Abstract
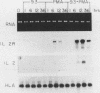
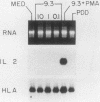
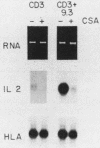
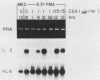
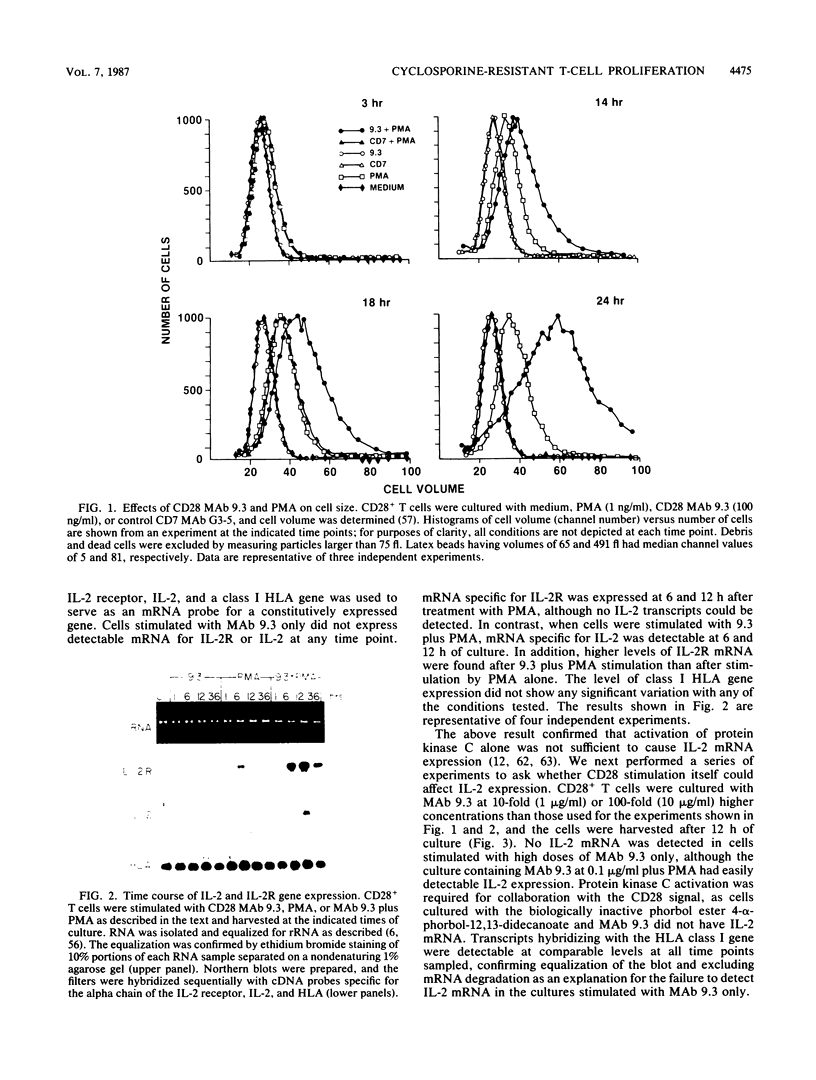
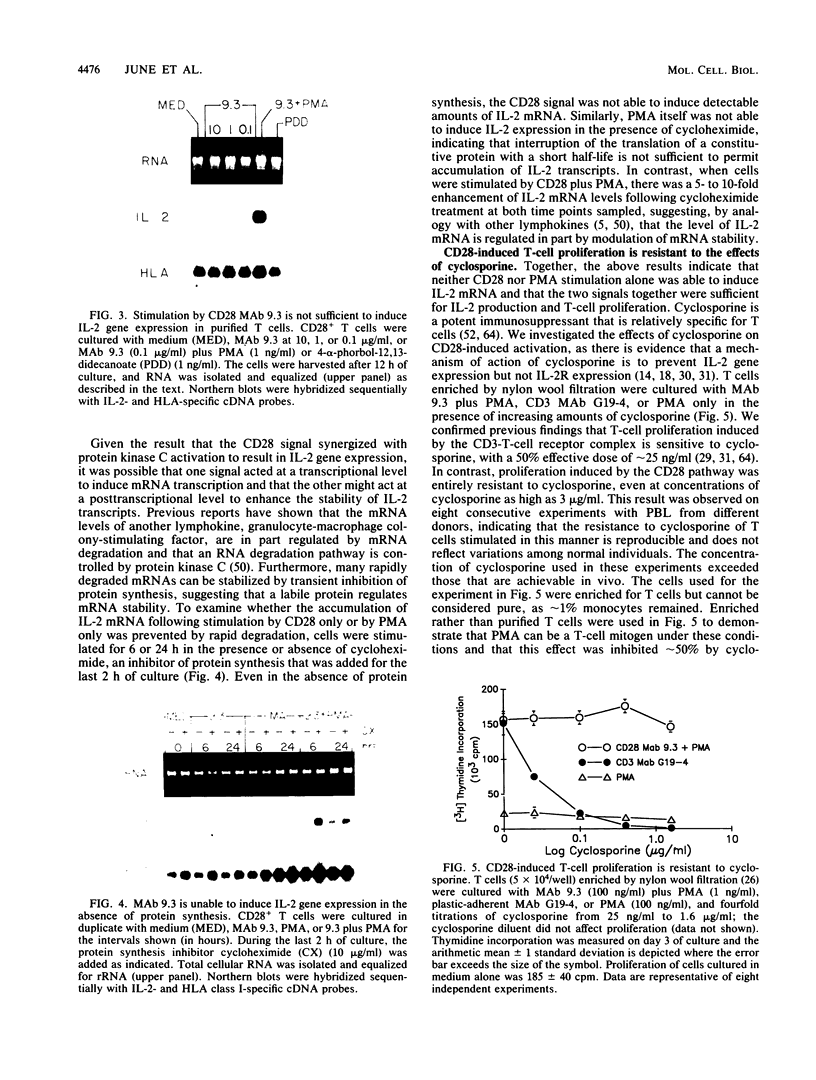
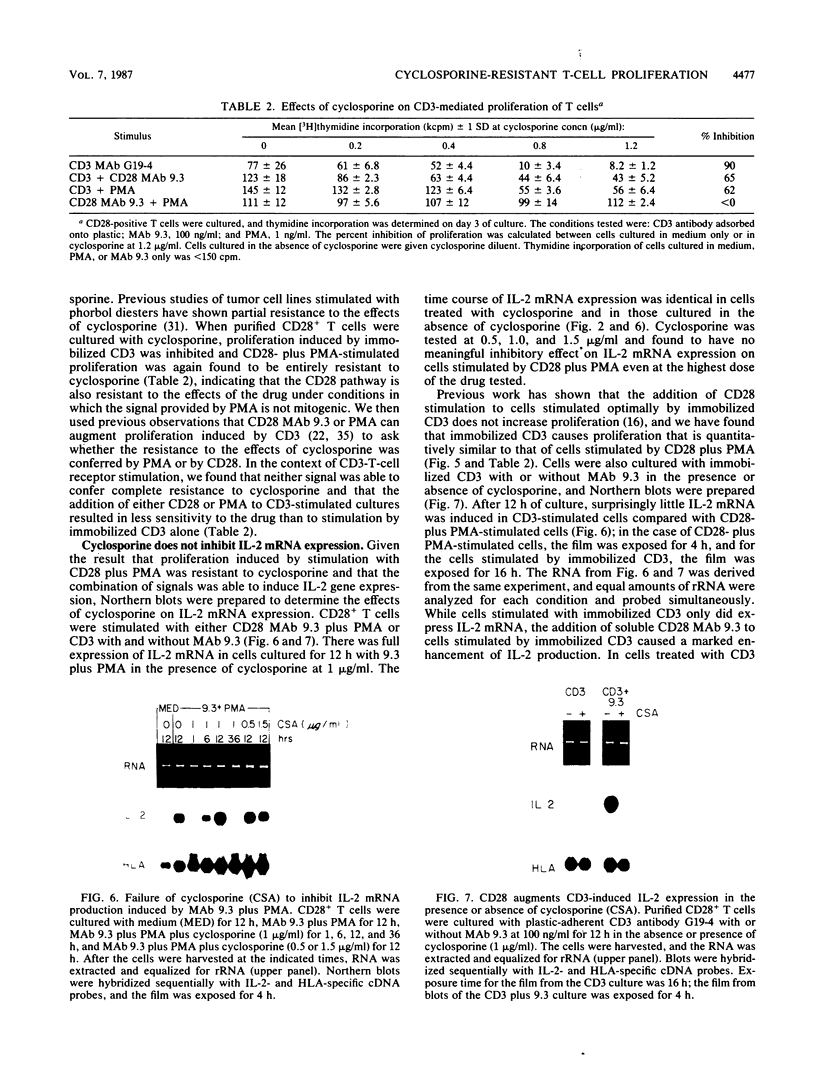
CD28 is a homodimeric glycoprotein expressed on the surface of a major subset of human T cells that has recently been identified as a member of the immunoglobulin supergene family. The binding of monoclonal antibodies to the CD28 antigen on purified T cells does not result in proliferation; however, previous studies have shown that the combination of CD28 stimulation and protein kinase C activation by phorbol myristate acetate (PMA) results in T-cell proliferation that is independent of both accessory cells and activation of the T-cell receptor-CD3 complex. In the present study, effects of stimulation by anti-CD28 on cell cycle progression and on the interleukin 2 (IL-2) and IL-2 receptor system have been investigated on primary cultures of purified peripheral-blood CD28+ T cells. There was no measurable effect on cell size or on DNA synthesis after stimulation of resting (G0) cells by CD28 alone. After 3 h of activation of T cells by PMA alone, a slight (8%) increase in cell volume occurred that did not progress to DNA synthesis. In contrast, T-cell stimulation by CD28 in combination with PMA resulted in a progressive increase in cell volume in approximately 100% of cells at 12 to 14 h after stimulation. Northern blot (RNA blot) analysis revealed that CD28 stimulation alone failed to cause expression of the alpha chain of the IL-2 receptor or of IL-2 mRNA, and in accord with previous studies, stimulation by PMA alone resulted in the accumulation of IL-2 receptor transcripts but no detectable IL-2 mRNA. In contrast, T-cell stimulation by the combination of CD28 and PMA resulted in the appearance of IL-2 transcripts and enhanced expression of IL-2 receptor mRNA. Functional studies revealed that the proliferation induced by CD28 and PMA stimulation was entirely resistant to cyclosporine, in contrast to T-cell activation induced by the CD3-T-cell receptor complex. Cyclosporine was found not to affect the accumulation of IL-2 mRNA after CD28 plus PMA stimulation, although there was no detectable IL-2 mRNA after stimulation by CD3 in the presence of the drug. Furthermore, stimulation by CD28 in combination with immobilized CD3 antibodies caused a striking enhancement of IL-2 mRNA expression that was, in part, resistant to the effects of cyclosporine. These studies indicate that the CD28 molecule synergizes with protein kinase C activation to induce IL-2 gene expression and demonstrate that stimulation by the CD28 pathway can cause vigorous T-cell proliferation even in the presence of cyclosporine and that cyclosporine does not prevent transcription of 16-2 mRNA, as has been suggested previously. Moreover, these findings suggest that a potential role for the CD28 molecule in vivo may be to augment IL-2 production after stimulation of the CD3-T-cell receptor molecular complex and thereby to amplify an antigen-specific immune response. Finally, these results provide further evidence that the CD28 molecule triggers T-cell proliferation in a manner that differs biochemically from CD3-T-cell receptor-induced proliferation.
Full text
PDF






Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Birchenall-Sparks M. C., Farrar W. L., Rennick D., Kilian P. L., Ruscetti F. W. Regulation of expression of the interleukin-2 receptor on hematopoietic cells by interleukin-3. Science. 1986 Jul 25;233(4762):455–458. doi: 10.1126/science.3088729. [DOI] [PubMed] [Google Scholar]
- Blackman M. A., Tigges M. A., Minie M. E., Koshland M. E. A model system for peptide hormone action in differentiation: interleukin 2 induces a B lymphoma to transcribe the J chain gene. Cell. 1986 Nov 21;47(4):609–617. doi: 10.1016/0092-8674(86)90625-2. [DOI] [PubMed] [Google Scholar]
- Cantrell D. A., Smith K. A. The interleukin-2 T-cell system: a new cell growth model. Science. 1984 Jun 22;224(4655):1312–1316. doi: 10.1126/science.6427923. [DOI] [PubMed] [Google Scholar]
- Caput D., Beutler B., Hartog K., Thayer R., Brown-Shimer S., Cerami A. Identification of a common nucleotide sequence in the 3'-untranslated region of mRNA molecules specifying inflammatory mediators. Proc Natl Acad Sci U S A. 1986 Mar;83(6):1670–1674. doi: 10.1073/pnas.83.6.1670. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Chouaib S., Welte K., Mertelsmann R., Dupont B. Prostaglandin E2 acts at two distinct pathways of T lymphocyte activation: inhibition of interleukin 2 production and down-regulation of transferrin receptor expression. J Immunol. 1985 Aug;135(2):1172–1179. [PubMed] [Google Scholar]
- Clark S. C., Arya S. K., Wong-Staal F., Matsumoto-Kobayashi M., Kay R. M., Kaufman R. J., Brown E. L., Shoemaker C., Copeland T., Oroszlan S. Human T-cell growth factor: partial amino acid sequence, cDNA cloning, and organization and expression in normal and leukemic cells. Proc Natl Acad Sci U S A. 1984 Apr;81(8):2543–2547. doi: 10.1073/pnas.81.8.2543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Damle N. K., Mohagheghpour N., Hansen J. A., Engleman E. G. Alloantigen-specific cytotoxic and suppressor T lymphocytes are derived from phenotypically distinct precursors. J Immunol. 1983 Nov;131(5):2296–2300. [PubMed] [Google Scholar]
- Davis L., Lipsky P. E. Signals involved in T cell activation. II. Distinct roles of intact accessory cells, phorbol esters, and interleukin 1 in activation and cell cycle progression of resting T lymphocytes. J Immunol. 1986 May 15;136(10):3588–3596. [PubMed] [Google Scholar]
- DeFranco A. L., Kung J. T., Paul W. E. Regulation of growth and proliferation in B cell subpopulations. Immunol Rev. 1982;64:161–182. doi: 10.1111/j.1600-065x.1982.tb00423.x. [DOI] [PubMed] [Google Scholar]
- Depper J. M., Leonard W. J., Krönke M., Noguchi P. D., Cunningham R. E., Waldmann T. A., Greene W. C. Regulation of interleukin 2 receptor expression: effects of phorbol diester, phospholipase C, and reexposure to lectin or antigen. J Immunol. 1984 Dec;133(6):3054–3061. [PubMed] [Google Scholar]
- Efrat S., Kaempfer R. Control of biologically active interleukin 2 messenger RNA formation in induced human lymphocytes. Proc Natl Acad Sci U S A. 1984 May;81(9):2601–2605. doi: 10.1073/pnas.81.9.2601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elliott J. F., Lin Y., Mizel S. B., Bleackley R. C., Harnish D. G., Paetkau V. Induction of interleukin 2 messenger RNA inhibited by cyclosporin A. Science. 1984 Dec 21;226(4681):1439–1441. doi: 10.1126/science.6334364. [DOI] [PubMed] [Google Scholar]
- Farrar W. L., Ruscetti F. W. Association of protein kinase C activation with IL 2 receptor expression. J Immunol. 1986 Feb 15;136(4):1266–1273. [PubMed] [Google Scholar]
- Geppert T. D., Lipsky P. E. Accessory cell independent proliferation of human T4 cells stimulated by immobilized monoclonal antibodies to CD3. J Immunol. 1987 Mar 15;138(6):1660–1666. [PubMed] [Google Scholar]
- Gmünder H., Lesslauer W. A 45-kDa human T-cell membrane glycoprotein functions in the regulation of cell proliferative responses. Eur J Biochem. 1984 Jul 2;142(1):153–160. doi: 10.1111/j.1432-1033.1984.tb08263.x. [DOI] [PubMed] [Google Scholar]
- Granelli-Piperno A., Andrus L., Steinman R. M. Lymphokine and nonlymphokine mRNA levels in stimulated human T cells. Kinetics, mitogen requirements, and effects of cyclosporin A. J Exp Med. 1986 Apr 1;163(4):922–937. doi: 10.1084/jem.163.4.922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grinstein S., Cohen S., Goetz J. D., Rothstein A., Gelfand E. W. Characterization of the activation of Na+/H+ exchange in lymphocytes by phorbol esters: change in cytoplasmic pH dependence of the antiport. Proc Natl Acad Sci U S A. 1985 Mar;82(5):1429–1433. doi: 10.1073/pnas.82.5.1429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hadden J. W., Hadden E. M., Haddox M. K., Goldberg N. D. Guanosine 3':5'-cyclic monophosphate: a possible intracellular mediator of mitogenic influences in lymphocytes. Proc Natl Acad Sci U S A. 1972 Oct;69(10):3024–3027. doi: 10.1073/pnas.69.10.3024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara T., Fu S. M., Hansen J. A. Human T cell activation. II. A new activation pathway used by a major T cell population via a disulfide-bonded dimer of a 44 kilodalton polypeptide (9.3 antigen). J Exp Med. 1985 Jun 1;161(6):1513–1524. doi: 10.1084/jem.161.6.1513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hara T., Fu S. M. Human T cell activation. I. Monocyte-independent activation and proliferation induced by anti-T3 monoclonal antibodies in the presence of tumor promoter 12-o-tetradecanoyl phorbol-13 acetate. J Exp Med. 1985 Apr 1;161(4):641–656. doi: 10.1084/jem.161.4.641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Herrmann F., Cannistra S. A., Levine H., Griffin J. D. Expression of interleukin 2 receptors and binding of interleukin 2 by gamma interferon-induced human leukemic and normal monocytic cells. J Exp Med. 1985 Sep 1;162(3):1111–1116. doi: 10.1084/jem.162.3.1111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Imboden J., Weyand C., Goronzy J. Antigen recognition by a human T cell clone leads to increases in inositol trisphosphate. J Immunol. 1987 Mar 1;138(5):1322–1324. [PubMed] [Google Scholar]
- Julius M. H., Simpson E., Herzenberg L. A. A rapid method for the isolation of functional thymus-derived murine lymphocytes. Eur J Immunol. 1973 Oct;3(10):645–649. doi: 10.1002/eji.1830031011. [DOI] [PubMed] [Google Scholar]
- June C. H., Ledbetter J. A., Rabinovitch P. S., Martin P. J., Beatty P. G., Hansen J. A. Distinct patterns of transmembrane calcium flux and intracellular calcium mobilization after differentiation antigen cluster 2 (E rosette receptor) or 3 (T3) stimulation of human lymphocytes. J Clin Invest. 1986 Apr;77(4):1224–1232. doi: 10.1172/JCI112425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katzen D., Chu E., Terhost C., Leung D. Y., Gesner M., Miller R. A., Geha R. S. Mechanisms of human T cell response to mitogens: IL 2 induces IL 2 receptor expression and proliferation but not IL 2 synthesis in PHA-stimulated T cells. J Immunol. 1985 Sep;135(3):1840–1845. [PubMed] [Google Scholar]
- Kay J. E., Benzie C. R. Lymphocyte activation by OKT3: cyclosporine sensitivity and synergism with phorbol ester. Immunology. 1986 Feb;57(2):195–199. [PMC free article] [PubMed] [Google Scholar]
- Krönke M., Leonard W. J., Depper J. M., Arya S. K., Wong-Staal F., Gallo R. C., Waldmann T. A., Greene W. C. Cyclosporin A inhibits T-cell growth factor gene expression at the level of mRNA transcription. Proc Natl Acad Sci U S A. 1984 Aug;81(16):5214–5218. doi: 10.1073/pnas.81.16.5214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Landegren U., Wigzell H. Cyclosporin A inhibits a discrete step in T-lymphocyte stimulation. Scand J Immunol. 1985 Sep;22(3):279–284. doi: 10.1111/j.1365-3083.1985.tb01882.x. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Gentry L. E., June C. H., Rabinovitch P. S., Purchio A. F. Stimulation of T cells through the CD3/T-cell receptor complex: role of cytoplasmic calcium, protein kinase C translocation, and phosphorylation of pp60c-src in the activation pathway. Mol Cell Biol. 1987 Feb;7(2):650–656. doi: 10.1128/mcb.7.2.650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledbetter J. A., June C. H., Grosmaire L. S., Rabinovitch P. S. Crosslinking of surface antigens causes mobilization of intracellular ionized calcium in T lymphocytes. Proc Natl Acad Sci U S A. 1987 Mar;84(5):1384–1388. doi: 10.1073/pnas.84.5.1384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ledbetter J. A., June C. H., Martin P. J., Spooner C. E., Hansen J. A., Meier K. E. Valency of CD3 binding and internalization of the CD3 cell-surface complex control T cell responses to second signals: distinction between effects on protein kinase C, cytoplasmic free calcium, and proliferation. J Immunol. 1986 Jun 1;136(11):3945–3952. [PubMed] [Google Scholar]
- Ledbetter J. A., Martin P. J., Spooner C. E., Wofsy D., Tsu T. T., Beatty P. G., Gladstone P. Antibodies to Tp67 and Tp44 augment and sustain proliferative responses of activated T cells. J Immunol. 1985 Oct;135(4):2331–2336. [PubMed] [Google Scholar]
- Ledbetter J. A., Parsons M., Martin P. J., Hansen J. A., Rabinovitch P. S., June C. H. Antibody binding to CD5 (Tp67) and Tp44 T cell surface molecules: effects on cyclic nucleotides, cytoplasmic free calcium, and cAMP-mediated suppression. J Immunol. 1986 Nov 15;137(10):3299–3305. [PubMed] [Google Scholar]
- Leonard W. J., Depper J. M., Crabtree G. R., Rudikoff S., Pumphrey J., Robb R. J., Krönke M., Svetlik P. B., Peffer N. J., Waldmann T. A. Molecular cloning and expression of cDNAs for the human interleukin-2 receptor. Nature. 1984 Oct 18;311(5987):626–631. doi: 10.1038/311626a0. [DOI] [PubMed] [Google Scholar]
- Lum L. G., Orcutt-Thordarson N., Seigneuret M. C., Hansen J. A. In vitro regulation of immunoglobulin synthesis by T-cell subpopulations defined by a new human T-cell antigen (9.3). Cell Immunol. 1982 Sep 1;72(1):122–129. doi: 10.1016/0008-8749(82)90289-1. [DOI] [PubMed] [Google Scholar]
- Martin P. J., Ledbetter J. A., Morishita Y., June C. H., Beatty P. G., Hansen J. A. A 44 kilodalton cell surface homodimer regulates interleukin 2 production by activated human T lymphocytes. J Immunol. 1986 May 1;136(9):3282–3287. [PubMed] [Google Scholar]
- Meuer S. C., Hussey R. E., Fabbi M., Fox D., Acuto O., Fitzgerald K. A., Hodgdon J. C., Protentis J. P., Schlossman S. F., Reinherz E. L. An alternative pathway of T-cell activation: a functional role for the 50 kd T11 sheep erythrocyte receptor protein. Cell. 1984 Apr;36(4):897–906. doi: 10.1016/0092-8674(84)90039-4. [DOI] [PubMed] [Google Scholar]
- Monroe J. G., Havran W. L., Cambier J. C. Enrichment of viable lymphocytes in defined cycle phases by sorting on the basis of pulse width of axial light extinction. Cytometry. 1982 Jul;3(1):24–27. doi: 10.1002/cyto.990030107. [DOI] [PubMed] [Google Scholar]
- Moretta A., Pantaleo G., Lopez-Botet M., Moretta L. Involvement of T44 molecules in an antigen-independent pathway of T cell activation. Analysis of the correlations to the T cell antigen-receptor complex. J Exp Med. 1985 Sep 1;162(3):823–838. doi: 10.1084/jem.162.3.823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Novogrodsky A., Patya M., Rubin A. L., Stenzel K. H. Agents that increase cellular cAMP inhibit production of interleukin-2, but not its activity. Biochem Biophys Res Commun. 1983 Jul 18;114(1):93–98. doi: 10.1016/0006-291x(83)91598-x. [DOI] [PubMed] [Google Scholar]
- Pantaleo G., Olive D., Poggi A., Kozumbo W. J., Moretta L., Moretta A. Transmembrane signalling via the T11-dependent pathway of human T cell activation. Evidence for the involvement of 1,2-diacylglycerol and inositol phosphates. Eur J Immunol. 1987 Jan;17(1):55–60. doi: 10.1002/eji.1830170110. [DOI] [PubMed] [Google Scholar]
- Parker P. J., Coussens L., Totty N., Rhee L., Young S., Chen E., Stabel S., Waterfield M. D., Ullrich A. The complete primary structure of protein kinase C--the major phorbol ester receptor. Science. 1986 Aug 22;233(4766):853–859. doi: 10.1126/science.3755547. [DOI] [PubMed] [Google Scholar]
- Reed J. C., Sabath D. E., Hoover R. G., Prystowsky M. B. Recombinant interleukin 2 regulates levels of c-myc mRNA in a cloned murine T lymphocyte. Mol Cell Biol. 1985 Dec;5(12):3361–3368. doi: 10.1128/mcb.5.12.3361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Segel G. B., Cokelet G. R., Lichtman M. A. The measurement of lymphocyte volume: importance of reference particle deformability and counting solution tonicity. Blood. 1981 May;57(5):894–899. [PubMed] [Google Scholar]
- Selvaraj P., Plunkett M. L., Dustin M., Sanders M. E., Shaw S., Springer T. A. The T lymphocyte glycoprotein CD2 binds the cell surface ligand LFA-3. 1987 Mar 26-Apr 1Nature. 326(6111):400–403. doi: 10.1038/326400a0. [DOI] [PubMed] [Google Scholar]
- Shapiro D. N., Adams B. S., Niederhuber J. E. Antigen-specific T cell activation results in an increase in cytoplasmic free calcium. J Immunol. 1985 Oct;135(4):2256–2261. [PubMed] [Google Scholar]
- Shaw G., Kamen R. A conserved AU sequence from the 3' untranslated region of GM-CSF mRNA mediates selective mRNA degradation. Cell. 1986 Aug 29;46(5):659–667. doi: 10.1016/0092-8674(86)90341-7. [DOI] [PubMed] [Google Scholar]
- Shevach E. M. The effects of cyclosporin A on the immune system. Annu Rev Immunol. 1985;3:397–423. doi: 10.1146/annurev.iy.03.040185.002145. [DOI] [PubMed] [Google Scholar]
- Sood A. K., Pereira D., Weissman S. M. Isolation and partial nucleotide sequence of a cDNA clone for human histocompatibility antigen HLA-B by use of an oligodeoxynucleotide primer. Proc Natl Acad Sci U S A. 1981 Jan;78(1):616–620. doi: 10.1073/pnas.78.1.616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Storb R., Deeg H. J., Farewell V., Doney K., Appelbaum F., Beatty P., Bensinger W., Buckner C. D., Clift R., Hansen J. Marrow transplantation for severe aplastic anemia: methotrexate alone compared with a combination of methotrexate and cyclosporine for prevention of acute graft-versus-host disease. Blood. 1986 Jul;68(1):119–125. [PubMed] [Google Scholar]
- Storb R., Thomas E. D. Graft-versus-host disease in dog and man: the Seattle experience. Immunol Rev. 1985 Dec;88:215–238. doi: 10.1111/j.1600-065x.1985.tb01160.x. [DOI] [PubMed] [Google Scholar]
- Thompson C. B., Challoner P. B., Neiman P. E., Groudine M. Expression of the c-myb proto-oncogene during cellular proliferation. 1986 Jan 30-Feb 5Nature. 319(6052):374–380. doi: 10.1038/319374a0. [DOI] [PubMed] [Google Scholar]
- Thompson C. B., Scher I., Schaefer M. E., Lindsten T., Finkelman F. D., Mond J. J. Size-dependent B lymphocyte subpopulations: relationship of cell volume to surface phenotype, cell cycle, proliferative response, and requirements for antibody production to TNP-Ficoll and TNP-BA. J Immunol. 1984 Nov;133(5):2333–2342. [PubMed] [Google Scholar]
- Touraine J. L., Hadden J. W., Touraine F., Hadden E. M., Estensen R., Good R. A. Phorbol myristate acetate: a mitogen selective for a T-lymphocyte subpopulation. J Exp Med. 1977 Feb 1;145(2):460–465. doi: 10.1084/jem.145.2.460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wedner H. J., Dankner R., Parker C. W. Cyclic GMP and lectin-induced lymphocyte activation. J Immunol. 1975 Dec;115(6):1682–1687. [PubMed] [Google Scholar]
- Weiss A., Imboden J., Shoback D., Stobo J. Role of T3 surface molecules in human T-cell activation: T3-dependent activation results in an increase in cytoplasmic free calcium. Proc Natl Acad Sci U S A. 1984 Jul;81(13):4169–4173. doi: 10.1073/pnas.81.13.4169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss A., Manger B., Imboden J. Synergy between the T3/antigen receptor complex and Tp44 in the activation of human T cells. J Immunol. 1986 Aug 1;137(3):819–825. [PubMed] [Google Scholar]
- Weiss A., Shields R., Newton M., Manger B., Imboden J. Ligand-receptor interactions required for commitment to the activation of the interleukin 2 gene. J Immunol. 1987 Apr 1;138(7):2169–2176. [PubMed] [Google Scholar]
- Weiss A., Wiskocil R. L., Stobo J. D. The role of T3 surface molecules in the activation of human T cells: a two-stimulus requirement for IL 2 production reflects events occurring at a pre-translational level. J Immunol. 1984 Jul;133(1):123–128. [PubMed] [Google Scholar]
- Wiesinger D., Borel J. F. Studies on the mechanism of action of cyclosporin A. Immunobiology. 1980 Jan;156(4-5):454–463. doi: 10.1016/S0171-2985(80)80078-7. [DOI] [PubMed] [Google Scholar]
- Williams J. M., Deloria D., Hansen J. A., Dinarello C. A., Loertscher R., Shapiro H. M., Strom T. B. The events of primary T cell activation can be staged by use of Sepharose-bound anti-T3 (64.1) monoclonal antibody and purified interleukin 1. J Immunol. 1985 Oct;135(4):2249–2255. [PubMed] [Google Scholar]
- Williams J. M., Ransil B. J., Shapiro H. M., Strom T. B. Accessory cell requirement for activation antigen expression and cell cycle progression by human T lymphocytes. J Immunol. 1984 Dec;133(6):2986–2995. [PubMed] [Google Scholar]
- Wiskocil R., Weiss A., Imboden J., Kamin-Lewis R., Stobo J. Activation of a human T cell line: a two-stimulus requirement in the pretranslational events involved in the coordinate expression of interleukin 2 and gamma-interferon genes. J Immunol. 1985 Mar;134(3):1599–1603. [PubMed] [Google Scholar]
- Yamada H., Martin P. J., Bean M. A., Braun M. P., Beatty P. G., Sadamoto K., Hansen J. A. Monoclonal antibody 9.3 and anti-CD11 antibodies define reciprocal subsets of lymphocytes. Eur J Immunol. 1985 Dec;15(12):1164–1168. doi: 10.1002/eji.1830151204. [DOI] [PubMed] [Google Scholar]