Abstract
A new concept of immunogenic cell death (ICD) has recently been proposed. The immunogenic characteristics of this cell death mode are mediated mainly by molecules called ‘damage-associated molecular patterns' (DAMPs), most of which are recognized by pattern recognition receptors. Some DAMPs are actively emitted by cells undergoing ICD (e.g. calreticulin (CRT) and adenosine triphosphate (ATP)), whereas others are emitted passively (e.g. high-mobility group box 1 protein (HMGB1)). Recent studies have demonstrated that these DAMPs play a beneficial role in anti-cancer therapy by interacting with the immune system. The molecular pathways involved in translocation of CRT to the cell surface and secretion of ATP from tumor cells undergoing ICD are being elucidated. However, it has also been shown that the same DAMPs could contribute to progression of cancer and promote resistance to anticancer treatments. In this review, we will critically evaluate the beneficial and detrimental roles of DAMPs in cancer therapy, focusing mainly on CRT, ATP and HMGB1.
Keywords: calreticulin, ATP, HMGB1, immunogenic cell death, TLRs
Facts
Immunogenic cell death (ICD) is characterized by secretion, release or surface exposure of damage-associated molecular patterns (DAMPs).
Surface-exposed calreticulin (CRT), actively secreted adenosine triphosphate (ATP) and passively released high-mobility group box 1 protein (HMGB1), the key DAMPs of ICD, play a beneficial role in anti-cancer therapy by activating the immune system.
Depending on the cell death inducer, a specific signaling pathway is activated, leading to CRT surface exposure and ATP secretion.
Of note, that different conditions of ATP (e.g. different extracellular concentrations) and HMGB1 (e.g. different redox states) could have different biological effects contributing to tumor metastasis and tumor progression.
Open Questions
Which redox state of HMGB1 is responsible for the pro- and anti-tumorigenic activities?
What determines the difference between DAMPs that drive cancer progression and those that inhibit tumor growth?
Large-scale clinical studies are necessary to determine the prognostic value of ICD associated with DAMP emission in different types of cancers.
More than 20 years ago the ‘danger theory' was proposed. This theory explains why potent immune responses are elicited against tissue transplants, ischemia-related injuries, tumors and autoimmune diseases, none of which involve microbial components.1 It states that the immune system can distinguish between dangerous and innocuous endogenous signals. In response to trauma, ischemia and other types of tissue and cell damage, and death, certain molecules that perform predominantly non-immunological functions inside the cell are released or secreted from damaged or dead/dying cells or exposed on their surface, thereby initiating an immune response in the absence of infection. These immunostimulatory molecules were later named damage-associated molecular patterns (DAMPs). They can be derived from any compartment of the cell (Figure 1).2, 3 The modification of DAMPs by proteolysis and oxidation, which often accompanies cell death, modulates their immunostimulatory potential.4, 5 Most of the DAMPs that resemble pathogen-associated molecular patterns (PAMPs) stimulate the immune system through a broad family of membrane-bound or cytoplasmic pattern-recognition receptors, which include Toll-like receptors (TLRs), NOD-like receptors and RIG-I-like receptors.2, 3, 6, 7, 8
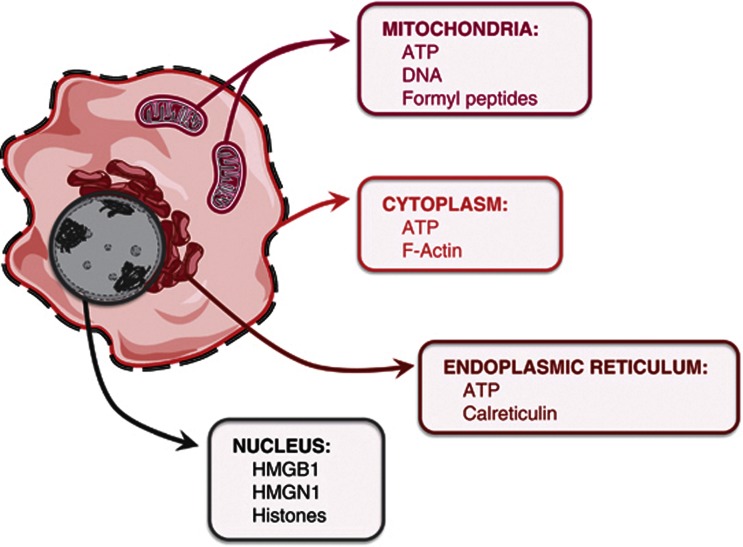
Figure 1.
DAMPs are derived from different compartments of the cells. For example, they can come from mitochondria (DNA, formyl peptides and ATP), nucleus (HMGB1, high-mobility group box 1 protein; HMGN1, high mobility group nucleosome binding protein 1; histones), ER (calreticulin and ATP) and cytoplasm (ATP and F-actin)2, 74, 78
In the past several years it has been shown that DAMPs can be emitted actively (calreticulin (CRT) and adenosine triphosphate (ATP)) or passively (high-mobility group box 1 protein (HMGB1)) from tumor cells undergoing immunogenic apoptosis, that is, immunogenic cell death (ICD). It has been shown that ICD induction is stressor-dependent9 and requires reactive oxygen species (ROS)-based endoplasmic reticulum (ER) stress.10, 11 Therefore ICD inducers are classified as Type I (e.g. anthracyclines, mitoxantrone, oxaliplatin, cyclophosphamide, UVC and γ-radiation) or Type II (e.g. hypericin-based photodynamic therapy (PDT)).12 Type I ICD inducers trigger ICD through targets that are not associated with the ER but stimulate ICD-associated immunogenicity through secondary or ‘collateral' ER stress effects. In contrast, Type II ICD inducers selectively target the ER to induce cell death and ICD-associated immunogenicity in an ER-focused manner. It has been shown that DAMPs have a beneficial role in experimental models of anti-cancer therapy because of their interaction with the immune system. Tumor cells killed in vitro and implanted subcutaneously into syngenic immunocompetent mice can mediate an ‘anti-cancer vaccine effect' in the absence of any adjuvants or immunostimulatory substances.11, 13, 14 In this experimental model, considerable proportions of mice inoculated in this way are protected against subsequent re-challenge with live cancer cells and develop fewer tumors at the re-challenge site. It has been shown that surface exposure of CRT, secretion of ATP and passive release of HMGB1 are required for the activation of potent anti-cancer immunity.11, 13, 14 However, in the spontaneous mammary murine tumor model it has been shown that the adaptive immune system is not needed for the therapeutic efficacy of immunogenic chemotherapeutics such as oxaliplatin and doxorubicin.15 Also, recent studies have shown that some of these DAMPs (e.g. HMGB1) contribute to the progression of cancer16 and promote resistance to anti-cancer treatments.17, 18, 19, 20, 21 In this review, we will discuss the beneficial and detrimental roles of DAMPs in cancer therapy, including the mechanisms of emission of three vital DAMPs, namely, surface-exposed CRT, secreted ATP and released HMGB1.
Role of CRT Surface Exposure During ICD in Cancer Treatment
CRT is a soluble protein in the lumen of the ER, traditionally regarded as a Ca2+ homeostasis regulator and an ER chaperone.22 It is involved in several functions inside and outside the ER, such as regulation of chaperone activity and Ca2+ homeostasis, assembly of the major histocompatibility complex class I molecules, and cellular proliferation and migration.3, 23 It has been shown that certain anti-cancer modalities induce ICD of tumor cells characterized by surface exposure of CRT before signs of apoptosis (i.e. exposure of PS).13 Surface-exposed CRT facilitates tumor antigen presentation and tumor-specific cytotoxic T-lymphocyte responses. It has been shown that the exposure of CRT on the cell surface of cancer cells in response to anthracyclines is dependent on the ER-stress response24 associated with ROS production.10, 25 Several CRT translocation pathways have been described. The first pathway described is induced by anthracyclines and is dependent on PERK-mediated eukaryotic initiation factor 2α-phosphorylation (eIF2α-P), the secretory pathway and caspase-8-mediated BCAP31 (BAP31)-dependent activation of BAX or BAK proteins.10 However, recently it has been shown that only PERK, BAX or BAK and the secretory pathway are required for the surface translocation of CRT in response to hypericin-photodynamic therapy (PDT).11 In this case, PERK governed the trafficking of ecto-CRT by regulating the proximal secretory pathway.11 eIF2α-phosphorylation and caspase-8 signaling were not required for CRT exposure. The pathway that is used for CRT translocation strongly depends on the apoptotic stage during which the exposure takes place.12 Indeed, either knockdown of CRT or vaccination of mice with cells deficient in any of the proteins required for CRT exposure reduced the immunogenicity of the cancer cells.11, 13 All these data indicate that cancer cells undergoing ICD induce protective immunity against the viable cancer cells in the prophylactic anti-cancer vaccination models in mice.
But what about the role of surface-exposed CRT in anti-cancer treatment in humans, where the situation could be different? It has been shown that anthracycline treatment of human prostate cancer, ovarian cancer and acute lymphoblastic leukemia cell lines (and also primary tumor cells) induces key features of ICD (such as CRT exposure and release of HMGB1) like those in mouse cancer cells.26 The authors showed that the cells undergoing ICD induced maturation of dentritic cells (DCs), which stimulated tumor-specific IFN-γ-producing T cells. Another study showed that CRT exposure correlates with the therapeutic benefit of vaccination with DCs pulsed with primary lymphoma cells undergoing ICD (triggered by heat shock, γ-irradiation and ultraviolet C-light) in indolent non-Hodgkin's lymphoma patients.27 However, leukemic cells isolated from some patients with acute myeloid leukemia (AML) exhibited CRT surface exposure regardless of chemotherapy, and this exposure was not modulated by in vivo chemotherapy, including anthracyclines.28 The authors also detected high levels of exposed CRT on malignant myeloblasts, and the level was positively correlated with the ability of autologous T cells to secrete IFN-γ in response to DCs, suggesting that the presence of CRT on the surface of leukemia cells facilitates cellular anti-cancer immune responses in AML patients. Apoptotic AML cells collected during in vivo therapy with all-trans retinoic acid showed increased exposure of CRT during spontaneous in vitro apoptosis, suggesting that in vivo pharmacotherapy can induce ICD.29 But the authors detected a wide variation of the level of CRT exposure between patients, which probably depends on individual patient characteristics.29 It has also been shown that serum CRT levels are significantly higher in lung cancer patients after chemotherapy compared to untreated patients.30 However, in this study correlation of CRT with efficacy of anti-cancer therapy was not examined. In conclusion, there is still no conclusive clinical data on the association between CRT surface exposure and favorable outcome of anti-cancer therapy. Large-scale studies are needed to determine the prognostic value of immunogenic cell death associated with surface translocation/secretion of CRT in patients suffering from different types of cancers.
Role of Extracellular Emission of ATP During ICD in Cancer Treatment
During the past few years, it has become clear that cells undergoing different types of cell death passively release or actively secrete ATP.2, 11, 31, 32, 33 Several mechanisms of ATP secretion in dying/dead/stressed cells have been reported.11, 34, 35, 36 Like the exposure of CRT, the mechanism for ATP secretion in ICD strongly depends on the apoptotic stage and the type of stress or cell death stimulus that induces ICD.12 In PDT-induced ICD, ATP was actively secreted from cells during the pre-apoptotic stage, and this secretion was dependent on the classical as well as the PERK-regulated secretory pathway and PI3K-dependent exocytosis, but it was independent of BAX/BAK.11
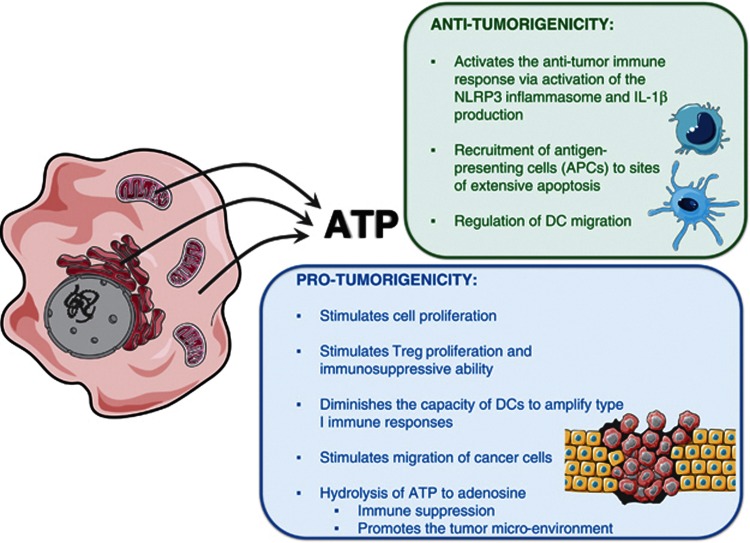
It has been shown that ATP released from dying tumor cells is required for the immune response to cancer cells.37 Cancer cells undergoing ICD in response to anthracycline secrete ATP in an autophagy-dependent manner.32, 35, 36 The authors showed that suppression of autophagy inhibited the early apoptotic secretion of ATP from dying cells killed by chemotherapeutics and compromised the tumor-specific immune response in vivo (Figure 2).32, 35, 36 Although all these data suggest that ATP functions as an efficient adjuvant in anti-cancer treatment, no published clinical trials have confirmed this yet.
Figure 2.
ATP is actively secreted by dying tumor cells in the early apoptotic stage and could be involved in pro- and anti-tumorigenic activities
It is important to note that extracellular ATP, besides acting as a DAMP that stimulates anti-tumor immune responses, can also function as a ‘find me' signal facilitating rapid attraction of antigen-presenting cells to sites of extensive apoptotic cell death.31 It has been reported that extracellular ATP is involved in the regulation of cell migration of DCs38, 39, 40 and also prostate cancer cells.41 Thus, by stimulating cell migration (e.g. cancer cells), extracellular ATP could also modulate cancer invasion and metastasis and thereby have a negative effect on anti-cancer treatment. This possibility has to be tested in experimental cancer models.
Extracellular ATP can also stimulate cell proliferation42 and it could be a source of immunosuppressive agents such as adenosine.12 Pro-inflammatory ATP can be hydrolyzed to immunosuppressive adenosine,43, 44 which could lead to immune suppression and development of a tumor-promoting microenvironment that reduces the efficacy of anti-tumor immune responses (Figure 2).44, 45, 46 Another interesting topic for future research is to understand the factors or conditions that determine the biological responses of cells to extracellular ATP. The concentration of extracellular ATP might be an important determinant of the final outcome: it has been shown that different concentrations of extracellular ATP could have different biological effects.47 For example, 250 nM extracellular ATP does not modulate Treg functions, whereas at 1 mM it ‘turns on' T regulatory cells (Tregs) and stimulates their proliferation and immunosuppressive ability in a contact-dependent manner.47 The stimulation of Tregs by ATP may also represent a mechanism by which tumors avoid the immune system. In addition, it has also been shown that under conditions that cause prolonged release of low amounts of ATP, this nucleotide has a more subtle modulatory function, inducing a distorted DC maturation and diminishing the ability of mature DCs to initiate type 1 immune responses.40, 48 All these add another angle of complexity to the role of ATP in immunogenicity.
Role of Extracellular HMGB1 in Cancer Treatment
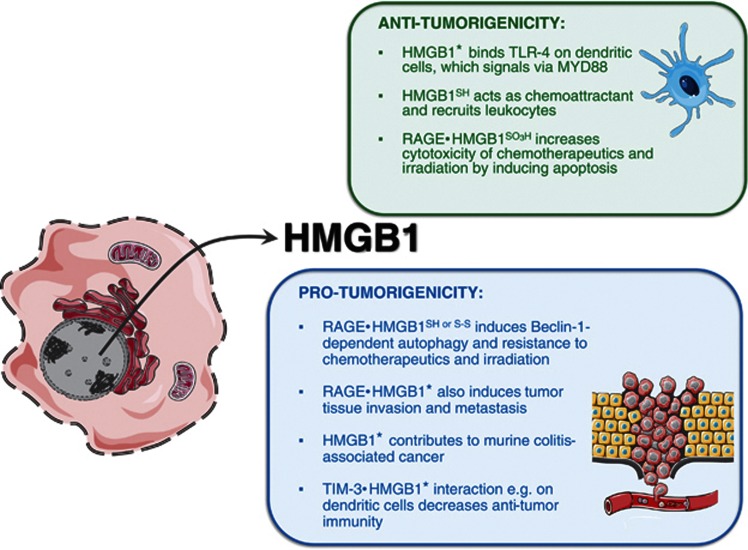
HMGB1 is a non-histone chromatin-binding protein expressed constitutively by all eukaryotic cells. In the nucleus it mediates different functions, such as stabilization of nucleosomes, DNA repair and recombination, and transcription.49 Although the nuclear function of HMGB1 was described in 1975,50 its extracellular immunomodulatory role was discovered only 24 years later.51 The authors found that HMGB1 was secreted as a cytokine by cultured macrophages after stimulation with endotoxin, TNF or IL-1, and that administration of antibodies against HMGB1 attenuated endotoxin lethality in mice.51 This study for the first time pointed to a previously unrecognized immunomodulatory role of HMGB1 in experimental mouse models of sepsis and in septic patients. It is now well established that extracellular HMGB1 participates in many biological processes, such as angiogenesis, myogenesis, chemotaxis and wound healing.52, 53, 54, 55, 56, 57, 58, 59 To fulfill its functions, extracellular HMGB1 binds to various receptors, including TLR2, TLR4, TLR9 and the receptor for advanced glycosylation end products (RAGE).60 Further down we will analyze its pro- and anti-tumorigenic activities and emphasize that HMGB1 is one of the DAMPs that have dual and opposite effects on cancer treatment. In cancer therapy, HMGB1 is a double-edged sword (Figure 3).
Figure 3.
Passive release of HMGB1 from the nucleus of tumor cells during the late stages of apoptotis (e.g. secondary necrosis) induces both pro- and anti-tumorigenic activities, which may depend on the redox state of HMGB1. The redox states are indicated by SH (fully reduced), S-S (disulfide bonds), SO3H (oxidized) and ★ (unknown)
HMGB1 is associated with cell injury (and acts as a DAMP). Initially, it was proposed that HMGB1 is passively released only from necrotic cells, whereas apoptotic and secondary necrotic cells retain it in the nucleus.61 However, it has become clear that most anti-cancer agents and inducers of apoptosis and ICD can promote HMGB1 release. It has been shown that extracellular HMGB1 is required for the immunogenicity of ICD.62 These authors used a prophylactic tumor vaccination model, in which immunization of mice with HMGB1 depletes CT26 cancer cells or co-injection of anti-HMGB1 antibody compromises the ability of mice to resist re-challenge with live CT26 cancer cells. An effective immune response against cancer cells undergoing ICD requires that DCs have functional TLR4 and intact signaling through its adaptor MYD88 so that antigen from dying tumor cells can be processed and cross-presented efficiently.62 Retrospective clinical analysis confirmed these data. In breast cancer patients, loss-of-function single-nucleotide polymorphism (SNP; Asp299Gly) in the Tlr-4 gene (reduces binding of HMGB-1 to TLR-4) correlated with early relapse after anthracycline treatment.62 In addition, it was recently shown that levels of HMGB1 increase significantly in the plasma of breast cancer patients who respond to neoadjuvant chemotherapy with epirubicin/docetaxel, but not in non-responders.63 Similarly, loss-of-function SNP mutation in the Tlr-4 gene (Thr399Ile) was associated with the worst overall survival of patients with head and neck squamous cell carcinomas after anthracycline administration64 and of patients with colorectal cancer receiving oxaliplatin-based therapy.65 Also, several other studies pointed to the contribution of extracellular HMGB1 to the efficacy of anti-cancer treatment. For example, it has been shown that HMGB-1 released from dying tumor cells treated with adenoviral vector, radiation and temozolomide led to infiltration of DCs and to regression of TLR-2-dependent brain tumors in a mouse model.66 Although all these studies point to the anti-cancer role of HMGB1, it is necessary to mention the other face of HMGB1: its pro-tumorigenic properties.
Several studies have indicated that HMGB1 is involved in tumor tissue invasion and metastasis. It has been shown that blockade of RAGE–HMGB1 decreased growth and metastasis of both implanted tumors and tumors developing spontaneously in susceptible mice.67 HMGB1 also mediates the development of murine colitis and colitis-associated cancer.68 The authors showed in the colitis-associated cancer model that a neutralizing anti-HMGB1 antibody decreased tumor incidence and size. Recently, it has been shown that the HMGB1 released from dying cells is critical for tumor progression in an established mouse model of prostate cancer. In this model, HMGB1 was required for the activation and intratumoral accumulation of T cells expressing cytokine lymphotoxin-α1β2 (LT) on their surface.16 These tumor-activated T cells were essential for promoting the progression of preneoplasia to invasive carcinoma in an LTβ receptor-dependent manner.16 Extracellular HMGB1 enhances re-growth and metastasis of cancer cells that have survived chemotherapy.69 Another study showed that HMGB1 could decrease anti-tumor immunity via interaction with T-cell immunoglobulin domain and mucin domain 3 (TIM-3).20 Tumor-associated DCs in mouse tumors and cancer patients showed strong expression of TIM-3, which interacted with HMGB1 and in that way decreased the therapeutic efficacy of DNA vaccination by diminishing the immunogenicity of DAMPs (e.g. nucleic acids) released from dying cancer cells. Moreover, the authors showed that cisplatin, a non-immunogenic chemotherapeutic,70 might compromise the innate anti-tumor responses of DCs by acting together with TIM-3-dependent inhibitor pathways.20
All these various functions of HMGB1 could be partially explained by a switching among mutually exclusive redox states. Recent studies have found that reduced cysteines make HMGB1 a chemoattractant, whereas a disulfide bond makes it a proinflammatory cytokine, and further cysteine oxidation to sulfonates mediated by ROS abrogates both activities.4 The different redox states of HMGB1 could be analyzed by liquid chromatography tandem mass spectrometric analysis and by electrophoretic mobility of HMGB1 in the presence and in the absence of dithiothreitol.4 It has also been shown that the redox state of extracellular HMGB1 critically regulates the autophagic response to anti-cancer agents and thereby influences anti-tumor efficacy. Reducible HMGB1 binds to RAGE, induces Beclin-1-dependent autophagy, and promotes resistance to chemotherapeutic agents or ionizing radiation, while oxidized HMGB1 increases the cytotoxicity of these chemotherapeutics via induction of apoptosis.71
From one point of view all these modifications could occur after the release of HMGB1 within the injured tissues. A possible scenario was proposed for muscle injury:4 all-thiol-HMGB1 is released first to recruit leukocytes, which in turn produce disulfide-HMGB1 directly by secretion and/or indirectly by ROS-mediated partial oxidation of extracellular HMGB1. Sustained ROS production induces the terminal oxidation of HMGB1, which gets inactivated during the resolution of inflammation. However, the extracellular redox state in cancer may be highly variable.72 Different cancer cell types or different stages of progression of the same cancer cell type might generate unique extracellular redox states.72 This redox state variability associated with cancer cells may partially explain the diversity of HMGB1 effects in anti-tumour immunity.12
But from another point of view, modification of HMGB1 (and possibly of other DAMPs) could be caused by activation of a certain biochemical pathway during a specific cell death modality. For example, it has been shown that caspase activity in apoptosis is responsible for modification of the immunogenic form of HMGB1 into the non-immunogenic oxidized HMGB1 form.73 In fact, the biochemical processes activated during the different cell death modalities (e.g. in apoptosis versus necroptosis) may profoundly affect the composition and properties of the released DAMPs.74
It is also important to take into account that properties of the intrinsic tumor microenvironment, such as the hypoxia often present within tumors,75 could modify the action of ICD stressors and modulate the immunomodulatory properties of DAMPs. It was recently shown that hypoxia leads to HMGB1 release, which contributes to tumor invasiveness.76 It is well known that hypoxic conditions lead to increased production of ROS, which not only mediate hypoxia-induced HMGB1 release77 but could also modify the redox state of HMGB1. However, in these studies71, 76, 77 no distinction was made between all-thiol-HMGB1 and disulfide-HMGB1. It is important to analyze whether these three different redox states of HMGB1 (all-thiol-HMGB1, disulfide-HMGB1 and oxidized HMGB1) can explain the different functions of HMGB1 in anti-cancer treatment.
Future Perspectives
The insights from the last several years increasingly support the idea that DAMPs represent one of the main characteristics of ICD that are required to induce anti-tumor immunity. Most studies are focusing on experimental mouse models, but it is necessary to explore the possibility of designing large-scale studies in order to determine the prognostic value of ICD associated with DAMP emission in patients suffering from different types of cancers. But it should be kept in mind that under certain conditions, the presence of DAMPs in the tumor microenvironment can promote tumor metastasis and progression. The main question concerns the difference between DAMPs that drive cancer progression and those that inhibit tumor growth. To answer that question, it will be essential to characterize the post-translational modifications of DAMPs, the chemotherapeutic drugs, and the types of tumors and their microenvironment.
Acknowledgments
We thank Dr. A Bredan for editing the manuscript. This work was supported by project grants from the Fund for Scientific Research Flanders (FWO-Vlaanderen, G.0728.10, 3G060713, 3G0A5413 to DVK; 3G067512 to DVK and OK; G.0642.10N to OK and CB), by an individual research grant from FWO-Vlaanderen (31507110 to DVK) and by the Federal Research Programme (IAP 7/30 to CB). DVK is a senior postdoctoral researcher and TLA is a doctoral fellow, both paid by fellowships from FWO-Vlaanderen. Vandenabeele's group is supported by VIB, Ghent University (GROUP-ID Consortium of the UGent MRP initiative), FWO-Vlaanderen (G.0875.11, G.0973.11, G.0A45.12N), Federal Research Programme (IAP 7/32), European Research Programme FP6 ApopTrain (MRTN-CT-035624), FP7 Apo-Sys 200767, and the Euregional PACTII. PV holds a Methusalem Grant (BOF09/01M00709) from the Flemish Government.
Glossary
- AML
acute myeloid leukemia
- ATP
adenosine triphosphate
- CRT
calreticulin
- DAMPs
damage-associated molecular patterns
- DC
dendritic cells
- eIF2α
eukaryotic initiation factor 2α
- ER
endoplasmic reticulum
- HMGB1
high-mobility group box 1 protein
- ICD
immunogenic cell death
- IL
interleukin
- LT
lymphotoxin
- MHC
major histocompatibility complex
- MYD88
myeloid differentiation primary response gene 88
- NLRs
NOD-like receptors
- PAMPs
pathogen-associated molecular patterns
- PDT
photodynamic therapy
- PERK
PKR-like ER kinase
- PI3K
phosphoinositide 3-kinase
- PS
phosphatidylserine
- RAGE
receptor for advanced glycation end products
- RLRs
RIG-I-like receptors
- ROS
reactive oxygen species
- TIM-3
T-cell immunoglobulin domain and mucin domain 3
- TLRs
Toll-like receptors
- TNF
tumor necrosis factor
- Tregs
T regulatory cells
The authors declare no conflict of interest.
Footnotes
Edited by G Melino
References
- Matzinger P. Tolerance, danger, and the extended family. Annu Rev Immunol. 1994;12:991–1045. doi: 10.1146/annurev.iy.12.040194.005015. [DOI] [PubMed] [Google Scholar]
- Krysko DV, Agostinis P, Krysko O, Garg AD, Bachert C, Lambrecht BN, et al. Emerging role of damage-associated molecular patterns derived from mitochondria in inflammation. Trends Immunol. 2011;32:157–164. doi: 10.1016/j.it.2011.01.005. [DOI] [PubMed] [Google Scholar]
- Garg AD, Nowis D, Golab J, Vandenabeele P, Krysko DV, Agostinis P. Immunogenic cell death, DAMPs and anticancer therapeutics: an emerging amalgamation. Biochim Biophys Acta. 2010;1805:53–71. doi: 10.1016/j.bbcan.2009.08.003. [DOI] [PubMed] [Google Scholar]
- Venereau E, Casalgrandi M, Schiraldi M, Antoine DJ, Cattaneo A, De Marchis F, et al. Mutually exclusive redox forms of HMGB1 promote cell recruitment or proinflammatory cytokine release. J Exp Med. 2012;209:1519–1528. doi: 10.1084/jem.20120189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang H, Lundback P, Ottosson L, Erlandsson-Harris H, Venereau E, Bianchi ME, et al. Redox modification of cysteine residues regulates the cytokine activity of high mobility group box-1 (HMGB1) Mol Med. 2012;18:250–259. doi: 10.2119/molmed.2011.00389. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Zitvogel L, Kepp O, Kroemer G. Decoding cell death signals in inflammation and immunity. Cell. 2010;140:798–804. doi: 10.1016/j.cell.2010.02.015. [DOI] [PubMed] [Google Scholar]
- Palm NW, Medzhitov R. Pattern recognition receptors and control of adaptive immunity. Immunol Rev. 2009;227:221–233. doi: 10.1111/j.1600-065X.2008.00731.x. [DOI] [PubMed] [Google Scholar]
- Krysko DV, Kaczmarek A, Krysko O, Heyndrickx L, Woznicki J, Bogaert P, et al. TLR-2 and TLR-9 are sensors of apoptosis in a mouse model of doxorubicin-induced acute inflammation. Cell Death Differ. 2011;18:1316–1325. doi: 10.1038/cdd.2011.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dudek AM, Garg AD, Krysko DV, De Ruysscher D, Agostinis P.Inducers of immunogenic cancer cell death Cytokine Growth Factor Rev 2013. e-pub ahead of print, 4 February 2013. doi: pii: S1359-6101(13)00006-3. 10.1016/j.cytogfr.2013.01.005 [DOI] [PubMed]
- Panaretakis T, Kepp O, Brockmeier U, Tesniere A, Bjorklund AC, Chapman DC, et al. Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. EMBO J. 2009;28:578–590. doi: 10.1038/emboj.2009.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garg AD, Krysko DV, Verfaillie T, Kaczmarek A, Ferreira GB, Marysael T, et al. A novel pathway combining calreticulin exposure and ATP secretion in immunogenic cancer cell death. EMBO J. 2012;31:1062–1079. doi: 10.1038/emboj.2011.497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Krysko DV, Garg AD, Kaczmarek A, Krysko O, Agostinis P, Vandenabeele P. Immunogenic cell death and DAMPs in cancer therapy. Nat Rev Cancer. 2012;12:860–875. doi: 10.1038/nrc3380. [DOI] [PubMed] [Google Scholar]
- Obeid M, Tesniere A, Ghiringhelli F, Fimia GM, Apetoh L, Perfettini JL, et al. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007;13:54–61. doi: 10.1038/nm1523. [DOI] [PubMed] [Google Scholar]
- Casares N, Pequignot MO, Tesniere A, Ghiringhelli F, Roux S, Chaput N, et al. Caspase-dependent immunogenicity of doxorubicin-induced tumor cell death. J Exp Med. 2005;202:1691–1701. doi: 10.1084/jem.20050915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciampricotti M, Hau CS, Doornebal CW, Jonkers J, de Visser KE. Chemotherapy response of spontaneous mammary tumors is independent of the adaptive immune system. Nat Med. 2012;18:344–346. doi: 10.1038/nm.2652. [DOI] [PubMed] [Google Scholar]
- He Y, Zha J, Wang Y, Liu W, Yang X, Yu P. Tissue damage-associated ‘danger signals' influence T cell responses that promote the progression of pre-neoplasia to cancer. Cancer Res. 2012;73:629–639. doi: 10.1158/0008-5472.CAN-12-2704. [DOI] [PubMed] [Google Scholar]
- Tang D, Kang R, Livesey KM, Cheh CW, Farkas A, Loughran P, et al. Endogenous HMGB1 regulates autophagy. J Cell Biol. 2010;190:881–892. doi: 10.1083/jcb.200911078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L, Yang M, Kang R, Wang Z, Zhao Y, Yu Y, et al. HMGB1-induced autophagy promotes chemotherapy resistance in leukemia cells. Leukemia. 2011;25:23–31. doi: 10.1038/leu.2010.225. [DOI] [PubMed] [Google Scholar]
- Liu L, Yang M, Kang R, Wang Z, Zhao Y, Yu Y, et al. DAMP-mediated autophagy contributes to drug resistance. Autophagy. 2011;7:112–114. doi: 10.4161/auto.7.1.14005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiba S, Baghdadi M, Akiba H, Yoshiyama H, Kinoshita I, Dosaka-Akita H, et al. Tumor-infiltrating DCs suppress nucleic acid-mediated innate immune responses through interactions between the receptor TIM-3 and the alarmin HMGB1. Nat Immunol. 2012;13:832–842. doi: 10.1038/ni.2376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heijmans J, Buller NV, Hoff E, Dihal AA, van der Poll T, van Zoelen MA, et al. Rage signalling promotes intestinal tumourigenesis. Oncogene. 2012;32:1202–1206. doi: 10.1038/onc.2012.119. [DOI] [PubMed] [Google Scholar]
- Gelebart P, Opas M, Michalak M. Calreticulin, a Ca2+-binding chaperone of the endoplasmic reticulum. Int J Biochem Cell Biol. 2005;37:260–266. doi: 10.1016/j.biocel.2004.02.030. [DOI] [PubMed] [Google Scholar]
- Gold LI, Eggleton P, Sweetwyne MT, Van Duyn LB, Greives MR, Naylor SM, et al. Calreticulin: non-endoplasmic reticulum functions in physiology and disease. FASEB J. 2010;24:665–683. doi: 10.1096/fj.09-145482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garg AD, Kaczmarek A, Krysko O, Vandenabeele P, Krysko DV, Agostinis P. ER stress-induced inflammation: does it aid or impede disease progression. Trends Mol Med. 2012;18:589–598. doi: 10.1016/j.molmed.2012.06.010. [DOI] [PubMed] [Google Scholar]
- Garg AD, Krysko DV, Vandenabeele P, Agostinis P. The emergence of phox-ER stress induced immunogenic apoptosis. OncoImmunology. 2012;1:786–788. doi: 10.4161/onci.19750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fucikova J, Kralikova P, Fialova A, Brtnicky T, Rob L, Bartunkova J, et al. Human tumor cells killed by anthracyclines induce a tumor-specific immune response. Cancer Res. 2011;71:4821–4833. doi: 10.1158/0008-5472.CAN-11-0950. [DOI] [PubMed] [Google Scholar]
- Zappasodi R, Pupa SM, Ghedini GC, Bongarzone I, Magni M, Cabras AD, et al. Improved clinical outcome in indolent B-cell lymphoma patients vaccinated with autologous tumor cells experiencing immunogenic death. Cancer Res. 2010;70:9062–9072. doi: 10.1158/0008-5472.CAN-10-1825. [DOI] [PubMed] [Google Scholar]
- Wemeau M, Kepp O, Tesniere A, Panaretakis T, Flament C, De Botton S, et al. Calreticulin exposure on malignant blasts predicts a cellular anticancer immune response in patients with acute myeloid leukemia. Cell Death Dis. 2010;1:e104. doi: 10.1038/cddis.2010.82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fredly H, Ersvaer E, Gjertsen BT, Bruserud O. Immunogenic apoptosis in human acute myeloid leukemia (AML): primary human AML cells expose calreticulin and release heat shock protein (HSP) 70 and HSP90 during apoptosis. Oncol Rep. 2011;25:1549–1556. doi: 10.3892/or.2011.1229. [DOI] [PubMed] [Google Scholar]
- Liu R, Gong J, Chen J, Li Q, Song C, Zhang J, et al. Calreticulin as a potential diagnostic biomarker for lung cancer. Cancer Immunol Immunother. 2012;61:855–864. doi: 10.1007/s00262-011-1146-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elliott MR, Chekeni FB, Trampont PC, Lazarowski ER, Kadl A, Walk SF, et al. Nucleotides released by apoptotic cells act as a find-me signal to promote phagocytic clearance. Nature. 2009;461:282–286. doi: 10.1038/nature08296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martins I, Tesniere A, Kepp O, Michaud M, Schlemmer F, Senovilla L, et al. Chemotherapy induces ATP release from tumor cells. Cell Cycle. 2009;8:3723–3728. doi: 10.4161/cc.8.22.10026. [DOI] [PubMed] [Google Scholar]
- Ayna G, Krysko DV, Kaczmarek A, Petrovski G, Vandenabeele P, Fesus L. ATP release from dying autophagic cells and their phagocytosis are crucial for inflammasome activation in macrophages. PLoS One. 2012;7:e40069. doi: 10.1371/journal.pone.0040069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chekeni FB, Elliott MR, Sandilos JK, Walk SF, Kinchen JM, Lazarowski ER, et al. Pannexin 1 channels mediate ‘find-me' signal release and membrane permeability during apoptosis. Nature. 2010;467:863–867. doi: 10.1038/nature09413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Michaud M, Martins I, Sukkurwala AQ, Adjemian S, Ma Y, Pellegatti P, et al. Autophagy-dependent anticancer immune responses induced by chemotherapeutic agents in mice. Science. 2011;334:1573–1577. doi: 10.1126/science.1208347. [DOI] [PubMed] [Google Scholar]
- Martins I, Michaud M, Sukkurwala AQ, Adjemian S, Ma Y, Shen S, et al. Premortem autophagy determines the immunogenicity of chemotherapy-induced cancer cell death. Autophagy. 2012;8:3. doi: 10.4161/auto.19009. [DOI] [PubMed] [Google Scholar]
- Ghiringhelli F, Apetoh L, Tesniere A, Aymeric L, Ma Y, Ortiz C, et al. Activation of the NLRP3 inflammasome in dendritic cells induces IL-1beta-dependent adaptive immunity against tumors. Nat Med. 2009;15:1170–1178. doi: 10.1038/nm.2028. [DOI] [PubMed] [Google Scholar]
- Idzko M, Dichmann S, Ferrari D, Di Virgilio F, la Sala A, Girolomoni G, et al. Nucleotides induce chemotaxis and actin polymerization in immature but not mature human dendritic cells via activation of pertussis toxin-sensitive P2y receptors. Blood. 2002;100:925–932. doi: 10.1182/blood.v100.3.925. [DOI] [PubMed] [Google Scholar]
- Myrtek D, Idzko M. Chemotactic activity of extracellular nucleotideson human immune cells. Purinergic Signal. 2007;3:5–11. doi: 10.1007/s11302-006-9032-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- la Sala A, Sebastiani S, Ferrari D, Di Virgilio F, Idzko M, Norgauer J, et al. Dendritic cells exposed to extracellular adenosine triphosphate acquire the migratory properties of mature cells and show a reduced capacity to attract type 1 T lymphocytes. Blood. 2002;99:1715–1722. doi: 10.1182/blood.v99.5.1715. [DOI] [PubMed] [Google Scholar]
- Zhang Y, Gong LH, Zhang HQ, Du Q, You JF, Tian XX, et al. Extracellular ATP enhances in vitro invasion of prostate cancer cells by activating Rho GTPase and upregulating MMPs expression. Cancer Lett. 2010;293:189–197. doi: 10.1016/j.canlet.2010.01.010. [DOI] [PubMed] [Google Scholar]
- Ryu JK, Choi HB, Hatori K, Heisel RL, Pelech SL, McLarnon JG, et al. Adenosine triphosphate induces proliferation of human neural stem cells: role of calcium and p70 ribosomal protein S6 kinase. J Neurosci Res. 2003;72:352–362. doi: 10.1002/jnr.10507. [DOI] [PubMed] [Google Scholar]
- Robson SC, Sevigny J, Zimmermann H. The E-NTPDase family of ectonucleotidases: structure function relationships and pathophysiological significance. Purinergic Signal. 2006;2:409–430. doi: 10.1007/s11302-006-9003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beavis PA, Stagg J, Darcy PK, Smyth MJ. CD73: a potent suppressor of antitumor immune responses. Trends Immunol. 2012;33:231–237. doi: 10.1016/j.it.2012.02.009. [DOI] [PubMed] [Google Scholar]
- Ohta A, Gorelik E, Prasad SJ, Ronchese F, Lukashev D, Wong MK, et al. A2A adenosine receptor protects tumors from antitumor T cells. Proc Natl Acad Sci USA. 2006;103:13132–13137. doi: 10.1073/pnas.0605251103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pellegatti P, Raffaghello L, Bianchi G, Piccardi F, Pistoia V, Di Virgilio F. Increased level of extracellular ATP at tumor sites: in vivo imaging with plasma membrane luciferase. PLoS One. 2008;3:e2599. doi: 10.1371/journal.pone.0002599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trabanelli S, Ocadlikova D, Gulinelli S, Curti A, Salvestrini V, de Paula Vieira R, et al. Extracellular ATP exerts opposite effects on activated and regulatory CD4+ T cells via purinergic P2 receptor activation. J Immunol. 2012;189:1303–1310. doi: 10.4049/jimmunol.1103800. [DOI] [PubMed] [Google Scholar]
- la Sala A, Ferrari D, Corinti S, Cavani A, Di Virgilio F, Girolomoni G. Extracellular ATP induces a distorted maturation of dendritic cells and inhibits their capacity to initiate Th1 responses. J Immunol. 2001;166:1611–1617. doi: 10.4049/jimmunol.166.3.1611. [DOI] [PubMed] [Google Scholar]
- Tang D, Kang R, Zeh HJ, 3rd, Lotze MT. High-mobility group box 1 and cancer. Biochim Biophys Acta. 2010;1799:131–140. doi: 10.1016/j.bbagrm.2009.11.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andersson U, Tracey KJ. HMGB1 is a therapeutic target for sterile inflammation and infection. Annu Rev Immunol. 2011;29:139–162. doi: 10.1146/annurev-immunol-030409-101323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang H, Bloom O, Zhang M, Vishnubhakat JM, Ombrellino M, Che J, et al. HMG-1 as a late mediator of endotoxin lethality in mice. Science. 1999;285:248–251. doi: 10.1126/science.285.5425.248. [DOI] [PubMed] [Google Scholar]
- Palumbo R, Sampaolesi M, De Marchis F, Tonlorenzi R, Colombetti S, Mondino A, et al. Extracellular HMGB1, a signal of tissue damage, induces mesoangioblast migration and proliferation. J Cell Biol. 2004;164:441–449. doi: 10.1083/jcb.200304135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schlueter C, Weber H, Meyer B, Rogalla P, Roser K, Hauke S, et al. Angiogenetic signaling through hypoxia: HMGB1: an angiogenetic switch molecule. Am J Pathol. 2005;166:1259–1263. doi: 10.1016/S0002-9440(10)62344-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitola S, Belleri M, Urbinati C, Coltrini D, Sparatore B, Pedrazzi M, et al. Cutting edge: extracellular high mobility group box-1 protein is a proangiogenic cytokine. J Immunol. 2006;176:12–15. doi: 10.4049/jimmunol.176.1.12. [DOI] [PubMed] [Google Scholar]
- Sorci G, Riuzzi F, Arcuri C, Giambanco I, Donato R. Amphoterin stimulates myogenesis and counteracts the antimyogenic factors basic fibroblast growth factor and S100B via RAGE binding. Mol Cell Biol. 2004;24:4880–4894. doi: 10.1128/MCB.24.11.4880-4894.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Straino S, Di Carlo A, Mangoni A, De Mori R, Guerra L, Maurelli R, et al. High-mobility group box 1 protein in human and murine skin: involvement in wound healing. J Invest Dermatol. 2008;128:1545–1553. doi: 10.1038/sj.jid.5701212. [DOI] [PubMed] [Google Scholar]
- Degryse B, Bonaldi T, Scaffidi P, Muller S, Resnati M, Sanvito F, et al. The high mobility group (HMG) boxes of the nuclear protein HMG1 induce chemotaxis and cytoskeleton reorganization in rat smooth muscle cells. J Cell Biol. 2001;152:1197–1206. doi: 10.1083/jcb.152.6.1197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porto A, Palumbo R, Pieroni M, Aprigliano G, Chiesa R, Sanvito F, et al. Smooth muscle cells in human atherosclerotic plaques secrete and proliferate in response to high mobility group box 1 protein. FASEB J. 2006;20:2565–2566. doi: 10.1096/fj.06-5867fje. [DOI] [PubMed] [Google Scholar]
- Chavakis E, Hain A, Vinci M, Carmona G, Bianchi ME, Vajkoczy P, et al. High-mobility group box 1 activates integrin-dependent homing of endothelial progenitor cells. Circ Res. 2007;100:204–212. doi: 10.1161/01.RES.0000257774.55970.f4. [DOI] [PubMed] [Google Scholar]
- Yanai H, Ban T, Taniguchi T. High-mobility group box family of proteins: ligand and sensor for innate immunity. Trends Immunol. 2012;33:633–640. doi: 10.1016/j.it.2012.10.005. [DOI] [PubMed] [Google Scholar]
- Scaffidi P, Misteli T, Bianchi ME. Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature. 2002;418:191–195. doi: 10.1038/nature00858. [DOI] [PubMed] [Google Scholar]
- Apetoh L, Ghiringhelli F, Tesniere A, Obeid M, Ortiz C, Criollo A, et al. Toll-like receptor 4-dependent contribution of the immune system to anticancer chemotherapy and radiotherapy. Nat Med. 2007;13:1050–1059. doi: 10.1038/nm1622. [DOI] [PubMed] [Google Scholar]
- Arnold T, Michlmayr A, Baumann S, Burghuber C, Pluschnig U, Bartsch R, et al. Plasma HMGB-1 after the initial dose of epirubicin/docetaxel in cancer. Eur J Clin Invest. 2013;43:286–291. doi: 10.1111/eci.12043. [DOI] [PubMed] [Google Scholar]
- Bergmann C, Bachmann HS, Bankfalvi A, Lotfi R, Putter C, Wild CA, et al. Toll-like receptor 4 single-nucleotide polymorphisms Asp299Gly and Thr399Ile in head and neck squamous cell carcinomas. J Transl Med. 2011;9:139. doi: 10.1186/1479-5876-9-139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tesniere A, Schlemmer F, Boige V, Kepp O, Martins I, Ghiringhelli F, et al. Immunogenic death of colon cancer cells treated with oxaliplatin. Oncogene. 2010;29:482–491. doi: 10.1038/onc.2009.356. [DOI] [PubMed] [Google Scholar]
- Curtin JF, Liu N, Candolfi M, Xiong W, Assi H, Yagiz K, et al. HMGB1 mediates endogenous TLR2 activation and brain tumor regression. PLoS Med. 2009;6:e10. doi: 10.1371/journal.pmed.1000010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taguchi A, Blood DC, del Toro G, Canet A, Lee DC, Qu W, et al. Blockade of RAGE-amphoterin signalling suppresses tumour growth and metastases. Nature. 2000;405:354–360. doi: 10.1038/35012626. [DOI] [PubMed] [Google Scholar]
- Maeda S, Hikiba Y, Shibata W, Ohmae T, Yanai A, Ogura K, et al. Essential roles of high-mobility group box 1 in the development of murine colitis and colitis-associated cancer. Biochem Biophys Res Commun. 2007;360:394–400. doi: 10.1016/j.bbrc.2007.06.065. [DOI] [PubMed] [Google Scholar]
- Luo Y, Chihara Y, Fujimoto K, Sasahira T, Kuwada M, Fujiwara R, et al. High mobility group box 1 released from necrotic cells enhances regrowth and metastasis of cancer cells that have survived chemotherapy. Eur J Cancer. 49:741–751. doi: 10.1016/j.ejca.2012.09.016. [DOI] [PubMed] [Google Scholar]
- Martins I, Kepp O, Schlemmer F, Adjemian S, Tailler M, Shen S, et al. Restoration of the immunogenicity of cisplatin-induced cancer cell death by endoplasmic reticulum stress. Oncogene. 2011;30:1147–1158. doi: 10.1038/onc.2010.500. [DOI] [PubMed] [Google Scholar]
- Tang D, Kang R, Cheh CW, Livesey KM, Liang X, Schapiro NE, et al. HMGB1 release and redox regulates autophagy and apoptosis in cancer cells. Oncogene. 2010;29:5299–5310. doi: 10.1038/onc.2010.261. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chaiswing L, Oberley TD. Extracellular/microenvironmental redox state. Antioxid Redox Signal. 2010;13:449–465. doi: 10.1089/ars.2009.3020. [DOI] [PubMed] [Google Scholar]
- Kazama H, Ricci JE, Herndon JM, Hoppe G, Green DR, Ferguson TA. Induction of immunological tolerance by apoptotic cells requires caspase-dependent oxidation of high-mobility group box-1 protein. Immunity. 2008;29:21–32. doi: 10.1016/j.immuni.2008.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaczmarek A, Vandenabeele P, Krysko DV. Necroptosis: the release of damage-associated molecular patterns and its physiological relevance. Immunity. 2013;38:209–223. doi: 10.1016/j.immuni.2013.02.003. [DOI] [PubMed] [Google Scholar]
- Harrison L, Blackwell K. Hypoxia and anemia: factors in decreased sensitivity to radiation therapy and chemotherapy. Oncologist. 2004;9:31–40. doi: 10.1634/theoncologist.9-90005-31. [DOI] [PubMed] [Google Scholar]
- Yan W, Chang Y, Liang X, Cardinal JS, Huang H, Thorne SH, et al. High-mobility group box 1 activates caspase-1 and promotes hepatocellular carcinoma invasiveness and metastases. Hepatology. 2012;55:1863–1875. doi: 10.1002/hep.25572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsung A, Klune JR, Zhang X, Jeyabalan G, Cao Z, Peng X, et al. HMGB1 release induced by liver ischemia involves Toll-like receptor 4 dependent reactive oxygen species production and calcium-mediated signaling. J Exp Med. 2007;204:2913–2923. doi: 10.1084/jem.20070247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang D, Postnikov YV, Li Y, Tewary P, de la Rosa G, Wei F, et al. High-mobility group nucleosome-binding protein 1 acts as an alarmin and is critical for lipopolysaccharide-induced immune responses. J Exp Med. 2012;209:157–171. doi: 10.1084/jem.20101354. [DOI] [PMC free article] [PubMed] [Google Scholar]