Abstract
Human milk oligosaccharides (HMO) constitute the third most abundant class of molecules in breast milk. Since infants lack the enzymes required for milk glycan digestion, this group of carbohydrates passes undigested to the lower part of the intestinal tract, where they can be consumed by specific members of the infant gut microbiota. We review proposed mechanisms for the depletion and metabolism of HMO by two major bacterial genera within the infant intestinal microbiota, Bifidobacterium and Bacteroides
Keywords: Bacteroides, Bifidobacterium, human milk oligosaccharides, infant gut
Introduction
From the moment a newborn leaves the mother’s womb, it is exposed to an environment full of microbes. An apparently chaotic succession of bacteria colonizes the sterile niche of the neonatal gut, and over the course of the first year or two of life the intestine achieves an adult-like microbial composition. Aerobic or facultative anaerobic bacteria that are capable of growth within the oxygen-replete intestine are favoured early in an infant’s life. Anaerobic bacteria with limited or no aerotolerance, such as Bifidobacterium, Bacteroides or Clostridium, become established as oxygen levels decrease over the ensuing months [1]. Many additional factors such as sanitary conditions, delivery mode or antibiotic use drive the fluctuations of the microbial community during the first months [1]. Little is known of the mechanisms that connect these and other relevant factors to microbiota assembly.
Culture-independent enumeration using 16S ribosomal RNA sequences has revealed that consumption of breast milk influences the microbial landscape of the infant gut [2,3]. Through lactation, the mother provides the infant with compounds that may act as prebiotics, which selectively modify gut microbial composition. As potential prebiotics, these compounds could provide beneficial effects in the gut and contribute to the well-being of the newborn [4]. In vitro fermentation studies have clearly demonstrated that human milk oligosaccharides (HMO), a very abundant class of molecules in breast milk, play a major role in promoting the growth of bifidobacteria and Bacteroides [5]. While most infants are well equipped to digest lactose present in breast milk, degradation of HMO requires an extensive set of glycoside hydrolases and intestinal membrane transporters that the infant lacks in the small intestine. Milk glycans pass undigested to the lower part of the intestinal tract and can enrich the gut microbiota with members that consume complex carboxydrates [6].
More than 200 different oligosaccharide structures have been found in the milk of a single donor. Each oligosaccharide has a common lactose reduced end, elongated with units of N-acetyllactosamine (Galβ1-4GlcNAc) or lacto-N-biose I units (LNB) (Galβ1-3GlcNAc). In addition, these glycans can be fucosylated and/or sialylated (Fig. 1 shows a summary of the linkages found in branched and linear HMO) [7]. Fucosyl linkages in these structures are dependent on the action of at least three different fucosyltransferases in the mother’s mammary glands, and the presence of a specific type of fucosyltransferases is determined by the mother’s secretor and Lewis genes [8]. For example, non-secretor mothers (i.e. those that lack a functional fucosyltransferase-2) are unable to modify HMO with an α-1-2-linked fucose residue. Recent reviews highlight the complexity of HMO and variation in HMO content over time and between individuals [7,9].
FIG. 1.
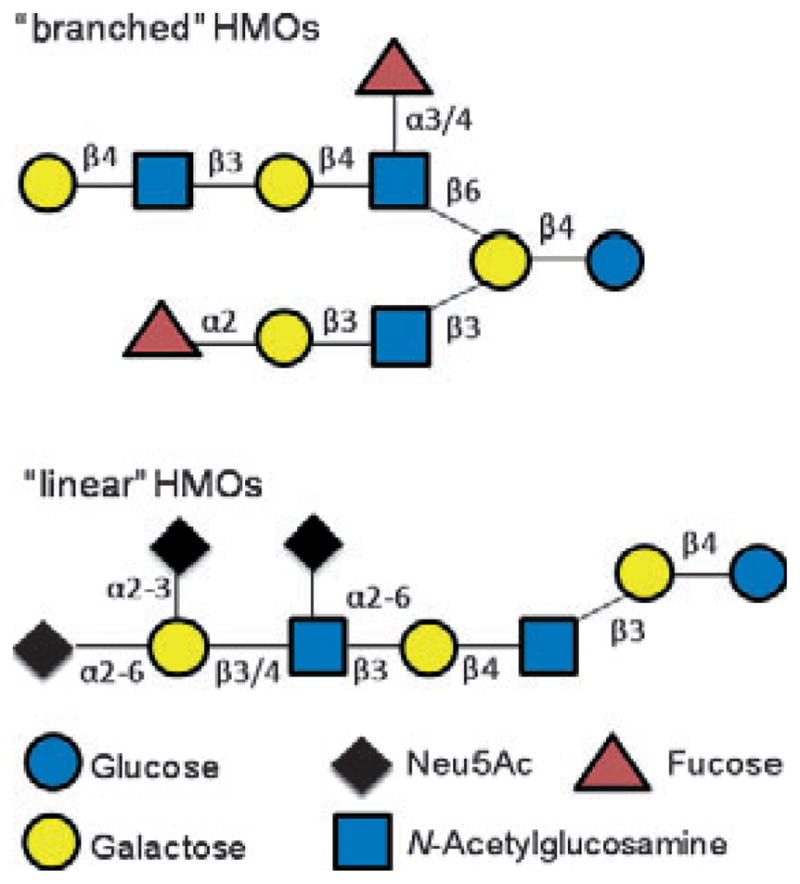
Summary of HMO structures, based on the work of Wu et al. [7].
To date, two major groups of commensals found in the infant gut, Bifidobacterium and Bacteroides, have been described as effective consumers of breast milk glycans [5]. The availability of the genome sequences of several Bacteroides and Bifidobacterium strains and the development of analytical methods to monitor the depletion of specific HMO, such as HPLC chip/time of flight MS technology, have recently expanded our knowledge of the different strategies employed by microbes to consume HMO.
Consumption of HMO by Bifidobacterium
Bifidobacterium acquisition and metabolism of HMO have been topics of interest in recent years, since this genus has been found in high concentration in breast-fed infant faeces [10]. Many bifidobacterial genomes encode a large proportion of oligosaccharide processing and transporting genes clustered within conserved loci. These loci typically contain regulatory elements, ABC transporters, carbohydrate binding proteins and glycoside hydrolases. Genome sequence from the infant commensal Bifidobacterium longum subsp. infantis (Bif. infantis), a strain adept at HMO consumption, reveals the existence of five HMO-related loci [11]. The largest of these clusters (43 kb) contains several glycoside hydrolases, ABC transporters and extracellular solute binding proteins. Absence of solute binding proteins in this locus results in the lack of ability to grow in HMO [12]. Matrix assisted laser desorption ionization Fourier transform ion cyclotron resonance (MALDI-FTICR) MS analysis of glycan consumption during growth in breast milk glycans has revealed that Bif. infantis efficiently consumes a subset of HMO. Lacto-N-tetraose (Galβ1-3GlcNAcβ1-3Galβ1-4Glc) and its isomer lacto-N-neotetraose (Galβ1-4GlcNAcβ1-3Galβ1-4Glc), which are considered short-chain oligosaccharides and are consistently present during lactation in very high concentration, are imported into the Bif. infantis cell [8]. Specific intracellular glycoside hydrolases, such as fucosidases, sialidases or galactosidases, degrade the oligosaccharides to generate monosaccharides that enter the fructose-6-phosphate phosphoketolase central metabolic pathway.
Analysis of other bifidobacterial genomes suggests a broad range of strategies for consuming milk glycans different to the one described for Bif. infantis. Strains belonging to the species Bif. bifidum express the enzyme lacto-N-biosidase, which is absent in the Bif. infantis genome. Expression of extracellular lacto-N-biosidase results in the cleavage of lacto-N-tetraose outside the cells into two disaccharides. The resulting LNB and lactose units are then imported. Once imported, the action of lacto-N-biose phosphorylase converts LNB into the monosaccharides galactose and N-acetylglucosamine. Other strains of bifidobacteria, such as those belonging to the species Bif. breve or Bif. longum subsp. longum, are unable to use diverse HMO but can grow using short chain oligosaccharides, such as lacto-N-neotetraose or LNB [13].
Consumption of HMO by Bacteroides
Members of the genus Bacteroides constitute a prevalent genus within the adult Western gut microbiota, and recent studies confirm that Bacteroides may also dominate the intestinal microbiota of some infants [3]. Bacteroides are known for their ability to utilize a wide variety of oligosaccharides from the intestinal environment, including dietary polysaccharides such as xylan, fructan or starch and host-derived glycans, especially those associated with intestinal mucus. This extraordinary saccharolytic ability is attributable to specialized machinery encoded by the polysaccharide utilization loci (PULs) within the Bacteroides genomes [14]. PULs often encode sensor regulators, glycoside hydrolases and homologues of two outer membrane proteins involved in binding and importing starch (SusC and SusD). Each PUL within a Bacteroides genome appears to be responsible for sensing and harvesting a distinct class of oligosaccharide or polysaccharide. PULs are likely to be involved in the consumption of HMO.
HMO use has been reported for Bacteroides fragilis and B. vulgatus. Analysis of HMO consumption by MALDI-FTICR MS shows that, unlike the bias toward use of short-chain glycans exhibited by Bifidobacterium species, Bacteroides efficiently consume a broad range of HMO glycans [5]. The more efficient use of HMO by B. fragilis compared with B. vulgatus suggests that some Bacteroides species may be better adapted to take advantage of HMO during infant gut colonization [15]. Whether HMO or other selective forces have driven the evolution of the HMO use pathways and the extent of pathway conservation between species are important issues that remain to be determined.
The Bacteroides HMO utilization model is summarized in Fig. 2. Bacteroides bind complex oligosaccharides, such as mucin glycans, on the cell surface prior to an initial surface hydrolysis, which enables the transit of a resulting oligosaccharide through the outer membrane SusC-like porins into the periplasm for further degradation [15]. We hypothesize that, due to high structural similarity found between mucin glycans and HMOs, Bacteroides may follow a similar strategy to consume HMOs.
FIG. 2.

Model of accessing HMO by Bacteroides.
Expression of glycoside hydrolases and transporters appears to be a common theme for milk glycan consumption by Bacteroides and Bifidobacteria [14]. Table 1 summarizes the carbohydrate active enzymes putatively involved in HMO consumption encoded in the genomes of four HMO-utilizing strains: B. fragilis, B. vulgatus, Bif. bifidum and Bif. infantis.
TABLE 1.
Annotated gene coding for glycoside hydrolases putatively involved in HMO degradation as described at the CAZy database (http://www.cazy.org)
| GH2 | GH16 | GH18 | GH20 | GH29 | GH33 | GH95 | |
|---|---|---|---|---|---|---|---|
| Bacteroides fragilis ATCC25285 | 15 | 6 | 2 | 12 | 9 | 3 | 4 |
| Bacteroides vulgatus ATCC8483 | 25 | 1 | 2 | 8 | 8 | 2 | 4 |
| Bifidobacterium infantis ATCC15697 | 2 | – | 1 | 3 | 3 | 2 | 1 |
| Bifidobacterium bifidum NCIMB 4117 | 3 | – | – | 4 | 1 | 1 | 1 |
The selected glycoside hydrolase families include the following activities: GH2, α-galactosidase; GH16, endo-β-1,4-galactosidase; GH18, endo-β-N-acetylglycosaminidase; GH20, β-hexosaminidase; GH29, α-1,3/4-fucosidase; GH33, sialidase; GH95, α-1,2-fucosidase.
Conclusions
Over the course of human evolution, it is likely that significant selective pressure has shaped the composition of breast milk to provide the maximum advantage for the newborn. Since microbes appear to compete for infant intestinal colonization yet differ in their relative ability to confer fitness upon the host (e.g. via maturation of the neonatal immune system or by providing protection against pathogens), it seems likely that milk content may be optimized to promote beneficial species. The ability of certain Bifidobacterium strains to efficiently use HMO suggests that production of milk oligosaccharides by the mother may be a strategy to ensure the presence of this group of bacteria in the infant gut. The consumption of HMO by Bacteroides species suggests that milk glycans may be attracting multiple groups of intestine adapted mutualists to the infant intestine. Bacteroides play an essential role within the distal gut in the degradation of the fibre consumed by the adult host. We hypothesize that by providing HMO the mother ensures the presence of this group of bacteria in the infant intestine, conferring stability and adaptability to the microbiota during the transition to a post-weaning diet, when Bacteroides are required as a major component of the intestinal microbial community. This possibility is supported by recent metagenomic studies that have revealed an abundance of plant polysaccharide-degrading glycoside hydrolases within the gut microbiomes of breast-fed infants [2,3].
While the beneficial influence of breast milk in infant health is well supported, much work remains to characterize the important molecules and to understand how they connect to infant health. Current efforts should focus on attaining a mechanistic understanding of the interaction between host, breast milk and microbial composition. Increased knowledge of how oligosaccharides may influence the gut composition will be required for developing therapies with specific glycans and microbes to optimize the microbial community when breast milk is not available and to maximize each newborn’s chance for a healthy life.
Acknowledgments
We thank Sara Fisher for editing the manuscript. This work was funded in part by grants from the National Institutes of Health NIDDK (R01-DK085025 and K01-DK077053).
Footnotes
Transparency Declaration
The authors declare that they have no conflicts of interest.
References
- 1.Adlerberth I. Factors influencing the establishment of the intestinal microbiota in infancy. Nestle Nutr Workshop Ser Pediatr Program. 2008;62:13–29. doi: 10.1159/000146245. [DOI] [PubMed] [Google Scholar]
- 2.Koening JE, Spor A, Scalfone N, et al. Succession of microbial consortia in the developing infant gut microbiome. Proc Natl Acad Sci U S A. 2011;108:4578–4585. doi: 10.1073/pnas.1000081107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vaishampayan PA, Kuehl JV, Froula JL, Morgan JL, Ochman H, Francino MP. Comparative metagenomics and population dynamics of the gut microbiota in mother and infant. Genome Biol Evol. 2010;2:53–66. doi: 10.1093/gbe/evp057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Roberfroid M. Prebiotics: the concept revisited. J Nut. 2007;137:830S–837S. doi: 10.1093/jn/137.3.830S. [DOI] [PubMed] [Google Scholar]
- 5.Marcobal A, Barboza M, Froehlich JW, et al. Consumption of human milk oligosaccharides by gut-related microbes. J Agric Food Chem. 2010;58:5334–5340. doi: 10.1021/jf9044205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.German JB, Freeman SL, Lebrilla CB, Mills DA. Human milk oligosaccharides: evolution, structures and bioselectivity as substrates for intestinal bacteria. Nestle Nutr Workshop Ser Pediatr Program. 2008;62:205–218. doi: 10.1159/000146322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wu S, Tao N, German JB, Grimm R, Lebrilla CB. Development of an annotated library of neutral human milk oligosaccharides. J Prot Res. 2010;9:4138–4151. doi: 10.1021/pr100362f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ninonuevo MR, Perkins PD, Francis J, et al. Daily variations in oligosaccharides of human milk determined by microfluidic chips and mass spectrometry. J Agric Food Chem. 2008;56:618–626. doi: 10.1021/jf071972u. [DOI] [PubMed] [Google Scholar]
- 9.Chichlowski M, German JB, Lebrilla CB, Mills D. The influence of milk oligosaccharides on micobiota of infants: opportunities for formula. Annu Rev Food Sci Technol. 2011;2:1–21. doi: 10.1146/annurev-food-022510-133743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ouwehand AC, Salminen S, Isolauri E. Probiotics: an overview of beneficial effects. Anton Leeuw Int J G. 2002;82:279–289. [PubMed] [Google Scholar]
- 11.Sela DA, Chapman J, Adeuya A, et al. The genome sequence of Bifidobacterium longum subsp infantis reveals adaptations for milk utilization within the infant microbiome. Proc Natl Acad Sci U S A. 2008;105:18964–18969. doi: 10.1073/pnas.0809584105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.LoCascio RG, Desai P, Sela DA, Weimer B, Mills DA. Comparative genomic hybridization of Bifidobacterium longum strains reveals broad conservation of milk utilization genes in subsp. infantis. Appl Environ Microbiol. 2010;76:7373–7381. doi: 10.1128/AEM.00675-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Sela DA, Mills DA. Nursing our microbiota: molecular linkages between bifidobacteria and milk oligosaccharides. Trends Microbiol. 2010;18:298–307. doi: 10.1016/j.tim.2010.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hooper LV, Midtvedt T, Gordon JI. How host–microbial interactions shape the nutrient environment of the mammalian intestine. Annu Rev Nutr. 2002;22:283–307. doi: 10.1146/annurev.nutr.22.011602.092259. [DOI] [PubMed] [Google Scholar]
- 15.Xu J, Mahowald MA, Ley RE, et al. Evolution of symbiotic bacteria in the distal human intestine. PLoS Biol. 2007;5:1574–1586. doi: 10.1371/journal.pbio.0050156. [DOI] [PMC free article] [PubMed] [Google Scholar]


