Abstract
When corresponding areas of the two eyes view dissimilar images, stable perception gives way to visual competition wherein perceptual awareness alternates between those images. Moreover, a given image can remain visually dominant for several seconds at a time even when the competing images are swapped between the eyes multiple times each second. This perceptual stability across eye swaps has led to the widespread belief that this unique form of visual competition, dubbed stimulus rivalry, is governed by eye-independent neural processes at a purely binocular stage of cortical processing. We tested this idea by investigating the influence of stimulus rivalry on the buildup of the threshold elevation aftereffect, a form of contrast adaptation thought to transpire at early cortical stages that include eye-specific neural activity. Weaker threshold elevation aftereffects were observed when the adapting image was engaged in stimulus rivalry than when it was not, indicating diminished buildup of adaptation during stimulus-rivalry suppression. We then confirmed that this reduction occurred, in part, at eye-specific neural stages by showing that suppression of an image at a given moment specifically diminished adaptation associated with the eye viewing the image at that moment. Considered together, these results imply that eye-specific neural events at early cortical processing stages contribute to stimulus rivalry. We have developed a computational model of stimulus rivalry that successfully implements this idea.
Keywords: ambiguous stimuli, binocular rivalry, bistable perception, flicker-and-swap rivalry
Binocular rivalry is a compelling form of perceptual bistability that is frequently touted as a potent tool for studying neural correlates of visual perception (1). In conventional binocular rivalry, an observer simultaneously views a dissimilar image with each eye for a prolonged period, resulting in a perceptual cycle during which the images alternately reach awareness for seconds at a time. In a highly influential paper, Logothetis et al. (2) demonstrated that yoking each image to a given eye is unnecessary. In a rivalry variant dubbed stimulus rivalry, the images are swapped rapidly and repetitively between the two eyes, yet periods of uninterrupted visibility still last several seconds, much longer than the brief durations that a given image is present in a given eye. This remarkable survival of image dominance across eye swaps is widely regarded as unassailable evidence that competition between eye-independent neural representations, rather than between incompatible monocular neural signals, governs perception in stimulus rivalry (2–5). In this paper, we suggest a revision of this interpretation by demonstrating psychophysically that perception during stimulus rivalry is reflected in neural events at early, monocular processing stages and by presenting a concise computational model that includes competition between monocular representations as a key factor behind stimulus rivalry. Within the context of this model, the sustained image dominance that defines stimulus rivalry is better understood as alternating eye dominance that keeps pace with the eye swaps.
We performed two psychophysical experiments that both exploit visual adaptation as an inferential tool for probing the neural processes underlying stimulus rivalry. In experiment 1, we investigated whether perceptual suppression during stimulus rivalry dampens the effectiveness of one of the rival images at inducing a threshold elevation aftereffect (TEAE). This aftereffect, characterized by impaired detection of a pattern whose orientation and spatial frequency are similar to those of the inducing image, is widely believed to be caused by adaptation of neurons in early visual cortical areas, including V1 (6–8). Based on this belief, a number of studies have assessed the vulnerability of the TEAE to suppression induced by different visual disappearance phenomena (1), with the aim being to infer the loci of neural events underlying those phenomena (9). Accordingly, the reported weakening of the TEAE by conventional binocular-rivalry suppression implies attenuated neural responses in early cortical areas during suppression in conventional rivalry (10–12), whereas the undiminished TEAE observed following suppression produced by motion-induced blindness implicates a more central neural process in that form of suppression (13). Here, we extended this strategy to suppression associated with stimulus rivalry.
Results
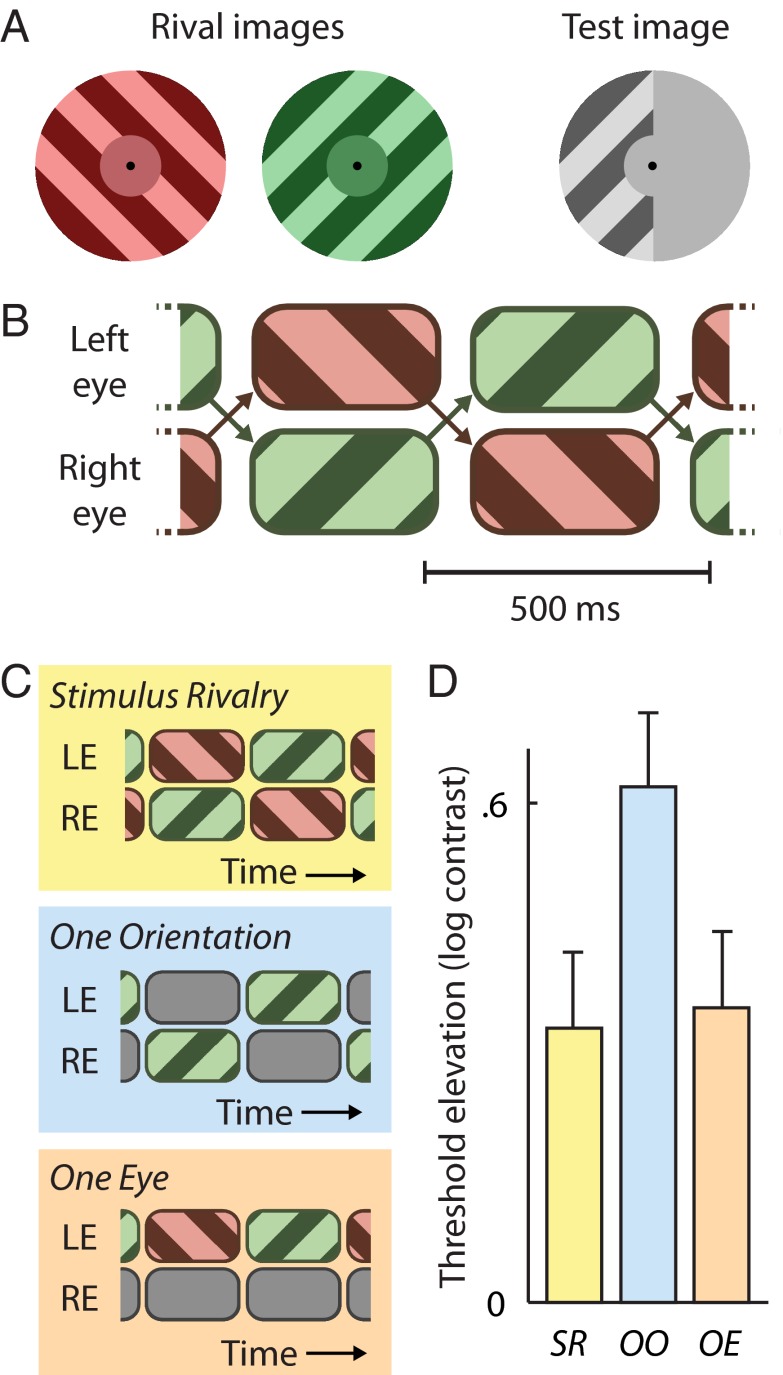
Fig. 1 A and B schematically shows our experimental design. In accordance with the standard method for generating stimulus rivalry, two rival grating patches exchanged eyes approximately three times each second, while rapidly flickering on and off at 18 Hz (a procedure commonly used to facilitate stimulus rivalry) at the same time. We verified that observers reliably experienced periods of unchanging perceptual dominance that lasted several seconds, and that therefore spanned multiple eye swaps (Methods). Following adaptation to this display, we briefly presented to one eye only a low-contrast test grating of the same orientation as one of the two images shown during stimulus rivalry. The test grating filled only one randomly chosen half of the circular display, and the observer judged which half of the display this was, with contrast varied adaptively to find the detection threshold. The magnitude of the TEAE was indexed relative to thresholds measured on nonadapted baseline trials that did not involve rivalry.
Fig. 1.
Experiment 1. Effect of stimulus-rivalry suppression on formation of the TEAE. (A) Schematic of colored rival gratings (Left) and achromatic test grating (Right). The test grating matched one of the rival gratings in orientation and spatial frequency but covered only one-half of the stimulus area, either left or right. The observer’s responses (left vs. right) following each test presentation guided an adaptive procedure to an estimate of the observer’s contrast detection threshold. (B) Stimulus-rivalry presentation sequence. (C) Detection thresholds following stimulus rivalry (yellow) were compared with thresholds following exposure to two other display sequences, both constructed by omitting part of the stimulus-rivalry sequence. In the One Orientation condition (blue), exposure to the adapting orientation matches that produced in the Stimulus Rivalry condition, but in the One Eye condition (orange), exposure to the adapting orientation is reduced relative to stimulus rivalry. LE, left eye; RE, right eye. (D) TEAE (y axis) in the Stimulus Rivalry (SR) condition is significantly weaker than that in the One Orientation (OO) condition but matches the TEAE in the One Eye (OE) condition. Both results indicate that stimulus-rivalry suppression attenuates contrast adaptation.
To investigate whether TEAE formation was affected by stimulus-rivalry suppression, we compared the “Stimulus Rivalry” condition described above with a “One Orientation” condition, during which we removed part of the stimulus-rivalry sequence (Fig. 1C). Specifically, we replaced the rival image whose orientation did not match that of the test image with a blank display, thus leaving only the grating whose orientation did match, flickering and exchanging eyes periodically. Because of the TEAE’s tight orientation tuning (6), this second grating is the one expected to cause a TEAE in our experiment. Importantly, although retinal exposure to this adapting grating is identical in both conditions, this grating is perceptually suppressed much of the time during the Stimulus Rivalry condition but not during the One Orientation condition. Thus, if stimulus-rivalry suppression leaves TEAE formation unaffected, we expect both conditions to produce TEAEs of similar magnitude, whereas if stimulus-rivalry suppression attenuates TEAE formation, we expect a weaker TEAE following stimulus-rivalry adaptation.
In a second comparison condition, called the “One Eye” condition (Fig. 1C), we again presented only part of the stimulus-rivalry display, this time leaving the part presented to the eye that would receive the subsequent test grating but presenting a blank display to the other eye. Monocular test images such as ours show the strongest TEAEs after presenting an adapter to the same eye, but a TEAE can still be observed, although notably reduced, after adapting the opposite eye, indicating a mix of monocular and binocular contributions to the TEAE (6, 14). This means that any TEAE measured in our Stimulus Rivalry condition likely reflects a compound effect of same-eye and opposite-eye exposure to the adapting grating. In the One Eye condition, therefore, one would predict a comparatively weaker TEAE, because opposite-eye exposure is abolished in that condition but same-eye exposure remains unchanged. However, this prediction changes if one supposes that perceptual suppression in the Stimulus Rivalry condition attenuates TEAE buildup: This attenuation would counteract the effect of added retinal exposure during stimulus rivalry; thus, the resulting TEAE may not be stronger than in the One Eye condition. In other words, if the One Eye condition does not yield a smaller TEAE than the Stimulus Rivalry condition, this constitutes complementary evidence that perceptual suppression diminishes TEAE formation.
Data from 10 observers (Fig. 1D) show a significant effect of condition on TEAE strength [one-way, repeated-measures ANOVA: F(2,18) = 6.9, P < 0.01]. Indeed, the TEAE was stronger following one-orientation adaptation than following stimulus-rivalry adaptation [two-tailed paired t test: t(9) = 2.8, P = 0.02]. Given that retinal exposure to the adapting grating was identical in both conditions, whereas perceptual suppression occurred only during stimulus rivalry, this difference points to reduced TEAE formation during perceptual suppression of the adapting grating. This conclusion is further supported by a comparison between the Stimulus Rivalry and One Eye conditions, which yields no TEAE difference despite the fact that the more extensive retinal exposure in the Stimulus Rivalry condition would predict an enhanced TEAE there [two-tailed paired t test: t(9) = 0.49, P = 0.64].
This pattern of results implies that suppression associated with stimulus rivalry attenuates the buildup of contrast adaptation generally thought to transpire within early cortical areas, a surprising outcome in light of the prevailing view that stimulus rivalry arises within higher tier areas of the visual hierarchy. This outcome led us to wonder whether part of the attenuation in contrast adaptation occurring during stimulus rivalry can be traced back to neurons predominantly responsive to input from one eye rather than responding equally to both. The involvement of these so-called “monocular neurons” would be consistent with the knowledge that neurons of this type are common in early cortical areas, but the involvement of monocular neurons would undermine the common assumption that stimulus rivalry relies on competition at binocular processing stages exclusively.
In the case of conventional binocular rivalry, testing whether suppression reduces adaptation at monocular processing levels would typically involve measuring differential adaptation between an eye that has received a dominant adapter during rivalry and an eye that has received a suppressed adapter. In the case of stimulus rivalry, however, this approach is precluded by the fact that the perceptually dominant image is being continually swapped between eyes (Fig. 2A, Upper). As a result, the total amount of time a given eye receives the dominant image is almost equal for both eyes across any considerable viewing period; hence, no differential monocular adaptation can be measured afterward, regardless of any involvement of monocular neurons. Fig. 2A (Lower) shows a way around this limitation. Before each stimulus-rivalry viewing period (lasting 60 s), we designated an eye that should receive the dominant image for a larger proportion of the time during that viewing period. This was then achieved by letting each change in perceptual dominance that the observer reported during this viewing period instantly trigger a change in the rhythm of the eye-exchange sequence, such that every subsequent cycle in this sequence consisted of a long interval (427 ms) during which the designated eye viewed the dominant image and a short interval (213 ms) during which it viewed the suppressed image. This asymmetrical rhythm continued until another perceptual change was reported; at that point, the rhythm was adjusted based on the new dominant image. Fig. 2B further illustrates this manipulation.
Fig. 2.
Experiment 2. Effect of stimulus-rivalry suppression on adaptation of monocular neurons. (A) During ordinary stimulus rivalry, perceptual dominance is distributed approximately equally across the two eyes (Upper), precluding differential accumulation of monocular adaptation in the processing streams of the left and right eyes. A designated eye can be made to receive the dominant image for a larger proportion of the time by online adjustment of the eye-exchange cycle in response to perceptual reports (Lower), thus allowing the possibility of differential accumulation of monocular adaptation. (B) During ordinary stimulus rivalry (Top row), the eye-exchange cycle was symmetrical, but during biased stimulus rivalry (Middle and Bottom rows), each period of the exchange cycle consisted of a short interval viewing one eye/orientation combination and a long interval viewing the opposite eye/orientation combination. By selecting whether the strategy illustrated in the Middle row or in the Bottom row was used, it was possible to force the dominant stimulus to be imaged within a designated eye for a relatively large proportion of the total viewing duration of stimulus rivalry. (C) Extended periods of stimulus rivalry were followed by binocular rivalry between the same two rival images. Because binocular-rivalry dominance durations are influenced by monocular adaptation, they can reveal buildup of differential monocular adaptation during biased stimulus rivalry.
This biasing maneuver did not affect stimulus rivalry: Dominance periods still spanned many eye swaps (mean dominance duration was 2.2 s, the same as during ordinary stimulus rivalry, produced by symmetrically timed swaps). Critically, the method of yoking the rhythm of the eye-exchange cycle to perceptual dominance reports did successfully route the dominant image to the designated eye. During viewing periods when the left eye was the designated eye, the dominant image spent an estimated 63% of the time in the left eye vs. 37% in the right eye, and when the right eye was the designated eye, these percentages were 40% and 60%, respectively (Methods). For comparison, the percentages were 50% and 50%, respectively, during ordinary stimulus rivalry. Thus, if the buildup of adaptation of monocular neurons is indeed attenuated during stimulus-rivalry suppression, this biasing maneuver should result in differential accumulation of adaptation in the two eyes’ monocular streams.
To test for such differential adaptation, each extended viewing period was followed by a period of conventional binocular rivalry using the same two grating images (Fig. 2C), with their eye configuration counterbalanced across viewing periods. It is well established that dominance during conventional binocular rivalry is temporarily biased away from an eye that has been exposed to its rival image before the onset of rivalry, implying that binocular-rivalry dominance durations are shortened by accumulated adaptation (15–17). In one typical study, a minute of adaptation to one of the rival images in isolation temporarily changed a balanced cycle, with equal predominance for both eyes, to a cycle in which the nonadapted eye dominated about 70% of the time (15). Such results render conventional rivalry well suited as a gauge of differential monocular adaptation in this experiment. If stimulus-rivalry suppression does diminish the accumulation of monocular adaptation, we expect more binocular-rivalry dominance for the eye that received the suppressed image for a larger proportion of the time during the immediately preceding period of stimulus rivalry than for the eye that mostly received the dominant image. Compared with the work cited above, however, we do expect relatively modest effects on binocular rivalry here, given that our adapting stimulus is a rivaling display with only a slight asymmetry in eye dominance rather than an uninterrupted monocular stimulus like that used in earlier studies.
Fig. 3 shows results from all eight observers tested in this experiment. The balance between left-eye dominance and right-eye dominance during a period of binocular-rivalry viewing was significantly affected by the preceding stimulus-rivalry period [Fig. 3A; one-way, repeated-measures ANOVA: F(2,14) = 8.8, P < 0.01]. Specifically, the left eye dominated for a smaller proportion of the time following stimulus-rivalry periods with more left-eye dominance (Fig. 3A, white) than following stimulus-rivalry periods with more right-eye dominance [Fig. 3A, black; two-tailed paired t test: t(7) = 3.4, P = 0.01], with the proportion following neutral stimulus rivalry (Fig. 3A, gray) falling between these two extremes. (The proportions of right-eye dominance equal 1 minus the left-eye proportions shown in this plot.) An analysis of the durations of individual dominance periods, rather than of predominance ratio, showed the same effect (Fig. 3B). Binocular-rivalry dominance durations differed significantly between an eye that had previously enjoyed more stimulus-rivalry dominance (Fig. 3B, white), an eye that had enjoyed less (Fig. 3B, black), and following neutral stimulus rivalry (Fig. 3B, gray) [one-way, repeated-measures ANOVA on normalized durations: F(2,14) = 3.7, P = 0.05; Methods]. Indeed, an eye’s dominance durations were significantly longer if the eye had enjoyed less stimulus-rivalry dominance than if it had enjoyed more [two-tailed paired t test: t(7) = 4.7, P < 0.01].
Fig. 3.
Results of experiment 2. Perceptual dominance of the eye that received the dominant image for a larger proportion of the time during stimulus rivalry (SR) was reduced during subsequent binocular rivalry (BR), both in overall ratio (A) and in individual durations (B), indicating that monocular adaptation associated with this eye was elevated, compared with the eye that mostly received the suppressed image during stimulus rivalry. (C) This difference in predominance between the two eyes wore off over the course of binocular-rivalry viewing, consistent with an adaptation-based account. (D) Ocular imbalance in binocular-rivalry dominance correlated on an observer-to-observer basis with the magnitude of the imposed imbalance in ocular dominance during stimulus rivalry.
These results clearly show that binocular-rivalry dominance in our experiment is biased away from the eye that previously dominated more during stimulus rivalry, but is this the result of monocular adaptation? If so, we expect the biasing effect to wear off over the course of binocular-rivalry viewing, just as it does after adapting to a nonrivaling stimulus (15, 18, 19). For Fig. 3C, we separated the six dominance periods that comprised each interval of binocular-rivalry viewing into three consecutive pairs, for each pair calculating the proportion of dominance time for the eye that dominated more during the preceding stimulus-rivalry period (i.e., the “designated eye”). Consistent with an adaptation-based account, this proportion started off below 0.5 for the first percept pair [two-tailed t test: t(7) = 4.08, P < 0.01] and then rose significantly across consecutive pairs [we obtained each observer’s slope of proportion vs. pair using linear regression and then compared this set of slopes with 0; two-tailed t test: t(7) = 2.55, P < 0.05].
In a final analysis, we investigated whether the magnitude of the imbalance in ocular dominance imposed during stimulus rivalry predicted the magnitude of the imbalance observed during subsequent binocular rivalry, as expected if the two were causally related. For each observer, we calculated an estimate of the proportion of time that the designated eye ended up receiving the dominant image during stimulus rivalry, an estimate that varied between observers due to differences in their perceptual dynamics during stimulus rivalry (Methods). Indeed, comparing across observers, this estimate correlates significantly with the imbalance observed during binocular rivalry (Fig. 3D; Pearson’s correlation). This again indicates that biasing stimulus-rivalry dominance in favor of one eye preferentially biases subsequent binocular-rivalry dominance in favor of the other eye, further supporting the notion that suppression during stimulus rivalry attenuates monocular adaptation.
The results of Fig. 3 cannot be attributed to eye-independent, image-based effects. Although our manipulation successfully influenced which eye dominated most during stimulus rivalry, it did not cause one particular rival image to remain dominant for more time than the other [two-tailed paired t test comparing image predominance ratio during stimulus-rivalry periods involving mostly left-eye dominance vs. those involving mostly right-eye dominance: t(7) = 0.26, P = 0.8; 52% red predominance and 48% green predominance in both cases]. Moreover, even if our manipulation did cause an asymmetry in image dominance, this would not lead to any systematic patterns in Fig. 3 because we counterbalanced which eye viewed which image during binocular rivalry. Neither can our results be attributed to an imbalance on the input side, with one image being presented more to one eye than to the other due to our biasing maneuver. In other words, although our manipulation involved routing the dominant image via a designated eye, this eye still received both images equally because both images took turns being dominant [two-tailed paired t test on the proportion of time the red image spent in the left eye during stimulus-rivalry periods with mainly left-eye dominance vs. periods with mainly right-eye dominance: t(7) = 1.5, P = 0.18; 51% and 49%, respectively]. As an extra verification that input differences cannot explain our results, we repeated this experiment with four of the original observers, this time not adjusting the eye swap rhythm to incoming perceptual reports but, instead, replaying the sequences of eye swaps previously used in the main experiment. The resulting presentation sequences were thus identical to those of the main experiment, including any imbalances that might exist, but were no longer yoked to perceptual state. All effects shown in Fig. 3 now disappeared (Methods), confirming that these specifically depend on the contingency between eye configuration and stimulus-rivalry dominance state.
Considered together, these results conclusively show that stimulus-rivalry suppression is accompanied by reduced buildup of adaptation within an eye’s monocular stream and, by implication, within monocular neurons, subsequently producing a temporary bias in binocular-rivalry dominance in favor of the eye that enjoyed less dominance during stimulus rivalry. Importantly, this outcome also implies that the monocular neurons whose adaptation is diminished during stimulus-rivalry suppression overlap with those involved in binocular rivalry, because without overlap, there would be no carryover from stimulus rivalry to binocular rivalry.
Discussion
Stimulus rivalry’s characteristic maintenance of image dominance across multiple eye swaps has promoted the widely held view that stimulus rivalry arises from competition at higher, eye-independent processing stages. In the most concrete implementation of that view, Wilson (3) extended a standard binocular-rivalry model, centered on eye-specific, orientation-tuned neural populations, with a second pair of orientation-tuned populations that respond equally to input from both eyes, located after the eye-specific populations in the processing stream (Fig. 4A, blue shows the extension). In this extended model, conventional binocular rivalry arises from competition between the eye-specific populations, but the ongoing on/off stimulus flicker that is part of the stimulus-rivalry presentation sequence effectively shuts down this between-eye competition. Consequently, both eyes’ signals proceed to the eye-independent stage, where rivalry is then resolved. Although explaining maintained image dominance across eye swaps, this model predicts that neural responses during stimulus rivalry should not be modulated at monocular stages. Our present evidence of just such modulation led us to consider whether stimulus rivalry can be accounted for without assuming relocation of competition to a binocular level.
Fig. 4.
Accommodating the present findings in models of stimulus rivalry. (A) This schematic shows an existing stimulus-rivalry model developed by Wilson (3). The first stage (black) is a standard binocular-rivalry model with orientation-tuned monocular populations (striped circles) that interact via mutual inhibition (connecting lines with dots at the ends), and to that is added a second stage consisting of binocular, eye-independent populations (blue), again connected via mutual inhibition. In this hybrid model, inhibition at the monocular stage is silenced during stimulus rivalry, leaving only competition at the eye-independent stage. This exclusion of between-eye inhibition explains stimulus rivalry’s apparent immunity to eye swaps but does not fit well with the present evidence that stimulus rivalry modulates monocular neurons. (B) Starting from the same binocular-rivalry model, we extended it with within-eye cross-orientation inhibition (purple) and between-eye iso-orientation inhibition (orange). This model variant still explains stimulus rivalry, and it is consistent with our findings because it does not exclude competition at a monocular stage. Eqs. 1–3 in Methods define the components and interactions comprising this model, and those can be summarized as follows. Activity levels in the orientation-tuned populations are indicated by variable names Eorientation,eye (e.g., E45,L for the population sensitive to right-tilted gratings presented to the left eye). Each of these populations receives three kinds of inhibition, indicated by Iorientation,eye (e.g., I−45,L for inhibition arising from the population sensitive to left-tilted gratings presented to the left eye), corresponding to this figure’s black, purple, and orange lines. Each of these populations furthermore exhibits self-adaptation, indicated by Horientation,eye. (C) Model of B exhibits stimulus rivalry’s characteristic periods of stimulus dominance that span several eye swaps, showing that relocation of competition to a binocular stage is not necessary to explain stimulus rivalry. a.u., arbitary units.
To pursue an answer to that question, we turned to the standard binocular-rivalry model that Wilson (3) used as his starting point (Fig. 4B, black part of schematic). Without any extensions, this model neither favors nor disfavors maintained image dominance across eye swaps because it includes no interaction between the image pair presented before an eye swap and the pair with opposite eye configuration presented afterward (i.e., Fig. 4B shows no black connections between the outermost population pair and the innermost population pair; cf. ref. 20). We changed this by including what can be construed as within-eye response normalization (21): inhibition between monocular populations associated with the same eye but differing in orientation preference (Fig. 4B, purple). The addition of this component is consistent with empirical evidence for this kind of inhibition (22–26), and it provides a mechanism for stimulus rivalry that does not exclude monocular neurons. Specifically, when an image is presented to a given eye after an eye swap, its response is now diminished by the within-eye inhibitory response that has developed during the period leading up to the eye swap, and because this inhibition will have reached a higher level if the eye was dominant before the eye swap, this results in a tendency for dominance to switch eyes at the moment of the eye swap. In other words, stimulus rivalry in this model is better understood as alternating eye dominance across eye swaps than as maintained image dominance, but the resulting perceptual experience is the same.
One important feature of stimulus rivalry is its dependence on rapid stimulus flicker. Without flicker, swapping the images between eyes causes perceptual dominance to switch instantly to the other image, thus staying with the same eye (27, 28). To capture this flicker dependence in our model, we also included inhibitory connections between opposite-eye populations tuned to the same orientation (Fig. 4B, orange). Such between-eye iso-orientation suppression is indicated by empirical work (25, 29), and it promotes maintained eye dominance across eye swaps in a fashion fully analogous to the one described above (i.e., via an inhibitory interaction between responses to the image that dominated before an eye swap and responses to the same image presented to the other eye afterward). Because this latter type of inhibition in our model is stronger than the within-eye inhibition discussed in the previous paragraph, it is usually able to tip the balance toward maintained eye dominance. However, because it also has a faster decay, its strength quickly wanes during the brief stimulus-off period that occurs right before a flickering stimulus swaps eyes, resulting in a net tendency for maintained image dominance there. In other words, whereas the Wilson model (3) requires ongoing flicker to produce stimulus rivalry, our model generates stimulus rivalry even if ongoing flicker is replaced by a single blank interval just before the eye swap. This is consistent with experimental work showing that stimulus rivalry still occurs without flicker as long as this last blank period is present (30, 31).
There is ample empirical evidence for the two inhibitory components added to our extended model, but is it also plausible that between-eye iso-orientation suppression is stronger in most situations, whereas within-eye suppression only becomes the dominant factor when the stimulus is interrupted, as is the case in our conceptualization? In our model, this situation arises through a difference between the two components in both their maximum strength and their time scale of decay during stimulus absence. Although it is not straightforward to arrive at estimates of these characteristics from empirical work, there is some evidence that between-eye iso-orientation suppression is indeed the stronger of the two (25, 32). Regarding the crossover in strength for interrupted stimuli, an intriguing possibility is suggested by work that points to a type of within-eye inhibition that is strongest right after stimulus onset and for rapidly changing stimuli but becomes weaker during sustained stimulation (22, 33).
This revised model provides evidence that stimulus rivalry can be explained without assuming relocation of competition to a purely binocular level. Combined with our experimental evidence, this points to the involvement in stimulus rivalry of early, monocular processing stages. However, these results do not argue against an additional role of binocular neurons, and eye-dependent and eye-independent mechanisms may jointly determine stimulus rivalry (34). Indeed, in the case of conventional binocular rivalry, there is solid evidence for both kinds of contributions (32, 35–38), and binocular rivalry is widely viewed as the culmination of neural events distributed over multiple stages of processing comprising feedforward and feedback connections (4). What the present results indicate is that during stimulus rivalry, just like during binocular rivalry, those stages include very early ones in which interocular competition transpires.
According to our conceptualization, neural events associated with stimulus rivalry engage the same mechanisms mediating competitive interactions underlying conventional binocular rivalry. In this respect, our results provide a tidier, parsimonious view of rivalry created by dissimilar stimulation of the two eyes, whether that stimulation involves eye swapping or not. Our results also provide a ready account of why dominance phases of stimulus rivalry and binocular rivalry become entrained when the two forms of stimulation appear at spatially adjacent regions of the visual field (39) and why the two forms of rivalry exhibit the same dependence on contrast (2). Still, there are ways in which the two differ, including their dependence on stimulus size (40) and on spatial and temporal frequency (2, 28, 31). The reasons for those differences have yet to be worked out, but one possibility relates the two forms of rivalry to preferential activation of the magno- and parvocellular pathways formed at the earliest stages of visual processing (31). Another question for future research is how our results would generalize to stimulus rivalry between complex images of objects or natural scenes. We purposefully used gratings here because those are the patterns traditionally used in work on stimulus rivalry (2, 28) and because gratings are well tailored for inducing TEAEs (6, 10). However, it is natural to wonder whether comparable results would be found using complex images that selectively activate neurons in high-tier visual areas.
To end on a cautionary note, we often read (and ourselves have written) that competition evidenced during rivalry, stimulus rivalry included, reveals the operation of selection processes associated with consciousness (41). Although not overruling this conviction, the present results emphasize that important neural determinants of the competition involved in rivalry transpire at very early stages of visual processing whose activity is unlikely to be sufficient for conscious experience (42, 43). This realization, in turn, suggests discretion when inferring strong links between neural concomitants of rivalry dominance and those of consciousness.
Methods
Experiment 1.
Stimuli.
Adapting stimuli were a red and green sinusoidal grating (1.5 cycles per degree) presented within an annular aperture (inner diameter of 1.0°, outer diameter of 5.5°) and oriented at −45° and 45° from vertical, respectively. They swapped eyes every 320 ms and flickered on/off at 18.75 Hz. In addition, each grating slowly drifted at 2.5° per second in one of the two directions perpendicular to the grating’s orientation (i.e., diagonally upward or diagonally downward) but always with the same vertical motion component in both eyes. This drift precluded the buildup of afterimages, and pilot experiments suggested it promoted stimulus rivalry. Observers were instructed to fixate a central mark present throughout the experiment. To aid fusion, the gratings were embedded within an area of fusible random pixel noise (circular inner diameter of 6°, square outer diameter of 6°), and fusible lines extended outward vertically and horizontally from the centers of the outer edges of this area (0.092° width). Mean gray (see below) filled the screen outside of the fusion aids and between the fusion texture and the stimuli, as well as filling the stimulus area during the off-periods of the flicker, but the center area between the fixation mark and the stimulus annulus was always black (0 cd/m2). The test stimulus was a grayscale copy of one of the adapting stimuli, but with either its left half or right half erased. In addition, the contrast of the test stimulus smoothly decreased toward its outer edges following a Gaussian profile (σ = 0.275°).
Participants and procedure.
Ten observers (four female, mean age of 34.5 y, six unaware of the purpose of the experiment) participated in this experiment. During testing, an observer was seated in front of a computer screen with a head/chin rest stabilizing the observer’s head position immediately in front of a mirror stereoscope used for dichoptic presentation. All experiments complied with procedures approved by the Institutional Review Board Office at Seoul National University, including written consent for observer participation.
Before the experiment, each observer performed heterochromatic flicker photometry to find the green luminance perceptually equivalent to maximum red luminance (16.12 cd/m2). The mean luminance of the red grating throughout the experiment was half of maximum, and that of the green grating was half of the equivalent green luminance. The mean luminance of the test grating, as well as the luminance of the background, was the average of these two values.
Before the experiment, we also tested whether each observer reliably experienced slow and irregular perceptual alternations during stimulus rivalry, rather than fast and regular alternations entrained to the eye swaps. In 5-min sessions, perceptual tracking data were collected during 15-s trials of either conventional binocular rivalry, stimulus rivalry, or binocularly fused presentation of the red and green grating alternating on the screen at the same rate as stimulus-rivalry eye swaps (i.e., yoked fast, regular alternations). These trial types were randomly interleaved. Observers had one key to indicate the perception of each grating and a third key to indicate fast, regular perceptual alternations. All observers reliably reported slow, irregular alternations during binocular rivalry and fast, regular alternations during yoked, fusible presentation. As the criterion for good stimulus rivalry, we required slow, irregular alternations being reported over 90% of the time during stimulus-rivalry trials. Two of our original 12 observers were excluded on the basis of this criterion.
In experiment 1, the three test conditions (Stimulus Rivalry, One Orientation, and One Eye; Fig. 1C) were blocked in separate sessions. Each session started with 30 s of mean-luminance adaptation, followed by consecutive 7-s periods of grating adaptation. Grating motion direction reversed between consecutive 7-s adaptation periods to avoid motion aftereffects. The first eight adaptation periods of each session were separated only by a 0.4-s blank (initial adaptation buildup); all periods after that (top-up adaptation) were separated by a 0.4-s blank as well as a spatial two-alternative, forced-choice detection task, where observers determined which side of the display contained the test image. The test stimulus was ramped up and back down linearly across seven monitor frames in total (at 75 Hz), with peak contrast adjusted adaptively in four randomly interleaved QUEST staircases (44). The moment of test grating presentation was indicated by a middle-frequency auditory tone (750 Hz, 100 ms), and feedback was also given using two different auditory tones right after observers responded, without any imposed response window (400 Hz, 300 ms for incorrect; 1,000 Hz, 100 ms for correct). The eye and orientation of the test grating were fixed for each observer and counterbalanced across observers, and the same eye and orientation were used for the One Eye condition and One Orientation condition, respectively. Detection thresholds were estimated by averaging the output 75% correct thresholds from all four staircases within a session. Sessions were separated by at least 5 min to eliminate across-session adaptation effects.
To avoid saturation of the TEAE (10), experiment 1 was preceded by a pilot experiment in which we determined detection thresholds at a range of adapting contrast levels using the One Orientation adapter stimulus (Fig. 1C). Based on the resulting detection threshold curve of each observer, we selected an adapter contrast that was not only high enough to yield a robust TEAE but sufficiently low for the TEAE not to be saturated. This contrast was used for the ensuing experiment with three adaptation conditions. In addition, we reduced the duration of the adaptation periods to 3.5 s for three observers who showed saturation at very low contrasts in this threshold curve. Threshold elevation in Fig. 1D was defined relative to baseline threshold at 0% adapter contrast estimated from a Naka–Rushton function fitted to this curve.
Data analysis.
We analyzed the TEAEs using a one-way, repeated-measures ANOVA with post hoc paired t tests as planned comparisons between the Stimulus Rivalry and One Orientation conditions and between the Stimulus Rivalry and One Eye conditions. All P values of the post hoc t tests survived a Bonferroni correction for multiple (planned) comparisons.
Experiment 2.
Stimuli.
The spatial properties and contrast of the stimulus-rivalry stimuli were the same as during experiment 1. For conventional binocular rivalry, we used the same gratings, except that contrast was reduced by half in an attempt to obtain the same effective contrast in the absence of flicker. Eye configuration during binocular rivalry was randomized within a session and counterbalanced within each adaptation condition. The temporal stimulus characteristics during ordinary stimulus rivalry were the same as in experiment 1; however, during biased stimulus rivalry, each perceptual report determined the durations that applied to the two eye configurations until the next perceptual report (see below). Eye swaps always occurred at the end of a stimulus-off phase of the flicker sequence, and as soon as the designated duration for the current eye configuration had passed.
During biased stimulus rivalry, a single period of asymmetrical eye swapping consisted of 427 ms of viewing one eye configuration and 213 ms of viewing the other. This timing keeps the duration of a single period the same as in the unbiased condition (640 ms) while imposing a strongly asymmetrical duty cycle of 2:1. We have no reason to believe that this particular timing is essential for our results, but stimulus rivalry will likely break up if the timing of the eye swaps deviates much further from what is standard (28, 30).
Participants and procedure.
Eight participants from experiment 1 (four female, mean age of 34.6 y, four unaware of the purpose of the experiment) also participated in experiment 2. Sessions were organized into 16 trials, each composed of 60 s of stimulus rivalry, a 1-s blank screen, six perceptual reversals of binocular-rivalry tracking, and a deadaptation period. During this latter period, a yellow disk alternately covered the stimulus areas of the left eye and right eye for 0.5 s (with the other eye receiving a mean gray field). This initially resulted in a perceived periodic color change caused by the two eyes having been adapted to different colors during binocular rivalry, and observers terminated the deadaptation period, thereby starting the next trial, when this color change was no longer perceived, indicating recovery from color adaptation.
Four different conditions were administered during different 60-s stimulus-rivalry periods: In addition to ordinary stimulus rivalry and the two bias directions, we had a “fused” condition in which a fusible untextured disk covered the stimulus area, flickering at 18.75 Hz and alternating between average green and average red every 320 ms. This condition was included to verify that adaptation to ordinary stimulus rivalry, by itself, does not importantly change binocular-rivalry dynamics, relative to adaptation to a nonrivalry display. Indeed, the balance in eye dominance during binocular rivalry was similar following ordinary stimulus rivalry and following this fused stimulus, with both values being intermediate between the values observed following the two biased conditions. Adaptation to ordinary stimulus rivalry did cause a longer mean binocular-rivalry dominance duration of 2.5 s, relative to 2.0 s following adaptation to the fused stimulus. All four conditions were randomized across the 16 trials of a session, with the limitation that each set of 4 consecutive trials consisted of one occurrence of each condition, with ordinary stimulus rivalry and the fused condition presented first and third (in random order) and the two biased conditions presented second and fourth (in random order). In this way, opposite bias conditions never occurred in direct succession, preventing counteracting adaptation on consecutive trials. Grating drift direction (identical for stimulus rivalry and binocular rivalry within a trial) remained the same during such a block of 4 trials and alternated between such blocks.
During stimulus rivalry, observers had two buttons to indicate the two grating percepts and a third button to report fast, regular alternations. They reported this third option 2.5% of the time, on average, during ordinary stimulus rivalry vs. 2.1% of the time during biased stimulus rivalry [not significantly different; two-sided paired t test: t(7) = 1.21, P = 0.26]. As soon as fast, regular alternations were reported during biased stimulus rivalry, this reset the eye swap sequence to the ordinary, symmetrical rhythm until a different percept was reported. During binocular rivalry, observers again had two buttons for the two grating percepts, as well as a third button for perceptual mixtures. They reported such mixtures, on average, 6.3% of the viewing time following ordinary stimulus rivalry vs. 6.4% of the time following biased stimulus rivalry [not significantly different; two-sided paired t test: t(7) = 0.10, P = 0.92].
Data analysis.
For Fig. 3 A and B, we tested with one-way, repeated-measures ANOVAs and planned paired t tests between conditions of opposite stimulus-rivalry bias. Before comparing dominance durations, we normalized within observers, but across conditions, to account for duration differences that exist between eyes, between orientations, and between positions of the dominance period within an alternation sequence (e.g., the first period tended to be shorter than the rest). To this end, we divided all durations by a mean that was calculated separately for each combination of eye, image, and rank number of the dominance period. For analyses in Fig. 3 involving predominance ratios, those ratios were calculated after discarding periods of mixed perception so that the relative predominances of the left eye and right eye, or of the designated eye and nondesignated eye, added up to 1. Equivalent results were obtained when periods of mixed perception were not excluded from the analyses (Tables S1–S3).
To ensure that our results were not due to input differences between adaptation conditions, rather than to percept-contingent differences, four observers participated in a “replay” condition in which we repeated the exact same stimulus sequences previously presented during the main experiment 2. The observed biasing effects of adaptation on binocular rivalry should now disappear because the association between perceptual reports and the eye-exchange cycle is no longer present. Indeed, predominance ratios no longer showed the expected effect of adaptation condition [instead, showing an effect in the opposite direction; F(2,6) = 7.8, P = 0.02], although the main experiment 2 did show the expected effect even when using only data from these four observers [F(2,6) = 8.6, P = 0.02]. Likewise, percept durations showed no effect during replay [F(2,6) = 0.98, P = 0.4], but this is less telling because this effect also fell short of significance in the main experiment when including only data from these four observers [F(2,6) = 3.27, P = 0.1].
For Fig. 3D, we indexed the dominance imbalance during stimulus rivalry in terms of the estimated proportion of time that the dominant image spent in the designated eye. This estimate involves two assumptions. First, there is a certain motor delay between an actual change in perceptual dominance and the resulting key press. We assumed a reaction time of 400 ms (45), but the biasing maneuver stays effective at routing the dominant image to a given eye at considerably longer response times, as long as the motor delay is short relative to perceptual dominance durations. The estimate also involves an assumption regarding the infrequent periods where the observer reported fast and predictable perceptual alternations between the two gratings: periods of maintained eye dominance. Because we cannot tell from our data which of the eyes is dominant during such a period, we assumed that it was either eye equally often. One might suspect that, instead, the nondesignated eye would experience more periods of maintained eye dominance due to its lower adaptation state. Interestingly, if we follow this scenario and assume that all periods of fast and predictable alternations concern eye dominance of the nondesignated eye, the correlation of Fig. 3D improves even further (r = 0.90, P < 0.01).
Model Simulations.
For our model simulations, we started with the basic system of differential equations from Wilson (3), to which we added two additional components: inhibition between same-eye populations tuned to orthogonal orientations and inhibition between opposite-eye populations tuned to the same orientation.
By adding these additional inhibitory connections to the model of Wilson (3), the firing rate of the neural population responding to the right-tilted grating presented to the left eye is described by
 |
Here, E45,L is the firing rate of a population responding to a grating tilted 45° from vertical, presented to the left eye. V45,L is the input strength to that same population, set to 10 when the stimulus is present and to 0 when it is not. H45,L is the adaptation level of this population. Variables of the form IA,B indicate inhibition that this population receives from populations tuned to orientation A presented to eye B. For instance, I−45,R originates from the population tuned to left-tilted gratings presented to the right eye. These inhibition components are scaled by gxx, g=x, and gx=, three constants that apply, respectively, to inhibition acting across orientations and eyes, within orientations but across eyes, and across orientations but within eyes. Their respective values are 0.90, 0.55, and 0.25. τ is a time constant with a value of 11 ms and [C]+ is a threshold nonlinearity that evaluates to C for C > 0 and to 0 otherwise. The equations for the three remaining excitatory populations are formed analogously.
The evolution of the inhibition components is described as in the original Wilson model (3). For all inhibition components, the general form is:
 |
where I is inhibition originating from an excitatory population that responds to gratings of a given orientation and eye of origin, and E is the associated excitatory response to such gratings. Time constant τI has a different value for the three kinds of inhibition, being 11 ms for cross-orientation inhibition acting across eyes, 4 ms for iso-orientation inhibition acting across eyes, and 26 ms for cross-orientation inhibition acting within eyes.
Finally, adaptation of the excitatory populations is also described as in the original model, with the general form being:
 |
Here, H is the adaptation state of an excitatory population that responds to gratings of a given orientation and eye of origin, and E is the associated excitatory response to such gratings. Then, h is the adaptation gain, which has a value of 2.4. The adaptation time constant τH has a value of 4 s. In all simulations, we used an explicit Euler integration method with a step size of 0.25 ms.
How do these equations fit into the diagram of Fig. 4B? Note first that this diagram depicts mutual inhibition by reciprocal connections (black, orange, and purple lines) between two populations of neurons. Eq. 1 expands this circuitry by explicitly defining inhibitory components as separate sets of units (whose activity is termed I), each of which can be construed as inhibitory interneurons lying between two populations of excitatory neurons, with the time constants of those inhibitory units being defined by Eq. 2. The self-adaptation within the populations of excitatory units, specified in Eq. 3, is not depicted in the diagram of Fig. 4B.
Supplementary Material
Acknowledgments
We thank André Noest and Tomas Knapen for helpful discussions during the early stages of this project; and Nikos Logothetis, Robert O’Shea, George Sperling, and Hugh Wilson for comments on an earlier draft of this paper. J.B. is supported by a Veni grant from the Netherlands Organization for Scientific Research. H.S., S.-H.L, and R.B. are supported by the World Class University program through the National Research Foundation of Korea funded by the Ministry of Education, Science, and Technology (Grant R31-10089), and R.B. is supported by the National Institutes of Health (Grant P30-EY008126).
Footnotes
The authors declare no conflict of interest.
See QnAS on page 8320.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1305393110/-/DCSupplemental.
References
- 1.Blake R, Logothetis N. Visual competition. Nat Rev Neurosci. 2002;3(1):13–21. doi: 10.1038/nrn701. [DOI] [PubMed] [Google Scholar]
- 2.Logothetis NK, Leopold DA, Sheinberg DL. What is rivalling during binocular rivalry? Nature. 1996;380(6575):621–624. doi: 10.1038/380621a0. [DOI] [PubMed] [Google Scholar]
- 3.Wilson HR. Computational evidence for a rivalry hierarchy in vision. Proc Natl Acad Sci USA. 2003;100(24):14499–14503. doi: 10.1073/pnas.2333622100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tong F, Meng M, Blake R. Neural bases of binocular rivalry. Trends Cogn Sci. 2006;10(11):502–511. doi: 10.1016/j.tics.2006.09.003. [DOI] [PubMed] [Google Scholar]
- 5.Pearson J, Tadin D, Blake R. The effects of transcranial magnetic stimulation on visual rivalry. J Vis. 2007;7(7):2.1–2.11. doi: 10.1167/7.7.2. [DOI] [PubMed] [Google Scholar]
- 6.Blakemore C, Campbell FW. On the existence of neurones in the human visual system selectively sensitive to the orientation and size of retinal images. J Physiol. 1969;203(1):237–260. doi: 10.1113/jphysiol.1969.sp008862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Vautin RG, Berkley MA. Responses of single cells in cat visual cortex to prolonged stimulus movement: neural correlates of visual aftereffects. J Neurophysiol. 1977;40(5):1051–1065. doi: 10.1152/jn.1977.40.5.1051. [DOI] [PubMed] [Google Scholar]
- 8.Movshon JA, Lennie P. Pattern-selective adaptation in visual cortical neurones. Nature. 1979;278(5707):850–852. doi: 10.1038/278850a0. [DOI] [PubMed] [Google Scholar]
- 9.Blake R, He S. Visual adaptation as a tool for studying the neural correlates of conscious visual awareness. In: Clifford C, Rhodes G, editors. Fitting the Mind to the World. New York: Oxford Univ Press; 2005. pp. 281–307. [Google Scholar]
- 10.Blake R, Tadin D, Sobel KV, Raissian TA, Chong SC. Strength of early visual adaptation depends on visual awareness. Proc Natl Acad Sci USA. 2006;103(12):4783–4788. doi: 10.1073/pnas.0509634103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Yang E, Hong S-W, Blake R. Adaptation aftereffects to facial expressions suppressed from visual awareness. J Vis. 2010;10(12):24–1–13. doi: 10.1167/10.12.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Brascamp JW, van Boxtel JJ, Knapen TH, Blake R. A dissociation of attention and awareness in phase-sensitive but not phase-insensitive visual channels. J Cogn Neurosci. 2010;22(10):2326–2344. doi: 10.1162/jocn.2009.21397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Montaser-Kouhsari L, Moradi F, Zandvakili A, Esteky H. Orientation-selective adaptation during motion-induced blindness. Perception. 2004;33(2):249–254. doi: 10.1068/p5174. [DOI] [PubMed] [Google Scholar]
- 14.Blake R, Overton R, Lema-Stern S. Interocular transfer of visual aftereffects. J Exp Psychol Hum Percept Perform. 1981;7(2):367–381. doi: 10.1037//0096-1523.7.2.367. [DOI] [PubMed] [Google Scholar]
- 15.Blake R, Overton R. The site of binocular rivalry suppression. Perception. 1979;8(2):143–152. doi: 10.1068/p080143. [DOI] [PubMed] [Google Scholar]
- 16.Wade NJ, de Weert CM. Aftereffects in binocular rivalry. Perception. 1986;15(4):419–434. doi: 10.1068/p150419. [DOI] [PubMed] [Google Scholar]
- 17.Kang M-S, Blake R. What causes alternations in dominance during binocular rivalry? Atten Percept Psychophys. 2010;72(1):179–186. doi: 10.3758/APP.72.1.179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Van Boxtel J, Alais D, Van Ee R. Retinotopic and non-retinotopic stimulus encoding in binocular rivalry and the involvement of feedback. J Vis. 2008;8(5):17.1–17.10. doi: 10.1167/8.5.17. [DOI] [PubMed] [Google Scholar]
- 19.de Jong MC, Knapen T, van Ee R. Opposite influence of perceptual memory on initial and prolonged perception of sensory ambiguity. PLoS ONE. 2012;7(1):e30595. doi: 10.1371/journal.pone.0030595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Noest AJ, van Wezel RJ. Dynamics of temporally interleaved percept-choice sequences: Interaction via adaptation in shared neural populations. J Comput Neurosci. 2012;32(1):177–195. doi: 10.1007/s10827-011-0347-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Carandini M, Heeger D. Normalization as a canonical neural computation. Nat Rev Neurosci. 2011;13:1–12. doi: 10.1038/nrn3136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Baker DH, Meese TS, Summers RJ. Psychophysical evidence for two routes to suppression before binocular summation of signals in human vision. Neuroscience. 2007;146(1):435–448. doi: 10.1016/j.neuroscience.2007.01.030. [DOI] [PubMed] [Google Scholar]
- 23.Bonds AB. Role of inhibition in the specification of orientation selectivity of cells in the cat striate cortex. Vis Neurosci. 1989;2(1):41–55. doi: 10.1017/s0952523800004314. [DOI] [PubMed] [Google Scholar]
- 24.Walker GA, Ohzawa I, Freeman RD. Binocular cross-orientation suppression in the cat’s striate cortex. J Neurophysiol. 1998;79(1):227–239. doi: 10.1152/jn.1998.79.1.227. [DOI] [PubMed] [Google Scholar]
- 25.Moradi F, Heeger D. Inter-ocular contrast normalization in human visual cortex. J Vis. 2009;9(3):13.1–13.22. doi: 10.1167/9.3.13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Roeber U, Wong E, Freeman A. Cross-orientation interactions in human vision. J Vis. 2008;8(3):15.1–15.11. doi: 10.1167/8.3.15. [DOI] [PubMed] [Google Scholar]
- 27.Blake R, Westendorf DH, Overton R. What is suppressed during binocular rivalry? Perception. 1980;9(2):223–231. doi: 10.1068/p090223. [DOI] [PubMed] [Google Scholar]
- 28.Lee SH, Blake R. Rival ideas about binocular rivalry. Vision Res. 1999;39(8):1447–1454. doi: 10.1016/s0042-6989(98)00269-7. [DOI] [PubMed] [Google Scholar]
- 29.Ding J, Sperling G. A gain-control theory of binocular combination. Proc Natl Acad Sci USA. 2006;103(4):1141–1146. doi: 10.1073/pnas.0509629103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Van Boxtel J, Knapen T, Erkelens C, Van Ee R. Removal of monocular interactions equates rivalry behavior for monocular, binocular, and stimulus rivalries. J Vis. 2008b;8(15):13.1–13.17. doi: 10.1167/8.15.13. [DOI] [PubMed] [Google Scholar]
- 31.Denison RN, Silver MA. Distinct contributions of the magnocellular and parvocellular visual streams to perceptual selection. J Cogn Neurosci. 2012;24(1):246–259. doi: 10.1162/jocn_a_00121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Stuit S, Paffen C, Smagt M, Verstraten F. What is grouping during binocular rivalry? Front Hum Neurosci. 2011;5:1–9. doi: 10.3389/fnhum.2011.00117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Li B, Peterson MR, Thompson JK, Duong T, Freeman RD. Cross-orientation suppression: Monoptic and dichoptic mechanisms are different. J Neurophysiol. 2005;94(2):1645–1650. doi: 10.1152/jn.00203.2005. [DOI] [PubMed] [Google Scholar]
- 34.Bhardwaj R, O’Shea RP. Temporal analysis of image-rivalry suppression. PLoS ONE. 2012;7(9):e45407. doi: 10.1371/journal.pone.0045407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pearson J, Clifford CG. Determinants of visual awareness following interruptions during rivalry. J Vis. 2004;4(3):196–202. doi: 10.1167/4.3.6. [DOI] [PubMed] [Google Scholar]
- 36.Brascamp J, et al. Flash suppression and flash facilitation in binocular rivalry. J Vis. 2008;7(12):12.1–2.12. doi: 10.1167/7.12.12. [DOI] [PubMed] [Google Scholar]
- 37.Bartels A, Logothetis N. Binocular rivalry: A time dependence of eye and stimulus contributions. J Vis. 2010;10(12):3–1–14. doi: 10.1167/10.12.3. [DOI] [PubMed] [Google Scholar]
- 38.Keliris GA, Logothetis NK, Tolias AS. The role of the primary visual cortex in perceptual suppression of salient visual stimuli. J Neurosci. 2010;30(37):12353–12365. doi: 10.1523/JNEUROSCI.0677-10.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Pearson J, Clifford CW. When your brain decides what you see: Grouping across monocular, binocular, and stimulus rivalry. Psychol Sci. 2005;16(7):516–519. doi: 10.1111/j.0956-7976.2005.01566.x. [DOI] [PubMed] [Google Scholar]
- 40.Bonneh Y, Sagi D, Karni A. A transition between eye and object rivalry determined by stimulus coherence. Vision Res. 2001;41(8):981–989. doi: 10.1016/s0042-6989(01)00013-x. [DOI] [PubMed] [Google Scholar]
- 41.Crick F, Koch C. Consciousness and neuroscience. Cereb Cortex. 1998;8(2):97–107. doi: 10.1093/cercor/8.2.97. [DOI] [PubMed] [Google Scholar]
- 42.Rees G, Kreiman G, Koch C. Neural correlates of consciousness in humans. Nat Rev Neurosci. 2002;3(4):261–270. doi: 10.1038/nrn783. [DOI] [PubMed] [Google Scholar]
- 43.Tong F. Primary visual cortex and visual awareness. Nat Rev Neurosci. 2003;4(3):219–229. doi: 10.1038/nrn1055. [DOI] [PubMed] [Google Scholar]
- 44.Watson AB, Pelli DG. QUEST: A Bayesian adaptive psychometric method. Percept Psychophys. 1983;33(2):113–120. doi: 10.3758/bf03202828. [DOI] [PubMed] [Google Scholar]
- 45.van Dam LC, van Ee R. Retinal image shifts, but not eye movements per se, cause alternations in awareness during binocular rivalry. J Vis. 2006;6(11):1172–1179. doi: 10.1167/6.11.3. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.