulsive responding for food on a delay discounting (DD) task increased in both males and females. This increased impulsive choice when PCP access was discontinued was used to infer a withdrawal effect due to physical dependence on PCP that was of greater magnitude in males than females (Carroll et al. 2009b). This is the only study we are aware of showing increased impulsive choice for another substance during drug withdrawal, and it may have important implications for treatment of abstinent human drug abusers. That increased impulsive choice during drug withdrawal differs in males and females (Carroll et al. 2009b) suggests that withdrawal effects may be hormonally related and would vary with during phases of the menstrual cycle. While sex differences and hormonal effects have been well characterized during other phases of drug abuse (see Anker and Carroll 2012), there is little information about these factors in withdrawal effects. There is also little known about sex differences with PCP and other hallucingens/dissociative anesthetics. In the epidemiological data PCP and hallucinogens are often combined, with no specific information about individual drug or whether there are sex differences in the use of these drugs (SAMHSA 2011). The goal of the present study was to compare PCP withdrawal effects on impulsive behavior for a nondrug reinforcer, saccharin (SACC) in female monkeys during the follicular and luteal phases of the menstrual cycle.
Oral PCP has been used in studies of withdrawal with monkeys because it is safely consumed in sufficient quantities with little chance of overdose; thus, withdrawal episodes can be safely repeated over time with no physical signs of distress. The results of withdrawal studies with PCP in monkeys (Carroll 1987, 1989; Carroll and Carmona 1991; Carroll et al. 1994; 2009b, Perry et al. 2006), are consistent with withdrawal studies in rats using other drugs (e.g., Barr and Phillips 1999; Corrigall et al. 1989; LeSage et al. 2006) generally reveal a suppression in behavior. For example, in most of the previous studies in which food-maintained behavior was maintained under high fixed-ratio (FR) schedules responding for food was reduced during drug withdrawal. In contrast, the present research showed consistent increases in responding for SACC under the DD schedule when highly preferred foods were used in a mouse paradigm, opiate withdrawal increased the motivation to eat under a PR schedule (Rouibi and Contarino 2012). Similarly, increased operant behavior for food in rats was reported during early opiate withdrawal (Cooper et al. 2010).
In the present study the drug withdrawal model used is a DD schedule with SACC as the reinforcer revealed that during PCP withdrawal, measures of impulsive choice for SACC significantly increased, more in males than females (Carroll et al. 2009b), while SACC intake was not changed. In that study a reverse condition was also found; food restriction (partial food withdrawal) increased impulsive choice for PCP self-administration under the DD schedule. Both PCP withdrawal and partial food withdrawal produced greater impulsive choice for another reinforcer, suggesting in general, that males are more responsive than females to loss of reinforcement associated with a change in schedule maintained by drug or food reinforcement. The greater withdrawal effect in males vs. females found with the impulsivity measures was similar to that previously reported for PCP withdrawal measured on a baseline of FR responding for food in male and female monkeys (Perry et al. 2006) and in withdrawal studies in which rats were injected with drugs such as ethanol, pentobarbital (Devaud and Chadda 2001), morphine (Cicero et al. 2002; Suzuki et al. 1985), and methaqualone (Suzuki et al. 1988). The present use of an altered operant conditioning baseline as a measure of drug withdrawal effects is consistent with observational measures and further detects a mild form of physical dependence, lacking behavioral signs, that could result in relapse or substitution of another addictive substance.
The main goal of the present study was to extend the finding of increased impulsive choice for SACC during PCP withdrawal in males compared with females (Carroll et al. 2009b) to a comparison of withdrawal effects in females during the follicular vs luteal phase of the menstrual cycle. The follicular phase is characterized by higher estrogen, lower progesterone, while estrogen levels during the luteal phase can occasionally be equivalent to levels seen during the follicular phase, but with higher progesterone. Changes in withdrawal effects during different hormonal conditions may inform us on the optimal phase of the menstrual cycle for quitting drug abuse in women. For example, initial clinical studies on menstrual phase and smoking cessation in women have reported that when behavioral therapy (Allen et al 2008) or behavioral therapy plus bupropion are used, a more favorable outcome occurs during the luteal phase (Mazure et al. 2011). In contrast, Franklin et al. (2007) and Carpenter et al. (2008) found better treatment results with agonist (nicotine replacement) therapy during the follicular vs luteal phase.
Phase of the menstrual cycle influences the subjective effects of drugs in humans and nonhuman primates (Evans and Foltin 2006, 2010; Hudson and Stamp 2011; Lynch and Sofuoglu 2010; Mello 2010). For example, self-report measures for smoked cocaine, such as liking, are greater during the follicular phase when estradiol levels are high than during the luteal phase when progesterone levels are elevated (Evans et al. 2002; Evans and Foltin 2006; Justice and de Wit 1999; Sofuoglu et al. 1999; White et al. 2002, but see Lukas et al 1996; Mendelson et al. 2001; Collins et al. 2007). However, no cycle effects were found with intranasal (Collins et al. 2007; Lukas et al. 1996) or i.v. cocaine (Mendelson et al. 2001). In a recent study smoked cocaine self-administration was compared during the follicular and luteal vs follicular phase when women were given micronized progesterone, and similar amounts of cocaine were self-administered under all 3 conditions (Reed et al. 2011). Mello et al (2007) reported increased progressive-ratio responding for cocaine during the follicular vs late luteal phase in cynomologous monkeys. In contrast, we found a small increase in orally self-administered PCP during the luteal phase (compared with the follicular phase) in female rhesus monkeys (Newman et al. 2006). These differences in drug intake by phase could be due to effects of cocaine vs PCP.
Thus, another goal of the present research was to compare oral PCP self-administration (during baseline conditions) in female monkeys during the follicular and luteal phases. There have been very few studies of drug self-administration and drug withdrawal as a function of menstrual cycle phase in nonhuman primates. As in the previous study (Carroll et al. 2009b) PCP self-administration and withdrawal effects were evaluated in one component of the daily session, while impulsive choice on a delay discounting task for SACC (0.3% w/v) served as the measure of PCP withdrawal effects. We used two concentrations of PCP because previous results indicate more pronounced withdrawal effects that emerge at higher PCP concentrations or self-administered doses (Carroll et al. 2009b), but sex (Becker and Hu 2008; Lynch et al. 2002), and hormonal (Newman et al. 2006) differences are more likely to emerge at lower drug doses.
Material and methods
Animals
Seven adult female rhesus monkeys (Macacca mulatta) were used in a within-subjects experiment. The monkeys were previously trained to self-administer orally-delivered PCP under fixed ratio (FR) progressive-ratio (PR), and DD schedules (Carroll et al. 2009b). They were maintained at 85–90% of their free-feeding body weights that ranged from 5.5 to 10.0 kg, and they had unlimited access to water except during the time out periods 2 h prior to and 1.5 h after each daily session.
Monkeys were housed in individual home cages in quad formation (2-story, 2 side-by-side cages) in a room containing 12 monkeys. Each monkey’s cage had an operant conditioning work panel mounted on a side wall that was used to record responses and dispense PCP, water, SACC and food. The monkeys were housed in a room that was temperature- (23°C) and humidity-controlled with the lights on from 0600 to 1800 h. Enrichment devices such as plastic toys, movies, and fruit and vegetables were available during the intersession period. The experiment was conducted in accordance with the Principles of Laboratory Animal Care (National Research Council 2011) and approved by the University of Minnesota Institutional Care and Use Committee under Protocol Number 1007A85493. Laboratory facilities were accredited by the American Association for the Assessment and Accreditation of Laboratory Animal Care.
Apparatus
The quad cages were used for both housing and testing were 83 cm wide, 76 cm high, and 100 cm deep (Lab Products, Maywood, NJ, USA). The operant conditioning panel mounted to the side of each cage contained 2 brass drinking spouts, 2 green stimulus lights above the drinking spouts, and a center red stimulus light indicated the start of a delay-discounting trial. The green light was steadily illuminated when water or saccharin were available, and it blinked when a drug solution was available above the drinking spouts. These devices were mounted on a panel that was attached to the outside of the cage with circular cut-outs in the cage wall allowing the lights and drinking spouts to protrude through the cage wall. The drinking devices were mounted within a circular clear Plexiglas plate that allowed the monkeys to see 4 stimulus lights mounted behind the plate. Two small white lights were illuminated when a lip contact response was made for water or SACC, and 2 small green lights indicated a lip contact for PCP. Med-PC software (Med-PC for Windows) and interfaces (Med Associates, St. Albans, VT, USA), located in an adjacent room were used to control the experiment and record data.
Procedure
The experimental design and procedures were similar to those reported by Newman et al. (2006, 2008) and Carroll et al. (2009b). There were 2 components in each day: Component 1- PCP self-administration under FR 16, and Component 2 - a DD schedule for SACC deliveries contingent on FR 8 lip-contact responses. This sequence was conducted 7 days a week as described below (See Table 1).
Table 1.
Timetable of daily procedures
| 8:00–9:45 | Time out, no food or liquid |
| 9:45–11:45 | Component 1 drug and water concurrent FR 16 schedules |
| 11:45–12:15 | Time out, no food or liquid |
| 12:15–13:00 | Component 2 SACC delay discounting (45 min) |
| FR 8 for each SACC delivery (immediate or delayed) | |
| 13:00–14:30 | Time out, food, no liquid |
| 14:30–8:00 | Intersession, only water available FR 1 |
Experimental design
There were 4 conditions that were compared under PCP self-administration and withdrawal phases. 1) Water baseline (Wat-BL), 2) PCP available during luteal phase (PCP-L) or follicular phase (PCP-F), 3) The drug-available period immediately preceding PCP withdrawal (PCP Pre-WD), and 4) water substitution or withdrawal (Wat-WD) by substituting water for PCP. Menstrual cycle days 7–11 were used for the mid-follicular phase, and Days 24–28 for the late-luteal. The first part of the hyphenated abbreviation refers to liquid consumed during session – and the second is the menstrual phase (follicular or luteal) or the operational procedure, such as withdrawing PCP and replacing it with water (WD) or obtaining baseline measures with water available in both drinking spouts (Wat). Thus, PCP Pre-WD occurred during the early-follicular (Days 2–6) or mid-luteal (Days 19–23) phase. Condition 1, Wat-BL, was obtained once during the follicular and luteal phases, at a pre-experimental time when no drug withdrawal conditions had been tested for several months. The Wat-BL data were collected after several weeks of water access. The monkeys were stabilized on PCP, then water was substituted for several weeks to obtain a baseline for DD SACC without the influence of PCP or PCP withdrawal. The other 3 conditions (2–4) were repeated in order 2) PCP-F or PCP-L, 3) PCP Pre-WD, and 4) Wat-WD over several menstrual cycles to assess variability across repeated withdrawal episodes. Conditions 2–4 were not randomized because that is the sequence necessary to produce withdrawal effects and to study PCP intake during follicular and luteal phases. However, the 0.25 and 0.5 mg/ml PCP concentrations were tested in random order. The 3 repeated conditions were tested during the follicular and luteal menstrual cycle phases which were sampled in nonsystematic order. As Table 3 indicates, only 1 menstrual phase could be studied each month, and the entire procedure to test one menstrual cycle could last from over a month to up to 2 months.
Table 3.
Experimental design for PCP withdrawal
PCP and water availability for self-administration during Component 1 (9:45 am – 11:45 pm)
| Baseline 10 days | Withdrawal 10 days | Recovery 14 – 27 days | |||
|---|---|---|---|---|---|
| Spout 1 | Spout 2 | Spout 1 | Spout 2 | Spout 1 | Spout 2 |
| PCP FR 16.25, .5 mg/ml | H2O FR 16 | H2O FR 16 | H2O FR 16 | PCP FR 16.25, .5 mg/ml | H2O FR 16 |
Daily procedures
Component 1: PCP self-administration component
Monkeys were trained to self-administer PCP (0.25 mg/ml) and water under concurrent FR 16 schedules during daily 2-h sessions from 0945 to 1145 h (see Table 1), 7 days/week. Completion of the FR requirements for liquid deliveries on each of the 2 drinking spouts were independent of each other. Lip contact responses were counted on the two spouts, and when the FR was completed on a spout, liquid was delivered at that spout. PCP and water were available on alternating sides, each under a FR 16 schedule, with corresponding stimulus lights (PCP – flashing green, water – solid green) to signal the available liquid. All monkeys had previous experience with this schedule (Carroll et al.. 2009b; Newman et al. 2006; Perry et al. 2006). This component was followed by a 30-min (1145–1215 h) when no food or liquid was available.
Component 2: Delay discounting saccharin component
This procedure has previously been described in detail (Carroll et al. 2009b), and the DD component occurred from 1215 to 1300 h (Table 1). Initially, there were 2 forced-choice trials to ensure sampling on the right and left side, and those were followed by 45 choice trials for up to 45 min. A SACC solution was available from both drinking spouts, small-immediate on one spout vs large-delayed on the other unitl 45 choice trials were completed or 45 min elapsed. Lip contacts on the drinking spouts, with a solid green light above each spout, were reinforced by a 0.6 ml delivery of SACC. On the “immediate” spout one delivery was contingent upon the completion of a FR 8 requirement for lip contacts and an additional FR 1 for 1 delivery of SACC. On the delayed side, after completion of FR 8, and then a FR 1 for each of 6 SACC deliveries were available. Following the SACC delivery on either spout the lights above the spouts were extinguished, and the center red light was illuminated to signal the next choice trial.
There was never a delay for the SACC delivery on the immediate spout; however, on the first day the first delivery on the delayed spout started at 10 sec. Subsequently, during the 45 choice trials each response on the delayed spout increased the delay by 1 sec, and each response on the immediate spout decreased the delay on the delayed spout by 1 sec. Thus, a delay was calculated after each SACC delivery for all 45 trials and averaged to yield a mean adjusted delay (MAD). On subsequent days the delay started where it left off on the previous day. Sides were reversed daily for the delayed and immediate spouts. The self-adjusting delay procedure has been used previously with rats (See review by Perry and Carroll 2008; Carroll et al. 2008, 2010) and monkeys (Carroll et al. 2009b, 2010), and MAD values have been used to indicate low (high MAD) or high (low MAD) impulsive choice for a substance.
PCP withdrawal conditions
Behavior was allowed to stabilize for at least 10 sessions (days) with PCP and water available during Component 1 (see Table 3). Subsequently PCP was replaced with water for 10 days. The 10-day PCP withdrawal period was based on previous findings that detected differences in PCP intake between follicular and luteal phases (Newman et al. 2006). The 10-day PCP withdrawal period was timed to occur during the mid-follicular (Days 7–11) or the late luteal phase (Days 24–28) of the menstrual cycle, allowing at least another 7 days back on PCP before another 10-day withdrawal period was implemented. Subsequently, the concurrent PCP and water condition was reinstated for 14–27 days until behavior stabilized (see Table 3).
Menstrual cycle phase determination
Menstrual cycle phase was determined by observation of menses onset and confirmation by examination of vaginal cytology. Monkeys were checked for bleeding each day, and vaginal swabbing was performed 1 h before the session 3 times per week. The method of Mauro et al. (1970) was used to classify cytology of the vaginal epithelium. Mensis phase was defined by a predominance of parabasal and anucleated/cornified cell types. The follicular phase was described by superficial, intermediate, and anucleated/cornified cell types, and the luteal phase was indicated by the presence of parabasal, superficial, and intermediate and anucleated/cornified epithelial cells. Blood samples were not taken to obtain estradiol and progesterone levels because it would involve anesthetizing the animal and could result in behavioral disruptions the next day during a hormonal phase (follicular or luteal). We used bleeding to document the onset of menses, and swabbed the animals at least 5–6 days a week for vaginal cytology to confirm menstrual cycle status.
Drugs
PCP (phencyclidine HCl) was obtained from the National Institute on Drug Abuse (Research Triangle Institute, Research Triangle Park, NC). The PCP solutions were prepared with tap water, stored in a locked refrigerator, and placed in the reservoirs each day at room temperature.
Data Analysis
Within each PCP concentration 4 experimental conditions were compared: 1) baseline water (Water-BL) deliveries on both spouts during a pre-experimental extended period, 2) a baseline of PCP access that occurred during the follicular (PCP-F) or luteal phase (PCP-L) of the menstrual cycle, 3) a pre-PCP withdrawal (PCP Pre-WD) condition immediately before PCP withdrawal,, and 4) the PCP withdrawal period (Water-WD) when water was substituted for PCP. These 4 conditions were compared in a 2-factor, repeated measures ANOVA with experimental phase and menstrual phase as the factors. Separate ANOVA were conducted for the 0.25 and 0.5 mg/ml PCP concentrations. Dependent measures such as PCP and water deliveries, mg/kg PCP intake, SACC deliveries, and the impulsive choice measure, mean adjusted delay (MAD), were compared within subjects for both the 0.25 and 0.5 mg/ml PCP concentrations, follicular and luteal phases, and across 4 repeated measures conditions. Data for each monkey consisted of a mean of the 4 days during the mid-follicular or late luteal phase when PCP was available or when PCP had been replaced with water for a 10-day withdrawal period. Means for the 4 days of each cycle were used for data analysis and presentation, as there were no increasing or decreasing trends in the daily patterns of intake over the 4 days. These data were then averaged over the 5–7 withdrawal periods and PCP self-administration periods that corresponded with each hormonal phase. Additional comparisons were made on liquid deliveries (PCP or water) during Component 1, and SACC deliveries and MAD scores during Component 2 across the 0.25 and 0.5 mg/ml concentrations during the PCP-F and PCP-L conditions. The mean data for each condition for the 7 monkeys were analyzed with repeated-measures analyses of variance (ANOVA; GB-Stat Dynamic Microsystems, Inc., Silver Spring, MD, USA). Additional two-condition comparisons were made using Fischers Least Significant Differences, Bonferroni-correlated t-tests. An overall significance level of p<0.05 or p <0.05 x number of comparisons for multiple comparisons, was observed.
Results
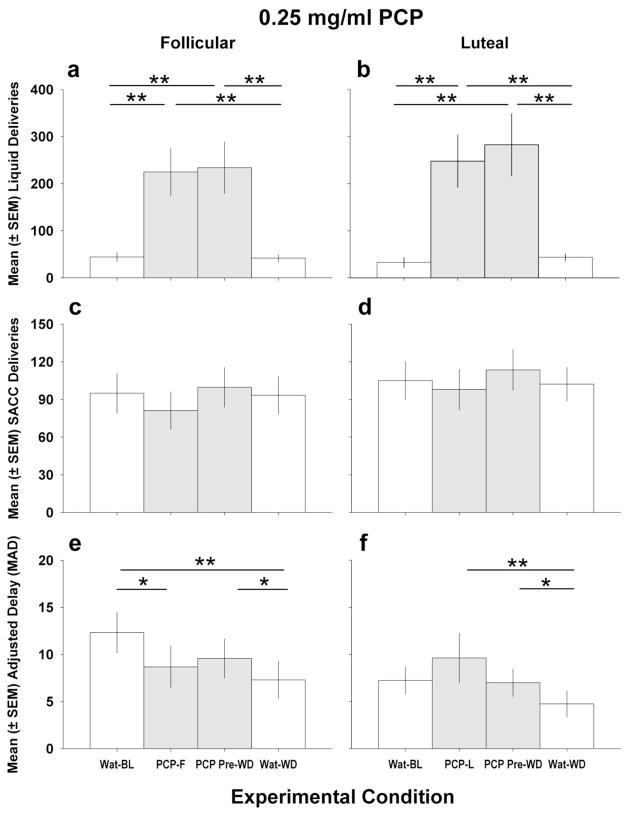
Figure 1 summarizes the results of withdrawing access to 0.25 mg/ml PCP during the follicular and luteal phases. The results (Fig 1a,b) show that PCP was consumed in excess of water during both the follicular and luteal phases (F 3,55 = 18.8802, p < 0.001), indicating that PCP was functioning as a reinforcer. Post hoc tests for both follicular and luteal phases indicated significant differences between Wat-BL and PCP-F, PCP-L, and PCP Pre-WD conditions (p<0.01). The upper frames (Fig 1a,b) show the water and PCP liquid deliveries that were measured in the FR self-administration Component 1 over 4 experimental conditions: 1) water baseline (Wat-BL), 2) PCP availability during the follicular or luteal phases (PCP-F, PCP-L), 3) PCP intake immediately prior to the withdrawal phase (PCP Pre-WD), and 4) when PCP had been withdrawn and water was substituted for the drug (Wat-WD). Note that only the last 2 conditions (PCP-Pre-WD and Wat-WD) were conducted sequentially in time, the first 2 conditions were obtained as baseline measures once during the follicular and luteal phases before the experiment began (Wat-BL), or during several follicular or luteal phases (PCP-F, PCP-L) when PCP withdrawal was not taking place during that particular menstrual cycle phase.
Fig 1.
Mean (±SEM) values are presented for seven female monkeys during the follicular phase (left frames) or luteal phase (right frames) of their menstrual cycle during four experimental conditions: water baseline (Wat –BL) with access only to water for an extended period, PCP access during the follicular (F) or luteal (L) phase) (PCP-F or PCP-L), PCP (0.25 mg/ml) pre-withdrawal (PCP Pre-WD) immediately prior to withdrawal, and during withdrawal when water was substituted for PCP (wat-WD). The first term in the x-axis labels refers to the substance that was available for oral consumption during sessions, and the second term indicates baseline (BL) during the follicular (F) or luteal (L) phase or withdrawal (WD) of PCP. Frames a and b represent water or PCP deliveries earned under component 1 during the follicular and luteal phase of the menstrual cycle, respectively, when liquids were available under independent concurrent FR 8 schedules for 2 h. Frames c and d indicate the number of SACC deliveries, during the follicular (F) or luteal (L) phase, earned during component 2 under a delay-discounting task whereby the monkeys could earn one SACC delivery immediately under FR 8 or 6 deliveries after a delay, with the 1 or 6 deliveries each under an FR 8 schedule. Frames e and f depict the mean adjusted delay (MAD) calculated from the 45 choice trials under DD schedule during the follicular (F) or luteal (L) phase of the menstrual cycle, respectively. Significant differences across experimental conditions are indicated by * (p < 0.05) and ** (p < 0.01), and significant differences between the follicular and luteal phases are indicated by # (p < 0.05), ## (p < 0.01).
Data shown in Fig 1c,d represent SACC deliveries earned during the DD Component 2. The SACC deliveries were not significantly different across the experimental conditions, indicating that changes in PCP or water intake during the preceding Component 1 (Fig 1a, b) did not affect SACC intake during the subsequent Component 2, nor did the availability of water or PCP during the 4 experimental conditions shown on the x-axis. Fig 1e,f shows the impulsive choice measure for SACC across the 4 experimental conditions.
There was a main effect of experimental condition (F3, 55 = 12.6672, p < .001), and post-hoc tests indicated that MADs were significantly lower during PCP withdrawal (Wat-WD) than they were during the PCP-available condition immediately preceding withdrawal (PCP Pre-WD) for both the follicular (Fig 1e) and luteal (Fig 1f) phase indicating greater impulsive choice for SACC during PCP withdrawal. The MAD values are an average of their self-adjusted delays over the 45 choice trials per session were lower during water substitution or PCP withdrawal (Wat-WD) indicating higher impulsive choice for SACC (Fig 1e, f) compared with the PCP-available condition immediately preceding PCP withdrawal (PCP Pre-WD) during both the follicular (1e) and luteal (1f) phases.
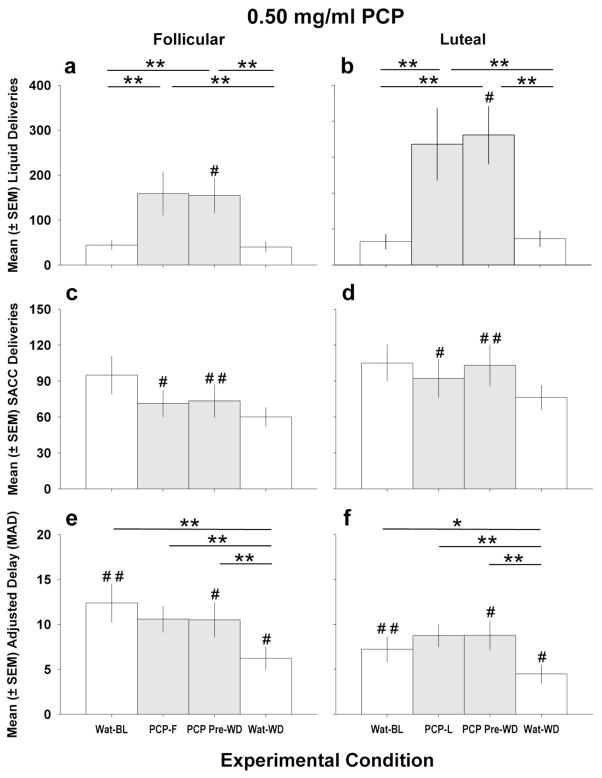
Fig 2 shows similar mean (±SEM) data for the 4 experimental conditions when 0.5 mgml PCP was available during phases shown on the x-axis label: 1) PCP-F and PCP-L phases, 3) PCP Pre-WD, and 4) Wat-WD. The results for 0.5 mg/ml PCP (Fig 2a, b) indicate that PCP deliveries exceeded those for water during the Component 1. In addition, PCP intake was higher during the luteal (Fig 2b) vs follicular phase (Fig 2a) in all 7 animals; however, this effect was significant only under the PCP access condition immediately prior to withdrawal (PCP Pre-WD) (p<0.05) when the monkeys would have been in their mid-luteal phase. Statistical analyses indicated a main effect of experimental condition (F 3, 55 = 5.8035, p<0.05), and menstrual phase (F1, 55 = 5.3033, p<0.05) on on PCP deliveries. Post-hoc tests indicated that PCP was functioning as a reinforcer compared to the water baselines (Fig 2a, b), as there were significant differences between the water baseline - Wat-BL (F and L) and PCP-available (PCP-F and PCP-L) conditions, respectively (Ps < 0.01), as well as when water was substituted for PCP (PCP Pre-WD vs Wat-WD).
Fig 2.
Fig 2 is the same as described for Fig 1 except the PCP concentration was 0.5 mg/ml. All other aspects of Fig 2 are as noted in Fig. 1.
The SACC deliveries during Component 2 in the SACC available, follicular and luteal DD components (Fig 2c, d) did not differ significantly across experimental conditions within a menstrual phase, but SACC intake was higher in the luteal phase than the follicular phase (F 1, 55 = 15.2147, p < 0.001) during PCP-L vs PCP-F conditions and during the PCP Pre-WD condition immediately prior to PCP withdrawal (Ps < 0.05 and 0.01), respectively. Thus, both PCP (Fig 2b) and SACC (Fig 2d) intake increased during the luteal compared with the follicular phase.
The mean adjusted delay (MAD) values obtained in Component 2 and shown in Fig 2e and f, which serve as measures of impulsive choice, showed a similar pattern for withdrawal of 0.5 mg/ml PCP as previously reported for 0.5 mg/ml PCP (Fig 1). However, there were also menstrual cycle phase differences in MADs with 0.25 mg/ml PCP (Fig 2e, f). The MAD values (Fig 2e, f) for the SACC-reinforced DD task significantly varied by experimental condition (F 3, 55 = 2.9906, p < 0.05), and there was an experimental condition x menstrual phase interaction (F 3,55 = 10.6554, p < 0.05). Lower MAD values (sec), indicating greater impulsive choice, during both follicular and luteal were found when comparing Wat-WD vs PCP Pre-WD, and PCP-F or PCP-L baselines (p<0.01) and the Wat-BL water baselines in both follicular (p < 0.05) and luteal (p < 0.05, p < 0.01) phases (Fig 2e, f). The effect size of the difference in the impulsive choice measure during pre-withdrawal (PCP Pre-WD) vs withdrawal (Wat-WD) was 0.77 in the follicular phase and 0.51 in the luteal phase.
Table 4 shows that when PCP intake (mg/kg) was compared between the 0.25 mg/ml and 0.5 mg/ml concentrations, intake was significantly higher when 0.5 mg/ml (vs 0.25 mg/ml) PCP was available (F 1,27 = 11.2767, p < 0.01), and post hoc tests of these comparisons revealed significant concentration differences under the PCP-F and PCP Pre-WD luteal conditions (ps < 0.05). However, Table 4 also shows that when adjusted for body weight the amount consumed during the PCP-F and PCP-L baselines or the PCP Pre-WD conditions, immediately before the F or L withdrawal periods, was not significantly different. This may have been due to small changes in body weight that occur over the menstrual cycle. In the present study body weights were obtained monthly and not synchronized with individual monkeys’ menstrual cycles. Overall, the PCP concentration comparison indicates greater sensitivity of menstrual cycle phases (luteal > follicular) for PCP and SACC intake at 0.5 mg/ml PCP, and the withdrawal–induced increases in impulsive choice for SACC were also greater at the 0.5 (vs 0.25 mg/ml) concentration.
Table 4.
Mean (±SEM) PCP intake adjusted for body weight (mg/kg) during PCP-F, PCP-L phases, and PCP Pre-WD baseline conditions before follicular (F – WD) or luteal (L – WD) phase PCP withdrawal and in a previous study of PCP self-administration using 3-h sessions (Newman et al. 2006) with 0.25 and 0.5 mg/ml PCP concentrations
| PCP concentration | Menstrual phase | PCP Pre-WD baseline | Newman et al. 2006 | |||
|---|---|---|---|---|---|---|
| (mg/ml) | PCP–F | PCP–L | Pre-F-WD | Pre-L–WD | F | L |
| 0.25 | 4.4 * | 4.8 | 4.5 | 5.5 * | 4.0* | 4.8* |
| 0.5 | 6.3 * | 6.7 | 6.1 | 7.2 * | 6.0* | 6.6* |
= 0.5 > 0.25 mg/kg intake (p < 0.05)
Discussion
The results indicate that in female monkeys PCP withdrawal effects expressed as increased impulsive choice for SACC, are greater during the luteal (vs follicular) phase of the menstrual cycle; however, this may be a function of the higher PCP intake (mg/kg) in the luteal (vs follicular) phase. Greater withdrawal effects during the luteal phase is consistent with previous results showing enhanced withdrawal effects in males vs females using impulsive choice (Carroll et al. 2009) and other measures (Perry et al. 2006)l. Impulsive choice for SACC was greater when monkeys were maintained and withdrawn from the larger PCP concentration (0.5 mg/ml) compared with the 0.25 concentration in the present and previous (Carroll et al. 2009b) studies. Thus, higher baseline drug intakes are more likely than lower intakes to reveal withdrawal effects on an impulsive choice measure for SACC. The current results were consistent with those reported when both female and male monkeys tested in a similar procedure at the 0.5 mg/ml PCP concentration (Carroll et al. 2009b). In that study effect sizes were 1.5 in females and 1.6 in males compared with females in the current study with 0.77 in the follicular phase and 0.5 in the luteal phase. However, in the previous study with males and females.
The present and previous (Carroll et al. 2009b) results concur on the finding of increased impulsive choice for one substance during withdrawal of another substance and continue to emphasize that increased impulsive choice for a nondrug reinforcer is another dimension of drug withdrawal effects. In the present study impulsive choice for SACC was greater during Wat-WD compared with the pre-experimental water baseline (Wat-BL) condition (Fig 1e, 2 e, f) suggesting that increased impulsive choice reflects a withdrawal effect than a difference in water vs PCP access in Component 1 on impulsive choice for SACC in Component 2. That lowered MADs (higher impulsive choice) for SACC occurred during Wat-WD vs Wat-BL suggests that reduction in MAD is an acute response to PCP withdrawal.
While previous withdrawal effects were expressed as a decrease in behavior maintained by a high FR schedule for lab chow (e.g., Perry et al. 2006), the present and recent (Carroll et al. 2009b) finding of increased impulsive choice is consistent with animal work demonstrating increases in other behaviors such as intake of preferred foods (Rouibi and Contarino 2010; Cooper et al. 2010) or physical exercise (Miladi-Gorgi et al. 2011) during opiate withdrawal. A similar finding was reported by Giordano et al. (2002) in which cessation of one form of drug abuse led to impulsive behavior for another drug or excessive ingestion of preferred foods. However, opposite effects (increased anhedonia in a rodent model) were reported during withdrawal from chronic amphetamine exposure (Der-Avakian and Markou 2010). Studies of responding for natural rewards in drug-dependent, withdrawn human subjects show mixed results; reduced food intake in heroin dependent individuals (Lubman et al. 2009; Zijlstra et al. 2009), and increased craving or preference for liquid or solid sweets in opioid-dependent individuals (Morabia et al. 1989; Weiss 1982). Further work is needed to fully characterize this potential interchangeability between drug- seeking and impulsivity for nondrug reinforcers using parallel preclinical and clinical models. PCP withdrawal may have been altering behavioral economic systems (e.g., substitution of nondrug for drug reinforcers) that are associated with impulsivity for nondrug reinforcers. In future studies it would be useful to extend the withdrawal model to examine nondrug reinforcers that are more healthy than SACC or other sweet substances, such as physical exercise (e.g., Miladi-Gorgi et al. 2011). The findings from this animal work also suggest that humans encountering mild withdrawal effects leading to continued drug use, and impulsivity to resume drug use could be redirected to increase healthy behaviors.
One feature that is consistent across animal studies of withdrawal-induced suppression of operant conditioning behavioral baselines is that there was no observable evidence that mild PCP withdrawal produced malaise that may have been reflected by inactivity or anxiety/stress that might have been indicated by threats, or retreat that could explain the decreased responding under the high FR schedule for food (e.g., Perry et al. 2006). In the present study with visual observation it was impossible to distinguish the withdrawal phase from the drug-available phase. Furthermore, in previous PCP withdrawal studies using high FR schedules when monkeys were offered food from the experimenter or changed to a lower FR schedule for food, they readily consumed the food, while continuing to show a suppression in food-maintained responding at high FR schedules (Carroll and Carmona 1991). In the current study there was no change in daily food and water intake, body weight, or activity level during the baseline water (Wat-BL) or 3 repeated experimental phases. Thus, PCP withdrawal was not expressed as a physiological withdrawal syndrome. The present results are also consistent with previous findings that indicated elevated PCP self-administration during the late luteal phase compared with early or mid-follicular phase of the menstrual cycle (Newman et al. 2006). In the present study PCP deliveries were significantly higher during the luteal PCP Pre-WD baseline than during the Pre-WD baseline in the follicular phase at 0.5 mg/ml PCP (Fig 2c, d). However, PCP intakes (mg/kg) during the follicular and luteal phases were consistently but not significantly higher during the luteal vs follicular phase (Table 4). Similar PCP intakes were also reported in a previous PCP self-administration study in female monkeys using 0.25 mg/ml PCP (4.7 mg/kg) and 0.5 mg/ml PCP (9.2 mg/kg) where menstrual cycles were allowed to vary randomly (Perry et al. 2006) and to those reported for 0.25 and 0.5 mg/ml PCP in female monkeys during follicular and luteal phases (Newman et al. 2006).
The elevation in PCP intake during the luteal phase is in contrast to reports that the proestrus phase (higher estrogen) is related to elevated cocaine self-administration in rats (Anker and Carroll 2010, Carroll et al. 2010) and the follicular phase (higher estrogen) in monkeys (Mello et al. 2011). Exogenously-administered estrogen is also associated with increased drug intake and positive effect of stimulant drugs seen in female vs male rats (Anker and Carroll 2010, 2011), but there is less known about the effect of ovarian hormones on self-administration of a variety of drug classes in nonhuman primates. There is also little known about the role of estrogen and progesterone on impulsivity for a drug reinforcer in rats or nonhuman primates. A recent study in female rats indicated that estradiol increased impulsivity for food in an effort-discounting task in rats to a greater extent in the ovariectomzed (OVX) group compared with the pre-OVX or sham controls (Uban et al. 2012). The effect sizes (2–4) were larger than in the present monkey study, but it concurred with the present results that impulsivity for a nondrug reinforcer is subject to hormonal influences (Uban et al. 2012). Hormonal status, impulsivity, type of drug and pharmacological class of the self-administered drug are important factors to consider for treatment of drug abuse.
In humans fluctuations of estrogen and progesterone correspond with several differences in other physiological and subjective effects of stimulant drugs, particularly stimulants (Evans 2007; Terner and de Wit 2006). However, self-administration of other drugs of abuse such as alcohol (Freitag and Adesso 1993; Hay et al. 1984; Holdstock and de Wit 2000; Nyberg et al. 2004; Sutker et al. 1987), nicotine (Allen et al. 1999, 2004; Snively et al. 2000), marijuana (Lex et al. 1984), and opioids (e.g., Gear et al. 1996) does not vary as consistently with phase of the menstrual cycle in humans as does self administration of stimulants such as cocaine and methamphetamine (see review by Anker and Carroll 2011). Other drug classes may not have been studied extensively enough for a clear pattern of results to emerge, or they are not responsive to hormonal fluctuations over the menstrual cycle.
The present results focused on an impulsive-choice measure to assess drug withdrawal effects; however, varied reactivity to stress and craving during different menstrual cycle phases also varies consistently with stimulant abuse and withdrawal effects in humans, and overlap may exist between stress and impulsivity measures (Sinha 2008, Sinha et al. 2007). For example, the follicular phase women showed higher systolic/diastolic blood pressure measures, possibly related to stress, as they scored higher on self-report measures of anxiety and drug craving after exposure to stressful or drug-related stimuli than women in the mid-follicular phase (Sinha et al. 2007) when estrogen levels are highest. In contrast, the mid-luteal phase, was associated with high positive affective responses to cocaine and enhanced cue- and stress-induced cocaine craving. Thus, estrogen may facilitate both the positively reinforcing and aversive aspects of cocaine abuse in women depending on the stimulus that primes craving (e.g., cues, drug)..
In contrast, others have found no differences in the physiological and subjective responses to cocaine during different phases of the menstrual cycle (Lukas et al. 1996; Mendelson et al. 1999). Progesterone, the predominant hormone during the luteal phase, has opposite effects on subjective measures in women after stimulant administration, compared with estrogen. For example, women treated with progesterone showed a decrease in positive, subjective effects of smoked (Evans and Foltin 2006; Sofuoglu et al. 2002) and iv cocaine (Sofuoglu et al. 2004) compared with placebo-treated controls. High plasma levels of progesterone were associated with decreased craving after drug- or stress-related cues in cocaine-dependent women (Sinha et al. 2007). In the present study it is possible that elevated progesterone during the luteal phase alleviated the stress associated with PCP withdrawal and allowed impulsive choice for SACC to occur in the luteal phase compared to the follicular phase.
The implication for human females cycling through menstrual phases during chronic periods of drug use and abstinence is that vulnerability to relapse may vary with cycle phase. The present results suggest that in impulsive behavior directed toward nondrug reinforcers peaks during the luteal phase of the menstrual cycle. An examination of withdrawal effects in humans, mainly focusing on smoking cessation, indicates that symptoms such as anxiety, depression, irritability, and poor concentration increased and are most severe during the luteal phase (Perkins et al. 2000; Pomerleau 1992). Cigarette craving in dependent women (Franklin et al. 2004), and cue-induced anxiety, physical stress responses, and cocaine craving (Sinha et al. 2007) are related to the late luteal phase. Furthermore, clinical work on smoking cessation during specific phases of the menstrual cycle indicated that the follicular phase (low progesterone) may be associated with more favorable outcomes when nicotine replacement therapy is used (Franklin et al. 2007; Carpenter et al. 2008). In contrast, quit attempts during the follicular phase (vs luteal) result in worse smoking cessation outcomes when behavioral strategies, or when non-nicotine (e.g., buproprion) or no medications are used to treat smoking cessation (Allen et al. 1999, 2004, 2008, 2009; Mazure et al. 2011). Thus, timing quit attempts to specific phases of the menstrual cycle as a function of the type of abused drug and treatment strategy is potentially important to a successful treatment outcome.
In summary, the present results indicated that withdrawal from PCP measured by elevated impulsive choice for SACC under a DD task was greater in female monkeys during the luteal than the follicular phase of the menstrual cycle. Thus, the risk of relapse to this form of drug abuse might be higher during the luteal phase and could lead to impulsive behavior directed toward another drug or nondrug substance. Once PCP access was restored, impulsive choice for SACC returned to lower levels indicating an acute and reversible effect of drug withdrawal. Females withdrawn from PCP were more at risk for these withdrawal effects in the luteal (vs follicular) phase of the menstrual cycle. The impact of drug withdrawal effects on impulsive choice for nondrug reinforcers may be important in designing treatment strategies that coordinate cessation attempts with the hormonal cycle. There may also be a benefit to directing impulsivity during withdrawal toward of healthy, alternative activities.
Table 2.
Menstrual phases studied for PCP baseline during follicular (PCP-F) and luteal (PCP-L) phases, PCP pre-withdrawal (Pre-WD), and PCP withdrawal (Wat-WD)

|
Acknowledgments
The authors are grateful to John Eckstrom and Lisa Tsackert for their technical assistance and Natalie Zlebnik for reviewing an earlier version of this manuscript and assisting with the figures. This research was supported by the National Institute on Drug Abuse grants R01 DA002486-32 and K05 DA015267-10 (MEC).
References
- Allen SS, Bade, Center B, Finstad D, Hatsukami D. Menstrual phase effects on smoking relapse. Addiction. 2008;103:809–821. doi: 10.1111/j.1360-0443.2008.02146.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen SS, Allen AM, Pomerleau CS. Influence of phase-related variability in premenstrual symptomatology, mood, smoking withdrawal, and smoking behavior during ad libitum smoking, on smoking cessation outcome. Addict Behav. 2009;34:107–111. doi: 10.1016/j.addbeh.2008.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Allen SS, Hatsukami DK, Christianson D, Nelson D. Withdrawal and pre-menstrual symptomatology during the menstrual cycle in short-term smoking abstinence: effects of menstrual cycle on smoking abstinence. Nicotine Tob Res. 1999;1:129–142. doi: 10.1080/14622299050011241. [DOI] [PubMed] [Google Scholar]
- Allen SS, Hatsukami DK, Bade T, Center B. Transdermal nicotine use in postmenopausal women: does the treatment efficacy differ in women using and not using hormone replacement therapy? Nicotine Tob Res. 2004;6:777–788. doi: 10.1080/1462220042000274239. [DOI] [PubMed] [Google Scholar]
- Anker JJ, Carroll ME. Females are more vulnerable to drug abuse than males: evidence from preclinical studies and role of ovarian hormones. In: Neill JC, Kulkarni J, editors. Biological Basis of Sex Differences in Psychopharmacology. Vol. 8. Springer; London, UK: 2011. pp. 73–96. Current Topics in Behavioral Neurosciences. [DOI] [PubMed] [Google Scholar]
- Anker JJ, Carroll ME. The role of progestins in the behavioral effects of cocaine and other drugs of abuse: human and animal research. Neurosci Biobehav Rev. 2010;35:315–333. doi: 10.1016/j.neubiorev.2010.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barr AM, Phillips AG. Withdrawal following repeated exposure to d-amphetamine decreases responding for a sucrose solution as measured by a progressive ratio schedule of reinforcement. Psychopharmacology. 1999;141:99–106. doi: 10.1007/s002130050812. [DOI] [PubMed] [Google Scholar]
- Becker JB, Hu M. Sex differences in drug abuse. Frontiers in Neurobiology. 2008;29:36–47. doi: 10.1016/j.yfrne.2007.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carpenter MJ, Saladin ME, Leinback AS, Larowe SD, Upadhyaya HP. Menstrual phase effects on smoking cessation: a pilot feasibility study. J Womens Health (Larchmt) 2008;17:293–301. doi: 10.1089/jwh.2007.0415. [DOI] [PubMed] [Google Scholar]
- Carroll ME. A quantitative assessment of phencyclidine dependence produced by oral self-administration in rhesus monkeys. J Pharmacol Exp Ther. 1987;242:405–412. [PubMed] [Google Scholar]
- Carroll ME. Oral self-administration of N-allylnormetazocine (SKF-10,047) stereoisomer in rhesus monkeys: Substitution during phencyclidine self-administration and withdrawal. Pharmacol Biochem Behav. 1989;30:493–500. doi: 10.1016/0091-3057(88)90486-8. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Anker JJ. Sex differences and ovarian steroid hormones in animal models of drug dependence. Horm Behav. 2010;58:44–56. doi: 10.1016/j.yhbeh.2009.10.001. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Anker JJ, Mach JL, Newman JL, Perry JL. Delay discounting as a predictor of drug abuse. In: Madden GJ, Bickel WK, editors. Impulsivity: The Behavioral and Neurological Science of Discounting. American Psychological Association; Washington, DC: 2010. pp. 243–272. [Google Scholar]
- Carroll ME, Anker JJ, Perry JL. Modeling risk factors for nicotine and other drug abuse in the preclinical laboratory, Drug and Alcohol Dependence, Supplemental Issue: Women and Smoking: Understanding Socioeconomic Influences. Dr Alc Dep. 2009a;93:343–348. doi: 10.1016/j.drugalcdep.2008.11.011. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Carmona GN. Effects of food FR and food deprivation on disruptions in food-maintained performance of monkeys during phencyclidine withdrawal. Psychopharmacology. 1991;104:143–149. doi: 10.1007/BF02244169. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Carmona GN, Rodefer JS. Phencyclidine (PCP) self-administration and withdrawal in rhesus monkeys: Effects of buprenorphine and dizocilpine (MK-801) pretreatment. Pharmacol Biochem Behav. 1994;48:723–732. doi: 10.1016/0091-3057(94)90339-5. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Lynch WJ, Roth ME, Morgan AD, Cosgrove KP. Sex and estrogen influence drug abuse. Tr Pharmacol Sci. 2004;25:273–279. doi: 10.1016/j.tips.2004.03.011. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Mach JL, LaNasa RM, Newman JL. Impulsivity as a behavioral measure of withdrawal of orally delivered PCP and nondrug rewards in male and female monkeys. Psychopharmacology. 2009b;207:85–98. doi: 10.1007/s00213-009-1636-y. [DOI] [PubMed] [Google Scholar]
- Carroll ME, Morgan AD, Anker JJ, Perry JL. Selective breeding for differential saccharin intake as an animal model of drug abuse. Behav Pharmacol. 2008;19:435–460. doi: 10.1097/FBP.0b013e32830c3632. [DOI] [PubMed] [Google Scholar]
- Cicero TJ, Nock B, Meyer ER. Gender-linked differences in the expression of physical dependence in the rat. Pharmacol Biochem Behav. 2002;72:691–697. doi: 10.1016/s0091-3057(02)00740-2. [DOI] [PubMed] [Google Scholar]
- Collins SL, Evans SM, Foltin RW, Haney M. Intranasal cocaine in humans: effects of sex and menstrual cycle. Pharmacol Biochem Behav. 2007;86:117–124. doi: 10.1016/j.pbb.2006.12.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cooper ZD, Shi YG, Woods JH. Reinforcer-dependnet enhancement of operant responding in opioid-withdrawn rats. Psychopharmacology. 2010;212:369–378. doi: 10.1007/s00213-010-1966-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corrigall WA, Herling S, Coen KM. Evidence for a behavioral deficit during withdrawal from chronic nicotine treatment. Pharmacol Biochem Behav. 1989;33:559–562. doi: 10.1016/0091-3057(89)90387-0. [DOI] [PubMed] [Google Scholar]
- Der-Avakian A, Markou A. Withdrawal from chronic exposure to amphetamine, but not nicotine, leads to an immediate and enduring deficit in motivated behavior without affecting social interaction in rats. Behav Pharmacol. 2010;21:359–368. doi: 10.1097/FBP.0b013e32833c7cc8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Devaud LL, Chadda R. Sex differences in rats in the development of and recovery from ethanol dependence assessed by changes in seizure susceptibility. Alc Clin Exp Res. 2001;25:1689–1696. [PubMed] [Google Scholar]
- Evans SM. The role of estradiol and progesterone in modulating the subjective effects of stimulants in humans. Exp Clin Psychopharmacol. 2007;15:418–426. doi: 10.1037/1064-1297.15.5.418. [DOI] [PubMed] [Google Scholar]
- Evans SM, Foltin RW. Does the response to cocaine differ as a function of sex or hormonal status in human and non-human primates? Horm Behav. 2010;58:13–21. doi: 10.1016/j.yhbeh.2009.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans SM, Foltin RW. Exogenous progesterone attenuates the subjective effects of smoked cocaine in women, but not in men. Neuropsychopharmacology. 2006;31:659–674. doi: 10.1038/sj.npp.1300887. [DOI] [PubMed] [Google Scholar]
- Evans SM, Haney M, Foltin RW. The effects of smoked cocaine during follicular and luteal phases of the menstrual cycle in women. Psychopharmacology. 2002;159:397–406. doi: 10.1007/s00213-001-0944-7. [DOI] [PubMed] [Google Scholar]
- Franklin TR, Ehrman R, Lynch KG, Harper D, Sciortino N, O’Brien CP, Childress AR. Menstrual Cycle Phase at Quit Date Predicts Smoking Status in an NRT Treatment Trial: A Retrospective Analysis. Journal of Women’s Health. 2007;17:287–292. doi: 10.1089/jwh.2007.0423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Franklin T, Napier K, Ehrman R, Gariti P, O’Brien CP, Childress AR. Retrospective study: influence of menstrual cycle on cue-induced cigarette craving. Nic Tob Res. 2004;6:171–175. doi: 10.1080/14622200310001656984. [DOI] [PubMed] [Google Scholar]
- Freitag WJ, Adesso VJ. Mood effects of alcohol and expectancies across the menstrual cycle. Alcohol. 1993;10:291–298. doi: 10.1016/0741-8329(93)90008-c. [DOI] [PubMed] [Google Scholar]
- Gear RW, Miaskowski C, Gordon NC, Paul SM, Heller PH, Levine JD. Kappa-opioids produce significantly greater analgesia in women than in men. Nat Med. 1996;2:1248–1250. doi: 10.1038/nm1196-1248. [DOI] [PubMed] [Google Scholar]
- Giordano LA, Bickel WK, Loewenstein G, Jacobs EA, Marsch L, Badger GJ. Mild opioid deprivation increases the degree that opioid-dependent outpatients discount delayed heroin and money. Psychopharmacology. 2002;163:174–182. doi: 10.1007/s00213-002-1159-2. [DOI] [PubMed] [Google Scholar]
- Hay WM, Nathan PE, Heermans HW, Frankenstein W. Menstrual cycle, tolerance and blood alcohol level discrimination ability. Addict Behav. 1984;9:67–77. doi: 10.1016/0306-4603(84)90008-x. [DOI] [PubMed] [Google Scholar]
- Holdstock L, de Wit H. Effects of ethanol at four phases of the menstrual cycle. Psychopharmacology (Berl) 2000;150:374–382. doi: 10.1007/s002130000461. [DOI] [PubMed] [Google Scholar]
- Husdon A, Stamp JA. Ovarian hormones and propensity to drug relapse: a review. Neurosci Biobehav Rev. 2011;35:427–436. doi: 10.1016/j.neubiorev.2010.05.001. [DOI] [PubMed] [Google Scholar]
- Justice AJ, de Wit H. Acute effects of d-amphetamine during the follicular and luteal phases of the menstrual cycle in women. Psychopharmacology (Berl) 1999;145:67–75. doi: 10.1007/s002130051033. [DOI] [PubMed] [Google Scholar]
- LeSage MG, Burroughs D, Pentel PR. Effects of nicotine withdrawal on performance under a progressive-ratio schedule of sucrose pellet delivery in rats. Pharmacol Biochem Behav. 2006;83:585–591. doi: 10.1016/j.pbb.2006.03.021. [DOI] [PubMed] [Google Scholar]
- Lex BW, Mendelson JH, Bavli S, Harvey K, Mello NK. Effects of acute marijuana smoking on pulse rate and mood states in women. Psychopharmacology (Berl) 1984;84:178–187. doi: 10.1007/BF00427443. [DOI] [PubMed] [Google Scholar]
- Lubman DI, Yucel M, Kettle JW, Scaffidi A, Mackenzie T, Simmons JG, Allen NB. Responsiveless to drug cues and natural rewards inopiate addiction: associations with later heroin use. Arch Gen Psychiatry. 2009;66:205–212. doi: 10.1001/archgenpsychiatry.2008.522. [DOI] [PubMed] [Google Scholar]
- Lukas SE, Sholar M, Lundahl LH, Lamas X, Kouri E, Wines JD, Kragie L, Mendelson JH. Sex differences in plasma cocaine levels and subjective effects after acute cocaine administration in human volunteers. Psychopharmacology (Berl) 1996;125:346–354. doi: 10.1007/BF02246017. [DOI] [PubMed] [Google Scholar]
- Lynch WJ, Roth ME, Carroll ME. Biological basis of sex differences in drug abuse: preclinical and clinical studies. Psychopharmacology. 2002;164:121–137. doi: 10.1007/s00213-002-1183-2. [DOI] [PubMed] [Google Scholar]
- Lynch WJ, Sofuoglu M. Role of progesterone in nicotine addiction: evidence from initiation to relapse. Exp Clin Psychopharm. 2010;18:451–461. doi: 10.1037/a0021265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauro J, Serrone D, Somsin P, Stein AA. Cyclic vaginal cytologic patterns in the Macaca mulatta. Acto Cytol. 1970;14:348–352. [PubMed] [Google Scholar]
- Mazure CM, Toll B, McKee SA, Wu R, O’Malley SS. Menstrual cycle phase at quit date and smoking abstinence at 6 weeks in an open label trial of bupropion. Dr Alc Dep. 2011;114:68–72. doi: 10.1016/j.drugalcdep.2010.07.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mello NK. Hormones, nicotine, and cocaine: Clinical studies. Hormones and Behavior. 2010;58:57–71. doi: 10.1016/j.yhbeh.2009.10.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mello NK, Knudson IM, Kelly M, Fivel PA, Mendelson JH. Effects of Progesterone and Testosterone on Cocaine Self-Administration and Cocaine Discrimination by Female Rhesus Monkeys. Neuropsychopharm. 2011;36:2187–2199. doi: 10.1038/npp.2011.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mello NK, Knudson IM, Mendelson JH. Sex and menstrual cycle effects on progressive ratio measures of cocaine self-administration in cynomolgus monkeys. Neuropsychopharm. 2007;32:1956–1966. doi: 10.1038/sj.npp.1301314. [DOI] [PubMed] [Google Scholar]
- Mendelson JH, Mello NK, Sholar MB, Siegel AJ, Kaufman MJ, Levin JM, Renshaw PF, Cohen BM. Cocaine pharmacokinetics in men and in women during the follicular and luteal phases of the menstrual cycle. Neuropsychopharmacology. 1999;21:294–303. doi: 10.1016/S0893-133X(99)00020-2. [DOI] [PubMed] [Google Scholar]
- Mendelson JH, Scholar MB, Siegel AJ, Mello NK. Effects of cocaine on luteinizing hormone in women during the follicular and luteal phases of the menstrual cycle and in men. J Pharmacol Exp Ther. 2001;296:972–979. [PubMed] [Google Scholar]
- Miladi-Gorji H, Rashidy-Pour A, Fathollahi Y. Anxiety profile in morphine-dependent and withdrawn rats: Effect of voluntary exercise. Physiol Behav. 2012;105:195–202. doi: 10.1016/j.physbeh.2011.08.010. [DOI] [PubMed] [Google Scholar]
- Morabia A, Fabre J, Chee E, Zeger S, Orsat E, Robert A. Diet and opiate addiction: a quantitative assessment of the diet of noninstitutionalized opiate addicts. Br J Addict. 1989;84:173–180. doi: 10.1111/j.1360-0443.1989.tb00566.x. [DOI] [PubMed] [Google Scholar]
- National Research Council of the National Academies. Guide for the Care and Use of Laboratory Animals. 8. National Academies Press; Washington, D.C: 2011. p. 217. [Google Scholar]
- Newman JL, Thorne JJ, Batulis DK, Carroll ME. Effects of menstrual cycle phase on the reinforcing effects of phencyclidine (PCP) in rhesus monkeys. Pharmacol Biochem Behav. 2006;85:584–591. doi: 10.1016/j.pbb.2006.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman JL, Perry JL, Carroll ME. Effects of altering reinforcer magnitude and reinforcement schedule on phencyclidine (PCP) self-administration in monkeys using an adjusting delay task. Pharmacol Biochem Behav. 2008;90:778–786. doi: 10.1016/j.pbb.2008.05.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newman JL, Thorne JJ, Batulis DK, Carroll ME. Effects of menstrual cycle phase on the reinforcing effects of phencyclidine (PCP) in rhesus monkeys. Pharmacol Biochem Behav. 2006;85:584–591. doi: 10.1016/j.pbb.2006.10.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nyberg S, Wahlstrom G, Backstrom T, Sundstrom Poromaa I. Altered sensitivity to alcohol in the late luteal phase among patients with premenstrual dysphoric disorder. Psychoneuroendocrinology. 2004;29:767–777. doi: 10.1016/S0306-4530(03)00121-5. [DOI] [PubMed] [Google Scholar]
- Perkins KA, Levine M, Marcus M, Schiffman S, D’Amico D, Miller A, Keins A, Ashcom J, Broge M. Tobacco withdrawal in women and menstrual cycle phase. J Consult Clin Psychol. 2000;68:176–180. doi: 10.1037/0022-006X.68.1.176. [DOI] [PubMed] [Google Scholar]
- Perry JL, Carroll ME. The role of impulsive behavior in drug abuse. Psychopharmacology. 2008;200:1–26. doi: 10.1007/s00213-008-1173-0. [DOI] [PubMed] [Google Scholar]
- Perry JL, Normile LM, Morgan AD, Carroll ME. Sex differences in physical dependence on orally self-administered phencyclidine (PCP) rhesus monkeys (Macaca mulatta) Exp Clin Psychopharm. 2006;1:68–78. doi: 10.1037/1064-1297.14.1.68. [DOI] [PubMed] [Google Scholar]
- Pomerleau CS, Garcia AW, Pomerleau OF, Cameron OG. The effects of menstrual phase and nicotine abstinence on nicoting intake and on biochemical and subjective measures in women smokers: a preliminary report. Psychoneuroendocrinology. 1992;17:627–638. doi: 10.1016/0306-4530(92)90021-x. [DOI] [PubMed] [Google Scholar]
- Reed SC, Evans SM, Bedi G, Rubin E, Foltin RW. The effects of oral micronized progesterone on smoked cocaine self-administration in women. Horm Behav. 2011;59:227–235. doi: 10.1016/j.yhbeh.2010.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rouibi K, Contarino A. Increased motivation to eat in opiate-withdrawn mice. Psychopharmacology. 2012;221:675–684. doi: 10.1007/s00213-011-2612-x. [DOI] [PubMed] [Google Scholar]
- Sinha R. Chronic stress, drug use, and vulnerability to addiction. Ann N Y Acad Sci. 2008;1141:105–130. doi: 10.1196/annals.1441.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha R, Fox H, Hong KI, Sofuoglu M, Morgan PT, Bergquist KT. Sex steroid hormones, stress response, and drug craving in cocaine-dependent women: implications for relapse susceptibility. Exp Clin Psychopharmacol. 2007;15:445–452. doi: 10.1037/1064-1297.15.5.445. [DOI] [PubMed] [Google Scholar]
- Snively TA, Ahijevych KL, Bernhard LA, Wewers ME. Smoking behavior, dysphoric states and the menstrual cycle: results from single smoking sessions and the natural environment. Psychoneuroendocrinology. 2000;25:677–691. doi: 10.1016/s0306-4530(00)00018-4. [DOI] [PubMed] [Google Scholar]
- Sofuoglu M, Babb DA, Hatsukami DK. Effects of progesterone treatment on smoked cocaine response in women. Pharmacol Biochem Behav. 2002;72:431–435. doi: 10.1016/s0091-3057(02)00716-5. [DOI] [PubMed] [Google Scholar]
- Sofuoglu M, Dudish-Poulsen S, Nelson D, Pentel PR, Hatsukami DK. Sex and menstrual cycle differences in the subjective effects from smoked cocaine in humans. Exp Clin Psychopharmacol. 1999;7:274–283. doi: 10.1037//1064-1297.7.3.274. [DOI] [PubMed] [Google Scholar]
- Sofuoglu M, Mitchell E, Kosten TR. Effects of progesterone treatment on cocaine responses in male and female cocaine users. Pharmacol Biochem Behav. 2004;78:699–705. doi: 10.1016/j.pbb.2004.05.004. [DOI] [PubMed] [Google Scholar]
- Substance Abuse and Mental Health Services Administration (SAMHSA) Results from the 2011 National Survey on Drug Use and Health: Summary of National Findings. U.S. Dept of Health and Human Services; Rockville MD: 2011. [Google Scholar]
- Sutker PB, Goist KC, Jr, King AR. Acute alcohol intoxication in women: relationship to dose and menstrual cycle phase. Alcohol Clin Exp Res. 1987;11:74–79. doi: 10.1111/j.1530-0277.1987.tb01266.x. [DOI] [PubMed] [Google Scholar]
- Suzuki T, Koike Y, Misawa M. Sex differences in physical dependence on methaqualone in the rat. Pharmacol Biochem Behav. 1988;30:483–488. doi: 10.1016/0091-3057(88)90484-4. [DOI] [PubMed] [Google Scholar]
- Suzuki T, Koike Y, Yoshii T, Yanaura S. Sex differences in the induction of physical dependence on pentobarbital in the rat. Jap J Pharmacol. 1985;39:453–459. doi: 10.1254/jjp.39.453. [DOI] [PubMed] [Google Scholar]
- Terner JM, de Wit H. Menstrual cycle phase and responses to drugs of abuse in humans. Drug Alcohol Depend. 2006;84:1–13. doi: 10.1016/j.drugalcdep.2005.12.007. [DOI] [PubMed] [Google Scholar]
- Uban KA, Rummel J, Floresco SB, Galea LAM. Estradiol modulates effort-based decision making in female rats. Neuropsychopharm. 2010;37:390–401. doi: 10.1038/npp.2011.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss G. Food fantasies of incarcerated drug users. Int J Addict. 1982;17:905–912. doi: 10.3109/10826088209056337. [DOI] [PubMed] [Google Scholar]
- White TL, Justice AJ, de Wit H. Differential subjective effects of D-amphetamine by gender, hormone levels and menstrual cycle phase. Pharmacol Biochem Behav. 2002;73:729–741. doi: 10.1016/s0091-3057(02)00818-3. [DOI] [PubMed] [Google Scholar]
- Zijlstra F, Veltman DJ, Booij J, van den Brink W, Franken IH. Neurobiological substrates of cue-elicited craving and anhedonia in reently abstinent opioid-dependent males. Dr Alc Dep. 2009;99:183–192. doi: 10.1016/j.drugalcdep.2008.07.012. [DOI] [PubMed] [Google Scholar]