Abstract
Therapeutic hypothermia has shown neuroprotective promise, but whether it can be used to improve outcome in stroke has yet to be determined in patients. Recombinant tissue plasminogen activator (rt-PA) is only given to a minority of patients with acute ischemic stroke, and is not without risk, namely significant brain hemorrhage. We explored whether mild hypothermia, in combination with rt-PA, influences the safety of rt-PA. Mice were subjected to middle cerebral artery occlusion (MCAO) using a filament model, followed by 24 hours reperfusion. Two paradigms were studied. In the first paradigm, cooling and rt-PA treatment began at the same time upon reperfusion, whereas in the second paradigm, cooling began soon after ischemia onset, and rt-PA began after re-warming and upon reperfusion. Experimental groups included: tPA treatment at normothermia (37°C), rt-PA treatment at hypothermia (33°C), no rt-PA at normothermia, and no rt-PA treatment at hypothermia. Infarct size, neurological deficit scores, blood brain barrier (BBB) permeability, brain hemorrhage, and expression of endogenous tissue plasminogen activator (tPA) and its inhibitor, plasminogen activator inhibitor (PAI-1) were assessed. For both paradigms, hypothermia reduced infarct size and neurological deficits compared to normothermia, regardless of whether rt-PA was given. rt-PA treatment increased brain hemorrhage and BBB disruption compared to normothermia, and this was prevented by cooling. However, mortality was higher when rt-PA and cooling were administered at the same time, beginning 1–2 hours post MCAO. Endogenous tPA expression was reduced in hypothermic mice, whereas PAI-1 levels were unchanged by cooling. In the setting of rt-PA treatment, hypothermia reduces brain hemorrhage, and BBB disruption, suggesting that combination therapy with mild hypothermia and rt-PA appears safe.
Introduction
Thrombolysis with recombinant tissue plasminogen activator (rt-PA) is effective in patients with acute ischemic stroke within 3–4.5 hours of symptom onset. However, the benefit of pharmacologic thrombolysis is associated with an approximately 10-fold increase risk of symptomatic intracranial hemorrhage (ICH) (NINDS, 1995; Hacke et al., 2008). Ischemia causes microvascular damage and disruption of the blood brain barrier (BBB), which can lead to hemorrhagic transformation. The use of thrombolytic agents is well known to exacerbate hemorrhagic transformation, and cause neurological deterioration which may lead to death.
At the experimental level, hypothermia is highly effective in reducing injury from stroke and other cerebral insults. Mild to moderate hypothermia (temperature decreases to 32–35°C) has been used increasingly in clinical settings, because it provides significant neuroprotection with fewer systemic complications compared to deep hypothermia. Although the effect of therapeutic hypothermia on ischemic stroke has not yet been examined in large clinical trials, findings in patients suffering cardiac arrest indicate that mild hypothermia improves neurological outcome (Bernard et al., 2002; HACA, 2002), and a few smaller studies in ischemic stroke suggest that it is feasible (Liu and Yenari, 2009; Hemmen et al., 2010). Lowering body temperature is known to have many salutary effects in ameliorating ischemic brain injury, including the reduction of brain edema and BBB disruption (Yenari and Han, 2012).
In the present study, we investigated the effects of hypothermia on rt-PA-induced hemorrhagic transformation and BBB disruption in experimental stroke with reperfusion to study the safety of combination rt-PA and therapeutic hypothermia. For this purpose we chose to use the filament model, which, in contrast to clot models, allows reperfusion at predefined time points; thus, enabling us to separate the actions of tissue plasminogen activator (tPA) on the BBB from the effects of early reperfusion due to thrombolysis and resultant reduced lesions size. Further, we explored two different paradigms that might conceivably be applied clinically. In the first paradigm, cooling and rt-PA were begun simultaneously, with cooling somewhat delayed to model a scenario where a patient might be treated in the emergency department. This paradigm was somewhat similar to the ICTus-L study (Hemmen et al., 2010), where patients received IV rt-PA, and then were randomized to cooling treatment. This paradigm would also test the safety of administering rt-PA at lower body temperatures where the thrombolytic actions of rt-PA and the body's endogenous hemostatic systems may be altered (Yenari et al., 1995; Meunier et al., 2012). The second paradigm envisioned immediate cooling followed by delayed rt-PA treatment, envisioning a clinical scenario where a stroke victim may receive cooling in the field, then brought to emergency department for rt-PA treatment.
Materials and Methods
Stroke model
Experiments were performed according to animal protocols approved by institutional panels on laboratory animal care. Male C57/BL6 mice (Jackson Labs) weighing 25–30 g were anesthetized with 5% isoflurane by facemask, followed by 2–3% for maintenance, and subjected to transient middle cerebral artery occlusion (MCAO) using previously published protocols (Yenari et al., 2006; Zheng et al., 2008). Briefly, a 6-0 uncoated monofilament suture was inserted into the internal carotid artery to occlude the middle and anterior cerebral arteries (MCA, ACA), then removed to allow reperfusion. Physiological parameters were monitored and kept in the normal range, except for temperature as described. During surgery, rectal temperature was kept between 37–38°C, which corresponds to a brain temperature between 38–39°C (Yenari et al., 2000). Mild hypothermia, defined as a rectal temperature of 30°C, corresponding to a brain temperature of 33°C, was induced using cooling blankets, as previously published by our group (Yenari et al., 2000). These temperature correlations were confirmed in pilot experiments before the start of the current study, and matched patterns previously published by our group.
Neurological deficits were assessed 24 hours after ischemia onset using a modified scale based on that originally published by Bederson et al. (1986). In our lab, the scale used is: 0, no detectable deficit; 1, flexion of the contralateral forelimb; 2, circling to the contralateral side; 3, falling to the contralateral side; 4, death (Zheng et al., 2008). After deficit scores were assessed, animals were euthanized and brains harvested for various assays as outlined below.
Experimental groups and drug treatments
Animals given rt-PA (Alteplase, Genentech, So.) were given a total dose of 10 mg/kg, with 10% as a bolus and the remainder over 30 minutes intravenously via a tail vein injection. The relatively high dose of rt-PA was chosen on the basis of the approximately 10-fold difference in fibrin-specific activity between human and rodent system (Korninger and Collen, 1981), as well as previous thrombolytic doses used in studies of rt-PA in rodents (Brinker et al., 1999; Chopp et al., 1999; Meng et al., 1999). Controls were given normal saline as a vehicle in volumes and routes identical to that of the rt-PA animals.
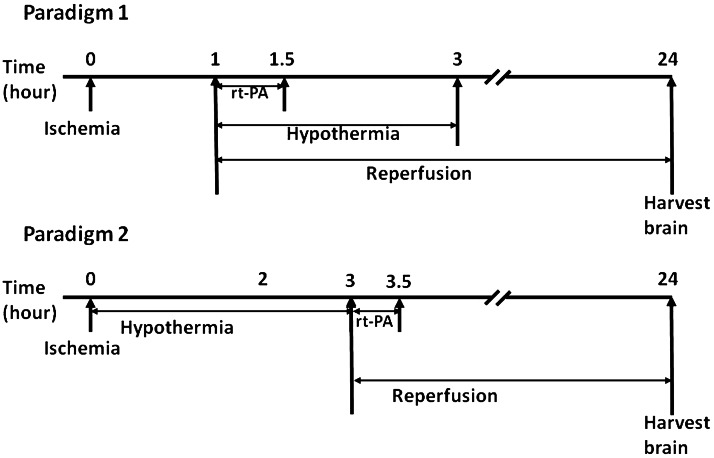
Two paradigms were used (Fig. 1). The first paradigm applied simultaneous cooling and rt-PA treatment. Pilot studies involved occluding the MCA for 2 and 3 hours, to best replicate likely clinical scenarios when rt-PA might be given in acute ischemic stroke patients. Cooling and rt-PA were both commenced upon reperfusion. However, this resulted in unacceptably high mortality rates (nearly 100%), and thus, the duration of MCAO was reduced to 1 hour. Four experimental groups were studied in the first paradigm: TN: rt-PA treatment at normothermia and begun 1 hour after MCAO and at the onset of reperfusion (37°C); TH: rt-PA treatment and hypothermia (33°C) begun immediately after reperfusion with cooling maintained for 3 h; NN:vehicle infusion at normothermia). NH: vehicle infusion with hypothermia applied in the same manner as the TH group. In the second paradigm, cooling was preceded by rt-PA treatment. These mice were subjected to 3 hours MCAO as they had no difficulty surviving the observation period. rt-PA was administered 3 hours post MCAO during reperfusion and after rewarming. Cooling began immediately after MCAO. Three experimental groups were studied. TN: rt-PA treatment at normothermia; TH: rt-PA treatment plus hypothermia; and NN: vehicle at normothermia. Because no adverse effect of cooling alone was observed in the first paradigm, the NH group was not studied again in paradigm 2, to minimize the numbers of animals studied.
FIG. 1.
Diagrams showing the two experimental paradigms studied. In paradigm #1, animals underwent 1 hour middle cerebral artery occlusion (MCAO) followed by recombinant tissue plasminogen activator (rt-PA) treatment and cooling (simultaneous treatment). In paradigm #2, animals underwent 3 hours MCAO and 3 hours cooling following by rt-PA treatment at normothermia (staggered treatment).
Infarct size, intracerebral hemorrhage scores
Twenty four hours later, animals were euthanized; brains were removed, and cut into 2 mm coronal slices. Sections were photographed and inspected for gross hemorrhage. Five brain sections were visually inspected for gross hemorrhage and scored by a rater blinded to treatment using methods previously published by our group (Yenari et al., 1997a, 1997b, 2006). Only cerebral hemorrhages visible to the naked eye were scored: 0, if there was no gross hemorrhage, 1=small amount of hemorrhage (present on no more than 1 brain section), 2=medium amount of hemorrhage (present on 2–3 sections), 3=large amount of hemorrhage (present on >3 sections, but no subarachnoid blood), 4=severe hemorrhage=same as 3 but brain also has subarachnoid blood.
Sections were then immersed in 2,3,5-triphenyltetrazolium chloride (TTC; Sigma) at 37°C for 20 minutes. Infarct volume was determined using previously published methods, including a method to correct for the presence of edema (Swanson et al., 1990; Yenari et al., 2006). Afterwards, brains were fixed and prepared for histochemistry.
BBB disruption assay
BBB disruption was assessed by quantifying the amount of extravasated Evans blue dye (EBD). 100 μL of 4% EBD in phosphate-buffered saline (PBS) was infused via the tail vein 3 hours before euthanasia (n=6/group). At the time of euthanasia, mice were anesthetized and transcardially perfused with PBS. The extent of BBB disruption was by computing the volume of dye extravasation by tracing the region of blue-stained tissue from coronal sections, normalized to the infarct volume to correct for smaller lesion sizes among treated animals (Yenari et al., 2006).
To validate the above method of estimating the volume of EBD extravasation, an established spectrophotometric method was carried out (Belayev et al., 1996). Brains of six animals were injected with 0 to 100 μL intrastriatally under anesthesia using a steretaxic frame. Twenty four hours later, brains were harvested and sectioned as before. After photographing brain sections, sections were homogenized in 50% trichloroacetate. The extracted dye was diluted in ethanol (1:3), and fluorescence was determined on a spectrophotometer (excitation:620 nm, emission:680 nm). External standards in the same solvent (100–500 ng/mL) were prepared and used to calculate the amount of dye present in the brain samples, and expressed per gram of tissue (Belayev et al., 1996). Spectrophotometric measurements were compared to those derived from manual tracings.
Immunohistochemistry
To characterize the patterns of endogenous tPA expression and that of its endogenous inhibitor, plasminogen activator inhibitor (PAI-1), a separate set of animals not treated with rt-PA was subjected to MCAO (6 at normothermia and 6 at hypothermia with cooling started immediately after MCAO and maintained for 23 hours, as in Paradigm 1), then perfused transcardially with normal saline followed by 3% paraformaldehyde. The animals' brains were postfixed in 3% paraformaldehyde and a 30% sucrose solution for 1 day at 4°C. 25-micrometer sections were cut onto glass slides in the coronal plane by using a cryostat. Tissue sections were treated with proteinase K (1:2 dilution; Dako Corp.) for 15 minutes and given three 5-minute washes in PBS. The sections were blocked in PBS containing 5% normal serum, 1% bovine serum albumin, and 0.3% Triton X-100 for 1 hour at room temperature, after which they were incubated in primary antibodies diluted in blocking solution at 4°C overnight. Tissue sections on slides were washed with PBS and incubated for 2 hours at room temperature (light shielded) in secondary antibodies diluted in blocking solution, the slides were covered and the tissue was examined with the aid of a fluorescence microscope (Carl Zeiss). Primary antibodies against tPA (1:500,PAM-3; American Diagnostica, Inc.) or PAI-1 (1:500, #1062; American Diagnostica) were applied. Sections were then reacted with biotinylated secondary antibodies then reacted with DAB and counterstained with hematoxylin and eosin.
Adjacent sections were also double labeled to identify the source of tPA and PAI-1 using double immunofluorescent staining. Primary murine antibodies used to identify neurons (NEUN; 1:300 dilution; Sigma Chemical Corp.), astrocytes (GFAP, 1:100 dilution; Pharmingen), or macrophages/microglia (ED1, 1:200 dilution; Research Diagnostics) were combined with primary antibodies against tPA or PAI-1 as described above. The secondary antibodies were Cy-3–conjugated goat anti–rabbit IgG (1:400 dilution; Jackson ImmunoResearch), and fluorescein isothiocyanate–conjugated anti–mouse IgG (1:100 dilution; Jackson ImmunoResearch).
Western blot analysis
Using similar experimental conditions as those used for immunostaining, tissue samples from the ipsilateral ischemic hemisphere as well as of corresponding regions from the contralateral side were prepared for Western blot analysis. Five brains were used for each condition. Samples were treated with 10×lysis buffer (PBS, 1% nonidet P-40, 0.5% sodium deoxycholate, 0.1% SDS, and the protease inhibitors phenylmethylsulfonylfluoride, aprotinin, and sodium orthovandate) and homogenized with the aid of a sonicator (Dremel). To determine protein concentrations, we followed the bicinchonic acid method by using a BCA kit (Pierce).
Immunoblots for tPA and PAI-1 were carried out. Tissue extracts were subjected to electrophoresis in 12.5% and 15% polyacryl-amide gels and transferred to nitrocellulose membranes (Millipore). Membranes were stained with Ponceau Red to ensure equal protein loading. After blocking for 2 hours, the membranes were incubated in primary antibodies against tPA (mouse polyclonal antibody; 1:500 dilution; Chemicon), PAI-1 (rabbit polyclonal antibody, 1:500 dilution; Chemicon), followed by incubation in horseradish peroxidase–conjugated secondary antibody (Amersham Parmarcia Biotech). The gels were developed using the enhanced chemiluminescence system (Amersham Parmarcia Bio-tech). Densitometric measurements were made from the protein bands by using Gelscope (Imageline) and normalized to the total protein from Ponceau red stains.
Statistics, randomization and data analysis
All data were analyzed by investigators blinded to treatment groups. Animals were randomized to experimental groups to minimize bias. Data were expressed as mean±SE. ANOVA followed by Neuman-Keuls was used for studies involving greater than two groups of normally distributed data, Student's t-test for studies involving two groups and nonparametric tests (Mann Whitney) were applied when data were not normally distributed. Fisher exact test was applied to hemorrhage scores and mortality data. Values of p<0.05 were considered significant.
Results
Infarct size, neurological deficit
A total of 37 animals were studied in paradigm 1, and a total of 34 in paradigm 2. An additional eight animals were studied in paradigm 1 to optimize the dosing of rt-PA and cooling paradigms. An additional 10 animals were studied to determine correlation curves for the BBB disruption methods and brain:rectal correlation. Ten additional animals from paradigm 2 in the NH and NN groups were studied to generate tissue for Western blots.
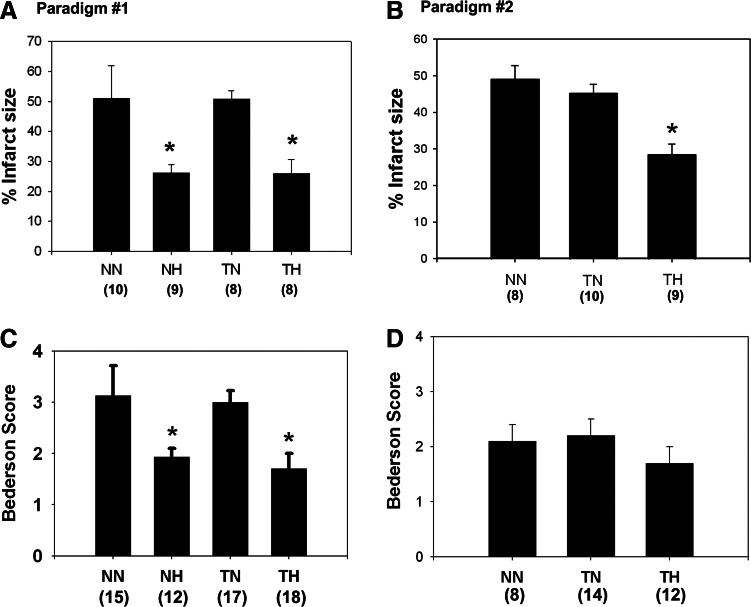
The distribution of animals studied in the various experimental groups is provided in Figure 2C and D (numbers in parenthesis). Of these animals, those that survived to 24 hours post ischemia were included in the analysis in Figure 2A and B. All included mice showed ischemic lesions and neurological deficits. Treatment with hypothermia significantly reduced infarct size compared to tPA-treated and normothermic controls (Fig. 2) in both paradigms. Treatment with rt-PA did not affect infarct size, but this is not surprising considering the model used. Neurological deficits were similarly affected by hypothermia, particularly in paradigm #1, with trends observed in paradigm #2. It should be noted that the higher deficit scores in paradigm #1 reflect the higher mortality. rt-PA treatment and hypothermia did not affect mortality within each paradigm; however, mortality was higher in paradigm 1 among rt-PA treated animals (TN∼53% & TH∼55%) compared to paradigm 2 (∼25% in groups given rt-PA, and 0% in untreated controls) and amongst nontreated animals in paradigm 1 (∼25–30%). These differences in mortality were not statistically different; however, it should be pointed out that we observed more brain hemorrhage in this murine model compared to similar experiences in rat models. The reasons for these differences are not clear, but do point out species differences in their sensitivity to brain ischemia.
FIG. 2.
Hypothermia decreased infarct size and improves neurological deficits. (A, B) Therapeutic cooling led to smaller infarct sizes in both paradigms, regardless of whether animals were give rt-PA. (C, D) Therapeutic cooling led to improved neurological deficit scores amongst animals that received cooling in paradigm #1. Nonsignificant trends were observed in paradigm #2. Numbers of animals included in the data presented are in parentheses. *p<0.05, NN, no rt-PA/normothermia; NH, no rt-PA/hypothermia; TN, rt-PA/normothermia; TH, rt-PA/hypothermia.
Brain hemorrhage
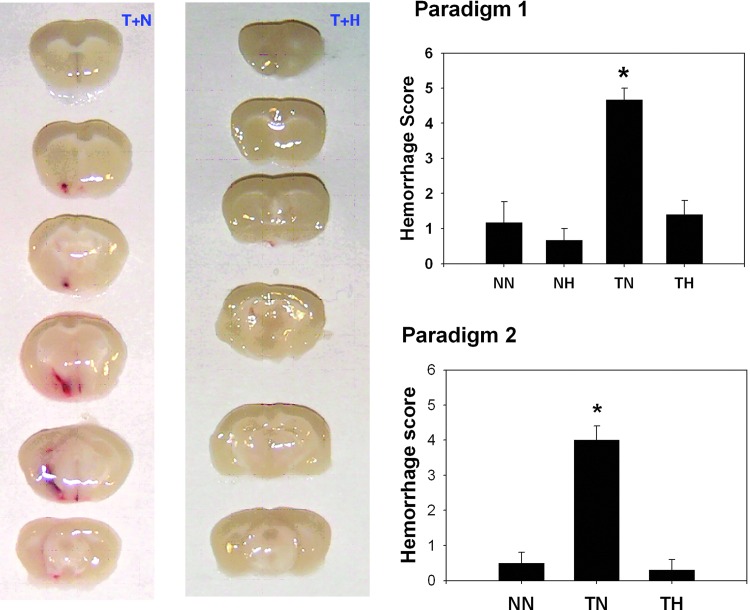
rt-PA treatment at normothermia (TN) increased brain hemorrhage scores compared to no rt-PA at normothermia in both paradigms (Fig. 3). Hypothermia reduced hemorrhage scores to that observed in normothermic controls (NN). There was a trend suggesting that hemorrhage scores were higher amongst rt-PA treated hypothermic animals (TN) compared to hypothermic animals treated with vehicle (NH) in Paradigm 1.
FIG. 3.
Thrombolysis increased cerebral hemorrhage after MCAO, and this was decreased by hypothermia. The left side of the figure shows representative gross brain sections of animals treated with rt-PA at normothermia (T+N) and rt-PA plus hypothermia (T+H) from paradigm #2. Graphs on the right side of the figure show hemorrhage scores for each paradigm. In each paradigm, rt-PA treatment at normothermia increased brain hemorrhage, and this was reduced by cooling. *p<0.05. Color images available online at www.liebertpub.com/ther
BBB disruption
From brains of animals of paradigm 2, BBB disruption was assessed 24 hours following MCAO and reperfusion. Among normothermic animals treated with rt-PA, more BBB disruption as estimated by the areas of EB extravasation were seen compared to those at hypothermia and vehicle treated controls (Fig. 4). Since hypothermia may lessen the extent of BBB disruption by reducing infarct sizes, we corrected for this potential confound by expressing the extent of BBB disruption relative to the infarct sizes and found that the reduced proportionate amount of Evan's Blue dye extravasation still remained at hypothermia. Comparison of the tracing method to the spectrophotometric method showed strong correlation between the two approaches.
FIG. 4.
Hypothermia decreased blood brain barrier (BBB) disruption. The left side of the figure shows representative gross brain sections of animals perfused with Evan's blue dye (EBD) to delineate regions of BBB leakage. Shown are animals treated with rt-PA at normothermia (T+N) and rt-PA plus hypothermia (T+H) from paradigm #2. The graph in the upper right corner shows high correlation between a well established spectrophotometic method of estimating BBB leakage by measuring the amount of EBD in brain lysate (X-axis) and by computing blue areas on brain sections multiplied by the section thickness (Y-axis). The graph in the bottom right corner shows volumes of EBD extravasation normalized to infarct size (paradigm #2). Rt-PA treatment at normothermia (TN) increased the extent of EBD leakage compared to no rt-PA at normothermia (NN). Combined rt-PA at hypothermia (TH) reduced EBD leakage to levels similar to the NN group (*p<0.05). Color images available online at www.liebertpub.com/ther
Endogenous tPA/PAI-1 expression
Using brains from paradigm 2 where no exogenous rt-PA was given (NN and NH groups), immunostains for endogenous tPA showed expression within neuron-like cells of the ipsilateral ischemic cortex. The majority of tPA staining colocalized to neurons, as well as a few astrocytes and ED1 positive cells (Fig. 5A). Similar patterns were observed for PAI-1. Hypothermia reduced the amount of endogenous tPA staining (Fig. 5B). In contrast, immunostains of adjacent sections with PAI-1 failed to show any differences between the two conditions. Western blots demonstrated similar expression patterns (Fig. 5C).
FIG. 5.
Endogenous expression of rt-PA and plasminogen activator inhibitor (PAI-1), effect of hypothermia. (A) Immunostains of a brain from a normothermic animal not given rt-PA (NN), 24 hours after MCAO show that the majority of cells positive for endogenous rt-PA (red) are neurons (NeuN stain, green). (B) Immunostains of endogenous tPA and its endogenous inhibitor PAI-1 (plasminogen activator inhibitor-1) are shown from a normothermic animal (37C, NN group) and hypothermic animal (33C, NH group) after MCAO. Endogenous rt-PA (left panels) is markedly decreased in the hypothermia brain, whereas PAI-1 levels are not affected by cooling (right panels). (C) Western blots of ischemic (ipsi) and contralateral nonischemic (contra) brain samples are shown for hypothermic (33C) and normothermic (37C) animals not given rt-PA. Similar to patterns shown in the immunostains, Western blots show decreased rt-PA protein, but no differences in PAI-1 by hypothermia. Quantification of the blots show decreased endogenous tPA expression with hypothermia (HT) compared to normothermia (NT) in brains exposed to MCAO. No significant differences were observed for PAI-1. *p<0.01.
Discussion
We show here that combination therapy with rt-PA and therapeutic cooling can be used safely. In contrast to the concern that rt-PA plus cooling could exacerbate hemorrhage, BBB disruption and brain hemorrhage was actually reduced by hypothermia. Cooling also led to smaller infarct size and better neurological outcome, which is consistent with previous reports in the literature (van der Worp et al., 2007).
While cooling saw reduction in brain hemorrhage in both paradigms, a better outcome was observed in paradigm 2, where treatments were staggered, rather than given simultaneously. This was a somewhat unexpected observation, as paradigm 1 would have been predicted to lead to a better outcome because the duration of MCAO was shorter (1 hour), and both treatments were begun at the same time. Nevertheless, this paradigm led to a higher mortality rate. Paradigm 2 would have predicted a worse outcome, since the duration of ischemia was longer (3 hours), and rt-PA was started later; however, cooling was begun sooner and did not overlap with rt-PA treatment. Since lower temperature is known to inhibit the endogenous hemostatic system (Martini, 2007; Meunier et al., 2012), it is possible that simultaneous rt-PA treatment at lower temperature led to an increased bleeding tendency. Animals that died early were noted to have early brain or wound site hemorrhage, which was not captured in the brain hemorrhage analysis which was performed at 24 hours. Hypothermia is known to cause coagulopathies, and this has been documented even in moderate cooling (Martini, 2007). Thus, staggering treatments so that rt-PA is given at normothermia may improve safety of rt-PA. However, we did not measure coagulation parameters. Thus, we can only speculate whether cooling had any effect on coagulation and fibrinolysis. It is also possible that these differences are due to the fact that cooling was started sooner in Paradigm #2. Early cooling is associated with the chance of a better outcome (van der Worp et al., 2007), and would also leave more viable tissue that would benefit from recanalization. Independent of rt-PA treatment, our data showed a beneficial effect of cooling in terms of reducing lesion size and improving neurological scores. Our data also indicate that rt-PA treatment led to increased brain hemorrhage and BBB disruption compared to no rt-PA treatment. Further, hypothermia could reduce brain hemorrhage and BBB disruption to levels seen in normothermic animals not treated with rt-PA.
Laboratory studies of hypothermia in models of primary brain hemorrhage failed to show the same robust neuroprotective effects compared to cerebral ischemia (MacLellan et al., 2002, 2004, 2006, 2009). Reasons for this have been attributed to the development of hypertension in the primary brain hemorrhage models, but could also be a result of coagulopathies that can develop during even moderate cooling (Martini, 2007; Meunier et al., 2012). Hypothermia to 32C led to reduced fibrinogen and impaired thrombin generation (Martini, 2007). While therapeutic cooling in mild to moderate ranges was not thought to increase brain hemorrhage risk (Resnick et al., 1994), spontaneous brain hemorrhage has complicated this intervention in a case of cardiac arrest (Cho et al., 2013).
It is not yet clear from previous studies examining the feasibility of combination treatment with thrombolysis and therapeutic cooling whether hypothermia and rt-PA is synergistic (Bederson et al., 1986; Meden et al., 1994; Tang et al., 2009). Using models of embolic stroke, two different studies tested the neuroprotective and thrombolytic efficacy of combination therapy. Using a rat embolic model, Meden et al. (1994) showed that, at 2 hours after embolization, rt-PA could significantly reduce infarct volume. Three-hour hypothermia (32°C) begun immediately after embolization reduced the infarct volume even more. However, the combination of hypothermia and rt-PA treatment did not show further protection. Interestingly, angiograms showed that improved recanalization was best seen in hyperthermic animals, but there was no difference in recanalization between normothermia and hypothermia, where infarct size was actually increased. In another similar study of thromboembolic stroke (Bederson et al., 1986), rt-PA was given 1 or 3 hours after embolization, and cooling (33°C) was started 1 hour after embolization and maintained for 4 hours. Animals receiving rt-PA all had better recovery of cerebral perfusion. However, animals in all hypothermic groups had less injury, regardless of whether they received rt-PA. Thus, neither study truly demonstrated the superiority of combination of rt-PA and hypothermia over either therapy alone. Some clinical studies of combination rt-PA and therapeutic cooling have been reported and suggest that this approach is both feasible and safe (Martin-Schild et al., 2008; Hemmen et al., 2010), but efficacy data from prospective trials are not yet available.
Reasons for this lack of synergistic efficacy are still not clear, but it is possible that tPA itself has neurotoxic properties (Wang et al., 1998). While early experiments in an animal model of embolic stroke (Zivin et al., 1985) and clinical trials in stroke patients (NINDS, 1995; Lees et al., 2010) clearly demonstrated that rt-PA as a thrombolytic reduced the extent of the neurologic damage, when given a few hours after the onset of cerebral ischemia. However, a few laboratory studies have demonstrated that excessive endogenous tPA within the brain actually promotes neuronal death (Wang et al., 1998; Nagai et al., 1999; Yepes et al., 2000; Cinelli et al., 2001). In pathologic situations, such as cerebral ischemia, excessive increases in vascular permeability lead to an abnormal opening of the BBB with the passage of potentially harmful substances from the blood into the brain and the development of vasogenic edema.
Other experimental studies support the neurotoxic effects of both endogenous tPA (Yepes et al., 2009) and exogenously administered rt-PA (Harston et al., 2010). These effects may be due to enzymatic degradation of the basal lamina and subsequently, a damaged extracellular matrix interaction leading to cell death (Chen and Strickland, 1997). Thus, rt-PA, when contained in the intravascular space, has the potential to improve outcome from stroke due to its fibrinolytic properties, but when allowed to enter the brain through a disrupted BBB, can cause and/or worsen brain cell damage (Kaur et al., 2004).
Although one study has suggested that tPA deficiency increases cerebrovascular fibrin deposition with subsequent worsening of brain injury following transient MCAO (Tabrizi et al., 1999), several animals models have demonstrated that both genetic deficiency of tPA (Wang et al., 1998; Nagai et al., 1999) and its inhibition with the tPA inhibitor neuroserpin (Yepes et al., 2000) are associated with a significant increase in neuronal survival and a decrease in infarct volume following either transient or permanent MCAO (Cinelli et al., 2001).
The reduction of plasminogen activity either by plasminogen gene inactivation or treatment with α2-antiplasmin resulted in a significant decrease in BBB permeability (Yepes et al., 2003). These same investigators also demonstrated that as early as 1 hour after MCAO, there is a robust surge in tPA activity adjacent to the blood vessels of the necrotic core, followed by an increase in vascular permeability in the same area 5 hours later. These results demonstrate that in cerebral ischemia, there is a link between endogenous perivascular tPA and increases in vascular permeability. In our study, we found that treatment with rt-PA increased BBB permeability and hemorrhage compared to animals not given rt-PA. Therapeutic cooling reduced both of these measures in the setting of rt-PA treatment. When we studied the effect of cooling on endogenous tPA and its inhibitor PAI-1 in the brain, we also found that cooling decreased tPA expression but did not affect PAI-1 levels. This suggests that an additional mechanism of BBB protection and brain hemorrhage reduction might involve suppression of endogenous tPA.
BBB disruption and brain hemorrhage following stroke has also been linked to matrix metalloproteinases (MMP) activation (Rosenberg et al., 1996). MMPs can promote proteolytic degradation of the vascular basement membrane with subsequent BBB breakdown and vasogenic edema, and studies have shown that inhibition or deficiency of several MMPs improve outcome from stroke and reduce BBB disruption (Asahi et al., 2000; Lapchak et al., 2000). A link between tPA, MMPs, and BBB permeability following MCAO was suggested by the observation that thrombolytic treatment with rt-PA in an animal model of embolic stroke is associated with significant increases in MMP-9 expression, cerebrovascular permeability, and vasogenic edema (Aoki et al., 2002; Wang et al., 2003; Zhao et al., 2007). Consistent with this, we and others have shown that therapeutic hypothermia leads to the suppression of MMPs (Wagner et al., 2003; Hamann et al., 2004; Lee et al., 2005, 2009; Truettner et al., 2005; Nagel et al., 2008).
It also deserves mention that we utilized a commonly studied filament model of experimental stroke, rather than an embolic model. While a limitation of this study is that we cannot comment on the thrombolytic efficacy of rt-PA alone or in combination, a disadvantage of the embolic model is that reproducibility is low and the timing of reperfusion cannot be controlled. Thus, in this study, we can make useful conclusions regarding safety measures, such as the extent of BBB disruption and brain hemorrhage as well as mortality. However, our model cannot be used to test the thrombolytic efficacy of rt-PA treatment. Yet, we were still able to detect differences in lesion volume when cooling was applied and suggests the robustness of this as a neuroprotectant.
In sum, we show that combination therapy with rt-PA and therapeutic hypothermia is safe, and does not increase the risk of rt-PA related cerebral complications. These data may be useful in the design of future clinical trials. Combination therapy of hypothermia to expand the therapeutic window of thrombolytic use should certainly be explored. Treatment with rt-PA or clot retrieving devices cannot be easily or safely implemented in the field. Thus, hypothermia could be initiated in the field while patients are transported to centers for therapeutic recanalization.
Acknowledgments
This work was supported by grants from National Institutes of Health (NS40516) and Veteran's Merit Award to M.Y. Grants to M.Y. were administered by Northern California Institute for Research and Education, and supported by resources of Veterans Affairs Medical Center, San Francisco, California.
Disclosure Statement
All authors declare that no competing financial interests exist.
References
- Aoki T. Sumii T. Mori T. Wang X. Lo EH. Blood-brain barrier disruption and matrix metalloproteinase-9 expression during reperfusion injury: mechanical versus embolic focal ischemia in spontaneously hypertensive rats. Stroke. 2002;33:2711–2717. doi: 10.1161/01.str.0000033932.34467.97. [DOI] [PubMed] [Google Scholar]
- Asahi M. Asahi K. Jung JC. del Zoppo GJ. Fini ME. Lo EH. Role for matrix metalloproteinase 9 after focal cerebral ischemia: effects of gene knockout and enzyme inhibition with BB-94. J Cereb Blood Flow Metab. 2000;20:1681–1689. doi: 10.1097/00004647-200012000-00007. [DOI] [PubMed] [Google Scholar]
- Bederson JB. Pitts LH. Tsuji M. Nishimura MC. Davis RL. Bartkowski H. Rat middle cerebral artery occlusion: evaluation of the model and development of a neurologic examination. Stroke. 1986;17:472–476. doi: 10.1161/01.str.17.3.472. [DOI] [PubMed] [Google Scholar]
- Belayev L. Busto R. Zhao W. Ginsberg MD. Quantitative evaluation of blood-brain barrier permeability following middle cerebral artery occlusion in rats. Brain Res. 1996;739:88–96. doi: 10.1016/s0006-8993(96)00815-3. [DOI] [PubMed] [Google Scholar]
- Bernard SA. Gray TW. Buist MD. Jones BM. Silvester W. Gutteridge G. Smith K. Treatment of comatose survivors of out-of-hospital cardiac arrest with induced hypothermia. N Engl J Med. 2002;346:557–563. doi: 10.1056/NEJMoa003289. [DOI] [PubMed] [Google Scholar]
- Brinker G. Pillekamp F. Hossmann KA. Brain hemorrhages after rt-PA treatment of embolic stroke in spontaneously hypertensive rats. Neuroreport. 1999;10:1943–1946. doi: 10.1097/00001756-199906230-00027. [DOI] [PubMed] [Google Scholar]
- Chen ZL. Strickland S. Neuronal death in the hippocampus is promoted by plasmin-catalyzed degradation of laminin. Cell. 1997;91:917–925. doi: 10.1016/s0092-8674(00)80483-3. [DOI] [PubMed] [Google Scholar]
- Cho HJ. Kyong YY. Oh YM. Choi SM. Choi KH. Oh JS. Therapeutic hypothermia complicated by spontaneous brain stem hemorrhage. Am J Emerg Med. 2013;31:266 e1–3. doi: 10.1016/j.ajem.2012.04.017. [DOI] [PubMed] [Google Scholar]
- Chopp M. Zhang RL. Zhang ZG. Jiang Q. The clot thickens—thrombolysis and combination therapies. Acta Neurochir Suppl. 1999;73:67–71. doi: 10.1007/978-3-7091-6391-7_11. [DOI] [PubMed] [Google Scholar]
- Cinelli P. Madani R. Tsuzuki N. Vallet P. Arras M. Zhao CN. Osterwalder T. Rulicke T. Sonderegger P. Neuroserpin, a neuroprotective factor in focal ischemic stroke. Mol Cell Neurosci. 2001;18:443–457. doi: 10.1006/mcne.2001.1028. [DOI] [PubMed] [Google Scholar]
- HACA. Mild therapeutic hypothermia to improve the neurologic outcome after cardiac arrest. N Engl J Med. 2002;346:549–556. doi: 10.1056/NEJMoa012689. [DOI] [PubMed] [Google Scholar]
- Hacke W. Kaste M. Bluhmki E. Brozman M. Davalos A. Guidetti D. Larrue V. Lees KR. Medeghri Z. Machnig T. Schneider D. von Kummer R. Wahlgren N. Toni D. Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med. 2008;359:1317–1329. doi: 10.1056/NEJMoa0804656. [DOI] [PubMed] [Google Scholar]
- Hamann GF. Burggraf D. Martens HK. Liebetrau M. Jager G. Wunderlich N. DeGeorgia M. Krieger DW. Mild to moderate hypothermia prevents microvascular basal lamina antigen loss in experimental focal cerebral ischemia. Stroke. 2004;35:764–769. doi: 10.1161/01.STR.0000116866.60794.21. [DOI] [PubMed] [Google Scholar]
- Harston GW. Sutherland BA. Kennedy J. Buchan AM. The contribution of L-arginine to the neurotoxicity of recombinant tissue plasminogen activator following cerebral ischemia: a review of rtPA neurotoxicity. J Cereb Blood Flow Metab. 2010;30:1804–1816. doi: 10.1038/jcbfm.2010.149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hemmen TM. Raman R. Guluma KZ. Meyer BC. Gomes JA. Cruz-Flores S. Wijman CA. Rapp KS. Grotta JC. Lyden PD. Intravenous thrombolysis plus hypothermia for acute treatment of ischemic stroke (ICTuS-L): final results. Stroke. 2010;41:2265–2270. doi: 10.1161/STROKEAHA.110.592295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur J. Zhao Z. Klein GM. Lo EH. Buchan AM. The neurotoxicity of tissue plasminogen activator? J Cereb Blood Flow Metab. 2004;24:945–963. doi: 10.1097/01.WCB.0000137868.50767.E8. [DOI] [PubMed] [Google Scholar]
- Korninger C. Collen D. Studies on the specific fibrinolytic effect of human extrinsic (tissue-type) plasminogen activator in human blood and in various animal species in vitro. Thromb Haemost. 1981;46:561–565. [PubMed] [Google Scholar]
- Lapchak PA. Chapman DF. Zivin JA. Metalloproteinase inhibition reduces thrombolytic (tissue plasminogen activator)-induced hemorrhage after thromboembolic stroke. Stroke. 2000;31:3034–3040. doi: 10.1161/01.str.31.12.3034. [DOI] [PubMed] [Google Scholar]
- Lee JE. Yoon YJ. Moseley ME. Yenari MA. Reduction in levels of matrix metalloproteinases and increased expression of tissue inhibitor of metalloproteinase-2 in response to mild hypothermia therapy in experimental stroke. J Neurosurg. 2005;103:289–297. doi: 10.3171/jns.2005.103.2.0289. [DOI] [PubMed] [Google Scholar]
- Lee KM. Jang JH. Park JS. Kim DS. Han HS. Effect of mild hypothermia on blood brain barrier disruption induced by oleic acid in rats. Genes Genomics. 2009;31:89–98. [Google Scholar]
- Lees KR. Bluhmki E. von Kummer R. Brott TG. Toni D. Grotta JC. Albers GW. Kaste M. Marler JR. Hamilton SA. Tilley BC. Davis SM. Donnan GA. Hacke W. Allen K. Mau J. Meier D. del Zoppo G. De Silva DA. Butcher KS. Parsons MW. Barber PA. Levi C. Bladin C. Byrnes G. Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet. 2010;375:1695–1703. doi: 10.1016/S0140-6736(10)60491-6. [DOI] [PubMed] [Google Scholar]
- Liu L. Yenari MA. Clinical application of therapeutic hypothermia in stroke. Neurol Res. 2009;31:331–335. doi: 10.1179/174313209X444099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacLellan C. Shuaib A. Colbourne F. Failure of delayed and prolonged hypothermia to favorably affect hemorrhagic stroke in rats. Brain Res. 2002;958:192–200. doi: 10.1016/s0006-8993(02)03702-2. [DOI] [PubMed] [Google Scholar]
- MacLellan CL. Clark DL. Silasi G. Colbourne F. Use of prolonged hypothermia to treat ischemic and hemorrhagic stroke. J Neurotrauma. 2009;26:313–323. doi: 10.1089/neu.2008.0580. [DOI] [PubMed] [Google Scholar]
- MacLellan CL. Davies LM. Fingas MS. Colbourne F. The influence of hypothermia on outcome after intracerebral hemorrhage in rats. Stroke. 2006;37:1266–1270. doi: 10.1161/01.STR.0000217268.81963.78. [DOI] [PubMed] [Google Scholar]
- MacLellan CL. Girgis J. Colbourne F. Delayed onset of prolonged hypothermia improves outcome after intracerebral hemorrhage in rats. J Cereb Blood Flow Metab. 2004;24:432–440. doi: 10.1097/00004647-200404000-00008. [DOI] [PubMed] [Google Scholar]
- Martin-Schild S. Hallevi H. Shaltoni H. Barreto AD. Gonzales NR. Aronowski J. Savitz SI. Grotta JC. Combined neuroprotective modalities coupled with thrombolysis in acute ischemic stroke: a pilot study of caffeinol and mild hypothermia. J Stroke Cerebrovasc Dis. 2008;18:86–96. doi: 10.1016/j.jstrokecerebrovasdis.2008.09.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martini WZ. The effects of hypothermia on fibrinogen metabolism and coagulation function in swine. Metabolism. 2007;56:214–221. doi: 10.1016/j.metabol.2006.09.015. [DOI] [PubMed] [Google Scholar]
- Meden P. Overgaard K. Pedersen H. Boysen G. Effect of hypothermia and delayed thrombolysis in a rat embolic stroke model. Acta Neurol Scand. 1994;90:91–98. doi: 10.1111/j.1600-0404.1994.tb02686.x. [DOI] [PubMed] [Google Scholar]
- Meng W. Wang X. Asahi M. Kano T. Asahi K. Ackerman RH. Lo EH. Effects of tissue type plasminogen activator in embolic versus mechanical models of focal cerebral ischemia in rats. J Cereb Blood Flow Metab. 1999;19:1316–1321. doi: 10.1097/00004647-199912000-00004. [DOI] [PubMed] [Google Scholar]
- Meunier JM. Chang W-TW. Bluett B. Wenker E. Lindsell CJ. Shaw GJ. Temperature Affects Thrombolytic Efficacy Using rt-PA and Eptifibatide, an In Vitro Study. Ther Hypothermia Temp Manag. 2012;2:112–118. doi: 10.1089/ther.2012.0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagai N. Urano T. Endo A. Takahashi H. Takada Y. Takada A. Neuronal degeneration and a decrease in laminin-like immunoreactivity is associated with elevated tissue-type plasminogen activator in the rat hippocampus after kainic acid injection. Neurosci Res. 1999;33:147–154. doi: 10.1016/s0168-0102(98)00125-4. [DOI] [PubMed] [Google Scholar]
- Nagel S. Su Y. Horstmann S. Heiland S. Gardner H. Koziol J. Martinez-Torres FJ. Wagner S. Minocycline and hypothermia for reperfusion injury after focal cerebral ischemia in the rat: effects on BBB breakdown and MMP expression in the acute and subacute phase. Brain Res. 2008;1188:198–206. doi: 10.1016/j.brainres.2007.10.052. [DOI] [PubMed] [Google Scholar]
- NINDS. Tissue plasminogen activator for acute ischemic stroke. The National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group. N Engl J Med. 1995;333:1581–1587. doi: 10.1056/NEJM199512143332401. [DOI] [PubMed] [Google Scholar]
- Resnick DK. Marion DW. Darby JM. The effect of hypothermia on the incidence of delayed traumatic intracerebral hemorrhage. Neurosurgery. 1994;34:252–255. doi: 10.1227/00006123-199402000-00007. discussion 255–256. [DOI] [PubMed] [Google Scholar]
- Rosenberg GA. Navratil M. Barone F. Feuerstein G. Proteolytic cascade enzymes increase in focal cerebral ischemia in rat. J Cereb Blood Flow Metab. 1996;16:360–366. doi: 10.1097/00004647-199605000-00002. [DOI] [PubMed] [Google Scholar]
- Swanson RA. Morton MT. Tsao-Wu G. Savalos RA. Davidson C. Sharp FR. A semiautomated method for measuring brain infarct volume. J Cereb Blood Flow Metab. 1990;10:290–293. doi: 10.1038/jcbfm.1990.47. [DOI] [PubMed] [Google Scholar]
- Tabrizi P. Wang L. Seeds N. McComb JG. Yamada S. Griffin JH. Carmeliet P. Weiss MH. Zlokovic BV. Tissue plasminogen activator (tPA) deficiency exacerbates cerebrovascular fibrin deposition and brain injury in a murine stroke model: studies in tPA-deficient mice and wild-type mice on a matched genetic background. Arterioscler Thromb Vasc Biol. 1999;19:2801–2806. doi: 10.1161/01.atv.19.11.2801. [DOI] [PubMed] [Google Scholar]
- Tang XN. Liu L. Yenari MA. Combination therapy with hypothermia for treatment of cerebral ischemia. J Neurotrauma. 2009;26:325–331. doi: 10.1089/neu.2008.0594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Truettner JS. Alonso OF. Dalton Dietrich W. Influence of therapeutic hypothermia on matrix metalloproteinase activity after traumatic brain injury in rats. J Cereb Blood Flow Metab. 2005;25:1505–1516. doi: 10.1038/sj.jcbfm.9600150. [DOI] [PubMed] [Google Scholar]
- van der Worp HB. Sena ES. Donnan GA. Howells DW. Macleod MR. Hypothermia in animal models of acute ischaemic stroke: a systematic review and meta-analysis. Brain. 2007;130:3063–3074. doi: 10.1093/brain/awm083. [DOI] [PubMed] [Google Scholar]
- Wagner S. Nagel S. Kluge B. Schwab S. Heiland S. Koziol J. Gardner H. Hacke W. Topographically graded postischemic presence of metalloproteinases is inhibited by hypothermia. Brain Res. 2003;984:63–75. doi: 10.1016/s0006-8993(03)03088-9. [DOI] [PubMed] [Google Scholar]
- Wang X. Lee SR. Arai K. Tsuji K. Rebeck GW. Lo EH. Lipoprotein receptor-mediated induction of matrix metalloproteinase by tissue plasminogen activator. Nat Med. 2003;9:1313–317. doi: 10.1038/nm926. [DOI] [PubMed] [Google Scholar]
- Wang YF. Tsirka SE. Strickland S. Stieg PE. Soriano SG. Lipton SA. Tissue plasminogen activator (tPA) increases neuronal damage after focal cerebral ischemia in wild-type and tPA-deficient mice. Nat Med. 1998;4:228–231. doi: 10.1038/nm0298-228. [DOI] [PubMed] [Google Scholar]
- Yenari MA. Beaulieu C. Steinberg GK. Moseley ME. Diffusion-weighted magnetic resonance imaging characteristics of hemorrhagic transformation in experimental embolic stroke. J Neuroimaging. 1997a;7:227–231. doi: 10.1111/jon199774227. [DOI] [PubMed] [Google Scholar]
- Yenari MA. de Crespigny A. Palmer JT. Roberts S. Schrier SL. Albers GW. Moseley ME. Steinberg GK. Improved perfusion with rt-PA and hirulog in a rabbit model of embolic stroke. J Cereb Blood Flow Metab. 1997b;17:401–411. doi: 10.1097/00004647-199704000-00005. [DOI] [PubMed] [Google Scholar]
- Yenari MA. Han HS. Neuroprotective mechanisms of hypothermia in brain ischaemia. Nat Rev Neurosci. 2012;13:267–278. doi: 10.1038/nrn3174. [DOI] [PubMed] [Google Scholar]
- Yenari MA. Onley D. Hedehus M. deCrespigny A. Sun GH. Moseley ME. Steinberg GK. Diffusion- and perfusion-weighted magnetic resonance imaging of focal cerebral ischemia and cortical spreading depression under conditions of mild hypothermia. Brain Res. 2000;885:208–219. doi: 10.1016/s0006-8993(00)02942-5. [DOI] [PubMed] [Google Scholar]
- Yenari MA. Palmer JT. Bracci PM. Steinberg GK. Thrombolysis with tissue plasminogen activator (tPA) is temperature dependent. Thromb Res. 1995;77:475–481. doi: 10.1016/0049-3848(95)93883-2. [DOI] [PubMed] [Google Scholar]
- Yenari MA. Xu L. Tang XN. Qiao Y. Giffard RG. Microglia potentiate damage to blood-brain barrier constituents: improvement by minocycline in vivo and in vitro. Stroke. 2006;37:1087–1093. doi: 10.1161/01.STR.0000206281.77178.ac. [DOI] [PubMed] [Google Scholar]
- Yepes M. Roussel BD. Ali C. Vivien D. Tissue-type plasminogen activator in the ischemic brain: more than a thrombolytic. Trends Neurosci. 2009;32:48–55. doi: 10.1016/j.tins.2008.09.006. [DOI] [PubMed] [Google Scholar]
- Yepes M. Sandkvist M. Moore EG. Bugge TH. Strickland DK. Lawrence DA. Tissue-type plasminogen activator induces opening of the blood-brain barrier via the LDL receptor-related protein. J Clin Invest. 2003;112:1533–1540. doi: 10.1172/JCI19212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yepes M. Sandkvist M. Wong MK. Coleman TA. Smith E. Cohan SL. Lawrence DA. Neuroserpin reduces cerebral infarct volume and protects neurons from ischemia-induced apoptosis. Blood. 2000;96:569–576. [PubMed] [Google Scholar]
- Zhao BQ. Tejima E. Lo EH. Neurovascular proteases in brain injury, hemorrhage and remodeling after stroke. Stroke. 2007;38:748–752. doi: 10.1161/01.STR.0000253500.32979.d1. [DOI] [PubMed] [Google Scholar]
- Zheng Z. Kim JY. Ma H. Lee JE. Yenari MA. Anti-inflammatory effects of the 70 kDa heat shock protein in experimental stroke. J Cereb Blood Flow Metab. 2008;28:53–63. doi: 10.1038/sj.jcbfm.9600502. [DOI] [PubMed] [Google Scholar]
- Zivin JA. Fisher M. DeGirolami U. Hemenway CC. Stashak JA. Tissue plasminogen activator reduces neurological damage after cerebral embolism. Science. 1985;230:1289–1292. doi: 10.1126/science.3934754. [DOI] [PubMed] [Google Scholar]