Abstract
The effect of Korean red ginseng (KRG) on diabetic renal damage was investigated using streptozotocin (STZ)-induced diabetic rats. The diabetic rats showed loss of body weight gain, and increases in kidney weight and urine volume, whereas the oral administration of KRG at a dose of 100 or 250 mg/kg of body weight per day for 28 d prevented these diabetes-induced physiological abnormalities. Among the kidney function parameters, elevated plasma levels of urea nitrogen and creatinine in diabetic control rats tended to be lowered in KRG-treated rats. In addition, administration of KRG at a dose of 100 mg/kg body weight in the diabetic rats showed significant decreases in serum glucose and tumor necrosis factor-α (TNF-α), implying that KRG might prevent the pathogenesis of diabetic complications caused by impaired glucose metabolism and oxidative stress. KRG also significantly reduced advanced glycation end product (AGE) formation and secretion from kidney of diabetic rats. Furthermore, KRG decreased the levels of N-(carboxymethyl) lysine and expression of AGE receptor. KRG also reduced the overexpression of cyclooxygenase-2 and inducible nitric oxide synthase in the kidney via deactivation of nuclear factor-kappa B. We also found that KRG prevented STZ-induced destruction of glomerular structure and significantly suppressed high glucose-induced fibronectin production. Taken together, KRG ameliorates abnormalities associated with diabetic nephropathy through suppression of inflammatory pathways activated by TNF-α and AGEs. These findings indicate that KRG has a beneficial effect on pathological conditions associated with diabetic nephropathy.
Keywords: Panax ginseng, Streptozotocin, Advanced glycation end products, Nuclear factor-kappa B, Tumor necrosis factor-α
INTRODUCTION
Diabetes mellitus is rapidly becoming a global health issue and may overtake cancer during the next two decades because it covertly affects multiple organ systems during the often long period between onset and diagnosis. A number of complications are associated with poorly controlled hyperglycemia. Diabetic nephropathy (DN) is one of the most common complications of diabetes mellitus. DN is a leading cause of end-stage renal disease. Other than angiotensin-converting enzyme inhibitors and angiotensin receptor blockers, there are few resources in the armamentarium with which treats patients with overt DN. Research points toward a multifactorial etiology and a complex interplay among several pathogenic pathways that can contribute to the declining kidney function in individuals with diabetes [1]. Studies on the pathological mechanisms of DN have revealed that advanced glycation end products (AGEs) play an important role in the pathogenesis of DN through interacting with receptors for advanced glycation end products (RAGE), which activate a series of intracellular signaling pathways. AGEs and RAGE have therefore been considered two potential targets [2].
Ginseng has been used as an herbal medicine and nutritional supplement in East Asia for thousands of years and has gained popularity in the West because of its various pharmacological properties. In recent years, accumulating in vitro and in vivo evidence has indicated that ginseng possesses significant hypoglycemic activities [3-7]. Park et al. [8] reported that the protopanaxadiol derivatives of ginseng significantly reduced the level of glucose-induced fibronectin up-regulation in primary cultured rat mesangial cells. The aim of the present study was to evaluate the preventive effects of Korean red ginseng (KRG) on DN and to elucidate the mechanisms underlying such effects.
MATERIALS AND METHODS
Chemicals
KRG extract was obtained from the Korea Ginseng Corporation (Daejeon, Korea), and dissolved in distilled water. Antibodies against phospho-Erk1/2, phospho-JNK1/2, inducible nitric oxide synthase (iNOS), cyclooxygenase-2 (COX-2), phospho-inhibitor of κB (IκB), IκB, nuclear factor-kappa B (NF-κB), fibronectin (FN) and anti-actin were from Santa Cruz Biotechnology (Santa Cruz, CA, USA). Antibodies against N-(carboxymethyl) lysine (CML) and OxiSelect AGE kit were from Cell Biolabs (San Diego, CA, USA). Monloney murine leukemia virus (MMLV) reverse transcriptase, Taq polymerase and oligo-dT primer were supplied by Promega (Madison, WI, USA). Protein extraction kit, Easy-Blue total RNA extraction kit, and ECL-reagent kit were from Intron Biotechnology (Beverly, MA, USA). Other reagent and chemicals were analytical grade.
Animal treatment and chemistry analysis
The animal experiment protocol was reviewed and approved by Institutional Animal Ethics Committee of Kyung Hee University. Six-week-old SD-Rats (Orient Bio, Seongnam, Korea) were housed in temperature (22±2℃) and humidity-controlled (50±5%) room with a cycle of 12 h light/12 h darkness and free access to food and water. Rats were randomly divided into the following four groups: the normal control group (N); streptozotocin (STZ)-induced diabetic group (STZ); STZ-treated rats were administrated with 100 or 250 mg/kg/d of KRG. A single dose of 65 mg/kg STZ prepared in citrate buffer (0.1 M, pH 4.) was injected intraperitoneally to induced hyperglycemia. Rats were considered to be diabetic if they had more than 250 mg/dL of plasma glucose concentration. In the treatment group, rats were orally administered once a day for 28 d starting 7 d before STZ injection. Blood was collected through cardiac puncture at 28th day, and centrifuged at 3,000 g for 20 min to obtain plasma. Plasma was stored at -70℃ until assays were performed. Urine was obtained using a metabolic cage at 27th day. The plasma concentrations of blood urea nitrogen (BUN), and creatinine (Cr) were determined using commercial kits (Stanbio Laboratory, Boerne, TX, USA) and an automatic analyzer (SmartLab, Mannheim, Germany). Urine samples were collected 24 h after fasting using metabolic cages and used for estimation of glucose and AGE. Renal AGEs were determined using the OxiSelect AGE kit according to the manufacturer’s instruction. The quantity of CML adduct in kidney protein sample was determined by comparing its absorbance with that of a known AGE-bovine serum albumin standard curve and normalized to total protein content in the kidney.
Reverse transcriptase polymerase chain reaction
To determine the levels of gene expression of iNOS, COX-2, and RAGE in kidney tissue, reverse transcriptase polymerase chain reaction (RT-PCR) technique was utilized. Total RNA was isolated form rat kidney using the Easy-Blue kit according to the manufacturer’s instruction. From each sample, total RNA (20 μg) was reversely transcribed into cDNA using MMLV reverse transcriptase and oligo-dT 15 primers. The cDNA fragment was amplified by PCR using the following specific primers: iNOS forward 5’-ATGGCTTCCCCTTGGAAGTTTCTC-3’, reverse 5’-TCCAGGCCATCTT GGTGGCAAAGA-3’; COX-2 forward 5’-TCAAGACACATCAGAAGCGA-3’, reverse 5’-TACCTGAGTGTCTTTGATTG-3’; RAGE forward 5’-CCTGGATACCAACTACTGCTTC-3’, reverse 5’- TACTCCTTGGAGGCCATGTA-3’; GAPDH forward 5’-ACCCCCAAT GTATCCGTTGT-3’, reverse 5’-TACTCCTTGGAGGCCATGTA-3’. PCR reactions were performed in a total volume of 20 μL consisting of 2 μL of the cDNA product, 0.2 mM of each dNTP, 20 pmol of each primer, and 0.8 unit of Taq polymerase. PCR was performed at 95℃ for 30 s followed by 60℃ for 30 s and 72℃ for 1 min. The last cycle was followed by a final extension step at 72℃ for 10 min. The PCR products were electrophoresed in 1% agarose gels at 80 V and stained with 0.5 μg/mL ethidium bromide and scanning densitometry was performed with I-MAX gel image analysis system (Core-Bio, Seoul, Korea). GAPDH was amplified as a control gene.
Western blot analysis
After sacrifice, the kidneys was immediately removed and instantly soaked in liquid nitrogen and stored at -70℃. Protein extracts were prepared using a protein extraction kit and then insoluble proteins were removed by centrifugation at 3,000 g for 20 min. The protein concentration of cell lysates was measured by Bradford method (Bio-Rad, Hercules, CA, USA). Equal amounts of protein (40 μg) were resolved by 8% sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred to a polyvinylidene fluoride membrane (Millipore, Beverly, MA, USA). The membrane was further incubated with specific antibodies such as CML, pErk1/2, pJNK1/2, iNOS, COX-2, pIκB, NF-κB, FN, and actin. The membrane was continuously incubated with appropriate secondary antibodies coupled to horseradish peroxidase and developed in the enhanced chemiluminescence Western detection reagents (Amersham, Uppsala, Sweden). The expressions of the above antibodies were normalized by actin protein expression in the same sample. The immunoreactive bands were visualized and quantified by a densitometric analysis.
Histological analysis
The kidney tissue was removed and fixed in 10 % neutral buffered formalin, embedded in paraffin, sectioned with 5 μM thickness (Leica, Wetzlar, Germany), and then stained with hematoxylin-eosin. Sections were examined under bright field microscope (Olympus, Tokyo, Japan).
Statistical analysis
Results are representative of at least three independent experiments done in triplicate and are expressed as mean±SD. Comparisons between groups were analyzed using Student’s t-test.
RESULTS AND DISCUSSION
Effects of Korean red ginseng on body weight and blood/urine parameters
STZ is a naturally occurring chemical that is particularly toxic to the insulin-producing beta cells of the pancreas in mammals. It is used in medicine to treat certain cancers of the Islets of Langerhans and is used in medical research to produce an animal model of diabetes [9]. In our study, a single dose of 65 mg/kg STZ prevented weight gain and caused renal hypertrophy. The kidney weight of STZ control rats was increased by 32% relative to that of normal rats, and KRG prevented from renal hypertrophy in a dose-dependent manner (Table 1).
Table 1.
The body and kidney weight of each experimental group
| N | STZ | STZ+KRG (mg/kg) | ||
|---|---|---|---|---|
|
| ||||
| 100 | 250 | |||
|
| ||||
| Body weight (g) | 392.5±14.8 | 299.8±17.6*** | 293.4±13.8 | 315.8±13.6 |
| Kidney weight (g) | 2.60±0.11 | 3.44±0.15*** | 3.14±0.08 | 2.94±0.08# |
Data represent the mean±SEM.
N, normal; STZ, streptozotocin; KRG, Korean red ginseng.
***p<0.001 vs. N and #p<0.05 vs.STZ.
A single administration of STZ damaged β cells, resulting in insulin levels that were 31% lower than those of normal rats. The blood glucose levels were consequently elevated by 2.09 times relative to the levels in normal rats. These abnormalities were improved when STZ-induced diabetic rats were treated with KRG extract for 28 d (Table 2). The significantly elevated urine volume and glucose levels were also reduced in KRG-treated rats. However, the modestly elevated BUN and Cr levels in the STZ control rats were not statistically different from the levels in the KRG-treated rats. In addition, STZ dramatically increased the levels of plasma tumor necrosis factor-α (TNF-α) by 4.2 times compared to control rats (Table 2).
Table 2.
The blood and urine chemistry of each experimental group
| N | STZ | STZ+KRG (mg/kg) | ||
|---|---|---|---|---|
|
| ||||
| 100 | 250 | |||
|
| ||||
| Plasma parameters | ||||
| Glucose (mg/dL) | 221.20±11.2 | 461.7±61.7* | 335.5±60.4 | 272.3±60.6# |
| Insulin (ng/mL) | 11.2±0.2 | 7.8±1.3* | 13.5±0.8# | 10.1±1.1 |
| AGE (μg/dL) | 1.09±0.5 | 1.66±0.01* | 1.26±0.07# | 1.36±0.3 |
| TNF-α (pg/mL) | 20.0±0.02 | 84.0±6.3** | 19.0±0.44### | 35.0±16.7# |
| BUN (mg/dL) | 6.53±0.3 | 7.91±0.3 | 6.92±0.4 | 7.3±0.2 |
| Cr (mg/dL) | 0.75±0.03 | 0.84±0.01* | 0.80±0.02 | 0.86±0.01 |
| Urine parameters | ||||
| Volume (mL) | 10.0±2.5 | 190.0±6.2*** | 105.70±21.9# | 152.0±2.7### |
| Glucose (mg/dL) | 10.9±1.6 | 668.5±26.7*** | 329.6±64.5# | 427.1±29.5# |
| AGE (μg/mL) | 1.86±0.12 | 3.1±0.4** | 2.02±0.4# | 3.15±1.2 |
Data represent the mean±SEM.
N, normal; STZ, streptozotocin; KRG, Korean red ginseng; AGE, advanced glycation end product; TNF-α, tumor necrosis factor-α; BUN, blood urea nitrogen.
*p<0.05, **p<0.01, ***p<0.001 vs. N and #p<0.05, ### p<0.001 vs. STZ.
TNF-α is a cytokine involved in systemic inflammation and is a member of a group of cytokines that stimulate the acute phase reaction. When STZ-induced diabetic rats were treated with 100 mg/kg KRG, the TNF-α level in the plasma was decreased to the level of control rats, indicating that KRG has beneficial metabolic effects through the suppression of TNF-α- mediated inflammatory signaling pathways.
Effects of Korean red ginseng on blood/urine advanced glycation end products and renal protein expression
AGEs are the result of a chain of chemical reactions after an initial glycation reaction. AGEs can be formed external to the body (exogenously) by heating (e.g., cooking) [10] or inside the body (endogenously) through normal metabolism and aging. Under certain pathologic conditions (e.g., oxidative stress due to hyperglycemia in patients with diabetes), AGE formation can be increased beyond normal levels. AGEs are now known to play a role as proinflammatory mediators in gestational diabetes [11]. AGEs may be more or less, reactive than the initial sugars from which they were derived. They are absorbed by the body during digestion with approximately 30% efficiency. Many cells in the body (for example, endothelial cells, smooth muscle, and cells of the immune system) from tissues such as the lung, liver, kidney, and peripheral blood bear the RAGE that, when binding by AGEs, contributes to age- and diabetes-related chronic inflammatory diseases such as atherosclerosis, asthma, arthritis, myocardial infarction, nephropathy, retinopathy, periodontitis, and neuropathy [12,13]. There may be some chemicals, such as aminoguanidine, that limit the formation of AGEs by reacting with 3-deoxyglucosone [14]. In this study, we investigated the effects of KRG extract on AGEs formation. As shown in Table 2, the AGEs levels in the plasma and urine were found to be increased by 53% and 67%, respectively, when compared to those in STZ control rats. This pathogenic state of high glucose-induced AGE formation and secretion into the urine was ameliorated in KRG-treated rats, although a dose-dependent effect was not observed. Increased levels of AGEs in the kidney of STZ control rats were markedly decreased in the KRG-treated rats (Fig. 1A).
Fig. 1. Effects of Korean red ginseng (KRG) extract on (A) advanced glycation end products (AGEs), (B) N-(carboxymethyl) lysine (CML), and (C) receptors for advanced glycation end products (RAGE) protein and gene expressions in kidney of SD rats. N, normal rats; STZ, streptozotocin, diabetic control rats; KRG, diabetic rats treated with KRG (100 or 250 mg/kg/d). *p<0.05, **p<0.01, ***p<0.001 compared with STZ.
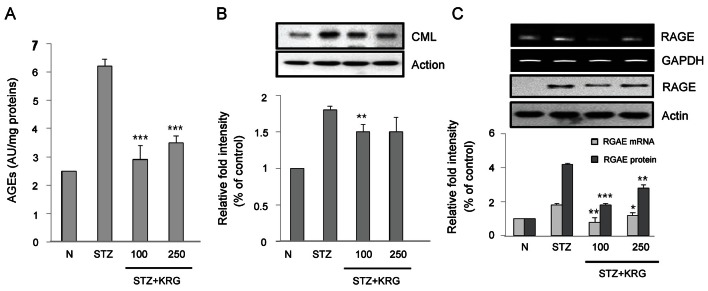
Next, the renal mRNA, protein contents of AGEs, CML, a major AGE in human tissues, and RAGE were determined by RT-PCR and Western blot analysis, respectively. The gene and protein expression levels of CML and RAGE in KRG-treated rats were significantly reduced relative to the expression levels in the STZ control rats (Fig. 1B, C). These results suggest that KRG may have a beneficial effect on diabetic complications by preventing AGE formation and inhibiting the binding of AGEs to their receptor, because the binding of AGEs to RAGE contributes to diabetes-related chronic inflammatory diseases such as nephropathy [2].
Effects of Korean red ginseng on renal inflammatory biomarkers
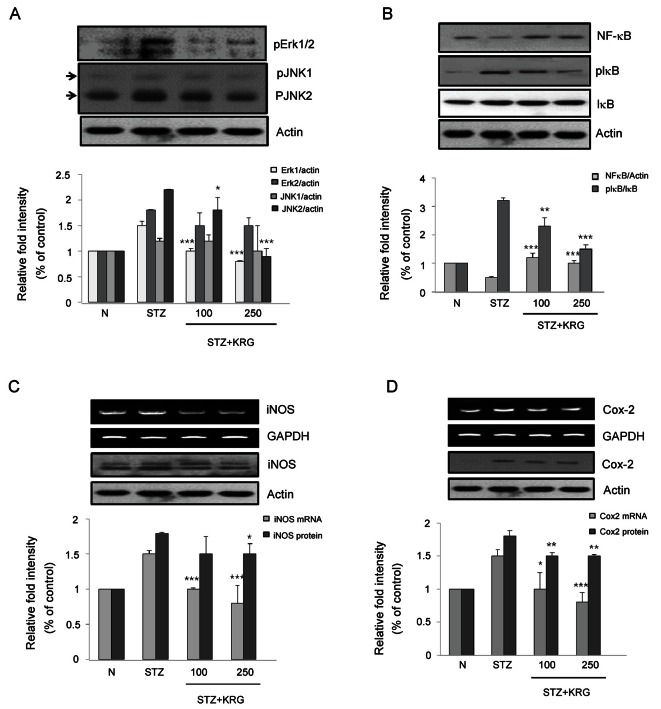
Table 2 shows that the KRG extract dramatically decreased the plasma levels of TNF-α, a cytokine involved in systemic inflammation, indicating that TNF-α-mediated inflammatory pathways could be modulated by KRG. This anti-inflammatory activity might be one of the mechanisms of the renal protective effect of KRG. Of the three major mitogen-activated protein kinases (MAPKs), the phosphorylated forms of Erk and Jun were overexpressed in the kidneys of STZ-treated control rats. Both of these MAPKs were significantly suppressed in KRG-treated diabetic rats (Fig. 2A).
Fig. 2. Effects of Korean red ginseng (KRG) extract on (A) mitogen-activated protein kinases (MAPKs), (B) nuclear factor-kappa B (NF-κB), (C) inducible nitric oxide synthase (iNOS), and (D) cyclooxygenase-2 (COX-2) protein and gene expressions in kidney of SD rats. N, normal rats; STZ, streptozotocin, diabetic control rats; KRG, diabetic rats treated with KRG (100 or 250 mg/kg/d). *p<0.05, **p<0.01, ***p<0.001 compared with STZ. IκB, inhibitor of κB.
Next, effects of KRG on the NF-κB-mediated inflammation cascade were investigated because NF-κB is a well-known transcription factor involved in the inflammatory response [15]. NF-κB activity is under the control of signaling from extracellular stimuli such as lipopolysaccharide and TNF-α. IκB molecules sequester NF-κB in the cytosol of resting cells and prevent its nuclear localization and transcriptional function [15]. The phosphorylation of IκB molecules by inhibitor of κB kinase promotes IκB degradation and releases NF-κB, which then translocates to the nucleus to promote the transcription of target genes, such as iNOS and COX-2 [15]. As shown in Fig. 2B, IκB was significantly phosphorylated and thus NF-κB was released in the renal tissue of STZ control rats. Therefore, when the cells were activated by extracellular stimuli, the cytosolic NF-κB protein level would be decreased because activated NF-κB is free to enter the nucleus (Fig. 2B). This activated NF-κB signaling pathway was blocked by KRG treatment. The gene and protein expression levels of iNOS and COX-2, known target molecules for NF-κB, were significantly suppressed in KRG-treated rats relative to the expression levels in STZ control rats (Fig. 2C, D). These findings further demonstrate that the MAPK and NF-κB pathways are activated in diabetic kidneys and suggest that KRG could suppress the activation of the MAPK and NF-κB pathways during diabetic renal injury.
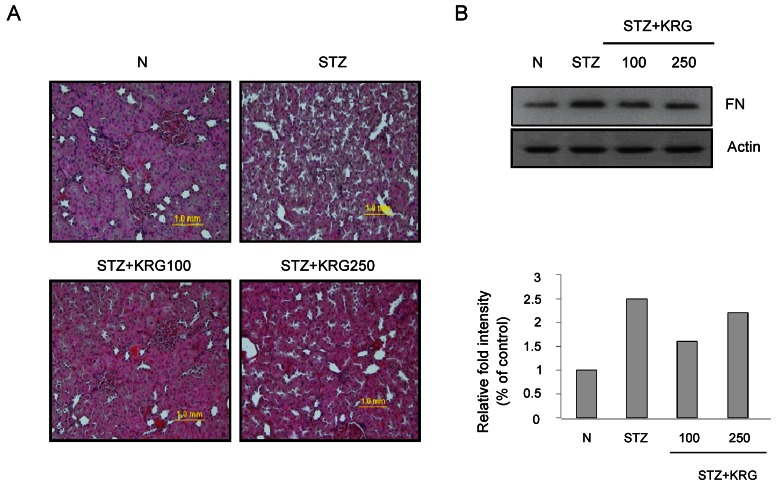
Effects of Korean red ginseng on renal morphology and fibronectin protein expression
DN is a morbid microvascular complication associated with diabetes and is the most common cause of end-stage renal disease. In DN, the accumulation of extracellular matrix (ECM) components in the glomerular mesangium and tubulointerstitium causes early glomerular hypertrophy, as well as later glomerulosclerosis and tubulointerstitial fibrosis [16,17]. Glomerular mesangial cells (GMCs) play important roles in the physiological and pathological processes of the kidneys. GMC proliferation and hypertrophy, ECM accumulation, and the consequent renal fibrosis have been recognized as major pathogenic events in the progression of renal failure in individuals with DN [18,19]. As one of the important components of ECM, FN is often used as an index to evaluate the extent of matrix accumulation. The inhibition of FN production is regarded as an effective strategy to ameliorate DN. Here, the glomerular histology of SD rats treated with STZ or STZ plus KRG extract was examined. The representative glomerular histology of H&E-stained sections is shown in Fig. 3A. Compared with age-matched control rats, STZ-induced rats exhibited prominent destruction of the glomerular structure. In the KRG-treated group, the level of glomerular destruction was significantly less than that in STZ-induced diabetic rats (Fig. 3A). The accumulation of ECM, represented by increased FN expression, in the kidneys was observed in the diabetic group, and was significantly reduced by KRG treatment (Fig. 3B).
Fig. 3. Glomerular injury in streptozotocin (STZ)-induced diabetic kidney. (A) Glomerular histopathology analysis by H&E staining. The pictures display representative glomeruli of H&E-stained sections in control (N), diabetic (STZ), and Korean red ginseng (KRG) groups at an original magnification of 100. (B) The protein expression levels of fibronectin (FN) in the rat kidneys were detected by Western blot analysis.
Collectively, these results confirmed the presence of renal injury characterized by renal hypertrophy, ECM accumulation, glomerulosclerosis, and renal dysfunction in STZ-induced diabetic rats as expected. KRG could ameliorate the renal injury and down-regulate FN expression in STZ-induced diabetic rats.
In summary, we demonstrated that KRG prevented the formation of AGEs and the expression of TNF-α, thus blocking the MAPK and NF-κB-mediated inflammatory pathways. As a major complication of diabetes, DN often leads to end-stage renal failure and high mortality, and finding effective treatment and prevention methods for DN is a major challenge facing modern medicine. Our study provides valuable information needed for understanding preventive mechanism of KRG against STZ-induced renal injury.
Acknowledgments
This study was supported by the 2010 grant from the Korean Society of Ginseng.
References
- 1.Karihaloo A. Anti-fibrosis therapy and diabetic nephropathy. Curr Diab Rep. 2012;12:414–422. doi: 10.1007/s11892-012-0290-7. [DOI] [PubMed] [Google Scholar]
- 2.Zhou X, Wang B, Zhu L, Hao S. A novel improved therapy strategy for diabetic nephropathy: targeting AGEs. Organogenesis. 2012;8:18–21. doi: 10.4161/org.19332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yang CY, Wang J, Zhao Y, Shen L, Jiang X, Xie ZG, Liang N, Zhang L, Chen ZH. Anti-diabetic effects of Panax notoginseng saponins and its major anti-hyperglycemic components. J Ethnopharmacol. 2010;130:231–236. doi: 10.1016/j.jep.2010.04.039. [DOI] [PubMed] [Google Scholar]
- 4.Yuan HD, Kim SJ, Chung SH. Beneficial effects of IH-901 on glucose and lipid metabolisms via activating adenosine monophosphate-activated protein kinase and phosphatidylinositol-3 kinase pathways. Metabolism. 2011;60:43–51. doi: 10.1016/j.metabol.2009.12.024. [DOI] [PubMed] [Google Scholar]
- 5.Lee HJ, Lee YH, Park SK, Kang ES, Kim HJ, Lee YC, Choi CS, Park SE, Ahn CW, Cha BS, et al. Korean red ginseng (Panax ginseng) improves insulin sensitivity and attenuates the development of diabetes in Otsuka Long-Evans Tokushima fatty rats. Metabolism. 2009;58:1170–1177. doi: 10.1016/j.metabol.2009.03.015. [DOI] [PubMed] [Google Scholar]
- 6.Luo JZ, Luo L. Ginseng on hyperglycemia: effects and mechanisms. Evid Based Complement Alternat Med. 2009;6:423–427. doi: 10.1093/ecam/nem178. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 7.Vuksan V, Sung MK, Sievenpiper JL, Stavro PM, Jenkins AL, Di Buono M, Lee KS, Leiter LA, Nam KY, Arnason JT, et al. Korean red ginseng (Panax ginseng) improves glucose and insulin regulation in well-controlled, type 2 diabetes: results of a randomized, double-blind, placebo-controlled study of efficacy and safety. Nutr Metab Cardiovasc Dis. 2008;18:46–56. doi: 10.1016/j.numecd.2006.04.003. [DOI] [PubMed] [Google Scholar]
- 8.Park MJ, Bae CS, Lim SK, Kim DI, Lim JC, Kim JC, Han HJ, Moon JH, Kim KY, Yoon KC, et al. Effect of protopanaxadiol derivatives in high glucose-induced fibronectin expression in primary cultured rat mesangial cells: role of mitogen-activated protein kinases and Akt. Arch Pharm Res. 2010;33:151–157. doi: 10.1007/s12272-010-2237-3. [DOI] [PubMed] [Google Scholar]
- 9.Rossini AA, Like AA, Chick WL, Appel MC, Cahill GF Jr. Studies of streptozotocin-induced insulitis and diabetes. Proc Natl Acad Sci U S A. 1977;74:2485–2489. doi: 10.1073/pnas.74.6.2485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Koschinsky T, He CJ, Mitsuhashi T, Bucala R, Liu C, Buenting C, Heitmann K, Vlassara H. Orally absorbed reactive glycation products (glycotoxins): an environmental risk factor in diabetic nephropathy. Proc Natl Acad Sci U S A. 1997;94:6474–6479. doi: 10.1073/pnas.94.12.6474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Pertynska-Marczewska M, Glowacka E, Sobczak M, Cypryk K, Wilczynski J. Glycation endproducts, soluble receptor for advanced glycation endproducts and cytokines in diabetic and non-diabetic pregnancies. Am J Reprod Immunol. 2009;61:175–182. doi: 10.1111/j.1600-0897.2008.00679.x. [DOI] [PubMed] [Google Scholar]
- 12.Barlovic DP, Soro-Paavonen A, Jandeleit-Dahm KA. RAGE biology, atherosclerosis and diabetes. Clin Sci (Lond) 2011;121:43–55. doi: 10.1042/CS20100501. [DOI] [PubMed] [Google Scholar]
- 13.Sukkar MB, Ullah MA, Gan WJ, Wark PA, Chung KF, Hughes JM, Armour CL, Phipps S. RAGE: a new frontier in chronic airways disease. Br J Pharmacol. 2012;167:1161–1176. doi: 10.1111/j.1476-5381.2012.01984.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Wells-Knecht KJ, Zyzak DV, Litchfield JE, Thorpe SR, Baynes JW. Mechanism of autoxidative glycosylation: identification of glyoxal and arabinose as intermediates in the autoxidative modification of proteins by glucose. Biochemistry. 1995;34:3702–3709. doi: 10.1021/bi00011a027. [DOI] [PubMed] [Google Scholar]
- 15.Baker RG, Hayden MS, Ghosh S. NF-κB, inflammation, and metabolic disease. Cell Metab. 2011;13:11–22. doi: 10.1016/j.cmet.2010.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Mauer SM, Steffes MW, Ellis EN, Sutherland DE, Brown DM, Goetz FC. Structural-functional relationships in diabetic nephropathy. J Clin Invest. 1984;74:1143–1155. doi: 10.1172/JCI111523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Adler S. Structure-function relationships associated with extracellular matrix alterations in diabetic glomerulopathy. J Am Soc Nephrol. 1994;5:1165–1172. doi: 10.1681/ASN.V551165. [DOI] [PubMed] [Google Scholar]
- 18.Schena FP, Gesualdo L. Pathogenetic mechanisms of diabetic nephropathy. J Am Soc Nephrol. 2005;16 Suppl 1:S30–S33. doi: 10.1681/asn.2004110970. [DOI] [PubMed] [Google Scholar]
- 19.Ichinose K, Kawasaki E, Eguchi K. Recent advancement of understanding pathogenesis of type 1 diabetes and potential relevance to diabetic nephropathy. Am J Nephrol. 2007;27:554–564. doi: 10.1159/000107758. [DOI] [PubMed] [Google Scholar]