Abstract
Descriptive epidemiology of the myelodysplastic syndromes (MDS) is always interesting and may reveal time-dependent and geographical variations, as well as occupational exposure. Epidemiological data in Greece are not available by now. We have collected and analyzed medical records of all patients with a documented diagnosis of MDS, performed by an expert hematologist and/or hematopathologist, in the geographical area of Western Greece, during the 20-year period, defined between 1990 and 2009. We have then calculated and described demographic and clinical features of the diagnosed MDS patient population, and assessed the incidence and prevalence rates of MDS in Western Greece, during the above-mentioned period. A total of 855 patients with newly diagnosed MDS have been identified. Refractory anemia was the most common subtype in both FAB and WHO classification systems and in both genders. Del-5q and RARS were more commonly encountered among females, and the dysplastic subtype of chronic myelomonocytic leukemia among males. Trisomy 8 was the most common single cytogenetic abnormality. The crude mean annual incidence rate of MDS was 6.0 per 100,000 inhabitants aged ≥15 years old (all subtypes according to FAB), and it was 4.8 per 100,000 when CMML and RAEB-T were excluded. Crude incidence rate was higher in rural than in urban areas, but this finding was not confirmed after age standardization. Age-standardized mean annual incidence rate in men was 7.9/100,000 and in women 3.4/100,000. A continuously increasing incidence rate of MDS has been observed throughout the study period.
Keywords: Myelodysplastic syndromes, Epidemiology, Incidence, Classification, Western Greece
Introduction
Myelodysplastic syndromes (MDS) are acquired clonal hematopoietic stem cell disorders, characterized by ineffective hematopoiesis, peripheral blood cytopenias, and increased risk of leukemic transformation. The incidence of MDS is not worldwide known, and this is attributed to many reasons. First, there are frequent diagnostic inconsistencies, leading to erroneous classification of many cases with borderline features, as other diseases, such as aplastic anemia, anemia of inflammation, myeloproliferative neoplasms, and acute myelogenous leukemia. Second, there is occasional difficulty in making the correct diagnosis, especially when morphologic features are subtle or inadequate [1]. Indeed, dysplastic features in the peripheral blood or bone marrow (BM) may accompany a large number of chronic diseases or conditions, particularly in the elderly, of inflammatory, endocrine, autoimmune, or neoplastic etiology, or they may be a consequence of advanced age alone [2]. Third, the diagnosis of MDS in very elderly patients may not be confirmed, due to reluctance of patients and/or physicians to perform a BM examination [3]. Another reason for lacking data is that systemic registration of MDS has only recently been started, even in countries with well-organized and active cancer registries.
The incidence of MDS varies significantly by geographical area, and this has been attributed to genetic, occupational, lifestyle, and environmental factors, not fully elucidated as yet [4]. According to published epidemiological studies, the mean annual incidence varies between 1/100,000 inhabitants in Japan [5] and 12.6 in the area of Bournemouth, UK (1981–1990) [6]. Data on the incidence and prevalence of MDS in Greece have not been published by now.
The objective of the present study was to describe demographic and clinical features of patients with MDS in Western Greece, during the previous two decades, and to estimate incidence and prevalence rates in a demographically well-defined population of the same area.
Materials and methods
Western Greece is defined geographically as the area southern of Epirus, western of Fokida and Eurytania in the Greek mainland, as well as western of Korinthia and Arcadia, and northern of Messinia in Peloponnese (Fig. 1). In this area, consisting of three prefectures, Achaia, Ilia, and Etolia and Akarnania, there is one major, fully occupied hospital-referral center, the University Hospital of Patras, a second major, referral hospital, “St Andrew” Hospital, and two smaller hospitals with a Department of Hematology. The University Hospital possesses an expert Hematopathology laboratory and accepts patients from the whole area, referred by physicians of smaller hospitals and private practice. Patients are also referred from neighbor territories, such as the Ionian islands (Kefallinia, Zakynthos, Ithaca, and Lefkas), and the closest parts of Korinthia, Fokida, and Messinia. In our area patients with hematological dyscrasias are not treated or followed by other specialists or private practice centers.
Fig. 1.
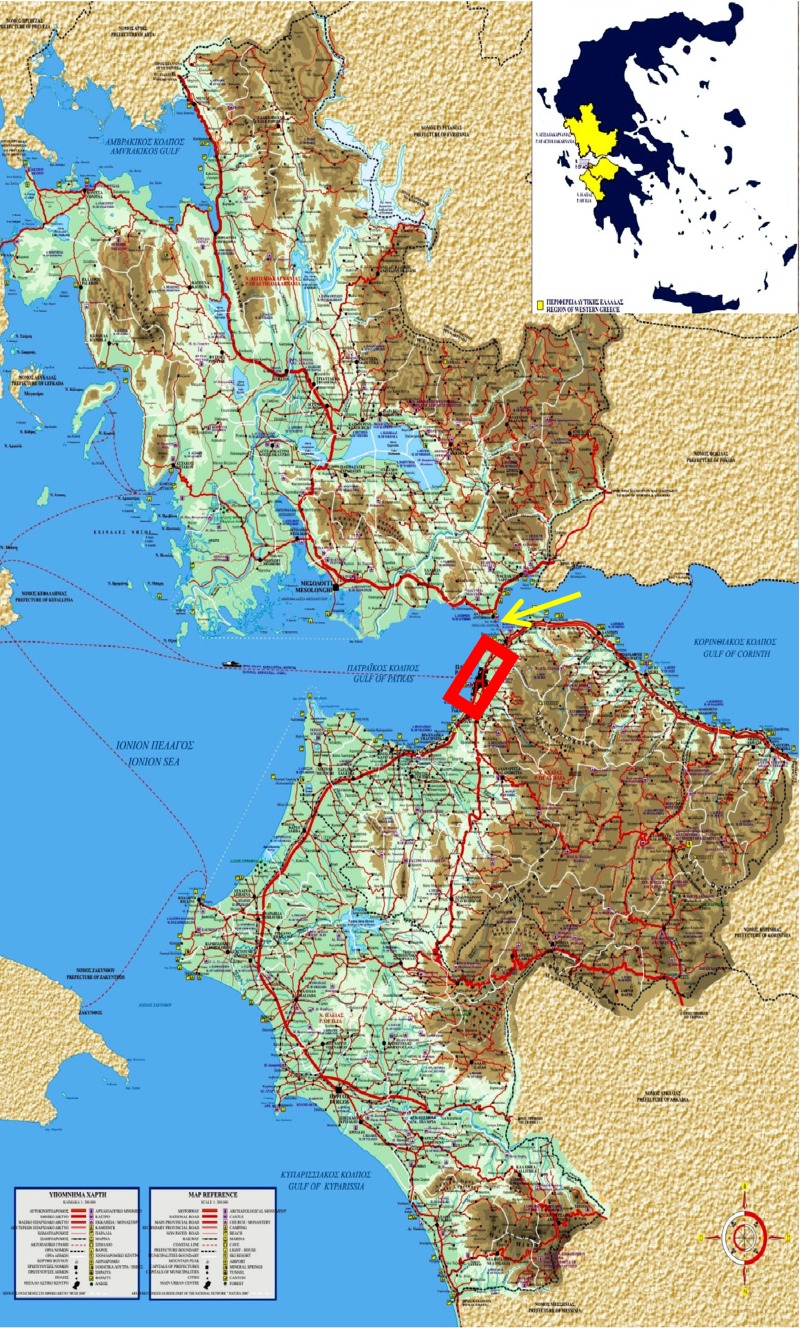
Map of Greece, where the area of Western Greece (prefectures of Achaia, Ilia, and Etolia/Akarnania) is depicted in yellow (inserted picture). In the enlarged map of Western Greece, the red rectangle indicates the urban area of Patras, regional capital of Western Greece. The yellow arrow depicts the position of the Rion-Antirion bridge, which connects Peloponnese with continental Greece
Medical records and electronic databases of the four above-mentioned hospitals were reviewed, and all patients with MDS diagnosed between 1.1.1990 and 31.12.2009 were registered. Only patients with a documented diagnosis of MDS, confirmed by BM examination (aspiration and/or biopsy), were included in the analysis. The study has been approved by the Ethical and Scientific Council of the participating hospitals.
Demographic data, as well as disease information (FAB and WHO classification, cytogenetics, laboratory findings at diagnosis, IPSS, transfusion dependency, WPSS, and leukemic evolution) were collected and analyzed for all patients (aged ≥15 years) diagnosed during the study period. Pediatric MDS cases have not been included in this analysis.
Patients residing in the three prefectures of Western Greece were then selected for the estimation of epidemiological indices. The adult population of the area was considered stable during the study period and consisted of 603,543 inhabitants. Demographic data were based on the 2001 national census, provided by the General Secretariat of the Hellenic Statistical Authority.
Statistical analysis
Statistical analysis was performed by using the Statistical Software IBM SPSS Statistics 20. t test (Student’s test) was used for the comparison of means, Chi square (χ 2) test for the comparison of percentages between subgroups, and analysis of variance was performed to compare means between several subgroups. The comparison of age-standardized incidence rates was performed using the comparative morbidity figure (CMF). The standard error and 95 % confidence intervals (95 % CI) of CMF were calculated according to the method of Breslow and Day [7, 8].
Results
Demographic features
Totally, 855 patients were diagnosed with an MDS between 1.1.1990 and 31.12.2009 in Western Greece. Among them, 606 (71 %) were males and 249 (29 %) females (M/F ratio 2.43:1). Mean age at diagnosis was 73.1 ± 18.9 years (median 74) and it was not significantly different between men and women, in each MDS subtype, and overall. However, age at diagnosis was significantly different by FAB subtype, and patients with RAEB-T were younger (p = 0.008).
Among the 855 patients, 721 (84.3 %) were living in Western Greece [Achaia (N = 371), Ilia (N = 175), Etolia/Akarnania (N = 175)], and 134 (15.7 %) in neighbor prefectures, [Kefallinia and Zakynthos (N = 41), Korinthia (N = 28), Fokida (N = 15), Messinia (N = 14), and other (N = 36)]. Four hundred and eighty-three patients (56.7 %) were living in rural or semi-urban areas, whereas 341 (39.9 %) resided in urban areas. For 29 patients (0.3 %) the area of residence was unknown.
Occupation was known in 640 patients (75 %) and almost half of them were farmers [N = 279 (44 %)]. Other frequently encountered occupations were housekeeping [N = 84 (13 %)], merchantry [N = 37 (6 %)], public administration [N = 21 (3 %)], industrial worker [N = 21 (3 %)], policeman [N = 16 (2.5 %)], office employee [N = 13 (2 %), and teacher [N = 12 (2 %)].
Clinical and laboratory features
Presenting symptoms were known in 597 patients (70 %) and the most common were fatigue in 330 (55 %), fever or infection in 88 (15 %), and bleeding in 50 (8 %). In 126 patients (21 %) diagnosis was suspected by routine blood tests, while they were completely asymptomatic.
Totally, 833 patients (97.4 %) had primary, and 22 (2.6 %) secondary or therapy-related MDS, following previous exposure to chemo-/radiotherapy. Moreover, 67 cases (7.8 %) were fibrotic MDS, based on their substantial increase of BM reticulin fibers, and additional 38 (4.4 %) had hypoplastic-type MDS (BM cellularity ≤30 %).
Baseline hematological parameters were available in 776 patients (90.8 %) and 63.5 % had moderate to severe anemia (hemoglobin <10 g/dl), 33.4 % had neutropenia (ANC <1.5x109/μl), and 40.9 % had thrombocytopenia (PLT <100 × 109/μl). Moreover, 35.4 % of the patients had various bicytopenias and 12.1 % were pancytopenic. The presence and the various combinations of cytopenias at diagnosis are summarized in Table 1.
Table 1.
Cytopenias and hematological parameters at diagnosis in 776 patients with MDS
| Cytopenia at diagnosis | N (%) | |
|---|---|---|
| Anemia (Hb <10 g/dl) | 493 (63.5) | |
| Neutropenia (ANC <1,500/mm3) | 259 (33.4) | |
| Thrombocytopenia (PLT < 100,000 mm3) | 317 (40.9) | |
| Anemia and neutropenia | 161 (20.7) | |
| Anemia and thrombocytopenia | 207 (26.7) | |
| Neutropenia and thrombocytopenia | 141 (18.2) | |
| Pancytopenia | 95 (12.2) | |
| Laboratory values at presentation | Mean | P |
| Hemoglobin—men (N = 549) | 9.6 g/dl | <0.0001 |
| Hemoglobin—women (N = 227) | 9.1 g/dl | |
| Hemoglobin—all patients | 9.5 g/dl | |
| White blood cells—men | 7.88 × 109/l | 0.096 |
| White blood cells—women | 6.66 × 109/l | |
| White blood cells—all patients | 7.52 × 109/l | |
| Abs. neutrophil count—men | 4.10 × 109/l | 0.191 |
| Abs. neutrophil count—women | 3.55 × 109/l | |
| Abs. neutrophil count—all patients | 3.94 × 109/l | |
| Platelet count—men | 154 × 109/l | 0.156 |
| Platelet count—women | 169 × 109/l | |
| Platelet count—all patients | 158 × 109/l | |
FAB and WHO classification
The distribution according to FAB classification was as follows: refractory anemia (RA) 313 patients, RA with ringed sideroblasts (RARS) 82, RA with excess of blasts (RAEB) 287, RAEB in transformation (RAEB-T) 34, chronic myelomonocytic leukemia (CMML) 123, and unclassified MDS 16. RARS was more frequent in women, yet this difference was not statistically significant.
According to the WHO classification 176 patients had RA, 16 Del(5q) syndrome, 53 RARS, 122 refractory cytopenia with multilineage dysplasia (RCMD), 24 RCMD with ringed sideroblasts (RCMD-RS), 162 RAEB-1, 123 RAEB-2, 52 CMML-dysplastic (CMML-D), 71 CMML-proliferative (CMML-P), 16 other hybrid myelodysplastic/myeloproliferative disorders (MDS/MPD, including four with RARS-T), six unclassified MDS, and 34 AML. Del-5q syndrome (p = 0.006) and RARS (p = 0.039) were more frequent in women, whereas CMML-D was more common in men (p = 0.002, Table 2).
Table 2.
Distribution according to FAB and WHO classification, IPSS, and WPSS
| Group | Νo. | Percent | Men | Percent | Women | Percent | p value |
|---|---|---|---|---|---|---|---|
| FAB classification | |||||||
| RA | 313 | 36.6 | 214 | 35.3 | 99 | 39.8 | 0.223 |
| RARS | 82 | 9.6 | 51 | 8.4 | 31 | 12.4 | 0.073 |
| RAEB | 287 | 33.6 | 212 | 35.0 | 75 | 30.1 | 0.170 |
| RAEB-T | 34 | 4.0 | 23 | 3.8 | 11 | 4.4 | 0.277 |
| CMML | 123 | 14.5 | 93 | 15.3 | 30 | 12.0 | 0.213 |
| Unclassified | 16 | 1.9 | 13 | 2.1 | 3 | 1.2 | 0.447 |
| WHO-2001 classification | |||||||
| RA | 176 | 20.66 | 121 | 20.0 | 55 | 22.1 | 0.491 |
| Del (5q) | 16 | 1.9 | 6 | 1.0 | 10 | 4.0 | 0.006 |
| RCMD | 122 | 14.3 | 87 | 14.4 | 35 | 14.1 | 0.900 |
| RARS | 53 | 6.2 | 31 | 5.1 | 22 | 8.8 | 0.039 |
| RCMD-RS | 24 | 2.8 | 16 | 2.6 | 8 | 3.2 | 0.649 |
| RAEB-1 | 162 | 18.9 | 121 | 20.0 | 41 | 16.5 | 0.234 |
| RAEB-2 | 123 | 14.4 | 91 | 15.0 | 32 | 12.9 | 0.415 |
| AML | 34 | 4.0 | 24 | 4.0 | 10 | 4.0 | 0.277 |
| CMML-D | 52 | 6.1 | 47 | 7.8 | 5 | 2.0 | 0.002 |
| CMML-P | 71 | 8.3 | 46 | 7.6 | 25 | 10.0 | 0.241 |
| MDS/MPD (incl. RARS-T) | 16 | 1.9 | 12 | 2.0 | 4 | 1.6 | 0.243 |
| Unclassified/unknown | 6 | 0.5 | 4 | 0.7 | 2 | 0.8 | 0.741 |
| Total | 855 | 100 | 606 | 100 | 249 | 100 | – |
| IPSS risk group categorization | |||||||
| Low | 236 | 39.9 | 172 | 39.2 | 64 | 42.1 | 0.557 |
| Intermediate-1 | 193 | 32.6 | 152 | 34.6 | 41 | 27.0 | 0.128 |
| Intermediate-2 | 105 | 17.8 | 76 | 17.3 | 29 | 19.1 | 0.622 |
| High | 57 | 9.6 | 39 | 8.9 | 18 | 11.8 | 0.293 |
| Subtotal | 591 | 100 | 439 | 100 | 152 | 100.0 | – |
| Unknown IPSS | 264 | – | 167 | – | 97 | – | n.s. |
| WPSS risk group categorization | |||||||
| Very low | 138 | 29.4 | 92 | 26.8 | 46 | 36.2 | 0.047 |
| Low | 87 | 18.5 | 61 | 17.8 | 26 | 20.5 | 0.504 |
| Intermediate | 75 | 16.0 | 66 | 19.2 | 9 | 7.1 | 0.001 |
| High | 127 | 27.0 | 94 | 27.4 | 33 | 26.0 | 0.761 |
| Very high | 43 | 9.1 | 30 | 8.8 | 13 | 10.2 | 0.620 |
| Subtotal | 470 | 100 | 343 | 100 | 127 | 100.0 | – |
| WPSS unknown | 207 | – | 132 | – | 75 | – | n.s. |
| WPSS not applicable | 178 | – | 131 | – | 47 | – | n.s. |
| Total | 855 | – | 606 | – | 249 | – | – |
No significant difference between men and women, according to FAB and to IPSS was observed. Del-5q syndrome and RARS were more frequent in women (4 % vs. 1 %, p = 0.006, and 8.8 % vs. 5.1 %, p = 0.039, respectively), whereas CMML-D was more frequent in men (7.8 % vs. 2 %, p = 0.002). WPSS very low subgroup was more frequent in women (36.2 % vs. 26.8 %, p = 0.047), whereas intermediate subgroup was more frequent in men (19.2 % vs. 7.1 %, p = 0.001).
Cytogenetic abnormalities
Cytogenetic analysis was performed in 611 patients (71 %), but in 20 (3.3 %) it could not be assessed due to absence of metaphases. Therefore, karyotype was available in 591 patients and it was normal in 364 (61.6 %) and abnormal in 227 (38.4 %). The most commonly detected cytogenetic abnormalities were: trisomy 8 (8.3 %), loss of Υ (5.8 %), and multiple (≥3) or complex chromosomal abnormalities (7.6 %). Del(5q) was found in 2.7 % and chromosome 7 abnormalities in 3 % of the patients. Loss of chromosome Y was considered a clonal abnormality, when >50 % of a minimum of 20 metaphases analyzed were demonstrating −Y. The kind of the detected chromosomal abnormalities among the total population tested, as well as among patients with a detected abnormal karyotype, is demonstrated in Table 3.
Table 3.
Cytogenetics in 591 patients with MDS
| Patient population: | All patients | CMML and RAEBT excluded | ||||
|---|---|---|---|---|---|---|
| Karyotype | N | % | N | % | ||
| Normal | 364 | 61.6 | (−) | 294 | 60.7 | (−) |
| Trisomy 8 | 49 | 8.3 | (21.6) | 35 | 7.2 | (18.4) |
| Loss of Y | 34 | 5.8 | (15.0) | 28 | 5.8 | (14.7) |
| Del(5q) | 16 | 2.7 | (7.0) | 16 | 3.3 | (8.4) |
| Del(20q) | 13 | 2.2 | (5.7) | 12 | 2.5 | (6.3) |
| Chromosome 7 aberrations | 18 | 3.0 | (7.9) | 17 | 3.5 | (8.9) |
| Trisomy 21 | 6 | 1.0 | (2.6) | 3 | 0.6 | (1.6) |
| Other single aberrations | 19 | 3.2 | (8.4) | 19 | 3.9 | (10.0) |
| Del(5q) plus add. abnormalities | 7 | 1.2 | (3.1) | 7 | 1.4 | (3.7) |
| Double abnormalities | 20 | 3.4 | (8.8) | 17 | 3.5 | (8.9) |
| Multiple/complex aberrations | 45 | 7.6 | (19.8) | 36 | 7.4 | (18.9) |
| Total | 591 | 100.0 | (100.0) | 484 | 100.0 | (100.0) |
Italic figures demonstrate percentages among patients with an abnormal karyotype
Prognostic classification according to IPSS and WPSS
IPSS could be assessed in 591 patients (69 %) and it was low in 236, intermediate-1 in 193, intermediate-2 in 105, and high in 57 patients. No difference in the distribution of men and women was found [low—(i.e., low/Int-1) vs. high-risk (i.e., Int-2/high)], p = 0.269].
WPSS could be assessed in 470 patients. In 207 patients cytogenetic results were unavailable, and therefore the WPSS could not be calculated, whereas in additional 178, WPSS was not applicable, either for a disease entity not assigned to the WHO classification (177), or for missing transfusion requirements (1). Patient distribution according to IPSS and WPSS is shown in Table 2.
Leukemic evolution
After a mean duration of dysplastic phase of 26 months (range 0.1–204 months), disease progressed to a more aggressive MDS or AML in 275 patients (32 %), whereas no progression was observed in 503 (59 %). In 77 patients (9 %), progression status was unknown. These patients were equally distributed by gender (p = 0.494) and by residence area (p = 0.287), therefore the unknown cases could be excluded, and the rate of disease evolution was again similar between the two genders (p = 0.174) or by residence area (p = 0.988).
Disease progression paralleled to the IPSS and WPSS risk group categorization (Table 5) and was lower in patients <70 years (p < 0.0001). Moreover, patients with RA and RARS had lower progression rate than those with RCMD and RCMD/RS (p < 0.0001) and patients with RAEB-1 had also lower progression rate, than those with RAEB-2 (p = 0.004). Disease progression was not significantly different between CMML-D and CMML-P (p = 0.405). The rates of disease progression are presented in Table 4.
Table 5.
Comparison between evolution rates to AML by WHO subtype and by age
| WHO subtype | Total N | Evolved (N) | Percent | Not evolved (N) | Percent | Unknown (N) | Percent | p value |
|---|---|---|---|---|---|---|---|---|
| RA/RARS | 229 | 18 | 7.9 | 189 | 82.5 | 22 | 9.6 | <0.0001 |
| RCMD/RCMD-RS | 162 | 41 | 25.3 | 106 | 65.4 | 15 | 9.2 | |
| RAEB-1 | 162 | 70 | 43.2 | 80 | 49.4 | 12 | 7.4 | 0.004 |
| RAEB-2 | 123 | 65 | 52.8 | 41 | 33.3 | 17 | 13.8 | |
| CMML-D | 52 | 19 | 36.5 | 31 | 59.6 | 2 | 3.8 | 0.405 |
| CMML-P | 71 | 29 | 40.8 | 35 | 49.3 | 7 | 9.6 | |
| Other subtypes | 56 | 33 | 58.9 | 21 | 37.5 | 2 | 3.6 | – |
| Age <70 years | 241 | 105 | 43.6 | 123 | 51.0 | 13 | 5.4 | <0.0001 |
| Age ≥70 years | 614 | 170 | 27.7 | 380 | 61.9 | 64 | 10.4 |
Table 4.
Disease progression and evolution by MDS subtype according to FAB and WHO classification, and by IPSS and WPSS prognostic category
| FAB classification | Patients evolved | Percent |
|---|---|---|
| RA | 39/282 | 13.8 |
| RARS | 20/76 | 26.3 |
| RAEB | 136/257 | 52.9 |
| RAEB-T | 27/33 | 81.8 |
| CMML | 48/114 | 42.1 |
| Unclassified | 5/16 | 31.3 |
| Total | 275/778 | 35.3 |
| Unknown | 77 | |
| WHO classification | ||
| RA | 13/158 | 8.2 |
| Del (5q) | 3/16 | 18.8 |
| RCMD | 23/110 | 20.9 |
| RARS | 5/49 | 10.2 |
| RCMD-RS | 15/22 | 68.2 |
| RAEB-1 | 70/150 | 46.7 |
| RAEB-2 | 65/106 | 61.3 |
| CMML-D | 19/50 | 38.0 |
| CMML-P | 29/64 | 45.3 |
| MDS/MPD | 8/16 | 50.0 |
| Unclassified | 1/6 | 16.7 |
| IPSS (N = 575) | ||
| Low | 38/228 | 16.7 |
| Int-1 | 76/189 | 40.2 |
| Int-2 | 65/103 | 63.1 |
| High | 33/55 | 60.0 |
| WPSS (Ν = 444) | ||
| Very low | 11/131 | 8.4 |
| Low | 18/81 | 22.2 |
| Intermediate | 33/77 | 42.9 |
| High | 69/114 | 60.5 |
| Very high | 18/41 | 43.9 |
Incidence and prevalence rates
Incidence and prevalence rates were estimated for the population of Achaia, Ilia, and Etolia and Akarnania separately, and for the whole adult population of Western Greece (603,543 inhabitants). During the 20-year study period (1990–2009) totally, 721 MDS patients were identified and registered. Among them, 386 (53.5 %) were residents of rural, and 315 (43.7 %) of urban areas, whereas for 20 patients (2.8 %) the residential area was unknown.
On 31.12.2009, 147 patients with MDS were alive, corresponding to a point prevalence of 24 cases per 100,000 inhabitants (0.024 %). The crude mean annual incidence of MDS in the adult population of Western Greece during the period 1990–2009 was 6.0 (95 % CI 5.6–6.4), while the age-standardized incidence rate was 5.7 (95 % CI 5.3–6.1). However, when CMML and RAEB-T were excluded (not considered MDS according to WHO classification), the crude mean annual incidence rate was estimated 4.8 (95 % CI 4.8–5.2) per 100,000 (Table 5).
The age-specific mean annual incidence rate of MDS increased with age, starting from 0.1/100,000 in the age group 15–39 years, 0.6 (40–49 years), 2.1 (50–59 years), 10 (60–69 years), 28.4 (70–79 years), and 34.7 in the age group >80 years (Table 6). There was a significant variation in the crude incidence rates among the different areas, which ranged from 2.8/100,000 inhabitants in the urban areas of Etolia and Akarnania to 8.8/100,000 inhabitants in the rural areas of Achaia. Moreover, there was an increased incidence of MDS in Achaia in comparison to other prefectures (Table 7).
Table 6.
Age-specific incidence rates of MDS in Western Greece, 1990–2009, by gender and by residence in a rural or urban area
| Men and women | Men | Women | Rural areas | Urban areas | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Age group (years) | N | Rate | N | Rate | N | Rate | N | Rate | N | Rate |
| 15–39 | 4 | 0.1 | 2 | 0.1 | 2 | 0.1 | 1 | 0.1 | 3 | 0.1 |
| 40–49 | 11 | 0.6 | 9 | 0.9 | 2 | 0.2 | 7 | 1.0 | 4 | 0.3 |
| 50–59 | 33 | 2.1 | 22 | 2.8 | 11 | 1.4 | 16 | 2.5 | 17 | 1.9 |
| 60–69 | 164 | 10.0 | 106 | 13.7 | 58 | 6.3 | 79 | 9.8 | 83 | 9.9 |
| 70–79 | 342 | 28.4 | 251 | 46.0 | 91 | 13.8 | 192 | 30.4 | 140 | 24.6 |
| ≥80 | 167 | 34.7 | 116 | 56.2 | 51 | 18.5 | 91 | 33.3 | 68 | 32.7 |
| Total (crude) | 721 | 6.0 | 506 | 8.3 | 215 | 3.6 | 386 | 7.7 | 315 | 4.5 |
| Total crude (CMML and RAEB-T excluded) | 585 | 4.8 | 405 | 6.7 | 180 | 3.0 | 309 | 6.1 | 260 | 3.7 |
Table 7.
Crude and age-standardized incidence rates of MDS in the population of people aged 15 years and older in Western Greece during the period 1990–2009 by prefecture, by rural, or urban area and by gender, comparative morbidity figures (CMF) and 95 % confidence intervals
| Subgroup | Crude (95 % CI) | Age-standardized (95 % CI) | ||
|---|---|---|---|---|
| Prefecture | ||||
| Achaia | Rural areas | 8.8 (7.3–10.3) | 7.2 (5.9–8.5) | |
| Urban areas | 5.6 (4.8–6.4) | 6.8 (6.0–7.6) | ||
| Overall | 6.9 (6.2–7.6) | 7.3 (6.6–8.0) | ||
| Ilia | Rural areas | 7.2 (5.9–8.5) | 5.1 (4.1–6.1) | |
| Urban areas | 3.7 (2.7–4.7) | 4.1 (3.1–5.1) | ||
| Overall | 5.7 (4.9–6.5) | 5.0 (4.2–5.8) | ||
| Etolia and Akarnania | Rural areas | 7.0 (5.8–8.3) | 5.2 (4.2–6.2) | |
| Urban areas | 2.8 (2.0–3.6) | 3.0 (2.2–3.8) | ||
| Overall | 4.8 (4.1–5.5) | 4.3 (3.6–5.0) | ||
| Comparison between subgroups | CMF (95 % CI) | |||
| All prefectures | Rural areas | 7.7 (6.9–8.5) | 5.9 (5.1–6.7) | 1.13 (0.96–1.33) |
| Urban areas | 4.5 (4.0–5.0) | 5.2 (4.7–5.7) | ||
| Gender | Men | 8.3 (7.6–9.0) | 7.9 (7.1–8.7) | 2.32 (1.96–2.75) |
| Women | 3.6 (3.1–4.1) | 3.4 (2.9–3.9) | ||
| All subgroups | Total | 6.0 (5.6–6.4) | 5.7 (5.3–6.1) | |
| Sex-standardized incidence rate (95 % CI) | 5.9 (5.6–6.3) | |||
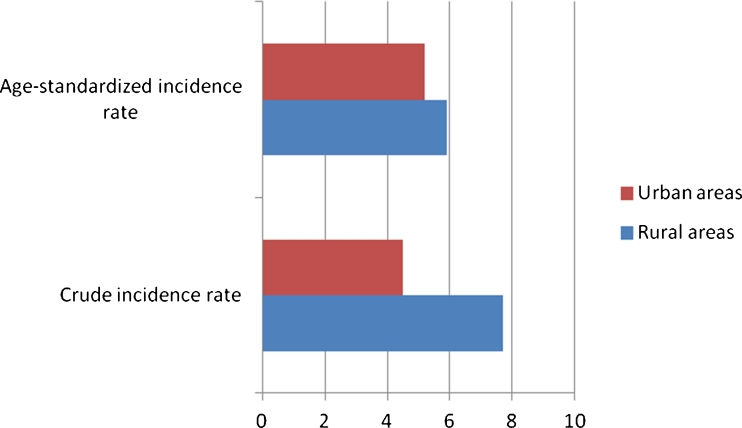
Given that the rural population is more senile than the urban, and that the incidence of MDS increases with age, we consequently performed a direct standardization method, to compare the incidence between rural and urban areas, by using as a standard population the whole adult population of Greece in 2001. Age-standardized incidence of MDS in rural areas was 5.9 (95 % CI 5.1–6.7) whereas, in urban areas was 5.2 (95 % CI 4.7–5.7). The CMF (ratio of incidence in rural/urban areas) was 1.13 (95 % CI 0.96–1.33). Therefore, we did not find a statistically significant difference in the incidence of MDS between rural and urban areas after age standardization. Crude incidence, as opposed to age-standardized incidence of MDS in rural and in urban areas, is depicted in Fig. 2. We also calculated age-standardized incidence rates of MDS for each prefecture, and the difference (incidence in Achaia, compared to that of the other prefectures) was even more remarkable. Age-standardized incidence of MDS in men was 7.9 (95 % CI 7.1–8.7) and in women 3.4 (95 % CI 2.9–3.9). The CMF (ratio of incidence in men/women) was 2.32 (95 % CI 1.96–2.75). Sex-standardized incidence of MDS was 5.9 (95 % CI 5.5–6.3) per 100,000 inhabitants (Table 7).
Fig. 2.
Comparison between crude and age-standardized incidence rate of MDS in rural and urban areas of SW Greece (1990–2009)
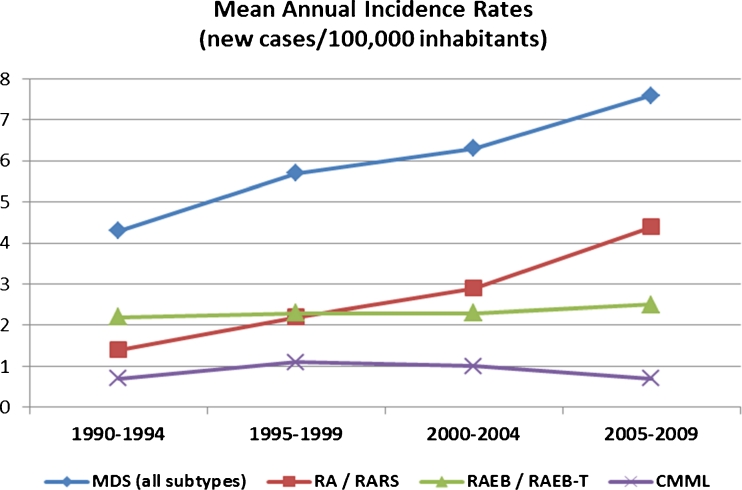
We finally estimated the temporal trends in the incidence in the same population throughout the study period, by dividing it in 5-year intervals. The number of new MDS cases gradually increased from 130 during the quinquennium 1990–1994, to 171 during 1995–1999, 190 during 2000–2004, and eventually 230 during 2005–2009. The crude mean annual incidence rate increased from 4.3 (95 % CI 3.6–5.0) per 100,000 inhabitants in the first period (1990–1994), to 5.7 (95 % CI 4.9–6.6) during 1995–1999, 6.3 (95 % CI 5.4–7.2) during 2000–2004, and finally 7.6 (95 % CI 6.6–8.6) in the last period (2005–2009). Thus, a statistically significant increase of the overall incidence rate of MDS was observed throughout the study period. To elucidate which MDS subtypes mainly increased or whether all MDS subtypes proportionally increased, we calculated the crude mean annual incidence rates among patients with lower-risk MDS (FAB/RA-RARS), higher-risk MDS (RAEB/RAEBT/unclassified) and CMML separately. We found that the observed increase in the annual incidence of MDS in our area was almost entirely attributed to the lower-risk MDS, whose incidence rose from 1.4/100,000 inhabitants in the first period (1990–1994) to 4.4/100,000 in the last quinquennium 2005–2009, demonstrating 214 % increase. The incidence of higher-risk MDS and of CMML was practically unchanged throughout the 20-year period. The temporal trend of mean annual incidence rate of the whole MDS population and of the higher- and lower-risk cases, as well as that of CMML, through 1990–2009, is depicted in Fig. 3.
Fig. 3.
Temporal trend of mean annual incidence rates of MDS in SW Greece through 1990–2009 in 5-year periods
Discussion
Epidemiological studies on MDS are rare worldwide, but particularly in Greece there are no data about the epidemiology of these diseases. This is the first study which attempts to describe demographic and clinical features of patients with MDS, and to estimate incidence and prevalence rates in a well-defined geographical area of Greece.
A clear predominance of MDS in men has already been reported. In two studies male to female ratio was estimated 1.9 [9], and 1.62 [10], but in our study it was even higher (2.43). In our patient population the mean age at diagnosis was not significantly different between men and women, either overall or by subtype. In addition, there was a remarkably high representation of farmers. According to occupational data for the Hellenic population of Western Greece during 2007–2009 (provided by the Hellenic Statistical Authority), persons occupied with agricultural activities constituted 21 %, i.e., smaller than half of what we found. Nevertheless, a safe comparison cannot be made, because this percentage refers to persons of productive age, whereas our patients were mostly elderly and declared being farmers even after their retirement. This difference may also depict an under-representation of occupations associated with higher socioeconomic status, since some of these patients might have been treated in private centers of Athens. This possibility however may be valid only for a small minority of our patients. Alternatively, it could suggest a true association between MDS and agricultural activity, but the investigation of this hypothesis exceeds the limits of descriptive epidemiology.
We found that only 2.6 % of the patients had secondary (therapy-related) MDS. This percentage is lower than what has been previously reported (5.3–12.5 %) [9, 11–13], although in a Spanish study no secondary MDS cases were identified [14]. The distribution according to FAB and WHO classification is consistent with the results of previous studies. All MDS subtypes, excluding Del-5q and RARS, were more frequent in men [15]. RARS was more frequent in women, in both classification systems, yet this difference was statistically significant only for the WHO classification. Conversely, CMML-D was significantly more frequent in men, as has been previously reported [16].
In our registry, karyotype is missing mostly in patients diagnosed in the early 1990s, and, generally, the later the diagnosis was made, the more cytogenetic studies are available, given the increased sensitization of experts about MDS. Loss of chromosome Y was highly represented, but surprisingly, an even higher representation of trisomy 8 was observed. Del(5q) was the third most common single abnormality, and it should be marked that seven elderly patients, in whom Del-5q plus additional evolutionary clones were detected at baseline, were referred when their clinical situation had worsened. In these patients there was a period of macrocytic anemia, not requiring transfusions for 1–6 years before their referral, and therefore they might initially be true cases of Del-5q syndrome, diagnosed during disease progression. Distribution according to IPSS and WPSS as well as rate of leukemic transformation were in agreement with reports of previous studies [17] and were not significantly different in relation to gender, residence area, or geographical prefecture.
Incidence rates of MDS in Europe have been reported to vary between 2.1/100,000 inhabitants in the UK during 1984–1986 [18], 3.2 in France (1980–1990) [9], 3.5 in South Thames (1999–2000) [10], 3.6 in Sweden (1978–1982) [19], 4.1 (1975–1990) [11] and 4.9 (1996–2005) [13] in Germany (Düsseldorf), 7.7 in the Basque country (1993–1996) [12], 8.1 in Spain (1994–1998) [14], 9.3 in Somerset, UK [20], and 12.6 in Bournemouth, UK (1981–1990) [6]. A recent study from Romania found a very low incidence of 0.3/100,000 [21]. In the United States the incidence of MDS has been reported 3.4/100,000 (SEER, 2001–2003) [22] and 3.3/100,000 (SEER and NAACCR, 2001–2003) [23]. However, recent studies suggest that many MDS cases are undetected by cancer registries and constructed algorithms, and that the true incidence of MDS might be higher [24]. Accordingly, the incidence of MDS in a non-profit healthcare system in western Washington State rose from 6.9 to 10.2 when possible MDS cases were identified from chart review [25]. Finally, a study in Japan reported a very low incidence rate of 1/100,000 inhabitants [26]. The comparison of the various incidence rates is difficult, because of great differences in age (excluding pediatric MDS cases leads to higher incidence rate), in classification (American studies have adopted ICD classification of MDS), and in time (starting 30 years ago but not referring to the same period, Table 8).
Table 8.
Comparison between incidence rates of MDS from several epidemiologic studies
| Authors | Country (area) | Population (inhabitants) | Age of study population | Period | Type of MDS included | Incidence rate (95 % CI) per 100,000/year |
|---|---|---|---|---|---|---|
| Radlund et al. [19] | Sweden (Jönköping) | 224,000 | >19 years old | 1978–1992 | FAB classification | 3.2 (1978–1982)] |
| 4.1 (1983–1987)] 3.6 mean | ||||||
| 3.5 (1988–1992)] | ||||||
| Maynadié et al. [9] | France (Côte d’ Or, Burgundy) | 493,931 | All ages | 1980–1990 | FAB classification | 3.2 (crude) |
| 1.7 (age adjusted) | ||||||
| Williamson et al. [6] | UK (Bournemouth) | 214,500 | 1981–1990 | FAB classification | 12.6 (crude) | |
| Cartwright RA-Leukemia Research Fund Centre [18] | UK (East Dorset) | 11,000,000 | 1984–1986 | 2.1 (age adjusted) | ||
| Aul C et al. [10] | Germany (Düsseldorf) | 575,000 | 1975–1990 | FAB classification | 1.4–4.1 (crude) | |
| Germing U et al. [29] | Germany (Düsseldorf) | 575,000 | 1991–2001 | FAB classification | 4.9 (crude) | |
| Neukirchen J et al. [13] | Germany | 575,000 | All ages | 2002–2005 | WHO classification | 3.4 (crude) |
| (Düsseldorf) | FAB classification | 4.15 (crude) | ||||
| Shimizu Η et al. [26] | Japan (nationwide) | 160,000,000 | Sept. 1991 | 1.0 (crude) | ||
| Bauduer et al. [12] | France (Pays Basque) | 290,000 | All ages | 1993–1996 | FAB classification | 7.7 |
| Phillips MJ et al. [20] | UK (Somerset) | 1985–1993 | 9.3 | |||
| Iglesias Gallego M et al. [14] | Spain (Ourense) | 346,913 | All ages | 1994–1998 | FAB classification | 8.1 |
| Phekoo J et al. [10] | UK (South Thames) | 5,499,841 | ≥16 years old | 1999–2000 | FAB classification | 3.5 |
| Ma X et al. [22] | USA (SEER-17 regions) | 75,000,000 | All ages | 2001–2003 | ICD-O3 classificationa | 3.4 (3.3–3.5) |
| Rollison DE et al. [23] | USA (NAACCR, SEER) | 759,270,956 | All ages | 2001–2004 | ICD-O3 classificationa | 3.3 |
| Gologan R [21] | Romania (Bucharest area) | 5,560,566 | >16 years old | 1985–2004 | FAB classification | 0.3 |
| Present study | Western Greece (Achaia, Ilia, Etolia and Akarnania) | 603,543 | ≥15 years old | 1990–2009 | FAB classification excluding CMML/RAEB-T | 6.0 (5.6–6.4) (crude) |
| 5.7 (5.3–6.1) (age adjusted) | ||||||
| 4.8 (4.4–5.2) |
aCMML and RAEB-T excluded
The crude incidence rate of MDS was higher in rural than in urban areas, but this difference did not persist after age standardization. In contrast, another study found higher incidence of MDS in men residing in urban areas [9].
We also observed an increased incidence of MDS in Achaia, compared to the other prefectures, and this difference became more prominent after age standardization. Possible explanations might be the easier access of patients residing in Achaia to the referral centers, leading to minimization of under-diagnosed cases, and the declaration of Patras, capital of Achaia, and of the whole region of Western Greece, residential area of patients’ relatives, as residential area of patients living in distantly located, particularly rural areas. This might have led to underestimation of the incidence in the other prefectures of the region, as this has been observed in the urban areas of Etolia/Akarnania. Other possible explanations might be the restricted access of distantly living inhabitants to specialized centers, and the empirical treatment of less severe cases by family physicians, thus evading correct diagnosis, and the movement of some patients to referral centers outside Western Greece, particularly in Athens, where their relatives live and work.
Increasing incidence of MDS has been reported by McNally et al. [27] and by Aul et al. [11, 28] during the first period of the Düsseldorf study, but incidence rate reached a plateau in 1986, without further increase during the second period (1991–2001) [29]. Neukirchen et al. also calculated the incidence of MDS in Düsseldorf during the period 1996–2005 and found no further increase in the period 2002–2005 [13]. In our study, a continuously increasing incidence of MDS has been observed throughout the 20-year period, but this entirely concerned the lower-risk categories. This could be attributed to a better quality of health care, provided to the elderly population, and consequently to the prolongation of life expectancy. Another most possible explanation might be the higher degree of suspicion and awareness of the primary medical care providers, probably also as a result of the broader use of automatic blood analyzers, and the increasing referral of early-MDS cases, for evaluation to the specialized Hematological Departments, cases which were not usually referred in the past. Alternatively, it could reflect a true increase, related to environmental factors. Therefore, further epidemiological research with case–control studies is required, in order to investigate the possible contribution of environmental, occupational, and lifestyle factors to the continuous increase of MDS, particularly of the lower-risk categories, in our area.
Acknowledgments
Conflict of interest
The authors declare that they have no conflict of interest.
References
- 1.Bennett JM. The myelodysplastic syndromes. Pathobiology and clinical management. New York: Marcel Dekker Inc; 2002. p. 15. [Google Scholar]
- 2.Lipschitz DA, Udupa KB, Milton KY, Thompson CO. Effect of age on hematopoiesis in man. Blood. 1984;63:502–509. [PubMed] [Google Scholar]
- 3.Germing U, Neukirchen J, Haas R. The epidemiology of myelodysplastic syndromes. Clin Leukemia. 2008;2(Feb):34–38. doi: 10.3816/CLK.2008.n.004. [DOI] [Google Scholar]
- 4.Strom SS, Velez-Bravo V, Estey EH. Epidemiology of myelodysplastic syndromes. Semin Hematol. 2008;45:8–13. doi: 10.1053/j.seminhematol.2007.10.003. [DOI] [PubMed] [Google Scholar]
- 5.Shimizu H, Matsushita Y, Aoki K, Nomura T, Yoshida Y, Mizoguchi H. Prevalence of the myelodysplastic syndromes in Japan. Int J Hematol. 1995;61:17–22. doi: 10.1016/0925-5710(94)00339-G. [DOI] [PubMed] [Google Scholar]
- 6.Williamson PJ, Kruger AR, Reynolds PJ, Hamblin TJ, Oscier DG. Establishing the incidence of myelodysplastic syndrome. Br J Haematol. 1994;87:743–745. doi: 10.1111/j.1365-2141.1994.tb06733.x. [DOI] [PubMed] [Google Scholar]
- 7.Breslow NE, Day NE (1987) Rates and rate standardisation. In: Heseltine E, technical editor for IARC. Statistical methods in cancer research. Volume II—the design and analysis of cohort studies. Lyon: IARC Scientific Publications 48–79 [PubMed]
- 8.Bouyer J, Hemon D, Cordier S, Derrienic F, Stucker I, Stengel B, Clavel J (1993) Standardisation des mesures de risque et d’association. In: Epidémiologie. Principes et méthodes quantitatives. Paris: Les editions INSERM 209–226
- 9.Maynadié M, Verret C, Moskovtchenko P, Mugneret F, Petrell T, Caillot D, Carli PM. Epidemiological characteristics of myelodysplastic syndromes in a well-defined French population. Br J Cancer. 1996;74:288–290. doi: 10.1038/bjc.1996.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Phekoo KJ, Richards MA, Moller H, Schey SA. The incidence and outcome of myeloid malignancies in 2,112 adult patients in South East England. Haematologica. 2006;91:1400–1404. [PubMed] [Google Scholar]
- 11.Aul C, Gattermann N, Schneider W. Age-related incidence and other epidemiological aspects of myelodysplastic syndromes. Br J Haematol. 1992;82:358–367. doi: 10.1111/j.1365-2141.1992.tb06430.x. [DOI] [PubMed] [Google Scholar]
- 12.Bauduer F, Ducout L, Dastugue N, Capdupuy C, Renoux M. Epidemiology of myelodysplastic syndromes in a French general hospital of the Basque country. Leuk Res. 1998;22:205–208. doi: 10.1016/S0145-2126(97)00159-8. [DOI] [PubMed] [Google Scholar]
- 13.Neukirchen J, Schoonen WM, Strupp C, Gattermann N, Aul C, Haas R, Germing U. Incidence and prevalence of myelodysplastic syndromes: data from the Düsseldorf MDS registry. Leuk Res. 2011;35:1591–1596. doi: 10.1016/j.leukres.2011.06.001. [DOI] [PubMed] [Google Scholar]
- 14.Iglesias Gallego M, Sastre Moral JL, Gayoso Diz P, Garcia Costa A, Ros Forteza S, Mayan Santos JM. Incidence and characteristics of myelodysplastic syndromes in Ourense (Spain) between 1994–1998. Haematologica/Journal of Hematology. 2003;88:1197–1199. [PubMed] [Google Scholar]
- 15.Van den Berghe H, Vermaelen K, Merucci C, Barbieri D, Tricot G. The 5q-anomaly. Cancer Genet Cytogenet. 1985;17:189–225. doi: 10.1016/0165-4608(85)90016-0. [DOI] [PubMed] [Google Scholar]
- 16.Ribera JM, Cervantes F, Rozman C. A multivariate analysis of prognostic factors in chronic myelomonocytic leukemia according to the FAB criteria. Br J Haematol. 1987;65:307–311. doi: 10.1111/j.1365-2141.1987.tb06858.x. [DOI] [PubMed] [Google Scholar]
- 17.Greenberg P, Cox C, LeBeau MM, et al. International scoring system for evaluating prognosis in myelodysplastic syndromes. Blood. 1997;89:2079–2088. [PubMed] [Google Scholar]
- 18.Cartwright RA. Incidence and epidemiology of the myelodysplastic syndromes. In: Mufti GJ, Galton DAG, editors. The myelodysplastic syndromes. Edinburgh: Churchill Livingstone; 1992. pp. 23–31. [Google Scholar]
- 19.Radlund A, Thiede T, Hansen S, Carlsson M, Engquist L. Incidence of myelodysplastic syndromes in a Swedish population. Eur J Haematol. 1995;54:153–156. doi: 10.1111/j.1600-0609.1995.tb00206.x. [DOI] [PubMed] [Google Scholar]
- 20.Phillips MJ, Cull GM, Ewings M. Establishing the incidence of myelodysplasia syndrome. Br J Haematol. 1994;88:896–897. doi: 10.1111/j.1365-2141.1994.tb05138.x. [DOI] [PubMed] [Google Scholar]
- 21.Gologan R. Demo-geographical data of myelodysplastic syndrome based on a large sample of patients from a Romanian Hematological Center. J BUON. 2010;15:547–555. [PubMed] [Google Scholar]
- 22.Ma X, Does M, Raza A, Mayne ST. Myelodysplastic syndromes: incidence and survival in the United States. Cancer. 2007;109:1536–1542. doi: 10.1002/cncr.22570. [DOI] [PubMed] [Google Scholar]
- 23.Rollison DE, Howlader N, Smith MT, Strom SS, Merrit WD, Ries LA, Edward BK, List AF. Epidemiology of myelodysplastic syndromes and chronic myeloproliferative disorders in the United States, 2001–2004, using data from the NAACCR and SEER programs. Blood. 2008;112:45–52. doi: 10.1182/blood-2008-01-134858. [DOI] [PubMed] [Google Scholar]
- 24.Cogle CR, Craig BM, Rollison DE, List AF. Incidence of the myelodysplastic syndromes using a novel claims-based algorithm: high number of uncaptured cases by cancer registries. Blood. 2011;117:7121–7125. doi: 10.1182/blood-2011-02-337964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.De Roos AJ, Deeg HJ, Onstad L, Kopecky KJ, Aiello EJ, Yong M, Fryzek J, Davis S. Incidence of myelodysplastic syndromes within a nonprofit healthcare system in western Washington state 2005–2006. Am J Hematol. 2010;85:765–770. doi: 10.1002/ajh.21828. [DOI] [PubMed] [Google Scholar]
- 26.Shimizu H, Matsushita Y, Aoki K, Nomura T, Yoshida Y, Mizoguchi H. Prevalence of the myelodysplastic syndromes in Japan. Int J Hematol. 1995;61:17–22. doi: 10.1016/0925-5710(94)00339-G. [DOI] [PubMed] [Google Scholar]
- 27.McNally RJQ, Roman E, Cartwright RA. Leukemias and lymphomas: time trends in the UK, 1984–93. Cancer Causes Control. 1999;10:35–42. doi: 10.1023/A:1008859730818. [DOI] [PubMed] [Google Scholar]
- 28.Aul C, Giagounidis A, Germing U. Epidemiological features of myelodysplastic syndromes: results from regional cancer surveys and hospital-based statistics. Int J Hematol. 2001;73:405–410. doi: 10.1007/BF02994001. [DOI] [PubMed] [Google Scholar]
- 29.Germing U, Strupp C, Kundgen A, Bowen D, Aul C, Haas R, Gattermann N. No increase in age-specific incidence of myelodysplastic syndromes. Haematologica. 2004;89:905–910. [PubMed] [Google Scholar]