Abstract
Background
Data suggest that weight, and specifically BMI, plays a role in breast cancer development and outcome. We hypothesized there would be a correlation between BMI and clinical outcome in patients with early stage HER2-positive breast cancer enrolled in the N9831 adjuvant trial.
Methods
Patients were grouped according to baseline BMI: normal, BMI < 25; overweight, 25 ≤ BMI < 30; and obese, BMI ≥30. Disease-free survival (DFS) was estimated by the Kaplan-Meier method. Comparisons between treatment arms A, B, and C (chemotherapy +/− trastuzumab) were performed using a stratified Cox proportional hazards model.
Results
Analysis was completed on 3017 eligible patients. Obese patients were more likely to be older and postmenopausal (p<0.0001 for both), have larger tumors (p=0.002) and positive lymph nodes (p=0.004). In the pooled analysis cohort, differences in DFS among the BMI groups were statistically significant (5-year DFS rates were 82.5%, 78.6%, and 78.5% for normal weight, overweight and obese women, respectively; logrank p-value = 0.02). The adjusted HR comparing the DFS of overweight to normal women was 1.30 (95% CI: 1.06 to 1.61) and obese to normal women was 1.31 (95% CI: 1.07 to 1.59). There were no statistically significant differences in DFS by weight group for women within any trial arm.
Conclusion
Patients with early stage HER2 positive breast cancer and normal BMI had a better 5-year DFS compared with overweight and obese women. Adjuvant trastuzumab improves clinical outcome regardless of BMI.
Keywords: obesity, BMI, adjuvant therapy, trastuzumab
Introduction
Obesity, as defined as a body mass index (BMI) ≥30 according to the World Health Organization, is a significant public health issue and has been associated with increased death rates for essentially all cancers, including breast cancer. 1–3 Previous studies have also shown obesity (high BMI) is a risk factor for the development of new cases of breast cancer and also negatively affects disease-free survival (DFS) after a diagnosis of this disease.4–9 Previous studies have also shown that obese patients, when compared to non-obese patients, tend to have increased age, larger tumor size and increased number of axillary lymph node involvement.10, 11 In view of the established relationship between estrogenic levels and obesity, most of the studies have examined the relationship of DFS with BMI and hormone receptor (HR) status estrogen receptor positive, endocrine therapy, and most recently triple negative breast cancer.7–9, 12–15 These studies have shown that among post-menopausal women, an increased BMI is associated with ER+/PR+ status and also an increased risk of breast cancer up to 33%.8, 9 However, data on newer specific subtypes of breast cancer, such as human epidermal growth factor 2 (HER2)-positive disease are not available. HER2-positive breast cancer, defined as protein overexpression or gene amplification, affects approximately 15–20% of patients with invasive breast cancer.16 Moreover, a potential impact of BMI on the efficacy of adjuvant trastuzumab had not been reported, which prompted our study. We hypothesized this could be particularly relevant, based on the cross-signaling pathways between HER2 and other metabolic pathways such as the estrogen and insulin growth factor pathways.17
Our group has conducted a large adjuvant trial (North Central Cancer Treatment Group, now part of the Alliance, N9831) which enrolled over 3500 patients with a diagnosis of HER2-positive early stage breast cancer, allowing us the opportunity to explore the relationship of BMI, HER2-positive breast cancer, and treatment with chemotherapy ± trastuzumab on patient outcome. Our independent N9831 trial, the joint efficacy analysis of the N9831 with the National Surgical Adjuvant Breast Project (NSABP) B-31 clinical trial, as well as other well conducted trials, demonstrated that adding trastuzumab to standard chemotherapy significantly improves disease-free and overall survival in the overall group of patients with HER2-positive early stage breast cancer, and provide the basis for an analysis of potential impact of BMI on various pathological and outcome parameters.18–21
This report presents baseline BMI and its relation to tumor characteristics and disease-free survival (DFS) in patients treated with chemotherapy alone or chemotherapy with trastuzumab who participated in the N9831 adjuvant clinical/translational trial.
Patients and Methods
Patients
The randomized phase III NCCTG N9831 trial enrolled 3505 women aged ≥18 years with primary, operable, node-positive or high risk node negative HER2 positive adenocarcinoma of the breast who were eligible for adjuvant chemotherapy.18, 19 All tumors were required to be HER2-positive by either fluorescence in situ hybridization (FISH) showing gene amplification (≥ 2.0 ratio) or immunohistochemistry (IHC) assay showing a strongly positive (3+, 10% of cells with circumferential membrane staining) score. Complete resection of the primary tumor and axillary-node evaluation were required. All patients gave written, informed consent
Eligible patients enrolled in N9831 were randomly assigned to one of three arms. In arm A (the control arm) patients received doxorubicin and cyclophosphamide (AC; 60/600 mg/m2 every 3 weeks × 4) followed by paclitaxel (P; 80 mg/m2 weekly × 12), in arm B (the sequential arm) patients received similar AC followed by paclitaxel, followed by trastuzumab (4 mg/kg loading dose, then 2mg/kg for 52 weeks), and in arm C (the concurrent arm) patients received AC followed by paclitaxel concurrently with trastuzumab (4 mg/kg loading dose, then 2mg/kg for 11 weeks), then trastuzumab alone (2mg/kg for 40 additional weeks). After completion of chemotherapy, radiation therapy and/or hormonal therapy were given, to all patients, as clinically indicated.18, 19
Methods
BMI (defined as weight in kilograms divided by the square of the height in meters) was calculated from baseline body surface area (BSA) and weight obtained from the baseline case report forms, height was not recorded. The formula used to compute BMI from BSA and weight was BMI = (weight3)/(1296 × BSA4).20 Each study site used their own method for determining a woman’s BSA that was reported on the baseline case report form, from which we calculated the BMI.
Statistical Analyses
The primary analysis of the report herein was to define whether DFS differed by BMI and whether there was a treatment by BMI interaction. The primary analysis categorized BMI as normal weight (BMI < 25), overweight (25 ≤ BMI < 30), and obese (BMI ≥ 30). A second analysis was performed that treated BMI as a continuous variable.
DFS, the primary endpoint, was defined as the time from registration to the first disease-related event. A disease-related event was local, regional or distant breast cancer recurrence, contralateral breast cancer, a new primary cancer (except squamous or basal cell carcinoma of the skin, carcinoma in situ of the cervix, or lobular carcinoma in situ of the breast), or death from any cause.18 DFS was estimated by the Kaplan-Meier methods and DFS curves among different groups were compared with a log-rank test. Comparisons were made of DFS among BMI groups for women pooled across the three treatment arms and within each treatment arm.
Patients were grouped according to BMI status based on their baseline BMI value. Categorical variables between groups (e.g. normal versus overweight versus obese) were compared with a chi-square test. Continuous variables were compared between groups with a two-sample t-test. A stratified Cox proportional hazards analysis, stratified by hormone receptor status and nodal status, and adjusted for race and age was used to generate hazard ratios and 95% confidence intervals (CIs). In addition to determining the effect of treatment and BMI status on DFS, a Cox model was used to assess the potential interaction between treatment arm and BMI status.
A secondary analysis was performed using breast-cancer specific survival (BCSS) as an endpoint. An event was a death due to breast cancer and deaths due to other causes were censored. A two-sided p-value less than 0.05 was considered statistically significant. Statistical analyses were performed using SAS version 9.2.
Results
There were 3505 women enrolled on N9831 and this analysis was performed on an analysis cohort of 3017 women. The 488 women excluded from the original cohort were (1) N = 283 who were not found to have HER2-positive tumors upon central laboratory testing, (2) N = 61 who were ineligible for study entry, (3) N = 28 who withdrew consent prior to receiving any protocol directed treatment, and (4) N = 116 who were lost to follow-up. Within the analysis cohort, 877 women (29%) were classified as normal BMI, 842 women (28%) were classified as overweight, and 1298 women (43%) were classified as obese.
Patient and Tumor Characteristics by BMI Groups
Patient characteristics for the 3017 eligible patients are shown in Table 1. Overweight and obese women were more likely to be older, post-menopausal, and non-white compared to normal weight women. In addition, overweight and obese women were more likely to have larger tumors. Normal and overweight women were more likely to have breast cancer lymph node involvement compared to obese women. A greater proportion of obese women had hormone receptor positive disease compared to overweight and normal weight women but this difference was not statistically significant. No statistically significant differences were observed in predominant tumor histology and histologic tumor grade among the normal weight, overweight, and obese groups.
Table 1.
Patient Characteristics by BMI Groups
| Characteristic | BMI < 25 N=877 |
25 ≤ BMI< 30 N = 842 |
BMI ≥ 30 N=1298 |
p-value |
|---|---|---|---|---|
|
| ||||
| Median age, years (min, max) | 46 (19, 79) | 50 (22, 80) | 51 (23, 82) | <0.0001 |
|
| ||||
| Age Group, n (%) | ||||
| <40 | 209 (24) | 128 (15) | 182 (14) | <0.0001 |
| 40–49 | 326 (37) | 279 (33) | 380 (29) | |
| 50–59 | 235 (27) | 277 (33) | 479 (37) | |
| ≥ 60 | 107 (12) | 158 (19) | 257 (20) | |
|
| ||||
| Race, n (%) | ||||
| White | 779 (89) | 698 (83) | 1055 (81) | <0.0001 |
| Other | 98 (11) | 114 (17) | 243 (19) | |
|
| ||||
| Menopausal Status, n (%) | ||||
| Pre-menopausal or < 50 | 562 (64) | 440 (52) | 608 (47) | <0.0001 |
| Post-menopausal or ≥ 50 | 315 (36) | 402 (48) | 690 (53) | |
|
| ||||
| ER/PR Status, n (%) | ||||
| ER or PR Positive | 458 (52) | 439 (52) | 721 (56) | 0.19 |
| ER and PR negative | 419 (48) | 403 (48) | 577 (45) | |
|
| ||||
| Nodal Status, n (%) | ||||
| Node Negative | 105 (12) | 98 (12) | 200 (15) | 0.016 |
| Node Positive | 772 (88) | 744 (88) | 1098 (85) | |
|
| ||||
| Predominant Tumor Histology, n (%) | ||||
| Ductal | 827 (94) | 796 (95) | 1229 (95) | 0.94 |
| Lobular | 27 (3) | 24 (3) | 41 (3) | |
| Mucinous | 5 (0.6) | 1 (0.1) | 6 (0.5) | |
| Papillary | 1 (0.1) | 2 (0.2) | 2 (0.2) | |
| Medullary | 2 (0.2) | 3 (0.4) | 6 (0.5) | |
| Intraductal | 1 (0.1) | 1 (0.1) | 1 (0.1) | |
| Other | 13 (1) | 13 (2) | 13 (1) | |
| Missing | 1 | 2 | 0 | |
|
| ||||
| Histologic Tumor Grade (Elston/SBR), n (%) | ||||
| Well/Intermediate | 260 (30) | 241 (29) | 357 (28) | 0.55 |
| Poor | 617 (70) | 601 (71) | 941 (72) | |
|
| ||||
| Path Tumor Size, n (%) | ||||
| < 2 cm | 352 (40) | 259 (31) | 391 (30) | <0.0001 |
| ≥ 2 cm | 525 (60) | 583 (69) | 907 (70) | |
Treatment Characteristics
Table 2 compares the treatment characteristics (type of surgery, use of hormonal therapy, and timing of trastuzumab) among the three groups determined by baseline BMI classifications. Obese women were more likely to undergo breast conserving surgery (BCS) compared to normal weight and overweight women. The differences in the proportions of women who received hormonal treatment among the three groups were not significant. The proportions of normal weight, overweight, and, obese women did not differ across treatment groups (p=0.86).
Table 2.
Treatment Summary by BMI
| Treatment | BMI < 25 N=877 |
25 ≤ BMI < 30 N = 842 |
BMI ≥ 30 N=1298 |
p-value |
|---|---|---|---|---|
|
| ||||
| Surgery, n(%) | ||||
| Breast Conserving | 296 (34) | 302 (36) | 565 (44) | <0.0001 |
| Mastectomy | 581 (66) | 540 (64) | 733 (56) | |
|
| ||||
| Received Hormonal Treatment, n(%) | ||||
| Yes | 447 (51) | 422 (50) | 702 (54) | 0.12 |
| No | 426 (49) | 417 (50) | 587 (46) | |
| Missing | 4 | 3 | 9 | |
|
| ||||
| Treatment arm, n (%) | ||||
| A: No trastuzumab | 297 (34) | 281 (33) | 455 (35) | 0.86 |
| B: Sequential trastuzumab | 304 (35) | 305 (36) | 443 (34) | |
| C: Concurrent trastuzumab | 276 (32) | 256 (30) | 400 (31) | |
BMI and Survival
There were 647 disease-related events in the analysis cohort of 3017 women. At the time of analysis, 360 women in the analysis cohort had died of which 292 deaths were due to breast cancer. The median follow-up for women who remain disease-free was 5.3 years.
In the pooled analysis cohort (i.e. pooling patients across the three treatment arms), the difference in DFS among normal weight, overweight, and obese women was statistically significant (Table 3; Figure 1): the adjusted HR comparing the DFS of overweight women to normal weight women was 1.30 (95% CI: 1.06 to 1.61) and the adjusted HR comparing the DFS of obese women to normal weight women was 1.31 (1.07 to 1.59). The 5-year DFS rates were 82.5%, 78.6% and 78.5% for normal weight, overweight, and obese women, respectively. However, the DFS did not appear to differ significantly between overweight and obese women for the pooled cohort.
Table 3.
DFS within Treatment Arms Based on BMI
| Arm* | Subgroup | N/# Events 3017/647 |
HR (95% CI) | p-value | DFS 5 yr |
|---|---|---|---|---|---|
|
| |||||
| Pooled: Arms A, B, and C | BMI < 25 | 877/161 | 1 (ref) | 0.025 | 82.5 |
| 25≤BMI<30 | 842/195 | 1.27 (1.03, 1.57) | 78.6 | ||
| BMI≥30 | 1298/291 | 1.28 (1.05, 1.55) | 78.5 | ||
|
| |||||
| Arm A: No trastuzumab | BMI < 25 | 297/69 | 1 (ref) | 0.54 | 77.1 |
| 25≤BMI<30 | 281/82 | 1.20 (0.87, 1.65) | 73.3 | ||
| BMI≥30 | 455/122 | 1.11 (0.83, 1.50) | 73.3 | ||
|
| |||||
| Arm B: Sequential trastuzumab | BMI < 25 | 304/49 | 1 (ref) | 0.10 | 85.1 |
| 25≤BMI<30 | 305/68 | 1.40 (0.97, 2.03) | 77.9 | ||
| BMI≥30 | 443/96 | 1.42 (1.00, 2.01) | 80.3 | ||
|
| |||||
| Arm C: Concurrent trastuzumab | BMI < 25 | 276/43 | 1 (ref) | 0.54 | 85.4 |
| 25≤BMI<30 | 256/45 | 1.17 (0.77, 1.78) | 85.1 | ||
| BMI≥30 | 400/73 | 1.23 (0.84, 1.81) | 78.9 | ||
Stratified by Hormone Receptor Status and Nodal Status
Arm B interaction p-value = 0.24
Arm C interaction p-value = 0.69
Figure 1.

Disease-free survival by BMI group for patients pooled across treatment arms
The effect of treatment did not statistically differ by BMI classified as normal, overweight, and obese. The treatment arm, by obesity status interaction terms, was not significant, although this analysis is underpowered. Table 3 contains a summary of the DFS by the three BMI groups within each treatment arm and figures 2A, 2B, and 2C display the DFS among the BMI groups within each treatment arm: the control group that did not receive trastuzumab (Arm A), the group that received trastuzumab sequentially (Arm B) and the group that received trastuzumab concurrently (Arm C), respectively. It appears as though normal weight women had better DFS experience than overweight and obese women on the control arm (Figure 2A) and the sequential trastuzumab arm (Figure 2B), although the differences among the three groups were not statistically significant. Of note, there does not appear to be a difference in the DFS experience among the three BMI groups on the concurrent trastuzumab arm (Figure 2C); in particular, the normal weight women do not appear to have a better DFS than the other two groups.
Figure 2.


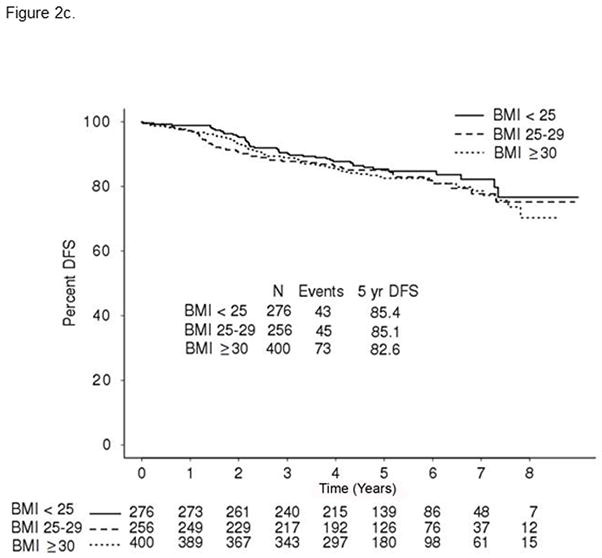
Figure 2a: Disease-free survival BMI group for treatment Arm A: No trastuzumab
Figure 2b: Disease-free survival BMI group for treatment Arm B: Sequential trastuzumab
Figure 2c: Disease-free survival BMI group for treatment Arm C: Concurrent trastuzumab
We also analyzed BMI as a continuous variable (results not shown). The BMI by treatment variable interaction terms were again all non-significant.
The conclusions were similar for the BCSS analysis. In the pooled analysis, there were differences among the three BMI groups but they were not statistically significant: the 5-year BCSS rates were 93.0%, 91.3%, and 91.3% for normal weight, overweight, and obese women (p-value = 0.13). The effect of treatment did not differ by BMI group, the treatment arm by BMI group interaction terms were not statistically significant. The survival curves of the three BMI group for each treatment arm appear similar to those shown in Figure 2 (data not shown). The differences in BCSS for the weight groups within each arm were not statistically significant: p-value = 0.29 for arm A, 0.11 for Arm B, and 0.67 for Arm C.
Discussion
This is the first study that specifically looks at the relationship of BMI with the HER2-positive biological subtype of early stage breast cancer and its association with adjuvant therapy including trastuzumab. Our data from the N9831 study demonstrated an association between BMI, certain breast cancer features, and clinical outcome. In regards to breast cancer features, overweight and obese patients with HER2-positive tumors were more likely to be older than 50 years, post-menopausal, and more likely to have larger tumors. However, normal and overweight women were the ones more likely to have positive lymph node involvement. We also noted that there was a trend (although not statistically significant), for a greater proportion of obese women to have hormone receptor negative disease compared to non-obese women, which was a somewhat surprising finding compared to what we had expected.
Regarding the analysis of BMI and clinical outcome, our study demonstrated that, when taking into account all patients across the 3 treatment arms, there is a statistically significant correlation between rising BMI and worse clinical outcome in patients with early stage HER2 positive breast cancer. In this overall study population, those patients with normal BMI had a better 5-year DFS compared with overweight and obese women (82.5% vs. 78.5%). Considering that overweight and obese patients in this cohort were more likely to be post-menopausal, these results are similar to the experience reported by many national and international groups, especially in women with early stage hormone receptor positive breast cancer.4, 22, 23
We also analyzed the relationship of BMI with the type of adjuvant therapy received. One of the major weaknesses of the study is that it is underpowered to be able to detect small statistical differences between the 3 different treatment arms and their interaction with the 3 BMI groups and their respective clinical outcome. Although results did not reach statistical significance, we did note that on those patients who did not receive adjuvant trastuzumab (arm A), normal weight women had better DFS experience than overweight and obese women. Such difference was no longer noted in the concurrent trastuzumab arm (C). Despite the caveats previously mentioned, this probably represents an important finding of this study: that adjuvant trastuzumab seems to improve clinical outcome of all patients regardless of their BMI. Although DFS might have been unduly affected by co-morbidities and new events, the fact that the addition of trastuzumab seemed to negate much of the impact of obesity argues that this is not the case and that there is some real interaction between BMI and breast cancer outcomes.
Sparano et al recently evaluated the relation between BMI and outcomes in 6885 patients enrolled in 3 adjuvant trials coordinated by the Eastern Cooperative Oncology Group (E1199, E5188, and E3189).12 When evaluated as a categorical variable, obesity was associated with inferior DFS (HR, 1.24; 95% confidence interval [CI], 1.06–1.46; P = .0008) and OS (HR, 1.37; 95% CI, 1.13–1.67; P = .002) in hormone receptor-positive disease, but not other subtypes (HER2 positive or triple negative). However, the outcome difference may be due to the smaller sample size (n = 940), as only the patients from the E1199 trial had HER2 testing routinely performed, and the fact that the multivariate analysis for interaction of BMI and clinical outcome (DFS, OS and BCSS), was only analyzed in the context of obese (BMI ≥ 30 Kg/m2; n = 302) vs. non-obese (BMI < 30 Kg/m2; n = 597) patients. We also noted that we lost statistical significance when analysis was not taking into account the 3 different BMI subgroups (data not shown).
The exact biologic cause-effect relationship between increased BMI and risk of breast cancer development, tumor characteristics and risk of disease recurrence in patients with early stage breast cancer in HER2-positive and other subtypes of breast cancer is a matter requiring additional studies. Several hypotheses to explain the relationship of BMI etiology with breast cancer incidence and characteristics have been brought forth including sex steroids, insulin level, insulin-like growth factors and their downstream Ras and MAP kinase pathways, adipokines, hypoxia, genetic susceptibility, migrating adipose stem cells, and leptin, amongst others. 22, 24–30 It has also been hypothesized that increased concentrations of circulating estrogen may contribute to these differences, partially due to the peripheral aromatization of androstenedione to estrogen in adipose tissue. 26, 31 The increased concentration of circulating estrogens could also be due to lower levels of sex hormone-binding globulin (SHBG) in obese postmenopausal women.27 Insulin-like growth factor receptor (IGFR) signaling is involved in the growth and resistance to therapy of breast cancer cells. Expression of IGFR has been correlated with obesity, with increased IGF-binding protein 3 expression correlated with increasing BMI.24, 28 IGF also plays an important role in signaling with HER2 with cross-talk between IGFR and HER2 present in trastuzumab resistant cells.32 Leptin and its associated receptor are overexpressed in breast cancer and are involved in cross talk with the HER2 pathway and have been associated with obesity and worse outcomes. 22, 29, 33 Future studies to expand the literature regarding the effect of weight reduction towards normal BMI (and influence pathways that may impact cellular effects of obesity such as insulin and exercise) for patients diagnosed with breast cancer (and other cancers) while obese deserve significant priority. 22, 34, 35
Putting our data in clinical perspective, we observed that adjuvant trastuzumab led to an improvement in DFS in all patients, regardless of their BMI, as shown in Figure 1a, 1b, 2a, and 2b. However, the patients with the best clinical outcome were those with normal weight treated with concurrent (Arm C) trastuzumab and chemotherapy (5-year DFS of 85.4%), followed by the other patients also in the concurrent trastuzumab treatment but grouped as either overweight or obese (5-year DFS of 83.6% and 82.6% respectively), then normal weight patients without trastuzumab therapy (5-year DFS of 77.1%), and then overweight/obese patients not receiving trastuzumab (5-year DFS of 73.3%). It remains uncertain whether weight loss intervention programs after a breast cancer diagnosis could meaningfully reduce the risk of recurrence. However, these data support the concept that having a normal BMI at the time of diagnosis and being treated with concurrent anti-HER2 therapy plus chemotherapy leads to the best long-term outcome.
Acknowledgments
Support from NIH CA25224 and CA114740, and the Breast Cancer Research Foundation.
References
- 1.Obesity: preventing and managing the global epidemic. Report of a WHO consultation. World Health Organ Tech Rep Ser. 2000;894:i–xii. 1–253. [PubMed] [Google Scholar]
- 2.Ballard-Barbash R, Hunsberger S, Alciati MH, et al. Physical activity, weight control, and breast cancer risk and survival: clinical trial rationale and design considerations. J Natl Cancer Inst. 2009;101(9):630–43. doi: 10.1093/jnci/djp068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Ogden CL, Carroll MD, Kit BK, Flegal KM. Prevalence of obesity and trends in body mass index among US children and adolescents, 1999–2010. JAMA. 2012;307(5):483–90. doi: 10.1001/jama.2012.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Protani M, Coory M, Martin JH. Effect of obesity on survival of women with breast cancer: systematic review and meta-analysis. Breast Cancer Res Treat. 2010;123(3):627–35. doi: 10.1007/s10549-010-0990-0. [DOI] [PubMed] [Google Scholar]
- 5.Reeves GK, Pirie K, Beral V, Green J, Spencer E, Bull D. Cancer incidence and mortality in relation to body mass index in the Million Women Study: cohort study. BMJ. 2007;335(7630):1134. doi: 10.1136/bmj.39367.495995.AE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Morimoto LM, White E, Chen Z, et al. Obesity, body size, and risk of postmenopausal breast cancer: the Women’s Health Initiative (United States) Cancer Causes Control. 2002;13(8):741–51. doi: 10.1023/a:1020239211145. [DOI] [PubMed] [Google Scholar]
- 7.Ewertz M, Gray KP, Regan MM, et al. Obesity and risk of recurrence or death after adjuvant endocrine therapy with letrozole or tamoxifen in the breast international group 1-98 trial. J Clin Oncol. 2012;30(32):3967–75. doi: 10.1200/JCO.2011.40.8666. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Suzuki R, Orsini N, Saji S, Key TJ, Wolk A. Body weight and incidence of breast cancer defined by estrogen and progesterone receptor status--a meta-analysis. Int J of Cancer J Int du Cancer. 2009;124(3):698–712. doi: 10.1002/ijc.23943. [DOI] [PubMed] [Google Scholar]
- 9.Suzuki R, Rylander-Rudqvist T, Ye W, Saji S, Wolk A. Body weight and postmenopausal breast cancer risk defined by estrogen and progesterone receptor status among Swedish women: A prospective cohort study. Int J of Cancer J Int du Cancer. 2006;119(7):1683–9. doi: 10.1002/ijc.22034. [DOI] [PubMed] [Google Scholar]
- 10.Loi S, Milne RL, Friedlander ML, et al. Obesity and outcomes in premenopausal and postmenopausal breast cancer. Cancer Epidemiol Biomarkers Prev. 2005;14(7):1686–91. doi: 10.1158/1055-9965.EPI-05-0042. [DOI] [PubMed] [Google Scholar]
- 11.Haakinson DJ, Leeds SG, Dueck AC, et al. The Impact of Obesity on Breast Cancer: A Retrospective Review. Ann Surg Oncol. 2012;19(9):3012–18. doi: 10.1245/s10434-012-2320-8. [DOI] [PubMed] [Google Scholar]
- 12.Sparano JA, Wang M, Zhao F, et al. Obesity at diagnosis is associated with inferior outcomes in hormone receptor-positive operable breast cancer. Cancer. 2012;118(23):5937–46. doi: 10.1002/cncr.27527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ademuyiwa FO, Groman A, O’Connor T, Ambrosone C, Watroba N, Edge SB. Impact of body mass index on clinical outcomes in triple-negative breast cancer. Cancer. 2011;117(18):4132–40. doi: 10.1002/cncr.26019. [DOI] [PubMed] [Google Scholar]
- 14.Phipps AI, Buist DS, Malone KE, et al. Breast density, body mass index, and risk of tumor marker-defined subtypes of breast cancer. Ann Epidemiol. 2012;22(5):340–8. doi: 10.1016/j.annepidem.2012.02.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Phipps AI, Chlebowski RT, Prentice R, et al. Body size, physical activity, and risk of triple-negative and estrogen receptor-positive breast cancer. Cancer Epidemiol Biomarkers Prev. 2011;20(3):454–63. doi: 10.1158/1055-9965.EPI-10-0974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Perez EA, Roche PC, Jenkins RB, et al. HER2 testing in patients with breast cancer: poor correlation between weak positivity by immunohistochemistry and gene amplification by fluorescence in situ hybridization. Mayo Clin Proc. 2002;77(2):148–54. doi: 10.4065/77.2.148. [DOI] [PubMed] [Google Scholar]
- 17.Gee JM, Robertson JF, Gutteridge E, et al. Epidermal growth factor receptor/HER2/insulin-like growth factor receptor signalling and oestrogen receptor activity in clinical breast cancer. Endocr Relat Cancer. 2005;12 (Suppl 1):S99–S111. doi: 10.1677/erc.1.01005. [DOI] [PubMed] [Google Scholar]
- 18.Perez EA, Suman VJ, Davidson NE, et al. Sequential versus concurrent trastuzumab in adjuvant chemotherapy for breast cancer. J Clin Oncol. 2011;29(34):4491–7. doi: 10.1200/JCO.2011.36.7045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Perez EA, Romond EH, Suman VJ, et al. Four-Year Follow-Up of Trastuzumab Plus Adjuvant Chemotherapy for Operable Human Epidermal Growth Factor Receptor 2-Postive Breast Cancer: Joint Analysis of Data from NCCTG N9831 and NSABP B-31. J Clin Oncol. 2011;29(25):3366–73. doi: 10.1200/JCO.2011.35.0868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mosteller RD. Simplified calculation of body-surface area. N Engl J Med. 1987;317(17):1098. doi: 10.1056/NEJM198710223171717. [DOI] [PubMed] [Google Scholar]
- 21.Peto R, Davies C, Godwin J, et al. Comparisons between different polychemotherapy regimens for early breast cancer: meta-analyses of long-term outcome among 100,000 women in 123 randomised trials. Lancet. 2012;379(9814):432–44. doi: 10.1016/S0140-6736(11)61625-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chlebowski RT. Obesity and breast cancer outcome: adding to the evidence. J Clin Oncol. 2012;30(2):126–8. doi: 10.1200/JCO.2011.39.7877. [DOI] [PubMed] [Google Scholar]
- 23.Ewertz M, Jensen MB, Gunnarsdottir KA, et al. Effect of obesity on prognosis after early-stage breast cancer. J Clin Oncol. 2011;29(1):25–31. doi: 10.1200/JCO.2010.29.7614. [DOI] [PubMed] [Google Scholar]
- 24.Goodwin PJ, Ennis M, Pritchard KI, et al. Insulin-like growth factor binding proteins 1 and 3 and breast cancer outcomes. Breast Cancer Res Treat. 2002;74(1):65–76. doi: 10.1023/a:1016075709022. [DOI] [PubMed] [Google Scholar]
- 25.Goodwin PJ, Ennis M, Pritchard KI, et al. Insulin- and obesity-related variables in early-stage breast cancer: correlations and time course of prognostic associations. J Clin Oncol. 2012;30(2):164–71. doi: 10.1200/JCO.2011.36.2723. [DOI] [PubMed] [Google Scholar]
- 26.Key TJ, Appleby PN, Reeves GK, et al. Body mass index, serum sex hormones, and breast cancer risk in postmenopausal women. J Natl Cancer Inst. 2003;95(16):1218–26. doi: 10.1093/jnci/djg022. [DOI] [PubMed] [Google Scholar]
- 27.Potischman N, Swanson CA, Siiteri P, Hoover RN. Reversal of relation between body mass and endogenous estrogen concentrations with menopausal status. J Natl Cancer Inst. 1996;88(11):756–8. doi: 10.1093/jnci/88.11.756. [DOI] [PubMed] [Google Scholar]
- 28.Probst-Hensch NM, Steiner JH, Schraml P, et al. IGFBP2 and IGFBP3 protein expressions in human breast cancer: association with hormonal factors and obesity. Clin Cancer Res. 2010;16(3):1025–32. doi: 10.1158/1078-0432.CCR-09-0957. [DOI] [PubMed] [Google Scholar]
- 29.Surmacz E. Obesity hormone leptin: a new target in breast cancer? Breast Cancer Res. 2007;9(1):301. doi: 10.1186/bcr1638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kabat GC, Kim M, Caan BJ, et al. Repeated measures of serum glucose and insulin in relation to postmenopausal breast cancer. Int J of Cancer J Int du Cancer. 2009;125(11):2704–10. doi: 10.1002/ijc.24609. [DOI] [PubMed] [Google Scholar]
- 31.Roberts DL, Dive C, Renehan AG. Biological mechanisms linking obesity and cancer risk: new perspectives. Annu Rev Med. 2010;61:301–16. doi: 10.1146/annurev.med.080708.082713. [DOI] [PubMed] [Google Scholar]
- 32.Nahta R, Yuan LX, Zhang B, Kobayashi R, Esteva FJ. Insulin-like growth factor-I receptor/human epidermal growth factor receptor 2 heterodimerization contributes to trastuzumab resistance of breast cancer cells. Cancer Res. 2005;65(23):11118–28. doi: 10.1158/0008-5472.CAN-04-3841. [DOI] [PubMed] [Google Scholar]
- 33.Goodwin PJ, Ennis M, Fantus IG, et al. Is leptin a mediator of adverse prognostic effects of obesity in breast cancer? J Clin Oncol. 2005;23(25):6037–42. doi: 10.1200/JCO.2005.02.048. [DOI] [PubMed] [Google Scholar]
- 34.Center for Disease Control. Overweight and Obesity: The Obesity Epidemic. 2011. pp. 1–3. [Google Scholar]
- 35.Kushi LH, Doyle C, McCullough M, et al. American Cancer Society Guidelines on nutrition and physical activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J Clin. 2012;62(1):30–67. doi: 10.3322/caac.20140. [DOI] [PubMed] [Google Scholar]


