Abstract
AR-67 (7-t-butyldimethylsilyl-10-hydroxycamptothecin) is a lipophilic camptothecin analog, currently under early stage clinical trials. Transporters are known to have an impact on the disposition of camptothecins and on the response to chemotherapeutics in general due to their expression in tumor tissues. Therefore, we investigated the interplay between the breast cancer resistance protein (BCRP), multidrug resistant protein 1 (MDR1), and organic anion-transporting polypeptide (OATP) 1B1/1B3 transporters and AR-67 and their impact on the toxicity profile of AR-67. Using cell lines expressing the aforementioned transporters, we showed that the lipophilic AR-67 lactone form is a substrate for efflux transporters BCRP and MDR1. Additionally, OATP1B1 and OATP1B3 facilitated the uptake of AR-67 carboxylate in SLCO1B1- and SLCO1B3-transfected cell systems compared with the mock-transfected ones. Notably, both BCRP and MDR1 conferred resistance to AR-67 lactone. Prompted by recent studies showing increased OATP1B3 expression in certain cancer types, we investigated the effect of OATP1B3 expression on cell viability after exposure to AR-67 carboxylate. OATP1B3-expressing cells had increased carboxylate uptake as compared with mock-transfected cells but were not sensitized because the intracellular amount of lactone was 50-fold higher than that of carboxylate and comparable between OATP1B3-expressing and OATP1B3-nonexpressing cells. In conclusion, BCRP- and MDR1-mediated efflux of AR-67 lactone confers resistance to AR-67, but OATP1B3-mediated uptake of the AR-67 carboxylate does not sensitize OATP1B3-expressing tumor cells.
Introduction
AR-67 (7-t-butyldimethylsilyl-10-hydroxycamptothecin, also known as DB-67) (Fig. 1) is a third-generation camptothecin analog that belongs to the class of 7-silylcamptothecins (Bom et al., 2000; Curran et al., 2000). Similar to other camptothecins, AR-67 undergoes pH-dependent but reversible hydrolysis of the lipophilic lactone to the hydrophilic carboxylate (Bom et al., 2000). Although both lactone and carboxylate forms interact with DNA (Staker et al., 2002), they have different transport characteristics. The lactone passively diffuses into the cell and is considered the pharmacologically active form. In contrast, the negatively charged carboxylate requires transporter-mediated uptake, and it is often considered an inactive form. Preclinical studies have demonstrated the high lipophilicity and an apparent blood stability of the lactone form of AR-67 compared with the camptothecins approved by the U.S. Food and Drug Administration (Bom et al., 2001).
Fig. 1.
pH-dependent interconversion between the lactone and carboxylate form of the camptothecin analog AR-67.
A common link between drug disposition and drug efficacy are transporter proteins, which could play a pivotal role in both the disposition and efficacy or toxicity of camptothecin analogs. As AR-67 exists in equilibrium between the hydrophobic lactone and hydrophilic carboxylate forms, both influx and efflux transporters could potentially play roles in both metabolic clearance and tumor sensitivity. Intracellular drug concentration will be influenced by the balance between cellular efflux, potentially resulting in resistance, and cellular uptake, potentially resulting in sensitivity. Metabolic clearance, on the other hand, may result from vectorial transport, where both influx and efflux transporters contribute to clearance in the same direction.
The effect of transporters on the pharmacokinetic and pharmacodynamic profile of topotecan and irinotecan, the camptothecins approved by the Food and Drug Administration, has been demonstrated in previous studies. Topotecan and the active irinotecan metabolite SN-38 (7-ethyl-10-hydroxy-camptothecin) have been identified as breast cancer resistance protein (BCRP) substrates (Nakatomi et al., 2001; de Vries et al., 2007) whereas transport mediated by multidrug resistant protein 1 (MDR1) has been reported for topotecan and irinotecan (Luo et al., 2002; de Vries et al., 2007). Notably, expression of BCRP in established cancer cell lines and tumor biopsy samples has been associated with resistance to camptothecins (Kawabata et al., 2001; Candeil et al., 2004). Among the uptake transporters, organic anion-transporting polypeptide (OATP) 1B1 has been implicated in the transport of irinotecan and SN-38, which has also been identified as an OATP1B3 substrate (Nozawa et al., 2005; Yamaguchi et al., 2008). However, little is known about the potential interactions between AR-67 and transporters or the implications of these interactions on the antitumor activity of AR-67 and its pharmacokinetic profile.
In this study, we explored the interaction of AR-67 with BCRP and MDR1 and with OATP1B3 and OATP1B1. First, we determined whether expression of the efflux transporters BCRP and MDR1 would have an impact on the cytotoxic profile of the lipophilic AR-67 lactone in vitro. Additionally, we examined the effect of OATP1B3 expression on the intracellular amounts of AR-67 lactone and carboxylate. Based on recent studies reporting increased expression of OATP1B3 in tumor tissues (Muto et al., 2007; Pressler et al., 2011), we tested whether increased intracellular AR-67 uptake, facilitated by OATP1B3, would potentiate the antitumor activity of AR-67 in vitro. To address these questions, we used established cancer cell lines that expressed functional forms of the BCRP, MDR1, OATP1B3, and OATP1B1 transporters.
Materials and Methods
Cell Lines and Reagents.
Madin-Darby canine kidney II (MDCKII) cells were obtained from the European Collection of Cell Cultures (Porton Down, Wiltshire, UK). Cells were mock transfected with pcDNA3.1 vector or with pcDNA3.1-ATP-binding cassette (ABC) G2 (Wang et al., 2012). Human ovarian cancer cell line 8 (OVCAR-8) and its derivative cell line expressing human MDR1 (NCI/ADR-RES) were obtained from the U.S. National Cancer Institute (NCI) Developmental Therapeutics Program. HeLa (cervical adenocarcinoma) cells were stably transfected with OATP1B3 cDNA and RKO (colon carcinoma) cells were stably transfected with OATP1B1 cDNA inserted in the multiple cloning site of the pIRESneo2 vector (Clontech, Mountain View, CA) (Monks et al., 2007; Daily et al., 2010). MDCKII cells were cultured in minimum essential media (MEM) supplemented with 5% fetal bovine serum (FBS) and geneticin (G418, 800 μg/ml), and the OVCAR-8 and NCI/ADR-RES cells were cultured in Dulbecco’s modified Eagle medium supplemented with 10% FBS. The HeLa and RKO cell lines were grown in Dulbecco’s modified Eagle medium supplemented with 10% FBS and geneticin (G418, 800 μg/ml). Additionally, nonessential amino acids (1%) were added to the RKO growth media. Penicillin (100 U/ml) and streptomycin (100 μg/ml) were added in the media. All of the aforementioned materials were supplied by GIBCO/Invitrogen Corporation (Carlsbad, CA). Transporter expression was validated by Western blot analysis, and transporter function was verified with prototypical substrates (found in the Supplemental Materials and Methods).
Preparation of Drug Stock and Working Solutions.
AR-67 (DB-67; Novartis, New York, NY) was solubilized in dimethylsulfoxide at a concentration of 1 mg/ml (AR-67 lactone stock solution) and stored at −80°C. AR-67 carboxylate working solutions were prepared by dilution of one volume of lactone stock solution with 9 volumes of 0.005 N NaOH and were allowed to convert to carboxylate overnight at 4°C (AR-67 carboxylate stock solution) (Horn et al., 2006). Working solutions were stored at −80°C after their preparation and at 4°C during the experimental procedure.
Intracellular Amount of AR-67 in BCRP and MDR1 Expressing Cell Lines.
To examine the effect of ABC efflux transporters BCRP and MDR1 on the intracellular amount of AR-67, cells were seeded in six-well plates. The next day, the medium was replaced, and the cells were incubated in serum-free medium (Opti-MEM) with 0.125, 0.25, 0.5, 0.75, and 1 µM AR-67 lactone for 5 minutes at 37°C. The incubation time was selected to ensure linearity in the transport process. At the end of the incubation, the medium was aspirated, and cells were washed twice with Opti-MEM (4°C). The cells were lysed with 200 µl of 0.5 N NaOH and placed on a rocker (4°C) for 10 minutes. Extraction and quantification of AR-67 was performed as indicated in a high-performance liquid chromatography (HPLC) fluorescence method under conditions that ensure system suitability (Horn et al., 2006). Protein was quantified using the BCA Protein Assay Kit (Pierce/Thermo Fisher Scientific, Rockford, IL). In inhibition studies, cells were preincubated with 5 µM GF120918 (N-4-[2-(1,2,3,4-tetrahydro-6,7-dimethoxy-2-isoquinolinyl)ethyl]-phenyl)-9,10-dihydro-5-methoxy-9-oxo-4-acridine carboxamide hydrochloride) (de Bruin et al., 1999; Evers et al., 2000) for 1 hour before incubation with 1 µM AR-67 lactone (for 5 minutes).
Transepithelial Flux of AR-67.
MDCKII-pcDNA and MDCKII-BCRP cells were grown on Corning Transwell 3414 membrane inserts (3.0-μm pore size, 24-mm diameter; Corning Glassworks, Corning, NY). When the transepithelial electrical resistance exceeded 200 Ω·cm2s in both cell lines, the medium was aspirated and replaced with 1.8 ml of serum-free Opti-MEM with or without GF120918 (5 µM) on both the apical and basolateral sides. Cells were placed at 37°C in a humidified 5% CO2 incubator for 1 hour. Afterward, 0.2 ml of 50 µM AR-67 lactone was added to the donor side for a final concentration of 5 µM, and 0.2 ml of medium was added to the receiver side. Cells were then placed on a rocker and incubated at 37°C and 5% CO2. The amount of AR-67 was quantified in 50 µl samples using the HPLC method described previously. The apparent permeability and efflux were calculated as previously described elsewhere (Kalvass and Pollack, 2007).
Intracellular Uptake of AR-67 Carboxylate.
The effect of the expression of OATP1B3 and OATP1B1 on the intracellular uptake of AR-67 was studied using the HeLa-pIRES/OATP1B3 and RKO-pIRES/OATP1B1 cell lines using serum-free medium (Opti-MEM). To evaluate the dose-dependent OATP-mediated uptake of AR-67, OATP-transfected cell lines were incubated with 0.5, 1.0, 1.5, 2.0, and 3.0 µM AR-67 carboxylate for 1 minute. The incubation time was selected to ensure linearity in the transport process as indicated by preliminary studies. The pH of the carboxylate-containing media was 7.2. In inhibition studies, Hela-pIRES/OATP1B3 and RKO-pIRES/OATP1B1 cells were incubated with 1 µM AR-67 carboxylate for 5 and 10 minutes, respectively. The inhibition of the transporter-mediated uptake was studied by preincubation with bromosulfophthalein (BSP) (50 µM) (Seithel et al., 2007; Kalliokoski and Niemi, 2009) for 10 minutes, keeping the concentration of BSP constant during substrate incubation.
At the end of the incubation period, the cells were washed twice with Opti-MEM (4°C) before being lysed; AR-67, calculated as the sum of the two forms, was extracted from the cell lysate and quantified as described earlier (Horn et al., 2006).
When quantification of each of the AR-67 forms separately was required, the cells were incubated with AR-67 carboxylate or lactone (1 μM) for 0.083, 0.5, 1, 3, 6, 12, 24, and 48 hours at 37°C and washed twice with Opti-MEM (4°C) at the end of the incubation period. Phosphate-buffered saline was added; the cells were scrapped off and lysed by sonication. AR-67 lactone and carboxylate were extracted and quantified using HPLC (Horn et al., 2006).
Drug Stability in Culture Media.
AR-67 interconversion was studied in cell culture media at pH 7.4 (37°C) in a humidified atmosphere of 5% CO2. AR-67 carboxylate (1 μM) or lactone (1 μM) were added into the media at t = 0 hours. The AR-67 carboxylate and lactone concentrations were quantified at 0, 0.5, 1, 2, 4, and 6 hours using HPLC (Horn et al., 2006)
Cytotoxicity Assays.
Cells were seeded in 96-well plates at initial densities that ensured their exponential growth for the duration of the experiment. To study the effect of AR-67 lactone and AR-67 carboxylate on cell viability, the cells were seeded and allowed to adhere overnight. Twenty-four hours later, the cell culture medium was replaced with drug-containing medium. The final concentration of dimethylsulfoxide in cell culture media never exceeded 1%. Cells expressing BCRP and MDR1 were exposed to AR-67 lactone for 72 hours. The effect of BCRP and MDR1 on AR-67 lactone-induced cytotoxicity was studied in the presence of the dual BCRP/MDR1 inhibitor GF120918. MDCKII-pcDNA/BCRP cells were pretreated for 30 minutes with GF120918 (1 μM) followed by cotreatment with GF120918 (1 μM) and AR-67 lactone. OVCAR-8 and NCI/ADR-RES cells were pretreated for 30 minutes with GF120918 (5 μM) followed by cotreatment with GF120918 (5 μM) and AR-67 lactone.
The cytotoxic effect of AR-67 carboxylate on the OATP-expressing cell lines after various exposure times was also evaluated. In these cases, cells were treated with AR-67 carboxylate for 5 and 30 minutes, 1 and 3 hours. At the end of the incubation period, the drug-containing medium was removed, and the cells were washed once with ice-cold phosphate-buffered saline. Afterward, drug-free medium was added in the wells, and the cells were allowed to grow for 48 hours, at which point their viability was assessed.
Cell viability was assessed with the CellTiter 96 AQueous Non-Radioactive Cell Proliferation Assay kit (Promega/Fisher Scientific) following the manufacturer’s instructions. The viability of the HeLa- and RKO-OATP-transfected cell lines was evaluated by using a resazurin assay (Alamar Blue).
Data Analysis and Statistics.
GraphPad Prism (version 5.02 for Windows; GraphPad Software Inc., San Diego, CA) was employed for the graphical representation of the results, data analyses, and statistical comparisons.
To calculate the kinetic parameters for the OATP1B3 and OATP1B1 transporters, AR-67 intracellular uptake was determined as described previously. Transporter-mediated uptake was corrected by subtracting the intracellular amount in mock-transfected cells, and error propagation was applied. Data were modeled using the Michaelis-Menten equation which provided us with a satisfactory fitting:
where V (ng of total AR-67/μg protein/min) is the rate of transport of AR-67 lactone or carboxylate, C (µM) is the concentration of AR-67 carboxylate added in the well, and the Michaelis-Menten rate constant (Km, µM) is the concentration of AR-67 lactone or carboxylate at which the transporter activity is equal to its maximum value (Vmax).
Dose-response data were also fitted using nonlinear regression analysis. The viability of the treated cells was normalized with the viability of untreated (control) cells. IC50 values were estimated as the dose of the cytotoxic drug that induces response equal to half of the maximum response observed.
Results
AR-67 Lactone Is a Substrate of BCRP and MDR1.
BCRP and MDR1-expressing cell lines with confirmed expression and function (Supplemental Figs. 1 and 2, respectively) were used to assess the interaction of AR-67 lactone with these efflux transporters. Initial studies assessed the time-dependent intracellular AR-67 amount (unpublished data), which demonstrated a time-dependent increase in intracellular amounts within the first 10 minutes. The intracellular amount of total AR-67 was then measured following 5-minute incubation with a range of AR-67 lactone concentrations. The intracellular AR-67 amounts in MDCKII-BCRP (Fig. 2A) and NCI/ADR-RES cell lines (Fig. 2B) were lower than those in the MDCKII-pcDNA and OVCAR-8 cells, respectively. The transporter effect was reversed when the cells were pretreated with GF120918 (Fig. 2, C and D), which is known to inhibit the drug efflux mediated by the ABCG2 and ABCB1 gene products (de Bruin et al., 1999; Evers et al., 2000).
Fig. 2.
Effect of BCRP and MDR1 expression on the intracellular AR-67 amounts in MDCKII-pcDNA/BCRP and OVCAR-8/NCI/ADR-RES cells. (A) MDCKII-pcDNA/BCRP and (B) OVCAR-8/NCI/ADR-RES cells were incubated with 0.125, 0.25, 0.5, 0.75 and 1 μM of AR-67 lactone for 5 minutes, and the AR-67 intracellular amount was evaluated. The effect of the dual BCRP/MDR1 inhibitor GF120918 was tested by treating (C) MDCKII-pcDNA/BCRP and (D) OVCAR-8/NCI/ADR-RES cells without (open bars) and with (solid bars) GF120918 (5 µM for 1 hour) before incubating with 1 µM AR-67 lactone for 5 minutes. At the end of the incubation periods, the cells were lysed, and the intracellular AR-67 was quantified using HPLC as mentioned in the Materials and Methods section. Data are represented as mean (n = 3) ± S.D. Statistical analysis was performed using unpaired t test, statistical significance for **P < 0.01; ***P < 0.001.
BCRP-mediated efflux of AR-67 lactone, in the presence and absence of GF120918, was also evaluated using polarized MDCKII-transfected cells grown on Transwell inserts. GF120918 had no effect on drug transfer from the apical to the basolateral side in MDCKII-pcDNA cells (Fig. 3A), but increased the transfer in MDCKII-BCRP cells (Fig. 3B). This is consistent with the apical expression of BCRP in polarized MDCKII cells (Grube et al., 2007; Wang et al., 2012).
Fig. 3.
Effect of BCRP on the vectorial transport of AR-67 in MDCKII-pcDNA/BCRP cells. Transepithelial transport of 5 µM AR-67 lactone in MDCKII-pcDNA and MDCKII-BCRP cells apical to basolateral (A > B; A and B) and basolateral to apical (B > A; C and D) in the presence (□) and absence (▿) of the inhibitor GF120918 (5 µM). AR-67 was quantified using HPLC as determined in the Materials and Methods section. Data are represented as the mean (n = 3) ± S.D.
GF120918 also attenuated AR-67 transfer from the basolateral to the apical side in MDCKII-pcDNA (Fig. 3C). However, the effect on MDCKII-BCRP cells was more significant (Fig. 3D). This is consistent with the determination of the apparent permeability and efflux ratio values estimated (Supplemental Table 1) for transport across BCRP-expressing cell monolayers. Collectively, these data suggest that AR-67 lactone is a substrate for MDR1 and BCRP.
Our attempt to perform similar studies with AR-67 carboxylate yielded insignificant intracellular AR-67 amounts in monolayers and drug transfer across polarized cells (unpublished data).
AR-67 Carboxylate Is Transported by OATP1B1 and OATP1B3.
Previous in vivo pharmacokinetic studies demonstrated that AR-67 carboxylate has a relatively higher clearance than the lactone form (Adane et al., 2010). Given the anionic nature of the carboxylate, we reasoned that rapid clearance may be through its increased uptake in the liver by organic anion transporters. Here, we assessed the uptake of AR-67 carboxylate in cells transfected with either SLCO1B1 or SLCO1B3. The expression of the OATP1B1 and OATP1B3 transporters was confirmed using reverse-transcription polymerase chain reaction analysis and immunohistochemistry while their function was evaluated using prototype substrates (Supplemental Fig. 3).
Initial time-dependent studies showed rapid uptake of AR-67 carboxylate in both transporter-expressing cell lines but not in the mock-transfected cells (unpublished data). Subsequently, concentration-dependent uptake studies were conducted after incubation with AR-67 carboxylate for 1 minute. The intracellular amount of total AR-67 was significantly higher in the OATP1B3 (HeLa-OATP1B3) and OATP1B1 (RKO-OATP1B1) expressing cells than that in the mock-transfected ones (Fig. 4). The concentration-dependent uptake of AR-67 carboxylate by OATP1B3 (Fig. 4A) is consistent with saturable kinetics. The transporter-mediated uptake was diminished by the addition of BSP (Fig. 4, C and D) (Seithel et al., 2007; Kalliokoski and Niemi, 2009).
Fig. 4.
Effect of OATP1B3 and OATP1B1 expression on the intracellular AR-67 amounts in HeLa-pIRES/OATP1B3 and RKO-pIRES/OATP1B1 cells. (A) HeLa-pIRES/OATP1B3 and (B) RKO-pIRES/OATP1B1 cells were incubated with 0.5, 1.0, 1.5, 2.0, and 3.0 µM of AR67 carboxylate for 1 minute to study the dose-dependent uptake of the carboxylate form. The estimated Km values for OATP1B3 and OATP1B1 were 0.32 (0.0–0.8316) μM and 2.58 (0.0–6.253) μM, respectively. They were obtained by using the Michaelis-Menten equation to fit the transporter-mediated uptake of AR-67, as described in the Materials and Methods section, and are reported as the mean (95% confidence interval). (C) HeLa-pIRES/OATP1B3 and (D) RKO-pIRES/OATP1B1 cells were incubated with 1 μM of AR-67 carboxylate for 5 and 10 minutes, respectively (open bars). The inhibitory effect of BSP (50 μM) was studied by pretreating for 10 minutes before exposure to AR-67 carboxylate as described previously for all cell lines (solid bars). Intracellular AR-67 was quantified using HPLC, as determined in the Materials and Methods section. Data are represented as the mean (n = 3) ± S.D. Statistical analysis was performed using unpaired t test, statistical significance for **P < 0.01; ***P < 0.001.
Kinetic analysis also demonstrated higher affinity of AR-67 carboxylate for OATP1B3 than OATP1B1, with estimated Km values of 0.32 and 2.58 μM, respectively. Additionally, incubation with the AR-67 lactone form did not result in significant uptake differences between OATP1B3 or OATP1B1 and mock-transfected cells (unpublished data).
BCRP and MDR-1 Confer Resistance to AR-67 Lactone.
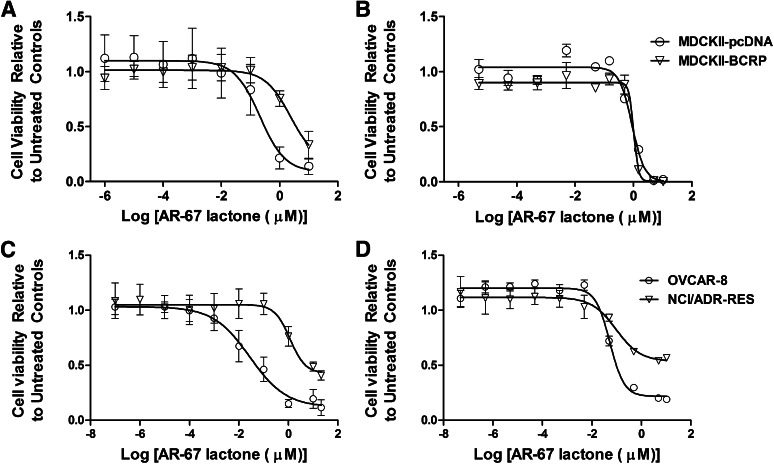
To evaluate the significance of the interaction between AR-67 lactone and the efflux transporters BCRP and MDR1, we assessed their potential for conferring drug resistance in the context of in vitro cytotoxicity studies. We found that BCRP confers a 10-fold resistance as measured by the differences in the estimated IC50 values between MDCKII-pcDNA and MDCKII-BCRP cells (Fig. 5A). The BCRP effect was negated in the presence of the transporter inhibitor GF120918 (Fig. 5B). Similarly, AR-67 lactone was significantly more cytotoxic to the OVCAR-8 than the NCI/ADR-RES cells as demonstrated by a 100-fold lower IC50 values (Fig. 5C). The transporter effect on the cytotoxic profile of AR-67 lactone was eliminated in the presence of GF120918 (Fig. 5D).
Fig. 5.
BCRP and MDR1 decrease the cytotoxicity of AR-67 lactone. (A) After 72-hour drug incubation, IC50 values were estimated in MDCKII-pcDNA (0.21 μM, [0.11–0.40]) and MDCKII-BCRP (2.37 μM, [1.41–4.00]; P < 0.01, unpaired Student’s t test). (B) After pretreatment (30 minutes) and in the presence of GF120918 and AR-67 lactone (72 hours), IC50 values were obtained in MDCKII-pcDNA (0.88 μM, [0.72–1.09]) and MDCKII-BCRP (1.02 μM, [0.79–1.32]). (C) Similarly, IC50 values were estimated in OVCAR-8 (0.027 μM, [0.018–0.043]) and NCI/ADR-RES (1.16 μM, [0.79–1.70]; P < 0.0001, unpaired Student’s t test). (D) After pretreatment (30 minutes) and in the presence of GF120918 and AR-67 lactone (72 hours), IC50 values were obtained in OVCAR-8 (0.053 μM, [0.044–0.064]) and NCI/ADR-RES cells (0.095 μM, [0.044–0.21]). The AR-67 lactone doses ranged from 10−7 to 21 µM. IC50 parameters and best-fit lines were estimated by nonlinear regression analysis. Data are plotted as the mean ± S.D. (n = 3), and IC50 values (μM) are reported as the mean (95% confidence interval).
To determine whether sensitivity differences between transporter expressing and nonexpressing cells were due to variability in the expression of the Top1 enzyme, we assessed its expression in the MDCKII-pcDNA/BCRP and OVCAR-8/NCI/ADR-RES cell lines (Supplemental Fig. 4). Our results indicated comparable Top1 protein expression between the control and transporter-expressing cell lines.
OATP1B3 Expression Does Not Sensitize Cells to AR-67 Carboxylate.
Our study showed that OATP1B3 and OATP1B1, which are highly expressed in the liver (Abe et al., 1999; Konig et al., 2000a,b; Abe et al., 2001), have the potential of transporting AR-67 carboxylate (Fig. 4). However, several recent studies have reported that OATP1B3 is also expressed in tumor tissues (Monks et al., 2007; Muto et al., 2007; Lee et al., 2008; Lockhart et al., 2008; Pressler et al., 2011). To determine whether active uptake of the carboxylate could sensitize OATP1B3-expressing cells, we conducted cytotoxicity studies (Fig. 6). The equivalent Top1 expression in HeLa-pIRES and HeLa-OATP1B3 was verified by Western blot analysis (Supplemental Fig. 4). Initially, the cytotoxicity of AR-67 carboxylate was assessed after 48 hours of continuous drug exposure (Fig. 6D), but owing to the reformation of the lactone upon incubation (Fig. 7A) we also determined the cytotoxicity after 0.5, 1.0, and 3.0 hours (Fig. 6, A–C) of drug treatment, which allowed for relatively greater carboxylate exposure. As expected by the action of camptothecins during the S-phase, longer incubation resulted in lower IC50 values (Fig. 6E). However, OATP1B3-expressing cells were not more sensitive than mock-transfected cells.
Fig. 6.
Time-dependent cytotoxic effect of AR-67 carboxylate on HeLa-pIRES and HeLa-OATP1B3 cell lines. (A–D) HeLa-pIRES and HeLa-OATP1B3 cell lines were exposed to AR-67 carboxylate for 30 minutes (A), 1 hour (B), 3 hours (C), and 48 hours (D), as described in the Materials and Methods section. The AR-67 carboxylate doses ranged from 10−3 to 210 µM. (E) Summary of the cytotoxicity of AR-67 carboxylate on HeLa-pIRES and HeLa-OATP1B3 cells studied under the experimental conditions described in panels A–D. IC50 values to reflect the effect of AR-67 on cell viability were used. Cell viability was assessed at the end of the treatment as described in the Materials and Methods section. Nonlinear regression analysis was performed to model the data (solid line). Data points on the graphs are reported as the mean (n = 3) ± S.D., and IC50 values (μM) are reported as the mean (95% confidence interval).
Fig. 7.
Stability of AR-67 in cell culture media. The time course of AR-67 interconversion was studied in cell culture media (pH 7.4, 37°C, 5% CO2) after the addition of 1 μM of AR-67 carboxylate (A) or 1 μM of AR-67 lactone (B). Data are represented as the percentage ratio concentration of carboxylate or lactone over total drug.
Time-Dependent Intracellular AR-67 Uptake after Exposure to Lactone and Carboxylate.
To gain a better understanding of the apparent lack of sensitivity of OATP1B3 cells to AR-67 carboxylate (Fig. 6), we determined the intracellular exposure to each AR-67 form. The intracellular uptake of AR-67 was assessed over 48 hours after exposing the cells to carboxylate (1 μM; Fig. 8A) or lactone (1 μM; Fig. 8B). The lactone form was found to dominate the intracellular compartment regardless of which form was added in the cell culture media (Fig. 8). As expected, the intracellular AR-67 carboxylate amount was higher in the HeLa-OATP1B3 cells than in the HeLa-pIRES cells at all times and irrespective of the extracellular AR-67 form. However, the intracellular lactone amount was not different between the two cell lines, although it was significantly higher than the respective carboxylate one.
Fig. 8.
Time course of AR-67 carboxylate and lactone intracellular amount in HeLa-pIRES and HeLa-OATP1B3 cells after incubation with AR-67 carboxylate or lactone. HeLa-pIRES and HeLa-OATP1B3 cells were incubated with 1 μM of the AR-67 forms, carboxylate (A) and lactone (B). AR-67 forms intracellularly were quantified by HPLC as described in the Materials and Methods section. Data are represented as the mean (n = 3) ± S.D.
The ratio of lactone to carboxylate increased from about 1, at 5 minutes, to 34 in the HeLa-pIRES and to 24 in the HeLa-OATP1B3 cell line after 48 hour of incubation with AR-67 carboxylate (Fig. 8A). As expected, when AR-67 lactone was added in the cell culture media, it diffused rapidly into both cell lines and was about 100 times higher than the intracellular carboxylate at the 5-minute time point (Fig. 8B). Although the amount of the intracellular carboxylate increased by almost 5-fold during the 48-hour incubation, lactone dominated, and the lactone amounts were significantly higher than the carboxylate respective ones (Fig. 8B). Importantly, at steady state the intracellular lactone and carboxylate reached the same ratio regardless of which form was added in the cell culture media (Fig. 8). Although carboxylate is the form that prevails in cell culture medium after carboxylate (Fig. 7A) or lactone addition (Fig. 7B), this study demonstrates that the cells are exposed to significantly higher lactone amounts.
Notably, after 5 minutes of incubation with AR-67 carboxylate, intracellular AR-67 carboxylate accounted for 42 and 55% of the total drug in HeLa-pIRES and HeLa1B3 cells, respectively. This is the only time point with high intracellular AR-67 carboxylate amounts in the OATP1B3 cells compared with the respective lactone ones (Fig. 8A). To determine whether this difference would be adequate to sensitize the OATP1B3 cells to carboxylate, we studied the cytotoxic effect of AR-67 carboxylate in Hela-pIRES and HeLa-OATP1B3 after drug exposure for 5 minutes, which was followed by 48 hours of growth. Although the IC50 values of carboxylate and lactone were lower in HeLa-OATP1B3 cells as compared with HeLa-pIRES-transfected cells, the differences were not statistically significant. As shown in Fig. 9A, the relatively increased exposure of OATP1B3-expressing cells to AR-67 carboxylate over the 5-minute drug exposure did not result in significant sensitization.
Fig. 9.
Effect of AR-67 carboxylate and lactone on HeLa-pIRES and HeLa-OATP1B3 cell lines. (A) HeLa-pIRES and HeLa-OATP1B3 cells were treated with AR-67 carboxylate for 5 minutes, cells were washed, drug-free medium was added into the wells, and the cells were allowed to grow for 48 hours before assessing cell viability. The estimated IC50 values were 40.32 μM (16.36–99.33) and 13.08 μM (8.29–20.62) for HeLa-pIRES and HeLa-OATP1B3 cells, respectively. (B) HeLa-pIRES and HeLa-OATP1B3 cells were treated with AR-67 lactone for 5 minutes, cells were washed, and drug-free medium was added into the wells. The estimated IC50 values were 1.08 μM (0.20–5.70) and 0.23 μM (0.028–1.93) for HeLa-pIRES and HeLa-OATP1B3 cells, respectively. Data analysis to obtain IC50 values was performed using nonlinear regression (solid line). Data are presented as the mean (n = 3) ± S.D., and IC50 values (μM) are reported as the mean (95% confidence interval).
To verify this result we then assessed the effect of drug exposure on DNA damage by the induction of phosphorylated histone 2AX (γ-H2AX) (Furuta et al., 2003; Huang et al., 2004). In accordance with the lack of a transporter effect on cell viability (Fig. 9A), the degree of γ-H2AX activation after treatment with AR-67 carboxylate (Supplemental Fig. 5) did not differ between the HeLa-pIRES and the HeLa-OATP1B3 transfected cells. For comparison, the cytotoxic effect of lactone was also studied under the same experimental conditions with similar results in both cell lines (Fig. 9B). Overall, HeLa-pIRES and HeLa-OATP1B3 were more sensitive to the lactone than the carboxylate when the IC50 values were used to evaluate the cytotoxic effect of AR-67 (Fig. 9).
Discussion
In this study, we demonstrated that BCRP and MDR1 transport AR-67 lactone. The MDCKII-BCRP cell line was found to be more resistant to AR-67 lactone treatment than the mock-transfected cells, and MDR1 expression conferred resistance to the same anticancer agent using OVCAR-8 and NCI/ADR-RES cells. Additionally, we studied the interaction between AR-67 carboxylate with the uptake transporters OATP1B3 and OATP1B1 and showed that these transporters mediate its transport. However, the OATP1B3-expressing cell line did not show increased sensitivity to AR-67 carboxylate in in vitro systems. In an effort to study the factors that play a role on the antitumor activity of AR-67 in vitro, we quantified both AR-67 forms intracellularly after treatment with either AR-67 lactone or carboxylate. Interestingly, our intracellular uptake data indicated that the AR-67 lactone was favored intracellularly and overcame the OATP1B3-mediated increased intracellular uptake of AR-67 carboxylate by 50-fold. This may explain why the uptake of AR-67 carboxylate does not sensitize OATP1B3-expressing cells.
Based on the physicochemical properties of the AR-67 lactone and carboxylate, we expected that both efflux and uptake transporters would interact with AR-67. Studies have shown that MDR1 (and possibly BCRP) transport the substrates that have the potential for partitioning into the lipid bilayer and are mostly lipid-soluble moieties with planar aromatic rings (Sharom, 2008), such as AR-67 lactone. Conversely, OATP1B1 and OATP1B3 proteins selectively transport anions (Kalliokoski and Niemi, 2009), such as AR-67 carboxylate. Therefore, in our studies we adjusted the pH to ensure the exposure of cells to AR-67 lactone or carboxylate, as needed. Furthermore, the exposure and incubation times for transport and cell viability assays were considered in the context of lactone/carboxylate conversion kinetics.
Our studies showed that AR-67 lactone is a substrate of BCRP and MDR1 (Figs. 2 and 3). This is in accord with previous reports showing similar results with topotecan (de Vries et al., 2007; Sharom, 2008). Other camptothecin analogs have also been shown to be substrates of BCRP, which was reported to transport irinotecan, SN-38, and SN-38 glucuronide (Maliepaard et al., 1999; Nakatomi et al., 2001; Luo et al., 2002). Our studies also showed that AR-67 carboxylate is transported by OATP1B1 and OATP1B3 (Fig. 4). Previous studies have shown that irinotecan and its metabolite SN-38 are substrates of OATP1B1 (Nozawa et al., 2005). However, Yamaguchi et al. (2008) showed that SN-38, but not irinotecan, can be taken up by OATP1B3. Finally, in vitro studies have shown that, among the lipophilic camptothecins, gimatecan and karenitecin but not lurtotecan are substrates of OATP1B1 (Oostendorp et al., 2009), although the report did not indicate which form was transported.
After identifying AR-67 lactone as a BCRP substrate, we used the MDCKII-pcDNA/BCRP cell lines to demonstrate that BCRP expression confers resistance to treatment with the lactone form (Fig. 5A). Our findings agree with findings from other studies on camptothecins. BCRP-expressing lung cancer cell lines appear to be more resistant to SN-38 than their parent cell line (Kawabata et al., 2001). Moreover, studies in metastatic hepatic tumor tissue have indicated that BCRP might be implicated in the development of resistance specifically to irinotecan-based chemotherapy in vivo (Candeil et al., 2004). Despite the significant expression of BCRP in certain cancer types and especially in a small subpopulation of primitive stem cells (Katayama et al., 2009), definitive clinical relevance of BCRP expression in tumor tissues has yet to be proven. Additionally, as a result of the MDR1-mediated efflux of AR-67 in NCI/ADR-RES cells (Fig. 2, B and D), as compared with OVCAR-8 cells, a resistance phenotype was observed (Fig. 5C). Previous studies have been suggestive of the prognostic role of MDR1 in leukemia patients treated with anthracyclines (Guerci et al., 1995; Smeets et al., 1997), which are established substrates of MDR1. However, further studies are warranted to evaluate the impact of an MDR1-AR-67 interaction on clinical response in cancer patients.
Both in vitro and clinical studies have indicated that OATP1B3 facilitates the cytotoxic effect of paclitaxel (Smith et al., 2007; Svoboda et al., 2011). Here, we tested whether OATP1B3 would sensitize cells after exposure to the anionic AR-67 carboxylate. Our studies showed that the observed cytotoxic effect was not different between HeLa-pIRES and HeLa-OATP1B3 cells, irrespective of the exposure time to the carboxylate (Fig. 6). However, the cytotoxic effect of AR-67 carboxylate increased in a time-dependent manner, consistent with its mechanism of action targeting cells primarily in their S-phase. In addition, comparable activation of the DNA damage marker γ-H2AX (Supplemental Fig. 5) in HeLa-pIRES and HeLa-OATP1B3 cells after 5-minute exposure to AR-67 carboxylate was consistent with the absence of OATP1B3 effect on cell viability (Fig. 9A).
The quantification of the intracellular levels of both AR-67 forms suggests that the lipophilic lactone diffuses freely in the cell while the transfer of the carboxylate form into the cytoplasm is a slow, transporter-mediated process, limited by saturable kinetics (Fig. 8). Ultimately, the intracellular lactone/carboxylate steady-state amounts, independent of the extracellular lactone/carboxylate kinetics (Fig. 7), suggest that the AR-67 carboxylate-induced cytotoxic effect is restricted due to its physicochemical properties. Notably, the same intracellular lactone-carboxylate amount ratio was observed when cells were exposed to either lactone or carboxylate suggesting that the interconversion between the two forms depended strictly on intracellular factors (Fig. 8). Rapid conversion of carboxylate to the lactone form in the cytoplasm due to protein-binding cannot be excluded. Moreover, although the intracellular pH would favor carboxylate formation, the AR-67 intracellular distribution pattern could be similar to the one observed for the third-generation lipophilic camptothecin gimatecan, which partitions into lysosomes where the lactone form is favored due to low pH conditions (Croce et al., 2004).
The microenvironment of solid tumors is characterized by hypoxia and low pH as a result of disorganized vasculature and an underdeveloped lymphatic system (Gillies et al., 2002; Cardone et al., 2005). Previous studies have shown increased transport activity of BCRP and MDR1 at acidic conditions (Thews et al., 2006; Breedveld et al., 2007) and pH-sensitive transport activity of OATP1B3 (Leuthold et al., 2009). Therefore, further studies on the AR-67 cytotoxic effect under low pH conditions are necessary.
The first-in-human study showed that approximately 87% of total AR-67 quantified in the blood was in the lactone form (Arnold et al., 2010). However, pharmacokinetic modeling of preclinical data demonstrated that the apparent stability was due to increased carboxylate clearance (Adane et al., 2010). Our findings suggest that OATP1B1 and OATP1B3 hepatic expression is the likely mechanism for this clearance, and thus it may play a key role in the disposition of AR-67. These studies, therefore, demonstrate how transporters can play a key role in drug disposition without having a significant role in cellular uptake and tumor responsiveness, which is a static system.
Multiple studies have underlined the importance of efflux and uptake transporters in the disposition of anticancer agents. Furthermore, genetic polymorphisms in ABCG2 and ABCB1 that were identified in patients receiving irinotecan and diflomotecan, respectively, resulted in altered pharmacokinetics and pharmacodynamics (Sai et al., 2003; de Jong et al., 2004; Sparreboom et al., 2004; Zhou et al., 2005). Additionally, SLCO1B1 polymorphisms possibly related with the toxicity profile of irinotecan and other cytotoxic agents have been reported (Xiang et al., 2006; Han et al., 2008; Innocenti et al., 2009). Similarly, the clinical impact of in vitro identified OATP1B3 polymorphisms need to be explored further in well-designed large-scale human trials (Letschert et al., 2004; Smith et al., 2007). In line with these studies and our results, future studies should explore further the potential role of BCRP, MDR1, OATP1B1, and OATP1B3 polymorphisms on the transport of AR-67.
In conclusion, our study demonstrated that AR-67 lactone is a substrate of the ATP-binding cassette transporters MDR1 and BCRP. Additionally, both BCRP- and MDR1-mediated efflux of AR-67 lactone conferred resistance using in vitro models. The clinical significance of these findings needs to be explored further. Although our in vitro work suggests that OATP1B3 expression might not increase the efficacy of AR-67 given to cancer patients expressing the transporter in the tumor tissue, we demonstrated that AR-67 carboxylate can be transported by the OATP1B1 and OATP1B3 transporters. Taking into consideration the liver-specific expression of these uptake transporters (Abe et al., 1999; Konig et al., 2000a) and the impact of carboxylate clearance on exposure to AR-67 (Adane et al., 2010), we reason that they may be playing a key role in the elimination of AR-67.
Supplementary Material
Abbreviations
- ABC
ATP-binding cassette
- AR-67
7-t-butyldimethylsilyl-10-hydroxycamptothecin (DB-67)
- BCRP
breast cancer resistance protein (ABCG2)
- BSP
bromosulfophthalein
- FBS
fetal bovine serum
- GF120918
N-4-[2-(1,2,3,4-tetrahydro-6,7-dimethoxy-2-isoquinolinyl)ethyl]-phenyl)-9,10-dihydro-5-methoxy-9-oxo-4-acridine carboxamide hydrochloride
- HPLC
high-performance liquid chromatography
- Km
Michaelis-Menten rate constant
- MDCKII
Madin-Darby canine kidney strain II
- MDR1
multidrug resistant protein 1 (ABCB1)
- MEM
minimum essential media
- NCI
National Cancer Institute
- OATP
organic anion-transporting polypeptide
- OVCAR-8
human ovarian cancer cell line 8
- RKO
colon carcinoma cells
- SN-38
7-ethyl-10-hydroxy-camptothecin
- Vmax
maximum transport rate
- γ-H2AX
phosphorylated histone 2AX
Authorship Contributions
Participated in research design: Tsakalozou, Adane, Leggas.
Conducted experiments: Tsakalozou, Adane, Kuo, Daily.
Performed data analysis: Tsakalozou, Adane, Kuo, Daily, Leggas.
Wrote or contributed to the writing of the manuscript: Tsakalozou, Adane, Kuo, Daily, Moscow, Leggas.
Footnotes
This work was supported in part by the National Institutes of Health National Cancer Institute [Grant CA-123867].
 This article has supplemental material available at dmd.aspetjournals.org.
This article has supplemental material available at dmd.aspetjournals.org.
References
- Abe T, Kakyo M, Tokui T, Nakagomi R, Nishio T, Nakai D, Nomura H, Unno M, Suzuki M, Naitoh T, et al. (1999) Identification of a novel gene family encoding human liver-specific organic anion transporter LST-1. J Biol Chem 274:17159–17163 [DOI] [PubMed] [Google Scholar]
- Abe T, Unno M, Onogawa T, Tokui T, Kondo TN, Nakagomi R, Adachi H, Fujiwara K, Okabe M, Suzuki T, et al. (2001) LST-2, a human liver-specific organic anion transporter, determines methotrexate sensitivity in gastrointestinal cancers. Gastroenterology 120:1689–1699 [DOI] [PubMed] [Google Scholar]
- Adane ED, Liu Z, Xiang TX, Anderson BD, Leggas M. (2010) Factors affecting the in vivo lactone stability and systemic clearance of the lipophilic camptothecin analogue AR-67. Pharm Res 27:1416–1425 [DOI] [PubMed] [Google Scholar]
- Arnold SM, Rinehart JJ, Tsakalozou E, Eckardt JR, Fields SZ, Shelton BJ, DeSimone PA, Kee BK, Moscow JA, Leggas M. (2010) A phase I study of 7-t-butyldimethylsilyl-10-hydroxycamptothecin in adult patients with refractory or metastatic solid malignancies. Clin Cancer Res 16:673–680 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bom D, Curran DP, Kruszewski S, Zimmer SG, Thompson Strode J, Kohlhagen G, Du W, Chavan AJ, Fraley KA, Bingcang AL, et al. (2000) The novel silatecan 7-tert-butyldimethylsilyl-10-hydroxycamptothecin displays high lipophilicity, improved human blood stability, and potent anticancer activity. J Med Chem 43:3970–3980 [DOI] [PubMed] [Google Scholar]
- Bom D, Curran DP, Zhang J, Zimmer SG, Bevins R, Kruszewski S, Howe JN, Bingcang A, Latus LJ, Burke TG. (2001) The highly lipophilic DNA topoisomerase I inhibitor DB-67 displays elevated lactone levels in human blood and potent anticancer activity. J Control Release 74:325–333 [DOI] [PubMed] [Google Scholar]
- Breedveld P, Pluim D, Cipriani G, Dahlhaus F, van Eijndhoven MA, de Wolf CJ, Kuil A, Beijnen JH, Scheffer GL, Jansen G, et al. (2007) The effect of low pH on breast cancer resistance protein (ABCG2)-mediated transport of methotrexate, 7-hydroxymethotrexate, methotrexate diglutamate, folic acid, mitoxantrone, topotecan, and resveratrol in in vitro drug transport models. Mol Pharmacol 71:240–249 [DOI] [PubMed] [Google Scholar]
- Candeil L, Gourdier I, Peyron D, Vezzio N, Copois V, Bibeau F, Orsetti B, Scheffer GL, Ychou M, Khan QA, et al. (2004) ABCG2 overexpression in colon cancer cells resistant to SN38 and in irinotecan-treated metastases. Int J Cancer 109:848–854 [DOI] [PubMed] [Google Scholar]
- Cardone RA, Casavola V, Reshkin SJ. (2005) The role of disturbed pH dynamics and the Na+/H+ exchanger in metastasis. Nat Rev Cancer 5:786–795 [DOI] [PubMed] [Google Scholar]
- Croce AC, Bottiroli G, Supino R, Favini E, Zuco V, Zunino F. (2004) Subcellular localization of the camptothecin analogues, topotecan and gimatecan. Biochem Pharmacol 67:1035–1045 [DOI] [PubMed] [Google Scholar]
- Curran DP, Josien H, Bom D, Gabarda AE, Du W. (2000) The cascade radical annulation approach to new analogues of camptothecins. Combinatorial synthesis of silatecans and homosilatecans. Ann N Y Acad Sci 922:112–121 [DOI] [PubMed] [Google Scholar]
- Daily A, Monks NR, Leggas M, Moscow JA. (2010) Abrogation of microcystin cytotoxicity by MAP kinase inhibitors and N-acetyl cysteine is confounded by OATPIB1 uptake activity inhibition. Toxicon 55:827–837 [DOI] [PubMed] [Google Scholar]
- de Bruin M, Miyake K, Litman T, Robey R, Bates SE. (1999) Reversal of resistance by GF120918 in cell lines expressing the ABC half-transporter, MXR. Cancer Lett 146:117–126 [DOI] [PubMed] [Google Scholar]
- de Jong FA, Marsh S, Mathijssen RH, King C, Verweij J, Sparreboom A, McLeod HL. (2004) ABCG2 pharmacogenetics: ethnic differences in allele frequency and assessment of influence on irinotecan disposition. Clin Cancer Res 10:5889–5894 [DOI] [PubMed] [Google Scholar]
- de Vries NA, Zhao J, Kroon E, Buckle T, Beijnen JH, van Tellingen O. (2007) P-glycoprotein and breast cancer resistance protein: two dominant transporters working together in limiting the brain penetration of topotecan. Clin Cancer Res 13:6440–6449 [DOI] [PubMed] [Google Scholar]
- Evers R, Kool M, Smith AJ, van Deemter L, de Haas M, Borst P. (2000) Inhibitory effect of the reversal agents V-104, GF120918 and Pluronic L61 on MDR1 Pgp-, MRP1- and MRP2-mediated transport. Br J Cancer 83:366–374 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuta T, Takemura H, Liao ZY, Aune GJ, Redon C, Sedelnikova OA, Pilch DR, Rogakou EP, Celeste A, Chen HT, et al. (2003) Phosphorylation of histone H2AX and activation of Mre11, Rad50, and Nbs1 in response to replication-dependent DNA double-strand breaks induced by mammalian DNA topoisomerase I cleavage complexes. J Biol Chem 278:20303–20312 [DOI] [PubMed] [Google Scholar]
- Gillies RJ, Raghunand N, Karczmar GS, Bhujwalla ZM. (2002) MRI of the tumor microenvironment. J Magn Reson Imaging 16:430–450 [DOI] [PubMed] [Google Scholar]
- Grube M, Reuther S, Meyer Zu Schwabedissen H, Köck K, Draber K, Ritter CA, Fusch C, Jedlitschky G, Kroemer HK. (2007) Organic anion transporting polypeptide 2B1 and breast cancer resistance protein interact in the transepithelial transport of steroid sulfates in human placenta. Drug Metab Dispos 35:30–35 [DOI] [PubMed] [Google Scholar]
- Guerci A, Merlin JL, Missoum N, Feldmann L, Marchal S, Witz F, Rose C, Guerci O. (1995) Predictive value for treatment outcome in acute myeloid leukemia of cellular daunorubicin accumulation and P-glycoprotein expression simultaneously determined by flow cytometry. Blood 85:2147–2153 [PubMed] [Google Scholar]
- Han JY, Lim HS, Shin ES, Yoo YK, Park YH, Lee JE, Kim HT, Lee JS. (2008) Influence of the organic anion-transporting polypeptide 1B1 (OATP1B1) polymorphisms on irinotecan-pharmacokinetics and clinical outcome of patients with advanced non-small cell lung cancer. Lung Cancer 59:69–75 [DOI] [PubMed] [Google Scholar]
- Horn J, Jordan SL, Song L, Roberts MJ, Anderson BD, Leggas M. (2006) Validation of an HPLC method for analysis of DB-67 and its water soluble prodrug in mouse plasma. J Chromatogr B Analyt Technol Biomed Life Sci 844:15–22 [DOI] [PubMed] [Google Scholar]
- Huang X, Okafuji M, Traganos F, Luther E, Holden E, Darzynkiewicz Z. (2004) Assessment of histone H2AX phosphorylation induced by DNA topoisomerase I and II inhibitors topotecan and mitoxantrone and by the DNA cross-linking agent cisplatin. Cytometry A 58:99–110 [DOI] [PubMed] [Google Scholar]
- Innocenti F, Kroetz DL, Schuetz E, Dolan ME, Ramírez J, Relling M, Chen P, Das S, Rosner GL, Ratain MJ. (2009) Comprehensive pharmacogenetic analysis of irinotecan neutropenia and pharmacokinetics. J Clin Oncol 27:2604–2614 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalliokoski A, Niemi M. (2009) Impact of OATP transporters on pharmacokinetics. Br J Pharmacol 158:693–705 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalvass JC, Pollack GM. (2007) Kinetic considerations for the quantitative assessment of efflux activity and inhibition: implications for understanding and predicting the effects of efflux inhibition. Pharm Res 24:265–276 [DOI] [PubMed] [Google Scholar]
- Katayama R, Koike S, Sato S, Sugimoto Y, Tsuruo T, Fujita N. (2009) Dofequidar fumarate sensitizes cancer stem-like side population cells to chemotherapeutic drugs by inhibiting ABCG2/BCRP-mediated drug export. Cancer Sci 100:2060–2068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawabata S, Oka M, Shiozawa K, Tsukamoto K, Nakatomi K, Soda H, Fukuda M, Ikegami Y, Sugahara K, Yamada Y, et al. (2001) Breast cancer resistance protein directly confers SN-38 resistance of lung cancer cells. Biochem Biophys Res Commun 280:1216–1223 [DOI] [PubMed] [Google Scholar]
- König J, Cui Y, Nies AT, Keppler D. (2000a) Localization and genomic organization of a new hepatocellular organic anion transporting polypeptide. J Biol Chem 275:23161–23168 [DOI] [PubMed] [Google Scholar]
- König J, Cui Y, Nies AT, Keppler D. (2000b) A novel human organic anion transporting polypeptide localized to the basolateral hepatocyte membrane. Am J Physiol Gastrointest Liver Physiol 278:G156–G164 [DOI] [PubMed] [Google Scholar]
- Lee W, Belkhiri A, Lockhart AC, Merchant N, Glaeser H, Harris EI, Washington MK, Brunt EM, Zaika A, Kim RB, et al. (2008) Overexpression of OATP1B3 confers apoptotic resistance in colon cancer. Cancer Res 68:10315–10323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letschert K, Keppler D, König J. (2004) Mutations in the SLCO1B3 gene affecting the substrate specificity of the hepatocellular uptake transporter OATP1B3 (OATP8). Pharmacogenetics 14:441–452 [DOI] [PubMed] [Google Scholar]
- Leuthold S, Hagenbuch B, Mohebbi N, Wagner CA, Meier PJ, Stieger B. (2009) Mechanisms of pH-gradient driven transport mediated by organic anion polypeptide transporters. Am J Physiol Cell Physiol 296:C570–C582 [DOI] [PubMed] [Google Scholar]
- Lockhart AC, Harris E, Lafleur BJ, Merchant NB, Washington MK, Resnick MB, Yeatman TJ, Lee W. (2008) Organic anion transporting polypeptide 1B3 (OATP1B3) is overexpressed in colorectal tumors and is a predictor of clinical outcome. Clin Exp Gastroenterol 1:1–7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luo FR, Paranjpe PV, Guo A, Rubin E, Sinko P. (2002) Intestinal transport of irinotecan in Caco-2 cells and MDCK II cells overexpressing efflux transporters Pgp, cMOAT, and MRP1. Drug Metab Dispos 30:763–770 [DOI] [PubMed] [Google Scholar]
- Maliepaard M, van Gastelen MA, de Jong LA, Pluim D, van Waardenburg RC, Ruevekamp-Helmers MC, Floot BG, Schellens JH. (1999) Overexpression of the BCRP/MXR/ABCP gene in a topotecan-selected ovarian tumor cell line. Cancer Res 59:4559–4563 [PubMed] [Google Scholar]
- Monks NR, Liu S, Xu Y, Yu H, Bendelow AS, Moscow JA. (2007) Potent cytotoxicity of the phosphatase inhibitor microcystin LR and microcystin analogues in OATP1B1- and OATP1B3-expressing HeLa cells. Mol Cancer Ther 6:587–598 [DOI] [PubMed] [Google Scholar]
- Muto M, Onogawa T, Suzuki T, Ishida T, Rikiyama T, Katayose Y, Ohuchi N, Sasano H, Abe T, Unno M. (2007) Human liver-specific organic anion transporter-2 is a potent prognostic factor for human breast carcinoma. Cancer Sci 98:1570–1576 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakatomi K, Yoshikawa M, Oka M, Ikegami Y, Hayasaka S, Sano K, Shiozawa K, Kawabata S, Soda H, Ishikawa T, et al. (2001) Transport of 7-ethyl-10-hydroxycamptothecin (SN-38) by breast cancer resistance protein ABCG2 in human lung cancer cells. Biochem Biophys Res Commun 288:827–832 [DOI] [PubMed] [Google Scholar]
- Nozawa T, Minami H, Sugiura S, Tsuji A, Tamai I. (2005) Role of organic anion transporter OATP1B1 (OATP-C) in hepatic uptake of irinotecan and its active metabolite, 7-ethyl-10-hydroxycamptothecin: in vitro evidence and effect of single nucleotide polymorphisms. Drug Metab Dispos 33:434–439 [DOI] [PubMed] [Google Scholar]
- Oostendorp RL, van de Steeg E, van der Kruijssen CM, Beijnen JH, Kenworthy KE, Schinkel AH, Schellens JH. (2009) Organic anion-transporting polypeptide 1B1 mediates transport of Gimatecan and BNP1350 and can be inhibited by several classic ATP-binding cassette (ABC) B1 and/or ABCG2 inhibitors. Drug Metab Dispos 37:917–923 [DOI] [PubMed] [Google Scholar]
- Pressler H, Sissung TM, Venzon D, Price DK, Figg WD. (2011) Expression of OATP family members in hormone-related cancers: potential markers of progression. PLoS ONE 6:e20372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sai K, Kaniwa N, Itoda M, Saito Y, Hasegawa R, Komamura K, Ueno K, Kamakura S, Kitakaze M, Shirao K, et al. (2003) Haplotype analysis of ABCB1/MDR1 blocks in a Japanese population reveals genotype-dependent renal clearance of irinotecan. Pharmacogenetics 13:741–757 [DOI] [PubMed] [Google Scholar]
- Seithel A, Eberl S, Singer K, Auge D, Heinkele G, Wolf NB, Dörje F, Fromm MF, König J. (2007) The influence of macrolide antibiotics on the uptake of organic anions and drugs mediated by OATP1B1 and OATP1B3. Drug Metab Dispos 35:779–786 [DOI] [PubMed] [Google Scholar]
- Sharom FJ. (2008) ABC multidrug transporters: structure, function and role in chemoresistance. Pharmacogenomics 9:105–127 [DOI] [PubMed] [Google Scholar]
- Smeets M, Raymakers R, Vierwinden G, Pennings A, van de Locht L, Wessels H, Boezeman J, de Witte T. (1997) A low but functionally significant MDR1 expression protects primitive haemopoietic progenitor cells from anthracycline toxicity. Br J Haematol 96:346–355 [DOI] [PubMed] [Google Scholar]
- Smith NF, Marsh S, Scott-Horton TJ, Hamada A, Mielke S, Mross K, Figg WD, Verweij J, McLeod HL, Sparreboom A. (2007) Variants in the SLCO1B3 gene: interethnic distribution and association with paclitaxel pharmacokinetics. Clin Pharmacol Ther 81:76–82 [DOI] [PubMed] [Google Scholar]
- Sparreboom A, Gelderblom H, Marsh S, Ahluwalia R, Obach R, Principe P, Twelves C, Verweij J, McLeod HL. (2004) Diflomotecan pharmacokinetics in relation to ABCG2 421C>A genotype. Clin Pharmacol Ther 76:38–44 [DOI] [PubMed] [Google Scholar]
- Staker BL, Hjerrild K, Feese MD, Behnke CA, Burgin AB, Jr, Stewart L. (2002) The mechanism of topoisomerase I poisoning by a camptothecin analog. Proc Natl Acad Sci USA 99:15387–15392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Svoboda M, Wlcek K, Taferner B, Hering S, Stieger B, Tong D, Zeillinger R, Thalhammer T, Jager W. (2011) Expression of organic anion-transporting polypeptides 1B1 and 1B3 in ovarian cancer cells: relevance for paclitaxel transport. Biomed Pharmacother 65:417–426 [DOI] [PubMed] [Google Scholar]
- Thews O, Gassner B, Kelleher DK, Schwerdt G, Gekle M. (2006) Impact of extracellular acidity on the activity of P-glycoprotein and the cytotoxicity of chemotherapeutic drugs. Neoplasia 8:143–152 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang L, Leggas M, Empey PE, McNamara PJ. (2012) Stereoselective interaction of pantoprazole with ABCG2. II. In vitro flux analysis. Drug Metab Dispos 40:1024–1031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xiang X, Jada SR, Li HH, Fan L, Tham LS, Wong CI, Lee SC, Lim R, Zhou QY, Goh BC, et al. (2006) Pharmacogenetics of SLCO1B1 gene and the impact of *1b and *15 haplotypes on irinotecan disposition in Asian cancer patients. Pharmacogenet Genomics 16:683–691 [DOI] [PubMed] [Google Scholar]
- Yamaguchi H, Kobayashi M, Okada M, Takeuchi T, Unno M, Abe T, Goto J, Hishinuma T, Mano N. (2008) Rapid screening of antineoplastic candidates for the human organic anion transporter OATP1B3 substrates using fluorescent probes. Cancer Lett 260:163–169 [DOI] [PubMed] [Google Scholar]
- Zhou Q, Sparreboom A, Tan EH, Cheung YB, Lee A, Poon D, Lee EJ, Chowbay B. (2005) Pharmacogenetic profiling across the irinotecan pathway in Asian patients with cancer. Br J Clin Pharmacol 59:415–424 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.