Abstract
Nebulin—a giant sarcomeric protein—plays a pivotal role in skeletal muscle contractility by specifying thin filament length and function. Although mutations in the gene encoding nebulin (NEB) are a frequent cause of nemaline myopathy, the most common non-dystrophic congenital myopathy, the mechanisms by which mutations in NEB cause muscle weakness remain largely unknown. To better understand these mechanisms, we have generated a mouse model in which Neb exon 55 is deleted (NebΔExon55) to replicate a founder mutation seen frequently in patients with nemaline myopathy with Ashkenazi Jewish heritage. NebΔExon55 mice are born close to Mendelian ratios, but show growth retardation after birth. Electron microscopy studies show nemaline bodies—a hallmark feature of nemaline myopathy—in muscle fibres from NebΔExon55 mice. Western blotting studies with nebulin-specific antibodies reveal reduced nebulin levels in muscle from NebΔExon55 mice, and immunofluorescence confocal microscopy studies with tropomodulin antibodies and phalloidin reveal that thin filament length is significantly reduced. In line with reduced thin filament length, the maximal force generating capacity of permeabilized muscle fibres and single myofibrils is reduced in NebΔExon55 mice with a more pronounced reduction at longer sarcomere lengths. Finally, in NebΔExon55 mice the regulation of contraction is impaired, as evidenced by marked changes in crossbridge cycling kinetics and by a reduction of the calcium sensitivity of force generation. A novel drug that facilitates calcium binding to the thin filament significantly augmented the calcium sensitivity of submaximal force to levels that exceed those observed in untreated control muscle. In conclusion, we have characterized the first nebulin-based nemaline myopathy model, which recapitulates important features of the phenotype observed in patients harbouring this particular mutation, and which has severe muscle weakness caused by thin filament dysfunction.
Keywords: nebulin, nemaline myopathy, muscle fibre weakness, thin filament function
Introduction
Nemaline myopathy is the most common non-dystrophic congenital myopathy (Sanoudou and Beggs, 2001), with hallmark features that include muscle weakness and the presence of nemaline bodies in skeletal muscle fibres (North et al., 1997). To date, seven genes have been implicated in nemaline myopathy. Strikingly, six of these genes code for proteins of the skeletal muscle thin filament: alpha tropomyosin 3 and beta tropomyosin (TPM3 and TPM2), nebulin (NEB), actin alpha 1 (ACTA1), troponin T type 1 (TNNT1), and cofilin 2 (CFL2). The seventh implicated gene, KBTBD13, encodes a ubiquitin ligase whose function in skeletal muscle is currently unknown (Sambuughin et al., 2010, 2012). With mutations in NEB likely accounting for >50% of nemaline myopathy cases (Pelin et al., 1999), NEB is the most frequently affected gene in nemaline myopathy. To date, 64 different mutations in NEB have been reported in nemaline myopathy probands (Pelin et al., 1999; Lehtokari et al., 2006, 2009).
Nebulin is a giant sarcomeric protein (∼800 kDa); its C-terminus is anchored in the Z-disk and its N-terminus is located close to the thin filament pointed end. Thus, a single nebulin molecule spans nearly the entire length of the thin filament (Fig. 1). Insights into the role of nebulin’s functions made a leap forward when two full nebulin knockout mouse models were published (Bang et al., 2006; Witt et al., 2006). Nebulin plays important roles in sarcomeric structure and contractile performance. It stabilizes the thin filament and specifies its minimal length (Bang et al., 2006; Witt et al., 2006; Castillo et al., 2009; Gokhin et al., 2009; Pappas et al., 2010). Recent evidence also suggests that nebulin modulates both the kinetics of actomyosin interaction (Bang et al., 2009; Chandra et al., 2009) and the calcium sensitivity of force generation (Chandra et al., 2009). Skeletal muscle fibres of patients with nemaline myopathy with NEB mutations might develop muscle weakness due to loss of these functions of nebulin; their myofibres contain shorter thin filaments (Ottenheijm et al., 2009b), have altered crossbridge cycling kinetics (Ottenheijm et al., 2010; Lawlor et al., 2011; Ochala et al., 2011), and their calcium-sensitivity of force generation is reduced (Ottenheijm et al., 2010). However, studies on muscle fibres from patients are limited due to, for example, the small size of diagnostic muscle biopsies and the heterogeneity of the study populations. Consequently, our understanding of the mechanisms underlying muscle weakness in patients with nemaline myopathy with nebulin mutations remains incomplete. To improve this understanding requires the development of mouse models that harbour mutations in Neb that are found in patients.
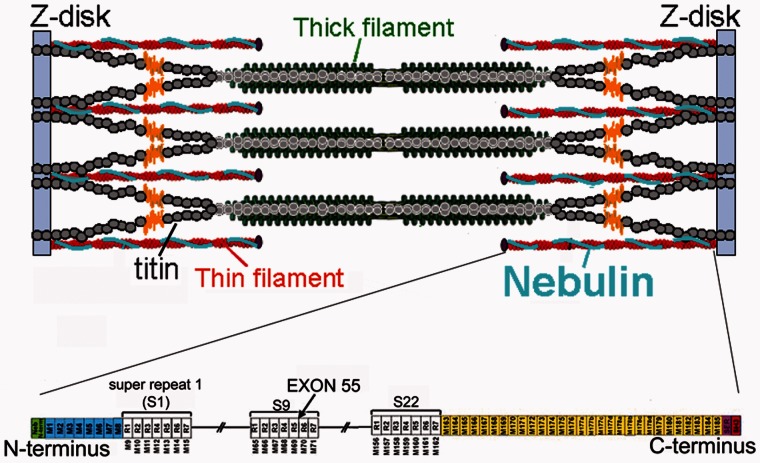
Figure 1.
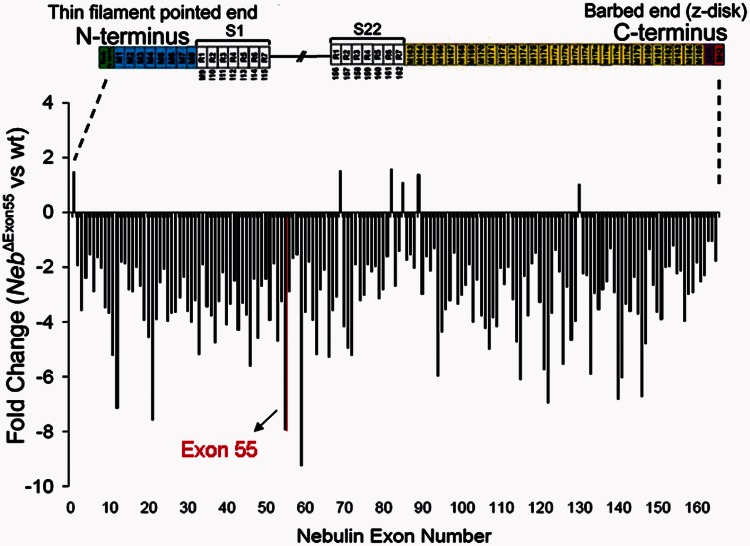
Top: Schematic of the location of nebulin in the sarcomere. Bottom: Schematic of the nebulin protein. Nebulin has a highly modular structure, within the central region (M9-M162) seven modular repeats arranged into 22 super-repeats. Exon 55 codes for 35 amino acids that are part of repeat 5 and 6 (super-repeat 9). Note that exon 55 in the human and murine nebulin gene are equivalent.
Here, we have generated a mouse in which Neb exon 55 is deleted (Fig. 1)—an in-frame deletion—to model a founder mutation frequently seen in patients with nemaline myopathy and which causes autosomal recessive nemaline myopathy (Anderson et al., 2004; Lehtokari et al., 2006, 2009). This model (NebΔExon55) allowed, for the first time, for a detailed and comprehensive investigation of the disease mechanisms caused by a nebulin mutation and for the evaluation of potential treatments. Our findings reveal that homozygous mice have a phenotype that recapitulates important features observed previously in patients with nemaline myopathy with deletion of NEB exon 55, including severe muscle weakness and, importantly we show that muscle strength can be restored by a novel fast skeletal troponin activator that, at submaximal force levels, augments the response of the thin filament to calcium.
Materials and methods
Generation of NebΔExon55 mice
NebΔExon55 mice were designed to mimic the 2502-bp deletion that has previously been found to cause nemaline myopathy in humans (Anderson et al., 2004). Note that Neb exon 55 is the murine equivalent of the exon deleted in human patients (Kazmierski et al., 2003). The construct replaced exon 55 and parts of introns 54 and 55 with a flippase recognition target (FRT) flanked neomycin cassette. No differences in phenotype (life-span, body weight, and nebulin protein levels) were found when the neomycin cassette was removed with a flippase (FLP) deleter strain [B6; SJL-Tg(ACTFLPe)9205Dym/J- purchased from Jackson Laboratories]. Animals were maintained on a C57/BL6 background. All experiments were approved by IACUC and followed the NIH Guidelines ‘Using Animals in Intramural Research’ for animal use.
A targeting construct was assembled by cloning arms of homology from C57/BL6 genomic DNA (purchased from Jackson Labs) into a pCKOB vector. The vector was introduced into C57BL/6J-Tyrc-2J/J embryonic stem cells by electroporation. The deletion was introduced by homologous recombination and selected by negative selection against thymidine kinase (using Ganciclovir) and positive selection for neomyosin resistance (G418). The surviving embryonic stem cell clones were verified by long PCR using primers (P1) 5’-ATTGACAATGCCGATTAACTACTGC-3’ and (P2) 5’-CTGGCACTCTGTCGATACCC-3’ (left arm) (P3) 5’-ACACGCGTCACCTTAATATGC-3’ and (P4) 5’-CCCAGCTGCTTACGGTAGCC-3’ and used to produce NebΔEx55 mice by injection into C57/BL6 blastocyst. Genotyping was performed using tail digests using GoTaq® Green Master Mix (Promega). The following primer sets were used: (P5)-reverse 5’-GAAAGGAACTCTGTCCTCTGG-3’; (P6) WT forward 5’-GCATTCTTGCTCTTTCTTGTATGG-3’; (P3) NebΔEx55 forward 5’-ACACGCGTCACCTTAATATGC-3’; (P7) NebΔEx55 neo removed forward 5’-ACACCCAGGCAGAAGCTAGG-3’.
Histochemistry and electron microscopy
Cross sections (5 μm) of isopentane-frozen quadriceps muscles were taken midway down the length of the muscle and stained with haematoxylin and eosin or Gomori trichrome using standard histochemical techniques. For Gomori trichrome staining, sections were stained in Harris haematoxylin (Richard Allen Scientific) for 5 min, rinsed, and then stained in Gomori One-Step Light Green Solution (Newcomer Supply) for 10 min. Following an additional rinse and differentiation in 0.2% acetic acid (Newcomer Supply) for 2 min, sections were dehydrated and coverslipped using standard techniques. Light microscopic images were captured using an Olympus DP72 camera and cellSens Standard software (Olympus). For the determination of fibre size in quadriceps muscle sections, immunofluorescence was performed using anti-dystrophin antibodies, and a representative area of the muscle was photographed using a Zeiss ImagerZ1 microscope with an Axiocam HRC camera. The minFeret diameter (the smallest diameter across the myofibre) was measured manually in sections from four wild-type mice (range 653 to 862 fibres measured) and three NebΔExon55 mice (range 1003 to 1343 fibres measured) using Zeiss Axiovision 4.8 software. For electron microscopy, glutaraldehyde-fixed portions of tibialis cranialis muscle were submitted to the Microbiology and Molecular Genetic Electron Microscopy Facility at the Medical College of Wisconsin. Tissue was then fixed in 2.5% glutaraldehyde in 0.1 M cacodylate buffer pH 7.4 for 60 min, washed 3 × 10 min in 0.1 M cacodylate buffer, post-fixed with 1% potassium ferrocyanide reduced OsO4 for 3 h on ice, dehydrated through graded methanol, and embedded in EMbed 812 resin (hard). Ultrathin sections (60 nm) were contrasted with uranyl acetate and lead citrate and viewed in a Hitachi H600 TEM.
Gel electrophoresis and western blotting
Gel electrophoresis of nebulin was with 1% agarose gels and has been described in detail previously (Warren et al., 2003). Muscle samples from 20 wild-type and 20 NebΔExon55 mice were flash frozen in liquid nitrogen after dissection. Samples were pulverized to a fine powder, solubilized and loaded on 1% agarose gels. Gels were then stained with Coomassie blue and scanned using an Epson expressions scanner.
For western blots, samples were loaded in 0.8% SDS-agarose gels and run at 15 mA/gel for 3 h. Gels were transferred onto 0.45 μm polyvinylidene fluoride membranes (Imobilon) using a Bio-Rad semi-dry transfer unit at 152 mA/gel for 2.5 h. Blots were stained using Ponceau S to visualize total protein, dried and imaged. Blots were then rehydrated, blocked with 50% Odyssey Blocking Buffer in PBS for 1 h and incubated with Rabbit anti-nebulin N-terminus (Myomedix) overnight. Primary antibody was then removed and CF680 goat anti-rabbit secondary (Biotium Inc.) was added. Blots were scanned and analysed using an Odyssey infrared imaging system (Li-Cor Biosciences).
Nebulin transcriptional profiling
Day 4 longissimus dorsi muscle was isolated from wild-type and NebΔEx55 mice and stored in RNAlater (Ambion). RNA was isolated using RNeasy® Fibrous Tissue Mini Kit (Qiagen). A custom microarray was prepared by spotting 50-mer oligonucleotides in triplicate onto GAPSII amino-silane coated slides (Corning) with a robotic arrayer (Virtek). Oligonucleotide sequences for every murine nebulin exon were selected with 45–55% GC content, Tm near 70°C, low self-complementarity, and specificity based on NCBI BLAST searches with no off-target stretches longer than 25 nt with identity greater than 80%; for exons shorter than 50 nt the selected sequence spans an exon–exon junction. Sequence-specificity was confirmed by BLAT against the current mouse genome (GRCm38/mm10). For optimizing hybridization conditions, 5 bp mismatch controls were incorporated. The array had been validated by analysing transcripts isolated from human muscle with known nebulin exon composition. RNA was reverse transcribed and amplified using SenseAmp (Genisphere), and SuperScript® III (Invitrogen) and dye-coupled with Alexa Fluor® 555 or AlexaFluor 647 (Invitrogen). Sample pairs were hybridized with SlideHyb™ Glass Array Hybridization Buffer #1 (Ambion) in a GeneTAC Hybridization Station (Genomic Solutions); slides were scanned with an ArrayWoRX scanner and the results analysed using the R package CARMA (Greer et al., 2006).
Immunofluorescence confocal scanning laser microscopy
Small muscle strips were dissected and permeabilized overnight at ∼4°C in relaxing solution (in mM; 20 BES, 10 EGTA, 6.56 MgCl2, 5.88 NaATP, 1 DTT, 46.35 K-propionate, 15 creatine phosphate, pH 7.0 at 20°C) containing 1% (v/v) Triton™ X-100. Immunolabelling and confocal scanning laser microscopy was performed as described previously (Ottenheijm et al., 2009b). Primary antibodies: anti-α-actinin (mouse monoclonal, A7811, Sigma-Aldrich), anti-tropomodulin-1 (Witt et al., 2006) Alexa Fluor® 488 conjugated phalloidin (A12379, Invitrogen), anti-CapZα (mouse monoclonal, 5B12.3, DSHB), and rabbit anti-nebulin N-terminus (Myomedix). Secondary antibodies: Alexa Fluor® 594 (goat anti-mouse, Invitrogen), and Alexa Fluor® 488 (goat anti-rabbit, Invitrogen). Secondary antibodies did not stain when used without primary antibodies (data not shown). Images were produced using a Bio-Rad MRC 1024 confocal laser scanning microscope with the LaserSHARP 2000 software package (Hercules). From the acquired images, thin filament lengths were determined using ImageJ software (National Institutes of Health).
Permeabilized fibre contractility
For contractility experiments we used tibialis cranialis muscle; the equivalent of m. tibialis anterior in humans, which has been shown to be selectively affected in patients with nemaline myopathy with nebulin mutations (Jungbluth et al., 2004). The procedures for permeabilized muscle fibre contractility were as described previously (Ottenheijm et al., 2009a, b, 2010, 2011), with minor modifications. In short, small strips dissected from muscle from six wild-type and six NebΔExon55 mice were permeabilized, or skinned, overnight at ∼4°C in relaxing solution (in mM; 20 BES, 10 EGTA, 6.56 MgCl2, 5.88 NaATP, 1 DTT, 46.35 K-propionate, 15 creatine phosphate, pH 7.0 at 20°C) containing 1% (v/v) Triton™ X-100. The skinning procedure renders the membranous structures in the muscle fibres permeable, which enables activation of the myofilaments with exogenous Ca2+. Preparations were washed thoroughly with relaxing solution and stored in 50% glycerol/relaxing solution at −20°C. Small muscle fibre bundles (diameter ∼0.07 mm) were dissected from the permeabilized strips, and were attached to a strain gauge and a high-speed motor using aluminium foil clips. Experiments were performed at 20°C. The XY (width) and XZ (depth, using a prism) dimensions of bundle diameters were measured with a ×40 objective. The muscle bundle cross-sectional area was calculated from the average of three width and depth measurements made along the length of the muscle bundle. The preparation was activated at pCa 4.5 (pCa = −log of molar free Ca2+ concentration) to obtain maximal Ca2+-activated force. Maximal tension was determined by dividing the force generated at pCa 4.5 by cross-sectional area. To determine force–sarcomere length relations, the maximal force generated at various sarcomere lengths was determined. Sarcomere length was measured with an on-line laser-diffraction system (Granzier and Wang, 1993).
To determine the force–pCa relation, muscle preparations were sequentially bathed in solutions with pCa values ranging from 4.5 to 9.0 and the steady-state force was measured. Measured force values were normalized to the maximal force obtained at pCa 4.5. The obtained force–pCa data were fit to the Hill equation, providing pCa50 (pCa giving 50% maximal active tension) and the Hill coefficient, nH, an index of myofilament cooperativity.
To determine a dose-response curve for the fast skeletal troponin activator CK-2066260 (Cytokinetics Inc) tissue was exposed to pCa solutions of 6.25 with increasing concentrations (1, 2, 5, 10 and 20 μM) of CK-2066260 dissolved in 1% dimethylsulphoxide (DMSO) as vehicle. Note that 1% DMSO did not affect muscle fibre contractility (data not shown). CK-2066260 enhanced submaximal force generation with a maximal effect at 5 μM (data not shown), which is in accordance with previously reported dose-response curves of its analogue CK-2017357 (Russell et al., 2012). Accordingly, experiments were conducted with either CK-2066260 (5 μM) or vehicle alone (1% DMSO). Force–pCa relations were determined as described above.
To measure the rate of tension redevelopment (ktr), we used the large slack/release approach (Brenner and Eisenberg, 1986), to disengage force generating crossbridges from the thin filaments, which were isometrically activated. Three different bathing solutions were used during the experimental protocols: a relaxing solution, a pre-activating solution with low EGTA (ethylene glycol tetraacetic acid) concentration, and an activating solution. The composition of these solutions was as described previously (Stienen et al., 1996). Fast activation of the fibres was achieved by transferring the permeabilized fibres from the pre-activation solution containing a low concentration of EGTA (pCa 9.0) to a pCa 4.5 activating solution. Once the steady-state was reached, a slack equivalent to 10% of the muscle length was rapidly induced at one end of the muscle using the motor. This was followed immediately by an unloaded shortening lasting 30 ms. The remaining bound cross-bridges were mechanically detached by rapidly (1 ms) restretching the muscle fibre to its original length, after which tension redevelops. The rate constant of monoexponential tension redevelopment (ktr) was determined by fitting the rise of tension to the following equation: F = Fss(1 − e-ktr.t), where F is force at time t and ktr is the rate constant of tension redevelopment.
To determine force simultaneously with ATP consumption rate, we used the system described by Stienen et al. (1996). To measure the ATPase activity, a near UV light was projected through the quartz window of the bath (30 μl volume and temperature controlled at 20°C) and detected at 340 nm. The maximum activation buffer (pCa 4.5) contained 10 mM phosphoenol pyruvate, with 4 mg/ml pyruvate kinase (500 U/mg), 0.24 mg/ml lactate dehydrogenase (870 U/mg) and 20 µM diadenosine-5’ pentaphosphate (A2P5). For efficient mixing, the solution in the bath was continuously stirred by means of motor-driven vibration of a membrane positioned at the base of the bath. ATPase activity of the permeabilized fibre bundles was measured as follows: ATP regeneration from ADP is coupled to the breakdown of phosphoenol pyruvate to pyruvate and ATP catalysed by pyruvate kinase, which is linked to the synthesis of lactate catalysed by lactate dehydrogenase. The breakdown of NADH, which is proportional to the amount of ATP consumed, is measured online by UV absorbance at 340 nm. The ratio of light intensity at 340 nm (sensitive to NADH concentration), and the light intensity at 410 nm (reference signal), is obtained by means of an analogue divider. After each recording, the UV absorbance signal of NADH was calibrated by multiple rapid injections of 0.25 nmol ADP (0.025 μl of 10 mM ADP) into the bathing solution, with a stepper motor-controlled injector. The slope of the [NADH] versus time trace during steady-state tension development of a calcium-induced contraction (Fig. 8B) was determined from a linear fit and the value divided by the fibre volume (in mm3) to determine the fibre’s ATPase rate. ATPase rates were corrected for the basal ATPase measured in relaxing solution. The ATPase rate was divided by tension (force/cross-sectional area) to determine the tension cost.
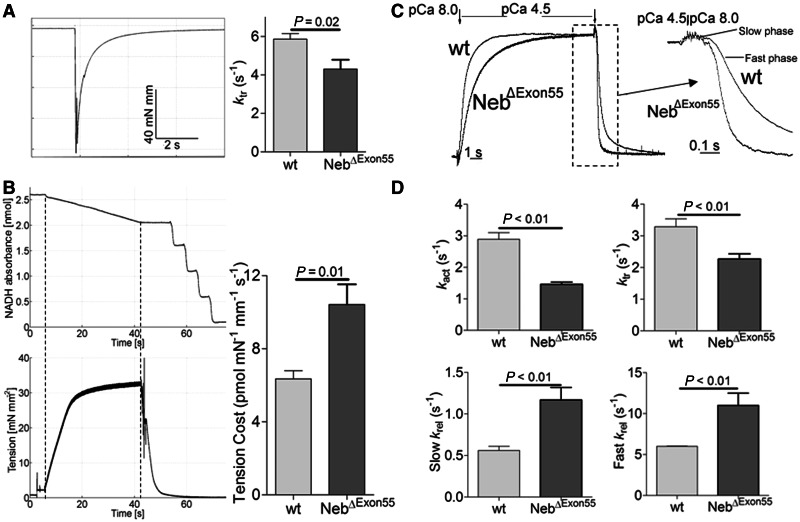
Figure 8.
(A) ktr measurements in 6-day-old NebΔExon55 and wild-type (wt) fibres. Left: Example of a ktr protocol in a NebΔExon55 fibre preparation; right: ktr is significantly lower in NebΔExon55 compared with wild-type fibres. (B) Tension cost measurements in NebΔExon55 and wild-type fibres. Left: Example of maximally activated NebΔExon55 fibres with developed force at the bottom and [ATP] at the top. The slope of the [ATP] versus time trace was divided by fibre volume (in mm3) to determine ATP consumption rate. ATP consumption rate was normalized to tension to determine the tension cost. Right: Tension cost is significantly higher in NebΔExon55 compared to wild-type fibres. (C) An example of a protocol to determine crossbridge cycling kinetics in a myofibril. Myofibrils are rapidly activated (pCa 4.5), followed by rapid relaxation (pCa 8.0) after steady state tension is reached. From the force tracings, the rate constants of activation (kact) and relaxation are determined. Relaxation consists of an initial slow phase (slow krel) followed by a fast phase (fast krel). ktr is determined during a separate protocol that is similar to that applied on fibres. (D) Both kact and ktr are significantly lower in NebΔExon55 compared with wild-type myofibrils, whereas slow and fast krel are significantly higher in NebΔExon55 compared with wild-type myofibrils.
Myofibril contractility
Myofibrils were prepared from flash frozen and then thawed tibialis cranialis muscles from 4–7 day old wild-type (n = 5) and and homozygous NebΔExon55 (n = 5) mice, and placed in ice-cold rigor solution containing (mM): NaCl 132, KCl 5, MgCl2 1, Tris 10, EGTA 5 (pH 7.1). The muscles were incubated for 3 h in the same solution with 1% Triton™-100. Triton was then removed and the muscles sheared in rigor solution to produce myofibril suspensions. In the present experiments we used previously published techniques (Tesi et al., 2002). Briefly, to study myofibrillar force development a small volume of the myofibril suspension was transferred to a temperature-controlled chamber (15°C) filled with relaxing solution (pCa 8.0) and mounted on an inverted microscope. Single myofibrils or bundles of few myofibrils (25–80 μm long, 1–4 μm wide) were mounted horizontally between two glass microtools. One tool was connected to a length-control motor that could produce rapid (<1 ms) length changes. The second tool was a calibrated cantilevered force probe (2–6 nm/nN; frequency response 2–5 kHz). Force was measured from the deflection of the image of the force probe projected on a split photodiode. Average sarcomere length and myofibril diameter were measured from image analysis. The initial sarcomere length of the preparations was adjusted to ∼2.2 μm. Myofibrils were activated and relaxed by rapidly translating the interface between two flowing streams of different pCa solutions across the length of the preparation. The solution change took place with a time constant of 2–3 ms and was complete in < 5 ms. Activating and relaxing solutions, calculated as previously described (Tesi et al., 2002), were at pH 7.0 and contained 10 mM total EGTA (CaEGTA/EGTA ratio set to obtain pCa 8.0 and 4.5), 5 mM MgATP, 1 mM free Mg2+, 10 mM MOPS, propionate and sulphate to adjust the final solution to an ionic strength of 200 mM and monovalent cation concentration of 155 mM. Creatine phosphate (10 mM) and creatine kinase (200 U/ml) were added to all solutions. Contaminant inorganic phosphate (Pi) from spontaneous breakdown of MgATP and creatine phosphate was reduced to <5 μM by an inorganic phosphate scavenging system (purine-nucleoside-phosphorylase with substrate 7-methyl-guanosine). All solutions to which the muscles and myofibrils were exposed contained a cocktail of protease inhibitors including leupeptin (10 μM), pepstatin (5 μM), PMSF (200 μM) and E64 (10 μM), as well as NaN3 (500 μM) and DTE (2 mM).
Statistical analyses
Data are presented as mean ± SEM. For statistical analyses, t-tests were used. The effect of CK-2066260 was investigated within individual muscle fibre preparations; a paired t-test was performed in these analyses. P < 0.05 was considered to be statistically significant.
Results
NebΔExon55 mice display a phenotype that resembles nemaline myopathy
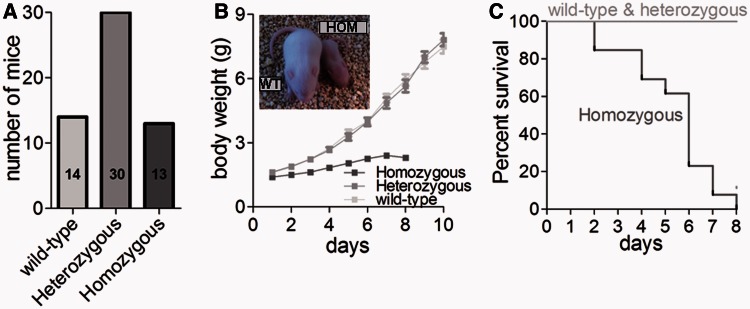
Using homologous recombination, we removed exon 55 from the nebulin gene. Mice heterozygous for the targeted allele are viable and fertile and survive into adulthood with no obvious clinical or contractile phenotype. Homozygous offspring (referred to as NebΔExon55) are produced at expected Mendelian genetic ratios (∼1/4 of total number of pups using heterozygous breedings, Fig. 2A), with a weight at birth that is not different from that of heterozygous and wild-type littermates. However, NebΔExon55 mice show severe growth retardation after birth (Fig. 2B) and typically do not survive past the first week (Fig. 2C).
Figure 2.
(A) Homozygous (NebΔExon55) mice are born close to Medelian ratio: 13 homozygous mice out of total 57 pups when using heterozygous breedings. (B) Body weight of NebΔExon55 mice at birth is not different from that of HET and wild-type (WT) littermates. NebΔExon55 mice show severe growth retardation after birth, and (C) typically do not survive past the first week (survival curve based on 57 pups). Inset: Photograph of a NebΔExon55 (HOM) and a wild-type littermate mouse illustrating the growth retardation in NebΔExon55 mice.
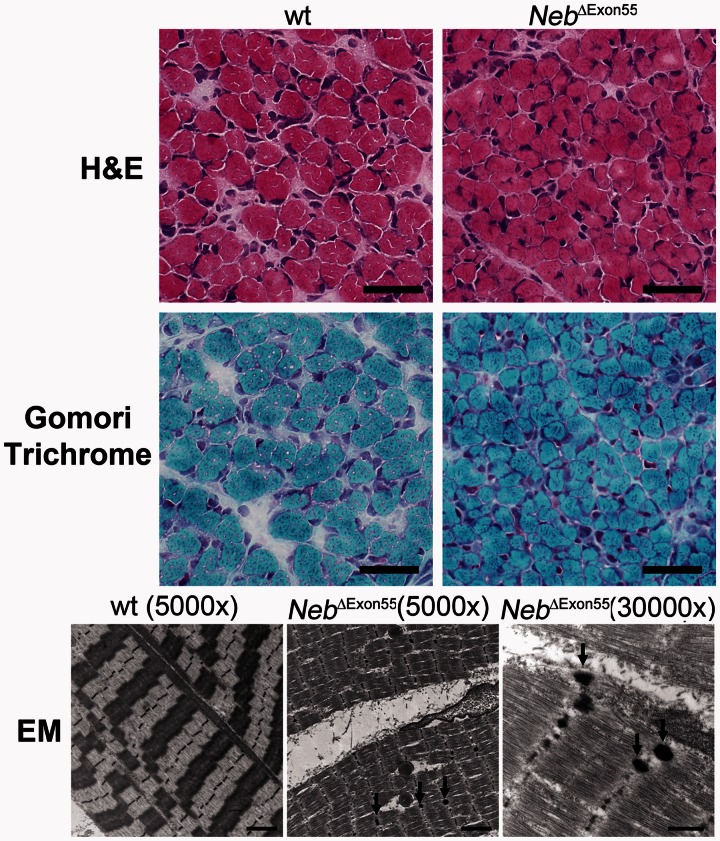
Nemaline myopathy in humans is characterized by the presence of nemaline bodies (often called nemaline rods when they are rod-shaped), which is often accompanied by myofibre hypotrophy. To test whether these histological characteristics of nemaline myopathy are present in NebΔExon55 mice, we first evaluated sections of quadriceps muscle stained with haematoxylin and eosin or Gomori trichrome. As shown in Fig. 3A, the haematoxylin and eosin stained sections from 6-day-old wild-type and NebΔExon55 mice reveal smaller muscle fibres in NebΔExon55 mice. Quantitative analysis revealed that where average fibre minFeret diameter was 13.0 ± 0.4 μm in wild-type mice; this was significantly reduced to 10.1 ± 0.3 μm in NebΔExon55 mice (P < 0.01). Discrete populations of large and small fibres were not observed in NebΔExon55 mouse muscle. The number of central nuclei was <1% in both wild-type and NebΔExon55 muscle, and NebΔExon55 muscle showed no signs of fibrosis. Nemaline bodies were not visible at the light microscopic level on Gomori trichrome stain (Fig. 3A, lower panel) of quadriceps muscle, but electron microscopy revealed electron-dense nemaline bodies at the location of the Z-band (arrows, Fig. 3B) in the tibialis cranialis (the murine equivalent of tibialis anterior in humans) muscles of NebΔExon55 mice. These nemaline bodies displayed electron density equivalent to normal Z-bands, which differentiates them from the Z-band streaming that can be found in a variety of muscle disorders. Nemaline bodies were not seen in wild-type mouse muscle. Thus, NebΔExon55 mice display a histological phenotype that resembles nemaline myopathy, including the presence of nemaline bodies and hypotrophic fibres.
Figure 3.
Histological findings in 6-day-old wild-type and NebΔExon55 mice. Haematoxylin and eosin (H&E) stained sections of quadriceps muscle from wild-type (wt) and NebΔExon55 mice reveal smaller muscle fibres in NebΔExon55 mice. Nemaline rods were not visible at the light microscopic level on Gomori trichrome stain, but electron-dense nemaline bodies (arrows) were seen on electron microscopy (EM) in the tibialis cranialis muscle from all NebΔExon55 mice. Nemaline bodies were not seen in wild-type mouse muscle. Scale bar = 40 µm for haematoxylin and eosin and Gomori trichrome images, 2 µm for ×5000 electron microscopy images, and 500 nm for the ×30 000 electron microscopy image.
Deletion of exon 55 results in severely reduced nebulin protein levels
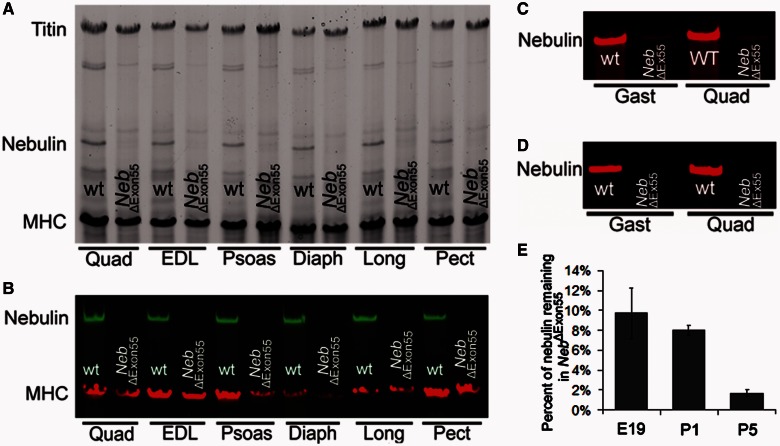
NEB exon 55 encodes 35 amino acids and represents ∼4 kDa at the protein level. As deletion of exon 55 maintains the reading frame 3’ of exon 55, we expected the NebΔExon55 mice to express a nebulin isoform that is ∼4 kDa smaller than wild-type nebulin. However, SDS-agarose gel analysis showed that, in 5-day-old NebΔExon55 mice, nebulin proteins levels were severely reduced in all muscle types tested (Fig. 4A). Western blot experiments using an antibody directed against the nebulin N-terminus confirmed these findings (Fig. 4B). To evaluate whether the low nebulin protein levels at Day 5 after birth were preceded by a gradual decrease from normal levels at birth, we also evaluated nebulin levels at Day 1 after birth (Fig. 4C) and at embryonic Day 19 (Fig. 4D). These experiments showed that nebulin protein levels were severely reduced in NebΔExon55 muscle at both time points. Nebulin protein levels in NebΔExon55 quadriceps muscle (normalized to myosin heavy chain) was ∼10% of wild-type values in embryonic Day 19 tissue, 8% of wild-type values in post-natal Day 1 tissue, and decreased to ∼2% of wild-type values in post-natal Day 5 tissue (Fig. 4E). Thus, deletion of exon 55 results in severly reduced nebulin protein levels.
Figure 4.
(A) SDS-agarose gel electrophoresis reveals severely reduced nebulin protein levels in NebΔExon55 mice in all skeletal muscle types tested, whereas myosin heavy chain (MHC) and titin levels are unaltered. Muscles are obtained from 5-day-old mice. (B) Quantitative western blotting studies with an antibody against nebulin’s N-terminus shows that nebulin protein levels are ∼2% of wild-type (wt) levels. Western blotting studies with the same antibody on muscle from embryonic Day 19 (E19) mice (C) and 1-day-old (P1) mice (D) and show that nebulin protein is 10% of wild-type values at Day 19 (E19) and 8% of wild-type values at Day 1 (E). *P < 0.05. Quad = m. quadriceps; EDL = extensor digitorum longus; Psoas = m. psoas major; Diaph = m. diaphragm; Long = m. longissimus dorsi; Pect = m. pectoralis major; Gast = m. gastrocnemius.
To evaluate whether the reduced nebulin protein levels were caused by alterations at the level of DNA transcription or of messenger RNA translation, we used a homemade nebulin exon microarray to test which of the 166 murine nebulin exons are included in the full-length nebulin messenger RNA. As shown in Fig. 5, in addition to the expected reduction of exon 55 messenger RNA, transcription of nearly all nebulin exons was significantly reduced in NebΔExon55 muscle. Thus, these results suggest that the low nebulin protein levels in NebΔExon55 mice are not caused by defects in the translation of nebulin messenger RNA into protein but rather by changes in transcription of the mutant nebulin gene or in the stability of the mutant nebulin messenger RNA.
Figure 5.
Microarray-based gene expression analysis of nebulin exon expression of m. longissimus dorsi in NebΔExon55 mice compared with wild-type (wt) mice at 4 days of age. The array contains all 166 murine nebulin exons. Nearly all nebulin exons are downregulated in NebΔExon55 mice.
NebΔExon55 mice have shorter thin filament lengths
Nebulin plays an important role in specifying thin filament length, and shorter thin filaments clearly contribute to reduced sarcomeric force generation in human nebulin-deficient nemaline myopathy (Ottenheijm et al., 2009b). Therefore, we measured thin filament length in NebΔExon55 muscle fibres. Using immunofluorescence confocal scanning laser microscopy we determined the location of tropomodulin (a thin filament pointed end capping protein) relative to the Z-disk and we measured the width of the band obtained using the fluorescently labelled actin-binding protein phalloidin.
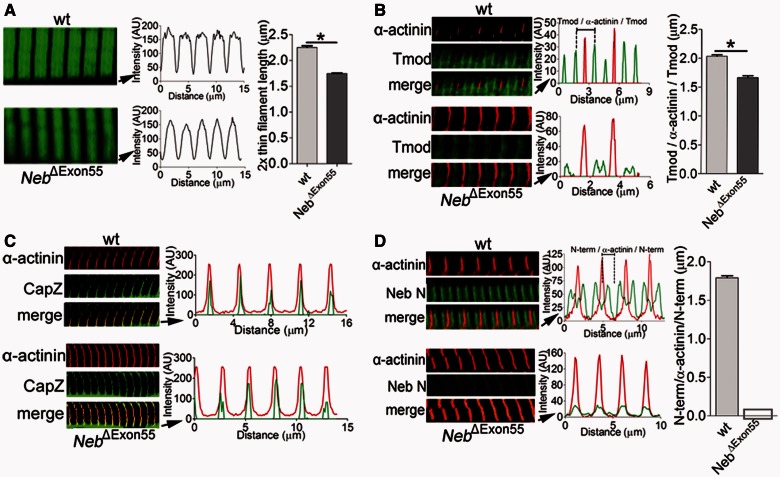
Figure 6A shows that the staining pattern of fluorescently labelled phalloidin differed between myofibrils from NebΔExon55 and wild-type tibialis cranialis muscle; wild-type myofibrils showed broad actin labelling with uniform intensity (except for the Z-disk area where actin filaments overlap), whereas in NebΔExon55 myofibrils the labelling was narrower, and intensity gradually decreased from the Z-disk towards the middle of the sarcomere. Densitometric analysis revealed that the width at half-maximal intensity was 2.25 ± 0.03 μm for wild-type and 1.75 ± 0.01 μm for NebΔExon55 myofibrils (Fig. 6A).
Figure 6.
(A) Left: Actin staining with phalloidin-Alexa Fluor® 488 in Day 6 tibialis cranialis myofibrils from NebΔExon55 and wild-type mice shows broad staining in wild-type myofibrils (top), whereas this staining is narrowed in NebΔExon55 myofibrils (bottom). Right: Analysis of phalloidin line scan intensities revealed significantly reduced average thin filament lengths in NebΔExon55 myofibrils. (B) Left: NebΔExon55 and wild-type myofibrils stained for α-actinin and tropomodulin (Tmod1). Right: Overlay of line scan intensity profile of α-actinin and tropomodulin. The distance between tropomodulin staining (measured across the Z-disk, and indicated as Tmod-α-actinin-Tmod) is significantly reduced in NebΔExon55 myofibrils. (C) CapZ staining of NebΔExon55 and wild-type myofibrils. Staining of NebΔExon55 myofibrils is similar to the staining pattern of wild-type myofibrils. (D) Left: Six day old NebΔExon55 and wild-type myofibrils stained for α-actinin and nebuln’s N-terminus (Neb N). Right: Analysis of line scan intensities. Note that Neb N staining is nearly absent in NebΔExon55 myofibrils. *P < 0.05.
Tropomodulin staining in wild-type myofibrils showed a distinct doublet in the middle of the sarcomere, 2.03 ± 0.03 µm apart, measured across the Z-disk, whereas in myofibrils from NebΔExon55 mice this value was only 1.67 ± 0.03 µm (Fig. 6B). These findings support the results from the phalloidin experiments and suggest that thin filament lengths are reduced from ∼1.0 µm in wild-type myofibrils to ∼0.85 µm in NebΔExon55 myofibrils.
The experiments with fluorescently labelled tropomodulin revealed that thin filaments are shorter in NebΔExon55 myofibrils and suggested that capping at the thin filament pointed end by tropomodulin remains intact in these myofibrils. Immunofluorescence experiments on CapZ (a thin filament barbed end capping protein) showed that capping at the barbed end also remains intact in NebΔExon55 myofibrils (Fig. 6C).
Finally, the data in Fig. 6D, in which an antibody against nebulin’s N-terminus was used, show that nebulin is nearly absent from NebΔExon55 myofibrils, thereby confirming the western blot experiments (Fig. 4). Thus, immunofluorescence confocal microscopy experiments show that in NebΔExon55 muscle thin filament capping is intact but that thin filament length is significantly reduced.
NebΔExon55 mice display severe muscle weakness
Force–sarcomere length relation
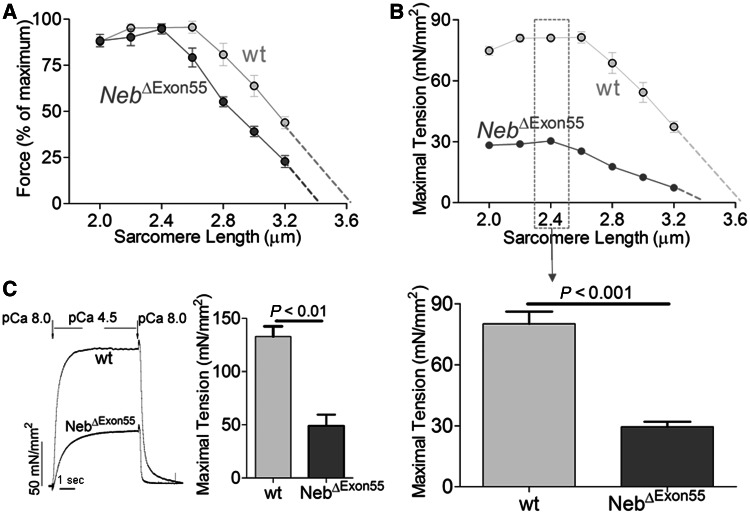
Because sarcomeres generate force in proportion to thick and thin filament overlap, a well-defined thin filament length is an important determinant of muscle function. This is illustrated by the force–sarcomere length relationship, which is characterized by a force plateau at optimal filament overlap, followed by a descending limb at higher sarcomere lengths as filament overlap decreases. Shortened thin filaments will reduce thick and thin filament overlap and impair the sarcomere’s force generating capacity, which is reflected by a leftward shift of the force–sarcomere length relationship (Granzier et al., 1991). Hence we studied whether the reduced thin filament length in NebΔExon55 myofibrils affects the force generating capacity of muscle by determining the force–sarcomere length relationship in permeabilized fibre preparations. Fibres from wild-type muscle showed a characteristic force plateau up to a sarcomere length of ∼2.6 μm, followed by a linear descending limb (Fig. 7A). Note that the descending limb of wild-type fibres intercepts the x-axis at ∼3.6 μm. Assuming that thick filaments are constant at ∼1.6 μm, this would suggest that thin filament length is ∼1.0 μm, which is in-line with thin filament lengths as determined by our confocal microscopy studies (Fig. 6A and B). Importantly, fibres from NebΔExon55 muscle showed a leftward-shift of the force–sarcomere length relationship, indicating shorter thin filament lengths (Fig. 7A). The descending limb of NebΔExon55 fibres intercepts the x-axis at a sarcomere length of ∼3.4 μm, a length that is consistent with thin filament lengths of ∼0.9 μm, which is close to the length as determined by the confocal microscopy studies (0.85 μm).
Figure 7.
(A) The force–sarcomere length relation of tibialis cranialis fibres from 6-day-old NebΔExon55 and wild-type (wt) mice, with force normalized to maximal force at the optimal length: the relation is shifted leftward in NebΔExon55 fibres suggesting shorter thin filaments in NebΔExon55 fibres. (B) Top: The maximal tension–sarcomere length relation of NebΔExon55 and wild-type fibres, illustrating that NebΔExon55 fibres develop at all sarcomere lengths a reduced level of force. This indicates that other factors, in addition to shorter thin filaments, contribute to the weakness of NebΔExon55 muscle. Bottom: At a sarcomere length of 2.4 μm, maximal tension is reduced in fibres from NebΔExon55 muscle to ∼38% of wild-type values. (C) Left: Force tracing of activated single myofibrils from wild-type and NebΔExon55 muscle. Right: Maximal tension of NebΔExon55 myofibrils is reduced to ∼36% of wild-type values.
Considering that thin filament length is an important determinant of a muscle’s force generating capacity, we also plotted tension (force normalized to fibre cross-sectional area) versus sarcomere length (Fig. 7B). Importantly, at a sarcomere length of 2.4 μm, a length on the plateau of the force–sarcomere length relation of both genotypes, absolute maximal tension was reduced in fibres from NebΔExon55 muscle to ∼38% of wild-type values (Fig. 7B). To study whether the reduced maximal tension of NebΔExon55 fibres is caused by a reduction in myofibrillar fractional area or rather by changes in myofibrillar function, we also determined the contractile force of single myofibrils isolated from NebΔExon55 and wild-type muscle fibres. As shown in Fig. 7C, the maximal tension generated by NebΔExon55 myofibrils was reduced to ∼36% of wild-type values, a reduction that is similar in magnitude to that observed in NebΔExon55 fibres. Thus, these findings indicate that muscle from NebΔExon55 mice is significantly weakened, that this weakening is primarily caused by changes in myofibrillar function, and that reduction of thin filament length contributes to weakness especially at longer sarcomere lengths.
Crossbridge cycling kinetics
Recent work has led to the notion that nebulin is important for the tuning of crossbridge cycling kinetics to maximize force generation (Chandra et al., 2009; Ottenheijm and Granzier, 2010). To determine whether changes in crossbridge cycling kinetics contribute to the weakness observed in NebΔExon55 fibres, we determined the tension cost and the rate constant of force redevelopment, ktr, in fibres from NebΔExon55 muscle and compared results to those from wild-type muscle. To determine ktr, fibres were first isometrically activated at pCa 4.5 and when a steady force was reached, crossbridges were disengaged by performing a quick release, a brief period of unloaded shortening, and then a rapid restretch to the original length. Following restretch, force rebuilds with a time course that can be fit to a monoexponential with rate constant ktr (see Fig. 8A, left panel, for a typical force response). As shown in Fig. 8A, ktr was significantly lower in the NebΔExon55 fibres than in the wild-type fibres (4.3 ± 0.5 versus 5.9 ± 0.3 s−1, NebΔExon55 versus wild-type, respectively). Tension cost was determined by the simultaneous measurement of the breakdown of NADH and force during contraction, with NADH levels enzymatically coupled to ATP utilization (see ‘Materials and methods’ section). An example of a maximally-activated NebΔExon55 fibre preparation with [NADH] falling linearly during the tension plateau is shown in Fig. 8B. The slope of the [NADH] versus time curve was normalized by the fibre volume to obtain ATP consumption rates that can be compared for differently sized muscle preparations. By normalizing ATP consumption rates to the tension generated and fibre volume, the tension cost can be determined. As shown in Fig. 8B, the tension cost was significantly higher in NebΔExon55 fibres compared with wild-type fibres (10.4 ± 1.1 versus 6.4 ± 0.4 pmol/mN/mm/s, NebΔExon55 versus wild-type, respectively).
Next, we determined the activation and relaxation kinetics of single myofibrils isolated from NebΔExon55 and wild-type muscle fibres. Due to their small size (diameter: 1–2 μm) and when combined with a rapid solution switching mechanism (Tesi et al., 2002) force activation and relaxation kinetics can be determined. As shown in Fig. 8C and D, the rate constants of activation (kact) and of force redevelopment following a rapid-release rapid-restretch protocol (ktr) were significantly reduced in NebΔExon55 myofibrils; kact: 1.46 ± 0.07 versus 2.89 ± 0.21 s-1, NebΔExon55 versus wild-type, respectively; ktr: 2.27 ± 0.16 versus 3.29 ± 0.25 s-1, NebΔExon55 versus wild-type, respectively. Note that the magnitude of ktr reduction in myofibrils was in the same order as observed in NebΔExon55 fibres. The relaxation phase of myofibrils consists of an initial slow phase for which we determined the rate constant ‘slow krel’ – which is thought to reflect the apparent rate constant of detachment of force bearing crossbridges, gapp (tension cost is also proportional to gapp) (Poggesi et al., 2005). The subsequent fast relaxation phase is characterized by the ‘fast krel’. Force relaxation kinetics in the NebΔExon55 myofibrils were significantly faster than in wild-type myofibrils: slow krel: 1.17 ± 0.15 versus 0.56 ± 0.05 s-1, NebΔExon55 versus wild-type, respectively; fast krel: 11.0 ± 1.5 versus 6.0 ± 0.04 s-1 NebΔExon55 versus wild-type, respectively (Fig. 8D). Note that the magnitude of increase in slow krel in NebΔExon55 myofibrils is close to that of the increase in tension cost in fibres.
Thus, these mechanical studies on both fibres and single myofibrils show that crossbridge cycling kinetics are significantly altered in muscle from NebΔExon55 mice.
Calcium sensitivity of force
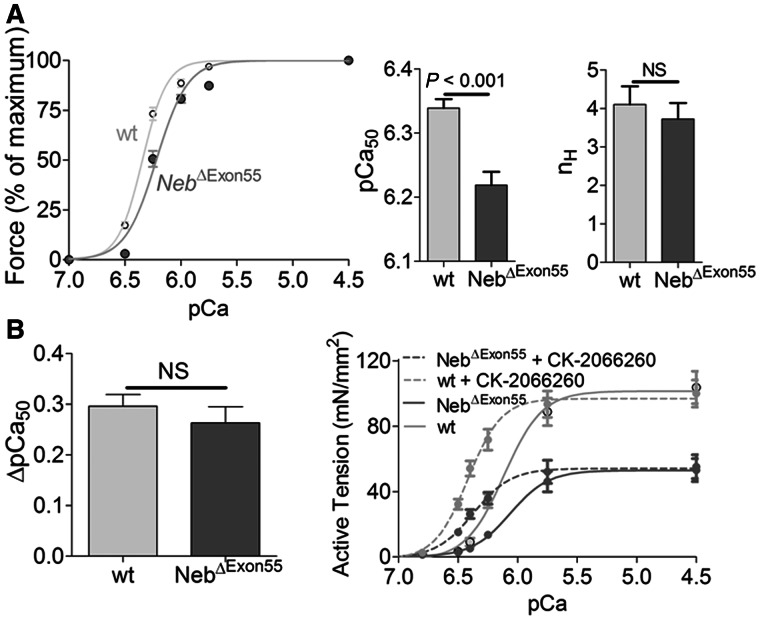
The calcium sensitivity of force generation is also an important parameter of muscle function as it reflects the response of force to submaximal activation levels, levels at which muscle typically operates in vivo. To determine the calcium sensitivity of force, permeabilized fibres from tibialis cranialis muscle were exposed to incremental calcium concentrations and the force response was recorded. As shown in Fig. 9A, the force–pCa curve of NebΔExon55 fibres was shifted rightward (reflected by a lower pCa50 in NebΔExon55 fibres: 6.22 ± 0.02 versus 6.34 ± 0.01, NebΔExon55 versus wild-type, respectively), indicating that the calcium sensitivity of force was significantly reduced in fibres from NebΔExon55 mice compared to those from wild-type mice. The cooperativity of activation (nH) was not different between groups. Thus, the calcium sensitivity of force is reduced in NebΔExon55 muscle.
Figure 9.
(A) Force–Ca2+ characteristics of tibialis cranialis fibres from 5-day-old NebΔExon55 and wild-type mice. Left: Note the rightward shift of the force–pCa relationship in NebΔExon55 compared with wild-type fibres. Middle: The Ca2+ concentration needed for 50% of maximal force generation was significantly higher (i.e. lower pCa50) in NebΔExon55 versus wild-type fibres, whereas no difference was found in the Hill coefficient (nH, right). (B) The effect of the fast troponin activator CK-2066260 on the force–Ca2+ characteristics in NebΔExon55 and wild-type fibres. Left: 5 μM of CK-2066260 induced a large increase in the pCa50, an increase that was comparable between NebΔExon55 and wild-type fibres. Right: In NebΔExon55 fibres, 5 μM CK-2066260 increased active tension at submaximal (Ca2+) to values that exceeded those of untreated wild-type fibres. NS = not significant.
Next, we studied the ability of the novel compound CK-2066260 to restore the calcium sensitivity of force in NebΔExon55 fibres from tibialis cranialis muscle. CK-2066260 is a recently developed skeletal muscle troponin activator that amplifies the response of the thin filament to calcium in fast skeletal muscle fibres (Russell et al., 2012). As shown in Fig. 9B, 5 μM of CK-2066260 induced a profound increase in the pCa50, an increase that was comparable between NebΔExon55 and wild-type fibres (ΔpCa50: 0.26 ± 0.03 versus 0.29 ± 0.02, NebΔExon55 versus wild-type, respectively). Importantly, in NebΔExon55 fibres, CK-2066260 increased active tension at submaximal (Ca2+) to values that exceeded those of untreated wild-type fibres (Fig. 9C). These findings show the promise of fast skeletal troponin activation in restoring muscle strength in nebulin-based nemaline myopathy.
Discussion
We generated the first mouse model for nebulin-based nemaline myopathy by targeted deletion of Neb exon 55. Our findings show that muscle from NebΔExon55 mice is severely weakened and contains nemaline rods, both hallmark features of nemaline myopathy. The muscle weakness in NebΔExon55 mice is caused by hypotrophy of muscle fibres, and by the muscle fibres having shorter thin filaments, changes in crossbridge cycling kinetics and reduced calcium sensitivity of force generation. A novel drug that facilitates calcium binding to the thin filament significantly augmented muscle force to levels that exceed those observed in untreated control muscle. As we discuss in detail below, the overall phenotype of this new mouse model recapitulates important features observed previously in patients with nemaline myopathy with deletion of Neb exon 55.
Deletion of exon 55 leads to severely reduced nebulin protein levels
Nebulin is a giant protein expressed at high levels in skeletal muscle. Most of the molecule is comprised of centrally located modules (M9 to M162) that are organized into seven-module super-repeats that match the repeat of the actin filament (Fig. 1). This precise arrangement is thought to allow each nebulin module to interact with a single monomer of the actin filament (Labeit et al., 1991; Labeit and Kolmerer, 1995), and each nebulin super-repeat to associate with a single tropomyosin (Tm)/troponin (Tn) complex (Jin and Wang, 1991; McElhinny et al., 2003; Ogut et al., 2003). NEB exon 55 encodes 35 amino acids that are part of modules 69 and 70 in super-repeat 9 (Fig. 1). The absence of exon 55 in the nebulin transcript does not generate a frameshift, and the transcript is predicted to encode a protein that is 35 amino acids smaller than the normal gene product. However, as two of the encoded 35 amino acid modules of nebulin are interrupted by the deletion of exon 55, this results in the disruption of the seven-module set of super-repeat 9 (Labeit and Kolmerer, 1995). It has been proposed that this disruption causes a mismatch between nebulin and its binding sites on actin (Anderson et al., 2004), thereby preventing the incorporation of mutant nebulin molecules and leading to their degradation (Ottenheijm et al., 2009b). In line with this proposition, a study on five patients homozygous for deletion of NEB exon 55 showed that the muscle fibres of these patients have nebulin levels that are <15% of normal values (Ottenheijm et al., 2009b). The present work shows that nebulin protein was severely reduced in all muscle types tested from NebΔExon55 mice, including in embryonic muscle (Fig. 4), suggesting that muscle loading after birth did not greatly accelerate the loss of nebulin protein. Hence, the proteomic phenotype of NebΔExon55 mice resembles that of the full nebulin knockout models (Bang et al., 2006; Witt et al., 2006), although nebulin protein levels in the NebΔExon55 mice are slightly higher and more similar to those observed in patients (Ottenheijm et al., 2009b). The nebulin exon microarray studies (Fig. 5) revealed the downregulation of nearly all nebulin exons at the RNA level, not only exon 55. Thus, the low nebulin protein levels appear to be caused by defects at the transcript level, rather than by degradation of mutant nebulin protein. The mechanisms underlying the more severe reduction of nebulin levels in NebΔExon55 mice compared to humans with the same mutation is unclear. An explanation could be that most patients with NEB exon 55 deletion also have very little nebulin protein and that they die in utero. However, the rate of miscarriage in the families that carry the NEB exon 55 mutation appears to be within the normal range (Beggs et al., in preparation) making this explanation unlikely. Alternatively, nebulin isoform composition might play a role: if inclusion of exon 55 is mandatory in the mouse and less so in humans, then this would more severely affect nebulin protein levels in NebΔExon55 mice. Transcript studies in the mouse showed no differential splicing of exon 55 (Buck et al., 2010), consistent with the absence of nebulin expression in any of the muscle types that we studied (Fig. 4A and B). Thus if alternative splicing of human nebulin transcripts were to occur that can produce some functional nebulin isoforms devoid exon 55, this could explain why nebulin levels in NebΔExon55 mice are more severely reduced than in humans with the same mutation.
Severe weakness of muscle fibres caused by thin filament dysfunction
A hallmark feature of patients with nemaline myopathy is muscle weakness, greatly affecting daily life activities and negatively impacting the quality of life (North et al., 1997). At the structural level, nemaline myopathy is characterized by myofibrillar disarray originating from the Z-disk and culminating in nemaline rods, which consist of thin filament and Z-disk proteins (Engel and Gomez, 1967; Yamaguchi et al., 1978, 1982; Morris et al., 1990; North, et al., 1997; Sanoudou and Beggs, 2001). Importantly, the number of nemaline rods in muscle biopsies does not correlate with muscle weakness in patients with nemaline myopathy (Shimomura and Nonaka, 1989; Ryan et al., 2003). This suggests that myofibrillar disarray and nemaline rods are a secondary phenotype and are not the sole contributors to the muscle weakness associated with nemaline myopathy. The NebΔExon55 model has a relative paucity of rod bodies yet muscle weakness is severe, supporting the notion that muscle weakness in nemaline myopathy with nebulin mutations is caused primarily by changes in myofilament function.
The maximal tension generated by muscle fibres from NebΔExon55 mice was severely reduced (Fig. 7B and 9B), and based on electron microscopy studies this reduction is unlikely to be due to myofibrillar disarray (Fig. 3B). This conclusion is supported by the studies on the maximal tension generation of single myofibrils (Fig. 7C): the magnitude of tension reduction in NebΔExon55 myofibrils was similar to that in NebΔExon55 fibres (62% and 64%, respectively). Thus, the weakness of muscle fibres in NebΔExon55 mice can be attributed to changes in myofibrillar function, rather than by myofibrillar disarray or by a reduced myofibrillar fractional area. Note that the maximal tension generated by myofibrils was higher than that by muscle fibres, both in wild-type and NebΔExon55 mice, and that this is caused by the tension in fibres being normalized to a cross-sectional area that comprises myofibrils plus non-contractile material such as mitochondria, glycogen granules and sarcoplasmic reticulum. The 64% reduction in tension generation by fibres from NebΔExon55 muscle, combined with the reduction in fibre diameter of 23%, results in a total force loss of >80% in NebΔExon55 fibres when compared with wild-type fibres. This severe muscle fibre weakness in NebΔExon55 mice explains why the mice die within several days of birth, as this will severely hamper both feeding (the stomachs of NebΔExon55 mice are typically devoid of milk) and breathing. That many patients homozygous for deletion of NEB exon 55 have been shown to survive past the neonatal phase (Anderson et al., 2004; Lehtokari et al., 2006, 2009; Ottenheijm et al., 2009b, 2010) is likely to be partly due to the extensive medical care that includes gastro-nasal feeding and mechanical ventilation. It should be noted that some patients with deletion of NEB exon 55, those with the typical form of nemaline myopathy, remain ambulant into adolescence or beyond. Future studies should address whether these patients have higher nebulin protein levels and a better preserved myofibrillar function.
The reduction in tension that we found in the mouse might be due to any of the myofibrillar functions that nebulin has been proposed to play: (i) specifying thin filament length; (ii) regulating the interaction between myosin-based crossbridges and the thin filament; and (iii) tuning the response of the thin filament to calcium (Bang et al., 2009; Chandra et al., 2009; Ottenheijm and Granzier, 2010;). Our work on the NebΔExon55 mice indicates that all three mechanisms are disturbed upon deletion of Neb exon 55, and contribute to the development of muscle weakness.
In skeletal muscle, thin filament lengths are fine-tuned at ∼1.1–1.3 µm (depending on species and muscle type) (Littlefield and Fowler, 2008) to overlap with thick filaments and to meet the muscle’s contractile requirements (Burkholder et al., 1994; Littlefield and Fowler, 2008). The extent of overlap between thick and thin filaments determines the sarcomere’s force generating capacity: short thin filaments reduce overlap and impair force generation. Thus, thin filament length is a key aspect of muscle function, and recent work indicates that nebulin is involved in specifying this length (Bang et al., 2006; Witt et al., 2006). Our immunofluorescence confocal microscopy studies revealed that thin filament length is significantly reduced from 1.0 μm in wild-type muscle to 0.85 μm in NebΔExon55 muscle. These structural studies were corroborated by mechanical studies, which revealed a leftward shift (at sarcomere lengths of 2.4 μm and longer) of the force–sarcomere length relation in NebΔExon55 muscle fibres. Thus, deletion of Neb exon 55 results in shorter thin filament lengths, contributing to muscle weakness at longer sarcomere lengths.
Weakness of NebΔExon55 fibres was also apparent at sarcomere lengths shorter than 2.4 μm (Fig. 7B), suggesting that the reduction of thin filament length is not the only mechanism for weakness in NebΔExon55 mice. Our findings show that crossbridge cycling kinetics in NebΔExon55 fibres are altered, and that this contributes to muscle weakness. NebΔExon55 muscle fibres had significantly higher tension cost compared to wild-type fibres (Fig. 8B), which indicates that the crossbridge detachment rate is higher (Stienen et al., 1996). In combination with the observation that ktr was slower in NebΔExon55 fibres (Fig. 8A), these findings lead to the conclusion that the rate of crossbridge attachment is slower in NebΔExon55 fibres. Consequently, the fraction of force generating crossbridges is significantly reduced in NebΔExon55 muscle. Importantly, these findings on NebΔExon55 fibres were strongly supported by results from studies on single myobrils (Fig. 8C and D). The advantages of studies on myofibrils include that, due to their small size, they instantly equilibrate with the bathing solution making it possible to study force relaxation kinetics. Furthermore, as myofibrillar damage typically results in breakage of the myofibrils during activation, the data obtained from myofibrils reflect direct effects of the mutated protein on crossbridge cycling kinetics without potential confounding effects of myofibrillar damage. Importantly, the magnitude of the ktr changes in myofibrils were similar to that in fibres and the changes in slow krel of myofibrils are similar to that of the tension cost measured in fibres. Thus, measurement at two distinct levels of organization (myofibrils and fibres) and using distinct techniques (tension cost measurements and force relaxation kinetics) support the hypothesis that alterations in crossbridge cycling kinetics greatly contribute to the observed weakness of NebΔExon55 muscle.
The studies discussed above were carried out at a maximal activating calcium level. However, in vivo, skeletal muscle typically is submaximally activated. Thus, submaximal parameters of muscle function provide relevant physiological information. To test whether submaximal force generation was affected in NebΔExon55 fibres, we exposed permeabilized fibres to various calcium concentrations and determined the ensuing force level. We found that the calcium sensitivity of force generation was significantly reduced in NebΔExon55 fibres, suggesting that in addition to maximal force generation, the capacity for submaximal force generation is greatly impaired in NebΔExon55 mice.
In summary, muscle of NebΔExon55 mice is nebulin-deficient and produces weakness by shortening thin filament lengths, reducing the fraction of crossbridges in the force generating state, and reducing the force response to submaximal calcium concentrations.
Troponin activation: a therapeutic mechanisms to improve muscle strength?
To date, no therapy exists that enhances force generation in patients with nemaline myopathy with nebulin mutations. Strategies to restore thin filament length or crossbridge cycling kinetics currently do not exist for skeletal muscle and, in part due to the extremely large size of the nebulin gene and protein, effective genetic strategies to combat these effects through gene therapy are likely far off in the future. However, the lower calcium sensitivity of force generation in patients with nemaline myopathy with nebulin mutations (Ottenheijm et al., 2010) might offer a more immediate therapeutic target, since a fast skeletal muscle troponin activator has been developed that amplifies the response of the thin filament to calcium in fast skeletal muscle fibres (Russell et al., 2012). The fast skeletal muscle troponin activator ‘tirasemtiv’ (formerly CK-2017357) was shown to greatly increase the calcium-sensitivity of force generation in healthy rat skeletal muscle fibres (Russell et al., 2012), and is currently in phase IIa clinical studies for generalized myasthenia gravis, and IIb clinical trials for amyotrophic lateral sclerosis (Shefner et al., 2012).
Here we took advantage of the novel NebΔExon55 model to test the ability of the fast skeletal troponin activator CK-2066260 to improve muscle fibre strength in NebΔExon55 fibres. CK-2066260 is a close structural analogue of tirasemtiv and the pharmacology of the two compounds is the same in vitro and in vivo. Our findings indicate that CK-2066260 greatly increases force generation in NebΔExon55 fibres to levels that even exceed those observed in untreated muscle cells from wild-type mice (Fig. 9B). Thus, fast skeletal troponin activation might offer a therapeutic strategy for the restoration of muscle strength in patients with nemaline myopathy with deletion of nebulin exon 55, and possibly in patients with other nebulin mutations. Future studies should address whether the in vivo administration of troponin activators to nebulin knockout mouse models, such as the NebΔExon55 mice, improves their survival.
In conclusion, we have generated a mouse model in which Neb exon 55 is deleted to model a founder mutation seen frequently in patients with nemaline myopathy. This is the first nebulin-based model for nemaline myopathy. Our data indicate that the phenotype of NebΔExon55 mice recapitulates important features that we observed previously in patients harbouring this particular mutation. This severe muscle weakness, which characterizes the phenotype of both the mouse model and patients, can be restored by a novel compound that augments the response of the thin filament to calcium.
Funding
This work was supported by generous support by ‘A Foundation Building Strength for Nemaline Myopathy’ a VIDI grant (016.126.319) from the Netherlands Organization for Scientific Research to C.O., by the EU-FP7 and ERARE (SarcoSi and NEMMYOP) to S.L. and C.O., by PRIN 2010-2011 of the Ministero Università e Ricerca (MIUR), and by NIH R01 AR053897 to H.G., K08 AR059750, L40 AR057721 to M.W.L., R01 AR044345 to A.H.B., and grant MDA201302 from the Muscular Dystrophy Association (USA). Electron microscopy was performed at the Microbiology and Molecular Genetic EM Facility at MCW, which is supported by grants from the NIH (1S10RR022412-01) and ‘Advancing a Healthier Wisconsin Program’ from the MCW.
Acknowledgements
We gratefully acknowledge the Genetic Engineering of Mouse models (GEMM) core and its Director Dr. Tom Doetschman for making the NebΔExon55 mouse model. Immunofluorescence microscopy for muscle fibre size was performed using the Children’s Research Institute’s Imaging Core Facility at MCW.
References
- Anderson SL, Ekstein J, Donnelly MC, Keefe EM, Toto NR, LeVoci LA, et al. Nemaline myopathy in the Ashkenazi Jewish population is caused by a deletion in the nebulin gene. Hum Genet. 2004;115:185–90. doi: 10.1007/s00439-004-1140-8. [DOI] [PubMed] [Google Scholar]
- Bang ML, Caremani M, Brunello E, Littlefield R, Lieber RL, Chen J, et al. Nebulin plays a direct role in promoting strong actin-myosin interactions. FASEB J. 2009;23:4117–25. doi: 10.1096/fj.09-137729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bang ML, Li X, Littlefield R, Bremner S, Thor A, Knowlton KU, et al. Nebulin-deficient mice exhibit shorter thin filament lengths and reduced contractile function in skeletal muscle. J Cell Biol. 2006;173:905–16. doi: 10.1083/jcb.200603119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenner B, Eisenberg E. Rate of force generation in muscle: correlation with actomyosin ATPase activity in solution. Proc Natl Acad Sci USA. 1986;83:3542–6. doi: 10.1073/pnas.83.10.3542. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buck D, Hudson BD, Ottenheijm CA, Labeit S, Granzier H. Differential splicing of the large sarcomeric protein nebulin during skeletal muscle development. J Struct Biol. 2010;170:325–33. doi: 10.1016/j.jsb.2010.02.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burkholder TJ, Fingado B, Baron S, Lieber RL. Relationship between muscle fiber types and sizes and muscle architectural properties in the mouse hindlimb. J Morphol. 1994;221:177–90. doi: 10.1002/jmor.1052210207. [DOI] [PubMed] [Google Scholar]
- Castillo A, Nowak R, Littlefield KP, Fowler VM, Littlefield RS. A nebulin ruler does not dictate thin filament lengths. Biophys J. 2009;96:1856–65. doi: 10.1016/j.bpj.2008.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandra M, Mamidi R, Ford S, Hidalgo C, Witt C, Ottenheijm C, et al. Nebulin alters cross-bridge cycling kinetics and increases thin filament activation: a novel mechanism for increasing tension and reducing tension cost. J Biol Chem. 2009;284:30889–96. doi: 10.1074/jbc.M109.049718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engel AG, Gomez MR. Nemaline (Z disk) myopathy: observations on the origin, structure, and solubility properties of the nemaline structures. J Neuropathol Exp Neurol. 1967;26:601–19. doi: 10.1097/00005072-196710000-00007. [DOI] [PubMed] [Google Scholar]
- Gokhin DS, Bang ML, Zhang J, Chen J, Lieber RL. Reduced thin filament length in nebulin-knockout skeletal muscle alters isometric contractile properties. Am J Physiol Cell Physiol. 2009;296:C1123–32. doi: 10.1152/ajpcell.00503.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Granzier HL, Akster HA, Ter Keurs HE. Effect of thin filament length on the force-sarcomere length relation of skeletal muscle. Am J Physiol. 1991;260:C1060–70. doi: 10.1152/ajpcell.1991.260.5.C1060. [DOI] [PubMed] [Google Scholar]
- Granzier HL, Wang K. Interplay between passive tension and strong and weak binding cross-bridges in insect indirect flight muscle. A functional dissection by gelsolin-mediated thin filament removal. J Gen Physiol. 1993;101:235–70. doi: 10.1085/jgp.101.2.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greer KA, McReynolds MR, Brooks HL, Hoying JB. CARMA: a platform for analyzing microarray datasets that incorporate replicate measures. BMC Bioinformatics. 2006;7:149. doi: 10.1186/1471-2105-7-149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin JP, Wang K. Cloning, expression, and protein interaction of human nebulin fragments composed of varying numbers of sequence modules. J Biol Chem. 1991;266:21215–23. [PubMed] [Google Scholar]
- Jungbluth H, Sewry CA, Counsell S, Allsop J, Chattopadhyay A, Mercuri E, et al. Magnetic resonance imaging of muscle in nemaline myopathy. Neuromuscul Disord. 2004;14:779–84. doi: 10.1016/j.nmd.2004.08.005. [DOI] [PubMed] [Google Scholar]
- Kazmierski ST, Antin PB, Witt CC, Huebner N, McElhinny AS, Labeit S, et al. The complete mouse nebulin gene sequence and the identification of cardiac nebulin. J Mol Biol. 2003;328:835–46. doi: 10.1016/s0022-2836(03)00348-6. [DOI] [PubMed] [Google Scholar]
- Labeit S, Gibson T, Lakey A, Leonard K, Zeviani M, Knight P, et al. Evidence that nebulin is a protein-ruler in muscle thin filaments. FEBS Lett. 1991;282:313–6. doi: 10.1016/0014-5793(91)80503-u. [DOI] [PubMed] [Google Scholar]
- Labeit S, Kolmerer B. The complete primary structure of human nebulin and its correlation to muscle structure. J Mol Biol. 1995;248:308–15. doi: 10.1016/s0022-2836(95)80052-2. [DOI] [PubMed] [Google Scholar]
- Lawlor MW, Ottenheijm CA, Lehtokari VL, Cho K, Pelin K, Wallgren-Pettersson C, et al. Novel mutations in NEB cause abnormal nebulin expression and markedly impaired muscle force generation in severe nemaline myopathy. Skelet Muscle. 2011;1:23. doi: 10.1186/2044-5040-1-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehtokari VL, Greenleaf RS, DeChene ET, Kellinsalmi M, Pelin K, Laing NG, et al. The exon 55 deletion in the nebulin gene—one single founder mutation with world-wide occurrence. Neuromuscul Disord. 2009;19:179–81. doi: 10.1016/j.nmd.2008.12.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lehtokari VL, Pelin K, Sandbacka M, Ranta S, Donner K, Muntoni F, et al. Identification of 45 novel mutations in the nebulin gene associated with autosomal recessive nemaline myopathy. Hum Mutat. 2006;27:946–56. doi: 10.1002/humu.20370. [DOI] [PubMed] [Google Scholar]
- Littlefield RS, Fowler VM. Thin filament length regulation in striated muscle sarcomeres: pointed-end dynamics go beyond a nebulin ruler. Semin Cell Dev Biol. 2008;19:511–9. doi: 10.1016/j.semcdb.2008.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McElhinny AS, Kazmierski ST, Labeit S, Gregorio CC. Nebulin: the nebulous, multifunctional giant of striated muscle. Trends Cardiovasc Med. 2003;13:195–201. doi: 10.1016/s1050-1738(03)00076-8. [DOI] [PubMed] [Google Scholar]
- Morris EP, Nneji G, Squire JM. The three-dimensional structure of the nemaline rod Z-band. J Cell Biol. 1990;111:2961–78. doi: 10.1083/jcb.111.6.2961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- North KN, Laing NG, Wallgren-Pettersson C. Nemaline myopathy: current concepts. The ENMC International Consortium and Nemaline Myopathy. J Med Genet. 1997;34:705–13. doi: 10.1136/jmg.34.9.705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ochala J, Lehtokari VL, Iwamoto H, Li M, Feng HZ, Jin JP, et al. Disrupted myosin cross-bridge cycling kinetics triggers muscle weakness in nebulin-related myopathy. FASEB J. 2011;25:1903–13. doi: 10.1096/fj.10-176727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ogut O, Hossain MM, Jin JP. Interactions between nebulin-like motifs and thin filament regulatory proteins. J Biol Chem. 2003;278:3089–97. doi: 10.1074/jbc.M205853200. [DOI] [PubMed] [Google Scholar]
- Ottenheijm CA, Granzier H. Lifting the nebula: novel insights into skeletal muscle contractility. Physiology (Bethesda) 2010;25:304–10. doi: 10.1152/physiol.00016.2010. [DOI] [PubMed] [Google Scholar]
- Ottenheijm CA, Hidalgo C, Rost K, Gotthardt M, Granzier H. Altered contractility of skeletal muscle in mice deficient in titin's M-band region. J Mol Biol. 2009a;393:10–26. doi: 10.1016/j.jmb.2009.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottenheijm CA, Hooijman P, DeChene ET, Stienen GJ, Beggs AH, Granzier H. Altered myofilament function depresses force generation in patients with nebulin-based nemaline myopathy (NEM2) J Struct Biol. 2010;170:334–43. doi: 10.1016/j.jsb.2009.11.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottenheijm CA, Lawlor MW, Stienen GJ, Granzier H, Beggs AH. Changes in cross-bridge cycling underlie muscle weakness in patients with tropomyosin 3-based myopathy. Hum Mol Genet. 2011;20:2015–25. doi: 10.1093/hmg/ddr084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ottenheijm CA, Witt CC, Stienen GJ, Labeit S, Beggs AH, Granzier H. Thin filament length dysregulation contributes to muscle weakness in nemaline myopathy patients with nebulin deficiency. Hum Mol Genet. 2009b;18:2359–69. doi: 10.1093/hmg/ddp168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pappas CT, Krieg PA, Gregorio CC. Nebulin regulates actin filament lengths by a stabilization mechanism. J Cell Biol. 2010;189:859–70. doi: 10.1083/jcb.201001043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelin K, Hilpela P, Donner K, Sewry C, Akkari PA, Wilton SD, et al. Mutations in the nebulin gene associated with autosomal recessive nemaline myopathy. Proc Natl Acad Sci USA. 1999;96:2305–10. doi: 10.1073/pnas.96.5.2305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Poggesi C, Tesi C, Stehle R. Sarcomeric determinants of striated muscle relaxation kinetics. Pflugers Arch. 2005;449:505–17. doi: 10.1007/s00424-004-1363-5. [DOI] [PubMed] [Google Scholar]
- Russell AJ, Hartman JJ, Hinken AC, Muci AR, Kawas R, Driscoll L, et al. Activation of fast skeletal muscle troponin as a potential therapeutic approach for treating neuromuscular diseases. Nat Med. 2012;18:452–5. doi: 10.1038/nm.2618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan MM, Ilkovski B, Strickland CD, Schnell C, Sanoudou D, Midgett C, et al. Clinical course correlates poorly with muscle pathology in nemaline myopathy. Neurology. 2003;60:665–73. doi: 10.1212/01.wnl.0000046585.81304.bc. [DOI] [PubMed] [Google Scholar]
- Sambuughin N, Swietnicki W, Techtmann S, Matrosova V, Wallace T, Goldfarb L, et al. KBTBD13 interacts with Cullin 3 to form a functional ubiquitin ligase. Biochem Biophys Res Commun. 2012;421:743–9. doi: 10.1016/j.bbrc.2012.04.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sambuughin N, Yau KS, Olive M, Duff RM, Bayarsaikhan M, Lu S, et al. Dominant mutations in KBTBD13, a member of the BTB/Kelch family, cause nemaline myopathy with cores. Am J Hum Genet. 2010;87:842–7. doi: 10.1016/j.ajhg.2010.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanoudou D, Beggs AH. Clinical and genetic heterogeneity in nemaline myopathy—a disease of skeletal muscle thin filaments. Trends Mol Med. 2001;7:362–8. doi: 10.1016/s1471-4914(01)02089-5. [DOI] [PubMed] [Google Scholar]
- Shefner J, Cedarbaum JM, Cudkowicz ME, Maragakis N, Lee J, Jones D, et al. Safety, tolerability and pharmacodynamics of a skeletal muscle activator in amyotrophic lateral sclerosis. Amyotroph Lateral Scler. 2012;13:430–8. doi: 10.3109/17482968.2012.684214. [DOI] [PubMed] [Google Scholar]
- Shimomura C, Nonaka I. Nemaline myopathy: comparative muscle histochemistry in the severe neonatal, moderate congenital, and adult-onset forms. Pediatr Neurol. 1989;5:25–31. doi: 10.1016/0887-8994(89)90005-2. [DOI] [PubMed] [Google Scholar]
- Stienen GJ, Kiers JL, Bottinelli R, Reggiani C. Myofibrillar ATPase activity in skinned human skeletal muscle fibres: fibre type and temperature dependence. J Physiol. 1996;493(Pt 2):299–307. doi: 10.1113/jphysiol.1996.sp021384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tesi C, Piroddi N, Colomo F, Poggesi C. Relaxation kinetics following sudden Ca(2+) reduction in single myofibrils from skeletal muscle. Biophys J. 2002;83:2142–51. doi: 10.1016/S0006-3495(02)73974-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warren CM, Krzesinski PR, Greaser ML. Vertical agarose gel electrophoresis and electroblotting of high-molecular-weight proteins. Electrophoresis. 2003;24:1695–702. doi: 10.1002/elps.200305392. [DOI] [PubMed] [Google Scholar]
- Witt CC, Burkart C, Labeit D, McNabb M, Wu Y, Granzier H, et al. Nebulin regulates thin filament length, contractility, and Z-disk structure in vivo. EMBO J. 2006;25:3843–55. doi: 10.1038/sj.emboj.7601242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamaguchi M, Robson RM, Stromer MH, Dahl DS, Oda T. Actin filaments form the backbone of nemaline myopathy rods. Nature. 1978;271:265–7. doi: 10.1038/271265a0. [DOI] [PubMed] [Google Scholar]
- Yamaguchi M, Robson RM, Stromer MH, Dahl DS, Oda T. Nemaline myopathy rod bodies. Structure and composition. J Neurol Sci. 1982;56:35–56. doi: 10.1016/0022-510x(82)90059-4. [DOI] [PubMed] [Google Scholar]