Abstract
Neutrophils have long been known to participate in acute inflammation, but a role in chronic inflammatory and autoimmune diseases is now emerging. These cells are key players in the recognition and elimination of pathogens, but they also sense self components, including nucleic acids and products of sterile tissue damage. While this normally contributes to tissue repair, it can also lead to the release of highly immunogenic products that can trigger and/or amplify autoimmune pathogenic loops. Understanding the mechanisms that underlie neutrophil activation, migration, survival and their various forms of death in health and disease might provide us with new approaches to treat chronic inflammatory conditions.
Introduction
Neutrophils are key players in the recognition and elimination of pathogens, but they also sense sterile tissue damage and contribute to its repair. These cells circulate through the vascular compartment in a resting state while equipped with a dangerous array of microbicidal weapons. To prevent inappropriate migration and accumulation in tissues, neutrophils need to be `primed' by cytokines, chemoattractants and growth factors or interact with activated endothelial cells before leaving the blood [reviewed in [1•,2••]]. Regardless of the trigger that prompts their recruitment to tissues, neutrophils end up dying in action. A sophisticated set of events follows to ensure that neutrophil burial ultimately leads to resolution of inflammation. Massive numbers of resting neutrophils also die quietly every day to maintain homeostasis. Our review will cover recent insights into how non-infectious types of neutrophil activation and death might contribute to the pathogenesis of chronic human autoimmune and inflammatory diseases.
Neutrophil life and death
Humans produce large numbers of neutrophils, up to 1011, every day in the bone marrow. Fully differentiated neutrophils are subsequently released into the circulation, where they represent 60–70% of the white blood cell pool. The number of neutrophils is kept constant, which implies that every day up to 1011 neutrophils die. Neutrophils die spontaneously in the steady state through apoptosis, which leads to the proper disposal of their potentially dangerous cargo. The signals that prompt the initiation of this process are poorly characterized but seem to be independent of classic death receptor pathways. In mice, upregulation of the chemokine receptor CXCR4 by senescent neutrophils dictates their homing to the bone marrow, where apoptosis and ensuing phagocytosis by stromal macrophages takes place [1•]. Ex vivo, senescent neutrophils accumulate reactive oxygen species (ROS), which inhibits the ROS-PI3P-Akt axis and marks the initiation of programmed cell death [3].
In addition to senescence-dependent apoptosis, neutrophils undergo apoptosis in response to death receptor ligation, exposure to TNFα and post-phagocytosis [4]. Neutrophils also die through necrosis and pyroptosis, which prompt the release of danger signals and cytokines. Programmed necrosis, a form of death characterized by cytoplasmic vacuolization, ROS production and autophagosome/secondary granule fusion, also affects neutrophils. This process is caspase-independent and requires PI3K and RIP1 kinase activation [5]. Although the contribution of programmed neutrophil necrosis to inflammatory diseases has not been explored, vacuolized neutrophils can be found in vivo in patients with infections as well as autoimmune diseases, such as rheumatoid arthritis, vasculitis and psoriasis [6].
There are, in addition, granulocyte-specific forms of death that might be beneficial to control infections but could lead to chronic inflammation. Thus, upon in vitro activation with PMA, microbes or cytokines, neutrophils undergo NETosis. This process is characterized by chromatin depolymerization, breakdown of nuclear and granular membranes, subsequent mixing of nuclear and cytoplasmic granular cargo and extrusion of this material as fibrous structures called neutrophil extracellular traps (NETs) [7•]. NETs kill extracellular microorganisms in vitro and might represent a powerful way to control their proliferation in vivo. NET release is dependent on ROS production. Accordingly, neutrophils from patients with chronic granulomatous disease (CGD), who carry mutations in NADPH oxidase complex-encoding genes, cannot release NETs [8]. Upstream of NADPH oxidase, the Raf–MEK–ERK pathway as well as the translocation of two enzymes, neutrophil elastase (NE) and myeloperoxidase (MPO), from cytoplasmic granules into the nucleus contribute to NET formation [9]. NE induces histone degradation, which initiates chromatin decondensation [10]. Histone citrullination and autophagy may also play a role in NET formation [11,12].
NETosis is not always synonymous of death. Thus, neutrophils exposed in vivo to gram-positive bacteria extrude chromatin-DNA through an exocytosis-related process that spares the integrity of the plasma membrane and is oxidant-independent. Interestingly, eventually enucleated neutrophils retain for some time their migratory and phagocytic capacity [13,14]. In addition to chromatin DNA, living granulocytes can expel mitochondria as a source of extracellular DNA [15,16]. As reviewed below, these processes might be associated to a number of human diseases.
Certain inflammatory conditions extend the life span of neutrophils. Cytokines such as G-CSF, GM-CSF, TNF, type I and type II interferons (IFN) as well as bacterial products such as LPS promote neutrophil survival [reviewed in [17]], which might favor the chronicity of inflammatory diseases. Although neutrophils are terminally differentiated cells, among the molecules that regulate their survival are cell cycle regulators. These include survivin, cyclin-dependent kinases, and PCNA, a nuclear factor involved in DNA replication and repairing of proliferating cells [18].
Elucidating the pathways that control neutrophil life span might lead to improved treatments for a wide array of diseases. It will also contribute to understand how to circumvent important side effects of therapeutic agents, including chemotherapy, that by inducing neutropenia render patients susceptible to infections.
Human neutrophils express a wide array of pattern recognition receptors
Neutrophils are equipped with a wide spectrum of receptors for shared pathogen-associated molecular patterns (PAMPs). These `pattern recognition receptors' (PPRs) include toll-like receptors (TLRs), NACHT-leucine-rich repeat receptors (NLRs), cytoplasmic nucleic acid sensors, and C-type lectin receptors.
Human neutrophils express both surface and endosomal TLRs, which include TLR7–9 but not TLR3. TLR7 message is expressed at very low levels in resting neutrophils, but is induced upon exposure to cytokines such as type I IFN [19,20•]. They also express a wide array of cytoplasmic PPRs. The RNA sensors MDA5 and RIG-I are not only constitutively expressed in the cytosol [21,22], but also stored in secretory vesicles from where they translocate to the cell surface, a process that does not lead to downstream signaling [23]. The cytosolic DNA sensors IFN-inducible protein 16 (IFI-16), leucin-rich repeat interacting protein-1 (LRRFIP1) and DDX41 are also found in neutrophils. Activation of these receptors leads to phosphorylation and nuclear translocation of IRF3 and subsequent IFNβ transcription [24]. Intriguingly, while human and mouse neutrophils transcribe Type I IFNs, the corresponding proteins have not been documented in neutrophil cultures [19,20•,21,25]. This conundrum remains to be explained.
Human neutrophils express most receptors for the Fc portion of IgG. Engagement of these receptors by opsonized pathogens permits their internalization and ultimate elimination. FcγRs are also important, however, for the internalization of self nucleic acid-containing immune complexes (ICs) into endosomal compartments [26], which could contribute to autoimmune pathology [20•].
Neutrophils sense and release DAMPS
PRRs recognize not only PAPMs, but also damage-associated molecules (DAMPs) released during non-apoptotic cell death. DAMPs contribute to sterile inflammation, especially if chemical (i.e. oxidation) or other stress-induced modifications occur at the time of cell death. Among the best characterized DAMPs are high mobility box group 1 (HMGB1), heat shock (HSPs) and S100 proteins; ATP, nucleic acids, crystals such as uric acid, cytokines such as IL1 and IL33, as well as mitochondrial components. DAMPs can also derive from extracellular sources, as in the case of hyaluronan (HA), a component of the extracellular matrix [27••].
Direct recognition of DAMPs contributes to neutrophil-mediated pathology. Mitochondrial-derived DAMPs (mtDAMP) such as formyl peptides and mtDNA activate neutrophils, for example, through formyl peptide receptor-1 and TLR9, respectively. These DAMPs circulate in the blood of patients with severe trauma and induce neutrophil chemotaxis in vitro. In vivo, inoculation of mtDAMP into the trachea of rats causes significant lung injury associated with massive neutrophil infiltration into the airways [28•,29].
Neutrophils also represent a rich source of DAMPs that, through multiple mechanisms, lead to nucleic acid-induced immune activation and might contribute to autoimmunity Fig. 1. During necrosis and NETosis, for example, they release large amounts of HMGB1 and anti-microbial peptides such as LL-37. These cationic molecules bind self-nucleic acids and increase their immunogenicity. Thus, neutrophil-derived HMGB1-and/or LL37-DNA complexes induce type I IFN production upon binding TLR9 in plasmacytoid DCs (pDCs) [20•,30••]. LL37/RNA complexes also activate myeloid DCs (mDCs) through TLR7 and TLR8 binding [31•]. These molecules are abundant in psoriatic skin and released in large amounts by systemic lupus erythematosus (SLE) neutrophils upon activation in vitro [20•,30••,32•]. They also bind DNA within SLE ICs, which in turn induce type I IFN production upon internalization by pDCs [33].
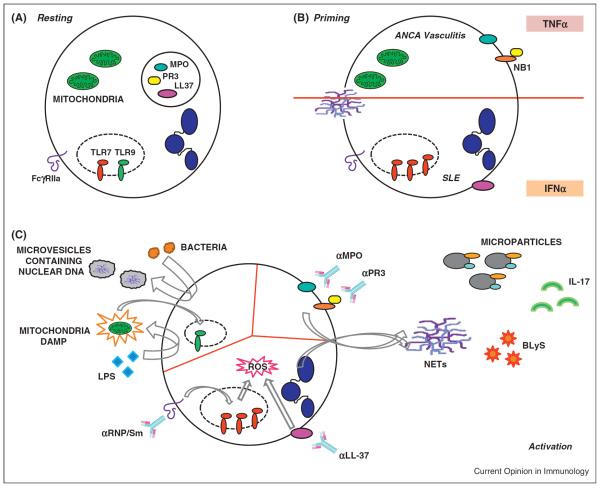
Figure 1.
(A) Under resting conditions (upper left), neutrophils constitutively express FcgR and endosomal TLRs. Proteases and antimicrobial peptides are stored in cytoplasmic granules. (B) Upon priming with cytokines such as TNFα, granular proteins are translocated to the plasma membrane. IFNα induces TR7 expression and promotes the translocation of anti-microbial peptides (i.e. LL37) to the plasma membrane. (C) Upper left: activation with bacterial can lead to the release of microparticles containing chromatin/DNA. This can eventually lead to total enucleation. Mitochondria from damaged cells can be internalized and activate endosomal TLR9; LPS-activation in turn leads to the extrusion of mitochondrial-derived DAMPs that can activate other cells. Upper right: activation with ANCA-MPO/PR3 might leads to NETosis and/or the release of microparticles displaying surface ANCA-related antigens. In addition, this type of activation can induce secretion of B cell-activating factors (BLyS/BAFF) and/or cytokines such as IL17. Bottom: activation with SLE-derived anti-RNP antibodies or anti-LL37 antibodies leads to extrusion of immunogenic DNA in the form of NETs.
Neutrophils can be a source of nucleic acid-containing DAMPs also while alive. Thus, activated neutrophils and eosinophils release DNA from mitochondria [16,15]. These organelles are evolutionary related to saprophytic bacteria and contain CpG repeats within their DNA. Indeed, mtDNA activates nucleic acid sensors both in cell autonomous [34] and non-cell autonomous manner [29]. Thus, neutrophils can concomitantly sense and release DAMPs, and this interplay might contribute to chronic inflammation in humans [35].
Neutrophils in human chronic inflammatory and autoimmune diseases
Neutrophils infiltrate the tissues of patients with inflammatory and autoimmune diseases, including the psoriatic skin, rheumatoid arthritis (RA) synovial tissue and fluid, glomeruli from SLE and anti-neutrophil cytoplasmic antibody-related (ANCA) vasculitides, thrombi from patients with deep venous thrombosis (DVT), etc. Because they are short-lived and difficult to manipulate in vitro, studies on neutrophils have lagged well behind those of other leukocytes, especially lymphocytes, in these diseases.
ANCA-associated vasculitides (AVV)
Some of the most potent enzymes stored in neutrophil granules become the target of immune attack in patients with ANCA-associated vasculitides (AVV), a heterogeneous group of diseases that include Wegener's granulomatosis (WG), microscopic polyangiitis (MPA), and Churg–Strauss Syndrome (CSS). These patients develop severe kidney and/or respiratory tract involvement owing to necrotizing small vessel inflammation [reviewed in [36]]. The best characterized ANCA antigens are PR3 and MPO, and there is solid evidence to support that these neutrophil proteins drive autoimmunity in AAV. Thus, splenocyte transfer from MPO−/− mice immunized with MPO into Rag−/− mice induces the development of MPO–ANCA and reproduces the kidney lesions of AAV patients and in the recipient mice [37]. ANCA antigens are secreted during degranulation as well as translocated to the cell surface upon priming with proinflammatory cytokines [38]. In the case of PR3, the membrane antigen NB1 (CD177) acts as a bridging molecule to keep the translocated molecule bound to the cell surface [39]. This NB1/PR3 complex associates with Mac-1 (CD11b/CD18) and becomes a `signaling platform' for neutrophil activation upon PR3–ANCA binding [40].
A third ANCA target, LAMP-2, has been recently identified. LAMP-2 is a heavily glycosylated type I membrane protein expressed on the surface of granules that also contain MPO and PR3. Autoantibodies against LAMP-2 activate neutrophils in vitro by inducing actin-network reorganization and degranulation, which triggers micro-vascular endothelial cell apoptosis. These antibodies cause pauci-immune necrotizing glomerulonephritis when injected into rats.
How neutrophil granular proteins become antigenic in AAV remains an unresolved question. In the case of LAMP-2, its ANCA epitope shares 100% homology with the bacterial adhesin FimH, and rats immunized with FimH develop kidney lesions and antibodies that cross-react with human LAMP-2. Some cases of AAV might therefore result from cross-reactivity triggered by infectious agents [41]. In humans, polymorphic variants of PR3 have recently been shown to confer susceptibility to PR3 AVV, but the underlying mechanisms responsible for this association are not known [42].
ANCA-activated neutrophils release toxic factors that play a pivotal role in the damage of micro-vessels. Some of these factors are well characterized (ROS and lytic enzymes) while others are starting to be elucidated. ANCA-activation leads, for example, to the release of microparticles displaying ANCA-autoantigens on their surface. These microparticles adhere to the endothelium and cause damage by promoting ROS, IL6, IL8 and thrombin generation [43]. In vitro, TNFα-primed neutrophils undergo NETosis when exposed to ANCA. NET-derived DNA/protein complexes stick to endothelial cells causing capillary inflammation and cell death. Indeed, aggregates of extracellular DNA and circulating MPO/DNA complexes are found in kidney biopsies and blood of AVV patients, respectively [44•].
ANCA-neutrophil interplay has also been linked to the release of IL-17, which is elevated in AVV serum and plays an important role in renal tissue damage in various mouse models of glomerulonephritis. Kidneys from AVV-patients express high levels of IL-17 mRNA and neutrophils, which are found in large numbers in AVV glomeruli and tubulointerstitium, seem to be one of the cellular sources of this cytokine [45], which in turn might attract more neutrophils to the inflammatory sites.
ANCA-activated neutrophils also release BLyS, which could contribute to the amplification of ANCA responses by inducing B cell survival and plasma cell differentiation [46]. The pathogenic role of B cells in AAV is indeed supported by recent clinical trials showing that B cell depletion is an effective therapy for AVV [47].
Systemic lupus erythematosus (SLE)
SLE is a systemic autoimmune disease where breakdown of tolerance to nuclear antigens and inefficient removal of ICs leads to tissue damage. The skin, blood vessels, kidneys, central nervous system and joints may become the target of SLE-induced inflammation. Neutrophil counts are decreased in SLE blood, but the presence of these cells in inflamed tissues from patients is well documented. Finding neutrophils in kidney biopsies, for example, is considered a sign of SLE glomerular activity, and detection of neutrophil-derived proteins (i.e. lipocalin) in the urine is a surrogate marker of disease activity [48]. Moreover, treatments that increase granulopoiesis, such as G-CSF administration, induce lupus flares [49]. Despite this evidence, the role of neutrophils in SLE pathogenesis has not been elucidated.
Genomic studies also support the contribution of neutrophils to human SLE. Early onset disease, for example, is characterized by the expression of a type I IFN-inducible signature in most hemopoietic cells. Activated pDCs are thought to be the source of IFN, as high doses of steroids that kill pDCs in vitro erase this signature in vivo [50]. pDC activation leading to IFN production occurs upon internalization of SLE ICs through FcγRIIa and subsequent binding to endosomal TLR7–9 [51]. Recent evidence supports that IC activation of neutrophils, which also express FcγRs and endosomal TLRs, contribute to sustain IFN production by pDCs [20•]. Thus, exposure of SLE neutrophils to anti-RNP ICs in vitro induces the release of DNA and DNA modifying proteins, including HMGB1 and LL-37. Supernatants from these cultures activate pDCs in a TLR9-dependent manner and lead to the secretion of high levels of type I IFN [20•]. In addition, as described above for ANCA, SLE autoantibodies trigger neutrophil activation and NET release through the recognition of anti-microbial peptides displayed on their surface [32•]. Furthermore, a population of low-density neutrophils that is expanded in SLE blood [52] seems to spontaneously release NETs that activate pDCs and induce endothelial cell death in vitro [53]. The role of NETosis in SLE has recently been questioned in an MRL/lpr murine model of SLE made deficient in ROS production through the ablation of the Nox gene [54]. This is not representative however of the human disease, as type I IFN does not seem to play a role and DNA signaling through TLR9 is protective in this model.
Common genetic variants linked to IC-internalization by neutrophils and pDCs, such as FCGRIIA, predispose to SLE [55]. Rare genetic variants associated with SLE encode components of the classical complement activation pathway (i.e. C1q) [56], and proteins involved in the degradation of extracellular DNA (i.e. DNAse1 and DNase1L3). These rare variants are highly penetrant for disease development and risk of nephritis [57]. Interestingly, C1q is found within NETs and anti-C1q autoantibodies from SLE patients bind to and protect NETs from degradation [58]. Mutations in DNAse1 and DNase1L3 might also prevent NET degradation. All these mechanisms could explain why sera from a fraction of SLE patients fail to degrade NETs in vitro [59•] and provide evidence for an amplification loop linking autoantibody-mediated neutrophil and pDC activation with type I IFN production at the core of SLE pathogenesis.
Thrombosis
It is widely accepted that neutrophils contribute to thrombosis in the context of cardiovascular diseases such as myocardial infarction, sepsis and other pathological processes. The mechanisms underlying the pro-coagulant effect of neutrophils remain however elusive.
Activated platelets induce neutrophils to release nucleosomes as well as serine proteases, which co-localize on the cell surface. Membrane bound nucleosome/protease complexes induce tissue factor- and factor XII-dependent coagulation, which might represent a physiological mechanism to halt microbe invasion [60].
In addition, neutrophil products extruded during NETosis contribute to their pro-coagulant effect. Perfusion of PMA-induced NETs with blood causes platelet adhesion, activation, aggregation and subsequent thrombus formation. The main NET components that favor platelet aggregation/activation are histones, as these proteins recapitulate the effect of NETs [61].
Using a mouse model of deep venous thrombosis (DVP) and intravital two-photon microscopy, monocytes, neutrophils and platelets have been shown to cooperate in thrombus formation. Thrombosis is initiated by crawling blood monocytes and neutrophils adhering to venous endothelium. Thrombus-resident neutrophils are then indispensable for thrombus propagation through the release of NETs [62•], which activate Factor XII and sustain coagulation.
Neutrophilic dermatoses
Neutrophils are the predominant cell type in the affected skin of patients with `neutrophilic dermatoses', a heterogeneous group of diseases of unknown etiology that include common (i.e. pyoderma gangrenosum) as well as rare syndromes (i.e. Behcet's and Sweet syndromes). Among them, chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature (CANDLE syndrome) is owing to mutations in the proteasome subunit β8 (PSMB8) [63•,64]. Nucleated, immature neutrophils accumulate in the skin of these patients as a dense interstitial infiltrate mixed with areas of cellular debris or karyorrhexis. Although the connection between the immunoproteasome and neutrophil activation is not clear, proteasome pharmacological inhibition can induce neutrophilic dermatosis [64]. CANDLE patients also display a prominent upregulation of blood interferon-inducible genes. Whether neutrophils play a role in the activation of the IFN pathway in this disease, as it might be the case in SLE, remains to be explored.
Concluding remarks
Neutrophils are highly sophisticated cells that sense, kill and die in ways designed to benefit the host. Their role in human inflammatory and autoimmune diseases has been neglected, but evidence is now accumulating that suggests considering them as therapeutic targets.
Because of their critical role in anti-microbial defense, strategies aimed at depleting neutrophils would render patients too susceptible to infections. In fact, neutropenia is a side effect of several recently approved biologicals intended to treat RA, juvenile arthritis and autoinflammatory diseases, among others. Whether some of the beneficial effects of these drugs rely on decreasing the number of peripheral neutrophils is not known.
Understanding the mechanisms that underlie neutrophil activation, migration, survival and their eclectic forms of death in health and disease will provide us with safer and perhaps more relevant approaches to treat chronic inflammatory conditions without the permanent threat of infection. Significant advances in the past decade have expanded our knowledge on the interesting and complex neutrophil biology. We look forward to the next decade to learn how to use this knowledge for the benefit of patients.
References and recommended reading
Papers of particular interest, published within the period of review, have been highlighted as:
• of special interest
•• of outstanding interest
- 1•.Summers C, Rankin SM, Condliffe AM, Singh N, Peters AM, Chilvers ER. Neutrophil kinetics in health and disease. Trends Immunol. 2010;31:318–324. doi: 10.1016/j.it.2010.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]; Comprehensive review of neutrophil kinetics and the signals that regulate the peripheral neutrophil pool.
- 2••.Nathan C. Neutrophils and immunity: challenges and opportunities. Nat Rev Immunol. 2006;6:173–182. doi: 10.1038/nri1785. [DOI] [PubMed] [Google Scholar]; Beautiful and insightful review of the multiple and fundamental roles of neutrophil in the immune system.
- 3.Xu Y, Loison F, Luo HR. Neutrophil spontaneous death is mediated by down-regulation of autocrine signaling through GPCR, PI3Kgamma ROS, and actin. Proc Natl Acad Sci USA. 2010;107:2950–2955. doi: 10.1073/pnas.0912717107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Geering B, Simon HU. Peculiarities of cell death mechanisms in neutrophils. Cell Death Differ. 2011;18:1457–1469. doi: 10.1038/cdd.2011.75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Galluzzi L, Vitale I, Abrams JM, Alnemri ES, Baehrecke EH, Blagosklonny MV, Dawson TM, Dawson VL, El-Deiry WS, Fulda S, et al. Molecular definitions of cell death subroutines: recommendations of the nomenclature committee on cell death 2012. Cell Death Differ. 2012;19:107–120. doi: 10.1038/cdd.2011.96. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Mihalache CC, Yousefi S, Conus S, Villiger PM, Schneider EM, Simon HU. Inflammation-associated autophagy-related programmed necrotic death of human neutrophils characterized by organelle fusion events. J Immunol. 2011;186:6532–6542. doi: 10.4049/jimmunol.1004055. [DOI] [PubMed] [Google Scholar]
- 7•.Papayannopoulos V, Zychlinsky A. NETs: a new strategy for using old weapons. Trends Immunol. 2009;30:513–521. doi: 10.1016/j.it.2009.07.011. [DOI] [PubMed] [Google Scholar]; Very comprehensive review of NETosis.
- 8.Bianchi M, Hakkim A, Brinkmann V, Siler U, Seger RA, Zychlinsky A, Reichenbach J. Restoration of NET formation by gene therapy in CGD controls aspergillosis. Blood. 2009;114:2619–2622. doi: 10.1182/blood-2009-05-221606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hakkim A, Fuchs TA, Martinez NE, Hess S, Prinz H, Zychlinsky A, Waldmann H. Activation of the Raf–MEK–ERK pathway is required for neutrophil extracellular trap formation. Nat Chem Biol. 2011;7:75–77. doi: 10.1038/nchembio.496. [DOI] [PubMed] [Google Scholar]
- 10.Papayannopoulos V, Metzler KD, Hakkim A, Zychlinsky A. Neutrophil elastase and myeloperoxidase regulate the formation of neutrophil extracellular traps. J Cell Biol. 2010;191:677–691. doi: 10.1083/jcb.201006052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li P, Li M, Lindberg MR, Kennett MJ, Xiong N, Wang Y. PAD4 is essential for antibacterial innate immunity mediated by neutrophil extracellular traps. J Exp Med. 2010;207:1853–1862. doi: 10.1084/jem.20100239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Remijsen Q, Vanden Berghe T, Wirawan E, Asselbergh B, Parthoens E, De Rycke R, Noppen S, Delforge M, Willems J, Vandenabeele P. Neutrophil extracellular trap cell death requires both autophagy and superoxide generation. Cell Res. 2011;21:290–304. doi: 10.1038/cr.2010.150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Pilsczek FH, Salina D, Poon KK, Fahey C, Yipp BG, Sibley CD, Robbins SM, Green FH, Surette MG, Sugai M, et al. A novel mechanism of rapid nuclear neutrophil extracellular trap formation in response to Staphylococcus aureus. J Immunol. 2010;185:7413–7425. doi: 10.4049/jimmunol.1000675. [DOI] [PubMed] [Google Scholar]
- 14.Yipp BG, Petri B, Salina D, Jenne CN, Scott BN, Zbytnuik LD, Pittman K, Asaduzzaman M, Wu K, Meijndert HC, et al. Infection-induced NETosis is a dynamic process involving neutrophil multitasking in vivo. Nat Med. 2012 doi: 10.1038/nm.2847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Yousefi S, Gold JA, Andina N, Lee JJ, Kelly AM, Kozlowski E, Schmid I, Straumann A, Reichenbach J, Gleich GJ, et al. Catapult-like release of mitochondrial DNA by eosinophils contributes to antibacterial defense. Nat Med. 2008;14:949–953. doi: 10.1038/nm.1855. [DOI] [PubMed] [Google Scholar]
- 16.Yousefi S, Mihalache C, Kozlowski E, Schmid I, Simon HU. Viable neutrophils release mitochondrial DNA to form neutrophil extracellular traps. Cell Death Differ. 2009;16:1438–1444. doi: 10.1038/cdd.2009.96. [DOI] [PubMed] [Google Scholar]
- 17.Witko-Sarsat V, Pederzoli-Ribeil M, Hirsch E, Sozzani S, Cassatella MA. Regulating neutrophil apoptosis: new players enter the game. Trends Immunol. 2011;32:117–124. doi: 10.1016/j.it.2011.01.001. [DOI] [PubMed] [Google Scholar]
- 18.Witko-Sarsat V, Mocek J, Bouayad D, Tamassia N, Ribeil JA, Candalh C, Davezac N, Reuter N, Mouthon L, Hermine O, et al. Proliferating cell nuclear antigen acts as a cytoplasmic platform controlling human neutrophil survival. J Exp Med. 2010;207:2631–2645. doi: 10.1084/jem.20092241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Hayashi F, Means TK, Luster AD. Toll-like receptors stimulate human neutrophil function. Blood. 2003;102:2660–2669. doi: 10.1182/blood-2003-04-1078. [DOI] [PubMed] [Google Scholar]
- 20•.Garcia-Romo GS, Caielli S, Vega B, Connolly J, Allantaz F, Xu Z, Punaro M, Baisch J, Guiducci C, Coffman RL, et al. Netting neutrophils are major inducers of type I IFN production in pediatric systemic lupus erythematosus. Sci Transl Med. 2011;3:73ra20. doi: 10.1126/scitranslmed.3001201. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper describes interplay between neutrophils and pDCs that might contribute to a type I IFN self-amplification loop in human lupus.
- 21.Tamassia N, Le Moigne V, Rossato M, Donini M, McCartney S, Calzetti F, Colonna M, Bazzoni F, Cassatella MA. Activation of an immunoregulatory and antiviral gene expression program in poly(I:C)-transfected human neutrophils. J Immunol. 2008;181:6563–6573. doi: 10.4049/jimmunol.181.9.6563. [DOI] [PubMed] [Google Scholar]
- 22.Prince LR, Whyte MK, Sabroe I, Parker LC. The role of TLRs in neutrophil activation. Curr Opin Pharmacol. 2011;11:397–403. doi: 10.1016/j.coph.2011.06.007. [DOI] [PubMed] [Google Scholar]
- 23.Berger M, Hsieh CY, Bakele M, Marcos V, Rieber N, Kormann M, Mays L, Hofer L, Neth O, Vitkov L, et al. Neutrophils express distinct RNA receptors in a non-canonical way. J Biol Chem. 2012;287:19409–19417. doi: 10.1074/jbc.M112.353557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Tamassia N, Bazzoni F, Le Moigne V, Calzetti F, Masala C, Grisendi G, Bussmeyer U, Scutera S, De Gironcoli M, Costantini C, et al. IFN-beta expression is directly activated in human neutrophils transfected with plasmid DNA and is further increased via TLR-4-mediated signaling. J Immunol. 2012;189:1500–1509. doi: 10.4049/jimmunol.1102985. [DOI] [PubMed] [Google Scholar]
- 25.Denny MF, Yalavarthi S, Zhao W, Thacker SG, Anderson M, Sandy AR, McCune WJ, Kaplan MJ. A distinct subset of proinflammatory neutrophils isolated from patients with systemic lupus erythematosus induces vascular damage and synthesizes type I IFNs. J Immunol. 2010;184:3284–3297. doi: 10.4049/jimmunol.0902199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Means TK, Latz E, Hayashi F, Murali MR, Golenbock DT, Luster AD. Human lupus autoantibody-DNA complexes activate DCs through cooperation of CD32 and TLR9. J Clin Invest. 2005;115:407–417. doi: 10.1172/JCI23025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27••.Chen GY, Nunez G. Sterile inflammation: sensing and reacting to damage. Nat Rev Immunol. 2010;10:826–837. doi: 10.1038/nri2873. [DOI] [PMC free article] [PubMed] [Google Scholar]; Great review on DAMPs and their sensors.
- 28•.Zhang Q, Raoof M, Chen Y, Sumi Y, Sursal T, Junger W, Brohi K, Itagaki K, Hauser CJ. Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature. 2010;464:104–107. doi: 10.1038/nature08780. [DOI] [PMC free article] [PubMed] [Google Scholar]; Very interesting work describing the mechanisms responsible for neutrophil activation by mitochondrial DAMPs.
- 29.Zhang Q, Itagaki K, Hauser CJ. Mitochondrial DNA is released by shock and activates neutrophils via p38 map kinase. Shock. 2010;34:55–59. doi: 10.1097/SHK.0b013e3181cd8c08. [DOI] [PubMed] [Google Scholar]
- 30••.Lande R, Gregorio J, Facchinetti V, Chatterjee B, Wang YH, Homey B, Cao W, Su B, Nestle FO, Zal T, et al. Plasmacytoid dendritic cells sense self-DNA coupled with antimicrobial peptide. Nature. 2007;449:564–569. doi: 10.1038/nature06116. [DOI] [PubMed] [Google Scholar]; First description of the role of cationic anti-microbial peptides in modifying mammalian DNA to increase its interferogenic. capacity.
- 31•.Ganguly D, Chamilos G, Lande R, Gregorio J, Meller S, Facchinetti V, Homey B, Barrat FJ, Zal T, Gilliet M. Self-RNA-antimicrobial peptide complexes activate human dendritic cells through TLR7 and TLR8. J Exp Med. 2009;206:1983–1994. doi: 10.1084/jem.20090480. [DOI] [PMC free article] [PubMed] [Google Scholar]; First description of the role of cationic anti-microbial peptides in modifying mammalian RNA to activate myeloid DCs.
- 32•.Lande R, Ganguly D, Facchinetti V, Frasca L, Conrad C, Gregorio J, Meller S, Chamilos G, Sebasigari R, Riccieri V, et al. Neutrophils activate plasmacytoid dendritic cells by releasing self-DNA-peptide complexes in systemic lupus erythematosus. Sci Transl Med. 2011;3:73ra19. doi: 10.1126/scitranslmed.3001180. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper describes the presence of anti-LL37 antibodies in a fraction of SLE patients. These antibodies bind to neutrophils and induce the release of immunogenic DNA.
- 33.Tian J, Avalos AM, Mao SY, Chen B, Senthil K, Wu H, Parroche P, Drabic S, Golenbock D, Sirois C, et al. Toll-like receptor 9-dependent activation by DNA-containing immune complexes is mediated by HMGB1 and RAGE. Nat Immunol. 2007;8:487–496. doi: 10.1038/ni1457. [DOI] [PubMed] [Google Scholar]
- 34.Oka T, Hikoso S, Yamaguchi O, Taneike M, Takeda T, Tamai T, Oyabu J, Murakawa T, Nakayama H, Nishida K, et al. Mitochondrial DNA that escapes from autophagy causes inflammation and heart failure. Nature. 2012;485:251–255. doi: 10.1038/nature10992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Makam M, Diaz D, Laval J, Gernez Y, Conrad CK, Dunn CE, Davies ZA, Moss RB, Herzenberg LA, Tirouvanziam R. Activation of critical, host-induced, metabolic and stress pathways marks neutrophil entry into cystic fibrosis lungs. Proc Natl Acad Sci USA. 2009;106:5779–5783. doi: 10.1073/pnas.0813410106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Kallenberg CG. Pathophysiology of ANCA-associated small vessel vasculitis. Curr Rheumatol Rep. 2010;12:399–405. doi: 10.1007/s11926-010-0138-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xiao H, Heeringa P, Hu P, Liu Z, Zhao M, Aratani Y, Maeda N, Falk RJ, Jennette JC. Antineutrophil cytoplasmic autoantibodies specific for myeloperoxidase cause glomerulonephritis and vasculitis in mice. J Clin Invest. 2002;110:955–963. doi: 10.1172/JCI15918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Falk RJ, Terrell RS, Charles LA, Jennette JC. Anti-neutrophil cytoplasmic autoantibodies induce neutrophils to degranulate and produce oxygen radicals in vitro. Proc Natl Acad Sci USA. 1990;87:4115–4119. doi: 10.1073/pnas.87.11.4115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.von Vietinghoff S, Tunnemann G, Eulenberg C, Wellner M, Cristina Cardoso M, Luft FC, Kettritz R. NB1 mediates surface expression of the ANCA antigen proteinase 3 on human neutrophils. Blood. 2007;109:4487–4493. doi: 10.1182/blood-2006-10-055327. [DOI] [PubMed] [Google Scholar]
- 40.Jerke U, Rolle S, Dittmar G, Bayat B, Santoso S, Sporbert A, Luft F, Kettritz R. Complement receptor Mac-1 is an adaptor for NB1 (CD177)-mediated PR3-ANCA neutrophil activation. J Biol Chem. 2011;286:7070–7081. doi: 10.1074/jbc.M110.171256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kain R, Exner M, Brandes R, Ziebermayr R, Cunningham D, Alderson CA, Davidovits A, Raab I, Jahn R, Ashour O, et al. Molecular mimicry in pauci-immune focal necrotizing glomerulonephritis. Nat Med. 2008;14:1088–1096. doi: 10.1038/nm.1874. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lyons PA, Rayner TF, Trivedi S, Holle JU, Watts RA, Jayne DR, Baslund B, Brenchley P, Bruchfeld A, Chaudhry AN, et al. Genetically distinct subsets within ANCA-associated vasculitis. Engl J Med. 2012;367:214–223. doi: 10.1056/NEJMoa1108735. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Hong Y, Eleftheriou D, Hussain AA, Price-Kuehne FE, Savage CO, Jayne D, Little MA, Salama AD, Klein NJ, Brogan PA. Antineutrophil cytoplasmic antibodies stimulate release of neutrophil microparticles. J Am Soc Nephrol. 2012;23:49–62. doi: 10.1681/ASN.2011030298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44•.Kessenbrock K, Krumbholz M, Schonermarck U, Back W, Gross WL, Werb Z, Grone HJ, Brinkmann V, Jenne DE. Netting neutrophils in autoimmune small-vessel vasculitis. Nat Med. 2009;15:623–625. doi: 10.1038/nm.1959. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper describes how neutrophil activation with TNF leads to NET formation upon subsequent exposure to ANCA from patients with AVV.
- 45.Velden J, Paust HJ, Hoxha E, Turner JE, Steinmetz OM, Wolf G, Jabs WJ, Ozcan F, Beige J, Heering PJ, et al. Renal IL-17 expression in human ANCA-associated glomerulonephritis. Am J Physiol Renal Physiol. 2012;302:F1663–F1673. doi: 10.1152/ajprenal.00683.2011. [DOI] [PubMed] [Google Scholar]
- 46.Holden NJ, Williams JM, Morgan MD, Challa A, Gordon J, Pepper RJ, Salama AD, Harper L, Savage CO. ANCA-stimulated neutrophils release BLyS and promote B cell survival: a clinically relevant cellular process. Ann Rheum Dis. 2011;70:2229–2233. doi: 10.1136/ard.2011.153890. [DOI] [PubMed] [Google Scholar]
- 47.Stone JH, Merkel PA, Spiera R, Seo P, Langford CA, Hoffman GS, Kallenberg CG, St Clair EW, Turkiewicz A, Tchao NK, et al. Rituximab versus cyclophosphamide for ANCA-associated vasculitis. N Engl J Med. 2010;363:221–232. doi: 10.1056/NEJMoa0909905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Brunner HI, Mueller M, Rutherford C, Passo MH, Witte D, Grom A, Mishra J, Devarajan P. Urinary neutrophil gelatinase-associated lipocalin as a biomarker of nephritis in childhood-onset systemic lupus erythematosus. Arthritis Rheum. 2006;54:2577–2584. doi: 10.1002/art.22008. [DOI] [PubMed] [Google Scholar]
- 49.Vasiliu IM, Petri MA, Baer AN. Therapy with granulocyte colonystimulating factor in systemic lupus erythematosus may be associated with severe flares. J Rheumatol. 2006;33:1878–1880. [PubMed] [Google Scholar]
- 50.Guiducci C, Gong M, Xu Z, Gill M, Chaussabel D, Meeker T, Chan JH, Wright T, Punaro M, Bolland S, et al. TLR recognition of self nucleic acids hampers glucocorticoid activity in lupus. Nature. 2010;465:937–941. doi: 10.1038/nature09102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Ronnblom L, Pascual V. The innate immune system in SLE: type I interferons and dendritic cells. Lupus. 2008;17:394–399. doi: 10.1177/0961203308090020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pascual V, Chaussabel D, Banchereau J. A genomic approach to human autoimmune diseases. Annu Rev Immunol. 2010;28:535–571. doi: 10.1146/annurev-immunol-030409-101221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Villanueva E, Yalavarthi S, Berthier CC, Hodgin JB, Khandpur R, Lin AM, Rubin CJ, Zhao W, Olsen SH, Klinker M, et al. Netting neutrophils induce endothelial damage, infiltrate tissues, and expose immunostimulatory molecules in systemic lupus erythematosus. J Immunol. 2011;187:538–552. doi: 10.4049/jimmunol.1100450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Campbell AM, Cashgaian M, Schlomchik MJ. NADPH Oxydase inhibits the pathogenesis if systemic lupus erythematosus. Sci Transl Med. 2012;4:141–147. doi: 10.1126/scitranslmed.3004801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Moser KL, Kelly JA, Lessard CJ, Harley JB. Recent insights into the genetic basis of systemic lupus erythematosus. Genes Immun. 2009;10:373–379. doi: 10.1038/gene.2009.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Cheng MH, Anderson MS. Monogenic autoimmunity. Annu Rev Immunol. 2012;30:393–427. doi: 10.1146/annurev-immunol-020711-074953. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Al-Mayouf SM, Sunker A, Abdwani R, Abrawi SA, Almurshedi F, Alhashmi N, Al Sonbul A, Sewairi W, Qari A, Abdallah E, et al. Loss-of-function variant in DNASE1L3 causes a familial form of systemic lupus erythematosus. Nat Genet. 2011;43:1186–1188. doi: 10.1038/ng.975. [DOI] [PubMed] [Google Scholar]
- 58.Leffler J, Martin M, Gullstrand B, Tyden H, Lood C, Truedsson L, Bengtsson AA, Blom AM. Neutrophil extracellular traps that are not degraded in systemic lupus erythematosus activate complement exacerbating the disease. J Immunol. 2012;188:3522–3531. doi: 10.4049/jimmunol.1102404. [DOI] [PubMed] [Google Scholar]
- 59•.Hakkim A, Furnrohr BG, Amann K, Laube B, Abed UA, Brinkmann V, Herrmann M, Voll RE, Zychlinsky A. Impairment ofneutrophil extracellular trap degradation is associated with lupus nephritis. Proc Natl Acad Sci USA. 2010;107:9813–9818. doi: 10.1073/pnas.0909927107. [DOI] [PMC free article] [PubMed] [Google Scholar]; First description of a link between NETosis and SLE pathogenesis.
- 60.Massberg S, Grahl L, von Bruehl ML, Manukyan D, Pfeiler S, Goosmann C, Brinkmann V, Lorenz M, Bidzhekov K, Khandagale AB, et al. Reciprocal coupling of coagulation and innate immunity via neutrophil serine proteases. Nat Med. 2010;16:887–896. doi: 10.1038/nm.2184. [DOI] [PubMed] [Google Scholar]
- 61.Fuchs TA, Brill A, Duerschmied D, Schatzberg D, Monestier M, Myers DD, Jr, Wrobleski SK, Wakefield TW, Hartwig JH, Wagner DD. Extracellular DNA traps promote thrombosis. Proc Natl Acad Sci USA. 2010;107:15880–15885. doi: 10.1073/pnas.1005743107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62•.von Bruhl ML, Stark K, Steinhart A, Chandraratne S, Konrad I, Lorenz M, Khandoga A, Tirniceriu A, Coletti R, Kollnberger M, et al. Monocytes, neutrophils, and platelets cooperate to initiate and propagate venous thrombosis in mice in vivo. J Exp Med. 2012;209:819–835. doi: 10.1084/jem.20112322. [DOI] [PMC free article] [PubMed] [Google Scholar]; Interesting paper describing cell interactions that initiate and sustain thrombosis and the central role of neutrophils in these processes.
- 63•.Liu Y, Ramot Y, Torrelo A, Paller AS, Si N, Babay S, Kim PW, Sheikh A, Lee CC, Chen Y, et al. Mutations in proteasome subunit beta type 8 cause chronic atypical neutrophilic dermatosis with lipodystrophy and elevated temperature with evidence of genetic and phenotypic heterogeneity. Arthritis Rheum. 2012;64:895–907. doi: 10.1002/art.33368. [DOI] [PMC free article] [PubMed] [Google Scholar]; This paper reports the first association between neutrophilic dermatosis, and mutations in a subunit of the immunoproteasome.
- 64.Tanguy-Schmidt A, Avenel-Audran M, Croue A, Lissandre S, Dib M, Zidane-Marinnes M, Moles MP, Hunault-Berger M. Bortezomib-induced acute neutrophilic dermatosis. Ann Dermato Venereol. 2009;136:443–446. doi: 10.1016/j.annder.2008.11.021. [DOI] [PubMed] [Google Scholar]