Abstract
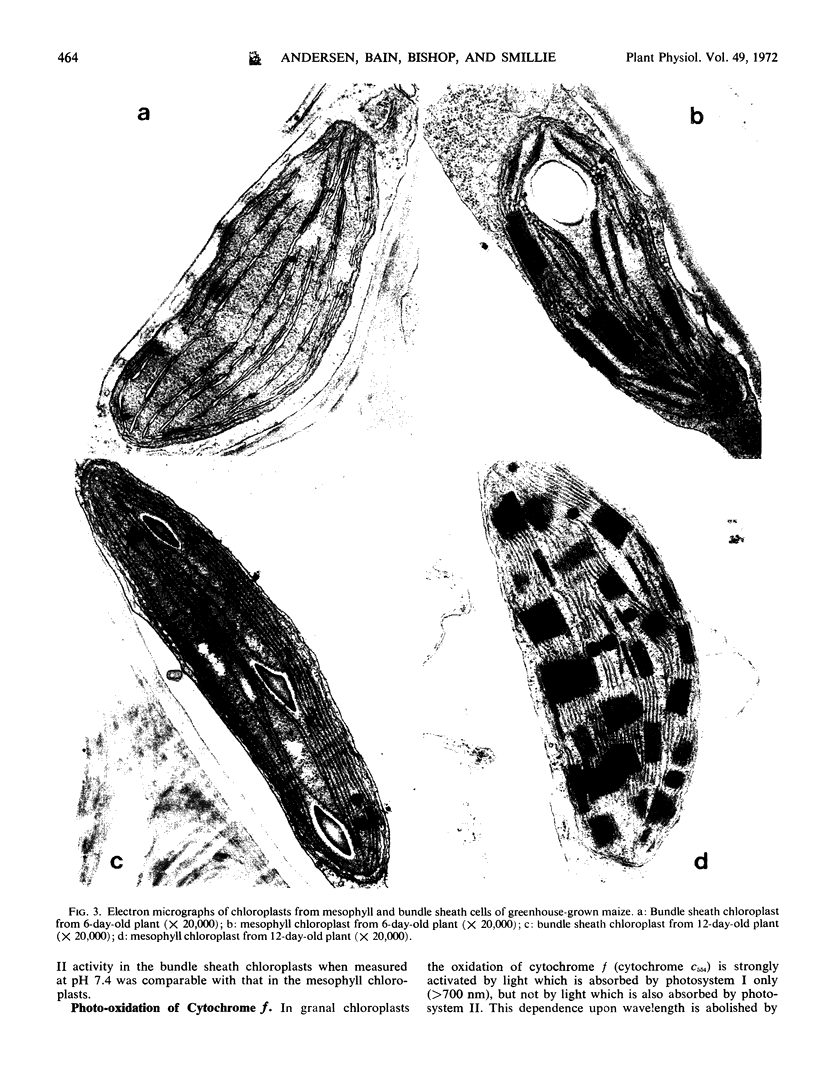
The photochemical activities of chloroplasts isolated from bundle sheath and mesophyll cells of maize (Zea mays var. DS606A) have been measured. Bundle sheath chloroplasts are almost devoid of grana, except in very young leaves, while mesophyll chloroplasts contain grana at all stages of leaf development.
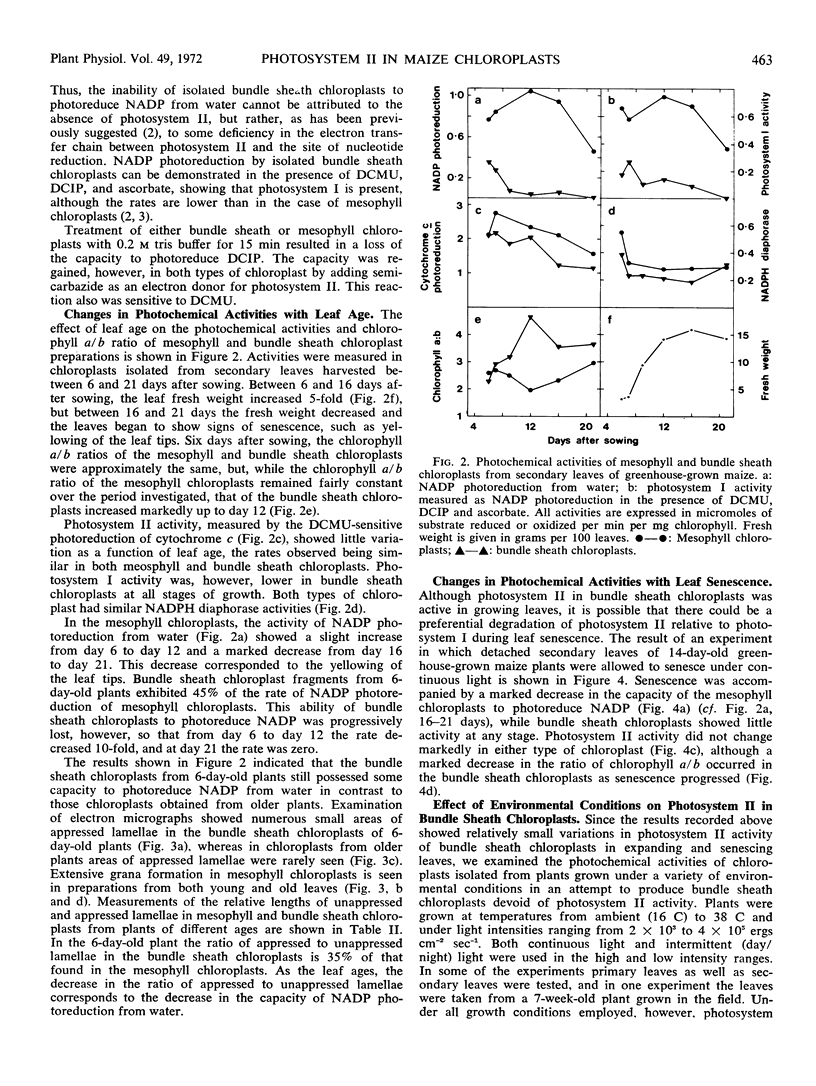
Chloroplast fragments isolated from bundle sheath cells showed a light-dependent reduction of potassium ferricyanide, 2, 6-dichlorophenolindophenol, mammalian cytochrome c, plastocyanin, and Euglena cytochrome c552. These activities were inhibited by 3-(3, 4-dichlorophenyl)-1, 1-dimethylurea at 1.25 micromolar. However, the photoreduction of NADP from water was extremely low or absent, except in chloroplasts from very young leaves, and the capacity for NADP reduction appeared to be related to the degree of grana formation.
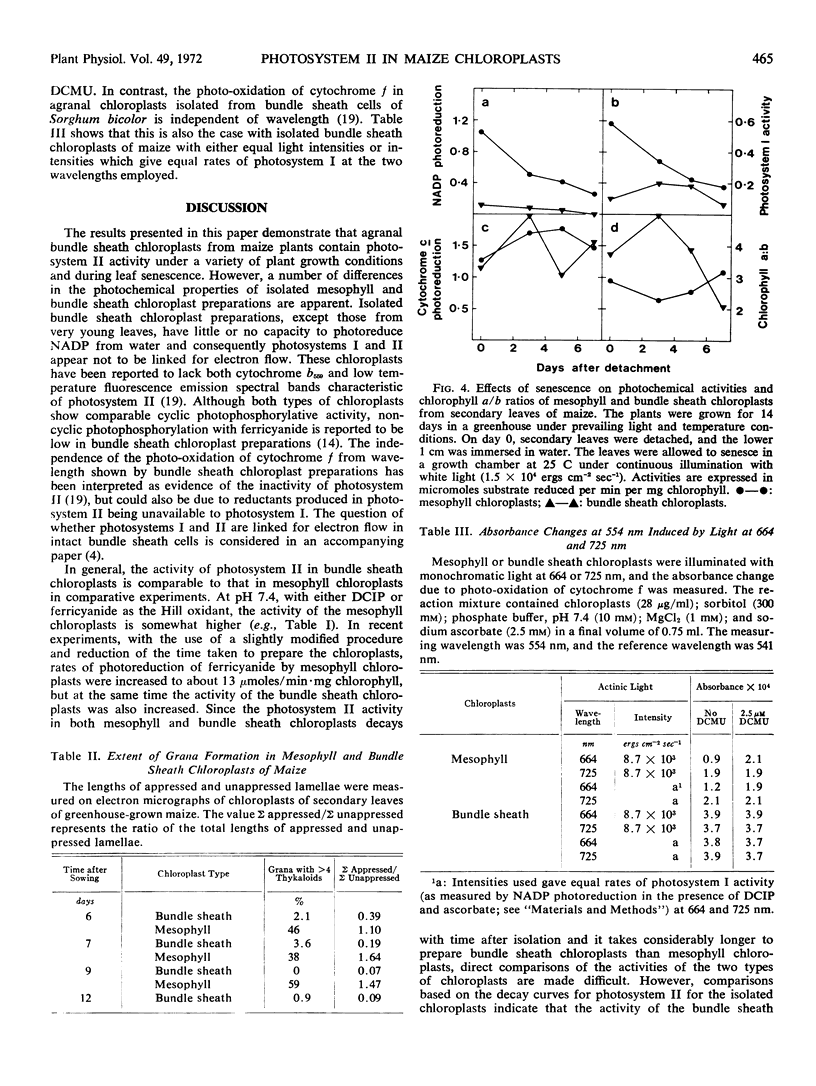
Photosystem I activity was present in bundle sheath chloroplast preparations at all stages of leaf growth and senescence examined. However, the activity was lower than in isolated mesophyll chloroplasts. NADPH diaphorase activity was comparable in both types of chloroplast.
Chloroplasts isolated from bundle sheath cells of plants grown under a variety of conditions, including continuous and intermittent light, high and low light intensities, and high temperature, exhibited photosystem II activity.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arnon D. I. COPPER ENZYMES IN ISOLATED CHLOROPLASTS. POLYPHENOLOXIDASE IN BETA VULGARIS. Plant Physiol. 1949 Jan;24(1):1–15. doi: 10.1104/pp.24.1.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bishop D. G., Andersen K. S., Smillie R. M. Incomplete membrane-bound photosynthetic electron transfer pathway in agranal chloroplasts. Biochem Biophys Res Commun. 1971 Jan 8;42(1):74–81. doi: 10.1016/0006-291x(71)90364-0. [DOI] [PubMed] [Google Scholar]
- Bishop D. G., Andersen K. S., Smillie R. M. Photoreduction and Oxidation of Cytochrome f in Bundle Sheath Cells of Maize. Plant Physiol. 1972 Apr;49(4):467–470. doi: 10.1104/pp.49.4.467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodenough U. W., Armstrong J. J., Levine R. P. Photosynthetic Properties of ac-31, a Mutant Strain of Chlamydomonas reinhardi Devoid of Chloroplast Membrane Stacking. Plant Physiol. 1969 Jul;44(7):1001–1012. doi: 10.1104/pp.44.7.1001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Homann P. H., Schmid G. H. Photosynthetic reactions of chloroplasts with unusual structures. Plant Physiol. 1967 Nov;42(11):1619–1632. doi: 10.1104/pp.42.11.1619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Izawa S., Good N. E. Effect of Salts and Electron Transport on the Conformation of Isolated Chloroplasts. II. Electron Microscopy. Plant Physiol. 1966 Mar;41(3):544–552. doi: 10.1104/pp.41.3.544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohad I., Siekevitz P., Palade G. E. Biogenesis of chloroplast membranes. II. Plastid differentiation during greening of a dark-grown algal mutant (Chlamydomonas reinhardi). J Cell Biol. 1967 Dec;35(3):553–584. doi: 10.1083/jcb.35.3.553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Polya G. M., Osmond C. B. Photophosphorylation by mesophyll and bundle sheath chloroplasts of c(4) plants. Plant Physiol. 1972 Feb;49(2):267–269. doi: 10.1104/pp.49.2.267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sane P. V., Goodchild D. J., Park R. B. Characterization of chloroplast photosystems 1 and 2 separated by a non-detergent method. Biochim Biophys Acta. 1970 Aug 4;216(1):162–178. doi: 10.1016/0005-2728(70)90168-4. [DOI] [PubMed] [Google Scholar]
- Wiessner W., Amelunxen F. Beziehungen zwischen submikroskopischer Chloroplastenstruktur und Art der Kohlenstoffquelle unter phototrophen Ernährungsbedingungen bei Chlamydobotrys stellata. Arch Mikrobiol. 1969;66(1):14–24. [PubMed] [Google Scholar]
- Wiessner W., Amelunxen F. Umwandlungen der submikroskopischen Chloroplastenstruktur parallel zur Veränderung der stoffwechselphysiologischen Leistung von Chlamydobotrys stellata. Arch Mikrobiol. 1969;67(4):357–369. [PubMed] [Google Scholar]
- Woo K. C., Anderson J. M., Boardman N. K., Downton W. J., Osmond C. B., Thorne S. W. Deficient Photosystem II in Agranal Bundle Sheath Chloroplasts of C(4) Plants. Proc Natl Acad Sci U S A. 1970 Sep;67(1):18–25. doi: 10.1073/pnas.67.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]