Abstract
Ginsenoside (G) Rp1 is a ginseng saponin derivative with anti-cancer and anti-inflammatory activities. In this study, we examined the mechanism by which G-Rp1 inhibits inflammatory responses of cells. We did this using a strategy in which DNA constructs containing cyclooxygenase (COX)-2 and inducible nitric oxide synthase (iNOS) promoters were transfected into HEK293 cells. G-Rp1 strongly inhibited the promoter activities of COX-2 and iNOS; it also inhibited lipopolysaccharide induced upregulation of COX-2 and iNOS mRNA levels in RAW264.7 cells. In HEK293 cells G-Rp1 did not suppress TANK binding kinase 1-, Toll-interleukin-1 receptor-domain-containing adapter-inducing interferon-β (TRIF)-, TRIFrelated adaptor molecule (TRAM)-, or activation of interferon regulatory factor (IRF)-3 and nuclear factor (NF)-кB by the myeloid differentiation primary response gene (MyD88)-induced. However, G-Rp1 strongly suppressed NF-кB activation induced by IкB kinase (IKK)β in HEK293 cells. Consistent with these results, G-Rp1 substantially inhibited IKKβ-induced phosphorylation of IкBɑ and p65. These results suggest that G-Rp1 is a novel anti-inflammatory ginsenoside analog that can be used to treat IKKβ/NF-кB-mediated inflammatory diseases.
Keywords: Panax ginseng, Ginsenoside Rp1, Cyclooxygenase 2, Nitric oxide synthase type II, Promoter activity, Nuclear factor-кB
INTRODUCTION
Nitric oxide (NO) and prostaglandin E2 (PGE2) are representative inflammatory mediators produced by macrophages under inflammatory conditions [1]. These molecules are generated by the activation of inducible NO synthase (iNOS) and cycooxygenase-2 (COX-2). Thus, these molecules are primarily controlled by transcriptional and translational regulation by surface receptors such as Toll like receptors (TLR) and their counter adaptor and signaling molecules such as TANK binding kinase (TBK)1, Toll–interleukin-1 receptor-domain-containing adapter-inducing interferon-β (TRIF), TRIF-related adaptor molecule (TRAM), and myeloid differentiation primary response gene (88) (MyD88). The activation of cellular environments finally leads to the functional up-regulation of transcription factors (e.g., nuclear factor [NF]-кB, interferon regulatory factor [IRF]-3, and activator protein [AP]-1) [2]. Excessive release of NO and PGE2 is also linked to the onset of serious inflammatory diseases such as rheumatoid arthritis and arteriosclerosis [3]. Inhibition of NO and PGE2 release, therefore, is considered to be an important therapeutic target.
Ginseng (Panax ginseng C. A. Meyer) is an herbal medicine that has long been used to treat diseases such as infection, cancer, arthritis, and atherosclerosis. Recently, numerous scientific studies have identified the functionally active phytochemical components in ginseng and the molecular mechanisms of their therapeutic effects. Furthermore, various disease models have been created to expand the effects of ginseng derived extracts or components [4]. The major pharmacological components of ginseng are thought to be ginseng saponins (called ginsenosides). Recent ginseng researchers have now begun to study the use of individual ginsenosides or specific ginsenoside-rich fractions. For example, it has been reported that ginsenoside (G)-Rg3 can indirectly suppress tumor growth [5], and it has been launched as an anti-cancer drug. G-F1 is presently added to in cosmetic biomaterials that are used for their anti-wrinkling effects [6]. G-Rb1 has also been shown to exhibit anti-arthritic activity [7].
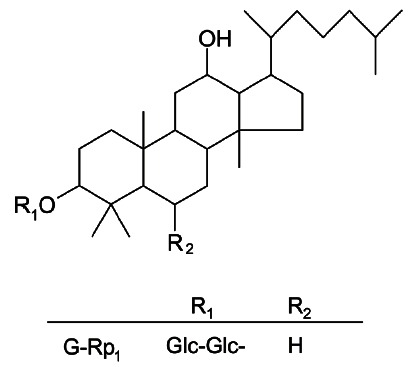
Nonetheless, the development of ginseng-derived ginsenosides as valuable drugs or remedies is now seen as limited because the components are already widely used. To avoid such an application problem, we have tried to prepare novel ginsenoside-derived compounds that improve chemical stability and mass production. Given these goals, G-Rp1 (Fig. 1) was prepared on a large scale from crude ginsenosides (e.g., G-Rg5 and G-Rk1) by means of reduction by hydrogenation [8] and proved to be chemically stable [9]. Through aggressive efforts, it has been demonstrated that G-Rp1 has strong anti-cancer and anti-inflammatory properties [8,10,11]
Fig. 1. The chemical structure of ginsenoside (G)-Rp1.
Although it is speculated that G-Rp1 has anti-inflammatory activity, the exact inhibitory target of this compound has not yet been identified. In this study, we explored the mechanism by which G-Rp1 exerts its regulatory effects. To this end, we used a reporter gene assay system in HEK293 cells. Cells were transfected with transcription factor-stimulatory adaptors and signaling molecules.
MATERIALS AND METHODS
Materials
G-Rp1 (purity, 97%), a racemic mixture of R- and S-enantiomers in a 1:1.3 ratio, was prepared using established protocols [9]. The stock solution (20 mM) of G-Rp1 was prepared using 100% dimethyl sulfoxide. Lipopolysaccharide (LPS, Esherichia coli 0111:B4), ionomycin A, and phorbol-12-myristate-13-acetate (PMA) were purchased from Sigma (St. Louis, MO, USA). Fetal bovine serum was obtained from Gibco (Grand Island, NY, USA). All other chemicals were Sigma grade. Phospho-specific or total antibodies to p65 and IкBɑ were purchased from Cell Signaling (Beverly, MA, USA).
Cell culture
RAW264.7 cells (the American Type Culture Collection, Rockville, MD, USA) were maintained in complete RPMI1640 medium (supplemented with 100 U/mL of penicillin and 100 μg/mL of streptomycin, and 10% fetal bovine serum). HEK293 cells (the American Type Culture Collection) were maintained in DMEM medium (supplemented with 100 U/mL of penicillin, 100 μg/mL of streptomycin, and 10% fetal bovine serum).
Reverse transcription and real-time polymerase chain reaction
Total RNA from LPS-treated-RAW264.7 cells (5×106 cells/mL) was prepared by adding TRIzol Reagent (Gibco) according to the manufacturer’s protocol [12]. The total RNA solution was stored at -70℃ until used. Semi-quantitative real-time (RT) reactions were conducted using MuLV reverse transcriptase. Total RNA (1 μg) was incubated with oligo-dT15 for 5 min at 70℃ and then mixed with a 5X first-strand buffer, 10 mM dNTPs, and 0.1 M DTT. The reaction mixture was further incubated for 5 min at 37℃ and for 60 min after the addition of MuLV reverse transcriptase (2 U). Reactions were terminated after 10 min at 70℃, and total RNA was depleted by adding RNase H. The polymerase chain reaction (PCR) was conducted with the incubation mixture (2 μl cDNA, 4 μM 5’ and 3’ primers, a 10X buffer [ 10 mM Tris-HCl, pH 8.3, 50 mM KCl, 0.1% Triton X-100], 250 μ M of dNTP, 25 mM of MgCl2, and 1 unit of Taq polymerase [Promega, Madison, WI, USA]). The following incubation conditions were used: a 30 sec denaturation time at 94℃, an annealing time of 30 sec between 55 and 60℃, an extension time of 45 sec at 72℃, and a final extension of 5 min at 72℃. For RT-PCR analysis, one microgram of RNA was submitted to reverse transcription with the Molony murine leukemia virus reverse transcriptase (Invitrogen, Carlsbad, CA, USA). Two microliters of cDNA obtained from each sample were submitted to a qPCR assay using the SYBR green Master mix method (Applied Biosystems, Foster City, CA, USA) in the ABO sequence detection system, The results were normalized with the 18S transcript. The primers (Bioneer, Daejeon, Korea) used in this experiment are indicated in Table 1.
Table 1.
Primers used for quantitative real-time and semiquantitative real-time polymerase chain reaction (RT-PCR) analyses
| Gene | Primer sequences |
|---|---|
|
| |
| For semiquantitative RT-PCR | |
| IFN-β | F 5'- :CCA CCA CAG CCC TCT CCA TCA ACT AT-3' |
| R 5'- CAA GTG GAG AGC AGT TGA GGA CAT C-3' | |
| GAPDH | F 5'-CACTCACGGCAAATTCAACGGCAC-3' |
| R 5'-GACTCCACGACATACTCAGCAC-3' | |
| For quantitative real-time RT-PCR | |
| TNF-α | F 5'-TGCCTATGTCTCAGCCTCTTC-3' |
| R 5'-GAGGCCATTTGGGAACTTCT-3' | |
| COX-2 | F 5'-GGGAGTCTGGAACATTGTGAA-3' |
| R 5'-GCACATTGTAAGTAGGTGGACTGT-3' | |
| iNOS | F 5`-GGAGCCTTTAGACCTCAACAGA-3` |
| R 5`-TGAACGAGGAGGGTGGTG-3` | |
| GAPDH | F 5'- CAATGAATACGGCTACAGCAAC-3' |
| R 5'- AGGGAGATGCTCAGTGTTGG-3' | |
IFN-β, interferon-β; TNF-α, tumor necrosis factor-α; COX-2, cyclooxygenase-2; iNOS, inducible nitric oxide synthase; F, forward; R, reverse.
Immunoblotting
Cells (5×106 cells/mL) were lysed in lysis buffer (in mM: 20 TRIS-HCl, pH 7.4, 2 EDTA, 2 EGTA, 50 β-glycerophosphate, 1 sodium orthovanadate, 1 dithiothreitol, 1% Triton X-100, 10% glycerol, 10 μg/mL leupeptin, 10 μg/mL aprotinin, 10 μg/mL pepstatin, 1 benzimidine, and 2 hydrogen peroxide) for 30 min rotating at 4℃. Lysates were clarified by centrifugation at 16,000 ×g for 10 min at 4℃. Soluble cell lysates were immunoblotted, and phospho- and total levels of IкBɑ and p65 were visualized as previously reported [13].
Luciferase reporter gene activity assay
HEK293 cells (1×106 /mL) were transfected with 1 μg of plasmids with COX-2 promoter-Luc, iNOS-promoter-Luc, NF-кB-Luc or IFN-β-promoter-Luc in the absence or presence of adaptors or enzyme DNAs as well as β-galactosidase using a PEI method in a 12-well plate. The cells were used for experiments 48 h after transfection. Luciferase assays were performed using the Luciferase Assay System (Promega) [14,15].
Briefly, the transfected cells treated with G-Rp1 were lysed in the culture dishes with reporter lysis buffer. Lysates were centrifuged at maximum speed for 10 min in an Eppendorf microcentrifuge. Ten μL of the supernatant fraction were incubated with 50 μL of luciferase substrate, and the relative luciferase activity was determined with a Luminoskan Ascent system (Thermo Labsystems, Helsinki, Finland). Luciferase activity was normalized to β-galactosidase activity.
Statistical analysis
Student’ s t-test and one-way ANOVA were used to determine the statistical significance of differences between values for the various experimental and control groups. Data expressed as means± standard errors are taken from at least three independent experiments each performed in triplicate (Figs. 2, 3A, 3B, 4, and 5A). The rest of the data are representative of three different experiments with similar results. A p-value of 0.05 or less were considered to be statistically significant.
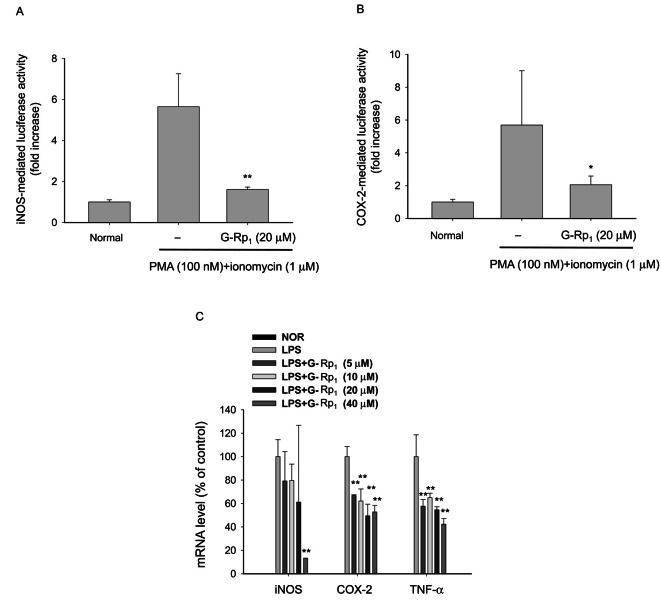
Fig. 2. Effect of ginsenoside (G)-Rp1 on the promoter activity of inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX)-2 genes. (A,B) HEK293 cells co-transfected with the plasmid constructs iNOS-Luc or COX-2-Luc (1 μg/mL each) and β-gal (as a transfection control) were treated with G-Rp1 (20 μM) in the presence or absence of phorbol-12-myristate-13-acetate (PMA, 100 nM)/ionomycin A (1 μM) for 12 h. Luciferase activity was determined by luminometry. (C) RAW264.7 cells (5×106 cells/mL) pre-treated with G-Rp1 for 1 h were stimulated in the absence or presence of lipopolysaccharide (1 μg/mL) for 6 h. After preparing mRNA, the levels of iNOS, COX-2, tumor necrosis factor (TNF)-ɑ, and GAPDH were determined by real-time polymerase chain reaction. NOR, normal. *p<0.05 and **p<0.01 compared to control.
Fig. 3. Effect of ginsenoside (G)-Rp1 on interferon regulatory factor-3 activation and interferon (IFN)-β expression. (A,B) HEK293 cells co-transfected with the plasmid constructs IFN-β-promoter-Luc (1 μg/mL each) and β-gal (as a transfection control) were treated with G-Rp1 (20 μM) in the presence or absence of DNA constructs for Toll–interleukin-1 receptor-domain-containing adapter-inducing interferon-β (TRIF) or TANK binding kinase (TBK)1 for 12 h. Luciferase activity was determined by luminometry. (C) RAW264.7 cells (5×106 cells/mL) pre-treated with G-Rp1 for 1 h were stimulated in the absence or presence of LPS (1 μg/mL) for 1 h. After preparing mRNA, the levels of IFN-β and GAPDH were determined by semiquantitative real-time polymerase chain reaction. LPS, lipopolysaccharide.
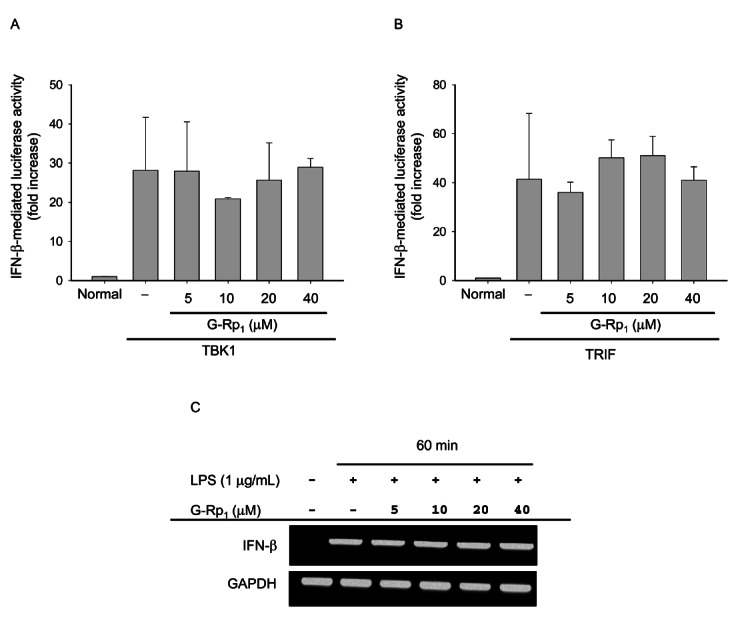
Fig. 4. Effect of ginsenoside (G)-Rp1 on nuclear factor (NF)-кB activation induced by adaptor molecules. (A,B,C, and D) HEK293 cells co-transfected with the plasmid constructs NF-кB-Luc (1 μg/mL each) and β-gal (as a transfection control) were treated with G-Rp1 in the presence or absence of DNA constructs containing TANK binding kinase (TBK)1, Toll–interleukin-1 receptor-domain-containing adapter-inducing interferon-β (TRIF), TRIF-related adaptor molecule (TRAM) or myeloid differentiation primary response gene (88) (MyD88) for 12 h. Luciferase activity was determined by luminometry.
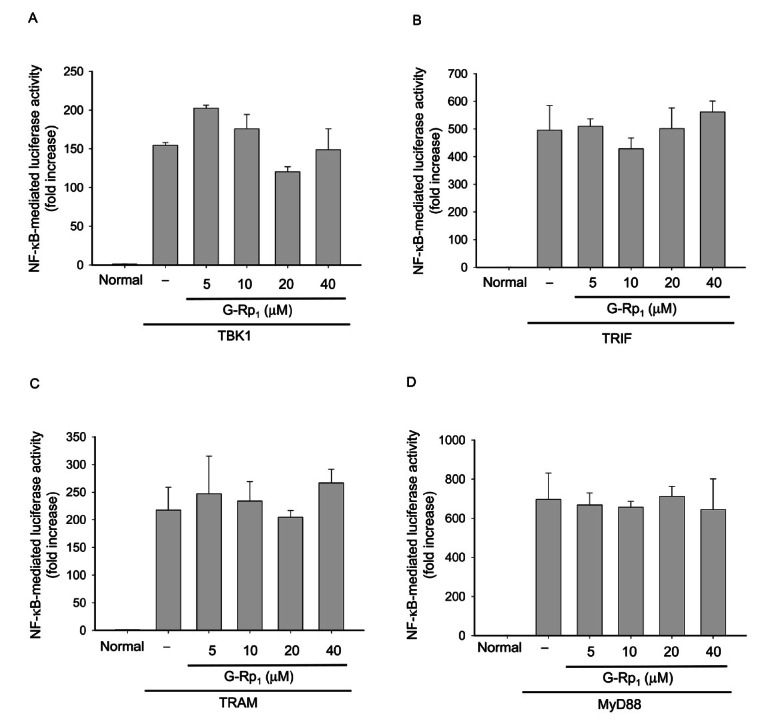
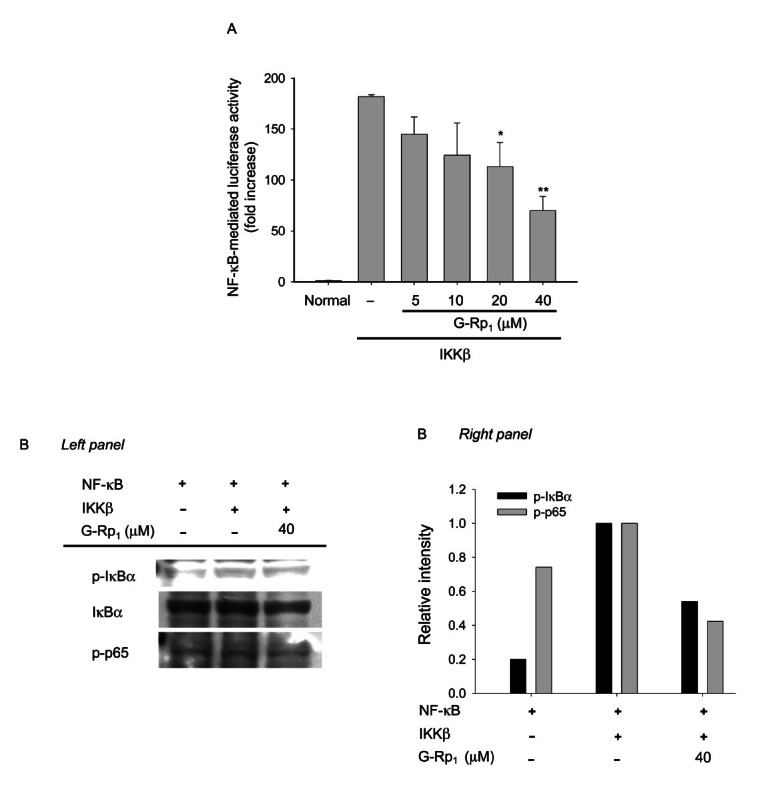
Fig. 5. Effect of ginsenoside (G)-Rp1 on nuclear factor (NF)-кB activation induced by by IкB kinase (IKK)β. (A) HEK293 cells co-transfected with the plasmid constructs NF-кB-Luc (1 μg/mL each) and β-gal (as a transfection control) were treated with G-Rp1 in the presence or absence of DNA constructs containing IKKβ for 12 h. Luciferase activity was determined by luminometry. (B) HEK293 cells (5×106 cells/mL) transfected with IKKβ (1 μg/mL) were incubated with G-Rp1 for 1 h. After preparing the nuclear fraction, the phosphorylation or total levels of IкBɑ and p65 were determined by phospho-specific or total antibodies. *p<0.05 and **p<0.01 compared to control.
RESULTS AND DISCUSSION
G-Rp1 (Fig. 1) has been shown to inhibit the production of IL-1β in LPS-activated macrophages in a dose dependent manner [16]. In addition, it has been reported that β1-integrin (CD29)-mediated cell-cell adhesion, which is frequently seen in inflammatory responses [17], can be suppressed by this compound. Therefore, in this study, we aimed to investigate the anti-inflammatory mechanism of G-Rp1. To do this, we used a reporter gene assay to examine the ability of this compound to suppress transcriptional activation of iNOS and COX-2, major inflammatory genes.
Transcriptional activation was set up by transfection of luciferase gene-containing DNA constructs with iNOS and COX-2 promoters. Indeed, transfection of each DNA increased its promoter activity, as assessed by luciferase activity, up to 6 fold upon stimulation with PMA and ionomycin A (Fig. 2). Interestingly, G-Rp1 strongly inhibited their promoter activities up to 90% at 20 μM (Fig. 2A, B), without affecting cell viability up to 40 μM (data not shown). Additionally, this compound dose-dependently suppressed the mRNA expression of iNOS, COX-2, and tumor necrosis factor (TNF)-ɑ induced by LPS stimulation of RAW264.7 cells (Fig. 2C), suggesting that G-Rp1 works properly. The observed results were similar to a previous report in which G-Rp1 diminished IL-1β production at the transcriptional level [16].
Transcriptional control by G-Rp1 was shown to be mediated by suppression of NF-кB activation [18]. In addition, AP-1 and IRF-3 have been suggested to play a critical role in the expression of iNOS and COX-2 genes by direct or indirect activation. Therefore, we next evaluated the involvement of these transcription factors in G-Rp1-mediated inhibition of transcriptional activation.
First, we tested whether G-Rp1 modulated an IFN-β-mediated indirect pathway in which IFN-β produced by the TRIF/TBK1/IRF-3 pathway can stimulate, autonomously, the production of PGE2 and NO in macrophages or other inflammatory cells. As Fig. 3 shows, however, G-Rp1 did not suppress IRF-3 activation, assessed by luciferase activity assay using an IRF-3 binding DNA sequence that contained the IFN-β-promoter construct with a luciferase gene. In particular, upregulation of IFN-β promoter activity by stimulation with TRIF, a major adaptor molecule involved in TLR4 signaling, or TBK1, a major signaling enzyme to activate IKKe and counter transcription factor IRF-3, was not attenuated by G-Rp1 treatment (Fig. 3). Moreover, G-Rp1 did not suppress mRNA expression of IFN-β (Fig. 3C), a major outcome of IRF-3 activation [19], suggesting that G-Rp1 is not involved in blocking the TRIF/TBK1/IRF-3 pathway for IFN-β production. Furthermore, G-Rp1 did not block the luciferase activity that was mediated by activation of AP-1 stimulated by PMA [20-22] and co-transfection with other stimulatory molecules such as TRIF and TBK1 (data not shown).
Recently, we reported that G-Rp1 suppresses LPS-mediated NF-кB activation in RAW264.7 cells. Therefore, we investigated whether G-Rp1 can block NF-кB-mediated promoter activity that had been indirectly increased by various adaptors and signaling molecules in HEK293 cells. Several stimuli and proteins that had been cotransfected into HEK293 cells were found to trigger NF-кB activation and subsequently increase luciferase activity [21]. In particular, PMA and TNF-ɑ are regarded as good inducers, while MyD88, TRIF, TBK1, and IKKβ are considered to be strong activating proteins of the NF-кB pathway. Indeed, under our conditions, cotransfection of or treatment with these molecules enhanced NF-кB-mediated luciferase activity.
However, G-Rp1 treatment did not block MyD88-, TRIF-, TRAM-, and TBK1-mediated NF-кB activation and their subsequent luciferase activities (Fig. 4), although there seems to be an inhibitory pattern when HEK293 cells were treated with 20 μM G-Rp1 under TBK1 co-transfection conditions (Fig. 4A). However, since a higher concentration (40 μM) of G-Rp1 failed to show NF-кB inhibitory activity, we concluded that G-Rp1 is not able to strongly suppress NF-кB activation that is mediated by such adaptor molecules. Unlike the latter results, G-Rp1 strongly inhibited IKKβ-induced NF-кB activation in HEK293 cells (Fig. 5A). In agreement with this, G-Rp1 suppressed the phosphorylation of IкBɑ and p65 (Fig. 5B), suggesting that IKKβ is the true target of G-Rp1 in its NF-кB suppressive activity. Indeed, G-Rp1 strongly down-regulated the release of NF-кB-mediated inflammatory gene products. For example, G-Rp1 suppressed the production of other inflammatory mediators such as NO (down to 42±2% at 40 μM) and TNF-ɑ (53±2% at 40 μM) [16]. This compound strongly blocked LPS-mediated IL-1β release [16]. The importance of NF-кB in inflammatory responses was also confirmed by treatment with parthenolide and BAY 11-7082, two strong and selective NF-кB inhibitors with distinct anti-inflammatory properties [21]. Therefore, we speculated that NF-кB itself or its activation pathway induced by LPS or other inflammatory signals may be the critical target of G-Rp1.
Recently, the anti-inflammatory feature of ginsenosides or their structural analogs such as compound K, G-Rh1, G-Rb1, G-Rb2, G-Rh2, G-Re, and 20(S)-protopanaxatriol have been revealed [23]. The main anti-inflammatory effects of these compounds are to suppress NO generation, TNF-ɑ release, and PGE2 production both at transcriptional and translational levels [24]. As we reported previously, the inhibitory target of these compounds is mostly found to be the NF-кB pathway, not the activation of glucocorticoid receptor [16,25]. There was no correlation between the structures of these compounds and their anti-inflammatory activities. However, a unique carbohydrate moiety at the R1 position of ginsenosides seems to be crucial for strong anti-inflammatory activity [17,26]. Similar structure-activity relationships were found for G-Rp1 and its structural analogs including G-Rp2, G-Rp1, and G-Rp3, as reported previously [17,26].
Recently, we demonstrated that G-Rp1 is available after oral administration since this compound strongly blocked melanoma cell metastasis [10]. Moreover, when this compound was orally administered, it was chemically stable and identifiable in blood (data not shown). Furthermore, G-Rp1 is a novel patentable ginsenoside derivative. Acute toxicity of this compound was found only at doses greater than 2.5 g/kg. These points open the possibility of developing G-Rp1 as a novel ginsenoside-derived drug with anti-cancer and anti-inflammatory properties. Continued studies will be focused on exploring the pharmacological value of G-Rp1 by performing extensive pre-clinical studies.
In conclusion, we found that G-Rp1 inhibits prompoter activities of iNOS and COX-2 as well as the mRNA levels of these genes. In particular, this compound down-regulates NF-кB-mediated luciferase activity that is mediated by IKKβ, but not by other adaptor and signaling molecules. In agreement with these findings, G-Rp-1 strongly diminished the phosphorylation of IkBɑ and p65. Since G-Rp1 is known to be safe and orally effective [10], our results suggest that G-Rp1 can be used as a novel drug for the treatment of inflammatory diseases.
Acknowledgments
This study was supported by 2008 research grant from Kangwon National University.
References
- 1.Bird S, Zou J, Wang T, Munday B, Cunningham C, Secombes CJ. Evolution of interleukin-1beta. Cytokine Growth Factor Rev. 2002;13:483–502. doi: 10.1016/S1359-6101(02)00028-X. [DOI] [PubMed] [Google Scholar]
- 2.Kim HS, Moon EY. Reactive oxygen species-induced expression of B cell activating factor (BAFF) is independent of Toll-like receptor 4 and myeloid differentiation primary response gene 88. Biomol Ther. 2009;17:144–150. [Google Scholar]
- 3.Namkoong S, Kim YM. Therapeutic application of nitric oxide in human diseases. Biomol Ther. 2010;18:351–362. [Google Scholar]
- 4.Gillis CN. Panax ginseng pharmacology: a nitric oxide link? Biochem Pharmacol. 1997;54:1–8. doi: 10.1016/S0006-2952(97)00193-7. [DOI] [PubMed] [Google Scholar]
- 5.Kwon HY, Kim EH, Kim SW, Kim SN, Park JD, Rhee DK. Selective toxicity of ginsenoside Rg3 on multidrug resistant cells by membrane fluidity modulation. Arch Pharm Res. 2008;31:171–177. doi: 10.1007/s12272-001-1137-y. [DOI] [PubMed] [Google Scholar]
- 6.Lee EH, Cho SY, Kim SJ, Shin ES, Chang HK, Kim DH, Yeom MH, Woe KS, Lee J, Sim YC, et al. Ginsenoside F1 protects human HaCaT keratinocytes from ultraviolet-B-induced apoptosis by maintaining constant levels of Bcl-2. J Invest Dermatol. 2003;121:607–613. doi: 10.1046/j.1523-1747.2003.12425.x. [DOI] [PubMed] [Google Scholar]
- 7.Kim HA, Kim S, Chang SH, Hwang HJ, Choi YN. Anti-arthritic effect of ginsenoside Rb1 on collagen induced arthritis in mice. Int Immunopharmacol. 2007;7:1286–1291. doi: 10.1016/j.intimp.2007.05.006. [DOI] [PubMed] [Google Scholar]
- 8.Cho JY, Yoo ES, Baik KU, Park MH, Han BH. In vitro inhibitory effect of protopanaxadiol ginsenosides on tumor necrosis factor (TNF)-alpha production and its modulation by known TNF-alpha antagonists. Planta Med. 2001;67:213–218. doi: 10.1055/s-2001-12005. [DOI] [PubMed] [Google Scholar]
- 9.Kumar A, Kumar M, Panwar M, Samarth RM, Park TY, Park MH, Kimura H. Evaluation of chemopreventive action of ginsenoside Rp1. Biofactors. 2006;26:29–43. doi: 10.1002/biof.5520260104. [DOI] [PubMed] [Google Scholar]
- 10.Park TY, Park MH, Shin WC, Rhee MH, Seo DW, Cho JY, Kim HM. Anti-metastatic potential of ginsenoside Rp1, a novel ginsenoside derivative. Biol Pharm Bull. 2008;31:1802–1805. doi: 10.1248/bpb.31.1802. [DOI] [PubMed] [Google Scholar]
- 11.Kumar A, Kumar M, Park TY, Park MH, Takemoto T, Terado T, Kitano M, Kimura H. Molecular mechanisms of ginsenoside Rp1-mediated growth arrest and apoptosis. Int J Mol Med. 2009;24:381–386. doi: 10.3892/ijmm_00000243. [DOI] [PubMed] [Google Scholar]
- 12.Kim DH, Choi JH, Park HJ, Park JH, Lee KT. Costunolide induces apoptosis via modulation of cyclin-dependent kinase in HL-60 human leukemia cells. Biomol Ther. 2010;18:178–183. [Google Scholar]
- 13.Cho JY, Fox DA, Horejsi V, Sagawa K, Skubitz KM, Katz DR, Chain B. The functional interactions between CD98, beta1-integrins, and CD147 in the induction of U937 homotypic aggregation. Blood. 2001;98:374–382. doi: 10.1182/blood.V98.2.374. [DOI] [PubMed] [Google Scholar]
- 14.Jung KK, Lee HS, Cho JY, Shin WC, Rhee MH, Kim TG, Kang JH, Kim SH, Hong S, Kang SY. Inhibitory effect of curcumin on nitric oxide production from lipopolysaccharide-activated primary microglia. Life Sci. 2006;79:2022–2031. doi: 10.1016/j.lfs.2006.06.048. [DOI] [PubMed] [Google Scholar]
- 15.Kim EH, Son RH, Myoung HJ, Mar W, Kim WK, Nam KW. The inhibitory effect of baicalin on the short-term food intake in C57BL/ 6J mice. Biomol Ther. 2010;18:171–177. [Google Scholar]
- 16.Kim BH, Lee YG, Park TY, Kim HB, Rhee MH, Cho JY. Ginsenoside Rp1, a ginsenoside derivative, blocks lipopolysaccharide-induced interleukin-1beta production via suppression of the NF-kappaB pathway. Planta Med. 2009;75:321–326. doi: 10.1055/s-0028-1112218. [DOI] [PubMed] [Google Scholar]
- 17.Kim BH, Cho JY. Regulatory role of ginsenoside Rp1, a novel ginsenoside derivative, on CD29-mediated cell adhesion. Planta Med. 2009;75:316–320. doi: 10.1055/s-0028-1112213. [DOI] [PubMed] [Google Scholar]
- 18.Hardy K, Hunt NH. Effects of a redox-active agent on lymphocyte activation and early gene expression patterns. Free Radic Biol Med. 2004;37:1550–1563. doi: 10.1016/j.freeradbiomed.2004.07.020. [DOI] [PubMed] [Google Scholar]
- 19.Palsson-McDermott EM, O’Neill LA. Signal transduction by the lipopolysaccharide receptor, Toll-like receptor-4. Immunology. 2004;113:153–162. doi: 10.1111/j.1365-2567.2004.01976.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee JY, Lee YG, Lee J, Yang KJ, Kim AR, Kim JY, Won MH, Park J, Yoo BC, Kim S, et al. Akt Cys-310-targeted inhibition by hydroxylated benzene derivatives is tightly linked to their immunosuppressive effects. J Biol Chem. 2010;285:9932–9948. doi: 10.1074/jbc.M109.074872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Lee YG, Chain BM, Cho JY. Distinct role of spleen tyrosine kinase in the early phosphorylation of inhibitor of kappaB alpha via activation of the phosphoinositide-3-kinase and Akt pathways. Int J Biochem Cell Biol. 2009;41:811–821. doi: 10.1016/j.biocel.2008.08.011. [DOI] [PubMed] [Google Scholar]
- 22.Kim JY, Lee YG, Kim MY, Byeon SE, Rhee MH, Park J, Katz DR, Chain BM, Cho JY. Src-mediated regulation of inflammatory responses by actin polymerization. Biochem Pharmacol. 2010;79:431–443. doi: 10.1016/j.bcp.2009.09.016. [DOI] [PubMed] [Google Scholar]
- 23.Park EK, Choo MK, Han MJ, Kim DH. Ginsenoside Rh1 possesses antiallergic and anti-inflammatory activities. Int Arch Allergy Immunol. 2004;133:113–120. doi: 10.1159/000076383. [DOI] [PubMed] [Google Scholar]
- 24.Park EK, Shin YW, Lee HU, Kim SS, Lee YC, Lee BY, Kim DH. Inhibitory effect of ginsenoside Rb1 and compound K on NO and prostaglandin E2 biosyntheses of RAW264.7 cells induced by lipopolysaccharide. Biol Pharm Bull. 2005;28:652–656. doi: 10.1248/bpb.28.652. [DOI] [PubMed] [Google Scholar]
- 25.Lee YJ, Jin YR, Lim WC, Park WK, Cho JY, Jang S, Lee SK. Ginsenoside-Rb1 acts as a weak phytoestrogen in MCF-7 human breast cancer cells. Arch Pharm Res. 2003;26:58–63. doi: 10.1007/BF03179933. [DOI] [PubMed] [Google Scholar]
- 26.Park TY, Cho JY. Inhibitory effect of ginsenoside-Rp1, a novel ginsenoside derivative, on the functional activation of macrophage-like cells. Biomol Ther. 2008;16:370–376. [Google Scholar]