Abstract
Rationale
Several studies have suggested that modulation of the glutamatergic system via metabotropic glutamate receptors (mGlu) could be a new way to achieve antipsychotic-like activity. LSP1-2111, the group III mGlu receptor orthosteric agonist, with a high affinity towards mGlu4 receptors, was previously shown to exhibit antipsychotic-like action in animal models displaying positive symptoms of schizophrenia.
Objectives
Here, we decided to investigate the possible role of LSP1-2111 in models of negative (social interaction) and cognitive (NOR) symptoms of psychosis. We also investigated the involvement of 5-HT1A receptors in the LSP1-2111-induced antipsychotic effects. Apart from the above-mentioned models of negative and cognitive symptoms, MK-801 and amphetamine-induced hyperactivity tests, plus the DOI-induced head twitches in mice as models for positive symptoms of psychosis, were used in this part of the investigations.
Results
LSP1-2111 (0.5, 2, and 5 mg/ kg) dose-dependently inhibited MK-801-induced deficits in social interaction and NOR tests. The effects of the drug were antagonized by 5-HT1A antagonist, WAY100635 (0.1 mg/kg). A similar inhibition of LSP1-2111-induced effects was observed in models of positive symptoms of schizophrenia. Moreover, the concomitant administration of subeffective doses of LSP1-2111 (0.3-0.5 mg/kg) with a subeffective dose of 5-HT1A agonist, (R)-(+)-8-Hydroxy-DPAT (0.01 mg/kg), induced a clear antipsychotic-like effect in all of the procedures used.
Conclusions
Altogether, we propose that the activation of group III mGlu receptors may be a promising target for the development of novel antipsychotic drugs, towards not only positive but also negative and cognitive symptoms. The action of the compound is 5-HT1A-dependent.
Keywords: Amphetamine, Animal model, Antipsychotic, Behavior, Serotonin receptor, Glutamate, Schizophrenia, Metabotropic glutamate receptor, MK-801, Object recognition
Introduction
Psychotic disorders constitute the group of mental diseases affecting nearly 5 million of the population in Europe, which places them in the sixth place among the most frequently diagnosed psychiatric disorders (Andlin-Sobocki and Rössler 2005). However, it is regarded as the most serious psychiatric illness that, in most cases, affects young people between 18 and 35 years of age, in their most effective period of life, excluding them from a normal existence, such as being able to start a family and continue with employment (Cramer et al. 1992; Mueser and McGurk 2004).
Metabotropic glutamate receptor (mGlu) ligands have been recently considered as an alternative to the presently used neuroleptic therapy, possibly improving antipsychotic treatment. The family of mGlu receptors is divided into three classes according to the sequence homology, pharmacology, and the second messenger system they activate. Group I involves mGlu1 and mGlu5 subtypes, group II consists of mGlu2 and mGlu3 receptors, and mGlu4, mGlu6, mGlu7, and mGlu8 constitute the third group of mGlu receptors (Pin and Duvoisin 1995).
Key evidence for the involvement of mGlu receptors in psychosis originated in the clinical report by Patil et al. (2007), showing that the treatment with LY2140023 (an mGlu2/3 agonist) was safe and well-tolerated, plus effective in reducing both positive and negative symptoms when compared to the placebo (Patil et al. 2007).
However, the results of the subsequent studies appeared to be inconclusive because LY2140023 monohydrate and the active control olanzapine did not separate from the placebo in the treatment of patients with acutely exacerbated schizophrenia (Kinon et al. 2011). That is why the project was stopped (August 29, 2012; Lilly press release). However, the very recent report from Addex (November 5, 2012; Addex press release) demonstrated a successful phase 2a clinical study with ADX71149, the mGlu2 receptor PAM. These studies do not shatter the credibility of group II mGlu ligands as the new target in antipsychotic therapy due to the effect of the placebo observed in schizophrenia clinical trials representing a growing problem that interferes with the signal detection for treatments (Alphs et al. 2012). The important matter is that mGlu receptor ligands seem to possess the unique characteristic of not targeting dopamine receptors (Cartmell et al. 2000a, b; Weinberger 2007).
Recently, the third and the largest group of mGlu receptors has started to be broadly investigated in the field of neuropsychopharmacology. The first brain-penetrating agent selective towards this group, ACPT-I, was characterized in 1997 (Acher et al. 1997). The papers published showed the efficacy of the compound in several behavioral models of the positive symptoms of schizophrenia, such as MK-801 and amphetamine-induced hyperlocomotion, as well as in DOI-induced head twitches in mice (Pałucha-Poniewiera et al. 2008). Later on, a successor of ACPT-I, which displayed a higher potency at mGlu4 over mGlu7/mGlu8 receptors by 30-fold, LSP1-2111, was shown to be active in the same models (Wierońska et al. 2012b). The results indicate that group III mGlu receptors, which share the same presynaptic expression as group II mGlu receptors, meaning that the stimulation of both groups inhibits the release of glutamate (Cartmell and Schoepp 2000), may be considered as a new target for future antipsychotics. However, there is no data about the efficacy of the compounds in the models of negative and cognitive symptoms of psychosis. The mechanism of action of the drugs has also been poorly investigated so far. Therefore, in the present study we focused on the efficacy of LSP1-2111 in models of negative and cognitive disturbances. In addition, the involvement of the serotonergic system in antipsychotic activity, with a special consideration for the 5-HT1A receptors, was reported several times (see Ohno 2011 for review). Not only the antipsychotic effect of 5-HT1A agonist, 8-OH-DPAT, was reported (Bubeníková-Valesová et al. 2007), but also activity towards that subtype of the serotonergic receptor was established for some atypical antipsychotics (Meltzer et al. 2003). Therefore, in the present study we used WAY100635, a 5-HT1A antagonist (Fletcher et al. 1996), and (R)-(+)-8-Hydroxy-DPAT hydrobromide, a 5-HT1A agonist (Wood et al. 2000), which were co-administered with effective and subeffective doses of LSP1-2111, respectively, in order to determine the role of the serotonergic receptor in LSP1-2111-mediated action.
The social interaction test was performed as predictive for negative symptoms of the disease and novel object recognition (NOR) as predictive for cognitive disturbances. The standard models of positive symptoms of schizophrenia, such as the MK-801 or amphetamine-induced hyperactivity test and DOI-induced head twitches were also used in our experiments. In the interaction studies, the action of the effective dose of LSP1-2111 (5 mg/kg) was blocked by WAY100635 (0.1 mg/kg) administration while the subeffective doses of LSP1-2111 (0.3–0.5 mg/kg, depending on the experimental design) were enhanced by the subeffective dose of (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 mg/kg). Our result shows that the proposed antipsychotic action of LSP1-2111 involves the 5-HT1A receptors.
Materials and methods
Animals and housing
Male Albino Swiss mice (20–25 g) were used both for the hyperactivity tests and DOI-induced head twitches. Male Wistar rats (200–220 g) were used in the social interaction and novel object recognition tests. The animals were kept under a 12:12 light–dark cycle at a room temperature of 19–21 °C with free access to food and water. Each experimental group consisted of eight to ten animals/dose, and the animals were used only once in each test. All of the animals were experimentally naive prior to testing. For all the experiments in mice, the compounds were injected at a volume of 10 mL/kg and in rats at a volume of 1 mL/kg. The experiments were performed by an observer blind to the treatment and were conducted according to the procedures approved by the Animal Care and Use Committee at the Institute of Pharmacology, Polish Academy of Sciences in Krakow.
Drugs
LSP1-2111 Acher WO2007 052169 was synthesized in Dr. Francine Acher’s laboratory and was dissolved in water, and then the pH was adjusted with NaOH to 7.0 before the stock solution was dissolved in a saline (Acher et al. 2007). MK-801, WAY100635, (R)-(+)-8-Hydroxy-DPAT hydrobromide (Tocris), DOI and d-amphetamine sulfate (Sigma-Aldrich, St. Louis, USA) were dissolved in 0.9 % NaCl.
The psychostimulants (MK-801, amphetamine, and DOI) were administered at the same doses and schedule as in our previous studies (Pałucha-Poniewiera et al. 2008; Wierońska et al. 2011, 2012a, b). LSP1-2111 and WAY100635 were administered 45 min before the experiments. The administration schedule of the compounds was adapted from the other studies, but also was based on our long-lasting experience with the ligands used. The doses and routes of administration of LSP1-2111 used in the present study were the same as those used in our previous studies concerning the mechanisms of anxiolytic and antipsychotic action of this compound (see Wierońska et al. 2010, 2012a, b). A dose of WAY100635 was taken from the studies of Homberg et al. (2008), Iijima et al. (2007), Olivier et al. (1998, 2008). The administration schedule of (R)-(+)-8-Hydroxy-DPAT hydrobromide was adapted from the studies of Bubeníková-Valesová et al. (2007), Vinkers et al. (2009), and Yu and Lewander (1997). LSP1-2111 was administered intraperitoneally while WAY100635 and (R)-(+)-8-Hydroxy-DPAT hydrobromide were given subcutaneously (Homberg et al. 2008; Olivier et al. 1998; Yu and Lewander 1997). In the interaction studies, all animals received two injections: when one of the drugs was omitted, a vehicle was given instead. The control animals received injections of the appropriate vehicles.
MK-801-induced deficits in social interaction in rats
Social interaction tests were performed according to the method described by Satow et al. (2009), using a circle made of wood, 90 cm in diameter, divided into 10 × 10-cm squares by faint yellow lines. Each social interaction test between two rats was carried out during the light phase of the light/dark cycle. The rats were selected from separate housing cages to make a pair for the study. The body weights of the paired rats were matched within 20 g of variance. The study was conducted 3.5 h after the subcutaneous administration of MK-801 at 0.1 mg/kg, s.c. Each pair of rats was diagonally placed in opposite corners of the box so that they faced away from each other. Saline was administered as the vehicle. LSP1-2111 (0.5, 2, and 5 mg/kg) and WAY100635 (0.1 mg/kg) were administered 45 min before the social interaction test (together with LSP1-2111 at a dose of 5 mg/kg), and (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 mg/kg) was administered 15 min before the test, 30 min after LSP1-2111 (0.5 mg/kg). The behavior of the animals was monitored and recorded on a video recorder located outside the box over a 10-min period. The test box was wiped clean between each trial. The social interaction between the two rats was determined as the total time spent participating in social behavior such as sniffing, genital investigation, chasing, and fighting each other. The number of episodes was counted as a separate paradigm. The treatment groups included eight to ten animals.
Novel object recognition
The method was adapted from Horiguchi et al. (2011a, b) and Dere et al. (2007). The animals were trained and tested in a black wooden circular open field (100 cm in diameter, 35-cm high) with the floor divided into 20-cm square sections. The open field was in a dark room illuminated only by a 25-W bulb. On the first day (adaptation), the animals were allowed to explore the open field for 10 min. On the next day (training, T1), the animals were administered with the tested drugs, placed in the apparatus and allowed to explore two identical objects (cylinder-shaped objects with walls painted white, 7 cm in diameter, 11-cm high) for the time required to complete 15 s of exploration of either object. For the retention trial (T2) conducted 1 h later, one of the objects presented in T1 was replaced with a novel object (a prism-shaped object with walls painted black, 5-cm wide, 14-cm high). The rats were returned to the open field for 5 min, and the duration of exploration (i.e., sitting in close proximity to the objects, sniffing, or touching them) of each object was measured separately by a trained observer. All drugs were administered before the training (T1) session. MK-801 (0.1 mg/kg, s.c.) was given 30 min before the session, LSP1-2111 (0.5, 2, and 5 mg/kg, i.p.) was injected 45 min before the MK-801 injections, WAY 100635 (0.1 mg/kg, s.c.) was co-administered with LSP1-2111 (5 mg/kg), and (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 mg/kg, s.c.) was injected 15 min before the test, 30 min after LSP1-2111 (2 mg/kg). All injections were given at a volume of 1 mL/kg of the body weight. The treatment groups included eight animals.
Locomotor activity of habituated mice
The locomotor activity was recorded individually for each animal in OPTO-M3 locomotor activity cages (Columbus Instrument) linked online to a compatible PC. Each cage (13 × 23 × 15 cm) was surrounded with an array of photocell beams. Interruptions of these photobeams resulted in horizontal activity then defined as ambulation scores.
The locomotor activity of habituated mice was performed as a control experiment for the studies on hyperactivity induced by MK-801 or amphetamine. The measurements were performed as follows: the mice were placed into actometers for an acclimatization period of 30 min before they were then administered with an investigated compound and placed again into the same cages. After the correct time that was dependent on the action of compounds (see above), the ambulation scores were measured for 60 min. The treatment groups included eight to ten animals.
MK-801- or amphetamine-induced hyperactivity
The locomotor activity was recorded for each animal in locomotor activity cages (according to Rorick-Kehn et al. 2007; Monn et al. 1999; with the small modifications used in our previous studies: Pałucha-Poniewiera et al. 2008; Wierońska et al. 2011, 2012a, b). The mice were placed individually into actometers for an acclimatization period of 30 min before they were administered with i.p. injections of the LSP1-2111 (subeffective dose, 0.3 or effective dose, 5 mg/kg), then placed again in the same cages. WAY100635 (0.1 and 3 mg/kg) was given 45 min before the test (it was co-administered with LSP1-2111 at a dose of 5 mg/kg), and (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 and 0.1 mg/kg) was given 15 min before the test (30 min after the administration of LSP1-2111 at a dose of 0.3 mg/kg). The mice which were not given WAY100635 or (R)-(+)-8-Hydroxy-DPAT hydrobromide were administered with the saline. Then the mice were intraperitoneally injected with MK-801 at a dose of 0.3 mg/kg or amphetamine at a dose of 3 mg/kg and once again returned to the same cage. From then on, the ambulation scores were counted for 60 min. All of the groups were compared with the MK-801 or amphetamine control group. The experiment also included a control group that was not treated with MK-801 or amphetamine. The treatment groups included eight to ten animals.
Head twitch test
The experiments were performed according to our previous studies (Pałucha-Poniewiera et al. 2008; Wierońska et al. 2011, 2012b). Briefly, in order to habituate mice to the experimental environment, each animal was transferred to a 12-cm (diameter) × 20-cm (height) glass cage, lined with sawdust, 30 min before the treatment. The head twitches of the mice were induced by DOI (2.5 mg/kg, i.p.). Immediately after the treatment, the number of head twitches was counted during a 20-min session. LSP1-2111 was administered in effective and subeffective doses (5 or 0.3 mg/kg, i.p., respectively), WAY100635 was administered at a dose 0.1 mg/kg, s.c. and (R)-(+)-8-Hydroxy-DPAT hydrobromide at a dose of 0.01 mg/kg s.c. The compounds were given at the time schedule described above. The treatment groups included eight to ten animals.
Statistical analysis
The data are presented as the means ± S.E.M. Statistical analysis of the data was performed using the Statistica 10 package (StatSoft, Inc., OK, USA). A one-way ANOVA followed by the Tukey’s post hoc comparison was used in the dose-dependence studies while a two-way ANOVA, followed by Newman–Keuls post hoc comparison test, was used in the interaction studies. A P value of at least p < 0.05 was considered as statistically significant.
Results
The effect of LSP1-2111 on the MK-801-induced deficits in the social interaction test
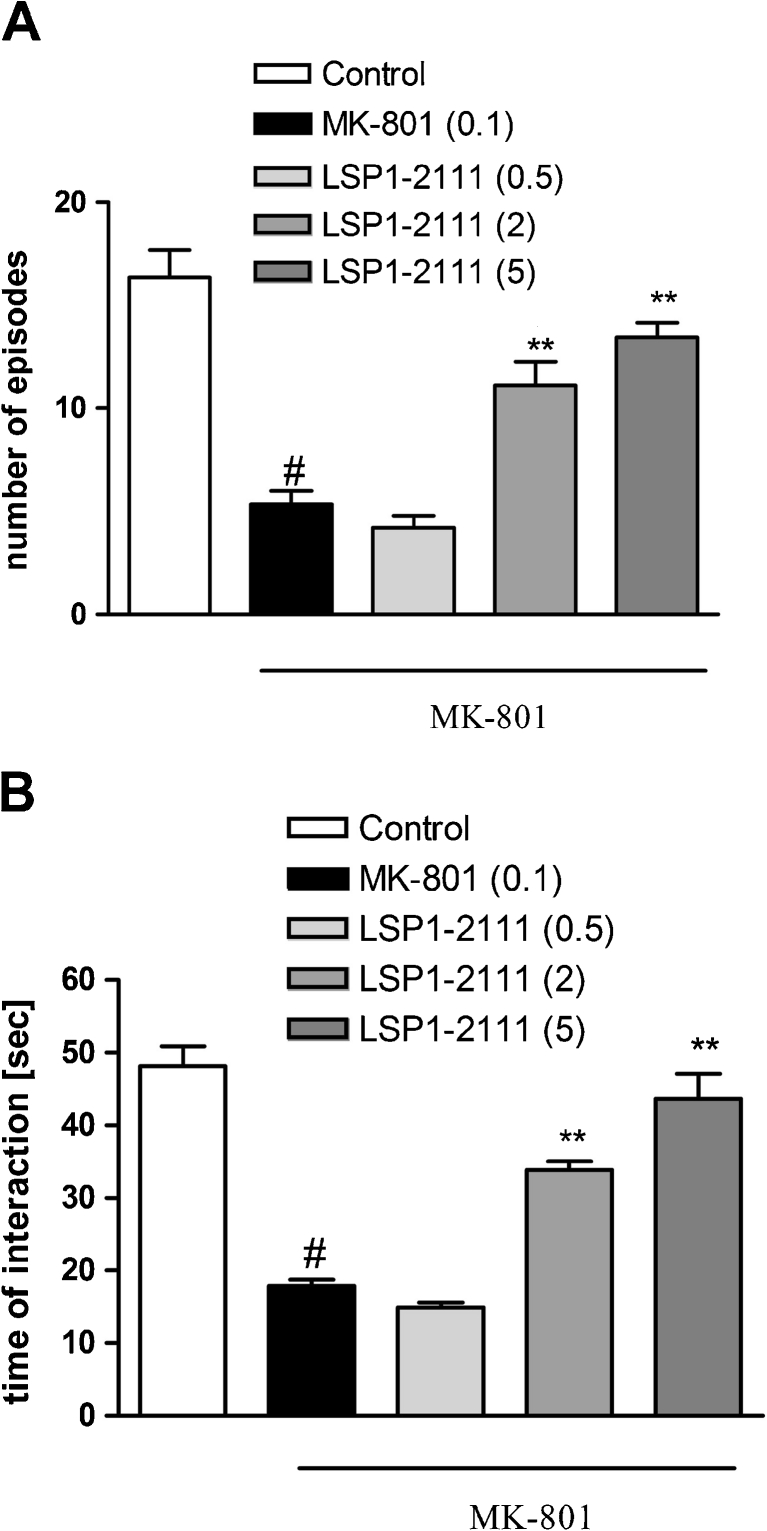
Subcutaneous administration of MK-801 at 0.1 mg/kg significantly decreased the total duration of the social interaction between two naive rats and the total number of social episodes when compared to the vehicle-treated group. LSP1-2111 administered 45 min before the test significantly improved social withdrawal induced by MK-801 (Fig. 1a, b), without affecting the behavior of the animals when administered on its own (Fig. 2c, d).
Fig. 1.
Effects of LSP1-2111 on MK-801-induced deficits in social interaction. The number of episodes of social contacts (a) and time of social interaction (b) were measured. Data are presented as means ± SEM. Doses in milligrams are indicated in parentheses. # P < 0.001 versus controls, **P < 0.001 versus MK-801-treated group
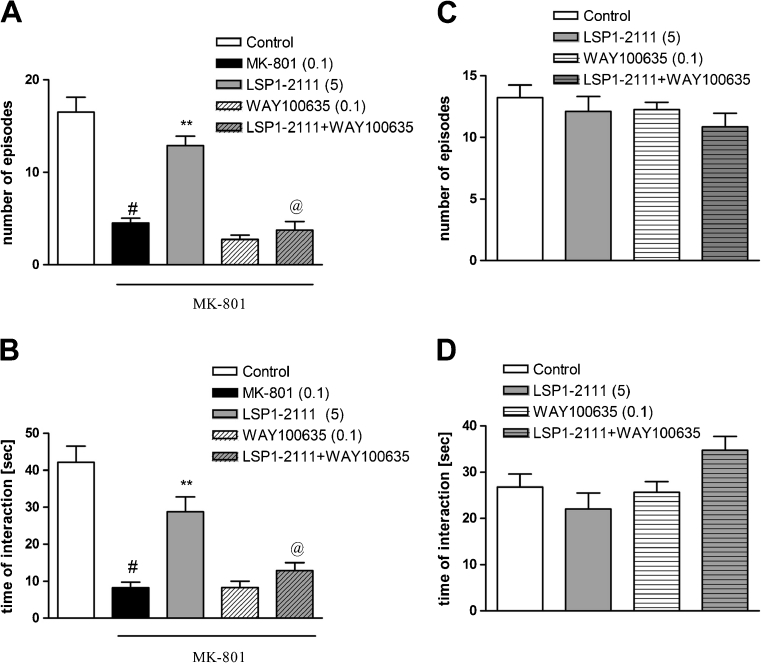
Fig. 2.
Effects on MK-801-induced deficits in social interaction. The number of episodes of social contacts (a, c) and time of social interaction (b, d) were measured. a, b Effects of combined administration of LSP1-2111 and WAY100635 c, d control experiments with the same group treatment. Data are presented as means ± SEM. Doses in milligrams are indicated in parentheses. # P < 0.001 versus controls, **P < 0.001 versus MK-801-treated group and at least @ P < 0.003 versus LSP1-2111-treated group
One-way ANOVA followed by Tukey’s post hoc comparison revealed that LSP1-2111 at the doses of 2 and 5 mg/kg reversed MK-801-induced deficits in the social interaction, increasing both the number of episodes [F (3.32) = 30.32; P < 0.001] and total duration of interactions [F (3.32) = 49; P < 0.001]. Lowest dose (0.5 mg/kg) was ineffective (Fig. 1a, b).
The effect of the combined administration of WAY100635 and LSP1-2111 in the social interaction test in rats
WAY100635 was given at a dose of 0.1 mg/kg, s.c., 45 min before the test, and LSP1-2111 was given at a dose of 5 mg/kg, i.p. also 45 min before the test. WAY100635, which itself was inactive, when given with LSP1-2111, antagonized LSP1-2111-induced antipsychotic effect in the social interaction test (Fig. 2a, b). Two-way ANOVA main effects analysis of the number of episodes revealed the significant effect of LSP1-2111 [F (1.33) = 16.83; P < 0.0002], a significant effect of WAY100635 [F (1.33) = 38.5; P < 0.001] and significant LSP1-2111xWAY100635 interaction [F (1.33) = 36.66, P < 0.001]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111 when compared to the MK-801-treated animals, P < 0.0001, and also significant LSP1-2111xWAY100635 interaction, P < 0.0001, when compared to LSP1-2111-treated group. Two-way ANOVA analysis of the time of interaction revealed significant effect of LSP1-2111 [F (1.33) = 6.42; P < 0.01], significant effect of WAY100635 [F (1.33) = 8.7; P < 0.005] and significant effect of LSP1-2111xWAY100635 interaction [F (1.33) = 21.3; P < 0.0001]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111 when compared to the MK-801-treated animals, P < 0.0001, and also significant LSP1-2111xWAY100635 interaction, P < 0.0005, when compared to LSP1-2111-treated rats (Fig. 2a, b).
The control experiment with the groups of LSP1-2111, WAY100635, and LSP1-2111+WAY100635 revealed that LSP1-2111, WAY100635, or LSP1-2111+WAY100635 interaction had not any influence on the number of episodes and time of interaction [F (1.34) = 0.78] (Fig. 2c, d).
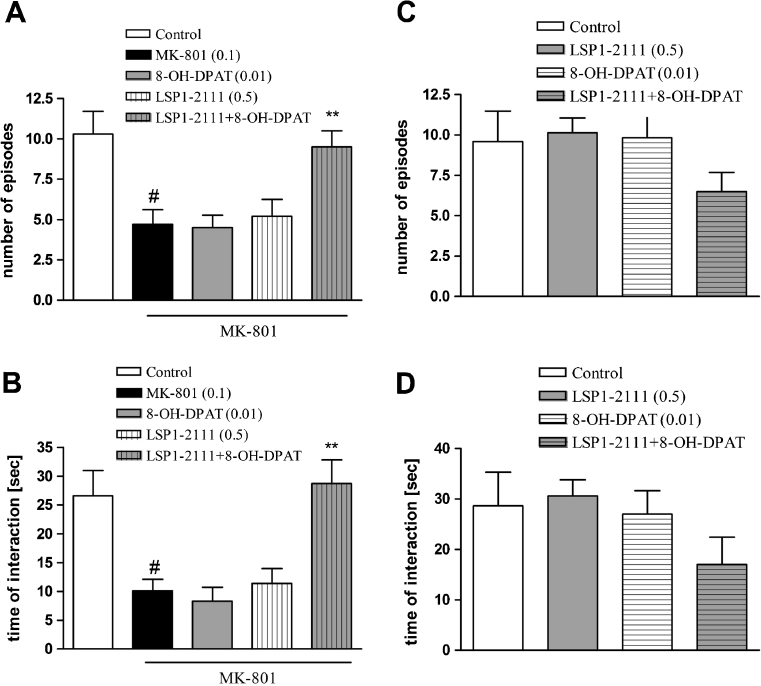
The effect of combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and a subeffective dose of LSP1-2111 in the social interaction test in rats
(R)-(+)-8-Hydroxy-DPAT hydrobromide was given at a dose of 0.01 mg/kg, s.c., 15 min before the test, and LSP1-2111 was given at a dose of 0.5 mg/kg, i.p. 45 min before the test. Concomitant administration of the subeffective doses of 5-HT1A and mGlu4 activators induced clear antipsychotic effect in the social interaction test (Fig. 3a, b). Two-way ANOVA main effects analysis of the number of episodes revealed the significant effect of LSP1-2111 [F (1.36) = 8.5; P < 0.005], a significant effect of (R)-(+)-8-Hydroxy-DPAT hydrobromide [F (1.36) = 4.7; P < 0.03], and significant LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction [F (1.36) = 5.7; P < 0.02]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide only, P < 0.002, when compared to all other groups. Two-way ANOVA analysis of the time of interaction revealed a significant effect of LSP1-2111 [F (1.36) = 5.5; P < 0.01], significant effect of (R)-(+)-8-Hydroxy-DPAT hydrobromide [F (1.36) = 11.05; P < 0.001] and significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction [F (1.36) = 12.2; P < 0.001]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction only, P < 0.001, when compared to all the other groups (Fig. 3a, b).
Fig. 3.
Effects on MK-801-induced deficits in social interaction. The number of episodes of social contacts (a, c) and time of social interaction (b, d) were measured. a, b Effects of combined administration of LSP1-2111 and (R)-(+)-8-Hydroxy-DPAT hydrobromide c, d control experiments with the same group treatment. Data are presented as means ± SEM. Doses in milligrams are indicated in parentheses. # P < 0.001 versus controls, at least **P < 0.002 versus MK-801-treated group
The control experiment with the groups of LSP1-2111, (R)-(+)-8-Hydroxy-DPAT hydrobromide, and LSP1-2111+(R)-(+)-8-Hydroxy-DPAT hydrobromide revealed that neither LSP1-2111, (R)-(+)-8-Hydroxy-DPAT hydrobromide, nor LSP1-2111+(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction had any influence on the behavior of animals [F (1.36) = 0.86] (Fig. 3c, d).
The effect of LSP1-2111 on the MK-801-induced deficits in the novel object recognition test
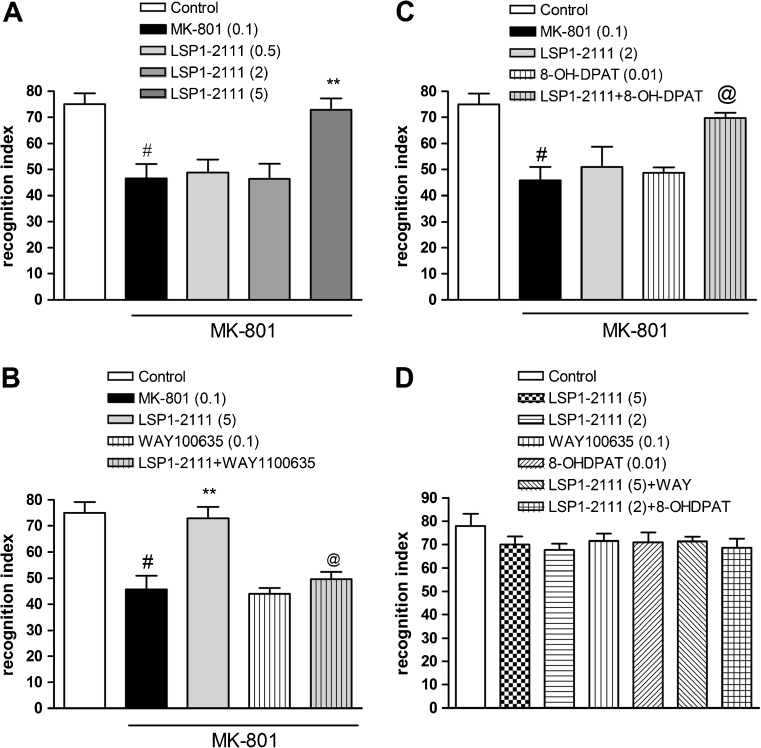
Subcutaneous administration of MK-801 at 0.1 mg/kg significantly decreased the recognition index when compared with the vehicle-treated group. LSP1-2111 given 45 min before the test significantly improved deficits in the novel object recognition induced by MK-801. One-way ANOVA followed by Tukey’s post hoc comparison revealed that LSP1-2111 at a dose of 5 mg/kg reversed MK-801-induced deficit [F (3.28) = 5.9; P < 0.01] (Fig. 4a), without affecting the behavior of the animals when administered alone (Fig. 4d). Lower doses (0.5 and 2 mg/kg) were ineffective (Fig. 4a).
Fig. 4.
Effects on MK-801-induced deficits in NOR. The dose-dependent effect of LSP1-2111 (a), the combined administration of LSP1-2111 (5 mg/kg) with WAY100635 (0.1 mg/kg) (b), and the coadministration of LSP1-2111 with (R)-(+)-8-Hydroxy-DPAT hydrobromide (c) are presented. d Control experiments with the same group treatments. Data are presented as means ± SEM. Doses in milligrams are indicated in parentheses. # P < 0.001 versus controls, **P < 0.005 versus MK-801-treated group and @ P < 0.003
The effect of combined administration of WAY100635 and LSP1-2111 in the novel object recognition test in rats
WAY100635 was given at a dose of 0.1 mg/kg, s.c., 45 min before the test, and LSP1-2111 was given at a dose of 5 mg/kg, i.p. also 45 min before the test. WAY100635, which itself was inactive, antagonized LSP1-2111-induced antipsychotic effect in the NOR test (Fig. 4b). Two-way ANOVA main effects revealed statistical effect of LSP1-2111 [F (1.28) = 17.7; P < 0.0002], statistical effect of WAY100635 [F (1.28) = 10.32; P < 0.003], and statistical effect of LSP1-2111xWAY100635 interaction [F (1.28) = 7.7; P < 0.01]. Post hoc Newman–Keuls comparison revealed that WAY100635 antagonized LSP1-2111-induced effect, decreasing recognition index in the statistically significant way, P < 0.0003, compared to LSP1-2111-treated animals (Fig. 4b). The control experiment with the groups of LSP1-2111, WAY100635, and LSP1-2111 + WAY100635 revealed that neither of the combination interaction had any influence on the recognition index [F (1.28) = 0.92] (Fig. 4d).
The effect of combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and a subeffective dose of LSP1-2111 in the novel object recognition test in rats
(R)-(+)-8-Hydroxy-DPAT hydrobromide was given at a dose of 0.01 mg/kg, s.c., 15 min before the test, and LSP1-2111 was given at a dose of 2 mg/kg, i.p. 45 min before the test. Concomitant administration of the subeffective doses of 5-HT1A and mGlu4 activators induced clear antipsychotic effect in the NOR test (Fig. 4c). The two-way ANOVA main effects revealed the statistical effect of LSP1-2111 [F (1.28) = 5.6; P < 0.02], statistical effect of (R)-(+)-8-Hydroxy-DPAT hydrobromide [F (1.28) = 11.04; P < 0.01], and statistical effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction [F (1.28) = 6.8; P < 0.01]. Post hoc Newman–Keuls comparison revealed the statistical effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction only, P < 0.001 (Fig. 4c). The control experiment with the groups of LSP1-2111, (R)-(+)-8-Hydroxy-DPAT hydrobromide, and LSP1-2111 + (R)-(+)-8-Hydroxy-DPAT hydrobromide revealed that neither of the combination interactions had any influence on the recognition index [F (1.28) = 0.87] (Fig. 4d).
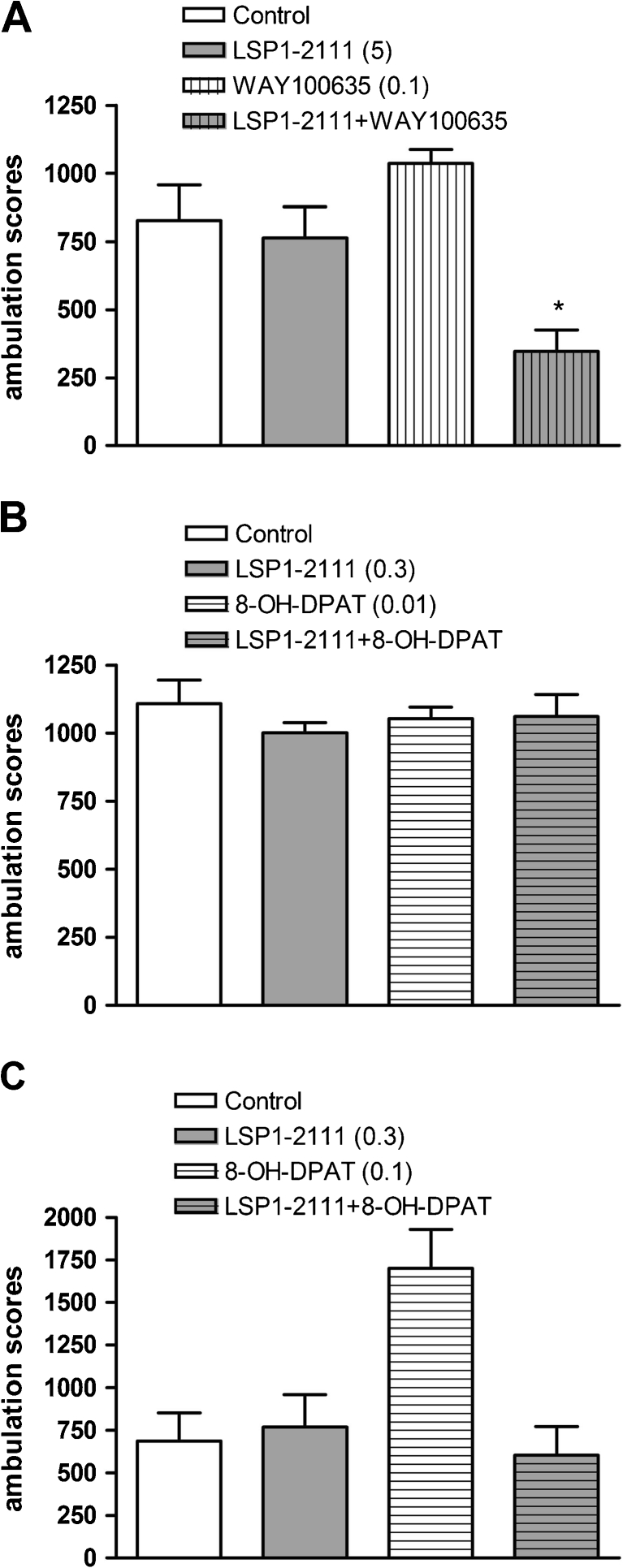
The effects of LSP1-2111, WAY100635, and (R)-(+)-8-Hydroxy-DPAT hydrobromide on locomotor activity in mice habituated to activity meters
Two-way ANOVA revealed that LSP1-2111 (5 and 0.3 mg/kg, 45 min before the test), (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 mg/kg, 15 min before the test), and WAY100635 (0.1 mg/kg, 45 min before the test) did not change the locomotor activity of mice adapted to activity meters for 30 min (Fig. 5a). No statistically significant effect from the co-administration of LSP1-2111 (0.3 mg/kg) with (R)-(+)-8-Hydroxy-DPAT hydrobromide (0.01 mg/kg) was observed (Fig. 5b). However, a significant reduction of locomotor activity after the combined co-administration of LSP1-2111 (5 mg/kg) and WAY100635 (0.1 mg/kg) 45 min before the measurements [F (1.31) = 9.94; P < 0.003] (Fig. 5a, b) was noticed, as well as the increase of the ambulation scores after the administration of 8-OH-DPAT at the higher dose of 0.1 mg/kg (Fig. 5c).
Fig. 5.
Locomotor activity of mice habituated to locomotor activity cages. The bars represent means ± SEM. Ambulation scores were measured for 60 min after an appropriate time of drug administration. Doses in milligrams are indicated in parentheses. *P < 0.003 versus control group
The effect of MK-801 and amphetamine on locomotor activity in mice
MK-801, at a dose of 0.3 mg/kg, produced a robust increase in the ambulation scores (up to 574–889 % of the control, P < 0.0009) within 60 min of the experimental session than the saline-treated animals (Figs. 6a, b and 7a, b). Similar results were observed after the administration of amphetamine at a dose of 3 mg/kg, i.p. The increase in ambulation scores was observed as going up to 536–688 %, P < 0.0005, compared to the vehicle-treated animals (Fig. 8a, b).
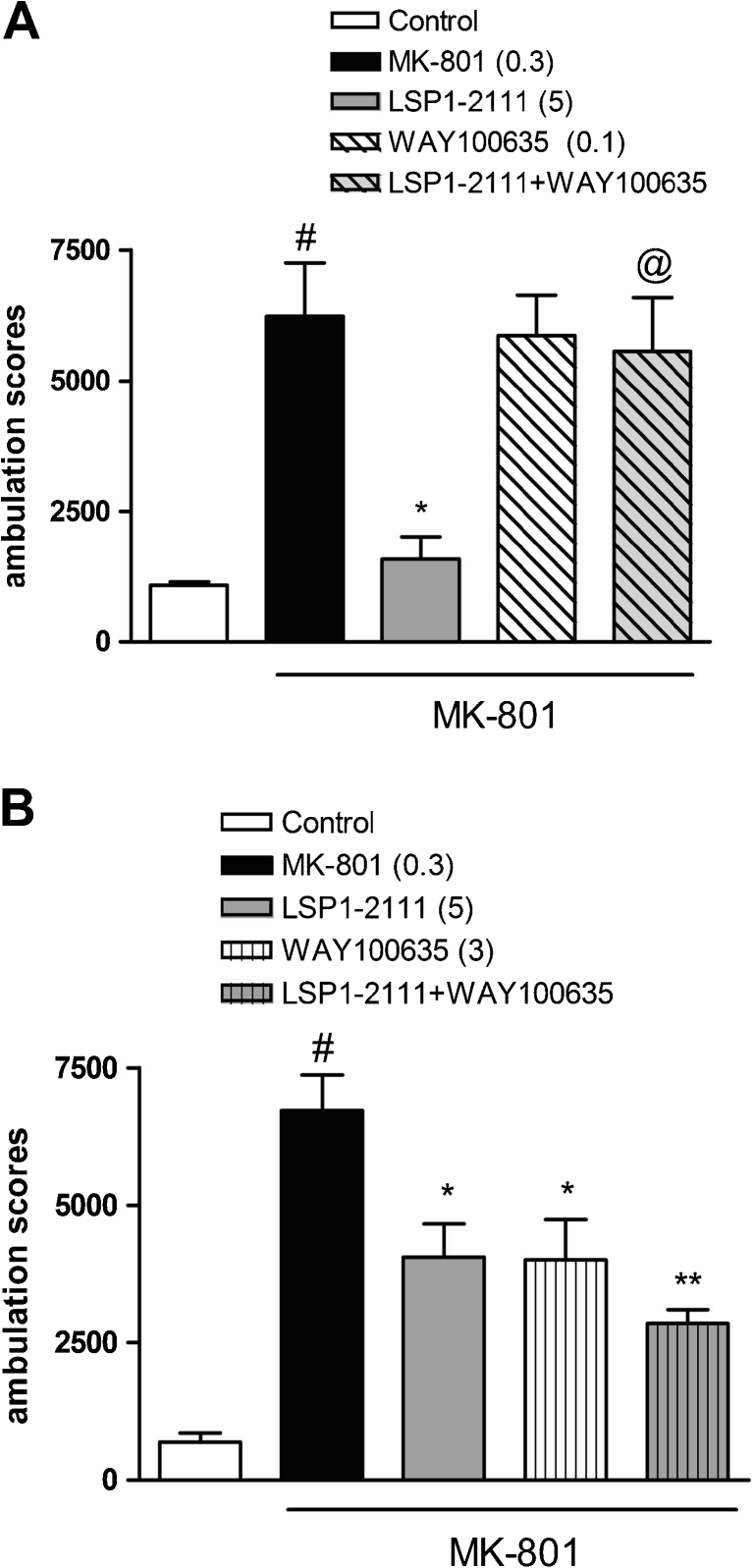
Fig. 6.
The effect of the combined administration of WAY100635 and LSP1-2111 on MK-801-induced hyperactivity in mice. Concomitant administration of LSP1-2111 with WAY100635, 5HT1A receptor antagonist at a low dose at 0.1 mg/kg (and at the higher dose 3 mg/kg (b). LSP1-2111 and WAY100635 were given 45 min before MK-801. Doses in milligrams are indicated in parentheses. Locomotor activity was measured immediately after psychostimulant agent administration. Bars represent means ± SEM. # P < 0.001 versus the control group,**P < 0.01 versus MK-801 and at least @ P < 0.02 versus the MK-801 + LSP1-2111-treated group
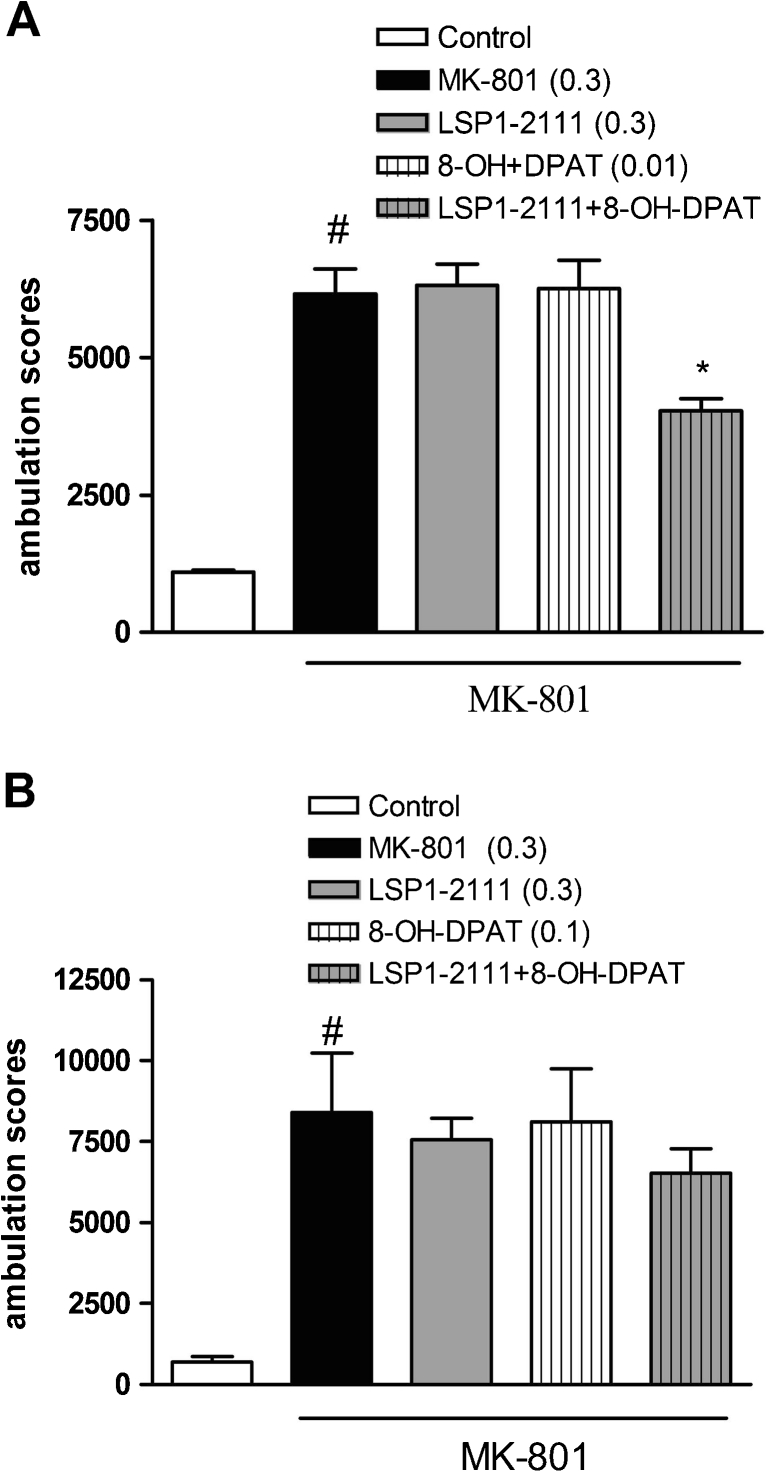
Fig. 7.
The effect of the combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and LSP1-2111 on MK-801-induced hyperactivity in mice. Concomitant administration of LSP1-2111 with (R)-(+)-8-Hydroxy-DPAT, agonist at a low dose of 0.01 mg/kg (a) and at the higher dose of 0.1 mg/kg (b). LSP1-2111 was given 45 min, and (R)-(+)-8-Hydroxy-DPAT hydrobromide was given 15 min before MK-801 administration. Doses in milligrams are indicated in parentheses. Locomotor activity was measured immediately after psychostimulant agent administration. The bars represent means ± SEM. # P < 0.001 versus the control group, *P < 0.01 versus MK-801-treated group
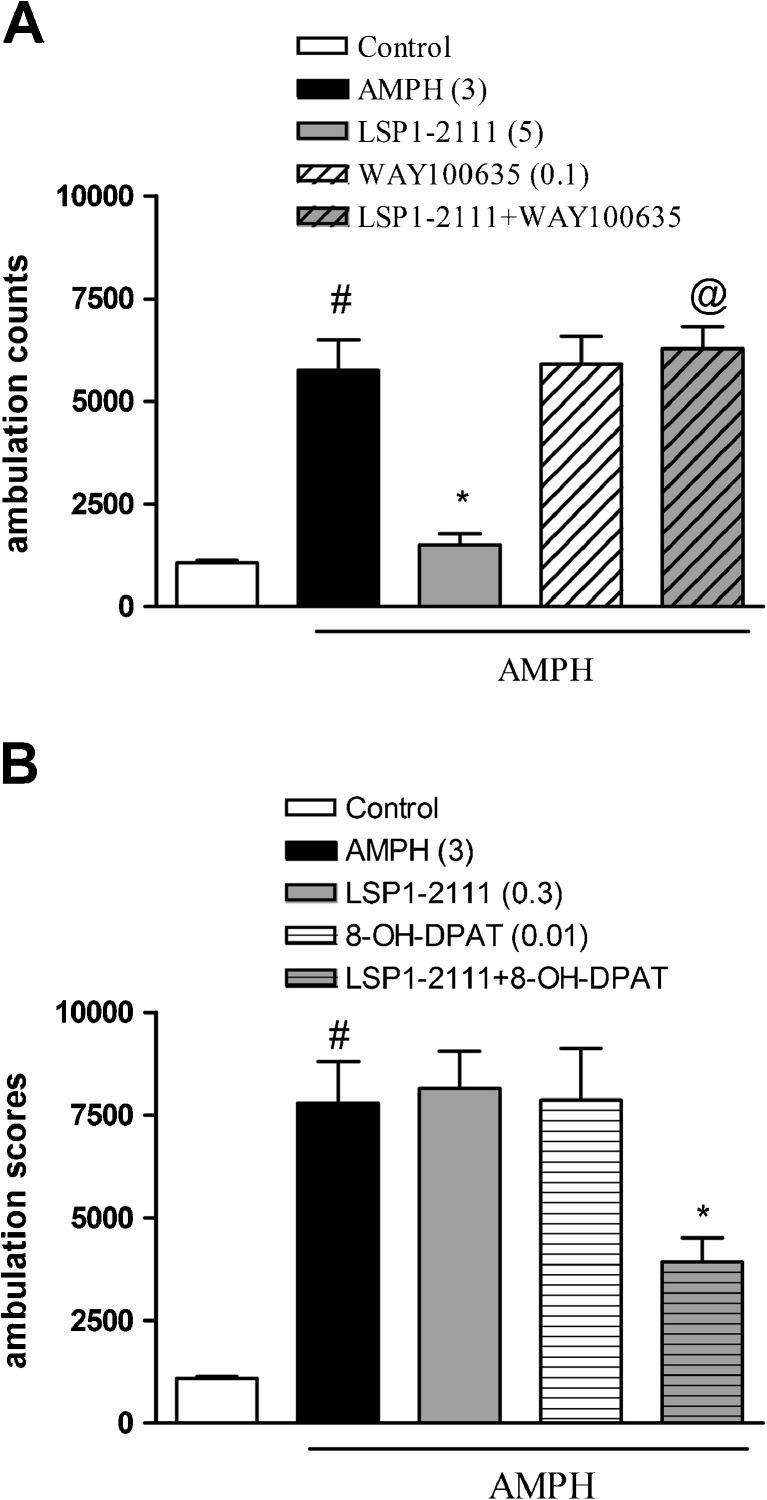
Fig. 8.
The effect of the combined administration of WAY100635 and (R)-(+)-8-Hydroxy-DPAT hydrobromide on LSP1-2111 action in amphetamine-induced hyperactivity in mice. Locomotor activity of mice habituated to activity cages. Concomitant administration of LSP1-2111 with WAY100635, 5HT1A receptor antagonist (at a dose of 0.1 mg/kg) (a) and 8-OH-DPAT, agonist (at a dose of 0.01 mg/kg) (b). LSP1-2111 and WAY100635 were given 45 min, and (R)-(+)-8-Hydroxy-DPAT hydrobromide was given 15 min before amphetamine administration. Doses in milligrams are indicated in parentheses. Locomotor activity was measured immediately after psychostimulant agent administration. The bars represent means ± SEM. # P < 0.001 versus the control group; **P < 0.01 versus amphetamine group; and @ P < 0.009 versus the AMPH + LSP1-2111-treated group
The effect of the combined administration of WAY100635 and LSP1-2111 on MK-801-induced hyperactivity in mice
WAY100635, when given at the ineffective dose of 0.1 mg/kg with LSP1-2111 (5 mg/kg), antagonized LSP1-2111-induced an antipsychotic effect in the hyperactivity test (Fig. 6a). The two-way ANOVA main effects analysis revealed the significant effect of LSP1-2111 [F (1.29) = 8.65; P < 0.006], a significant effect of WAY100635 [F (1.29) = 4.58; P < 0.04], and significant LSP1-2111xWAY100635 interaction [F (1.29) = 6.66; P < 0.015]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111 when compared to the MK-801-treated animals, P < 0.002, and also significant LSP1-2111xWAY100635 interaction, P < 0.002, when compared to LSP1-2111 (Fig. 6a). Such an antagonistic effect was not observed when WAY100635 was administered at a higher dose of 3 mg/kg, which produced a statistically significant antipsychotic effect (F (1.40) = 18.31; P < 0.01) (Fig. 6b).
The effect of the combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and LSP1-2111 on MK-801- induced hyperactivity in mice
Concomitant administration of the subeffective doses of 5-HT1A and mGlu4 activators induced clear antipsychotic effect in the hyperactivity test (Fig. 7a). The two-way ANOVA main effects analysis revealed the significant effect of the 8-OH-DPATxLSP1-2111 interaction [F(1.30) = 4.43, P < 0.04]. Post-hoc Newman-Keuls analysis revealed the significant effect of 8-OH-DPATxLSP1-2111 when compared to the MK-801-treated animals, P < 0.01 (Fig. 6b). The attenuation of the MK-801-induced hyperlocomotion reached 40 %. Such an addictive effect was not observed when (R)-(+)-8-Hydroxy-DPAT hydrobromide was given at a higher dose of 0.1 mg/kg (Fig. 7b).
The effect of the combined administration of WAY100635 and LSP1-2111 on amphetamine-induced hyperactivity in mice
WAY100635, when given with LSP1-2111, antagonized LSP1-2111-induced antipsychotic effect in the amphetamine-induced hyperactivity test (Fig. 8a). The two-way ANOVA main effects analysis revealed the significant effect of LSP1-2111 [F (1.31) = 10.23; P < 0.003], the significant effect of WAY100635 [F (1.31) = 16.52; P < 0.0003] and significant LSP1-2111xWAY100635 interaction [F (1.31) = 14.43, P < 0.0006]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111 when compared to the AMPH treated animals, P < 0.0001, and significant LSP1-2111xWAY100635 interaction, P < 0.0001, when compared to LSP1-2111 (Fig. 8a).
The effect of the combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and a subeffective dose of LSP1-2111 on amphetamine-induced hyperactivity in mice
Concomitant administration of the subeffective doses of 5-HT1A agonist and LSP1-2111 induced clear antipsychotic effect in the amphetamine-induced hyperactivity test (Fig. 8b). The two-way ANOVA main effects analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction [F (1.29) = 4.78, P < 0.03]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide when compared to AMPH treated animals, P < 0.009 (Fig. 8b). The attenuation of the AMPH-induced hyperlocomotion reached 50 %.
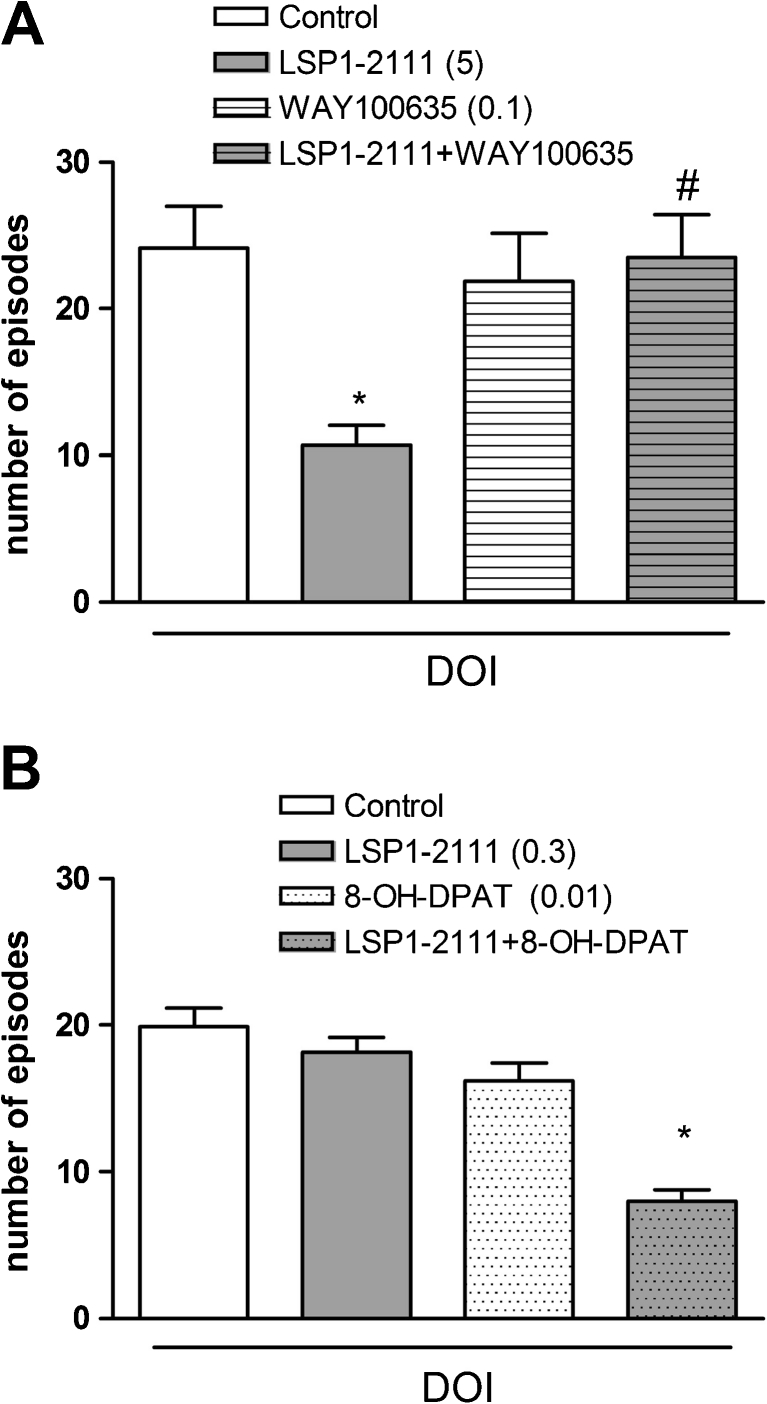
The effect of the combined administration of WAY100635 and LSP1-2111 on DOI-induced head twitches in mice
LSP1-2111, administered i.p. at a dose of 5 mg/kg, produced a significant decrease in the number of DOI-induced head twitches in mice [F (1.28) = 4.38, P < 0.04]. WAY100635, when given with LSP1-2111, antagonized LSP1-2111-induced antipsychotic effect in the DOI-induced head twitches test. The effect of LSP1-2111xWAY100635 interaction was significant as well [F (1.28) = 7.11, P < 0.012]. Post hoc Newman–Keuls analysis revealed a significant LSP1-2111 effect, P < 0.01 when compared to the controls, and significant LSP1-2111xWAY100635 interaction when compared to LSP1-2111-treated group, P < 0.009 (Fig. 9a).
Fig. 9.
DOI-induced head twitches in mice. LSP1-2111 was given 45 min before the test. WAY100635 (a) was also injected 45 min while (R)-(+)-8-Hydroxy-DPAT hydrobromide (b) 15 min before the test. The number of head twitches was measured immediately after DOI administration. The bars represent means±SEM. *P < 0.01 versus the DOI control group, and at least # P < 0.009 versus the LSP1-2111 group
The effect of the combined administration of (R)-(+)-8-Hydroxy-DPAT hydrobromide and a subeffective dose of LSP1-2111 on DOI-induced head twitches in mice
Concomitant administration of the subeffective doses of 5-HT1A and LSP1-2111 induced clear antipsychotic effect in the DOI-induced head twitch test. The two-way ANOVA main effects analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide interaction [F (1.30) = 8.6, P < 0.006]. Post hoc Newman–Keuls analysis revealed the significant effect of LSP1-2111x(R)-(+)-8-Hydroxy-DPAT hydrobromide when compared to the DOI-treated animals, P < 0.0001 (Fig. 9b). The attenuation of the DOI-induced head twitches reached 40 %.
Discussion
In the present manuscript, we report two major findings. Firstly, LSP1-2111, the group III mGlu receptors’ orthosteric agonist, shows activity in reversing the behavioral deficits predictive of negative and cognitive symptoms of psychosis in the animal models. Secondly, serotonin acting via 5-HT1A receptors (of possibly presynaptic placement) is involved in the action of the compound in the models of cognitive, negative, and positive symptoms of the disease.
LSP1-2111 is a substance with a 2-year history that binds group III mGlu receptors with preferential activity towards the mGlu4 subtype (EC50 = 2.2 ± 0.27 for mGlu4 vs. 52.87 ± 20.66 for mGlu7 and 65.97 ± 11.81 for mGlu8 receptors) (Beurrier et al. 2009). Concomitantly, it was shown that LSP1-2111 exhibited no off-target interaction towards other families of ionotropic and G-protein coupled receptors, and transporters (Flor and Acher 2012).
The antipsychotic-like action of LSP1-2111 was demonstrated by us recently in models predictive of positive symptoms of schizophrenia, such as MK-801 or amphetamine hyperlocomotions, and DOI-induced head twitches in mice (Wierońska et al. 2011, 2012b). In the present experiments we show, that the drug is also active in an animal model of negative symptoms. We used the social interaction test, described earlier by Satow et al. (2009), with small modifications. The number of social episodes between rats and the total duration of those contacts were measured as the parameters indicating social behavior.
MK-801, an NMDA channel blocker, disrupted the social interactions decreasing both the number of social episodes between rats and the duration of interaction. These deficits were reversed in a dose-dependent manner after LSP1-2111 administration, with a clear effect observed at a doses of 2 and 5 mg/kg. The drug did not influence the basal social interaction, indicating that positive effects of the compound in the test were not due to apparent behavioral activation or inhibition.
Apart from negative symptoms of schizophrenia, the efficacy of the orthosteric agonists of group III of the mGlu receptors towards cognitive disturbances had not been investigated previously and, as such, we decided to focus also on that aspect of LSP1-2111-induced action. We used the novel object recognition test, which is based on the rodents’ tendency to explore unknown objects in their environment, thus spending more time on sniffing the novel versus the familiar object (Dere et al. 2007). In our experiments, the animals were familiarized with two objects and, after a 1-h delay, they were presented with one familiar and one additional object, which was regarded as a novel object, delays longer than 1 h led to weak or no NOR effects. Because the animals must discriminate between the exposed previously familiar object and novel object that is presented for the first time, the NOR is considered to be a recognition memory task (Barak and Weiner 2011).
In the studies described herein, MK-801 was used to induce the disruption in the NOR performance. LSP1-2111 reversed the MK-801-induced deficit at the dose of 5 mg/kg. Simultaneously, the compound did not influence the behavior of the animals when given alone.
As our recent data show that selective positive mGlu4 receptor allosteric modulators, Lu AF21934 and Lu AF32615, were effective in tests used to investigate positive, negative, and cognitive symptoms (Sławińska et al. 2013, submitted), we are inclined to assume that the effectiveness of LSP1-2111 may be predominantly related to the stimulation of mGlu4 receptors. We are aware that we cannot exclude the involvement of mGlu8 stimulation in the LSP1-2111-induced action; however, because of a 30-fold higher affinity of the compound towards mGlu4 vs. mGlu8 receptor, the compound would mainly stimulate mGlu4 receptors especially at low doses used (Beurrier et al. 2009). However, further studies with mGlu8 selective ligands are highly desired. Although LSP1-2111 displays a similar potency on mGlu4 and mGlu6 receptors, the exclusive expression of the mGlu6 receptor in the retina precludes any action of this compound outside this structure. On the basis of our previous studies, we can state that the stimulation of the mGlu7 receptor is not involved in the antipsychotic-like effect of LSP1-2111 as AMN082 (a mGlu7 PAM) did not induce such an effect (Wierońska et al. 2012b).
The propsychotic effects of NMDA receptor antagonist can be explained by the impairment of the NMDA receptor function at the GABAergic interneurons. The subsequent loss of inhibitory control over glutamatergic pyramidal neurons leads to glutamatergic hyperactivity, which is responsible for behavioral deficits described as positive, negative, and cognitive disturbances (Javitt 1987; Conn et al. 2009). The effectiveness of group II mGlu receptor agonists (Amitai and Markou 2010; Patil et al. 2007; Uslaner et al. 2009) and mGlu5 receptor stimulators (Kanuma et al. 2010) may result from counteracting the glutamatergic hyperactivity (Linden and Schoepp 2006). The prevention of glutamatergic hyperactivity by inhibiting glutamate release via stimulation of mGlu4 receptors might explain the antipsychotic-like effects of LSP1-2111.
As the major limitation of antipsychotic therapy presently used is its poor efficacy towards negative and cognitive symptoms, the results presented here suggest that stimulation of the mGlu4 receptor may represent a promising therapeutic strategy for the future.
In the second part of our investigations, we explored the possible involvement of the serotonergic system in the mechanisms of the antipsychotic-like action of LSP1-2111, focusing on the role of 5-HT1A receptors. The involvement of that subtype in psychosis was postulated several times for a number of reasons. Firstly, the increased 5-HT1A receptor level was observed in the postmortem study of the cerebral cortex and hippocampus of schizophrenic patients (Burnet et al. 1996; Simpson et al. 1996). Secondly, selected atypical antipsychotics, such as, clozapine or perospiron, were shown to be partial agonists of 5-HT1A receptors (Ichikawa et al. 2002; Hagiwara et al. 2008). Thirdly, an agonist of 5-HT1A receptor, 8-OH-DPAT, at the low doses, induced antipsychotic-like effects in the prepulse inhibition, the MK-801-induced hyperlocomotion (Bubeníková-Valesová et al. 2007), and in the social interaction deficits (Boulay et al. 2004). However, 8-OH-DPAT used in those studies stimulated not only 5-HT1A receptors but also had a moderate affinity towards 5-HT7 receptors (Wood et al. 2000). In the present experiments, we used a more active enantiomer of the compound, e.g., (R)-(+)-8-OH-DPAT, a full agonist of 5-HT1A receptors. The second compound used, WAY100635, was a selective, silent antagonist of 5-HT1A receptors (IC50 = 2.2 nM). Both ligands were shown to be active at the somatodendritic 5-HT1A receptors located on the dorsal raphe serotonergic neurons and at the postsynaptic receptors (Fletcher et al. 1996; Yu and Lewander 1997). In addition, the action of the compounds is believed to be dose-dependent, e.g., lower doses are supposed to preferentially bind somatodendritic autoreceptors as shown in several papers before (Ago et al. 2003; Sakaue et al. 2000; Artigas et al. 1996; Barnes and Sharp 1999). Therefore, in the present studies we used low doses of the compounds (WAY100635 at 0.1 mg/kg and (R)-(+)-8-OH-DPAT at 0.01 mg/kg) that did not have any activity on its own. Our previous experiments revealed that WAY100635 at the dose of 0.1 mg/kg effectively antagonized the antidepressant and anxiolytic effects of mGlu ligands (Stachowicz et al. 2009; Pałucha-Poniewiera et al. 2010; Wierońska et al. 2010), and the dose of (R)-(+)-8-OH-DPAT was based on the study of Yu and Lewander (1997), and also our preliminary experiments that showed that the dose of 0.01 mg/kg was the minimal ineffective dose (the antipsychotic-like effects were observed at the dose of 0.025, data not shown).
The pretreatment of animals with WAY100635 (0.1 mg/kg) fully antagonized the action of LSP1-2111 that was administered at the highest effective dose of 5 mg/kg. WAY100635 inhibited the action of LSP1-2111 in: hyperactivity, head twitches, social interactions, and NOR tests. Concomitantly, in all of these procedures the clear antipsychotic-like action was evident after co-administration of subeffective doses of LSP1-2111 and (R)-(+)-8-OH-DPAT. The control experiments eliminated the potential false effect as neither of the combinations of LSP1-2111 with the 5HT1A receptor agonist/antagonist influenced basal behavioral activity in any way that could influence the results. Only the combination of LSP1-2111 with WAY100635 decreased the basal locomotor activity of mice habituated to locomotor activity cages; although, as the WAY100635 reversed the effect of LSP1-2111 on MK-801 or amphetamine hyperactivity and increased rather than decreased the ambulation scores, this aspect of interaction may be disregarded in our considerations. The important thing is, that those interactions between 5-HT1A-mGlu4 receptors were not observed when higher doses of 5-HT1A receptor ligands were administered, as shown in the MK-801-induced hyperactivity test as an example.
On the basis of the results described above, the serotonergic profile of the antipsychotic-like effectiveness of LSP1-2111 is beginning to emerge, and the 5-HT1A receptor seems to play a substantial role in its action. This phenomenon can be explained at least in two ways.
It has been shown that the non-competitive NMDA receptor antagonists may enhance the expression of serotonergic stimulation, which may contribute to the pathogenesis of schizophrenia (Dall'Olio et al. 1999). Accordingly, the stimulation of somatodendritic 5-HT1A receptors by DPAT may abolish abnormal serotonergic activity and thus induce an antipsychotic-like effect. As the support for this statement the decrease of concentration of 5-hydroxyindoleacetic acid (5-HIAA), that indicates a decrease in turnover of 5-HT (Larsson et al. 1990), and the reduced extracellular 5-HT level in the striatum (Allen et al. 1997), were observed after 8-OH-DPAT administration.
The pattern of expression of 5-HT1A receptors may also constitute a key culprit of this phenomenon. Nearly 50 % of pyramidal neurons in the cortex are positive for the 5-HT1A receptors which are expressed on the axonal hillocks (Wedzony et al. 2008). These heteroreceptors would inhibit the glutamate release after activation, and this aspect of serotonergic agonist action may be important in light of the glutamatergic theory of psychosis. Thus, synergism between LSP1-2111 and (R)-(+)-8-OH-DPAT actions may develop due to the activation of mGlu4/8 autoreceptors and 5-HT1A heteroreceptors expressed on glutamatergic terminals. Stimulation of both subtypes may play in concert resulting in the inhibition of glutamate release. Although the detailed colocalization studies on the 5-HT1A-mGlu4/8 receptors are lacking, we can speculate that those receptors may colocalize. It is known that relationships between mGlu2/3 and 5-HT2A receptors exist (González-Maeso et al. 2008) and are involved in the antipsychotic activity of group II mGlu receptor ligands (Amitai and Markou 2010; Marek et al. 2000; Uslaner et al. 2009). Therefore, it can be hypothesized that the dimerization between 5-HT1A and mGlu4 receptors also occur and may be involved in the interactions between the LSP1-2111 and 5-HT1A receptor ligands observed here. Together with the effects of the stimulation of somatodendritic 5-HT1A receptors described above in raphe nuclei (point 1), 5-HT1A and mGlu4/8 receptors may represent a promising tandem (or triplet) in schizophrenia research.
The fact that the (R)-(+)-8-OH-DPAT at the ten times higher dose of 0.1 mg/kg did not act synergistically with LSP1-2111 may confirm our hypothesis that the activation of presynaptic 5-HT1A receptors could be responsible for the interaction with LSP1-2111. The higher doses of (R)-(+)-8-OH-DPAT may induce a distinct behavioral change that could be associated with the 5-HT syndrome, related to 5-HT1A selective activation such as changes in locomotor activity, flat body posture, and forepaw treading (Blanchard et al. 1997; Yu and Lewander 1997; Goodwin et al. 1986, 1987; Larsson et al. 1990).
Also, the fact that WAY100635 at a dose of 3 mg/kg did not block the action of LSP1-2111 further enhances the probability that presynaptic 5-HT1A receptors are mainly involved in the interactions described in this paper. This dose of WAY100635 produced an antipsychotic effect confirming that data of Wedzony et al. (2000), who described such an effect of WAY100135.
To summarize: the proper activity of pyramidal neurons and the functional integrity between both the glutamatergic and serotonergic system may be crucial for efficient antipsychotic treatment. The data suggest that cooperation between group III Glu receptor and 5-HT1A receptor agonists may be efficient in attenuating not only positive symptoms but may be especially important in other key features of schizophrenia poorly controlled by dopamine-based antipsychotics, namely social functioning and cognitive disturbances.
Acknowledgements
The work was supported by the project ModAll Nr UDA-POIG.01.03.01-12-100/08-00 given to A.P.
References
- Acher FC, Tellier FJ, Azerad R, Brabet IN, Fagni L, Pin JP. Synthesis and pharmacological characterization of aminocyclopentanetricarboxylic acids: new tools to discriminate between metabotropic glutamate receptor subtypes. J Med Chem. 1997;40(19):3119–3129. doi: 10.1021/jm970207b. [DOI] [PubMed] [Google Scholar]
- Acher F, Selvam C, Triballeau N, Pin JP, Bertrand HO (2007) Hypo-phosphorous acid derivatives and their therapeutical applications. U.S. Patent Application WO2007052169
- Ago Y, Koyama Y, Baba A, Matsuda T. Regulation by 5-HT1A receptors of the in vivo release of 5-HT and DA in mouse frontal cortex. Neuropharmacology. 2003;45(8):1050–1056. doi: 10.1016/S0028-3908(03)00304-6. [DOI] [PubMed] [Google Scholar]
- Allen AR, Singh A, Zhuang ZP, Kung MP, Kung HF, Lucki I. The 5-HT1A receptor antagonist p-MPPI blocks responses mediated by postsynaptic and presynaptic 5-HT1A receptors. Pharmacol Biochem Behav. 1997;57(1–2):301–307. doi: 10.1016/S0091-3057(96)00339-5. [DOI] [PubMed] [Google Scholar]
- Alphs L, Benedetti F, Fleischhacker WW, Kane JM. Placebo-related effects in clinical trials in schizophrenia: what is driving this phenomenon and what can be done to minimize it? Int J Neuropsychopharmacol. 2012;15(7):1003–1014. doi: 10.1017/S1461145711001738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amitai N, Markou A. Effects of metabotropic glutamate receptor 2/3 agonism and antagonism on schizophrenia-like cognitive deficits induced by phencyclidine in rats. Eur J Pharmacol. 2010;639(1–3):67–80. doi: 10.1016/j.ejphar.2009.12.040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andlin-Sobocki P, Rössler W. Cost of psychotic disorders in Europe. Eur J Neurol. 2005;12(Suppl 1):74–77. doi: 10.1111/j.1468-1331.2005.01198.x. [DOI] [PubMed] [Google Scholar]
- Artigas F, Romero L, de Montigny C, Blier P. Acceleration of the effect of selected antidepressant drugs in major depression by 5-HT1A antagonist. Trends Neurosci. 1996;19(9):378–383. doi: 10.1016/S0166-2236(96)10037-0. [DOI] [PubMed] [Google Scholar]
- Barak S, Weiner I. Putative cognitive enhancers in preclinical models related to schizophrenia: the search for an elusive target. Pharmacol Biochem Behav. 2011;99(2):164–189. doi: 10.1016/j.pbb.2011.03.011. [DOI] [PubMed] [Google Scholar]
- Barnes NM, Sharp T. A review of central 5-HT receptors and their function. Neuropharmacology. 1999;38(8):1083–1152. doi: 10.1016/S0028-3908(99)00010-6. [DOI] [PubMed] [Google Scholar]
- Beurrier C, Lopez S, Révy D, Selvam C, Goudet C, Lhérondel M, Gubellini P, Kerkerian-LeGoff L, Acher F, Pin JP, Amalric M. Electrophysiological and behavioral evidence that modulation of metabotropic glutamate receptor 4 with a new agonist reverses experimental parkinsonism. FASEB J. 2009;23(10):3619–3628. doi: 10.1096/fj.09-131789. [DOI] [PubMed] [Google Scholar]
- Blanchard RJ, Griebel G, Guardiola-Lemaître B, Brush MM, Lee J, Blanchard DC. An ethopharmacological analysis of selective activation of 5-HT1A receptors: the mouse 5-HT1A syndrome. Pharmacol Biochem Behav. 1997;57(4):897–908. doi: 10.1016/S0091-3057(96)00472-8. [DOI] [PubMed] [Google Scholar]
- Boulay D, Depoortre R, Louis C, Perrault G, Griebel G, Soubrié P. SSR181507, a putative atypical antipsychotic with dopamine D2 antagonist and 5-HT1A agonist activities: improvement of social interaction deficits induced by phencyclidine in rats. Neuropharmacology. 2004;46(8):1121–1129. doi: 10.1016/j.neuropharm.2004.02.008. [DOI] [PubMed] [Google Scholar]
- Bubeníková-Valesová V, Votava M, Pálenícek T, Horácek J. The opposite effect of a low and a high dose of serotonin-1A agonist on behavior induced by MK-801. Neuropharmacology. 2007;52(4):1071–1078. doi: 10.1016/j.neuropharm.2006.11.004. [DOI] [PubMed] [Google Scholar]
- Burnet PW, Eastwood SL, Harrison PJ. 5-HT1A and 5-HT2A receptor mRNAs and binding site densities are differentially altered in schizophrenia. Neuropsychopharmacology. 1996;15(5):442–455. doi: 10.1016/S0893-133X(96)00053-X. [DOI] [PubMed] [Google Scholar]
- Cartmell J, Schoepp DD. Regulation of neurotransmitter release by metabotropic glutamate receptors. J Neurochem. 2000;75(3):889–907. doi: 10.1046/j.1471-4159.2000.0750889.x. [DOI] [PubMed] [Google Scholar]
- Cartmell J, Monn JA, Schoepp DD. Attenuation of specific PCP-evoked behaviors by the potent mGlu2/3 receptor agonist, LY379268 and comparison with the atypical antipsychotic, clozapine. Psychopharmacology (Berlin) 2000;148(4):423–429. doi: 10.1007/s002130050072. [DOI] [PubMed] [Google Scholar]
- Cartmell J, Monn JA, Schoepp DD. The mGlu(2/3) receptor agonist LY379268 selectively blocks amphetamine ambulations and rearing. Eur J Pharmacol. 2000;400(2–3):221–224. doi: 10.1016/S0014-2999(00)00423-4. [DOI] [PubMed] [Google Scholar]
- Conn PJ, Lindsley CW, Jones CK. Activation of metabotropic glutamate receptors as a novel approach for the treatment of schizophrenia. Trends Pharmacol Sci. 2009;30(1):25–31. doi: 10.1016/j.tips.2008.10.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cramer P, Bowen J, O'Neill M. Schizophrenics and social judgement. Why do schizophrenics get it wrong? Br J Psychiatry. 1992;160:481–487. doi: 10.1192/bjp.160.4.481. [DOI] [PubMed] [Google Scholar]
- Dall'Olio R, Gaggi R, Bonfante V, Gandolfi O. The non-competitive NMDA receptor blocker dizocilpine potentiates serotonergic function. Behav Pharmacol. 1999;10(1):63–71. doi: 10.1097/00008877-199902000-00006. [DOI] [PubMed] [Google Scholar]
- Dere E, Huston JP, De Souza Silva MA. The pharmacology, neuroanatomy and neurogenetics of one-trial object recognition in rodents. Neurosci Biobehav Rev. 2007;31(5):673–704. doi: 10.1016/j.neubiorev.2007.01.005. [DOI] [PubMed] [Google Scholar]
- Fletcher A, Forster EA, Bill DJ, Brown G, Cliffe IA, Hartley JE, Jones DE, McLeachan A, Stanhope KJ, Critchley DJ, Childs KJ, Middlefell VC, Lanfumey L, Corradetti R, Lapore AM, Gozlan H, Hamon M, Douris CT. Electrophysiological, biochemical, neurohormonal and behavioral studies with WAY100635, a potent, selective and silent 5-HT1A receptor antagonist. Behav Brain Res. 1996;73:337–353. doi: 10.1016/0166-4328(96)00118-0. [DOI] [PubMed] [Google Scholar]
- Flor PJ, Acher FC. Orthosteric versus allosteric GPCR activation: the great challenge of group-III mGluRs. Biochem Pharmacol. 2012;84(4):414–424. doi: 10.1016/j.bcp.2012.04.013. [DOI] [PubMed] [Google Scholar]
- González-Maeso J, Ang RL, Yuen T, Chan P, Weisstaub NV, López-Giménez JF, Zhou M, Okawa Y, Callado LF, Milligan G, Gingrich JA, Filizola M, Meana JJ, Sealfon SC. Identification of a serotonin/glutamate receptor complex implicated in psychosis. Nature. 2008;452(7183):93–97. doi: 10.1038/nature06612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodwin GM, De Souza RJ, Wood AJ, Green AR. The enhancement by lithium of the 5-HT1A mediated serotonin syndrome produced by 8-OH-DPAT in the rat: evidence for a post-synaptic mechanism. Psychopharmacology (Berlin) 1986;90(4):488–493. doi: 10.1007/BF00174066. [DOI] [PubMed] [Google Scholar]
- Goodwin GM, De Souza RJ, Green AR. Attenuation by electroconvulsive shock and antidepressant drugs of the 5-HT1A receptor-mediated hypothermia and serotonin syndrome produced by 8-OH-DPAT in the rat. Psychopharmacology (Berlin) 1987;91(4):500–505. doi: 10.1007/BF00216018. [DOI] [PubMed] [Google Scholar]
- Hagiwara H, Fujita Y, Ishima T, Kunitachi S, Shirayama Y, Iyo M, Hashimoto K. Phencyclidine-induced cognitive deficits in mice are improved by subsequent subchronic administration of the antipsychotic drug perospirone: role of serotonin 5-HT1A receptors. Eur Neuropsychopharmacol. 2008;18(6):448–454. doi: 10.1016/j.euroneuro.2007.11.005. [DOI] [PubMed] [Google Scholar]
- Homberg JR, De Boer SF, Raas HS, Olivier JD, Verheul M, Ronken E, Cools AR, Ellenbroek BA, Schoffelmeer AN, Vanderschuren LJ, De Vries TJ, Cuppen E. Adaptations in pre- and postsynaptic 5-HT1A receptor function and cocaine supersensitivity in serotonin transporter knockout rats. Psychopharmacology (Berlin) 2008;200(3):367–380. doi: 10.1007/s00213-008-1212-x. [DOI] [PubMed] [Google Scholar]
- Horiguchi M, Huang M, Meltzer HY. The role of 5-hydroxytryptamine 7 receptors in the phencyclidine-induced novel object recognition deficit in rats. J Pharmacol Exp Ther. 2011;338(2):605–614. doi: 10.1124/jpet.111.180638. [DOI] [PubMed] [Google Scholar]
- Horiguchi M, Huang M, Meltzer HY. Interaction of mGlu2/3 agonism with clozapine and lurasidone to restore novel object recognition in subchronic phencyclidine-treated rats. Psychopharmacology (Berlin) 2011;217(1):13–24. doi: 10.1007/s00213-011-2251-2. [DOI] [PubMed] [Google Scholar]
- Ichikawa J, Li Z, Dai J, Meltzer HY. Atypical antipsychotic drugs, quetiapine, iloperidone, and melperone, preferentially increase dopamine and acetylcholine release in rat medial prefrontal cortex: role of 5-HT1A receptor agonism. Brain Res. 2002;956(2):349–357. doi: 10.1016/S0006-8993(02)03570-9. [DOI] [PubMed] [Google Scholar]
- Iijima M, Shimazaki T, Ito A, Chaki S. Effects of metabotropic glutamate 2/3 receptor antagonists in the stress-induced hyperthermia test in singly housed mice. Psychopharmacology (Berlin) 2007;190(2):233–239. doi: 10.1007/s00213-006-0618-6. [DOI] [PubMed] [Google Scholar]
- Javitt DC. Negative schizophrenic symptomatology and the PCP (phencyclidine) model of schizophrenia. Hillside J Clin Psychiatry. 1987;9(1):12–35. [PubMed] [Google Scholar]
- Kanuma K, Aoki T, Shimazaki Y. Recent patents on positive allosteric modulators of the metabotropic glutamate 5 receptor as a potential treatment for schizophrenia. Recent Pat CNS Drug Discov. 2010;5(1):23–34. doi: 10.2174/157488910789753512. [DOI] [PubMed] [Google Scholar]
- Kinon BJ, Zhang L, Millen BA, Osuntokun OO, Williams JE, Kollack-Walker S, Jackson K, Kryzhanovskaya L, Jarkova N, the HBBI Study Group A multicenter, inpatient, phase 2, double-blind, placebo-controlled dose-ranging study of LY2140023 monohydrate in patients with DSM-IV schizophrenia. J Clin Psychopharmacol. 2011;31(3):349–355. doi: 10.1097/JCP.0b013e318218dcd5. [DOI] [PubMed] [Google Scholar]
- Larsson LG, Rényi L, Ross SB, Svensson B, Angeby-Möller K. Different effects on the responses of functional pre- and postsynaptic 5-HT1A receptors by repeated treatment of rats with the 5-HT1A receptor agonist 8-OH-DPAT. Neuropharmacology. 1990;29(2):85–91. doi: 10.1016/0028-3908(90)90047-U. [DOI] [PubMed] [Google Scholar]
- Linden AM, Schoepp DD. Metabotropic glutamate receptor targets for neuropsychiatric disorders. Drug Discov Today Ther Strateg. 2006;3:507–517. doi: 10.1016/j.ddstr.2006.10.018. [DOI] [Google Scholar]
- Marek GJ, Wright RA, Schoepp DD, Monn JA, Aghajanian GK. Physiological antagonism between 5-hydroxytryptamine(2A) and group II metabotropic glutamate receptors in prefrontal cortex. J Pharmacol Exp Ther. 2000;292(1):76–87. [PubMed] [Google Scholar]
- Meltzer HY, Li Z, Kaneda Y, Ichikawa J. Serotonin receptors: their key role in drugs to treat schizophrenia. Prog Neuropsychopharmacol Biol Psychiatry. 2003;27(7):1159–1172. doi: 10.1016/j.pnpbp.2003.09.010. [DOI] [PubMed] [Google Scholar]
- Monn JA, Valli MJ, Massey SM, Hansen MM, Kress TJ, Wepsiec JP, Harkness AR, Grutsch JL, Jr, Wright RA, Johnson BG, Andis SL, Kingston A, Tomlinson R, Lewis R, Griffey KR, Tizzano JP, Schoepp DD. Synthesis, pharmacological characterization, and molecular modeling of heterobicyclic amino acids related to (+)-2-aminobicyclo[3.1.0] hexane-2,6-dicarboxylic acid (LY354740): identification of two new potent, selective, and systemically active agonists for group II metabotropic glutamate receptors. J Med Chem. 1999;42(6):1027–1040. doi: 10.1021/jm980616n. [DOI] [PubMed] [Google Scholar]
- Mueser KT, McGurk SR. Schizophrenia. Lancet. 2004;363(9426):2063–2072. doi: 10.1016/S0140-6736(04)16458-1. [DOI] [PubMed] [Google Scholar]
- Ohno Y. Therapeutic role of 5-HT1A receptors in the treatment of schizophrenia and Parkinson's disease. CNS Neurosci Ther. 2011;17(1):58–65. doi: 10.1111/j.1755-5949.2010.00211.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olivier B, Zethof TJ, Ronken E, van der Heyden JA. Anxiolytic effects of flesinoxan in the stress-induced hyperthermia paradigm in singly-housed mice are 5-HT1A mediated. Eur J Pharmacol. 1998;342:177–182. doi: 10.1016/S0014-2999(97)01482-9. [DOI] [PubMed] [Google Scholar]
- Olivier JD, Cools AR, Olivier B, Homberg JR, Cuppen E, Ellenbroek BA. Stress-induced hyperthermia and basal body temperature are mediated by different 5-HT(1A) receptor populations: a study in SERT knockout rats. Eur J Pharmacol. 2008;590(1–3):190–197. doi: 10.1016/j.ejphar.2008.06.008. [DOI] [PubMed] [Google Scholar]
- Pałucha-Poniewiera A, Kłodzińska A, Stachowicz K, Tokarski K, Hess G, Schann S, Frauli M, Neuville P, Pilc A. Peripheral administration of group III mGlu receptor agonist ACPT-I exerts potential antipsychotic effects in rodents. Neuropharmacology. 2008;55(4):517–524. doi: 10.1016/j.neuropharm.2008.06.033. [DOI] [PubMed] [Google Scholar]
- Pałucha-Poniewiera A, Brański P, Lenda T, Pilc A. The antidepressant-like action of metabotropic glutamate 7 receptor agonist N, N'-bis(diphenylmethyl)-1,2-ethanediamine (AMN082) is serotonin-dependent. J Pharmacol Exp Ther. 2010;334(3):1066–1074. doi: 10.1124/jpet.110.169730. [DOI] [PubMed] [Google Scholar]
- Patil ST, Zhang L, Martenyi F, Lowe SL, Jackson KA, Andreev BV, Avedisova AS, Bardenstein LM, Gurovich IY, Morozova MA, Mosolov SN, Neznanov NG, Reznik AM, Smulevich AB, Tochilov VA, Johnson BG, Monn JA, Schoepp DD. Activation of mGlu2/3 receptors as a new approach to treat schizophrenia: a randomized Phase 2 clinical trial. Nat Med. 2007;13(9):1102–1107. doi: 10.1038/nm1632. [DOI] [PubMed] [Google Scholar]
- Pin JP, Duvoisin R. The metabotropic glutamate receptors: structure and functions. Neuropharmacology. 1995;34(1):1–26. doi: 10.1016/0028-3908(94)00129-G. [DOI] [PubMed] [Google Scholar]
- Rorick-Kehn LM, Johnson BG, Knitowski KM, Salhoff CR, Witkin JM, Perry KW, Griffey KI, Tizzano JP, Monn JA, McKinzie DL, Schoepp DD. In vivo pharmacological characterization of the structurally novel, potent, selective mGlu2/3 receptor agonist LY404039 in animal models of psychiatric disorders. Psychopharmacology (Berlin) 2007;193(1):121–136. doi: 10.1007/s00213-007-0758-3. [DOI] [PubMed] [Google Scholar]
- Sakaue M, Somboonthum P, Nishihara B, Koyama Y, Hashimoto H, Baba A, Matsuda T. Postsynaptic 5-hydroxytryptamine(1A) receptor activation increases in vivo dopamine release in rat prefrontal cortex. Br J Pharmacol. 2000;129(5):1028–1034. doi: 10.1038/sj.bjp.0703139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Satow A, Suzuki G, Maehara S, Hikichi H, Murai T, Murai T, Kawagoe-Takaki H, Hata M, Ito S, Ozaki S, Kawamoto H, Ohta H. Unique antipsychotic activities of the selective metabotropic glutamate receptor 1 allosteric antagonist 2-cyclopropyl-5-[1-(2-fluoro-3-pyridinyl)-5-methyl-1H-1,2,3-triazol-4-yl]-2,3-dihydro-1H-isoindol-1-one. J Pharmacol Exp Ther. 2009;330(1):179–190. doi: 10.1124/jpet.109.151118. [DOI] [PubMed] [Google Scholar]
- Simpson MD, Lubman DI, Slater P, Deakin JF. Autoradiography with [3H](R)-(+)-8-Hydroxy-DPAT hydrobromide reveals increases in 5-HT(1A) receptors in ventral prefrontal cortex in schizophrenia. Biol Psychiatry. 1996;39(11):919–928. doi: 10.1016/0006-3223(95)00026-7. [DOI] [PubMed] [Google Scholar]
- Sławińska A, Wieronska JM, Stachowicz K, Łasoń-Tyburkiewicz M, Gruca P, Papp M, Kusek M, Tokarski K, Doller D, Pilc A (2013) The antipsychotic-like effects of mGlu4 receptor positive allosteric modulators in rodents. JPET, submitted [DOI] [PMC free article] [PubMed]
- Stachowicz K, Kłodzińska A, Palucha-Poniewiera A, Schann S, Neuville P, Pilc A. The group III mGlu receptor agonist ACPT-I exerts anxiolytic-like but not antidepressant-like effects, mediated by the serotonergic and GABA-ergic systems. Neuropharmacology. 2009;57(3):227–234. doi: 10.1016/j.neuropharm.2009.06.005. [DOI] [PubMed] [Google Scholar]
- Uslaner JM, Smith SM, Huszar SL, Pachmerhiwala R, Hinchliffe RM, Vardigan JD, Hutson PH. Combined administration of an mGlu2/3 receptor agonist and a 5-HT 2A receptor antagonist markedly attenuate the psychomotor-activating and neurochemical effects of psychostimulants. Psychopharmacology (Berlin) 2009;206(4):641–651. doi: 10.1007/s00213-009-1644-y. [DOI] [PubMed] [Google Scholar]
- Vinkers CH, de Jong NM, Kalkman CJ, Westphal KG, van Oorschot R, Olivier B, Korte SM, Groenink L. Stress-induced hyperthermia is reduced by rapid-acting anxiolytic drugs independent of injection stress in rats. Pharmacol Biochem Behav. 2009;93(4):413–418. doi: 10.1016/j.pbb.2009.05.017. [DOI] [PubMed] [Google Scholar]
- Wedzony K, Maćkowiak M, Zajaczkowski W, Fijał K, Chocyk A, Czyrak A. WAY 100135, an antagonist of 5-HT1A serotonin receptors, attenuates psychotomimetic effects of MK-801. Neuropsychopharmacology. 2000;23(5):547–559. doi: 10.1016/S0893-133X(00)00150-0. [DOI] [PubMed] [Google Scholar]
- Wedzony K, Chocyk A, Maćkowiak M. A search for colocalization of serotonin 5-HT2A and 5-HT1A receptors in the rat medial prefrontal and entorhinal cortices—immunohistochemical studies. J Physiol Pharmacol. 2008;59(2):229–238. [PubMed] [Google Scholar]
- Weinberger DR. Schizophrenia drug says goodbye to dopamine. Nat Med. 2007;13(9):1018–1019. doi: 10.1038/nm0907-1018. [DOI] [PubMed] [Google Scholar]
- Wierońska JM, Stachowicz K, Pałucha-Poniewiera A, Acher F, Brański P, Pilc A. Metabotropic glutamate receptor 4 novel agonist LSP1-2111 with anxiolytic, but not antidepressant-like activity, mediated by serotonergic and GABAergic systems. Neuropharmacology. 2010;59(7–8):627–634. doi: 10.1016/j.neuropharm.2010.08.008. [DOI] [PubMed] [Google Scholar]
- Wierońska JM, Kusek M, Tokarski K, Wabno J, Froestl W, Pilc A. The GABA B receptor agonist CGP44532 and the positive modulator GS39783 reverse some behavioral changes related to positive syndromes of psychosis in mice. Br J Pharmacol. 2011;163(5):1034–1047. doi: 10.1111/j.1476-5381.2011.01301.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wierońska JM, Stachowicz K, Brański P, Pałucha-Poniewiera A, Pilc A. On the mechanism of anti-hyperthermic effects of LY379268 and LY487379, group II mGlu receptors activators, in the stress-induced hyperthermia in singly housed mice. Neuropharmacology. 2012;62(1):322–331. doi: 10.1016/j.neuropharm.2011.07.042. [DOI] [PubMed] [Google Scholar]
- Wierońska JM, Stachowicz K, Acher F, Lech T, Pilc A. Opposing efficacy of group III mGlu receptor activators, LSP1-2111 and AMN082, in animal models of positive symptoms of schizophrenia. Psychopharmacology (Berlin) 2012;220(3):481–494. doi: 10.1007/s00213-011-2502-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wood M, Chaubey M, Atkinson P, Thomas DR. Antagonist activity of meta-chlorophenylpiperazine and partial agonist activity of 8-OH-DPAT at the 5-HT(7) receptor.Eur. J Pharmacol. 2000;396(1):1–8. doi: 10.1016/s0014-2999(00)00213-2. [DOI] [PubMed] [Google Scholar]
- Yu H, Lewander T. Pharmacokinetic and pharmacodynamic studies of (R)-8-hydroxy-2-(di-n-propylamino)tetralin in the rat. Eur Neuropsychopharmacol. 1997;7(3):165–172. doi: 10.1016/S0924-977X(96)00395-1. [DOI] [PubMed] [Google Scholar]