Abstract
The baculovirus expression system is one of the most successful and widely used eukaryotic protein expression methods. This short review will summarise the role of bacterial artificial chromosomes (BACS) as an enabling technology for the modification of the virus genome. For many years baculovirus genomes have been maintained in E. coli as bacterial artificial chromosomes, and foreign genes have been inserted using a transposition-based system. However, with recent advances in molecular biology techniques, particularly targeting reverse engineering of the baculovirus genome by recombineering, new frontiers in protein expression are being addressed. In particular, BACs have facilitated the propagation of disabled virus genomes that allow high throughput protein expression. Furthermore, improvement in the selection of recombinant viral genomes inserted into BACS has enabled the expression of multiprotein complexes by iterative recombineering of the baculovirus genome.
1. Baculovirus Protein Expression
1.1. Baculoviruses
The Baculoviridae is a family of viruses with a circular dsDNA genome, ranging in size from 80 kb to 180 kb, that infect arthropods. Virus particles are rod-shaped, with a lipid envelope derived from the host cell. The family is divided into four genera, based on the comparison of a subset of core genes conserved between all baculoviruses [1, 2]. The viruses used for recombinant protein expression are Autographa californica multiple nucleopolyhedrosis virus(AcMNPV) and Bombyx mori nucleopolyhedrosis virus(BmNPV) and both are in the Alphabaculovirus genus. Both viruses are pathogens of the larval stages of lepidopteran species and have an infection cycle that involves ingestion and infection of the cells of the mid-gut, followed by systemic infection. In contrast to vertebrate herpesviruses, which have a similarly sized, circular DNA genome, and which are largely cryptic infections except in immunocompromised individuals, infection of caterpillars with AcMNPV and BmNPV results in a short duration, acute, systemic infection with liquefaction of the host (Figure 1). Once the host is liquefied, the virus remains in the environment encased in a protein matrix formed from the virally encoded polyhedrin protein until it is ingested by the next caterpillar and the alkali environment of the insect mid-gut triggers dissociation of the polyhedrin coat and release of the virus [3, 4].
Figure 1.
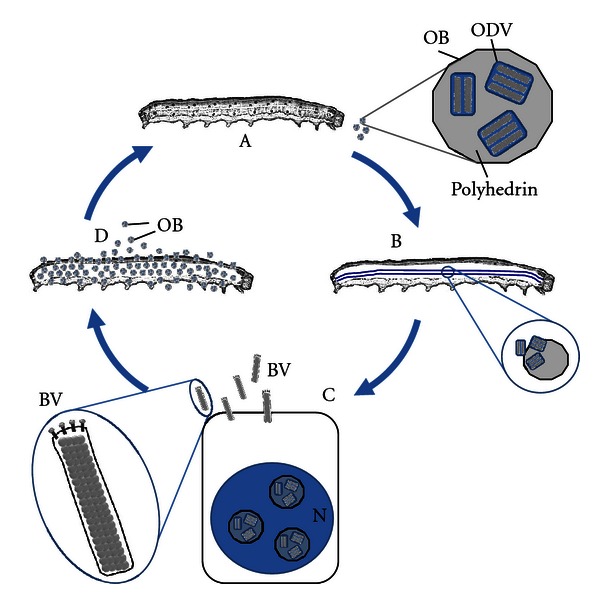
Key stages in the infection cycle of AcMNPV. (A) Infection is initiated by the ingestion of a virus occlusion body (OB). This consists of multiple virus nucleocapsids surrounded by a single lipid envelope (ODV) embedded in a protein matrix formed by the virally encoded polyhedrin protein. (B) The occlusion body is dissolved by the alkaline environment of the insect mid-gut, releasing ODV which initiate a primary infection in the midgut epithelial cells. (C) Virus enters cells and replicates in the nucleus. Two different forms of infectious virus are produced in infected cells. Budded virus (BV) is released at the cell surface and mediates systemic infection of the insect via the tracheal system, and ODV remains embedded in occlusion bodies. (D) Late stages of virus infection trigger liquefaction of the host, releasing the environmentally stable proteinaceous occlusion bodies. Polyhedrin protein is nonessential for the infection of cells in continuous culture in the laboratory and its high level of synthesis makes its promoter ideal for the high-level production of recombinant protein.
Reflecting the complex nature of their genome, baculovirus infections have a complex multiphase replication cycle that involves a distinct series of transcriptional steps. AcMNPV enters the host cell by a fusion process, mediated by the GP64 protein on the virus surface [5–10], and the nucleocapsid is trafficked to the nucleus. The virus has a genome of 133 894 bp double-stranded (ds) DNA and encodes 154 putative ORFs [11]; following nuclear entry, some of the corresponding genes are transcribed by the host RNA polymerase II and are termed immediate early genes. These genes encode proteins with a range of functions including some that act as transactivators for the expression of other viral genes that start to be produced late (6–12 hours) and very late (16–76 hours) post-infection [12–20]. Two forms of mature virus particles are produced in infected cells: budded virus is released from the cell from about 8 hours post-infection [21, 22] and is responsible for cell-to-cell spread within the infected caterpillar. Occluded virus is produced later in infection and is the form of the virus preserved in polyhedra, and is responsible for infection of the next host.
With a few exceptions early in infection, there is a global down-regulation of cellular transcription in baculovirus-infected cells [23–25]. The molecular basis for how this is mediated is unclear, but given the number of genes affected (mRNA corresponding to >10,000 ESTs is transcriptionally downregulated within 12 hours post-infection [25]), this is unlikely to be a targeted programme of downregulation. The consequence of transcriptional shutdown is that cellular protein levels deteriorate in baculovirus infections between 10–12 hours postinfection and cell lysis usually occurs sometime after 76 hours postinfection. AcMNPV overcomes this transcriptional downregulation by expressing its own DNA-dependent RNA polymerase complex, which is responsible for the transcription of late and very late genes [26–33]. Coupled to a high gene copy number as a result of the replication of the virus genome, this polymerase potentiates high level synthesis of recombinant protein. The polyhedrin protein accumulates to high levels in infected cells and polyhedra can represent up to 30% of the dry weight of infected caterpillars [34]. A lot of the initial and continuing interest in baculoviruses is related to their potential as environmentally safe, targeted, biopesticides for the control of insect pests [34–37]. However, AcMNPV and BmNPV are also widely used in academic and industrial research and biopharmaceutical production.
1.2. Production of Recombinant Proteins in the Baculovirus System
Early experiments in protein expression using the baculovirus genome as a vector were based on the replacement of the coding sequence for the polyhedrin protein with another gene. This approach has two advantages; firstly, it capitalises on the very high expression level usually observed for polyhedrin. Secondly, as polyhedra are readily observed under a light microscope in cells that express native polyhedrin, recombinant viruses can be selected based on a polyhedra negative phenotype, allowing for selection of pure recombinant virus stocks. As the virus genome was too large for routine direct manipulation at that time, early protein expression studies took advantage of the fact that homologous recombination in baculovirus-infected insect cells is relatively efficient. Replication of the baculovirus genome involves expression of proteins that promote homologous recombination [38, 39]. Transfer vectors were constructed in which an expression cassette was flanked with regions homologous to sequences in the baculovirus genome. Cotransfection of the transfer vector and virus DNA into insect cells allowed selection of recombinant virus. The first recombinant protein successfully expressed to high levels using this approach was human beta interferon [40]. Subsequent modifications to this system included the insertion of the LacZ coding sequence into the polyhedrin locus of the virus genome to allow blue-white screening and linearization of the genome in an adjacent essential gene; both modifications facilitated selection of recombinant viruses [41–43]. The use of dual promoter vectors for the expression of LacZ, another protein from the polyhedron, and one other genetic locus (p10 locus) within the virus genome was also demonstrated [44–46].
Perhaps the best demonstration of the utility of recombinant protein production using the baculovirus system is by its widespread use. A search of PubMed with the terms “((baculovirus OR AcMNPV) AND recombinant protein)” returns 7281 peer-reviewed publications, between the initial description of human interferon expression in 1983, and December 2011 (Figure 2). There are now several other eukaryotic systems that are used for the expression of recombinant proteins but the baculovirus system remains a popular choice with 233 and 261 publications with the same search terms in 2009 and 2010, respectively. Recombinant proteins expressed range from small peptides to multiprotein complexes assembled from multiple subunits [47–50]. As expression is in eukaryotic cells, recombinant proteins have authentic posttranslational processing with respect to phosphorylation, palmitoylation, isoprenylation, myristoylation, and folding [51–54]. Glycoproteins expressed in the system are also usually modified by the addition of carbohydrates by the N-glycosylation pathway, but the insect cells used generally lack the capability to synthesize the terminal highly branched galactose and sialic acid carbohydrates found on proteins expressed in mammalian cells [55]. These problems can be overcome to a certain extent by using modified cells engineered to express enzymes in the human glycosylation pathway [56–58].
Figure 2.
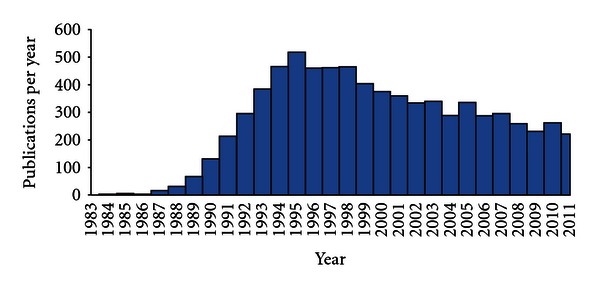
Annual publications 1983–2011 containing the search terms baculovirus or AcMNPV and recombinant protein.
2. Early Use of Bacterial Artificial Chromosomes in the Baculovirus System
While the selection of viruses produced by homologous recombination in insect cells was a highly effective way to recover recombinant viruses, it required a reasonable level of training in virology in order to be able to complete routinely. The development of baculovirus genomes in which a bacterial artificial chromosome (BAC) sequence and transposon target site were inserted at the polyhedrin locus in the virus genome allowed the propagation of the virus genome in E. coli as a bacmid [59]. This system allowed the transposition of a short sequence of DNA containing the promoter for the polyhedrin gene, a coding sequence, and a polyadenylation signal sequence into the BAC DNA located at the polyhedrin locus of the AcMNPV genome. The system was designed such that successful transposition interrupted the coding sequence of the LacZα fragment, allowing selection of recombinants in E. coli by standard blue-white screening. This removed the need for plaque selection of recombinant viruses, which made the system more accessible to non-virology laboratories. The bacmid system was successfully marketed by Invitrogen as “Bac-to-Bac” and has been adopted by many laboratories as a method for the efficient production of recombinant baculovirus. Notable variations on this core technology include the introduction of an antibiotic resistance gene expressed in E. coli with the transposed element, and the inclusion of the sacB gene on the transfer vector, to select for recombinant bacmid and counter-select the transfer vector which has not recombined, respectively [60]. These variations allow high-throughput selection of recombinants produced using the core approach.
Since the initial description of application of the bacmid technology to AcMNPV, the approach has since been applied to other related baculoviruses [61–64]. In most cases, the role of the BAC modified virus genome has been to provide a stable reference genome for sequencing studies. But in the case of BmNPV, a similar protein expression system to the more widely used AcMNPV system has also been developed.
3. Recent Developments in Baculovirus Research Enabled by BACs
3.1. Improved Understanding of Baculovirus Biology
In addition to facilitating insertion of foreign coding sequences into the baculovirus genome, maintenance of the virus genome as a bacmid in E. coli has also enabled the use of advanced reverse engineering approaches. In particular, the use of the recET/lambda-red-based recombineering approach [65] has been enthusiastically adopted by laboratories frustrated with the problem of producing null mutants in virus genes using conventional approaches. The core problem is that disruption of scientifically interesting genes often results in the production of a virus that has low or no viability. With conventional techniques, the generation of mutations in these genes either relied on conditional mutants [66, 67] or recombination in insect cells where mutations were complemented with transgenes in the virus or cellular genome [8, 68, 69]. Although effective, these approaches were time consuming and, in some cases, failed to recover the required mutation [70].
The great advantage of the BAC platform as a basis of mutagenesis is that recovery of the mutant genome does not rely on the ability of the virus to replicate in insect cells, only on the viability of the BAC sequence. Therefore, there is a similar potential for mutagenesis by homologous recombination as in insect cells, but it is possible to separate out failure of the resulting virus to replicate from failure of the recombination reaction. Bideshi and Federici first reported application of recET-based recombination to generate deletion mutants in the AcMNPV bacmid [71] using the E. coli strain BJ5183 [72]. This mutation involved the disruption of the AcMNPV helicase gene; subsequent studies have used similar approaches for the disruption of a range of genes involved in a number of other virus functions (Table 1). The approach has been particularly useful to characterise mutations involved in virus assembly and cell-to-cell spread, where release of the infectious virus can be separated from genome replication.
Table 1.
Summary of null mutants in AcMNPV generated in BACmid DNA by ET recombination.
| Virus function | Gene deleted | References |
|---|---|---|
| DNA replication | helicase (p143), lef3, lef5, lef6, lef11, alk-exo, dna-pol | [70, 71, 94–100] |
| Transcription | ie-0 and ie-1 | [101] |
| Cell entry | ac23, gp64 | [47, 102, 103] |
| Infectivity in insects | ac96, p74 | [104, 105] |
| Nucleocapsid assembly | p6.9, ac53, vlf-1, 38K, ac101, ac142, ac144, ac109 | [106–112] |
| Nucleocapsid release from the nucleus | exon0, ac66, vp80 | [22, 85, 113–115] |
| Formation of enveloped virus | ac142, p48, ac76, odv-e18, odv-e56 | [47, 116–118] |
| Interaction with host | egt, chiA, v-cath, IAP2 | [47] |
| Unknown, essential genes | ac92, ac-fgf, ac11, ac-pe | [47, 119] |
| Unknown, non-essential genes | me53, ac18, ac-ctx | [47, 120, 121] |
To date, ~25% (40 out of an estimated 156) AcMNPV genes have been disrupted using the ET recombination method, demonstrating its utility for the routine modification of viral genomes maintained as bacmids. Similar approaches have been used with BmNPV [73, 74] and with other dsDNA viruses that can be maintained as bacmids in E. coli, including herpesviruses [75–81]. Therefore, the BAC recET system is becoming a valuable approach that has been used to aid in the understanding the mechanism of infection, assembly, replication, and host interaction of a broad range of different viruses.
3.2. Improved Quality of Biopharmaceuticals
One of the problems with using baculovirus-based systems for the production of biopharmaceuticals has been the consequence that a live virus is used in the protein production process and must be purified away from any recombinant protein produced. Although baculoviruses are naturally present at high quantities in the environment and AcMNPV is easily inactivated with nonionic detergents and binary ethylenimine [82] and does not replicate in vertebrate cells [83, 84], removal of baculovirus and its DNA from protein products is generally required prior to licensing. One recent approach used an insect cell line expressing AcMNPV VP80 from a transgene integrated into the cellular chromosomal DNA and a bacmid-derived AcMNPV vp80 null mutant [22]. As VP80 is essential for the maturation of the virus nucleocapsid in the nucleus [85], the null mutant was only able to produce viable progeny viruses in the complementing cell line. This cell line was used for amplification of the viruses and a nontransgenic cell line used for protein expression to recover recombinant protein free of viable baculovirus.
3.3. Recombineering for High-Throughput Production of Proteins
In addition to improving understanding of the virus biology, the bacmid platform, coupled to recET mutagenesis, has also been applied to improving the baculovirus system for protein production. Although many recombinant baculoviruses capable of expressing recombinant protein have been generated with the BAC-Tn7 transposase-based system [86], there are indications that genes inserted into the polyhedrin locus of the virus genome using this system are unstable, with recombinant protein expression declining with passage number [61, 87]. Although this can be addressed by genetic linkage of transgenes with sequences that promote retention of the foreign gene sequences, [88, 89] some laboratories maintain that insertion of genes directly into the AcMNPV genome in insect cells results in more stable protein expression. One of the problems with the latter approach has always been that recombination in insect cells required both a viable virus genome and a suitable transfer vector. Therefore, the viable virus genome inevitably led to a proportion of parental virus genomes that did not express the foreign gene and the approach was not suitable for high-throughput protein production.
The problem of selection of recombinant viruses was partially solved by Kitts et al. [42, 90], who developed techniques allowing selection of recombinant baculovirus using linearised virus DNA and lacZ expression. However, although it was a significant improvement on previous approaches, there was still a residual background of replicating viruses. The problem was finally solved by Zhao et al. [91], who used the bacmid-based recET recombineering system to make a deletion in an essential gene (orf1629) for virus replication in insect cells that is located next to the polyhedrin locus. This mutation was only possible because the defective viral genome could be maintained in E. coli as a bacmid. As the virus genome could only replicate in insect cells once recombination with the transfer vector had occurred, the problem of background virus replication was eliminated. In practice, there is probably a low level of replication of the defective genome through complementation unless virus genomes are purified by plaque-cloning methods. However, this is not significant enough to interfere with most high-throughput applications. Recent developments to this approach have combined the knockout deletion in orf1629 with deletions of chiA, v-cath, and p10 genes, with the aim of improving expression levels and trafficking of recombinant proteins [92, 93]. These modified defective genomes are commercially available as flashBac (Oxford Expression Technologies, UK) and BacMagic (Merck).
4. Engineering Stable Expression of Protein Complexes
Although many protein expression experiments focus on the production of one specific recombinant protein, there is an increasing recognition that many, if not most, biological processes are completed by complexes formed of several interacting proteins. In response, there is an increasing focus on the expression and purification of these complexes using recombinant systems. The AcMNPV based-baculovirus expression system has proved to be particularly useful for the expression of functional complexes [49, 122–124]. As a eukaryotic system, baculovirus-based expression has all the appropriate chaperones and other posttranslational modification activities that facilitate the assembly of large and complex protein assemblies.
The most straightforward approach to the production of protein complexes using the baculovirus system is the co-infection of the same batch of cells with two or more recombinant baculoviruses, each expressing a different foreign protein. The principle is that cells coinfected with the two viruses will express all the foreign proteins, allowing assembly of the complex. This approach has been used successfully for the production of many complexes and is often used to assess the potential for proteins to interact to form complexes [125–128]. However, as a method to reproducibly synthesize optimal amounts of recombinant protein, it is reliant on careful titering of infectious virus and cell numbers as the relative proportion of individual viruses present in each cell is determined by the Poisson distribution. This, in effect, means that a batch of cells infected with multiple viruses actually consists of multiple subpopulations in which the ratio of expression of different subunits in the complex varies. Furthermore, there is evidence that the Poisson distribution alone may not be sufficient to explain all variation in the level of subunit expression, and that infection with large numbers of different viruses per cell may inhibit protein expression [129]. An approach that circumvents these problems is to express multiple proteins from the same virus backbone so that all infected cells produce the recombinant proteins at the same relative ratio. Transfer vectors allowing the expression of multiple proteins from the same baculovirus genome were first constructed some time ago [130–133]. However, due to the need to insert multiple genes into the same vector, and the fact that the upper size of the vector restricts the number of genes that can be inserted, cloning strategies for these vectors, especially for large complexes, are not straightforward.
The first attempt to address this problem, based on the manipulation of bacmid DNA was by Berger et al. [134]. In these experiments, a LoxP recombination target site was inserted at the position of the chiA and v-cath genes that are adjacent in the baculovirus genome in the bacmid, using the recET recombineering approach. As the bacmid already contained the Tn7 transposase target site, the LoxP site allowed insertion of foreign genes at a second locus by Cre-mediated recombination. The resulting system is referred to as “Multibac” in the literature. The advantage of this system is that it is possible to “mix and match” the sequences inserted at the polyhedrin and chiA-v-cath loci [135, 136]. By combining with transfer vectors that are modified to allow repeated insertions of dual expression cassettes by conventional ligation or ligation-independent cloning approaches, expression of multiple protein subunits from the same baculovirus is possible [134, 135]. This partially solves the problem of large transfer vectors as genes are split between two loci, effectively doubling the limit imposed by maximum transfer vector size. Furthermore, as the system was developed specifically to allow automation of protein production by robotics, many different combinations of subunits can be rapidly tested for any one complex, which is particularly useful for structural studies [135]. Although in principle this system can be used for the expression of protein complexes with a large number of different subunits, early experiments suggested that there was some genetic instability, possibly due to repeated DNA elements [136]. This was not supported in subsequent experiments which suggested that 5 different recombinant proteins were still expressed after 4 low multiplicity of infection in vitro passages in 90% of cells [135]. However, studies on transposon insertions with a single insert predict that effects of gene deletion in baculovirus experiments may not be apparent before 5–10 passages anyway [87]. Overall, there is no clear evidence that inserts in the modified system are any less stable than the original Tn7 transposase-based bacmid system [59].
An alternative system for the production of multiprotein complexes from baculovirus genomes engineered to co-express protein subunits avoids both the transfer vector size problem and the potential for genetic instability in insect cells [47]. Instead of using site-specific transposase or recombinase activities to integrate genes into the baculovirus genome, the approach takes advantage of improved selection of recombinants to use the recET recombineering approach to directly insert cassettes for foreign gene expression at different genetic loci in the baculovirus genome (Figure 3).
Figure 3.
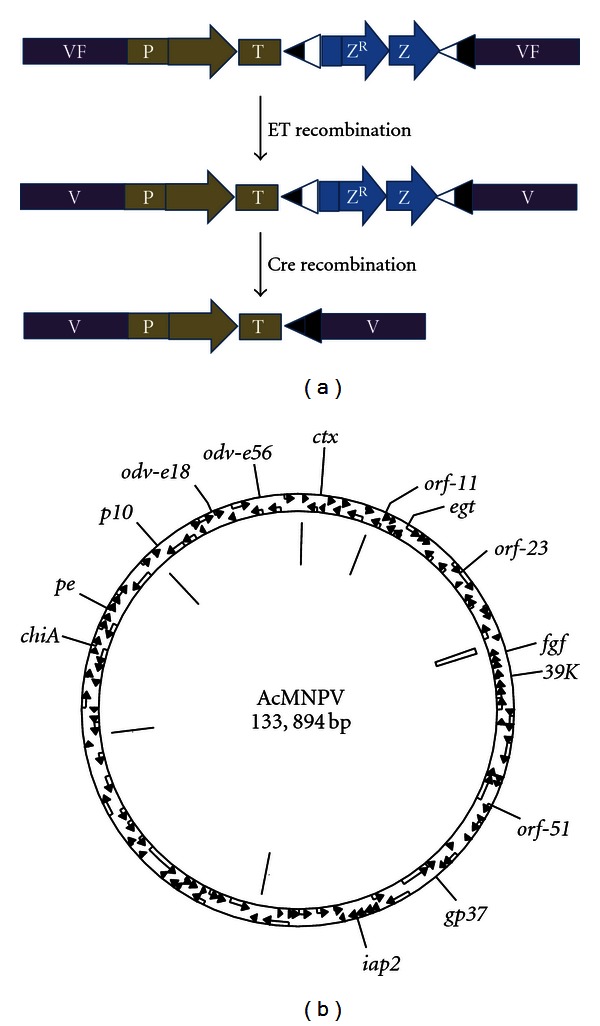
Iterative modification of AcMNPV to insert multiple single-locus expression cassettes. (a) Strategy used for repeated modification of the same bacmid to express multiple different recombinant proteins. ET recombination is targeted by viral flanking sequences (VF) to homologous sequences in the bacmid. Selection in E. coli is achieved using a bipartite marker consisting of a Zeocin resistance (ZR) and LacZα (Z), flanked by partially defective loxP sites. Following Cre-mediated recombination, this marker is removed, allowing a subsequent modification of the same DNA. (b) Loci within the AcMNPV successfully modified using the iterative modification strategy.
In order for the BAC-based recET approach to be viable for the routine insertion of genes for protein expression, it was necessary to significantly improve the ease with which recombinants could be selected. To achieve this, a bipartite selection system that allowed at least a 21-fold increase in the efficiency of selection of recombinants was developed [47]. By coupling this selection to self inactivating loxP sites, it was possible to achieve iterative modifications at multiple sites in the same genome using the same selection. Using this system coexpression of protein complexes containing two, four, and different recombinant proteins was demonstrated.
5. Conclusions and Future Perspectives
The use of bacterial artificial chromosomes continues to have a major impact on the development of studies for baculovirus and other large DNA viruses. Coupled to the recET recombineering approaches [65, 137, 138], the BAC platform is a powerful tool for understanding both the basic biology and for developing the industrial applications of baculovirus systems. The combination of null mutants with complementation has provided interesting insights into the role of viral proteins in virus replication, host interaction, and assembly. Current studies have been relatively crude and have resulted in deletions in the virus genome. As genes are arranged in both directions on the circular dsDNA AcMNPV genome, the impact of these deletions on flanking genes is a potential problem. Future studies may very well involve more seamless modification of the virus genome.
In terms of the applied use of baculovirus systems, BACs have facilitated protein expression for a number of years. The new recombineering approaches have also opened up new ways to produce virion-free biopharmaceuticals [22], and for the efficient production of multiprotein complexes [47, 134]. However, these are only beginning to scratch the surface of what is now possible with the baculovirus system. The demonstration that the routine integration of transgenes at any locus in the AcMNPV genome is now possible [47] opens the possibility of modifying background genomes to facilitate the expression environment for specific recombinant proteins.
Acknowledgment
P. Roy and R. Noad are listed as coinventors on a Patent Application (GB2009002647) by London School of Hygiene and Tropical Medicine that relates to the iterative modification of AcMNPV BAC DNA.
References
- 1.Jehle JA, Blissard GW, Bonning BC, et al. On the classification and nomenclature of baculoviruses: a proposal for revision. Archives of Virology. 2006;151(7):1257–1266. doi: 10.1007/s00705-006-0763-6. [DOI] [PubMed] [Google Scholar]
- 2.Miele SA, Garavaglia MJ, Belaich MN, Ghiringhelli PD. Baculovirus: molecular insights on their diversity and conservation. International Journal of Evolutionary Biology. 2011;2011:15 pages. doi: 10.4061/2011/379424.379424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Volkman LE, Summers MD. Autographa californica nuclear polyhedrosis virus: comparative infectivity of the occluded, alkali-liberated, and nonoccluded forms. Journal of Invertebrate Pathology. 1977;30(1):102–103. doi: 10.1016/0022-2011(77)90045-3. [DOI] [PubMed] [Google Scholar]
- 4.Engelhard EK, Kam-Morgan LNW, Washburn JO, Volkman LE. The insect tracheal system: a conduit for the systemic spread of Autographa californica M nuclear polyhedrosis virus. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(8):3224–3227. doi: 10.1073/pnas.91.8.3224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Li Z, Blissard GW. The pre-transmembrane domain of the Autographa californica multicapsid nucleopolyhedrovirus GP64 protein is critical for membrane fusion and virus infectivity. Journal of Virology. 2009;83(21):10993–11004. doi: 10.1128/JVI.01085-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mangor JT, Monsma SA, Johnson MC, Blissard GW. A GP64-null baculovirus pseudotyped with vesicular stomatitis virus G protein. Journal of Virology. 2001;75(6):2544–2556. doi: 10.1128/JVI.75.6.2544-2556.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hefferon KL, Oomens AGP, Monsma SA, Finnerty CM, Blissard GW. Host cell receptor binding by baculovirus GP64 and kinetics of virion entry. Virology. 1999;258(2):455–468. doi: 10.1006/viro.1999.9758. [DOI] [PubMed] [Google Scholar]
- 8.Monsma SA, Oomens AGP, Blissard GW. The GP64 envelope fusion protein is an essential baculovirus protein required for cell-to-cell transmission of infection. Journal of Virology. 1996;70(7):4607–4616. doi: 10.1128/jvi.70.7.4607-4616.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Blissard GW, Wenz JR. Baculovirus gp64 envelope glycoprotein is sufficient to mediate pH-dependent membrane fusion. Journal of Virology. 1992;66(11):6829–6835. doi: 10.1128/jvi.66.11.6829-6835.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kadlec J, Loureiro S, Abrescia NGA, Stuart DI, Jones IM. The postfusion structure of baculovirus gp64 supports a unified view of viral fusion machines. Nature Structural and Molecular Biology. 2008;15(10):1024–1030. doi: 10.1038/nsmb.1484. [DOI] [PubMed] [Google Scholar]
- 11.Ayres MD, Howard SC, Kuzio J, Lopez-Ferber M, Possee RD. The complete DNA sequence of Autographa californica nuclear polyhedrosis virus. Virology. 1994;202(2):586–605. doi: 10.1006/viro.1994.1380. [DOI] [PubMed] [Google Scholar]
- 12.Schultz KLW, Wetter JA, Fiore DC, Friesen PD. Transactivator IE1 is required for baculovirus early replication events that trigger apoptosis in permissive and nonpermissive cells. Journal of Virology. 2009;83(1):262–272. doi: 10.1128/JVI.01827-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Olson VA, Wetter JA, Friesen PD. The highly conserved basic domain I of baculovirus IE1 is required for hr enhancer DNA binding and hr-dependent transactivation. Journal of Virology. 2003;77(10):5668–5677. doi: 10.1128/JVI.77.10.5668-5677.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Pullen SS, Friesen PD. The CAGT motif functions as an initiator element during early transcription of the baculovirus transregulator ie-1. Journal of Virology. 1995;69(6):3575–3583. doi: 10.1128/jvi.69.6.3575-3583.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nissen MS, Friesen PD. Molecular analysis of the transcriptional regulatory region of an early baculovirus gene. Journal of Virology. 1989;63(2):493–503. doi: 10.1128/jvi.63.2.493-503.1989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Jarvis DL, Weinkauf C, Guarino LA. Immediate-early baculovirus vectors for foreign gene expression in transformed or infected insect cells. Protein Expression and Purification. 1996;8(2):191–203. doi: 10.1006/prep.1996.0092. [DOI] [PubMed] [Google Scholar]
- 17.Kovacs GR, Guarino LA, Summers MD. Novel regulatory properties of the IE1 and IE0 transactivators encoded by the baculovirus Autographa californica multicapsid nuclear polyhedrosis virus. Journal of Virology. 1991;65(10):5281–5288. doi: 10.1128/jvi.65.10.5281-5288.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jarvis DL, Fleming JAGW, Kovacs GR, Summers MD, Guarino LA. Use of early baculovirus promoters for continuous expression and efficient processing of foreign gene products in stably transformed lepidopteran cells. Nature Biotechnology. 1990;8(10):950–955. doi: 10.1038/nbt1090-950. [DOI] [PubMed] [Google Scholar]
- 19.Guarino LA, Summers MD. Functional mapping of a trans-activating gene required for expression of a baculovirus delayed-early gene. Journal of Virology. 1986;57(2):563–571. doi: 10.1128/jvi.57.2.563-571.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jiang SS, Chang IS, Huang LW, et al. Temporal transcription program of recombinant Autographa californica multiple nucleopolyhedrosis virus. Journal of Virology. 2006;80(18):8989–8999. doi: 10.1128/JVI.01158-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Volkman LE, Summers MD, Hsieh CH. Occluded and nonoccluded nuclear polyhedrosis virus grown in Trichoplusia ni: comparative neutralization, comparative infectivity, and in vitro growth studies. Journal of Virology. 1976;19(3):820–832. doi: 10.1128/jvi.19.3.820-832.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Marek M, van Oers MM, Devaraj FF, Vlak JM, Merten OW. Engineering of baculovirus vectors for the manufacture of virion-free biopharmaceuticals. Biotechnology and Bioengineering. 2011;108(5):1056–1067. doi: 10.1002/bit.23028. [DOI] [PubMed] [Google Scholar]
- 23.Ooi BG, Miller LK. Regulation of host RNA levels during baculovirus infection. Virology. 1988;166(2):515–523. doi: 10.1016/0042-6822(88)90522-3. [DOI] [PubMed] [Google Scholar]
- 24.Nobiron I, O’Reilly DR, Olszewski JA. Autographa californica nucleopolyhedrovirus infection of Spodoptera frugiperda cells: a global analysis of host gene regulation during infection, using a differential display approach. Journal of General Virology. 2003;84(11):3029–3039. doi: 10.1099/vir.0.19270-0. [DOI] [PubMed] [Google Scholar]
- 25.Salem TZ, Zhang F, Xie Y, Thiem SM. Comprehensive analysis of host gene expression in Autographa californica nucleopolyhedrovirus-infected Spodoptera frugiperda cells. Virology. 2011;412(1):167–178. doi: 10.1016/j.virol.2011.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Beniya H, Funk CJ, Rohrmann GF, Weaver RF. Purification of a virus-induced RNA polymerase from Autographa californica nuclear polyhedrosis virus-infected Spodoptera frugiperda cells that accurately initiates late and very late transcription in vitro. Virology. 1996;216(1):12–19. doi: 10.1006/viro.1996.0029. [DOI] [PubMed] [Google Scholar]
- 27.Yang CL, Stetler DA, Weaver RF. Structural comparison of the Autographa californica nuclear polyhedrosis virus-induced RNA polymerase and the three nuclear RNA polymerases from the host, Spodoptera frugiperda . Virus Research. 1991;20(3):251–264. doi: 10.1016/0168-1702(91)90079-b. [DOI] [PubMed] [Google Scholar]
- 28.Huh NE, Weaver RF. Identifying the RNA polymerases that synthesize specific transcripts of the Autographa californica nuclear polyhedrosis virus. Journal of General Virology. 1990;71(1):195–201. doi: 10.1099/0022-1317-71-1-195. [DOI] [PubMed] [Google Scholar]
- 29.Fuchs LY, Woods MS, Weaver RF. Viral transcription during Autographa californica nuclear polyhedrosis virus infection: a novel RNA polymerase induced in infected Spodoptera frugiperda cells. Journal of Virology. 1983;48(3):641–646. doi: 10.1128/jvi.48.3.641-646.1983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Passarelli AL, Guarino LA. Baculovirus late and very late gene regulation. Current Drug Targets. 2007;8(10):1103–1115. doi: 10.2174/138945007782151324. [DOI] [PubMed] [Google Scholar]
- 31.Knebel-Mörsdorf D, Quadt I, Li Y, Montier L, Guarino LA. Expression of baculovirus late and very late genes depends on LEF-4, a component of the viral RNA polymerase whose guanyltransferase function is essential. Journal of Virology. 2006;80(8):4168–4173. doi: 10.1128/JVI.80.8.4168-4173.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Guarino LA, Xu B, Jin J, Dong W. A virus-encoded RNA polymerase purified from baculovirus-infected cells. Journal of Virology. 1998;72(10):7985–7991. doi: 10.1128/jvi.72.10.7985-7991.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Crouch EA, Cox LT, Morales KG, Passarelli AL. Inter-subunit interactions of the Autographa californica M nucleopolyhedrovirus RNA polymerase. Virology. 2007;367(2):265–274. doi: 10.1016/j.virol.2007.05.026. [DOI] [PubMed] [Google Scholar]
- 34.Miller LK, Lingg AJ, Bulla LA. Bacterial, viral, and fungal insecticides. Science. 1983;219(4585):715–721. doi: 10.1126/science.219.4585.715. [DOI] [PubMed] [Google Scholar]
- 35.Miller LK. Genetically engineered insect virus pesticides: present and future. Journal of Invertebrate Pathology. 1995;65(3):211–216. doi: 10.1006/jipa.1995.1032. [DOI] [PubMed] [Google Scholar]
- 36.Inceoglu AB, Kamita SG, Hinton AC, et al. Recombinant baculoviruses for insect control. Pest Management Science. 2001;57(10):981–987. doi: 10.1002/ps.393. [DOI] [PubMed] [Google Scholar]
- 37.Szewczyk B, Hoyos-Carvajal L, Paluszek M, Skrzecz I, Lobo De Souza M. Baculoviruses—re-emerging biopesticides. Biotechnology Advances. 2006;24(2):143–160. doi: 10.1016/j.biotechadv.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 38.Crouch EA, Passarelli AL. Genetic requirements for homologous recombination in Autographa californica nucleopolyhedrovirus. Journal of Virology. 2002;76(18):9323–9334. doi: 10.1128/JVI.76.18.9323-9334.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Martin DW, Weber PC. DNA replication promotes high-frequency homologous recombination during Autographa californica multiple nuclear polyhedrosis virus infection. Virology. 1997;232(2):300–309. doi: 10.1006/viro.1997.8573. [DOI] [PubMed] [Google Scholar]
- 40.Smith GE, Summers MD, Fraser MJ. Production of human beta interferon in insect cells infected with a baculovirus expression vector. Molecular and Cellular Biology. 1983;3(12):2156–2165. doi: 10.1128/mcb.3.12.2156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pennock GD, Shoemaker C, Miller LK. Strong and regulated expression of Escherichia coli beta-galactosidase in insect cells with a baculovirus vector. Molecular and Cellular Biology. 1984;4(3):399–406. doi: 10.1128/mcb.4.3.399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kitts PA, Ayres MD, Possee RD. Linearization of baculovirus DNA enhances the recovery of recombinant virus expression vectors. Nucleic Acids Research. 1990;18(19):5667–5672. doi: 10.1093/nar/18.19.5667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zuidema D, Klinge-Roode EC, van Lent JWM, Vlak JM. Construction and analysis of an Autographa californica nuclear polyhedrosis virus mutant lacking the polyhedral envelope. Virology. 1989;173(1):98–108. doi: 10.1016/0042-6822(89)90225-0. [DOI] [PubMed] [Google Scholar]
- 44.Vialard J, Lalumiere M, Vernet T, et al. Synthesis of the membrane fusion and hemagglutinin proteins of measles virus, using a novel baculovirus vector containing the β-galactosidase gene. Journal of Virology. 1990;64(1):37–50. doi: 10.1128/jvi.64.1.37-50.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vlak JM, Schouten A, Usmany M, et al. Expression of cauliflower mosaic virus gene I using a baculovirus vector based upon the p10 gene and a novel selection method. Virology. 1990;179(1):312–320. doi: 10.1016/0042-6822(90)90299-7. [DOI] [PubMed] [Google Scholar]
- 46.Zuidema D, Schouten A, Usmany M, et al. Expression of cauliflower mosaic virus gene I in insect cells using a novel polyhedrin-based baculovirus expression vector. Journal of General Virology. 1990;71(10):2201–2209. doi: 10.1099/0022-1317-71-10-2201. [DOI] [PubMed] [Google Scholar]
- 47.Noad RJ, Stewart M, Boyce M, Celma CC, Willison KR, Roy P. Multigene expression of protein complexes by iterative modification of genomic bacmid DNA. BMC Molecular Biology. 2009;10, article 87 doi: 10.1186/1471-2199-10-87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Loudon PT, Hirasawa T, Oldfield S, Murphy M, Roy P. Expression of the outer capsid protein VP5 of two bluetongue viruses, and synthesis of chimeric double-shelled virus-like particles using combinations of recombinant baculoviruses. Virology. 1991;182(2):793–801. doi: 10.1016/0042-6822(91)90620-q. [DOI] [PubMed] [Google Scholar]
- 49.French TJ, Marshall JJA, Roy P. Assembly of double-shelled, viruslike particles of bluetongue virus by the simultaneous expression of four structure proteins. Journal of Virology. 1990;64(12):5695–5700. doi: 10.1128/jvi.64.12.5695-5700.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.French TJ, Roy P. Synthesis of bluetongue virus (BTV) corelike particles by a recombinant baculovirus expressing the two major structural core proteins of BTV. Journal of Virology. 1990;64(4):1530–1536. doi: 10.1128/jvi.64.4.1530-1536.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Fuchs B, Hecker D, Scheidtmann KH. Phosphorylation studies on rat p53 using the baculovirus expression system. Manipulation of the phosphorylation state with okadaic acid and influence on DNA binding. European Journal of Biochemistry. 1995;228(3):625–639. doi: 10.1111/j.1432-1033.1995.0625m.x. [DOI] [PubMed] [Google Scholar]
- 52.Nyunoya H, Akagi T, Ogura T, Maeda S, Shimotohno K. Evidence for phosphorylation of trans-activator p40(x) of human T-cell leukemia virus type 1 produced in insect cells with a baculovirus expression vector. Virology. 1988;167(2):538–544. [PubMed] [Google Scholar]
- 53.Maekawa S, Matsuura Y, Nakamura S. Expression and myristoylation of NAP-22 using a baculovirus transfer vector system. Biochimica et Biophysica Acta. 1994;1218(1):119–122. doi: 10.1016/0167-4781(94)90113-9. [DOI] [PubMed] [Google Scholar]
- 54.Kloc M, Reddy B, Crawford S, Etkin LD. A novel 110-kDa maternal CAAX box-containing protein from Xenopus is palmitoylated and isoprenylated when expressed in baculovirus. The Journal of Biological Chemistry. 1991;266(13):8206–8212. [PubMed] [Google Scholar]
- 55.Jarvis DL, Finn EE. Biochemical analysis of the N-glycosylation pathway in baculovirus-infected lepidopteran insect cells. Virology. 1995;212(2):500–511. doi: 10.1006/viro.1995.1508. [DOI] [PubMed] [Google Scholar]
- 56.Hillar A, Jarvis DL. Re-visiting the endogenous capacity for recombinant glycoprotein sialylation by baculovirus-infected Tn-4h and DpN1 cells. Glycobiology. 2010;20(10):1323–1330. doi: 10.1093/glycob/cwq099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Harrison RL, Jarvis DL. Protein N-glycosylation in the baculovirus-insect cell expression system and engineering of insect cells to produce “mammalianized” recombinant glycoproteins. Advances in Virus Research. 2006;68:159–191. doi: 10.1016/S0065-3527(06)68005-6. [DOI] [PubMed] [Google Scholar]
- 58.Hill DR, Aumiller JJ, Shi X, Jarvis DL. Isolation and analysis of a baculovirus vector that supports recombinant glycoprotein sialylation by SfSWT-1 cells cultured in serum-free medium. Biotechnology and Bioengineering. 2006;95(1):37–47. doi: 10.1002/bit.20945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Luckow VA, Lee SC, Barry GF, Olins PO. Efficient generation of infectious recombinant baculoviruses by site-specific transposon-mediated insertion of foreign genes into a baculovirus genome propagated in Escherichia coli . Journal of Virology. 1993;67(8):4566–4579. doi: 10.1128/jvi.67.8.4566-4579.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Airenne KJ, Peltomaa E, Hytönen VP, Laitinen OH, Ylä-Herttuala S. Improved generation of recombinant baculovirus genomes in Escherichia coli . Nucleic acids research. 2003;31(17, article e101) doi: 10.1093/nar/gng102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Pijlman GP, Dortmans JCFM, Vermeesch AMG, et al. Pivotal role of the non-hr origin of DNA replication in the genesis of defective interfering baculoviruses. Journal of Virology. 2002;76(11):5605–5611. doi: 10.1128/JVI.76.11.5605-5611.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Wang H, Deng F, Pijlman GP, et al. Cloning of biologically active genomes from a Helicoverpa armigera single-nucleocapsid nucleopolyhedrovirus isolate by using a bacterial artificial chromosome. Virus Research. 2003;97(2):57–63. doi: 10.1016/j.virusres.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 63.Simón O, Williams T, Asensio AC, et al. Sf29 gene of Spodoptera frugiperda multiple nucleopolyhedrovirus is a viral factor that determines the number of virions in occlusion bodies. Journal of Virology. 2008;82(16):7897–7904. doi: 10.1128/JVI.00099-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Miao Y, Zhang Y, Nakagaki K, et al. Expression of spider flagelliform silk protein in Bombyx mori cell line by a novel Bac-to-Bac/BmNPV baculovirus expression system. Applied Microbiology and Biotechnology. 2006;71(2):192–199. doi: 10.1007/s00253-005-0127-2. [DOI] [PubMed] [Google Scholar]
- 65.Datsenko KA, Wanner BL. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proceedings of the National Academy of Sciences of the United States of America. 2000;97(12):6640–6645. doi: 10.1073/pnas.120163297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Carstens EB, Chan H, Yu H, Williams GV, Casselman R. Genetic analyses of temperature-sensitive mutations in baculovirus late expression factors. Virology. 1994;204(1):323–337. doi: 10.1006/viro.1994.1537. [DOI] [PubMed] [Google Scholar]
- 67.Partington S, Yu H, Lu A, Carstens EB. Isolation of temperature sensitive mutants of Autographa californica nuclear polyhedrosis virus: phenotype characterization of baculovirus mutants defective in very late gene expression. Virology. 1990;175(1):91–102. doi: 10.1016/0042-6822(90)90189-x. [DOI] [PubMed] [Google Scholar]
- 68.Liu G, Carstens EB. Site-directed mutagenesis of the AcMNPV p 143 gene: effects on baculovirus DNA replication. Virology. 1999;253(1):125–136. doi: 10.1006/viro.1998.9485. [DOI] [PubMed] [Google Scholar]
- 69.Thomas CJ, Gooday GW, King LA, Possee RD. Mutagenesis of the active site coding region of the Autographa californica nucleopolyhedrovirus chiA gene. Journal of General Virology. 2000;81(5):1403–1411. doi: 10.1099/0022-1317-81-5-1403. [DOI] [PubMed] [Google Scholar]
- 70.Lin G, Blissard GW. Analysis of an Autographa californica nucleopolyhedrovirus lef-11 knockout: Lef-11 is essential for viral DNA replication. Journal of Virology. 2002;76(6):2770–2779. doi: 10.1128/JVI.76.6.2770-2779.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Bideshi DK, Federici BA. The Trichoplusia ni granulovirus helicase in unable to support replication of Autographa californica multicapsid nucleopolyhedrovirus in cells and larvae of T. ni . Journal of General Virology. 2000;81(6):1593–1599. doi: 10.1099/0022-1317-81-6-1593. [DOI] [PubMed] [Google Scholar]
- 72.Takahashi N, Yoshikura H, Kobayashi I. An Escherichia coli strain, BJ5183, that shows highly efficient conservative (two-progeny) DNA double-strand break repair of restriction breaks. Gene. 2003;303(1-2):89–97. doi: 10.1016/s0378-1119(02)01107-1. [DOI] [PubMed] [Google Scholar]
- 73.Shen H, Chen K, Yao Q, et al. Characterization of Bombyx mori nucleopolyhedrovirus orf74, a novel gene involved in virulence of virus. Virus Genes. 2009;38(3):487–494. doi: 10.1007/s11262-009-0350-5. [DOI] [PubMed] [Google Scholar]
- 74.Huang J, Hao B, Deng F, Sun X, Wang H, Hu Z. Open reading frame Bm21 of Bombyx mori nucleopolyhedrovirus is not essential for virus replication in vitro, but its deletion extends the median survival time of infected larvae. Journal of General Virology. 2008;89(4):922–930. doi: 10.1099/vir.0.83504-0. [DOI] [PubMed] [Google Scholar]
- 75.Wang Y, Sathish N, Hollow C, Yuan Y. Functional characterization of Kaposi’s sarcoma-associated herpesvirus open reading frame K8 by bacterial artificial chromosome-based mutagenesis. Journal of Virology. 2011;85(5):1943–1957. doi: 10.1128/JVI.02060-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Sathish N, Yuan Y. Functional characterization of Kaposi’s sarcoma-associated herpesvirus small capsid protein by bacterial artificial chromosome-based mutagenesis. Virology. 2010;407(2):306–318. doi: 10.1016/j.virol.2010.08.017. [DOI] [PubMed] [Google Scholar]
- 77.Zhu FX, Li X, Zhou F, Gao SJ, Yuan Y. Functional characterization of Kaposi’s sarcoma-associated herpesvirus ORF45 by bacterial artificial chromosome-based mutagenesis. Journal of Virology. 2006;80(24):12187–12196. doi: 10.1128/JVI.01275-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yao H, Osterrieder N, O’Callaghan DJ. Generation and characterization of an EICP0 null mutant of equine herpesvirus 1. Virus Research. 2003;98(2):163–172. doi: 10.1016/j.virusres.2003.09.007. [DOI] [PubMed] [Google Scholar]
- 79.Petherbridge L, Howes K, Baigent SJ, et al. Replication-competent bacterial artificial chromosomes of Marek’s disease virus: novel tools for generation of molecularly defined herpesvirus vaccines. Journal of Virology. 2003;77(16):8712–8718. doi: 10.1128/JVI.77.16.8712-8718.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Trapp S, Osterrieder N, Keil GM, Beer M. Mutagenesis of a bovine herpesvirus type 1 genome cloned as an infectious bacterial artificial chromosome: analysis of glycoprotein E and G double deletion mutants. Journal of General Virology. 2003;84(2):301–306. doi: 10.1099/vir.0.18682-0. [DOI] [PubMed] [Google Scholar]
- 81.Tischer BK, Kaufer BB, Sommer M, Wussow F, Arvin AM, Osterrieder N. A self-excisable infectious bacterial artificial chromosome clone of varicella-zoster virus allows analysis of the essential tegument protein encoded by ORF9. Journal of Virology. 2007;81(23):13200–13208. doi: 10.1128/JVI.01148-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Rueda P, Fominaya J, Langeveld JPM, Bruschke C, Vela C, Casal JI. Effect of different baculovirus inactivation procedures on the integrity and immunogenicity of porcine parvovirus-like particles. Vaccine. 2000;19(7-8):726–734. doi: 10.1016/s0264-410x(00)00259-0. [DOI] [PubMed] [Google Scholar]
- 83.Boyce FM, Bucher NLR. Baculovirus-mediated gene transfer into mammalian cells. Proceedings of the National Academy of Sciences of the United States of America. 1996;93(6):2348–2352. doi: 10.1073/pnas.93.6.2348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Carbonell LF, Miller LK. Baculovirus interaction with nontarget organisms: a virus-borne reporter gene is not expressed in two mammalian cell lines. Applied and Environmental Microbiology. 1987;53(7):1412–1417. doi: 10.1128/aem.53.7.1412-1417.1987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Marek M, Merten OW, Galibert L, Vlak JM, van Oers MM. Baculovirus VP80 protein and the F-actin cytoskeleton interact and connect the viral replication factory with the nuclear periphery. Journal of Virology. 2011;85(11):5350–5362. doi: 10.1128/JVI.00035-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Luckow VA. Baculovirus systems for the expression of human gene products. Current Opinion in Biotechnology. 1993;4(5):564–572. doi: 10.1016/0958-1669(93)90078-b. [DOI] [PubMed] [Google Scholar]
- 87.Pijlman GP, van Schinjndel JE, Vlak JM. Spontaneous excision of BAC vector sequences from bacmid-derived baculovirus expression vectors upon passage in insect cells. Journal of General Virology. 2003;84(10):2669–2678. doi: 10.1099/vir.0.19438-0. [DOI] [PubMed] [Google Scholar]
- 88.Pijlman GP, de Vrij J, van den End FJ, Vlak JM, Martens DE. Evaluation of baculovirus expression vectors with enhanced stability in continuous cascaded insect-cell bioreactors. Biotechnology and Bioengineering. 2004;87(6):743–753. doi: 10.1002/bit.20178. [DOI] [PubMed] [Google Scholar]
- 89.Pijlman GP, Roode EC, Fan X, et al. Stabilized baculovirus vector expressing a heterologous gene and GP64 from a single bicistronic transcript. Journal of Biotechnology. 2006;123(1):13–21. doi: 10.1016/j.jbiotec.2005.10.022. [DOI] [PubMed] [Google Scholar]
- 90.Kitts PA, Possee RD. A method for producing recombinant baculovirus expression vectors at high frequency. BioTechniques. 1993;14(5):810–817. [PubMed] [Google Scholar]
- 91.Zhao Y, Chapman DA, Jones IM. Improving baculovirus recombination. Nucleic Acids Research. 2003;31(2, article E6) doi: 10.1093/nar/gng006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Hitchman RB, Possee RD, Crombie AT, et al. Genetic modification of a baculovirus vector for increased expression in insect cells. Cell Biology and Toxicology. 2010;26(1):57–68. doi: 10.1007/s10565-009-9133-y. [DOI] [PubMed] [Google Scholar]
- 93.Hitchman RB, Possee RD, King LA. Baculovirus expression systems for recombinant protein production in insect cells. Recent Patents on Biotechnology. 2009;3(1):46–54. doi: 10.2174/187220809787172669. [DOI] [PubMed] [Google Scholar]
- 94.Vanarsdall AL, Mikhailov VS, Rohrmann GF. Characterization of a baculovirus lacking the DBP (DNA-binding protein) gene. Virology. 2007;364(2):475–485. doi: 10.1016/j.virol.2007.03.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Yu M, Carstens EB. Identification of a domain of the baculovirus Autographa californica multiple nucleopolyhedrovirus single-strand DNA-binding protein LEF-3 essential for viral DNA replication. Journal of Virology. 2010;84(12):6153–6162. doi: 10.1128/JVI.00115-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Su J, Lung O, Blissard GW. The Autographa californica multiple nucleopolyhedrovirus lef-5 gene is required for productive infection. Virology. 2011;416(1-2):54–64. doi: 10.1016/j.virol.2011.04.019. [DOI] [PubMed] [Google Scholar]
- 97.Lin G, Blissard GW. Analysis of an Autographa californica multicapsid nucleopolyhedrovirus lef-6-null virus: LEF-6 is not essential for viral replication but appears to accelerate late gene transcription. Journal of Virology. 2002;76(11):5503–5514. doi: 10.1128/JVI.76.11.5503-5514.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Okano K, Vanarsdall AL, Rohrmann GF. Characterization of a baculovirus lacking the alkaline nuclease gene. Journal of Virology. 2004;78(19):10650–10656. doi: 10.1128/JVI.78.19.10650-10656.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Okano K, Vanarsdall AL, Rohrmann GF. A baculovirus alkaline nuclease knockout construct produces fragmented DNA and aberrant capsids. Virology. 2007;359(1):46–54. doi: 10.1016/j.virol.2006.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Vanarsdall AL, Okano K, Rohrmann GF. Characterization of the replication of a baculovirus mutant lacking the DNA polymerase gene. Virology. 2005;331(1):175–180. doi: 10.1016/j.virol.2004.10.024. [DOI] [PubMed] [Google Scholar]
- 101.Stewart TM, Huijskens I, Willis LG, Theilmann DA. The Autographa californica multiple nucleopolyhedrovirus ie0-ie1 gene complex is essential for wild-type virus replication, but either IE0 or IE1 can support virus growth. Journal of Virology. 2005;79(8):4619–4629. doi: 10.1128/JVI.79.8.4619-4629.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Lung OY, Cruz-Alvarez M, Blissard GW. Ac23, an envelope fusion protein homolog in the baculovirus Autographa californica multicapsid nucleopolyhedrovirus, is a viral pathogenicity factor. Journal of Virology. 2003;77(1):328–339. doi: 10.1128/JVI.77.1.328-339.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Lung O, Westenberg M, Vlak JM, Zuidema D, Blissard GW. Pseudotyping Autographa californica multicapsid nucleopolyhedrovirus (AcMNPV): F proteins from group II NPVs are functionally analogous to AcMNPV GP64. Journal of Virology. 2002;76(11):5729–5736. doi: 10.1128/JVI.76.11.5729-5736.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Fang M, Nie Y, Harris S, Erlandson MA, Theilmann DA. Autographa californica multiple nucleopolyhedrovirus core gene ac96 encodes a per os infectivity factor (pif-4) Journal of Virology. 2009;83(23):12569–12578. doi: 10.1128/JVI.01141-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zhou W, Yao L, Xu H, Yan F, Qi Y. The function of envelope protein p74 from Autographa californica multiple nucleopolyhedrovirus in primary infection to host. Virus Genes. 2005;30(2):139–150. doi: 10.1007/s11262-004-5623-4. [DOI] [PubMed] [Google Scholar]
- 106.Wang M, Tuladhar E, Shen S, et al. Specificity of baculovirus P6.9 basic DNA-binding proteins and critical role of the C terminus in virion formation. Journal of Virology. 2010;84(17):8821–8828. doi: 10.1128/JVI.00072-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Liu C, Li Z, Wu W, et al. Autographa californica multiple nucleopolyhedrovirus ac53 plays a role in nucleocapsid assembly. Virology. 2008;382(1):59–68. doi: 10.1016/j.virol.2008.09.003. [DOI] [PubMed] [Google Scholar]
- 108.Vanarsdall AL, Okano K, Rohrmann GF. Characterization of the role of very late expression factor 1 in baculovirus capsid structure and DNA processing. Journal of Virology. 2006;80(4):1724–1733. doi: 10.1128/JVI.80.4.1724-1733.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Vanarsdall AL, Okano K, Rohrmann GF. Characterization of a baculovirus with a deletion of vlf-1. Virology. 2004;326(1):191–201. doi: 10.1016/j.virol.2004.06.003. [DOI] [PubMed] [Google Scholar]
- 110.Wu W, Lin T, Pan L, et al. Autographa californica multiple nucleopolyhedrovirus nucleocapsid assembly is interrupted upon deletion of the 38K gene. Journal of Virology. 2006;80(23):11475–11485. doi: 10.1128/JVI.01155-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Vanarsdall AL, Pearson MN, Rohrmann GF. Characterization of baculovirus constructs lacking either the Ac 101, Ac 142, or the Ac 144 open reading frame. Virology. 2007;367(1):187–195. doi: 10.1016/j.virol.2007.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Lin L, Wang J, Deng R, Ke J, Wu H, Wang X. ac109 is required for the nucleocapsid assembly of Autographa californica multiple nucleopolyhedrovirus. Virus Research. 2009;144(1-2):130–135. doi: 10.1016/j.virusres.2009.04.010. [DOI] [PubMed] [Google Scholar]
- 113.Fang M, Dai X, Theilmann DA. Autographa californica multiple nucleopolyhedrovirus EXON0 (ORF141) is required for efficient egress of nucleocapsids from the nucleus. Journal of Virology. 2007;81(18):9859–9869. doi: 10.1128/JVI.00588-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Dai X, Stewart TM, Pathakamuri JA, Li Q, Theilmann DA. Autographa californica multiple nucleopolyhedrovirus exon0 (orf141), which encodes a RING finger protein, is required for efficient production of budded virus. Journal of Virology. 2004;78(18):9633–9644. doi: 10.1128/JVI.78.18.9633-9644.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Ke J, Wang J, Deng R, Wang X. Autographa californica multiple nucleopolyhedrovirus ac66 is required for the efficient egress of nucleocapsids from the nucleus, general synthesis of preoccluded virions and occlusion body formation. Virology. 2008;374(2):421–431. doi: 10.1016/j.virol.2007.12.033. [DOI] [PubMed] [Google Scholar]
- 116.McCarthy CB, Dai X, Donly C, Theilmann DA. Autographa californica multiple nucleopolyhedrovirus ac142, a core gene that is essential for BV production and ODV envelopment. Virology. 2008;372(2):325–339. doi: 10.1016/j.virol.2007.10.019. [DOI] [PubMed] [Google Scholar]
- 117.Yuan M, Wu W, Liu C, et al. A highly conserved baculovirus gene p48 (ac103) is essential for BV production and ODV envelopment. Virology. 2008;379(1):87–96. doi: 10.1016/j.virol.2008.06.015. [DOI] [PubMed] [Google Scholar]
- 118.Hu Z, Yuan M, Wu W, Liu C, Yang K, Pang Y. Autographa californica multiple nucleopolyhedrovirus ac76 is involved in intranuclear microvesicle formation. Journal of Virology. 2010;84(15):7437–7447. doi: 10.1128/JVI.02103-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Nie Y, Fang M, Theilmann DA. Autographa californica multiple nucleopolyhedrovirus core gene ac92 (p33) is required for efficient budded virus production. Virology. 2011;409(1):38–45. doi: 10.1016/j.virol.2010.09.023. [DOI] [PubMed] [Google Scholar]
- 120.de Jong J, Arif BM, Theilmann DA, Krell PJ. Autographa californica multiple nucleopolyhedrovirus me53 (ac140) is a nonessential gene required for efficient budded-virus production. Journal of Virology. 2009;83(15):7440–7448. doi: 10.1128/JVI.02390-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wang Y, Wu W, Li Z, et al. ac18 is not essential for the propagation of Autographa californica multiple nucleopolyhedrovirus. Virology. 2007;367(1):71–81. doi: 10.1016/j.virol.2007.05.017. [DOI] [PubMed] [Google Scholar]
- 122.Urakawa T, Roy P. Bluetongue virus tubules made in insect cells by recombinant baculoviruses: expression of the NS1 gene of bluetongue virus serotype 10. Journal of Virology. 1988;62(11):3919–3927. doi: 10.1128/jvi.62.11.3919-3927.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Loudon PT, Roy P. Assembly of five bluetongue virus proteins expressed by recombinant baculoviruses: inclusion of the largest protein VP1 in the core and virus-like particles. Virology. 1991;180(2):798–802. doi: 10.1016/0042-6822(91)90094-r. [DOI] [PubMed] [Google Scholar]
- 124.Stewart M, Bhatia Y, Athmaran TN, et al. Validation of a novel approach for the rapid production of immunogenic virus-like particles for bluetongue virus. Vaccine. 2010;28(17):3047–3054. doi: 10.1016/j.vaccine.2009.10.072. [DOI] [PubMed] [Google Scholar]
- 125.Smith RH, Levy JR, Kotin RM. A simplified baculovirus-AAV expression vector system coupled with one-step affinity purification yields high-titer rAAV stocks from insect cells. Molecular Therapy. 2009;17(11):1888–1896. doi: 10.1038/mt.2009.128. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Negrete A, Yang LC, Mendez AF, Levy JR, Kotin RM. Economized large-scale production of high yield of rAAV for gene therapy applications exploiting baculovirus expression system. Journal of Gene Medicine. 2007;9(11):938–948. doi: 10.1002/jgm.1092. [DOI] [PubMed] [Google Scholar]
- 127.Overton HA, Fujii Y, Price IR, Jones IM. The protease and gag gene products of the human immunodeficiency virus: authentic cleavage and post-translational modification of an insect cell expression system. Virology. 1989;170(1):107–116. doi: 10.1016/0042-6822(89)90357-7. [DOI] [PubMed] [Google Scholar]
- 128.Zeng CQY, Wentz MJ, Cohen J, Estes MK, Ramig RF. Characterization replicase activity of double-layered and single-layered rotavirus-like particles expressed from baculovirus recombinants. Journal of Virology. 1996;70(5):2736–2742. doi: 10.1128/jvi.70.5.2736-2742.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Mena JA, Ramírez OT, Palomares LA. Population kinetics during simultaneous infection of insect cells with two different recombinant baculoviruses for the production of rotavirus-like particles. BMC Biotechnology. 2007;7, article 39 doi: 10.1186/1472-6750-7-39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Emery VC, Bishop DHL. The development of multiple expression vectors for high level synthesis of eukaryotic proteins: expression of LCMV-N and acNPV polyhedrin protein by a recombinant baculovirus. Protein Engineering, Design and Selection. 1987;1(4):359–366. doi: 10.1093/protein/1.4.359. [DOI] [PubMed] [Google Scholar]
- 131.Weyer U, Possee RD. A baculovirus dual expression vector derived from the Autographa californica nuclear polyhedrosis virus polyhedrin and p10 promoters: co-expression of two influenza virus genes in insect cells. Journal of General Virology. 1991;72(12):2967–2974. doi: 10.1099/0022-1317-72-12-2967. [DOI] [PubMed] [Google Scholar]
- 132.Belyaev AS. High-level expression of five foreign genes by a single recombinant baculovirus. Gene. 1995;156(2):229–233. doi: 10.1016/0378-1119(95)00050-g. [DOI] [PubMed] [Google Scholar]
- 133.Belyaev AS, Roy P. Development of baculovirus triple and quadruple expression vectors: co-expression of three or four bluetongue virus proteins and the synthesis of bluetongue virus-like particles in insect cells. Nucleic Acids Research. 1993;21(5):1219–1223. doi: 10.1093/nar/21.5.1219. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Berger I, Fitzgerald DJ, Richmond TJ. Baculovirus expression system for heterologous multiprotein complexes. Nature Biotechnology. 2004;22(12):1583–1587. doi: 10.1038/nbt1036. [DOI] [PubMed] [Google Scholar]
- 135.Fitzgerald DJ, Schaffitzel C, Berger P, et al. Multiprotein expression strategy for structural biology of eukaryotic complexes. Structure. 2007;15(3):275–279. doi: 10.1016/j.str.2007.01.016. [DOI] [PubMed] [Google Scholar]
- 136.Fitzgerald DJ, Berger P, Schaffitzel C, Yamada K, Richmond TJ, Berger I. Protein complex expression by using multigene baculoviral vectors. Nature Methods. 2006;3(12):1021–1032. doi: 10.1038/nmeth983. [DOI] [PubMed] [Google Scholar]
- 137.Oppenheim AB, Rattray AJ, Bubunenko M, Thomason LC, Court DL. In vivo recombineering of bacteriophage λ by PCR fragments and single-strand oligonucleotides. Virology. 2004;319(2):185–189. doi: 10.1016/j.virol.2003.11.007. [DOI] [PubMed] [Google Scholar]
- 138.Copeland NG, Jenkins NA, Court DL. Recombineering: a powerful new tool for mouse functional genomics. Nature Reviews Genetics. 2001;2(10):769–779. doi: 10.1038/35093556. [DOI] [PubMed] [Google Scholar]


