Saunders et al. use GRO-seq to map the RNA Pol II genomic distribution in early Drosophila embryos. The authors find that whereas some genes activated by the transcription factor Zelda are regulated by Pol II recruitment, others are regulated at the elongation step. Widespread pausing is detected throughout the bone morphogenetic protein (BMP) signaling pathway, with BMP target genes more highly paused than pathway components. This study suggests a new paradigm for Pol II pausing—that the major effect of pausing is on transcriptional levels, rather than timing.
Keywords: Drosophila embryo, axis patterning, zygotic transcription, promoter-proximal pausing, GRO-seq, BMP, Dpp signaling pathway
Abstract
Cascades of zygotic gene expression pattern the anterior–posterior (AP) and dorsal–ventral (DV) axes of the early Drosophila embryo. Here, we used the global run-on sequencing assay (GRO-seq) to map the genome-wide RNA polymerase distribution during early Drosophila embryogenesis, thus providing insights into how genes are regulated. We identify widespread promoter-proximal pausing yet show that the presence of paused polymerase does not necessarily equate to direct regulation through pause release to productive elongation. Our data reveal that a subset of early Zelda-activated genes is regulated at the level of polymerase recruitment, whereas other Zelda target and axis patterning genes are predominantly regulated through pause release. In contrast to other signaling pathways, we found that bone morphogenetic protein (BMP) target genes are collectively more highly paused than BMP pathway components and show that BMP target gene expression requires the pause-inducing negative elongation factor (NELF) complex. Our data also suggest that polymerase pausing allows plasticity in gene activation throughout embryogenesis, as transiently repressed and transcriptionally silenced genes maintain and lose promoter polymerases, respectively. Finally, we provide evidence that the major effect of pausing is on the levels, rather than timing, of transcription. These data are discussed in terms of the efficiency of transcriptional activation required across cell populations during developmental time constraints.
Patterning the axes of the Drosophila embryo relies on the activation of zygotic gene networks in response to maternal cues. Specification of the anterior–posterior (AP) axis is dependent on a segmentation gene cascade, with the gap genes activated first, followed by the pair-rule genes, then the segment polarity genes, and, finally, the homeotic genes (Niessing et al. 1997; Sanson 2001). Patterning of the dorsal–ventral (DV) axis is initiated by a maternal Dorsal nuclear gradient, which divides the embryo into three major tissues (Stathopoulos and Levine 2004). Subsequent signaling interactions subdivide these tissues into specific cell types. For example, Dorsal-mediated repression restricts decapentaplegic (dpp) transcription to the dorsal ectoderm of the embryo. Although Dpp protein, encoding a bone morphogenetic protein (BMP) ligand, is initially uniform in the dorsal ectoderm, it is redistributed to form a gradient that patterns this region into distinct cell fates (O'Connor et al. 2006).
The intricate expression patterns of key zygotic developmental genes require tight regulation of transcription. For transcription to occur, the promoter region is remodeled to an open chromatin structure that is accessible to the core transcription machinery. General transcription factors melt the promoter DNA and enable the RNA polymerase to initiate transcription. The transition to productive transcription elongation is facilitated by post-translational modification of the C-terminal domain (CTD) of the largest subunit of RNA polymerase II (Pol II). Various transcription factors interact with the modified CTD to govern the elongation rate, cotranscriptional processing of the RNA, and, finally, transcription termination. Transcription activators or repressors bound to enhancers regulate the initiation step by affecting preinitiation complex recruitment through specific protein interactions and/or altering nucleosome occupancy of the promoter region (Kadonaga 2004).
Although the need for recruitment and initiation of the transcription complex at promoters is absolute, another major point of control is at the level of elongation via Pol II pausing proximal to the promoter. During pausing, the initiated polymerase transcribes ∼20–50 nucleotides (nt) and then is held from further elongation by pause-inducing factors (Saunders et al. 2006). Transcription factors can establish a promoter-proximally paused complex, vary the duration and stability of the pause, or change the rate of escape into productive elongation (Fuda et al. 2009). Promoter-proximal pausing was initially characterized on the human myc gene (Bentley and Groudine 1986), HIV (Kao et al. 1987), and the Drosophila heat-shock gene hsp70 (Gilmour and Lis 1986; Rougvie and Lis 1988). More recently, the use of Pol II chromatin immunoprecipitation (ChIP) followed by genomic approaches has found that promoter-associated Pol II is widespread in mammalian and Drosophila genomes (Adelman and Lis 2012), particularly for developmental genes in Drosophila (Muse et al. 2007; Zeitlinger et al. 2007; Levine 2011; Gaertner et al. 2012). Pausing appears to be important for maintaining promoters in an open conformation through competition between paused Pol II and the +1 nucleosome (Gilchrist et al. 2008, 2010). Other hypothesized functions for pausing describe it as (1) critical for robust and synchronous transcription responses (Boettiger and Levine 2009), (2) a post-initiation checkpoint that allows for integration of more information from signaling pathways, and (3) a checkpoint that ensures proper preprocessing of nascent RNAs or maturation of the transcription complex itself (Adelman and Lis 2012).
Recent technological advances in the mapping of nascent or short RNAs have allowed Pol II pausing to be visualized with higher resolution, thus uncovering a mechanistic insight into promoter regulation (Core et al. 2008; Nechaev et al. 2010; Churchman and Weissman 2011; Menet et al. 2012). The global run-on sequencing assay (GRO-seq) provides a measure of the density and orientation of transcriptionally competent polymerases across the genome (Core et al. 2008) and affords an attractive balance between resolution, comprehensive genome coverage, and sensitivity (Core et al. 2008, 2012; Chopra et al. 2011; Min et al. 2011). Here, we used GRO-seq to examine the pausing and transcription dynamics of key developmental genes during early Drosophila embryogenesis. Our data determine the mechanisms of transcriptional regulation of key gene sets deployed during early embryogenesis and clearly distinguish between promoters that are regulated primarily through increased escape from pausing versus increased recruitment. Finally, our data also provide insight into the biological impact of pausing on the long-term expression of genes.
Results
Widespread early zygotic transcription and pausing at active genes
To generate a high-resolution profile of gene transcription in early Drosophila embryos, we performed GRO-seq analysis (Core et al. 2008). GRO-seq involves next-generation sequencing of nuclear run-on RNA molecules representing the very 3′ portion of nascent transcripts from paused or elongating RNA polymerases. Mapping these sequencing reads reveals the distribution and orientation of transcriptionally competent polymerases across the genome (Core et al. 2008). Time points of 2–2.5 h and 3–3.5 h after egg laying (AEL) were chosen to coincide with transcription of the gene networks required for AP and DV axis patterning (Sanson 2001; Stathopoulos and Levine 2004). Two biological replicates were analyzed for each time point; Supplemental Figure S1 shows the high correlation between replicates (R2 = 0.92–0.98). The numbers of rRNA GRO-seq reads indicated that the 2- to 2.5-h and 3- to 3.5-h time points had similar transcriptional activity, allowing their direct comparison (see the Materials and Methods). Evidence that the embryos collected at each of our time points were correctly staged and not contaminated with incorrectly staged embryos is shown in Supplemental Figure S2 (see also the Materials and Methods).
Analysis of transcription levels in 2- to 2.5-h embryos, based on normalized gene body read counts (gene body reads per kilobase per million [gbRPKM]) (see the Materials and Methods), reveals a range of transcriptional activity across the genome (Fig. 1A). Using a conservative statistical cutoff for gene body read density, we found that at least 62% (8752 of 14,065) of genes are transcriptionally active in 2- to 2.5-h embryos (Fig. 1B), which is more than double a previous estimate (Lott et al. 2011). The top 500 most transcriptionally active genes are shown in Supplemental Table S1. Of the genes we assigned as active at 2–2.5 h (Fig. 1B), 23 are part of a set of 27 genes previously categorized as being strictly maternal and lacking zygotic transcription (Arbeitman et al. 2002). With the higher sensitivity of GRO-seq, we detected zygotic transcription of the 23 genes at variable levels (Supplemental Fig. S3A–C) and confirmed early zygotic expression of one of these, CycA, using nascent RNA in situ hybridization (Supplemental Fig. S3D). Similarly, analysis of another set of >5000 genes defined as being maternally expressed with no early zygotic transcription (Lott et al. 2011) revealed that only 288 of these lack zygotic transcription at either of our two time points, as detected by GRO-seq (Supplemental Table S2).
Figure 1.
Transcriptional activity within the early embryo. (A) Chart showing the distribution of gbRPKMs across all genes in the Drosophila genome (see the Materials and Methods). (B) Pie chart displaying proportion of genes that are transcriptionally active (have a gbActPval of <0.01) or inactive (have a gbActPval of >0.01) at 2–2.5 h AEL. The number of active and inactive genes is displayed. (C) Chart showing the distribution of PIs across all genes in the Drosophila genome (see the Materials and Methods). (D) Pie chart displaying proportion of genes that are paused (have a pausing P-value of <0.01) or not paused (have a pausing P-value of >0.01) at 2–2.5 h AEL. (E) Chart showing the paused and not paused genes in D also separated depending on whether the genes are active or inactive. (F) UCSC genome browser images of GRO-seq data for an example of each class of gene at 2–2.5 h, with the percentage of genes in each category shown.
Next, we calculated the pausing index (PI) for each gene, which is the density of polymerases in the promoter region divided by that in the gene body (Core et al. 2008), to determine the number of genes that harbor paused polymerase in the early Drosophila embryo. The distribution of PI values across the genome at 2–2.5 h is shown in Figure 1C, with some genes having a very high PI value. Different scenarios can account for high PI values. In many cases, one paused polymerase occupies each promoter-proximal region at around position +40, although there is the potential to fit more where pausing is more dispersed and distal (Kwak et al. 2013). However, if only a few polymerases are present across the rest of the gene, then the polymerase density in the 250-base-pair (bp) promoter-proximal region will be vastly different from the polymerase density in a multikilobase gene body. Similarly, every gene copy could be occupied by a paused polymerase, but polymerases might only be present in the gene body in a subpopulation of cells, again leading to vastly different promoter and gene body polymerase densities. The complete list of paused genes at 2–2.5 h with the PI ranking is shown in Supplemental Table S3. Using a statistical cutoff of 0.01 for the PI (see the Materials and Methods), we found that more than half of the genes at 2–2.5 h AEL (55%; 7734 out of 14,065) have significant enrichment of GRO-seq density in the promoter-proximal region relative to the gene body (Fig. 1D). Our data also indicate that the majority (80%; 7036 out of 7734) of transcriptionally active genes have promoter-paused Pol II, whereas only a small proportion of inactive genes have paused polymerase (Fig. 1E). The four possible GRO-seq gene classes, as defined by the PI and gene body activity, and the proportion of genes in each are shown in Figure 1F. Due to the heterogeneity of the nucleus population analyzed, the paused and active profile for any given gene could be a mix of different pausing and activity states of the gene in expressing and nonexpressing cells across the embryo. To investigate whether actively transcribed genes are likely to be paused, we further analyzed the 7036 genes classified as paused and active from the 2- to 2.5-h embryo data set. Of these genes, 6253 are also active in a more homogenous population of S2 cells, with 4052 of these (65%) harboring paused Pol II (Core et al. 2012). This suggests that the paused and active profile that we identified in the embryo can be associated with individual genes bearing both paused and actively transcribing Pol II. We also note that some ubiquitously expressed genes are paused and active. It is also likely that paused Pol II is present when some genes are inactive (see below). Together, these data are consistent with Pol II pausing being important for the capability and maintenance of transcription activity.
The segmentation genes are regulated by pausing
To investigate transcriptional regulation of specific sets of genes, we initially focused on the segmentation genes, which pattern the AP axis. The classical gap, pair-rule, and segment polarity genes have moderate to high transcriptional activity at 2–2.5 h, whereas the homeotic genes have lower activity (Fig. 2A, Y-axis shows gbRPKM). To compare the proportion of polymerases in the promoter region of each segmentation gene, we color-coded the data points in the graph by the level of GRO-seq promoter reads (prRPKM) at 2–2.5 h (Fig. 2A). The gap, pair-rule, and segment polarity genes mostly have a very high polymerase density in the pause region, whereas the homeotic genes tend to have a lower density of polymerases in their promoters and, on average, in their gene bodies. The transcription profiles of individual examples from each set of genes are shown in Supplemental Figure S4, A–D. Thus, the segmentation genes in early embryos generally have a high density of promoter-proximal Pol II.
Figure 2.
Transcription dynamics of the segmentation genes. (A) Graph shows the gbRPKMs of the individual segmentation genes at 2–2.5 h. The points are color-coded for the 2- to 2.5-h prRPKMs; color denotes whether a gene is not paused (based on a pausing P-value of >0.01) or paused with a prRPKM within the top 0%–25%, 26%–50%, 51%–75%, or 76%–100% of paused genes. (B) Relative PIs of the gap, pair-rule, and segment polarity genes. Genes are categorized as not paused or paused with a PI within the top 25%, 25%–50%, or bottom 50% of all paused genes at 2–2.5 h or 3–3.5 h. See also Supplemental Table S4. (C,Ci) Graphs show the change in gbRPKM (C) versus the change in PI or prRPKM (Ci) from 2–2.5 h to 3–3.5 h for the gap, pair-rule, and segment polarity genes. The point highlighted in pink corresponds to ftz, which exhibits characteristics of regulation by polymerase recruitment and is excluded from the trend line.
By 3–3.5 h, the changes in transcriptional activity of the segmentation genes (Supplemental Fig. S4E) correlate with that of their mRNA levels observed after 3.5 h, as revealed by analysis of the modENCODE consortium RNA sequencing (RNA-seq) data (Supplemental Fig. S4F; The modENCODE Consortium et al. 2010; Graveley et al. 2011). Despite the altered transcriptional activity of some gap, pair-rule, and segment polarity genes at 3–3.5 h, these genes retain high promoter reads (Supplemental Fig. S4Ei). We visualized pausing of the segmentation genes by plotting their PI values, which show that, with the exception of some homeotic genes, all segmentation genes analyzed have promoter-paused Pol II at 2–2.5 h and 3–3.5 h (Fig. 2B; Supplemental Table S4).
The high PIs of the gap, pair-rule, and segment polarity genes prompted us to investigate whether they are regulated at the level of pause release. If a gene is regulated at release, as the gene body reads increase, the promoter reads remain similar or decrease, and the PI decreases (Supplemental Fig. S4G). Thus, there is a negative correlation between the change in gbRPKM versus the change in PI, and there is no correlation between the change in gene body read counts versus the change in prRPKM. When we plotted these data for the combined set of gap, pair-rule, and segment polarity genes, for most genes (except fushi tarazu [ftz]) (Fig. 2C,Ci, pink diamond), we found a strong negative correlation between the change in gbRPKM versus PI (Fig. 2C) and very little correlation between the change in gbRPKM versus prRPKM (Fig. 2Ci). This can also be seen simply when the raw reads are plotted: There are obvious changes in the gbRPKMs between 2–2.5 h and 3–3.5 h (Supplemental Fig. S4E) but little change in the prRPKMs (Supplemental Fig. S4H), as expected for regulation by pause release. Together, these data indicate that escape from the pause is an important regulatory step in the activation of most gap, pair-rule, and segment polarity genes during AP patterning.
Regulation of DV patterning genes
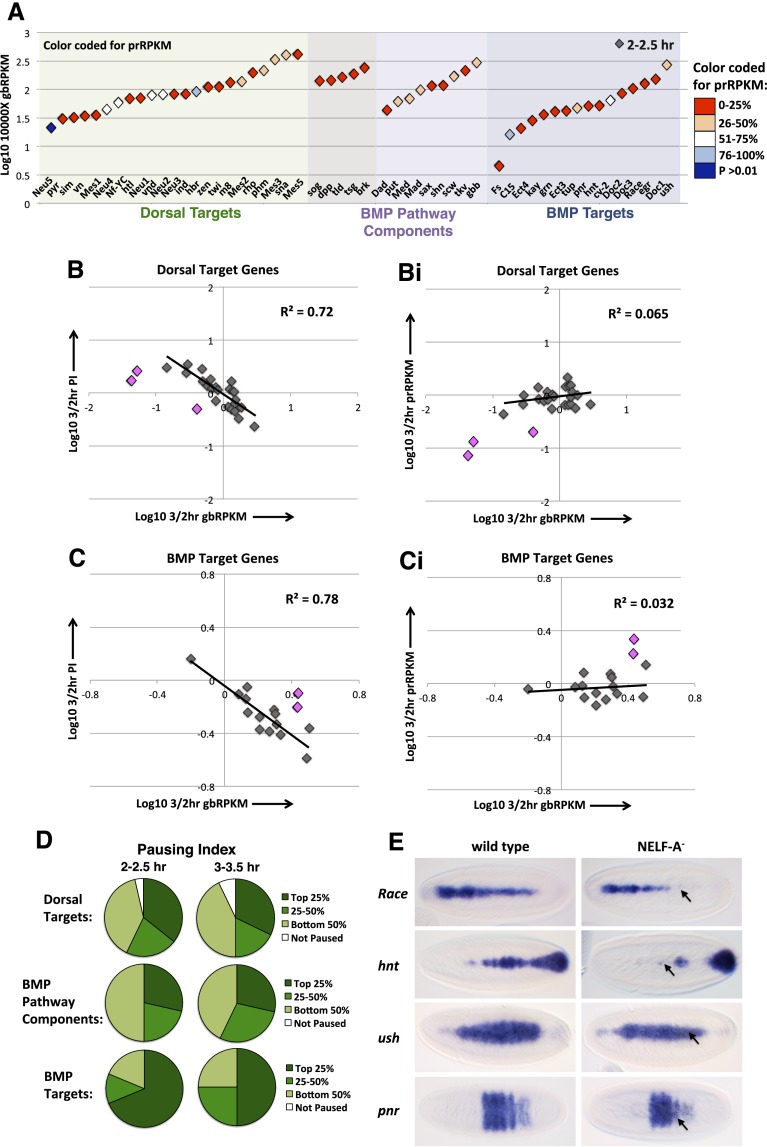
We next investigated the transcriptional dynamics and regulation of genes involved in DV patterning—in particular, targets of the Dorsal transcription factor, the BMP signaling pathway components, and BMP target genes (Stathopoulos and Levine 2004; O'Connor et al. 2006). Five of the BMP pathway components are Dorsal targets, and at 2–2.5 h, these two gene sets show, on average, slightly higher transcription levels than the BMP targets (Fig. 3A, Y-axis is gbRPKM). As described for the segmentation genes, many of the DV genes show high promoter read counts (Fig. 3A, color coding). University of California at Santa Cruz (UCSC) browser images for specific DV patterning genes are shown in Supplemental Figure S5, A–C. At 3–3.5 h, changes in the transcriptional activity of many of the DV genes were observed (Supplemental Fig. S5D), although most of these genes retain high promoter reads (Supplemental Fig. S5Di,E). The screw (scw) and twisted gastrulation (tsg) genes, encoding a BMP ligand and extracellular BMP-binding protein, respectively (see the X-axis of Supplemental Fig. S5G for the functions of BMP pathway components; O'Connor et al. 2006), and the Dorsal target gene Neuroectoderm-expressed 2 (Neu2) are highly transcribed at 2–2.5 h but shut off at 3–3.5 h (Supplemental Fig. S5D). These three genes even lose polymerases from the gene promoter, indicating complete transcriptional repression (Supplemental Fig. S5F). This repression results in a sharp decrease in the mRNA levels of these genes by 4–6 h AEL (Supplemental Fig. S5G; data not shown), as also observed by RNA in situ hybridization for scw transcripts (Arora et al. 1994).
Figure 3.
Transcription dynamics of the DV patterning genes. (A) Graph shows the gbRPKMs of the DV patterning genes at 2–2.5 h. The points are color coded for the 2- to 2.5-h prRPKMs; color denotes whether a gene is not paused (P > 0.01) or paused with a prRPKM within the top 0%–25%, 26%–50%, 51%–75%, or 76%–100% of paused genes. The five genes shaded in gray are both Dorsal targets and BMP pathway components. (B,Bi) Graphs show the change in gbRPKM (B) versus the change in PI or prRPKM (Bi) from 2–2.5 h to 3–3.5 h for the Dorsal target genes. Neu2, tsg, and zen (highlighted in pink) exhibit characteristics of regulation by polymerase recruitment and are excluded from the trend line. The gbRPKMs and prRPKMs of Mes1 and Neu1 do not change and are therefore absent from the chart. (C,Ci) As in B and Bi for the BMP target genes. Doc2 and C15 (highlighted in pink) exhibit characteristics of regulation by polymerase recruitment and are excluded from the trend line. (D) Pie charts show the proportion of DV patterning genes with PIs within the top 25%, or 25%–50% or bottom 50% of all paused genes at 2–2.5 h or 3–3.5 h. See also Supplemental Table S5. (E) RNA in situ hybridization for the Race, hnt, ush, and pnr mRNAs, activated in response to BMP signaling, in wild-type or NELF-A maternal and zygotic null embryos. Embryos are dorsal views with anterior to the left.
To address whether the DV genes are transcriptionally regulated at the level of pause release, we plotted the change in gbRPKM versus the change in prRPKM or PI. For the Dorsal and BMP target genes, there is generally a negative correlation between the changes in gbRPKM and PI (Fig. 3B,C) but little correlation between the changes in gbRPKM and prRPKM (Fig. 3Bi,Ci), consistent with regulation predominantly at the level of pause release (Supplemental Fig. S4G). Genes Neu2, tsg, zerknullt (zen), Dorsocross2 (Doc2), and C15, however, appear to be regulated at the recruitment step (Fig. 3B,Bi,C,Ci, pink diamonds). As mentioned above, five BMP pathway components are Dorsal targets that, with the exception of tsg, are regulated by pause release. Inference of the transcription regulatory mechanism of the remaining BMP pathway components was not possible due to half of the genes within this small category having similar transcriptional activity at both time points (data not shown).
Analysis of the PI values for DV genes reveals that all of the BMP pathway components and target genes are paused, as are most of the Dorsal targets (Fig. 3D; Supplemental Table S5). In particular, we were interested in how pausing distributed across the BMP pathway and its targets, as previous analysis of the NF-κB, JNK, and JAK–STAT signaling pathways and their gene targets in S2 cells found that a higher proportion of the signaling pathway components are paused compared with downstream targets (Gilchrist et al. 2012). In contrast, our data show that the BMP target genes exhibit stronger pausing than the BMP pathway components, with around two-thirds having PIs within the top 25% of paused genes across the genome at 2–2.5 h. At 3–3.5 h, the BMP target genes have slightly lower PIs overall compared with 2–2.5 h, but again, a greater proportion of BMP target genes are present in the top 25% paused category compared with BMP pathway components (Fig. 3D). In addition, in the early embryo, most targets of the NF-κB transcription factor Dorsal are paused (Fig. 3D), with one-third in the top 25% paused category, although we cannot compare this with pausing dynamics across the Toll–Dorsal signaling pathway, as these components are maternally expressed.
To determine whether the high pausing on signaling pathway targets in the early embryo reflects differences relating to the pathway–target set and/or system, we used our 2- to 2.5-h AEL data to analyze the subset of AMP genes examined by Gilchrist et al. (2012) in S2 cells in response to NF-κB signaling. Consistent with their findings, these genes are only modestly enriched for polymerase pausing in 2- to 2.5-h embryos, with only around half classified as paused based on the PI (Supplemental Fig. S5H; Supplemental Table S6). We therefore extended our analysis to a second embryonic signaling pathway active during our assayed time points: the STAT pathway (Zeidler et al. 2000). A large proportion of the STAT pathway components are also highly paused (Supplemental Fig. S5I; Supplemental Table S7). However, a set of STAT target genes (Tsurumi et al. 2011) is less paused than the STAT pathway components, as observed in S2 cells (Gilchrist et al. 2012), with a quarter lacking a pause in the early embryo (Supplemental Fig. S5I; Supplemental Table S7). Therefore, BMP and Dorsal target genes show higher pausing than the STAT signaling pathway targets and AMP genes tested. Thus, different signaling pathways and particular sets of targets might require different modes of regulation as a means to satisfy specific needs of the organism.
As many DV genes are paused, we investigated the expression patterns of a subset of these in embryos mutant for the NELF-A gene, encoding the A subunit of the pause-inducing negative elongation factor (NELF) complex (Narita et al. 2003; Wu et al. 2005). We found that the expression patterns of dpp and sog, encoding the main BMP ligand and its regulator, respectively (O'Connor et al. 2006), do not show major defects in embryos obtained from females carrying homozygous germline clones for the NELF-A[KG] mutation and lacking zygotic NELF-A activity (Supplemental Fig. S5J). This finding is similar to a previous observation that the expression of some pair-rule genes and the Dorsal target rhomboid is unaffected in the neuroectoderm in embryos lacking NELF subunits (Wang et al. 2010). However, we observed that BMP target gene expression is disrupted in approximately half of the embryos lacking maternal and zygotic NELF-A (Fig. 3E), suggesting that these genes are more dependent on paused Pol II for their activation.
Differential regulation of early Zelda-activated genes
The predominant mode of regulation identified for the AP and DV genes analyzed above is at the level of pause release; however, not all paused genes are regulated at this step. When we examined genes activated at the earliest stages of embryogenesis (1–2 h AEL) by Zelda (Zld), a key maternally deposited transcription factor involved in the activation of zygotic genes (Liang et al. 2008), most of these genes still have high transcription at 2–2.5 h (75% are in the top 25% most transcriptionally active category). However, many have lower promoter polymerase densities than the AP (excluding the homeotic) and DV genes (cf. color coding in Figs. 4A and 2A, 3A). As these genes are early activated Zld targets, their transcription levels and promoter polymerase densities are reduced at 3–3.5 h (Supplemental Fig. S6A,Ai). Analyses of how changes in gbRPKM correlate with that of prRPKM and PI revealed that two-thirds of these genes clearly sort into two groups: one that is regulated at the level of pause release (31% of the genes) (Fig. 4B,Bi) and one that is regulated by polymerase recruitment (37% of the genes) (Fig. 4C,Ci; Supplemental Fig. S6B–E). In terms of the latter, this gene set shows a positive correlation between gbRPKM and prRPKM but a much lower correlation between gbRPKM and PI (Fig. 4C,Ci; Supplemental Fig. S4G). The remaining one-third of Zld target genes could not be clearly assigned to either mode of regulation (Supplemental Fig. S6E). The PIs reveal that pausing is observed for the majority of Zld-regulated genes in the 2- to 2.5-h embryo, including those regulated by recruitment (Fig. 4D; Supplemental Table S8). Consistent with their mode of control, the recruitment-regulated genes then show lower pausing at 3–3.5 h, when their transcriptional activity has decreased (Fig. 4D). The observation that most of the recruitment-controlled genes are paused when active supports the concept that the major regulated step in transcription of paused genes is not always escape from the pause (Hah et al. 2011).
Figure 4.
Regulation of Zld target genes. (A) Graph shows gbRPKMs at 2–2.5 h for Zld target genes (Liang et al. 2008); the points are color-coded for prRPKM at 2–2.5 h, as detailed in the legend. Gene list is shown in Supplemental Figure S6E. (B,C) Graphs show the changes in gbRPKM versus PI (B,C) or prRPKM (Bi,Ci) from 2–2.5 h to 3–3.5 h for Zld genes regulated by pause release (B) or recruitment (C). Genes within these categories are listed in Supplemental Figure S6E. (D) Pie charts show proportion of Zld target genes with PIs within the top 25% or 25%–50% or bottom 50% of all paused genes at 2–2.5 h or 3–3.5 h. See also Supplemental Table S8.
We next considered whether the AP, DV, and Zld genes classified as paused and active in the heterogenous nucleus population from the embryo might be paused and inactive in nonexpressing cells. To address this, we analyzed the Zld genes regulated by pause release or recruitment using the GRO-seq data set from S2 cells, which represent a more homogenous embryonically derived cell population (Core et al. 2012). Almost all of the Zld genes regulated by pause release that are inactive in S2 cells have promoter-paused Pol II (Supplemental Fig. S6F). In contrast, the Zld genes regulated by Pol II recruitment are mostly not paused when inactive in S2 cells, as expected for activation at the level of polymerase recruitment. Similarly, a proportion of the AP and DV genes that we found to be regulated by pause release in the embryo is paused and inactive in S2 cells. Although the DV genes show the lowest proportion of paused and inactive genes in S2 cells, analysis of mutant embryos with altered nuclear Dorsal levels has previously shown that some Dorsal target genes are paused when inactive (Zeitlinger et al. 2007). Therefore, together, these data suggest that activation of some of the paused genes in the Drosophila embryo may involve a change from inactive and paused to active in particular cells, which is entirely consistent with a mechanism involving polymerase release from the pause (Chopra et al. 2009).
Pausing enables transcriptional plasticity
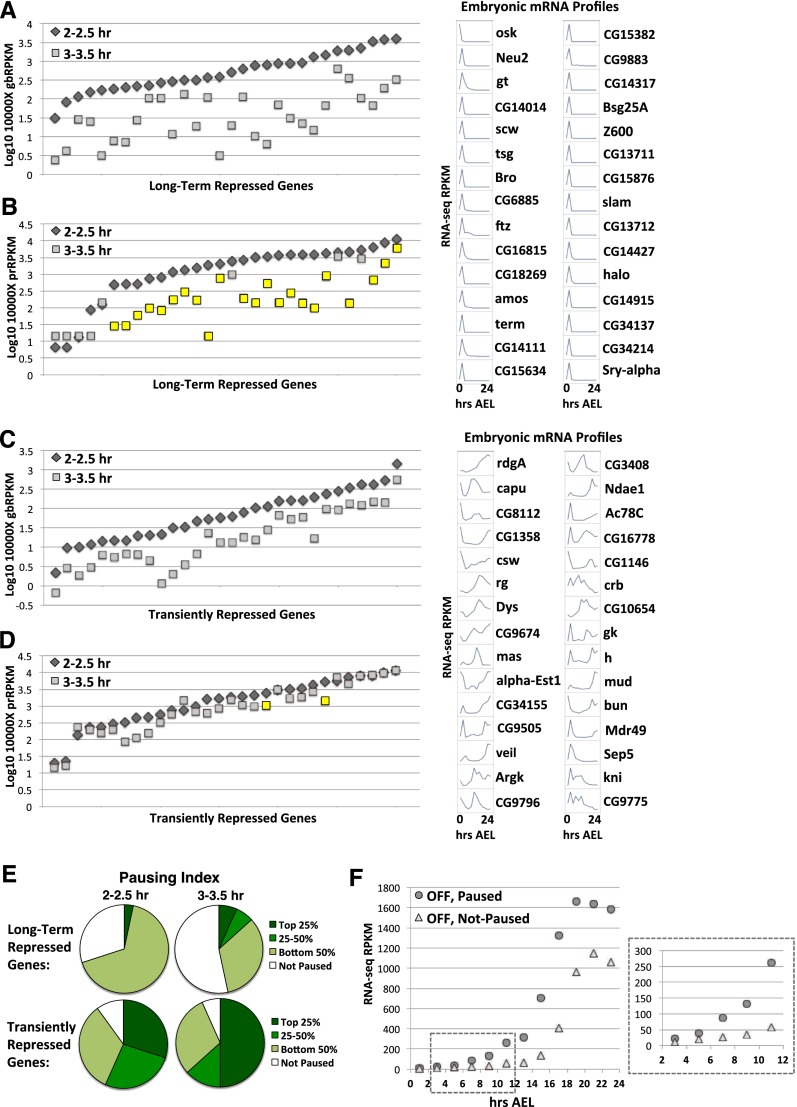
As some genes (scw and tsg) (Supplemental Fig. S5F) lose their promoter polymerases when they become transcriptionally repressed and others maintain promoter polymerases when inactive (class III genes), we further investigated the connection between promoter polymerase occupancy and transcriptional inactivity. First, we used RNA-seq data (The modENCODE Consortium et al. 2010) to select genes that are transcribed early but become shut off later in embryogenesis. Thirty genes that match this profile and have a statistically significant decrease in transcription from 2–2.5 to 3–3.5 h, including scw and tsg, are shown in Figure 5A (see also Supplemental Fig. S7A). The majority of these long-term repressed genes (73%; 22 out of 30) also have substantially reduced densities of promoter polymerases at 3–3.5 h (Fig. 5B). The genes in Figure 5, A and B, become completely shut off and do not reactivate transcription during embryogenesis. Therefore, we next used the RNA-seq data to select transiently repressed genes with a statistically significant decrease in transcription at 3–3.5 h as compared with 2–2.5 h but that then become more actively transcribed again sometime later in embryogenesis (Fig. 5C). In contrast to the long-term repressed genes, these transiently repressed genes have a similar level of polymerase density at their promoter at the two time points (Fig. 5D). Only two of the 30 transiently repressed genes (7%) have statistically significant promoter read changes, indicating that pausing is maintained at genes requiring adjustable transcription output. Investigation of the PIs for these two sets of genes reveals that there is qualitatively and quantitatively less pausing on the long-term repressed genes in early embryogenesis (Fig. 5E). These data suggest that high pausing set up on genes early facilitates flexibility in gene expression over long periods of development.
Figure 5.
Long-term consequences of pausing. (A,B) Graphs show the gbRPKMs (A) or prRPKMs (B) of long-term repressed genes at 2–2.5 h or 3–3.5 h. Genes are ordered by the 2- to 2.5-h gbRPKM (A) or prRPKM (B), low–high in both cases; therefore, the gene order differs in A and B. All gbRPKM changes are statistically significant (DESeq adjusted P-value < 0.2); statistically significant prRPKM changes are highlighted in yellow. The minicharts on the right display mRNA levels for each of the 30 genes during embryogenesis, derived from modENCODE data. Gene orders are listed in Supplemental Figure S7A. (C,D) As in A and B, but the data shown are for transiently repressed genes. (E) Pie charts show the proportion of long-term or transiently repressed genes with PIs within the top 25% or 25%–50% or bottom 50% of all paused genes at 2–2.5h or 3–3.5 h. (F) Graph shows the average RNA-seq RPKM for class III versus class IV zygotic genes throughout embryogenesis.
We also investigated the effect of pausing on the activation of zygotic genes in the embryo. For this, we focused on genes that are transcriptionally repressed in the early embryo and compared our class III (off, paused at 2–2.5 h) versus class IV (off, not paused at 2–2.5 h) genes (Fig. 1F) for the ability to become transcriptionally active during later embryogenesis. As shown in Figure 5F, genes that are paused at 2–2.5 h have, on average, higher transcription levels at every time point later in embryogenesis compared with genes that are completely unbound by polymerases at 2–2.5 h. However, the timing of transcription activation is similar between these two groups, with both increasing throughout embryogenesis (Fig. 5F). Therefore, the major effect of pausing appears to be on the levels of transcription rather than the timing of activation in that paused genes are not necessarily those destined to fire next in the sequence of developmental regulation. Consistent with this, genes that are paused and active during the early time points that we tested show, on average, higher mRNA levels later in embryogenesis than genes that are active but not paused during the same early time points (Supplemental Fig. S7B). These findings can be explained by synchronous gene activation in the presence of pausing (see the Discussion; Boettiger and Levine 2009). Together, our results suggest that pausing allows more robust transcriptional activity upon gene activation and is less prevalent on genes that are destined to be silenced.
Discussion
Our data detect extensive zygotic transcription in the early embryo, including of genes not previously known to be transcribed in the embryo, and show that most of the active genes display promoter-proximal pausing. We also reveal new mechanistic insight into transcription activation of specific gene sets. Some early Zld-activated genes are predominately regulated at the level of polymerase recruitment, whereas paused polymerase release is the main control point for others. Many Zld genes regulated at recruitment show promoter-proximal pausing, even though the regulated rate-limiting step for activation of these genes is polymerase recruitment. Similarly, many estrogen-responsive genes are paused in breast cancer cells, even though most are regulated through initiation rather than pausing (Hah et al. 2011). Therefore, on a subset of genes, pausing can be a prominent step in transcription without necessarily being the main point of regulation.
Zld has been described as a pioneer transcription factor that binds to a multitude of enhancers to establish or maintain an open chromatin structure that facilitates transcriptional activation of the zygotic genome (Harrison et al. 2011). Pioneer transcription factors can function actively to open chromatin and allow other factors to bind (Zaret and Carroll 2011). This may be how Zld functions at genes regulated by polymerase recruitment, with the chromatin-opening property of Zld at these enhancers bypassing the need to set up a paused polymerase to maintain an open chromatin conformation. Pioneer transcription factors can also function in a delayed, passive manner (Zaret and Carroll 2011), binding to an enhancer and priming it for later activation. This may be the role Zld is playing at the genes regulated by pause escape, with establishment of the pause as part of the priming mechanism.
Regulation at the level of pause release appears to be a major form of regulation for genes involved in AP and DV axis patterning. This is based on both the changes in our GRO-seq patterns during development and findings for individual genes in these sets (Wang et al. 2007; Zeitlinger et al. 2007; Chopra et al. 2009). In addition, the phenotypes of mutants in genes encoding pausing and escape factors support this conclusion. NELF-A mutant embryos arrest early due to cellularization defects or later due to abnormalities associated with germband retraction, including head defects and incomplete dorsal closure (Wang et al. 2010). The former phenotype may be due to a failure to activate the Zld targets involved in cellularization, whereas germband retraction defects are associated with disrupted BMP signaling (Frank and Rushlow 1996), and we showed defective BMP target gene expression in embryos lacking NELF-A. Similarly, mutations in lilliputian, a gene encoding a component of the super elongation complex (SEC) (Luo et al. 2012), phenocopy partial loss of dpp function (Su et al. 2001), and lilliputian mutant embryos have a pair-rule phenotype (Tang et al. 2001). The SEC contains the critical pause–release factor P-TEFb and stimulates productive elongation (Luo et al. 2012). Finally, embryos mutant for Spt5, which encodes a component of the pause-inducing DRB sensitivity-inducing factor (DSIF) complex (Wada et al. 1998), have disrupted expression patterns of pair-rule genes and segmentation defects (Jennings et al. 2004). Therefore, overall, the phenotypes associated with mutations in genes encoding factors involved in pausing regulation can be attributed, at least in part, to defects in the transcription activation of genes regulated by pause release.
Promoter polymerases are maintained or lost upon transcription down-regulation of transiently or long-term repressed genes, respectively. This suggests that pausing allows plasticity in gene expression during development. Transient repression can be mediated by repressors like Snail, Knirps, Runt, and Ftz, which appear to act by inhibiting the release of paused polymerases into productive elongation (Wang et al. 2007; Bothma et al. 2011; Li and Arnosti 2011). As a result, these genes will be poised for reactivation following relief of repression. In contrast, repressors that mediate complete gene silencing—e.g., Polycomb group repressors—might remove pausing as a prelude to chromatin compaction and long-term silencing. This is consistent with our observation that genes destined for silencing have lower pausing in early embryogenesis, possibly due to the presence of specific promoter elements that are unfavorable for high pausing. In support of Polycomb repressors removing pausing, Drosophila embryos mutant for extra sex combs, a PRC2 complex subunit, have enhanced pausing on a quarter of all genes, including those normally lacking a paused polymerase (Chopra et al. 2011). In addition, genes bound by Polycomb group complexes have reduced levels of paused polymerases in both the Drosophila embryo and mouse embryonic stem cells (Chopra et al. 2011; Min et al. 2011). Recent data suggest that, although the Polycomb-repressive H3K27me3 chromatin mark and paused Pol II can coexist on many genes during Drosophila development, for some genes, maintenance of the H3K27me3 mark correlates with loss of Pol II over time (Gaertner et al. 2012). It is also possible that loss of paused polymerase leads to chromatin condensation, followed by recruitment of Polycomb group repressors and gene silencing.
Last, we addressed whether pausing confers an advantage to gene activation. Our data suggest that the absence of pausing does not preclude early activation but tends to lead to lower mRNA levels. Therefore, the genes with paused Pol II are not necessarily the next genes to fire during the time course of development. Genes can be paused earlier than when they are activated and can maintain a paused Pol II through cycles of regulatory signals that activate or repress their transcription. A simple way to explain the positive influence of pausing on transcription levels relates to the idea that pausing facilitates transcriptional synchrony of gene activation (Boettiger and Levine 2009). In this scenario, within a cell population, pausing on a target gene will increase the probability of activation so that the majority of cells respond to a cue, leading to high mRNA levels. In contrast, if a cell population lacks pausing on a target gene, then, although cells can respond to the cue with the same initial timing, the need for polymerase recruitment would lower the probability of activation in any given cell. The resulting stochastic response would lead to a reduced mRNA level across the cell population, as we observed. Other functions proposed for pausing, such as chromatin accessibility, coupling of elongation to RNA processing events, or the organization of genes into chromatin domains (Li and Gilmour 2011; Adelman and Lis 2012), may contribute to the synchronous activation of genes in addition to being important for subsequent rounds of transcription.
Our data show that many Dorsal target genes are paused, as described previously (Zeitlinger et al. 2007), with pausing of these genes being linked to their synchronous activation (Boettiger and Levine 2009). Considering pausing in terms of promoting synchronous versus stochastic activation may also explain the widespread, high pausing that we detected on BMP target genes as well as pathway components. This is different from some other signaling pathways where pathway components are typically more paused than the target genes (Gilchrist et al. 2012). The BMP cascade is specialized in that there is a very narrow time window (∼1 h) in which localized pathway components must be activated, the gradient established, and then target genes induced before the cell movements associated with gastrulation commence (Stathopoulos and Levine 2004). Therefore, the BMP target genes, like the Dorsal targets, may be mostly paused to promote synchronous activation of genes in order to provide the efficiency in activation needed due to the time constraint. Consistent with this, although STAT also activates transcription in the early embryo, some of the STAT target genes that we tested show higher levels of expression later in embryogenesis, and these are the ones enriched in the lowest PI category for STAT targets (data not shown). It would be interesting to compare the pausing levels of the BMP pathway and target genes during wing imaginal disc patterning, which occurs over a much longer time period (Podos and Ferguson 1999). The short timing associated with early Drosophila embryogenesis in general may explain why our estimate of the proportion of paused genes (∼55%) exceeds the estimate of 30%–40% for human cell lines and mouse embryonic cell lines (Core et al. 2008; Min et al. 2011). We suggest that pausing conveys an advantage to gene activation across cell populations under time pressure conditions and/or during rapid differentiation.
Materials and methods
GRO-seq
Embryos were collected from young, well-fed yellow white flies at 25°C. Adult Drosophila flies were kept in multiple small collection bottles (∼700 adults per bottle), as, in our experience, adults are less prone to withholding embryos in these bottles than in large cages. Collection plates from the first 2 h were discarded to minimize collection of older, stored embryos. Flies were allowed to lay embryos on apple agar plates that were changed every 30 min, and the embryos were then aged for a further 2 h (for the 2- to 2.5-h time point) or 3 h (for the 3- to 3.5-h time point) at 25°C before immediate collection. Therefore, embryos were at least 2 or 3 h old, respectively. Collected embryos were rinsed thoroughly, flash-frozen, and stored at −80°C. Embryos were resuspended in cold buffer 1 (250 mM sucrose, 10 mM Tris at pH 7.5, 5 mM MgCl2, 0.1% Triton X-100, 1 mM EGTA, 5 mM DTT, 1× protease inhibitor cocktail [Roche], 1:5000 superasin [Ambion]) plus 3 mM CaCl2, loose dounced 20 times, poured over mesh to remove large debris, and tight dounced a further 10 times at 4°C. Nuclei were pelleted at 700g for 10 min at 4°C and washed twice in buffer 1 and then once in freezing buffer (50 mM Tris at pH 8.0, 25% glycerol, 5 mM MgAc2, 0.1 mM EDTA, 5 mM DTT, 1× protease inhibitor cocktail [Roche], 1:2500 superasin [Ambion]). Nuclei were then resuspended in freezing buffer, flash-frozen, and stored at −80°C. In each run-on reaction, 7 × 107 to 30 × 107 nuclei were used. The run-on RNA library was prepared as in Core et al. (2008), with 25 min of base hydrolysis, and sequenced on the Illumina Genome Analyzer in the Core Laboratories Center at Cornell University.
Data analysis
Data were analyzed as in Core et al. (2012). Reads from the replicates were combined to provide the counts presented here; all P-value cutoffs are 0.01. For genes with multiple isoforms, the isoform with the lowest pausing P-value was used. We note that some pauses were missed due to inaccurate annotations. For plotting gbRPKMs, values were multiplied by 10,000 for aesthetic purposes. To avoid zeros for Log scales, pseudo gbRPKMs were used, where +1 was added to all gene body or promoter read counts. For the continuums in Figure 1, A and C, the log10 pseudo values (gbRPKM or PI) were divided into 10% portions, and the total number of genes in each portion was calculated. Labels on the X-axes are the nonlog pseudo values of these categories. The Drosophilia melanogaster dm3 genome assembly was used for all analyses and UCSC genome browser images (http://genome.ucsc.edu; Kent et al. 2002). The processed Zld (Liang et al. 2008) and STAT (Tsurumi et al. 2011) data sets were obtained from the respective studies. Zld target genes were those down-regulated more than threefold in the absence of Zld (Liang et al. 2008). Zld targets were categorized as regulated at recruitment or pause release based on the relationship between the change in gene body reads versus the change in promoter reads (Supplemental Fig. S4G). STAT targets were the top 40 down-regulated genes in a STAT mutant background classified as active with GRO-seq at 2–2.5 h (Tsurumi et al. 2011). Adjusted P-values (Padj) were generated using DESeq (version 1.10.1) (Anders and Huber 2010) on the separate replicate data. The long-term and transiently repressed genes decreased in GRO-seq gene body counts from 2–2.5 h to 3–3.5 h with Padj < 0.2. The genes were categorized based on their modENCODE expression profile. The modENCODE RPKMs were downloaded from FlyBase (http://www.flybase.org). The long-term, and transiently repressed gene sets presented in Figure 5, A–D, were selected based on having statistically significant changes in transcription between 2–2.5 h and 3–3.5 h. For Figure 5F, maternally expressed mRNAs were removed based on the modENCODE data having a read count >100 at the 0- to 2-h time point. The GRO-seq data have been deposited in NCBI's Gene Expression Omnibus (GEO) (Edgar et al. 2002) and are accessible through GEO series accession number GSE41611 (http://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE41611). The link to the GRO-seq data on the UCSC genome browser is available from http://ashelab.com/datasets.
Normalization of GRO-seq reads from the 2- to 2.5-h and 3- to 3.5-h time points
The GRO-seq data presented are the uniquely mapped reads. Repetitive reads, like those mapping as rRNA, were excluded from the analysis. It is possible that more genes are transcriptionally active at 3–3.5 h than at 2–2.5 h. Therefore, GRO-seq on exactly the same number of nuclei at these two time points could yield more uniquely mapped sequencing reads at 3–3.5 h. If this were the case, gene activity at 3–3.5 h could be underestimated when reads are normalized to the total uniquely mapped reads. However, the rRNA genes will be active at both time points, and so we can expect these genes to be expressed at equal levels at both times. Therefore, to determine whether we needed to normalize the data between the time points, we determined the correction factor for normalizing to total transcription activity. To do this, we mapped all reads to the genome (including the rRNA) and expressed the unique reads (the presented GRO-seq data) as a fraction of the total transcriptome. With this method, we determined the correction factor to be 1.036. Since this value is essentially 1.0, no correction was required.
In situ hybridization
Nascent transcript in situs were performed as previously described (Ronshaugen and Levine 2004); mRNA in situs were performed using digU-labeled RNA probes and standard protocols. NELF-A[KG] flies are described in Wang et al. (2010).
Acknowledgments
We thank Mark Ashe and Annick Sawala for helpful discussions, Peter Gergen for the NELF flies, Tom Pettini and Matt Ronshaugen for help with the nascent RNA in situ, Leo Zeef for the DESeq analyses, and Mark Ashe and Andy Sharrocks for comments on the manuscript. This research was funded by a Wellcome Trust program grant to H.L.A. (092005/Z/10/Z), an NIH grant (GM25232) to J.T.L., an EMBO long-term fellowship to A.S. (ALTF 975-2008), and the Drosophila Core Research Facility at the Faculty of Life Sciences, University of Manchester, established through funds from the University and the Wellcome Trust (087742/Z/08/Z).
Footnotes
Supplemental material is available for this article.
Article is online at http://www.genesdev.org/cgi/doi/10.1101/gad.215459.113.
References
- Adelman K, Lis JT 2012. Promoter-proximal pausing of RNA polymerase II: Emerging roles in metazoans. Nat Rev Genet 13: 720–731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anders S, Huber W 2010. Differential expression analysis for sequence count data. Genome Biol 11: R106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbeitman MN, Furlong EE, Imam F, Johnson E, Null BH, Baker BS, Krasnow MA, Scott MP, Davis RW, White KP 2002. Gene expression during the life cycle of Drosophila melanogaster. Science 297: 2270–2275 [DOI] [PubMed] [Google Scholar]
- Arora K, Levine MS, O'Connor MB 1994. The screw gene encodes a ubiquitously expressed member of the TGF-β family required for specification of dorsal cell fates in the Drosophila embryo. Genes Dev 8: 2588–2601 [DOI] [PubMed] [Google Scholar]
- Bentley DL, Groudine M 1986. A block to elongation is largely responsible for decreased transcription of c-myc in differentiated HL60 cells. Nature 321: 702–706 [DOI] [PubMed] [Google Scholar]
- Boettiger AN, Levine M 2009. Synchronous and stochastic patterns of gene activation in the Drosophila embryo. Science 325: 471–473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bothma JP, Magliocco J, Levine M 2011. The snail repressor inhibits release, not elongation, of paused Pol II in the Drosophila embryo. Curr Biol 21: 1571–1577 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chopra VS, Hong JW, Levine M 2009. Regulation of Hox gene activity by transcriptional elongation in Drosophila. Curr Biol 19: 688–693 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chopra VS, Hendrix DA, Core LJ, Tsui C, Lis JT, Levine M 2011. The polycomb group mutant esc leads to augmented levels of paused Pol II in the Drosophila embryo. Mol Cell 42: 837–844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Churchman LS, Weissman JS 2011. Nascent transcript sequencing visualizes transcription at nucleotide resolution. Nature 469: 368–373 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Core LJ, Waterfall JJ, Lis JT 2008. Nascent RNA sequencing reveals widespread pausing and divergent initiation at human promoters. Science 322: 1845–1848 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Core LJ, Waterfall JJ, Gilchrist DA, Fargo DC, Kwak H, Adelman K, Lis JT 2012. Defining the status of RNA polymerase at promoters. Cell Rep 2: 1025–1035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edgar R, Domrachev M, Lash AE 2002. Gene Expression Omnibus: NCBI gene expression and hybridization array data repository. Nucleic Acids Res 30: 207–210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frank LH, Rushlow C 1996. A group of genes required for maintenance of the amnioserosa tissue in Drosophila. Development 122: 1343–1352 [DOI] [PubMed] [Google Scholar]
- Fuda NJ, Ardehali MB, Lis JT 2009. Defining mechanisms that regulate RNA polymerase II transcription in vivo. Nature 461: 186–192 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaertner B, Johnston J, Chen K, Wallaschek N, Paulson A, Garruss AS, Gaudenz K, De Kumar B, Krumlauf R, Zeitlinger J 2012. Poised RNA polymerase II changes over developmental time and prepares genes for future expression. Cell Rep 2: 1670–1683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilchrist DA, Nechaev S, Lee C, Ghosh SK, Collins JB, Li L, Gilmour DS, Adelman K 2008. NELF-mediated stalling of Pol II can enhance gene expression by blocking promoter-proximal nucleosome assembly. Genes Dev 22: 1921–1933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilchrist DA, Dos Santos G, Fargo DC, Xie B, Gao Y, Li L, Adelman K 2010. Pausing of RNA polymerase II disrupts DNA-specified nucleosome organization to enable precise gene regulation. Cell 143: 540–551 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilchrist DA, Fromm G, dos Santos G, Pham LN, McDaniel IE, Burkholder A, Fargo DC, Adelman K 2012. Regulating the regulators: The pervasive effects of Pol II pausing on stimulus-responsive gene networks. Genes Dev 26: 933–944 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilmour DS, Lis JT 1986. RNA polymerase II interacts with the promoter region of the noninduced hsp70 gene in Drosophila melanogaster cells. Mol Cell Biol 6: 3984–3989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graveley BR, Brooks AN, Carlson JW, Duff MO, Landolin JM, Yang L, Artieri CG, van Baren MJ, Boley N, Booth BW, et al. 2011. The developmental transcriptome of Drosophila melanogaster. Nature 471: 473–479 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hah N, Danko CG, Core L, Waterfall JJ, Siepel A, Lis JT, Kraus WL 2011. A rapid, extensive, and transient transcriptional response to estrogen signaling in breast cancer cells. Cell 145: 622–634 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harrison MM, Li XY, Kaplan T, Botchan MR, Eisen MB 2011. Zelda binding in the early Drosophila melanogaster embryo marks regions subsequently activated at the maternal-to-zygotic transition. PLoS Genet 7: e1002266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jennings BH, Shah S, Yamaguchi Y, Seki M, Phillips RG, Handa H, Ish-Horowicz D 2004. Locus-specific requirements for Spt5 in transcriptional activation and repression in Drosophila. Curr Biol 14: 1680–1684 [DOI] [PubMed] [Google Scholar]
- Kadonaga JT 2004. Regulation of RNA polymerase II transcription by sequence-specific DNA binding factors. Cell 116: 247–257 [DOI] [PubMed] [Google Scholar]
- Kao SY, Calman AF, Luciw PA, Peterlin BM 1987. Anti-termination of transcription within the long terminal repeat of HIV-1 by tat gene product. Nature 330: 489–493 [DOI] [PubMed] [Google Scholar]
- Kent WJ, Sugnet CW, Furey TS, Roskin KM, Pringle TH, Zahler AM, Haussler D 2002. The human genome browser at UCSC. Genome Res 12: 996–1006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kwak H, Fuda NJ, Core LJ, Lis JT 2013. Precise maps of RNA polymerase reveal how promoters direct initiation and pausing. Science 339: 950–953 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Levine M 2011. Paused RNA polymerase II as a developmental checkpoint. Cell 145: 502–511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li LM, Arnosti DN 2011. Long- and short-range transcriptional repressors induce distinct chromatin states on repressed genes. Curr Biol 21: 406–412 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li J, Gilmour DS 2011. Promoter proximal pausing and the control of gene expression. Curr Opin Genet Dev 21: 231–235 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liang HL, Nien CY, Liu HY, Metzstein MM, Kirov N, Rushlow C 2008. The zinc-finger protein Zelda is a key activator of the early zygotic genome in Drosophila. Nature 456: 400–403 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lott SE, Villalta JE, Schroth GP, Luo S, Tonkin LA, Eisen MB 2011. Noncanonical compensation of zygotic X transcription in early Drosophila melanogaster development revealed through single-embryo RNA-seq. PLoS Biol 9: e1000590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo Z, Lin C, Shilatifard A 2012. The super elongation complex (SEC) family in transcriptional control. Nat Rev Mol Cell Biol 13: 543–547 [DOI] [PubMed] [Google Scholar]
- Menet JS, Rodriguez J, Abruzzi KC, Rosbash M 2012. Nascent-Seq reveals novel features of mouse circadian transcriptional regulation. eLife 1: e00011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Min IM, Waterfall JJ, Core LJ, Munroe RJ, Schimenti J, Lis JT 2011. Regulating RNA polymerase pausing and transcription elongation in embryonic stem cells. Genes Dev 25: 742–754 [DOI] [PMC free article] [PubMed] [Google Scholar]
- The modENCODE Consortium, Roy S, Ernst J, Kharchenko PV, Kheradpour P, Negre N, Eaton ML, Landolin JM, Bristow CA, Ma L, et al. 2010. Identification of functional elements and regulatory circuits by Drosophila modENCODE. Science 330: 1787–1797 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muse GW, Gilchrist DA, Nechaev S, Shah R, Parker JS, Grissom SF, Zeitlinger J, Adelman K 2007. RNA polymerase is poised for activation across the genome. Nat Genet 39: 1507–1511 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Narita T, Yamaguchi Y, Yano K, Sugimoto S, Chanarat S, Wada T, Kim DK, Hasegawa J, Omori M, Inukai N, et al. 2003. Human transcription elongation factor NELF: Identification of novel subunits and reconstitution of the functionally active complex. Mol Cell Biol 23: 1863–1873 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nechaev S, Fargo DC, dos Santos G, Liu L, Gao Y, Adelman K 2010. Global analysis of short RNAs reveals widespread promoter-proximal stalling and arrest of Pol II in Drosophila. Science 327: 335–338 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niessing D, Rivera-Pomar R, La Rosee A, Hader T, Schock F, Purnell BA, Jackle H 1997. A cascade of transcriptional control leading to axis determination in Drosophila. J Cell Physiol 173: 162–167 [DOI] [PubMed] [Google Scholar]
- O'Connor MB, Umulis D, Othmer HG, Blair SS 2006. Shaping BMP morphogen gradients in the Drosophila embryo and pupal wing. Development 133: 183–193 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Podos SD, Ferguson EL 1999. Morphogen gradients: New insights from DPP. Trends Genet 15: 396–402 [DOI] [PubMed] [Google Scholar]
- Ronshaugen M, Levine M 2004. Visualization of trans-homolog enhancer-promoter interactions at the Abd-B Hox locus in the Drosophila embryo. Dev Cell 7: 925–932 [DOI] [PubMed] [Google Scholar]
- Rougvie AE, Lis JT 1988. The RNA polymerase II molecule at the 5′ end of the uninduced hsp70 gene of D. melanogaster is transcriptionally engaged. Cell 54: 795–804 [DOI] [PubMed] [Google Scholar]
- Sanson B 2001. Generating patterns from fields of cells. Examples from Drosophila segmentation. EMBO Rep 2: 1083–1088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saunders A, Core LJ, Lis JT 2006. Breaking barriers to transcription elongation. Nat Rev Mol Cell Biol 7: 557–567 [DOI] [PubMed] [Google Scholar]
- Stathopoulos A, Levine M 2004. Whole-genome analysis of Drosophila gastrulation. Curr Opin Genet Dev 14: 477–484 [DOI] [PubMed] [Google Scholar]
- Su MA, Wisotzkey RG, Newfeld SJ 2001. A screen for modifiers of decapentaplegic mutant phenotypes identifies lilliputian, the only member of the Fragile-X/Burkitt's Lymphoma family of transcription factors in Drosophila melanogaster. Genetics 157: 717–725 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang AH, Neufeld TP, Rubin GM, Muller HA 2001. Transcriptional regulation of cytoskeletal functions and segmentation by a novel maternal pair-rule gene, lilliputian. Development 128: 801–813 [DOI] [PubMed] [Google Scholar]
- Tsurumi A, Xia F, Li J, Larson K, LaFrance R, Li WX 2011. STAT is an essential activator of the zygotic genome in the early Drosophila embryo. PLoS Genet 7: e1002086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wada T, Takagi T, Yamaguchi Y, Ferdous A, Imai T, Hirose S, Sugimoto S, Yano K, Hartzog GA, Winston F, et al. 1998. DSIF, a novel transcription elongation factor that regulates RNA polymerase II processivity, is composed of human Spt4 and Spt5 homologs. Genes Dev 12: 343–356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Lee C, Gilmour DS, Gergen JP 2007. Transcription elongation controls cell fate specification in the Drosophila embryo. Genes Dev 21: 1031–1036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang X, Hang S, Prazak L, Gergen JP 2010. NELF potentiates gene transcription in the Drosophila embryo. PLoS ONE 5: e11498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu CH, Lee C, Fan R, Smith MJ, Yamaguchi Y, Handa H, Gilmour DS 2005. Molecular characterization of Drosophila NELF. Nucleic Acids Res 33: 1269–1279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zaret KS, Carroll JS 2011. Pioneer transcription factors: Establishing competence for gene expression. Genes Dev 25: 2227–2241 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zeidler MP, Bach EA, Perrimon N 2000. The roles of the Drosophila JAK/STAT pathway. Oncogene 19: 2598–2606 [DOI] [PubMed] [Google Scholar]
- Zeitlinger J, Stark A, Kellis M, Hong JW, Nechaev S, Adelman K, Levine M, Young RA 2007. RNA polymerase stalling at developmental control genes in the Drosophila melanogaster embryo. Nat Genet 39: 1512–1516 [DOI] [PMC free article] [PubMed] [Google Scholar]