Abstract
Itch is triggered by somatosensory neurons expressing the ion-channel TRPV1 but the mechanisms underlying this nociceptive response remain poorly understood. Here we showed that the neuropeptide, Nppb, is expressed in a subset of TRPV1-neurons and found that Nppb−/− mice selectively lose almost all behavioral responses to itch-inducing agents. Importantly, Nppb triggered potent scratching when injected intrathecally in wild type and Nppb−/− mice showing that this neuropeptide evokes itch when released from somatosensory neurons. Itch responses were blocked by toxin-mediated ablation of Nppb-receptor expressing cells but a second neuropeptide, GRP, still induced strong responses in the toxin treated animals. Thus our results define the primary pruriceptive neurons, characterize Nppb as an itch-selective neuropeptide and reveal the next two stages of this dedicated neuronal pathway.
Pruritic responses are triggered by somatosensory neurons, with several itch-inducing agents acting through pathway involving the ion-channel TRPV1 (1, 2) and the effector enzyme PLCβ3 (3). Recently, agonist induced silencing of TRPV1-expressing neurons was shown to result in a profound loss of all itch responses (2) but also affects thermosensation and pain (4). In contrast, a much more specific loss of pruriception was observed in mice lacking the gastrin releasing peptide (GRP) receptor (5) and in animals where the spinal interneurons expressing this receptor had been specifically targeted with a GRP-conjugated toxin (GRP-saporin) (6). Thus it was hypothesized that GRP is the neurotransmitter that initiates a labeled line for itch (5, 6).
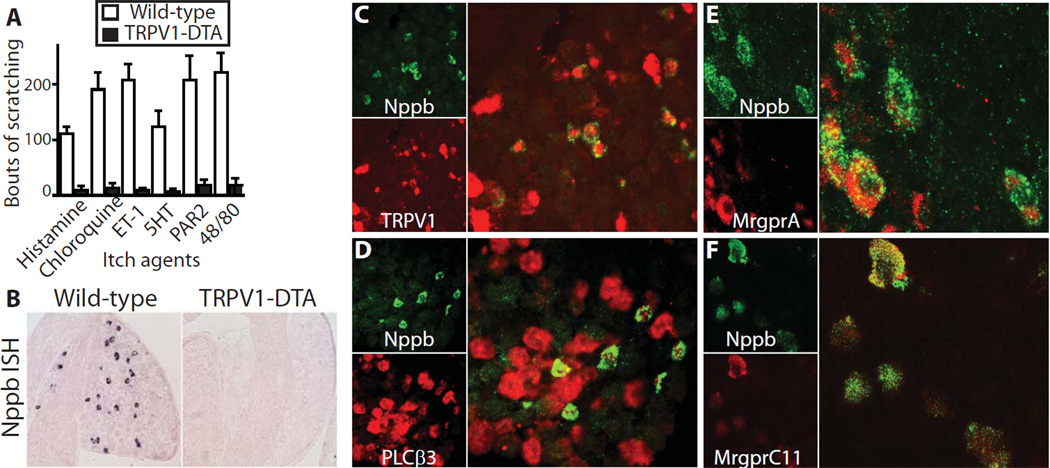
Previously, we generated mice that had lost all TRPV1-lineage neurons (7). These mice have major pruritic deficits (Fig. 1A) as well as a complete loss of thermosensory input (7) in keeping with previous reports using capsaicin-induced lesions (2, 4). To identify candidate molecules that might mediate itch signaling, we used a differential microarray based screen that identified many TRPV1-enriched transcripts (Table S1). Amongst these, the natriuretic polypeptide b (Nppb) is prominently expressed in a small subset of DRG neurons but is dramatically decreased in sensory ganglia from TRPV1-DTA animals (Fig. 1B). Double label ISH explicitly demonstrates that all Nppb-expressing neurons containTRPV1 (Fig. 1C) and PLCβ3 (Fig. 1D), which are critically required for histamine induced scratching in mice (2, 3). Moreover, double labeling (Fig. 1E, F) shows almost complete overlap between the expression of Nppb and two Mas related G protein coupled receptors that have recently been shown to detect specific pruritogens (8–10).
Fig. 1. Nppb is always co-expressed with itch related signaling molecules TRPV1 and PLCβ3.
TRPV1-DTA mice exhibit dramatically reduced scratch responses following intradermal injection of pruritic agents; histamine (100 µg in 10 µl); chloroquine (100 µg); endothelin 1 (1µM); methyl-serotonin (30 µg); PAR2 agonist SLIGRL-NH2 (100 µg) and compound 48/80 (100 µg). Itch inducing substances were injected intradermally into the shoulder of mice and numbers of scratching bouts were assessed over 30 minutes; data are mean ± s.e.m (n ≥ 7 animals) normalized to wild-type litter controls. (B) In situ hybridization (ISH) of sections through DRG illustrating loss of Nppb-expression in TRPV1-DTA animals; quantitation of Nppb expressing neurons revealed that 7 ± 0.6 % of NeuN-positive C4 DRG neurons express the neuropeptide in wild type mice (n=6). (C) Double label ISH of DRG demonstrates that Nppb (green) and TRPV1 (red) are co-expressed in the same sensory neurons; note that only a subset of TRPV1 expressing neurons contain Nppb. (D) All Nppb-positive neurons also express PLCβ3 (red) but many PLCβ3 neurons are Nppb negative. (E) Double label ISH also shows that Nppb-positive neurons (green) all express MrgprA receptors (including MrgprA3, the receptor for chloroquine; red), with more than 70% of MrgprA expressing-neurons also containing the neuropeptide. (F) Double label immunostaining demonstrates complete overlap between expression of MrgprC11 (red), the receptor for the pruritogen SLIGL-NH2 and Nppb (green) in somatosensory neurons. Also see Fig. S2 for additional characterization of neurons expressing Nppb.
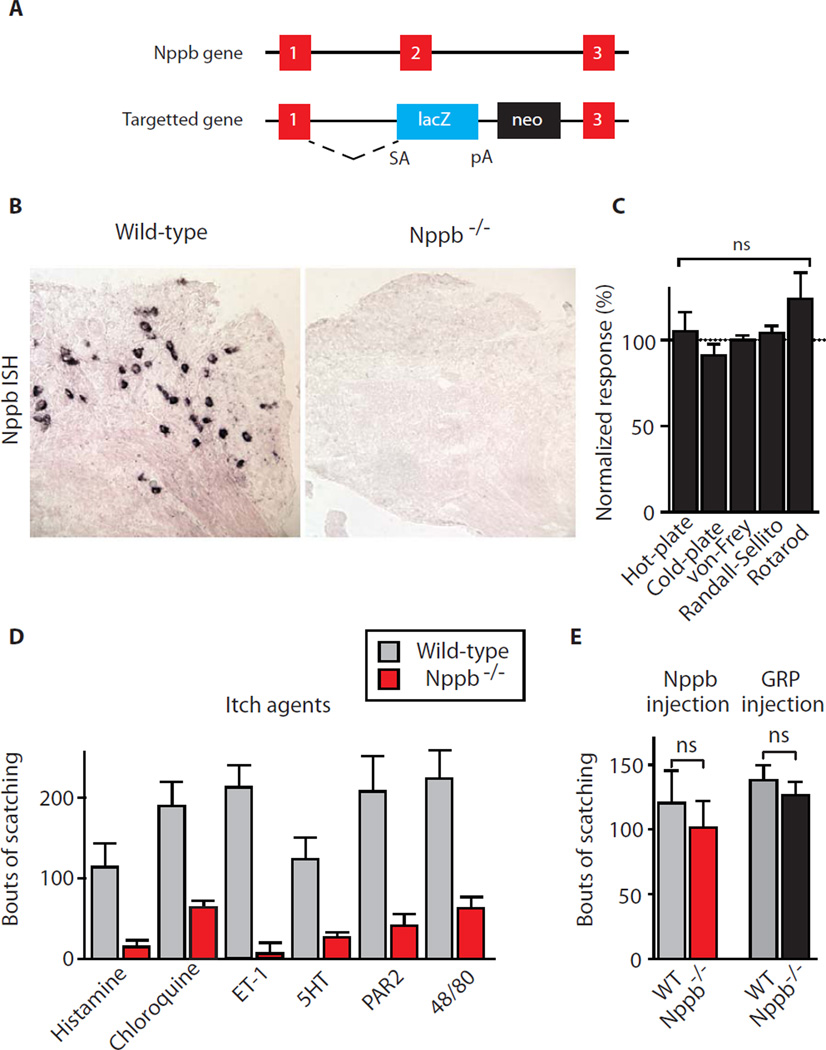
We generated Nppb−/− animals (Fig. 2A) and showed that these mutants displayed no detectable expression of Nppb (Fig. 2B). The mice were healthy, had normal numbers of nociceptive, touch, and proprioceptive neurons, and the distribution and number of dorsal horn interneurons was unaffected by gene disruption (Fig. S1). Nppb−/− mice retained normal responses to thermal, nocieptive, touch, and proprioceptive stimulation when tested using standard assays (Fig. 2C). We performed intradermal injections and recorded numbers of scratching bouts for substances that directly activate pruriceptors and with compound 48/80 that causes itch via an indirect route (11). All these agents (Table S2) reliably triggered multiple bouts of scratching in control animals (Fig. 2D, grey bars) but Nppb−/− mice were almost completely insensitive to the full range of pruritic substances tested (Fig. 2D, red bars).
Fig. 2. Generation and characterization of Nppb −/− mice.
(A) Schematic representation showing the disruption of the Nppb gene by insertion of a splice acceptor-lacZ cassette into the second exon used to generate Nppb−/− mice. (B) In situ hybridization of sections through DRG reveals that Nppb expression is lost in Nppb−/− animals. (C) Nppb −/− mice retained normal reactions to thermal, touch and proprioceptive stimuli, (D) but exhibit greatly attenuated responses to a range of pruritic agents (Table S2); data are mean ± s.e.m (n≥ 7 animals) normalized to wild-type litter controls. Significant differences between genotypes were determined using Student’s t-test with * indicating P < 0.001. (E) Intrathecal injection of Nppb (5µg in 10µl) into the lumbar 4–5 segment of control and Nppb −/− mice induced repeated bouts of scratching; injection of GRP (1nM in 10µl) also triggered itch responses in both mutant and control animals. Data represent mean values ± s.e.m. (n≥ 5 animals); no significant differences in response between genotypes were found (Student’s t-test). Typical behavioral responses of control and Nppb −/− mice to histamine, Nppb and the nociceptive neuropeptide substance P are illustrated in Videos S1 – 3.
How does Nppb induce this stereotyped scratch response? We reasoned that because this peptide is prominently expressed in somatosensory neurons the most plausible explanation for its role would be if it acted as a specific itch related neuromodulator (or neurotransmitter) in the spinal cord. Indeed intrathecal injection of Nppb induced profound scratching behavior in wild type animals (Fig. 2E, grey bar) demonstrating that spinal-Nppb is sufficient to induce itch even without activation of the peripheral neurons that express it. Intrathecal injection of Nppb into Nppb−/− mice also led to an equivalent phenotype (Fig. 2E, red bar). Loss of Nppb in sensory afferents was thus responsible for the pruriceptive deficits seen in mutant mice. Therefore we suggest that Nppb-expression delineates the subset of somatosensory neurons that detect pruritic agents and that central release of Nppb from these neurons is necessary for the itch response.
Because Nppb is responsible for transmitting the peripheral signals that trigger pruritic responses, its receptor Npra (12) should be expressed at the site of afferent fiber synaptic connections in the spinal dorsal horn and mark the secondary neurons in the itch response circuit. Therefore we used ISH to localize Npra expression in the dorsal horn and found that it is indeed expressed in a limited subset of neurons (most likely interneurons, see below), primarily in the outer layer i.e. lamina I (Fig. 3A left panel) corresponding to the terminal field of TRPV1 expressing sensory neurons (13).
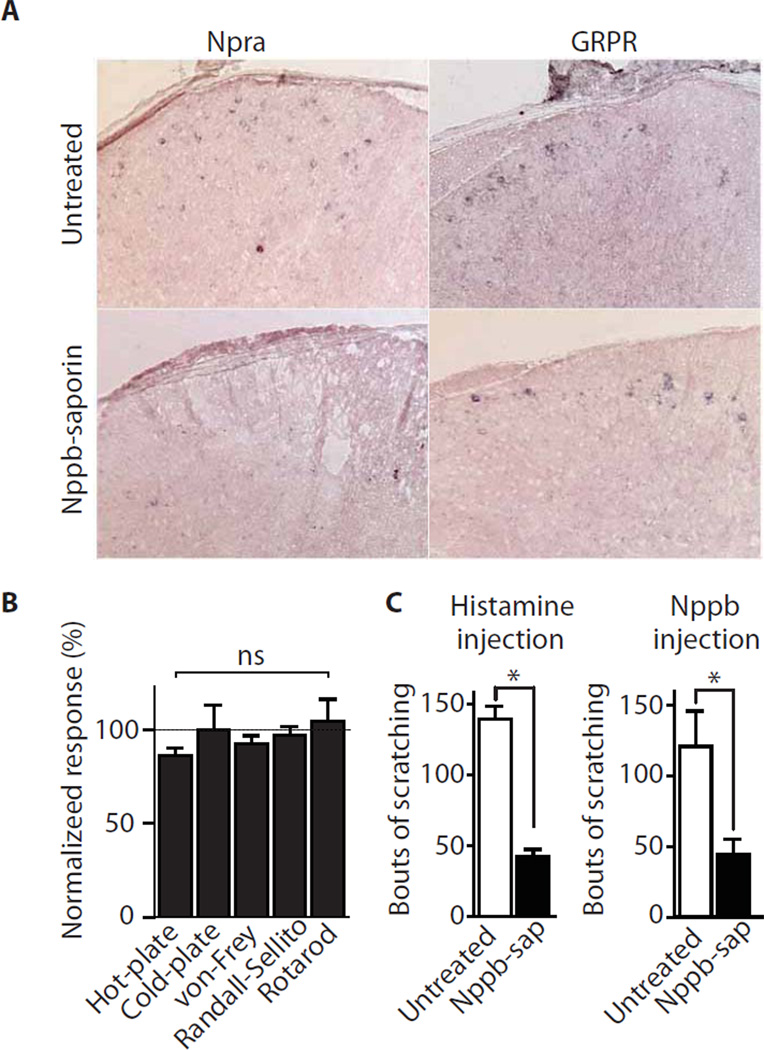
Fig. 3. Selective ablation of Npra receptor neurons in the spinal cord impairs pruriception.
(A) Expression of Npra in the dorsal spinal cord was assessed using ISH. In normal mice (upper panel), a significant subset of interneurons in the outer layer express Npra, however after intrathecal administration of Nppb-saporin (lower panel) few Npra neurons remained. In contrast, the number of GRP-receptor positive cells (right panels) were unaffected by Nppb-saporin. (B) Nppb-saporin treated mice retain normal reactions to thermal, touch and proprioceptive stimuli, but (C) exhibit greatly attenuated responses to intradermal injection of histamine (100 µg in 10 µl) or intrathecal administration of Nppb. Data represent means normalized against untreated controls ± s.e.m. (n≥ 5 animals). Significant differences between genotypes were determined using Student’s t-test with * indicating P < 0.01.
To examine whether the Npra neurons in the spinal cord function in the itch circuit and if they are selectively required for pruritogen induced scratching (rather than other somatosensory responses) we used a targeted-toxin cell ablation strategy (14). We injected an Nppb-saporin conjugate intrathecally into wild type mice to target their Npra expressing cells and assessed the effectiveness, specificity and behavioral consequences of administering this toxin. Approximately 70 % of Npra-positive neurons were eliminated after administration of toxin (Figs. 3A, S3). This targeting was highly selective, with neither cells expressing the GRP-receptor nor other dorsal horn interneurons affected by Nppb-saporin treatment (Figs. 3A, S3). Toxin injected mice displayed normal responses to thermal, touch, and painful stimulation (Fig. 3B). However, we observed a dramatic reduction in scratching evoked by histamine (Fig. 3C) indicating that these neurons are required for itch responses but not for other somatosensory pathways.
How do these findings fit with current models of itch behavior? The GRP-receptor has been shown to be a key element in the pruritic pathway (5, 6) with the suggestion that GRP might be the primary neurotransmitter for itch. However, this view has also been questioned (11, 15). We were unable to detect more than trace quantities of GRP expression in the DRG using a sensitive qPCR assay (Fig. 4A). Similarly, somatosensory neurons from GRP-reporter mice Tg(GRP-EGFP) were negative for EGFP expression (Figs. S4A). In contrast, ISH for GRP and analysis of EGFP expression in the GRP-reporter mice revealed that this neuropeptide is present in a population of neurons in the dorsal horn (Figs. 4B, S4A) as reported previously (15, 16). Therefore we concluded that GRP cannot act at the level of pruriception but must function downstream of Nppb and applied three complementary functional strategies to substantiate this hypothesis and dissect the itch response circuit.
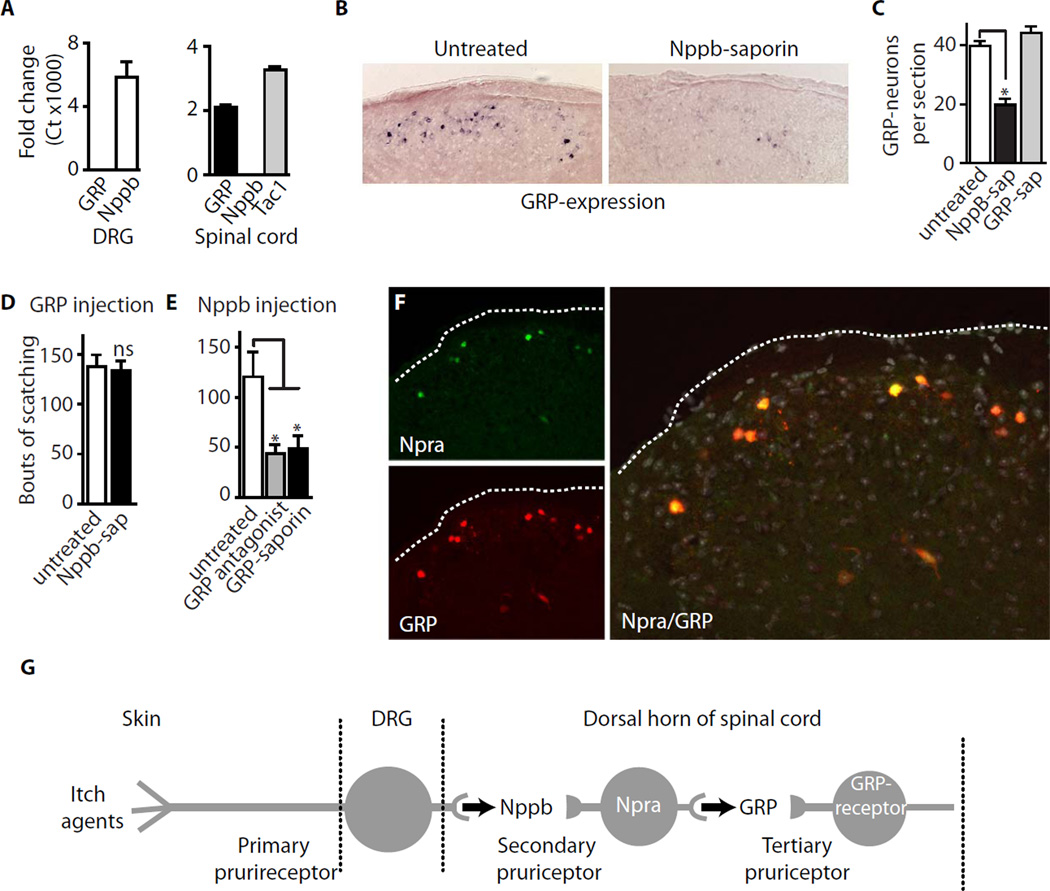
Fig. 4. GRP acts directly downstream of Nppb in the rodent pruriceptive circuit.
(A) qPCR was used to quantitate expression of GRP and Nppb relative to GAPDH in the DRG and spinal cord. GRP is robustly expressed in the spinal cord (at a level comparable with Tac1) but is almost undetectable in the DRG. Nppb is prominently expressed in DRG, but is not present in the spinal cord. Data represent mean ± s.e.m for triplicate cDNA preparations each analyzed in 2 separate PCR reactions. (B) ISH was used to analyze GRP expression in the dorsal horn of normal (left panel) and Nppb-saporin treated mice (right panel); note that many GRP expressing interneurons are lost on ablation of Npra-expressing cells. (C) A significant number of GRP-neurons was eliminated following Nppb-saporin (Nppb-sap) treatment. GRP-saporin (GRP-sap), which targets GRP-receptor neurons has no effect on the number of GRP-interneurons; data represent mean ± s.e.m. (n≥ 4 animals). Significant differences between groups were determined using Student’s t-test with * indicating P < 0.001. (D) Ablation of Npra-neurons had no effect on intrathecally administered GRP induced scratch responses. (E) Scratching induced by lumbar injection of Nppb was strongly attenuated by pretreatment with a selective GRP antagonist or by the ablation of GRPR-expressing neurons with GRP-saporin. Data (D, E) are mean ± s.e.m. (n≥ 6 animals) and * indicates P < 0.001 (Student’s t-test). (F) Double label immunohistochemistry was used to localize interneurons expressing Npra (green) and GRP-driven EGFP (red) in sections through the dorsal horn of Tg(GRP-EGFP) mice. Left panels show typical individual staining patterns while the right panel shows merged Npra and GRP images with the cell nuclei counterstained with DAPI (grey); the outline of the dorsal horn is dotted. (G) Model of the first three stages of the pruriceptive circuit with the critical neuropeptide used at each stage indicated.
First we demonstrated that GRP induced scratching was unaltered either by Nppb-knockout (Fig. 2E) or by the ablation of Npra-expressing neurons (Fig. 4D). Second, we showed that pharmacological inhibition of the GRP-receptor not only attenuated behavioral responses to the pruritic agent histamine or GRP injection (Fig. S4B), but also inhibited scratching after intrathecal administration of Nppb (Fig. 4E). Lastly, we tested mice with selective ablation of GRP-receptor expressing neurons and again found significantly reduced itch responses to Nppb (Fig. 4E). These data place GRP downstream of Nppb in the itch response circuit, suggest that the secondary pruriceptors are targets for one neuropeptide, Nppb and in turn signal through a second peptide, GRP. Indeed, just as this model predicts, all Npra-expressing neurons in the dorsal horn contain GRP (Fig. 4F) and Nppb-saporin treatment significantly reduces the number of GRP expressing cells (Fig. 4B, right panel).
Our results molecularly characterize the first three stations of an itch response pathway in mice (Fig. 4G), demonstrate that Nppb marks the primary sensory neurons and show that this peptide is both necessary and sufficient for transmission of peripheral signals that induce stereotypic itch responses. Unlike previously characterized receptors (8, 9) and signaling molecules (1–3) that affect the detection of particular itch-inducing agents, Nppb is necessary for responses to a wide range of pruritogens (i.e., compounds classed as inducing histamine and non-histamine related itch, Table S2). Our data also refine the role GRP and GRP-receptor cells play in the itch response pathway by placing them at later stages than had been hypothesized previously (5, 6). Many questions about itch remain unanswered, including its close relationship to sensing pain (17), its slow kinetics and long duration, as well as reports from human and non-human primate studies that different central pathways mediate histamine and non-hitamine itch (18–21). Because Nppb is critically required for pruriception in mice, future studies involving ablation and/or activation of the Nppb-expressing somatosensory neurons together with circuit tracing may reveal whether these cells directly trigger a spinal scratch reflex, are selective detectors for the sensation of itch or are in fact more broadly-tuned nociceptors. Such experiments will help reveal the central mechanism(s) for itch, explain the interactions between pruriception and other somatosensory signals and ultimately provide a powerful stimulus for the rational design of novel therapies to alleviate chronic itch.
Supplementary Material
Acknowledgments
We are grateful to Nick Ryba for encouragement and helpful advice and for valuable suggestions. The Nppb−/− mice used for this research project were generated ES cells obtained from the NCRR-NIH supported KOMP repository (www.komp.org) and engineered by the Wellcome Trust Sanger Institute and the Mouse Biology Program (www.mousebiology.org). We thank NIDCR GTF for help generating Nppb mutant mice. The mouse strain STOCK Tg(GRPEGFP) was obtained from the MMRRC, a NCRR-NIH funded strain repository, and was donated to the MMRRC by the NINDS funded GENSAT BAC transgenic project. This research was supported by the intramural research program of the NIH, NIDCR.
Footnotes
This manuscript has been accepted for publication in Science. This version has not undergone final editing. Please refer to the complete version of record at http://www.sciencemag.org/. The manuscript may not be reproduced or used in any manner that does not fall within the fair use provisions of the Copyright Act without the prior, written permission of AAAS.
References and Notes
- 1.Shim WS, et al. TRPV1 mediates histamine-induced itching via the activation of phospholipase A2 and 12-lipoxygenase. J Neurosci. 2007 Feb 28;27:2331. doi: 10.1523/JNEUROSCI.4643-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Imamachi N, et al. TRPV1-expressing primary afferents generate behavioral responses to pruritogens via multiple mechanisms. Proc Natl Acad Sci U S A. 2009 Jul 7;106:11330. doi: 10.1073/pnas.0905605106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Han SK, Mancino V, Simon MI. Phospholipase Cbeta 3 mediates the scratching response activated by the histamine H1 receptor on C-fiber nociceptive neurons. Neuron. 2006 Nov 22;52:691. doi: 10.1016/j.neuron.2006.09.036. [DOI] [PubMed] [Google Scholar]
- 4.Cavanaugh DJ, et al. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc Natl Acad Sci U S A. 2009 Jun 2;106:9075. doi: 10.1073/pnas.0901507106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sun YG, Chen ZF. A gastrin-releasing peptide receptor mediates the itch sensation in the spinal cord. Nature. 2007 Aug 9;448:700. doi: 10.1038/nature06029. [DOI] [PubMed] [Google Scholar]
- 6.Sun YG, et al. Cellular basis of itch sensation. Science. 2009 Sep 18;325:1531. doi: 10.1126/science.1174868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mishra SK, Tisel SM, Orestes P, Bhangoo SK, Hoon MA. TRPV1-lineage neurons are required for thermal sensation. EMBO J. 2011 Feb 2;30:582. doi: 10.1038/emboj.2010.325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Liu Q, et al. Sensory neuron-specific GPCR Mrgprs are itch receptors mediating chloroquine-induced pruritus. Cell. 2009 Dec 24;139:1353. doi: 10.1016/j.cell.2009.11.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Liu Q, et al. The distinct roles of two GPCRs, MrgprC11 and PAR2, in itch and hyperalgesia. Science signaling. 2011;4:ra45. doi: 10.1126/scisignal.2001925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Han L, et al. A subpopulation of nociceptors specifically linked to itch. Nat Neurosci. 2012 Dec 23;16:174. doi: 10.1038/nn.3289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.McNeil B, Dong X. Peripheral mechanisms of itch. Neuroscience bulletin. 2012 Apr;28:100. doi: 10.1007/s12264-012-1202-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Misono KS, et al. Structure, signaling mechanism and regulation of the natriuretic peptide receptor guanylate cyclase. The FEBS journal. 2011 Jun;278:1818. doi: 10.1111/j.1742-4658.2011.08083.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Caterina MJ, et al. Impaired nociception and pain sensation in mice lacking the capsaicin receptor. Science. 2000 Apr 14;288:306. doi: 10.1126/science.288.5464.306. [DOI] [PubMed] [Google Scholar]
- 14.Wiley RG, Lappi DA. Targeted toxins in pain. Advanced drug delivery reviews. 2003 Aug 28;55:1043. doi: 10.1016/s0169-409x(03)00102-9. [DOI] [PubMed] [Google Scholar]
- 15.Fleming MS, et al. The majority of dorsal spinal cord gastrin releasing peptide is synthesized locally whereas neuromedin B is highly expressed in pain- and itch-sensing somatosensory neurons. Mol Pain. 2012 Jul 9;8:52. doi: 10.1186/1744-8069-8-52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Li MZ, et al. Molecular mapping of developing dorsal horn-enriched genes by microarray and dorsal/ventral subtractive screening. Developmental biology. 2006 Apr 15;292:555. doi: 10.1016/j.ydbio.2006.01.033. [DOI] [PubMed] [Google Scholar]
- 17.Patel KN, Dong X. An itch to be scratched. Neuron. 2010 Nov 4;68:334. doi: 10.1016/j.neuron.2010.10.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Namer B, et al. Separate peripheral pathways for pruritus in man. J Neurophysiol. 2008 Oct;100:2062. doi: 10.1152/jn.90482.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Schmelz M, Schmidt R, Bickel A, Handwerker HO, Torebjork HE. Specific C-receptors for itch in human skin. J Neurosci. 1997 Oct 15;17:8003. doi: 10.1523/JNEUROSCI.17-20-08003.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Johanek LM, et al. A role for polymodal C-fiber afferents in nonhistaminergic itch. J Neurosci. 2008 Jul 23;28:7659. doi: 10.1523/JNEUROSCI.1760-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Davidson S, et al. Pruriceptive spinothalamic tract neurons: physiological properties and projection targets in the primate. J Neurophysiol. 2012 Sep;108:1711. doi: 10.1152/jn.00206.2012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gong S, et al. A gene expression atlas of the central nervous system based on bacterial artificial chromosomes. Nature. 2003 Oct 30;425:917. doi: 10.1038/nature02033. [DOI] [PubMed] [Google Scholar]
- 23.Yegen BC, et al. Inhibitory effects of gastrin releasing peptide on gastric emptying in rats. Regulatory peptides. 1996 Mar 22;61:175. doi: 10.1016/0167-0115(95)00151-4. [DOI] [PubMed] [Google Scholar]
- 24.Hoon MA, et al. Putative mammalian taste receptors: a class of taste-specific GPCRs with distinct topographic selectivity. Cell. 1999 Feb 19;96:541. doi: 10.1016/s0092-8674(00)80658-3. [DOI] [PubMed] [Google Scholar]
- 25.Adler E, et al. A novel family of mammalian taste receptors. Cell. 2000 Mar 17;100:693. doi: 10.1016/s0092-8674(00)80705-9. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.