Abstract
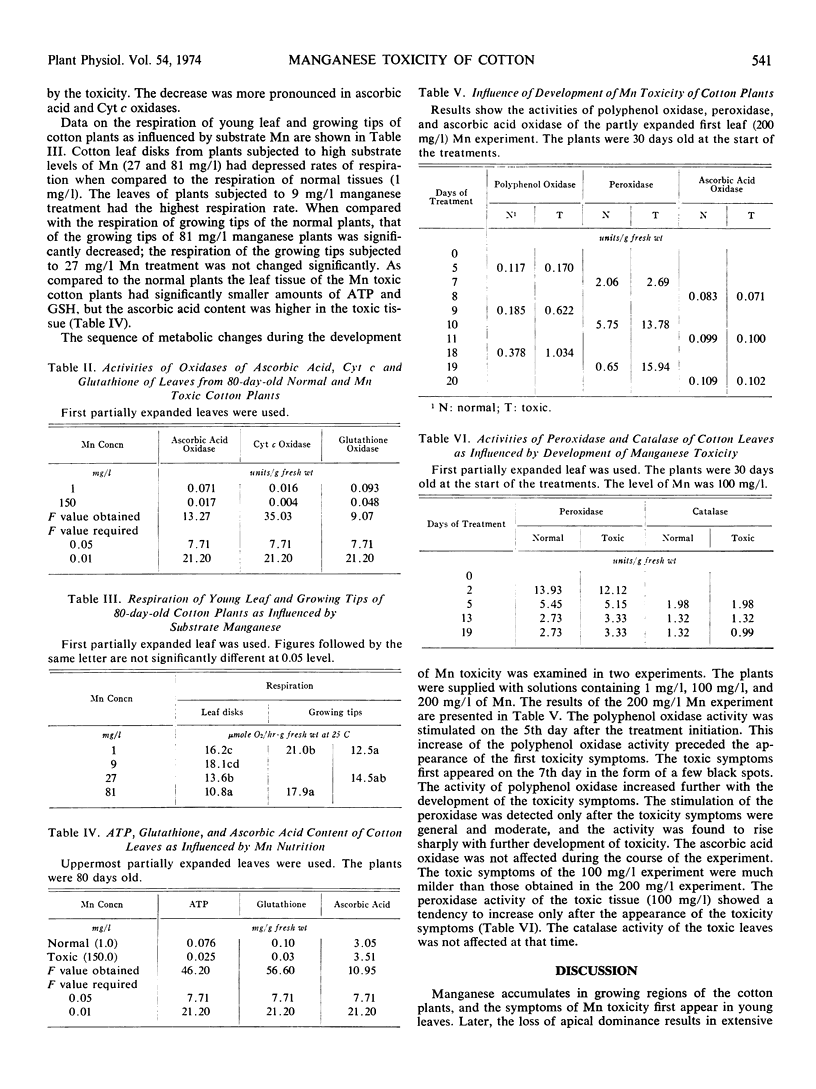
Cotton plants (Gossypium hirsutum. Linn. var. Sankar 4) were grown at normal and toxic levels of substrate manganese, and the altered metabolism of manganese toxic plants was studied. The tissues of plants exposed to toxic levels of manganese had higher activities of peroxidase and polyphenol oxidase, and the activities of catalase, ascorbic acid oxidase, glutathione oxidase and cytochrome c oxidase were lowered. In addition, the high manganese tissue had lower contents of ATP and glutathione but higher amounts of ascorbic acid. The respiration of the partially expanded leaves and the growing tips of toxic plants were depressed when compared to that of the normal tissues. The metabolic changes of manganese toxicity of cotton are placed in the following order: accumulation of manganese in the leaf tissue; a rise in respiration; stimulation of polyphenol oxidase; the appearance of initial toxicity symptoms; the evolution of ethylene and stimulation of peroxidase; the presence of severe toxicity symptoms; the depression of terminal oxidases and respiration; abscission of the growing tip and proliferation of the stem tissue. The early stimulation of polyphenol oxidase may be used to detect potential manganese toxicity.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- COOPERSTEIN S. J., LAZAROW A. A microspectrophotometric method for the determination of cytochrome oxidase. J Biol Chem. 1951 Apr;189(2):665–670. [PubMed] [Google Scholar]
- Fowler J. L., Morgan P. W. The relationship of the peroxidative indoleacetic Acid oxidase system to in vivo ethylene synthesis in cotton. Plant Physiol. 1972 Apr;49(4):555–559. doi: 10.1104/pp.49.4.555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- GALSTON A. W., BONNER J., BAKER R. S. Flavoprotein and peroxidase as components of the indoleacetic acid oxidase system of peas. Arch Biochem Biophys. 1953 Feb;42(2):456–470. doi: 10.1016/0003-9861(53)90373-7. [DOI] [PubMed] [Google Scholar]
- GOLDACRE P. L. Hydrogen peroxide in the enzymic oxidation of heteroauxin. Aust J Sci Res B. 1951 Aug;4(3):293–302. doi: 10.1071/bi9510293. [DOI] [PubMed] [Google Scholar]
- GORTNER W. A., KENT M. Indoleacetic acid oxidase and an inhibitor in pineapple tissue. J Biol Chem. 1953 Oct;204(2):593–603. [PubMed] [Google Scholar]
- GRUNERT R. R., PHILLIPS P. H. A modification of the nitroprusside method of analysis for glutathione. Arch Biochem. 1951 Feb;30(2):217–225. [PubMed] [Google Scholar]
- KENTEN R. H. The oxidation of indolyl-3-acetic acid by waxpod bean root sap and peroxidase systems. Biochem J. 1955 Jan;59(1):110–121. doi: 10.1042/bj0590110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan P. W., Hall W. C. Indoleacetic Acid Oxidizing Enzyme & Inhibitors from Light-Grown Cotton. Plant Physiol. 1963 Jul;38(4):365–370. doi: 10.1104/pp.38.4.365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morgan P. W., Joham H. E., Amin J. V. Effect of manganese toxicity on the indoleacetic Acid oxidase system of cotton. Plant Physiol. 1966 Apr;41(4):718–724. doi: 10.1104/pp.41.4.718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- RAY P. M., THIMANN K. V. The destruction of indoleacetic acid. I. Action of an enzyme from Omphalia flavida. Arch Biochem Biophys. 1956 Sep;64(1):175–192. doi: 10.1016/0003-9861(56)90253-3. [DOI] [PubMed] [Google Scholar]
- ROE J. H. Chemical determination of ascorbic, dehydroascorbic, and diketogulonic acids. Methods Biochem Anal. 1954;1:115–139. doi: 10.1002/9780470110171.ch5. [DOI] [PubMed] [Google Scholar]
- STREHLER B. L., TOTTER J. R. Determination of ATP and related compounds: firefly luminescence and other methods. Methods Biochem Anal. 1954;1:341–356. doi: 10.1002/9780470110171.ch13. [DOI] [PubMed] [Google Scholar]
- Taylor D. M., Morgan P. W., Joham H. E., Amin J. V. Influence of Substrate and Tissue Manganese on the IAA-Oxidase System in Cotton. Plant Physiol. 1968 Feb;43(2):243–247. doi: 10.1104/pp.43.2.243. [DOI] [PMC free article] [PubMed] [Google Scholar]



