Abstract
Ab therapy against surface Ags on tumor cells has demonstrated significant efficacy for some cancers. However, it is costly and patients frequently develop acquired resistance over time. In cases of Ab therapy resistance, T cell responses have been shown to be essential in controlling disease progression. Thus, vaccination that generates a sustained Ab response as well as a T cell response may be more effective and economical. Here, we have developed a vaccination strategy by targeting protein Ags to B cells via a CD19 single chain variable fragment miniAb. By using the tumor-associated Ag (TAA) her-2/neu extracellular domain (ECD), we showed that the co-engagement of CD19 and BCR induced full B cell activation to produce a high titer of Abs and enhanced CD4 Th2 response and CD8 T cell activation and differentiation. These Abs competitively inhibited humanized her-2/neu Ab binding and were capable of activating the complement and inhibiting human breast cancer growth in vitro. Therapeutic efficacy was demonstrated in vivo using murine mammary carcinomas models. Furthermore, four different ECDs of her-2/neu could be targeted to B cells to generate Abs against particular domains with different anti-tumor properties. This approach may offer a new avenue for vaccine development with significantly lower cost which may be usable not only for cancer therapy but also for infectious agents.
Introduction
There is currently great interest in activating the immune system for cancer therapy (1). However, developing effective cancer vaccines has proven to be a daunting task (2). The recent approval of the first therapeutic cancer vaccine by the US Food and Drug Administration (FDA), Sipuleucel-T, a vaccine for the treatment of asymptomatic metastatic castrate-resistant advanced prostate cancer with a modest clinical benefit in some patients (3), has re-energized research into more potent cancer vaccine development. Currently many cancer vaccine platforms have been evaluated in pre-clinical animal models or clinical trials (4), including protein or peptide-pulsed dendritic cell (DC)-based vaccines (5). The DC-based vaccine platform normally requires leukapheresis and further in vitro expansion of DCs. Drawbacks of this approach include large-scale preparation of clinical grade DCs, the choice of DC subsets (6), and DC-related trafficking. In contrast, anti-tumor mAb therapy has achieved clinical promise and now is widely used in oncology patient care (7, 8). Thus, it would be desirable if tumor vaccines could elicit long-lasting anti-tumor humoral responses as well as T cell responses.
B cells are capable of eliciting anti-tumor responses by the production of Abs as well as serving as APCs to induce CD4 T cell responses (9, 10). In addition, B cells can present Ag to cross-prime CD8 T cells for expansion and activation (11). Ag activation of B cells has been shown to enhance the expression of costimulatory molecules, principally CD86, which is essential for B cells’ ability to break T cell tolerance. However, the role of B cells in tumor development has been controversial. Previous studies showed that therapeutic depletion of B cells enhances B16 melanoma growth in mice (12). In contrast, in a skin squamous carcinoma model, B cells, predominantly the Abs produced by B cells, promote tumor progression via triggering chronic inflammation (13). Nevertheless, the ability of B cells to induce both Ab production and T cell responses makes B cells an ideal cell subset for cancer vaccine development.
CD19 is a B cell-specific member of the Ig superfamily expressed at almost every stage of B cell development except after differentiation into plasma cells (14). CD19 is also considered a co-receptor for BCR; co-engagement of BCR and CD19 reduces the B cell activation threshold (15). Our previous studies also showed that CD19 on the B cell surface is important for B cell Ag presentation (16, 17). Targeting of Ags to B cells via CD19 led to more efficient Ag presentation by B cells and potent CD4 and CD8 T cell activation. In addition, co-ligation of CD19 and the BCR potently activates B cells to induce Ag-specific Ab responses that may specifically target tumor cells (17).
Her-2/neu has been an attractive target for cancer immunotherapy (18). The Her-2/neu chimeric humanized Ab trastuzumab (Herceptin) has been approved to treat metastatic her-2/neu overexpressing breast cancers (19). Despite the great success of Herceptin therapy, the major limitation of immunotherapy with trastuzumab is the development of drug resistance usually within one year from the beginning of treatment arising from various mechanisms (20-22). It appears that CD8 T cell responses are effective against these tumors (23). In addition, Herceptin cost per patient could be as much as US$70,000 per year (24). Clearly, generating sustained and active immune responses to the her-2/neu protein is essential to this existing approach. Here, we constructed CD19 single chain variable fragment (scFv) miniAbs as a means to target Ags to B cells and found that this approach elicits not only augmented Ab responses but also T cell responses. More importantly, Herceptin-like Abs and enhanced CD8 cytolytic activity were elicited in immunized mice. These Ab and T cell responses could significantly delay tumor progression in both prophylactic and therapeutic settings and prolong the survival of tumor-bearing mice. In addition, this targeting approach could generate Ab responses against four different domains of Her-2/neu extracellular domains (ECD) with different anti-tumor properties. Thus this approach offers a new avenue for effective vaccine development.
Materials and Methods
Mice and cell lines
Balb/c, OVA TCR Tg OT-I, and OT-II Rag-deficient mice were purchased from the Jackson Laboratory (Bar Harbor, ME). C57Bl/6 mice were purchased from the NCI (Frederick, MD). All experimental mice were housed under specific pathogen-free conditions in the animal facility of University of Louisville and treated in accordance with the guidelines of the Institutional Animal Care and Use Committee (IACUC) of the University of Louisville.
Balb/c mammary carcinoma cell D2F2/E2 expressing human her-2/neu was kindly provided by Dr. Wei-Zen Wei (Karmanos Cancer Institute, Detroit, MI). Human her-2/neu expressing C57Bl/6 mammary tumor line EO771 (EO771/E2) was generated by stable transfection with human full-length her-2/neu cDNA plasmid (25). Human breast cancer cell SKBR-3 and human ovarian cancer cell SKOV3 from ATCC were maintained in DMEM supplemented with 10% heat-inactivated FCS, 2mM L-glutamine, 100U/ml penicillin, and 100 μg/ml streptomycin. Rat anti-mouse CD19 hybridoma (1D3) from ATCC was maintained in MEM supplemented with 10% FBS. BL21(DE3)Plyss competent cells were purchased from Novagen.
Generation of CD19 scFv miniAb and fusion proteins
To generate CD19 scFv, total RNA was extracted from 1D3 and first strand cDNA was synthesized. VH and VL were amplified using primers: Vkappa start: GAAGATCTCCACCATGGACATTCAGCTGACCCAGTCTCCA; Jkappa with linker: AGAGCCACCTCCGCCCCGTTTCAGTTCCAGCTTGGTGCC; VH start with linker:GGCGGAGGTGGCTCTARGTSMARCTGVWGSARTCWGG;VH stop:ACTAGTCGACTCATGAGGACACGGTGACCATGGTTCCTGGGCCCC. The single chain Fv (VL-VH) was then synthesized by overlapping PCR. PCR product was sequenced and further cloned into pET-20b (+) vectors in an NcoI site.
The full-length human c-ErbB-2 (Her-2/neu) cDNA was isolated from plasmid pCMV-ErbB-2 as a 4.4-kb EcoRI restriction fragment and was kindly provided by Dr. Wei. The pET-20b(+)-anti-CD19-scFv-c-ErbB-2 cDNA constructs that encode the herceptin-binding domain (from residues 475 to 652, designated as P3-4) were generated. Four different extracellular domains of ErbB -2 were also cloned into pET-20b (+) vectors using primers summarized in Supplemental Table I. The short OVA fragment containing OVA257-264 and OVA323-339 encoding sequence (residues 241-386) was amplified from plasmid pCMV-OVA and subcloned in-frame between the sal1 and xho1 restriction sites of pET-20b(+)-scFv using primers shown in Supplemental Table 1. pET-20b (+) constructs were transfected into BL21(DE3)Plyss cells and induced with 0.1mM IPTG. The proteins were purified using His-Select Nickel Affinity Gel (Sigma) and LPS contamination was removed by Detoxi-Gel™ Endotoxin Removing Columns (Thermo Scientific). The proteins were dialyzed in PBS and analyzed with SDS-PAGE and Western Blot (WB). Endotoxin level was <0.5EU/mg as measured by LAL assay (Associations of Cape Cod, Inc).
Conjugation of protein P3-4 to anti-CD19 mAb
Rat anti-mouse CD19 mAb (IgG2a) mAb was reduced in 20 mM dithiothreitol (DTT; Bio-Rad, Hercules, CA) at room temperature for 30 min and then separated from the reducing agent over a desalting column. LPS-free recombinant protein P3-4 was activated with succinimidyl 4-(N-maleimidomethyl) cyclohexane-1-carboxylate (SMCC; Pierce) according to the manufacturer's protocol and mixed with the reduced mAb for 1 hour at room temperature and then incubated overnight at 4°C. The unconjugated anti-CD19 mAb or free protein P3-4 was removed by size-exclusive column. The conjugates were examined for B-cell binding as assessed by flow cytometry.
Fusion protein binding assay and confocal microscopy
For in vitro binding assay, splenocytes were incubated with proteins scFv, P3-4, scFv-p3-4, OVA-biotin, scFv-OVA-biotin or biotin-labeled scFv-D1, scFv-D2, scFv-D3, scFv-D4 and then stained with oregon-green-labeled anti-Her2 Ab or PE-streptavidin and APC-anti-mouse B220. Cells were washed and assessed by flow cytometry. For the in vivo binding assay, mice were injected i.v. with biotinylated fusion proteins scFv, P3-4, scFv-p3-4, OVA or scFv-OVA. Peripheral blood was drawn at 10 min after injection. Cells were stained with APC-anti-mouse B220 and PE-streptavidin and assessed by flow cytometry.
For determination of protein (scFv, scFv-p3-4, parental CD19 mAb) KD values, B cells were incubated with different concentrations of proteins and then assayed by flow cytometry. Kd values were calculated by the following equation: 1/(F-Fback)=1/Fmax+(KD/Fmax)(1/[scFv], where F=fluorescence unit, Fback=background fluorescence and Fmax was calculated from plot (26).
For co-localization assays, B cells were incubated with scFv-p3-4 for 30 min and stained with biotinylated anti-her-2/neu followed by Alexa Fluor 594-streptavidin and Alexa Fluor 488-anti-IgM for 30 min at 4°C. Cells were analyzed on a Nikon confocal microscope.
In vitro B cell culture
Purified B cells were stimulated with scFv, P3-4, scFv-p3-4 or CD19 mAb conjugated P3-4 (1 μg/ml) for 24 h and then harvested to detect surface marker expression by flow cytometer. For cytokine assays, B cells were incubated for 48 h with different fusion proteins (20 μg/ml) and supernatants were harvested to measure cytokine levels by ELISA.
Detection of her-2/neu Abs by ELISA
Ninety-six-well plates were coated with recombinant her-2/neu P1-4 protein (1 μg/well) overnight at 4°C and blocked with 0.5% BSA/PBS. Pre- or post-immune sera from mice were diluted and further reacted with goat anti–mouse IgM or IgG horseradish peroxidase (HRP) conjugates (Southern Biotech, Birmingham, AL). The assays were subsequently developed by the addition of ABTS 1 Component Microwell Substrate (BioFX Laboratories, Owings Mills, MD) and the OD450 was determined. To measure IgG isotype Abs, sera were diluted at 1:100 and further reacted with HRP-conjugated goat anti–mouse IgG1, IgG2a and IgG2b (Southern Biotech, Birmingham, AL). Ab concentrations were determined by generating a standard curve using serial dilutions of Herceptin.
Ab competitive inhibition assay
Micro-titer plates were coated with 1 μg/well recombinant her-2/neu P3-4 for overnight and then blocked with 0.5% BSA/PBS for 1 h at RT. Serially diluted immune sera (1:20, 1:40, 1:80) were added into wells for 1 h at RT and followed by biotin-Herceptin. Pre-immune sera were used as controls. The wells were incubated with streptavidin-HRP and ABTS 1 Component Microwell Substrate. OD450 was measured. The percent inhibition was calculated using formula: (ODpre-ODpost)/ODpre×100%
Complement activation assay
1 × 106 SKOV3 tumor cells were suspended in 100 μl HBSS with calcium and magnesium and incubated with inactivated-post/pre-immune sera (1:10 dilution) for 30 min at 37°C. SCID mouse sera as a source of complement were added and followed by FITC-anti-mouse C3 (MP Biomedical, Inc.) and measured by flow cytometry.
In vitro tumor cell growth inhibition assay and western blot (WB)
1 × 104 SKBR-3 cells were placed into the wells of the Acea 16-well plates for 24 h. Ten μl of heat-inactivated pre- or post- immune sera were added to wells and incubated for indicated times. Herceptin was used as positive control. The inhibition of tumor cell growth was calculated by measuring the relative decrease in current impedance among wells containing post-immune serum and wells containing pre-immune serum only as described previously (27, 28). The percent of inhibition was calculated using formula: (Cell indexpre–Cell indexpost)/Cell indexpre ×100%
For WB, SKBR-3 cells were treated with medium, heat-inactivated immune serum (1:10) or Herceptin (2 μg/ml) for 3 h at 37 °C. Cells were lysed and immunoblot was performed with rabbit anti- Phospho-Akt Ab (Cell Signaling Technology Inc. Danvers, MA) or mouse anti-β-actin Ab (Sigma, St. Louis, MO). The blots were visualized using the ECL prime WB detection reagents (GE Healthcare Biosciences, Pittsburgh, PA).
T cell proliferation assay
For in vitro proliferation assays, splenocytes from OT-I or OT-II Tg mice were labeled with 10 μM CFSE (Molecular Probes, Carlsbad, CA) and then stimulated with varying concentrations of fusion proteins OVA or scFv-OVA. Three days later, turnover of T cells was examined by flow cytometry. For 3H-thymidine incorporation assays, splenocytes from immunized mice were cultured in 96-well plates (5×105 cells/well) and stimulated with scFv-p3-4 (20 μg/ml) for 72 h. 3H-thymidine was added 16 h before 3 days culture and proliferation was measured by Scintillation counter (Packard). Stimulation index (SI) was used to show fold increase. SI was calculated using formula: cpmexp/cpmcontrol. For in vivo proliferation assays, CD8 or CD4 T cells purified from OT-I or OT-II Tg mice were labeled with 10 μM CFSE. T cells (2×106/mouse) were then adoptively transferred into recipient mice. OVA or scFv–OVA fusion proteins were injected into mice i.v. 24 h after adoptive transfer. Recipient mice were sacrificed after 3 days and the turnover of T cells was examined by flow cytometry.
Intracellular cytokine staining
Intracellular cytokine staining was performed using BD Cytofix cytoperm kit with BD Golgiplug (BD Pharmingen, San Diego, CA) according to the manufacturer's protocol. For IFN-γ staining, cells were stimulated with OVA, scFv-OVA or scFv-p3-4 for 3 days, and then re-stimulated with PMA plus ionomycin for 4 h in the presence of Golgiplug and then stained with APC– or FITC-conjugated mAbs against mouse CD8 or CD4 and PE-conjugated anti-mouse IFN-γ (Biolegend). For intracellular IL-4 staining, cells were stimulated with ConA (3 μg/ml) for 2 days, followed by mouse IL-2 (10 ng/ml) and IL-4 (50 ng/ml) for 3 days. Cells were re-stimulated with immobilized CD3 (10 μg/ml) and soluble CD28 mAbs (2 μg/ml) in the presence of Golgiplug for 5 h. Cells were surface stained with anti-mouse CD4-APC and intracellularly stained with anti-mouse IL-4-FITC (eBiosciences).
In vivo cytotoxicity assay
B cells loaded with or without scFv-p3-4 were used as target cells for in vivo cytotoxicity assay. In brief, B cells were pulsed with scFv-p3-4 (10 μg/million cells) and then labeled with 2.5 μM CFSE (CFSEhigh). Un-pulsed B cells were labeled with 0.25 μM CFSE (CFSElow). The mixed B cells at a ratio of 1:1 were injected into mice immunized with different regimens. Mice were killed after 24 h of target cell transfer. Specific cytotoxicity was determined by detecting the differentially fluorescent-labeled target cell populations by flow cytometry. The percentage of cytotoxicity was determined as follows: (1-CFSEhigh/CFSE low) × 100%.
Flow cytometry
Splenocytes were incubated with anti-CD16/CD32 Fc receptor blocker for 10 min on ice and then washed and stained with indicated fluorochrome-conjugated mAbs. Cells were collected with a FACSCalibur flow cytometer (BD Immunocytometry Systems, San Jose, CA) and analyzed using FlowJo software (TreeStar, Ashland, OR).
Mouse immunization and tumor challenge
Six- to 8-week-old Balb/c or C57Bl/6 mice were immunized i.v. with scFv, P3-4, or scFv-p3-4 at 50 μg/mouse/injection on days 0, 7 and 14. A group of mice immunized with PBS was used as control. On days 7 and 21, the sera were collected for her-2/neu Ab measurement. For tumor therapy in the prophylactic setting, mice were immunized 4 times and challenged by s.c. injection in the flank with 1 × 105 D2F2/E2 (Balb/c) tumor cells. In the therapeutic setting, Balb/c or C57Bl/6 mice were first challenged with 1 × 105 D2F2/E2 or 5 × 105 EO771/E2 tumor cells. When palpable tumors formed, mice were treated with different regimens for 4 times at 1-week intervals. Tumor diameter was measured by calipers twice per week. Mice were killed when tumors reached 15 mm in diameter. In some experiments, survival was monitored up to 100 days beyond tumor implantation. In some experiments, mice were injected intraperitoneally with CD8 mAb (clone 2.43; 500 μg/mouse) or CD4 mAb (clone GK1.5; 250 μg/mouse) or isotype control mAb (250 μg/mouse) 3 days prior to immunization.
Statistical analysis
Unpaired T-test analysis was used to determine whether the differences between T and B cell-mediated immune responses induced by scFv-p3-4 versus scFv or P3-4 were significant. A two-way ANOVA and Kaplan-Meier survival analysis were used to determine significance for in vivo tumor therapy. P values less than 0.05 were considered significant.
Results
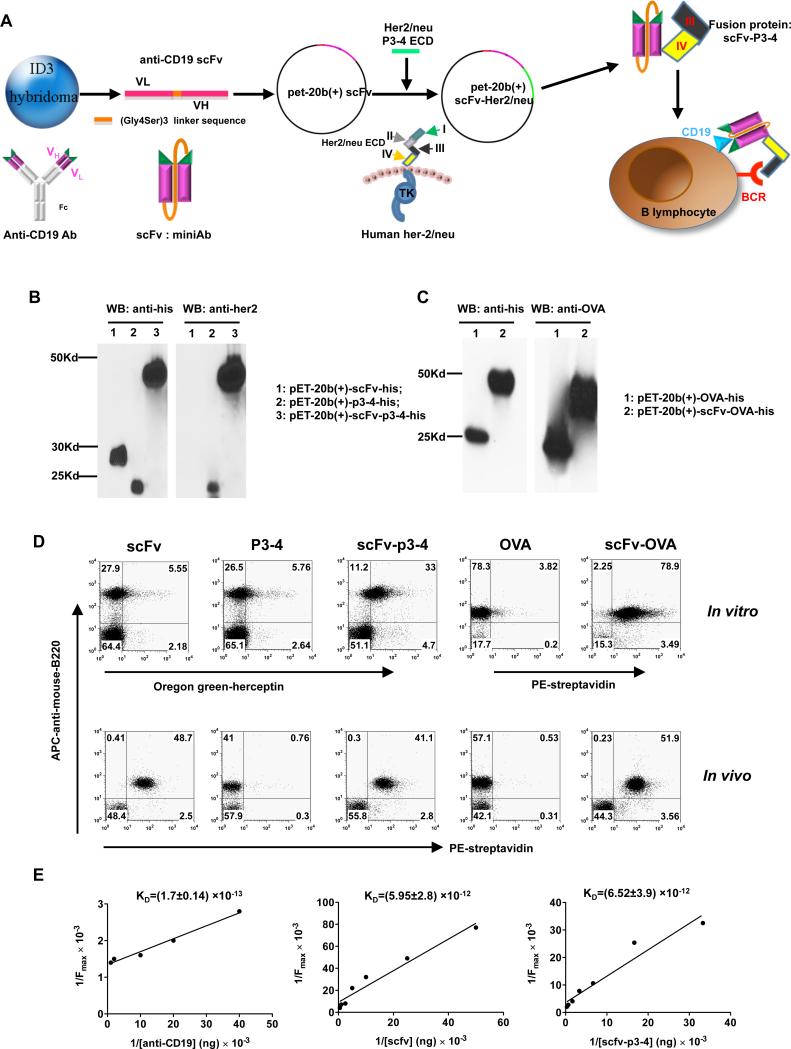
Generation of anti-CD19 scFv fusion proteins that specifically bind to B cells
Our previous studies have demonstrated that targeting of Ags via CD19 led to enhanced Ag-specific T cell responses and broke immune tolerance (17). However, if whole Abs were used, these could potentially stimulate the production of inflammatory cytokines resulting in serious adverse effects. Single chain Ab variable region fragments (scFv) are potentially useful as therapeutic reagents (29-31) less likely to engender inflammatory responses. Single chain miniAbs are recombinant monovalent Abs lacking the constant part of both heavy and light chains. These molecules retain their Ag recognition ability and can be easily expressed in a prokaryotic system or mammalian cell lines. We generated anti-CD19 scFv miniAb from 1D3 rat anti-mouse CD19 hybridoma cells. Candidate Ag genes such as her-2/neu ECD can be ligated with anti-CD19 scFv to make a fusion protein that specifically targets B cell CD19 and simultaneously engages the BCR (Fig. 1A). Tumor-associated Ag (TAA) her-2/neu ECD and surrogate Ag OVA were chosen to fuse with anti-CD19 scFv. The anti-CD19 scFv-her2/neu ECD cDNA constructs which contain cDNA that encodes the Herceptin-binding domain (from residues 475 to 652 amino acid, designated as P3-4) were generated. Similarly, truncated OVA cDNA (residues 241 to 386) was also ligated with scFv plasmids. Subsequently, these recombinant proteins were produced, purified, and characterized. As indicated in Fig. 1B, recombinant her-2/neu P3-4, anti-CD19 scFv-p3-4 (scFv-p3-4) fusion proteins were blotted positively with both His-Tag and her-2/neu Abs. The recombinant anti-CD19 scFv miniAb protein was blotted positively with His-Tag Ab but not with her-2/neu Ab nor OVA Ab. Similarly, recombinant proteins OVA and scFv-OVA reacted with His-Tag Ab and OVA Ab, respectively (Fig. 1C).
Figure 1. CD19 scFv miniAb specially binds to B cells with high binding affinity.
(A) Schematic view of fusion protein constructs. (B, C) Lysates of BL21 (DE3)Plyss transfected with expression plasmids for CD19-scFv, her-2/neu P3-4, CD19-scFv-p3-4 (B), OVA and CD19-scFv-OVA(C) were purified and then western blotted using Abs against His-Tag, her-2/neu or OVA. (D) For in vitro B cell binding, splenocytes were incubated with scFv, P3-4, scFv-p3-4, biotin-OVA, biotin-scFv-OVA followed by APC-anti-mouse B220 and Oregon-green-Herceptin or PE-streptavidin. Cells were washed and assessed by flow cytometer. For in vivo binding, biotin-labeled-proteins were i.v. injected into mice. Peripheral blood was drawn at 10 min after injection. Cells were stained with APC-anti-mouse B220 and PE-streptavidin and assessed by flow cytometer. (E) Determination of protein KD values by Line weaver-Burk analysis.
To verify that the scFv, scFv-p3-4 and scFv-OVA proteins retained the Ag-binding activity of the parental Ab, we measured specific binding to B cells in vitro and in vivo. For the in vitro binding assay, splenocytes were incubated with protein scFv, P3-4, scFv-p3-4, biotin-OVA, or biotin-scFv-OVA. For the in vivo binding assay, biotin-labeled-proteins were i.v. injected into mice. Peripheral blood was drawn at 10 min after injection. The successful targeting B cells was observed by the identification of double positive cells (Fig. 1D). These results indicate that anti-CD19 scFv miniAb with or without Ag tagged is capable of binding to B cells specifically. We also measured the protein Kd values by Lineweaver-Burk analysis. The results revealed that scFv and scFv-p3-4 proteins retained high binding affinity to B cells (Fig. 1E).
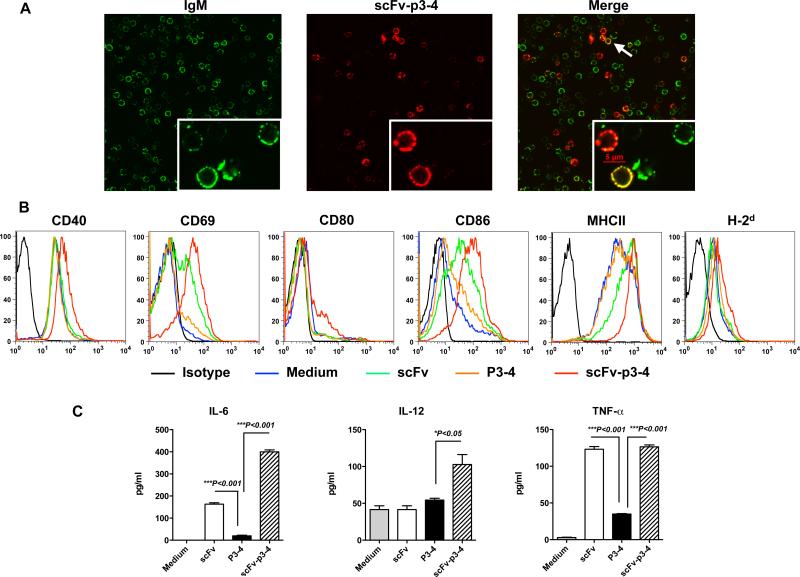
Fusion protein scFv-p3-4 activates B cells to produce low levels of pro-inflammatory cytokines
To determine whether Ag binding to B cells via anti-CD19 scFv was targeted to the BCR, B cells were incubated with anti-CD19 scFv-p3-4 fusion protein and stained with biotinylated anti-Her-2/neu followed by Alexa Fluor 594-streptavidin (red) and Alexa Fluor 488-anti-IgM (green). As shown in Figure 2A, Ag linked to anti-CD19 scFv co-localized with surface IgM on B cells. To examine whether the fusion protein scFv-p3-4 could activate B cells, purified B cells were incubated with fusion proteins scFv, P3-4, scFv-p3-4. Expression of surface markers on B cells was assessed by flow cytometry. Fusion protein scFv-p3-4, but not P3-4, significantly up-regulated the expression levels of surface markers including CD40, CD69, CD80, CD86, MHC class II, and MHC class I molecule (Fig. 2B). Further cytokine measurement indicated that scFv-p3-4 stimulated low levels of pro-inflammatory cytokines including IL-6, IL-12P40, and TNF-α (Fig. 2C). scFv alone also stimulated moderate expression levels of CD69, CD86, and MHC class II but the levels were significantly lower than these stimulated by scFv-p3-4. In addition, scFv alone stimulated IL-6 and TNF-α production but not IL-12. These results suggest that Ag targeted to B cells via CD19 molecule can co-engage the BCR and stimulate full B cell activation.
Figure 2. Ag bound to B cells via CD19 is co-localized with the BCR and co-engagement of the BCR and CD19 activates B cells.
(A) Purified B cells were incubated with scFv-p3-4 fusion protein for 30 min and stained with biotinylated anti-her2/neu followed by Alexa Fluor 594-streptavidin (red) and Alexa Fluor 488-anti-IgM (green). Cells were cytospin to slides and images were acquired. (B) B cells were stimulated with fusion proteins as indicated for 24 h. Surface markers were assessed by flow cytometry. Data shown are one from 3 independent experiments. (C) Supernatants from B cells stimulated with fusion proteins scFv, p3-4, scFv-p3-4, or medium alone for 48 h were harvested and assayed for cytokines IL-6, IL-12p40, and TNF-α by ELISA. Representative of 3 experiments. Error bars represent s.e.m.
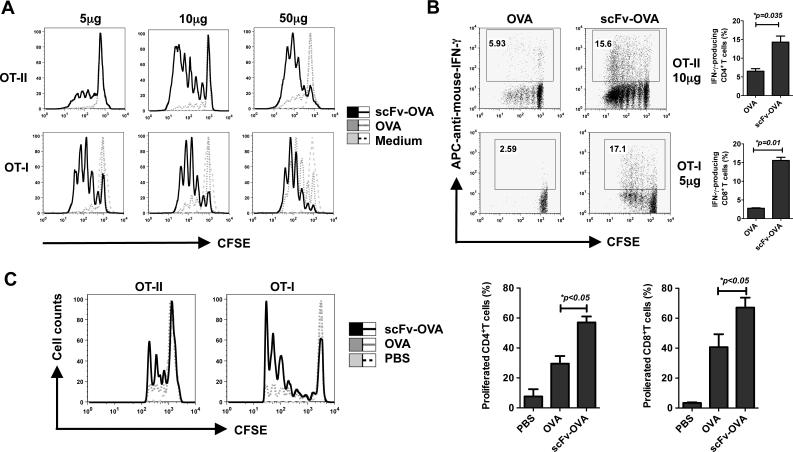
Targeting surrogate Ag OVA to B cells via CD19 scFv stimulates augmented Ag-specific CD4 and CD8 T-cell responses
To determine whether targeting Ag to B cells increases CD4 and CD8 T-cell responses, we generated the fusion protein scFv-OVA for Ag presentation. For an in vitro Ag presentation assay, OT-II CD4+ T cells or OT-I CD8+T cells were used as readout of OVA Ag presentation. Indeed, CD4+ or CD8+ T cells underwent significantly more proliferation in response to scFv-OVA as compared to OVA stimulation (Fig. 3A). In addition, both CD4+ T and CD8+ T cells produced large amounts of IFN-γ upon scFv-OVA stimulation (Fig. 3B). Next, we examined whether this strategy would lead to enhanced T-cell proliferation in vivo. Mice were administered i.v. with 2×106 CFSE-labeled naive OT-I or OT-II cells. The next day, mice were injected with soluble OVA or scFv-OVA. As an additional control, PBS was injected into another group of mice. As shown in Fig. 3C, both CD4+ and CD8+ T cells underwent at least 4 divisions within the first 3 days of exposure to scFv-OVA in vivo. In contrast, OT-I or OT-II T cells responded significantly less to the same amount of soluble OVA. These results suggest that scFv-OVA fusion protein induces potent T cell proliferation and differentiation both in vitro and in vivo.
Figure 3. T-cell responses mediated by scFv-OVA.
(A, B) Splenocytes from OT-I or OT-II mice were labeled with CFSE and then stimulated with different concentrations of OVA or scFv-OVA for 3 days. The turnover of T cells and intracellular IFN-γ staining from splenic cells were examined by flow cytometer. Cells were gated on CD8+ or CD4+ populations. T-cell proliferation shown in overlay histogram was from medium, OVA or scFv-OVA (A). The IFN-γ production of CD4+and CD8+ was shown in (B). (C) 2×106 CFSE-labeled naive CD4 OT-II or CD8 OT-I T cells were i.v. adoptively transferred into naive C57Bl/6 mice (n=3). The next day, mice were injected with a single dose of OVA or scFv-OVA (for OT- I T cells, 5 μg protein/mouse; for OT- II T cells, 20 μg protein/mouse) or PBS. Recipient mice were killed after 3 days and turnover of T cells from splenic cells was examined by flow cytometer. Cells were gated on CFSE-positive population. T-cell proliferation shown in overlay histogram was from PBS, OVA, or scFv-OVA. Representative of 3 experiments.
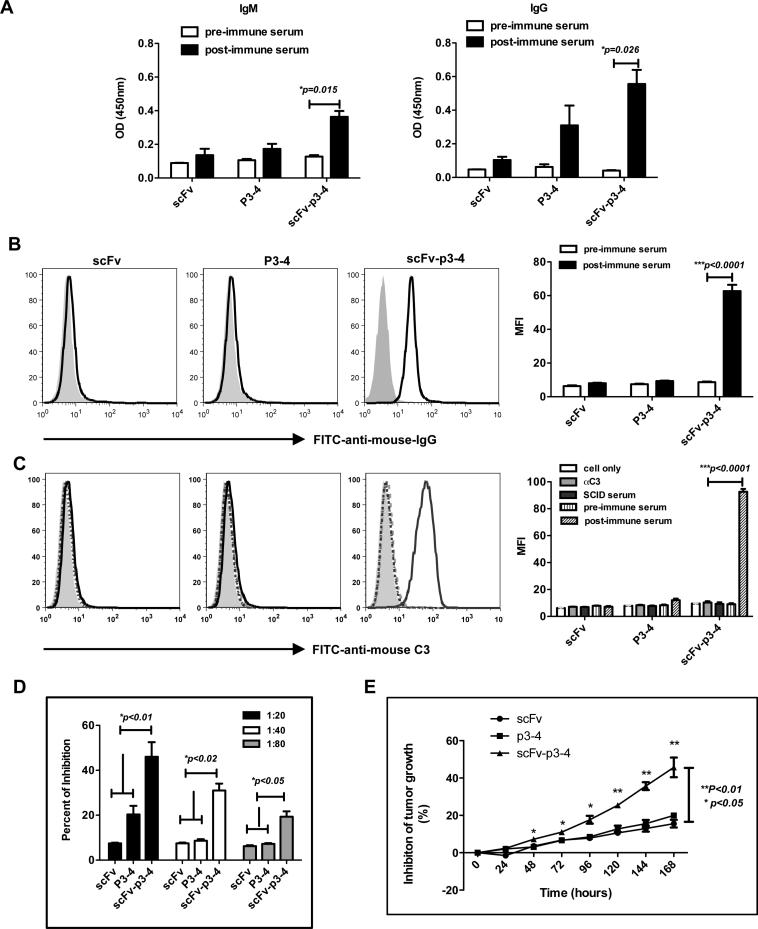
Herceptin-like anti-tumor Abs are elicited by immunization with fusion protein scFv-p3-4
We next tested whether targeting TAA her-2/neu Ag to B cells could elicit anti-tumor Abs. As depicted in Fig. 4A, mice immunized with scFv-p3-4 secreted large amounts of her-2/neu Abs. P3-4 or scFv protein immunization did not elicit any appreciable level of her-2/neu Ab. The Ab specificity was further confirmed with her-2/neu-expressing human ovarian cell line SKOV-3 (Fig. 4B). Next we determined whether immune sera are capable of activating complement, one of the mechanisms for anti-tumor Ab-mediated tumor killing. Immune sera from scFv-p3-4 immunized mice showed potent complement activation (Fig. 4C). Since P3-4 contains the Herceptin-binding domain, we examined whether Abs from mice immunized with scFv-p3-4 have Herceptin-like activity. Competitive inhibition assay was performed in solid phase immunoassay with recombinant her-2/neu protein (P3-4) as the target Ag. The results revealed that the post-immune sera from mice immunized with scFv-p3-4, but not scFv or P3-4, were capable of competing with Herceptin binding (Fig. 4D). Approximately 50% inhibition was achieved when the immune sera were diluted at 1:20 (Fig. 4D).
Figure 4. scFv-p3-4 fusion protein elicits enhanced her-2/neu-specific humoral responses.
(A) Mice (n=3) were immunized with scFv, P3-4, scFv-p3-4 (50 μg/mouse) for 3 times at a 1-week interval, mice were bled at day 7 (IgM) and day 21 (IgG). The sera were measured for her2/neu-specific Abs by ELISA. (B) SKOV-3 tumor cells were stained with sera (1:20) from pre- and post- scFv, P3-4 or scFv-p3-4-immunized mice followed with secondary anti-mouse IgG-FITC and examined by flow cytometer. Summarized mean fluorescent intensity (MFI) was shown in bar graph. (C) SKOV3 tumor cells were incubated with heat inactivated post-immune serum followed by SCID mice serum. FITC-anti-mouse C3 antibody was added. C3 deposition was measured by flow cytometry. Summarized MFI is shown in the bar graph. (D) Ab competitive inhibition assay shows that sera from scFv-p3-4-immunized mice are capable of inhibiting Herceptin-mediated binding. Percent inhibition is shown. (E) 1 × 104 SKBR-3 cells were placed into the wells of the Acea 16-well plates for 24 h. Ten μl of heat-inactivated fusion protein immunized serum or pre-immune serum were added to cells and incubated for 170 h. The inhibition of tumor cell growth was calculated by measuring the relative decrease in current impedance among wells containing immune serum and wells containing pre-immune serum. Representative of 3 experiments.
The biological properties of Herceptin were first described for their ability to inhibit her-2/neu-positive human breast cancer cell growth in vitro (32). Inhibition of tumor cell growth in vitro is also an early and important indication of efficacy in vivo. As shown in Fig. 4E, sera from mice vaccinated with scFv-p3-4 significantly inhibited growth of human breast cancer cell SKBR-3. In contrast, sera from mice immunized with scFv or P3-4 showed minimal inhibition of tumor cell growth. Taken together, these results suggest that targeting her-2/neu p3-4 to B cells via CD19 scFv induces potent Ab response that activates complement and inhibits human breast cancer cell growth in vitro.
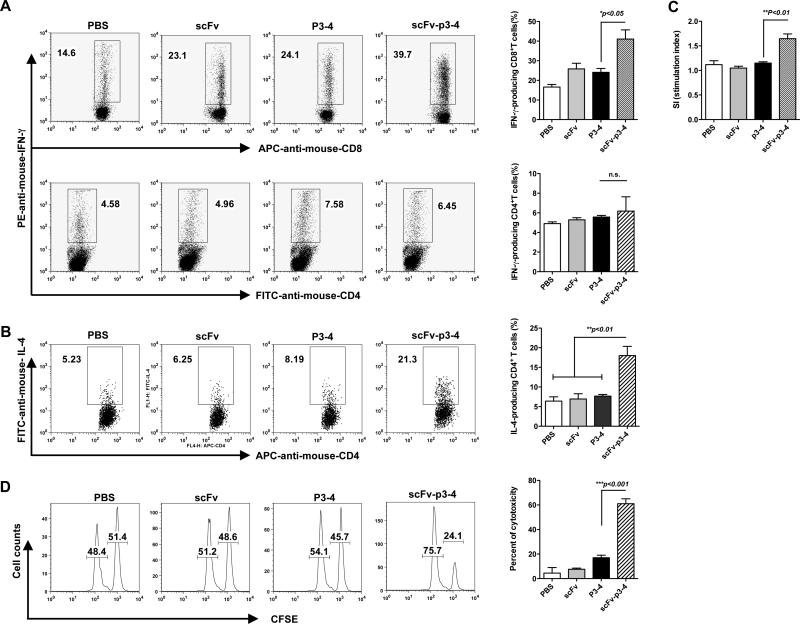
Fusion protein scFv-p3-4 stimulates enhanced CD4 Th2 responses and her-2/neu-specific CD8 T cell responses with augmented cytolytic activity in vivo
To determine whether anti-her-2/neu T- cell responses were elicited by this vaccination strategy, splenocytes from mice vaccinated with different fusion proteins were harvested and stimulated with scFv-p3-4 protein. We found that IFN-γ-producing CD8+ T cells were significantly increased in scFv-p3-4 fusion protein immunized mice as compared to those from scFv-, P3-4-immunized or unimmunized mice (Fig. 5A). However, IFN-γ-producing CD4+ T cells were not significantly different among all groups of immunized mice (Fig. 5A). Since mice immunized with fusion protein scFv-p3-4 elicited potent humoral responses, we next examined IL-4 production from CD4+ T cells. As shown in Fig. 5B, CD4+ T cells from scFv-p3-4 immunized mice secreted significantly more IL-4 as compared to these from other fusion protein immunized mice. In addition, fusion protein scFv-p3-4, but not scFv or P3-4, prompted enhanced T cell proliferation as measured by 3H-thymidine incorporation (Fig. 5C). To determine the cytolytic activity against her-2/neu positive target cells, an in vivo cytotoxicity assay was performed. As shown in Fig. 5D, scFv-p3-4-immunized mice exhibited the highest cytolytic activity (mean=60%) versus less than 20% cytotoxicity in mice immunized with scFv or P3-4 alone (P<0.001).
Figure 5. Increased IFN-γ-producing CD8 T cells, IL-4-producing CD4 T cells, T cell proliferation, and augmented in vivo cytolytic activity elicited by targeting of her-2/neu to B cells.
(A, C) BALB/c mice (n=3) were immunized with scFv, P3-4 or scFv-p3-4 for 4 times at a 1-week interval. Splenocytes from immunized mice were stimulated with scFv-p3-4 (20 μg/ml) for 3 days. Intracellular IFN-γ of CD4 and CD8 (A) was carried out and 3H-thymidine proliferation were measured (C). (B) IL-4 production. Cells from immunized mice (n=3-5) as described above were stimulated with ConA, IL-2, and IL-4 and then re-stimulated with immobilized CD3 and soluble CD28 mAbs. Intracellular IL-4 staining was performed. (D) In vivo cytotoxicity. CFSE labeled syngeneic B cells pulsed with (CFSE high) or without scFv-p3-4 (CFSE low) were injected intravenously into immunized mice. Mice were sacrificed 24 h after transfer and splenocytes were harvested and assessed by flow cytometry. Cells were gated on CFSE-positive cells. Representative histograms and summarized % of cytotoxicity were shown. Data are representative of 3 experiments.
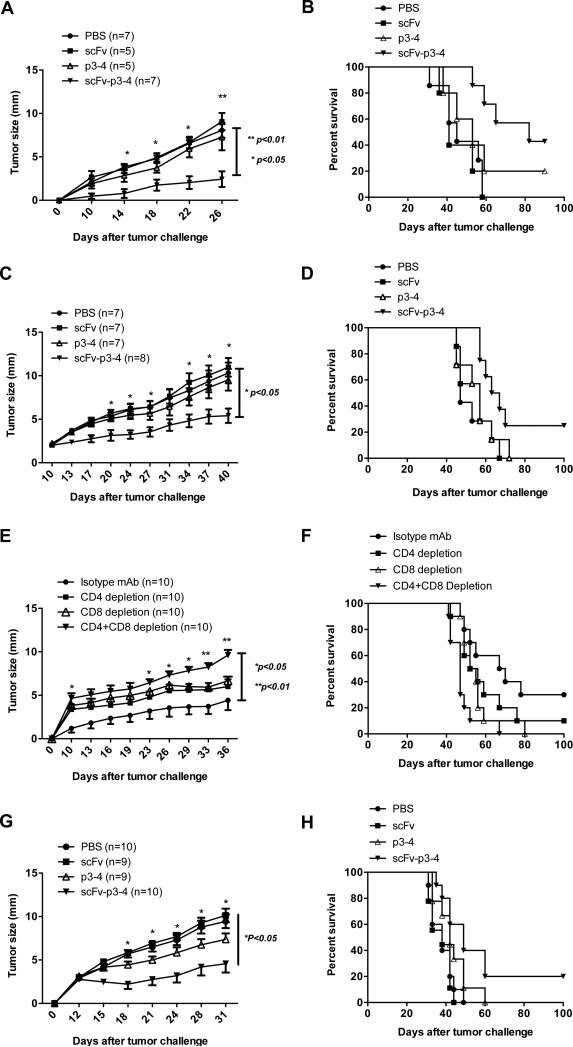
Vaccination with fusion protein scFv-p3-4 induces significant anti-tumor effects
Since fusion protein scFv-p3-4 stimulated her-2/neu Ab response as well as enhanced CD8 T-cell responses in immunized mice, our next step was to determine whether anti-tumor immunity could be established by this vaccination approach. For the tumor prophylactic experiment, Balb/c mice were immunized i.v. with scFv, P3-4, or scFv-p3-4 on days 0,7,14 and 21. Mice immunized with different regimens were then challenged on day 28 by s.c. injection in the flank with 1 × 105 syngeneic D2F2/E2 murine breast cancer cells that express human her-2/neu. As shown in Fig. 6A, mice immunized with scFv-p3-4 had a significantly delayed tumor progression compared with mice immunized with scFv, p3-4 or PBS control mice. In addition, these immunized mice achieved approximately 40% greater long-term, tumor-free survival (Fig. 6B). For the tumor therapeutic experiment, mice were first challenged with 1×105 syngeneic her-2/neu-expressing D2F2/E2 tumor cells. Ten days after tumor inoculation, tumor-bearing mice were treated with fusion proteins scFv-p3-4, scFv, P3-4 or PBS at 1-week intervals. As shown in Fig. 6C, the tumor-bearing mice treated with scFv-p3-4 had a significantly lower tumor burden compared with scFv, p3-4-treated mice or PBS control mice. In addition, these mice achieved approximately 25% long-term, tumor-free survival at day 100 (Fig. 6D). To gain insight into the cellular mechanisms of this vaccine, CD4+ and/or CD8+ T cells were depleted before mice were vaccinated. Her-2/neu Ab was not formed when CD4+ T cells were depleted (data not shown). As shown in Fig. 6E, mice depleted of both CD4+ and CD8+ cells completely lost scFv-p3-4-induced anti-tumor protection. Mice depleted of CD4+ or CD8+ cells showed increased tumor burden but not significantly different from isotype mAb-treated mice. These data suggest that both CD4+ and CD8+ are necessary for the scFv-p3-4-elicited anti-tumor immunity. To further confirm this therapeutic effect, C57Bl/6 mice implanted with human her-2/neu-expressing murine mammary carcinoma EO771 were treated with different regimens. Similar to the D2F2/E2 Babl/c model, the tumor-bearing mice treated with scFv-p3-4 had a significantly lower tumor burden compared with scFv, p3-4-treated mice or PBS control mice (Fig. 6G). In addition, these mice achieved approximately 20% long-term, tumor-free survival at day 100 (Figure 6H).
Figure 6. Reduced tumor burden and enhanced tumor-free survival upon scFv-p3-4 vaccination.
(A, B) BALB/c mice were vaccinated with scFv, P3-4 or scFv-p3-4 for 4 times at a 1-week interval. PBS-immunized mice were used as control. At day 28, immunized mice were challenged s.c. with 1×105 D2F2/E2 tumor cells. Tumor growth (A) and survival (B) were recorded. (C, D) BALB/c mice were challenged s.c. with 1×105 D2F2/E2 tumor cells. The mice were treated with scFv, P3-4 or scFv-p3-4 for 4 times at a 1-week interval when the tumor size (diameter) reached 2-3mm. Tumor growth (C) and survival (D) were recorded. (E, F) BALB/c mice were first injected intraperitoneally with anti-CD4 mAb (250μg), anti-CD8 (500μg) or isotype control mAb (250μg) on day -3. Mice were then immunized with different fusion proteins. The depletion mAbs or isotype control mAb was further injected 3 days before boost immunization. After 4 times immunization, mice were challenged s.c. with 1×105 D2F2/E2 tumor cells. Tumor growth (E) and survival (F) were recorded. (G, H) C57Bl/6 mice were challenged s.c. with 5×105 EO771/E2 tumor cells. Tumor-bearing mice were treated with scFv, P3-4 or scFv-p3-4 for 4 times at a 1-week interval when the tumor size (diameter) reached 2-3mm. Tumor growth (G) and survival (H) were recorded.
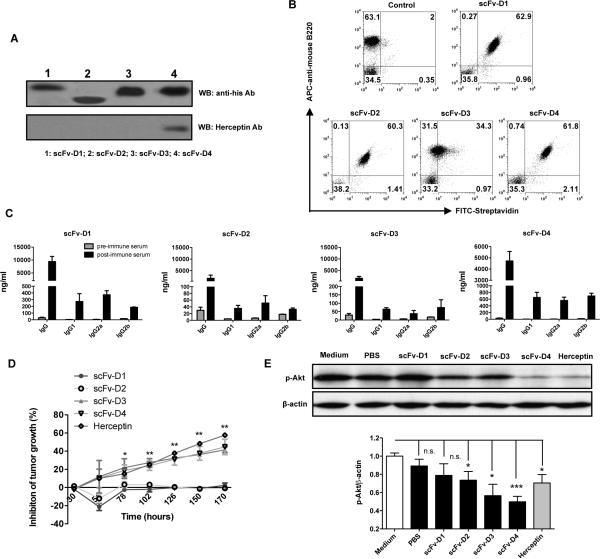
Targeting different ECDs of Her-2/neu to B cells elicits potent Ab responses
Since the her-2/neu ECD contains four different domains, we next investigated whether targeting different her-2/neu ECD domains to B cells could generate Abs specific to particular domains. Four different her-2/neu ECD domains were fused with CD19 scFv to generate fusion proteins. All fusion proteins blotted positively with His-Tag Ab but only scFv-D4 domain fusion protein blotted positively with Herceptin Ab (Fig. 7A). This is consistent with a previous report that the Herceptin-binding domain is located in the her-2/neu ECD D4 domain (33). All fusion proteins bound to B cells with high affinity except scFv-D3 showed rather lower binding affinity to B cells as compared to other fusion proteins (Fig. 7B). Nevertheless, mice immunized with the four fusion proteins generated varying levels of IgG Ab levels with different isotypes (Fig. 7C). Interestingly, in vitro human breast cancer growth inhibition assays indicated that sera from scFv-D3 and scFv-D4 immune mice were as effective as Herceptin in causing growth inhibition (Fig. 7D). Despite a high titer of IgG Ab levels in mice immunized with scFv-D1, the serum did not show any inhibitory effect directly on human breast cancer cells (Fig. 7D). Furthermore, SKBR-3 human breast cancer cells constitutively express high levels of phospho-Akt. Herceptin treatment significantly inhibited p-Akt levels (Fig. 7E). Immune sera from scFv-D4 immunized mice showed similar inhibitory effects. Sera from scFv-D2 and scFv-D3 also showed a significant inhibitory effect on p-Akt levels. However, sera from scFv-D1 immunized mice did not show any effect on p-Akt level (Fig. 7E). These data suggest that targeting different her-2/neu ECDs to B cells via CD19 is capable of generating Abs. However, these Abs could have differential biological effects.
Figure 7. Abs elicited by targeting her-2/neu different ECD domains via CD19 scFV have differential anti-tumor effect in vitro.
(A) Purified fusion proteins scFv-D1, scFv-D2, scFv-D3 and scFv-D4 were blotted with his tag mAb or her-2/neu Ab. (B) For in vitro B cell binding, splenocytes were incubated with biotinylated scFv-D1, scFv-D2, scFv-D3, and scFv-D4 followed by APC-anti-mouse B220 and FITC-streptavidin. (C) Mice were immunized with scFv-D1, scFv-D2, scFv-D3 and scFv-D4 (50 μg/mouse) for 4 times at 1-week intervals. Sera were collected at day 28 and then measured for her2/neu-specific Abs by ELISA. (D) 1 × 104 SKBR-3 cells were placed into the wells of the Acea 16-well plates for 24 h. Heat-inactivated immune serum (1:20), pre-immune serum (1:20), or Herceptin (10 μg/ml) was added to wells and incubated for indicated times. The inhibition of tumor cell growth was calculated by measuring the relative decrease in current impedance among wells containing post-immune serum and wells containing pre-immune serum. (E) Immunoblotting of phosphorylated Akt (p-Akt) in post-immune serum-treated (1:10 dilution), Herceptin (2 μg/ml) or medium-treated SKBR-3 cells. β-Actin served as loading control. Densitometric quantification is also shown (n=4). * p<0.05, ** p<0.01, *** p<0.001. n.s. not significant
Discussion
The goal of this study was to generate a sustained anti-tumor Ab response as well as potent T cell responses. Although targeting Ags to DCs via lectins such as DEC205 and Clec9A has been shown to induce potent T cell responses (34-36), desirable B cell responses to any given Ag require direct contact between naïve B cells and intact Ag (37). Therefore we targeted Ags directly to B cells via a CD19 miniAb. We showed that this approach not only generates augmented humoral response but also potent T cell responses. The efficacy of this B cell-based vaccine was demonstrated in murine breast cancer models. It appears that CD4+ and CD8+ T cells are both required for the vaccine to be effective. In addition, this strategy can be used to generate Ab responses against any Ags of interest.
The uniqueness of the B cell-based vaccine approach is that Ags targeted to B cells elicit exaggerated Ag-specific Ab responses. DCs are conventionally considered as more potent APCs to induce both CD4+ and CD8+ T cell responses (38). They also indirectly promote B cell humoral responses. However, when Ags enter into DCs for the induction of Ab responses, Ags are processed and dominant epitopes are presented on the surface in the context of MHC class I or class II molecules. Generation of blocking or neutralizing Abs requires the presentation of intact Ag to B cells (37). We previously used intact CD19 mAb to target Ags specifically to B cells (16, 17). However, intact mAb conjugates could potentially induce severe inflammatory responses. Indeed, comparison study showed that whole CD19 mAb conjugates induced much more proinflammatory cytokines (Supplemental Figure). We therefore constructed CD19 scFv miniAb and showed that these fusion proteins have high binding affinity to B cells although Kd values were lower than that with intact CD19 mAb. The co-engagement of CD19 and the BCR by fusion protein CD19 scFv-p3-4 activates B cells to upregulate the surface molecules CD40, CD80, CD86, MHC class I and II molecules that are critical for T cell activation and stimulation of low levels of cytokine production including IL-6, TNF-α, and IL-12. Although Ag-specific B cells are normally scarce, non-Ag specific B cells bound with fusion proteins via CD19 may serve as Ag-specific B cells for Ag presentation and T cell activation. However, it is unknown whether Ag internalization and further processing are required for B cell Ag presentation. In contrast, engagement of CD19 alone only induces moderate B cell activation and the BCR alone did not significantly stimulate B cell activation. Thus, it appears that full B cell activation requires co-engagement of CD19 and the BCR by scFv-p3-4 fusion protein. Previous studies have shown that production of IL-6 correlates with B cell vaccine efficacy via direct stimulation of CD8+ T cell proliferation (39). IL-12 has also been shown to promote Th1 differentiation. Indeed, using the surrogate OVA Ag, we demonstrated that fusion protein CD19 scFv-OVA elicited augmented CD4+ and CD8+ T cell proliferation as well as effector differentiation as revealed by more IFN-γ production.
This B cell-based vaccine strategy was extended to use TAA her-2/neu. Anti-human her-2/neu Ab Herceptin has been widely used for metastatic breast cancer patient care (40). It costs as much as US$70,000 per patient per year (24). In addition, a subset of breast cancer patients is refractory to Ab therapy despite high levels of her-2/neu expression on tumor cells. Furthermore, many patients who initially respond to Ab therapy ultimately develop resistance leading to disease progression (41, 42). Previous studies demonstrated that her-2/neu-specific CD8 T cell responses could eradicate drug-refractory tumors (43). Thus it would be ideal if cancer vaccine against her-2/neu TAA could generate both Ab and T cell responses. The current study demonstrated that targeting her-2/neu TAA to B cells via CD19 scFv miniAb elicited potent Ab responses. These Abs competitively inhibited Herceptin-binding ability. More importantly, these her-2/neu Abs are capable of activating complement and inhibiting her-2/neu+ human breast cancer cell line SKBR-3 growth. Additionally, her-2/neu-specific CD8 T cells were significantly enhanced in mice vaccinated with an scFv-p3-4 fusion protein. Although IFN-γ-producing CD4 T cells were not significant different among all groups, IL-4-producing CD4 T cells were significantly increased in scFv-p3-4-immunized mice which is consistent with potent Ab response elicited in these mice. This is in contrast to a recent report in which human her-2/neu protein was targeted to DCs via DEC-205 (44). In this study, DEC-her-2 vaccination with polyI:C as adjuvant induces potent T cell immunity. However, both DEC-her-2 or control her-2 protein induced similar levels of her-2/neu Ab response (44). It is unknown whether these Abs had tumor inhibitory activity.
The therapeutic efficacy of B cell-based vaccine was demonstrated using two murine breast cancer models. Human her-2/neu-expressing D2F2/E2 cells are refractory to Ab treatment but tumors can be controlled by CD8 T cells mediated by her-2 DNA vaccination (23). In the current study, a B cell-based vaccine showed therapeutic efficacy in both prophylactic and therapeutic setting in terms of tumor progression. Tumor-free survival was also enhanced in these mice. Depletion of both CD4+ and CD8+ T cells completely abrogated the vaccine efficacy, suggesting that both CD4+ and CD8+ T cells are required. CD4+ depletion also completely abolished the her-2/neu Ab response (data not shown), suggesting that although co-engagement of CD19 and BCR by fusion protein activates B cells, potent Ab production and isotype switching require CD4+ T cell help. It is worth noting that there was no adjuvant included in the current studies. Since B cells express multiple TLRs (45), addition of a TLR agonist such as CpG or polyI:C may significantly increase Ab and T cell responses. The therapeutic efficacy of this B cell vaccine was further tested in EO771 mammary carcinoma model on C57Bl/6 mice. Similarly, the tumor progression was significantly decreased in the tumor-bearing mice vaccinated with B cell vaccine.
The B cell-based vaccine strategy can be further extended to other areas including infectious disease, particularly for control of viral infection. Previous studies demonstrate that targeting HIV envelope glycoprotein trimers to B cells via a proliferation-inducing ligand (APRIL) induces potent Ab responses (37). Immunodominance is the concept that an antigenic determinant causes it to be responsible for the major immune response in a host. Immunodominance can occur in T and B cells (46). Ab immunodominance is reflected in the fact that the IgG response normally is specific for a single epitope (46). However, combating pathogens or cancer may require Ab and T cell responses against multiple epitopes to circumvent immune selection and escape. Targeting different her-2/neu ECD domains to B cells via CD19 generated Abs against each domain to varying levels. This approach may also offer a new way to generate mAbs. Interestingly, these Abs have different anti-tumor properties. Thus targeting selected, multiple epitopes to B cells may generate broader Ab and T cell responses that can clear pathogens or control tumor progression and recurrence. In summary, targeting Ags to B cells via CD19 miniAb generates both T and B cell responses. This vaccination approach provides a cost effective way to generate a sustained Herceptin-like Ab response as well as anti-tumor T cell responses.
Supplementary Material
Acknowledgements
The authors thank Dr. J.W. Eaton for critical reading of this manuscript.
This work was supported by research funding from National Institutes of Health Grant R01 CA86412, the American Cancer Society Grant RSG-09-190-01, and the Kentucky Lung Cancer Research Board (to J.Y.).
References
- 1.Kirkwood JM, Butterfield LH, Tarhini AA, Zarour H, Kalinski P, Ferrone S. Immunotherapy of cancer in 2012. CA: a cancer journal for clinicians. 2012 doi: 10.3322/caac.20132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Schlom J. Therapeutic cancer vaccines: current status and moving forward. Journal of the National Cancer Institute. 2012;104:599–613. doi: 10.1093/jnci/djs033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Kantoff PW, Higano CS, Shore ND, Berger ER, Small EJ, Penson DF, Redfern CH, Ferrari AC, Dreicer R, Sims RB, Xu Y, Frohlich MW, Schellhammer PF. Sipuleucel-T immunotherapy for castration-resistant prostate cancer. N Engl J Med. 2010;363:411–422. doi: 10.1056/NEJMoa1001294. [DOI] [PubMed] [Google Scholar]
- 4.Krug LM, Ragupathi G, Hood C, George C, Hong F, Shen R, Abrey L, Jennings HJ, Kris MG, Livingston PO. Immunization with N-propionyl polysialic acid-KLH conjugate in patients with small cell lung cancer is safe and induces IgM antibodies reactive with SCLC cells and bactericidal against group B meningococci. Cancer Immunol Immunother. 2012;61:9–18. doi: 10.1007/s00262-011-1083-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Tacken PJ, de Vries IJ, Torensma R, Figdor CG. Dendritic-cell immunotherapy: from ex vivo loading to in vivo targeting. Nat Rev Immunol. 2007;7:790–802. doi: 10.1038/nri2173. [DOI] [PubMed] [Google Scholar]
- 6.Dudziak D, Kamphorst AO, Heidkamp GF, Buchholz VR, Trumpfheller C, Yamazaki S, Cheong C, Liu K, Lee HW, Park CG, Steinman RM, Nussenzweig MC. Differential antigen processing by dendritic cell subsets in vivo. Science. 2007;315:107–111. doi: 10.1126/science.1136080. [DOI] [PubMed] [Google Scholar]
- 7.Weiner LM, Murray JC, Shuptrine CW. Antibody-based immunotherapy of cancer. Cell. 2012;148:1081–1084. doi: 10.1016/j.cell.2012.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ferris RL, Jaffee EM, Ferrone S. Tumor antigen-targeted, monoclonal antibody-based immunotherapy: clinical response, cellular immunity, and immunoescape. J Clin Oncol. 2010;28:4390–4399. doi: 10.1200/JCO.2009.27.6360. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Constant SL. B lymphocytes as antigen-presenting cells for CD4+ T cell priming in vivo. J Immunol. 1999;162:5695–5703. [PubMed] [Google Scholar]
- 10.Guo K, Li J, Tang JP, Tan CP, Hong CW, Al-Aidaroos AQ, Varghese L, Huang C, Zeng Q. Targeting intracellular oncoproteins with antibody therapy or vaccination. Science translational medicine. 2011;3:99ra85. doi: 10.1126/scitranslmed.3002296. [DOI] [PubMed] [Google Scholar]
- 11.Heit A, Huster KM, Schmitz F, Schiemann M, Busch DH, Wagner H. CpG-DNA aided cross-priming by cross-presenting B cells. J Immunol. 2004;172:1501–1507. doi: 10.4049/jimmunol.172.3.1501. [DOI] [PubMed] [Google Scholar]
- 12.DiLillo DJ, Yanaba K, Tedder TF. B cells are required for optimal CD4+ and CD8+ T cell tumor immunity: therapeutic B cell depletion enhances B16 melanoma growth in mice. J Immunol. 2010;184:4006–4016. doi: 10.4049/jimmunol.0903009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Visser KE, Korets LV, Coussens LM. De novo carcinogenesis promoted by chronic inflammation is B lymphocyte dependent. Cancer cell. 2005;7:411–423. doi: 10.1016/j.ccr.2005.04.014. [DOI] [PubMed] [Google Scholar]
- 14.Fujimoto M, Fujimoto Y, Poe JC, Jansen PJ, Lowell CA, DeFranco AL, Tedder TF. CD19 regulates Src family protein tyrosine kinase activation in B lymphocytes through processive amplification. Immunity. 2000;13:47–57. doi: 10.1016/s1074-7613(00)00007-8. [DOI] [PubMed] [Google Scholar]
- 15.Carter RH, Fearon DT. CD19: lowering the threshold for antigen receptor stimulation of B lymphocytes. Science. 1992;256:105–107. [PubMed] [Google Scholar]
- 16.Yan J, Wolff MJ, Unternaehrer J, Mellman I, Mamula MJ. Targeting antigen to CD19 on B cells efficiently activates T cells. Int Immunol. 2005;17:869–877. doi: 10.1093/intimm/dxh266. [DOI] [PubMed] [Google Scholar]
- 17.Ding C, Wang L, Marroquin J, Yan J. Targeting of antigens to B cells augments antigen-specific T-cell responses and breaks immune tolerance to tumor-associated antigen MUC1. Blood. 2008;112:2817–2825. doi: 10.1182/blood-2008-05-157396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Baselga J, Swain SM. Novel anticancer targets: revisiting ERBB2 and discovering ERBB3. Nature reviews. Cancer. 2009;9:463–475. doi: 10.1038/nrc2656. [DOI] [PubMed] [Google Scholar]
- 19.Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344:783–792. doi: 10.1056/NEJM200103153441101. [DOI] [PubMed] [Google Scholar]
- 20.Berns K, Horlings HM, Hennessy BT, Madiredjo M, Hijmans EM, Beelen K, Linn SC, Gonzalez-Angulo AM, Stemke-Hale K, Hauptmann M, Beijersbergen RL, Mills GB, van de Vijver MJ, Bernards R. A functional genetic approach identifies the PI3K pathway as a major determinant of trastuzumab resistance in breast cancer. Cancer cell. 2007;12:395–402. doi: 10.1016/j.ccr.2007.08.030. [DOI] [PubMed] [Google Scholar]
- 21.Shattuck DL, Miller JK, Carraway KL, 3rd, Sweeney C. Met receptor contributes to trastuzumab resistance of Her2-overexpressing breast cancer cells. Cancer Res. 2008;68:1471–1477. doi: 10.1158/0008-5472.CAN-07-5962. [DOI] [PubMed] [Google Scholar]
- 22.Zhang S, Huang WC, Li P, Guo H, Poh SB, Brady SW, Xiong Y, Tseng LM, Li SH, Ding Z, Sahin AA, Esteva FJ, Hortobagyi GN, Yu D. Combating trastuzumab resistance by targeting SRC, a common node downstream of multiple resistance pathways. Nat Med. 2011;17:461–469. doi: 10.1038/nm.2309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Whittington PJ, Piechocki MP, Heng HH, Jacob JB, Jones RF, Back JB, Wei WZ. DNA vaccination controls Her-2+ tumors that are refractory to targeted therapies. Cancer Res. 2008;68:7502–7511. doi: 10.1158/0008-5472.CAN-08-1489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fleck LM. The costs of caring: Who pays? Who profits? Who panders? The Hastings Center report. 2006;36:13–17. doi: 10.1353/hcr.2006.0040. [DOI] [PubMed] [Google Scholar]
- 25.Radkevich-Brown O, Jacob J, Kershaw M, Wei WZ. Genetic regulation of the response to Her-2 DNA vaccination in human Her-2 transgenic mice. Cancer Res. 2009;69:212–218. doi: 10.1158/0008-5472.CAN-08-3092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Benedict CA, MacKrell AJ, Anderson WF. Determination of the binding affinity of an anti-CD34 single-chain antibody using a novel, flow cytometry based assay. Journal of immunological methods. 1997;201:223–231. doi: 10.1016/s0022-1759(96)00227-x. [DOI] [PubMed] [Google Scholar]
- 27.Li B, Allendorf DJ, Hansen R, Marroquin J, Ding C, Cramer DE, Yan J. Yeast beta-glucan amplifies phagocyte killing of iC3b-opsonized tumor cells via complement receptor 3-Syk-phosphatidylinositol 3-kinase pathway. J Immunol. 2006;177:1661–1669. doi: 10.4049/jimmunol.177.3.1661. [DOI] [PubMed] [Google Scholar]
- 28.Solly K, Wang X, Xu X, Strulovici B, Zheng W. Application of real-time cell electronic sensing (RT-CES) technology to cell-based assays. Assay Drug Dev Technol. 2004;2:363–372. doi: 10.1089/adt.2004.2.363. [DOI] [PubMed] [Google Scholar]
- 29.Gilliland LK, Norris NA, Marquardt H, Tsu TT, Hayden MS, Neubauer MG, Yelton DE, Mittler RS, Ledbetter JA. Rapid and reliable cloning of antibody variable regions and generation of recombinant single chain antibody fragments. Tissue Antigens. 1996;47:1–20. doi: 10.1111/j.1399-0039.1996.tb02509.x. [DOI] [PubMed] [Google Scholar]
- 30.Prechl J, Baiu DC, Horvath A, Erdei A. Modeling the presentation of C3d-coated antigen by B lymphocytes: enhancement by CR1/2-BCR co-ligation is selective for the co-ligating antigen. Int Immunol. 2002;14:241–247. doi: 10.1093/intimm/14.3.241. [DOI] [PubMed] [Google Scholar]
- 31.Ye Z, Hellstrom I, Hayden-Ledbetter M, Dahlin A, Ledbetter JA, Hellstrom KE. Gene therapy for cancer using single-chain Fv fragments specific for 4-1BB. Nat Med. 2002;8:343–348. doi: 10.1038/nm0402-343. [DOI] [PubMed] [Google Scholar]
- 32.Baselga J, Norton L, Albanell J, Kim YM, Mendelsohn J. Recombinant humanized anti-HER2 antibody (Herceptin) enhances the antitumor activity of paclitaxel and doxorubicin against HER2/neu overexpressing human breast cancer xenografts. Cancer Res. 1998;58:2825–2831. [PubMed] [Google Scholar]
- 33.Cho HS, Mason K, Ramyar KX, Stanley AM, Gabelli SB, Denney DW, Jr., Leahy DJ. Structure of the extracellular region of HER2 alone and in complex with the Herceptin Fab. Nature. 2003;421:756–760. doi: 10.1038/nature01392. [DOI] [PubMed] [Google Scholar]
- 34.Charalambous A, Oks M, Nchinda G, Yamazaki S, Steinman RM. Dendritic cell targeting of survivin protein in a xenogeneic form elicits strong CD4+ T cell immunity to mouse survivin. J Immunol. 2006;177:8410–8421. doi: 10.4049/jimmunol.177.12.8410. [DOI] [PubMed] [Google Scholar]
- 35.Idoyaga J, Lubkin A, Fiorese C, Lahoud MH, Caminschi I, Huang Y, Rodriguez A, Clausen BE, Park CG, Trumpfheller C, Steinman RM. Comparable T helper 1 (Th1) and CD8 T-cell immunity by targeting HIV gag p24 to CD8 dendritic cells within antibodies to Langerin, DEC205, and Clec9A. Proc Natl Acad Sci U S A. 2011;108:2384–2389. doi: 10.1073/pnas.1019547108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Caminschi I, Proietto AI, Ahmet F, Kitsoulis S, Shin Teh J, Lo JC, Rizzitelli A, Wu L, Vremec D, van Dommelen SL, Campbell IK, Maraskovsky E, Braley H, Davey GM, Mottram P, van de Velde N, Jensen K, Lew AM, Wright MD, Heath WR, Shortman K, Lahoud MH. The dendritic cell subtype-restricted C-type lectin Clec9A is a target for vaccine enhancement. Blood. 2008;112:3264–3273. doi: 10.1182/blood-2008-05-155176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Melchers M, Bontjer I, Tong T, Chung NP, Klasse PJ, Eggink D, Montefiori DC, Gentile M, Cerutti A, Olson WC, Berkhout B, Binley JM, Moore JP, Sanders RW. Targeting HIV-1 envelope glycoprotein trimers to B cells by using APRIL improves antibody responses. J Virol. 2012;86:2488–2500. doi: 10.1128/JVI.06259-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Steinman RM. Decisions about dendritic cells: past, present, and future. Annu Rev Immunol. 2012;30:1–22. doi: 10.1146/annurev-immunol-100311-102839. [DOI] [PubMed] [Google Scholar]
- 39.Vanden Bush TJ, Buchta CM, Claudio J, Bishop GA. Cutting Edge: Importance of IL-6 and cooperation between innate and adaptive immune receptors in cellular vaccination with B lymphocytes. J Immunol. 2009;183:4833–4837. doi: 10.4049/jimmunol.0900968. [DOI] [PubMed] [Google Scholar]
- 40.Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE, Jr., Davidson NE, Tan-Chiu E, Martino S, Paik S, Kaufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353:1673–1684. doi: 10.1056/NEJMoa052122. [DOI] [PubMed] [Google Scholar]
- 41.Nahta R. Pharmacological strategies to overcome HER2 cross-talk and Trastuzumab resistance. Current medicinal chemistry. 2012;19:1065–1075. doi: 10.2174/092986712799320691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Nahta R, O'Regan RM. Therapeutic implications of estrogen receptor signaling in HER2-positive breast cancers. Breast cancer research and treatment. 2012 doi: 10.1007/s10549-012-2067-8. [DOI] [PubMed] [Google Scholar]
- 43.Whittington PJ, Radkevich-Brown O, Jacob JB, Jones RF, Weise AM, Wei WZ. Her-2 DNA versus cell vaccine: immunogenicity and anti-tumor activity. Cancer Immunol Immunother. 2009;58:759–767. doi: 10.1007/s00262-008-0599-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wang B, Zaidi N, He LZ, Zhang L, Kuroiwa JM, Keler T, Steinman RM. Targeting of the non-mutated tumor antigen HER2/neu to mature dendritic cells induces an integrated immune response that protects against breast cancer in mice. Breast Cancer Res. 2012;14:R39. doi: 10.1186/bcr3135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Rawlings DJ, Schwartz MA, Jackson SW, Meyer-Bahlburg A. Integration of B cell responses through Toll-like receptors and antigen receptors. Nat Rev Immunol. 2012;12:282–294. doi: 10.1038/nri3190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yewdell JW, Bennink JR. Immunodominance in major histocompatibility complex class I-restricted T lymphocyte responses. Annu Rev Immunol. 1999;17:51–88. doi: 10.1146/annurev.immunol.17.1.51. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.