Abstract
In 1998, gastrointestinal stromal tumor (GIST) emerged as a distinct oncogenetic entity and subsequently became a paradigm of targeted therapies in solid tumors. Diagnosis of GIST relies on both histology and immunohistochemistry. Ninety-five percent of GISTs express either KIT or DOG-1. Approximately 80%–90% of GISTs harbor gain-of-function mutations of either KIT or platelet-derived growth factor receptor alpha polypeptide (PDGFRA) receptor tyrosine kinase (RTK). More than 100 different mutations have been described, some of which are associated with specific clinical and/or histological characteristics. Detection of KIT or PDGFRA mutations is recommended in advanced GISTs because they are highly predictive of tumor response to RTK inhibitors, as well as in KIT-negative cases to confirm diagnosis. In most cases, GISTs are sporadic, but in rare cases, they are related with genetic predisposition, such as neurofibromatosis type 1, Carney triad, Carney–Stratakis syndrome, and inherited KIT or PDGFRA germline mutations.
Keywords: gastrointestinal stromal tumors, KIT, PDGFRA, genetic predispositions, imatinib
Introduction
Until the late 1990s, gastrointestinal stromal tumors (GISTs) were often misdiagnosed as other gastrointestinal mesenchymal tumors, such as leiomyoma, leiomyosarcoma, or schwannoma. In 1998, Hirota et al1 demonstrated the critical role of KIT receptor tyrosine kinase (RTK)-activating mutations in GISTs, and KIT immunostaining became a major diagnostic tool. Only 3 years later, the KIT inhibitor imatinib was shown to have dramatic antitumor effects, and this rare and frequently undiagnosed tumor became the paradigm of targeted therapies. This review updates knowledge on GIST and highlights the knowledge on genetic alterations.
GIST pathogenesis
GISTs are thought to be derived from intestinal cells of Cajal or precursors because of their digestive origin and the expression of KIT.2 Physiologically, the cells of Cajal have an autonomous pacemaker function and coordinate gastrointestinal peristalsis throughout the gastrointestinal tract.3 Gain-of-function mutations of proto-oncogenes KIT or platelet-derived growth factor receptor alpha polypeptide (PDGFRA) play a critical role in GIST pathogenesis.1,4 They are found in 85% of GISTs, in either KIT for 75%–80% or PDGFRA for 5%–10%, and are mutually exclusive.5
Epidemiology
GISTs are the most frequent mesenchymal tumors of the digestive tract. The incidence peak and the median age at diagnosis is of about 60–65 years. However, the distribution by age is wide, and GIST can occur during childhood or at 90 years of age.6–8 Most epidemiological studies reported a slight male predominance.7–10 In western countries, the estimated annual incidence reported in population-based studies is between 11 and 15 per million people.9–11 This commonly accepted incidence was estimated from pathologic registry in all cases9–11 and is probably underestimated because GIST tumorlets (size of 1–10 mm) are found frequently in autopsies.12 GISTs are sporadic in their great majority but, in rare cases, occur in patients with genetic predisposition. These rare cases are described in detail in this review.
Location and symptoms at diagnosis
Typically, the tumor occurs in the stomach for 66% or in the small intestine for 25% of most cases, but they might occur along the entire length of the digestive tract from the esophagus to the anus and sometimes in the omentum and the mesentery.6,13 Therefore, most GISTs arise from the bowel wall with a frequently slow and extra-luminal tumor development. Symptoms of the disease are nonspecific and reflect these features.7–9 GISTs can be revealed by gastrointestinal bleeding, solid mass at palpation, pain, abdominal discomfort, and early satiety, and acute abdomen emergency can sometimes occur. The diagnosis is also frequently made incidentally.7,8,14
In cases of advanced GISTs, most metastases arise in the liver and/or peritoneal cavity. Other metastases in lung, pleura, bones, or other areas are less frequent.7,8,14,15 Lymph node metastases are unusual, and lymphadenectomy is warranted only for evident nodal involvement or for rare pediatric GISTs.16
Risk assessment after curative surgery
Until last year, GISTs were often described as benign or of uncertain malignant potential. Subsequently, after the discovery and well characterization of the KIT role, it was suggested that all GISTs have malignant potential, and many prognostic factors have been assessed to estimate the risk of relapse after curative surgery.6,17–22 Nevertheless, to be used in routine practice, prognosis factors have to be robust, reproducible, and easy to assess. These considerations led to the adoption of a consensus risk classification based on the combination of the maximal tumor size and the mitotic count per 50 high-power fields (hpf) in 2002 (Table 1A).16,23 However, the relative good prognosis of patients with gastric GIST of all size with low mitotic activity called into question the concept of the generally malignant nature of GIST.7 Moreover, it was demonstrated that gastric GISTs have a better prognostic outlook than intestinal GISTs of similar size and mitotic index.24 Thus, Miettinen and Lasota24 proposed a new classification including the anatomic site as a third independent prognostic factor and defined a new subclass of benign GIST (Table 1B). More recently, a prognostic normogram using the same three factors was developed and validated in three independent large cohorts.25
Table 1A.
Classifications for risk assessment after curative surgery. Consensus approach published by Fletcher et al in 200223
| Risk group | Maximal tumor diameter | Mitotic count/50 hpf |
|---|---|---|
| Very low risk | <2 cm | <5 |
| Low risk | 2–5 cm | <5 |
| Intermediate risk | <5 cm | 6–10 |
| 5–10 cm | <5 | |
| High risk | 5–10 cm | 6–10 |
| >10 cm | any | |
| any | >10 |
Table 1B.
Classification with GIST location proposed by Miettinen and Lasota in 200624
| Risk roup | Maximal tumor diameter | Mitotic count/50 hpf a | Estimated rate of relapse according to GIST location | |||
|---|---|---|---|---|---|---|
|
| ||||||
| Gastric | Jejuno ileal | Duodenal | Rectal | |||
| 1 | ≤2 cm | ≤5 | 0 | 0 | 0 | 0 |
| 2 | >2–≤5 cm | ≤5 | 1.9% | 4.3% | 8.3% | 8.5% |
| 3a | >5–≤10 cm | ≤5 | 3.6% | 24% | –b | –b |
| 3b | >10 cm | ≤5 | 12% | 52% | 34% | 57% |
| 4 | ≤2 cm | >5 | 0 | 50% | – | 54% |
| 5 | >2–≤5 cm | >5 | 16% | 73% | 50% | 52% |
| 6a | >5–≤10 cm | >5 | 55% | 85% | –b | –b |
Notes:
The mitotic count was made in a 5 mm2 surface, which corresponds to 20–25 fields at ×400 magnification with recent microscopes;
Subgroups with insufficient number of patients to estimate the rate of relapse risk.
Abbreviations: GIST, gastrointestinal stromal tumors; hpf, high-power fields.
Treatment in advanced GIST
GISTs are highly resistant to conventional chemotherapy and radiotherapy, and during many years, the only curative treatment was surgery even in cases of advanced disease. Understanding the molecular physiology of GIST has allowed development of targeted therapies. Today, two small molecule tyrosine kinase inhibitors with activity against KIT and PDGFRA, among others, are approved in patients with GIST: imatinib (Glivec®, Gleevec®; Novartis, Basel, Switzerland) and sunitinib (Sutent®; Pfizer, New York, NY).
In patients with advanced GIST, imatinib efficacy was initially described in a single patient.26 After rapid development, it is today the first-line reference treatment in advanced GISTs, achieves disease control in 70%–85% of patients with a median progression-free survival (PFS) of 20–24 months and a median overall survival (OS) of 50–55 months.27,28 Nevertheless, imatinib is not curative, and its interruption results in rapid progression in most patients.29 Under imatinib, secondary resistance occurred in 15%–20% of patients each year.
Recently, retrospective studies suggested a potential benefit of secondary metastases resection in patients with a good response under imatinib.30–32 Prospective randomized trials should soon evaluate such strategy and allow recommendations to be made. In the opposite, patients with disease progression did not seem to benefit from surgery and should be considered for other targeted therapies.30–32 Among them, sunitinib has proved its efficacy with a median PFS of 6–8 months, and it is approved in second-line treatment.33,34 In third-line treatment, promising results have been reported with sorafenib (Nexavar®; Bayer, Leverkusen, Germany),35 and many other targeted therapies like nilotinib (Tasigna®; Novartis) or dasatinib (Sprycel®; Bristol-Myers Squibb, New York, NY) are in development at present.36,37
Adjuvant treatment
In cases of localized GIST, surgery remains the only curative treatment. After such resection, results of a randomized Phase III trial demonstrated that imatinib taken daily for 1 year significantly improved recurrence-free survival compared with placebo, and at 1-year follow-up, the hazard ratio was 0.35 (95% CI: 0.22–0.53; P < 0.0001).38 Two studies in progress are investigating treatment duration: the EORTC trial 62024 (no adjuvant treatment vs 2 years of imatinib) and the SSG trial XVIII (1 year vs 3 years of imatinib). In waiting results of those studies, 1-year imatinib is today the standard treatment in adjuvant setting, and its indication has been approved recently in Europe in patients who are at significant risk of relapse.
Diagnostic criteria
Most GISTs are suspected with endoscopy or computed tomography (CT) scan or during surgery. However, definitive diagnosis relies on pathology, which is based on both histology and immunohistochemistry. As a result of advances in understanding GISTs pathogenesis and the rapid development of effective targeted therapies, international consensus criteria have been published for the diagnostic of GIST.16,23,39
Endoscopy and imagery
GIST can be found incidentally or after digestive symptoms during gastroscopy, colonoscopy, capsule endoscopy, or enteroscopy.40,41 GIST is a submucosal tumor and is observed at endoscopy as a bulge underneath the mucosa of the gastrointestinal tract. The digestive mucosa is of normal appearance, but a mucosal ulceration secondary to tumor growth is common and can induce gastrointestinal bleeding. Superficial mucosal biopsies are generally noncontributive. Endoscopic ultrasound is the most important tool to assess its layer of origin, particularly in cases of small GIST.39 It allows it to equally perform guided fine-needle aspiration or trucut biopsy. These two techniques are safe and comparable but have only limited value for GIST diagnostic because of a high rate of technical failure.40,41 In cases of localized GIST, the best tissue acquisition technique remains surgical resection, and multiple core needle biopsies are recommended only in large tumor cases, which involve multivisceral resection.39 When submucosal tumor is less than 2 cm in size, and so probably a GIST of low risk, one standard approach is to follow it with endoscopic ultrasound, reserving resection for growing tumors. For tumors larger than 2 cm or rectal tumors of worse prognosis, biopsy/excision should be made for definitive diagnostic.39
The diagnostic of GIST is often suggested on CT scan. Typically, tumors are described as a large well-circumscribed tumor arising from the stomach or small bowel. They are predominantly extra-luminal with lower signal intensity than that of the contrast material-enhanced liver.14 In most cases, large tumors have a heterogeneously enhancing soft-tissue rim surrounding a necrotic center, and small tumors are more homogeneous. Central gas, calcification, or fluid–fluid level suggest intratumoral hemorrhage is possible. For preoperative staging, CT scans and 18F-fluoro-deoxyglucose positron emission tomography (PET) are both useful.14,42,43 Although less precise for anatomic detail than CT scans, PET can reveal small metastases and is useful in cases of locally advanced GIST when early assessment of therapy effectiveness is of special concern.39,42–45 Like for rectal adenocarcinoma, magnetic resonance imaging provides better preoperative detail of perineal region and is recommended in cases of rectal GIST.39
Standard histologic examination
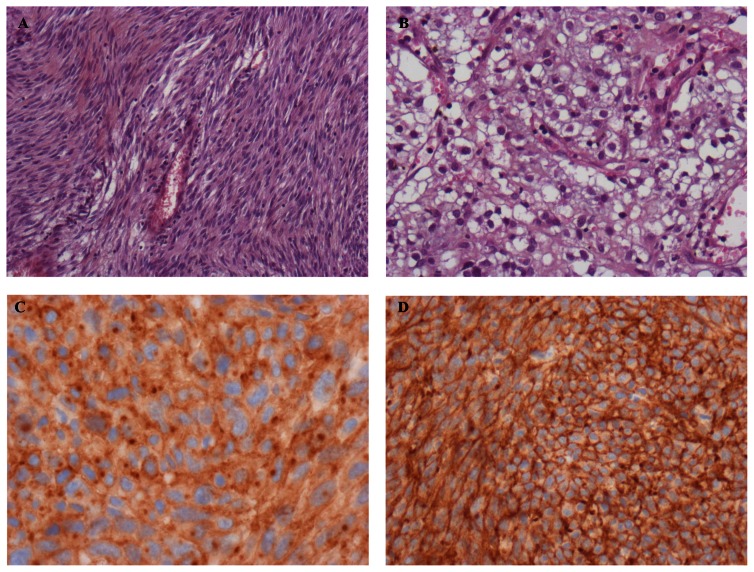
Histologically, GISTs can be most often classified in three categories: the spindle cell type that represents 70% of cases and is composed of relatively uniform eosinophilic cells arranged in short fascicles or whorls (Figure 1A), the epithelioid type that represents 20% of cases and is composed of rounded cells with eosinophilic or clear cytoplasm (Figure 1B), and the mixed type that contains the both cell types together. In uncommon cases, GISTs show a prominent myxoid stroma, a paraganglioma-like pattern, or a cytoplasmic pleomorphism.23 Necrosis and/or hemorrhage area are frequent in large GISTs, and the necrosis percent had been suggested as a prognostic factor in some studies.6 Tumor rupture, whatever the cause, should be recorded because of its adverse prognostic value.39
Figure 1.
Histologic features of gastrointestinal stromal tumors (GISTs). A) Spindle cell type. B) Epithelioid cell type. C) CD117 (KIT) immunostaining with cytoplasmic dot- like (Golgi) pattern and cytoplasmic pattern. D) GIST positive with DOG-1 immunostaining.
In resected GISTs, standard histologic description must be completed by the mitotic count, which was evaluated in 50 hpf in the 2002 consensus risk classification.23 However, in the last classification proposed by Miettinen and Lasota,24 the mitotic count was made in a 5 mm2 surface, which corresponds to 20–25 fields at 400× magnification by using a modern wide-field microscope with wide-field eye-pieces. Evaluation of adjuvant imatinib indication is of major importance for routine practice.
Immunohistochemistry
Ninety-five percent of GISTs show KIT immunostaining (CD117), but some variability in pattern exists. Most GISTs show unequivocal diffuse and strong cytoplasmic positivity. A cytoplasmic dot-like (Golgi pattern) staining is present in almost half of the cases and may be exclusive or associated with cytoplasmic pattern (Figure 1C).23 Interestingly, Golgi staining is correlated with KIT mutation and is significantly more frequent in GISTs with homozygous mutations.46 These results are consistent with experimental data in which KIT mutant proteins remain within endoplasmic reticulum or Golgi compartments and do not reach the cell membrane.47 Stromal mast cells are a valuable internal positive control for KIT immunostaining. Although highly specific, KIT immunostaining can be positive in other tumors, in particular in small-cell lung cancer, testicular teratocarcinoma, melanoma, and angiosarcoma.48–50 In these tumors, KIT mutations are uncommon with the exception of melanoma,49 and overexpression is mostly associated with genomic amplification of wild-type gene.48,50
Other immunohistochemical analyses are frequently used in GISTs diagnostic: 70% of GISTs show immunopositivity for CD34, 40% for smooth muscle actin (SMA), 5% for S-100 protein, and 2% for desmin.16,23 CD34 and SMA immunopositivity seem to vary according to GIST location.51 Expression of PKC-θ was also used for GIST identification.4 However, the low specificity of these markers limits their relevance in routine practice of GISTs. By contrast, DOG-1 is useful for diagnosis of GISTs. DOG-1 was characterized using gene microarrays, and its protein function is unknown. It is expressed almost ubiquitously in GISTs irrespective of the type of activating mutations, is rarely expressed in other soft tissue tumors, and is positive in KIT-immunonegative GISTs with PDGFRA mutation (Figure 1D).52
Activating mutations
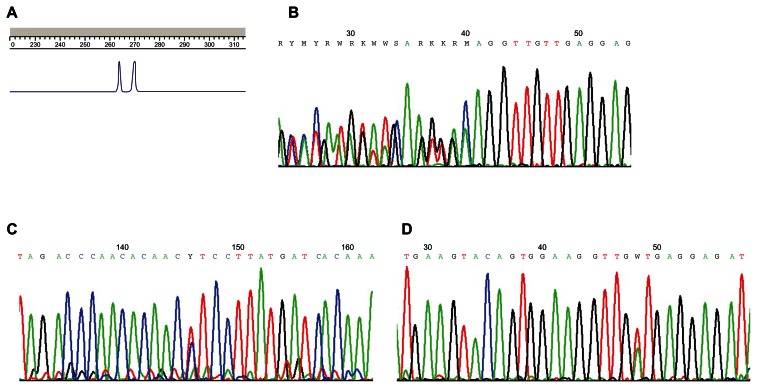
The screening of KIT and PDFGRA mutations may be performed on either formalin-fixed, paraffin-embedded, or frozen samples. Bouin fixation should be avoided since it may impair the molecular analysis feasibility.16 A histological examination to spot tumor areas must be performed before all DNA extraction. Because of the relative tumoral homogeneity of GISTs, tumor tissue microdissection is not required in most cases. There is no recommendation for optimal mutation screening technique. Direct sequencing was performed in most previous studies. Length analysis of polymerase chain reaction (PCR) products is more sensitive than direct sequencing for detection of deletions and insertions (Figure 2A).4 Moreover, this technique allows for rapid screening for the most frequently mutated exons and to guide direct sequencing. In absence of length modification, direct sequencing of all putative mutated exons must be done to search point mutations (Figures 2B–2D). Denaturing high-performance liquid chromatography is another very sensitive technique, which allows detection of deletions, insertions, and substitutions.53 The frequencies of KIT and PDGFRA mutations in advanced GISTs are detailed in Table 2.5 The search of mutations must be done in specialized laboratories implicated in a quality assurance program.39
Figure 2.
Diagnostic of KIT-activating mutations. A) Deletion of KIT exon 11 detects with length analysis of polymerase chain reaction products. The normal allele is on the right, and the mutated allele with two codons deleted is on the left. B) Sequencing of KIT exon 11 with delWK557–558 heterozygous deletion. C) Sequencing of KIT exon 11 with L576P point mutations. D) Sequencing of KIT exon 11 with V560D point mutations.
Table 2.
Activating KIT or PDFGRA mutations and therapeutic implications
| Activating mutations | Frequency at diagnosis5,6 | Predictive value for imatiniba | Secondary mutations of imatinib resistance90,93–97 | Predictive value for sunitinibb |
|---|---|---|---|---|
| KIT | ||||
| Exon 9 | 10% | 70%–75% | 58% | |
| Exon 11 | 67% | 90%–95% | 34% | |
| Exon 13 | 1% | ± | Yes | + |
| Exon 14 | 0% | Yes | + | |
| Exon 17 | 1% | ± | Yes | − |
| Exon 18 | 0% | Yes | − | |
| PDGFRA | 33%–60% | |||
| Exon 12 | 1% | + | ||
| Exon 14 | <1% | + | ||
| Exon 18 | 5% | ± excepted for D842V | Yes | − for D842V |
| Wild type | 12%–15% | 33%–73% | 56% | |
Diagnostic recommendations
In most cases, the diagnosis of GIST relies on concordant histology and KIT immunopositivity. A central review by an expert in sarcoma pathology should be made for equivocal cases. In the rare cases of KIT-negative tumors (5%), molecular analysis for KIT and PDGFRA has a diagnostic interest, and other markers such as DOG-1 have to be considered.16,23,39
Mutational analysis for activating mutations is strongly recommended in all advanced GISTs because of their predictive value.39 This specific point will be discussed in detail in the next section.
Type of mutations and specificities
KIT and PDGFRA mutations
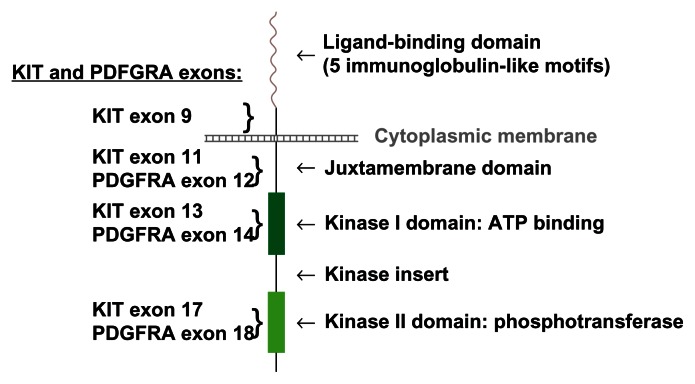
More than 80% of GISTs contain activating mutations of KIT or PDGFRA.5 Both receptors are members of the type III receptor protein-tyrosine kinase family and encode a transmembrane RTK. Binding of ligand (stem cell factor [SCF] for KIT) results in dimerization of the receptor, activation of its kinase activity, and phosphorylation of several proteins implicated in signaling pathways known to promote cell growth and survival.54 Both receptors activate the downstream phosphatidylinositol-3-kinase (PI3K)-AKT, mitogen-activated protein (MAP)-kinase, and Janus kinase-signal transducer and activator of transcription (JAK-STAT3) signaling cascades. 54,55 Activating mutations consist of in-frame deletions, insertions, or missense mutations, and result in constitutive receptor activation independent of ligand binding. Mutations can occur in the extracellular domain (KIT exon 9), in the juxtamembrane domain (KIT exon 11 and PDGFRA exon 12), in the kinase I domain (KIT exon 13 and PDGFRA exon 14), and in the kinase II domain (KIT exon 17 and PDGFRA exon 18).5
KIT mutations are found in 75%–80% of GISTs, and 60% of them are within the exon 11 of KIT,5,6,55 which comprises 33 codons (codons 550–582). KIT exon 11 encodes an intracytoplasmic juxtamembrane domain, which has an autoinhibitory function.56 Normally, after dimerization, transphosphorylation of two tyrosine residues (Tyr568 and Tyr570) in the juxtamembrane segment occurs, and the loop formed by exon 11 is converted from a compact inactive conformation to an extended active conformation.57,58 Mutations occurring in exon 11 disrupt this normal autoinhibitory function and promote spontaneous kinase activation.57,59 More than 90 mutations of exon 11 have been published:60–69 85% are in-frame deletions or insertions and 15% are missense mutations. The most frequent mutation is a short deletion in the proximal part of exon 11, delWK557–558, accounting for 8%–25% of KIT exon 11 mutations.60–69 Other KIT mutations are located within exon 9 in 10% of cases, consisting in most cases of in-frame insertions or within exons 13 or 17 in 1% for each.55PDGFRA mutations are found in 5%–7% of GISTs. They are located within exon 18 in 5% of GISTs and within exon 12 or 14 in 1% of GISTs.5 The most frequent exon 18 mutation is a substitution, D842V, and accounts for 30%–80% of PDGFRA mutations.55,63,65,66,70KIT or PDGFRA mutations are heterozygous in 95% of mutated GISTs.6 In rare cases, homozygous mutations could result from a loss of KIT wild-type allele and duplication of the mutant.71
KIT transcriptional regulation
To better understand the critical role of KIT in GIST pathogenesis, many studies have focused on KIT regulation. KIT contains 21 exons, and two alternative splicing sites have been described. One is located at the 3′ end of exon 9 and results in the transcription of two isoforms, GNNK− and GNNK+.72 Although the GNNK− isoform was the predominant isoform and has been reported to have a greater oncogenic potential than the GNNK+,73 the GNNK−/+ ratio was similar in GISTs than in normal interstitial cells of Cajal and control mast cells.74 In heterozygous mutated GISTs, similar amounts of both allele and both transcripts were found, and so, the oncogenic effects of KIT in GISTs do not seem related to a variation in transcripts ratio.
Interestingly, KIT messenger RNA (mRNA) amounts were higher in heterozygous mutated GISTs than in homozygous mutated and nonmutated GISTs.74,75 Using quantitative PCR and fluorescence in-situ hybridization (FISH), it was demonstrated that KIT amplification was not implicated in KIT overexpression in the majority of GISTs. More, despite variable expressions between tumors, analysis of both quantified mRNA with real-time PCR (RT-PCR) and protein levels with Western blot show a close correlation between these two regulation levels.75 A fraction of KIT was phosphorylated and activated in all GISTs, but this amount was not correlated with the mutational GISTs status.76 These data suggested a transcriptional regulation of KIT overexpression in GISTs.
Subsequent analysis of SCF production by RT-PCR, immunohistochemistry, and enzyme-linked immunosorbent assay demonstrated that KIT ligand and both its transcript isoforms were present in almost all tested GISTs and in three primary cultures.76 Indeed, SCF is produced by different cell types, and SCF mRNA and protein were detected in almost all normal digestive control samples. Previous in vitro data have reported that SCF treatment of GIST cells with heterozygous KIT mutation induced a stronger KIT tyrosine phosphorylation,77 while it did not in GIST cells with homozygous KIT mutation.78 Thus, SCF may activate wild-type KIT in nonmutated GISTs and modulate the fraction of activated KIT in heterozygous GISTs. These results suggest that an autocrine/paracrine mechanism may play a role in GISTs pathogenesis.76
KIT translational regulation
Two forms of KIT protein are described in GISTs.79 The 125 kDa form is the precursor of the 145 kDa form and lacks the complex glycosylation necessary for its cell surface targeting. 80 In vitro data suggested that intracellular signaling activated by the 125-kDa mutant form was sufficient to drive neoplasia KIT.80 KIT immunohistochemistry staining show that paranuclear dot-like patterns were significantly more frequent in homozygous mutated GISTs than in heterozygous or nonmutated GISTs.47,81 These results are supported by Western blot analyses. While the phosphorylated mature form of KIT is found in almost all GISTs irrespective of mutational status, the phosphorylated immature form is detected in GISTs with KIT mutation but not or weakly in nonmutated GISTs.47,77 In NIH3T3 cells infected with wild-type or mutated exon 11 KIT complementary DNA, KIT was predominantly found in the perinuclear region in mutated cells, whereas it was diffusely present in the cytoplasm and at the plasma membrane in wild-type cells. Mutant KIT was mainly retained in an phosphorylated immature form within Golgi and endoplasmic reticulum compartments, whereas wild-type KIT was expressed in a nonphosphorylated form at the plasma membrane. Moreover, stimulation with SCF and inhibition with imatinib have different effects according to mutational KIT status. In wild-type cells, SCF treatment significantly decreased the cell surface expression of KIT and imatinib restored it. In mutated cells, SCF treatment had no effect on KIT membrane expression, whereas imatinib blocked mutant KIT phosphorylation, restored normal maturation, and increased membrane expression.47 Activating KIT mutations induce an alteration of normal maturation and intracellular trafficking of receptor, and are associated with an intracellular activation of its immature form. These results are important because they suggest that only targeted therapies able to enter cells may be effective against mutated KIT. They also constitute a possible explanation of the absence of correlations between KIT immunohistochemistry staining and imatinib efficacy.82 We currently do not know if wild-type and mutant KIT might activate different signaling pathways according to their intracellular location and interactions with other proteins or if both allele have exactly the same activation pathways.
Location and prognosis
Many studies described that GISTs location was associated with type of mutation. GISTs with KIT exon 9 mutation arise predominantly in small intestine and colon, and those with PDGFRA mutations most often originate from the stomach. 6,63,65,83 Recently, we reported that GISTs with a deletion of the two tyrosine residues of exon 11 (delTyr) arise in the small intestine, colon, or rectum in about 70% of cases, whereas those with delWK557–558 occur in the stomach in about 75% of cases, and that this difference was highly significant.84 These data suggest possible different types of oncogenic events driving kit mutations in the different parts of the digestive tract.
GIST prognostic also seems to be associated with type of mutation. Tumors with PDGFRA mutations are most often benign than those with KIT mutations,83 and those with KIT exon 9 or 11 mutations have been described to be associated with malignant behavior.55 The type of mutations in a same exon may equally be prognostic. Exon 11 deletions and deletions affecting codons 557–558 of KIT exon 11 were described to be independent adverse prognostic factors in patients with GIST.64,68,85 Conversely, in another study, GISTs in which the last part of exon 11 (codons 562–579) were deleted were most frequently associated with malignancy than GISTs with deletion of the first part of exon 11 (codons 550–561).6,69 Subsequently, we reported that GISTs with delWK557–558 and GISTs with delTyr did not differ in the risk of relapse after curative surgery and both convey a poor prognosis.84 Thus, the prognostic value of the type of KIT exon 11 mutations remains unclear, and more data are required. Recently, it was suggested that the homozygous/heterozygous status of mutant exon 11 KIT may be equally prognosis. In two small series, GISTs with homozygous exon 11 mutations were associated with a malignant behavior in about 90% of cases.71,81 This worsened prognosis could be explained by at least two hypotheses: on the one hand, homozygous status with the loss of the wild-type allele may have a highly intrinsic oncogenic effect (by loss of regulatory mechanisms), and on the other hand, homozygous status may be only a marker of late-stage tumors that have undergone multiple genetic events.71,81 Because these studies are all retrospective and have included relatively few patients, their results have to be taken with caution and require confirmation. In future, analysis of international adjuvant trials according to mutations type will probably permit progress in the understanding of their prognostic value.
Predictive value
The mutational status of KIT or PDGFRA is highly predictive of clinical response to imatinib. In randomized trials, disease control rates (partial response and stable disease) assessed by Response Evaluation Criteria In Solid Tumours (RECIST) criteria were 90%–95% in KIT exon 11 mutations, 70%–75% in KIT exon 9, 33%–60% in PDGFRA, and 33%–73% in wild-type GISTs.66,86 Patients with exon 11 mutations have a significant longer PFS and OS than patients with exon 9 mutations or wild-type GIST, and there was no difference between patients with exon 9 mutations and those with wild-type GIST.66,86 Imatinib is a competitive inhibitor of adenosine triphosphate (ATP) and can only bind to the inactive conformation of KIT. In inactive conformation of KIT, imatinib is too large to bind to an ATP-binding pocket and is blocked by an α-helical conformation of KIT of exon 11. Modification in juxtamembrane conformation caused by exon 11 mutations allows an imatinib binding and an inhibition of KIT activity, giving a structural explanation for the predictive value of exon 11 mutations.56 Some authors have proposed that deletion of codon 565 could be a predictive factor among patients harboring exon 11 mutations.66 Nevertheless, the outcome of nonresectable and metastatic GISTs with delWK557–558 and delTyr under imatinib was similar in terms of response rates, PFS, and OS in one of our study. Moreover, all patients included had an objective response or a stable disease under imatinib, and median PFS were of about 20 months.84 Thus, the predictive value of the type of exon 11 mutations remains still to be determined.
In the meta-analysis of the two Phase III trials that compared 400 mg daily and 800 mg daily of imatinib, objective response rates and OS were not significantly different between the two arms.87 However, there was a small significant PFS advantage for the 800-mg arm. In multivariate analysis, exon 9 mutations was the only significant factor for the benefit of 800 mg daily dose (P = 0.012).87 This high dose is today the standard recommended dose in patients with exon 9 mutation, whereas the 400 mg dose is recommended in all other cases.39 For KIT mutations within exons 13 and 17, no definitive conclusion can be made because of their low frequency. Interestingly, when these mutations were located within kinase domains, disease control was obtained in the majority of patients treated with imatinib in prospective trials. 66,86 Concerning PDGFRA mutations, concordant in vitro data and clinical results support that the D842V substitution and other mutations within the kinase domain activation loop (exon 18) are predictive of primary resistance to imatinib, whereas it is efficient in cases of juxtamembrane domain mutations.66,86,88
In short, almost all GIST with exon 11 mutations responded to imatinib, patients with exon 9 mutations should receive a 800 mg daily dose of imatinib, and D842V PDGFRA mutation is predictive of primary resistance.
Secondary resistance and sunitinib in second line
Imatinib is not curative, and secondary resistance occurred in 15%–20% of patients each year. Many different mechanisms of secondary resistance have been described: genomic amplification and overexpression of KIT or PDFGRA;89,90 switch to another kinase activation with overexpression of the AXL RTK that stimulates oncogenic cell cycling;91 and levels of ABCB1 and ABCC1 proteins that are implicated in multidrug resistance and in ATP-dependent elimination of imatinib.92 However, the most frequent mechanism consists in the acquisition of a more novel mutation within KIT or PDGFRA.90,93–97 In all described cases, more novel mutation occurred in the same RTK than the primary mutation (either KIT or PDGFRA), and no secondary mutations have been identified in wild-type GISTs.96 Moreover, in the same patient, different secondary mutations can occur in different metastases and/or in the same metastases.98 These secondary mutations involved either the ATP-binding pocket of the kinase I domain (KIT exons 13 and 14) or the kinase II domain activation loop (KIT exons 17 and 18).90,93–97 Similarly to the predictive value of exon 11 mutation, structural and functional understanding of KIT-imatinib interaction can explain occurrence sites of secondary mutations. Imatinib binding can be directly inhibited by mutations involving the ATP-binding pocket,99,100 and mutations involving the activation loop could strongly increase the proportion of KIT in active conformation, decreasing the possibility of KIT to bind to inactive forms.96 Interestingly, clonal evolution consecutive to novel mutation acquisition can be found as an enhancing nodule within a previously responding low-signal lesion on contrast-enhanced CT scans many months before progression defined by RECIST criteria.97,101
As for imatinib in first line, response to second-line sunitinib is correlated with the type of primary mutation.102 Nevertheless, at the opposite, disease control rate (for at least 6 months) were higher in exon 9 mutations (58%) and wild-type GISTs (56%) than in exon 11 mutations (34%). PFS and OS were also significantly longer for patients with exon 9 mutations and patients with wild-type GIST than for those with exon 11 mutations.102 After progression under imatinib, sunitinib efficacy is also influenced by the type of secondary mutation. Among patients with a secondary KIT mutation identified, PFS was significantly longer for the 18 patients with KIT exons 13 or 14 mutations than for the 13 patients with KIT exons 17 or 18 mutations (7.8 months vs 2.3 months; P = 0.016). Disease control rate and OS were equally better in patients subgroup with exons 13 or 14 mutations.102 Moreover, these results were supported by in vitro data. Sunitinib was efficient against secondary ATP-binding pocket mutations but has inferior potency than imatinib against some activation loop mutations. Moreover, the D842V PDGFRA mutation, classically resistant to imatinib, confers also a sunitinib resistance in vitro.102
Genetic predisposition
GIST can occur in patients with genetic predisposition in rare cases. Neurofibromatosis type 1 (NF-1) confers an increased risk of developing GISTs as many other tumors.103 GISTs are associated with two rare tumor syndromes, the Carney triad and the Carney–Stratakis syndrome, and germline mutations have been identified in rare families with multiple occurrences of GIST among relatives. Some clinical, pathological, and/or biological features are associated with each of these genetic predispositions.
Neurofibromatosis type 1
Neurofibromatosis type 1 (NF-1) is one of the most frequent autosomal dominant disorders and affects 1/3000 individuals worldwide.104 The NF1 gene encodes the neurofibromin, a large protein that acts as a negative regulator of the Ras protein.105 Inactivating mutation of NF1 results in the hyperactivation of Ras and of downstream pathways such as MAP-kinase, which is implicated in antiapoptotic and growth signaling.106 Café-au-lait spots, axillary and inguinal freckling, neurofibroma, optic glioma, and iris Lisch nodules are among the primary clinical features of NF-1 and are used as major criteria for the clinical diagnosis.107 Other manifestations included skeletal, vascular, neurological, and psychiatric disorders.108 Other malignancies like astrocytomas and pheochromocytoma occur with increased frequency in patients with NF-1.103,108,109
The risk for GIST in patients with NF-1 is estimated to be as high as 7%.103 Median age at diagnosis is 50 years, and almost 20% of patients are aged less than 40 years. Like sporadic GISTs, there is a slight male predominance (60% of cases). Typically, tumors occur in the small intestines (duodenum, jejunum, or ileum) and are multiple in 60% of cases. Their size is variable, ranging from 10–0.1 cm in published series.110–112 The majority of them are small and have a low mitotic index. They are considered to have a good prognosis, but malignant behavior and subsequent disease-related death have been reported.112 Histologically, the great majority are of spindle cell phenotype, and almost all are positive for KIT immunohistochemistry. Nevertheless, KIT or PDGFRA mutation are usually absent in these GISTs.110–112 Thus, pathogenesis pathways involved in their development are different for sporadic GISTs, and some particular biological features have been described. NF-1-related GISTs are characterized by a low level of constitutive KIT autophosphorylation despite comparable KIT expression, a strong expression and activation of MAP-kinase pathways, a less strong activation of PI3K-AKT and JAK-STAT3 pathways than in sporadic GISTs, and the near absence of neurofibromin expression.113,114 Mitotic recombination or other genetic mechanisms resulting in the loss of wild-type allele and NF1 heterozygosity may be the key molecular event underlying GIST development in NF-1 patients.113,115 There are no clear data about their imatinib sensitivity, but in vitro data suggest that NF-1-related GISTs are not well controlled by imatinib, similarly to wild-type GISTs in general.113
Carney triad and Carney–Stratakis syndrome
Carney triad is a very rare tumor syndrome that was described in 1977.116 A heritable disorder was suspected because of multifocal tumors in multiple organs and the young age of patients. however, it was not a familial disorder. Typically, Carney triad-associated functioning includes extra-adrenal paraganglioma, pulmonary chondroma, and epithelioid gastric leiomyosarcoma, which were subsequently referred to as GIST.117 This disorder has a strong female predilection. In 1999, based on the analysis of 79 cases, adrenocortical adenoma and esophageal leiomyoma were added to the Carney syndrome, and the presence of two of the three initial components was defined sufficient for diagnostic as only 20% of cases had the third component.118 Analysis of features of gastric tumors in 104 patients with Carney syndrome show major differences from sporadic gastric GISTs.119 All patients had gastric tumors and at least one of the other Carney syndrome components. Median age at diagnosis was 18 years (ranged from 6 to 53 years), and about 90% of patients were female. Multiple gastric tumors were found in each patient; they were preferentially localized in the antrum (61%) with a mean size of 6.3 cm (ranged from 0.2 to 18 cm). Histologically, two-thirds were of epithelioid phenotype and all were KIT positive. Interestingly, there was no correlation of the assessed risk using the 2002 classification23 with subsequent malignant behavior. Lymph nodes were frequently involved. After a median follow-up of 21 years, 15 patients (14%) died of gastric tumor evolution or therapeutic complication. Among the 16 patients treated by imatinib, the majority had no tumor response (62%).119KIT or PDGFRA mutations are not found in gastric tumors of Carney syndrome, and the molecular events implicated in their pathogenesis are not known at present.120 Because of the clinical, pathological, and biological differences observed between gastric tumors of Carney syndrome and sporadic gastric GISTs, it was concluded that these tumors should not be labeled as GISTs but as gastric stromal tumors.119
In 2002, a new syndrome distinct from the Carney triad was described and named Carney–Stratakis syndrome. This syndrome was defined as an association of multiple paraganglioma and gastric GISTs occurring in an autosomal-dominant manner with incomplete penetrance.121 Gastric tumors of this syndrome have been described from five families. Clinical and pathological features of gastric tumors were very similar than those described for gastric stromal tumors of Carney triad.121 Subsequently, molecular abnormalities were investigated in 11 patients from nine unrelated families. Patients had gastric GIST and/or paraganglioma, and both tumor types were found in almost all families.122 No KIT or PDGFRA mutations were found, but eight patients from seven families had a germline mutation in one of the three succinate dehydrogenase subunit-encoding genes (SDHB, SDHC, or SDHD). As in NF-1, allelic marker analysis suggested loss of wild-type allele of SDH gene in gastric stromal tumors and paragangliomas.122 These genes are implicated in mitochondrial tumor suppressor pathways, and SDH mutations have been described in sporadic or familial pheochromocytomas or extra-adrenal paragangliomas.123
Particular cases of pediatric GISTs
Pediatric GISTs are rare, and account for 1%–2% of all GISTs.124,125 Some of them can occur in a genetic predisposition matter (NF-1, Carney–Stratakis syndrome, KIT, or PDGFRA germline mutation), but most are sporadic. Pediatric GISTs are characterized by strong female predominance, gastric location in most cases, frequent multiple synchronous tumors and/or lymph node metastases, predominant epithelioid morphology, and the absence of KIT or PDGFRA mutations in most cases. Long survival may occur, even in the presence of metastases.124,125 Imatinib treatment may be less effective, and sunitinib, sorafenib, or third-generation tyrosine kinase inhibitors (dasatinib and nilotinib) have higher efficacy than imatinib in vitro.126 KIT downstream targets are activated in wild-type pediatric GISTs as in sporadic adult GISTs.126 However, gene expression analysis showed that wild-type pediatric GISTs (in Carney triad or not) had a distinct transcriptional signature than wild-type adult GISTs.125,126
Thus, pediatric GISTs (and most GISTs of adults under 30 years) are identical to Carney triad GISTs, clinically, morphologically, and genetically, and may represent a forme fruste of this syndrome or, alternatively, an independent subclass of GISTs with a same molecular pathogenesis. Data from paragangliomas and pheochromocytomas suggested that negative staining for SDHB was a marker of mitochondrial complex II dysfunction, and this was regardless of which protein was disrupted.127,128 Thus, subsequently of the discovery of the role of SDH mutations in Carney–Stratakis syndrome, some authors evaluated SDHB immunostaining in GISTs. Further supporting the hypothesis of a same pathogenesis, GISTs of Carney triad and pediatric wild-type GIST were negative for SDHB staining even without SDH mutations, while all GISTs with KIT mutations and NF-1-related GISTs were positive.129,130 Moreover, in sporadic adult GISTs, about 3% of tumors were negative for SDHB staining,129 and a germline mutation in SDHB, C, or D was found in 16% of KIT/PDGFRA wild-type GIST.130 Therefore, abnormalities of mitochondrial complex 2, attributable to SDH germline mutation or not, may be implicated in another oncogenic pathway than KIT/PDGFRAs. Additional studies on SDHB are necessary before use in routine practice.
Germline mutations of KIT or PDGFRA
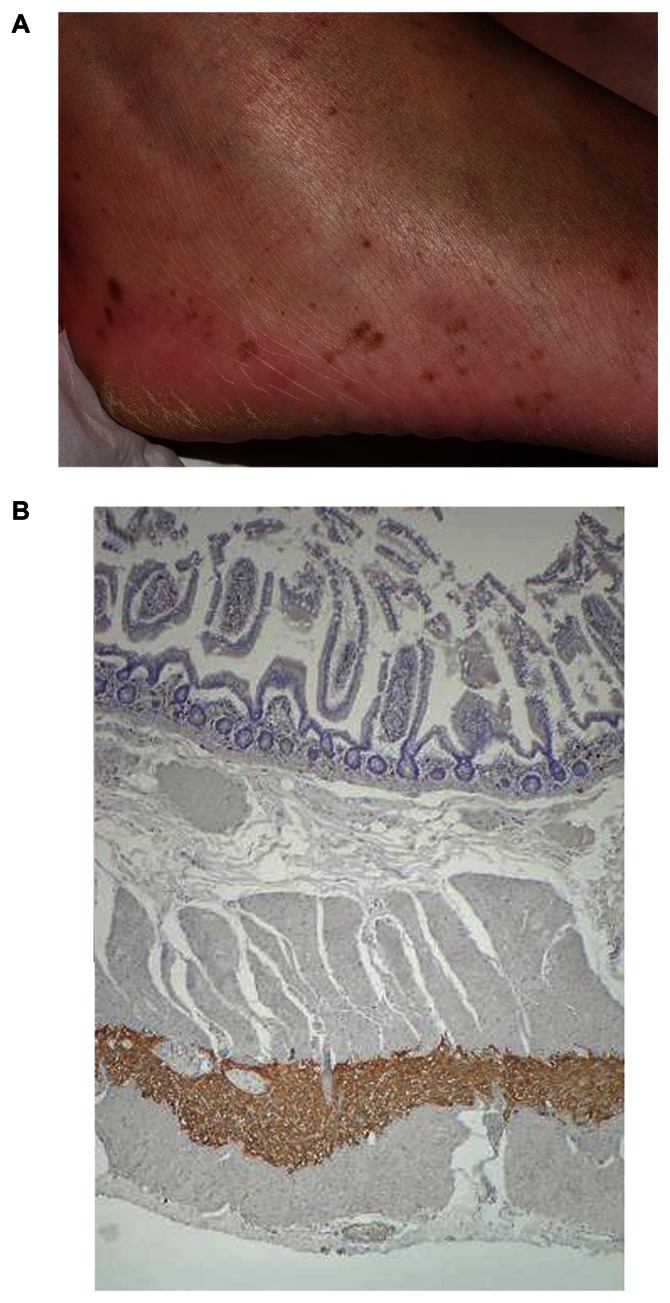
The first family with a germline KIT mutation has been described in 1998, only a few months after the first report of activating KIT mutations in most of sporadic GISTs.3,131 Since this date, many germline KIT or PDGFRA mutations have been identified in rare kindreds with multiple occurrences of GIST among blood relatives. Today, 16 different germline mutations involving exons 8, 11, 13, or 17 of KIT or exons 12 or 14 of PDGFRA have been reported to our knowledge (Table 3).131–150 In all publications, the pattern of inheritance is autosomal dominant with a penetrance of almost 100%. GISTs are multiple in most cases and frequently multifocal. They occurred at earlier ages than corresponding sporadic GISTs, commonly in young adults, and median age at diagnosis is approximately 40–50 years. Some authors have suggested a possible genetic anticipation because of decreasing age at diagnosis across generations.141,144,150 However, it was not reported previously, and earlier ages at diagnosis might be only due to greater interest for GISTs and/or screening of relatives. Various clinical features have been reported in these families and are described according to activating germline mutation in Table 3. Skin lesions features appeared to be frequent (Figure 4A), in particular in cases of KIT exon 11 mutation. The molecular mechanism of their occurrence is unclear, but, interestingly, a decrease in the number of pigmented lesions has been noted in one patient treated with imatinib given an additional proof of their relationship with activating KIT mutations.151
Table 3.
Germline mutations and clinical features
| Germline mutations | No. of relatives with GISTsa | GIST location | Particular clinical features | Imatinib responseb |
|---|---|---|---|---|
| KIT | ||||
| Exon 11 | ||||
| W557R144 | 15 | G, SI | Skin hyperpigmentation, dysphagia | |
| V559A135,138,142,147,148 | 10 | G, SI | Skin hyperpigmentation, lentigines, urticaria pigmentosa, nevi | –c |
| Del559131 | 7 | –c | Skin hyperpigmentation | –c |
| V560G138 | 4 | SI | Skin hyperpigmentation | |
| L576-P577 insQL134 | 3 | G, SI | Skin hyperpigmentation | –c |
| Q575_P577delinsH132 | 2 | R | –c | PR or SD |
| Del579133,137,145 | 27 | G, SI, C | Skin hyperpigmentation, nevi | SD |
| Exon 13 | ||||
| K642E140,149 | 3 | O, G, SI, R | Lentigines, dysphagia (unpublished data) | SD |
| Exon 17 | ||||
| D820Y141,146,150 | 15 | G, SI, R | Dysphagia | PR or SD |
| N822Y136 | 4 | G, SI | –c | PR or SD |
| PDGFRA | ||||
| Exon 12 | ||||
| V561D139 | 1 | G | Fibrous polyps and lipomas of the small intestine | –c |
| Exon 14 | ||||
| N846Y143 | 7 | G | Large hands | –c |
Notes:
Confirmed or suspected by medical record or death certificate.
In all cases in patients with advanced GIST.
Not described.
Abbreviations: GIST, gastrointestinal stromal tumors; O, esophagi; G, gastric; SI, small intestine; C, colon; R, rectum; PR, partial response; SD, stable disease.
Figure 4.
Clinical and histological features in patients with KIT germline mutations. A) Diffuse lentigines in a patient with KIT exon 13 germline mutation. B) Hyperplasia of interstitial cells of Cajal in the myenteric plexus layer of small intestine of the same patient.
Histologically, a diffuse proliferation of interstitial cells of Cajal is found in the myenteric plexus layer of gastrointestinal tract of affected cases (Figure 4B). This proliferation is a polyclonal hyperplasia, while GISTs are monoclonal.152 This is of particular interest because it demonstrates that activating KIT or PDGFRA mutations are necessary but not sufficient for GIST pathogenesis. Moreover, GISTs from a same patient with germline mutation are derived from independent clones and show different cytogenetic abnormalities, suggesting that different progression pathways can lead to GIST from polyclonal hyperplasia.150,152 This point is supported by analysis of cytogenetic abnormalities in sporadic GISTs. In them, losses at chromosomes 1p, 14q, and 22q are frequent and characteristic of the abnormalities.153 Three major cytogenetic pathways associated with GIST location and behaviors have been suggested from these alterations.154 Nevertheless, apart from activating mutations of KIT or PDFGRA, our understanding of fine molecular alterations implicated in GISTs pathogenesis are limited. At present, there is no recommendation for screening, follow-up, and treatment of patients harboring germline mutation. All patients will develop GISTs, but not all of these tumors will be malignant. Imatinib chemoprevention is questionable as patients may have long-term side effects, and it is not clear that there is any role for lifelong therapy. In the absence of a method allowing the determination of GIST evolution, aggressive treatment such as repeated surgery should be as limited as possible, and it is probable that only growing or large GISTs should be resected. The National Institutes of Health is currently sponsoring a research study focusing on learning more about hereditary GISTs: the Project Flag (http://www.projectflag.org/).
Three murine models of GIST harboring germline KIT mutations have been developed using expression of KIT mutant in transgenic knock-in mice. KIT mutations used are three known human germline mutations and involved exons 11 (558delV corresponding to human 559delV), 13 (D818Y corresponding to human D820Y), or 17 (K641E corresponding to human K642E). Heterozygous and homozygous mice developed diffuse hyperplasia of interstitial cells of Cajal in the digestive tract (from esophagus to large intestine) and GIST-like cecal tumor.155–157 Homozygous mice developed larger cecal tumor and died due to gastrointestinal obstruction after some weeks.156,157 There is no clear explanation for the differences observed in GIST location between murine model and human. However, these models are promising to improve our knowledge of KIT oncogenic signaling pathways. Moreover, mouse models will be useful for the preclinical investigation of new targeted therapies and overcome secondary resistance mutations.158,159
Conclusion
GISTs receive today considerable attention because of substantial advances in the understanding of their pathogenesis and development of efficient targeted therapies. The diagnostic of this disease relies on concordant histology and KIT immunopositivity. Mutational analysis for activating KIT and PDGFRA mutations is strongly recommended in KIT-negative tumors and in all advanced GISTs. In the future, patients’ treatment will probably be determined according to the predictive value of primary and/or secondary mutations. Activating mutations of KIT or PDGFRA protooncogenes are present in more than 80% of GISTs. Nevertheless, molecular analysis of GISTs occurring in patients with genetic predisposition have shown that some GISTs or GIST-like tumors are secondary to inactivating mutations of tumor suppressor genes like SDH or NF1.
Figure 3.
Schematic representation of KIT and PDGFRA receptor tyrosine-kinase.
Abbreviations: ATP, adenosine triphosphate; PDGFRA, platelet-derived growth factor receptor alpha polypeptide.
Acknowledgment
This work was supported by grants from AROLD.
Footnotes
Disclosure
Jean-François Emile serves as a consultant or advisor for Novartis and receives honoraria from Novartis.
References
- 1.Hirota S, Isozaki K, Moriyama Y, et al. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279(5350):577–580. doi: 10.1126/science.279.5350.577. [DOI] [PubMed] [Google Scholar]
- 2.Kindblom LG, Remotti HE, Aldenborg F, Meis-Kindblom JM. Gastrointestinal pacemaker cell tumor (GIPACT): gastrointestinal stromal tumors show phenotypic characteristics of the interstitial cells of cajal. Am J Pathol. 1998;152(5):1259–1269. [PMC free article] [PubMed] [Google Scholar]
- 3.Lee HT, Hennig GW, Fleming NW, et al. The mechanism and spread of pacemaker activity through myenteric interstitial cells of cajal in human small intestine. Gastroenterology. 2007;132(5):1852–1865. doi: 10.1053/j.gastro.2007.02.049. [DOI] [PubMed] [Google Scholar]
- 4.Heinrich MC, Corless CL, Duensing A, et al. PDGFRA activating mutations in gastrointestinal stromal tumors. Science. 2003;299(5607):708–710. doi: 10.1126/science.1079666. [DOI] [PubMed] [Google Scholar]
- 5.Rubin BP, Heinrich MC, Corless CL. Gastrointestinal stromal tumour. Lancet. 2007;369(9574):1731–1741. doi: 10.1016/S0140-6736(07)60780-6. [DOI] [PubMed] [Google Scholar]
- 6.Emile JF, Théou N, Tabone S, et al. Clinicopathologic, phenotypic, and genotypic characteristics of gastrointestinal mesenchymal tumors. Clin Gastroenterol Hepatol. 2004;2(7):597–605. doi: 10.1016/s1542-3565(04)00243-5. [DOI] [PubMed] [Google Scholar]
- 7.Miettinen M, Sobin LH, Lasota J. Gastrointestinal stromal tumors of the stomach. A clinicopathologic, immunohistochemical, and molecular genetic study of 1765 cases with long-term follow-up. Am J Surg Pathol. 2005;29(1):52–68. doi: 10.1097/01.pas.0000146010.92933.de. [DOI] [PubMed] [Google Scholar]
- 8.Miettinen M, Makhlouf H, Sobin LH, Lasota J. Gastrointes tinal stromal tumors of the jejunum and ileum. A clinicopathologic, immunohistochemical, and molecular genetic study of 906 cases before imatinib with long-term follow-up. Am J Surg Pathol. 2006;30(4):477–489. doi: 10.1097/00000478-200604000-00008. [DOI] [PubMed] [Google Scholar]
- 9.Nilsson B, Bümming P, Meis-Kindblom JM, et al. Gastrointestinal stromal tumors: the incidence, prevalence, clinical course, and prognostication in the preimatinib mesylate era. A population-based study in western Sweden. Cancer. 2005;103(4):821–829. doi: 10.1002/cncr.20862. [DOI] [PubMed] [Google Scholar]
- 10.Tryggvason G, Gislason HG, Magnusson MK, Jonasson JG. Gastrointestinal stromal tumors in Iceland, 1990–2003: the Icelandic GIST study, a population-based incidence and pathologic risk stratification study. Int J Cancer. 2005;117(2):289–293. doi: 10.1002/ijc.21167. [DOI] [PubMed] [Google Scholar]
- 11.Goettsch WG, Bos SD, Breekveldt-Postma N, Casparie M, Herings RM, Hogendoorn PC. Incidence of gastrointestinal stromal tumours is underestimated: results of a nation-wide study. Eur J Cancer. 2005;41(18):2868–2872. doi: 10.1016/j.ejca.2005.09.009. [DOI] [PubMed] [Google Scholar]
- 12.Agaimy A, Wünsch PH, Hofstaedter F, et al. Minute gastric sclerosing stromal tumors (GIST tumorlets) are common in adults and frequently show c-KIT mutations. Am J Surg Pathol. 2007;31(1):113–120. doi: 10.1097/01.pas.0000213307.05811.f0. [DOI] [PubMed] [Google Scholar]
- 13.Yamamoto H, Oda Y, Kawaguchi K, et al. c-kit and PDGFRA mutations in extragastrointestinal stromal tumor (gastrointestinal stromal tumor of the soft tissue) Am J Surg Pathol. 2004;28(4):479–488. doi: 10.1097/00000478-200404000-00007. [DOI] [PubMed] [Google Scholar]
- 14.Burkill GJ, Badran M, Al-Muderis O, et al. Malignant gastrointestinal stromal tumor: distribution, imaging features, and pattern of metastatic spread. Radiology. 2003;226(2):527–532. doi: 10.1148/radiol.2262011880. [DOI] [PubMed] [Google Scholar]
- 15.DeMatteo RP, Lewis JJ, Leung D, Mudan SS, Woodruff JM, Brennan MF. Two hundred gastrointestinal stromal tumors: recurrence patterns and prognostic factors for survival. Ann Surg. 2000;231(1):51–58. doi: 10.1097/00000658-200001000-00008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Blay JY, Bonvalot S, Casali P, et al. Consensus meeting for the management of gastrointestinal stromal tumors. Report of the GIST Consensus Conference of 2004 Mar 20–21, under the auspices of ESMO. Ann Oncol. 2005;16(4):566–578. doi: 10.1093/annonc/mdi127. [DOI] [PubMed] [Google Scholar]
- 17.Emory TS, Sobin LH, Lukes L, Lee DH, O’Leary TJ. Prognosis of gastrointestinal smooth-muscle (stromal) tumors: dependence on anatomic site. Am J Surg Pathol. 1999;23(1):82–87. doi: 10.1097/00000478-199901000-00009. [DOI] [PubMed] [Google Scholar]
- 18.Franquemont DW. Differentiation and risk assessment of gastrointestinal stromal tumors. Am J Clin Pathol. 1995;103(1):41–47. doi: 10.1093/ajcp/103.1.41. [DOI] [PubMed] [Google Scholar]
- 19.Hasegawa T, Matsuno Y, Shimoda T, Hirohashi S. Gastrointestinal stromal tumor: consistent CD117 immunostaining for diagnosis, and prognostic classification based on tumor size and MIB-1 grade. Hum Pathol. 2002;33(6):669–676. doi: 10.1053/hupa.2002.124116. [DOI] [PubMed] [Google Scholar]
- 20.Sabah M, Cummins R, Leader M, Kay E. Loss of heterozygosity of chromosome 9p and loss of p16INK4A expression are associated with malignant gastrointestinal stromal tumors. Mod Pathol. 2004;17(11):1364–1371. doi: 10.1038/modpathol.3800199. [DOI] [PubMed] [Google Scholar]
- 21.Nakamura N, Yamamoto H, Yao T, et al. Prognostic significance of expressions of cell-cycle regulatory proteins in gastrointestinal stromal tumor and the relevance of the risk grade. Hum Pathol. 2005;36(7):828–837. doi: 10.1016/j.humpath.2005.03.012. [DOI] [PubMed] [Google Scholar]
- 22.Imamura M, Yamamoto H, Nakamura N, et al. Prognostic significance of angiogenesis in gastrointestinal stromal tumor. Mod Pathol. 2007;20(5):529–537. doi: 10.1038/modpathol.3800767. [DOI] [PubMed] [Google Scholar]
- 23.Fletcher CD, Berman JJ, Corless C, et al. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol. 2002;33(5):459–465. doi: 10.1053/hupa.2002.123545. [DOI] [PubMed] [Google Scholar]
- 24.Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006;23(2):70–83. doi: 10.1053/j.semdp.2006.09.001. [DOI] [PubMed] [Google Scholar]
- 25.Gold JS, Gönen M, Gutiérrez A, et al. Development and validation of a prognostic nomogram for recurrence-free survival after complete surgical resection of localised primary gastrointestinal stromal tumour: a retrospective analysis. Lancet Oncol. 2009;10(11):1045–1052. doi: 10.1016/S1470-2045(09)70242-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Joenssu H, Roberts PJ, Sarlomo-Rikala M, et al. Effect of the tyrosine kinase inhibitor STI571 in a patient with a metastatic gastrointestinal stromal tumor. N Engl J Med. 2001;344(14):1052–1056. doi: 10.1056/NEJM200104053441404. [DOI] [PubMed] [Google Scholar]
- 27.Blanke CD, Rankin C, Demetri GD, et al. Phase III randomized, intergroup trial assessing imatinib mesylate at two dose levels in patients with unresectable or metastatic gastrointestinal stromal tumors expressing the kit receptor tyrosine kinase: S0033. J Clin Oncol. 2008;26(4):626–632. doi: 10.1200/JCO.2007.13.4452. [DOI] [PubMed] [Google Scholar]
- 28.Verweij J, Casali PG, Zalcberg J, et al. Progression-free survival in gastrointestinal stromal tumours with high-dose imatinib: randomised trial. Lancet. 2004;364(9440):1127–1134. doi: 10.1016/S0140-6736(04)17098-0. [DOI] [PubMed] [Google Scholar]
- 29.Blay JY, Le Cesne A, Ray-Coquard I, et al. Prospective multicentric randomized phase III study of imatinib in patients with advanced gastrointestinal stromal tumors comparing interruption versus continuation of treatment beyond 1 year: the French Sarcoma Group. J Clin Oncol. 2007;25(9):1107–1113. doi: 10.1200/JCO.2006.09.0183. [DOI] [PubMed] [Google Scholar]
- 30.DeMatteo RP, Maki RG, Singer S, Gonen M, Brennan MF, Antonescu CR. Results of tyrosine kinase inhibitor therapy followed by surgical resection for metastatic gastrointestinal stromal tumor. Ann Surg. 2007;245(3):347–352. doi: 10.1097/01.sla.0000236630.93587.59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Gronchi A, Fiore M, Miselli F, et al. Surgery of residual disease following molecular-targeted therapy with imatinib mesylate in advanced/metastatic GIST. Ann Surg. 2007;245(3):341–346. doi: 10.1097/01.sla.0000242710.36384.1b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mussi C, Ronellenfitsch U, Jakob J, et al. Post-imatinib surgery in advanced/metastatic GIST: is it worthwhile in all patients? Ann Oncol. 2010;21(2):403–408. doi: 10.1093/annonc/mdp310. [DOI] [PubMed] [Google Scholar]
- 33.Demetri GD, van Oosterom AT, Garrett CR, et al. Efficacy and safety of sunitinib in patients with advanced gastrointestinal stromal tumour after failure of imatinib: a randomised controlled trial. Lancet. 2006;368(9544):1329–1338. doi: 10.1016/S0140-6736(06)69446-4. [DOI] [PubMed] [Google Scholar]
- 34.George S, Blay JY, Casali PG, et al. Clinical evaluation of continuous daily dosing of sunitinib malate in patients with advanced gastrointestinal stromal tumour after imatinib failure. Eur J Cancer. 2009;45(11):1959–1968. doi: 10.1016/j.ejca.2009.02.011. [DOI] [PubMed] [Google Scholar]
- 35.Wiebe L, Kasza KE, Maki RG, et al. Activity of sorafenib (SOR) in patients (pts) with imatinib (IM) and sunitinib (SU)-resistant (RES) gastrointestinal stromal tumors (GIST): a phase II trial of the University of Chicago phase II consortium. J Clin Oncol. 2008;26(15S):10502. [Google Scholar]
- 36.Montemurro M, Schöffski P, Reichardt P, et al. Nilotinib in the treatment of advanced gastrointestinal stromal tumours resistant to both imatinib and sunitinib. Eur J Cancer. 2009;45(13):2293–2297. doi: 10.1016/j.ejca.2009.04.030. [DOI] [PubMed] [Google Scholar]
- 37.Evans TRJ, Morgan JA, van den Abbeele AD, et al. Phase I dose- escalation study of the SRC and multi-kinase inhibitor BMS-354825 in patients (pts) with GIST and other solid tumors. J Clin Oncol. 2005;23(16S):3034. [Google Scholar]
- 38.DeMatteo RP, Ballman KV, Antonescu CR, et al. Adjuvant imatinib mesylate after resection of localised, primary gastrointestinal stromal tumour: a randomised, double-blind, placebo-controlled trial. Lancet. 2009;373(9669):1097–1104. doi: 10.1016/S0140-6736(09)60500-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Casali PG, Jost L, Reichardt P, Schlemmer M, Blay JY. Gastrointestinal stromal tumors: ESMO clinical recommendations for diagnosis, treatment and follow-up. Ann Oncol. 2008;19(Suppl 2):ii35–ii38. doi: 10.1093/annonc/mdn080. [DOI] [PubMed] [Google Scholar]
- 40.Fernandez-Esparrach G, Sendino O, Solé M, et al. Endoscopic ultrasound- guided fine-needle aspiration and trucut biopsy in the diagnosis of gastric stromal tumors: a randomized crossover study. Endoscopy. 2010;42(4):292–299. doi: 10.1055/s-0029-1244074. [DOI] [PubMed] [Google Scholar]
- 41.Philipper M, Hollerbach S, Gabbert HE, et al. Prospective comparison of endoscopic ultrasound-guided fine-needle aspiration and surgical histology in upper gastrointestinal submucosal tumors. Endoscopy. 2010;42(4):300–305. doi: 10.1055/s-0029-1244006. [DOI] [PubMed] [Google Scholar]
- 42.Gayed I, Vu T, Iyer R, et al. The role of 18F-FDG PET in staging and early prediction of response to therapy of recurrent gastrointestinal stromal tumors. J Nucl Med. 2004;45(1):17–21. [PubMed] [Google Scholar]
- 43.Rosenbaum SJ, Stergar H, Antoch G, Veit P, Bockisch A, Kühl H. Staging and follow-up of gastrointestinal stromal tumors with PET/CT. Abdom Imaging. 2006;31(1):25–35. doi: 10.1007/s00261-005-0031-3. [DOI] [PubMed] [Google Scholar]
- 44.Stroobants S, Goeminne J, Seegers M, et al. 18FDG-Positron emission tomography for the early prediction of response in advanced soft tissue sarcoma treated with imatinib mesylate (Glivec®) Eur J Cancer. 2003;39(14):2012–2020. doi: 10.1016/s0959-8049(03)00073-x. [DOI] [PubMed] [Google Scholar]
- 45.Choi H, Charnsangavej C, Faria SC, et al. Correlation of computed tomography and positron emission tomography in patients with metastatic gastrointestinal stromal tumor treated at a single institution with imatinib mesylate: proposal of new computed tomography response criteria. J Clin Oncol. 2007;25(13):1753–1759. doi: 10.1200/JCO.2006.07.3049. [DOI] [PubMed] [Google Scholar]
- 46.Emile JF, Stock N, Corless CL, et al. Dotlike or Golgi-like KIT and PDGFRA staining in GISTs. Am J Surg Pathol. 2009;33(1):157–158. doi: 10.1097/PAS.0b013e3181883765. [DOI] [PubMed] [Google Scholar]
- 47.Tabone-Eglinger S, Subra F, El Sayadi H, et al. KIT mutations induce intracellular retention and activation of an immature form of the KIT protein in gastrointestinal stromal tumors. Clin Cancer Res. 2008;14(8):2285–2294. doi: 10.1158/1078-0432.CCR-07-4102. [DOI] [PubMed] [Google Scholar]
- 48.Sihto H, Sarlomo-Rikala M, Tynninen O, et al. KIT and platelet-derived growth factor receptor alpha tyrosine kinase gene mutations and KIT amplifications in human solid tumors. J Clin Oncol. 2005;23(1):49–57. doi: 10.1200/JCO.2005.02.093. [DOI] [PubMed] [Google Scholar]
- 49.Torres-Cabala CA, Wang WL, Trent J, et al. Correlation between KIT expression and KIT mutation in melanoma: a study of 173 cases with emphasis on the acral-lentiginous/mucosal type. Mod Pathol. 2009;22(11):1446–1456. doi: 10.1038/modpathol.2009.116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Dow N, Giblen G, Sobin LH, Miettinen M. Gastrointestinal stromal tumors: differential diagnosis. Semin Diagn Pathol. 2006;23(2):111–119. doi: 10.1053/j.semdp.2006.09.002. [DOI] [PubMed] [Google Scholar]
- 51.Miettinen M, Sobin LH, Sarlomo-Rikala M. Immunohistochemical spectrum of GISTs at different sites and their differential diagnosis with a reference to CD117 (KIT) Mod Pathol. 2000;13(10):1134–1142. doi: 10.1038/modpathol.3880210. [DOI] [PubMed] [Google Scholar]
- 52.West RB, Corless CL, Chen X, et al. The novel marker, DOG1, is expressed ubiquitously in gastrointestinal stromal tumors irrespective of KIT or PDGFRA mutation status. Am J Pathol. 2004;165(1):107–113. doi: 10.1016/S0002-9440(10)63279-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Emile JF, Lemoine A, Bienfait N, Terrier P, Azoulay D, Debuire B. Length analysis of polymerase chain reaction products: a sensitive and reliable technique for the detection of mutations in KIT exon 11 in gastrointestinal stromal tumors. Diagn Mol Pathol. 2002;11(2):107–112. doi: 10.1097/00019606-200206000-00007. [DOI] [PubMed] [Google Scholar]
- 54.Blume-Jensen P, Claesson-Welsh L, Siegbahn A, Zsebo KM, Westermark B, Heldin CH. Activation of the human c-kit product by ligand-induced dimerization mediates circular actin reorganization and chemotaxis. EMBO J. 1991;10(13):4121–4128. doi: 10.1002/j.1460-2075.1991.tb04989.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Corless CL, Fletcher JA, Heinrich MC. Biology of gastrointestinal tumors. J Clin Oncol. 2004;22(18):3813–3825. doi: 10.1200/JCO.2004.05.140. [DOI] [PubMed] [Google Scholar]
- 56.Mol CD, Dougan DR, Schneider TR, et al. Structural basis for the autoinhibition and STI-571 inhibition of c-Kit tyrosine kinase. J Biol Chem. 2004;279(30):31655–31663. doi: 10.1074/jbc.M403319200. [DOI] [PubMed] [Google Scholar]
- 57.Chan PM, Ilangumaran S, La Rose J, Chakrabartty A, Rottapel R. Autoinhibition of the kit receptor tyrosine kinase by the cytosolic juxtamembrane region. Mol Cell Biol. 2003;23(9):3067–3078. doi: 10.1128/MCB.23.9.3067-3078.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Kitayama H, Kanakura Y, Furitsu T, et al. Constitutively activating mutations of c-kit receptor tyrosine kinase confer factor-independent growth and tumorigenicity of factor-dependent hematopoietic cell lines. Blood. 1995;85(3):790–798. [PubMed] [Google Scholar]
- 59.Roskoski R., Jr Structure and regulation of Kit protein-tyrosine kinase – the stem cell factor receptor. Biochem Biophys Res Commun. 2005;338(3):1307–1315. doi: 10.1016/j.bbrc.2005.09.150. [DOI] [PubMed] [Google Scholar]
- 60.Ernst SI, Hubbs AE, Przygodzki RM, Emory TS, Sobin LH, O’Leary TJ. KIT mutation portends poor prognosis in gastrointestinal stromal/smooth muscle tumors. Lab Invest. 1998;78(12):1633–1636. [PubMed] [Google Scholar]
- 61.Taniguchi M, Nishida T, Hirota S, et al. Effect of c-kit mutation on prognosis of gastrointestinal stromal tumors. Cancer Res. 1999;59(17):4297–4300. [PubMed] [Google Scholar]
- 62.Debiec-Rychter M, Dumez H, Judson I, et al. Use of c-KIT/PDGFRA mutational analysis to predict the clinical response to imatinib in patients with advanced gastrointestinal stromal tumours entered on phase I and II studies of the EORTC Soft Tissue and Bone Sarcoma Group. Eur J Cancer. 2004;40(5):689–695. doi: 10.1016/j.ejca.2003.11.025. [DOI] [PubMed] [Google Scholar]
- 63.Penzel R, Aulmann S, Moock M, Schwarzbach M, Rieker RJ, Mechtersheimer G. The location of KIT and PDGFRA gene mutations in gastrointestinal stromal tumors is site and phenotype associated. J Clin Pathol. 2005;58(6):634–639. doi: 10.1136/jcp.2004.021766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Martin J, Poveda A, Llombart-Bosch A, et al. Deletions affecting codons 557–558 of the c-KIT gene indicate a poor prognosis in patients with completely resected gastrointestinal stromal tumors: a study by the Spanish Group for Sarcoma Research (GEIS) J Clin Oncol. 2005;23(25):6190–6198. doi: 10.1200/JCO.2005.19.554. [DOI] [PubMed] [Google Scholar]
- 65.Wardelmann E, Hrychyk A, Merkelbach-Bruse S, et al. Association of platelet-derived growth factor receptor alpha mutations with gastric primary site and epithelioid or mixed cell morphology in gastrointestinal stromal tumors. J Mol Diagn. 2004;6(3):197–204. doi: 10.1016/s1525-1578(10)60510-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Debiec-Rychter M, Sciot R, Le Cesne A, et al. KIT mutations and dose selection for imatinib in patients with advanced gastrointestinal stromal tumours. Eur J Cancer. 2006;42(8):1093–1103. doi: 10.1016/j.ejca.2006.01.030. [DOI] [PubMed] [Google Scholar]
- 67.Andersson J, Bümming P, Meis-Kindblom JM, et al. Gastrointestinal stromal tumors with exon 11 deletions are associated with poor prognosis. Gastroenterology. 2006;130(6):1573–1581. doi: 10.1053/j.gastro.2006.01.043. [DOI] [PubMed] [Google Scholar]
- 68.DeMatteo RP, Gold JS, Saran L, et al. Tumor mitotic rate, size, and location independently predict recurrence after resection of primary gastrointestinal stromal tumor (GIST) Cancer. 2008;112(3):608–615. doi: 10.1002/cncr.23199. [DOI] [PubMed] [Google Scholar]
- 69.Emile JF, Tabone-Eglinger S, Théou-Anton N, Lemoine A. Prognostic value of KIT exon 11 deletions in GISTs. Gastroenterology. 2006;131(3):976–977. doi: 10.1053/j.gastro.2006.07.042. [DOI] [PubMed] [Google Scholar]
- 70.Corless CL, Schroeder A, Griffith D, et al. PDGFRA mutations in gastrointestinal stromal tumors: frequency, spectrum and in vitro sensitivity to imatinib. J Clin Oncol. 2005;23(23):5357–5364. doi: 10.1200/JCO.2005.14.068. [DOI] [PubMed] [Google Scholar]
- 71.Lasota J, vel Dobosz AJ, Wasag B, et al. Presence of homozygous KIT exon 11 mutations is strongly associated with malignant clinical behavior in gastrointestinal stromal tumors. Lab Invest. 2007;87(10):1029–1041. doi: 10.1038/labinvest.3700628. [DOI] [PubMed] [Google Scholar]
- 72.Crosier PS, Ricciardi ST, Hall LR, Vitas MR, Clark SC, Crosier KE. Expression of isoforms of the human receptor tyrosine kinase c-kit in leukemic cell lines and acute myeloid leukemia. Blood. 1993;82(4):1151–1158. [PubMed] [Google Scholar]
- 73.Caruana G, Cambareri AC, Ashman LK. Isoforms of c-KIT differ in activation of signalling pathways and transformation of NIH3T3 fibroblasts. Oncogene. 1999;18(40):5573–5581. doi: 10.1038/sj.onc.1202939. [DOI] [PubMed] [Google Scholar]
- 74.Théou N, Tabone S, Saffroy R, et al. High expression of both mutant and wild–type alleles of c-kit in gastrointestinal stromal tumors. Biochim Biophys Acta. 2004;1688(3):250–256. doi: 10.1016/j.bbadis.2003.12.011. [DOI] [PubMed] [Google Scholar]
- 75.Tabone S, Théou N, Wozniak A, et al. KIT overexpression and amplification in gastrointestinal stromal tumors (GISTs) Biochim Biophys Acta. 2005;1741(1–2):65–72. doi: 10.1016/j.bbadis.2005.03.011. [DOI] [PubMed] [Google Scholar]
- 76.Théou-Anton N, Tabone S, Brouty-Boyé D, et al. Co expression of SCF and KIT in gastrointestinal stromal tumours (GISTs) suggests an autocrine/paracrine mechanism. Br J Cancer. 2006;94(8):1180–1185. doi: 10.1038/sj.bjc.6603063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Duensing A, Medeiros F, McConarty B, et al. Mechanisms of oncogenic KIT signal transduction in primary gastrointestinal stromal tumors (GISTs) Oncogene. 2004;23(22):3999–4006. doi: 10.1038/sj.onc.1207525. [DOI] [PubMed] [Google Scholar]
- 78.Lux ML, Rubin BP, Biase TL, et al. KIT extracellular and kinase domain mutations in gastrointestinal stromal tumors. Am J Pathol. 2000;156(3):791–795. doi: 10.1016/S0002-9440(10)64946-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rubin BP, Singer S, Tsao C, et al. KIT activation is a ubiquitous feature of gastrointestinal stromal tumors. Cancer Res. 2001;61(22):8118–8121. [PubMed] [Google Scholar]
- 80.Xiang Z, Kreisel F, Cain J, Colson A, Tomasson MH. Neoplasia driven mutant c-KIT is mediated by intracellular, not plasma membrane, receptor signaling. Mol Cell Biol. 2007;27(1):267–82. doi: 10.1128/MCB.01153-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Emile JF, Bachet JB, Tabone-Eglinger S, Terrier P, Vignault JM. GIST with homozygous KIT exon 11 mutations. Lab Invest. 2008;88(5):456–457. doi: 10.1038/labinvest.2008.13. [DOI] [PubMed] [Google Scholar]
- 82.Hornick JL, Fletcher CD. The role of KIT in the management of patients with gastrointestinal stromal tumors. Human Pathol. 2007;38(5):679–687. doi: 10.1016/j.humpath.2007.03.001. [DOI] [PubMed] [Google Scholar]
- 83.Lasota J, Dansonka-Mieszkowska A, Sobin LH, Miettinen M. A great majority of GISTs with PDGFRA mutations represent gastric tumors of low or no malignant potential. Lab Invest. 2004;84(7):874–883. doi: 10.1038/labinvest.3700122. [DOI] [PubMed] [Google Scholar]
- 84.Bachet JB, Hostein I, Le Cesne A, et al. Prognosis and predictive value of KIT exon 11 deletion in GISTs. Br J Cancer. 2009;101(1):7–11. doi: 10.1038/sj.bjc.6605117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Wardelmann E, Losen I, Hans V, et al. Deletion of Tryp-557 and Lys-558 in the juxtamembrane domain of the c-kit protooncogene is associated with metastatic behavior of gastrointestinal stromal tumors. Int J Cancer. 2003;106(6):887–895. doi: 10.1002/ijc.11323. [DOI] [PubMed] [Google Scholar]
- 86.Heinrich MC, Corless CL, Demetri GD, et al. Kinase mutations and imatinib response in patients with metastatic gastrointestinal stromal tumor. J Clin Oncol. 2003;21(23):4342–4349. doi: 10.1200/JCO.2003.04.190. [DOI] [PubMed] [Google Scholar]
- 87.Gastrointestinal Stromal Tumor Meta-Analysis Group (MetaGIST) Comparison of two doses of imatinib for the treatment of unresectable or metastastic gastrointestinal stromal tumors: a meta-analysis of 1640 patients. J Clin Oncol. 2010;28(7):1247–1253. doi: 10.1200/JCO.2009.24.2099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dewaele B, Wasag B, Cools J, et al. Activity of dasatinib, a dual SRC/ABL kinase inhibitor, and IPI-504, a heat shock protein 90 inhibitor, against gastrointestinal stromal tumor-associated PDGFRAD842V mutation. Clin Cancer Res. 2008;14(18):5748–5758. doi: 10.1158/1078-0432.CCR-08-0533. [DOI] [PubMed] [Google Scholar]
- 89.Miselli FC, Casieri P, Negri T, et al. c-KIT/PDGFRA gene status alterations possibly related to primary imatinib resistance in gastrointestinal stromal tumors. Clin Cancer Res. 2007;13(8):2369–2377. doi: 10.1158/1078-0432.CCR-06-1745. [DOI] [PubMed] [Google Scholar]
- 90.Debiec-Rychter M, Cools J, Dumez H, et al. Mechanisms of resistance to imatinib mesylate in gastrointestinal stromal tumors and activity of the PKC412 inhibitor against imatinib-resistant mutants. Gastroenterology. 2005;128(2):270–279. doi: 10.1053/j.gastro.2004.11.020. [DOI] [PubMed] [Google Scholar]
- 91.Mahadevan D, Cooke L, Riley C, et al. A novel tyrosine kinase switch is a mechanism of imatinib resistance in gastrointestinal stromal tumors. Oncogene. 2007;26(27):3909–3919. doi: 10.1038/sj.onc.1210173. [DOI] [PubMed] [Google Scholar]
- 92.Théou N, Gil S, Devocelle A, et al. Multidrug resistance proteins in gastrointestinal stromal tumors: site-dependent expression and initial response to imatinib. Clin Cancer Res. 2005;11(21):7593–7598. doi: 10.1158/1078-0432.CCR-05-0710. [DOI] [PubMed] [Google Scholar]
- 93.Chen LL, Trent JC, Wu EF, et al. A missense mutation in KIT kinase domain 1 correlates with imatinib resistance in gastrointestinal stromal tumors. Cancer Res. 2004;64(17):5913–5919. doi: 10.1158/0008-5472.CAN-04-0085. [DOI] [PubMed] [Google Scholar]
- 94.Antonescu CR, Besmer P, Guo T, et al. Acquired resistance to imatinib in gastrointestinal stromal tumor occurs through secondary gene mutation. Clin Cancer Res. 2005;11(11):4182–4190. doi: 10.1158/1078-0432.CCR-04-2245. [DOI] [PubMed] [Google Scholar]
- 95.Heinrich MC, Corless CL, Blanke CD, et al. Molecular correlates of imatinib resistance in gastrointestinal stromal tumors. J Clin Oncol. 2006;24(29):4764–4774. doi: 10.1200/JCO.2006.06.2265. [DOI] [PubMed] [Google Scholar]
- 96.Gramza AW, Corless CL, Heinrich MC. Resistance to tyrosine kinase inhibitors in gastrointestinal stromal tumors. Clin Cancer Res. 2009;15(24):7510–7518. doi: 10.1158/1078-0432.CCR-09-0190. [DOI] [PubMed] [Google Scholar]
- 97.Desai J, Shankar S, Heinrich MC, et al. Clonal evolution of resistance to imatinib in patients with metastatic gastrointestinal stromal tumors. Clin Cancer Res. 2007;13(18 Pt 1):5398–5405. doi: 10.1158/1078-0432.CCR-06-0858. [DOI] [PubMed] [Google Scholar]
- 98.Liegl B, Kepten I, Le C, et al. Heterogeneity of kinase inhibitor resistance mechanisms in GIST. J Pathol. 2008;216(1):64–74. doi: 10.1002/path. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Negri T, Pavan GM, Virdis E, et al. T670X KIT mutations in gastrointestinal stromal tumors: making sense of missense. J Natl Cancer Inst. 2009;101(3):194–204. doi: 10.1093/jnci/djn477. [DOI] [PubMed] [Google Scholar]
- 100.Roberts KG, Odell AF, Byrnes EM, et al. Resistance to c-KIT kinase inhibitors conferred by V654A mutation. Mol Cancer Ther. 2007;6(3):1159–1166. doi: 10.1158/1535-7163.MCT-06-0641. [DOI] [PubMed] [Google Scholar]
- 101.Shankar S, vanSonnenberg E, Desai J, Dipiro PJ, van Den Abbeele A, Demetri GD. Gastrointestinal stromal tumor: new nodule-within-a-mass pattern of recurrence after partial response to imatinib mesylate. Radiology. 2005;235(3):892–898. doi: 10.1148/radiol.2353040332. [DOI] [PubMed] [Google Scholar]
- 102.Heinrich MC, Owzar K, Corlesss CL, et al. Correlation of kinase genotype and clinical outcome in the North American Intergroup Phase III trial of imatinib mesylate for treatment of advanced gastrointestinal stromal tumor: CALGB 150105 Study by Cancer and Leukemia Group B and Southwest Oncology Group. J Clin Oncol. 2008;26(33):5360–5367. doi: 10.1200/JCO.2008.17.4284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Zöller ME, Rembeck B, Oden A, Samuelsson M, Angervall L. Malignant and benign tumors in patients with neurofibromatosis type 1 in a defined Swedish population. Cancer. 1997;79(11):2125–2131. [PubMed] [Google Scholar]
- 104.Friedman JM. Epidemiology of neurofibromatosis type 1. Am J Med Genet. 1999;89(1):1–6. [PubMed] [Google Scholar]
- 105.Martin GA, Viskochil D, Bollag G, et al. The GAP-related domain of the neurofibromatosis type 1 gene product interacts with ras p 21. Cell. 1990;63(4):843–849. doi: 10.1016/0092-8674(90)90150-d. [DOI] [PubMed] [Google Scholar]
- 106.Li Y, Bollag G, Clark R, et al. Somatic mutations in the neurofibromatosis 1 gene in human tumors. Cell. 1992;69(2):275–281. doi: 10.1016/0092-8674(92)90408-5. [DOI] [PubMed] [Google Scholar]
- 107.Neurofibromatosis. Conference statement. National Institutes of Health Consensus Development Conference. Arch Neurol. 1988;45(5):575–578. [PubMed] [Google Scholar]
- 108.Lee MJ, Stephenson DA. Recent developments in neurofibromatosis type 1. Curr Opin Neurol. 2007;20(2):135–141. doi: 10.1097/WCO.0b013e3280895da8. [DOI] [PubMed] [Google Scholar]
- 109.Opocher G, Conton P, Schiavi F, Macino B, Mantero F. Pheochromocytoma in von Hippel-Lindau disease and neurofibromatosis type 1. Fam Cancer. 2005;4(1):13–16. doi: 10.1007/s10689-004-6128-y. [DOI] [PubMed] [Google Scholar]
- 110.Takazawa Y, Sakurai S, Sakuma Y, et al. Gastrointestinal stromal tumors of neurofibromatosis type I (von Recklinhausen’s disease) Am J Surg Pathol. 2005;29(6):755–763. doi: 10.1097/01.pas.0000163359.32734.f9. [DOI] [PubMed] [Google Scholar]
- 111.Andersson J, Sihto H, Meis-Kindblom JM, Joensuu H, Nupponen N, Kindblom LG. NF1-associated gastrointestinal stromal tumors have unique clinical, phenotypic, and genotypic characteristics. Am J Surg Pathol. 2005;29(9):1170–1176. doi: 10.1097/01.pas.0000159775.77912.15. [DOI] [PubMed] [Google Scholar]
- 112.Miettinen M, Fetsch JF, Sobin LH, Lasota J. Gastrointestinal stromal tumors in patients with neurofibromatosis 1: a clinicopathologic and molecular genetic study of 45 cases. Am J Surg Pathol. 2006;30(1):90–96. doi: 10.1097/01.pas.0000176433.81079.bd. [DOI] [PubMed] [Google Scholar]
- 113.Maertens O, Prenen H, Debiec-Rychter M, et al. Molecular pathogenesis of multiple gastrointestinal stromal tumors in NF1 patients. Hum Mol Genet. 2006;15(6):1015–1023. doi: 10.1093/hmg/ddl016. [DOI] [PubMed] [Google Scholar]
- 114.Yamamoto H, Tobo T, Nakamori M, et al. Neurofibromatosis type 1-related gastrointestinal stromal tumors: a special reference to loss of heterozygosity at 14q and 22q. J Cancer Res Clin Oncol. 2009;135(6):791–798. doi: 10.1007/s00432-008-0514-z. [DOI] [PubMed] [Google Scholar]
- 115.Stewart DR, Corless CL, Rubin BP, et al. Mitotic recombination as evidence of alternative pathogenesis of gastrointestinal stromal tumours in neurofibromatosis type 1. J Med Genet. 2007;44(1):e61. doi: 10.1136/jmg.2006.043075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Carney JA, Sheps SG, Go VL, Gordon H. The triad of gastric leiomyosarcoma, functioning extra-adrenal paraganglioma and pulmonary chondroma. N Engl J Med. 1977;296(26):1517–1518. doi: 10.1056/NEJM197706302962609. [DOI] [PubMed] [Google Scholar]
- 117.Horenstein MG, Hitchcock TA, Tucker JA. Dual CD117 expression in gastrointestinal stromal tumor (GIST) and paraganglioma of Carney triad: a case report. Int J Surg Pathol. 2005;13(1):87–92. doi: 10.1177/106689690501300113. [DOI] [PubMed] [Google Scholar]
- 118.Carney JA. Gastric stromal sarcoma, pulmonary chondroma, and extra-adrenal paraganglioma (Carney triad): natural history, adrenocortical component and possible familial occurrence. Mayo Clin Proc. 1999;74(6):543–552. doi: 10.4065/74.6.543. [DOI] [PubMed] [Google Scholar]
- 119.Zhang L, Smyrk TC, Young WF, Jr, Stratakis CA, Carney JA. Gastric stromal tumors in Carney triad are different clinically, pathologically, and behaviorally from sporadic gastric gastrointestinal stromal tumors: findings in 104 cases. Am J Surg Pathol. 2010;34(1):53–64. doi: 10.1097/PAS.0b013e3181c20f4f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Stratakis CA, Carney JA. The triad of paragangliomas, gastric stromal tumours and pulmonary chondromas (Carney triad), and the dyad of paragangliomas and gastric stromal sarcomas (Carney-Stratakis syndrome): molecular genetics and clinical implications. J Int Med. 2009;266(1):43–52. doi: 10.1111/j.1365-2796.2009.02110.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Carney JA, Stratakis CA. Familial paraganglioma and gastric stromal sarcoma: a new syndrome distinct from the Carney triad. Am J Med Genet. 2002;108(2):132–139. doi: 10.1002/ajmg.10235. [DOI] [PubMed] [Google Scholar]
- 122.Pasini B, McWhinney SR, Bei T, et al. Clinical and molecular genetics of patients with the Carney-Stratakis syndrome and germline mutations of the genes coding for the succinate dehydrogenase subunits SDHB, SDHC, and SDHD. Eur J Hum Genet. 2008;16(1):79–88. doi: 10.1038/sj.ejhg.5201904. [DOI] [PubMed] [Google Scholar]
- 123.Amar L, Bertherat J, Baudin E, et al. Genetic testing in pheochromocytoma or functional paraganglioma. J Clin Oncol. 2005;23(24):8812–8818. doi: 10.1200/JCO.2005.03.1484. [DOI] [PubMed] [Google Scholar]
- 124.Miettinen M, Lasota J, Sobin LH. Gastrointestinal stromal tumors of the stomach in children and young adults: a clinicopathologic, imunohistochemical, and molecular genetic study of 44 cases with long-term follow-up and review of the literature. Am J Surg Pathol. 2005;29(10):1373–1381. doi: 10.1097/01.pas.0000172190.79552.8b. [DOI] [PubMed] [Google Scholar]
- 125.Prakash S, Sarran L, Socci N, et al. Gastrointestinal stromal tumors in children and young adults: a clinicopathologic, molecular, and genomic study of 15 cases and review of the literature. J Pediatr Hematol Oncol. 2005;27(4):179–187. doi: 10.1097/01.mph.0000157790.81329.47. [DOI] [PubMed] [Google Scholar]
- 126.Agaram NP, Laquaglia MP, Ustun B, et al. Molecular characterization of pediatric gastrointestinal stromal tumors. Clin Cancer Res. 2008;14(10):3204–3215. doi: 10.1158/1078-0432.CCR-07-1984. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.van Nederveen FH, Gaal J, Favrier J, et al. An immunohistochemical procedure to detect patients with paraganglioma and phaeochromocytoma with germline SDHB, SDHC or SDHD gene mutations: a retrospective and prospective analysis. Lancet Oncol. 2009;10(8):764–771. doi: 10.1016/S1470-2045(09)70164-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Gill AJ, Benn DE, Chou A, et al. Immunohistochemistry for SDHB triages genetic testing of SDHB, SDHC and SDHD in paraganglioma-phaeochromocytoma syndromes. Hum Pathol. 2010;41(6):805–814. doi: 10.1016/j.humpath.2009.12.005. [DOI] [PubMed] [Google Scholar]
- 129.Gill AJ, Chou A, Vilain R, et al. Immunohistochemistry for SDHB divides gastrointestinal stromal tumors (GISTs) into 2 distinct types. Am J Surg Pathol. 2010;34(5):636–644. doi: 10.1097/PAS.0b013e3181d6150d. [DOI] [PubMed] [Google Scholar]
- 130.Janeway KA, Kim S, Lodish M, et al. Succinate dehydrogenase in KIT/PDGFRA wild-type gastrointestinal stromal tumors. J Clin Oncol. 2010;28(15S):A10008. [Google Scholar]
- 131.Nishida T, Hirota S, Taniguchi M, et al. Familial gastrointestinal stromal tumours with germline mutation of the KIT gene. Nat Genet. 1998;19(4):323–324. doi: 10.1038/1209. [DOI] [PubMed] [Google Scholar]
- 132.Wozniak A, Rutkowski P, Sciot R, Ruka W, Michej W, Debiec-Rychter M. Rectal gastrointestinal stromal tumors associated with a novel germline KIT mutation. Int J Cancer. 2008;122(9):2160–2164. doi: 10.1002/ijc.23338. [DOI] [PubMed] [Google Scholar]
- 133.Lasota J, Miettinen M. A new familial GIST identified. Am J Surg Pathol. 2006;30(10):1342. doi: 10.1097/01.pas.0000213364.56498.3b. [DOI] [PubMed] [Google Scholar]
- 134.Carballo M, Roig I, Aguilar F, et al. Novel c-KIT germline mutation in a family with gastrointestinal stromal tumors and cutaneous hyperpigmentation. Am J Med Genet A. 2005;132(4):361–364. doi: 10.1002/ajmg.a.30388. [DOI] [PubMed] [Google Scholar]
- 135.Li FP, Fletcher JA, Heinrich MC, et al. Familial gastrointestinal stromal tumor syndrome: phenotypic and molecular features in a kindred. J Clin Oncol. 2005;23(12):2735–2743. doi: 10.1200/JCO.2005.06.009. [DOI] [PubMed] [Google Scholar]
- 136.Thalheimer A, Schlemmer M, Bueter M, et al. Familial gastrointestinal stromal tumors caused by the novel KIT exon 17 germline mutation N822Y. Am J Surg Pathol. 2008;32(10):1560–1565. doi: 10.1097/PAS.0b013e318172ce6f. [DOI] [PubMed] [Google Scholar]
- 137.Kleinbaum EP, Lazar AJ, Tamborini E, et al. Clinical, histopathologic, molecular and therapeutic findings in a large kindred with gastrointestinal stromal tumor. Int J Cancer. 2008;122(3):711–718. doi: 10.1002/ijc.23137. [DOI] [PubMed] [Google Scholar]
- 138.Kang DY, Park CK, Choi JS, et al. Multiple gastrointestinal stromal tumors: clinicopathologic and genetic analysis of 12 patients. Am J Surg Pathol. 2007;31(2):224–232. doi: 10.1097/01.pas.0000213318.66800.94. [DOI] [PubMed] [Google Scholar]
- 139.Pasini B, Matyakhina L, Bei T, et al. Multiple gastrointestinal stromal and other tumors caused by platelet-derived growth factor receptor alpha gene mutations: a case associated with a germline V561D defect. J Clin Endocrinol Metab. 2007;92(9):3728–3732. doi: 10.1210/jc.2007-0894. [DOI] [PubMed] [Google Scholar]
- 140.Graham J, Debiec-Rychter M, Corless CL, Reid R, Davidson R, White R. Imatinib in the management of multiple gastrointestinal stromal tumors associated with a germline KIT K642E mutation. Arch Pathol Lab Med. 2007;131(9):1393–1396. doi: 10.5858/2007-131-1393-IITMOM. [DOI] [PubMed] [Google Scholar]
- 141.O’Riain C, Corless CL, Heinrich MC, et al. Gastrointestinal stromal tumors: insights from a new familial GIST kindred with unusual genetic and pathologic features. Am J Surg Pathol. 2005;29(12):1680–1683. doi: 10.1097/01.pas.0000173024.79852.08. [DOI] [PubMed] [Google Scholar]
- 142.Kim HJ, Lim SJ, Park K, Yuh YJ, Jang SJ, Choi J. Multiple gastrointestinal stromal tumors with a germline c-kit mutation. Pathol Int. 2005;55(10):655–659. doi: 10.1111/j.1440-1827.2005.01885.x. [DOI] [PubMed] [Google Scholar]
- 143.Chompret A, Kannengiesser C, Barrois M, et al. PDGFRA germline mutation in a family with multiple cases of gastrointestinal stromal tumor. Gastroenterology. 2004;126(1):318–321. doi: 10.1053/j.gastro.2003.10.079. [DOI] [PubMed] [Google Scholar]
- 144.Robson ME, Glogowski E, Sommer G, et al. Pleomorphic characteristics of a germ-line KIT mutation in a large kindred with gastrointestinal stromal tumors, hyperpigmentation, and dysphagia. Clin Cancer Res. 2004;10(4):1250–1254. doi: 10.1158/1078-0432.ccr-03-0110. [DOI] [PubMed] [Google Scholar]
- 145.Tarn C, Merkel E, Canutescu AA, et al. Analysis of KIT mutations in sporadic and familial gastrointestinal stromal tumors: therapeutic implications through protein modeling. Clin Cancer Res. 2005;11(10):3668–3677. doi: 10.1158/1078-0432.CCR-04-2515. [DOI] [PubMed] [Google Scholar]
- 146.Hirota S, Nishida T, Isozaki K, et al. Familial gastrointestinal stromal tumors associated with dysphagia and novel type germline mutation of KIT gene. Gastroenterology. 2002;122(5):1493–1499. doi: 10.1053/gast.2002.33024. [DOI] [PubMed] [Google Scholar]
- 147.Maeyama H, Hidaka E, Ota H, et al. Familial gastrointestinal stromal tumor with hyperpigmentation: association with a germline mutation of the c-kit gene. Gastroenterology. 2001;120(1):210–215. doi: 10.1053/gast.2001.20880. [DOI] [PubMed] [Google Scholar]
- 148.Beghini A, Tibiletti MG, Roversi G, et al. Germline mutation in the juxtamembrane domain of the kit gene in a family with gastrointestinal stromal tumors and urticaria pigmentosa. Cancer. 2001;92(3):657–662. doi: 10.1002/1097-0142(20010801)92:3<657::aid-cncr1367>3.0.co;2-d. [DOI] [PubMed] [Google Scholar]
- 149.Isozaki K, Terris B, Belghiti J, Schiffmann S, Hirota S, Vanderwinden JM. Germline-activating mutation in the kinase domain of KIT gene in familial gastrointestinal stromal tumors. Am J Pathol. 2000;157(5):1581–1585. doi: 10.1016/S0002-9440(10)64795-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Veiga I, Silva M, Vieira J, et al. Hereditary gastrointestinal stromal tumors sharing the KIT exon 17 germline mutation p. Asp820Tyr develop through different cytogenetic pathways. Genes Chromosomes Cancer. 2010;49(2):91–98. doi: 10.1002/gcc.20720. [DOI] [PubMed] [Google Scholar]
- 151.Campbell T, Felsten L, Moore J. Disappearance of lentigines in a patient receiving imatinib treatment for familial gastrointestinal stromal tumor syndrome. Arch Dermatol. 2009;145(11):1313–1316. doi: 10.1001/archdermatol.2009.263. [DOI] [PubMed] [Google Scholar]
- 152.Chen H, Hirota S, Isozaki K, et al. Polyclonal nature of diffuse proliferation of interstitial cells of Cajal in patients with familial and multiple gastrointestinal stromal tumours. Gut. 2002;51(6):793–796. doi: 10.1136/gut.51.6.793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Chen Y, Tzeng CC, Liou CP, Chang MY, Li CF, Lin CN. Biological significance of chromosomal imbalance aberrations in gastrointestinal stromal tumors. J Biomed Sci. 2004;11(1):65–71. doi: 10.1007/BF02256550. [DOI] [PubMed] [Google Scholar]
- 154.Gunawan B, von Heydebreck A, Sander B, et al. An oncogenetic tree model in gastrointestinal stromal tumours (GISTs) identifies different pathways of cytogenetic evolution with prognostic implications. J Pathol. 2007;211(4):463–470. doi: 10.1002/path.2128. [DOI] [PubMed] [Google Scholar]
- 155.Sommer G, Agosti V, Ehlers I, et al. Gastrointestinal stromal tumors in a mouse by targeted mutation of the Kit receptor tyrosine kinase. Proc Natl Acad Sci U S A. 2003;100(11):6706–6711. doi: 10.1073/pnas.1037763100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Rubin BP, Antonescu CR, Scott-Browne JP, et al. A knock-in mouse model of gastrointestinal tumor harboring KIT K641E. Cancer Res. 2005;65(15):6631–6639. doi: 10.1158/0008-5472.CAN-05-0891. [DOI] [PubMed] [Google Scholar]
- 157.Nakai N, Ishikawa T, Nishitani A, et al. A mouse model of a human multiple GIST family with KIT-Asp820Tyr mutation generated by a knock-in strategy. J Pathol. 2008;214(3):302–311. doi: 10.1002/path.2296. [DOI] [PubMed] [Google Scholar]
- 158.Ishikawa T, Nakai N, Liu NN, et al. In vivo effect of imatinib on progression of cecal GIST-like tumors in exon 17-type c-kit kock-in mice. Lab Invest. 2009;89(10):1161–1168. doi: 10.1038/labinvest.2009.78. [DOI] [PubMed] [Google Scholar]
- 159.Rossi F, Ehlers I, Agosti V, et al. Oncogenic Kit signaling and therapeutic intervention in a mouse model of gastrointestinal stromal tumor. Proc Natl Acad Sci U S A. 2006;103(34):12843–12848. doi: 10.1073/pnas.0511076103. [DOI] [PMC free article] [PubMed] [Google Scholar]