Abstract
Angiogenesis, the development of new vessels from existing vasculature, plays a central role in tumor growth, survival, and progression. On the molecular level it is controlled by a number of pro- and anti-angiogenic cytokines, among which the vascular endothelial growth factors (VEGFs), together with their related VEGF-receptors, have an exceptional position. Therefore, the blockade of VEGF signaling in order to inhibit angiogenesis was deemed an attractive approach for cancer therapy and drugs interfering with the VEGF-ligands, the VEGF receptors, and the intracellular VEGF-mediated signal transduction were developed. Although promising in pre-clinical trials, VEGF-inhibition proved to be problematic in the clinical context. One major drawback was the generally high variability in patient response to anti-angiogenic drugs and the rapid development of therapy resistance, so that, in total, only moderate effects on progression-free and overall survival were observed. Biomarkers predicting the response to VEGF-inhibition might attenuate this problem and help to further individualize drug and dosage determination. Although up to now no definitive biomarker has been identified for this purpose, several candidates are currently under investigation. This review aims to give an overview of the recent developments in this field, focusing on the most prevalent tumor species.
Keywords: cancer, anti-angiogenic therapy, biomarkers, VEGF
1. Introduction
The growth and progression of tumors is crucially dependent on the supply of oxygen and the exchange of nutrients and metabolites with the surrounding tissue. As transport based on diffusion of these molecules is limited to very short distances of less than 3–4 mm only, a tumor exceeding this size needs to develop a vascular system in order to survive. The most important process driving this neovascularization is angiogenesis, the development of new vessels from the existing vascular system [1,2]. Physiologically, angiogenesis is active in normal adults during placenta formation and in wound healing [3,4], but deregulated angiogenesis can occur in diabetes, psoriasis, or rheumatoid arthritis [5]. Most importantly, angiogenesis is also implied in cancer. Starting early during tumor development [6,7] it is an important determinant of tumor aggressiveness and the degree of metastatic spread [8].
On the molecular level, angiogenesis is controlled predominantly by the relatively small family of vascular endothelial growth factors (VEGFs). It comprises VEGF-A (commonly named VEGF), VEGF-B, VEGF-C, VEGF-D, VEGF-E, and placenta growth factor (PlGF) [9–12]. Moreover, angiogenesis can be induced also via alternative pathways by other soluble factors such as fibroblast growth factor 1 and 2 (FGF1 and FGF2), angiopoietin or ephrin A1 and A2 [13,14]. While basal (blood) VEGF levels are necessary to sustain an intact vascular system [15], many tumors are characterized by elevated secretion of various VEGF isoforms [16]. This is considered to be a reaction of the cancer cells to a hypoxic and growth-factor rich environment [17–19] they are exposed to due to their high proliferation rate. The elevated VEGF levels may stimulate endothelial cells of nearby blood vessels to develop new vessels in order to supply the tumor with nutrients and oxygen and thus support its further growth [20–25].
The VEGF proteins are ligands for three tyrosine kinase receptors: VEGF receptor 1 (VEGFR-1, also called Flt-1), VEGF receptor 2 (VEGFR-2), and VEGF receptor 3 (VEGFR-3) [26–28]. Vascular endothelial cells predominantly express VEGFR-1 (with VEGF-A, VEGF-B, and PlGF as ligands) and VEGFR-2 (with VEGF-A as its main ligand [29]), whereas VEGFR-3 (binding VEGF-D and VEGF-E [30]) is mostly found in lymphatic endothelial cells controlling lymphangiogenesis, but can also be expressed in tumor vessels or chronic inflammatory wounds [16,31,32].
VEGF-A is the central factor in the promotion and regulation of tumor angiogenesis, VEGF signaling is mainly mediated by VEGFR-2 [33,34], while VEGFR-1 is believed to act as a decoy receptor, controlling VEGF availability [35,36]. VEGF-A/VEGFR-2 signaling plays an important role in both, physiological and pathophysiological processes, including burn injury [37], wound healing [38,39] and tumor angiogenesis. Importantly, it has been demonstrated as well that VEGF-A is a survival factor for endothelial cells [20,40–43].
2. Anti-Angiogenic Therapy
Considering the outstanding importance of angiogenesis for tumor growth and survival in general and the role of the VEGF-A/VEGFR-2 signaling system in particular, several drugs have been developed, which interfere with different angiogenic molecules. In principle, three different modes of action are possible: interception of the VEGF ligand, blockade of the VEGFR, or interruption of the intracellular VEGFR-mediated signaling.
Examples for drugs targeting the VEGF-A ligand are Bevacizumab and Aflibercept. Bevacizumab is a VEGF-binding humanized recombinant antibody, which inhibits the VEGF-VEGFR-interaction [44]. In the clinical context it has been used in lung [45–48], breast [49–53], colon [54–58], renal [59,60], gastric [61], pancreatic [62,63], and prostate cancer [64], as well as melanoma [65]. Aflibercept is a fusion protein of the human Fc part of IgG1 and the extracellular domain of VEGFR. As such it is able to quench VEGF-A and -B, and PlGF-1 and 2, effectively removing the soluble ligands from the VEGF-VEGFR-cascade. So far Aflibercept has been applied in ovarian [66], colorectal [67], lung [68], metastatic gynecologic soft-tissue [69], and urothelial metastatic transitional cell cancer [70], as well as melanoma [71] and glioblastoma [72].
Ramucirumab, a human antibody specific for the extracellular ligand-binding domain of VEGFR-2, belongs to the class of VEGFR-blocking drugs [73]. Ramucirumab has been used in studies for a multitude of different cancer types and has shown the best results for stable disease (only minor increases or decreases in tumor size) in renal, uterine, colorectal, and ovarian carcinoma [74].
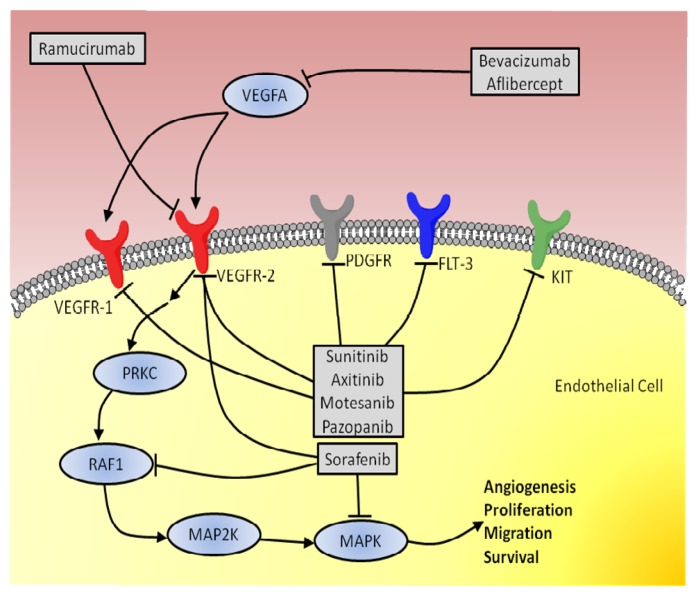
Small-molecule tyrosine kinase inhibitors (TKIs) inhibit ATP binding to the tyrosine domain of VEGFRs and therefore interrupt the VEGFR signal transduction. The most prominent members of this class of drugs are Sunitinib and Sorafenib, which are primarily used in renal [75] and gastrointestinal stromal tumors [76] or renal [77] and hepatocellular cancer [78], respectively. Further members include Motesanib, which is used in lung [79] and medullary thyroid cancer [80], or Pazopanib, which is approved for renal cell cancer and soft tissue sarcomas [81] (see Figure 1 for a brief overview).
Figure 1.
Overview of representative anti-angiogenic drugs and their targets in the angiogenic pathway. FLT-3: Fms-like tyrosine kinase 3; KIT: Mast/stem cell growth factor receptor; MAP2K: Mitogen-activated protein kinase kinase; MAPK: Mitogen-activated protein kinase; PDGFR: Platelet derived growth factor receptor; PRKC: Protein kinase C; RAF1: Proto-oncogene c-RAF; VEGFA: Vascular endothelial growth factor A; VEGFR-1: Vascular endothelial growth factor receptor 1; VEGFR-2: Vascular endothelial growth factor receptor 2.
However, although many studies seem to indicate a modest benefit of anti-angiogenic therapy, it generally suffers from a high variability in the response by the individual patient. Possible reasons for this observation may be the general capability of endothelial cells to form new vessels independently of an enhancement of the VEGF-A/VEGFR-2 signaling system. In a series of in vitro experiments, our group could demonstrate that cellular changes [82,83] induced by culturing endothelial cells under simulated microgravity trigger some cells to form tubes, which resemble the intima of blood vessels [43,84,85]. This suggests that some types of cancer cells may activate a mechanism inducing neighboring endothelial cells to provide a supply of oxygen and nutrients to a tumor when the VEGF/VEGFR signaling system is switched off by drugs. Therefore, it would be an important milestone to identify factors, which indicate the tendency of some tumor-subtypes in developing evasive strategies against anti-angiogenesis treatment. Knowledge of such factors will allow the prediction of and the adequate reaction to these effects. Hence a more individualized treatment of any patient could enhance the success of the therapy.
3. Biomarkers
Cancer cells are usually classified by biomarkers. A biomarker, as defined by the NIH, is “a characteristic objectively measured and evaluated as an indicator of normal biologic processes, pathogenic processes, or pharmacologic responses to a therapeutic intervention” [86]. Several types of biomarkers can be distinguished. Prognostic biomarkers help estimating the overall disease outcome, independent from therapy [87]. Predictive biomarkers on the other hand provide information about the response or survival of a certain patient under a specific treatment prior to therapy [88]. Furthermore, biomarkers can also have screening, diagnostic, pharmacodynamic, and safety-related properties or act as surrogate parameters. The use of biomarkers might therefore be a way to circumvent the drawbacks of indiscriminate anti-angiogenic therapy by enabling physicians to select patients with the highest likelihood for a positive response to a treatment. This review will focus on recent developments in biomarkers for anti-angiogenic therapy in the most prevalent cancers.
3.1. Biomarkers in Colorectal Cancer
Colorectal cancer is the second most frequent cause of death in North America and Europe with about 600,000 deaths and a rate of approximately 1.2 million new cases per year worldwide [89]. At the moment, no confirmed biomarkers are known allowing the prediction of anti-angiogenic therapy efficacy for this cancer. It was found in clinical trials that the survival benefit from adding Bevacizumab to standard chemotherapy was neither determined by K-ras, BRAF, or p53 mutation status nor by VEGF, p53 or thrombospondin 2-expression [90,91]. For that reason it is important to find new candidates to identify suitable colorectal tumor patients for VEGF-targeted therapeutic approaches.
3.1.1. Circulating Biomarkers
Blood is a relatively easily available material for the analysis of biomarkers and has a great potential for this application. Several studies have analyzed circulating molecules for their potential as predictive biomarkers for colorectal cancer. Cetin et al. [92] found that in a cohort of patients treated with FOLFIRI or XELOX in combination with Bevacizumab, serum LDH and neutrophil levels higher than the upper limit of normal (ULN) were independent predictors of short term survival. With a similar therapeutic regimen, Kopetz et al. [93] have screened a total of 37 plasma cytokines and circulating angiogenic factors for their use as biomarkers for treatment response or resistance to a FOLFIRI/Bevacizumab therapy. From this panel, elevated IL-8 levels at baseline were linked to a shorter progression-free survival (PFS).
Angiopoietin-2 is another potentially valuable circulating biomarker. It has been proposed to be involved in VEGF function and vascular remodeling. Interestingly, low serum angiopoietin-2-levels were found to be associated with a high overall survival (OS) of >90% after 18 months and a better response rate to anti-angiogenic therapy of >80% compared to high serum angiopoietin-2-levels in a study analyzing patients receiving Bevacizumab in combination with different chemotherapeutic regimens (FOLFOX, FOLFIRI, XELOX, XELIRI) [94].
Finally, circulating endothelial cells (CEC) have been found to predict the response to Bevacizumab-therapy in colorectal cancer. Blood from patients receiving FOLFOX4 and Bevacizumab was analyzed for endothelial cells before and during treatment. It could be shown that no correlation existed between the CEC levels and the outcome in a FOLFOX4 alone control group. However, CEC proved to be a strong indicator for the outcome of the Bevacizumab-based therapy. Patients with less than 65 CEC/4 mL blood at baseline (as determined by the CellSearch system), had a significantly longer median PFS and OS than patients with 65 CEC/4 mL or more. In addition, a low proportion of CXCR4-positive CEC of below 20% at baseline was also correlated to significantly longer PFS and OS [95,96]. These findings were confirmed by another study, which showed that a total number of 40 CEC/mL or less was connected to longer PFS [97]. The same group further investigated the course of CEC levels over the duration of treatment and the role of the fraction of apoptotic cells among them. They found that increases of both CEC and the apoptotic CEC subpopulation at the 6th cycle of Bevacizumab-based therapy were statistically significant indicators for better PFS [98].
3.1.2. Genetic Biomarkers
Genes involved in angiogenesis show a relatively high level of variation, ranging from silent SNPs to functional polymorphisms. The latter were tested in the VEGF, KDR, IL6, CXR1 and -2, P53, MMP2,-7,-9, and ICAM genes of patients with metastatic colorectal cancer receiving FOLFOX or XELOX with Bevacizumab using genomic DNA from peripheral blood. The IL-6 G-174C and P53 codon 72 polymorphisms were found to be correlated to a positive response to Bevacizumab therapy. Furthermore, PFS was significantly associated with MMP9 C-1562T and CXCR-1 G + 2607C [99].
In another study, the Nordic ACT trial (Bevacizumab + FOLFOX, XELOX; FOLFIRI, or XELIRI), the VEGFR-1 319 C/A SNP has been identified as significantly associated with response to Bevacizumab. Objective response rates differed significantly between the three genotypes (CC = 36% vs. CA = 40% vs. AA = 56%, p = 0.048, or CC + CA = 39% vs. AA = 56%, p = 0.015), indicating that the A-allele exerts a strong beneficial effect [100].
CD133, a trans-membrane protein isolated from colorectal cancer stem cells [101,102], has biomarker properties as well. Two SNPs have been identified as being useful in the prediction of PFS and OS in patients treated with FOLFOX/Bevacizumab or XELOX in first line [103]. Patients carrying either CC in both rs2286455 and rs3130 SNPs or a combination of CT with CT or TT exhibited a doubled PFS (16.5 months vs. 8.4 months, p = 0.010) after treatment with FOLFOX/Bevacizumab as compared to the rest.
3.1.3. Physiologic Biomarkers
Hypertension is a very common side effect of VEGF inhibitor medications, with general incidence rates of about 20% and grade 3 hypertension percentages of approximately 11% [56, 104]. Hurwitz et al. concluded in their recent paper that early treatment-related blood pressure increases do not predict clinical benefit from Bevacizumab based on PFS or OS outcomes. BP increases do not appear to have general prognostic importance for patients with advanced cancer [104]. Although the exact mechanism of hypertension induction by anti-angiogenic treatment is unknown, it is hypothesized that VEGF signaling influences NO-synthase activity. VEGF inhibitors might therefore reduce NO-production, leading to increased vasoconstriction and ultimately hypertension, which would be an indicator for a successful inhibition of angiogenesis [105]. This idea has been confirmed by different studies. Scartozzi et al. observed that Bevacizumab-induced grade 2–3 hypertension in patients receiving Irinotecan, Fluorouracil, and Bevacizumab was significantly associated with improved PFS (14.5 months vs. 3.1 months, p = 0.04) [106]. Österlund et al. described that hypertension was associated with prolonged PFS (10.5 months vs. 5.3 months, p = 0.008) and OS (25.8 months vs. 11.7 months, p < 0.001) in patients treated with Bevacizumab-containing chemotherapy [107]. Development of hypertension within three months was identified as an independent prognostic factor and no relation of hypertension to thromboembolic complications could be detected. In a retrospective analysis of patients with a therapeutic regimen of FOLFIRI, FOLFOX, XELOX, XELIRI, or FOLFOXIRI together with Bevacizumab, De Stefano et al. observed an induced grade 2–4 hypertension in 17.6% of the cases. Of the patients with induced arterial hypertension, 84.6% achieved a complete or partial response, whereas patients without these side effects only had 41.9% (p = 0.006). In addition and comparable to the other studies, hypertension was associated with improved PFS (15.1 months vs. 8.3 months, p = 0.04) [108].
3.2. Biomarkers in Breast Cancer
Among women, breast cancer is the malignancy with the highest occurrence at a rate of about 23% and a total number of approximately 1.4 million new cases per year worldwide. Furthermore, with about 460,000 cases per year it is also the most frequent cause of death due to cancer [89]. Unfortunately, so far anti-angiogenic therapy in general had only limited success. Most clinical studies showed no benefit in OS and PFS, the FDA withdrew its approval of Bevacizumab as a drug against metastatic HER2-negative breast cancer, and Sunitinib has failed altogether [109]. However, although anti-angiogenic therapy does not seem to be an option for every breast cancer patient, it is interesting to note that in each trial were patients who benefitted strongly from this regimen. So far no pattern has been identified on how to select these individuals, but efforts are ongoing to discover biomarkers for this purpose.
3.2.1. Circulating Biomarkers
In a study with combined Bevacizumab/Vinorelbine chemotherapy Burstein et al. could demonstrate that baseline VEGF plasma levels were associated with the time to progression (TTP). Patients with VEGF concentrations >32.6 pg/mL had a median TTP of 3.7 months, whereas patients with VEGF levels <32.6 pg/mL had a median TTP of 9.3 months (p = 0.003) [110]. In addition, in locally advanced breast cancer preoperatively treated by Docetaxel with or without Bevacizumab, low baseline serum concentrations of both VCAM-1 and E-selectin were significant (p = 0.033 and p = 0.035, respectively) predictors of clinical response in the form of operability [111]. Burstein et al. measured the VEGF, soluble VEGFR-2 (sVEGFR-2), soluble VEGFR-3 (sVEGFR-3), and soluble KIT (sKIT) plasma levels at baseline and during therapy in patients receiving Sunitinib after chemotherapy with and Anthracycline or Taxane. They found a trend for a connection of decreasing sVEGFR-3 levels (≥20%) with longer OS (p = 0.07). In addition, decreases of sKIT levels by ≥50% were significantly associated with longer TTP (p < 0.001) and OS (p = 0.0194) [112]. Finally, CEC proved to be predictors of anti-angiogenic therapy success in a cohort of patients receiving metronomic, i.e., repeated, low-dosed, chemotherapy in conjunction with Bevacizumab. A significant association of high baseline levels of CEC with overall response (p = 0.02), clinical benefit (p = 0.01), and improved PFS (p = 0.04) was observed in this study [113].
3.2.2. Genetic Biomarkers
So far, only polymorphisms in the VEGF and VEGFR2 genes have been analyzed for their potential to serve as a biomarker for anti-angiogenic therapy in breast cancer. Schneider et al. did a retrospective study on the ECOG2100 cohort, investigating five SNPs for VEGF and two for VEGFR2. They found that VEGFR2 polymorphisms did not show any influence on OS or any other clinical parameter. Two VEGF genotypes, on the other hand, VEGF-2578 AA and VEGF-1154 AA, were significantly associated with improved OS (p = 0.023 and p = 0.001, respectively) in the Paclitaxel/Bevacizumab combination arm of the study [114].
3.2.3. Physiologic Biomarkers
The role of anti-angiogenic therapy induced hypertension is subject of debate, as currently there are contradicting results found in the literature. Beside identifying two VEGF-SNPs as candidate biomarkers, Schneider et al. also observed in the same study that patients who developed grade 3–4 hypertension had an improved median OS of 38.7 months vs. 25.3 months of normotensive patients (p = 0.002) [114]. In contrast to this, a meta-analysis of clinical outcome under anti-angiogenic medication (including the RIBBON-1 trial, comparing Bevacizumab + chemotherapy vs. Bevacizumab + placebo) did not show a prognostic value of early hypertension [115]. Further investigations are needed to clarify this situation.
3.2.4. Tissue Biomarkers
Blood levels of VEGF and related molecules can be influenced by many different additional factors besides cancer and may therefore be misleading or difficult to interpret in an analysis of their predictive value. Hence it might be a better approach to directly determine the expression of candidate genes or proteins inside the tumor tissue itself. This, of course, poses some problems, like easy availability of ideally multiple samples over the course of the therapy.
Fountzilas et al. immunohistochemically investigated a panel of biomarker candidate proteins in patients receiving a first line Paclitaxel/Bevacizumab therapy against metastatic breast cancer. They found that high intra-tumoral expression of VEGFR-3 was associated with clinical response, whereas VEGFR1 overexpression was an indicator for poor survival [116].
3.3. Biomarkers in Thyroid Cancer
Although representing only a small portion of cancer cases with an incidence of about 213,000 new cases (which equates to approximately 1.7%) and a mortality of a bit over 35,000 cases per year worldwide, thyroid cancer is the most frequent malignant endocrine tumor [89,117]. For a long time radio-therapy was the classical way to fight this disease [118]. Besides certain subtypes, such as the progressive or advanced medullary (MTC), or the differentiated (DTC) thyroid cancers [119,120], especially distantly metastatic tumors are still difficult to treat with the traditional therapies [121,122]. Earlier studies have shown that targeting endothelial cells in SCID mice bearing a follicular thyroid ML-1 tumor by the tyrosine kinase inhibitor PTK787/ZK222584 reduced tumor growth and vascularization [123]. In addition, an Angs/Tie-2 system dysfunction was suggested to play an important role in thyroid tumorigenesis. The decrease of the concentration of the angiogenesis inhibitor Ang-1 in serum was a useful additional biomarker for the presence of thyroid cancer [124]. Liang et al. demonstrated that MMP2, PTTG, VEGF-C, CXCR4 and bFGF are potential cellular tumor markers for identifying thyroid cancer with greater risk for metastasis. The authors suggest that the combination of the angiogenic factors VEGF-C and bFGF favors progression in metastatic thyroid carcinoma [125]. Thus, treating thyroid cancer with the help of pharmacological inhibitors of angiogenesis was considered to be a promising new way of fighting thyroid cancer [126]. Subsequently, over the course of the last five years, the option of anti-angiogenic treatment has been explored [127–133]. The results were encouraging, but a considerable variation in the response of the various patients was observed. Therefore, predictive biomarkers are sought, which could indicate the success of an anti-angiogenic treatment in advance.
3.3.1. Circulating Biomarkers
Broutin et al. showed in 2011 [134] that cytokines are possible biomarkers for the tumor response towards Sunitinib treatment of in medullary thyroid carcinomas (MTC). A significant decrease of tumor growth and angiogenesis was observed after Sunitinib therapy in a mouse model, which is associated with significantly decreased serum IL-8 levels. In parallel, the serum of 27 patients with MTC showed significantly increased serum concentrations of IL-8 compared to the healthy donor population [134]. In addition, we found that IL-8 gene expression is involved in human thyroid cancer cell tumor formation [135]. Therefore, IL-8 appears very interesting as a therapeutic target and as a clinical biomarker for the success of an anti-angiogenic treatment.
Bass et al. analyzed a cohort of patients with progressive advanced thyroid cancer receiving Motesanib as anti-angiogenic therapy over a period of 48 weeks [136]. During this time, the group determined serum and plasma concentrations of sVEGFR1 and -2, PlGF, VEGF, bFGF, sKIT, sVCAM-1, angiopoietin-1 and 2, and enzyme activities of caspases-3 and -7. It was found that patients with baseline VEGF levels below 671 pg/mL had a significantly better PFS than patients with higher baseline VEGF concentrations (p = 0.0007). Furthermore, the study could show that not only absolute serum concentrations or activities but also their changes during treatment were a useful indicator for patient response to therapy. Increases of PlGF by more than 4.7 fold (p < 0.0001) and of caspase 3/7-activity by more than 2.1-fold (p < 0.0001) as well as changes by less than −1.6-fold of sVEGFR2 (p < 0.0001) were independent predictors enabling investigators to separate responders from non-responders [136]. In another study, Sorafenib was applied in advanced iodine-refractory differentiated thyroid cancer. The investigators analyzed serum thyroglobulin (Tg) levels and found that both Tg baseline levels as well as Tg response were useful in predicting the clinical outcome of anti-angiogenic therapy with Sorafenib [137]. Furthermore, Sorafenib and Sunitinib have been shown to be effective in patients with widely metastatic, progressive differentiated thyroid cancer. Logarithmic thyroglobulin (Log Tg) significantly correlated with response to this treatment [138]. Now, serum thyroglobulin levels are suggested to have a value as a surrogate marker of response. So far, Tg had been used as a diagnostic tool [139] or as a predictor of recurrence after thyroidectomy [140,141].
3.3.2. Tissue Biomarkers
Shaik and coauthors described a subtype of human papillary thyroid cancer (PTC), which is resistant to therapy with VEGF receptor 2 (VEGFR2)-inhibitor. In these poorly differentiated PTC cells, the beta-transducin-repeat-containing protein inhibits cell migration and decreases sensitivity to Sorafenib [142]. In various thyroid carcinomas, hypoxia-inducible factor-1alpha expression was found to be increased [143]. Zerilli et al. demonstrated that hypoxia-inducible factor-1alpha is expressed in papillary thyroid carcinomas and is not only regulated by hypoxia but also by the BRAF(V600E)-mediated signaling pathway [143]. BRAF(V600E) (serine/threonine-protein kinase B-raf) is an oncogene linked to angiogenesis. Its presence in a cancer cell favors angiogenesis [144]. Besides the two preceding proteins, a number of proteins, especially membrane proteins, may be indicators of successful and un-successful angiogenic therapy [145]. Therefore, our group applies the methods of free-flow IEF and mass spectrometry to screen and evaluate as many proteins of thyroid cancer cells as possible in comparison to their behavior [146–149].
3.4. Biomarkers in Renal Cancer
Renal cancer is among the 10 most frequently occurring cancers in western countries and accounts for more than 100,000 deaths worldwide per year [150]. Renal cell carcinomas (RCC) comprise approximately 90% of renal cancers with the most common histological subtype being clear cell RCC (ccRCC) accounting for 80% of the cases. Up to 70% of patients are presented with localized disease, and approximately one third of these will relapse with metastatic RCC following radical or partial nephrectomy. This clearly indicates the need for tools to maximize the benefit of drug treatment and evaluate the risk of relapse on an individual basis. Today, classification of RCC is largely based on morphology and despite the emergence of promising prognostic biomarkers in RCC, none have been routinely applied in the clinic. Furthermore, no predictive biomarkers are used to identify patients who might benefit from a given treatment.
Knowledge of ccRCC biology has led to a number of anti-angiogenic systemic therapies targeting VEGF either directly by inhibiting antibodies or via targeted tyrosine kinase inhibitors. First line adjuvant therapies include Sunitinib and Pazopanib which are effective in 70%–80% of cases. There are no biomarkers available that may discriminate between patients who will benefit from this treatment and those who will not.
3.4.1. Circulating Biomarkers
Plasma VEGF and soluble VEGFR-2 (sVEGFR-2) has been tested as predictive markers of anti-angiogenic treatment in phase III Treatment Approaches in Renal Cancer Global Evaluation Trial (TARGET). In this study high baseline levels of VEGF were associated with poor prognosis but baseline sVEGFR-2 and changes in VEGF or sVEGFR-2 could not predict the response to Sorafenib [151]. In phase II trials using Sunitinib or Pazopanib significant changes in sVEGFR-2 levels were demonstrated in patients showing objective tumor response to treatment compared to patients with stable or progressive disease [152,153]. In a recent study of an unselected population of advanced kidney cancer patients receiving Sunitinib, serum levels of circulating neutrophil gelatinase associated lipocalin (NGAL) and VEGF were strongly associated to an improved progression free survival in both univariate and bivariate analyses and performed better than the Motzer score considered golden standard [154]. NGAL is upregulated in cells during “stress” and is tightly coupled to matrix metalloproteinase 9 (MMP-9), involved in the degradation of the extracellular matrix [155], making these proteins relevant for further analyses.
High levels of CAIX protein was shown to correlate to the responsiveness of interleukin (IL)-2 with 78% of patients responding to IL-2 showing high expression (>85% tumor cells) of CAIX protein in tumors compared to 51% in non-responders. The value of CAIX as a predictive marker is currently under investigation [156,157]. Similarly, HIF-1 alpha expression has been implicated as a potential prognostic marker where high (>35%) tumor-immunostaining levels were shown to correlate to shorter survival [158]. In addition, HIF-2 alpha expression was correlated to a beneficial responsive to Sunitinib in 43 ccRCC samples [159]. Other promising protein markers include tumor-associated B7-H1 and insulin-like growth factor II mRNA binding protein 3, both of which have been independently validated and add value to existing nomograms in RCC [160,161], but have not found their way into the clinic. Recent studies suggest that in addition to using only single biomarkers it might be more promising to screen a whole panel of cytokines and angiogenic factors (CAFs). Zurita et al. were able to identify a candidate CAF signature that may help predict PFS benefit from Sorafenib treatment in patients suffering from metastatic RCC [162,163].
3.4.2. Genetic Biomarkers
Single Nucleotide Polymorphisms
Genome-wide association studies have reported SNPs that may increase the risk of developing RCC [164,165]. A large study comprising 397 patients with RCC treated with Pazopanib addressed the response to TKI therapy [166]. 27 SNPs in 13 genes were reported including genes related to angiogenesis VEGF, IL-8 and fibroblast growth factor 2. Two polymorphisms in IL-8 were significantly associated to a shorter PFS of 27 weeks compared to the wild type genotype (48 weeks). Notably, IL-8 has recently been suggested as to be involved in resistance to TKIs [167]. In another study the response and toxicity of Sunitinib were evaluated in patients with ccRCC. Two VEGFR-3 SNPs were associated with reduced PFS, a variant of CYP3A5*1 was associated with increased toxicity, however, the IL-8 variants described above were not found in this study [168]. Interestingly, a third study found that SNPs in CYP3A5 increased survival [169], indicating little concordance between these studies. To some extent these disparate observations may be explained by sample number, indicating the need for large studies to increase the statistical power. Perhaps an even greater challenge arises from the heterogeneity found within individual tumors. This point was demonstrated using multi-region genetic analysis, showing that up to two thirds of mutations found in one region of the tumor were not present in other regions of the same tumor, suggesting that both favorable and unfavorable conclusions can be made depending on the specific specimen sampling [170]. Clearly, heterogeneity is a feature of all cancers, but to what extent these differences impact the tumor phenotype and therapeutic targets is unknown. However, mRNA profiling and expression of protein biomarkers are likely more robust to these changes as they are upstream of these events.
3.5. Biomarkers in Prostate Cancer
Among men prostate cancer (PC) is the second most frequently occurring cancer with about 900,000 new cases and approximately 260,000 deaths per year worldwide. It also has by far the highest worldwide 5-year prevalence of roughly 24% [89]. In USA and Europe, PC is estimated to account for 25% of cancer diagnoses in males and 9% of cancer related deaths [171,172]. The diagnosis of PC includes measurement of serum prostate-specific antigen (PSA), rectal examination and morphological/histological evaluation of needle biopsy. The treatment of prostate cancer includes prostatectomy and radiation, while androgen withdrawal is used to delay the progression of metastatic disease [173]. However, over time tumors become resistant to androgen deprivation and develop into castrate resistance PC (CRPC) with high morbidity and mortality. Therefore, anti-angiogenic treatment was considered an alternative way to fight PC. But, similar to breast cancer, anti-angiogenic therapy of prostate cancer has shown only moderate to disappointing effects, with little or no improvement in OS resulting from addition of Bevacizumab to a standard Docetaxel and Prednisone therapeutic regimen [64]. Nevertheless, trials with different anti-angiogenic drugs are still ongoing and biomarkers for the assessment of therapeutic efficiency are needed.
3.6. Future Developments
So far, most analyses of possible biomarker candidates for the prognosis and prediction of anti-angiogenic therapy have been conducted with a rational approach (Table 1), concentrating on molecules which are more or less directly involved in angiogenic pathways. As summarized in this article, many factors, initially expected to yield clear results, proved to be not as robust for this purpose, with the most prominent example being VEGF. Many candidate parameters suffer from poor reproducibility across different tumor types and there are still not enough studies comparing the same markers in different cancers [174]. Therefore it might be necessary to think more “outside the box” and to employ a wider, assumption-free strategy by using the increasingly easier available techniques for genomic and proteomic analysis of the samples. Gene expression profiling, preferably of endothelial cells originating from the tumor tissue, might provide further insight into the different types of tumor vasculature and help select the appropriate medication [175]. Investigating the cancer cell proteome and secretome might also lead to the identification of new, so far neglected molecules which are more effective than the “classical” candidates [176,177]. Moreover, decisions about the type of therapy may in future be based on a multitude of parameters. A profile comprising a number of different potential markers could help to predict the benefit of an anti-angiogenic therapy more robustly and reliably than a single biomarker, as attempted with CAF screening [162].
Table 1.
Prognostic biomarkers for anti-angiogenic therapy
| Type | Parameter | Cancer | Finding | References |
|---|---|---|---|---|
| Circulating | Serum LDH and neutrophil levels | Colon | LDH and neutrophil levels > ULN predict short survival | [92] |
| IL-8 | Colon | Elevated IL-8 linked to shorter PFS | [93] | |
| Angiopoietin-2 | Colon | low serum levels associated with high OS | [94] | |
| Circulating endothelial cells (CEC) | Colon | CEC < 65/4mL associated with longer PFS and OS | [95–98] | |
| Breast | High baseline levels associate with improved OR and PFS | [113] | ||
| VEGF plasma levels | Breast | <32.6 pg/mL associated with longer median TTP | [112] | |
| Thyroid | baseline concentrations ≤671 pg/mL associated with improved PFS | [136] | ||
| Renal | High baseline levels associated with poor prognosis | [151] | ||
| PlGF and sVEGFR2 plasma levels and caspase 3/7 activity | Thyroid | Changes by more than 4.7, −1.6, and 2.1-fold, respectively, indicate response | [136] | |
| sVEGFR2 plasma levels | Renal | Significant changes associated with objective tumor response | [152, 153] | |
| Serum NGAL and VEGF levels | Renal | Associated with improved PFS | [154] | |
| VCAM-1 and E-selection serum levels | Breast | Low levels associated with improved clinical response | [111] | |
| sKIT plasma level | Breast | Decrease ≥ 50%associated with longer TTP | [112] | |
| Serum Tg levels | Thyroid | Predictor for clinical outcome | [137, 138] | |
| CAF screen | Renal | Predictor for PFS benefit | [162] | |
| Genetic | MMP9 C-1562T and CXCR-1 G + 2607C | Colon | Associated with longer PFS | [99] |
| VEGFR-1 319 C/A | Colon | A-allele has strong beneficial effect | [100] | |
| CD133 rs2286455, rs3130, and rs2240688 SNPs | Colon | Associated with PFS and OS | [103] | |
| VEGF-2578 AA and VEGF-1154 AA | Breast | Associated with improved OS | [114] | |
| ccB subtype | Renal | Associated with poor prognosis | [170] | |
| VEGFR-3 and CYP3A5*1 SNPs | Renal | Associated with increased Sunitinib toxicity | [168] | |
| Physiologic | Hypertension | Colon | Associated with improved PFS | [106–108] |
| Breast | Associated with improved OS | [113] | ||
| Tissue | Tumor VEGFR-3 expression | Breast | Overexpression associated with poor survival | [115] |
| Tumor BTRC expression | Thyroid | Mediates Sorafenib-resistance | [142] |
4. Conclusions
Anti-angiogenic therapy generally suffers from a high variability in the response by the individual patient. In order to select patients with the highest likelihood for a positive response to such a treatment, the availability of reliable predictive biomarkers for anti-angiogenic therapy will be a key factor. Although there are some promising preliminary results, no general or cancer-specific biomarker has yet emerged, which could help select patients with a positive prognosis for anti-angiogenic therapy. For its future it is therefore of vital importance to conduct larger systematic trials to translate the preclinical data into clinically usable systems and to switch from unselective therapy to a more individual drug selection based on the patients’ predispositions.
Acknowledgements
The authors would like to thank Peter Lindborg, Los Angeles, CA, USA, for editing the English language.
Abbreviations
- AMACR
Alpha-methylacyl-CoA racemase
- Ang
Angiopoietin
- ATP
Adenosine triphosphate
- BRAF
Raf murine sarcoma viral oncogene homolog B1
- CAF
Cytokine and angiogenic factor
- CAIX
Carbonic anhydrase IX (CAIX)
- CEC
Circulating endothelial cells
- CD
Cluster of differentiation
- CR
Castration resistance
- CT
Computer tomography
- CTC
Circulating tumor cells
- CXCR4
CXC motif, Chemokine receptor type 4
- DNA
Deoxyribonucleic acid
- DTC
Differentiated thyroid cancer
- FDA
Food and Drug Administration
- FDG
18F-fluorodeoxyglucose
- FGF
Fibroblast growth factor
- FLT-3
Fms-like tyrosine kinase 3
- Fluorouracil
Folinic acid
- FOLFIRI
Irinotecan combination therapy
- FOLFOX4
Oxaliplatin combination therapy
- HER2
Human Epidermal Growth Factor Receptor 2
- HIF
Hypoxia-inducible transcription factor
- ICAM
Intercellular adhesion molecule
- IEF
Isoelectric focusing
- IgG
Immunoglobulin G
- IL
Interleukin
- K-ras
Kirsten rat sarcoma viral oncogene homolog
- KDR
Kinase insert domain receptor
- KIT
Mast/stem cell growth factor receptor
- LDH
Lactate dehydrogenase
- MAPK
Mitogen-activated protein kinase
- MAP2K
Mitogen-activated protein kinase kinase
- mCRPC
Metastatic castration-resistant prostate cancer
- MMP
Matrix metalloproteinase
- MRI
Magnetic resonance imaging
- MTC
Medullary thyroid carcinoma
- NGAL
Neutrophil gelatinase associated lipocalin
- NIH
National Institutes of Health
- NO
Nitric oxide
- OS
Overall survival
- PC
Prostate cancer
- PET
Positron emission tomography
- PFS
Progression-free survival
- PlGF
Placenta growth factor
- PRKC
Protein kinase C
- PSA
Prostate-specific antigen
- PTC
Papillary thyroid cancer
- PTTG
Pituitary Tumor-Transforming Gene 1-Interacting Protein
- RAF1
Proto-oncogene c-RAF
- RCC
Renal cell carcinomas
- RIBBON-1
Regimens in Bevacizumab for Breast Oncology-1
- RNA
Ribonucleic acid
- SCID
Severe combined immunodeficiency
- SNP
Single Nucleotide Polymorphisms
- TARGET
Treatment Approaches in Renal Cancer Global Evaluation Trial
- Tg
Thyroglublin
- TKI
Tyrosine kinase inhibitor
- TNM
Tumor Node Metastasis system
- TTP
Time to progression
- ULN
Upper limit of normal
- VCAM
Vascular cell adhesion molecule
- VEGF
Vascular endothelial growth factors
- VHL
Von Hippel-Lindau tumor suppressor
- XELIRI
Capecitabine and Irinotecan combination therapy
- XELOX
Capecitabine and Oxaliplatin combination therapy.
Conflict of Interest
The authors declare no conflict of interest.
References
- 1.Straume O., Chappuis P.O., Salvesen H.B., Halvorsen O.J., Haukaas S.A., Goffin J.R., Bégin L.R., Foulkes W.D., Akslen L.A. Prognostic importance of glomeruloid microvascular proliferation indicates an aggressive angiogenic phenotype in human cancers. Cancer Res. 2002;62:6808–6811. [PubMed] [Google Scholar]
- 2.Folkman J. Tumor angiogenesis: Therapeutic implications. N. Engl. J. Med. 1971;285:1182–1186. doi: 10.1056/NEJM197111182852108. [DOI] [PubMed] [Google Scholar]
- 3.Arnold F., West D.C. Angiogenesis in wound healing. Pharmacol. Ther. 1991;52:407–422. doi: 10.1016/0163-7258(91)90034-j. [DOI] [PubMed] [Google Scholar]
- 4.Reynolds L.P., Redmer D.A. Angiogenesis in the placenta. Biol. Reprod. 2001;64:1033–1040. doi: 10.1095/biolreprod64.4.1033. [DOI] [PubMed] [Google Scholar]
- 5.Folkman J. Angiogenesis in cancer, vascular, rheumatoid and other disease. Nat. Med. 1995;1:27–31. doi: 10.1038/nm0195-27. [DOI] [PubMed] [Google Scholar]
- 6.Hanahan D., Folkman J. Patterns and emerging mechanisms of the angiogenic switch during tumorigenesis. Cell. 1996;86:353–364. doi: 10.1016/s0092-8674(00)80108-7. [DOI] [PubMed] [Google Scholar]
- 7.Skobe M., Rockwell P., Goldstein N., Vosseler S., Fusenig N.E. Halting angiogenesis suppresses carcinoma cell invasion. Nat. Med. 1997;3:1222–1227. doi: 10.1038/nm1197-1222. [DOI] [PubMed] [Google Scholar]
- 8.Sullivan R., Graham C.H. Hypoxia-driven selection of the metastatic phenotype. Cancer Metastasis. Rev. 2007;26:319–331. doi: 10.1007/s10555-007-9062-2. [DOI] [PubMed] [Google Scholar]
- 9.Ferrara N. Role of vascular endothelial growth factor. Am. J. Physiol. Cell Physiol. 2001;280:C1358–C1366. doi: 10.1152/ajpcell.2001.280.6.C1358. [DOI] [PubMed] [Google Scholar]
- 10.Ferrara N., Gerber H.P., LeCouter J. The biology of VEGF and its receptors. Nat. Med. 2003;9:666–669. doi: 10.1038/nm0603-669. [DOI] [PubMed] [Google Scholar]
- 11.Tammela T., Enholm B., Alitalo K., Paavonen K. The biology of vascular endothelial growth factors. Cardiovasc. Res. 2005;65:550–563. doi: 10.1016/j.cardiores.2004.12.002. [DOI] [PubMed] [Google Scholar]
- 12.Kowanetz M., Ferrara N. Vascular endothelial growth factor signaling pathways, therapeutic perspective. Clin. Cancer Res. 2006;12:5018–5022. doi: 10.1158/1078-0432.CCR-06-1520. [DOI] [PubMed] [Google Scholar]
- 13.Holderfield M.T., Hughes C.C. Crosstalk between vascular endothelial growth factor, notch, and transforming growth factor-beta in vascular morphogenesis. Circ. Res. 2008;102:637–652. doi: 10.1161/CIRCRESAHA.107.167171. [DOI] [PubMed] [Google Scholar]
- 14.Bergers G., Hanahan D. Modes of resistance to anti-angiogenic therapy. Nat. Rev. Cancer. 2008;8:592–603. doi: 10.1038/nrc2442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Carmeliet P., Ng Y.S., Nuyens D., Theilmeier G., Brusselmans K., Cornelissen I., Ehler E., Kakkar V.V., Stalmans I., Mattot V., et al. Impaired myocardial angiogenesis and ischemic cardiomyopathy in mice lacking the vascular endothelial growth factor isoforms VEGF(164) and VEGF(188) Nat. Med. 1999;5:495–502. doi: 10.1038/8379. [DOI] [PubMed] [Google Scholar]
- 16.Valtola R., Salven P., Heikkilä P., Taipale J., Joensuu H., Rehn M., Pihlajaniemi T., Weich H., de Waal R., Alitalo K. VEGFR-3 and its ligand VEGF-C are associated with angiogenesis in breast cancer. Am. J. Pathol. 1999;154:1381–1390. doi: 10.1016/S0002-9440(10)65392-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Roskoski R. Vascular endothelial growth factor (VEGF) signaling in tumor progression. Crit. Rev. Oncol. Hematol. 2007;62:179–213. doi: 10.1016/j.critrevonc.2007.01.006. [DOI] [PubMed] [Google Scholar]
- 18.Ruohola J.K., Valve E.M., Karkkainen M.J., Joukov V., Alitalo K., Härkönen P.L. Vascular endothelial growth factors are differentially regulated by steroid hormones and antiestrogens in breast cancer cells. Mol. Cell Endocrinol. 1999;149:29–40. doi: 10.1016/s0303-7207(99)00003-9. [DOI] [PubMed] [Google Scholar]
- 19.Dumont N., Arteaga C.L. Transforming growth factor-beta and breast cancer—Tumor promoting effects of transforming growth factor-beta. Breast Cancer Res. 2000;2:125–132. doi: 10.1186/bcr44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Gerber H.P., McMurtrey A., Kowalski J., Yan M., Keyt B.A., Dixit V., Ferrara N. Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 3′-kinase/Akt signal transduction pathway. Requirement for Flk-1/KDR activation. Requirement for Flk-1/KDR activation. J. Biol. Chem. 1998;273:30336–30343. doi: 10.1074/jbc.273.46.30336. [DOI] [PubMed] [Google Scholar]
- 21.Dor Y., Porat R., Keshet E. Vascular endothelial growth factor and vascular adjustments to perturbations in oxygen homeostasis. Am. J. Physiol. Cell Physiol. 2001;280:C1367–C1374. doi: 10.1152/ajpcell.2001.280.6.C1367. [DOI] [PubMed] [Google Scholar]
- 22.Infanger M., Faramarzi S., Grosse J., Kurth E., Ulbrich C., Bauer J., Wehland M., Kreutz R., Kossmehl P., Paul M., et al. Expression of vascular endothelial growth factor and receptor tyrosine kinases in cardiac ischemia/reperfusion injury. Cardiovasc. Pathol. 2007;16:291–299. doi: 10.1016/j.carpath.2007.04.001. [DOI] [PubMed] [Google Scholar]
- 23.Semenza G. Signal transduction to hypoxia-inducible factor 1. Biochem. Pharmacol. 2002;64:993–998. doi: 10.1016/s0006-2952(02)01168-1. [DOI] [PubMed] [Google Scholar]
- 24.Ferrara N., Davis-Smyth T. The biology of vascular endothelial growth factor. Endocr. Rev. 1997;18:4–25. doi: 10.1210/edrv.18.1.0287. [DOI] [PubMed] [Google Scholar]
- 25.Herbert S.P., Stainier D.Y. Molecular control of endothelial cell behavior during blood vessel morphogenesis. Nat. Rev. 2011;12:551–564. doi: 10.1038/nrm3176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Shibuya M., Yamaguchi S., Yamane A., Ikeda T., Tojo A., Matsushime H., Sato M. Nucleotide sequence and expression of a novel human receptor-type tyrosine kinase gene (flt) closely related to the fms family. Oncogene. 1990;5:519–524. [PubMed] [Google Scholar]
- 27.Terman B.I., Carrion M.E., Kovacs E., Rasmussen B.A., Eddy R.L., Shows T.B. Identification of a new endothelial cell growth factor receptor tyrosine kinase. Oncogene. 1991;6:1677–1683. [PubMed] [Google Scholar]
- 28.Karkkainen M.J., Mäkinen T., Alitalo K. Lymphatic endothelium, a new frontier of metastasis research. Nat. Cell Biol. 2002;4:E2–E5. doi: 10.1038/ncb0102-e2. [DOI] [PubMed] [Google Scholar]
- 29.Ferrara N. Vascular endothelial growth factor, basic science and clinical progress. Endocr. Rev. 2004;25:581–611. doi: 10.1210/er.2003-0027. [DOI] [PubMed] [Google Scholar]
- 30.Alitalo K., Tammela T., Petrova T.V. Lymphangiogenesis in development and human disease. Nature. 2005;438:946–953. doi: 10.1038/nature04480. [DOI] [PubMed] [Google Scholar]
- 31.Paavonen K., Puolakkainen P., Jussila L., Jahkola T., Alitalo K. Vascular endothelial growth factor receptor-3 in lymphangiogenesis in wound healing. Am. J. Pathol. 2000;156:1499–1504. doi: 10.1016/S0002-9440(10)65021-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tammela T., Zarkada G., Nurmi H., Jakobsson L., Heinolainen K., Tvorogov D., Zheng W., Franco C.A., Murtomäki A., Aranda E., et al. VEGFR-3 controls tip to stalk conversion at vessel fusion sites by reinforcing Notch signalling. Nat. Cell Biol. 2011;3:1202–1213. doi: 10.1038/ncb2331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Takahashi H., Shibuya M. The vascular endothelial growth factor (VEGF)/VEGF receptor system and its role under physiological and pathological conditions. Clin. Sci. 2005;109:227–241. doi: 10.1042/CS20040370. [DOI] [PubMed] [Google Scholar]
- 34.Olsson A.K., Dimberg A., Kreuger J., Claesson-Welsh L. VEGF receptor signaling—In control of vascular function. Nat. Rev. Mol. Cell Biol. 2006;7:359–371. doi: 10.1038/nrm1911. [DOI] [PubMed] [Google Scholar]
- 35.Park J.E., Chen H.H., Winer J., Houck K.A., Ferrara N. Placenta growth factor. Potentiation of vascular endothelial growth factor bioactivity, in vitro and in vivo, and high affinity binding to Flt-1 but not to Flk-1/KDR. J. Biol. Chem. 1994;269:25646–25654. [PubMed] [Google Scholar]
- 36.Hiratsuka S., Minowa O., Kuno J., Noda T., Shibuya M. Flt-1 lacking the tyrosine kinase domain is sufficient for normal development and angiogenesis in mice. Proc. Natl. Acad. Sci. USA. 1998;4:9349–9354. doi: 10.1073/pnas.95.16.9349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Infanger M., Schmidt O., Kossmehl P., Grad S., Ertel W., Grimm D. Vascular endothelial growth factor serum level is strongly enhanced after burn injury and correlated with local and general tissue edema. Burns. 2004;30:305–311. doi: 10.1016/j.burns.2003.12.006. [DOI] [PubMed] [Google Scholar]
- 38.Infanger M., Shakibaei M., Kossmehl P., Hollenberg S.M., Grosse J., Faramarzi S., Schulze-Tanzil G., Paul M., Grimm D. Intraluminal application of vascular endothelial growth factor enhances healing of microvascular anastomosis in a rat model. J. Vasc. Res. 2005;42:202–213. doi: 10.1159/000085176. [DOI] [PubMed] [Google Scholar]
- 39.Infanger M., Grosse J., Westphal K., Leder A., Ulbrich C., Paul M., Grimm D. Vascular Endothelial Growth Factor induces extracellular matrix proteins and osteopontin in the umbilical artery. Ann. Vasc. Surg. 2008;22:273–284. doi: 10.1016/j.avsg.2007.11.002. [DOI] [PubMed] [Google Scholar]
- 40.Alon T., Hemo I., Itin A., Pe’er J., Stone J., Keshet E. Vascular endothelial growth factor acts as a survival factor for newly formed retinal vessels and has implications for retinopathy of prematurity. Nat. Med. 1995;1:1024–1028. doi: 10.1038/nm1095-1024. [DOI] [PubMed] [Google Scholar]
- 41.Gerber H.P., Dixit V., Ferrara N. Vascular Endothelial Growth Factor induces expression of the antiapoptotic proteins Bcl-2 and A1 in vascular endothelial cells. J. Biol. Chem. 1998;273:13313–13316. doi: 10.1074/jbc.273.21.13313. [DOI] [PubMed] [Google Scholar]
- 42.Benjamin L.E., Golijanin D., Itin A., Pode D., Keshet E. Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J. Clin. Invest. 1999;103:159–165. doi: 10.1172/JCI5028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Infanger M., Kossmehl P., Shakibaei M., Baatout S., Witzing A., Grosse J., Bauer J., Cogoli A., Faramarzi S., Derradji H., et al. Induction of three-dimensional assembly and increase in apoptosis of human endothelial cells by simulated microgravity. Impact of vascular endothelial growth factor. Apoptosis. 2006;11:749–764. doi: 10.1007/s10495-006-5697-7. [DOI] [PubMed] [Google Scholar]
- 44.Ferrara N., Hillan K., Gerber H.P., Novotny W. Discovery and development of Bevacizumab, an anti VEGF antibody for treating cancer. Nat. Rev. Drug Discov. 2004;3:391–398. doi: 10.1038/nrd1381. [DOI] [PubMed] [Google Scholar]
- 45.Reck M., von Pawel J., Zatloukal P., Ramlau R., Gorbounova V., Hirsh V., Leighl N., Mezger J., Archer V., Moore N., et al. BO17704 Study Group. Overall survival with cisplatin-gemcitabina and Bevacizumab or placebo as first line therapy for non-squamous NSCLC: Results from a randomized phase III trial (AVAiL) Ann. Oncol. 2010;21:1804–1809. doi: 10.1093/annonc/mdq020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Sandler A., Gray R., Perry M.C., Brahmer J., Schiller J.H., Dowlati A., Lilenbaum R., Johnson D.H. Paclitaxel-Carboplatin alone or with Bevacizumab for non-small-cell lung cancer. N. Engl. J. Med. 2006;355:2542–2550. doi: 10.1056/NEJMoa061884. [DOI] [PubMed] [Google Scholar]
- 47.Johnson D.H., Fehrenbacher L., Novotny W.F., Herbst R.S., Nemunaitis J.J., Jablons D.M., Langer C.J., DeVore R.F., 3rd, Gaudreault J., Damico L.A., et al. Randomized phase II trial comparing Bevacizumab plus Carboplatin and Paclitaxel with Carboplatin and Paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J. Clin. Oncol. 2004;22:2184–2191. doi: 10.1200/JCO.2004.11.022. [DOI] [PubMed] [Google Scholar]
- 48.Herbst R.S., O’Neill V.J., Fehrenbacher L., Belani C.P., Bonomi P.D., Hart L., Melnyk O., Ramies D., Lin M., Sandler A. Phase II study of efficacy and safety of Bevacizumab in combination with chemotherapy or Erlotinib compared with chemotherapy alone for treatment of recurrent or refractory non-small-cell lung cancer. J. Clin. Oncol. 2007;25:4743–4750. doi: 10.1200/JCO.2007.12.3026. [DOI] [PubMed] [Google Scholar]
- 49.Miller K.D., Chap L.I., Holmes F.A., Cobleigh M.A., Marcom P.K., Fehrenbacher L., Dickler M., Overmoyer B.A., Reimann J.D., Sing A.P., et al. Randomized phase III trial of Capecitabine compared with Bevacizumab plus Capecitabine in patient with previously treated metastatic breast cancer. J. Clin. Oncol. 2005;23:792–799. doi: 10.1200/JCO.2005.05.098. [DOI] [PubMed] [Google Scholar]
- 50.Miller K., Wang M., Gralow J., Dickler M., Cobleigh M., Perez E.A., Shenkier T., Cella D., Davidson N.E. Paclitaxel with Bevacizumab versus Paclitaxel alone in metastatic breast cancer. N. Engl. J. Med. 2007;357:2666–2676. doi: 10.1056/NEJMoa072113. [DOI] [PubMed] [Google Scholar]
- 51.Robert N.J., Dieras V., Glaspy J., Brufsky A., Bondarenko I., Lipatov O., Perez E., Yardley D., Zhou X., Phan S. RIBBON-1, randomized, double blind, placebo controlled phase III trial of chemotherapy with or without Bevacizumab for first line treatment of HER2 negative locally recurrent or metastatic breast cancer. ASCO Present. J. Clin. Oncol. 2009;27:15s. doi: 10.1200/JCO.2010.28.0982. [DOI] [PubMed] [Google Scholar]
- 52.Miles D.W., Chan A., Dirix L.Y., Cortés J., Pivot X., Tomczak P., Delozier T., Sohn J.H., Provencher L., Puglisi F., et al. Phase III study of Bevacizumab plus Docetaxel campared with placebo plus docetaxel for the first line treatment of human epidermal growth factor receptor-2-negative metastatic breast cancer. J. Clin. Oncol. 2010;28:3239–3247. doi: 10.1200/JCO.2008.21.6457. [DOI] [PubMed] [Google Scholar]
- 53.Brufsky A.M., Hurvitz S., Perez E., Swamy R., Valero V., O’Neill V., Rugo H.S. RIBBON-2, a randomized, double-blind, placebo-controlled, phase III trial evaluating the efficacy and safety of Bevacizumab in combination with chemotherapy for second-line treatment of human epidermal growth factor receptor 2-negative metastatic breast cancer. J. Clin. Oncol. 2011;29:4286–4293. doi: 10.1200/JCO.2010.34.1255. [DOI] [PubMed] [Google Scholar]
- 54.Kabbinavar F., Hurwitz H.I., Fehrenbacher L., Meropol N.J., Novotny W.F., Lieberman G., Griffing S., Bergsland E. Phase II randomized trial comparing Bevacizumab plus Fluorouracil(FU)/Leucovorin(LV) with FU/LV alone in patients with metastatic colorectal cancer. J. Clin. Oncol. 2003;21:60–65. doi: 10.1200/JCO.2003.10.066. [DOI] [PubMed] [Google Scholar]
- 55.Kabbinavar F.F., Schulz J., McCleod M., Patel T., Hamm J.T., Hecht J.R., Mass R., Perrou B., Nelson B., Novotny W.F. Addition of Bevacizumab to bolus Fluorouracil and Leucovorin in first line metastatic colorectal cancer, results of randomized phase II trial. J. Clin. Oncol. 2005;23:3697–3705. doi: 10.1200/JCO.2005.05.112. [DOI] [PubMed] [Google Scholar]
- 56.Hurwitz H., Fehrenbacher L., Novotny W., Cartwright T., Hainsworth J., Heim W., Berlin J., Baron A., Griffing S., Holmgren E., et al. Bevacizumab plus Irinotecan, Flouorouracil and Leucovorin for metastatic colorectal cancer. N. Engl. J. Med. 2004;350:2335–2342. doi: 10.1056/NEJMoa032691. [DOI] [PubMed] [Google Scholar]
- 57.Saltz L.B., Clarke S., Díaz-Rubio E., Scheithauer W., Figer A., Wong R., Koski S., Lichinitser M., Yang T.S., Rivera F., et al. Bevacizumab in combination with oxaliplatin based chemotherapy as first line therapy in metastatic colorectal cancer: A randomized phase III study. J. Clin. Oncol. 2008;26:2013–2019. doi: 10.1200/JCO.2007.14.9930. [DOI] [PubMed] [Google Scholar]
- 58.Tebbutt N.C., Wilson K., Gebski V.J., Cummins M.M., Zannino D., van Hazel G.A., Robinson B., Broad A., Ganju V., Ackland S.P., et al. Capecitabine, Bevacizumab and Mitomycin in first line treatment of metastatic colorectal cancer: Results of the Australian gastrointestinal trials group randomized phase III MAX study. J. Clin. Oncol. 2010;28:3191–3198. doi: 10.1200/JCO.2009.27.7723. [DOI] [PubMed] [Google Scholar]
- 59.Rini B.I., Halabi S., Rosenberg J.E., Stadler W.M., Vaena D.A., Archer L., Atkins J.N., Picus J., Czaykowski P., Dutcher J., et al. Phase III trial of Bevacizumab plus interferon alfa versus interferon alfa monotherapy in patients with metastatic renal cell carcinoma: Final results of CALGB 90206. J. Clin. Oncol. 2010;28:1–7. doi: 10.1200/JCO.2009.26.5561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Escudier B., Pluzanska A., Koralewski P., Ravaud A., Bracarda S., Szczylik C., Chevreau C., Filipek M., Melichar B., Bajetta E., et al. AVOREN Trial investigators. Bevacizumab plus interferon alfa-2a for treatment of metastatic renal cell carcinoma: A randomized double blind phase III trial. Lancet. 2007;370:2103–2111. doi: 10.1016/S0140-6736(07)61904-7. [DOI] [PubMed] [Google Scholar]
- 61.Ohtsu A., Shah M.A., van Cutsem E., Rha S.Y., Sawaki A., Park S.R., Lim H.Y., Yamada Y., Wu J., Langer B., et al. Bevacizumab in combination with chemotherapy as first-line therapy in advanced gastric cancer: A randomized, double-blind, placebo-controlled phase III study. J. Clin. Oncol. 2011;29:3968–3976. doi: 10.1200/JCO.2011.36.2236. [DOI] [PubMed] [Google Scholar]
- 62.Kindler H.L., Niedzwiecki D., Hollis D., Sutherland S., Schrag D., Hurwitz H., Innocenti F., Mulcahy M.F., O’Reilly E., Wozniak T.F., et al. Gemcitabine plus Bevacizumab compared with Gemcitabine plus placebo in patients with advanced pancreatic cancer: Phase III trial of the cancer and leukemia group B (CALGB 80303) J. Clin. Oncol. 2010;28:3617. doi: 10.1200/JCO.2010.28.1386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Van Cutsem E., Vervenne W.L., Bennouna J., Humblet Y., Gill S., van Laethem J.L., Verslype C., Scheithauer W., Shang A., Cosaert J., et al. Phase III trial of Bevacizumab in combination with Gemcitabine and Erlotinib in patients with metastatic pancreatic cancer. J. Clin. Oncol. 2009;27:2231–2237. doi: 10.1200/JCO.2008.20.0238. [DOI] [PubMed] [Google Scholar]
- 64.Kelly W.K., Halabi S., Carducci M., George D., Mahoney J.F., Stadler W.M., Morris M., Kantoff P., Monk J.P., Kaplan E., et al. Randomized, double-blind, placebo-controlled phase III trial comparing docetaxel and prednisone with or without bevacizumab in men with metastatic castration-resistant prostate cancer: CALGB 90401. J. Clin. Oncol. 2012;30:1534–1540. doi: 10.1200/JCO.2011.39.4767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kim K.B., Sosman J.A., Fruehauf J.P., Linette G.P., Markovic S.N., McDermott D.F., Weber J.S., Nguyen H., Cheverton P., Chen D., et al. BEAM: A randomized phase II study evaluating the activity of Bevacizumab in combination with Carboplatinum plus Paclitaxel in patients with previously untreated advanced melanoma. J. Clin. Oncol. 2012;30:34–41. doi: 10.1200/JCO.2011.34.6270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Tew W.P., Colombo N., Ray-Coquard I., Oza A., del Campo J., Scambia G., Spriggs D. VEGF-Trap for patients (pts) with recurrent platinum-resistant epithelial ovarian cancer (EOC), preliminary results of a randomized, multicenter phase II study. ASCO Meet. Abstr. 2007;25:5508. [Google Scholar]
- 67.Tang P., Cohen S.J., Bjarnason G.A., Kollmannsberger C., Virik K., MacKenzie M.J., Brown J., Wang L., Chen A.P., Moore M.J. Phase II trial of aflibercept (VEGF Trap) in previously treated patients with metastatic colorectal cancer (MCRC): A PMH phase II consortium trial. ASCO Meet. Abstr. 2008;26:4027. [Google Scholar]
- 68.Massarelli E., Miller V.A., Leighl N.B., Rosen P.J., Albain K.S., Hart L.L., Melnyk O., Sternas L., Ackerman J., Herbst R.S. Phase II study of the efficacy and safety of intravenous (IV) AVE0005 (VEGF Trap) given every 2 weeks in patients (Pts) with platinum- and erlotinib-resistant adenocarcinoma of the lung (NSCLA) ASCO Meet. Abstr. 2007;25:7627. [Google Scholar]
- 69.Townsley C., Hirte H., Hoskins P., Buckanovich R., Mackay H., Welch S., Wang L., Polintan R., Chen A., Oza A.M. A phase II study of aflibercept (VEGF trap) in recurrent or metastatic gynecologic soft-tissue sarcomas: A study of the Princess Margaret Hospital Phase II Consortium. ASCO Meet. Abstr. 2009;27:5591. [Google Scholar]
- 70.Twardowski P., Stadler W.M., Frankel P., Lara P.N., Ruel C., Chatta G., Heath E.I., Quinn D.I., Gandara D.R. Phase II study of aflibercept (VEGFTrap) in patients with recurrent or metastatic transitional cell carcinoma (TCC) of the urothelium: A California Cancer Consortium trial. ASCO Meet. Abstr. 2009;27:e16030. doi: 10.1016/j.urology.2010.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Tarhini A.A., Christensen S., Frankel P., Margolin K., Ruel C., Shipe-Spotloe J., DeMark M., Kirkwood J.M. Phase II study of aflibercept (VEGF trap) in recurrent inoperable stage III or stage IV melanoma of cutaneous or ocular origin. ASCO Meet. Abstr. 2009;27:9028. [Google Scholar]
- 72.De Groot J.F., Wen P.Y., Lamborn K., Chang S., Cloughesy T.F., Chen A.P., DeAngelis L.M., Mehta M.P., Gilbert M.R., Yung W.K., et al. Phase II single arm trial of aflibercept in patients with recurrent temozolomide-resistant glioblastoma: NABTC 0601. ASCO Meet. Abstr. 2008;26:2020. [Google Scholar]
- 73.Lu D., Jimenez X., Zhang H., Bohlen P., Witte L., Zhu Z. Selection of high affinity human neutralizing antibodies to VEGFR2 from a large antibody phage display library for antiangiogenesis therapy. Int. J. Cancer. 2002;97:393–399. doi: 10.1002/ijc.1634. [DOI] [PubMed] [Google Scholar]
- 74.Spratlin J. Ramucirumab (IMC-1121B), Monoclonal antibody inhibition of vascular endothelial growth factor receptor-2. Curr. Oncol. Rep. 2011;13:97–102. doi: 10.1007/s11912-010-0149-5. [DOI] [PubMed] [Google Scholar]
- 75.Wood L. Sunitinib malate for the treatment of renal cell carcinoma. Expert Opin. Pharmacother. 2012;13:1323–1336. doi: 10.1517/14656566.2012.689130. [DOI] [PubMed] [Google Scholar]
- 76.Sekkate S., Kairouani M., Abahssain H., Serji B., Boutayeb S., Mrabti H., Errihani H. Gastrointestinal stromal tumors. Presse. Med. 2012;41:917–926. doi: 10.1016/j.lpm.2011.11.020. [DOI] [PubMed] [Google Scholar]
- 77.Escudier B., Eisen T., Stadler W.M., Szczylik C., Oudard S., Staehler M., Negrier S., Chevreau C., Desai A.A., Rolland F., et al. Sorafenib for treatment of renal cell carcinoma, Final efficacy and safety results of the phase III treatment approaches in renal cancer global evaluation trial. J. Clin. Oncol. 2009;27:3312–3318. doi: 10.1200/JCO.2008.19.5511. [DOI] [PubMed] [Google Scholar]
- 78.Zhu A.X. Development of sorafenib and other molecularly targeted agents in hepatocellular carcinoma. Cancer. 2008;112:250–259. doi: 10.1002/cncr.23175. [DOI] [PubMed] [Google Scholar]
- 79.Scagliotti G.V., Vynnychenko I., Park K., Ichinose Y., Kubota K., Blackhall F., Pirker R., Galiulin R., Ciuleanu T.E., Sydorenko O., et al. International, randomized, placebo-controlled, double-blind phase III study of motesanib plus carboplatin/paclitaxel in patients with advanced nonsquamous non-small-cell lung cancer: MONET1. J. Clin. Oncol. 2012;30:2829–2836. doi: 10.1200/JCO.2011.41.4987. [DOI] [PubMed] [Google Scholar]
- 80.Coxon A., Bready J., Kaufman S., Estrada J., Osgood T., Canon J., Wang L., Radinsky R., Kendall R., Hughes P., et al. Anti-tumor activity of motesanib in a medullary thyroid cancer model. J. Endocrinol. Invest. 2012;35:181–190. doi: 10.3275/7609. [DOI] [PubMed] [Google Scholar]
- 81.Gennigens C., Jerusalem G. Pazopanib (Votrient) in the management of renal cell cancer and soft tissue sarcomas. Rev. Med. Liege. 2012;67:437–442. [PubMed] [Google Scholar]
- 82.Grimm D., Wise P., Lebert M., Richter P., Baatout S. How and why does the proteome respond to microgravity? Expert Rev. Proteomics. 2011;8:13–27. doi: 10.1586/epr.10.105. [DOI] [PubMed] [Google Scholar]
- 83.Pietsch J., Bauer J., Egli M., Infanger M., Wise P., Ulbrich C., Grimm D. The effects of weightlessness on the human organism and mammalian cells. Curr. Mol. Med. 2011;11:350–364. doi: 10.2174/156652411795976600. [DOI] [PubMed] [Google Scholar]
- 84.Grimm D., Infanger M., Westphal K., Ulbrich C., Pietsch J., Kossmehl P., Vadrucci S., Baatout S., Flick B., Paul M., et al. A delayed type of three-dimensional growth of human endothelial cells under simulated weightlessness. Tissue Eng. Part. A. 2009;15:2267–2275. doi: 10.1089/ten.tea.2008.0576. [DOI] [PubMed] [Google Scholar]
- 85.Grimm D., Bauer J., Ulbrich C., Westphal K., Wehland M., Infanger M., Aleshcheva G., Pietsch J., Ghardi M., Beck M., et al. Different responsiveness of endothelial cells to vascular endothelial growth factor and basic fibroblast growth factor added to culture media under gravity and simulated microgravity. Tissue Eng. Part A. 2010;16:1559–1573. doi: 10.1089/ten.TEA.2009.0524. [DOI] [PubMed] [Google Scholar]
- 86.De Gruttola V.G., Clax P., DeMets D.L., Downing G.J., Ellenberg S.S., Friedman L., Gail M.H., Prentice R., Wittes J., Zeger S.L. Considerations in the evaluation of surrogate endpoints in clinical trials. Summary of a National Institutes of Health workshop. Control. Clin. Trials. 2001;22:485–502. doi: 10.1016/s0197-2456(01)00153-2. [DOI] [PubMed] [Google Scholar]
- 87.Oldenhuis C.N., Oosting S.F., Gietema J.A., de Vries E.G. Prognostic versus predictive value of biomarkers in oncology. Eur. J. Cancer. 2008;44:946–953. doi: 10.1016/j.ejca.2008.03.006. [DOI] [PubMed] [Google Scholar]
- 88.McShane L.M., Altman D.G., Sauerbrei W., Taube S.E., Gion M., Clark G.M. Statistics Subcommittee of NCI-EORTC Working Group on Cancer Diagnostics. Reporting recommendations for tumor MARKer prognostic studies (REMARK) Breast Cancer Res. Treat. 2006;100:229–235. doi: 10.1007/s10549-006-9242-8. [DOI] [PubMed] [Google Scholar]
- 89.Ferlay J., Shin H.R., Bray F., Forman D., Mathers C., Parkin D.M. GLOBOCAN 2008 v1.2, Cancer Incidence and Mortality Worldwide, IARC CancerBase No. 10 [Internet] Lyon, France: International Agency for Research on Cancer; 2010. [(accessed on 15 February 2013)]. Available online: http://globocan.iarc.fr. [Google Scholar]
- 90.Ince W.L., Jubb A.M., Holden S.N., Holmgren E.B., Tobin P., Sridhar M., Hurwitz H.I., Kabbinavar F., Novotny W.F., Hillan K.J., et al. Association of k-ras, b-raf, and p53 status with the treatment effect of Bevacizumab. J. Natl. Cancer Inst. 2005;97:981–989. doi: 10.1093/jnci/dji174. [DOI] [PubMed] [Google Scholar]
- 91.Jubb A.M., Hurwitz H.I., Bai W., Holmgren E.B., Tobin P., Guerrero A.S., Kabbinavar F., Holden S.N., Novotny W.F., Frantz G.D., et al. Impact of vascular endothelial growth factor-A expression, thrombospondin-2 expression, and microvessel density on the treatment effect of Bevacizumab in metastatic colorectal cancer. J. Clin. Oncol. 2006;24:217–227. doi: 10.1200/JCO.2005.01.5388. [DOI] [PubMed] [Google Scholar]
- 92.Cetin B., Kaplan M.A., Berk V., Ozturk S.C., Benekli M., Isıkdogan A., Ozkan M., Coskun U., Buyukberber S. Prognostic factors for overall survival in patients with metastatic colorectal carcinoma treated with vascular endothelial growth factor-targeting agents. Asian Pac. J. Cancer Prev. 2012;13:1059–1063. doi: 10.7314/apjcp.2012.13.3.1059. [DOI] [PubMed] [Google Scholar]
- 93.Kopetz S., Hoff P.M., Morris J.S., Wolff R.A., Eng C., Glover K.Y., Adinin R., Overman M.J., Valero V., Wen S., et al. Phase II trial of infusional fluorouracil, Irinotecan, and Bevacizumab for metastatic colorectal cancer, efficacy and circulating angiogenic biomarkers associated with therapeutic resistance. J. Clin. Oncol. 2010;28:453–459. doi: 10.1200/JCO.2009.24.8252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Goede V., Coutelle O., Neuneier J., Reinacher-Schick A., Schnell R., Koslowsky T.C., Weihrauch M.R., Cremer B., Kashkar H., Odenthal M., et al. Identification of serum angiopoietin-2 as a biomarker for clinical outcome of colorectal cancer patients treated with Bevacizumab-containing therapy. Br. J. Cancer. 2010;103:1407–1414. doi: 10.1038/sj.bjc.6605925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Matsusaka S., Suenaga M., Mishima Y., Takagi K., Terui Y., Mizunuma N., Hatake K. Circulating endothelial cells predict for response to Bevacizumab-based chemotherapy in metastatic colorectal cancer. Cancer Chemother. Pharmacol. 2011;68:763–768. doi: 10.1007/s00280-010-1543-2. [DOI] [PubMed] [Google Scholar]
- 96.Matsusaka S., Mishima Y., Suenaga M., Terui Y., Kuniyoshi R., Mizunuma N., Hatake K. Circulating endothelial progenitors and CXCR4-positive circulating endothelial cells are predictive markers for Bevacizumab. Cancer. 2011;117:4026–4032. doi: 10.1002/cncr.25977. [DOI] [PubMed] [Google Scholar]
- 97.Ronzoni M., Manzoni M., Mariucci S., Loupakis F., Brugnatelli S., Bencardino K., Rovati B., Tinelli C., Falcone A., Villa E., et al. Circulating endothelial cells and endothelial progenitors as predictive markers of clinical response to Bevacizumab-based first-line treatment in advanced colorectal cancer patients. Ann. Oncol. 2010;21:2382–2389. doi: 10.1093/annonc/mdq261. [DOI] [PubMed] [Google Scholar]
- 98.Manzoni M., Mariucci S., Delfanti S., Rovati B., Ronzoni M., Loupakis F., Brugnatelli S., Tinelli C., Villa E., Falcone A., et al. Circulating endothelial cells and their apoptotic fraction are mutually independent predictive biomarkers in Bevacizumab-based treatment for advanced colorectal cancer. J. Cancer Res. Clin. Oncol. 2012;138:1187–1196. doi: 10.1007/s00432-012-1190-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Singh H., Pohl A., El-Khoueiry A., Lurje G., Zhang W., Yang D., Ning Y., Shriki J., Iqbal S., Lenz H. Use of genetic variants to predict clinical outcome in patients (pts) with metastatic colorectal cancer (mCRC) treated with first-line 5-FU or capecitabine in combination with oxaliplatin and Bevacizumab (FOLFOX/BV or XELOX/BV) J. Clin. Oncol. 2009;27:15s. [Google Scholar]
- 100.Hansen T.F., Christensen R.D., Andersen R.F., Garm Spindler K.L., Johnsson A., Jakobsen A. The predictive value of single nucleotide polymorphisms in the VEGF system to the efficacy of first-line treatment with Bevacizumab plus chemotherapy in patients with metastatic colorectal cancer: Results from the Nordic ACT trial. Int. J. Colorectal Dis. 2012;27:715–720. doi: 10.1007/s00384-011-1382-6. [DOI] [PubMed] [Google Scholar]
- 101.O’Brien C.A., Pollett A., Gallinger S., Dick J.E. A human colon cancer cell capable of initiating tumour growth in immunodeficient mice. Nature. 2007;445:106–110. doi: 10.1038/nature05372. [DOI] [PubMed] [Google Scholar]
- 102.Ricci-Vitiani L., Lombardi D.G., Pilozzi E., Biffoni M., Todaro M., Peschle C., De Maria R. Identification and expansion of human colon-cancer-initiating cells. Nature. 2007;445:111–115. doi: 10.1038/nature05384. [DOI] [PubMed] [Google Scholar]
- 103.Pohl A., Zhang W., Yang D., Lurje G., Ning Y., Khambata-Ford S., Langer C., Kahn M., Teo J.L., Lenz H.J. Association of CD133 polymorphisms and clinical outcome in metastatic colorectal cancer (mCRC) patients (pts) treated with either first-line 5-FU + Bevacizumab (BV) or second-line Irinotecan (IR)/Cetuximab (CB) or IR alone. J. Clin. Oncol. 2009;27:15s. [Google Scholar]
- 104.Hurwitz H.I., Douglas P.S., Middleton J.P., Sledge G.W., Johnson D.H., Reardon D.A., Chen D., Rosen O. Analysis of early hypertension and clinical outcome with bevacizumab: results from seven phase iii studies. Oncologist. 2013;18:273–280. doi: 10.1634/theoncologist.2012-0339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Jubb A.M., Harris A.L. Biomarkers to predict the clinical efficacy of Bevacizumab in cancer. Lancet Oncol. 2010;11:1172–1183. doi: 10.1016/S1470-2045(10)70232-1. [DOI] [PubMed] [Google Scholar]
- 106.Scartozzi M., Galizia E., Chiorrini S., Giampieri R., Berardi R., Pierantoni C., Cascinu S. Arterial hypertension correlates with clinical outcome in colorectal cancer patients treated with first-line Bevacizumab. Ann. Oncol. 2009;20:227–230. doi: 10.1093/annonc/mdn637. [DOI] [PubMed] [Google Scholar]
- 107.Österlund P., Soveri L.M., Isoniemi H., Poussa T., Alanko T., Bono P. Hypertension and overall survival in metastatic colorectal cancer patients treated with Bevacizumab-containing chemotherapy. Br. J. Cancer. 2011;104:599–604. doi: 10.1038/bjc.2011.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.De Stefano A., Carlomagno C., Pepe S., Bianco R., de Placido S. Bevacizumab-related arterial hypertension as a predictive marker in metastatic colorectal cancer patients. Cancer Chemother. Pharmacol. 2011;68:1207–1213. doi: 10.1007/s00280-011-1604-1. [DOI] [PubMed] [Google Scholar]
- 109.Wehland M., Bauer J., Infanger M., Grimm D. Target-based anti-angiogenic therapy in breast cancer. Curr. Pharm. Des. 2012;18:4244–4257. doi: 10.2174/138161212802430468. [DOI] [PubMed] [Google Scholar]
- 110.Burstein H.J., Chen Y.H., Parker L.M., Savoie J., Younger J., Kuter I., Ryan P.D., Garber J.E., Chen H., Campos S.M., et al. VEGF as a marker for outcome among advanced breast cancer patients receiving anti-VEGF therapy with Bevacizumab and vinorelbine chemotherapy. Clin. Cancer Res. 2008;14:7871–7877. doi: 10.1158/1078-0432.CCR-08-0593. [DOI] [PubMed] [Google Scholar]
- 111.Baar J., Silverman P., Lyons J., Fu P., Abdul-Karim F., Ziats N., Wasman J., Hartman P., Jesberger J., Dumadag L., et al. A vasculature-targeting regimen of preoperative docetaxel with or without Bevacizumab for locally advanced breast cancer: Impact on angiogenic biomarkers. Clin. Cancer Res. 2009;15:3583–3590. doi: 10.1158/1078-0432.CCR-08-2917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Burstein H.J., Elias A.D., Rugo H.S., Cobleigh M.A., Wolff A.C., Eisenberg P.D., Lehman M., Adams B.J., Bello C.L., DePrimo S.E., et al. Phase II study of Sunitinib malate, an oral multitargeted tyrosine kinase inhibitor, in patients with metastatic breast cancer previously treated with an Anthracycline and a Taxane. J. Clin. Oncol. 2008;26:1810–1816. doi: 10.1200/JCO.2007.14.5375. [DOI] [PubMed] [Google Scholar]
- 113.Calleri A., Bono A., Bagnardi A., Quarna J., Mancuso P., Rabascio C., Dellapasqua S., Campagnoli E., Shaked Y., Goldhirsch A., et al. Predictive potential of angiogenic growth factors and circulating endothelial cells in breast cancer patients receiving metronomic chemotherapy plus Bevacizumab. Clin. Cancer Res. 2009;15:7652–7657. doi: 10.1158/1078-0432.CCR-09-1493. [DOI] [PubMed] [Google Scholar]
- 114.Schneider B.P., Wang M., Radovich M., Sledge G.W., Badve S., Thor A., Flockhart D.A., Hancock B., Davidson N., Gralow J., et al. ECOG 2100. Association of vascular endothelial growth factor and vascular endothelial growth factor receptor-2 genetic polymorphisms with outcome in a trial of paclitaxel compared with paclitaxel plus Bevacizumab in advanced breast cancer: ECOG 2100. J. Clin. Oncol. 2008;26:4672–4678. doi: 10.1200/JCO.2008.16.1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Hurwitz H., Douglas P.S., Middleton J.P.W., Sledge G., Johnson D.H., Reardon D.A., Chen D., Rosen O. Analysis of early hypertension (HTN) and clinical outcome with Bevacizumab (BV) J. Clin. Oncol. 2010;28:15s. doi: 10.1634/theoncologist.2012-0339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Fountzilas G., Kourea H.P., Bobos M., Televantou D., Kotoula V., Papadimitriou C., Papazisis K.T., Timotheadou E., Efstratiou I., Koutras A., et al. Paclitaxel and Bevacizumab as first line combined treatment in patients with metastatic breast cancer, the Hellenic Cooperative Oncology Group experience with biological marker evaluation. Anticancer Res. 2011;31:3007–3018. [PubMed] [Google Scholar]
- 117.Busnardo B., de Vido D. The epidemiology and etiology of differentiated thyroid carcinoma. Biomed. Pharmacother. 2000;54:322–326. doi: 10.1016/S0753-3322(00)80056-6. [DOI] [PubMed] [Google Scholar]
- 118.Okuieff P., Chen Y., Maguire D.J., Huser A.K. Molecular markers of radiation-related normal tissue toxicity. Cancer Metastasis Rev. 2008;27:363–374. doi: 10.1007/s10555-008-9138-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Gilliland F.D., Hunt W.C., Morris D.M., Key C.R. Prognostic factors for thyroid carcinoma. A population-based study of 15,698 cases from the Surveillance, Epidemiology and End Results (SEER) program 1973–1991. Cancer. 1997;79:564–573. doi: 10.1002/(sici)1097-0142(19970201)79:3<564::aid-cncr20>3.0.co;2-0. [DOI] [PubMed] [Google Scholar]
- 120.Durante C., Haddy N., Baudin E., Leboulleux S., Hartl D., Travagli J.P., Caillou B., Ricard M., Lumbroso J.D., de Vathaire F., et al. Long-term outcome of 444 patients with distant metastases from papillary and follicular thyroid carcinoma, benefits and limits of radioiodine therapy. J. Clin. Endocrinol. Metab. 2006;91:2892–2899. doi: 10.1210/jc.2005-2838. [DOI] [PubMed] [Google Scholar]
- 121.Ain K.B., Lee C., Williams K.D. Phase II trial of thalidomide for therapy of radioiodine-unresponsive and rapidly progressive thyroid carcinomas. Thyroid. 2007;17:663–670. doi: 10.1089/thy.2006.0289. [DOI] [PubMed] [Google Scholar]
- 122.Schönberger J., Bauer J., Spruss T., Weber G., Chahoud I., Eilles C., Grimm D. Establishment and characterization of the follicular thyroid carcinoma cell line ML-1. J. Mol. Med. 2000;78:102–110. doi: 10.1007/s001090000085. [DOI] [PubMed] [Google Scholar]
- 123.Schoenberger J., Grimm D., Kossmehl P., Infanger M., Kurth E., Eilles C. Effects of PTK787/ZK222584, a tyrosine kinase inhibitor, on the growth of a poorly differentiated thyroid carcinoma, an animal study. Endocrinology. 2004;145:1031–1038. doi: 10.1210/en.2003-1228. [DOI] [PubMed] [Google Scholar]
- 124.Niedzwiecki S., Stepien T., Kopec K., Kuzdak K., Komorowski J., Krupinski R., Stepien H. Angiopoietin 1 (Ang-1), angiopoietin 2 (Ang-2) and Tie-2 (a receptor tyrosine kinase) concentrations in peripheral blood of patients with thyroid cancers. Cytokine. 2006;36:291–295. doi: 10.1016/j.cyto.2007.02.008. [DOI] [PubMed] [Google Scholar]
- 125.Liang H., Zhong Y., Luo Z., Huang Y., Lin H., Zhan S., Xie K., Li Q.Q. Diagnostic value of 16 cellular tumor markers for metastatic thyroid cancer, an immunohistochemical study. Anticancer Res. 2011;31:3433–3440. [PubMed] [Google Scholar]
- 126.Antonelli A., Fallahi P., Ferrari S.M., Ruffilli I., Santini F., Minuto M., Galleri D., Miccoli P. New targeted therapies for thyroid cancer. Curr. Genomics. 2011;12:626–631. doi: 10.2174/138920211798120808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Schlumberger M.J., Elisei R., Bastholt L., Wirth L.J., Martins R.G., Locati L.D., Jarzab B., Pacini F., Daumerie C., Droz J.P., et al. Phase II study of safety and efficacy of motesanib in patients with progressive or symptomatic, advanced or metastatic medullary thyroid cancer. J. Clin. Oncol. 2009;27:3794–3801. doi: 10.1200/JCO.2008.18.7815. [DOI] [PubMed] [Google Scholar]
- 128.Pennell N.A., Daniels G.H., Haddad R.I., Ross D.S., Evans T., Wirth L.J., Fidias P.H., Temel J.S., Gurubhagavatula S., Heist R.S., et al. A phase II study of gefitinib in patients with advanced thyroid cancer. Thyroid. 2008;18:317–323. doi: 10.1089/thy.2007.0120. [DOI] [PubMed] [Google Scholar]
- 129.Sherman S.I., Wirth L.J., Droz J.P., Hofmann M., Bastholt L., Martins R.G., Licitra L., Eschenberg M.J., Sun Y.N., Juan T., et al. Motesanib Thyroid Cancer Study Group. Motesanib diphosphate in progressive differentiated thyroid cancer. N. Engl. J. Med. 2008;359:31–42. doi: 10.1056/NEJMoa075853. [DOI] [PubMed] [Google Scholar]
- 130.Kloos R.T., Ringel M.D., Knopp M.V., Hall N.C., King M., Stevens R., Liang J., Wakely P.E., Jr, Vasko V.V., Saji M., et al. Phase II trial of sorafenib in metastatic thyroid cancer. J. Clin. Oncol. 2009;27:1675–1684. doi: 10.1200/JCO.2008.18.2717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Cohen E.E., Rosen L.S., Vokes E.E., Kies M.S., Forastiere A.A., Worden F.P., Kane M.A., Sherman E., Kim S., Bycott P., et al. Axitinib is an active treatment for all histologic subtypes of advanced thyroid cancer, results from a phase II study. J. Clin. Oncol. 2008;26:4708–4713. doi: 10.1200/JCO.2007.15.9566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Wells S.A., Jr, Gosnell J.E., Gagel R.F., Moley J., Pfister D., Sosa J.A., Skinner M., Krebs A., Vasselli J., Schlumberger M. Vandetanib for the treatment of patients with locally advanced or metastatic hereditary medullary thyroid cancer. J. Clin. Oncol. 2010;28:767–772. doi: 10.1200/JCO.2009.23.6604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Schneider T.C., Abdulrahman R.M., Corssmit E.P., Morreau H., Smit J.W., Kapiteijn E. Long-term analysis of the efficacy and tolerability of sorafenib in advanced radio-iodine refractory differentiated thyroid carcinoma, final results of a phase II trial. Eur. J. Endocrinol. 2012;167:643–650. doi: 10.1530/EJE-12-0405. [DOI] [PubMed] [Google Scholar]
- 134.Broutin S., Ameur N., Lacroix L., Robert T., Petit B., Oumata N., Talbot M., Caillou B., Schlumberger M., Dupuy C., et al. Identification of soluble candidate biomarkers of therapeutic response to sunitinib in medullary thyroid carcinoma in preclinical models. Clin. Cancer Res. 2011;17:2044–2054. doi: 10.1158/1078-0432.CCR-10-2041. [DOI] [PubMed] [Google Scholar]
- 135.Grosse J., Wehland M., Pietsch J., Schulz H., Saar K., Hübner N., Eilles C., Bauer J., Abou-El-Ardat K., Baatout S., et al. Gravity-sensitive signaling drives 3-dimensional formation of multicellular thyroid cancer spheroids. FASEB J. 2012;26:5124–5140. doi: 10.1096/fj.12-215749. [DOI] [PubMed] [Google Scholar]
- 136.Bass M.B., Sherman S.I., Schlumberger M.J., Davis M.T., Kivman L., Khoo H.M., Notari K.H., Peach M., Hei Y.J., Patterson S.D. Biomarkers as predictors of response to treatment with motesanib in patients with progressive advanced thyroid cancer. J. Clin. Endocrinol. Metab. 2010;95:5018–5027. doi: 10.1210/jc.2010-0947. [DOI] [PubMed] [Google Scholar]
- 137.Marotta V., Ramundo V., Camera L., del Prete M., Fonti R., Esposito R., Palmieri G., Salvatore M., Vitale M., Colao A., et al. Sorafenib in advanced iodine-refractory differentiated thyroid cancer, efficacy, safety and exploratory analysis of role of serum thyroglobulin and FDG-PET. Clin. Endocrinol. 2012;78:760–767. doi: 10.1111/cen.12057. [DOI] [PubMed] [Google Scholar]
- 138.Cabanillas M.E., Waguespack S.G., Bronstein Y., Williams M.D., Feng L., Hernandez M., Lopez A., Sherman S.I., Busaidy N.L. Treatment with tyrosine kinase inhibitors for patients with differentiated thyroid cancer, the M. D. Anderson experience. J. Clin. Endocrinol. Metab. 2010;95:2588–2595. doi: 10.1210/jc.2009-1923. [DOI] [PubMed] [Google Scholar]
- 139.Lee E.K., Chung K.W., Min H.S., Kim T.S., Kim T.H., Ryu J.S., Jung Y.S., Kim S.K., Lee Y.J. Preoperative serum thyroglobulin as a useful predictive marker to differentiate follicular thyroid cancer from benign nodules in indeterminate nodules. J. Korean Med. Sci. 2012;27:1014–1018. doi: 10.3346/jkms.2012.27.9.1014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Yim J.H., Kim E.Y., Bae Kim W., Kim W.G., Kim T.Y., Ryu J.S., Gong G., Hong S.J., Yoon J.H., Shong Y.K. Long-term consequence of elevated thyroglobulin in differentiated thyroid cancer. Thyroid. 2013;23:58–63. doi: 10.1089/thy.2011.0487. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Webb R.C., Howard R.S., Stojadinovic A., Gaitonde D.Y., Wallace M.K., Ahmed J., Burch H.B. The utility of serum thyroglobulin measurement at the time of remnant ablation for predicting disease-free status in patients with differentiated thyroid cancer: A meta-analysis involving 3947 patients. J. Clin. Endocrinol. Metab. 2012;97:2754–2763. doi: 10.1210/jc.2012-1533. [DOI] [PubMed] [Google Scholar]
- 142.Shaik S., Nucera C., Inuzuka H., Gao D., Garnaas M., Frechette G., Harris L., Wan L., Fukushima H., Husain A., et al. SCF(β-TRCP) suppresses angiogenesis and thyroid cancer cell migration by promoting ubiquitination and destruction of VEGF receptor 2. J. Exp. Med. 2012;209:1289–1307. doi: 10.1084/jem.20112446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Zerilli M., Zito G., Martorana A., Pitrone M., Cabibi D., Cappello F., Giordano C., Rodolico V. BRAF(V600E) mutation influences hypoxia-inducible factor-1alpha expression levels in papillary thyroid cancer. Mod. Pathol. 2010;23:1052–1060. doi: 10.1038/modpathol.2010.86. [DOI] [PubMed] [Google Scholar]
- 144.Bottos A., Martini M., di Nicolantonio F., Comunanza V., Maione F., Minassi A., Appendino G., Bussolino F., Bardelli A. Targeting oncogenic serine/threonine-protein kinase BRAF in cancer cells inhibits angiogenesis and abrogates hypoxia. Proc. Natl. Acad. Sci. USA. 2012;109:E353–E359. doi: 10.1073/pnas.1105026109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Grimm D., Bauer J., Pietsch J., Infanger M., Eucker J., Eilles C., Schoenberger J. Diagnostic and therapeutic use of membrane proteins in cancer cells. Curr. Med. Chem. 2011;18:176–190. doi: 10.2174/092986711794088344. [DOI] [PubMed] [Google Scholar]
- 146.Pietsch J., Kussian R., Sickmann A., Bauer J., Weber G., Nissum M., Westphal K., Egli M., Grosse J., Schönberger J., et al. Application of free-flow IEF to identify protein candidates changing under microgravity conditions. Proteomics. 2010;10:904–913. doi: 10.1002/pmic.200900226. [DOI] [PubMed] [Google Scholar]
- 147.Pietsch J., Sickmann A., Weber G., Bauer J., Egli M., Wildgruber R., Infanger M., Grimm D. A proteomic approach to analysing spheroid formation of two human thyroid cell lines cultured on a random positioning machine. Proteomics. 2011;11:2095–2104. doi: 10.1002/pmic.201000817. [DOI] [PubMed] [Google Scholar]
- 148.Pietsch J., Sickmann A., Weber G., Bauer J., Egli M., Wildgruber R., Infanger M., Grimm D. Metabolic enzyme diversity in different human thyroid cell lines and their sensitivity to gravitational forces. Proteomics. 2012;12:2539–2546. doi: 10.1002/pmic.201200070. [DOI] [PubMed] [Google Scholar]
- 149.Pietsch J., Riwaldt S., Bauer J., Sickmann A., Weber G., Grosse J., Infanger M., Eilles C., Grimm D. Interaction of proteins identified in human thyroid cells. Int. J. Mol. Sci. 2013;14:1164–1178. doi: 10.3390/ijms14011164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Ferlay J., Shin H.R., Bray F., Forman D., Mathers C., Parkin D.M. Estimates of worldwide burden of cancer in 2008, GLOBOCAN 2008. Int. J. Cancer. 2010;15:2893–2917. doi: 10.1002/ijc.25516. [DOI] [PubMed] [Google Scholar]
- 151.Bukowski R.M., Eisen T., Szczylik C., Stadler W.M., Simantov R., Shan M., Elting J., Pena C., Escudier B. Final results of the randomized phase III trial of sorafenib in advanced renal cell carcinoma, Survival and biomarker analysis. J. Clin. Oncol. 2007;25:15s. [Google Scholar]
- 152.Deprimo S.E., Bello C.L., Smeraglia J., Baum C.M., Spinella D., Rini B.I., Michaelson M.D., Motzer R.J. Circulating protein biomarkers of pharmacodynamic activity of Sunitinib in patients with metastatic renal cell carcinoma, Modulation of VEGF and VEGF-related proteins. J. Transl. Med. 2007;5:32. doi: 10.1186/1479-5876-5-32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Genega E.M., Ghebremichael M., Najarian R., Fu Y., Wang Y., Argani P., Grisanzio C., Signoretti S. Carbonic anhydrase IX expression in renal neoplasms, Correlation with tumor type and grade. Am. J. Clin. Pathol. 2010;134:873–879. doi: 10.1309/AJCPPPR57HNJMSLZ. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Porta C., Paglino C., de Amici M., Quaglini S., Sacchi L., Imarisio I., Canipari C. Predictive value of baseline serum vascular endothelial growth factor and neutrophil gelatinase-associated lipocalin in advanced kidney cancer patients receiving Sunitinib. Kidney Int. 2010;77:809–815. doi: 10.1038/ki.2009.552. [DOI] [PubMed] [Google Scholar]
- 155.Kjeldsen L., Johnsen A.H., Sengeløv H., Borregaard N. Isolation and primary structure of NGAL, a novel protein associated with human neutrophil gelatinase. J. Biol. Chem. 1993;268:10425–10432. [PubMed] [Google Scholar]
- 156.Pantuck A.J., Fang Z., Liu X., Seligson D.B., Horvath S., Leppert J.T., Belldegrun A.S., Figlin R.A. Gene expression and tissue microarray analysis of interleukin-2 complete responders in patients with metastatic renal cell carcinoma. J. Clin. Oncol. 2005;23:15s. [Google Scholar]
- 157.Atkins M., Regan M., McDermott D., Mier J., Stanbridge E., Youmans A., Febbo P., Upton M., Lechpammer M., Signoretti S. Carbonic anhydrase IX expression predicts outcome of interleukin 2 therapy for renal cancer. Clin. Cancer Res. 2005;11:3714–3721. doi: 10.1158/1078-0432.CCR-04-2019. [DOI] [PubMed] [Google Scholar]
- 158.Klatte T., Seligson D.B., Riggs S.B., Leppert J.T., Berkman M.K., Kleid M.D., Yu H., Kabbinavar F.F., Pantuck A.J., Belldegrun A.S. Hypoxia-inducible factor 1 alpha in clear cell renal cell carcinoma. Clin. Cancer Res. 2007;13:7388–7393. doi: 10.1158/1078-0432.CCR-07-0411. [DOI] [PubMed] [Google Scholar]
- 159.Patel P.H., Chadalavada R.S., Ishill N.M., Patil S., Reuter V.E., Motzer R.J., Chaganti R.S. Hypoxia-inducible factor (HIF) 1a and 2a levels in cell lines and human tumor predicts response to Sunitinib in renal cell carcinoma (RCC) J. Clin. Oncol. 2008;26:15s. [Google Scholar]
- 160.Hoffmann N.E., Sheinin Y., Lohse C.M., Parker A.S., Leibovich B.C., Jiang Z., Kwon E.D. External validation of IMP3 expression as an independent prognostic marker for metastatic progression and death for patients with clear cell renal cell carcinoma. Cancer. 2008;112:1471–1479. doi: 10.1002/cncr.23296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Thompson R.H., Kuntz S.M., Leibovich B.C., Dong H., Lohse C.M., Webster W.S., Sengupta S., Frank I., Parker A.S., Zincke H., et al. Tumor B7-H1 is associated with poor prognosis in renal cell carcinoma patients with long-term follow-up. Cancer Res. 2006;66:3381–3385. doi: 10.1158/0008-5472.CAN-05-4303. [DOI] [PubMed] [Google Scholar]
- 162.Zurita A.J., Jonasch E., Wang X., Khajavi M., Yan S., Du D.Z., Xu L., Herynk M.H., McKee K.S., Tran H.T., et al. A cytokine and angiogenic factor (CAF) analysis in plasma for selection of sorafenib therapy in patients with metastatic renal cell carcinoma. Ann. Oncol. 2012;23:46–52. doi: 10.1093/annonc/mdr047. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Tran H.T., Liu Y., Zurita A.J., Lin Y., Baker-Neblett K.L., Martin A.M., Figlin R.A., Hutson T.E., Sternberg C.N., Amado R.G., et al. Prognostic or predictive plasma cytokines and angiogenic factors for patients treated with pazopanib for metastatic renal-cell cancer: A retrospective analysis of phase 2 and phase 3 trials. Lancet Oncol. 2012;13:827–837. doi: 10.1016/S1470-2045(12)70241-3. [DOI] [PubMed] [Google Scholar]
- 164.Purdue M.P., Johansson M., Zelenika D., Toro J.R., Scelo G., Moore L.E., Prokhortchouk E., Wu X., Kiemeney L.A., Gaborieau V., et al. Genome-wide association study of renal cell carcinoma identifies two susceptibility loci on 2p21 and 11q13.3. Nat. Genet. 2011;43:60–65. doi: 10.1038/ng.723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Wu X., Scelo G., Purdue M.P., Rothman N., Johansson M., Ye Y., Wang Z., Zelenika D., Moore L.E., Wood C.G., et al. A genome-wide association study identifies a novel susceptibility locus for renal cell carcinoma on 12p11.23. Hum. Mol. Genet. 2012;21:456–462. doi: 10.1093/hmg/ddr479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Xu C.F., Bing N.X., Ball H.A., Rajagopalan D., Sternberg C.N., Hutson T.E., de Souza P., Xue Z.G., McCann L., King K.S., et al. Pazopanib efficacy in renal cell carcinoma, evidence for predictive genetic markers in angiogenesis related and exposure-related genes. J. Clin. Oncol. 2011;29:2557–2564. doi: 10.1200/JCO.2010.32.9110. [DOI] [PubMed] [Google Scholar]
- 167.Huang D., Ding Y., Zhou M., Rini B.I., Petillo D., Qian C.N., Kahnoski R., Futreal P.A., Furge K.A., Teh B.T. Interleukin-8 mediates resistance to anti-angiogenic agent Sunitinib in renal cell carcinoma. Cancer Res. 2010;70:1063–1071. doi: 10.1158/0008-5472.CAN-09-3965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Climent M.A., Arranz J.A., Gallardo E., Puente J., Bellmunt J., Mellado B., Martínez E., Moreno F., Font A., Robledo M., et al. Single nucleotide polymorphism associations with response and toxic effects in patients with advanced renal-cell carcinoma treated with first-line Sunitinib, a multicentre, observational, prospective study. Lancet Oncol. 2011;12:1143–1150. doi: 10.1016/S1470-2045(11)70266-2. [DOI] [PubMed] [Google Scholar]
- 169.Van der Veldt A.A., Eechoute K., Gelderblom H., Gietema J., Guchelaar H.J., van Erp N.P., van den Eertwegh A.J., Haanen J.B., Mathijssen R.H., Wessels J.A. Genetic polymorphisms associated with a prolonged progression-free survival in patients with metastatic renal cell cancer treated with Sunitinib. Clin. Cancer Res. 2011;17:620–629. doi: 10.1158/1078-0432.CCR-10-1828. [DOI] [PubMed] [Google Scholar]
- 170.Brannon A.R., Reddy A., Seiler M., Arreola A., Moore D.T., Pruthi R.S., Wallen E.M., Nielsen M.E., Liu H., Nathanson K.L., et al. Molecular stratification of clear cell renal cell carcinoma by consensus clustering reveals distinct subtypes and survival patterns. Genes Cancer. 2010;1:152–163. doi: 10.1177/1947601909359929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Jemal A., Siegel R., Ward E., Hao Y., Xu J., Thun M.J. Cancer statistics, 2009. CA Cancer J. Clin. 2009;59:225–249. doi: 10.3322/caac.20006. [DOI] [PubMed] [Google Scholar]
- 172.Ferlay J., Autier P., Boniol M., Heanue M., Colombet M., Boyle P. Estimates of the cancer incidence and mortality in Europe in 2006. Ann. Oncol. 2007;18:581–592. doi: 10.1093/annonc/mdl498. [DOI] [PubMed] [Google Scholar]
- 173.Bradford T.J., Tomlins S.A., Wang X., Chinnaiyan A.M. Molecular markers of prostate cancer. Urol. Oncol. 2006;24:538–551. doi: 10.1016/j.urolonc.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 174.Lambrechts D., Lenz H.J., de Haas S., Carmeliet P., Scherer S.J. Markers of response for the antiangiogenic agent bevacizumab. J. Clin. Oncol. 2013;31:1219–1230. doi: 10.1200/JCO.2012.46.2762. [DOI] [PubMed] [Google Scholar]
- 175.François P., Bertos N., Laferrière J., Sadekova S., Souleimanova M., Zhao H., Finak G., Meterissian S., Hallett M.T., Park M. Gene-expression profiling of microdissected breast cancer microvasculature identifies distinct tumor vascular subtypes. Breast Cancer Res. 2012;14:R120. doi: 10.1186/bcr3246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Carpi A., Mechanick J.I., Saussez S., Nicolini A. Thyroid tumor marker genomics and proteomics, diagnostic and clinical implications. J. Cell Physiol. 2010;224:612–619. doi: 10.1002/jcp.22187. [DOI] [PubMed] [Google Scholar]
- 177.Pavlou M.P., Diamandis E.P. The cancer cell secretome, a good source for discovering biomarkers? J. Proteomics. 2010;73:1896–1906. doi: 10.1016/j.jprot.2010.04.003. [DOI] [PubMed] [Google Scholar]