Abstract
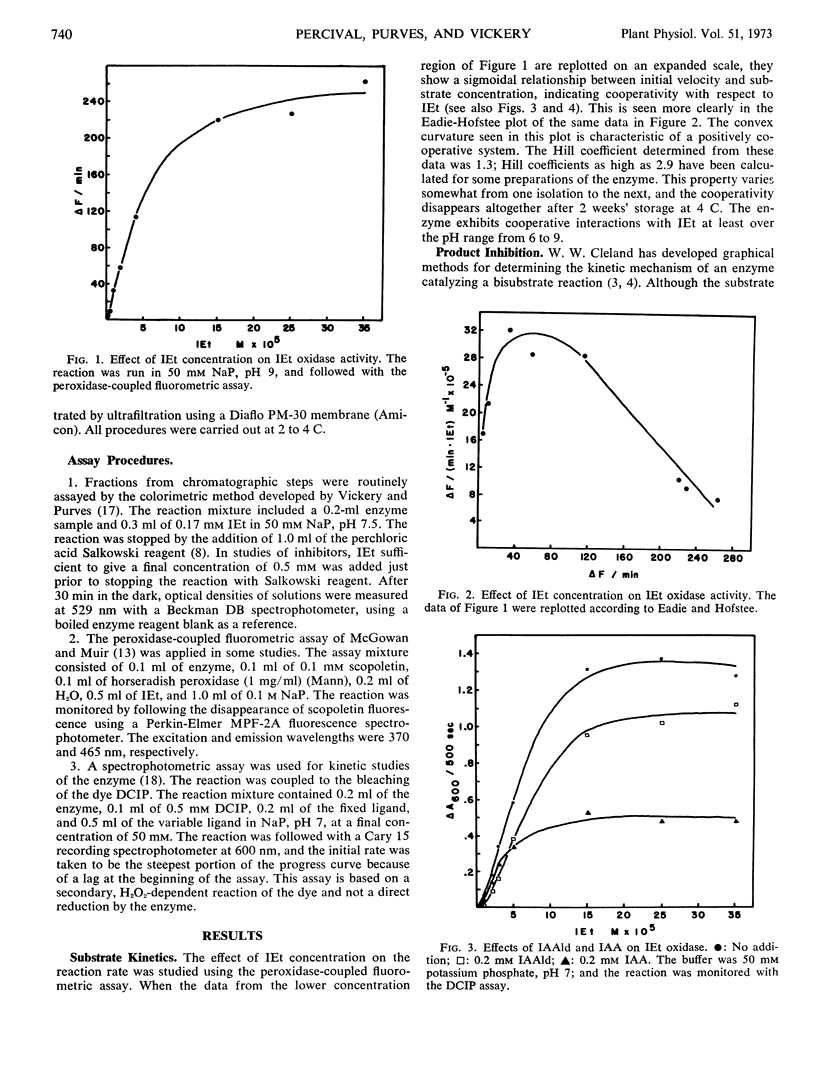
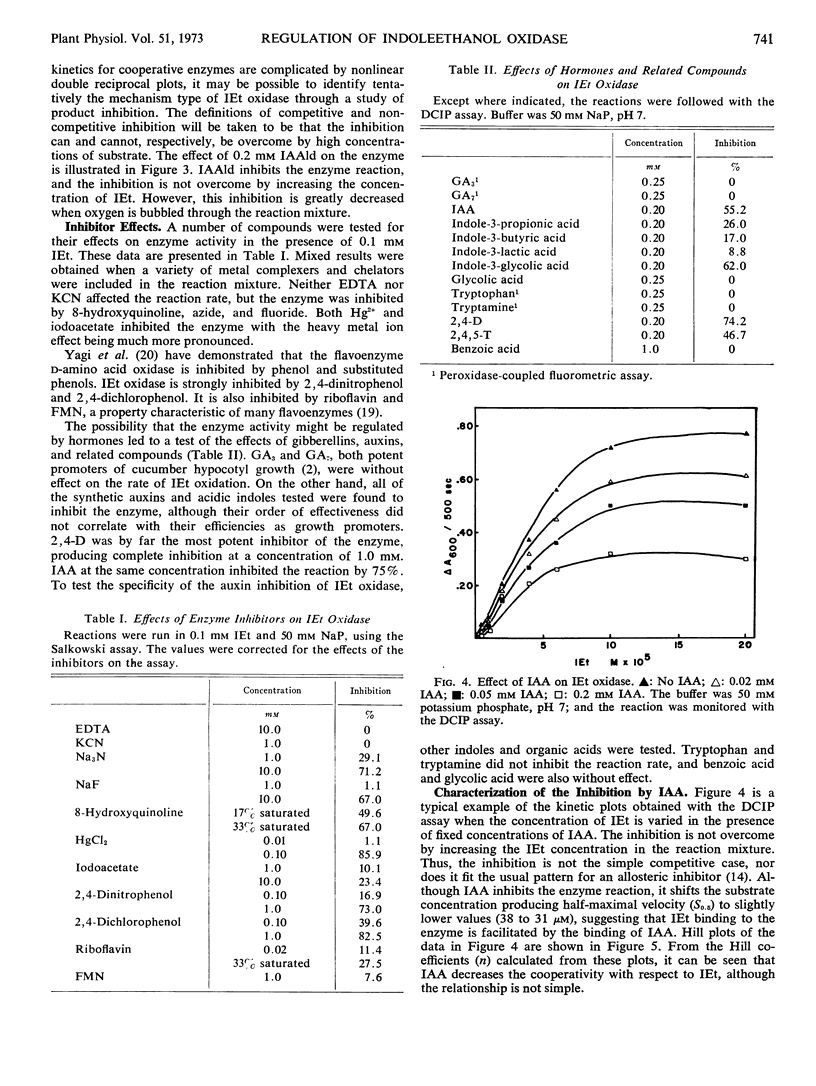
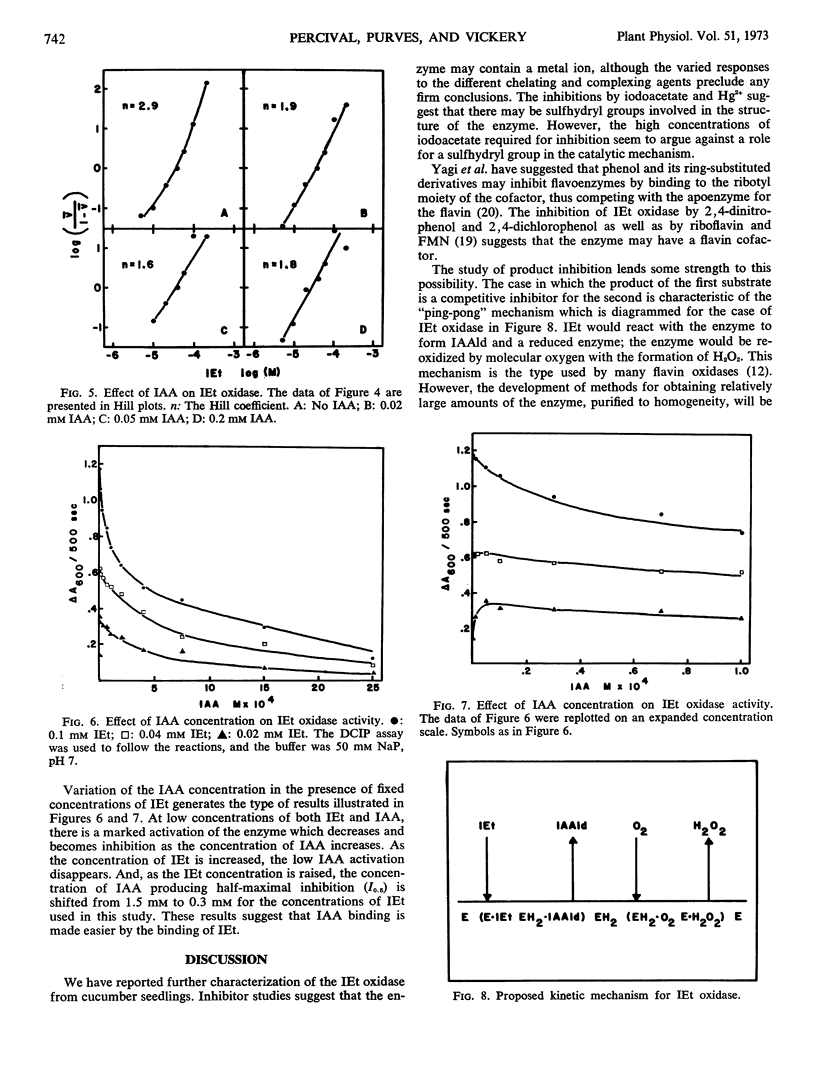
We report the further characterization of indole-3-ethanol oxidase from cucumber seedlings. The effects of various inhibitors suggest that the enzyme may be a flavoprotein with a metal ion and sulfhydryl groups required for full activity. Indole-3-acetaldehyde, a product of the reaction, inhibits the enzyme. This inhibition is overcome by O2 but not by indole-3-ethanol, indicating that the kinetic mechanism of the enzyme is a ping-pong Bi-Bi. The enzyme undergoes cooperative interactions with indoleethanol, yielding Hill coefficients as high as 2.96. Gibberellins are without effect on the enzyme, but it is inhibited by several acidic indoles possessing growth-promoting activity and by two synthetic auxins, 2,4-dichlorophenoxyacetic acid and 2,4,5-trichlorophenoxyacetic acid. Increasing concentrations of indoleacetic acid (IAA) brought about a slight reduction in the indoleethanol concentration producing halfmaximal velocity. Increasing levels of indoleethanol decreased the concentration of IAA required for half-maximal inhibition. At low concentrations of indoleethanol, low levels of IAA activated rather than inhibited. The effect of IAA was not overcome at higher levels of indoleethanol. These results may be interpreted as showing that IAA is a noncompetitive inhibitor which binds to that conformation of the enzyme which also binds indoleethanol. The significance of these interactions for the regulation of IAA biosynthesis is discussed.
Full text
PDF

Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andreae W. A., Good N. E. The Formation of Indoleacetylaspartic Acid in Pea Seedlings. Plant Physiol. 1955 Jul;30(4):380–382. doi: 10.1104/pp.30.4.380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. I. Nomenclature and rate equations. Biochim Biophys Acta. 1963 Jan 8;67:104–137. doi: 10.1016/0006-3002(63)91800-6. [DOI] [PubMed] [Google Scholar]
- CLELAND W. W. The kinetics of enzyme-catalyzed reactions with two or more substrates or products. II. Inhibition: nomenclature and theory. Biochim Biophys Acta. 1963 Feb 12;67:173–187. doi: 10.1016/0006-3002(63)91815-8. [DOI] [PubMed] [Google Scholar]
- FURUYA M., GALSTON A. W., STOWE B. B. Isolation from peas of co-factors and inhibitors of indolyl-3-acetic acid oxidase. Nature. 1962 Feb 3;193:456–457. doi: 10.1038/193456a0. [DOI] [PubMed] [Google Scholar]
- Gordon S. A., Weber R. P. COLORIMETRIC ESTIMATION OF INDOLEACETIC ACID. Plant Physiol. 1951 Jan;26(1):192–195. doi: 10.1104/pp.26.1.192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goren R., Tomer E. Effects of Seselin and Coumarin on Growth, Indoleacetic Acid Oxidase, and Peroxidase, with Special Reference to Cucumber (Cucumis sativa L.) Radicles. Plant Physiol. 1971 Feb;47(2):312–316. doi: 10.1104/pp.47.2.312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labarca C., Nicholls P. B., Bandurski R. S. A partial characterization of indoleacetylinositols from ZEA mays. Biochem Biophys Res Commun. 1965 Sep 8;20(5):641–646. doi: 10.1016/0006-291x(65)90448-1. [DOI] [PubMed] [Google Scholar]
- Lee T. T. Promotion of indoleacetic Acid oxidase isoenzymes in tobacco callus cultures by indoleacetic Acid. Plant Physiol. 1971 Jul;48(1):56–59. doi: 10.1104/pp.48.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MONOD J., WYMAN J., CHANGEUX J. P. ON THE NATURE OF ALLOSTERIC TRANSITIONS: A PLAUSIBLE MODEL. J Mol Biol. 1965 May;12:88–118. doi: 10.1016/s0022-2836(65)80285-6. [DOI] [PubMed] [Google Scholar]
- McGowan R. E., Muir R. M. Purification and Properties of Amine Oxidase from Epicotyls of Pisum sativum. Plant Physiol. 1971 May;47(5):644–648. doi: 10.1104/pp.47.5.644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vickery L. E., Purves W. K. Isolation of Indole-3-ethanol Oxidase from Cucumber Seedlings. Plant Physiol. 1972 May;49(5):716–721. doi: 10.1104/pp.49.5.716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- YAGI K., OZAWA T., OKADA K. Mechanism of inhibition of D-amino acid oxidase. II. Inhibitory actions of benzene derivatives. Biochim Biophys Acta. 1959 Sep;35:102–110. doi: 10.1016/0006-3002(59)90339-7. [DOI] [PubMed] [Google Scholar]



