Abstract
Advanced glycation end products (AGEs) and their receptors are strongly implicated in the development of diabetes complications. When stimulated by AGEs, the receptors for AGEs (RAGEs) induce inflammation and are thought to fuel disease progression. Soluble circulating RAGE (sRAGE) may counteract the detrimental effects of RAGE. We measured sRAGE in stored plasma from a random sample of 1,201 participants in the Atherosclerosis Risk in Communities (ARIC) Study who were aged 47–68 years, had normal kidney function, and had no history of cardiovascular disease. In cross-sectional analyses, black race, male sex, higher BMI, and higher C-reactive protein were independently associated with low sRAGE. The racial difference was striking, with blacks approximately three times more likely to have low sRAGE compared with whites even after adjustment. During ~18 years of follow-up, there were 192 incident coronary heart disease events, 53 ischemic strokes, 213 deaths, and 253 cases of diabetes (among the 1,057 persons without diabetes at baseline). In multivariable Cox models comparing risk in the first quartile with that in the fourth quartile of baseline sRAGE, low levels of sRAGE were significantly associated with risk of diabetes (hazard ratio 1.64 [95% CI 1.10–2.44]), coronary heart disease (1.82 [1.17–2.84]), and mortality (1.72 [1.11–2.64]) but not ischemic stroke (0.78 [0.34–1.79]). In conclusion, we found that low levels of sRAGE were a marker of future chronic disease risk and mortality in the community and may represent an inflammatory state. Racial differences in sRAGE deserve further examination.
Advanced glycation end products (AGEs) are strongly implicated in the development of diabetic vascular disease (1–3) and are of particular interest as novel biomarkers because they are a postulated etiologic link between hyperglycemia and diabetes complications (3–8). It is also thought that AGEs contribute to the development of vascular disease in nondiabetic people through their pro-oxidant activities (4,5). AGEs bind a variety of receptors, and circulating levels of AGE receptors are thought to be influenced by a number of endogenous (e.g., glucose, inflammation) and exogenous (e.g., smoking, diet) factors. The most widely studied AGE receptor is receptor for AGEs (RAGE), which is expressed in the vasculature, retina, kidney, and inflammatory cells. RAGE is considered a multiligand receptor of the immunoglobulin superfamily, binding, in addition to AGEs, S100/calgranulins, high-mobility group box-1, amyloid-β peptide, and other molecules (9). The COOH terminus of the protein is located on the extracellular surface. The NH2-terminus of RAGE is essential in activating proinflammatory nuclear factor (NF)-κB–mediated signaling. When stimulated by AGEs, RAGE induces inflammation, contributes to tissue injury, and fuels the progression of chronic disease through NF-kB–mediated signaling (10–13). The soluble receptor for AGEs (sRAGE) is the isoform of RAGE found in serum and is primarily formed by proteolytic cleavage of RAGE and secondarily by endogenously secreted RAGE (esRAGE). esRAGE can be measured independently, comprises roughly one-quarter of total serum RAGE (14,15), and is highly correlated with total sRAGE levels (14,16). sRAGE has been described as a “sponge” for AGEs and may have protective functions, as it lacks the NH2-terminus and cannot activate NF-κB signaling. Levels are primarily dependent on cell-surface RAGE levels (17).
Recent studies have demonstrated inverse associations of serum sRAGE with clinical outcomes in persons with diabetes or kidney disease (18–21) and inverse cross-sectional associations with measures of coronary heart disease or atherosclerosis in more general populations (15,22–24). However, few prospective studies have been conducted in diverse, community-based populations using robust ELISA methods for measurement of sRAGE. The objective of this study was to characterize the association of sRAGE with risk of diabetes, coronary heart disease, stroke, and all-cause mortality in a community-based population.
RESEARCH DESIGN AND METHODS
The Atherosclerosis Risk in Communities (ARIC) Study is a community-based prospective cohort of 15,792 middle-aged adults from four U.S. communities. The first examination of participants (visit 1) took place during 1987–1989, with three follow-up visits taking place: each approximately every 3 years. A fifth examination is currently ongoing (2011–2013). The study population for the current study is comprised of a subsample of participants who attended visit 2 during 1990–1992. A random sample of 1,289 participants with normal kidney function (estimated glomerular filtration rate >60 mL/min/1.73 m2) was selected from the 14,348 participants who attended visit 2. The final sample sizes used in the current study were 1,201 after excluding those who were missing sRAGE or covariates of interest and those with a history of cardiovascular disease and 1,057 after further excluding persons with diabetes defined by a fasting glucose ≥126 mg/dL, a nonfasting glucose ≥200 mg/dL, an HbA1c value ≥6.5%, a self-reported physician diagnosis of diabetes, or diabetes medication use at visit 2.
Measurement of sRAGE.
sRAGE was measured in 2010 by ELISA (R&D Systems, Minneapolis, MN) from plasma samples that had been in storage since collection from 1990 to 1992. The intra- and interassay coefficients of variation for the assay were 2.8 and 9.6%, respectively. sRAGE has been shown to be highly stable when measured from stored samples and robust to multiple freeze-thaw cycles (25).
Other variables of interest.
Plasma lipid levels (26,27), BMI (28), and blood pressure (29) were measured according to published methods. C-reactive protein was measured in 2008 from stored plasma samples using an immunoturbidimetric assay on the Siemens (Dade Behring, Deerfield, IL) BNII analyzer (Dade Behring). Hypertension was defined as average systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 Hg, or blood pressure–lowering medication use in the past 2 weeks. Participants reported their age, race, and smoking status.
Surveillance for incident diabetes.
During each ARIC visit, information was obtained on self-reported diabetes, glucose-lowering medication use, and serum glucose. After visit 4, diabetes cases were identified exclusively on the basis of information on self-reported diagnosis or diabetes medication use obtained during the annual telephone calls to all participants. We combined these two sources of information to define incident diabetes according to a standard time-to-diabetes definition based on the visit-based information (30,31) and self-reported information on diagnosed diabetes or glucose-lowering medication use obtained during a median of 12 years of annual telephone calls after visit 4 (32).
Surveillance for incident coronary heart disease, stroke, and all-cause mortality.
The ascertainment of deaths and classification of cardiovascular events have previously been described (33,34). Briefly, potential cardiovascular hospitalizations were reported annually by participants and also identified through community-wide hospital surveillance. Trained personnel abstracted hospital records related to possible cardiovascular events (34). Silent myocardial infarctions, as detected by means of electrocardiography during the visits, were identified and recorded. We defined newly diagnosed coronary heart disease as a definite or probable myocardial infarction, a death from coronary heart disease, a cardiac procedure, or electrocardiographic evidence of a silent myocardial infarction. We also examined definite or probable ischemic stroke. Adjudicated follow-up data for cardiovascular events were available up to 1 January 2010.
Statistical analysis.
Baseline characteristics of the study population were calculated overall and by quartiles of baseline sRAGE levels. We evaluated crude cross-sectional associations between sRAGE and the continuous variables using Spearman correlations and compared mean sRAGE levels across categorical variables using t tests. We constructed kernel density plots to visually compare sRAGE distributions by race group, diabetes, and hypertension status. We used multivariable logistic regression models to evaluate the cross-sectional associations of participant characteristics with low sRAGE (lowest quartile) at baseline. We also conducted supplemental cross-sectional analyses using Poisson regression, since the odds ratio (OR) from the logistic regression model is a nonconservative estimate of the prevalence ratio in this setting; the prevalence of the outcome is 25% by definition (quartile 1 of sRAGE). For prospective analyses, adjusted hazard ratios (HRs) and corresponding 95% CIs for each outcome were estimated using Cox proportional hazards models. Baseline sRAGE was modeled continuously using linear and restricted cubic spline (35) models (centered at the 50th percentile of sRAGE) and in quartiles, with quartile 4 as the reference group. Model 1 was adjusted for age, sex, and race. Model 2 was adjusted for Framingham Risk Score (which includes age, sex, current smoking status, systolic blood pressure, total cholesterol, HDL cholesterol, and diabetes status [except for models of diabetes]) plus C-reactive protein (milligrams per liter), BMI (weight in kilograms divided by the square of height in meters), and race. Models of incident diabetes excluded persons with a history of diabetes at baseline (N = 144). We conducted sensitivity analyses with additional adjustment for family history of diabetes, estimated glomerular filtration rate, and fasting glucose and analyses of risk of cardiovascular outcomes and death after excluding persons with diabetes (diagnosed or undiagnosed) at baseline.
RESULTS
Cross-sectional associations with low sRAGE.
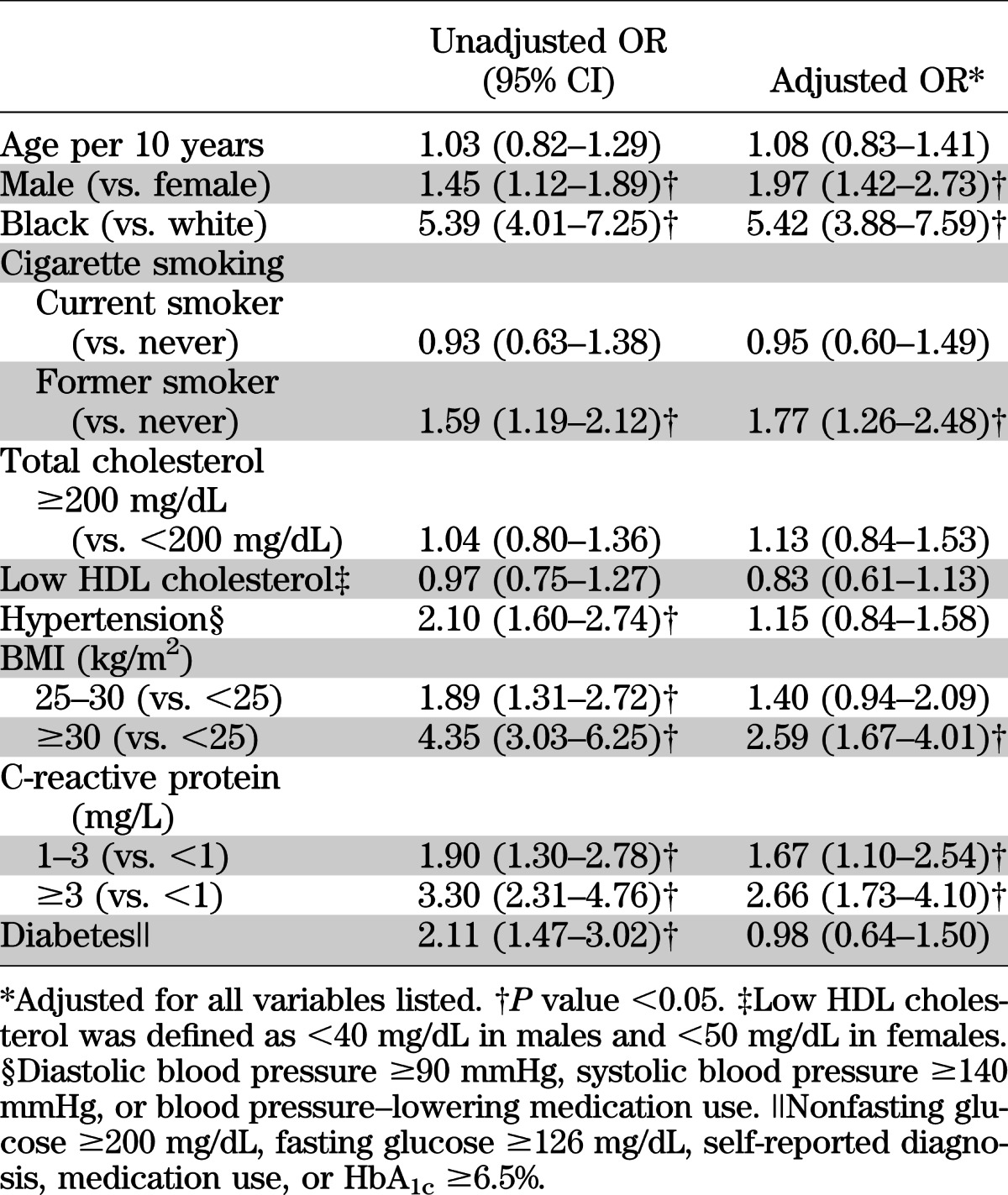
Compared with the higher quartiles, the lowest quartile of sRAGE had a higher proportion of men, black participants, and participants with diabetes or hypertension (Table 1). Mean levels of C-reactive protein and BMI also were highest in the lowest quartile of sRAGE. The distribution of sRAGE in blacks was substantially shifted toward lower levels compared with whites; mean (SD) sRAGE in blacks was 756 (413) pg/mL compared with 1,122 (469) pg/mL in whites (P < 0.0001) (Supplementary Fig. 1D). There also was a significant difference in mean sRAGE comparing persons with and without diabetes (905 vs. 1,052 pg/mL, P = 0.0114) (Supplementary Fig. 1C). The strongest crude (Spearman) correlations between baseline sRAGE and the continuous variables examined were for BMI (r = −0.32), C-reactive protein (r = −0.25), HbA1c (r = −0.22), fasting glucose (r = −0.22), and systolic blood pressure (r = −0.18) (Supplementary Table 1). All other correlations were low to moderate (|r| < 0.12). After simultaneous adjustment for each of the risk factors in a multivariable logistic regression model, male sex, black race, former smoking, obesity (BMI ≥30 kg/m2), and C-reactive protein remained significantly associated with low sRAGE (Table 2). In this model, diabetes was no longer significantly associated with low sRAGE after adjustment for the other variables. Race remained highly associated with sRAGE. The prevalence of low sRAGE among blacks was almost three times the prevalence in whites, even after adjustment (Supplementary Table 2).
TABLE 1.
Baseline characteristics of the study population by quartiles of sRAGE (N = 1,201)
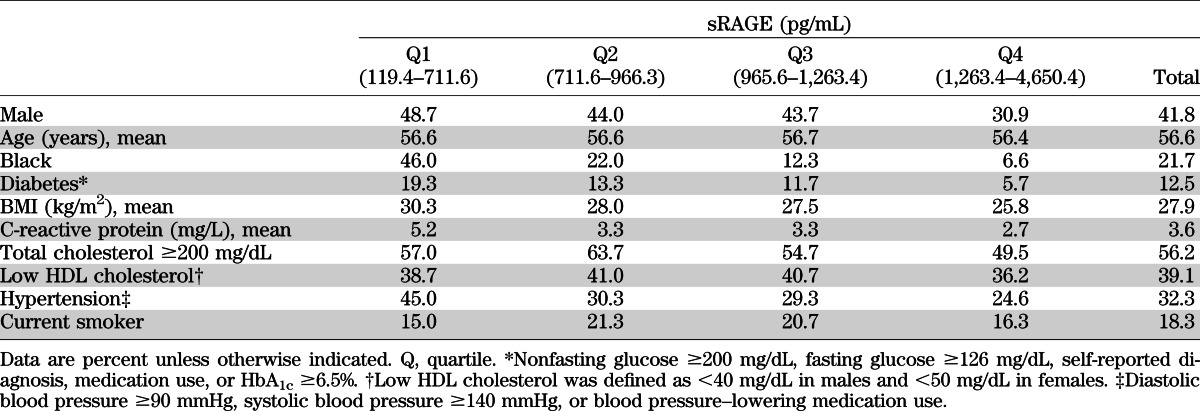
TABLE 2.
Unadjusted and adjusted* ORs for the lowest quartile of sRAGE
Prospective associations of sRAGE with outcomes.
The median follow-up time was ~18 years. In the study population of 1,057 persons without diabetes or cardiovascular disease at baseline, there were 253 cases of incident diabetes. In the study population of 1,201 persons without cardiovascular disease at baseline, there were 192 cases of incident coronary heart disease, 53 cases of ischemic stroke, and 213 total deaths. Low baseline levels of sRAGE were associated with risk of diabetes, coronary heart disease, and all-cause mortality (Table 3 and Fig. 1). Adjustment for traditional risk factors attenuated but did not eliminate these associations. Compared with persons in the highest quartile of sRAGE, persons in the lowest quartile had a significant and independent increased risk of diabetes (HR 1.64 [95% CI 1.10–2.44]), coronary heart disease (1.82 [1.17–2.84]), or all-cause mortality (1.72 [1.11–2.64]). There was no significant association of baseline sRAGE and incident ischemic stroke before or after adjustment. The shape of the association of sRAGE with incident diabetes was roughly linear (Fig. 1), with lower sRAGE levels associated with higher risk across the range of the exposure. For coronary heart disease, the slope appeared steeper for sRAGE levels below the median (965.6 pg/mL) (P value = 0.016 for slope from the piece-wise linear model for coronary heart disease before the median). The P value for the slope from the piece-wise linear model for mortality was <0.001 before the first knot (711.1 pg/mL). The results for coronary heart disease, stroke, and all-cause mortality were not appreciably altered after exclusion of persons with diagnosed or undiagnosed diabetes at baseline (Supplementary Table 3). Results were also similar after additional adjustment for family history of diabetes, kidney function, and fasting glucose. Censoring of incident cases of diabetes that occurred during follow-up but prior to the onset of coronary heart disease also did not change the associations observed with incident coronary heart disease (data not shown).
TABLE 3.
HRs for incident diabetes, coronary heart disease, ischemic stroke, and mortality by sRAGE quartile at baseline
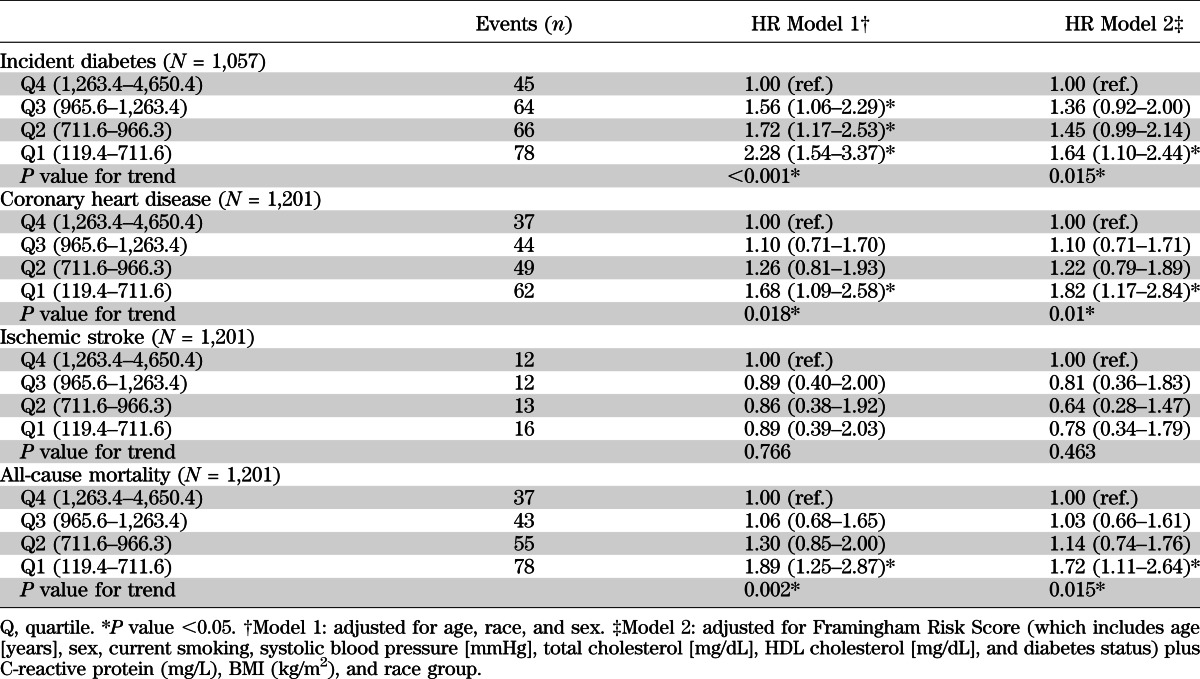
FIG. 1.
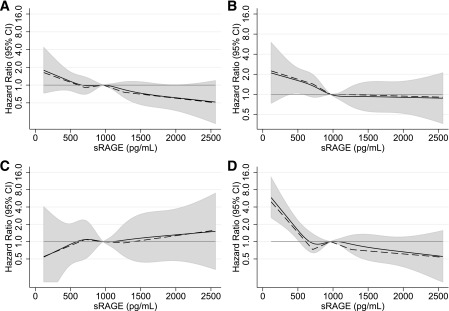
Adjusted HRs (95% CI) for baseline sRAGE and incident diabetes (A), coronary heart disease (B), ischemic stroke (C), and all-cause mortality (D). Adjusted HRs are from Cox proportional hazards models with adjustment for Framingham Risk Score (which includes age [years], sex, current smoking, systolic blood pressure [mmHg], total cholesterol [mg/dL], HDL cholesterol [mg/dL], and diabetes status) plus C-reactive protein (mg/L), BMI (kg/m2), and race group. Baseline sRAGE was modeled using restricted cubic splines (solid lines) with knots at the 5th, 27.5th, 50th, 72.5th, and 95th percentiles and piece-wise linear splines (dashed lines) with knots at the quartiles of sRAGE (711.1, 965.6, 1,263.8 pg/mL). Both models are centered at the 50th percentile (sRAGE 965.6 pg/mL). The shaded areas are the CIs for the restricted cubic spline models. The graphs are truncated at the 99th percentile of sRAGE.
Because we observed substantial racial differences in baseline levels of sRAGE, we conducted race-specific sensitivity analyses. In analyses stratified by race (Supplementary Tables 4 and 5), our results remained significant in whites but the trends for the associations of quartiles of sRAGE with incident diabetes, stroke, and all-cause mortality in blacks were not significant, likely owing to the small number of black participants in this study (N = 261). The association of sRAGE with incident coronary heart disease in blacks was actually opposite the direction (Supplementary Table 4) of the association in whites but not in a graded fashion (i.e., no significant trend); however, there were only 36 total coronary heart disease events among blacks in our study population, suggesting that these results are highly unreliable.
DISCUSSION
During a median of 18 years of follow-up, we found that low levels of sRAGE at baseline were independently associated with risk of diabetes, coronary heart disease, and all-cause mortality, but not stroke, in this community-based population. We also found that blacks were substantially more likely to have low levels of sRAGE levels at baseline compared with whites and that this racial difference was not explained by demographic, metabolic, or inflammatory factors. Even after adjustment, blacks were approximately three times as likely to have low sRAGE (<711.6 pg/mL) compared with whites. The magnitude of this race difference is striking and deserves further examination. Male sex, BMI, and C-reactive protein were also independently associated with low sRAGE at baseline. After adjustment, sRAGE levels did not differ by diabetes status at baseline, suggesting that sRAGE may not be as specific to diabetes as previously thought. The robust associations of sRAGE with risk of coronary heart disease and mortality, in addition to incident diabetes, suggest that sRAGE may be a generalized marker of ill health.
The glycation and chemical modification of long-lived proteins through a series of slow reactions (Schiff, Amadori, and Maillard reactions) to form AGEs and their cross-linking of extracellular matrix materials such as collagen are thought to result in vascular stiffness (36,37) and vascular complications of diabetes (2,38–41). There is evidence from laboratory studies, particularly in animal models, that circulating RAGE (sRAGE) counteracts the detrimental effects of cellular RAGE by binding serum AGEs (42,43). Perhaps mechanisms that promote cleavage and shedding of full-length RAGE to create sRAGE may be beneficial to health. ADAM10 and other metalloproteinases cleave RAGE resulting in sRAGE shedding (44). Our results provide further evidence that low levels of sRAGE may be a marker of long-term chronic disease risk.
Few studies have characterized AGEs in serum and investigated their associations with long-term health outcomes in a general population. Previous studies have documented inverse correlations between sRAGE with C-reactive protein (45–47) and low levels of sRAGE among persons with coronary heart disease (22,47,48). Our finding of substantially lower sRAGE levels in blacks compared with whites suggests that racial differences in sRAGE are an important area for further investigation. Previous studies also have reported significantly lower levels of sRAGE in blacks compared with whites (14,22,49). Nonetheless, owing to small numbers of events in the subgroup of black participants in our study, we were unable to rigorously evaluate potential effect modification by race on the association of sRAGE with long-term outcomes.
Important limitations of this study include the reliance on a single measurement of sRAGE at baseline, which will vary within individuals over time. Indeed, there is evidence that aerobic exercise (50) and use of certain medications (51,52) may acutely affect circulating sRAGE levels. Because of sample size limitations and correspondingly low power in subgroups, we were unable to rigorously examine the associations of sRAGE in subpopulations or evaluate possible effect modification. Indeed, our power was particularly limited for analyses of incident ischemic stroke (N = 53 cases overall) and for race-stratified analyses (N = 261 black participants at baseline). Owing to the observational nature of this investigation, the possibility of residual confounding cannot be eliminated. Nonetheless, this report is one of the first to investigate sRAGE in a racially diverse, community-based cohort with long-term follow-up for clinical outcomes. Major additional strengths include the rigorous measurement of cardiovascular disease risk factors and the use of comprehensive surveillance and adjudication of cardiovascular events.
In summary, our study demonstrated significant, independent associations between low sRAGE and future risk of diabetes, coronary heart disease, and all-cause mortality but not ischemic stroke in the general population. Our observation of substantially lower levels of sRAGE in blacks compared with whites suggests underlying racial variation in sRAGE production. In cross-sectional analyses, we found that male sex, higher BMI, and higher C-reactive protein were independently associated with low sRAGE. Taken as a whole, our results suggest that low levels of sRAGE are a marker of future chronic disease risk and mortality in the general population and may represent an inflammatory state.
Supplementary Material
ACKNOWLEDGMENTS
This research was supported by National Institutes of Health/National Institute of Diabetes and Digestive and Kidney Diseases Grant R01-DK076770. The ARIC Study is carried out as a collaborative study supported by National Heart, Lung, and Blood Institute contracts (HHSN268201100005C, HHSN268201100006C, HHSN268201100007C, HHSN268201100008C, HHSN268201100009C, HHSN268201100010C, HHSN268201100011C, and HHSN268201100012C).
No potential conflicts of interest relevant to this article were reported.
E.S. wrote the manuscript and analyzed data. M.K.H. reviewed and edited the manuscript and contributed to the discussion. A.M.R. analyzed the data and reviewed and edited the manuscript. R.C.H. and C.M.B. were responsible for laboratory measurements and reviewed and edited the manuscript. J.C. helped design the study, contributed to discussion, and reviewed and edited the manuscript. B.C.A. designed the study, contributed to discussion, and reviewed and edited the manuscript. E.S. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Parts of this study were presented in abstract form at the 72nd Scientific Sessions of the American Diabetes Association, Philadelphia, Pennsylvania, 8–12 June 2012.
The authors thank the staff and participants of the ARIC Study for their important contributions.
Footnotes
This article contains Supplementary Data online at http://diabetes.diabetesjournals.org/lookup/suppl/doi:10.2337/db12-1528/-/DC1.
REFERENCES
- 1.Peppa M, Vlassara H. Advanced glycation end products and diabetic complications: a general overview. Hormones (Athens) 2005;4:28–37 [DOI] [PubMed] [Google Scholar]
- 2.Genuth S, Sun W, Cleary P, et al. DCCT Skin Collagen Ancillary Study Group Glycation and carboxymethyllysine levels in skin collagen predict the risk of future 10-year progression of diabetic retinopathy and nephropathy in the diabetes control and complications trial and epidemiology of diabetes interventions and complications participants with type 1 diabetes. Diabetes 2005;54:3103–3111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Monnier VM, Sell DR, Genuth S. Glycation products as markers and predictors of the progression of diabetic complications. Ann N Y Acad Sci 2005;1043:567–581 [DOI] [PubMed] [Google Scholar]
- 4.Brownlee M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 2005;54:1615–1625 [DOI] [PubMed] [Google Scholar]
- 5.Peppa M, Uribarri J, Vlassara H. Aging and glycoxidant stress. Hormones (Athens) 2008;7:123–132 [DOI] [PubMed] [Google Scholar]
- 6.Vlassara H, Striker G. Glycotoxins in the diet promote diabetes and diabetic complications. Curr Diab Rep 2007;7:235–241 [DOI] [PubMed] [Google Scholar]
- 7.REF 2Genuth S, Sun W, Cleary P, et al. DCCT Skin Collagen Ancillary Study Group Glycation and carboxymethyllysine levels in skin collagen predict the risk of future 10-year progression of diabetic retinopathy and nephropathy in the diabetes control and complications trial and epidemiology of diabetes interventions and complications participants with type 1 diabetes. Diabetes 2005;54:3103–3111 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Monnier VM, Cerami A. Nonenzymatic browning in vivo: possible process for aging of long-lived proteins. Science 1981;211:491–493 [DOI] [PubMed] [Google Scholar]
- 9.Schmidt AM, Yan SD, Yan SF, Stern DM. The biology of the receptor for advanced glycation end products and its ligands. Biochim Biophys Acta 2000;1498:99–111 [DOI] [PubMed] [Google Scholar]
- 10.Yan SF, Ramasamy R, Schmidt AM. Mechanisms of disease: advanced glycation end-products and their receptor in inflammation and diabetes complications. Nat Clin Pract Endocrinol Metab 2008;4:285–293 [DOI] [PubMed] [Google Scholar]
- 11.Yan SF, Ramasamy R, Schmidt AM. The receptor for advanced glycation endproducts (RAGE) and cardiovascular disease. Expert Rev Mol Med 2009;11:e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yan SF, Barile GR, D’Agati V, Du Yan S, Ramasamy R, Schmidt AM. The biology of RAGE and its ligands: uncovering mechanisms at the heart of diabetes and its complications. Curr Diab Rep 2007;7:146–153 [DOI] [PubMed] [Google Scholar]
- 13.Ramasamy R, Vannucci SJ, Yan SS, Herold K, Yan SF, Schmidt AM. Advanced glycation end products and RAGE: a common thread in aging, diabetes, neurodegeneration, and inflammation. Glycobiology 2005;15:16R–28R [DOI] [PubMed] [Google Scholar]
- 14.Colhoun HM, Betteridge DJ, Durrington P, et al. Total soluble and endogenous secretory receptor for advanced glycation end products as predictive biomarkers of coronary heart disease risk in patients with type 2 diabetes: an analysis from the CARDS trial. Diabetes 2011;60:2379–2385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Semba RD, Ferrucci L, Sun K, et al. Advanced glycation end products and their circulating receptors predict cardiovascular disease mortality in older community-dwelling women. Aging Clin Exp Res 2009;21:182–190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yang SJ, Kim S, Hwang SY, et al. Association between sRAGE, esRAGE levels and vascular inflammation: analysis with (18)F-fluorodeoxyglucose positron emission tomography. Atherosclerosis 2012;220:402–406 [DOI] [PubMed] [Google Scholar]
- 17.Busch M, Franke S, Rüster C, Wolf G. Advanced glycation end-products and the kidney. Eur J Clin Invest 2010;40:742–755 [DOI] [PubMed] [Google Scholar]
- 18.Basta G, Leonardis D, Mallamaci F, et al. Circulating soluble receptor of advanced glycation end product inversely correlates with atherosclerosis in patients with chronic kidney disease. Kidney Int 2010;77:225–231 [DOI] [PubMed] [Google Scholar]
- 19.Gross S, van Ree RM, Oterdoom LH, et al. Low levels of sRAGE are associated with increased risk for mortality in renal transplant recipients. Transplantation 2007;84:659–663 [DOI] [PubMed] [Google Scholar]
- 20.Kim JK, Park S, Lee MJ, et al. Plasma levels of soluble receptor for advanced glycation end products (sRAGE) and proinflammatory ligand for RAGE (EN-RAGE) are associated with carotid atherosclerosis in patients with peritoneal dialysis. Atherosclerosis 2012;220:208–214 [DOI] [PubMed] [Google Scholar]
- 21.Katakami N, Matsuhisa M, Kaneto H, et al. Serum endogenous secretory RAGE level is an independent risk factor for the progression of carotid atherosclerosis in type 1 diabetes. Atherosclerosis 2009;204:288–292 [DOI] [PubMed] [Google Scholar]
- 22.Lindsey JB, de Lemos JA, Cipollone F, et al. Association between circulating soluble receptor for advanced glycation end products and atherosclerosis: observations from the Dallas Heart Study. Diabetes Care 2009;32:1218–1220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Falcone C, Emanuele E, D’Angelo A, et al. Plasma levels of soluble receptor for advanced glycation end products and coronary artery disease in nondiabetic men. Arterioscler Thromb Vasc Biol 2005;25:1032–1037 [DOI] [PubMed] [Google Scholar]
- 24.Dimitriadis K, Tsioufis C, Kasiakogias A, et al. Soluble receptor for advanced glycation end-products levels are related to albuminuria and arterial stiffness in essential hypertension. Nutr Metab Cardiovasc Dis. 31 December 2011 [Epub ahead of print] [DOI] [PubMed]
- 25.Wittwer C, Lehner J, Fersching D, Siegele B, Stoetzer OJ, Holdenrieder S. Methodological and preanalytical evaluation of a RAGE immunoassay. Anticancer Res 2012;32:2075–2078 [PubMed] [Google Scholar]
- 26.Siedel J, Hägele EO, Ziegenhorn J, Wahlefeld AW. Reagent for the enzymatic determination of serum total cholesterol with improved lipolytic efficiency. Clin Chem 1983;29:1075–1080 [PubMed] [Google Scholar]
- 27.Operations Manual No. 10: Clinical Chemistry Determinations, Version 1.0. Chapel Hill, NC, University of North Carolina, 1987 [Google Scholar]
- 28.Operations Manual No. 2: Cohort Component Procedures, Version 1.0. Chapel Hill, NC, University of North Carolina, 1987 [Google Scholar]
- 29.Operations Manual No. 11: Sitting Blood Pressure, Version 1.0. Chapel Hill, NC, University of North Carolina, 1987 [Google Scholar]
- 30.Duncan BB, Schmidt MI, Pankow JS, et al. Atherosclerosis Risk in Communities Study Low-grade systemic inflammation and the development of type 2 diabetes: the atherosclerosis risk in communities study. Diabetes 2003;52:1799–1805 [DOI] [PubMed] [Google Scholar]
- 31.Wang L, Folsom AR, Zheng ZJ, Pankow JS, Eckfeldt JH, Investigators AS, ARIC Study Investigators Plasma fatty acid composition and incidence of diabetes in middle-aged adults: the Atherosclerosis Risk in Communities (ARIC) Study. Am J Clin Nutr 2003;78:91–98 [DOI] [PubMed] [Google Scholar]
- 32.Selvin E, Steffes MW, Zhu H, et al. Glycated hemoglobin, diabetes, and cardiovascular risk in nondiabetic adults. N Engl J Med 2010;362:800–811 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rosamond WD, Folsom AR, Chambless LE, et al. Stroke incidence and survival among middle-aged adults: 9-year follow-up of the Atherosclerosis Risk in Communities (ARIC) cohort. Stroke 1999;30:736–743 [DOI] [PubMed] [Google Scholar]
- 34.White AD, Folsom AR, Chambless LE, et al. Community surveillance of coronary heart disease in the Atherosclerosis Risk in Communities (ARIC) Study: methods and initial two years’ experience. J Clin Epidemiol 1996;49:223–233 [DOI] [PubMed] [Google Scholar]
- 35.Harrell FE. Regression Modeling Strategies With Applications to Linear Models, Logistic Regression, and Survival Analysis. New York, Springer, 2001, p. 18–27 [Google Scholar]
- 36.Goldin A, Beckman JA, Schmidt AM, Creager MA. Advanced glycation end products: sparking the development of diabetic vascular injury. Circulation 2006;114:597–605 [DOI] [PubMed] [Google Scholar]
- 37.Semba RD, Najjar SS, Sun K, Lakatta EG, Ferrucci L. Serum carboxymethyl-lysine, an advanced glycation end product, is associated with increased aortic pulse wave velocity in adults. Am J Hypertens 2009;22:74–79 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kilhovd BK, Juutilainen A, Lehto S, et al. Increased serum levels of advanced glycation endproducts predict total, cardiovascular and coronary mortality in women with type 2 diabetes: a population-based 18 year follow-up study. Diabetologia 2007;50:1409–1417 [DOI] [PubMed] [Google Scholar]
- 39.Kilhovd BK, Juutilainen A, Lehto S, et al. High serum levels of advanced glycation end products predict increased coronary heart disease mortality in nondiabetic women but not in nondiabetic men: a population-based 18-year follow-up study. Arterioscler Thromb Vasc Biol 2005;25:815–820 [DOI] [PubMed] [Google Scholar]
- 40.Nin JW, Jorsal A, Ferreira I, et al. Higher plasma levels of advanced glycation end products are associated with incident cardiovascular disease and all-cause mortality in type 1 diabetes: a 12-year follow-up study. Diabetes Care 2011;34:442–447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Monnier VM, Mustata GT, Biemel KL, et al. Cross-linking of the extracellular matrix by the maillard reaction in aging and diabetes: an update on “a puzzle nearing resolution”. Ann N Y Acad Sci 2005;1043:533–544 [DOI] [PubMed] [Google Scholar]
- 42.Wautier JL, Zoukourian C, Chappey O, et al. Receptor-mediated endothelial cell dysfunction in diabetic vasculopathy. Soluble receptor for advanced glycation end products blocks hyperpermeability in diabetic rats. J Clin Invest 1996;97:238–243 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Park L, Raman KG, Lee KJ, et al. Suppression of accelerated diabetic atherosclerosis by the soluble receptor for advanced glycation endproducts. Nat Med 1998;4:1025–1031 [DOI] [PubMed] [Google Scholar]
- 44.Zhang L, Bukulin M, Kojro E, et al. Receptor for advanced glycation end products is subjected to protein ectodomain shedding by metalloproteinases. J Biol Chem 2008;283:35507–35516 [DOI] [PubMed] [Google Scholar]
- 45.Chen YS, Yan W, Geczy CL, Brown MA, Thomas R. Serum levels of soluble receptor for advanced glycation end products and of S100 proteins are associated with inflammatory, autoantibody, and classical risk markers of joint and vascular damage in rheumatoid arthritis. Arthritis Res Ther 2009;11:R39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.McNair ED, Wells CR, Mabood Qureshi A, et al. Modulation of high sensitivity C-reactive protein by soluble receptor for advanced glycation end products. Mol Cell Biochem 2010;341:135–138 [DOI] [PubMed] [Google Scholar]
- 47.Mahajan N, Malik N, Bahl A, Dhawan V. Receptor for advanced glycation end products (RAGE) and its inflammatory ligand EN-RAGE in non-diabetic subjects with pre-mature coronary artery disease. Atherosclerosis 2009;207:597–602 [DOI] [PubMed] [Google Scholar]
- 48.Basta G, Del Turco SD, Navarra T, et al. Inverse association between circulating levels of soluble receptor for advanced glycation end-products and coronary plaque burden. J Atheroscler Thromb 2012;19:941–948 [DOI] [PubMed]
- 49.Hudson BI, Moon YP, Kalea AZ, et al. Association of serum soluble receptor for advanced glycation end-products with subclinical cerebrovascular disease: the Northern Manhattan Study (NOMAS). Atherosclerosis 2011;216:192–198 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Choi KM, Han KA, Ahn HJ, et al. Effects of exercise on sRAGE levels and cardiometabolic risk factors in patients with type 2 diabetes: a randomized controlled trial. J Clin Endocrinol Metab 2012;97:3751–3758 [DOI] [PubMed] [Google Scholar]
- 51.Forbes JM, Thorpe SR, Thallas-Bonke V, et al. Modulation of soluble receptor for advanced glycation end products by angiotensin-converting enzyme-1 inhibition in diabetic nephropathy. J Am Soc Nephrol 2005;16:2363–2372 [DOI] [PubMed] [Google Scholar]
- 52.Tan KC, Chow WS, Tso AW, et al. Thiazolidinedione increases serum soluble receptor for advanced glycation end-products in type 2 diabetes. Diabetologia 2007;50:1819–1825 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


