Abstract
The new model plant for temperate grasses, Brachypodium distachyon offers great potential as a tool for functional genomics. We have established a sodium azide-induced mutant collection and a TILLING platform, called “BRACHYTIL”, for the inbred line Bd21-3. The TILLING collection consists of DNA isolated from 5530 different families. Phenotypes were reported and organized in a phenotypic tree that is freely available online. The tilling platform was validated by the isolation of mutants for seven genes belonging to multigene families of the lignin biosynthesis pathway. In particular, a large allelic series for BdCOMT6, a caffeic acid O-methyl transferase was identified. Some mutants show lower lignin content when compared to wild-type plants as well as a typical decrease of syringyl units, a hallmark of COMT-deficient plants. The mutation rate was estimated at one mutation per 396 kb, or an average of 680 mutations per line. The collection was also used to assess the Genetically Effective Cell Number that was shown to be at least equal to 4 cells in Brachypodium distachyon. The mutant population and the TILLING platform should greatly facilitate functional genomics approaches in this model organism.
Introduction
Brachypodium distachyon is a valuable model plant for economically important temperate grasses such as wheat, barley and oat [1]. Attractive features are the small size, simple culture conditions and the small (272 Mb), diploid (2n = 10) and fully sequenced genome [2]. B. distachyon is currently being used as a model for the study of domestication in grasses [1], [2], [3], [4], [5], plant pathogen interactions, root and culm development, biomass production and cell wall biosynthesis. Important progress has been made in the development of efficient transformation protocols [6], [7], [8] and sequence-indexed T-DNA insertion collections [9]: the BrachyTAG collection at the John Innes Centre (5000 lines) ([10], http://www.brachytag.org/) and the USDA Brachypodium Genome Resources collection (8491 lines) [11]. To extend the panel of resources for functional genomics in this species, we have developed a mutagenized population and a TILLING (Targeting Induced Local Lesion IN Genome) platform. Chemically-induced mutants are complementary to insertion mutants in several aspects: (i) mutation rates are higher and hence screens can be done on smaller mutant populations, (ii) single base pair changes, as opposed to insertion mutants, more likely yield allelic series of partial loss-of-function, conditional or gain-of-function mutants and (iii) somaclonal variation is avoided in the absence of an in vitro culture step that is required for T-DNA-induced mutagenesis [12], [13]. An efficient method to identify new alleles for target genes is TILLING. This method developed a decade ago has been successfully applied to many plant species [14], [15]. In the TILLING method, seeds are mutagenized, the resulting M1 plants are self-fertilized, and the M2 generation of individuals is used to prepare DNA samples for mutational screening, while seeds of the M3 families can be stored and distributed. DNA samples are pooled and subjected to gene-specific PCR. The amplification products are incubated with an endonuclease that preferentially cleaves mismatches in heteroduplexes between wild type and mutant. Upon detection of a mutation in a pool, the individual DNA samples are similarly screened to identify the plant carrying the mutation. TILLING populations have been established for grasses including wheat, sorghum, barley, rice and oat [15], [16], [17], [18], [19], [20], [21], [22], [23] but to our knowledge not yet for Brachypodium distachyon. Here we report on the development of a mutagenized population and a TILLING platform and demonstrate the efficiency of the TILLING method with the identification of a series of allelic mutants for an O-methyl transferase, involved in the lignification of the internodes.
Materials and Methods
Plant Material and Growth Conditions
Brachypodium distachyon (L.) Beauv. inbred line Bd21-3 was kindly provided by John Vogel. Bd21-3 seeds were grown in a greenhouse under long-day conditions (18 h light, 400 watt sodium lamps). Day and night temperatures were 23°C and 18°C, respectively. The relative humidity was about 60%. Plants were grown in soil (one-liter pots) and watered twice a week.
Chemical Mutagenesis
For the production of mutants, dry seeds were pre-soaked in distilled water for 2 h. Portions of 5000 seeds were then suspended in 200 mL of fresh sodium azide (NaN3) solution diluted in phosphate buffer (0.1 M, pH 3) for 2 h under the hood and with gentle shaking. The seeds were washed 3 times in water for 1 h and then kept at +4°C for 72 h before sowing in pots. For establishment of the kill curve, 500 seeds were mutagenized with 0.5, 1, 1.5, 3 or 10 mM NaN3.
Genomic DNA Extraction and Pooling
Four M2 plants per family were grown for one month in a greenhouse. DNA was extracted from 3 cm-long portions of the median foliar part. The collected samples were pooled and placed in 96-well plates containing 2 steel beads per well. Samples were lyophilized and ground using a bead mill. Genomic DNAs were isolated using the DNeasy 96 Plant Kit (Qiagen, Hilden, Germany). All genomic DNAs were both quantified on a 1% agarose gel with λ DNA (Invitrogen, Carlsbad, CA, USA) as a concentration reference using a NanoDrop spectrophotometer 2000 c (Thermo Fisher Scientific, MA, USA). DNA concentration was normalized to 6 ng.µL−1 and pooled eightfold in a 96-well format.
PCR Amplification and Detection of Mutations
DNA amplification is based on nested-PCR. The first PCR amplification is a standard PCR reaction with target-specific primers and 10 ng of Brachypodium genomic DNA. One µl of the first PCR product served as a template for the second nested PCR amplification, with a combination of specific primers carrying M13 tail and M13 universal primers, M13F700 (5′-CACGACGTTGTAAAACGAC-3′) and M13R800 (5′-GGATAACATTTCACACAGG-3′), labelled at the 5′end with infra-red dyes IRD700 and IRD800 (LI-COR®, Lincoln, Nebraska, USA) respectively. Mutation detection was carried out as described previously except for the second PCR. This PCR was carried out using 0.05 µM of specific primers carrying M13 tail and 0.1 µM of M13 universal primers. The identity of the mutations was determined by sequencing.
Sequence Analysis Tools
The CODDLE software (Codons Optimized to Discover Deleterious Lesions, http://www.proweb.org/coddle/) was used to identify regions of the target gene in which G/C to A/T transitions are most likely to result in deleterious effects on the protein. The PARSESNP software (Project Aligned Related Sequences and Evaluate SNPs, http://www.proweb.org/parsesnp/) was used to illustrate the distribution of mutations within the gene and to indicate the nature of each single mutation. The SIFT software (Sorting Intolerant from Tolerant, http://sift.jcvi.org/www/SIFT_seq_submit2.html) was used to predict the impact of the mutation on the protein. Multiple sequence alignment of full-length protein sequences was performed with ClustalW software (http://www.ebi.ac.uk/Tools/clustalw2).
Phylogenetic Tree and 3D-structure Prediction
Protein sequences of COMT from several vascular plants were identified by protein blast (http://www.ncbi.nlm.nih.gov/and http://www.greenphyl.org) and aligned with Clustawl2 (pairwise alignment) at (http://www.ebi.ac.uk/Tools/msa/clustalw2). Phylogenetic analysis was performed with TreeTop (http://www.genebee.msu.su/services/phtree_reduced.html). Cluster algorithm was used and 100 bootstrap were done. Final construction of tree was made with TREX (Treeview Newick Viewer option) at http://www.trex.uqam.ca/. Bootstrap values are expressed in percentages and placed at nodes of the trees. The 3D-structure of the BdCOMT6 protein was predicted from the crystallized ryegrass enzyme [24] with the molecular modeling program Geno3D (http://geno3d-pbil.ibcp.fr/) and UCSF Chimera software (http://www.cgl.ucsf.edu/chimera/).
Lignin Analyses
All reagents and solvents were high-quality grade commercial reagents employed without further purification. Dried mature stems (3 month old) from wild-type (WT) plants or from azygous (mutagenized plants seggregating the WT allele for the studied locus) or homozygous mutant plants were ground to 0.5 mm before exhaustive extraction with water, then ethanol in a Soxhlet apparatus. The lignin content was measured on the recovered extractive-free samples and by the Klason method [25]. Lignin structure was evaluated by thioacidolysis performed from 10 to 20 mg of extractive-free sample put together with 0.5 mg of C21 internal standard in 10 mL of reagent (dioxane/ethanethiol 9/1 V/V, containing 0.2 M BF3 etherate) and incubated at 100°C for 4 h. After the reaction, the lignin-derived monomers were extracted with methylene chloride as previously described [26], the combined organic extracts were concentrated to about 2 mL and then 4 µL of the sample were silylated by 100 µL BSTFA and 5 µL pyridine before injection onto a DB1 supelco capillary columns (carrier gas helium, constant flow rate 1 mL/min) operating from 40 to 180°C at +30°C/min, then 180 to 260°C at +2°C/min and combined with an ion trap mass spectrometer (Varian Saturn2100) operating in the electron impact mode (70 eV), with ions detected on the 50–600 m/z range. The surface area of the p-hydroxyphenyl (H), guaiacyl (G), syringyl (S) and 5-hydroxyguaiacyl (5-OH G) monomers were measured on specific ion chromatograms, at m/z 239, 269, 299, and 357 respectively.
Results and Discussion
Production of a Mutagenized Collection
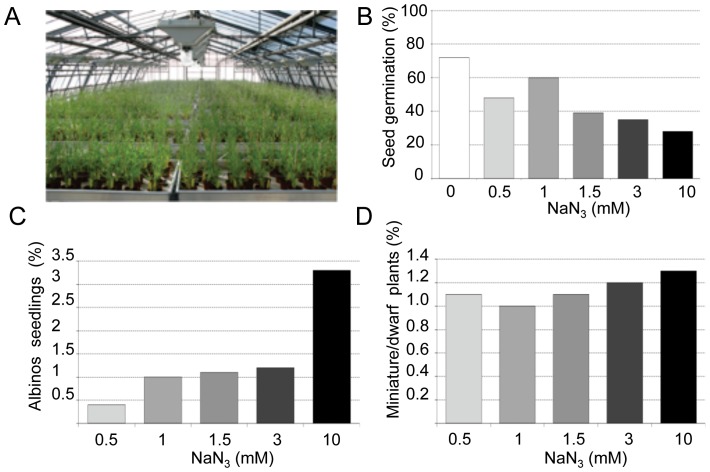
When grown under long-day conditions in our greenhouses (18 h light, Figure 1A), the life cycle was about 12 weeks. Sodium azide (NaN3) was used to mutagenize B. distachyon Bd21-3 seeds. This compound is commonly used for mutagenesis of grasses [15] and the frequency of chromosome breakage is relatively low. To establish a dose-response kill curve, we determined the fraction of M1 seeds that germinated after imbibition with increasing NaN3 concentrations (Figure 1B). The lowest germination frequency was 28% for 10 mM NaN3. The frequency of albino M2 seedlings (Figure 1C) and miniature/dwarf plants (Figure 1D) also showed a dose-dependent increase with for instance a maximum frequency of 3.3% and 1.3% albinos respectively for 10 mM and 3 mM NaN3 (Figure 1D). We concluded that 3 mM and 10 mM NaN3 provided a good compromise combining high mutation rate and sufficient germination frequency. These concentrations were used to generate the mutagenized population of 5530 individual families.
Figure 1. Production of Brachypodium mutants.
A. 3000 M1 mutagenized individual plants growing in greenhouse. B. Percentage of seeds germinating after 2 hours imbibition in different NaN3 concentrations. C. Percentage of M2 albino seedlings observed according to NaN3 concentrations. D. Percentage of M2 dwarf/miniature plants observed according to NaN3 concentrations. In each experiment (B–D) 100 seeds per concentration were sowed.
Plant Phenotyping
The phenotypes (shape, color and size) of a subset of mutants were recorded, using an ontology developed for sorghum [27], at three developmental stages: Germination/seedling stage, vegetative stage (before flowering) and reproductive stage (after flowering). Phenotypes were organized in a phenotypic tree available at http://urgv.evry.inra.fr/UTILLdb. Figure 2 and Table 1 illustrate the most striking phenotypes of greenhouse-grown mutants.
Figure 2. Examples of phenotypes detected in the Brachypodium mutant collection.
A. Spikelets of wild-type plants. B–D Aborted or abnormal spikelets in mutants. E. Ligule of wild-type plant. F. Abnormal ligule in mutant. G, H. Mis-shaped leaves in mutants. I. Crumple stem in mutant. J. Coloured stem in mutant. K. adventitious roots in mutant. L. Curved stems in mutant. M. Segregant phenotype for tiller formation in M2 plants compared to WT (right). N. Floppy stems in mutant.
Table 1. Types of phenotypes observed on 565 phenotyped lines (3360 plants).
| Major category | Phenotype sub-categorya | Number of families affected | % of families affected |
| Dead | Not emerged or early death | 340 | |
| Plant devlopment | Delay Plant development | 87 | 15.4 |
| Plantlet | Albino | 20 | 3.5 |
| Leaf | Size | 129 | 22.8 |
| Color | 121 | 21.4 | |
| Appearance | 125 | 22.1 | |
| Shape and arrangements | 9 | 1.6 | |
| Stem | Shape | 112 | 19.8 |
| Color | 19 | 3.3 | |
| Node | Shape | 1 | 0.2 |
| Spike | Flowering time | 52 | 9.2 |
| Spike organisation | 62 | 11 | |
| Reproductive organs | 15 | 2.6 | |
| Spike color | 62 | 11 | |
| Plant architecture | Architecture | 394 | 69.7 |
| Height and appearance | 146 | 25.8 | |
| Tillers | 70 | 12.4 | |
| Branching type | 47 | 8.3 |
phenotypes are available at http://urgv.evry.inra.fr/UTILLdb.
TILLING Genes Involved in Lignin Biosynthesis
B. distachyon is increasingly used as a model for bioenergy grasses [28], [29] Lignins are the main obstacle in the enzymatic conversion of cellulose into fermentable sugars. We therefore chose to screen the mutant collection for genes involved in lignin biosynthesis as a validation of the tilling method [29]. We focused on seven genes belonging to multigene families that are potentially involved in monolignol biosynthesis (Table 2): one caffeic acid O-methyltransferase gene (Bradi3g16530), two laccase genes (Bradi1g74320, Bradi1g66720), one 4-coumarate:coenzyme A ligase gene (Bradi3g05750) and three cytochrome P450 genes (Bradi2g31510, Bradi2g53470, Bradi3g43160).
Table 2. Tilled genes and mutation frequency in the mutant collection with 5530 M2 families screened.
| Target names | Accession number | Amplicon size (bp) | GC content (%) | Identified mutants | Mutation frequency |
| COMT | Bradi3g16530 | 1245 | 65 | 23 | 1/300 kb |
| Laccase | Bradi1g74320 | 2372 | 43 | 17 | 1/771 kb |
| Laccase | Bradi1g66720 | 2607 | 51 | 18 | 1/801 kb |
| 4CL | Bradi3g05750 | 735 | 68 | 17 | 1/239 kb |
| C4H | Bradi2g31510 | 643 | 65 | 14 | 1/254 kb |
| C4H | Bradi2g53470 | 728 | 63 | 12 | 1/335 kb |
| C4H | Bradi3g43160 | 829 | 61 | 27 | 1/170 kb |
| TOTAL | 9159 | 59 | 128 | 1/396 kb |
We identified a total of 128 mutations. The nucleotide changes induced by NaN3 are mainly G/A and C/T substitutions similar to those induced by ethyl methane sulfonate (EMS) for instance [30], [31]. Only 9% other mutations were detected (Table 3). We identified 63% non-synonymous mutations (including 53% and 5% inducing amino acid changes or stop codons, respectively). One line showed a mutation in a splicing region. Extrapolating from this small set of genes, we calculated an average mutation rate of one in around 400 kb (Table 2) or 700 mutations per genome for a genome size of 272 Mb [2], This frequency of induced mutations is similar to that found in EMS-mutagenized rice (1/294 kb, [23]) or in barley treated with NaN3 (1/374 kb, [22]) or EMS (1/500 kb, [20]).
Table 3. Frequencies of induced mutations types in tilled gene-coding regions.
| Accession number | Missensea | Nonsenseb | Splicingc | Silentd | Unusual nucleic transitione | |
| COMT | Bradi3g16530 | 13 | 0 | 0 | 10 | 0 |
| Laccase | Bradi1g74320 | 8 | 3 | 0 | 6 | 0 |
| Laccase | Bradi1g66720 | 8 | 0 | 1 | 5 | 3 |
| 4CL | Bradi3g05750 | 13 | 0 | 0 | 4 | 4 |
| C4H | Bradi2g31510 | 10 | 1 | 0 | 3 | 1 |
| C4H | Bradi2g53470 | 6 | 1 | 0 | 4 | 1 |
| C4H | Bradi3g43160 | 13 | 1 | 0 | 13 | 2 |
| TOTAL | 71 | 6 | 1 | 45 | 11 | |
| TOTAL (%) | 57.7 | 4.8 | 0.8 | 36.7 | 8.6 |
nucleic acid transition is a non-synonymous mutation and induce amino acid change in the translated protein.
nucleic acid transition produces a stop codon and may induce a truncated protein.
nucleic acid transition is located in splicing motif.
nucleic acid transition induces a synonymous mutation and then no change in the translated protein.
other nucleic acid transition than guanine to adenine and cytosine to thymine.
We also used our mutagenized population to estimate the ‘Genetically Effective Cell Number (GECN)’. This is the number of cells within the shoot meristem of the embryo that will contribute to the seed output. The GECN is usually estimated by determining the proportion of mutant seedlings in M2 families [32], [33]. We took advantage of our sequencing data on M2 families from individual M1 plants segregating mutations in 4 genes. In 584 lines from 30 different families (between 9 and 49 M2 independent lines were analyzed per family) we detected 38 homozygous mutations corresponding to a ratio of 1∶15.3 ( = 38/584). The 1∶15 ratio corresponds to a GECN of 4. This is higher than in Arabidopsis (GECN = 2) but in accordance with the number found in other grass species [33], [34].
Identification of a Caffeic acid O-methyltransferase (COMT) Gene Potentially Involved in Internode Lignification
B. distachyon lignins, like those in other grasses, are mainly composed of guaiacyl (G) and syringyl (S) units, with low amounts of p-hydroxyphenyl (H) units [35] [36]. These H, G and S lignin units respectively originate from the three monolignols, namely p-coumaryl, coniferyl, and sinapyl alcohols that differ only in the degree of methoxylation of the phenolic ring [37]. The main role of the COMT involved in lignification is the methylation of 5-hydroxyconiferaldehyde to produce sinapaldehyde, which is reduced by another enzyme to sinapyl alcohol, the precursor of S lignin units. The COMT enzyme belongs to the S-Adenosyl methionine (SAM)-dependent O-methyltransferases. It is active as a homodimer and does not need any metal ion as cofactor. The hallmark of transgenic or mutant angiosperms with strongly repressed COMT activity is a reduction of the amount of S lignin units (or of the S/G ratio) together with the appearance of easily detectable amounts of 5-hydroxyguaiacyl (5-OH G), which is present only in trace amounts in the WT [38] [39], [40], [41], [42]. Another trait of some COMT-mutant grass lines, referred to as the brown-midrib mutants, is a lower lignin level resulting in a higher enzymatic degradability [43], and reduced levels of p-coumaric acid (CA) ester-linked to the cell walls [44].
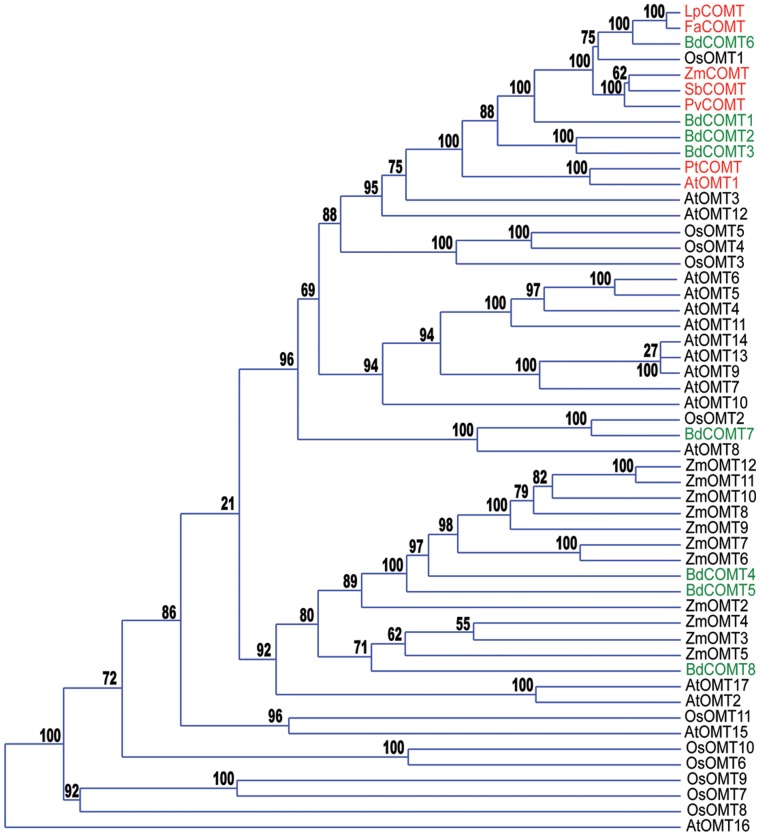
To unambiguously identify the B. distachyon COMT gene specifically involved in lignification, protein sequences of orthologs in several species were BLASTed onto the B. distachyon predicted proteome sequence. Eight proteins were identified: BdCOMT1 (Bradi1g14870), BdCOMT2 (Bradi2g02380), BdCOMT3 (Bradi2g02390), BdCOMT4 (Bradi2g19830), BdCOMT5 (Bradi2g19850), BdCOMT6 (Bradi3g16530), BdCOMT7 (Bradi3g55890), BdCOMT8 (Bradi4g20020). We performed a phylogenetic analysis with the most exhaustive list of encoded OMT proteins found in Oryza sativa, Arabidopsis thaliana and Zea mays genomes [45], [46], [47] (Figure 3, Information S1). In addition, we added to the phylogenetic analysis the protein sequences for which biological data (transgenics or mutants) support indisputably a role of the corresponding protein in lignification of several grass (maize, ryegrass, tall fescue, switchgrass, sorghum) and poplar [24], [39], [42], [48], [49], [50], [51]. AtOMT1 was clearly identified by our group as a unique gene involved in sinapyl alcohol biosynthesis in Arabidopsis since the knockout mutant displays a lignin devoid of S units [52]. Therefore, AtOMT1 is a reference model for dicot COMT proteins as well as maize ZmCOMT1 (BM3) is a reference for grasses [49]. The phylogenetic analysis shows that both dicot proteins (AtOMT1, PtOMT) cluster together whereas four members of BdCOMTs (BdCOMT3, BdCOMT2, BdCOMT1 and BdCOMT6) are grouped with the genuine grass COMT proteins, BdCOMT6 being the closest ortholog (figure 3). Consequently, BdCOMT6 was chosen for TILLING. It is worth noting, that despite no comt mutant was identified in rice to our knowledge this analysis suggests strongly that OsOMT1 (Os08g06100) is involved in monolignol formation.
Figure 3. Phylogenetic analysis of putative COMT proteins.
Phylogeny tree (phylogram) made with OMT proteins from Brachypodium (BdCOMT), rice (OsOMT), maize (ZmOMT) and Arabidopsis (AtOMT). The proteins known to be involved in lignification in ryegrass (LpCOMT), sorghum (SbCOMT), switchgrass (PvCOMT), fescue (FaCOMT) and poplar (PtCOMT) are included in the analysis and shown in red in the phylogram as well as Arabidopsis (AtOMT1) and Maize (ZmCOMT1) proteins. Brachypodium proteins (BdCOMT) are shown in green. Protein sequences are available in Information S1. Bootstrap values indicating the level of support for the displayed representation after re-sampling are shown on each node.
Mutations in BdCOMT6 Affect the Lignification of Mature Stems
We identified 25 lines, corresponding to 22 different mutations, among which eleven missense mutations cause changes in the BdCOMT6 amino acid sequence (Table 4). No induced stop codon mutation were identified. A first analysis using the SIFT software (using 0.05 as significant threshold) indicated that the substitutions caused by the mutations in lines Bd6840, Bd4688, Bd4604, Bd7480, Bd5139, Bd7549, Bd7391, Bd4142 and Bd4927 may partially or totally disrupt the COMT activity. Among them, mutations in lines Bd5139 and Bd7549 were found redundant, as well as those in lines Bd7480 and Bd4604, thereby reducing the number of mutations potentially affecting COMT activity to seven.
Table 4. Allelic series of mutations in BdCOMT6 gene identified by TILLING.
| Nucleic acid transitiona | Amino acid substitutionb | SIFTc | Family name |
| G37A | Asp13Asn | 0.18 | 5645 |
| G117A | Leu39Leu | – | 5714 |
| C498T | Asp166Asp | – | 5588 |
| G588A | Glu196Glu | – | 8240 |
| C600T | Tyr200Tyr | – | 5827 |
| G616A | Gly206Ser | 0.01 | 6840 |
| G638A | Gly213Asp | 0.26 | 3380 |
| G638A | Gly213Asp | 0.26 | 211 |
| G672A | Gly224Gly | – | 3725 |
| G708A | Gly236Gly | – | 5338 |
| G721A | Gly241Arg | 0.02 | 4688 |
| G737A | Gly246Asp | 0 | 4604 |
| G737A | Gly246Asp | 0 | 7480 |
| C762T | Pro254Pro | – | 5115 |
| G767A | Gly256Asp | 0 | 5139 |
| G767A | Gly256Asp | 0 | 7549 |
| C840T | Cys280Cys | – | 5348 |
| C854T | Pro285Lys | 0.02 | 7391 |
| G969A | Gly323Gly | – | 3730 |
| G976A | Glu326Lys | 0 | 4142 |
| G1013A | Glu338Asp | 0.14 | 5200 |
| G1063A | Ala355Thr | 0.01 | 4927 |
| G1071A | Glu357Glu | – | 185 |
Position of transition in mutants are relative to the starting ATG on the coding sequence.
Position of substitution in mutants are relative to the starting methionine of the encoded protein.
numbers are predictive score from the SIFT software (http://sift.bii.a-star.edu.sg/).
We next isolated homozygous lines for the all comt mutants, except for Bd7480. Indeed, this line failed to produce viable seeds at heterozygous stage. All genotyped plants were indistinguishable from WT plants when grown in the greenhouse except for homozygous Bd211, which was dwarfed. The dwarfism presumably is independent from the mutated COMT allele since Bd3380, which carries the same mutations was not dwarfed (Table 4). We therefore excluded Bd211 from the subsequent analyses. We next studied the lignin composition in mature stems using thioacidolysis (reviewed in [53]). Thioacidolysis identifies H, G and S thioethylated monomers from arylglycerol-β-ether-linked H, G and S units. In addition, it allows the identification of 5-OH G monomers as observed in maize bm3 mutants [54]. Among the various mutants analyzed (data not shown), three lines (Bd4142, Bd4604, and Bd5139), released 5-OH G thioacidolysis monomers in higher amounts, compared to the trace amounts detected in the control lines (Table 5). Together with the increased frequency of 5-OH G thioacidolysis monomers, we found that S monomer levels were substantially reduced (by 30 to 40% of the control value). In contrast, H monomers were obtained as minor components (3–6% range) and no substantial difference between mutants and controls was detected. Based on the S levels or S/G thioacidolysis ratio suggests, we hypothesize that COMT activity is lower in Bd5139 and Bd4142 compared to Bd4604. This hypothesis was further supported by the lignin level of the extractive-free stems as measured by the Klason method. Compared to the corresponding control, this level was reduced by 15%, 10% and 0% in Bd5139, Bd4142 and Bd4604 respectively (Figure 4).
Table 5. Relative frequency (% molar) of p-hydroxyphenyl H. guaiacyl G. syringyl S and 5-hydroxyguaiacyl 5-OH G monomers released by thioacidolysis of mature and extractive-free stems from control (Ctrl) and mutant Bd4142, Bd4604, Bd5139 lines.
| Culture | Sample (na) | % H | % G | % S | % 5-OH G | S/G molar ratio |
| 1 | Azygous Control (4) | 5.9 (0.3) | 37.9 (1.9) | 55.7 (1.0) | 0.5 (0.0) | 1.47 (0.15) |
| Bd4142 (4) | 6.2 (0.5) | 46.5 (3.1)* | 45.3 (3.3)* | 2.0 (0.3)* | 0.98 (0.14)* | |
| 2 | Wild-type Control (2) | 3.5 (0.3) | 30.8 (2.3) | 65.4 (2.7) | 0.2 (0.0) | 2.14 (0.25) |
| Bd4604 (2) | 3.2 (0.0) | 38.4 (0.3)* | 55.1 (3.3)* | 3.3 (0.3)* | 1.44 (0.02)* | |
| 3 | Wild-type Control (5) | 3.5 (0.5) | 31.4 (2.0) | 64.8 (2.4) | 0.3 (0.1) | 2.08 (0.22) |
| Bd5139 (5) | 4.0 (0.6) | 42.4 (0.4)* | 49.5 (1.4)* | 4.1 (1.5)* | 1.17 (0.04)* |
Ctrl (azygous line for Bd4142 and wild-type line for Bd4604 and Bd5139) and corresponding mutant samples were recovered from plants grown together and in identical conditions.
number of replicates. The data represent the means and SD (between brackets). Asterisks indicate significant differences compared to the corresponding Ctrl (ANOVA, value at P<0.01).
Figure 4. Klason lignin (KL) level of extractive-free mature stems of Bd4142, Bd4604 and Bd5139 lines as compared to control samples.
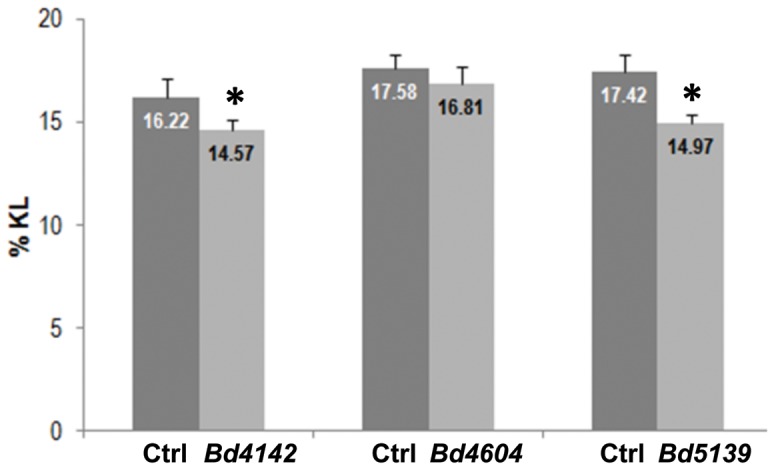
Control samples are either wild-type (Bd21-3) or azygous plants grown together with the corresponding mutants. The KL level is expressed as weight percentage of the extractive-free sample. Data are means and SD from 4 or 5 plants analyzed per line. Asterisks indicate significant difference compared to the control (ANOVA value at P<0.05).
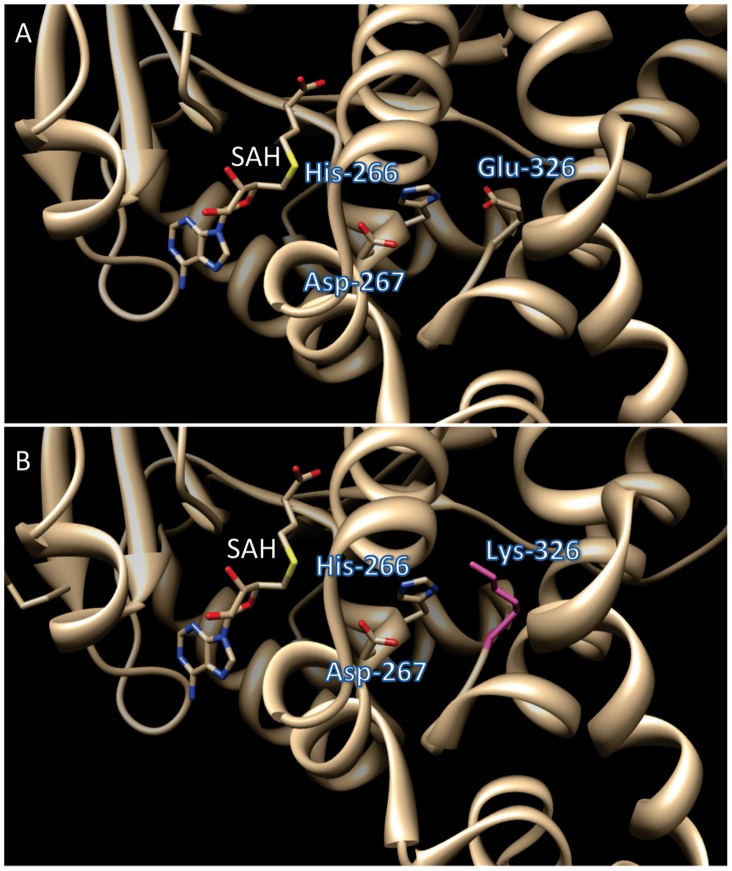
To further investigate how amino acid substitutions may affect BdCOMT6 activity, we used the 3-D structure, determined for the closely related (90% amino acid sequence identity) Lolium perenne OMT, LpOMT [24] to compare WT with mutant proteins. Both COMT enzymes belong to the plant type-1 family of SAM-dependent O-methyltransferases, have 360 amino acid residues and possess an auxiliary N-terminal domain that may functions in homodimerization [55], [24]. The Bd4142 mutation induces a Glu-326-Lys substitution (Figure 5). Glu-326 is thought to be one of the catalytic bases that activate the hydroxyl group of the substrate/ligand [24], whereby Glu-326 provides the hydrogen bond acceptor in an interaction with the His-266 and contributes indirectly to the deprotonation of the phenolic substrate [24]. The Glu-326-Lys substitution, inverts the charge of the aminoacid and therefore is expected to alter proper protein activity and explain the severe reduction of S units detected in this line (Table 5).
Figure 5. Structural representation of the amino acid substitution of BdCOMT6 protein in the line Bd4142.
A. 3-D model of wild-type protein highlighting three amino acids important for proper enzymatic activities. B. 3-D model of the protein in Bd4142 line in which Glu-326 is substitued by Lys-326. SAH, S-Adenosyl-L-homocysteine is shown to illustrate the proximity of the substitued amino acid in the substrat binding pocket.
The substitution observed in lines Bd5139 and Bd7549 is a glycine to aspartic acid at position 256, which is part of a loop facing a β-sheet carrying an amino acid involved in the substrate binding. It is worth noting that two residues (Phe-250 and Asp-248) in the vicinity of Gly-256 are also placed in this loop and are essential for stabilisation of the ligand (SAM/SAH). We speculate that the size and the charge modifications in this mutant could impact the β-sheet and destabilize the SAM binding site, disturbing its function as methyl donor and in consequence reduce the activity of the protein.
Finally, Bd4604 carries a Gly-246-Asp substitution. This residue is located at the periphery of the protein but still in the SAM/SAH binding domain and adjacent to a α-helical layer and a β-sheet both involved in SAM/SAH binding site conformation. The lateral chain of the aspartic acid in the mutant may turn towards residues involved in the binding of the cofactor. The size and charge differences may modify this site thus hampering the binding of SAH and the activity of the protein.
In conclusion, the three mutant lines that have an altered lignin content and/or composition share at least a mutation located in the vicinity of the SAM/SAH binding and catalytic domain. It is worth noting that the three COMT mutants do not show coloured leaf veins as observed in the brown-midrib mutants of maize, pearl millet or sorghum [56]. In addition, the levels of S units is still high in the mutants (as revealed by the 45 to 55% of S thioacidolysis monomers). This result suggests that enzymatic methoxylation at C5 of the phenolic ring of monolignol is still present in these lines. It remains to be shown whether it is due to residual enzymatic activity of the mutated proteins as shown in a similar allelic series in sorghum [57]. Finally, no grass COMT-deficient mutants or transgenic lines described so far are completely devoid of S units, in contrast to what has been reported in dicots, for example in the Arabidopsis Atcomt1 mutant [52]. This observation suggests that an alternative pathway may produce S units in grasses.
Conclusion
We have generated a large collection of chemically-induced mutants useful for forward genetics in B. distachyon. A subset of the phenotypes can be consulted at http://urgv.evry.inra.fr/UTILLdb. In addition, BRACHYTIL provides and efficient platform for reverse genetics. This study illustrates the power of this approach by the isolation of an allelic series for BdCOMT6 involved in monolignol biosynthesis. Next generation sequencing techniques will now greatly accelerate reverse genetics approaches in this collection.
Supporting Information
Amino acid sequences of putative COMT proteins used for phylogenetic analysis in Figure 3 .
(DOCX)
Acknowledgments
We are grateful to Wannes Woorend and Alexia Guillebaux for help with the TILLING of the 4CL and the COMT genes respectively. We thank Pierre Hilson for reading the manuscript and providing useful comments.
Funding Statement
This work was funded by the European FP7 project n° 211982 “RENEWALL” and the Agence Nationale de la Recherche (ANR) projects ANR-08-KBBE-004 “CELLWALL” and ANR-10-BLAN-1528 “PHENOWALL”. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1. Brkljacic J, Grotewold E, Scholl R, Mockler T, Garvin DF, et al. (2011) Brachypodium as a model for the grasses: today and the future. Plant Physiol 157: 3–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Initiative TIB (2010) Genome sequencing and analysis of the model grass Brachypodium distachyon. Nature 463: 763–768. [DOI] [PubMed] [Google Scholar]
- 3.Akhunov E, Sehgal S, Liang H, Wang S, Akhunova A, et al.. (2012) Comparative analysis of syntenic genes in grass genomes reveals accelerated rates of gene structure and coding sequence evolution in polyploid wheat. Plant Physiology. [DOI] [PMC free article] [PubMed]
- 4. Mayer KF, Martis M, Hedley PE, Simkova H, Liu H, et al. (2011) Unlocking the barley genome by chromosomal and comparative genomics. The Plant Cell 23: 1249–1263. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Murat F, Xu JH, Tannier E, Abrouk M, Guilhot N, et al. (2010) Ancestral grass karyotype reconstruction unravels new mechanisms of genome shuffling as a source of plant evolution. Genome research 20: 1545–1557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Păcurar DI, Thordal-Christensen H, Nielsen KK, Lenk I (2007) A high-throughput Agrobacterium-mediated transformation system for the grass model species Brachypodium distachyon L. Transgenic Research. 17: 965–975. [DOI] [PubMed] [Google Scholar]
- 7. Vain P, Worland B, Thole V, McKenzie N, Alves SC, et al. (2008) Agrobacterium-mediated transformation of the temperate grass Brachypodium distachyon (genotype Bd21) for T-DNA insertional mutagenesis. Plant Biotechnology Journal 6: 236–245. [DOI] [PubMed] [Google Scholar]
- 8. Vogel J, Hill T (2008) High-efficiency Agrobacterium-mediated transformation of Brachypodium distachyon inbred line Bd21–3. Plant Cell Reports 27: 471–478. [DOI] [PubMed] [Google Scholar]
- 9. Thole V, Alves SC, Worland B, Bevan MW, Vain P (2009) A protocol for efficiently retrieving and characterizing flanking sequence tags (FSTs) in Brachypodium distachyon T-DNA insertional mutants. Nature protocols 4: 650–661. [DOI] [PubMed] [Google Scholar]
- 10. Thole V, Worland B, Wright J, Bevan MW, Vain P (2010) Distribution and characterization of more than 1000 T-DNA tags in the genome of Brachypodium distachyon community standard line Bd21. Plant biotechnology journal 8: 734–747. [DOI] [PubMed] [Google Scholar]
- 11. Bragg JN, Wu J, Gordon SP, Guttman ME, Thilmony R, et al. (2012) Generation and Characterization of the Western Regional Research Center Brachypodium T-DNA Insertional Mutant Collection. PLoS ONE 7: e41916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Henikoff S, Till BJ, Comai L (2004) TILLING. Traditional mutagenesis meets functional genomics. Plant Physiology 135: 630–636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Wang TL, Uauy C, Robson F, Till B (2012) TILLING in extremis. Plant biotechnology journal 10: 761–772. [DOI] [PubMed] [Google Scholar]
- 14.Chawade A, Sikora P, Brautigam M, Larsson M, Vivekanand V, et al.. (2010) Development and characterization of an oat TILLING-population and identification of mutations in lignin and beta-glucan biosynthesis genes. BMC Plant Biology 10: (12 May 2010). [DOI] [PMC free article] [PubMed]
- 15. Kurowska M, Daszkowska-Golec A, Gruszka D, Marzec M, Szurman M, et al. (2011) TILLING - a shortcut in functional genomics. J Appl Genet 52: 371–390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Slade AJ, Knauf VC (2005) TILLING moves beyond functional genomics into crop improvement. Transgenic Research 14: 109–115. [DOI] [PubMed] [Google Scholar]
- 17. Slade AJ, McGuire C, Loeffler D, Mullenberg J, Skinner W, et al. (2012) Development of high amylose wheat through TILLING. BMC Plant Biology 12: 69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Rawat N, Sehgal SK, Joshi A, Rothe N, Wilson DL, et al. (2012) A diploid wheat TILLING resource for wheat functional genomics. BMC Plant Biology 12: 205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Till BJ, Reynolds SH, Weil C, Springer N, Burtner C, et al. (2004) Discovery of induced point mutations in maize genes by TILLING. BMC Plant Biology 4: 12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Gottwald S, Bauer P, Komatsuda T, Lundqvist U, Stein N (2009) TILLING in the two-rowed barley cultivar ‘Barke’ reveals preferred sites of functional diversity in the gene HvHox1. BMC research notes 2: 258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Weil CF (2009) TILLING in grass species. Plant physiology 149: 158–164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Talame V, Bovina R, Sanguineti MC, Tuberosa R, Lundqvist U, et al. (2008) TILLMore, a resource for the discovery of chemically induced mutants in barley. Plant Biotechnology Journal 6: 477–485. [DOI] [PubMed] [Google Scholar]
- 23. Till BJ, Cooper J, Tai TH, Colowit P, Greene EA, et al. (2007) Discovery of chemically induced mutations in rice by TILLING. BMC Plant Biology 7: 19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Louie GV, Bowman ME, Tu Y, Mouradov A, Spangenberg G, et al. (2010) Structure-function analyses of a caffeic acid O-methyltransferase from perennial ryegrass reveal the molecular basis for substrate preference. The Plant Cell 22: 4114–4127. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dence CW (1992) The determination of lignin. In: Lin S, Y, Dence CW, editors. Methods in Lignin Chemistry: Springer-Verlag. 33–61.
- 26. Lapierre C, Pollet B, Rolando C (1995) New insights into the molecular architecture of hardwood lignins by chemical degradative methods. Research on Chemical Intermediates 21: 397–412. [Google Scholar]
- 27. Xin Z, Wang ML, Barkley NA, Burow G, Franks C, et al. (2008) Applying genotyping (TILLING) and phenotyping analyses to elucidate gene function in a chemically induced sorghum mutant population. BMC Plant Biology 8: 103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Lee S, Warnick T, Pattathil S, Alvelo-Maurosa J, Serapiglia M, et al. (2012) Biological conversion assay using Clostridium phytofermentans to estimate plant feedstock quality. Biotechnology for Biofuels 5: 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Bouvier d’Yvoire M, Bouchabke-Coussa O, Voorend W, Antelme S, Cezard L, et al. (2013) Disrupting the cinnamyl alcohol dehydrogenase 1 gene (BdCAD1) leads to altered lignification and improved saccharification in Brachypodium distachyon. Plant J. 73: 496–508. [DOI] [PubMed] [Google Scholar]
- 30. Till BJ, Colbert T, Tompa R, Enns LC, Codomo CA, et al. (2003) High-throughput TILLING for functional genomics. Methods in molecular biology 236: 205–220. [DOI] [PubMed] [Google Scholar]
- 31. Till BJ, Reynolds SH, Greene EA, Codomo CA, Enns LC, et al. (2003) Large-scale discovery of induced point mutations with high-throughput TILLING. Genome research 13: 524–530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Bretagne-Sagnard B FG, Chupeau Y (1996) Induced albina mutations as a tool for genetic analysis and cell biology in flax (Linum usitatissimum). Journal of Experimental Botany 47: 189–194. [Google Scholar]
- 33. Page DR, Grossniklaus U (2002) The art and design of genetic screens: Arabidopsis thaliana. Nature reviews Genetics 3: 124–136. [DOI] [PubMed] [Google Scholar]
- 34. Gady AL, Hermans FW, Van de Wal MH, van Loo EN, Visser RG, et al. (2009) Implementation of two high through-put techniques in a novel application: detecting point mutations in large EMS mutated plant populations. Plant Methods 5: 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lapierre C (1993) Applications of new methods for the investigation of lignin structure. In: Jung HG, Buxton DR, Hatfield RD, Ralph J, editors. Forage cell wall structure and digestibility. Madison: American Society of Agronomy Inc. 133–163.
- 36.Barriere Y RC, Méchin V, Maltese S, Pichon M, Cardinal A, et al.. (2007) Genetics and genomics of lignification in grass cell walls based on maize as model species. Genes, Genomes and Genomics: 133–156.
- 37. Boerjan W, Ralph J, Baucher M (2003) Lignin biosynthesis. Annual Review of Plant Biology 54: 519–546. [DOI] [PubMed] [Google Scholar]
- 38. Goujon T, Sibout R, Pollet B, Maba B, Nussaume L, et al. (2003) A new Arabidopsis thaliana mutant deficient in the expression of O-methyltransferase impacts lignins and sinapoyl esters. Plant Molecular Biology 51: 973–989. [DOI] [PubMed] [Google Scholar]
- 39. Jung JH, Xiong Y, Kim JY, Fouad W, Vermerris W, et al. (2011) RNA Interference Suppresses Lignin Biosynthetic Genes Caffeic Acid 3-O-methyltransferase (COMT) and/or 4-Coumarate-CoA Ligase (4CL) in Sugarcane. In Vitro Cellular & Developmental Biology-Animal 47: S36–S36. [Google Scholar]
- 40. Robert D, Piersantelli D, Jouanin L, Ferret V, Pollet B, et al. (2000) Effects of CAD and COMT down-regulation on poplar tree lignin revealed by NMR spectroscopy. Abstracts of Papers of the American Chemical Society 219: U270–U270. [Google Scholar]
- 41.Ralph J, Lu F, Marita J, Hatfield R, Lapierre C, et al. 5-Hydroxyconiferyl alcohol as a monolignol in COMT-deficient angiosperms.; 2001 June 11–14 2001,; Nice, France. 27–30.
- 42. Chen L, Auh C-K, Dowling P, Bell J, Lehmann D, et al. (2004) Transgenic down-regulation of caffeic acid O-methyltransferase (COMT) led to improved digestibility in tall fescue (Festuca arundinacea). Functional Plant Biology 31: 235–245. [DOI] [PubMed] [Google Scholar]
- 43. Sattler SE, Funnell-Harris DL, Pedersen JF (2010) Brown midrib mutations and their importance to the utilization of maize, sorghum, and pearl millet lignocellulosic tissues. Plant Science 178: 229–238. [Google Scholar]
- 44. Marita JM, Vermerris W, Ralph J, Hatfield RD (2003) Variations in the cell wall composition of maize brown midrib mutants. Journal of Agricultural and Food Chemistry 51: 1313–1321. [DOI] [PubMed] [Google Scholar]
- 45. Kim BG, Kim DH, Hur HG, Lim J, Lim Y, et al. (2005) O-Methyltransferases from Arabidopsis thaliana. Agric Chem Biotechnol 48(3): 113–119. [Google Scholar]
- 46.Courtial A, Soler M, Chateigner-Boutin AL, Reymond M, Méchin V., et al. Breeding grasses for capacity to biofuel production or silage feeding value: an updated list of genes involved in maize secondary cell wall biosynthesis and assembly. Maydica (in press).
- 47. Hamberger B, Ellis M, Friedmann M, Souza CDA, Barbazuk B, et al. (2007) Genome-wide analyses of phenylpropanoid-related genes in Populus trichocarpa, Arabidopsis thaliana, and Oryza sativa: the Populus lignin toolbox and conservation and diversification of angiosperm gene families. Canadian Journal of Botany-Revue Canadienne De Botanique 85: 1182–1201. [Google Scholar]
- 48. Oliver AL, Pedersen JF, Grant RJ, Klopfenstein TJ (2005) Comparative Effects of the Sorghum bmr-6 and bmr-12 Genes: I. Forage Sorghum Yield and Quality. Crop Sci 45: 2234–2239. [Google Scholar]
- 49. Vignols F, Rigau J, Torres MA, Capellades M, Puigdomenech P (1995) The brown midrib3 (bm3) mutation in maize occurs in the gene encoding caffeic acid O-methyltransferase. Plant Cell 7: 407–416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Grand C, Parmentier P, Boudet A, Boudet AM (1985) Comparison of Lignins and of Enzymes Involved in Lignification in Normal and Brown Midrib (Bm3) Mutant Corn Seedlings. Physiologie Vegetale 23: 905–911. [Google Scholar]
- 51. Tschaplinski TJ, Standaert RF, Engle NL, Martin MZ, Sangha AK, et al. (2012) Down-regulation of the caffeic acid O-methyltransferase gene in switchgrass reveals a novel monolignol analog. Biotechnology for biofuels 5: 71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Goujon T, Sibout R, Pollet B, Maba B, Nussaume L, et al. (2003) A new Arabidopsis thaliana mutant deficient in the expression of O-methyltransferase impacts lignins and sinapoyl esters. Plant Mol Biol 51: 973–989. [DOI] [PubMed] [Google Scholar]
- 53.Lapierre C (2010) Determining lignin structure by chemical degradations. In: Heitner C, Dimmel D, Schmidt JA, editors. Lignin and lignans - Advances in Chemistry. Boca Raton, USA: CRC Press, Taylor & Francis Group. 11–48.
- 54. Lapierre C, Tollier MT, Monties B (1988) Occurrence of additional monomeric units in the lignins from internodes of a brown-midrib mutant of maize bm3. Comptes Rendus de l’Academie des Sciences, III (Sciences de la Vie) 307: 723–728. [Google Scholar]
- 55. Zubieta C, Ross JR, Koscheski P, Yang Y, Pichersky E, et al. (2003) Structural basis for substrate recognition in the salicylic acid carboxyl methyltransferase family. The Plant Cell 15: 1704–1716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Harrington MJ, Mutwil M, Barrière Y, Sibout R (2012) Chapter 3 - Molecular Biology of Lignification in Grasses. In: Lise J, Catherine L, editors. Advances in Botanical Research: Academic Press. 77–112.
- 57. Sattler S, Palmer N, Saballos A, Greene A, Xin Z, et al. (2012) Identification and Characterization of Four Missense Mutations in Brown midrib 12 (Bmr12), the Caffeic O-Methyltranferase (COMT) of Sorghum. BioEnergy Research 5: 855–865. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Amino acid sequences of putative COMT proteins used for phylogenetic analysis in Figure 3 .
(DOCX)