Abstract
Korean red ginseng (KRG) is reported to have anti-allergic properties, including beneficial effects on asthma and atopic dermatitis. However, its effect on allergic rhinitis has not been studied extensively. This study examined how KRG affected allergic inflammation of the nasal cavity in an allergic mouse model. A total of 40 Balb/c female mice were divided into four experimental groups according to treatment and allergic state: group 1 (G1), saline only; group 2 (G2), ovalbumin (OVA); group 3 (G3), OVA+KRG; and group 4 (G4), OVA+dexamethasone. Serum IgE levels were significantly lower in the KRG treatment group (G3) than in the allergic group (G2). However, serum IgG1 levels did not differ between G2 and G3. In the nasal lavage fluid, IL-4 and IL-5 levels were significantly lower in G3 than in G2 (p<0.05). H&E and Luna staining revealed that the eosinophil count was lower in G3 and G4 than in G2 (p<0.05). Immunohistochemical staining revealed that there were fewer IL-4-, IL- 5-, and MUC5AC-positive cells in G3 and G4 than in G2 (p<0.05). These results indicate that KRG reduces the nasal allergic inflammatory reaction in an allergic murine model by reducing Th2 cytokines.
Keywords: Panax ginseng, Korean red ginseng, Allergic rhinitis, Mice
INTRODUCTION
Allergic rhinitis is a common upper airway inflammatory disease in children and adults. The most frequent symptoms include nasal congestion, rhinorrhea, nasal itching, and sneezing, which can diminish patient quality of life and involve considerable costs to society. Additionally, these symptoms can have negative effects on sleep and school or work performance, especially in children [1-3]. Allergic rhinitis has also been associated with other medical conditions, such as sinusitis and asthma [4]. The worldwide prevalence of allergic rhinitis is estimated to be approximately 3% to 19%, and its frequency has increased steadily in recent years [4,5]. In Korea, the prevalence of allergic rhinitis ranges from 6% to 10%, furthermore 60% to 80% of asthmatic patients have the disease, which can seriously affect asthma severity and outcome [6-8].
Typically, a nasal corticosteroid spray and oral antihistamines are used to treat the symptoms of allergic rhinitis patients. However, some patients are unsatisfied with these treatments, and some may be unable to use these treatments for various reasons. Complementary and alternative medicine (CAM) has been a popular treatment modality among allergic disease patients [9]. In a study of the prevalence of CAM use, more than one-third of respondents reported using CAM [10], and in a report of specific alternative treatment modalities in adults with asthma or rhinosinusitis, 64% of participants reported using alternative treatments [9].
CAM treatments are thought to affect asthma by reducing inflammation. Korean red ginseng (KRG; the steamed root of Panax ginseng Meyer, family Araliaceae) is used frequently as a CAM in many countries. The major components of ginseng are saponins, also known as ginsenosides. To date, more than 40 ginsenosides have been identified, and they display various biological properties, including anti-inflammatory, anti-allergic, and anti-tumor activities [11-14]. Among these ginsenosides, Rg1, Re, Rf, Rb1, Rb2, Rc, and Rd have well-known biological effects [15].
The ginsenosides Rh1 and Rh2 have demonstrated anti-allergic and anti-inflammatory effects [16,17]. Anti-allergic effects of ginsenosides have been reported in murine asthma and atopic disease models [18-20]. The pathogenesis of allergic rhinitis is very similar to that of asthma and atopic rhinitis. However, the effect of KRG on allergic rhinitis has never been reported. Therefore, in this study we examined how KRG affects the allergic inflammatory reaction in an ovalbumin (OVA)-induced allergic mouse model.
MATERIALS AND METHODS
Animal
A total of 40 female Balb/c mice (6 wk of age) were purchased from Central Lab Animal Inc. (Seoul, Korea) and checked for infections. The average body weight of mice is 21 g. All study protocols adhered to the Gachon University Animal Research Guidelines. The mice were divided into four groups (n=10 per group): group 1 (G1), normal saline; group 2 (G2), OVA+saline; group 3 (G3), OVA+KRG; and group 4 (G4), OVA+dexamethsone.
Experimental design
To develop a control murine model (G1), 300 μL of saline was mixed with 1 mg of Al(OH)3 gel (Alum; Pierce Chemical Co., Rockford, IL, USA). Then, the mice were injected intraperitoneally with 300 μL of the mixture on days 1, 7, and 14. Next, the nasal cavity was dripped with 30 μL saline daily for 14 d.
Development of allergic murine model
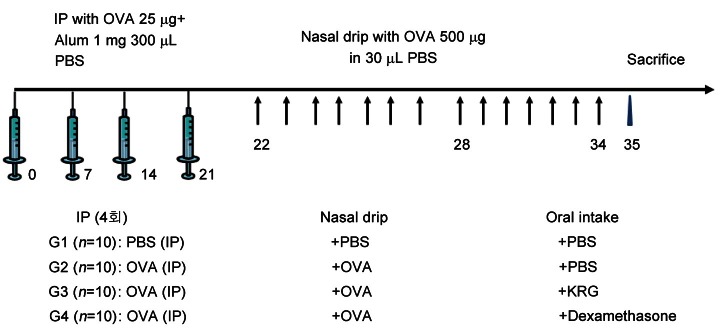
KRG was provided by the Korea Ginseng Corporation (Daejeon, Korea). To develop an allergic murine model, 25 μg of OVA (Sigma-Aldrich, St. Louis, MO, USA) and 1 mg of Al(OH)3 gel (Pierce Chemical Co.) were combined in 300 μL of PBS. Next, mice were injected intraperitoneally with 300 μL of the OVA mixture on days 1, 7, 14 and 21 to induce systemic sensitization. Then, the mice in the OVA group (G2) received a nasal drip of 500 μg OVA in 30 μL phosphate-buffered saline (PBS) daily for 14 d, beginning on day 22. Mice in the OVA+KRG group (G3) also received KRG (2 g/kg per d) according to the previous protocol [21] and were administered every morning from the 22th day to the 35th day for 14 d using orogastric gavage, after which OVA was dripped in the afternoon in the same way as the OVA group. In the OVA+dexamethasone group (G4), dexamethasone (1 mg/kg) was administered instead of KRG using the same method described for G3 (Fig. 1).
Fig. 1. Development of allergic murine model. Mice were injected intraperitoneally with 300 μL of the ovalbumin (OVA) mixture on days 1, 7, 14 and 21 to induce systemic sensitization. Then, the mice in the group 2 (G2) in the OVA G2 received a nasal drip of 500 μg OVA for 14 d, beginning on day 22. Mice in the group 3 (G3) also received Korean red ginseng (KRG) for 14 d, after which OVA was dripped in the same way as the group 2 (G2). In the group 4 (G4), dexamethasone was administered instead of KRG. Group 1 (G1), normal saline; G2, OVA+saline; G3, OVA+KRG; and G4: OVA+dexamethasone. IP, intraperitoneum injection; PBS, phosphate buffered saline.
Collection of serum and nasal lavage fluid
The mice were anesthetized with isoflurane 24 h after the last stimulation and then sacrificed. Approximately 300 to 400 μL of blood was obtained from the ophthalmic artery of each mouse by orbital puncture. The blood sample was stored at 4℃ for 2 to 4 h and then centrifuged (2,000 rpm, 4℃) to obtain serum. To collect the nasal lavage fluid (NALF), the head was extended, the anterior nares were positioned below the table, and an angiocatheter was placed in the trachea. Irrigation through the catheter was performed slowly with 1 mL of saline in the posterior to anterior direction. The lavage fluid was centrifuged (2,000 rpm, 4℃), and 0.5 mL of the supernatant was stored at -70℃.
Extraction of nasal specimen
The head of each mouse was removed and fixed in 10% paraformaldehyde for 24 h. The nasal specimen was decalcified by placing it in a surgipath (Decalcified II; Surgical Medical Industries Inc., Richmond, IL, USA) for 5 d. The head was then sectioned from behind the upper incisor to 2 mm in front of the incisive papilla of the hard palate. After sectioning, blocks were taken for paraffin embedding. Each paraffin block was cut at intervals of 4 μm.
Measurement of ovalbumin-specific IgE and IgG1 in serum
OVA-specific IgE antibody levels were measured using a mouse anti-OVA IgE enzyme-linked immunosorbent assay (ELISA) kit (Alpha Diagnostic International Inc., San Antonio, TX, USA) according to the manufacturer’s protocol. IgG1 was measured using a mouse anti-OVA IgG1 ELISA kit (Alpha Diagnostic International Inc.). Absorbance at 450 nm for OVA-specific IgE and IgG1 was measured using a microplate reader (U-2000; Hitachi, Tokyo, Japan).
Measurement of cytokines in the nasal lavage fluid
The cytokine levels in the NALF were measured using a mouse IL-4 ELISA kit (BioSource International, Camarillo, CA, USA), a mouse IL-5 ELISA kit (BioSource International), mouse IL-10 ELISA kit (BioSource International), and a mouse IFN-γ ELISA kit (PBL BioMedical Laboratory, Piscataway, NJ, USA). Briefly, the lavage fluid was applied to a 96-well microplate that had been coated with antibodies specific for mouse IL-4, IL-5, IL-10, or IFN-γ for 2 h at room temperature. After washing, the biotin conjugate was reacted for 1 h and washed out. Then, the streptavidin-HRP working solution was applied, serially stabilized chromogen was added, and the absorbance at 450 nm was measured.
H&E staining
The nasal specimen was stained with H&E to determine the number of inflammatory cells. Each slide was stained with hematoxylin for 45 s. After washing, the slide was treated with eosin for 20 s. The inflammatory cells were counted at 5 different sites under ×400 magnification.
Luna staining
After deparaffinization of the sample, the specimen was stained with hematoxylin-biebrich-scarlet solution for 5 min. After soaking with 70% ethanol for 5 to 10 s, the specimen was stained with 0.5% lithium carbonate and washed with water. The eosinophils were identified by their scarlet color.
Immunohistochemical staining
Nasal specimens were incubated with 1:200 dilutions of mouse monoclonal anti-human IL-4 antibody (BioVendor Laboratory Medicine Inc., Brno, Czech Republic), purified anti-mouse/human IL-5 antibody (BioLegend, San Diego, CA, USA), or monoclonal anti-human MUC5AC (Neomarker Co., Fremont, CA, USA) for 90 min at room temperature followed by incubation with goat anti-mouse immunoglobulin (Santa Cruz Biotechnology, Santa Cruz, CA, USA) for 1 h at room temperature. Immunoreactive proteins were visualized by incubation with streptavidin-conjugated peroxidase for 15 min. After decolorization using 3,3-diaminobenzidine tetrahydrochloride solution, the specimens were counterstained with hematoxylin. The control plates were processed similarly but without the primary antibody. The average ratio of the immunopositive area to the total area of the nasal specimen was determined under a microscope at ×100 magnification.
Statistical analysis
Statistical analyses of pathologic results in the nasal septum and cytokines in the NALF were conducted using the Kruskal-Wallis test (SPSS ver. 10.0; SPSS Inc., Chicago, IL, USA). Differences between groups were considered statistically significant at p<0.05.
RESULTS
Serum levels of ovalbumin-specific IgE and IgG1
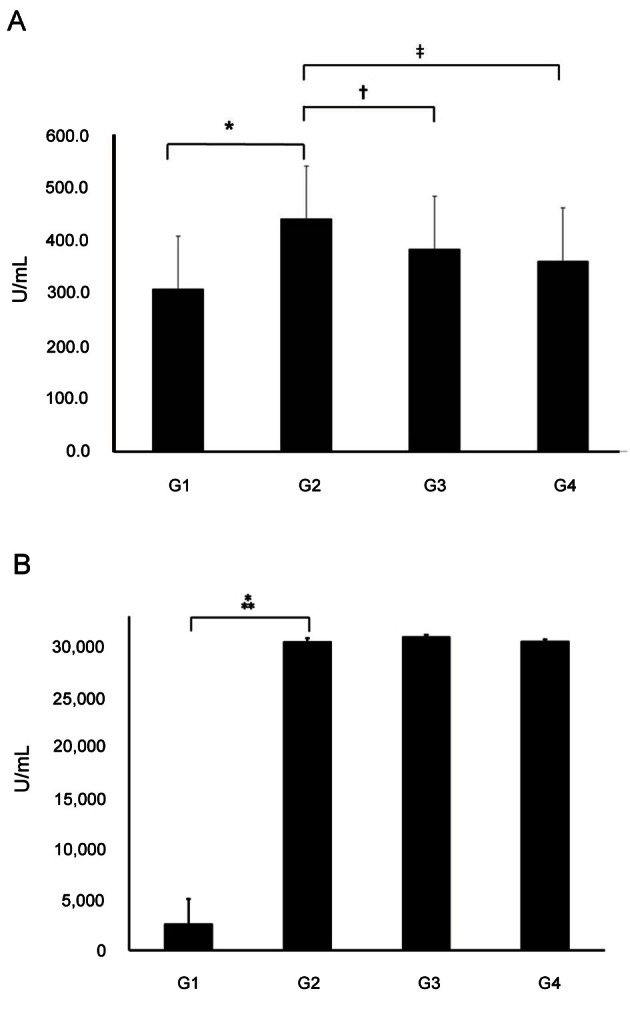
OVA-specific IgE levels were significantly higher in G2 (439.28±11.61 U/mL) than in G1 (306.93±11.15 U/mL) (p<0.01). IgE levels were significantly lower in G3 (382±7.77 U/mL) and G4 (359.77±11.04 U/mL) compared to G2 (p<0.01). IgG1 levels were significantly higher in G2 compared to G1. However, there were no significant differences in the IgG1 levels of G2 and G3 (Fig. 2).
Fig. 2. Serum levels of ovalbumin (OVA)-specific IgE and IgG1. (A) OVA-specific IgE levels were significantly higher in group 2 (G2) than in group 1 (G1). IgE levels were significantly lower in group 3 (G3) and group 4 (G4) compared to G2. (B) IgG1 levels were significantly higher in G2 compared to G1. However, there were no significant differences in the IgG1 levels of G2 and G3 (*p<0.001, †p=0.0022, ‡p=0.0002, ⁂p=0.0006). G1, normal saline; G2, OVA+saline; G3, OVA+Korean red ginseng; and G4, OVA+dexamethasone (graph: average±standard deviation).
Concentrations of cytokines in nasal lavage fluid
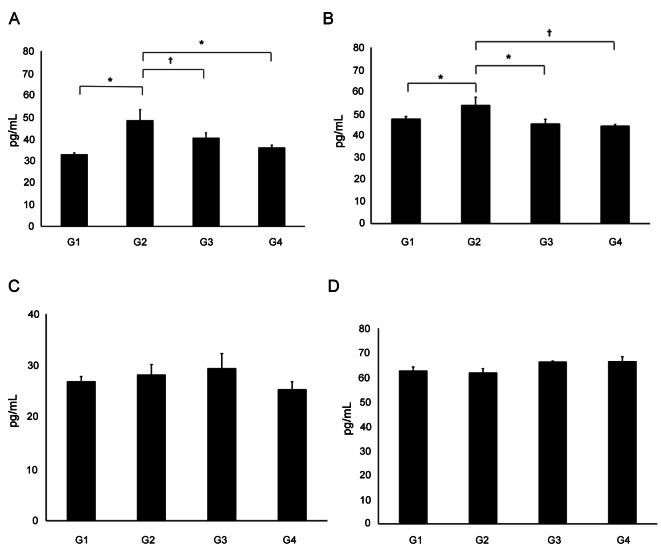
The concentration of IL-4 in the NALF was significantly elevated in G2 (46.62±5.33 pg/mL) compared to G1 (32.83±0.65 pg/mL). The IL-4 concentrations were also significantly lower in G3 (40.28±2.39 pg/mL) and G4 (37.12±2.62 pg/mL) than in G2 (p<0.05). The IL-5 concentration in G2 (53.64±3.71 pg/mL) was significantly higher than that in G1 (47.39±1.26 pg/mL), G3 (44.52±2.21 pg/mL) or G4 (44.23±0.83 pg/mL) (p<0.05 for all). However, there were no significant differences in IL-10 or IFN-γ concentrations between G2 and G3 (p>0.05) (Fig. 3).
Fig. 3. Concentration of cytokines in nasal lavage fluid. (A) The concentration of IL-4 in the nasal lavage fluid was significantly elevated in group 2 (G2) compared to group 1 (G1). The IL-4 concentrations were also significantly lower in group 3 (G3) and group 4 (G4) than in G2. (B) The IL-5 concentration in G2 was significantly higher than that in G1, G3 or G4. (C,D) There were no significant differences in IL-10 or IFN-γ concentrations between G2 and G3 (*p<0.01, †p<0.05). G1, normal saline; G2, ovalbumin (OVA)+saline; G3, OVA+Korean red ginseng; and G4, OVA+dexamethasone (graph: average±standard deviation).
H&E and Luna staining
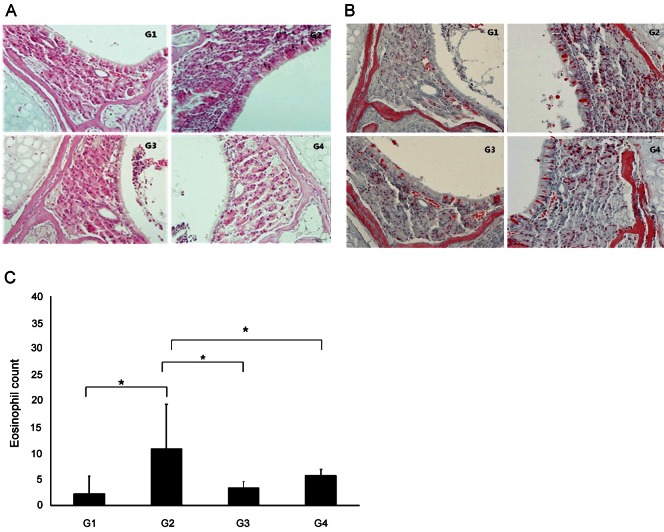
The eosinophil count was significantly higher in G2 (10.83±8.44%) than in G1 (2.25±3.30%) (p<0.05). The eosinophil count was also significantly lower in G3 (3.38±1.10%) and G4 (5.75±1.19%) than in G2 (p<0.05) (Fig. 4).
Fig. 4. H&E and Luna staining. The eosinophil count was significantly higher in group 2 (G2) than in group 1 (G1) (p<0.05) (A) H&E staining (×400), (B) Luna staining (×400). (C) The eosinophil count was also significantly lower in group 3 (G3) and group 4 (G4) than in G2 (*p<0.05). G1, normal saline; G2, ovalbumin (OVA)+saline; G3, OVA+ Korean red ginseng; G4, OVA+dexamethasone (black arrow, eosinophil; graph, average±standard deviation).
Immunohistochemical staining
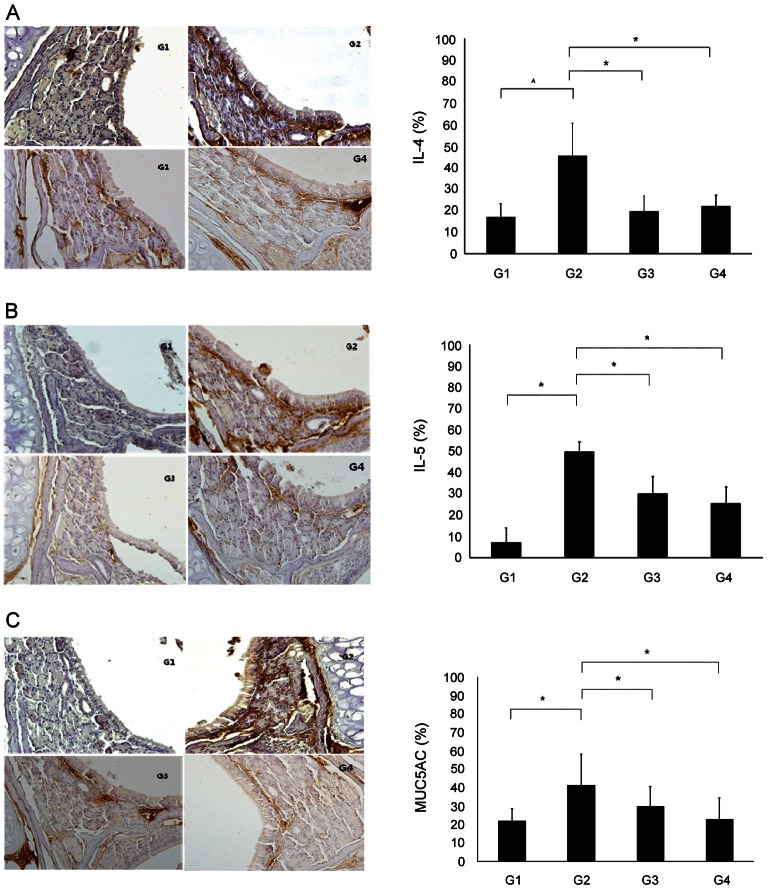
IL-4-, IL-5-, and MUC5AC-positive cells were found in the epithelium and submucosal gland, and the numbers of these cells were increased in the allergic mouse model in this study. The number of IL-4-positive cells was significantly higher in G2 (45.46±15.35%) than in G1 (17.08±6.09%) (p<0.05). Additionally, the number of these cells was significantly lower in G3 (19.53±7.40%) and G4 (22.08±5.11%) than in G2 (p<0.05). Significantly fewer IL-5-positive cells appeared in G3 (30.20±7.75%) and G4 (25.50±7.75%) compared to G2 (50.02±4.32) (p<0.05). MUC5AC staining revealed a significantly greater number of positive cells in G2 (41.53±16.46%) than in G1 (22.18±6.47%) (p<0.05). The number of positive cells was significantly lower in G3 (30.00±10.70%) and G4 (23.04±11.59%) than in G2 (41.53±16.46) (p<0.05) (Fig. 5).
Fig. 5. Immunohistochemical staining (×400). IL-4-, IL-5- and MUC5AC-positive cells were found in the epithelium and submucosal gland, and the numbers of these cells were increased in group 2 (G2). (A) The number of IL-4-positive cells was significantly higher in G2 than in group 1 (G1). Additionally, the number of these cells was significantly lower in group 3 (G3) and group 4 (G4) than in G2. (B) The number of IL-5-positive cells was significantly fewer IL-5-positive cells appeared in G3 and G4 compared to G2. (C) MUC5AC staining revealed a significantly greater number of positive cells in G2 than in G1. The number of positive cells was significantly lower in G3 and G4 than in G2 (*p<0.05). G1, normal saline; G2, ovalbumin (OVA)+saline; G3, OVA+ Korean red ginseng; G4, OVA+dexamethasone (graph: average±standard deviation).
DISCUSSION
Our results revealed that IgE levels were significantly lower in the KRG-treated group than in the control group. However, IgG1 levels did not decrease after KRG treatment. Previous studies have demonstrated various effects of ginsenosides on IgE and IgG production in OVA-sensitized mice. For example, orally administered ginseng extract significantly attenuated the increases in OVA-specific IgG levels in OVA-sensitized mice but had no effect on OVA-specific IgE levels in the splenocytes of the OVA-sensitized mice [22]. Meanwhile, the ginsenoside Rd can significantly increase the production of OVA-specific IgG, IgG1, and IgG2b levels in mice immunized with OVA, which exhibit immunological adjuvant activity [23]. In an mouse model of atopic dermatitis, the topical application of Rb1 did not reduce total serum IgE levels [24]. In contrast to these previous reports, our findings indicate that KRG can reduce total serum IgE production but does not appear to induce the production of IgG.
Allergic rhinitis is a localized type I hypersensitivity, expressed as IgE mediated immune reactions, in atopic patients. This reaction occurs through mast cell activation and degranulation following the cross-linking of IgE bound to its high-affinity receptor, FceRI, by a specific allergen [25]. IgG acts as blocking antibodies, binds to the high affinity receptor site instead of IgE and prevents the binding of IgE. The inflammatory mediators (histamine, kinins and proteases) released from mast cells act on the nasal mucosa to elicit the symptoms of sneezing, congestion and rhinorrhea within minutes of allergen exposure [26]. Thus, the elevation of an allergen-specific IgE reflects a hypersensitivity to that allergen. An influx of inflammatory cells, monocytes, T lymphocytes, basophils, and eosinophils to the nasal mucosa occurs within several hours after antigen exposure [26,27]. Tissue eosinophilia mediated by the TH2 cytokine IL-5 is a typical feature of allergic rhinitis, and the release of toxic products from eosinophils prolongs the allergic symptoms [28,29]. The Th1 cytokines IL-4, IL-5 and IL-13 promote eosinophil recruitment and prolonged tissue survival.
The ginseng extract prevented a reduction in the ratio of IFN-γ (Th1) to IL-4 (Th2) in splenocytes and increased the numbers of small intestinal CD8-, IFN-γ-, and IgA-positive cells in the OVA-sensitized mice [22]. The ginsenoside Rd from the roots of P. notoginseng not only significantly increased the production of the Th2 cytokines IL-4 and IL-5; it also strongly increased the production of the Th1 cytokines IL-2, IFN-γ, and TNF-α in the serum of the OVA-immunized mice. These findings suggest that Rd has immunological adjuvant activity and that it stimulated both Th1 and Th2 immune responses by regulating the production of Th1 and Th2 cytokines [23]. We found that the IL-4 and IL-5 concentrations in the NALF were significantly lower in KRG-treated mice than in the control mice, but the two groups exhibited similar IL-10 and IFN-γ concentrations. Thus, our results suggest that the major components of KRG, Rg1 and Rb1 (4 mg/g), decrease Th1 cytokine activation.
The ginsenoside Rb1 is reported to simultaneously stimulate Th1 and Th2 immune responses. In a mouse model of atopic dermatitis, the topical application of Rb1 on skin lesions improved the lesions. Rb1 also suppressed the mRNA expression of IL-4, IL-10 and nuclear factor of activated T cells in splenocytes [24]. An extract of the root of P. notoginseng, which contains the ginsenosides Rb1, Rg1, and Rd and the notoginsenoside R1, significantly inhibited the inflammatory response by blocking the production of NO, PGE2, TNF-α, and IL-1β [30] and by suppressing the NF-κB signaling pathway [30,31]. In another study, an extract from P. quinquefolius (American ginseng) inhibited nitric oxide synthase expression by blocking the activation of signal transducer and activator of transcription signaling [32]. Ginsan, a polysaccharide derived from P. ginseng, has an anti-asthmatic effect, decreasing IL-5 levels in the supernatant of cultured splenocytes, but it did not alter IFN-γ and serum IgE levels in an OVA-induced murine asthmatic model. Ginsan also reduced airway responsiveness, remodeling and eosinophilia [18]. An aqueous extract of KRG appears to have a potent inhibitory effect in oxazolone-induced mouse dermatitis [13]. In our study, only the cells that were immuno-positive for Th2 cytokines were reduced in number in the KRG group compared to the control. Therefore, the major mechanism of KRG activity is likely dependent on the inhibition of Th2 cytokines.
MUC5AC is known as a mucus gene that plays a protective role in the airway by capturing bacteria and viruses and preserving the humidity of the airway. In many airway inflammatory diseases, mucus hypersecretion has aggravated the airway obstruction and infection [33-35]. Thus, the decrease of MUCAC expression observed in this study indicates that allergic inflammation was decreased by KRG. In this study, H&E and Luna staining revealed fewer eosinophils in the KRG treatment group than in untreated controls. KRG reduced the eosinophil counts in the nasal epithelium, suggesting that it might be effective in preventing increases in eosinophil recuritment through anti-allergic activity. The oral administration of ginseng significantly decreased the numbers of mast cells and had a similar effect on the thicknesses of the basement membrane, epithelium, and subepithelial smooth muscle compared with the dexamethasone group in a murine model of chronic asthma [21]. Two ginsenosides, Rb1 and Rg1, have been shown to exhibit glucocorticoid-like activity. The ginsenoside Rg1 is known to be a functional ligand of the glucocorticoid receptor [36]. Rg1 can also down-regulate the glucocorticoid receptor, exhibits synergistic effects with cAMP [37] and promotes the immune response to OVA in Balb/c mice [38]. Our study used an extract of KRG that is composed mainly of Rg1 and Rb1 (4 mg/g). One possible mechanism for the effect of KRG on allergic rhinitis observed in this study is the glucocorticoid-like effect of these ginsenosides. The topical application of KRG and its constituent ginsenosides Rh2 and Rg3 significantly reduced the extensive mast cell infiltration of the ear in a 2,4,6-trinitro-1-chlorobenzene-induced model of atopic dermatitis. Together, these findings suggest that the local application of ginsenosides also inhibits the local activation of mast cells [19,20].
In conclusion, KRG suppressed allergic inflammation in a murine model of allergic rhinitis by inhibiting the Th2 cytokines IL-4 and IL-5. These findings suggest that KRG might be useful for the treatment of allergic rhinitis. Further study will be needed to assess how treatment with KRG affects patients with allergic rhinitis.
Acknowledgments
This study was supported by the 2010 grant from the Korean Society of Ginseng.
References
- 1.Nathan RA. The burden of allergic rhinitis. Allergy Asthma Proc. 2007;28:3–9. doi: 10.2500/aap.2007.28.2934. [DOI] [PubMed] [Google Scholar]
- 2.Ciprandi G, Klersy C, Cirillo I, Marseglia GL. Quality of life in allergic rhinitis: relationship with clinical, immunological, and functional aspects. Clin Exp Allergy. 2007;37:1528–1535. doi: 10.1111/j.1365-2222.2007.02809.x. [DOI] [PubMed] [Google Scholar]
- 3.Pratt EL, Craig TJ. Assessing outcomes from the sleep disturbance associated with rhinitis. Curr Opin Allergy Clin Immunol. 2007;7:249–256. doi: 10.1097/ACI.0b013e3280f3c09f. [DOI] [PubMed] [Google Scholar]
- 4.Skoner DP. Allergic rhinitis: definition, epidemiology, pathophysiology, detection, and diagnosis. J Allergy Clin Immunol. 2001;108(1 Suppl):S2–S8. doi: 10.1067/mai.2001.115569. [DOI] [PubMed] [Google Scholar]
- 5.Worldwide variation in prevalence of symptoms of asthma, allergic rhinoconjunctivitis, and atopic eczema: ISAAC. The International Study of Asthma and Allergies in Childhood (ISAAC) Steering Committee. Lancet. 1998;351:1225–1232. [PubMed] [Google Scholar]
- 6.Cho SH, Kim YK, Sohn JW, Kim WK, Lee SR, Park JK, Min KU, Ha MN, Ahn YO, Jee YK, et al. Prevalence of chronic rhinitis in Korean children and adolescents. Korean J Asthma Allergy Clin Immunol. 1999;19:452–458. [Google Scholar]
- 7.Suh M, Kim HH, Sohn MH, Kim KE, Kim C, Shin DC. Prevalence of allergic diseases among Korean school-age children: a nationwide cross-sectional questionnaire study. J Korean Med Sci. 2011;26:332–338. doi: 10.3346/jkms.2011.26.3.332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Park HS, Choi GS, Cho JS, Kim YY. Epidemiology and current status of allergic rhinitis, asthma, and associated allergic diseases in Korea: ARIA Asia-Pacific workshop report. Asian Pac J Allergy Immunol. 2009;27:167–171. [PubMed] [Google Scholar]
- 9.Blanc PD, Trupin L, Earnest G, Katz PP, Yelin EH, Eisner MD. Alternative therapies among adults with a reported diagnosis of asthma or rhinosinusitis: data from a population-based survey. Chest. 2001;120:1461–1467. doi: 10.1378/chest.120.5.1461. [DOI] [PubMed] [Google Scholar]
- 10.Tindle HA, Davis RB, Phillips RS, Eisenberg DM. Trends in use of complementary and alternative medicine by US adults: 1997-2002. Altern Ther Health Med. 2005;11:42–49. [PubMed] [Google Scholar]
- 11.Ernst E. Panax ginseng: an overview of the clinical evidence. J Ginseng Res. 2010;34:259–263. [Google Scholar]
- 12.Ro JY, Ahn YS, Kim KH. Inhibitory effect of ginsenoside on the mediator release in the guinea pig lung mast cells activated by specific antigen-antibody reactions. Int J Immunopharmacol. 1998;20:625–641. doi: 10.1016/s0192-0561(98)00062-9. [DOI] [PubMed] [Google Scholar]
- 13.Bae EA, Han MJ, Shin YW, Kim DH. Antiallergic and antipsoriatic effects of Korean red ginseng. J Ginseng Res. 2005;29:80–85. [Google Scholar]
- 14.Wakabayashi C, Hasegawa H, Murata J, Saiki I. In vivo antimetastatic action of ginseng protopanaxadiol saponins is based on their intestinal bacterial metabolites after oral administration. Oncol Res. 1997;9:411–417. [PubMed] [Google Scholar]
- 15.Kim SN, Ha YW, Shin H, Son SH, Wu SJ, Kim YS. Simultaneous quantification of 14 ginsenosides in Panax ginseng C.A. Meyer (Korean red ginseng) by HPLC-ELSD and its application to quality control. J Pharm Biomed Anal. 2007;45:164–170. doi: 10.1016/j.jpba.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 16.Park EK, Choo MK, Han MJ, Kim DH. Ginsenoside Rh1 possesses antiallergic and anti-inflammatory activities. Int Arch Allergy Immunol. 2004;133:113–120. doi: 10.1159/000076383. [DOI] [PubMed] [Google Scholar]
- 17.Park EK, Choo MK, Kim EJ, Han MJ, Kim DH. Antiallergic activity of ginsenoside Rh2. Biol Pharm Bull. 2003;26:1581–1584. doi: 10.1248/bpb.26.1581. [DOI] [PubMed] [Google Scholar]
- 18.Lim YJ, Na HS, Yun YS, Choi IS, Oh JS, Rhee JH, Cho BH, Lee HC. Suppressive effects of ginsan on the development of allergic reaction in murine asthmatic model. Int Arch Allergy Immunol. 2009;150:32–42. doi: 10.1159/000210378. [DOI] [PubMed] [Google Scholar]
- 19.Kim HS, Kim DH, Kim BK, Yoon SK, Kim MH, Lee JY, Kim HO, Park YM. Effects of topically applied Korean red ginseng and its genuine constituents on atopic dermatitis-like skin lesions in NC/Nga mice. Int Immunopharmacol. 2011;11:280–285. doi: 10.1016/j.intimp.2010.11.022. [DOI] [PubMed] [Google Scholar]
- 20.Bae EA, Han MJ, Shin YW, Kim DH. Inhibitory effects of Korean red ginseng and its genuine constituents ginsenosides Rg3, Rf, and Rh2 in mouse passive cutaneous anaphylaxis reaction and contact dermatitis models. Biol Pharm Bull. 2006;29:1862–1867. doi: 10.1248/bpb.29.1862. [DOI] [PubMed] [Google Scholar]
- 21.Babayigit A, Olmez D, Karaman O, Bagriyanik HA, Yilmaz O, Kivcak B, Erbil G, Uzuner N. Ginseng ameliorates chronic histopathologic changes in a murine model of asthma. Allergy Asthma Proc. 2008;29:493–498. doi: 10.2500/aap.2008.29.3137. [DOI] [PubMed] [Google Scholar]
- 22.Sumiyoshi M, Sakanaka M, Kimura Y. Effects of red ginseng extract on allergic reactions to food in Balb/c mice. J Ethnopharmacol. 2010;132:206–212. doi: 10.1016/j.jep.2010.08.012. [DOI] [PubMed] [Google Scholar]
- 23.Yang Z, Chen A, Sun H, Ye Y, Fang W. Ginsenoside Rd elicits Th1 and Th2 immune responses to ovalbumin in mice. Vaccine. 2007;25:161–169. doi: 10.1016/j.vaccine.2006.05.075. [DOI] [PubMed] [Google Scholar]
- 24.Park HJ, Byeon HE, Choi KW, Rhee DK, Lee KR, Pyo S. Inhibitory effects of ginsenoside Rb1 on atopic dermatitis-like skin lesions in mice. J Ginseng Res. 2010;34:363–368. [Google Scholar]
- 25.Gould HJ, Sutton BJ. IgE in allergy and asthma today. Nat Rev Immunol. 2008;8:205–217. doi: 10.1038/nri2273. [DOI] [PubMed] [Google Scholar]
- 26.Naclerio RM. Allergic rhinitis. N Engl J Med. 1991;325:860–869. doi: 10.1056/NEJM199109193251206. [DOI] [PubMed] [Google Scholar]
- 27.Ahmad N, Zacharek MA. Allergic rhinitis and rhinosinusitis. Otolaryngol Clin North Am. 2008;41:267–281. doi: 10.1016/j.otc.2007.11.010. [DOI] [PubMed] [Google Scholar]
- 28.Eliashar R, Levi-Schaffer F. The role of the eosinophil in nasal diseases. Curr Opin Otolaryngol Head Neck Surg. 2005;13:171–175. doi: 10.1097/01.moo.0000162258.03997.58. [DOI] [PubMed] [Google Scholar]
- 29.Rosenberg HF, Phipps S, Foster PS. Eosinophil trafficking in allergy and asthma. J Allergy Clin Immunol. 2007;119:1303–1310. doi: 10.1016/j.jaci.2007.03.048. [DOI] [PubMed] [Google Scholar]
- 30.Jung HW, Seo UK, Kim JH, Leem KH, Park YK. Flower extract of Panax notoginseng attenuates lipopolysaccharide-induced inflammatory response via blocking of NF-kappaB signaling pathway in murine macrophages. J Ethnopharmacol. 2009;122:313–319. doi: 10.1016/j.jep.2008.12.024. [DOI] [PubMed] [Google Scholar]
- 31.Kim BH, Lee YG, Park TY, Kim HB, Rhee MH, Cho JY. Ginsenoside Rp1, a ginsenoside derivative, blocks lipopolysaccharide-induced interleukin-1beta production via suppression of the NF-kappaB pathway. Planta Med. 2009;75:321–326. doi: 10.1055/s-0028-1112218. [DOI] [PubMed] [Google Scholar]
- 32.Ichikawa T, Li J, Nagarkatti P, Nagarkatti M, Hofseth LJ, Windust A, Cui T. American ginseng preferentially suppresses STAT/iNOS signaling in activated macrophages. J Ethnopharmacol. 2009;125:145–150. doi: 10.1016/j.jep.2009.05.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Evans CM, Kim K, Tuvim MJ, Dickey BF. Mucus hypersecretion in asthma: causes and effects. Curr Opin Pulm Med. 2009;15:4–11. doi: 10.1097/MCP.0b013e32831da8d3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kim S, Nadel JA. Role of neutrophils in mucus hypersecretion in COPD and implications for therapy. Treat Respir Med. 2004;3:147–159. doi: 10.2165/00151829-200403030-00003. [DOI] [PubMed] [Google Scholar]
- 35.Ali MS, Wilson JA, Bennett M, Pearson JP. Mucin gene expression in nasal polyps. Acta Otolaryngol. 2005;125:618–624. doi: 10.1080/00016480510027538. [DOI] [PubMed] [Google Scholar]
- 36.Chung E, Lee KY, Lee YJ, Lee YH, Lee SK. Ginsenoside Rg1 down-regulates glucocorticoid receptor and displays synergistic effects with cAMP. Steroids. 1998;63:421–424. doi: 10.1016/s0039-128x(98)00043-9. [DOI] [PubMed] [Google Scholar]
- 37.Lee YJ, Chung E, Lee KY, Lee YH, Huh B, Lee SK. Ginsenoside-Rg1, one of the major active molecules from Panax ginseng, is a functional ligand of glucocorticoid receptor. Mol Cell Endocrinol. 1997;133:135–140. doi: 10.1016/s0303-7207(97)00160-3. [DOI] [PubMed] [Google Scholar]
- 38.Sun J, Song X, Hu S. Ginsenoside Rg1 and aluminum hydroxide synergistically promote immune responses to ovalbumin in BALB/c mice. Clin Vaccine Immunol. 2008;15:303–307. doi: 10.1128/CVI.00448-07. [DOI] [PMC free article] [PubMed] [Google Scholar]