Abstract
OBJECTIVE
Type 2 diabetes in midlife or late life increases the risk of Alzheimer disease (AD), and type 1 diabetes has been associated with a higher risk of detrimental cognitive outcomes, although studies from older adults are lacking. We investigated whether individuals with AD were more likely to have a history of diabetes than matched controls from the general aged population.
RESEARCH DESIGN AND METHODS
Information on reimbursed diabetes medication (including both type 1 and 2 diabetes) of all Finnish individuals with reimbursed AD medication in 2005 (n = 28,093) and their AD-free control subjects during 1972–2005 was obtained from a special reimbursement register maintained by the Social Insurance Institute of Finland.
RESULTS
The prevalence of diabetes was 11.4% in the whole study population, 10.7% (n = 3,012) among control subjects, and 12.0% (n = 3,372) among AD case subjects. People with AD were more likely to have diabetes than matched control subjects (unadjusted OR 1.14 [95% CI 1.08–1.20]), even after adjusting for cardiovascular diseases (OR 1.31 [1.22–1.41]). The associations were stronger with diabetes diagnosed at midlife (adjusted OR 1.60 [1.34–1.84] and 1.25 [1.16–1.36] for midlife and late-life diabetes, respectively).
CONCLUSIONS
Individuals with clinically verified AD are more likely to have a history of clinically verified and medically treated diabetes than the general aged population, although the difference is small.
The prevalence of both type 2 diabetes and Alzheimer disease (AD) is higher in aged populations, and a recent meta-analysis, based on cohort studies, showed that diabetes in midlife or late life increases the risk of AD, independent of obesity and other vascular risk factors (1). Furthermore, impaired glucose regulation predisposes one to cognitive impairment and AD (2–4), and insulin resistance may indicate a higher predisposition to AD (5). Still, it is important to acknowledge that among people with diabetes, cognitive dysfunction may not be due to AD but a consequence of diabetes-related long-term complications and/or long-term hyperglycemia, recurrent hypoglycemic episodes, or disorders of lipid metabolism (6,7).
Previous studies have focused on the role of type 2 diabetes, and the associations between type 1 diabetes and AD have been studied less. Although learning and memory impairments and deficits in problem solving and mental flexibility have been found to be more common in individuals with type 1 diabetes than in the general population (8,9), to our knowledge, no data on older individuals or in a longitudinal life-course perspective have been reported. Possible explanations include the involvement of insulin in AD pathogenesis (10). Although memory functions are improved immediately after an intravenous insulin dose, chronic hyperinsulinemia and insulin resistance may have negative effects on cognition (11,12). The severity of dementia and cognitive decline seem to be more related to decreased insulin secretion than changes in glucose concentrations in patients with early-stage AD (13). Other suggested mechanisms linking type 2 diabetes and AD include inflammatory cytokines, oxidative stress, amyloid-β deposits, and microvascular disease resulting from T2D (14,15). The association between diabetes and cognitive decline among people who already have AD seems to be more complex, as indicated by inconsistent findings showing a faster (16), slower (17,18), and similar rate (19) of cognitive decline among AD patients with diabetes compared with those without diabetes.
The association between vascular risk factors, including diabetes and AD/dementia, has been suggested to be age dependent, i.e., midlife risk factors have a stronger association than late-life risk factors (20). However, many previous studies are limited by the length of follow-up time (21–26), with only a few studies including a follow-up of >10 years (27–29). Furthermore, self-reported measures of diabetes have been used in many previous studies (21–23,25).
We conducted a nationwide case-control study in order to assess whether the findings from previous smaller, possibly selected cohorts are generalizable to a population level. We investigated whether individuals with AD were more likely to have a history of clinically verified and medically treated diabetes, including both type 1 and type 2 diabetes in a register-based study containing all community-dwelling persons with a verified AD diagnosis, residing in Finland on 31 December 2005. We also assessed whether the associations were similar with diabetes diagnosed at midlife and late life.
RESEARCH DESIGN AND METHODS
Data sources
Data were obtained from the Finnish National Prescription Register and Special Reimbursement Register, maintained by the Social Insurance Institution (SII) of Finland. The National Prescription Register contains records of all reimbursed drug purchases of all Finnish residents living in noninstitutionalized settings. The Special Reimbursement Register includes information on all individuals who are entitled to reimbursement of medication for certain chronic diseases, such as AD or diabetes. To be included in the Special Reimbursement Register, the diagnosis must be based on explicit predefined criteria and written documentary evidence, including results of a diagnostic test, such as imaging or blood biochemistry, must be provided to the SII by a physician. Data from these registers have previously been applied in nationwide drug utilization studies (30). In Finland (population, 5.3 million), all citizens are covered by a tax-supported public health system and have access to health services, regardless of age, ethnic background, or socioeconomic status, and individual-level data on purchases of reimbursed prescription medicines and hospital visits are collected and updated continuously on statutory registers (31). Thus, these data are available on all individuals, provided that they have a social security number (i.e., all citizens and noncitizens living in Finland for at least 2 years but excluding persons who were living abroad for >1 year at the time of the study). Each resident of Finland is assigned a unique social security number, and this identification number was used to track information from registers. Changes in social security numbers in the 1970s mean that reliable automated linkage without individual checking and recoding of PINs is possible from 1972 onwards, and thus we included data from 1972 onwards only. Data linkage was performed by SII, and all data were de-identified before submission to the research team. No ethics committee approval was required as de-identified data were used and participants were not contacted.
Identification of cases with AD
All community-dwelling people with a verified AD diagnosis, residing in Finland on 31 December 2005 (n = 28,093) were identified from the Special Reimbursement Register, and a single age-, sex-, and region of residence–matched control subject per AD case subject (n of matched case-control pairs = 28,093) was identified. The age range was 42–101 years. AD diagnosis was based on the National Institute of Neurological and Communicative Disorders and Stroke and the Alzheimer's Disease and Related Disorders Association (NINCDS-ADRDA) and the DSM-IV criteria for AD (32,33). In brief, the main diagnostic criterion is progressive memory loss, supported by abnormal magnetic resonance imaging or cerebrospinal fluid biomarker findings. Alternative explanations for memory impairment, such as severe depression, metabolic disturbances (including hyperglycemia per se), and other forms of dementia, such as vascular dementia, needed to be excluded. The diagnoses were confirmed by a neurologist or geriatrician. Thus, our study population was restricted to clinically verified AD cases. The Finnish Current Care Guidelines recommend that all people with AD are treated with memantine or acetylcholinesterase inhibitors unless there is a specific contraindication (including gastric ulcer/intestinal tract operation <6 months previously or severe asthma) (34). Patients with mild or moderate AD are entitled to reimbursed AD medication, but the reimbursement is not withdrawn if/when the disease progresses.
Extraction of diabetes diagnoses
Information on reimbursed diabetes medication during 1972–2009 was retrieved from the Special Reimbursement Register. Diabetes is diagnosed on the basis of fasting capillary blood glucose concentration (SII reimbursement criterion cutoff is 7.0 mmol/L) or 2-h glucose concentration if an oral glucose tolerance test has been performed (cutoff 11.1 mmol/L) (35). Individuals with type 1 diabetes have been entitled to reimbursed insulin since 1964, whereas the criteria for reimbursed type 2 diabetes medication have varied over time. Type 2 diabetes was first mentioned in 1981 criteria stating that medication for those with type 2 diabetes is reimbursed if they have not benefited from lifestyle modification lasting at least 3 months. In 1994, the criteria for type 2 diabetes was updated to recommend 6-month dietary intervention as a first-line therapy and requesting that the pharmacotherapy for type 2 diabetes be used for at least 6 months, including a report of outcomes, before applying for special reimbursement. People with gestational diabetes have not been entitled to reimbursement, unless the need for pharmacotherapy is prolonged.
The reimbursement register does not contain information on the type of diabetes. Thus, we applied a robust age-based classification so that people who were <40 years of age when they were diagnosed with diabetes were classified as having type 1 diabetes and those who were at least 40 years of age were classified as having type 2 diabetes. This cutoff has been suggested as a simple and relatively reliable method for differentiating people with type 1 and type 2 diabetes (36). People with diabetes diagnosed at <65 years of age were categorized as having diabetes in midlife, and those who were at least 65 years of age when diabetes was diagnosed were classified as having late-life diabetes.
Statistical analyses
Statistical analyses were performed with Stata 12.0 (Stata Corp LP, College Station, TX). The difference in the mean age of AD diagnosis between those with and without diabetes was compared with the Student t test, and differences in the duration of diabetes and age at diabetes diagnosis between case and control subjects were compared with the Kruskal-Wallis test. The association of diabetes with AD was assessed in a case-control study including all 28,093 matched pairs. Crude ORs and pooled ORs according to the history of cardiovascular disease groups (no/yes) were calculated. Matching was taken into account by the Mantel-Haenszel method. Information on cardiovascular conditions (hyper-/hypotension, ischemic heart disease, familial hypercholesterolemia, embolisms and thrombosis, myocardial infarctions, heart failure and cardiac arrests, atherosclerosis, and aneurysms) was obtained from the Special Reimbursement Register and Finnish National Hospital Discharge Register (details available from A.-M.T.).
RESULTS
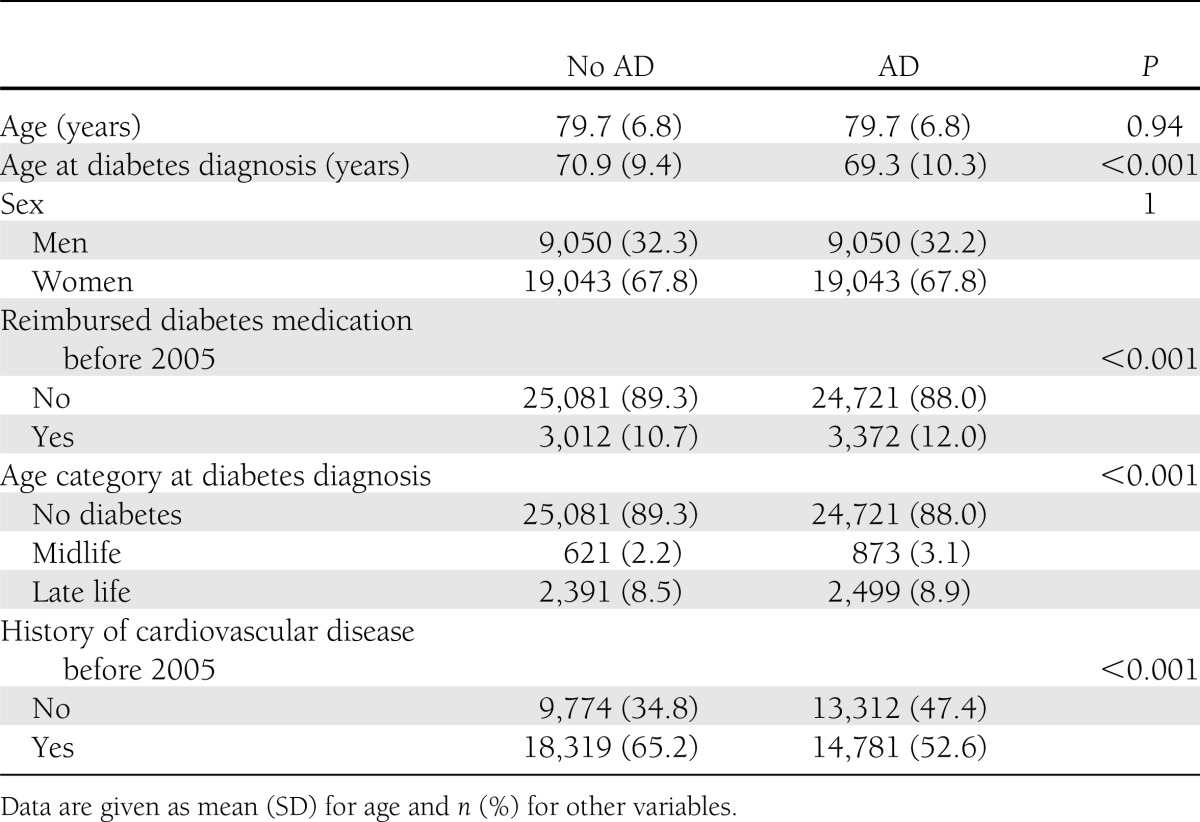
The sample is described in Table 1. The prevalence of diabetes with reimbursed medication in 2005 was 11.4% in the whole study population, 10.7% (n = 3,012) among control subjects (i.e., those without AD), and 12.0% (n = 3,372) among AD cases. The prevalence of cardiovascular diseases was higher in control subjects than AD case subjects. A history of cardiovascular diseases was more common among those with diabetes than those who did not have diabetes (OR 2.91 [95% CI 2.66–3.18], n = 5,159, 80.8%, and n = 27,941, 56.1%, respectively, including those with and without AD).
Table 1.
Sample description
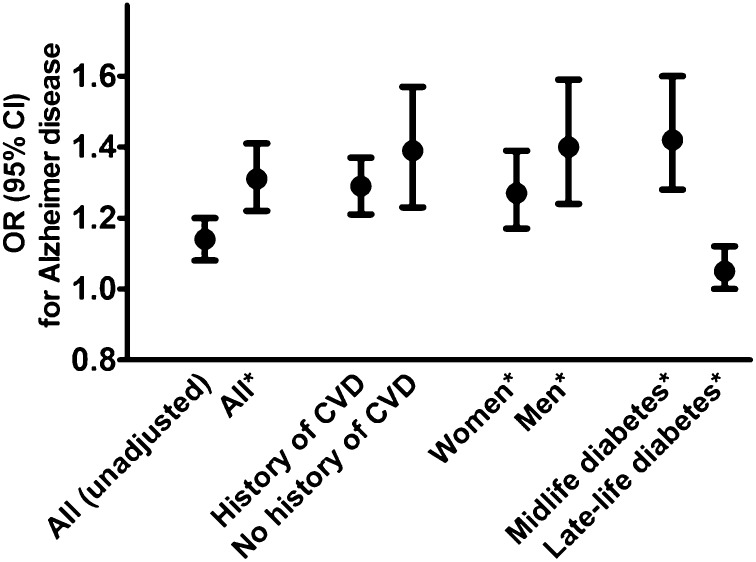
Associations between diabetes and AD are shown in Fig. 1. People with AD were more likely to have a history of clinically verified and medically treated diabetes (either type 1 or type 2 diabetes) in the unadjusted analysis (OR 1.14 [95% CI 1.08–1.20]), and the association was strengthened after adjusting for cardiovascular diseases (adjusted OR 1.31 [1.22–1.41]). The association between diabetes and AD was similar in individuals with no history of cardiovascular diseases (n = 23,086, OR 1.39 [1.23–1.57]) and those with cardiovascular diseases (n = 33,100, OR 1.29 [1.21–1.37]). The associations were similar in both sexes (adjusted OR 1.27 [1.17–1.39] and 1.40 [1.23–1.59] in women and men, respectively).
Figure 1.
Association between diabetes and incident AD in the whole study population, according to previous cardiovascular disease (CVD), sex, and onset age of diabetes. *Adjusted for cardiovascular diseases.
Both midlife and late-life diabetes were more common among those with AD (Table 1 and Fig. 1). Midlife diabetes was more strongly associated with AD than diabetes diagnosed in late life (crude OR 1.42 [95% CI 1.28–1.60] and 1.05 [1.00–1.12] for midlife and late-life diabetes, respectively). The associations were strengthened after adjusting for cardiovascular diseases (pooled OR 1.60 [1.34–1.84] and 1.25 [1.16–1.36] for midlife and late life, respectively).
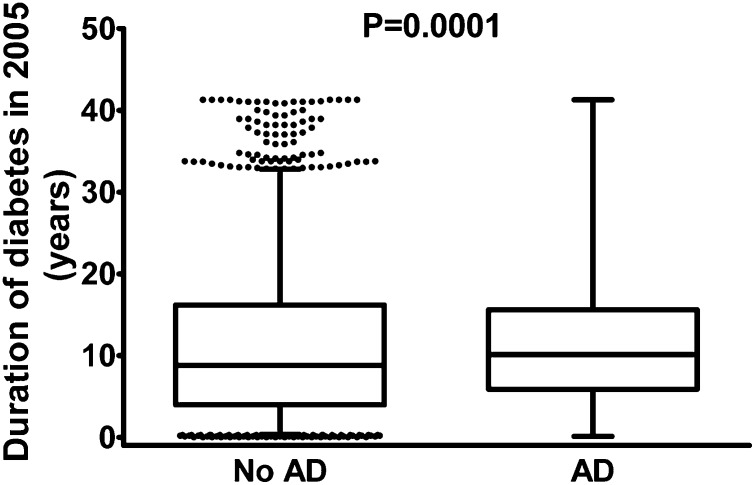
The median age at diabetes diagnosis was 71.4 years (interquartile range 64.6–77.0 years), ranging from 15.4 to 98.9 years. Those with AD were younger when diabetes was diagnosed (Table 1) and thus the duration of diabetes was longer in case subjects than control subjects (Fig. 2). There was also more variation in the diabetes duration among case subjects. Only 54 people were <40 years of age when they were diagnosed with diabetes, suggesting that the majority had type 2 diabetes. The exclusion of these 54 people who putatively had type 1 diabetes had no effect on the association (data not shown). Age at AD diagnosis between those with and without diabetes was similar (mean age 78.0 years, SD 0.1 for both groups).
Figure 2.
Duration of diabetes in control subjects and AD case subjects. Boxes represent the interquartile range and whiskers represent 95% CIs. Individual dots represent observations outside the 95% CIs.
Altogether, 757 (22.4%) of those with AD and diabetes received the entitlement of reimbursed diabetes medication after AD diagnosis. The median time interval was 2.0 years (interquartile range 0.7–3.9 years), and 35.9% (n = 272) of AD cases with diabetes received the decision on reimbursed medication within 1 year of AD diagnosis.
CONCLUSIONS
In this nationwide case-control study, people with clinically verified AD were more likely to have a history of clinically verified and medically treated diabetes than the general aged population. The association between diabetes and AD was similar in individuals with or without a history of cardiovascular disease, and adjustment for cardiovascular diseases strengthened the association. Our findings also showed that in Finland, AD patients who receive a diagnosis of diabetes often do so within 1–2 years of AD diagnosis. This is likely due to the exclusion of alternative diagnoses, which is required before receiving reimbursed AD medication.
Our findings and estimates are consistent with a previous meta-analysis on type 2 diabetes and AD (1). People with type 1 diabetes have also been suggested to have an increased risk of cognitive dysfunction (9). In our study, the mean age at diabetes diagnosis was ∼70 years, and only 52 of all 6,384 people with diabetes were <40 years of age when they were diagnosed, suggesting that the majority had type 2 diabetes. Considering the high average age (79.7 years), it is not surprising that only a small minority of the sample were <40 years of age when diagnosed with diabetes. Mortality among people with type 1 diabetes in Finland was significantly higher in the 1970s (37), so these individuals were less likely to survive to an older age than they would be today.
According to the hypothesis that midlife vascular risk may be more strongly associated with AD than late-life risk factors (20), we found a stronger association with midlife diabetes than late-life diabetes. This is in line with previous studies showing that long-term diabetes is more strongly associated with cognitive impairment and dementia (38) and that diabetes in midlife increases the risk of AD (29). Although this previous study found no association between late-life diabetes and AD (29), another recent study investigating the midlife and late-life vascular risk factors concluded that the association of diabetes was of similar magnitude in both age groups (39). The age-dependent effect may be due to smaller effects of the AD disease process on midlife factors than on factors assessed in late life. Midlife vascular risk factors may also be a more accurate reflection of the vascular load during adulthood (40).
The estimates from our study (unadjusted 95% CI 1.08–1.20) are comparable, although lower than in the meta-analysis by Profenno et al. (1) (95% CI 1.33–1.79), although our adjusted estimates (1.22–1.41) are consistent with the previous findings. This could be due to differences in the definition of diabetes. In half of the studies included in that meta-analysis, the diabetes diagnosis was based on self-report or use of diabetes medication (21–23,25), whereas in our study, all diabetes diagnoses were performed in healthcare settings and based on the assessment of blood glucose concentrations, which is mandatory for reimbursed diabetes medication in Finland. There were also differences in sample size: the conclusions of Profenno et al. (1) were based on 21,560 individuals, including 1,306 AD case subjects, whereas our sample included 28,093 AD case subjects. However, our study is limited in the sense that individuals with type 2 diabetes are underrepresented, as approximately one-third of type 2 diabetes is treated by lifestyle modifications only (36). Our sample would cover all individuals with type 1 diabetes (who were able to survive to their 70s or 80s), as the insulin replacement therapy is necessary, and the majority of people with advanced type 2 diabetes or those who did not benefit from lifestyle modification.
Our nationwide register-based study has some further limitations. We were not able to adjust for lifestyle or socioeconomic confounders, such as obesity, smoking, or education as these data were not available from the registers we used. However, we attempted to capture some variation in these factors by adjusting for cardiovascular diseases. Since there was no data on glycosylated hemoglobin concentrations or glucose concentrations after the reimbursement was accepted by SII, we could not assess whether those with better control of blood glucose concentrations had a different risk of AD. Diabetes is commonly undiagnosed, especially among older individuals (41), so the misclassification of people with undetected diabetes or diabetes treated with lifestyle modifications as not having diabetes is likely. This would lead to an underestimation of the association between diabetes and AD. This has implications for the interpretation of the results: our findings are not as informative on the association of all stages of diabetes as they are on the association of clinically verified and medically treated diabetes. Due to the lack of a systematic cognitive assessment of each person included in this study, it is likely that people with cognitive impairment and AD or other dementias were included in the control group. Thus, our results are likely an underestimation of the true association between diabetes and AD, although they are consistent with previous studies that were able to adjust for confounders or systematically assess the blood glucose concentrations of participants (1).
The strengths of the study are sample size, representativeness, length of follow-up, and verified diagnosis of AD. The study sample includes all community-dwelling individuals with AD residing in Finland in 2005. We included only noninstitutionalized people, but it is unlikely that the association between diabetes and AD would be different among institutionalized individuals. We had access to data collected over 30 years, whereas previous studies on type 2 diabetes (21–26), apart from a few exceptions (27–29), have included <10 years of follow-up. Since the information on diabetes medication, age at diabetes diagnosis, AD, and cardiovascular diseases was extracted from the registries, the results are not affected by recall bias. Although the register-based diagnoses of AD likely underestimate the true prevalence, these diagnoses are more reliable than AD diagnoses extracted from other sources of routine healthcare data, e.g., National Hospital Discharge Register, as the diagnoses in the Special Reimbursement Register are based on DSM-IV and NINCDS-ADRDA criteria whereas the accuracy of diagnoses from the National Hospital Discharge Register may vary considerably, depending on the facility (e.g., specialized geriatrics unit vs. general healthcare visit).
In conclusion, results from this unique nationwide case-control study show that individuals with AD are more likely to have a history of medically treated diabetes than the older population in general and that the association is independent of cardiovascular diseases.
Acknowledgments
A-.M.T., P.L., H.S., and S.H. acknowledge financial support by strategic funding of the University of Eastern Finland.
The funder had no role in study design, data collection or analysis, decision to publish, or preparation of the manuscript.
No potential conflicts of interest relevant to this article were reported.
A.-M.T. planned the research project, drafted the first version of the manuscript, performed statistical analyses, and accepted the final manuscript and made important intellectual contribution to draft versions. P.L., A.S., M.K., and M.U. accepted the final manuscript and made important intellectual contribution to draft versions. H.S. planned the research project and accepted the final manuscript and made important intellectual contribution to draft versions. S.H. planned the research project, drafted the first version of the manuscript, and accepted the final manuscript and made important intellectual contribution to draft versions. A.-M.T. is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
References
- 1.Profenno LA, Porsteinsson AP, Faraone SV. Meta-analysis of Alzheimer’s disease risk with obesity, diabetes, and related disorders. Biol Psychiatry 2010;67:505–512 [DOI] [PubMed] [Google Scholar]
- 2.Xu W, Qiu C, Winblad B, Fratiglioni L. The effect of borderline diabetes on the risk of dementia and Alzheimer’s disease. Diabetes 2007;56:211–216 [DOI] [PubMed] [Google Scholar]
- 3.Vanhanen M, Koivisto K, Kuusisto J, et al. Cognitive function in an elderly population with persistent impaired glucose tolerance. Diabetes Care 1998;21:398–402 [DOI] [PubMed] [Google Scholar]
- 4.Yaffe K, Blackwell T, Kanaya AM, Davidowitz N, Barrett-Connor E, Krueger K. Diabetes, impaired fasting glucose, and development of cognitive impairment in older women. Neurology 2004;63:658–663 [DOI] [PubMed] [Google Scholar]
- 5.Baker LD, Cross DJ, Minoshima S, Belongia D, Watson GS, Craft S. Insulin resistance and Alzheimer-like reductions in regional cerebral glucose metabolism for cognitively normal adults with prediabetes or early type 2 diabetes. Arch Neurol 2011;68:51–57 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.McCrimmon RJ, Ryan CM, Frier BM. Diabetes and cognitive dysfunction. Lancet 2012;379:2291–2299 [DOI] [PubMed] [Google Scholar]
- 7.Helkala EL, Niskanen L, Viinamaki H, Partanen J, Uusitupa M. Short-term and long-term memory in elderly patients with NIDDM. Diabetes Care 1995;18:681–685 [DOI] [PubMed] [Google Scholar]
- 8.Biessels GJ, Deary IJ, Ryan CM. Cognition and diabetes: a lifespan perspective. Lancet Neurol 2008;7:184–190 [DOI] [PubMed] [Google Scholar]
- 9.Brands AM, Biessels GJ, de Haan EH, Kappelle LJ, Kessels RP. The effects of type 1 diabetes on cognitive performance: a meta-analysis. Diabetes Care 2005;28:726–735 [DOI] [PubMed] [Google Scholar]
- 10.Neumann KF, Rojo L, Navarrete LP, Farías G, Reyes P, Maccioni RB. Insulin resistance and Alzheimer’s disease: molecular links & clinical implications. Curr Alzheimer Res 2008;5:438–447 [DOI] [PubMed] [Google Scholar]
- 11.De Felice FG, Vieira MN, Bomfim TR, et al. Protection of synapses against Alzheimer’s-linked toxins: insulin signaling prevents the pathogenic binding of Abeta oligomers. Proc Natl Acad Sci USA 2009;106:1971–1976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuljiš RO, Salković-Petrišić M. Dementia, diabetes, Alzheimer’s disease, and insulin resistance in the brain: progress, dilemmas, new opportunities, and a hypothesis to tackle intersecting epidemics. J Alzheimers Dis 2011;25:29–41 [DOI] [PubMed] [Google Scholar]
- 13.Burns JM, Donnelly JE, Anderson HS, et al. Peripheral insulin and brain structure in early Alzheimer disease. Neurology 2007;69:1094–1104 [DOI] [PubMed] [Google Scholar]
- 14.Haan MN. Therapy insight: type 2 diabetes mellitus and the risk of late-onset Alzheimer’s disease. Nat Clin Pract Neurol 2006;2:159–166 [DOI] [PubMed] [Google Scholar]
- 15.Akter K, Lanza EA, Martin SA, Myronyuk N, Rua M, Raffa RB. Diabetes mellitus and Alzheimer’s disease: shared pathology and treatment? Br J Clin Pharmacol 2011;71:365–376 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Regan C, Katona C, Walker Z, Hooper J, Donovan J, Livingston G. Relationship of vascular risk to the progression of Alzheimer disease. Neurology 2006;67:1357–1362 [DOI] [PubMed] [Google Scholar]
- 17.Sanz C, Andrieu S, Sinclair A, Hanaire H, Vellas B, REAL.FR Study Group Diabetes is associated with a slower rate of cognitive decline in Alzheimer disease. Neurology 2009;73:1359–1366 [DOI] [PubMed] [Google Scholar]
- 18.Mielke MM, Rosenberg PB, Tschanz J, et al. Vascular factors predict rate of progression in Alzheimer disease. Neurology 2007;69:1850–1858 [DOI] [PubMed] [Google Scholar]
- 19.Helzner EP, Luchsinger JA, Scarmeas N, et al. Contribution of vascular risk factors to the progression in Alzheimer disease. Arch Neurol 2009;66:343–348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kloppenborg RP, van den Berg E, Kappelle LJ, Biessels GJ. Diabetes and other vascular risk factors for dementia: which factor matters most? A systematic review. Eur J Pharmacol 2008;585:97–108 [DOI] [PubMed] [Google Scholar]
- 21.Arvanitakis Z, Wilson RS, Bienias JL, Evans DA, Bennett DA. Diabetes mellitus and risk of Alzheimer disease and decline in cognitive function. Arch Neurol 2004;61:661–666 [DOI] [PubMed] [Google Scholar]
- 22.Luchsinger JA, Tang MX, Stern Y, Shea S, Mayeux R. Diabetes mellitus and risk of Alzheimer’s disease and dementia with stroke in a multiethnic cohort. Am J Epidemiol 2001;154:635–641 [DOI] [PubMed] [Google Scholar]
- 23.MacKnight C, Rockwood K, Awalt E, McDowell I. Diabetes mellitus and the risk of dementia, Alzheimer’s disease and vascular cognitive impairment in the Canadian Study of Health and Aging. Dement Geriatr Cogn Disord 2002;14:77–83 [DOI] [PubMed] [Google Scholar]
- 24.Ott A, Stolk RP, van Harskamp F, Pols HA, Hofman A, Breteler MM. Diabetes mellitus and the risk of dementia: The Rotterdam Study. Neurology 1999;53:1937–1942 [DOI] [PubMed] [Google Scholar]
- 25.Peila R, Rodriguez BL, Launer LJ, Honolulu-Asia Aging Study Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: the Honolulu-Asia Aging Study. Diabetes 2002;51:1256–1262 [DOI] [PubMed] [Google Scholar]
- 26.Ahtiluoto S, Polvikoski T, Peltonen M, et al. Diabetes, Alzheimer disease, and vascular dementia: a population-based neuropathologic study. Neurology 2010;75:1195–1202 [DOI] [PubMed] [Google Scholar]
- 27.Akomolafe A, Beiser A, Meigs JB, et al. Diabetes mellitus and risk of developing Alzheimer disease: results from the Framingham Study. Arch Neurol 2006;63:1551–1555 [DOI] [PubMed] [Google Scholar]
- 28.Leibson CL, Rocca WA, Hanson VA, et al. Risk of dementia among persons with diabetes mellitus: a population-based cohort study. Am J Epidemiol 1997;145:301–308 [DOI] [PubMed] [Google Scholar]
- 29.Xu W, Qiu C, Gatz M, Pedersen NL, Johansson B, Fratiglioni L. Mid- and late-life diabetes in relation to the risk of dementia: a population-based twin study. Diabetes 2009;58:71–77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Bell JS, Laitinen ML, Lavikainen P, Lönnroos E, Uosukainen H, Hartikainen S. Use of strong opioids among community-dwelling persons with and without Alzheimer’s disease in Finland. Pain 2011;152:543–547 [DOI] [PubMed] [Google Scholar]
- 31.Furu K, Wettermark B, Andersen M, Martikainen JE, Almarsdottir AB, Sørensen HT. The Nordic countries as a cohort for pharmacoepidemiological research. Basic Clin Pharmacol Toxicol 2010;106:86–94 [DOI] [PubMed] [Google Scholar]
- 32.McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA work group under the auspices of department of health and human services task force on alzheimer's disease. Neurology 1984;34:939–944 [DOI] [PubMed] [Google Scholar]
- 33.American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders. 4th Ed. Washington DC, American Psychiatric Association, 1994 [Google Scholar]
- 34.Suhonen J, Pirttilä T, Erkinjuntti T, et al. Update on current care guidelines: the diagnosis and medical treatment of memory disorders. Duodecim 2010; 126:2167–2168 [in Finnish] [PubMed]
- 35.The Social Insurance Institution of Finland. Decision on medical criteria required for entitlement to a special reimbursement on 24.11.2011. 2011 [in Finnish]
- 36.Laakso M, Pyörälä K. Age of onset and type of diabetes. Diabetes Care 1985;8:114–117 [DOI] [PubMed] [Google Scholar]
- 37.Harjutsalo V, Forsblom C, Groop PH. Time trends in mortality in patients with type 1 diabetes: nationwide population based cohort study. BMJ 2011;343:d5364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Bruce DG, Davis WA, Casey GP, et al. Predictors of cognitive impairment and dementia in older people with diabetes. Diabetologia 2008;51:241–248 [DOI] [PubMed] [Google Scholar]
- 39.Kimm H, Lee PH, Shin YJ, et al. Mid-life and late-life vascular risk factors and dementia in Korean men and women. Arch Gerontol Geriatr 2011;52:e117–e122 [DOI] [PubMed] [Google Scholar]
- 40.Rönnemaa E, Zethelius B, Lannfelt L, Kilander L. Vascular risk factors and dementia: 40-year follow-up of a population-based cohort. Dement Geriatr Cogn Disord 2011;31:460–466 [DOI] [PubMed] [Google Scholar]
- 41.Gregg EW, Cadwell BL, Cheng YJ, Cowie CC, Williams DE, Geiss L, Engelgau MM, Vinicor F. Trends in the prevalence and ratio of diagnosed to undiagnosed diabetes according to obesity levels in the U.S. Diabetes Care 2004;27:2806–2812 [DOI] [PubMed]