Abstract
Cerebral angiostrongyliasis is an acute inflammation caused by the infection of the nematode Angiostrongylus cantonensis that results in eosinophilic meningitis. The current immunological assay of choice is an immunoblot that detects antibodies to a 31 kDa protein present in crude extracts of the female worm. Recently we have identified diagnostic targets from excretion and secretion products and determined the composition of the 31 kDa antigen after 2-D gel electrophoresis and mass spectrometry. Here we cloned and expressed five proteins in prokaryotic and eukaryotic systems. Recombinant proteins were purified and analysed by Western blot assays and among them 14-3-3, Lec5 and ES7 were recognized by Angiostrongylus-specific serum, although the signal was weak.
Keywords: 31 kDa antigen, Angiostrongylus, Eosinophilic meningitis, Recombinant protein
Introduction
In the genus Angiostrongylus two species are of medical importance: A. costaricensis, which lives in the mesenteric arteries of wild mice and causes abdominal angiostrongyliasis in humans, and A. cantonensis, which lives in the pulmonary arteries of rats and may cause eosinophilic meningitis or meningoencephalitis (EM) in humans. EM is an acute disease caused most often by the presence of A. cantonensis larvae in the meninges. The diagnosis is based on parasitological or molecular findings of either parasites or their DNA in cerebro-spinal fluid (CSF) or specific antibodies in the CSF or serum. Several studies have investigated the utility of various antigens, either crude or partially purified proteins, for sensitive and specific detection of A. cantonensis specific antibodies.1 The current immunological assay of choice is an immunoblot that detects antibodies to a 31 kDa protein present in crude extracts of the nematode.2 The 31 kDa antigen has been purified and used also in dot blot tests in regional hospitals in Thailand,3,4 but this process is laborious and results in a low yield of material, making standardization and distribution to other diagnostic centers difficult.
Therefore, with the ultimate goal of generating a recombinant protein antigen or antigens for angiostrongyliasis diagnosis, a proteomics approach was used to obtain amino acid sequence from the 31 kDa protein and from other potential diagnostic targets present in the excretory/secretory fraction of cultured A. cantonensis adults. The composition of the 31 kDa antigen was determined after 2-D gel electrophoresis and mass spectrometry. Amino acid sequence data were obtained from at least 3 different proteins: the 14-3-3 phosphoserine-binding protein (14-3-3), a protein containing a nascent polypeptide-associated complex domain (NAC), and the putative epsilon subunit of coatomer protein complex isoform 2 (Ep31). It was shown that the antigenicity of the native 31 kDa protein is dependent on the presence of carbohydrate moieties.5 Another study identified 17 proteins that may prove to be useful diagnostic targets for EM, including proteins with amino acid sequence homology with galectin-5 (Lec5), peroxiredoxin, hemoglobinases, heat shock proteins, protease inhibitors, and a putative protein (Es-7).6 Recombinant proteins from the 31 kDa protein or excretion and secretion products (ES) were expressed in prokaryotic and eukaryotic systems and evaluated as potential diagnostic targets.
Material and Methods
In this study five proteins were selected for recombinant expression: three identified from 31 kDa antigen (14-3-3, NAC, Ep31) and two identified from ES (Lec-5, Es-7). Genomic sequences were obtained by parallel tag sequencing using a Roche 454 GS FLX sequencer and the corresponding cDNAs were amplified by PCR (polymerase chain reaction) using Platinum Taq DNA polymerase. The amplification reaction was: DNA polymerase Pfx 1u, DNTPs 5 mM, buffer 1x, primers 10 pmol each, MgSO4 1.5 mM, cDNA 50 ng. PCR cycling conditions were: 95 °C for 5 min, 30 cycles of 95 °C for 30 s, the specific primer annealing temperature (Table 1) for 30 s, 72 °C for 1 min, followed by a final extension at 72 °C for 7 min.
Table 1.
The number of DNA base pairs sequenced for each protein coding locus, primer sequences, GenBank accession numbers of DNA sequences from which primers were designed, and annealing temperatures.
| Protein | Number of base pairs | Primersa | GenBank accession number | Annealing temperature |
| Ep31 | 877 | F: CACCATGTCTGGGGTTGATCGTTTGT R: TTAAGCGGCAACAAGTTCATCA |
FM207709.1 | 66 °C |
| ES7 | 648 | F: CACCATGCGGTCAATTCTGATCTATT R: TTAACTGACGTAGAGCCAGTGA |
FM207698.1 | 61 °C |
| Lec-5 | 969 | F:CACCATGAGGATGAAGGTGTTGCT R:CTTCATACTCCTGGAGCATCGTTG |
JN133965.1 | 59 °C |
| 14-3-3 | 925 | F:CACCATGACGGACAACAGGGGCGA R:TCAGTTGGCACCCTCTCCTTGTTC |
JN133967.1 | 71 °C |
| NAC | 583 | F:CACCATGGTTGCCGCGGTGGAAGT R:TTAACAAATAACTGAGAATCAA |
JN133968.1 | 55 °C |
F - foward, R - reverse
Recombinant proteins were expressed using either the 6xHis-tag Champion Pet200® prokaryotic expression system (Invitrogen, Life Technologies,Grand Island, NY) or pFastBac vector (Bac-To-Bac® Invitrogen) baculovirus expression system.
For prokaryotic expression Escherichia coli DE3 BL21 cells were used. Cells were grown in Luria-Bertani (LB) broth supplemented with kanamycin (100 mg/mL) with shaking (250 rpm) at 37 °C. At log phase IPTG (Isopropyl-β-D-thio-galactoside) (1 mM) was added and expression was performed for three hours. Cells were then centrifuged for 10 min at 3000 g and the pellet was suspended in lysis buffer (PBS, Pefabloc SC 1:100, leupeptin 1:1000, and pepstatin A 1:100, 0.1% of Triton X-100, pH 7.4). The lysed cells were sonicated for three pulses of 30 s each at 15% of amplitude. Soluble proteins were harvested by centrifugation at 20,000 g for 1 h. Recombinants were purified by affinity chromatography to nickel and eluted with imidazole (250 mM). Protein quantification was performed by the Bradford method.7
As post-translational modifications in eukaryotes are different from those in prokaryotes the recombinant proteins were then expressed in insect cells using a baculovirus expression system to produce recombinant forms of the proteins with more extensive post-translational modifications. Recombinant baculoviruses were constructed by homologous recombination using site specific transposition into DH10Bac E. coli cells. DNA from the constructed Bacmids was purified and used to transfect Sf-9 insect cells for recombinant virus production. Cells were cultivated in serum free medium and infected with the virus. Viability and diameter were monitored every day using a Vi-Cell XR cytometer. After cells reached about 17 µm and less than 50% viability they were harvested by centrifugation (1,500 g) and suspended in 50 mM Tris-HCl, pH 7.4, 1% Tween-20. Lysed cells were centrifuged at 20,000 g to pellet insoluble proteins. The recombinant proteins were purified on a cobalt column and eluted with an imidazole gradient.
SDS-PAGE using 12% polyacrylamide gels and Western blot analyses were used to confirm expression of the recombinant proteins. After electrophoresis the proteins were electro-transferred onto nitrocellulose membranes and blocked with 5% skim milk in PBS with 0.05% Tween-20 for 1 h at room temperature. Membranes were then incubated for 2 h with a pool of sera (1:200 dilution) prepared from either Angiostrongylus-specific control, serum from uninfected controls, and/or anti-6xHis monoclonal antibody (Invitrogen). After three washes, membranes were probed with a secondary peroxidase-conjugated anti-human IgG (Sigma, St. Louis, MO) (diluted 1:8000) or anti-mouse IgG (Sigma) for 1 h at room temperature. Diaminobenzidine (DAB) (Sigma) (0.05% DAB, 0.015% H2O2 in PBS, pH 7.4) was added as developer reagent. For specific Angiostrongylus control, a pool of 20 serum samples prepared from 20 patients histopathologically diagnosed with abdominal angiostrongyliasis (caused by Angiostrongyulus costaricensis) was used. Heterologous antigens have been used in the diagnosis of angiostrongyliasis since both A. cantonensis and A. costaricensis possess cross-reactive antigens that can be used to diagnose infections with either pathogen.8,9 Five serum samples collected from people with no travel history outside the United States were pooled and used as the ‘normal’ control serum pool.
PROSITE (prosite.expasy.org) was used to predict post-translational modifications such as glycosylation and lipidation.
Results and Discussion
A recombinant protein containing homology with the putative epsilon subunit of coatomer protein complex isoform 2 (Ep31) was expressed in the prokaryotic system and purified by nickel affinity chromatography. Western blot analysis using the anti-6His monoclonal antibody showed that Ep31 was expressed at the expected molecular mass, which was 36 kDa (33 kDa + an extra 3 kDa encoded by the plasmid) (Figure 1, A). The yield of the recombinant Ep31 was about 4 mg/ml. Purified Ep31, collected after the first round of imidazole elution, (Figure 2, lane 8) was used in a preliminary evaluation for antigenicity using the Angiostrongylus-specific serum pool. The recombinant Ep31 was recognized by both the normal and the Angiostrongylus-specific serum pools (Figure 1, B and C), which indicated a lack of specificity as a diagnostic antigen. Similarly, a recombinant form of ES-7 was expressed in the prokaryotic system and purified by nickel affinity chromatography. The molecular mass of the ES-7 protein was 26 kDa (23 kDa + 3 kDa encoded by the plasmid) (Figure 3). The yield of the recombinant ES-7 was about 0.8 mg/ml. Two purified ES-7 fractions were evaluated. Fractions 1 and 2 from the imidazole elution were analysed by Western blot (Figure 3, C and D). The Angiostrongylus-specific serum pool produced a weak reaction, in the absence of reactivity with the normal serum pool. This result indicated that ES-7 may have some diagnostic value; however titration of the antigen and tests for cross reactivity to other helminth infections are necessary.
Figure 1.
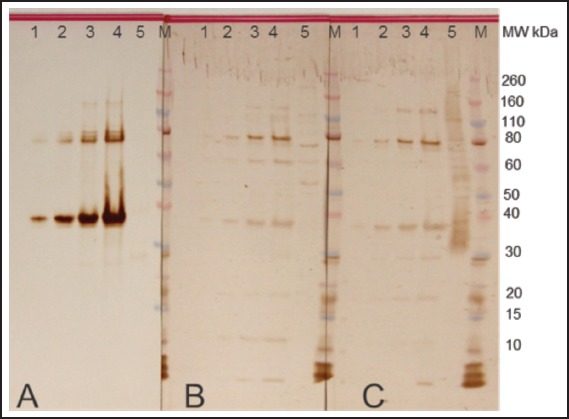
Expression of Ep31. Putative epsilon subunit of coatomer protein complex isoform 2 (Ep31) of Angiostrongylus cantonens expressed in E. coli. Western blot analyses were performed using monoclonal anti-His tag (A), normal human (B) and Angiostrongylus-specific (C) serum. Lane 1 - Ep31 0.5 µg /lane; lane 2 - Ep31 0.3 µg/lane; lane 3 - Ep31 0.2 µg/lane; lane 4 - Ep31 0.1 µg /lane; lane 5 - crude extract of A. cantonensis 0.8 µg/lane. M - molecular weight in kDa.
Figure 2.
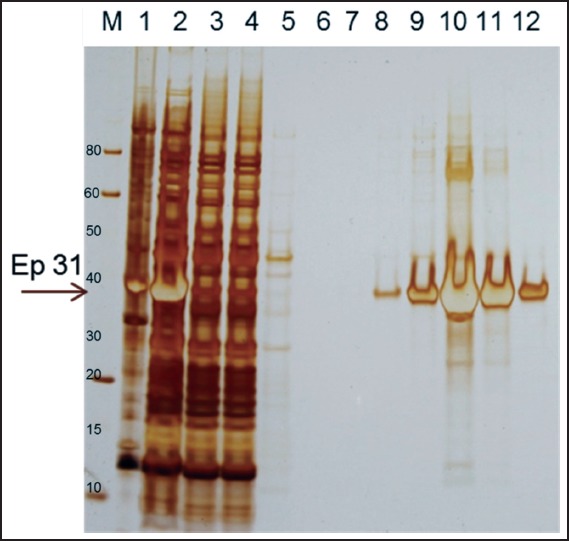
Purification of Ep31. The recombinant protein Ep31 purified by affinity chromatography to nickel. Lane 1 - Ep31 cell lysed insoluble proteins; lane 2 - Ep31 cell lysed soluble proteins; lane 3 - column unbound proteins; lanes 4 and 5 - column washes; lane 6 - Ep31 elution 1; lane 7 - Ep31 elution 2; lane 8 - Ep31 elution 3; lane 9 - Ep31 elution 4; lane 10 - Ep31 elution 5; lane 11 - Ep31 elution 6; lane 12 - Ep31 elution 7; M - molecular weight in kDa. The arrow indicates the Ep31.
Figure 3.
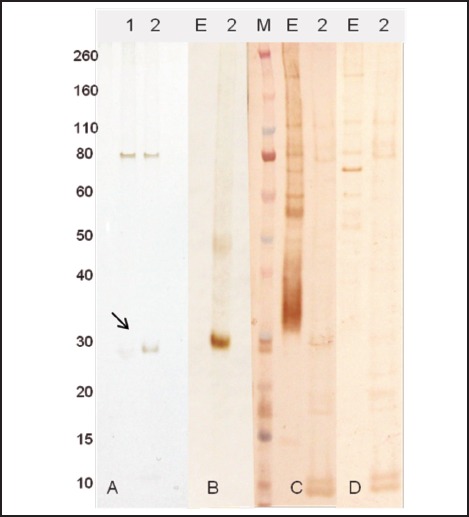
Expression and purification of ES7 protein. A - ES7 expressed in E. coli cells and purified by affinity chromatography to nickel: 1 - elution 1; 2 - elution 2. Western blot analyses were performed using monoclonal anti-His tag (B), Angiostrongylus-specific serum (C), and normal human serum (D). E - crude extract of A. cantonensis 0.8 µg. M - molecular weight in kDa. The arrow indicates the ES7 recombinant protein.
The recombinant proteins 14-3-3 and Lec-5 were expressed in E. coli and evaluated in the same manner. Each had the expected masses, 31 kDa (28 kDa + 3 kDa encoded by the plasmid) and 36 kDa (33 kDa + 3 kDa encoded by the plasmid), respectively (Figure 4). The recombinant proteins were assayed for antigenicity using Western blot (Figure 4). 14-3-3 was not preferentially recognized by the Angiostrongylus-specific serum pool, while Lec-5 was differentially recognized, although the specific reaction was weak (Figure 4, B, lane 8).
Figure 4.
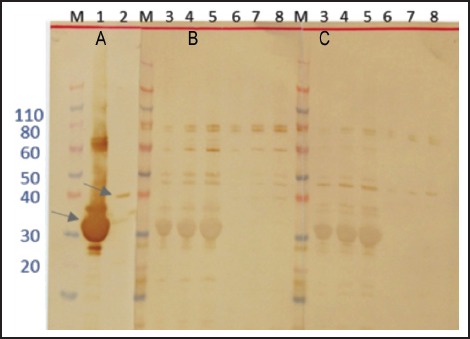
Expression and immunoblotting analyses of 14-3-3 and Lec5 recombinant proteins in E. coli cells. Western blot analyses were performed using monoclonal anti-His tag (A), Angiostrongylus-specific serum (B), and normal human serum (C). Lane 1 - 14-3-3 5 µg/lane; lane 2 - Lec5 0.8 µg/lane; lane 3 - 14-3-3 1 µg/lane; lane 4 - 14-3-3 3 µg/lane; lane 5 - 14-3-3 5 µg/lane; lane 6 - Lec5 0.1 µg/lane; lane 7 - Lec5 0.5µg/lane; lane 8 - Lec5 0.8 µg/lane M - molecular weight in kDa. The arrows indicate 14-3-3 (lane 1) and Lec5 (lane 2) recombinant proteins.
Four proteins were expressed in the prokaryotic system. Two identified from the ES sample, Lec-5, and ES-7, showed reactivity with the Angiostrongylus-specific serum pool, while 14-3-3 and Ep31 showed reactivity with both the Angiostrongylus-specific and the ‘normal’ serum pools. Analyses by the PROSITE program revealed sites for many post-translational modifications, including glycosylation and lipidation. These modifications have been implicated in the antigenicity of A cantonensis proteins as previous studies showed that oxidation of the carbohydrates on the native 31 kDa antigen reduced antibody reactivity,5 suggesting that sugar moieties might be needed for antibody recognition.
14-3-3 proteins are dimeric phosphoserine-binding proteins and members of a family of acidic regulatory molecules that participate in signal transduction, transport, and regulation of several eukaryotic biochemical processes.10,11 In some parasites, such as Echinococcus multilocularis and Schistosoma mansoni, 14-3-3 proteins have been shown to be immunogenic and therefore have been promoted as potential vaccine targets.12–14 For this reason, the A. cantonensis 14-3-3-protein was chosen for expression in the baculovirus system. SDS-PAGE and Western blot analyses revealed expression of a protein with the expected molecular mass (Figure 5). 14-3-3 was secreted during expression, which facilitated its purification. In the western Blot analysis, 14-3-3 was recognized by the Angiostrongylus-specific serum pool, though the reaction was very weak (Figure 5, B), which suggested that the carbohydrates incorporated during insect cell expression may not be the correct sugar moieties necessary for antigenicity.
Figure 5.

Expression of 14-3-3 recombinant protein in insect cells. Western blot analyses were performed using normal human serum (A), Angiostrongylus-specific serum (B), and monoclonal anti-His tag (C). Lane 1 - crude extract of A. cantonensis 0.5 µg/lane; lane 2 - 14-3-3 0.5 µg/lane. M - molecular weight in kDa.
Ultimately another expression system may be necessary to produce these recombinant glycoproteins. Further analysis of the glycosylation patterns of Angiostrongylus may be warranted.
Acknowledgements
This paper represents a contribution to the Rat Lungworm Disease Scientific Workshop held at the Ala Moana Hotel, Honolulu, Hawai‘i in August 2011. Funding for the workshop and for this publication was provided by the National Institute of Food and Agriculture, United States Department of Agriculture, through Award No. 2011-65213-29954.
Conflict of Interest
None of the authors identifies any conflict of interest.
References
- 1.Graeff-Teixeira C, da Silva AC, Yoshimura K. Update on eosinophilic meningoencephalitis and its clinical relevance. Clin Microbiol Rev. 2009;22(2):322–348. doi: 10.1128/CMR.00044-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Eamsobhana P, Yong HS. Immunological diagnosis of human angiostrongyliasis due to Angiostrongylus cantonensis (Nematoda: Angiostrongylidae) Int J Infect Dis. 2009;13(4):425–431. doi: 10.1016/j.ijid.2008.09.021. [DOI] [PubMed] [Google Scholar]
- 3.Eamsobhana P, Yoolek A, Suvouttho S, Suvuttho S. Purification of a specific immunodiagnostic Parastrongylus cantonensis antigen by electroelution from SDS-polyacrylamide gels. Southeast Asian J Trop Med Public Health. 2001;32:308–313. [PubMed] [Google Scholar]
- 4.Eamsobhana P, Yoolek A, Punthuprapasa P. Dot-blot ELISA for the immunological detection of specific antibody to Parastrongylus cantonensis. Trop Biomed. 2003;20:1–6. [Google Scholar]
- 5.Morassutti AL, Levert K, Perelygin A, da Silva AJ, Wilkins P, Graeff-Teixeira C. The 31 kDa antigen of Angiostrongylus cantonensis comprises distinct antigenic glycoproteins. Vector-Borne Zoonot Dis. 2012;12(11):961–968. doi: 10.1089/vbz.2011.0957. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Morassutti AL, Levert K, Pinto PM, da Silva AJ, Wilkins P, Graeff-Teixeira C. Characterization of Angiostrongylus cantonensis excretory-secretory proteins as potential diagnostic targets. Exp Parasitol. 2012;130(1):26–31. doi: 10.1016/j.exppara.2011.10.003. [DOI] [PubMed] [Google Scholar]
- 7.Bradford MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976;72:248–254. doi: 10.1006/abio.1976.9999. [DOI] [PubMed] [Google Scholar]
- 8.Dekumyoy P, Komalamisra C, Nuamtanong S, et al. Angiostrongyliasis: analysis of antigens of Angiostrongylus costaricensis adult worms versus IgG from infected patients with Angiostrongylus cantonensis. Southeast Asian J Trop Med Public Health. 2000;31(Suppl 1):48–53. [PubMed] [Google Scholar]
- 9.Ben R, Rodrigues R, Agostini AA, Graeff-Teixeira C. Use of heterologous antigens for the immunodiagnosis of abdominal angiostrongyliasis by an enzyme-linked immunosorbent assay. Mem Inst Oswaldo Cruz. 2010;105(7):914–917. doi: 10.1590/s0074-02762010000700013. [DOI] [PubMed] [Google Scholar]
- 10.Obsilova V, Nedbalkova E, Silhan J, et al. The 14-3-3 protein affects the conformation of the regulatory domain of human tyrosine hydroxylase. Biochemistry. 2008;47(6):1768–1777. doi: 10.1021/bi7019468. [DOI] [PubMed] [Google Scholar]
- 11.Mrowiec T, Schwappach B. 14-3-3 proteins in membrane protein transport. Biol Chem. 2006;387(9):1227–1236. doi: 10.1515/BC.2006.152. [DOI] [PubMed] [Google Scholar]
- 12.Schechtman D, Winnen R, Tarrab-Hazdai R, et al. Expression and immunolocalization of the 14-3-3 protein of Schistosoma mansoni. Parasitology. 2001;123(6):573–582. doi: 10.1017/s0031182001008769. [DOI] [PubMed] [Google Scholar]
- 13.Siles-Lucas M, Merli M, Gottstein B. 14-3-3 proteins in Echinococcus: their role and potential as protective antigens. Exp Parasitol. 2008;119(4):516–523. doi: 10.1016/j.exppara.2008.01.009. [DOI] [PubMed] [Google Scholar]
- 14.Wang Y, Cheng Z, Lu X, Tang C. Echinococcus multilocularis: proteomic analysis of the protoscoleces by bi-dimensional electrophoresis and mass spectrometry. Exp Parasitol. 2009;123(2):162–167. doi: 10.1016/j.exppara.2009.06.014. [DOI] [PubMed] [Google Scholar]


