Abstract
Oral squamous cell carcinoma (OSCC) ranks among the top ten most prevalent cancers worldwide. Like most head and neck squamous cell carcinomas (HNSCCs), OSCC is highly inflammatory and aggressive. However, the signaling pathways triggering the activation of its inflammatory processes remain elusive. G protein-coupled receptor signaling regulates the inflammatory response and invasiveness of cancers, but it remains unclear whether Gα12 is a critical player in the inflammatory cytokine pathway during the tumorigenesis of OSCC. This study was undertaken to determine the role of Gα12 signaling in the regulation of proinflammatory cytokines in their mediation of OSCC invasion. We found that both the transcription and protein levels of Gα12 are up-regulated in OSCC tumors. The elevated Gα12 expressions in OSCC patients also correlated with extra-capsular spread, an indicator of tumor invasiveness in HNSCCs. This clinical finding was supported by the studies of overexpression and RNAi knockdown of Gα12 in OSCC cells, which demonstrated that Gα12 promoted tumor cell migration and invasion. To understand how Gα12 modulates OSCC invasiveness, we analyzed key biological processes in microarray data upon depletion of Gα12 and found that cytokine- and other immune-related pathways were severely impaired. Importantly, the mRNA levels of IL-6 and IL-8 proinflammatory cytokines in clinical samples were found to be significantly correlated with the increased Gα12 levels, suggesting a potential role of Gα12 in modulating the IL-6 and IL-8 expressions. Supporting this hypothesis, overexpression or RNAi knockdown of Gα12 in OSCC cell lines both showed that Gα12 positively regulated the mRNA and protein levels of IL-6 and IL-8. Finally, we demonstrated that the Gα12 promotion of tumor cell invasiveness was suppressed by the neutralization of IL-6 and IL-8 in OSCC cells. Together, these findings suggest that Gα12 drives OSCC invasion through the up-regulation of IL-6 and IL-8 cytokines.
Introduction
Head and neck squamous cell carcinomas (HNSCCs) rank as the sixth most prevalent cancer worldwide, affecting up to 600,000 people each year [1], [2]. Among the various HNSCC subtypes, about 10% are accounted by oral squamous cell carcinoma (OSCC) [1]. However, the overall survival rate for OSCC patients remains poor (approximately 25% in 5-years) [1]. Similar to other subtypes of HNSCC, the development of OSCC is closely intertwined with behavioral and environmental risk factors, including the consumption of alcohol, tobacco, and betel nut as well as the infection by human papillomavirus [3]–[5]. These risk factors induce proinflammatory cytokine responses, which contribute to the high aggressiveness of malignancy associated with OSCC [6], [7]. A number of cytokines involved in proinflammation are known to be expressed in HNSCCs, including interleukin-1, interleukin-6 (IL-6), interleukin-8 (IL-8), tumor necrosis factor-α, and granulocyte-macrophage colony-stimulating factor [8], [9]. Specifically, the up-regulation of IL-6 and IL-8 has been suggested to play important roles in the development and progression of OSCC and other cancers [10]–[14]. The expression levels of IL-6 and IL-8 have been found to be elevated in the tumor, saliva, and serum of OSCC patients compared to the control groups [15]–[19]. Thus, IL-6 and IL8 has been proposed as potential biomarkers for the diagnosis and follow-up for OSCC patients [10], [12], [14]. However, the cellular regulatory molecules for the modulation of IL-6 and IL-8 responses in OSCC remain to be defined.
G protein-coupled receptors (GPCRs) are critical cell surface proteins involved in modulating inflammatory diseases and cancers [20], [21]. GPCR proteins are a large family of seven-transmembrane domain receptors, which sense external molecules and activate intracellular signal transduction pathways for various physiological responses, including proliferation, differentiation, and chemotaxis. Activation of GPCRs or overexpression of the GPCR signaling molecules are frequently found to play a critical role in promoting tumor growth, metastasis, and angiogenesis [20]–[23]. Most GPCRs transduce signals by activation of heterotrimeric G-proteins that are composed of Gα and Gβ/Gγ subunits. Gα12 is defined as one of the four classes of G protein α subunits, functioning in the regulation of cell motility through activating small GTP-binding proteins of Rho family [24]–[26]. Ours and other studies have demonstrated that the activation of Gα12 signaling plays a critical role in tumor progression and metastasis of nasopharyngeal carcinoma (NPC) as well as several other human cancers [27]–[29]. However, whether such a relationship also exists in OSCC tumorigenesis has not yet been reported.
Activation of Gα12 is known to contribute to inflammatory responses. For instance, Gα12 has been shown to mediate the sphingosine 1-phosphate (S1P) induction of cyclooxygenase-2 (COX-2) for the activation of nuclear factor-κB (NF-κB) [30]. Thus, this study was designed to investigate whether Gα12 modulates inflammatory responses in promoting tumor invasion in OSCC. Here, we demonstrate that Gα12 exerts an impact on proinflammatory cytokine signaling, which in turn contributes to OSCC invasiveness.
Materials and Methods
Clinical Samples
All participants provided written informed consent and the study, which was performed in adherence with the Declaration of Helsinki, has been approved by the Institutional Review Board at Chang Gung Memorial Hospital.
Microarray Analysis
The microarray gene expression data of OSCC tumor samples were retrieved from our previous published transcriptome profiling data of the Affymetrix Exon 1.0 ST array for 57 OSCC tumors and 22 non-cancerous controls [31] (Gene Expression Omnibus database under the accession number GSE25104). For the correlation analysis of gene expression levels and clinical features, we excluded two tumor samples and one control sample because their clinical information was incomplete. The cluster display was generated by Partek software (Partek Inc., Saint Louis, USA) with two-way data clustering. Each row and column represents an individual gene and sample, respectively. Normalized gene expression values were color coded in percentage relative to the mean: blue for values less than the mean and red for values greater than the mean. The association of the expression data with clinicopathological traits, including the presence of ECS (extra-capsular spread), tumor differentiation, pathologic T-status (pathological tumor status), N-status (pathological nodal status), pathological stage, tumor depth and lymphatic invasion, was analyzed by t-test. T-status, N-status and pathological stage of the tumors were determined according to the American Joint Committee on Cancer (AJCC). The relative gene expression level was determined using Robust Multiarray Average (RMA), a normalization approach used for normalizing Affymetrix data. The statistical analysis was conducted using R (www.r-project.org) and the SPSS software package 15.0 for Windows (SPSS Inc., Chicago, IL, USA).
To investigate the putative pathways regulated by Gα12 in OSCC, Affymetrix Exon 1.0 ST array was used to analyze the transcriptome profile of Gα12-depleted OSCC cells. OC-3 and HSC-3 cells transiently transfected with siRNA against Gα12 were harvested at 48 h post-transfection. Total RNA was isolated using Qiagen RNeasy Mini Kit (Qiagen, USA) for the array analysis according to the manufacture's standard protocol (Affymetrix, USA). The cluster display and Gene Ontology (GO) enrichment analysis were performed with Partek software (Partek Inc., Saint Louis, USA). The array data have been submitted to Gene Expression Omnibus database and are publically available under the accession number GSE44111.
Cell Culture
Four human OSCC cell lines, HSC-3, SCC25, OC-3 and CGHNC9 were used in this study. HSC-3 (JCRB0623) cells were originally obtained from the JCRB cell bank (Osaka, Japan). SCC25 (ATCC® CRL-1628™) cell line was purchased from Food Industry Research and Development Institute in Taiwan. OC-3 and CGHNC9 cells were originally established in Taiwan and authenticated using the experiments described before [32], [33].
HSC-3 was cultured in MEM (Invitrogen, USA), SCC25 in 1∶1 DMEM/F12 (Invitrogen, USA), OC-3 in 1∶1 DMEM/KSFM (keratinocyte serum-free medium) (Invitrogen, USA) and CGHNC9 in DMEM (Invitrogen, USA). All culture media were supplemented with 10% fetal bovine serum (Invitrogen, USA) and 50 units/ml penicillin and streptomycin (Invitrogen, USA). Cells were maintained in a 37°C incubator with 5% CO2.
Cell Transfection
To knock down Gα12, OSCC cells were seeded at a density of 5×104 per well in a 24-well culture plate 24 h before transfection with Gα12-siRNA (siGα12) or a non-targeting control siRNA (siCtrl) at a concentration of 50 nmol/L using DharmaFECT transfection reagents (Dharmacon, USA). To overexpress Gα12, OSCC cells were transiently transfected with Gα12 expression plasmids (pcDNA3-Gα12) or a mock control plasmid (pcDNA3) using Lipofectamin 2000 transfection reagents (Invitrogen, USA). The pcDNA3-Gα12 plasmid was obtained from the Missouri S&T cDNA Resource Center as previously described [27]. For functional assays, cells were collected at 1–3 days post-transfection.
Quantitative real-time Reverse Transcription-PCR (qPCR)
For reverse transcription of cellular mRNA to cDNA, an input of 2 µg of total RNA was used for the High-capacity cDNA Reverse transcription Kits (ABI Applied Biosystems, USA) according to the manufacturer's instructions. qPCR was performed on a Step One Real-time PCR system (ABI Applied Biosystems, USA) by using the KAPA SYBR FAST qPCR Kits (KAPA Biosystems, USA). The primer sequences of Gα12 used for qPCR were: forward-GTTTGTCGTCGTTGAGC, reverse-AGTAGTTTCACTCGCCC; for IL-8: forward-GGAGTGCTAAAGAACTTAGATG, reverse-TGGGGTCCAGACAGAG; for IL-6: forward-CAAAGATGTAGCCGCCC, reverse-GTTCAGGTTGTTTTCTGCC; for GAPDH: forward-CCTGCCAAATATGATGACATCAAG, reverse-ACCCTGTTGCTGTAGCCAAA.
Total RNA was purified using the Illustra RNAspin Mini RNA Isolation Kits (GE healthcare, USA) according to the manufacturer's instructions. The amount of target transcript was estimated by the respective standard curves and normalized to the amount of GAPDH transcript. The results were expressed as a relative fold change to the control. For semi-quantitative RT-PCR, the number of cycles for IL-6 and IL-8 was individually optimized. PCR products were analyzed on a 2% agarose gel.
Immunohistochemistry
To determine the protein expression level of Gα12, immunohistochemical staining of OSCC tissue sections was conducted as described previously [27] using anti-Gα12 primary antibody (1∶75; sc-409; Santa Cruz Biotechnology). The images of Gα12 staining were captured by a Leica DM2500 Upright Fluorescence Microscope at 20× objective.
Western Blotting
The harvested cells were washed twice with cold PBS buffer and lysed as described previously [27]. The total protein concentration was measured using the Bio-Rad Protein Assay (Bio-Rad, USA). Equal amounts of cell lysates were separated by SDS-PAGE and transferred to polyvinylidene fluoride membranes. The membrane was blocked and blotted with indicated primary antibodies: anti-Gα12 (Santa Cruz Biotechnology, USA), anti-GAPDH (glyceraldehyde-3-phosphate dehydrogenase) (Lab Frontier, USA). After washing, the membrane was incubated with HRP–conjugated secondary antibody (Jackson ImmunoResearch Laboratories, USA) and developed with enhanced chemiluminescence detection reagents (PerkinElmer, USA).
Migration and Matrigel Invasion Assay
OSCC cells transfected with indicated expression plasmids or siRNAs were harvested at 48 h post-transfection. Invasion capacity was analyzed in a Boyden chamber consisting of cell culture inserts with an 8 µm pore-sized PET membrane coated with matrigel (BD Biosciences, USA) according to the manufacturer's instructions. For the cell migration assay, the chamber without matrigel coating was used. After seeding cells into the chamber, cells were incubated for 16 h or 24 h at 37°C for migration or invasion, respectively. The recombinant human IL-6 was obtained from PeproTech (Rocky Hill, NJ). The neutralizing IL-6 and IL-8 antibodies and recombinant human IL-8 were obtained from R&D Systems (Minneapolis, MN). Migrated or invaded cells were stained with 0.1% crystal violet in 1% formaldehyde and 20% ethanol. The number of invaded cells was counted at least in five distinct fields for each duplicate chamber. The results were expressed as a relative percentage to control cells.
IL-6 and IL-8 ELISA
OSCC cells were seeded into 12-well plates (1×105 cells/well) and cultured overnight prior to the transfection of siRNA or plasmid DNA. At 24 h post-transfection, cells were transferred to serum-free medium for 24 h before being assayed for IL-6 and IL-8 protein levels in medium by ELISA. For the LPA treatment, cells at 24 h post-transfection were incubated in 10 µM LPA-containing (Sigma-Aldrich, USA) serum-depleted medium for 12 h. The conditioned media were centrifuged to remove cell debris for ELISA assays using DuoSet ELISA Development kit (R&D Systems, USA) according to the manufacturer's protocols. The quantification data were expressed as a relative fold change to control group.
Results
Gα12 is Significantly Up-regulated in OSCC Patients and Correlates with Extra-capsular Spread
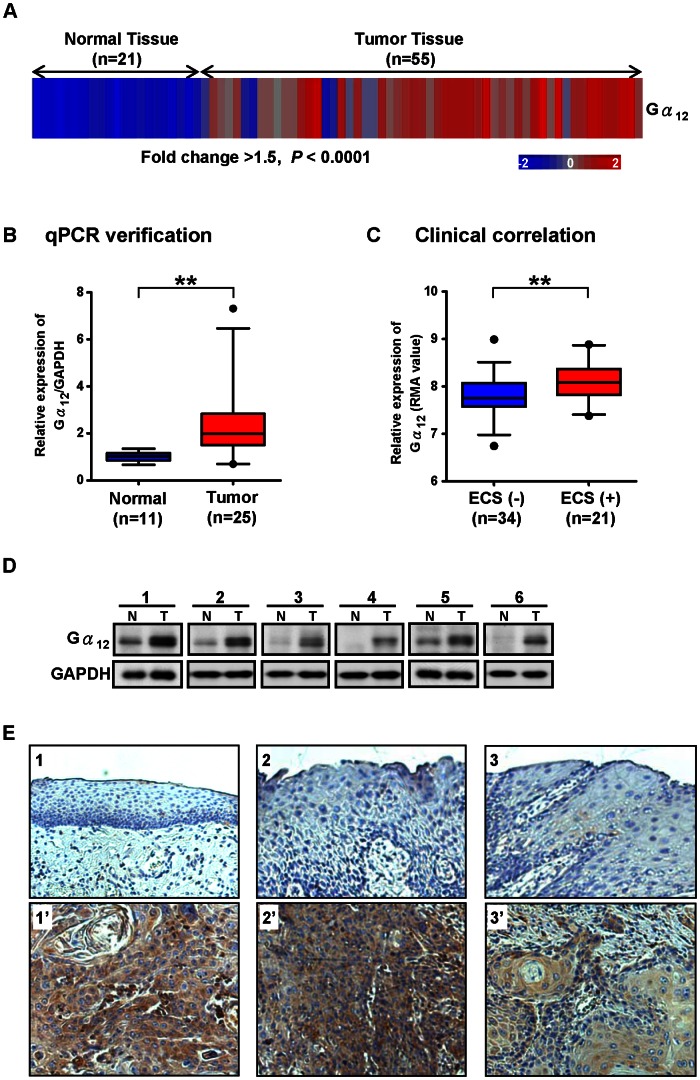
Our previous study of NPC has shown that Gα12 gene expression is up-regulated in tumor cells, and is also important in facilitating tumor invasiveness [27]. Thus, it was of interest to investigate whether Gα12 also played a role in the tumorigenesis of OSCC, a specific subtype of HNSCC. By analyzing our previous transcriptome profiling data for OSCC [31] (Gene Expression Omnibus database under the accession number GSE25104), we characterized the gene expression pattern of Gα12 in OSCC and normal mucosa tissues. The array results showed that Gα12 was significantly up-regulated in 55 OSCC tissues compared to 21 normal controls (P<10−10 with fold change cutoff of >1.5) (Figure 1A). To validate the array results, we performed quantitative RT-PCR (qPCR) analysis on 25 OSCC and 11 normal mucosa tissues. As suspected, Gα12 was significantly up-regulated in OSCC tissues compared to controls (P = 0.0036) (Figure 1B). Because the elevation of Gα12 expression is highly associated with tumor invasiveness in several human cancers [27]–[29], we examined the correlation between the Gα12 transcription level and clinicopathological characteristics, including tumor differentiation, pathologic T-status, N-status, pathological stage, tumor depth, ECS, and lymphatic invasion in 55 OSCC patients. We found evidence suggesting a significant correlation between higher Gα12 levels and ECS (P = 0.009) (Figure 1C). There appeared to be no discernible correlation between Gα12 and the other clinicopathological characteristics (data not shown). Since ECS is a known prognostic factor of tumor aggressiveness in HNSCC, including high loco-regional recurrence rates, distant metastasis, and poor prognosis [34]–[37], the correlation of Gα12 levels with ECS may suggest a role of Gα12 in OSCC invasiveness. To determine whether the Gα12 protein levels were also elevated in OSCC tumors as shown in its gene expression, we measured the Gα12 protein level in OSCC tumors by Western blotting and immunohistochemistry. Western blot analysis of OSCC tumor tissues revealed that Gα12 was markedly up-regulated compared to controls (Figure 1D). The results of immunohistochemical analysis also confirmed that of Western blot analysis. The representative micrographs of Gα12 staining showed clear immunoreactivity of Gα12 in most OSCC tumors (18 of the 20 cases, 90%) but the signal was either absent or weak in adjacent normal or precancerous tissues (Figure 1E). These results suggest that Gα12 expressions are up-regulated in OSCC tumors, and may be associated with tumor invasiveness.
Figure 1. The up-regulation of Gα12 in OSCC patients correlates with Extra-capsular spread.
(A) The Gα12 expression is significantly up-regulated in 55 OSCC tumors compared to 21 normal control tissues (fold change >1.5, P<10−10). The microarray data was analyzed by two-way clustering. Each column represents an individual clinical sample. Normalized gene expression values were color coded in percentage relative to the mean: blue for values less than the mean and red for values greater than the mean. (B) Quantitative RT-PCR (qPCR) analysis of Gα12 in 25 OSCC tumors compared to 11 normal mucosa tissues. The results were normalized to GAPDH expression levels and then analyzed by t-test, **P<0.01. Box plots display the median, 25th and 75th percentiles. Whiskers represent 5–95 percentiles and dots the outliers. (C) The box plot shows the relative gene expression values (RMA, log2) of Gα12 for extra-capsular spread (ECS) positive (+) and negative (−) patients. Statistical results were analyzed by t-test, **P<0.01. (D) Western blot analysis of Gα12 levels in 6 paired samples of OSCC and adjacent normal/pre-cancerous tissues. The Gα12 protein levels were found to be markedly up-regulated in OSCC tumor tissues compared to the GAPDH loading control. (E) Representative immunohistochemical images for Gα12 staining patterns in the paraffin-embedded section of OSCC biopsies. Gα12 immunoreactivity was detected primarily in the membrane and cytoplasm of OSCC (lower panel). In contrast, the adjacent normal and pre-cancerous oral tissues of individual patients showed very low immunoreactivity (upper panel). Original magnification, ×200.
Gα12 Modulates Cell Migration and Invasion Abilities of OSCC Cells
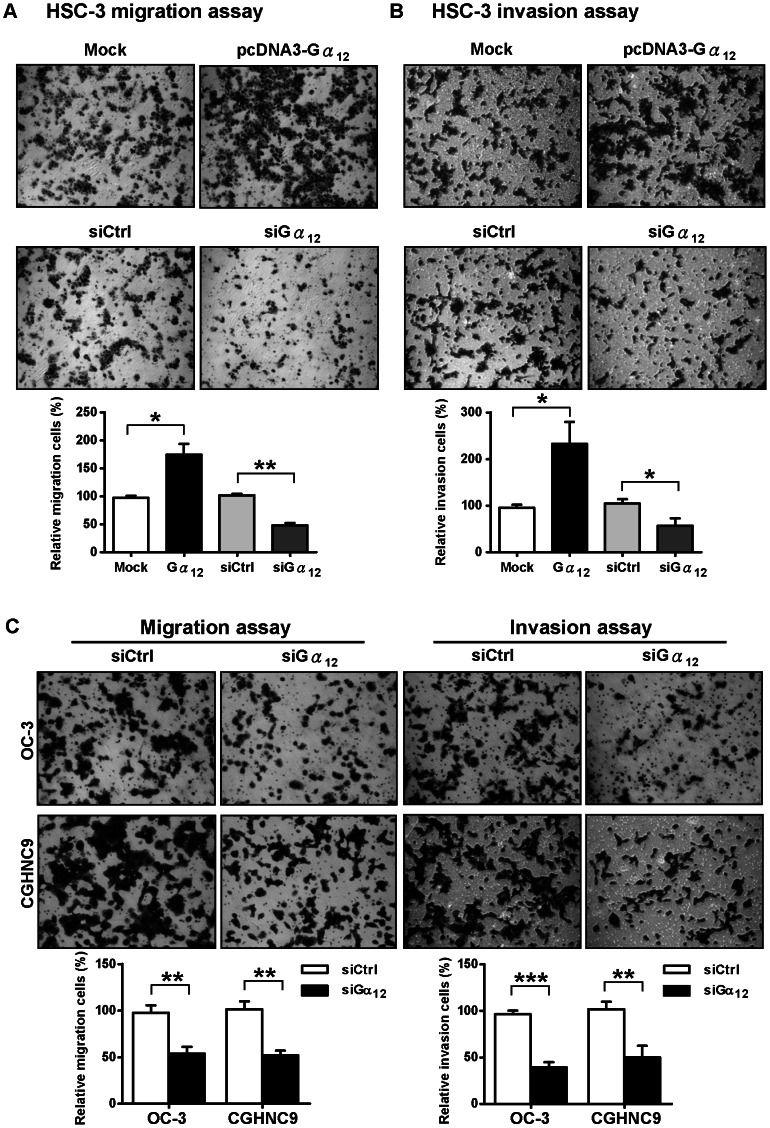
To validate the role of Gα12 in facilitating the invasive behavior of OSCC cells, we conducted transwell migration and invasion assays in cells overexpressing or depleted Gα12. The knockdown efficiency and overexpression level of Gα12 in these cell lines were demonstrated in Figure S1. As expected, the cell migration and invasion ability of OSCC tumor cells (HSC-3) was significantly increased by Gα12 overexpression and decreased by RNAi knockdown (Figure 2A and B). To further validate these results, we also depleted Gα12 by siRNA in two other OSCC cell lines (OC-3, and CGHNC9) and determined the effect on cell invasiveness. Results showed that the depletion of Gα12 also suppressed tumor cell migration and invasion ability in these two other OSCC cell lines (Figure 2C). Taken together, these data support the idea that Gα12 promotes the invasive behavior of OSCC cells.
Figure 2. Gα12 promotes OSCC cell migration and invasion.
(A) The transwell migration assay of Gα12 overexpressed (Gα12), or Gα12 depleted (siGα12) HSC-3 cells stained with crystal violet. The lower panel shows the quantitative results by three independent experiments. (B) The transwell invasion assay of Gα12 overexpressed (Gα12), or Gα12 depleted (siGα12) HSC-3 cells. The lower panel shows the quantitative results by three independent experiments. (C) Depletion of Gα12 in two other OSCC cell lines (OC-3 and CGHNC9) also decreased cell migration and invasion. The knockdown efficiency and overexpression level of Gα12 in four different OSCC cell lines used in this study are demonstrated in Figure S1. The bottom panel shows the quantitative results. All the quantitative values were calculated at least in five distinct fields of each chamber. The data are expressed as a relative percentage to the controls. The statistic results were analyzed by t-test, *P<0.05, **P<0.01, ***P<0.001. Error bars represent the standard deviation (SD) of the mean from three independent experiments.
Comparative Transcriptome Analysis Reveals the Enrichment of Immune-related Pathways in the Gα12-depleted OSCC Cells
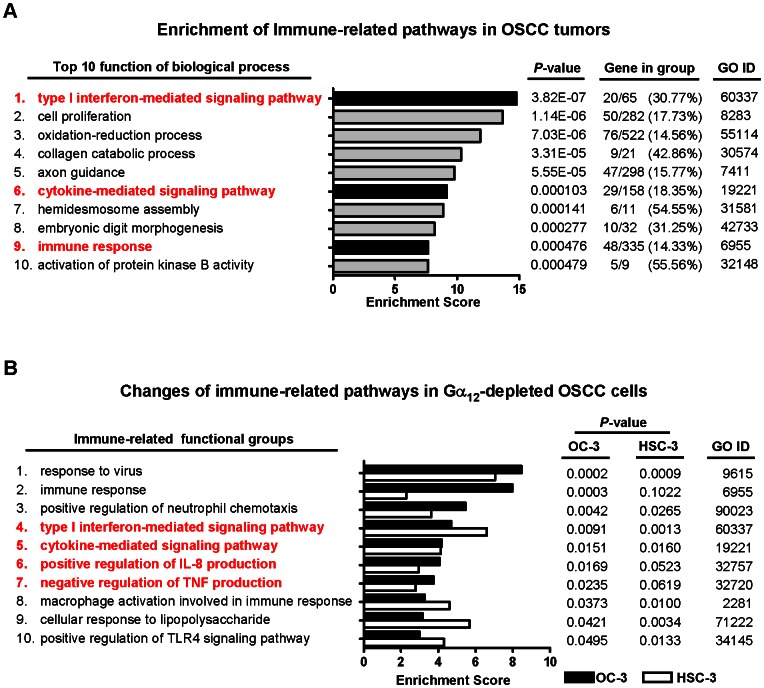
Several reports have suggested that the immune-related genes may serve as potential biomarkers or therapeutic targets for OSCC [6], [12], [19], but the underlying mechanisms still remained to be further investigated. We conducted gene ontology (GO) analysis for our previous transcriptome data of OSCC tumors to better understand the biological characteristics of the differentially expressed genes in OSCC. As noted by previous studies [12], [19], [38], cytokine and other immune-related signaling pathways were found to be highly enriched in the top ten GO terms (Figure 3A). Although GPCR signaling is important for the inflammatory response and invasiveness of cancers, it remains to be defined whether Gα12 is required for the inflammation-associated tumorigenesis in OSCC. To test this hypothesis, we performed microarray analysis for the Gα12-depleted HSC-3 and OC-3 OSCC cells. The differentially expressed genes were selected by an arbitrary fold-change cut-off of 2.0 for GO enrichment analysis. Similar to that found in OSCC clinical samples, comparative transcriptome analysis revealed that many immune-related functional groups, including cytokine and interferon-mediated pathways, were significantly changed in both cell lines (Figure 3B; for detail GO terms see Table S1), suggesting that Gα12 may play a critical role in modulating inflammatory cytokine responses during OSCC tumorigenesis.
Figure 3. Transcriptome analysis reveals changes of immune-related pathways in OSCC and in Gα12-depleted OSCC cell lines.
(A) Comparative transcriptome analysis of OSCC tumors reveals that cytokine and other immune-related functional groups are listed in the top ten GO terms. A total of 1,616 differently expressed genes selected by 1.5 fold change cut-off (positive false discovery rate q<10−8) in 55 OSCC tumors compared to 21 normal control tissues were analyzed by GO and pathway analysis tools. Functional groups of the inflammation-related pathways are highlight in red. (B) The immune-related signaling pathways are significantly impaired in the Gα12-depleted OC-3 and HSC-3 cell lines. An arbitrary 2.0 fold-change cut-off is used to filter the differentially expressed genes compared between Gα12-depleted and non-targeted siRNA control cells for the GO enrichment analysis. A total of 58 genes for HSC-3 cells and 218 genes for OC-3 cells were subjected to the analysis. The cytokine and interferon-mediated pathways (highlighted in red) were found in the GO terms for both cell lines. Detailed information of the GO terms is shown in Table S1.
Gα12 Correlates with IL-6 and IL-8 Expression Levels in OSCC Patients and Stimulates IL-6 and IL-8 Expressions in OSCC cell Lines
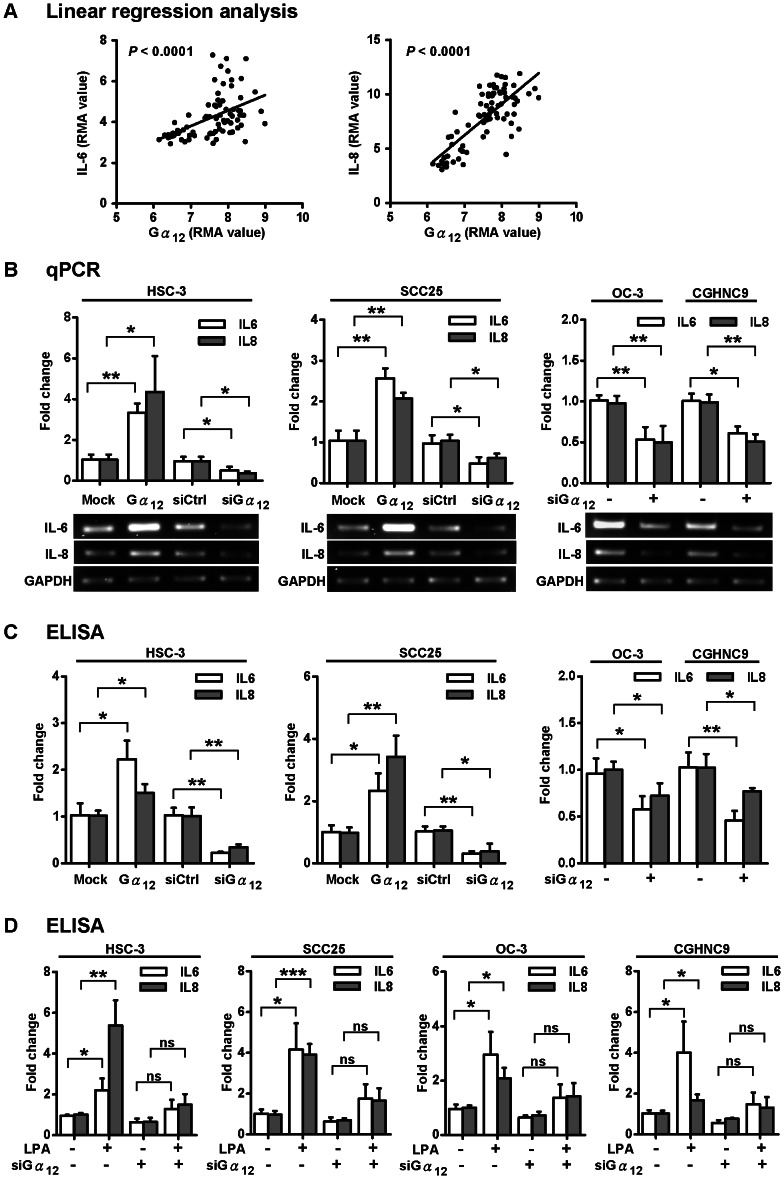
Since our transcriptome analysis of OSCC suggested that IL-6 and IL-8 cytokines were significantly up-regulated in OSCC patients (P<0.0001, with fold change 2.5 and 16.1 respectively), we hypothesized that these cytokine responses were associated with the up-regulation of Gα12. To test this idea, we analyzed the microarray data for the differential expression of genes that correlated with increased Gα12 levels in OSCC patients. Indeed, the expression levels of IL-6 and IL-8 were significantly correlated with the levels of Gα12 in a total of 76 OSCC and normal tissues (P<0.0001) (Figure 4A), suggesting that Gα12 might associate with the production of IL-6 and IL-8 in OSCC. To test this hypothesis, we investigated whether altering the Gα12 levels by gene silencing or transient overexpression would affect the IL-6 and IL-8 expression levels in OSCC cells. Results from studies in HSC3 and SCC25 cell lines demonstrated that the IL-6 and IL-8 levels were increased and decreased by the overexpression and depletion of Gα12, respectively (Figure 4B, left and middle panels). Similarly, we depleted the Gα12 expressions in OC3 and CGHNC9 cell lines and found that both the IL-6 and IL-8 gene expression levels were significantly decreased (Figure 4B, right panel). Additionally, we examined whether secreted cytokines in the culture supernatants were also affected in accordance with their mRNA expressions by the change of Gα12 levels. The ELISA assays of secreted cytokines from HSC-3 and SCC25 cells showed that the IL-6 and IL-8 protein levels were also increased and decreased by the overexpression and RNAi knockdown of Gα12, respectively (Figure 4C, left and middle panels). Depletion of Gα12 in OC3 and CGHNC9 cell lines (with high baseline levels of IL-6 and IL-8; see Figure S2) resulted in the decrease of IL-6 and IL-8 protein levels (Figure 4C, right panel). These results suggest that Gα12 is a critical modulator in stimulating the production of IL-6 and IL-8 in OSCC cells. To further substantiate this view, we treated the OSCC cells with lysophosphatidic acid (LPA) to investigate the role of Gα12 in modulation of IL-6 and IL-8 responses. LPA has been previously shown to promote cell proliferation and migration via stimulation of Gα12 signaling [39], [40] and can also elicit IL-6 and IL-8 production in various human cell types [41]–[43]. Indeed, our results showed that LPA stimulated IL-6 and IL-8 secretion into culture media from four different OSCC cell lines (HSC3, SCC25, OC3, and CGHNC9) but RNAi depletion of Gα12 suppressed the LPA-induced cytokine expressions (Figure 4D). Together, these results strongly suggest that the up-regulated Gα12 is a critical stimulus for the pro-inflammatory cytokine responses in OSCC.
Figure 4. Gα12-dependent regulation of IL-6 and IL-8 in OSCC.
(A) Dot plots of the linear regression analysis showing a positive correlation of gene expressions between Gα12 and IL-6/IL-8 in OSCC tumors and normal mucosa tissues. The relative expression scales are shown by RMA value in the microarray data. (B) IL-6 and IL-8 mRNA levels are positively regulated by Gα12 in OSCC cells. Gα12 levels in four different OSCC cell lines (HSC-3, SCC25, OC3, and CGHNC9) were altered by the transient overexpression or RNAi knockdown of Gα12. The qPCR results were normalized against GAPDH. The lower panel shows the electrophoresis image of the RT-PCR products. (C) The secreted proteins of IL-6 and IL-8 are up-regulated by Gα12 in OSCC cells. ELISA assay was used to measure IL-6 and IL-8 in the conditioned media of the Gα12-overexpressing or -depleted HSC-3, SCC25, OC-3, and CGHNC9 cells. (D) The LPA-induced IL-6 and IL-8 production is regulated by Gα12. IL-6 and IL-8 protein levels in conditioned media of OSCC cells were measured by ELISA assay. The baseline IL-6 and IL-8 levels in conditioned media from four different OSCC cell lines are shown in Figure S2. All the quantitative results are expressed as a fold change relative to the controls. The statistical results were analyzed by t-test, *P<0.05, **P<0.01, ***P<0.001. “ns” means no significance. Error bars represent SD of the mean from three independent experiments.
The role of Gα12 in the Stimulation of Cell Invasiveness Requires IL-6 and IL-8 in OSCC Cells
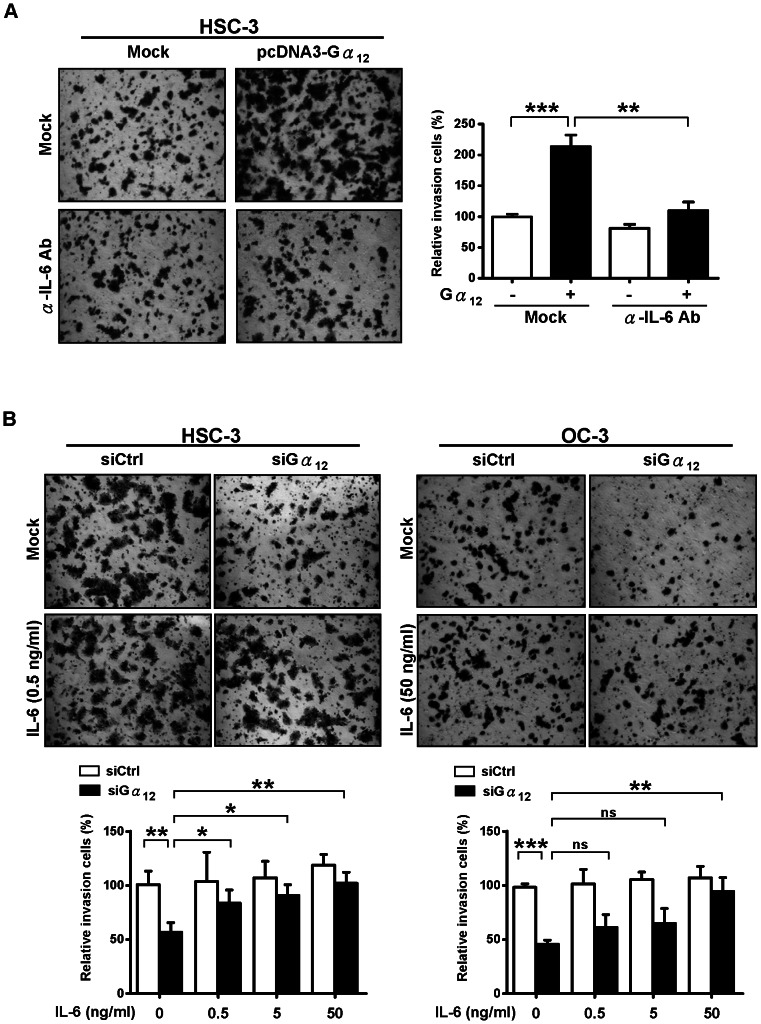
Although we have demonstrated the role of Gα12 in promotion of OSCC cell invasive behavior and pro-inflammatory cytokines expressions, we have not yet established the functional link between the Gα12-dependent cytokine response and cell invasiveness. To test whether the induction of proinflammatory cytokines was required for the promotion of tumor cell invasion, we neutralized the endogenous IL-6 in the Gα12-overexpressed HSC-3 cells and examined the tumor cell invasiveness via transwell invasion assays. Results showed that the anti-human IL-6 antibody, as compared to IgG control, significantly suppressed the cell invasion (about 80%) in cells transiently overexpressed with Gα12 (Figure 5A). To further test this idea, we first decreased the tumor cell invasiveness by depleting the endogenous Gα12, and then introduced recombinant IL-6 into the culture medium to determine if the cell invasiveness could be resumed by IL-6. The transwell invasion assays in HSC-3 and OC-3 cells suggested that the tumor cell invasiveness was restored by IL-6 (Figure 5B).
Figure 5. Gα12 promotes OSCC cell invasive behavior via the regulation of IL-6.
(A) The Gα12-induced cell invasion of HSC-3 is suppressed by the treatment of cells with anti-IL-6 antibody. Representative images of the transwell invasion assays of the Gα12-overexpressing HSC-3 cells treated with or without anti-IL-6 antibody for 24 h (left panel). Quantification of invasion is shown in the right panel. (B) Supplementation of culture media with IL-6 restores cell invasion in Gα12-depleted cells. The Gα12-depleted HSC-3 and OC-3 cell lines were treated with or without three different concentrations of recombinant human IL-6 (0.5 ng/ml, 5 ng/ml, and 50 ng/ml) for 24 h prior to transwell invasion assays. The quantitative results of three independent experiments were analyzed by t-test. *P<0.05, **P<0.01, ***P<0.001. “ns” means no significance. Error bars represent SD of the mean from three independent experiments.
Moreover, we also tested whether antibody neutralization of IL-8 suppresses the cell invasiveness enhanced by overexpression of Gα12. By using the anti-IL-8 antibody to neutralize IL-8 in HSC-3 and SCC25 cells overexpressing Gα12, we examined if the migration ability of the tumor cells was decreased. The results showed that the cell migration ability was significantly diminished by neutralizing the endogenous IL-8 in both HSC-3 and SCC25 cells (Figure S3), suggesting that IL-8 functions, similar to IL-6, as an important downstream effector of Gα12 signaling in regulating OSCC cell invasive behavior. To further substantiate this idea, we tested whether supplementation of recombinant IL-8 restores the migration ability decreased by the depletion of Gα12 in OSCC cells. As expected, the decreased migration ability of HSC-3 cells was restored by the recombinant IL-8 (Figure S4). Together, these results indicate that the Gα12 induction of cell invasiveness is regulated by the proinflammatory cytokines IL-6 and IL-8 in OSCC cells.
Discussion
Dysregulation of inflammatory pathways during tumorigenesis contributes to the invasive characteristics of cancer; therefore the associated mediators of these pathways become favorable targets in the search for new pharmacological medications. As in many other cancers, the development and invasiveness of OSCC is intimately related to chronic inflammation [6]–[9]. However, the signaling cascade that leads to the proinflammatory response has been poorly understood. In this study, we found evidence to support that the increase of Gα12 is an important stimulator for the production of proinflammatory cytokines IL-6 and IL-8 and tumor invasiveness in OSCC. We further showed that the Gα12-induced cell invasiveness was mediated through the regulation of IL-6 and IL-8 in OSCC.
Gα12 is known to regulate several cellular processes through the modulation of transcription factors, including NF-κB and AP-1 [23], [44]. For instance, it has been demonstrated that the coupling of Gα12 to S1P receptors induces COX-2 expression via NF-κB [30]. Since the S1P producing enzyme-sphingosine kinase 1 (SPHK1) has been reported to be up-regulated in HNSCC [45], it is possible that the S1P-NF-κB signaling might modulate the Gα12 induction of cytokine responses. Supporting this idea, our preliminary data showed that the S1P treatment of OSCC cells induced IL-6 and IL-8 expressions, but the RNAi-mediated depletion of Gα12 abolished the S1P induction of IL-6 and IL-8 (unpublished data). Future work will aim to test whether disruption of NF-κB diminishes the Gα12-induced cytokine expressions. The possible GPCRs that couple Gα12 in stimulating IL-6/IL-8 production in OSCC tumors also remain to be identified in the future.
In addition to the role in promoting tumor cell invasiveness, we speculate that the Gα12 induction of IL-6 and IL-8 production might also be involved in the drug resistance of OSCC, because several cytokines, including IL-6 and IL-8, are known to contribute to drug resistance in several different cancer types [46]–[48]. Drug resistance remains a critical hurdle for the success of cancer chemotherapy. Although chemotherapy is currently the primary treatment for both resectable and advanced OSCC patients, it has been reported to only marginally increase the survival rate for those patients [2], [3], [49]. Thus, it is important to investigate whether Gα12 signaling is linked to the drug resistance of OSCC. We are currently assessing if Gα12 could be a potential biomarker for the prediction of anti-cancer drug resistance by analyzing the correlation of Gα12 to the patients' responses to chemotherapy.
Another issue concerns the complex interplay between Gα12 and inflammatory cytokines. Although our data strongly suggests that Gα12 promotes inflammatory cytokines production in OSCC, we cannot exclude the possibility that the interplay is bidirectional or reciprocal because proinflammatory cytokines have been shown to transactivate S1P receptors via stimulation of S1P production, leading to the activation of G protein signaling [50]. Thus, it will be of interest in the future to investigate whether the increased cytokines inversely modulate the Gα12 signaling in OSCC.
Currently, due to the high invasiveness of OSCC, very few promising pharmacologic therapeutics have been developed and backed by clinical trials. Because Gα12 is involved in the inflammatory invasion of OSCC, we believe it can potentially become a useful therapeutic target or biomarker for the invasive OSCC, though further clinicopathological delineation of the association of Gα12 and IL-6/IL-8 functions are needed before Gα12 becomes an eligible prognostic marker for this disease.
Supporting Information
List of immune-related functional groups impaired in the Gα12-depleted OSCC cells.
(TIF)
Western blot analysis of siRNA knockdown efficiencies and overexpression levels of Gα12 in four different OSCC cell lines (HSC-3, SCC25, OC-3, and CGHNC9).
(TIF)
Baseline levels of IL-6 and IL-8 secreted in conditioned media from four different OSCC cell lines (HSC-3, SCC25, OC-3, and CGHNC9). Quantitative measurements of IL-6 and IL-8 were determined by ELISA assays.
(TIF)
IL-8 neutralizing antibody abolishes the Gα12-induced OSCC cell migration. (A), (B) Transwell migration assays of the Gα12-overexpressing HSC-3 and SCC25 cells treated with neutralizing antibody (10 µg/ml) against IL-8 for 16 h. Quantification of migration is shown in the right panel. Error bars represent SD of the mean from three independent experiments and analyzed by t-test. *P<0.05, **P<0.01.
(TIF)
Recombinant IL-8 restores the migration ability reduced by transiently depleted Gα12 in HSC-3 cells. Representative images show the migration of the Gα12-depleted HSC-3 cells through transwells. Cells treated with or without recombinant human IL-8 (1 ng/ml) for 16 h. Quantification of migration is shown in the right panel. Error bars represent SD of the mean from three independent experiments and analyzed by t-test. *P<0.05.
(TIF)
Acknowledgments
We thank the National Health Research Institutes Pathology Core for Gα12 immunohistochemistry staining, Dr. Lu-Hai Wang for critical comments on the article and James Steed and Duane Juang for English editing assistance.
Funding Statement
Funding was provided by National Health Research Institutes (http://www.nhri.org.tw/NHRI_WEB/nhriw001Action.do), Grant number: 01A1-MGPP02-014. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Ramqvist T, Dalianis T (2010) Oropharyngeal cancer epidemic and human papillomavirus. Emerging infectious diseases 16: 1671–1677. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Leemans CR, Braakhuis BJM, Brakenhoff R (2011) The molecular biology of head and neck cancer. Nature Reviews Cancer 11: 9–22. [DOI] [PubMed] [Google Scholar]
- 3. Pai SI, Westra WH (2009) Molecular pathology of head and neck cancer: implications for diagnosis, prognosis, and treatment. Annu Rev Pathol 4: 49–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Chen Y-J, Chang J, Liao C-T, Wang H-M, Yen T-C, et al. (2008) Head and neck cancer in the betel quid chewing area: recent advances in molecular carcinogenesis. Cancer Sci 99: 1507–1514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Chung C, Gillison M (2009) Human papillomavirus in head and neck cancer: its role in pathogenesis and clinical implications. Clinical cancer research 15: 6758–6762. [DOI] [PubMed] [Google Scholar]
- 6. Wang F, Arun P, Friedman J, Chen Z, Van Waes C (2009) Current and potential inflammation targeted therapies in head and neck cancer. Curr Opin Pharmacol 9: 389–395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Mantovani A, Allavena P, Sica A, Balkwill F (2008) Cancer-related inflammation. Nature 454: 436–444. [DOI] [PubMed] [Google Scholar]
- 8. Chen Z, Malhotra PS, Thomas GR, Ondrey FG, Duffey DC, et al. (1999) Expression of proinflammatory and proangiogenic cytokines in patients with head and neck cancer. Clinical cancer research 5: 1369–1379. [PubMed] [Google Scholar]
- 9. Pries R, Nitsch S, Wollenberg B (2006) Role of cytokines in head and neck squamous cell carcinoma. Expert review of anticancer therapy 6: 1195–1203. [DOI] [PubMed] [Google Scholar]
- 10. St John MAR, Li Y, Zhou X, Denny P, Ho C-M, et al. (2004) Interleukin 6 and interleukin 8 as potential biomarkers for oral cavity and oropharyngeal squamous cell carcinoma. Arch Otolaryngol Head Neck Surg 130: 929–935. [DOI] [PubMed] [Google Scholar]
- 11. Duffy S, Taylor JMG, Terrell J, Islam M, Li Y, et al. (2008) Interleukin-6 predicts recurrence and survival among head and neck cancer patients. Cancer 113: 750–757. [DOI] [PubMed] [Google Scholar]
- 12. Rao SK, Pavicevic Z, Du Z, Kim JG, Fan M, et al. (2010) Pro-inflammatory genes as biomarkers and therapeutic targets in oral squamous cell carcinoma. J Biol Chem 285: 32512–32521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Chen C-J, Sung W-W, Lin Y-M, Chen M-K, Lee C-H, et al. (2012) Gender difference in the prognostic role of interleukin 6 in oral squamous cell carcinoma. PLoS ONE 7: e50104–e50104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Culig Z (2013) Interleukin-6 as a therapy target in oral squamous carcinoma. Expert opinion on therapeutic targets 17: 53–59. [DOI] [PubMed] [Google Scholar]
- 15. Gokhale A, Haddad R, Cavacini L, Wirth L, Weeks L, et al. (2005) Serum concentrations of interleukin-8, vascular endothelial growth factor, and epidermal growth factor receptor in patients with squamous cell cancer of the head and neck. Oral Oncol 41: 70–76. [DOI] [PubMed] [Google Scholar]
- 16. Riedel F, Zaiss I, Herzog D, GÃtte K, Naim R, et al. (2005) Serum levels of interleukin-6 in patients with primary head and neck squamous cell carcinoma. Anticancer research 25: 2761–2765. [PubMed] [Google Scholar]
- 17. SahebJamee M, Eslami M, AtarbashiMoghadam F, Sarafnejad A (2008) Salivary concentration of TNFalpha, IL1 alpha, IL6, and IL8 in oral squamous cell carcinoma. Medicina oral, patología oral y cirugía bucal 13: E292–E295. [PubMed] [Google Scholar]
- 18. de Oliveira MV, Fraga CA, Gomez RS, Paula AM (2009) Immunohistochemical expression of interleukin-4, -6, -8, and -12 in inflammatory cells in surrounding invasive front of oral squamous cell carcinoma. Head Neck 31: 1439–1446. [DOI] [PubMed] [Google Scholar]
- 19. Wu JY, Yi C, Chung HR, Wang DJ, Chang WC, et al. (2010) Potential biomarkers in saliva for oral squamous cell carcinoma. Oral Oncol 46: 226–231. [DOI] [PubMed] [Google Scholar]
- 20. Dorsam RT, Gutkind JS (2007) G-protein-coupled receptors and cancer. Nat Rev Cancer 7: 79–94. [DOI] [PubMed] [Google Scholar]
- 21. Li S, Huang S, Peng S-B (2005) Overexpression of G protein-coupled receptors in cancer cells: involvement in tumor progression. Int J Oncol 27: 1329–1339. [PubMed] [Google Scholar]
- 22. Spiegelberg B, Hamm H (2007) Roles of G-protein-coupled receptor signaling in cancer biology and gene transcription. Current opinion in genetics & development 17: 40–44. [DOI] [PubMed] [Google Scholar]
- 23. Fraser C (2008) G protein-coupled receptor connectivity to NF-kappaB in inflammation and cancer. International reviews of immunology 27: 320–350. [DOI] [PubMed] [Google Scholar]
- 24. Neves SR, Ram PT, Iyengar R (2002) G protein pathways. Science 296: 1636–1639. [DOI] [PubMed] [Google Scholar]
- 25. Cotton M, Claing A (2009) G protein-coupled receptors stimulation and the control of cell migration. Cellular signalling 21: 1045–1053. [DOI] [PubMed] [Google Scholar]
- 26. Juneja J, Casey P (2009) Role of G12 proteins in oncogenesis and metastasis. British Journal of Pharmacology 158: 32–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Liu SC, Jen YM, Jiang SS, Chang JL, Hsiung CA, et al. (2009) G(alpha)12-mediated pathway promotes invasiveness of nasopharyngeal carcinoma by modulating actin cytoskeleton reorganization. Cancer Res 69: 6122–6130. [DOI] [PubMed] [Google Scholar]
- 28. Kelly P, Moeller BJ, Juneja J, Booden MA, Der CJ, et al. (2006) The G12 family of heterotrimeric G proteins promotes breast cancer invasion and metastasis. Proc Natl Acad Sci U S A 103: 8173–8178. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Kelly P, Stemmle LN, Madden JF, Fields TA, Daaka Y, et al. (2006) A role for the G12 family of heterotrimeric G proteins in prostate cancer invasion. J Biol Chem 281: 26483–26490. [DOI] [PubMed] [Google Scholar]
- 30. Ki SH, Choi MJ, Lee CH, Kim SG (2007) Galpha12 specifically regulates COX-2 induction by sphingosine 1-phosphate. Role for JNK-dependent ubiquitination and degradation of IkappaBalpha. J Biol Chem 282: 1938–1947. [DOI] [PubMed] [Google Scholar]
- 31. Peng C-H, Liao C-T, Peng S-C, Chen Y-J, Cheng A-J, et al. (2011) A novel molecular signature identified by systems genetics approach predicts prognosis in oral squamous cell carcinoma. PLoS ONE 6: e23452–e23452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Lin S-C, Liu C-J, Chiu C-P, Chang S-M, Lu S-Y, et al. (2004) Establishment of OC3 oral carcinoma cell line and identification of NF-kappa B activation responses to areca nut extract. Journal of oral pathology & medicine 33: 79–86. [DOI] [PubMed] [Google Scholar]
- 33. Lu Y-C, Chen Y-J, Wang H-M, Tsai C-Y, Chen W-H, et al. (2012) Oncogenic function and early detection potential of miRNA-10b in oral cancer as identified by microRNA profiling. Cancer prevention research 5: 665–674. [DOI] [PubMed] [Google Scholar]
- 34. Greenberg JS, Fowler R, Gomez J, Mo V, Roberts D, et al. (2003) Extent of extracapsular spread: a critical prognosticator in oral tongue cancer. Cancer 97: 1464–1470. [DOI] [PubMed] [Google Scholar]
- 35. Wenzel S, Sagowski C, Kehrl W, Metternich FU (2004) The prognostic impact of metastatic pattern of lymph nodes in patients with oral and oropharyngeal squamous cell carcinomas. European archives of oto-rhino-laryngology 261: 270–275. [DOI] [PubMed] [Google Scholar]
- 36. Shaw RJ, Lowe D, Woolgar JA, Brown JS, Vaughan ED, et al. (2010) Extracapsular spread in oral squamous cell carcinoma. Head Neck 32: 714–722. [DOI] [PubMed] [Google Scholar]
- 37. Liao CT, Lee LY, Huang SF, Chen IH, Kang CJ, et al. (2011) Outcome analysis of patients with oral cavity cancer and extracapsular spread in neck lymph nodes. Int J Radiat Oncol Biol Phys 81: 930–937. [DOI] [PubMed] [Google Scholar]
- 38. Tung CL, Lin ST, Chou HC, Chen YW, Lin HC, et al. (2013) Proteomics-based identification of plasma biomarkers in oral squamous cell carcinoma. J Pharm Biomed Anal 75: 7–17. [DOI] [PubMed] [Google Scholar]
- 39. Bian D, Mahanivong C, Yu J, Frisch SM, Pan ZK, et al. (2006) The G12/13-RhoA signaling pathway contributes to efficient lysophosphatidic acid-stimulated cell migration. Oncogene 25: 2234–2244. [DOI] [PubMed] [Google Scholar]
- 40. Radhika V, Hee Ha J, Jayaraman M, Tsim S-T, Dhanasekaran N (2005) Mitogenic signaling by lysophosphatidic acid (LPA) involves Galpha12. Oncogene 24: 4597–4603. [DOI] [PubMed] [Google Scholar]
- 41. Aki Y, Kondo A, Nakamura H, Togari A (2008) Lysophosphatidic acid-stimulated interleukin-6 and -8 synthesis through LPA1 receptors on human osteoblasts. Archives of oral biology 53: 207–213. [DOI] [PubMed] [Google Scholar]
- 42. Chen SU, Chou CH, Lee H, Ho CH, Lin CW, et al. (2008) Lysophosphatidic acid up-regulates expression of interleukin-8 and -6 in granulosa-lutein cells through its receptors and nuclear factor-kappaB dependent pathways: implications for angiogenesis of corpus luteum and ovarian hyperstimulation syndrome. J Clin Endocrinol Metab 93: 935–943. [DOI] [PubMed] [Google Scholar]
- 43. Hwang YS, Lee SK, Park KK, Chung WY (2012) Secretion of IL-6 and IL-8 from lysophosphatidic acid-stimulated oral squamous cell carcinoma promotes osteoclastogenesis and bone resorption. Oral Oncol 48: 40–48. [DOI] [PubMed] [Google Scholar]
- 44. Ho MKC, Su Y, Yeung WWS, Wong YH (2009) Regulation of transcription factors by heterotrimeric G proteins. Current Molecular Pharmacology 2: 19–31. [DOI] [PubMed] [Google Scholar]
- 45. Facchinetti MM, Gandini NA, Fermento ME, Sterin-Speziale NB, Ji Y, et al. (2010) The expression of sphingosine kinase-1 in head and neck carcinoma. Cells Tissues Organs 192: 314–324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Penson RT, Kronish K, Duan Z, Feller AJ, Stark P, et al. (2000) Cytokines IL-1beta, IL-2, IL-6, IL-8, MCP-1, GM-CSF and TNFalpha in patients with epithelial ovarian cancer and their relationship to treatment with paclitaxel. International journal of gynecological cancer 10: 33–41. [DOI] [PubMed] [Google Scholar]
- 47. Waugh DJJ, Wilson C (2008) The interleukin-8 pathway in cancer. Clinical cancer research 14: 6735–6741. [DOI] [PubMed] [Google Scholar]
- 48. Wang Y, Niu X, Qu Y, Wu J, Zhu Y, et al. (2010) Autocrine production of interleukin-6 confers cisplatin and paclitaxel resistance in ovarian cancer cells. Cancer Lett 295: 110–123. [DOI] [PubMed] [Google Scholar]
- 49. Freier K, Engel M, Lindel K, Flechtenmacher C, Mühling J, et al. (2008) Neoadjuvant concurrent radiochemotherapy followed by surgery in advanced oral squamous cell carcinoma (OSCC): a retrospective analysis of 207 patients. Oral Oncol 44: 116–123. [DOI] [PubMed] [Google Scholar]
- 50. Lebman D, Spiegel S (2008) Cross-talk at the crossroads of sphingosine-1-phosphate, growth factors, and cytokine signaling. Journal of lipid research 49: 1388–1394. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
List of immune-related functional groups impaired in the Gα12-depleted OSCC cells.
(TIF)
Western blot analysis of siRNA knockdown efficiencies and overexpression levels of Gα12 in four different OSCC cell lines (HSC-3, SCC25, OC-3, and CGHNC9).
(TIF)
Baseline levels of IL-6 and IL-8 secreted in conditioned media from four different OSCC cell lines (HSC-3, SCC25, OC-3, and CGHNC9). Quantitative measurements of IL-6 and IL-8 were determined by ELISA assays.
(TIF)
IL-8 neutralizing antibody abolishes the Gα12-induced OSCC cell migration. (A), (B) Transwell migration assays of the Gα12-overexpressing HSC-3 and SCC25 cells treated with neutralizing antibody (10 µg/ml) against IL-8 for 16 h. Quantification of migration is shown in the right panel. Error bars represent SD of the mean from three independent experiments and analyzed by t-test. *P<0.05, **P<0.01.
(TIF)
Recombinant IL-8 restores the migration ability reduced by transiently depleted Gα12 in HSC-3 cells. Representative images show the migration of the Gα12-depleted HSC-3 cells through transwells. Cells treated with or without recombinant human IL-8 (1 ng/ml) for 16 h. Quantification of migration is shown in the right panel. Error bars represent SD of the mean from three independent experiments and analyzed by t-test. *P<0.05.
(TIF)